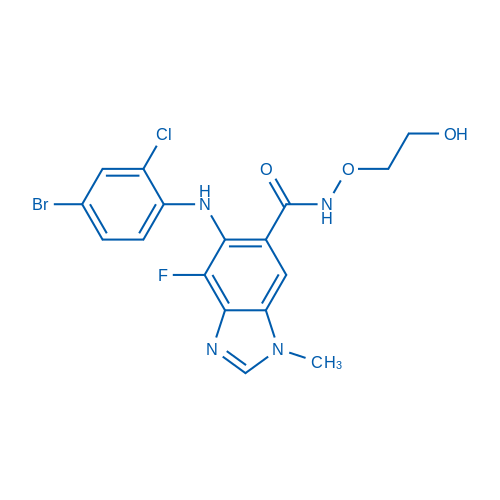
CAS No.: 606143-52-6
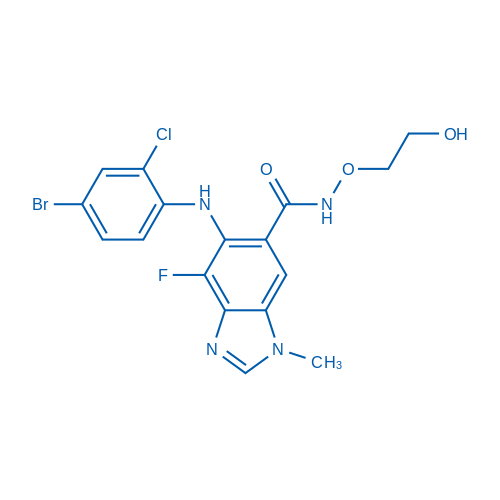
Selumetinib/司美替尼 Catalog No. CSN12386
Synonyms: 司美替尼;AZD6244;ARRY-142886
Selumetinib is a potent, highly selective MEK1 inhibitor with IC50 of 14 nM, also inhibits ERK1/2 phosphorylation with IC50 of 10 nM, no inhibition to p38α, MKK6, EGFR, ErbB2, ERK2, B-Raf, etc.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 ERK1
IC50:10nMERK2
IC50:10nMMEK1
IC50:14nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00085787 Advanced Cancer Phase 1 Completed - United States, Colorado ... more >> University of Colorado Cancer Center, Anschutz Cancer Center Aurora, Colorado, United States, 80010 United States, Minnesota Mayo Clinic Rochester Rochester, Minnesota, United States, 55905 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << NCT00780676 Breast Cancer Phase 2 Terminated(Closed early for fu... more >>tility.) Collapse << - United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << NCT02546661 Muscle Invasive Bladder Cancer Phase 1 Recruiting March 27, 2020 - - 更多
- 参考文献
- [1] Yeh TC, Marsh V, et al. Biological characterization of ARRY-142886 (AZD6244), a potent, highly selective mitogen-activated protein kinase kinase 1/2 inhibitor. Clin Cancer Res. 2007 Mar 1;13(5):1576-83.
- [2] Bernabé R, Patrao A, et al. Selumetinib in the treatment of non-small-cell lung cancer. Future Oncol. 2016 Nov;12(22):2545-2560.
- [3] Bartholomeusz C, Oishi T, et al. MEK1/2 inhibitor selumetinib (AZD6244) inhibits growth of ovarian clear cell carcinoma in a PEA-15-dependent manner in a mouse xenograft model. Mol Cancer Ther. 2012 Feb;11(2):360-9.
- [4] Denton CL, Gustafson DL, et al. Pharmacokinetics and pharmacodynamics of AZD6244 (ARRY-142886) in tumor-bearing nude mice. Cancer Chemother Pharmacol. 2011 Feb;67(2):349-60.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 606143-52-6 | 储存条件 |
|
|||||||||||
| 分子式 | C17H15BrClFN4O3 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 457.68 | 别名 | 司美替尼;AZD6244;ARRY-142886 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| human A101D cell | - | Growth inhibition assay | - | Inhibition of human A101D cell growth in a cell viability assay, IC50=240.33 nM. | SANGER |
| human A375 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human A375 cells expressing BRAF V600E mutant after 72 hrs by Cell titer-glo assay, IC50=31 nM. | 23474388 |
| human A549 cell | - | Growth inhibition assay | - | Inhibition of human A549 cell growth in a cell viability assay, IC50=214.13 nM. | SANGER |
| human AsPC-1 cell | - | Growth inhibition assay | - | Inhibition of human AsPC-1 cell growth in a cell viability assay, IC50=324.39 nM. | SANGER |
| human BHT-101 cell | - | Growth inhibition assay | - | Inhibition of human BHT-101 cell growth in a cell viability assay, IC50=106.93 nM. | SANGER |
| human BPH-1 cell | - | Growth inhibition assay | - | Inhibition of human BPH-1 cell growth in a cell viability assay, IC50=182.31 nM. | SANGER |
| human C2BBe1 cell | - | Growth inhibition assay | - | Inhibition of human C2BBe1 cell growth in a cell viability assay, IC50=272.59 nM. | SANGER |
| human C32 cell | - | Growth inhibition assay | - | Inhibition of human C32 cell growth in a cell viability assay, IC50=98.23 nM. | SANGER |
| human CAL-39 cell | - | Growth inhibition assay | - | Inhibition of human CAL-39 cell growth in a cell viability assay, IC50=332.26 nM. | SANGER |
| human CHP-212 cell | - | Growth inhibition assay | - | Inhibition of human CHP-212 cell growth in a cell viability assay, IC50=3.15 nM. | SANGER |
| human COLO-679 cell | - | Growth inhibition assay | - | Inhibition of human COLO-679 cell growth in a cell viability assay, IC50=341.19 nM. | SANGER |
| human CP50-MEL-B cell | - | Growth inhibition assay | - | Inhibition of human CP50-MEL-B cell growth in a cell viability assay, IC50=127.94 nM. | SANGER |
| human DOK cell | - | Growth inhibition assay | - | Inhibition of human DOK cell growth in a cell viability assay, IC50=147.08 nM. | SANGER |
| human DU-4475 cell | - | Growth inhibition assay | - | Inhibition of human DU-4475 cell growth in a cell viability assay, IC50=33.67 nM. | SANGER |
| human EoL-1-cell cell | - | Growth inhibition assay | - | Inhibition of human EoL-1-cell cell growth in a cell viability assay, IC50=144.76 nM. | SANGER |
| human H9 cell | - | Growth inhibition assay | - | Inhibition of human H9 cell growth in a cell viability assay, IC50=22.88 nM. | SANGER |
| human HCC2998 cell | - | Growth inhibition assay | - | Inhibition of human HCC2998 cell growth in a cell viability assay, IC50=269.07 nM. | SANGER |
| human H-EMC-SS cell | - | Growth inhibition assay | - | Inhibition of human H-EMC-SS cell growth in a cell viability assay, IC50=290.99 nM. | SANGER |
| human HL-60 cell | - | Growth inhibition assay | - | Inhibition of human HL-60 cell growth in a cell viability assay, IC50=24.59 nM. | SANGER |
| human HT-144 cell | - | Growth inhibition assay | - | Inhibition of human HT-144 cell growth in a cell viability assay, IC50=89.05 nM. | SANGER |
| human HTC-C3 cell | - | Growth inhibition assay | - | Inhibition of human HTC-C3 cell growth in a cell viability assay, IC50=214.61 nM. | SANGER |
| human HuP-T4 cell | - | Growth inhibition assay | - | Inhibition of human HuP-T4 cell growth in a cell viability assay, IC50=195.32 nM. | SANGER |
| human KU812 cell | - | Growth inhibition assay | - | Inhibition of human KU812 cell growth in a cell viability assay, IC50=211.68 nM. | SANGER |
| human KY821 cell | - | Growth inhibition assay | - | Inhibition of human KY821 cell growth in a cell viability assay, IC50=107.18 nM. | SANGER |
| human LAMA-84 cell | - | Growth inhibition assay | - | Inhibition of human LAMA-84 cell growth in a cell viability assay, IC50=456.22 nM. | SANGER |
| human LB2518-MEL cell | - | Growth inhibition assay | - | Inhibition of human LB2518-MEL cell growth in a cell viability assay, IC50=93.82 nM. | SANGER |
| human M14 cell | - | Growth inhibition assay | - | Inhibition of human M14 cell growth in a cell viability assay, IC50=36.89 nM. | SANGER |
| human MEL-HO cell | - | Growth inhibition assay | - | Inhibition of human MEL-HO cell growth in a cell viability assay, IC50=138.84 nM. | SANGER |
| human MIAPaCa2 cells | - | Proliferation assay | - | Antiproliferative activity against human MIAPaCa2 cells, IC50=142 nM. | 23474388 |
| human ML-2 cell | - | Growth inhibition assay | - | Inhibition of human ML-2 cell growth in a cell viability assay, IC50=293.63 nM. | SANGER |
| human MZ2-MEL cell | - | Growth inhibition assay | - | Inhibition of human MZ2-MEL cell growth in a cell viability assay, IC50=394.37 nM. | SANGER |
| human NCI-H1437 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H1437 cell growth in a cell viability assay, IC50=352.85 nM. | SANGER |
| human NCI-H747 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H747 cell growth in a cell viability assay, IC50=350.98 nM. | SANGER |
| human NKM-1 cell | - | Growth inhibition assay | - | Inhibition of human NKM-1 cell growth in a cell viability assay, IC50=385.8 nM | SANGER |
| human NOMO-1 cell | - | Growth inhibition assay | - | Inhibition of human NOMO-1 cell growth in a cell viability assay, IC50=31.97 nM. | SANGER |
| human ONS-76 cell | - | Growth inhibition assay | - | Inhibition of human ONS-76 cell growth in a cell viability assay, IC50=244.53 nM. | SANGER |
| human PSN1 cell | - | Growth inhibition assay | - | Inhibition of human PSN1 cell growth in a cell viability assay, IC50=366.09 nM. | SANGER |
| human RCM-1 cell | - | Growth inhibition assay | - | Inhibition of human RCM-1 cell growth in a cell viability assay, IC50=493.85 nM. | SANGER |
| human RKO cell | - | Growth inhibition assay | - | Inhibition of human RKO cell growth in a cell viability assay, IC50=248.38 nM. | SANGER |
| human RVH-421 cell | - | Growth inhibition assay | - | Inhibition of human RVH-421 cell growth in a cell viability assay, IC50=279.39 nM. | SANGER |
| human SH-4 cell | - | Growth inhibition assay | - | Inhibition of human SH-4 cell growth in a cell viability assay, IC50=166.48 nM. | SANGER |
| human SK-MEL-2 cell | - | Growth inhibition assay | - | Inhibition of human SK-MEL-2 cell growth in a cell viability assay, IC50=405.06 nM. | SANGER |
| human SK-N-AS cell | - | Growth inhibition assay | - | Inhibition of human SK-N-AS cell growth in a cell viability assay, IC50=92.83 nM. | SANGER |
| human SW620 cell | - | Growth inhibition assay | - | Inhibition of human SW620 cell growth in a cell viability assay, IC50=302.2 nM. | SANGER |
| human U-266 cell | - | Growth inhibition assay | - | Inhibition of human U-266 cell growth in a cell viability assay, IC50=487.74 nM. | SANGER |
| human UACC-257 cell | - | Growth inhibition assay | - | Inhibition of human UACC-257 cell growth in a cell viability assay, IC50=321.84 nM. | SANGER |
| human WM-115 cell | - | Growth inhibition assay | - | Inhibition of human WM-115 cell growth in a cell viability assay, IC50=267.54 nM. | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00085787 | Advanced Cancer | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado Cancer Center, Anschutz Cancer Center Aurora, Colorado, United States, 80010 United States, Minnesota Mayo Clinic Rochester Rochester, Minnesota, United States, 55905 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT00780676 | Breast Cancer | Phase 2 | Terminated(Closed early for fu... more >>tility.) Collapse << | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02546661 | Muscle Invasive Bladder Cancer | Phase 1 | Recruiting | March 27, 2020 | - |
| NCT00780676 | - | - | Terminated(Closed early for fu... more >>tility.) Collapse << | - | - |
| NCT02179671 | Locally Advanced or Metastatic... more >> Non-Small-Cell Lung Cancer (Stage IIIB-IV) Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Research Site Goodyear, Arizona, United States United States, District of Columbia Research Site Washington, District of Columbia, United States United States, Georgia Research Site Augusta, Georgia, United States Research Site Marietta, Georgia, United States United States, Kentucky Research Site Ashland, Kentucky, United States United States, Missouri Research Site St Louis, Missouri, United States United States, New York Research Site Mineola, New York, United States United States, North Carolina Research Site Huntersville, North Carolina, United States United States, Washington Research Site Spokane, Washington, United States Research Site Tacoma, Washington, United States Collapse << |
| NCT01229150 | Non Small Cell Lung Carcinoma | Phase 2 | Completed | - | United States, California ... more >> University of California, Davis Davis, California, United States, 95616 University of Southern California Health Sciences Campus Los Angeles, California, United States, 90033 City of Hope Medical Group South Pasadena, California, United States, 91030 United States, Illinois University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Maryland National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00936221 | Melanoma | Phase 2 | Completed | - | - |
| NCT01229150 | - | - | Completed | - | - |
| NCT00936221 | - | - | Completed | - | - |
| NCT01134601 | Non-Metastatic Adenocarcinoma ... more >>of the Rectum Collapse << | Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01160926 | Rectal Cancer | Phase 1 | Terminated(2 DLTs had been rep... more >>orted from first 4 patients on lowest possible dose cohort.) Collapse << | September 2016 | United Kingdom ... more >> The Christie NHS Foundation Trust Manchester, United Kingdom, M20 4BX Collapse << |
| NCT01750281 | Locally Advanced or Metastatic... more >> Non Small Cell Lung Cancer Stage IIIb - IV Collapse << | Phase 2 | Active, not recruiting | December 31, 2018 | - |
| NCT02143466 | Advanced Non Small Cell Lung C... more >>ancer Collapse << | Phase 1 | Recruiting | December 30, 2019 | - |
| NCT00866177 | Recurrent Melanoma ... more >> Stage III Skin Melanoma Stage IV Skin Melanoma Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01287130 | Colonic Neoplasms ... more >> Cancer of the Colon Colon Cancer Colon Neoplasms Colonic Cancer Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00710515 | Melanoma Mali... more >>gnant Melanoma Collapse << | Phase 1 | Completed | - | Netherlands ... more >> Research Site Amsterdam, Netherlands Research Site Maastricht, Netherlands United Kingdom Research Site Glasgow, United Kingdom Research Site Headington, United Kingdom Research Site London, United Kingdom Research Site Oxford, United Kingdom Collapse << |
| NCT01750281 | - | - | Active, not recruiting | - | - |
| NCT01143402 | - | - | Completed | - | - |
| NCT00866177 | - | - | Completed | - | - |
| NCT01166126 | Mucosal Melanoma ... more >> Recurrent Melanoma Stage IV Melanoma Collapse << | Phase 2 | Terminated | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00890825 | - | - | Completed | - | - |
| NCT01166126 | - | - | Terminated | - | - |
| NCT00514761 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | Brazil ... more >> Research Site Goiania, Brazil Research Site Rio de Janeiro, Brazil Research Site Sao Paulo, Brazil Collapse << |
| NCT01256359 | Melanoma | Phase 2 | Active, not recruiting | December 2018 | United Kingdom ... more >> Churchill Hospital Oxford, Oxfordshire, United Kingdom, OX3 7LJ Collapse << |
| NCT00372944 | Pancreatic Cancer | Phase 2 | Completed | - | United States, California ... more >> Research Site Alhambra, California, United States Research Site Bakersfield, California, United States Research Site Palm Springs, California, United States Research Site Santa Maria, California, United States United States, Florida Research Site Orlando, Florida, United States United States, Maryland Research Site Chevy Chase, Maryland, United States United States, Michigan Research Site Ann Arbor, Michigan, United States United States, New York Research Site New York, New York, United States United States, Tennessee Research Site Nashville, Tennessee, United States United States, Texas Research Site Temple, Texas, United States United States, Washington Research Site Seattle, Washington, United States Australia Research Site Heidelburg, Australia Bulgaria Research Site Plovdiv, Bulgaria Research Site Sofia, Bulgaria Research Site Varna, Bulgaria Hungary Research Site Budapest, Hungary Romania Research Site Cluj-Napoca, Romania Collapse << |
| NCT00890825 | Non Small Cell Lung Cancer | Phase 2 | Completed | - | - |
| NCT00600496 | Breast Cancer ... more >> Breast Neoplasms Colon Cancer Colonic Cancer Colon Neoplasms Lung Cancer Melanoma Kidney Cancer Collapse << | Phase 1 | Active, not recruiting | December 31, 2018 | United States, Michigan ... more >> Research Site Detroit, Michigan, United States, 48201 United States, Pennsylvania Research Site Philadelphia, Pennsylvania, United States, 19111 United States, Tennessee Research Site Nashville, Tennessee, United States, 37203 United States, Texas Research Site Houston, Texas, United States, 77030 Collapse << |
| NCT00372788 | Non-small Cell Lung Cancer | Phase 2 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States United States, Michigan Research Site Ann Arbor, Michigan, United States United States, New York Research Site New York, New York, United States United States, Tennessee Research Site Nashville, Tennessee, United States Bulgaria Research Site Plovdiv, Bulgaria Research Site Sofia, Bulgaria Research Site Varna, Bulgaria Romania Research Site Cluj-Napoca, Romania Collapse << |
| NCT01143402 | Iris Melanoma ... more >> Medium/Large Size Posterior Uveal Melanoma Ocular Melanoma With Extraocular Extension Recurrent Uveal Melanoma Small Size Posterior Uveal Melanoma Stage IV Uveal Melanoma Collapse << | Phase 2 | Completed | - | - |
| NCT01658943 | - | - | Completed | - | - |
| NCT01021748 | Locally Advanced or Metastatic... more >> Solid Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT00970359 | - | - | Completed | - | - |
| NCT01658943 | Pancreatic Acinar Cell Carcino... more >>ma Pancreatic Ductal Adenocarcinoma Recurrent Pancreatic Carcinoma Stage IV Pancreatic Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT01313039 | - | - | Completed | - | - |
| NCT00970359 | Thyroid Cancer | Not Applicable | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01021748 | - | - | Completed | - | - |
| NCT00338130 | Melanoma | Phase 2 | Completed | - | - |
| NCT01859182 | Adenocarcinoma of the Gallblad... more >>der Adenocarcinoma With Squamous Metaplasia of the Gallbladder Adult Primary Cholangiocellular Carcinoma Advanced Adult Primary Liver Cancer Cholangiocarcinoma of the Extrahepatic Bile Duct Localized Unresectable Adult Primary Liver Cancer Metastatic Extrahepatic Bile Duct Cancer Recurrent Adult Primary Liver Cancer Recurrent Extrahepatic Bile Duct Cancer Stage II Gallbladder Cancer Stage IIIA Gallbladder Cancer Stage IIIB Gallbladder Cancer Stage IVA Gallbladder Cancer Stage IVB Gallbladder Cancer Unresectable Extrahepatic Bile Duct Cancer Collapse << | Phase 2 | Withdrawn | - | - |
| NCT01160718 | Breast Cancer | Phase 2 | Completed | - | Belgium ... more >> Universitair Ziekenhuis Gent Ghent, Belgium, B-9000 U.Z. Gasthuisberg Leuven, Belgium, B-3000 Switzerland Kantonsspital Aarau Aarau, Switzerland, CH-5001 Kantonsspital Baden Baden, Switzerland, CH-5404 Universitaetsspital-Basel Basel, Switzerland, CH-4031 Inselspital Bern Bern, Switzerland, CH-3010 Spitalzentrum Biel Biel, Switzerland, CH-2501 Kantonsspital Graubuenden Chur, Switzerland, CH-7000 Brustzentrum Thurgau at Kantonsspital Frauenfeld Frauenfeld, Switzerland, 8501 Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland, CH-1011 Kantonsspital Luzern Luzerne, Switzerland, CH-6000 Oncology Institute of Southern Switzerland - Mendrisio Mendrisio, Switzerland, CH-6850 Hopital de Morges Morges, Switzerland, CH-1110 Kantonsspital - St. Gallen St. Gallen, Switzerland, CH-9007 Hopitaux Universitaires de Geneve Thonex-Geneve, Switzerland, CH-1226 Kantonsspital Winterthur Winterthur, Switzerland, CH-8400 City Hospital Triemli Zurich, Switzerland, CH-8063 Collapse << |
| NCT01364051 | Solid Neoplasm ... more >> Stage IV Cutaneous Melanoma AJCC v6 and v7 Collapse << | Phase 1 | Active, not recruiting | - | United States, Florida ... more >> Mayo Clinic in Florida Jacksonville, Florida, United States, 32224-9980 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT02685657 | Triple Negative Breast Cancer | Phase 2 | Unknown | - | - |
| NCT01333475 | Colorectal Neoplasms | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01313039 | Breast Cancer | Early Phase 1 | Completed | - | United States, Florida ... more >> University of Miami Sylvester Comprehensive Cancer Center Miami, Florida, United States, 33136 Collapse << |
| NCT02151084 | Biliary Tract Carcinoma ... more >> Gallbladder Carcinoma Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Cancer Centre Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT02337530 | Non-Small Cell Lung Cancer | Phase 2 | Active, not recruiting | December 31, 2018 | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia BCCA - Abbotsford Centre Abbotsford, British Columbia, Canada, V2S 0C2 BCCA - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, New Brunswick Regional Health Authority B, Zone 2 Saint John, New Brunswick, Canada, E2L 4L2 Canada, Ontario Juravinski Cancer Centre at Hamilton Health Sciences Hamilton, Ontario, Canada, L8V 5C2 Cancer Centre of Southeastern Ontario at Kingston Kingston, Ontario, Canada, K7L 5P9 Lakeridge Health Oshawa Oshawa, Ontario, Canada, L1G 2B9 Ottawa Hospital Research Institute Ottawa, Ontario, Canada, K1H 8L6 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 University Health Network Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec CHUM - Hopital Notre-Dame Montreal, Quebec, Canada, H2L 4M1 Collapse << |
| NCT01206140 | Recurrent Adult Soft Tissue Sa... more >>rcoma Stage III Adult Soft Tissue Sarcoma Stage IV Adult Soft Tissue Sarcoma Collapse << | Phase 2 | Completed | - | United States, California ... more >> Tower Cancer Research Foundation Beverly Hills, California, United States, 90211-1850 City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 USC / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 University of California Davis Phase 2 Consortium Sacramento, California, United States, 95817 United States, Pennsylvania Penn State Milton S Hershey Medical Center Hershey, Pennsylvania, United States, 17033-0850 University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT03259633 | - | - | - | - | - |
| NCT01206140 | - | - | Completed | - | - |
| NCT01333475 | - | - | Completed | - | - |
| NCT00604721 | - | - | Completed | - | - |
| NCT01217450 | Recurrent Colon Cancer ... more >> Recurrent Rectal Cancer Stage IVA Colon Cancer Stage IVA Rectal Cancer Stage IVB Colon Cancer Stage IVB Rectal Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute Medicine Branch Bethesda, Maryland, United States, 20892 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Wisconsin Clinical Cancer Center Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00463814 | Tumor Cancer | Phase 1 | Active, not recruiting | December 31, 2018 | United States, Colorado ... more >> Research Site Aurora, Colorado, United States, 80045 Netherlands Research Site Nijmegen, Netherlands, 6525 GA Research Site Utrecht, Netherlands, 3584 CX United Kingdom Research Site Sutton, United Kingdom, SM2 5PT Collapse << |
| NCT01362803 | Neurofibromatosis 1 ... more >> Neurofibromatosis Type 1 NF 1 Neurofibroma, Plexiform Collapse << | Phase 1 Phase 2 | Active, not recruiting | July 2, 2024 | United States, District of Col... more >>umbia Childrens National Medical Center Washington, District of Columbia, United States, 20010 United States, Maryland National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 United States, Pennsylvania Childrens Hospital, Philadelphia Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01222689 | Adenocarcinoma of the Pancreas... more >> Recurrent Pancreatic Cancer Stage III Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> UCSF-Mount Zion San Francisco, California, United States, 94115 United States, Ohio Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00604721 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Ohio Arthur G. James Cancer Hospital and Solove Research Institute at Ohio State University Medical Center Columbus, Ohio, United States, 43210 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37232 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT01586624 | Cancer Non Sm... more >>all Cell Lung Cancer Collapse << | Phase 1 | Active, not recruiting | March 2019 | United Kingdom ... more >> Cambridge Cancer Trials Centre (S4), Box 279, Addenbrooke's Hospital Hills Road, Cambridge, United Kingdom, CB2 0QQ The Christie NHS Foundation Trust Withington, Manchester, United Kingdom, M20 4BX Churchill Hospital Headington, Oxford, United Kingdom, OX3 7LJ The Freeman Hospital Newcastle, United Kingdom, NE7 7DN Collapse << |
| NCT01222689 | - | - | Completed | - | - |
| NCT00551070 | - | - | Active, not recruiting | - | - |
| NCT00553332 | - | - | Completed | - | - |
| NCT00551070 | Borderline Ovarian Epithelial ... more >>Tumor Low Grade Ovarian Serous Adenocarcinoma Primary Peritoneal Carcinoma Primary Peritoneal Low Grade Serous Adenocarcinoma Recurrent Borderline Ovarian Surface Epithelial-Stromal Tumor Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT02586987 | Lung Cancer M... more >>elanoma Head and Neck Carcinoma Gastroesophageal Cancer Breast Cancer Pancreatic Adenocarcinoma Colorectal Cancer Biliary Tract Cancer Collapse << | Phase 1 | Active, not recruiting | December 31, 2019 | United States, Illinois ... more >> Research Site Chicago, Illinois, United States, 60637 United States, Massachusetts Research Site Boston, Massachusetts, United States, 02215 United States, Nevada Research Site Las Vegas, Nevada, United States, 89169-3321 United States, New Jersey Research Site New Brunswick, New Jersey, United States, 8901 United States, North Carolina Research Site Charlotte, North Carolina, United States, 28204 United States, Pennsylvania Research Site Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00553332 | Liver and Intrahepatic Biliary... more >> Tract Cancer Recurrent Extrahepatic Bile Duct Cancer Unresectable Extrahepatic Bile Duct Cancer Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Michigan University of Michigan Cancer Center (UMCC) Research Base Ann Arbor, Michigan, United States, 48109-0352 Wayne State University Detroit, Michigan, United States, 48202 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Ohio Ohio State University Medical Center Columbus, Ohio, United States, 43210 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01519427 | Recurrent Melanoma ... more >> Stage IIIA Melanoma Stage IIIB Melanoma Stage IIIC Melanoma Stage IV Melanoma Collapse << | Phase 2 | Terminated(Study terminated du... more >>e to slow accrual) Collapse << | - | United States, Georgia ... more >> Emory University Atlanta, Georgia, United States, 30322 United States, New Jersey Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 United States, Virginia Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01029418 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Terminated(The phase II portio... more >>n was not conducted due to funding issue.) Collapse << | - | Singapore ... more >> National University Hospital Singapore Singapore, Singapore, 119074 National Cancer Centre Singapore Singapore, Singapore, 169610 Collapse << |
| NCT03649165 | Neurofibromatosis Type 1 (NF1)... more >>-Related Plexiform Neurofibromas (PNs) Healthy Participants Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02503358 | Stage IIIB Non-Small Cell Lung... more >> Cancer AJCC v7 Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 1 | Recruiting | September 1, 2019 | United States, Oregon ... more >> OHSU Knight Cancer Institute Recruiting Portland, Oregon, United States, 97239 Contact: Khaled A. Tolba 503-494-8666 tolba@ohsu.edu Principal Investigator: Khaled A. Tolba Collapse << |
| NCT01933932 | Locally Advanced or Metastatic... more >> Non Small Cell Lung Cancer Stage IIIb - IV Collapse << | Phase 3 | Active, not recruiting | December 31, 2018 | - |
| NCT01519427 | - | - | Terminated(Study terminated du... more >>e to slow accrual) Collapse << | - | - |
| NCT01011933 | - | - | Completed | - | - |
| NCT01933932 | - | - | Active, not recruiting | - | - |
| NCT01605916 | Neoplasms, Me... more >>tastatic Cancer, Non-Small Cell Lung Cancer Advanced Solid Malignancies Collapse << | Phase 1 | Completed | - | Japan ... more >> Research Site Fukuoka-shi, Japan Research Site Kashiwa-shi, Japan Research Site Nagoya-shi, Japan Collapse << |
| NCT01116271 | Colorectal Cancer | Phase 2 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States Research Site Palm Springs, California, United States United States, Colorado Research Site Aurora, Colorado, United States United States, Connecticut Research Site New Haven, Connecticut, United States United States, Delaware Research Site Newark, Delaware, United States United States, Florida Research Site Miami Beach, Florida, United States United States, New York Research Site New York, New York, United States United States, North Carolina Reserach Site Chapel Hill, North Carolina, United States Research Site Greenville, North Carolina, United States Research Site Washington, North Carolina, United States United States, Pennsylvania Research Site Philadelphia, Pennsylvania, United States United States, Washington Research Site Seattle, Washington, United States Collapse << |
| NCT01089101 | Low Grade Glioma ... more >> Recurrent Childhood Pilocytic Astrocytoma Recurrent Neurofibromatosis Type 1 Recurrent Visual Pathway Glioma Refractory Neurofibromatosis Type 1 Refractory Visual Pathway Glioma Collapse << | Phase 1 Phase 2 | Recruiting | - | United States, California ... more >> Children's Hospital Los Angeles Recruiting Los Angeles, California, United States, 90027 Contact: Girish Dhall 323-361-4629 Principal Investigator: Girish Dhall Lucile Packard Children's Hospital Stanford University Recruiting Palo Alto, California, United States, 94304 Contact: Michelle Monje 650-721-5750 Principal Investigator: Michelle Monje UCSF Medical Center-Mount Zion Active, not recruiting San Francisco, California, United States, 94115 UCSF Medical Center-Parnassus Recruiting San Francisco, California, United States, 94143 Contact: Anuradha Banerjee 877-827-3222 Sub-Investigator: Anuradha Banerjee Sub-Investigator: Michael D. Prados United States, Colorado Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Kathleen M. Dorris 720-777-8314 Principal Investigator: Kathleen M. Dorris United States, District of Columbia Children's National Medical Center Recruiting Washington, District of Columbia, United States, 20010 Contact: Eugene I. Hwang 202-476-5046 Principal Investigator: Eugene I. Hwang United States, Illinois Lurie Children's Hospital-Chicago Recruiting Chicago, Illinois, United States, 60611 Contact: Stewart Goldman 312-227-4844 Principal Investigator: Stewart Goldman Children's Memorial Outpatient Center in Lincoln Park Active, not recruiting Chicago, Illinois, United States, 60614 United States, Maryland National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: Katherine E. Warren 301-435-4683 warrenk@mail.nih.gov Principal Investigator: Katherine E. Warren United States, New York Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Ira J. Dunkel 212-639-2153 Principal Investigator: Ira J. Dunkel United States, North Carolina Duke University Medical Center Active, not recruiting Durham, North Carolina, United States, 27710 United States, Ohio Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Mariko D. DeWire-Schottmiller 513-803-1126 Principal Investigator: Mariko D. DeWire-Schottmiller United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC Recruiting Pittsburgh, Pennsylvania, United States, 15224 Contact: Ian F. Pollack 412-692-5881 Principal Investigator: Ian F. Pollack United States, Tennessee St. Jude Children's Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Giles W. Robinson 901-595-2907 Principal Investigator: Giles W. Robinson United States, Texas Baylor College of Medicine/Dan L Duncan Comprehensive Cancer Center Active, not recruiting Houston, Texas, United States, 77030 Texas Children's Hospital Recruiting Houston, Texas, United States, 77030 Contact: Patricia A. Baxter 832-824-4681 Principal Investigator: Patricia A. Baxter Collapse << |
| NCT02063230 | Solid Tumors | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Orlando, Florida, United States Collapse << |
| NCT01011933 | Endometrial Adenocarcinoma ... more >> Endometrial Adenosquamous Carcinoma Endometrial Clear Cell Adenocarcinoma Recurrent Uterine Corpus Carcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT00559949 | Recurrent Thyroid Gland Carcin... more >>oma Stage I Thyroid Gland Papillary Carcinoma Stage II Thyroid Gland Papillary Carcinoma Stage III Thyroid Gland Papillary Carcinoma Stage IV Thyroid Gland Papillary Carcinoma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Moffitt Cancer Center Tampa, Florida, United States, 33612 United States, Illinois University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, Tennessee Vanderbilt University/Ingram Cancer Center Nashville, Tennessee, United States, 37232 Canada, Ontario University Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT02393690 | Metastatic Thyroid Gland Carci... more >>noma Poorly Differentiated Thyroid Gland Carcinoma Recurrent Thyroid Gland Carcinoma Stage IV Thyroid Gland Follicular Carcinoma AJCC v7 Stage IV Thyroid Gland Papillary Carcinoma AJCC v7 Stage IVA Thyroid Gland Follicular Carcinoma AJCC v7 Stage IVA Thyroid Gland Papillary Carcinoma AJCC v7 Stage IVB Thyroid Gland Follicular Carcinoma AJCC v7 Stage IVB Thyroid Gland Papillary Carcinoma AJCC v7 Stage IVC Thyroid Gland Follicular Carcinoma AJCC v7 Stage IVC Thyroid Gland Papillary Carcinoma AJCC v7 Collapse << | Phase 2 | Recruiting | August 1, 2020 | United States, California ... more >> UC San Diego Moores Cancer Center Recruiting La Jolla, California, United States, 92093 Contact: Gerald Henderson 858-822-5223 gehenderson@ucsd.edu Principal Investigator: Gregory A. Daniels Hoag Memorial Hospital Recruiting Newport Beach, California, United States, 92663 Contact: Atessa Kiani 919-764-6586 atessa.kiani@hoag.org Principal Investigator: Michael J. Demeure United States, Colorado University of Colorado Hospital Recruiting Aurora, Colorado, United States, 80045 Contact: Paula Fisk 720-848-0676 paula.fisk@ucdenver.edu Principal Investigator: Bryan R. Haugen United States, District of Columbia MedStar Georgetown University Hospital Recruiting Washington, District of Columbia, United States, 20007 Contact: Lynette Wray 202-877-2749 lynette.wray@medstar.net Principal Investigator: Kenneth D. Burman United States, Massachusetts Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Lisa Freehafer 617-643-1751 lfreehafer@partners.org Principal Investigator: Lori J. Wirth United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Mabel Ryder 855-776-0015 Ryder.Mabel@mayo.edu Principal Investigator: Mabel Ryder United States, New York Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Jamie S. Walters 646-888-4450 waltersj@mskcc.org Principal Investigator: Alan L. Ho United States, North Carolina Duke University Medical Center Withdrawn Durham, North Carolina, United States, 27710 United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Mamdouh Beshara 614-292-4511 Mamdouh.Beshara@osumc.edu Principal Investigator: Jennifer A. Sipos United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Debra J. Nichols 713-792-0839 djnichols@mdanderson.org Principal Investigator: Ramona Dadu Collapse << |
| NCT02063230 | - | - | Completed | - | - |
| NCT03581487 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IV Lung Cancer Stage IVA Lung Cancer Stage IVB Lung Cancer Collapse << | Phase 1 Phase 2 | Not yet recruiting | April 16, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Not yet recruiting Houston, Texas, United States, 77030 Contact dlgibbon@mdanderson.org Collapse << |
| NCT02407405 | Neurofibromatosis Type 1 ... more >> Plexiform Neurofibroma Collapse << | Phase 2 | Recruiting | - | United States, Maryland ... more >> National Cancer Institute Pediatric Oncology Branch Recruiting Bethesda, Maryland, United States, 20892 Contact: Brigitte C. Widemann 800-411-1222 Principal Investigator: Brigitte C. Widemann National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: Brigitte C. Widemann 800-411-1222 Sub-Investigator: Brigitte C. Widemann Sub-Investigator: Diana L. Bradford Sub-Investigator: A P. Chen Sub-Investigator: John W. Glod Sub-Investigator: Geraldine O'Sullivan Coyne Sub-Investigator: Alessandra Brofferio Sub-Investigator: Edward W. Cowen Sub-Investigator: Bradford J. Wood Sub-Investigator: John (Jack) F. Shern Collapse << |
| NCT00559949 | - | - | Completed | - | - |
| NCT00588809 | Adult Acute Myeloid Leukemia W... more >>ith t(15;17)(q22;q12) Adult Acute Promyelocytic Leukemia (M3) Myelodysplastic Syndromes Myelodysplastic/Myeloproliferative Neoplasms Recurrent Adult Acute Myeloid Leukemia Secondary Acute Myeloid Leukemia Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637-1470 Collapse << |
| NCT01635023 | Healthy Volunteers | Phase 1 | Completed | - | United States, Kansas ... more >> Reseatch Site Overland Park, Kansas, United States Collapse << |
| NCT01061749 | Adult Solid Neoplasm | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 Collapse << |
| NCT00588809 | - | - | Completed | - | - |
| NCT00888134 | Adult Solid Neoplasm | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Cancer Center Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Massachusetts General Hospital Charlestown, Massachusetts, United States, 02129 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00888134 | - | - | Completed | - | - |
| NCT01085214 | - | - | Completed | - | - |
| NCT01605916 | - | - | Completed | - | - |
| NCT01085214 | Recurrent Plasma Cell Myeloma ... more >> Refractory Plasma Cell Myeloma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Moffitt Cancer Center Tampa, Florida, United States, 33612 United States, Georgia Emory University/Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Maryland University of Maryland/Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 Mark O Hatfield-Warren Grant Magnuson Clinical Center Bethesda, Maryland, United States, 20892 National Institutes of Health Bethesda, Maryland, United States, 20892 United States, Montana Billings Clinic Cancer Center Billings, Montana, United States, 59107 United States, North Carolina University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 United States, Tennessee Vanderbilt University/Ingram Cancer Center Nashville, Tennessee, United States, 37232 United States, Virginia Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT02056392 | - | - | Completed | - | - |
| NCT03109301 | Neoplasms, Nerve Tissue ... more >> Neurofibromatosis 1 Heredodegenerative Disorders, Nervous System Peripheral Nervous System Diseases Collapse << | Phase 2 | Recruiting | February 1, 2023 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01809210 | Locally Advanced or Metastatic... more >> NSCL Cancer Stage IIIB IV Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Glasgow, United Kingdom, G12 0YN Research Site London, United Kingdom, W1G 6AD Research Site Manchester, United Kingdom, M20 4BX Research Site Newcastle upon Tyne, United Kingdom, NE7 7DN Collapse << |
| NCT01843062 | Differentiated Thyroid Cancer | Phase 3 | Active, not recruiting | April 29, 2019 | - |
| NCT01146756 | Non Small Cell Lung Cancer | Phase 1 | Completed | - | United Kingdom ... more >> The Christie NHS Foundation Trust Manchester, Greater Manchester, United Kingdom, M20 4BX Collapse << |
| NCT01783197 | Non Small Cell Lung Cancer | Phase 1 | Active, not recruiting | December 2018 | Canada, British Columbia ... more >> BCCA - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Juravinski Cancer Centre at Hamilton Health Sciences Hamilton, Ontario, Canada, L8V 5C2 Cancer Centre of Southeastern Ontario at Kingston Kingston, Ontario, Canada, K7L 5P9 Ottawa Hospital Research Institute Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT01278615 | Recurrent Adult Diffuse Large ... more >>Cell Lymphoma Collapse << | Phase 2 | Terminated(Closed Prematurely.... more >>) Collapse << | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637 Decatur Memorial Hospital Decatur, Illinois, United States, 62526 NorthShore University HealthSystem-Evanston Hospital Evanston, Illinois, United States, 60201 Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 Illinois CancerCare-Peoria Peoria, Illinois, United States, 61615 Southern Illinois University Springfield, Illinois, United States, 62702 United States, Indiana Fort Wayne Medical Oncology and Hematology Inc-Parkview Fort Wayne, Indiana, United States, 46845 Indiana University/Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, Maryland University of Maryland/Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 United States, Massachusetts Tufts Medical Center Boston, Massachusetts, United States, 02111 University of Massachusetts Memorial Health Care Worcester, Massachusetts, United States, 01605 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Missouri Saint John's Mercy Medical Center Saint Louis, Missouri, United States, 63141 United States, New York Weill Medical College of Cornell University New York, New York, United States, 10065 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02093728 | Solid Tumours | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01278615 | - | - | Terminated(Closed Prematurely.... more >>) Collapse << | - | - |
| NCT01809210 | - | - | Completed | - | - |
| NCT02448290 | Gastric Adenocarcinoma | Phase 2 | Recruiting | October 2018 | Korea, Republic of ... more >> Samsung Medical center Recruiting Seoul, Korea, Republic of, 135-710 Contact: Yoonjeong Ahn 82-2-2148-7395 younjeong.ahn@samsung.com Principal Investigator: Jeeyun Lee, MD,PhD Collapse << |
| NCT03004105 | Malignant Neoplasm of Respirat... more >>ory and Intrathoracic Organ Carcinoma Advanced Lung Cancer Recurrent Nonsmall Cell Lung Cancer Collapse << | Phase 2 | Withdrawn(Inadequate funding) | May 2024 | - |
| NCT01242605 | Biliary Tract Neoplasms ... more >> Cholangiocarcinoma Gallbladder Neoplasms Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Hammersmith Hospital London, United Kingdom University College London Hospital London, United Kingdom The Christie Hospital Manchester, United Kingdom Collapse << |
| NCT02056392 | Solid Tumours | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT02188264 | Recurrent Colorectal Carcinoma... more >> Solid Neoplasm Stage IIIA Colorectal Cancer AJCC v7 Stage IIIB Colorectal Cancer AJCC v7 Stage IIIC Colorectal Cancer AJCC v7 Stage IVA Colorectal Cancer AJCC v7 Stage IVB Colorectal Cancer AJCC v7 Collapse << | Phase 1 | Active, not recruiting | - | United States, California ... more >> University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 United States, Colorado University of Colorado Hospital Aurora, Colorado, United States, 80045 United States, Missouri Siteman Cancer Center at West County Hospital Creve Coeur, Missouri, United States, 63141 Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, New Jersey Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Ohio Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 United States, Pennsylvania University of Pittsburgh Cancer Institute (UPCI) Pittsburgh, Pennsylvania, United States, 15232 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 Canada, Ontario University Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT03095248 | Neurofibromatosis 2 ... more >> Vestibular Schwannoma Meningioma Ependymoma Glioma Collapse << | Phase 2 | Recruiting | May 2021 | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Laura Fossett 513-636-2799 cancer@cchmc.org Collapse << |
| NCT01949870 | Inoperable Locally Advanced or... more >> Metastatic Biliary Tract Cancer Collapse << | Phase 1 | Terminated(change in the devel... more >>opment strategy) Collapse << | - | Japan ... more >> AZD6244 PhI Japanese Gem/ Kashiwa Shi, Chiba, Japan AZD6244 PhI Japanese Gem/ Chuo Ku, Tokyo, Japan Collapse << |
| NCT02025114 | Non-small Cell Lung Cancer (NS... more >>CLC) Collapse << | Phase 1 Phase 2 | Completed | - | Taiwan ... more >> National Cheng Kung University Hospital Tainan, Taiwan, 70403 Department of Oncology, National Taiwan University Hospital Taipei, Taiwan, 100 Taipei Veterans General Hospital Taipei, Taiwan, 11217 Collapse << |
| NCT01974752 | Metastatic Uv... more >>eal Melanoma Collapse << | Phase 3 | Completed | - | - |
| NCT01974752 | - | - | Completed | - | - |
| NCT03213691 | Advanced Malignant Solid Neopl... more >>asm Ann Arbor Stage III Childhood Non-Hodgkin Lymphoma Ann Arbor Stage IV Childhood Non-Hodgkin Lymphoma BRAF Gene Mutation GNA11 Gene Mutation GNAQ Gene Mutation Histiocytosis HRAS Gene Mutation KRAS Gene Mutation NF1 Gene Mutation NRAS Gene Mutation Recurrent Childhood Central Nervous System Neoplasm Recurrent Childhood Non-Hodgkin Lymphoma Recurrent Malignant Solid Neoplasm Recurrent Neuroblastoma Refractory Central Nervous System Neoplasm Refractory Malignant Solid Neoplasm Refractory Neuroblastoma Refractory Non-Hodgkin Lymphoma Collapse << | Phase 2 | Recruiting | September 30, 2024 | - |
| NCT02046850 | Solid Tumours | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT02238782 | Healthy Volunteers Bioavailabi... more >>lity Study Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Nottingham, United Kingdom Collapse << |
| NCT02839720 | Appearance Distress ... more >> Bothered by Itching Skin Cafe Au Lait Spot Cutaneous Neurofibroma Disfigurement Dysplasia Lisch Nodule Neurofibromatosis Type 1 NF1 Gene Mutation Optic Nerve Glioma Collapse << | Phase 2 | Recruiting | December 31, 2018 | United States, Alabama ... more >> University of Alabama at Birmingham Cancer Center Recruiting Birmingham, Alabama, United States, 35233 Contact: Bruce R. Korf 205-934-9411 bkorf@uabmc.edu Principal Investigator: Bruce R. Korf United States, Maryland National Institutes of Health Clinical Center Not yet recruiting Bethesda, Maryland, United States, 20892 Contact: Brigitte C. Widemann 301-496-0738 ext 7 widemanb@mail.nih.gov Principal Investigator: Brigitte C. Widemann United States, Texas UT Southwestern/Simmons Cancer Center-Dallas Not yet recruiting Dallas, Texas, United States, 75390 Contact: Lu Q. Le 214-648-7097 lu.le@utsouthwestern.edu Principal Investigator: Lu Q. Le Collapse << |
| NCT02238782 | - | - | Completed | - | - |
| NCT02063204 | Solid Tumours | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Orlando, Florida, United States Collapse << |
| NCT02322749 | Healthy Volunteers Bioequivale... more >>nce or Bioavailability Study Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT03392246 | Non-small Cell Lung Cancer | Phase 2 | Recruiting | June 30, 2025 | United States, Massachusetts ... more >> Massachusetts General Hospital Not yet recruiting Boston, Massachusetts, United States, 02214 Contact: Zofia Piotrowska, MD zpiotrowska@partners.org Principal Investigator: Zofia Piotrowska, MD Beth Israel Deaconess Medical Center Not yet recruiting Boston, Massachusetts, United States, 02215 Contact: Daniel Costa, MD dbcosta@bidmc.harvard.edu Principal Investigator: Daniel Costa, MD Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Sarah E Clifford 617-632-5438 SarahE_Clifford@DFCI.HARVARD.EDU Principal Investigator: Pasi A Janne, MD Collapse << |
| NCT02322749 | - | - | Completed | - | - |
| NCT01960374 | Healthy Volunteers Pharmacokin... more >>etic Study Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT01931761 | Solid Tumours | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Ruddington, United Kingdom Collapse << |
| NCT02583542 | Triple-Negative Breast Cancer ... more >> Squamous Cell Lung Cancer Non-squamous Cell Lung Cancer With KRAS Mutations Non-squamous Cell Lung Cancer With Wild-type KRAS Collapse << | Phase 1 Phase 2 | Recruiting | July 2019 | United Kingdom ... more >> University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, United Kingdom, B15 2TT Contact: Gary Middleton, Professor +44 (0) 1213713573 Gary.Middleton@uhb.nhs.uk Contact: Ceri Amstrong +44 (0) 1214143795 c.j.armstrong@bham.ac.uk Principal Investigator: Gary Middleton, Professor Barts Health NHS Trust Recruiting London, United Kingdom, EC1M 6BQ Contact: Peter Schmid +44 (0)20 7882 8498 p.schmid@qmul.ac.uk Contact: Louise Lim farahlouise.lim@bartshealth.nhs.uk Principal Investigator: Peter Schmid Sub-Investigator: Louise Lim University College London Hospitals NHS Foundation Trust Recruiting London, United Kingdom, W1T 7HA Contact: Martin Forster, Dr +44 (0) 2034475085 Martin.Forster@uclh.nhs.uk Contact: Magda Rocha +44 (0) 2034472921 Magda.PintoRocha@uclh.nhs.uk Principal Investigator: Martin Forster, Dr Christie NHS Foundation Trust Recruiting Manchester, United Kingdom, M20 4BX Contact: Yvonne Summers, Dr +44 (0) 161 446 8016 yvonne.summers@christie.nhs.uk Contact: Rachael Finney +44 (0) 1619182318 rachael.finney@christie.nhs.uk Principal Investigator: Yvonne Summers, Dr Collapse << |
| NCT03040986 | KRAS NP_004976.2:p.G12R ... more >> Stage III Pancreatic Cancer AJCC v6 and v7 Stage IV Pancreatic Cancer AJCC v6 and v7 Collapse << | Phase 2 | Suspended(Interim monitoring) | December 31, 2021 | United States, California ... more >> Los Angeles County-USC Medical Center Los Angeles, California, United States, 90033 USC / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 United States, Kentucky University of Kentucky/Markey Cancer Center Lexington, Kentucky, United States, 40536 United States, Maryland NCI - Center for Cancer Research Bethesda, Maryland, United States, 20892 United States, Michigan Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 United States, Pennsylvania University of Pittsburgh Cancer Institute (UPCI) Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT01949870 | - | - | Terminated(change in the devel... more >>opment strategy) Collapse << | - | - |
| NCT01974349 | Solid Tumours | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT03162627 | Malignant Neoplasm of Breast ... more >> Malignant Neoplasms of Digestive Organs Malignant Neoplasms of Female Genital Organs Malignant Neoplasms of Male Genital Organs Malignant Neoplasms of Thyroid and Other Endocrine Glands Collapse << | Phase 1 | Recruiting | August 2026 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact swestin@mdanderson.org Collapse << |
| NCT03705507 | Acute Lymphoblastic Leukemia ... more >> Acute Lymphoblastic Leukemia, Adult Acute Lymphoblastic Leukemia, Pediatric Acute Lymphoblastic Leukemia, in Relapse Acute Lymphoblastic Leukemia Recurrent Collapse << | Phase 1 Phase 2 | Recruiting | March 17, 2021 | United Kingdom ... more >> Birmingham Children's Hospital Recruiting Birmingham, United Kingdom Principal Investigator: Alice Norton Alder Hey Children's Hospital Recruiting Liverpool, United Kingdom Principal Investigator: Mark Caswell Department of Paediatric Oncology, Royal Marsden Hospital, Sutton Recruiting London Borough of Sutton, United Kingdom Principal Investigator: Donna Lancaster Haemato-Oncology Adult Unit, Royal Marsden Hospital, Sutton Recruiting London Borough of Sutton, United Kingdom Sub-Investigator: David Taussig University College Hospital Recruiting London, United Kingdom Principal Investigator: Adele Fielding The Christie Hospital Recruiting Manchester, United Kingdom Principal Investigator: Anna Castleton Freeman Hospital Recruiting Newcastle, United Kingdom Sub-Investigator: Tobias Menne Great North Children's Hospital, Royal Victoria Infirmary Recruiting Newcastle, United Kingdom Principal Investigator: Geoff Shenton Collapse << |
| NCT01752569 | AIDS-related Kaposi's Sarcoma | Phase 1 Phase 2 | Active, not recruiting | June 2018 | United Kingdom ... more >> Brighton and Sussex University Hospitals Brighton, United Kingdom, BN2 5BE Beatson West of Scotland Cancer Centre Glasgow, United Kingdom, G12 0YN Chelsea & Westminster Hospital London, United Kingdom, SW10 9NH The Christie Hospital Manchester, United Kingdom, M20 4BX Sheffield Teaching Hospitals NHS Foundation Trust Sheffield, United Kingdom, S10 2SJ Collapse << |
| NCT03745989 | Solid Tumors | Phase 1 | Not yet recruiting | May 6, 2022 | - |
| NCT03433183 | Malignant Peripheral Nerve She... more >>ath Tumors Neurofibromatosis 1 Collapse << | Phase 2 | Not yet recruiting | September 2021 | - |
| NCT03326388 | Neurofibromatosis Type 1 ... more >> Plexiform Neurofibroma Optic Nerve Glioma Collapse << | Phase 1 Phase 2 | Not yet recruiting | December 2023 | United Kingdom ... more >> Great Ormond Street Hospital NHS Foundatin Trust Not yet recruiting London, United Kingdom, WC1N 3JH Contact: Darren Hargrave, MB Bch 0207 405 9200 ext 8832 Darren.Hargrave@gosh.nhs.uk Contact: Patricia O'Hare, MB Bch BAO 0207 405 9200 ext 8832 Patricia.O'Hare@gosh.nhs.uk Collapse << |
| NCT02768766 | Uveal Melanoma | Phase 1 | Recruiting | January 2019 | United States, New York ... more >> Columbia University Medical Center Recruiting New York, New York, United States, 10032 Contact: Richard Carvajal, MD rdc2150@cumc.columbia.edu Principal Investigator: Richard Carvajal, MD Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Alexander N Shoushtari, M.D 646-888-4161 shoushta@mskcc.org United States, Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Sapna Patel, M.D 713-792-4955 sppatel@mdanderson.org Collapse << |
| NCT03326310 | Chronic Myeloid Leukemia ... more >> Myelofibroses Collapse << | Phase 1 | Recruiting | September 4, 2023 | United States, Illinois ... more >> The University of Chicago Recruiting Chicago, Illinois, United States, 60637 Contact: Melissa Fridstein 773-702-9885 mfridstein@medicine.bsd.uchicago.edu Collapse << |
| NCT02450656 | Colorectal Neoplasms ... more >> Gastrointestinal Neoplasms Pancreatic Neoplasms Carcinoma, Non-Small-Cell Lung Collapse << | Phase 1 Phase 2 | Recruiting | December 2019 | Netherlands ... more >> UMC St. Radboud Nijmegen Recruiting Nijmegen, Gelderland, Netherlands, 6525GA Contact: Carla ML van Herpen, MD, PhD +31243610353 Carla.vanHerpen@radboudumc.nl Principal Investigator: Carla ML van Herpen, MD, PhD Netherlands Cancer Institute - Antoni van Leeuwenhoek Hospital Recruiting Amsterdam, Netherlands, 1066CX Contact: F Opdam, MD, PhD Principal Investigator: F Opdam, MD, PhD Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| ERK1 | IC50:10nM | |
| ERK2 | IC50:10nM | |
| MEK1 | IC50:14nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
