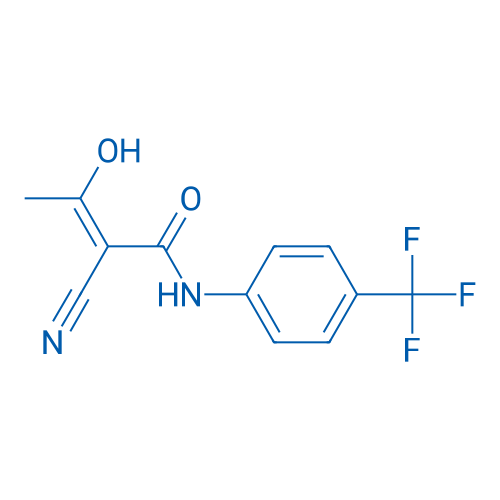
CAS No.: 163451-81-8
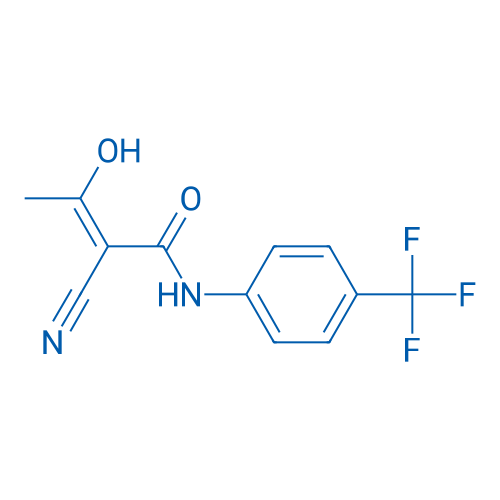
Teriflunomide/特立氟胺 Catalog No. CSN13699
Synonyms: A77 1726;HMR 1726;Flucyamide
Teriflunomide, the active metabolite of an approved antirheumatic drug leflunomide, is an emerging oral therapy for multiple sclerosis (MS). It reversibly inhibits dihydroorotate dehydrogenase, the rate-limiting step in the de novo synthesis of pyrimidines.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02509052 Recurrent Plasma Cell Myeloma ... more >> Refractory Plasma Cell Myeloma Collapse << Phase 1 Phase 2 Active, not recruiting June 2019 United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << NCT02559765 - - Completed - United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << NCT03709446 Breast Neoplasms ... more >> Breast Diseases Metastatic Triple Negative Breast Cancer Collapse << Phase 1 Phase 2 Not yet recruiting October 20, 2021 United States, New York ... more >> Icahn School of Medicine at Mount Sinai Not yet recruiting New York, New York, United States, 10029 Contact: Charles Shapiro, MD 212-241-3131 charles.shapiro@mssm.edu Contact: Joni Gomes, BSN 212-824-7076 joni.gomes@mssm.edu Collapse << - 更多
- 参考文献
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 163451-81-8 | 储存条件 |
|
|||||
| 分子式 | C12H9F3N2O2 | 运输 | 蓝冰 | |||||
| 分子量 | 270.21 | 别名 | A77 1726;HMR 1726;Flucyamide;SU 20;特立氟胺 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02509052 | Recurrent Plasma Cell Myeloma ... more >> Refractory Plasma Cell Myeloma Collapse << | Phase 1 Phase 2 | Active, not recruiting | June 2019 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << |
| NCT02559765 | - | - | Completed | - | United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << |
| NCT03709446 | Breast Neoplasms ... more >> Breast Diseases Metastatic Triple Negative Breast Cancer Collapse << | Phase 1 Phase 2 | Not yet recruiting | October 20, 2021 | United States, New York ... more >> Icahn School of Medicine at Mount Sinai Not yet recruiting New York, New York, United States, 10029 Contact: Charles Shapiro, MD 212-241-3131 charles.shapiro@mssm.edu Contact: Joni Gomes, BSN 212-824-7076 joni.gomes@mssm.edu Collapse << |
| NCT02342652 | Healthy Elderly Subjects (Age ... more >>≥55 Years) Collapse << | Phase 1 | Completed | - | Singapore ... more >> Singapore, Singapore Collapse << |
| NCT03302442 | - | - | Completed | - | France ... more >> Nantes University Hospital Nantes, France, 44093 Collapse << |
| NCT03535298 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Not yet recruiting | September 2023 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Tammy Skaramagas, BA 216-445-6724 skaramt1@ccf.org Principal Investigator: Daniel Ontaneda, MD, MSc United Kingdom University of Nottingham Not yet recruiting Nottingham, United Kingdom, NG7 2UH Contact: Sara Wilkins +44 115 9249924 ext 66816 Sara.wilkins@nuh.nhs.uk Principal Investigator: Nikos Evangelou, MD, DPhil Collapse << |
| NCT03500328 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Not Applicable | Recruiting | October 31, 2022 | - |
| NCT00273364 | Multiple Sclerosis | Phase 2 | Active, not recruiting | December 2018 | United States, Illinois ... more >> Northwestern University, Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT02776072 | - | - | Completed | - | - |
| NCT03399981 | - | - | Active, not recruiting | December 31, 2023 | United States, Massachusetts ... more >> Research Site Cambridge, Massachusetts, United States, 02142 Collapse << |
| NCT02651545 | - | - | Recruiting | November 30, 2020 | United States, New York ... more >> Buffalo General Hospital, D3 Recruiting Buffalo, New York, United States, 14203 Contact: Ralph HB Benedict, PhD 716-859-3484 benedict@buffalo.edu Collapse << |
| NCT03308994 | - | - | Recruiting | December 31, 2018 | France ... more >> Groupement des Hôpitaux de l'Institut Catholique de Lille (HICL) Recruiting Lille, Hauts-de-France, France, 59020 Contact: Arnaud Kwiatkowski, MD-PhD 33320874901 kwiatkowski.arnaud@ghicl.net University Hospital of Lille Recruiting Lille, Hauts-de-France, France, 59037 Contact: Patrick Vermersch, Professor 33320445765 patrick.vermersch@univ-lille2.fr University Hospital Center of Caen Recruiting Caen, Normandie, France, 14033 Contact: Gilles-Louis Defer, Professor 33231064617 defer-gi@chu-caen.fr Contact: Damien Chevanne, PhD 33231064617 chevanne-d@chu-caen.fr University Hospital of Rouen Recruiting Rouen, Normandie, France, 76031 Contact: Bertrand Bourre, MD 33232888990 Bertrand.Bourre@chu-rouen.fr Collapse << |
| NCT03122652 | Multiple Sclerosis | Phase 3 | Recruiting | September 25, 2021 | France ... more >> CHU de Bordeaux Recruiting Bordeaux, France, 33000 Contact: Bruno BROCHET, MD Principal Investigator: Bruno BROCHET, MD CHRU de Lille Recruiting Lille, France, 59000 Contact: Patrick VERMERSCH, MD Contact: , MD Principal Investigator: Patrick VERMERSCH Hospices Civils de Lyon Recruiting Lyon, France, 69677 Contact: Sandra VUKUSIC, MD Principal Investigator: Sandra VUKUSIC, MD Sub-Investigator: Françoise DURAND-DUBIEF, MD CHRU de Montpellier Recruiting Montpellier, France, 34000 Contact: Pierre LABAUGE, MD Principal Investigator: Pierre LABAUGE, MD CHU de Nantes Recruiting Nantes, France Contact: Sandrine WIERTLEWSKI Principal Investigator: Sandrine WIERTLEWSKI, MD CHU de Nice Recruiting Nice, France, 06000 Contact: Cassandre LANDES 492034126 ext 33 landes.c@chu-nice.fr Principal Investigator: Christine LEBRUN-FRENAY, MD, PhD CHU de Nîmes Recruiting Nîmes, France Contact: Eric THOUVENOT Principal Investigator: Eric THOUVENOT, MD APHP - Hôpital La Pitié Salpêtrière Recruiting Paris, France, 75013 Contact: Anne-Caroline PAPEIX, MD Principal Investigator: Anne-Caroline PAPEIX, MD Sub-Investigator: Céline LOUAPRE, MD Sub-Investigator: Elisabeth MAILLART, MD CHU de Rennes Recruiting Rennes, France, 35000 Contact: Emmanuelle LE PAGE, MD Principal Investigator: Emmanuelle LE PAGE, MD CHU de Strasbourg Recruiting Strasbourg, France, 67000 Contact: Jérôme DE SEZE, MD Principal Investigator: Jérôme DE SEZE, MD CHU de Toulouse Active, not recruiting Toulouse, France, 31000 Germany University hospital of Charité Not yet recruiting Berlin, Germany Contact: Jens WÜRFEL, MD Principal Investigator: Jens WÜRFEL, MD University Hospital of the Ruhr - University Bochum Not yet recruiting Bochum, Germany, D-44791 Contact: Carsten LUKAS, MD Principal Investigator: Carsten LUKAS, MD University of Heidelberg Not yet recruiting Heidelberg, Germany, 69120 Contact: Brigitte WILDEMANN, MD Principal Investigator: Brigitte WILDEMANN, MD University Medical Center of the Johannes Gutenberg Not yet recruiting Mainz, Germany, 55131 Contact: Frauke ZIPP, MD Principal Investigator: Frauke ZIPP, MD Technische Universität München Not yet recruiting Munich, Germany, 81675 Contact: Mark MÜHLAU, MD Principal Investigator: Mark MÜHLAU, MD Switzerland University Hospital Basel Not yet recruiting Basel, Switzerland, 4031 Contact: Ludwig Kappos, MD Principal Investigator: Ludwig Kappos, MD Turkey Hacettepe University Not yet recruiting Ankara, Turkey Contact: Rana Karabudak, MD Principal Investigator: Rana Karabudak, MD Mustafa Kemal University Active, not recruiting Antakya, Turkey Uludag University School of Medicine Active, not recruiting Bursa, Turkey Istanbul University Active, not recruiting Istanbul, Turkey Kocaeli University School of Medicine Active, not recruiting Kocaeli, Turkey Ondokuz Mayis University, Faculty of Medicine Active, not recruiting Samsun, Turkey United Kingdom Queen's Medical Centre Not yet recruiting Nottingham, United Kingdom Contact: Nikos Evangelou, MD Principal Investigator: Nikos Evangelou, MD Collapse << |
| NCT03277248 | Relapsing Multiple Sclerosis (... more >>RMS) Collapse << | Phase 3 | Recruiting | September 30, 2021 | United States, Arizona ... more >> TG Therapeutics RMS Investigational Trial site Recruiting Phoenix, Arizona, United States, 58018 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Colorado TG Therapeutics RMS Investigational Trial Site Recruiting Aurora, Colorado, United States, 80045 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Florida TG Therapeutics RMS Investigational Trial Site Recruiting Tampa, Florida, United States, 33612 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Kentucky TG Therapeutics RMS Investigational Trial Site Recruiting Lexington, Kentucky, United States, 40513 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Missouri TG Therapeutics RMS Investigational Trial Site Recruiting Chesterfield, Missouri, United States, 63017 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Nevada TG Therapeutics RMS Investigational Trial Site Recruiting Las Vegas, Nevada, United States, 89106 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, New Jersey TG Therapeutics RMS Investigational Trial Site Recruiting Teaneck, New Jersey, United States, 07666 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, New Mexico TG Therapeutics RMS Investigational Trial Site Recruiting Albuquerque, New Mexico, United States, 87131 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, New York TG Therapeutics RMS Investigational Trial Site Recruiting Patchogue, New York, United States, 11772 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Ohio TG Therapeutics RMS Investigational Trial site Recruiting Columbus, Ohio, United States, 43221 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Pennsylvania TG Therapeutics RMS Investigational Trial Site Recruiting Pittsburgh, Pennsylvania, United States, 15212 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Texas TG Therapeutics RMS Investigational Trial site Recruiting San Antonio, Texas, United States, 78258 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com United States, Washington TG Therapeutics RMS Investigational Trial site Recruiting Seattle, Washington, United States, 98122 Contact: TG Therapeutics Clinical Support Team 212-554-4484 RMSclinicalsupport@tgtxinc.com Collapse << |
| NCT03768648 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Not Applicable | Not yet recruiting | January 2019 | France ... more >> CHU de Bordeaux - Service de neurologie Not yet recruiting Bordeaux, France Contact: Bruno BROCHET, Prof 05 56 79 55 21 ext +33 bruno.brochet@chu-bordeaux.fr Contact: Mathilde DELOIRE, PhD 05 57 82 12 75 ext +33 mathilde.deloire@chu-bordeaux.fr Principal Investigator: Bruno BROCHET, Prof Sub-Investigator: Aurélie RUET, MD Sub-Investigator: Jean-Christophe OUALLET, MD Sub-Investigator: Cécile DULAU, MD Collapse << |
| NCT02679885 | - | - | Recruiting | December 2017 | United States, Connecticut ... more >> Multiple Sclerosis Treatment Center at Griffin Hospital Recruiting Derby, Connecticut, United States, 06418 Contact: Joseph Guarnaccia, MD 203-732-1290 mstreatmentcenters@griffinhealth.org Collapse << |
| NCT01239459 | Renal Impairment | Phase 1 | Completed | - | Germany ... more >> Sanofi-Aventis Administrative Office Berlin, Germany Collapse << |
| NCT03277261 | Relapsing Multiple Sclerosis (... more >>RMS) Collapse << | Phase 3 | Recruiting | September 30, 2021 | United States, California ... more >> TG Therapeutics RMS Investigational Trial Site Recruiting Carlsbad, California, United States, 92001 TG Therapeutics RMS Investigational Trial Site Recruiting Long Beach, California, United States, 90808 TG Therapeutics RMS Investigational Trial Site Recruiting Pasadena, California, United States, 91105 TG Therapeutics RMS Investigational Trial Site Recruiting Stanford, California, United States, 94305 United States, Florida TG Therapeutics RMS Investigational Trial Site Recruiting Miami, Florida, United States, 33136 United States, Illinois TG Therapeutics RMS Investigational Trial Site Recruiting Northbrook, Illinois, United States, 60062 United States, Kansas TG Therapeutics RMS Investigational Trial Site Recruiting Kansas City, Kansas, United States, 66160 United States, Michigan TG Therapeutics RMS Investigational Trial Site Recruiting Detroit, Michigan, United States, 48201 United States, New York TG Therapeutics RMS Investigational Trial Site Recruiting Amherst, New York, United States, 14226 United States, Ohio TG Therapeutics RMS Investigational Trial Site Recruiting Westerville, Ohio, United States, 43081 United States, Tennessee TG Therapeutics RMS Investigational Trial Site Recruiting Franklin, Tennessee, United States, 37064 TG Therapeutics RMS Investigational Trial Site Recruiting Knoxville, Tennessee, United States, 37922 United States, Texas TG Therapeutics RMS Investigational Trial Site Recruiting Dallas, Texas, United States, 75246 TG Therapeutics RMS Investigational Site Recruiting Round Rock, Texas, United States, 78681 Collapse << |
| NCT00811395 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Wien, Austria Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Spain Sanofi-Aventis Administrative Office Barcelona, Spain United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT01863888 | Multiple Sclerosis | Phase 3 | Completed | - | Belgium ... more >> Investigational Site Number 056001 Brussels, Belgium, 1070 Investigational Site Number 056002 Overpelt, Belgium, 3900 Investigational Site Number 056003 Sijsele-Damme, Belgium, 8340 Germany Investigational Site Number 276-003 Bad Mergentheim, Germany, 97980 Investigational Site Number 276-004 Hannover, Germany, 30625 Investigational Site Number 276-005 Marburg, Germany, 35043 Investigational Site Number 276-007 Mönchengladbach, Germany, 41061 Investigational Site Number 276-001 Münster, Germany, 48149 Investigational Site Number 276-002 Ulm, Germany, 89073 Netherlands Investigational Site Number 528001 Sittard-Geleen, Netherlands, 6162BG Collapse << |
| NCT00811395 | - | - | Completed | - | - |
| NCT00228163 | Multiple Sclerosis | Phase 2 | Completed | - | Canada ... more >> Investigational Site Number 16 Calgary, Canada, T2N 2T9 Investigational Site Number 10 Halifax, Canada, B3H 1V7 Investigational Site Number 15 London, Canada, N6A 5A5 Investigational Site Number 12 Montreal, Canada, H2L 4M1 Investigational Site Number 11 Montreal, Canada, H3A 2B4 Investigational Site Number 13 Ottawa, Canada, K1H 8L6 Investigational Site Number 18 Quebec, Canada, G1J 1Z4 Investigational Site Number 14 Toronto, Canada, M5B 1W8 Investigational Site Number 17 Vancouver, Canada, V6T 2B5 Investigational Site Number 19 Winnipeg, Canada, R3E 3N4 France Investigational Site Number 24 Clermont Ferrand Cedex 1, France, 63003 Investigational Site Number 21 Lyon Cedex 03, France, 69394 Investigational Site Number 30 Marseille Cedex 5, France, 13385 Investigational Site Number 23 Montpellier Cedex 5, France, 34000 Investigational Site Number 28 Nice Cedex, France, 06002 Investigational Site Number 27 Rennes Cedex, France, 35033 Collapse << |
| NCT02833714 | - | - | Terminated | - | United States, North Carolina ... more >> University of North Carolina Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT02425644 | Multiple Sclerosis | Phase 3 | Active, not recruiting | May 15, 2019 | - |
| NCT01881191 | - | - | Completed | - | United States, New York ... more >> Buffalo Neuroimaging Analysis Center Buffalo, New York, United States, 14203 Collapse << |
| NCT03368677 | - | - | Recruiting | November 2024 | Finland ... more >> Turku PET Centre Recruiting Turku, Finland Proper, Finland, 20520 Contact: Laura Airas, MD,professor 023130000 laura.airas@utu.fi Collapse << |
| NCT00883337 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01403376 | - | - | Completed | - | - |
| NCT01403376 | Multiple Sclerosis | Phase 2 | Completed | - | Austria ... more >> Investigational Site Number 040001 Wien, Austria, 1010 Canada Investigational Site Number 124005 Calgary, Canada, T2N 2T9 Investigational Site Number 124003 Greenfield Park, Canada, J4V 2J2 Investigational Site Number 124002 London, Canada, N6A 5A5 Investigational Site Number 124007 Montreal, Canada, H3A 2B4 Investigational Site Number 124008 Ottawa, Canada, K1H 8L6 Investigational Site Number 124001 Quebec, Canada, G1J 1Z4 Investigational Site Number 124009 Toronto, Canada, M5B 1W8 Germany Investigational Site Number 276003 Berlin, Germany, 10785 Investigational Site Number 276001 Essen, Germany, 45122 Investigational Site Number 276002 Münster, Germany, 48149 Russian Federation Investigational Site Number 643002 Nizhny Novgorod, Russian Federation, 603076 Ukraine Investigational Site Number 804002 Ivano-Frankovsk, Ukraine, 76008 Investigational Site Number 804001 Kharkiv, Ukraine, 61018 Collapse << |
| NCT00475865 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Vienna, Austria Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00751881 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00803049 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00489489 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy Spain Sanofi-Aventis Administrative Office Barcelona, Spain Collapse << |
| NCT00883337 | - | - | Completed | - | - |
| NCT00751881 | - | - | Completed | - | - |
| NCT00134563 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00475865 | - | - | Completed | - | - |
| NCT00803049 | - | - | Completed | - | - |
| NCT01252355 | - | - | Terminated(Sponsor decision to... more >> prematurely stop the study, not linked to any safety concern.) Collapse << | - | - |
| NCT02587195 | Multiple Sclerosis | Phase 3 | Active, not recruiting | January 18, 2019 | France ... more >> CHU de Besançon Besançon, France, 25030 CHU de Lille Lille, France, 59037 CHU de Montpellier Montpellier, France, 34295 CHU de Nice Nice, France, 06000 CHU de Strasbourg Strasbourg, France, 67098 Collapse << |
| NCT00134563 | - | - | Completed | - | - |
| NCT00622700 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01252355 | Multiple Sclerosis Relapse | Phase 3 | Terminated(Sponsor decision to... more >> prematurely stop the study, not linked to any safety concern.) Collapse << | - | - |
| NCT00489489 | - | - | Completed | - | - |
| NCT02792231 | Relapsing Multiple Scelrosis | Phase 3 | Recruiting | May 15, 2019 | - |
| NCT02792218 | Relapsing Multiple Sclerosis | Phase 3 | Active, not recruiting | May 16, 2019 | - |
| NCT02046629 | Multiple Sclerosis | Phase 1 | Completed | - | China ... more >> Investigational Site Number 156001 Shanghai, China, 200025 Collapse << |
| NCT02201108 | Multiple Sclerosis | Phase 3 | Active, not recruiting | September 2021 | - |
| NCT03198351 | - | - | Recruiting | February 1, 2022 | United States, California ... more >> Investigator Recruiting San Diego, California, United States, 92093 Collapse << |
| NCT00622700 | - | - | Completed | - | - |
| NCT01895335 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT01487096 | Multiple Sclerosis | Phase 2 | Completed | - | Canada, Ontario ... more >> Canada Toronto, Ontario, Canada France sanofi-aventis France Lyon, France Collapse << |
| NCT01895335 | - | - | Completed | - | - |
| NCT03464448 | - | - | Recruiting | October 2021 | United States, Michigan ... more >> University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Principal Investigator: Yang Mao-Draayer, MD/PHD Collapse << |
| NCT02490982 | - | - | Enrolling by invitation | December 2019 | Canada, Quebec ... more >> CHUM Montréal, Quebec, Canada, H2L 4M1 Collapse << |
| NCT01970410 | Multiple Sclerosis | Phase 4 | Active, not recruiting | June 2018 | United States, Arizona ... more >> Phoenix Neurological Associates, Ltd Phoenix, Arizona, United States, 85018 United States, New York Multiple Sclerosis Center of Northeastern New York Latham, New York, United States, 12110 United States, Oregon Providence Multiple Sclerosis Center Portland, Oregon, United States, 97225 Collapse << |
| NCT02263547 | Teriflunomide Elimination ... more >> Healthy Volunteers Collapse << | Phase 1 | Terminated(PK time points were... more >> not being met as expected. Determination that risks outweighed benefit.) Collapse << | - | United States, Florida ... more >> USF Carol and Frank Morsani Center for Advanced Healthcare Tampa, Florida, United States, 33612 Collapse << |
| NCT03561402 | - | - | Recruiting | February 28, 2019 | Canada, Quebec ... more >> Department of Pathology Recruiting Montréal, Quebec, Canada, H3A 2B4 Contact: David Haegert, MD Collapse << |
| NCT03526224 | - | - | Active, not recruiting | November 2019 | United States, New York ... more >> Buffalo Neuroimaging Analysis Center Buffalo, New York, United States, 14203 Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
