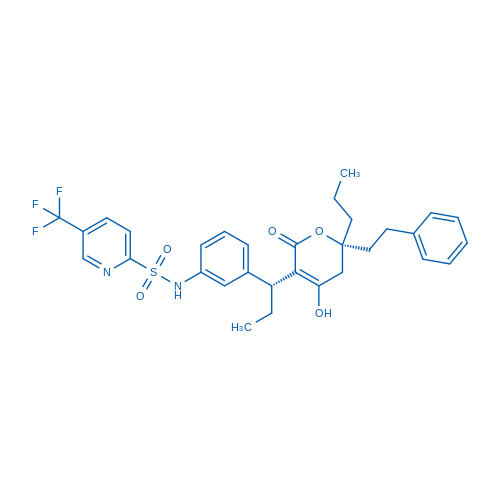
CAS No.: 174484-41-4
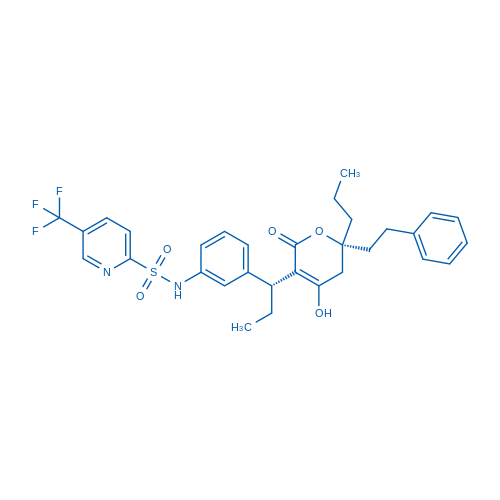
Tipranavir Catalog No. CSN12095
Synonyms: PNU-140690;brand name: Aptivus
Tipranavir, a nonpeptidic HIV protease inhibitor (NPPI), inhibits the enzymatic activity and dimerization of HIV-1 protease, exerts potent activity against multi-PI-resistant HIV-1 isolates.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00197145 Infection, Human Immunodeficie... more >>ncy Virus I Collapse << Phase 3 Terminated - - NCT00708162 HIV Infection Phase 3 Completed - - NCT00042289 HIV Infections Phase 4 Recruiting September 30, 2019 - - 更多
- 参考文献
- [1] Aoki M, Danish ML, et al. Loss of the protease dimerization inhibition activity of tipranavir (TPV) and its association with the acquisition of resistance to TPV by HIV-1. J Virol. 2012 Dec;86(24):13384-96.
- [2] Li F, Wang L, et al. Metabolism-mediated drug interactions associated with ritonavir-boosted tipranavir in mice. Drug Metab Dispos. 2010 May;38(5):871-8. doi: 10.1124/dmd.109.030817. Epub 2010 Jan 26.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 174484-41-4 | 储存条件 |
|
|
| 分子式 | C31H33F3N2O5S | 运输 | 蓝冰 | |
| 分子量 | 602.66 | 别名 | PNU-140690;brand name: Aptivus |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00197145 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 3 | Terminated | - | - |
| NCT00708162 | HIV Infection | Phase 3 | Completed | - | - |
| NCT00042289 | HIV Infections | Phase 4 | Recruiting | September 30, 2019 | - |
| NCT03631732 | HIV-1 Infection | Phase 3 | Recruiting | August 2020 | - |
| NCT00192660 | HIV-Associated Lipodystrophy S... more >>yndrome Cardiovascular Disease Collapse << | Phase 4 | Completed | - | Australia, New South Wales ... more >> St. Vincent's Hospital Sydney, New South Wales, Australia, 2010 Collapse << |
| NCT00197145 | - | - | Terminated | - | - |
| NCT02302547 | HIV | Phase 3 | Completed | - | France ... more >> Unité des Maladies Infectieuses, CHU de CAEN Caen, France, 14033 Service des Maladies Infectieuses, CHR Orléans La Source, ORLEANS CEDEX 2 CHR d'ORLEANS, France, 45067 Service d'Immunologie Clinique centre de Vaccination anti- VIH ANRS Hopital Henri- Mondor Creteil, France, 94010 Service de Médecine Interne et Maladies Infectieuses, Groupe Hospitalier La Rochelle, Cedex 01 La Rochelle, France, 17019 Service de Pneumologie, centre Hospitalier Fontenoy, CH de CHARTRES Le Coudray, France, 28630 CHU de NANCY Nancy, France, 54035 Service des maladies Infectieuses et tropicales, CH GEORGES RENON Niort, France, 79021 Service des Maladies Infectieuses et tropicales, APHP SAINT LOUIS Paris, France, 75475 Hospital Tenon Paris, France, 75970 Centre de diagnostic et thérapeutique, Hopital Hotel Dieu Paris, France, 94010 Consultation Maladies Infectieuses, Chu de Poitiers, Cedex Poitiers, France, 86021 Maladies Infectieuses, CHU de ROUEN Rouen, France, 76031 Service de Médecine Interne, CH de SAINTONGE- BP 326 Saintes, France, 17108 Médecine Interne, Hôpital FOCH Suresnes, France, 92151 Service Universitaire des Maladies Infectieuses et du Voyageur, CH DRON Tourcoing, France, 59200 Service de Medecine Interne et Maladies Infectieuses, CHRU BRETONNEAU, TOURS CEDEX9 Tours, France, 37044 Collapse << |
| NCT01814722 | - | - | Terminated(The trial was termi... more >>nated early due to slow recruitment.) Collapse << | - | - |
| NCT01814722 | - | - | Terminated(The trial was termi... more >>nated early due to slow recruitment.) Collapse << | - | - |
| NCT01923311 | Acquired Immune Deficiency Syn... more >>drome (AIDS) HIV Infections Collapse << | Phase 2 Phase 3 | Terminated | - | United States, Colorado ... more >> University of Colorado Denver Aurora, Colorado, United States, 80045 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Tennessee St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Italy Universita degli Studi di Pavia - Fondazione IRCCS Policlinico San Matteo Pavia, Italy, 27100 South Africa Be Part Yoluntu Centre Cape Town, South Africa, 7646 Rahima Moosa Mother and Child Hopsital Johannesburg, South Africa, 2112 Spain Hospital Universitario De Getafe Getafe, Madrid, Spain, 28095 Hospital 12 de Octubre Madrid, Spain, 28041 Thailand Thai Red Cross AIDS Research Centre (HIV-NAT) Bangkok, Thailand, 10330 Siriraj Hospital Bangkok, Thailand, 10700 Uganda Joint Clinical Research Centre Kampala, Uganda Collapse << |
| NCT01923311 | - | - | Terminated | - | - |
| NCT00708162 | - | - | Completed | - | - |
| NCT00966329 | HIV HIV Infec... more >>tions Collapse << | Phase 4 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Lluita contra la Sida Foundation, HIV Unit, Irsi Caixa Foundation Badalona, Barcelona, Spain, 08916 Lluita contra la Sida Foundation, HIV Unit Badalona, Barcelona, Spain, 08916 Collapse << |
| NCT01232361 | - | - | Completed | - | United States, Alabama ... more >> Univ. of Alabama Birmingham NICHD CRS (5096) Birmingham, Alabama, United States, 35294 United States, California Miller Children's Hospital Long Beach (5093) Long Beach, California, United States, 90806 Usc La Nichd Crs (5048) Los Angeles, California, United States, 90033 UCLA-Los Angeles/Brazil AIDS Consortium (LABAC) CR (3601) Los Angeles, California, United States, 90095 Univ of California, San Diego (UCSD) (4601) San Diego, California, United States, 92103 United States, Colorado Childrens Hospital (U. Colorado, Denver) NICHD CRS (5052) Denver, Colorado, United States, 80218-1088 United States, District of Columbia Children's National Med. Ctr. Washington DC NICHD CRS (5015) Washington, District of Columbia, United States, 20010 United States, Florida South Florida CDC Ft Lauderdale NICHD CRS (5055) Ft Lauderdal, Florida, United States, 33316 Univ of Miami Pediatric/Perinatal HIV/AIDS (4201) Miami, Florida, United States, 33136 United States, Illinois Chicago Children's CRS (4001) Chicago, Illinois, United States, 60611 Rush University Cook County Hospital NICHD CRS (5083) Chicago, Illinois, United States, 60612 United States, Maryland Johns Hopkins University NICHD CRS (5092) Baltimore, Maryland, United States, 21287 United States, Massachusetts Boston Medical Center Ped. HIV Program NICHD CRS (5011) Boston, Massachusetts, United States, 02118 WNE Maternal Pediatric Adolescent AIDS CRS (7301) Worcester, Massachusetts, United States, 01605 United States, Michigan Wayne State University/Children's Hospital of Michigan NICHD CRS (5041) Detroit, Michigan, United States, 48201 United States, New York Jacobi Med. Ctr. Bronx NICHD CRS (5013) Bronx, New York, United States, 10461 New York University NY (5012) New York, New York, United States, 10016 Columbia IMPAACT Center (4101) New York, New York, United States, 10032 SUNY Stony Brook (5040) Stony Brook, New York, United States, 11794-8111 United States, Tennessee St. Jude Childrens Research Hospital, Memphis (6501) Memphis, Tennessee, United States, 38105-2794 United States, Texas Texas Children's Hosp. CRS (3801) Houston, Texas, United States, 77030 Puerto Rico San Juan City Hosp. PR NICHD CRS (5031) San Juan, Puerto Rico, 00927 Collapse << |
| NCT00537394 | - | - | Completed | - | - |
| NCT02486510 | HIV-1 Infection | Early Phase 1 | Terminated(Futility criteria) | - | Spain ... more >> Hospital Universitario Ramon Y Cajal Madrid, Spain, 28034 Hospital Universitario La Paz Madrid, Spain, 28046 Collapse << |
| NCT01276236 | Kaposi's Sarcoma | Not Applicable | Unknown | April 2015 | United States, California ... more >> San Francisco General Hospital, Clinical Trials Unit San Francisco, California, United States, 94110 Collapse << |
| NCT01225406 | - | - | Completed | - | Thailand ... more >> Chulalongkorn University Bangkok, Thailand, 10330 HIV-NAT Bangkok, Thailand, 10330 Siriraj Hospital, Mahidol University Bangkok, Thailand, 10700 Prapokklao Chantaburi Chantaburi, Thailand, 22000 Nakornping Hospital Chiang Mai, Thailand, 50180 Chiang Rai Regional Hospital Chiang Rai, Thailand, 57000 Khon Kaen University Khon Kaen, Thailand, 40002 Bamrasnaradura Institute Nonthaburi, Thailand, 11000 Surin Hospital Surin, Thailand, 32000 Collapse << |
| NCT00537394 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00735072 | HIV Infection | Phase 4 | Completed | - | United States, California ... more >> University of California San Francisco - San Francisco General Hospital San Francisco, California, United States, 94110 Stanford University Stanford, California, United States, 94305 United States, Illinois Rush University - Stroger Hospital of Cook County Chicago, Illinois, United States, 60612 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 Collapse << |
| NCT02105987 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Completed | - | - |
| NCT00624195 | HIV Infections | Phase 2 Phase 3 | Completed | - | United States, California ... more >> HIV Neurobehavioral Research Center, University of California San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94110 United States, Maryland Johns Hopkins University- School of Medicine Baltimore, Maryland, United States, 21287 United States, Missouri Washington University St. Louis, Missouri, United States, 63110 United States, New York Mount Sinai Medical Center New York, New York, United States, 10024 Collapse << |
| NCT02195466 | Healthy | Phase 1 | Completed | - | - |
| NCT02227017 | Healthy | Phase 1 | Completed | - | - |
| NCT02105987 | - | - | Completed | - | - |
| NCT00307502 | HIV Infections | Phase 1 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Hospital de Figueres Figueras, Barcelona, Spain, 17600 Fundació Hospital-Asil de Granollers Granollers, Barcelona, Spain, 08400 Hospital de Vic Vic, Barcelona, Spain, 08500 Hospital Universitari Sant Joan de Reus Reus, Tarragona, Spain, 43201 Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT00624195 | - | - | Completed | - | - |
| NCT00735072 | - | - | Completed | - | - |
| NCT00615563 | - | - | Completed | - | - |
| NCT00447902 | HIV Infections | Phase 3 | Terminated | - | United States, California ... more >> 1182.99.32 Boehringer Ingelheim Investigational Site Beverly Hills, California, United States United States, Florida 1182.99.31 Boehringer Ingelheim Investigational Site Fort Lauderdale, Florida, United States United States, Rhode Island 1182.99.12 Boehringer Ingelheim Investigational Site Providence, Rhode Island, United States United States, Texas 1182.99.1 Boehringer Ingelheim Investigational Site Austin, Texas, United States 1182.99.4 Boehringer Ingelheim Investigational Site Houston, Texas, United States Argentina 1182.99.54001 Capital Federal, Argentina Brazil 1182.99.55002 Hospital DIA Sacomã - São Paulo, Brazil 1182.99.55004 Unidade de Referência em doenças Infecciosas Preveníveis Santo André, Brazil 1182.99.55001 Universidade Federal de Sao Paulo Sao Paulo, Brazil 1182.99.55003 Centro de Referência e Treinamento - DST/AIDS Vila Mariana - Sao Paulo, Brazil France 1182.99.3301A Boehringer Ingelheim Investigational Site Garches, France 1182.99.3306G Boehringer Ingelheim Investigational Site Nantes, France 1182.99.3306K Boehringer Ingelheim Investigational Site Nantes, France 1182.99.3310C Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3310D Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3310E Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3310F Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3310G Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3310H Boehringer Ingelheim Investigational Site Nice cedex 3, France 1182.99.3304A Boehringer Ingelheim Investigational Site Paris, France 1182.99.3304C Boehringer Ingelheim Investigational Site Paris, France 1182.99.3302A Boehringer Ingelheim Investigational Site Perpignan, France Germany 1182.99.4909 Boehringer Ingelheim Investigational Site Düsseldorf, Germany Italy 1182.99.3912 Boehringer Ingelheim Investigational Site Ancona, Italy 1182.99.3901 Boehringer Ingelheim Investigational Site Milano, Italy 1182.99.3911 Boehringer Ingelheim Investigational Site Milano, Italy 1182.99.3916 Boehringer Ingelheim Investigational Site Milano, Italy 1182.99.3906 Boehringer Ingelheim Investigational Site Pavia, Italy Spain 1182.99.3402 Barcelona, Spain 1182.99.3407 Madrid, Spain Collapse << |
| NCT00447902 | - | - | Terminated | - | - |
| NCT00294372 | HIV Infections | Phase 2 | Terminated | - | Germany ... more >> Boehringer Ingelheim Investigational Site Berlin, Germany Boehringer Ingelheim Investigational Site Bochum, Germany Boehringer Ingelheim Investigational Site Bonn, Germany Boehringer Ingelheim Investigational Site Erlangen, Germany Boehringer Ingelheim Investigational Site Frankfurt/Main, Germany Boehringer Ingelheim Investigational Site Hamburg, Germany Boehringer Ingelheim Investigational Site Hannover, Germany Boehringer Ingelheim Investigational Site Heidelberg, Germany Boehringer Ingelheim Investigational Site Mainz, Germany Boehringer Ingelheim Investigational Site Munchen, Germany Boehringer Ingelheim Investigational Site Ulm, Germany Collapse << |
| NCT02253849 | Healthy | Phase 1 | Completed | - | - |
| NCT02251171 | Healthy | Phase 1 | Completed | - | - |
| NCT02229760 | HIV Infections | Phase 1 Phase 2 | Terminated | - | - |
| NCT02249442 | Hepatic Insufficiency | Phase 1 | Completed | - | - |
| NCT00517192 | - | - | Terminated | - | - |
| NCT02259855 | Hepatic Insufficiency | Phase 1 | Completed | - | - |
| NCT00517192 | HIV Infections | Phase 3 | Terminated | - | - |
| NCT00056641 | HIV Infections | Phase 2 | Completed | - | - |
| NCT02251132 | Healthy | Phase 1 | Completed | - | - |
| NCT02253836 | Healthy | Phase 1 | Completed | - | - |
| NCT00440271 | HIV Infections | Phase 3 | Terminated | - | - |
| NCT00440271 | - | - | Terminated | - | - |
| NCT02245438 | Healthy | Phase 1 | Terminated | - | - |
| NCT02251873 | Healthy | Phase 1 | Completed | - | - |
| NCT02253888 | Healthy | Phase 1 | Completed | - | - |
| NCT02253823 | Healthy | Phase 1 | Completed | - | - |
| NCT00076999 | HIV Infections | Phase 1 | Completed | - | - |
| NCT00097799 | - | - | - | - | - |
| NCT00144105 | HIV Infections | Phase 2 | Terminated | - | - |
| NCT01068925 | HIV Infections | Phase 1 | Completed | - | United States, Texas ... more >> GSK Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00976950 | - | - | Completed | - | Romania ... more >> Boehringer Ingelheim Investigational site 9 Arad, Romania Boehringer Ingelheim Investigational site 13 Bacau, Romania Boehringer Ingelheim Investigational site 17 Brasov, Romania Boehringer Ingelheim Investigational site 18 Brasov, Romania Boehringer Ingelheim Investigational site 19 Brasov, Romania Boehringer Ingelheim Investigational site 1 Bucuresti, Romania Boehringer Ingelheim Investigational site 2 Bucuresti, Romania Boehringer Ingelheim Investigational site 3 Bucuresti, Romania Boehringer Ingelheim Investigational site 4 Bucuresti, Romania Boehringer Ingelheim Investigational site 5 Bucuresti, Romania Boehringer Ingelheim Investigational site 6 Bucuresti, Romania Boehringer Ingelheim Investigational site 7 Bucuresti, Romania Boehringer Ingelheim Investigational site 8 Bucuresti, Romania Boehringer Ingelheim Investigational site 16 Constanta, Romania Boehringer Ingelheim Investigational site 11 Craiova, Romania Boehringer Ingelheim Investigational site 12 Craiova, Romania Boehringer Ingelheim Investigational site 15 Galati, Romania Boehringer Ingelheim Investigational site 20 Tg.Mures, Romania Boehringer Ingelheim Investigational site 10 Timisoara, Romania Boehringer Ingelheim Investigational site 14 Vaslui, Romania Collapse << |
| NCT02245269 | Healthy | Phase 1 | Completed | - | - |
| NCT02243553 | Healthy | Phase 1 | Completed | - | - |
| NCT02249416 | Healthy | Phase 1 | Completed | - | - |
| NCT00976950 | - | - | Completed | - | - |
| NCT00805857 | - | - | Withdrawn | - | - |
| NCT02239835 | HIV Infections | Phase 2 | Terminated | - | - |
| NCT02251769 | Healthy | Phase 1 | Completed | - | - |
| NCT02251223 | HIV Infections | Phase 1 Phase 2 | Completed | - | - |
| NCT00615290 | - | - | Completed | - | France ... more >> Boehringer Ingelheim Investigational Site Agen, France Boehringer Ingelheim Investigational Site Aix En Provence, France Boehringer Ingelheim Investigational Site Amiens, France Boehringer Ingelheim Investigational Site Argenteuil, France Boehringer Ingelheim Investigational Site Aulnay Sous Bois, France Boehringer Ingelheim Investigational Site Auxerre, France Boehringer Ingelheim Investigational Site Belfort, France Boehringer Ingelheim Investigational Site Besancon Cedex, France Boehringer Ingelheim Investigational Site Beziers, France Boehringer Ingelheim Investigational Site Bondy, France Boehringer Ingelheim Investigational Site Bordeaux, France Boehringer Ingelheim Investigational Site Briis Sous Forges, France Boehringer Ingelheim Investigational Site Cahors, France Boehringer Ingelheim Investigational Site Cannes, France Boehringer Ingelheim Investigational Site Chartres, France Boehringer Ingelheim Investigational Site Corbeil Essonnes, France Boehringer Ingelheim Investigational Site Creil, France Boehringer Ingelheim Investigational Site Creteil, France Boehringer Ingelheim Investigational Site DAX, France Boehringer Ingelheim Investigational Site Digne, France Boehringer Ingelheim Investigational Site Garches, France Boehringer Ingelheim Investigational Site La Roche Sur Yon, France Boehringer Ingelheim Investigational Site Le Mans, France Boehringer Ingelheim Investigational Site Le Petit Quevilly, France Boehringer Ingelheim Investigational Site Libourne, France Boehringer Ingelheim Investigational Site Lyon, France Boehringer Ingelheim Investigational Site Mantes La Jolie, France Boehringer Ingelheim Investigational Site Marseille, France Boehringer Ingelheim Investigational Site Metz, France Boehringer Ingelheim Investigational Site Mulhouse, France Boehringer Ingelheim Investigational Site Nancy, France Boehringer Ingelheim Investigational Site Nantes, France Boehringer Ingelheim Investigational Site Nevers, France Boehringer Ingelheim Investigational Site Nice, France Boehringer Ingelheim Investigational Site Orleans, France Boehringer Ingelheim Investigational Site Paris, France Boehringer Ingelheim Investigational Site Perigueux, France Boehringer Ingelheim Investigational Site Poitiers, France Boehringer Ingelheim Investigational Site Rouen, France Boehringer Ingelheim Investigational Site Saint Brieuc, France Boehringer Ingelheim Investigational Site Saint Nazaire, France Boehringer Ingelheim Investigational Site Saint-mande, France 1182.127.3301 Boehringer Ingelheim Investigational Site Strasbourg, France Boehringer Ingelheim Investigational Site Strasbourg, France Boehringer Ingelheim Investigational Site Suresnes, France Boehringer Ingelheim Investigational Site Toulouse, France Boehringer Ingelheim Investigational Site Valenciennes, France Boehringer Ingelheim Investigational Site Villeneuve St G, France Collapse << |
| NCT00615290 | - | - | Completed | - | - |
| NCT00054717 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00933205 | - | - | - | - | - |
| NCT02251119 | Healthy | Phase 1 | Completed | - | - |
| NCT00531206 | - | - | Completed | - | - |
| NCT02226978 | Healthy | Phase 1 | Completed | - | - |
| NCT00054717 | - | - | Completed | - | - |
| NCT02249130 | HIV Infections | Phase 2 | Completed | - | - |
| NCT02253862 | Healthy | Phase 1 | Completed | - | - |
| NCT02248883 | Healthy | Phase 1 | Completed | - | - |
| NCT00034866 | HIV Infections | Phase 2 | Completed | - | - |
| NCT00076999 | - | - | Completed | - | - |
| NCT00144287 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00531206 | - | - | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Aachen, Germany Boehringer Ingelheim Investigational Site Berlin, Germany Boehringer Ingelheim Investigational Site Bremen, Germany Boehringer Ingelheim Investigational Site Dortmund, Germany Boehringer Ingelheim Investigational Site Düsseldorf, Germany Boehringer Ingelheim Investigational Site Erlangen, Germany Boehringer Ingelheim Investigational Site Frankfurt/Main, Germany Boehringer Ingelheim Investigational Site Freiburg, Germany Boehringer Ingelheim Investigational Site Gießen, Germany Boehringer Ingelheim Investigational Site Halle/Saale, Germany Boehringer Ingelheim Investigational Site Hamburg, Germany Boehringer Ingelheim Investigational Site Hannover, Germany Boehringer Ingelheim Investigational Site Homburg/Saar, Germany Boehringer Ingelheim Investigational Site Karlsruhe, Germany Boehringer Ingelheim Investigational Site Krefeld, Germany Boehringer Ingelheim Investigational Site Köln, Germany Boehringer Ingelheim Investigational Site Leipzig, Germany Boehringer Ingelheim Investigational Site Magdeburg, Germany Boehringer Ingelheim Investigational Site Mainz, Germany Boehringer Ingelheim Investigational Site München, Germany Boehringer Ingelheim Investigational Site Münster, Germany Boehringer Ingelheim Investigational Site Nürnberg, Germany Boehringer Ingelheim Investigational Site Oldenburg, Germany Boehringer Ingelheim Investigational Site Osnabrück, Germany Boehringer Ingelheim Investigational Site Saarbrücken, Germany Boehringer Ingelheim Investigational Site Stuttgart, Germany Boehringer Ingelheim Investigational Site Wuppertal, Germany Collapse << |
| NCT02238314 | HIV Infections | Phase 2 | Completed | - | - |
| NCT00144170 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00530920 | HIV Infections | Phase 2 | Completed | - | Germany ... more >> 1182.107.49002 Boehringer Ingelheim Investigational Site Berlin, Germany 1182.107.49004 Boehringer Ingelheim Investigational Site Berlin, Germany 1182.107.49003 Boehringer Ingelheim Investigational Site Frankfurt/Main, Germany 1182.107.49001 Boehringer Ingelheim Investigational Site München, Germany Italy 1182.107.39001 Boehringer Ingelheim Investigational Site Antella (fi), Italy 1182.107.39009 Boehringer Ingelheim Investigational Site Bari, Italy 1182.107.39007 Boehringer Ingelheim Investigational Site Ferrara, Italy 1182.107.39011 Boehringer Ingelheim Investigational Site Palermo, Italy Spain 1182.107.34001 Boehringer Ingelheim Investigational Site Barcelona, Spain 1182.107.34002 Boehringer Ingelheim Investigational Site Barcelona, Spain 1182.107.34003 Boehringer Ingelheim Investigational Site L'Hospitalet de Llobregat, Spain 1182.107.34004 Boehringer Ingelheim Investigational Site Madrid, Spain Collapse << |
| NCT00352066 | HIV Infection | Phase 1 | Completed | - | United States, Michigan ... more >> Jasper Clinic Inc Kalamazoo, Michigan, United States, 49007 Collapse << |
| NCT00144170 | - | - | Completed | - | - |
| NCT02251145 | Healthy | Phase 1 | Completed | - | - |
| NCT02245451 | Healthy | Phase 1 | Completed | - | - |
| NCT00275444 | HIV Infections | Phase 2 | Completed | - | - |
| NCT00146328 | HIV Infections | Phase 2 Phase 3 | Completed | - | - |
| NCT00062660 | - | - | - | - | - |
| NCT02251795 | Healthy | Phase 1 | Completed | - | - |
| NCT00530920 | - | - | Completed | - | - |
| NCT00344123 | HIV Infections | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21287 Collapse << |
| NCT02253875 | Healthy | Phase 1 | Completed | - | - |
| NCT00607958 | HIV Infections | Phase 4 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Collapse << |
| NCT00486330 | - | - | Completed | - | - |
| NCT00146328 | - | - | Completed | - | - |
| NCT02253797 | Healthy | Phase 1 | Completed | - | - |
| NCT02257021 | Healthy | Phase 1 | Completed | - | - |
| NCT02244190 | Healthy | Phase 1 | Completed | - | - |
| NCT02226991 | Healthy | Phase 1 | Completed | - | - |
| NCT00486330 | HIV Infections | Not Applicable | Completed | - | United States, Connecticut ... more >> Yale University School of Medicine AIDS Program New Haven, Connecticut, United States, 06510 Collapse << |
| NCT02251158 | Healthy | Phase 1 | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
