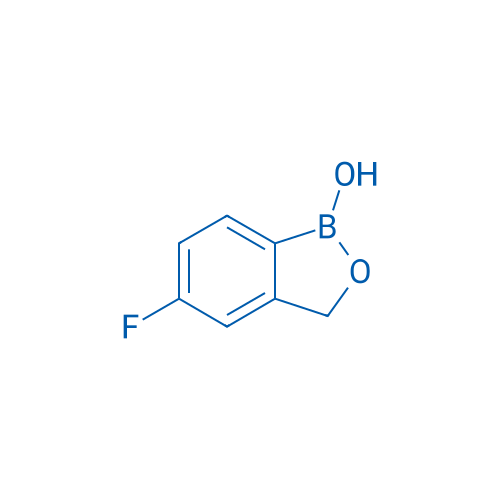
CAS No.: 174671-46-6
Tavaborole/他伐硼罗 Catalog No. CSN13870
Synonyms: AN-2690;Kerydin;他伐硼罗
AN-2690 is a broad-spectrum antifungal agent used to treat onychomycosis.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00680095 Onychomycosis Phase 1 Completed - United States, Virginia ... more >> The Education and Research Foundation, Inc. Lynchburg, Virginia, United States, 24501 Collapse << NCT00679965 Distal, Subungual Onychomycosi... more >>s Collapse << Phase 2 Completed - Mexico ... more >> Unidad de Investigación en Salud (UIS) Chihuahua, Mexico Hospital "Dr. Angel Leaño" Guadalajara, Mexico Instituto Dermatologico Jalisciense Guadalajara, Mexico Centro Dermatologico Pascua Mexico City, Mexico CIF-BIOTEC Medica Sur. Mexico City, Mexico IMIC Mexico City, Mexico Hospital Universitario Dr. José Eleuterio González Monterrey, Mexico MIRC / OCA Hospital Monterrey, Mexico ISSEMYM Toluca, Mexico Collapse << NCT01278394 Onychomycosis Phase 2 Completed - Mexico ... more >> Instituto Dermatologico Jalisciense Guadalajara, Mexico IMIC Mexico City, Mexico Collapse << - 更多
- 参考文献
- [1] Zhou XL, Tan M, Wang M, Chen X, Wang ED. Post-transfer editing by a eukaryotic leucyl-tRNA synthetase resistant to the broad-spectrum drug AN2690. Biochem J. 2010;430(2):325‐333
- [2] Baker SJ, Zhang YK, et al. Discovery of a new boron-containing antifungal agent, 5-fluoro-1,3-dihydro-1-hydroxy-2,1- benzoxaborole (AN2690), for the potential treatment of onychomycosis. J Med Chem. 2006 Jul 27;49(15):4447-50.
- [3] Gupta AK, Daigle D. Tavaborole (AN-2690) for the treatment of onychomycosis of the toenail in adults. Expert Rev Anti Infect Ther. 2014;12(7):735‐742
- [4] Markinson B, Ghannoum M, Winter T, Rycerz A, Rock F, Gupta AK. Examining the Benefits of the Boron-Based Mechanism of Action and Physicochemical Properties of Tavaborole in the Treatment of Onychomycosis. J Am Podiatr Med Assoc. 2018;108(1):12‐19
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 174671-46-6 | 储存条件 |
|
|||||||
| 分子式 | C7H6BFO2 | 运输 | 蓝冰 | |||||||
| 分子量 | 151.93 | 别名 | AN-2690;Kerydin;他伐硼罗 | |||||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
