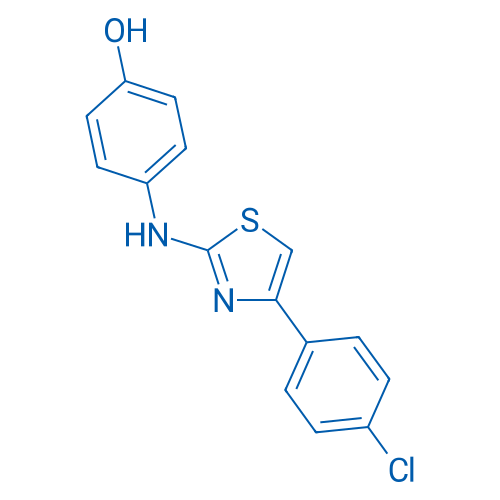
CAS No.: 312636-16-1
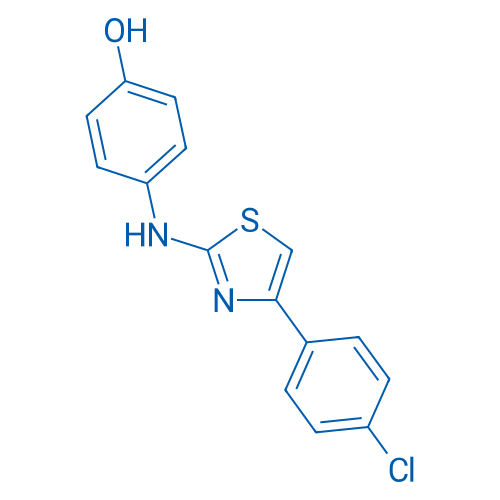
SKI II Catalog No. CSN18688
Synonyms: SphK-I2;Sphingosine Kinase Inhibitor 2
SKI II is an inhibitor of non ATP-competitive sphingosine kinase (SphK) with IC50 of 0.5 μM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 SPHK
IC50:0.5μM- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00976677 Recurrent Non-small Cell Lung ... more >>Cancer Stage IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Collapse << Phase 2 Terminated - - NCT00976677 - - Terminated - - NCT00951496 Malignant Ovarian Mixed Epithe... more >>lial Tumor Ovarian Brenner Tumor Ovarian Clear Cell Cystadenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Mucinous Cystadenocarcinoma Ovarian Serous Cystadenocarcinoma Stage IIA Fallopian Tube Cancer Stage IIA Ovarian Cancer Stage IIB Fallopian Tube Cancer Stage IIB Ovarian Cancer Stage IIC Fallopian Tube Cancer Stage IIC Ovarian Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Cancer Stage IIIA Primary Peritoneal Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Cancer Stage IIIB Primary Peritoneal Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Cancer Stage IIIC Primary Peritoneal Cancer Undifferentiated Ovarian Carcinoma Collapse << Phase 3 Active, not recruiting March 15, 2022 - - 更多
- 参考文献
- [1] French KJ, Upson JJ, et al. Antitumor activity of sphingosine kinase inhibitors. J Pharmacol Exp Ther. 2006 Aug;318(2):596-603.
- [2] French KJ, Schrecengost RS, et al. Discovery and evaluation of inhibitors of human sphingosine kinase. Cancer Res. 2003 Sep 15;63(18):5962-9.
- [3] Potì F, Ceglarek U, Burkhardt R, Simoni M, Nofer JR. SKI-II--a sphingosine kinase 1 inhibitor--exacerbates atherosclerosis in low-density lipoprotein receptor-deficient (LDL-R-/-) mice on high cholesterol diet. Atherosclerosis. 2015;240(1):212-215.
- [4] Liu Y, Zhu Z, Cai H, Liu Q, Zhou H, Zhu Z. SKI-II reverses the chemoresistance of SGC7901/DDP gastric cancer cells. Oncol Lett. 2014;8(1):367-373.
- [5] French KJ, Upson JJ, Keller SN, Zhuang Y, Yun JK, Smith CD. Antitumor activity of sphingosine kinase inhibitors. J Pharmacol Exp Ther. 2006;318(2):596-603.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 312636-16-1 | 储存条件 |
|
|||||||
| 分子式 | C15H11ClN2OS | 运输 | 蓝冰 | |||||||
| 分子量 | 302.78 | 别名 | SphK-I2;Sphingosine Kinase Inhibitor 2 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00976677 | Recurrent Non-small Cell Lung ... more >>Cancer Stage IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Collapse << | Phase 2 | Terminated | - | - |
| NCT00976677 | - | - | Terminated | - | - |
| NCT00951496 | Malignant Ovarian Mixed Epithe... more >>lial Tumor Ovarian Brenner Tumor Ovarian Clear Cell Cystadenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Mucinous Cystadenocarcinoma Ovarian Serous Cystadenocarcinoma Stage IIA Fallopian Tube Cancer Stage IIA Ovarian Cancer Stage IIB Fallopian Tube Cancer Stage IIB Ovarian Cancer Stage IIC Fallopian Tube Cancer Stage IIC Ovarian Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Cancer Stage IIIA Primary Peritoneal Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Cancer Stage IIIB Primary Peritoneal Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Cancer Stage IIIC Primary Peritoneal Cancer Undifferentiated Ovarian Carcinoma Collapse << | Phase 3 | Active, not recruiting | March 15, 2022 | - |
| NCT00003596 | Anal Cancer | Phase 3 | Completed | - | - |
| NCT00951496 | - | - | Active, not recruiting | - | - |
| NCT00872989 | Fallopian Tube Cancer ... more >> Ovarian Cancer Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00872989 | - | - | Completed | - | - |
| NCT00485667 | Postoperative Pain | Phase 1 | Completed | - | United Kingdom ... more >> Richmond Pharmacology Ltd London, United Kingdom, SW19 0RE Collapse << |
| NCT01814813 | Recurrent Glioblastoma ... more >> Recurrent Adult Brain Tumor Gliosarcoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00798603 | Lung Cancer | Phase 2 | Completed | - | - |
| NCT00798603 | - | - | Completed | - | - |
| NCT00313586 | - | - | Completed | - | - |
| NCT00313586 | Acute Myeloid Leukemia Arising... more >> From Previous Myelodysplastic Syndrome Adult Acute Myeloid Leukemia in Remission Adult Acute Myeloid Leukemia With Inv(16)(p13.1q22); CBFB-MYH11 Adult Acute Myeloid Leukemia With t(16;16)(p13.1;q22); CBFB-MYH11 Adult Acute Myeloid Leukemia With t(8;21)(q22;q22); RUNX1-RUNX1T1 Adult Acute Myeloid Leukemia With t(9;11)(p22;q23); MLLT3-MLL Adult Acute Promyelocytic Leukemia With t(15;17)(q22;q12); PML-RARA Alkylating Agent-Related Acute Myeloid Leukemia Chronic Myelomonocytic Leukemia de Novo Myelodysplastic Syndrome Previously Treated Myelodysplastic Syndrome Recurrent Adult Acute Myeloid Leukemia Secondary Acute Myeloid Leukemia Secondary Myelodysplastic Syndrome Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 2 | Completed | - | - |
| NCT00550836 | - | - | Completed | - | - |
| NCT00410813 | Breast Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00410813 | - | - | Completed | - | - |
| NCT00986674 | Recurrent Non-small Cell Lung ... more >>Cancer Stage IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00986674 | - | - | Completed | - | - |
| NCT01256398 | Acute Lymphoblastic Leukemia ... more >> Adult B Acute Lymphoblastic Leukemia With t(9;22)(q34.1;q11.2); BCR-ABL1 Untreated Adult Acute Lymphoblastic Leukemia Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00550836 | Pancreatic Cancer | Phase 2 | Completed | - | - |
| NCT00776100 | Lung Cancer | Phase 2 | Terminated(Study was terminate... more >>d prematurely due to slow accrual.) Collapse << | - | - |
| NCT00776100 | - | - | Terminated(Study was terminate... more >>d prematurely due to slow accrual.) Collapse << | - | - |
| NCT02101788 | Low Grade Ovarian Serous Adeno... more >>carcinoma Micropapillary Serous Carcinoma Ovarian Serous Adenocarcinoma Primary Peritoneal Serous Adenocarcinoma Recurrent Ovarian Carcinoma Recurrent Primary Peritoneal Carcinoma Collapse << | Phase 2 Phase 3 | Active, not recruiting | - | - |
| NCT02339571 | Recurrent Melanoma of the Skin... more >> Stage III Cutaneous Melanoma AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Collapse << | Phase 2 Phase 3 | Suspended(Scheduled Interim Mo... more >>nitoring) Collapse << | - | - |
| NCT02839343 | Pancreatic Adenocarcinoma ... more >> Borderline Resectable Adenocarcinoma of the Head of the Pancrease Collapse << | Phase 2 | Suspended(Interim analysis) | - | - |
| NCT02154490 | Recurrent Squamous Cell Lung C... more >>arcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 2 Phase 3 | Recruiting | April 1, 2022 | - |
| NCT01086605 | Breast Cancer | Phase 2 | Completed | - | - |
| NCT01515787 | Colorectal Cancer | Phase 2 Phase 3 | Recruiting | - | - |
| NCT03092674 | Acute Myeloid Leukemia ... more >> Myelodysplastic Syndrome Myelodysplastic Syndrome With Excess Blasts-2 Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 2 Phase 3 | Suspended(Other - Safety data ... more >>review, unscheduled) Collapse << | August 1, 2023 | - |
| NCT02498600 | Recurrent Fallopian Tube Carci... more >>noma Recurrent Ovarian Carcinoma Recurrent Primary Peritoneal Carcinoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00378703 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Carcinoma Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 2 | Completed | - | - |
| NCT01086605 | - | - | Completed | - | - |
| NCT01091454 | - | - | Completed | - | - |
| NCT01091454 | Triple-negative Breast Cancer | Phase 2 | Completed | - | - |
| NCT02642042 | KRAS Gene Mutation ... more >> Recurrent Non-Small Cell Lung Carcinoma Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 2 | Active, not recruiting | October 15, 2020 | - |
| NCT00684983 | HER2/Neu Positive ... more >> Recurrent Breast Carcinoma Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00792701 | Lung Cancer | Phase 2 | Completed | - | - |
| NCT00942357 | Endometrial Clear Cell Adenoca... more >>rcinoma Endometrial Serous Adenocarcinoma Stage IA Uterine Corpus Cancer Stage IB Uterine Corpus Cancer Stage II Uterine Corpus Cancer Stage IIIA Uterine Corpus Cancer Stage IIIB Uterine Corpus Cancer Stage IIIC Uterine Corpus Cancer Stage IVA Uterine Corpus Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00378703 | - | - | Completed | - | - |
| NCT01189266 | Anaplastic Astrocytoma ... more >> Anaplastic Oligoastrocytoma Brain Stem Glioma Childhood Glioblastoma Giant Cell Glioblastoma Gliosarcoma Untreated Childhood Anaplastic Astrocytoma Untreated Childhood Anaplastic Oligoastrocytoma Untreated Childhood Brain Stem Glioma Untreated Childhood Giant Cell Glioblastoma Untreated Childhood Gliosarcoma Collapse << | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT01105312 | Breast Cancer | Phase 1 Phase 2 | Completed | - | - |
| NCT01105312 | - | - | Completed | - | - |
| NCT02839707 | Fallopian Tube Clear Cell Aden... more >>ocarcinoma Fallopian Tube Endometrioid Adenocarcinoma Fallopian Tube Undifferentiated Carcinoma High Grade Fallopian Tube Serous Adenocarcinoma High Grade Ovarian Serous Adenocarcinoma Ovarian Clear Cell Adenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Seromucinous Carcinoma Ovarian Undifferentiated Carcinoma Primary Peritoneal High Grade Serous Adenocarcinoma Recurrent Fallopian Tube Carcinoma Recurrent Ovarian Carcinoma Recurrent Primary Peritoneal Carcinoma Collapse << | Phase 2 Phase 3 | Recruiting | June 30, 2023 | - |
| NCT00792701 | - | - | Completed | - | - |
| NCT03233711 | Anal Basaloid Carcinoma ... more >> Anal Canal Cloacogenic Carcinoma Anal Margin Squamous Cell Carcinoma Stage II Anal Canal Cancer AJCC v6 and v7 Stage IIB Anal Cancer AJCC v8 Stage III Anal Canal Cancer AJCC v6 and v7 Stage IIIA Anal Canal Cancer AJCC v6 and v7 Stage IIIB Anal Canal Cancer AJCC v6 and v7 Collapse << | Phase 2 | Recruiting | December 30, 2019 | - |
| NCT01412879 | Lymphoma | Phase 2 | Active, not recruiting | August 2021 | - |
| NCT00684983 | - | - | Active, not recruiting | - | - |
| NCT01653028 | Myxofibrosarcoma ... more >> Recurrent Adult Soft Tissue Sarcoma Recurrent Leiomyosarcoma Recurrent Liposarcoma Recurrent Malignant Peripheral Nerve Sheath Tumor Recurrent Undifferentiated Pleomorphic Sarcoma Stage III Soft Tissue Sarcoma AJCC v7 Stage IV Soft Tissue Sarcoma AJCC v7 Collapse << | Phase 2 | Completed | - | - |
| NCT01653028 | - | - | Completed | - | - |
| NCT01412879 | - | - | Active, not recruiting | - | - |
| NCT01167712 | Fallopian Tube Endometrioid Ad... more >>enocarcinoma Fallopian Tube Mucinous Adenocarcinoma Fallopian Tube Transitional Cell Carcinoma Malignant Ovarian Mixed Epithelial Tumor Ovarian Brenner Tumor Ovarian Clear Cell Adenocarcinofibroma Ovarian Endometrioid Adenocarcinoma Ovarian Mucinous Adenocarcinoma Ovarian Serous Adenocarcinoma Ovarian Transitional Cell Carcinoma Primary Peritoneal Serous Adenocarcinoma Stage IIA Fallopian Tube Cancer Stage IIA Ovarian Cancer Stage IIB Fallopian Tube Cancer Stage IIB Ovarian Cancer Stage IIC Fallopian Tube Cancer Stage IIC Ovarian Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Cancer Stage IIIA Primary Peritoneal Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Cancer Stage IIIB Primary Peritoneal Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Cancer Stage IIIC Primary Peritoneal Cancer Stage IV Fallopian Tube Cancer Stage IV Ovarian Cancer Stage IV Primary Peritoneal Cancer Undifferentiated Ovarian Carcinoma Collapse << | Phase 3 | Active, not recruiting | June 6, 2027 | - |
| NCT01896999 | Recurrent Classic Hodgkin Lymp... more >>homa Refractory Classic Hodgkin Lymphoma Collapse << | Phase 1 Phase 2 | Recruiting | - | - |
| NCT00775645 | Breast Cancer ... more >> Chemotherapeutic Agent Toxicity Fatigue Neuropathy Neurotoxicity Collapse << | Phase 3 | Completed | - | - |
| NCT01708954 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 2 | Active, not recruiting | March 17, 2020 | - |
| NCT02785952 | Recurrent Squamous Cell Lung C... more >>arcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 3 | Recruiting | April 1, 2022 | - |
| NCT00699491 | Male Breast Carcinoma ... more >> Recurrent Breast Carcinoma Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 1 Phase 2 | Completed | - | - |
| NCT01167712 | - | - | Active, not recruiting | - | - |
| NCT00045630 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Urethral Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00699491 | - | - | Completed | - | - |
| NCT00045630 | - | - | Completed | - | - |
| NCT01124695 | Breast Cancer | Phase 2 | Unknown | - | - |
| NCT00775645 | - | - | Completed | - | - |
| NCT01708954 | - | - | Active, not recruiting | - | - |
| NCT00887159 | Extensive Stage Small Cell Lun... more >>g Carcinoma Recurrent Small Cell Lung Carcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT00103181 | Breast Cancer | Not Applicable | Active, not recruiting | April 2020 | - |
| NCT00867321 | Liver Cancer | Phase 1 Phase 2 | Completed | - | - |
| NCT00867321 | - | - | Completed | - | - |
| NCT00238420 | - | - | Active, not recruiting | - | - |
| NCT00887159 | - | - | Completed | - | - |
| NCT00301821 | Lymphoma | Phase 2 | Completed | - | - |
| NCT00807768 | Endometrial Clear Cell Adenoca... more >>rcinoma Endometrial Serous Adenocarcinoma Fatigue Neurotoxicity Syndrome Obesity Stage I Uterine Corpus Cancer Stage II Uterine Corpus Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00807768 | - | - | Active, not recruiting | - | - |
| NCT01901094 | Stage II Breast Cancer ... more >> Stage IIIA Breast Cancer Collapse << | Phase 3 | Recruiting | - | - |
| NCT00086996 | Esophageal Cancer | Phase 2 | Completed | - | - |
| NCT00107380 | Lymphoma | Phase 2 | Completed | - | - |
| NCT00107380 | - | - | Completed | - | - |
| NCT00086996 | - | - | Completed | - | - |
| NCT00238420 | Bladder Urothelial Carcinoma ... more >> Stage I Bladder Cancer AJCC v6 and v7 Stage II Bladder Cancer AJCC v6 and v7 Stage III Bladder Cancer AJCC v6 and v7 Collapse << | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT00301821 | - | - | Completed | - | - |
| NCT00601627 | - | - | Completed | - | - |
| NCT00892177 | Glioblastoma Multiforme | Phase 2 | Active, not recruiting | - | - |
| NCT00331773 | Prostate Cancer | Phase 3 | Active, not recruiting | - | - |
| NCT02766335 | Recurrent Squamous Cell Lung C... more >>arcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | April 1, 2022 | - |
| NCT01081262 | Borderline Ovarian Mucinous Tu... more >>mor Ovarian Mucinous Cystadenocarcinoma Recurrent Fallopian Tube Carcinoma Recurrent Ovarian Carcinoma Stage IA Fallopian Tube Cancer Stage IA Ovarian Cancer Stage IB Fallopian Tube Cancer Stage IB Ovarian Cancer Stage IC Fallopian Tube Cancer Stage IC Ovarian Cancer Stage IIA Fallopian Tube Cancer Stage IIA Ovarian Cancer Stage IIB Fallopian Tube Cancer Stage IIB Ovarian Cancer Stage IIC Fallopian Tube Cancer Stage IIC Ovarian Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Cancer Stage IV Fallopian Tube Cancer Stage IV Ovarian Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01009918 | Breast Cancer ... more >> Cardiac Toxicity Collapse << | Phase 2 | Active, not recruiting | November 2018 | - |
| NCT00892177 | - | - | Active, not recruiting | - | - |
| NCT00454194 | - | - | Completed | - | - |
| NCT01835158 | Clear Cell Renal Cell Carcinom... more >>a Metastatic Renal Cell Cancer Stage III Renal Cell Cancer AJCC v7 Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00914121 | Healthy Subjects | Phase 1 | Completed | - | United States, Washington ... more >> Tacoma, Washington, United States, 98418 Collapse << |
| NCT00331773 | - | - | Active, not recruiting | - | - |
| NCT00454194 | Lung Cancer | Phase 2 | Completed | - | - |
| NCT00601627 | Pancreatic Cancer | Phase 2 | Completed | - | - |
| NCT01229943 | Pancreatic Gastrinoma ... more >> Pancreatic Neuroendocrine Tumor G1 Pancreatic Neuroendocrine Tumor G2 Pancreatic Vipoma Recurrent Pancreatic Neuroendocrine Carcinoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01841736 | Atypical Carcinoid Tumor ... more >> Foregut Carcinoid Tumor Hindgut Carcinoid Tumor Metastatic Carcinoid Tumor Metastatic Digestive System Neuroendocrine Tumor G1 Midgut Carcinoid Tumor Recurrent Digestive System Neuroendocrine Tumor G1 Regional Digestive System Neuroendocrine Tumor G1 Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01229943 | - | - | Active, not recruiting | - | - |
| NCT00796653 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00861705 | - | - | Active, not recruiting | - | - |
| NCT01013649 | Pancreatic Acinar Cell Carcino... more >>ma Pancreatic Ductal Adenocarcinoma Pancreatic Intraductal Papillary-Mucinous Neoplasm Stage I Pancreatic Cancer AJCC v6 and v7 Stage IA Pancreatic Cancer AJCC v6 and v7 Stage IB Pancreatic Cancer AJCC v6 and v7 Stage II Pancreatic Cancer AJCC v6 and v7 Stage IIA Pancreatic Cancer AJCC v6 and v7 Stage IIB Pancreatic Cancer AJCC v6 and v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT02196181 | BRAF V600E Mutation Present ... more >> BRAF V600K Mutation Present Stage III Cutaneous Melanoma AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Unresectable Melanoma Collapse << | Phase 2 | Recruiting | - | - |
| NCT00861705 | Estrogen Receptor Negative ... more >> HER2/Neu Negative Male Breast Carcinoma Progesterone Receptor Negative Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage IIIA Breast Cancer AJCC v7 Triple-Negative Breast Carcinoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01011478 | Colorectal Cancer ... more >> Precancerous Condition Collapse << | Phase 3 | Terminated | - | - |
| NCT02595905 | Breast Carcinoma Metastatic in... more >> the Brain Deleterious BRCA1 Gene Mutation Deleterious BRCA2 Gene Mutation Estrogen Receptor Negative HER2/Neu Negative Progesterone Receptor Negative Recurrent Breast Carcinoma Stage IV Breast Cancer AJCC v6 and v7 Triple-Negative Breast Carcinoma Collapse << | Phase 2 | Recruiting | - | - |
| NCT00770809 | - | - | Active, not recruiting | - | - |
| NCT01101451 | Cervical Adenocarcinoma ... more >> Cervical Adenosquamous Carcinoma Cervical Squamous Cell Carcinoma, Not Otherwise Specified Stage IA Cervical Cancer Stage IB Cervical Cancer Stage IIA Cervical Cancer Collapse << | Phase 3 | Recruiting | - | - |
| NCT00770809 | HER2/Neu Positive ... more >> Male Breast Carcinoma Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage IIIA Breast Cancer AJCC v7 Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00408408 | Breast Cancer | Phase 3 | Active, not recruiting | March 2018 | - |
| NCT00303628 | - | - | Terminated(The study was termi... more >>nated before reaching its accrual goal due to slow accrual.) Collapse << | - | - |
| NCT00303628 | Adenocarcinoma of the Rectum ... more >> Stage II Rectal Cancer Stage III Rectal Cancer Collapse << | Phase 3 | Terminated(The study was termi... more >>nated before reaching its accrual goal due to slow accrual.) Collapse << | April 2019 | - |
| NCT00408408 | - | - | Active, not recruiting | - | - |
| NCT00796653 | - | - | Completed | - | - |
| NCT01658943 | - | - | Completed | - | - |
| NCT01658943 | Pancreatic Acinar Cell Carcino... more >>ma Pancreatic Ductal Adenocarcinoma Recurrent Pancreatic Carcinoma Stage IV Pancreatic Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT01953588 | Estrogen Receptor-positive Bre... more >>ast Cancer HER2-negative Breast Cancer Invasive Ductal Breast Carcinoma Invasive Lobular Breast Carcinoma Recurrent Breast Cancer Stage II Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Collapse << | Phase 3 | Recruiting | - | - |
| NCT01498289 | Adenocarcinoma of the Gastroes... more >>ophageal Junction Esophageal Cancer Gastric Cancer Collapse << | Phase 2 | Active, not recruiting | March 2019 | - |
| NCT02500797 | Dedifferentiated Liposarcoma ... more >> Gastrointestinal Stromal Tumor Metastatic Liposarcoma Metastatic Undifferentiated Pleomorphic Sarcoma Pleomorphic Liposarcoma Stage III Bone Sarcoma AJCC v7 Stage III Soft Tissue Sarcoma AJCC v7 Stage IV Bone Sarcoma AJCC v7 Stage IV Soft Tissue Sarcoma AJCC v7 Stage IVA Bone Sarcoma AJCC v7 Stage IVB Bone Sarcoma AJCC v7 Unresectable Liposarcoma Collapse << | Phase 2 | Suspended(Scheduled Interim Mo... more >>nitoring) Collapse << | January 30, 2022 | - |
| NCT00003641 | - | - | Terminated(The third interim a... more >>nalysis showed that there was no evidence of benefit from treatment with interferon.) Collapse << | - | - |
| NCT02926638 | MET Positive ... more >>Recurrent Squamous Cell Lung Carcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | April 1, 2022 | - |
| NCT00003641 | Melanoma (Skin) | Phase 3 | Terminated(The third interim a... more >>nalysis showed that there was no evidence of benefit from treatment with interferon.) Collapse << | October 2025 | - |
| NCT00719303 | Fallopian Tube Clear Cell Aden... more >>ocarcinoma Fallopian Tube Endometrioid Adenocarcinoma Fallopian Tube Mucinous Adenocarcinoma Fallopian Tube Serous Adenocarcinoma Fallopian Tube Transitional Cell Carcinoma Malignant Ovarian Brenner Tumor Ovarian Clear Cell Adenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Mucinous Adenocarcinoma Ovarian Seromucinous Carcinoma Ovarian Serous Adenocarcinoma Ovarian Transitional Cell Carcinoma Primary Peritoneal Serous Adenocarcinoma Stage IIA Fallopian Tube Cancer AJCC v6 and v7 Stage IIA Ovarian Cancer AJCC V6 and v7 Stage IIB Fallopian Tube Cancer AJCC v6 and v7 Stage IIB Ovarian Cancer AJCC v6 and v7 Stage IIC Fallopian Tube Cancer AJCC v6 and v7 Stage IIC Ovarian Cancer AJCC v6 and v7 Stage IIIA Fallopian Tube Cancer AJCC v7 Stage IIIA Ovarian Cancer AJCC v6 and v7 Stage IIIA Primary Peritoneal Cancer AJCC v7 Stage IIIB Fallopian Tube Cancer AJCC v7 Stage IIIB Ovarian Cancer AJCC v6 and v7 Stage IIIB Primary Peritoneal Cancer AJCC v7 Stage IIIC Fallopian Tube Cancer AJCC v7 Stage IIIC Ovarian Cancer AJCC v6 and v7 Stage IIIC Primary Peritoneal Cancer AJCC v7 Stage IV Fallopian Tube Cancer AJCC v6 and v7 Stage IV Ovarian Cancer AJCC v6 and v7 Stage IV Primary Peritoneal Cancer AJCC v7 Undifferentiated Fallopian Tube Carcinoma Undifferentiated Ovarian Carcinoma Collapse << | Phase 3 | Recruiting | - | - |
| NCT01856192 | Ann Arbor Stage II Diffuse Lar... more >>ge B-Cell Lymphoma Ann Arbor Stage III Diffuse Large B-Cell Lymphoma Ann Arbor Stage IV Diffuse Large B-Cell Lymphoma CD20 Positive Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01522976 | - | - | Active, not recruiting | - | - |
| NCT01216683 | Lymphoma | Phase 2 | Active, not recruiting | April 2023 | - |
| NCT01522976 | Chronic Myelomonocytic Leukemi... more >>a Chronic Myelomonocytic Leukemia-1 Chronic Myelomonocytic Leukemia-2 Myelodysplastic Syndrome Myelodysplastic Syndrome With Excess Blasts Myelodysplastic Syndrome With Excess Blasts-1 Myelodysplastic Syndrome With Excess Blasts-2 Previously Treated Myelodysplastic Syndrome Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT02785913 | Recurrent Squamous Cell Lung C... more >>arcinoma Stage IV Squamous Cell Lung Carcinoma Collapse << | Phase 2 | Active, not recruiting | June 2020 | - |
| NCT02164916 | Colorectal Cancer | Phase 2 | Active, not recruiting | January 2020 | - |
| NCT01642251 | Extensive Stage Small Cell Lun... more >>g Carcinoma Large Cell Lung Carcinoma Neuroendocrine Carcinoma Small Cell Carcinoma Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT02164916 | - | - | Active, not recruiting | - | - |
| NCT02251470 | Stroke | Phase 1 Phase 2 | Completed | - | Germany ... more >> Hospital "BG-Kliniken Bergmannstrost" Halle, Saxony-Anhalt, Germany, D-06112 University Hospital Halle Halle, Saxony-Anhalt, Germany, D-06120 Collapse << |
| NCT02785939 | CCND1 Gene Amplification ... more >> CCND2 Gene Amplification CCND3 Gene Amplification CDK4 Gene Amplification Recurrent Squamous Cell Lung Carcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | April 1, 2022 | - |
| NCT02728843 | Parkinson's Disease | Phase 2 | Active, not recruiting | December 2019 | Canada, Ontario ... more >> Toronto Western Hospital Toronto, Ontario, Canada France CHU de Bordeaux, Centre Expert Parkinson Bordeaux, France Hôpital Henri Mondor Creteil, France Centre Hospitalier Régional Universitaire de Lille, Hôpital Roger Salengro Lille, France CHU Dupuytren Limoges, France Hôpital Neurologique Pierre Wertheimer Lyon, France CHRU de Montpellier - Hôpital Gui de Chauliac Montpellier, France CHU Pontchaillou Rennes, France CHU Charles Nicoll - Rouen Rouen, France Hôpitaux Universitaires de Strasbourg, Hôpital de Hautepierre Strasbourg, France CHU Purpan, Hôpital Pierre Paul Riquet Toulouse, France Germany Heinriche-Heine Universität Düsseldorf Dusseldorf, Germany UKSH Campus Kiel, Neurologie Kiel, Germany Universitätsklinikum Gießen und Marburg GmbH Marburg, Germany Klinikum rechts der Isar Munich, Germany United Kingdom Royal Devon & Exeter Hospital Exeter, Devon, United Kingdom Fairfield General Hospital Bury, United Kingdom Charing Cross Hospital London, United Kingdom Newcastle Clinical Ageing Research Unit Newcastle Upon Tyne, United Kingdom Derriford Hospital Plymouth, United Kingdom Collapse << |
| NCT02965378 | FGFR1 Gene Amplification ... more >> FGFR1 Gene Mutation FGFR2 Gene Amplification FGFR2 Gene Mutation FGFR3 Gene Amplification FGFR3 Gene Mutation Recurrent Squamous Cell Lung Carcinoma Stage IV Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | April 1, 2022 | - |
| NCT00853580 | Neurofibromatosis Type 1 | Phase 2 | Completed | - | United States, Alabama ... more >> The University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, California Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Illinois University of Chicago Chicago, Illinois, United States, 60637 United States, Maryland NIH Bethesda, Maryland, United States, 20892 United States, Massachusetts Children' Hospital Boston Boston, Massachusetts, United States, 02115 United States, Missouri Washington University - St. Louis Saint Louis, Missouri, United States, 63110 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19096 United States, Texas Childrens Medical Center - Univ. of Texas SW Medical Center Dallas, Texas, United States, 75235 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Australia, New South Wales The Children's Hospital at Westmead Westmead, New South Wales, Australia, 2145 Collapse << |
| NCT01229969 | Falls, Accidental | Not Applicable | Completed | - | United States, Georgia ... more >> Atlanta VA Medical Center Atlanta, Georgia, United States, 30033 Collapse << |
| NCT02906696 | Chronic Myelogenous Leukemia | Phase 2 | Recruiting | October 2022 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT00217737 | Colon Mucinous Adenocarcinoma ... more >> Colon Signet Ring Cell Adenocarcinoma Lynch Syndrome Stage IIA Colon Cancer AJCC v7 Stage IIB Colon Cancer AJCC v7 Stage IIC Colon Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT03368560 | Late Life Depression | Phase 1 | Withdrawn(Funding was not rece... more >>ived.) Collapse << | December 31, 2022 | - |
| NCT03220945 | Prostate Carcinoma | Not Applicable | Active, not recruiting | December 2019 | United States, Georgia ... more >> Emory University/Winship Cancer Institute Atlanta, Georgia, United States, 30322 Emory Saint Joseph's Hospital Atlanta, Georgia, United States, 30342 Collapse << |
| NCT00734292 | Mild to Moderate Asthma | Phase 2 | Completed | - | United States, California ... more >> Investigational Site Anaheim, California, United States, 92801 Investigational Site Mission Viejo, California, United States, 92691 United States, Colorado Investigational Site Denver, Colorado, United States, 80230 United States, Florida Investigational Site Valrico, Florida, United States, 33594 United States, Oregon Investigational Site Medford, Oregon, United States, 97504 Investigational Site Portland, Oregon, United States, 97213 United States, Virginia Investigational Site Richmond, Virginia, United States, 23229 United States, Wisconsin Investigational Site West Allis, Wisconsin, United States, 53227 Collapse << |
| NCT00319254 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Duarte, California, United States, 91010-3000 Pfizer Investigational Site Santa Monica, California, United States, 90404 United States, Florida Pfizer Investigational Site Tampa, Florida, United States, 33612 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 Australia, New South Wales Pfizer Investigational Site Darlinghurst, New South Wales, Australia, 2010 France Pfizer Investigational Site Dijon, France, 21034 Pfizer Investigational Site Saint-Herblain, France, 44805 Hong Kong Pfizer Investigational Site Pokfulam, Hong Kong Malta Pfizer Investigational Site Floriana, Malta, VLT 14 Poland Pfizer Investigational Site Lodz, Poland, 90-553 Pfizer Investigational Site Wroclaw, Poland, 51-124 Russian Federation Pfizer Investigational Site Moscow, Russian Federation, 115478 Ukraine Pfizer Investigational Site Dnipropetrovsk, Ukraine, 49102 Pfizer Investigational Site Sumy, Ukraine, 40005 Pfizer Investigational Site Uzhgorod, Ukraine, 88014 Collapse << |
| NCT02782403 | Leukemia | Phase 1 Phase 2 | Recruiting | March 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact pbose@mdanderson.org Collapse << |
| NCT02311998 | Leukemia | Phase 1 Phase 2 | Recruiting | April 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT02079675 | Non-constipation Irritable Bow... more >>el Syndrome Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> SK Chemicals Seoul, Korea, Republic of Collapse << |
| NCT00853580 | - | - | Completed | - | - |
| NCT00319254 | - | - | Completed | - | - |
| NCT03741491 | - | - | Not yet recruiting | April 2021 | - |
| NCT00887536 | Breast Cancer | Phase 3 | Active, not recruiting | January 2022 | - |
| NCT01547741 | Breast Cancer | Phase 3 | Active, not recruiting | May 2019 | - |
| NCT00382018 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT02003209 | Estrogen Receptor Positive ... more >> HER2/Neu Positive Progesterone Receptor Positive Stage IB Breast Cancer AJCC v7 Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage IIIA Breast Cancer AJCC v7 Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00382018 | - | - | Completed | - | - |
| NCT00874211 | - | - | Active, not recruiting | January 2019 | - |
| NCT00588770 | Recurrent Hypopharyngeal Squam... more >>ous Cell Carcinoma Recurrent Laryngeal Squamous Cell Carcinoma Recurrent Laryngeal Verrucous Carcinoma Recurrent Lip and Oral Cavity Squamous Cell Carcinoma Recurrent Metastatic Squamous Cell Carcinoma in the Neck With Occult Primary Recurrent Nasal Cavity and Paranasal Sinus Squamous Cell Carcinoma Recurrent Oral Cavity Verrucous Carcinoma Recurrent Oropharyngeal Squamous Cell Carcinoma Recurrent Salivary Gland Carcinoma Salivary Gland Squamous Cell Carcinoma Squamous Cell Carcinoma Metastatic in the Neck With Occult Primary Stage IV Hypopharyngeal Squamous Cell Carcinoma AJCC v7 Stage IV Major Salivary Gland Cancer AJCC v7 Stage IVA Laryngeal Squamous Cell Carcinoma AJCC v7 Stage IVA Laryngeal Verrucous Carcinoma AJCC v7 Stage IVA Lip and Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVA Major Salivary Gland Cancer AJCC v7 Stage IVA Nasal Cavity and Paranasal Sinus Squamous Cell Carcinoma AJCC v7 Stage IVA Oral Cavity Cancer AJCC v6 and v7 Stage IVA Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVB Laryngeal Squamous Cell Carcinoma AJCC v7 Stage IVB Laryngeal Verrucous Carcinoma AJCC v7 Stage IVB Lip and Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVB Major Salivary Gland Cancer AJCC v7 Stage IVB Nasal Cavity and Paranasal Sinus Squamous Cell Carcinoma AJCC v7 Stage IVB Oral Cavity Cancer AJCC v6 and v7 Stage IVB Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVC Laryngeal Squamous Cell Carcinoma AJCC v7 Stage IVC Laryngeal Verrucous Carcinoma AJCC v7 Stage IVC Lip and Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVC Major Salivary Gland Cancer AJCC v7 Stage IVC Nasal Cavity and Paranasal Sinus Squamous Cell Carcinoma AJCC v7 Stage IVC Oral Cavity Cancer AJCC v6 and v7 Stage IVC Oropharyngeal Squamous Cell Carcinoma AJCC v7 Tongue Carcinoma Untreated Metastatic Squamous Cell Carcinoma to Neck With Occult Primary Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00024167 | Prostate Cancer | Phase 3 | Terminated(Terminated due to s... more >>low accrual) Collapse << | - | - |
| NCT00003389 | - | - | Completed | - | - |
| NCT01439945 | - | - | Completed | - | - |
| NCT00003389 | Lymphoma | Phase 3 | Completed | - | - |
| NCT00058149 | Pancreatic Cancer | Phase 3 | Completed | - | - |
| NCT00860041 | - | - | Completed | - | - |
| NCT01439945 | Breast Cancer ... more >> Cancer Survivor Hot Flashes Collapse << | Phase 2 | Completed | - | - |
| NCT00024167 | - | - | Terminated(Terminated due to s... more >>low accrual) Collapse << | - | - |
| NCT02997228 | Colorectal Adenocarcinoma ... more >> Mismatch Repair Deficiency Stage IV Colorectal Cancer AJCC v7 Stage IVA Colorectal Cancer AJCC v7 Stage IVB Colorectal Cancer AJCC v7 Collapse << | Phase 3 | Recruiting | April 30, 2022 | - |
| NCT00070499 | Chronic Myelogenous Leukemia, ... more >>BCR-ABL1 Positive Chronic Phase of Disease Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01376349 | Breast Cancer ... more >> Gynecologic Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01376349 | - | - | Active, not recruiting | - | - |
| NCT01576328 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | United States, California ... more >> SC Clinical Research Garden Grove, California, United States, 92844 United States, Florida Diabetes Research Institute Miami, Florida, United States, 33136 Compass Research Orlando, Florida, United States, 32806 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States Tulane University Medical Center New Orleans, Louisiana, United States United States, Montana Big Sky Clinical Research Butte, Montana, United States, 59701 United States, Nevada Desert Endocrinology Clinical Research Center-Henderson Henderson, Nevada, United States, 89052 Alliance Against Diabetes/AAD Clinical Research Las Vegas, Nevada, United States, 89101 United States, New Hampshire Active Practices and Research Newington, New Hampshire, United States, 03801 United States, Ohio The Carl and Edyth Lindner Center for Research and Education at The Christ Hospital Cincinnati, Ohio, United States, 45219 Providence Health Partners - Center for Clinical Reseach Dayton, Ohio, United States, 45439 United States, Texas Dallas Diabetes and Endocrine Center Dallas, Texas, United States, 75230 West Houston Clinical Research Services Houston, Texas, United States Paragon Research Center San Antonio, Texas, United States, 78205 United States, Utah Wasatch Clinical Research Salt Lake City, Utah, United States United States, Virginia National Clinical Research - Norfolk, Inc Norfolk, Virginia, United States, 23502 National Clinical Research - Richmond, Inc. Richmond, Virginia, United States, 23294 United States, Washington Capital Clinical Research Center Olympia, Washington, United States, 98502 Collapse << |
| NCT00041067 | Breast Cancer | Phase 2 | Completed | - | - |
| NCT00041067 | - | - | Completed | - | - |
| NCT02193282 | ALK Gene Rearrangement ... more >> EGFR Exon 19 Deletion Mutation EGFR NP_005219.2:p.L858R Stage IB Non-Small Cell Lung Carcinoma AJCC v7 Stage II Non-Small Cell Lung Cancer AJCC v7 Stage IIA Non-Small Cell Lung Carcinoma AJCC v7 Stage IIB Non-Small Cell Lung Carcinoma AJCC v7 Stage IIIA Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 3 | Recruiting | - | - |
| NCT00009906 | Gastrointestinal Stromal Tumor | Phase 3 | Terminated | - | - |
| NCT00803062 | Cervical Adenocarcinoma ... more >> Cervical Adenosquamous Carcinoma Cervical Squamous Cell Carcinoma Recurrent Cervical Carcinoma Stage IVB Cervical Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00803062 | - | - | Completed | - | - |
| NCT01737905 | Asthma | Phase 3 | Completed | - | United States, California ... more >> West Coast Clinical Trials Global Cypress, California, United States, 90630 United States, Oregon The Clinical Research Institute of Southern Oregn, PC Medford, Oregon, United States, 97504 Transitional Clinical Research, Inc. Allergy Associates Research Center Portland, Oregon, United States, 97202 United States, Texas Western Sky Medical El Paso, Texas, United States, 79903 Sylvana Research Assocaites San Antonio, Texas, United States, 78229 United States, Washington ASTHMA, Inc. Clinical Research Center Seattle, Washington, United States, 98115 Collapse << |
| NCT00070499 | - | - | Active, not recruiting | - | - |
| NCT03419234 | Castration Levels of Testoster... more >>one Castration-Resistant Prostate Carcinoma Metastatic Prostate Carcinoma in the Soft Tissue Prostate Carcinoma Metastatic in the Bone Stage IV Prostate Adenocarcinoma AJCC v7 Collapse << | Phase 2 | Recruiting | April 1, 2025 | - |
| NCT03375320 | Atypical Carcinoid Tumor ... more >> Carcinoid Tumor Digestive System Neuroendocrine Neoplasm Enterochromaffin Cell Serotonin-Producing Pancreatic Neuroendocrine Tumor Functional Pancreatic Neuroendocrine Tumor Intermediate Grade Lung Neuroendocrine Neoplasm Low Grade Lung Neuroendocrine Neoplasm Lung Atypical Carcinoid Tumor Lung Carcinoid Tumor Metastatic Digestive System Neuroendocrine Tumor G1 Neuroendocrine Neoplasm Nonfunctional Pancreatic Neuroendocrine Tumor Pancreatic Neuroendocrine Tumor Stage IIIA Digestive System Neuroendocrine Tumor AJCC v7 Stage IIIB Digestive System Neuroendocrine Tumor AJCC v7 Stage IV Digestive System Neuroendocrine Tumor AJCC v7 Collapse << | Phase 3 | Recruiting | January 1, 2021 | - |
| NCT01012297 | Recurrent Uterine Corpus Sarco... more >>ma Stage IIIA Uterine Sarcoma Stage IIIB Uterine Sarcoma Stage IIIC Uterine Sarcoma Stage IVA Uterine Sarcoma Stage IVB Uterine Sarcoma Uterine Corpus Leiomyosarcoma Collapse << | Phase 3 | Terminated(The study was targe... more >>ted to accrue 130 patients, but closed early for futility.) Collapse << | - | - |
| NCT00110019 | - | - | Completed | - | - |
| NCT02201992 | ALK Gene Rearrangement ... more >> ALK Gene Translocation ALK Positive Stage IB Non-Small Cell Lung Carcinoma AJCC v7 Stage II Non-Small Cell Lung Cancer AJCC v7 Stage IIA Non-Small Cell Lung Carcinoma AJCC v7 Stage IIB Non-Small Cell Lung Carcinoma AJCC v7 Stage IIIA Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 3 | Recruiting | - | - |
| NCT00110019 | Mucosal Melanoma ... more >> Recurrent Melanoma Stage IIIA Skin Melanoma Stage IIIB Skin Melanoma Stage IIIC Skin Melanoma Stage IV Skin Melanoma Collapse << | Phase 3 | Completed | - | - |
| NCT00738881 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 3 | Terminated(Slow accrual) | - | - |
| NCT01012297 | - | - | Terminated(The study was targe... more >>ted to accrue 130 patients, but closed early for futility.) Collapse << | - | - |
| NCT00738881 | - | - | Terminated(Slow accrual) | - | - |
| NCT02443077 | Diffuse Large B-Cell Lymphoma ... more >>Activated B-Cell Type Diffuse Large B-Cell Lymphoma, Not Otherwise Specified High Grade B-Cell Lymphoma, Not Otherwise Specified Recurrent Burkitt Lymphoma Recurrent Diffuse Large B-Cell Lymphoma Refractory Burkitt Lymphoma Refractory Diffuse Large B-Cell Lymphoma Collapse << | Phase 3 | Suspended(Other - This protoco... more >>l requires a DTL.) Collapse << | - | - |
| NCT00390455 | Estrogen Receptor Positive ... more >> HER2 Positive Breast Carcinoma HER2/Neu Negative Progesterone Receptor Positive Recurrent Breast Carcinoma Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00390455 | - | - | Active, not recruiting | - | - |
| NCT01198145 | Diarrhea Gast... more >>rointestinal Complications Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT02185274 | - | - | Recruiting | October 2019 | Germany ... more >> Gynaecological department of the University hospital of Erlangen Recruiting Erlangen, Germany, 91054 Contact: Thomas Hildebrandt, MD 0049 91318533553 thomas.hildebrandt@uk-erlangen.de Principal Investigator: Thomas Hildebrandt, MD Institute for Sex Research and Forensic Psychiatry Recruiting Hamburg, Germany, 20246 Contact: Timo Nieder, PhD +49 40 741054232 t.nieder@uke.de Contact: Johannes Fuss, MD joh.fuss@zi-mannheim.de Principal Investigator: Timo Nieder, PhD Hormon- und Stoffwechselzentrum München Recruiting Munich, Germany, 80333 Contact: Cornelia Bernutz, MD cornelia.bernutz@gmx.de Principal Investigator: Cornelia Bernutz, MD Collapse << |
| NCT01198145 | - | - | Completed | - | - |
| NCT00296036 | Breast Cancer ... more >> Drug/Agent Toxicity by Tissue/Organ Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00296036 | - | - | Completed | - | - |
| NCT02912559 | Colon Adenocarcinoma ... more >> DNA Repair Disorder Lynch Syndrome Stage III Colon Cancer AJCC v7 Stage IIIA Colon Cancer AJCC v7 Stage IIIB Colon Cancer AJCC v7 Stage IIIC Colon Cancer AJCC v7 Collapse << | Phase 3 | Recruiting | July 1, 2020 | - |
| NCT00182754 | Metastatic Cancer ... more >> Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00880191 | Nausea and Vomiting ... more >> Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT03578081 | Malignant Neoplasm | Phase 3 | Recruiting | March 2020 | - |
| NCT00880191 | - | - | Completed | - | - |
| NCT00956813 | Breast Cancer ... more >> Hot Flashes Menopausal Symptoms Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00956813 | - | - | Completed | - | - |
| NCT00978458 | Brain and Central Nervous Syst... more >>em Tumors Neurotoxicity Collapse << | Phase 3 | Active, not recruiting | December 31, 2026 | - |
| NCT00006237 | Melanoma (Skin) | Phase 3 | Completed | - | - |
| NCT00182754 | - | - | Completed | - | - |
| NCT00006237 | - | - | Completed | - | - |
| NCT00075764 | - | - | Completed | - | - |
| NCT00075842 | Fatigue Sleep... more >> Disorders Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | United States, Colorado ... more >> Aurora Presbyterian Hospital Aurora, Colorado, United States, 80012 Boulder Community Hospital Boulder, Colorado, United States, 80301-9019 Penrose Cancer Center at Penrose Hospital Colorado Springs, Colorado, United States, 80933 Porter Adventist Hospital Denver, Colorado, United States, 80210 Presbyterian - St. Luke's Medical Center Denver, Colorado, United States, 80218 St. Joseph Hospital Denver, Colorado, United States, 80218 Rose Medical Center Denver, Colorado, United States, 80220 CCOP - Colorado Cancer Research Program Denver, Colorado, United States, 80224-2522 Swedish Medical Center Englewood, Colorado, United States, 80110 Sky Ridge Medical Center Lone Tree, Colorado, United States, 80124 Hope Cancer Care Center at Longmont United Hospital Longmont, Colorado, United States, 80502 St. Mary - Corwin Regional Medical Center Pueblo, Colorado, United States, 81004 North Suburban Medical Center Thornton, Colorado, United States, 80229 United States, Iowa Mercy Cancer Center at Mercy Medical Center - North Iowa Mason City, Iowa, United States, 50401 Siouxland Hematology-Oncology Associates, LLP Sioux City, Iowa, United States, 51101 St. Luke's Regional Medical Center Sioux City, Iowa, United States, 51104 United States, Kansas CCOP - Wichita Wichita, Kansas, United States, 67214 United States, Minnesota Fairview Ridges Hospital Burnsville, Minnesota, United States, 55337 Mercy and Unity Cancer Center at Mercy Hospital Coon Rapids, Minnesota, United States, 55433 Fairview Southdale Hospital Edina, Minnesota, United States, 55435 Mercy and Unity Cancer Center at Unity Hospital Fridley, Minnesota, United States, 55432 Hutchinson Area Health Care Hutchinson, Minnesota, United States, 55350 Meeker County Memorial Hospital Lichfield, Minnesota, United States, 55355 HealthEast Cancer Care at St. John's Hospital Maplewood, Minnesota, United States, 55109 Minnesota Oncology Hematology, PA - Maplewood Maplewood, Minnesota, United States, 55109 Virginia Piper Cancer Institute at Abbott - Northwestern Hospital Minneapolis, Minnesota, United States, 55407 Hennepin County Medical Center - Minneapolis Minneapolis, Minnesota, United States, 55415 CCOP - Metro-Minnesota Saint Louis Park, Minnesota, United States, 55416 St. Francis Cancer Center at St. Francis Medical Center Shakopee, Minnesota, United States, 55379 HealthEast Cancer Care at St. Joseph's Hospital St Paul, Minnesota, United States, 55102 United Hospital St. Paul, Minnesota, United States, 55102 HealthEast Cancer Care at Woodwinds Health Campus Woodbury, Minnesota, United States, 55125 Minnesota Oncology Hematology, PA - Woodbury Woodbury, Minnesota, United States, 55125 Collapse << |
| NCT00516503 | - | - | Completed | - | - |
| NCT00310180 | Breast Adenocarcinoma ... more >> Estrogen Receptor and/or Progesterone Receptor Positive HER2/Neu Negative Stage IA Breast Cancer AJCC v7 Stage IB Breast Cancer AJCC v7 Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage IIIB Breast Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00516503 | Chronic Myeloproliferative Dis... more >>orders Leukemia Lymphoma Lymphoproliferative Disorder Multiple Myeloma and Plasma Cell Neoplasm Myelodysplastic Syndromes Myelodysplastic/Myeloproliferative Neoplasms Neurotoxicity Pain Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00343382 | Breast Cancer ... more >> Vaginal Dryness Collapse << | Phase 3 | Completed | - | - |
| NCT00054418 | Breast Cancer ... more >> Osteoporosis Collapse << | Phase 3 | Completed | - | - |
| NCT00006721 | Lymphoma | Phase 3 | Active, not recruiting | June 2023 | - |
| NCT02445391 | Estrogen Receptor Negative ... more >> HER2/Neu Negative Invasive Breast Carcinoma Progesterone Receptor Negative Stage II Breast Cancer Stage IIA Breast Cancer Stage IIB Breast Cancer Stage III Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Triple-Negative Breast Carcinoma Collapse << | Phase 3 | Recruiting | May 31, 2024 | - |
| NCT00382070 | Breast Cancer | Phase 3 | Active, not recruiting | October 2018 | - |
| NCT00262847 | Fallopian Tube Clear Cell Aden... more >>ocarcinoma Fallopian Tube Endometrioid Adenocarcinoma Fallopian Tube Mucinous Adenocarcinoma Fallopian Tube Serous Adenocarcinoma Fallopian Tube Transitional Cell Carcinoma Malignant Ovarian Mixed Epithelial Tumor Ovarian Brenner Tumor Ovarian Clear Cell Adenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Mucinous Adenocarcinoma Ovarian Serous Adenocarcinoma Ovarian Transitional Cell Carcinoma Primary Peritoneal Serous Adenocarcinoma Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Cancer Stage IIIA Primary Peritoneal Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Cancer Stage IIIB Primary Peritoneal Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Cancer Stage IIIC Primary Peritoneal Cancer Stage IV Fallopian Tube Cancer Stage IV Ovarian Cancer Stage IV Primary Peritoneal Cancer Undifferentiated Fallopian Tube Carcinoma Undifferentiated Ovarian Carcinoma Collapse << | Phase 3 | Completed | - | - |
| NCT02446600 | BRCA Rearrangement ... more >> Deleterious BRCA1 Gene Mutation Deleterious BRCA2 Gene Mutation Fallopian Tube Clear Cell Adenocarcinoma Fallopian Tube Transitional Cell Carcinoma Fallopian Tube Undifferentiated Carcinoma Ovarian Clear Cell Adenocarcinoma Ovarian Endometrioid Tumor Ovarian Seromucinous Carcinoma Ovarian Serous Tumor Ovarian Transitional Cell Carcinoma Ovarian Undifferentiated Carcinoma Recurrent Fallopian Tube Carcinoma Recurrent Ovarian Carcinoma Recurrent Ovarian Endometrioid Adenocarcinoma Recurrent Primary Peritoneal Carcinoma Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00343382 | - | - | Completed | - | - |
| NCT00262847 | - | - | Completed | - | - |
| NCT00075764 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT00006721 | - | - | Active, not recruiting | - | - |
| NCT02194738 | Large Cell Lung Carcinoma ... more >> Lung Adenocarcinoma Stage IB Non-Small Cell Lung Carcinoma AJCC v7 Stage IB Squamous Cell Lung Carcinoma AJCC v7 Stage II Non-Small Cell Lung Cancer AJCC v7 Stage II Squamous Cell Lung Carcinoma AJCC v7 Stage IIA Non-Small Cell Lung Carcinoma AJCC v7 Stage IIA Squamous Cell Lung Carcinoma AJCC v7 Stage IIB Non-Small Cell Lung Carcinoma AJCC v7 Stage IIB Squamous Cell Lung Carcinoma AJCC v7 Stage IIIA Non-Small Cell Lung Cancer AJCC v7 Stage IIIA Squamous Cell Lung Carcinoma AJCC v7 Collapse << | Phase 3 | Recruiting | - | - |
| NCT02954874 | Estrogen Receptor Negative ... more >> HER2/Neu Negative Invasive Breast Carcinoma Progesterone Receptor Negative Stage 0 Breast Cancer AJCC v6 and v7 Stage I Breast Cancer AJCC v7 Stage IA Breast Cancer AJCC v7 Stage IB Breast Cancer AJCC v7 Stage II Breast Cancer AJCC v6 and v7 Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage III Breast Cancer AJCC v7 Stage IIIA Breast Cancer AJCC v7 Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Triple-Negative Breast Carcinoma Collapse << | Phase 3 | Recruiting | - | - |
| NCT00565851 | Clear Cell Adenocarcinoma ... more >> Fallopian Tube Clear Cell Adenocarcinoma Fallopian Tube Endometrioid Adenocarcinoma Fallopian Tube Mucinous Adenocarcinoma Fallopian Tube Serous Adenocarcinoma Fallopian Tube Undifferentiated Carcinoma Mucinous Adenocarcinoma Ovarian Brenner Tumor Ovarian Clear Cell Adenocarcinofibroma Ovarian Endometrioid Adenocarcinoma Ovarian Seromucinous Carcinoma Ovarian Serous Adenocarcinoma Ovarian Undifferentiated Carcinoma Primary Peritoneal Serous Adenocarcinoma Recurrent Fallopian Tube Carcinoma Recurrent Ovarian Carcinoma Recurrent Primary Peritoneal Carcinoma Undifferentiated Carcinoma Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01041781 | - | - | Terminated(DSMB recommendation... more >>) Collapse << | - | - |
| NCT01041781 | Lung Cancer | Phase 3 | Terminated(DSMB recommendation... more >>) Collapse << | - | - |
| NCT00746798 | West Nile Fever | Phase 2 | Completed | - | United States, California ... more >> Advanced Clinical Research Inst. Anaheim, California, United States, 92801 United States, Colorado Lynn Health Science Institute Colorado Springs, Colorado, United States, 80909 United States, Florida Miami Research South Miami, Florida, United States, 33143 United States, Idaho Advanced Clinical Research- Idaho Boise, Idaho, United States, 83642 Idaho Falls Infectious diseases Idaho Falls, Idaho, United States, 83404 United States, Kansas Johnson County Clinical Trials Lenexa, Kansas, United States, 66219 Vince & Associates Overland Park, Kansas, United States, 66211 United States, Missouri Bio-Kinetic Springfield, Missouri, United States, 65802 United States, Montana Big Sky Clinical Research Butte, Montana, United States, 59701 Infectious Disease Specialists, PC Missoula, Montana, United States, 59802 United States, North Dakota Odyssey Research Fargo, North Dakota, United States, 88104 United States, Oklahoma Lynn Health Science Institute Oklahoma City, Oklahoma, United States, 73112 United States, Texas Research Across America Dallas, Texas, United States, 75234 Benchmark Fort Worth, Texas, United States, 76135 United States, Utah Radiant Research Salt Lake City, Utah, United States, 84107 Collapse << |
| NCT03414970 | Ductal Breast Carcinoma ... more >> Invasive Breast Carcinoma Lobular Breast Carcinoma Medullary Breast Carcinoma Stage II Breast Cancer Stage IIA Breast Cancer Stage IIB Breast Cancer Stage IIIA Breast Cancer Tubular Breast Carcinoma Collapse << | Phase 3 | Recruiting | August 2035 | - |
| NCT01414608 | Cervical Adenocarcinoma ... more >> Cervical Adenosquamous Carcinoma Cervical Squamous Cell Carcinoma, Not Otherwise Specified Chemotherapeutic Agent Toxicity Cognitive Side Effects of Cancer Therapy Psychological Impact of Cancer Radiation Toxicity Sexual Dysfunction and Infertility Stage IB Cervical Cancer Stage IIA Cervical Cancer Stage IIB Cervical Cancer Stage IIIB Cervical Cancer Stage IVA Cervical Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00522392 | Stage I Multiple Myeloma ... more >> Stage II Multiple Myeloma Stage III Multiple Myeloma Collapse << | Phase 3 | Terminated(Slow accrual) | - | - |
| NCT00522392 | - | - | Terminated(Slow accrual) | - | - |
| NCT01196390 | Esophageal Adenocarcinoma ... more >> Gastroesophageal Junction Adenocarcinoma Stage IB Esophageal Cancer AJCC v7 Stage IIA Esophageal Cancer AJCC v7 Stage IIB Esophageal Cancer AJCC v7 Stage IIIA Esophageal Cancer AJCC v7 Stage IIIB Esophageal Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00499369 | Recurrent Colon Cancer ... more >> Recurrent Rectal Cancer Stage IVA Colon Cancer Stage IVA Rectal Cancer Stage IVB Colon Cancer Stage IVB Rectal Cancer Collapse << | Phase 3 | Terminated(Due to inadequate a... more >>ccrual, study was terminated and limited outcome data was reported.) Collapse << | - | - |
| NCT00569127 | Atypical Carcinoid Tumor ... more >> Carcinoid Tumor Colorectal Neuroendocrine Tumor G1 Gastric Neuroendocrine Tumor G1 Neuroendocrine Neoplasm Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT02085408 | Acute Myeloid Leukemia With Mu... more >>ltilineage Dysplasia Following Myelodysplastic Syndrome Adult Acute Megakaryoblastic Leukemia (M7) Adult Acute Minimally Differentiated Myeloid Leukemia (M0) Adult Acute Monoblastic Leukemia (M5a) Adult Acute Monocytic Leukemia (M5b) Adult Acute Myeloblastic Leukemia With Maturation (M2) Adult Acute Myeloblastic Leukemia Without Maturation (M1) Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Myelomonocytic Leukemia (M4) Adult Erythroleukemia (M6a) Adult Pure Erythroid Leukemia (M6b) Secondary Acute Myeloid Leukemia Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00569127 | - | - | Active, not recruiting | - | - |
| NCT00489411 | Neurotoxicity ... more >> Pain Peripheral Neuropathy Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00489411 | - | - | Completed | - | - |
| NCT00499369 | - | - | Terminated(Due to inadequate a... more >>ccrual, study was terminated and limited outcome data was reported.) Collapse << | - | - |
| NCT01107626 | Lung Cancer | Phase 3 | Active, not recruiting | May 8, 2020 | - |
| NCT00134056 | - | - | Completed | - | - |
| NCT00134056 | Metastatic Cancer ... more >> Prostate Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT01349881 | Colorectal Neoplasms | Phase 3 | Recruiting | July 2029 | - |
| NCT00746798 | - | - | Completed | - | - |
| NCT02465060 | Advanced Malignant Solid Neopl... more >>asm Bladder Carcinoma Breast Carcinoma Cervical Carcinoma Colon Carcinoma Colorectal Carcinoma Endometrial Carcinoma Esophageal Carcinoma Gastric Carcinoma Glioma Head and Neck Carcinoma Kidney Carcinoma Liver and Intrahepatic Bile Duct Carcinoma Lung Carcinoma Lymphoma Malignant Uterine Neoplasm Melanoma Ovarian Carcinoma Pancreatic Carcinoma Plasma Cell Myeloma Prostate Carcinoma Rectal Carcinoma Recurrent Bladder Carcinoma Recurrent Breast Carcinoma Recurrent Cervical Carcinoma Recurrent Colon Carcinoma Recurrent Colorectal Carcinoma Recurrent Esophageal Carcinoma Recurrent Gastric Carcinoma Recurrent Glioma Recurrent Head and Neck Carcinoma Recurrent Liver Carcinoma Recurrent Lung Carcinoma Recurrent Lymphoma Recurrent Malignant Solid Neoplasm Recurrent Melanoma Recurrent Ovarian Carcinoma Recurrent Pancreatic Carcinoma Recurrent Plasma Cell Myeloma Recurrent Prostate Carcinoma Recurrent Rectal Carcinoma Recurrent Skin Carcinoma Recurrent Thyroid Gland Carcinoma Recurrent Uterine Corpus Carcinoma Refractory Lymphoma Refractory Malignant Solid Neoplasm Refractory Plasma Cell Myeloma Skin Carcinoma Thyroid Gland Carcinoma Uterine Corpus Cancer Collapse << | Phase 2 | Recruiting | - | - |
| NCT00602641 | Plasma Cell Myeloma | Phase 3 | Active, not recruiting | - | - |
| NCT00326898 | Stage I Renal Cell Cancer ... more >> Stage II Renal Cell Cancer Stage III Renal Cell Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00326898 | - | - | Completed | - | - |
| NCT00114101 | DS Stage I Plasma Cell Myeloma... more >> DS Stage II Plasma Cell Myeloma DS Stage III Plasma Cell Myeloma Refractory Plasma Cell Myeloma Smoldering Plasma Cell Myeloma Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00602641 | - | - | Active, not recruiting | - | - |
| NCT00114101 | - | - | Active, not recruiting | - | - |
| NCT00433511 | Estrogen Receptor Negative ... more >> Estrogen Receptor Positive HER2/Neu Negative Male Breast Carcinoma Stage IA Breast Cancer Stage IB Breast Cancer Stage IIA Breast Cancer Stage IIB Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00081250 | Anorexia Cach... more >>exia Unspecified Adult Solid Tumor, Protocol Specific Weight Changes Collapse << | Phase 3 | Completed | - | - |
| NCT00376675 | Fatigue Unspe... more >>cified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | - |
| NCT00513747 | Leukemia | Phase 3 | Terminated(Insufficient accrua... more >>l) Collapse << | - | - |
| NCT00376675 | - | - | Completed | - | - |
| NCT00022633 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Urethral Cancer Collapse << | Phase 2 | Terminated(Permanently Closed ... more >>Due to Poor Accrual) Collapse << | - | - |
| NCT01185145 | Breast Cancer | Phase 2 | Active, not recruiting | August 2024 | United States, Colorado ... more >> Rocky Mountain Cancer Centers - Aurora Aurora, Colorado, United States, 80012 Rocky Mountain Cancer Centers - Boulder Boulder, Colorado, United States, 80303 Rocky Mountain Cancer Centers - Midtown Denver, Colorado, United States, 80128 Rocky Mountain Cancer Centers - Littleton Littleton, Colorado, United States, 80120 Rocky Mountain Cancer Centers - Sky Ridge Lone Tree, Colorado, United States, 80124 Rocky Mountain Cancer Centers - Thornton Thornton, Colorado, United States, 80260 Collapse << |
| NCT01242800 | Breast Cancer | Phase 3 | Active, not recruiting | June 30, 2027 | - |
| NCT01198158 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Carcinoma Stage III Renal Cell Cancer AJCC v7 Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 3 | Terminated | - | - |
| NCT01099449 | Chemotherapeutic Agent Toxicit... more >>y Colorectal Cancer Neuropathy Neurotoxicity Collapse << | Phase 3 | Completed | - | - |
| NCT00070564 | Breast Cancer | Phase 3 | Active, not recruiting | January 2027 | - |
| NCT00070564 | - | - | Active, not recruiting | - | - |
| NCT03018249 | Grade 1 Endometrial Endometrio... more >>id Adenocarcinoma Grade 2 Endometrial Endometrioid Adenocarcinoma Grade 3 Endometrial Endometrioid Adenocarcinoma Uterine Corpus Adenosarcoma Collapse << | Phase 2 | Active, not recruiting | December 31, 2020 | - |
| NCT00022633 | - | - | Terminated(Permanently Closed ... more >>Due to Poor Accrual) Collapse << | - | - |
| 靶点 | Description | IC50 |
|---|---|---|
| SPHK | IC50:0.5μM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
