CAS No.: 128517-07-7
Romidepsin/罗米地辛 Catalog No. CSN13496
Synonyms: FK 228;FR 901228;NSC 630176
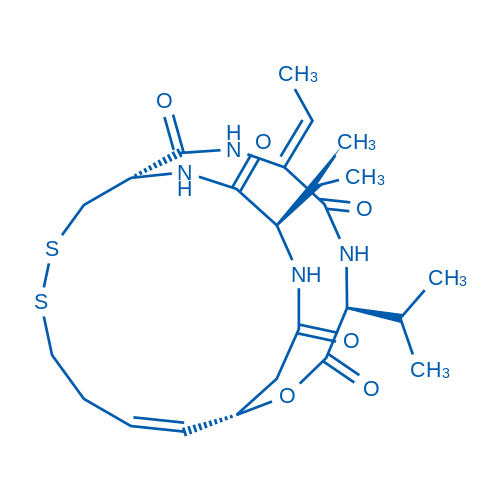
Romidepsin is a potent HDAC1 and HDAC2 inhibitor with IC50 of 36 nM and 47 nM, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 HDAC1
IC50:36nMHDAC2
IC50:47nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02661178 Healthy Volunteers Phase 1 Completed - United Kingdom ... more >> Hammersmith Medicines Research London, United Kingdom, NW10 7EW Collapse << NCT01663571 - - Terminated(Study is permanentl... more >>y closed to enrollment .Collection of private identifiable information is complete Analysis of private identifiable information is complete) Collapse << - United States, New York ... more >> NYU Langone Medical Center New York, New York, United States, 10016 Collapse << NCT03432741 Breast Adenocarcinoma ... more >> Recurrent Breast Carcinoma Recurrent Hodgkin Lymphoma Recurrent Mycosis Fungoides Recurrent Non-Hodgkin Lymphoma Recurrent Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Refractory Hodgkin Lymphoma Refractory Mycosis Fungoides Refractory Nodal Marginal Zone Lymphoma Refractory Non-Hodgkin Lymphoma Refractory Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Stage IV Breast Cancer AJCC v6 and v7 Collapse << Phase 1 Recruiting May 1, 2019 United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Clinical Trials Referral Office 855-776-0015 Principal Investigator: Grzegorz S. Nowakowski Collapse << - 更多
- 参考文献
- [1] Furumai R, Matsuyama A, et al. FK228 (depsipeptide) as a natural prodrug that inhibits class I histone deacetylases. Cancer Res. 2002 Sep 1;62(17):4916-21.
- [2] Aron JL, Parthun MR, et al. Depsipeptide (FR901228) induces histone acetylation and inhibition of histone deacetylase in chronic lymphocytic leukemia cells concurrent with activation of caspase 8-mediated apoptosis and down-regulation of c-FLIP protein. Blood. 2003 Jul 15;102(2):652-8. Epub 2003 Mar 20.
- [3] Sandor V, Senderowicz A, et al. P21-dependent g(1)arrest with downregulation of cyclin D1 and upregulation of cyclin E by the histone deacetylase inhibitor FR901228. Br J Cancer. 2000 Sep;83(6):817-25.
- [4] Horodeński J, Pawlowski Z, et al. [Atrophia gyrata choroideae et retinae]. Klin Oczna. 1973 Aug;43(8):929-32.
- [5] Jain S, Jirau-Serrano X, et al. Preclinical pharmacologic evaluation of pralatrexate and romidepsin confirms potent synergy of the combination in a murine model of human T-cell lymphoma. Clin Cancer Res. 2015 May 1;21(9):2096-106.
- [6] Li Z, Chan KK, et al. A subnanogram API LC/MS/MS quantitation method for depsipeptide FR901228 and its preclinical pharmacokinetics. J Pharm Biomed Anal. 2000 Feb;22(1):33-44.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 128517-07-7 | 储存条件 |
|
|||||||||
| 分子式 | C24H36N4O6S2 | 运输 | 蓝冰 | |||||||||
| 分子量 | 540.70 | 别名 | FK 228;FR 901228;NSC 630176;Depsipeptide;NSC 630176. depsipeptide;US brand name: Istodax;罗米地辛 | |||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 11z | 3-100 nM | Kinase Assay | - | reduces HDAC enzymatic activity (IC50 = 6.5 ± 0.6 nmol/L) | 20605144 |
| 1765-92 | - | Growth Inhibition Assay | - | IC50=1.77 nM | 18566246 |
| 402-91 | - | Growth Inhibition Assay | - | IC50=1.26 nM | 18566246 |
| 697 | - | Growth Inhibition Assay | - | IC50 = 2.5 nM | 21538216 |
| 697-R | - | Growth Inhibition Assay | - | IC50 = 8.6 nM | 21538216 |
| A549 | - | Growth Inhibition Assay | - | IC50=1.26±0.24 nM | 24954856 |
| A549 | 10–100 nM | Growth Inhibition Assay | 24/36/48 h | inhibits cell growth in both concentration- and time-dependent manner | 24485799 |
| A549 | 5 nM | Growth Inhibition Assay | 24/48/72 h | inhibits cell growth time-dependently | 22106282 |
| A549 | - | Growth Inhibition Assay | - | IC50=3.24 nM | 18566246 |
| ACH-2 | 1-9 nM | Function Assay | 24 h | induces HIV-1 Env expression | 25149467 |
| B104 | 2 nM | Function Assay | 24/48/72 h | increases the surface expression of CD20 | 20686505 |
| Brca1 Null | 1–20nM | Growth Inhibition Assay | 72 h | reduces cell viability alone and combined with cisplatin | 23010348 |
| Brca1 WT | 1–20nM | Growth Inhibition Assay | 72 h | reduces cell viability alone and combined with cisplatin | 23010348 |
| BT16 | 10 nM | Function Assay | 24/48/72 h | increases CDKN1C expression | 19221586 |
| CA46 | - | Apoptosis Assay | 6 h | induces blunt apoptosis | 23966164 |
| CD4 T | - | Growth Inhibition Assay | 48 h | EC50=4.5±1.0 nM | 24722454 |
| CD4 T | - | Growth Inhibition Assay | 48 h | CC50=107±126 nM | 24722454 |
| CD4+ T | - | Growth Inhibition Assay | - | EC50=3 nM | 24495105 |
| CO115 | 5 nM-50 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a concentration-dependent manner | 22924958 |
| DG75 | - | Apoptosis Assay | 6 h | induces no apoptosis | 23966164 |
| DpP75 | 1/10/100 nM | Apoptosis Assay | 48 h | induces blunt apoptosis | 23532732 |
| DpVp35 | 1/10/100 nM | Apoptosis Assay | 48 h | induces blunt apoptosis | 23532732 |
| DpVp50 | 1/10/100 nM | Apoptosis Assay | 48 h | induces blunt apoptosis | 23532732 |
| DTC1 | - | Growth Inhibition Assay | - | IC50=0.51 nM | 18566246 |
| EKVX | - | Growth Inhibition Assay | - | IC50=1.33±0.34 nM | 24954856 |
| Farage | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| FUJI | - | Growth Inhibition Assay | - | IC50=1.31 nM | 18566246 |
| G401 | 10 nM | Function Assay | 24/48/72 h | increases CDKN1C expression | 19221586 |
| Granta-519 | 5-40 nM | Growth Inhibition Assay | 24 h | IC50=58.5 nM | 20068080 |
| H146 | - | Growth Inhibition Assay | - | IC50=0.22±0.07 nM | 24954856 |
| H460 | - | Growth Inhibition Assay | - | IC50=2.58±0.80 nM | 24954856 |
| H526 | - | Growth Inhibition Assay | - | IC50=0.15±0.03 nM | 24954856 |
| HA | 0.625-10nM | Growth Inhibition Assay | 48 h | induces a significantly stronger inhibition of cell proliferation co-treated with bortezomib | 24771510 |
| HBL-2 | 2-10 nM | Growth Inhibition Assay | 24 h | IC50=4.3 nM | 20068080 |
| HCC95 | - | Growth Inhibition Assay | - | IC50=2.5±0.05 ng/ml | 19179890 |
| HCT116 | 5-5000 nM | Growth Inhibition Assay | 24 h | induces cell death in a concentration-dependent manner | 25492515 |
| HCT116 | - | Growth Inhibition Assay | - | IC50=1.00±0.00 nM | 24954856 |
| HCT116 | 5 nM-50 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a concentration-dependent manner | 22924958 |
| HCT116 | 20 nM | Function Assay | 8 h | modulates transcript levels for hundreds of genes in either direction | 20739454 |
| HCT116-p21-/- | - | Growth Inhibition Assay | - | IC50=1.26±0.37 nM | 24954856 |
| HFS | 5 nM | Growth Inhibition Assay | 24/48/72 h | inhibits cell growth time-dependently | 22106282 |
| HL60 | - | Growth Inhibition Assay | - | IC50=1.86 nM | 18566246 |
| HL-60 | 1-500 nM | Cytotoxicity Assay | 24 h | induces cytotoxicity in a concentration-dependent manner | 20624163 |
| HL-60 | 10 nM | Function Assay | 4/6/16 h | induces the generation of hydrogen peroxide from 4h | 20624163 |
| HL-60 | 10-500 nM | Function Assay | 4 h | decreases the histone deacetylase (HDAC) activity | 20624163 |
| hMSC-001F | - | Growth Inhibition Assay | - | IC50=>10 nM | 18566246 |
| HP100 | 1-500 nM | Cytotoxicity Assay | 24 h | induces cytotoxicity in a concentration-dependent manner | 20624163 |
| HP100 | 10 nM | Function Assay | 4/6/16 h | induces the generation of hydrogen peroxide from 4h | 20624163 |
| HP100 | 10-500 nM | Function Assay | 4 h | decreases the histone deacetylase (HDAC) activity | 20624163 |
| Hs68 | - | Growth Inhibition Assay | - | IC50=>10 nM | 18566246 |
| HuT78 | 1/10/100 nM | Apoptosis Assay | 48 h | induces apoptosis at 1 nM | 23532732 |
| HUT78 | - | Growth Inhibition Assay | - | IC50=1 nM | 21198545 |
| HuT-78 | - | Growth Inhibition Assay | - | IC50=1.73±0.44 nM | 24954856 |
| Jeko-1 | 2-50 nM | Growth Inhibition Assay | 24 h | IC50=11 nM | 20068080 |
| JJN3 | - | Growth Inhibition Assay | 24/48 h | EC50<1 nM; 48 h | 24030150 |
| JN-DSRCT-1 | - | Growth Inhibition Assay | - | IC50=1.25 nM | 18566246 |
| KAO | - | Growth Inhibition Assay | - | IC50=0.91 nM | 18566246 |
| KM-H2 | 1 nM-100 μM | Cytotoxicity Assay | 48 h | EC50=0.58 μM | 19233470 |
| L1236 | 1 nM-100 μM | Cytotoxicity Assay | 48 h | EC50=0.07 μM | 19233470 |
| L428 | 1 nM-100 μM | Cytotoxicity Assay | 48 h | EC50=0.43 μM | 19233470 |
| L540Cy | 1 nM-100 μM | Cytotoxicity Assay | 48 h | EC50=0.16 μM | 19233470 |
| LNCaP | 5 nM | Growth Inhibition Assay | 24/48/72 h | inhibits cell growth time-dependently | 22106282 |
| LOX-IMVI | - | Growth Inhibition Assay | - | IC50=0.87±0.03 nM | 24954856 |
| LY7/EBV | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| MCF-10A | - | Growth Inhibition Assay | - | IC50=0.17±0.01 nM | 24954856 |
| MCF7 | - | Growth Inhibition Assay | - | IC50=3.55 nM | 18566246 |
| MCF-7 | - | Growth Inhibition Assay | - | IC50=1.10±0.20 nM | 24954856 |
| MDA-MB-231 | - | Growth Inhibition Assay | - | IC50=0.68±0.14 nM | 24954856 |
| MDA-MB-435 | - | Growth Inhibition Assay | - | IC50=0.90±0.06 nM | 24954856 |
| MMRU | - | Growth Inhibition Assay | - | IC50=2.57 nM | 18566246 |
| MS-275 | 0.625-10nM | Growth Inhibition Assay | 48 h | induces a significantly stronger inhibition of cell proliferation co-treated with bortezomib | 24771510 |
| NCI/ADR-RES | 1–20nM | Growth Inhibition Assay | 72 h | reduces cell viability alone and combined with cisplatin | 23010348 |
| NCI-2882 | - | Growth Inhibition Assay | - | IC50=1.6±0.04 ng/ml | 19179890 |
| NCI-H1299 | - | Growth Inhibition Assay | - | IC50=4.6±0.2 ng/ml | 19179890 |
| NCI-H157 | - | Growth Inhibition Assay | - | IC50=1.6±0.02 ng/ml | 19179890 |
| NCI-H1650 | - | Growth Inhibition Assay | - | IC50=4.9±0.3 ng/ml | 19179890 |
| NCI-H1975 | - | Growth Inhibition Assay | - | IC50=1.3±0.04 ng/ml | 19179890 |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=2.9±0.2 ng/ml | 19179890 |
| NCI-H460 | - | Growth Inhibition Assay | - | IC50=2.1±0.07 ng/ml | 19179890 |
| NCI-H820 | - | Growth Inhibition Assay | - | IC50=2.4±0.1 ng/ml | 19179890 |
| NMS-2PC | - | Growth Inhibition Assay | - | IC50=0.81 nM | 18566246 |
| OCI-LY7 | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| OPM-2 | - | Growth Inhibition Assay | 24/48 h | EC50s=1 nM; 48 h | 24030150 |
| OVCAR-3 | 4/8/16 nM | Growth Inhibition Assay | 48 h | inhibits cell growth in a concentration-dependent manner | 20404564 |
| OVCAR-8 | 1–20nM | Growth Inhibition Assay | 72 h | reduces cell viability alone and combined with cisplatin | 23010348 |
| PC3 | - | Growth Inhibition Assay | - | IC50=1.65±0.35 nM | 24954856 |
| PC-3 | - | Growth Inhibition Assay | - | IC50=2.51 nM | 18566246 |
| Ramos | - | Apoptosis Assay | 6 h | induces extensive apoptosis | 23966164 |
| RKO | 5 nM-50 μM | Growth Inhibition Assay | 24 h | inhibits cell growth in a concentration-dependent manner | 22924958 |
| RPMI-8226 | - | Growth Inhibition Assay | 24/48 h | EC50s=1.8 nM; 48 h | 24030150 |
| S1 | - | Growth Inhibition Assay | - | IC50=7.67±0.29 nM | 24954856 |
| SF-295 | - | Growth Inhibition Assay | - | IC50=0.88±0.15 nM | 24954856 |
| SJSC | 10 nM | Function Assay | 24/48/72 h | increases CDKN1C expression | 19221586 |
| SK-BR-3 | - | Growth Inhibition Assay | - | IC50=1.00±0.35 nM | 24954856 |
| SKNMC | - | Growth Inhibition Assay | - | IC50=1.17 nM | 18566246 |
| SKOV-3 | 1–20nM | Growth Inhibition Assay | 72 h | reduces cell viability alone and combined with cisplatin | 23010348 |
| SKOV-3 | 4/8/16 nM | Growth Inhibition Assay | 48 h | inhibits cell growth in a concentration-dependent manner | 20404564 |
| ST486 | - | Apoptosis Assay | 6 h | induces extensive apoptosis | 23966164 |
| STM91-01 | 10 nM | Function Assay | 24/48/72 h | increases CDKN1C expression | 19221586 |
| SU-CCS-1 | - | Growth Inhibition Assay | - | IC50=0.89 nM | 18566246 |
| SU-DHL4 | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| SW480 | - | Growth Inhibition Assay | - | IC50=2.69 nM | 18566246 |
| SW620 | - | Growth Inhibition Assay | - | IC50=0.93±0.29 nM | 24954856 |
| SYO-1 | - | Growth Inhibition Assay | - | IC50=0.67 nM | 18566246 |
| THJ-16T | 1 nM | Growth Inhibition Assay | 24 h | inhibits cell growth | 20810568 |
| U266 | - | Growth Inhibition Assay | 24/48 h | EC50s=10 nM; 48 h | 24030150 |
| U2932 | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| U2932/EBV | 2.5-15 nM | Cell Viability Assay | 72 h | induces cytotoxicity in a concentration-dependent manner | 25790907 |
| UACC-62 | - | Growth Inhibition Assay | - | IC50=0.56±0.16 nM | 24954856 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02661178 | Healthy Volunteers | Phase 1 | Completed | - | United Kingdom ... more >> Hammersmith Medicines Research London, United Kingdom, NW10 7EW Collapse << |
| NCT01663571 | - | - | Terminated(Study is permanentl... more >>y closed to enrollment .Collection of private identifiable information is complete Analysis of private identifiable information is complete) Collapse << | - | United States, New York ... more >> NYU Langone Medical Center New York, New York, United States, 10016 Collapse << |
| NCT03432741 | Breast Adenocarcinoma ... more >> Recurrent Breast Carcinoma Recurrent Hodgkin Lymphoma Recurrent Mycosis Fungoides Recurrent Non-Hodgkin Lymphoma Recurrent Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Refractory Hodgkin Lymphoma Refractory Mycosis Fungoides Refractory Nodal Marginal Zone Lymphoma Refractory Non-Hodgkin Lymphoma Refractory Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 1 | Recruiting | May 1, 2019 | United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Clinical Trials Referral Office 855-776-0015 Principal Investigator: Grzegorz S. Nowakowski Collapse << |
| NCT03593018 | Relapsed Angioimmunoblastic T-... more >>Cell Lymphoma Refractory Angioimmunoblastic T-cell Lymphoma Collapse << | Phase 3 | Recruiting | December 2021 | Belgium ... more >> A. Z. Sint-Jan Brugge-Oostende AV Recruiting Bruges, Belgium Contact: Sylvia SNAUWAERT, MD Cliniques Universitaires de Bruxelles - Hôpital Erasme Recruiting Bruxelles, Belgium Contact: Alice WOLFROMM, MD Université Catholique de Louvain Saint-Luc Recruiting Bruxelles, Belgium Contact: Nicole STRAETMANS, MD CHU de Liège Recruiting Liège, Belgium Contact: Christophe BONNET, MD CHU UCL Namur - Site Godinne Recruiting Yvoir, Belgium Contact: Marc ANDRE, MD France Institut d'Hématologie de Basse Normandie Recruiting Caen, France Contact: Gandhi DAMAJ, MD CHU de Clermont-Ferrand - Hôpital Estaing Recruiting Clermont-Ferrand, France Contact: Olivier TOURNILHAC, MD CHU Henri Mondor Recruiting Créteil, France Contact: Jehan DUPUIS, MD CHU de Dijon Recruiting Dijon, France Contact: Olivier CASASNOVAS, MD CHU de Grenoble Recruiting Grenoble, France Contact: Rémy GRESSIN, MD CHRU de Lille Recruiting Lille, France Contact: Franck MORSCHHAUSER, MD CHU de Limoges Recruiting Limoges, France Contact: Céline KENNEL, MD CHU de Montpellier - Hôpital Saint-Eloi Recruiting Montpellier, France Contact: Guillaume CARTRON, MD CHU de Nantes - Hôtel Dieu Recruiting Nantes, France Contact: Steven LE GOUILL, MD Hôpital Necker Recruiting Paris, France Contact: Richard DELARUE, MD Hôpital Saint-Louis Recruiting Paris, France Contact: Catherine THIEBLEMONT, MD CHU Haut-Lévèque - Centre François Magendie Recruiting Pessac, France Contact: Kamal BOUABDALLAH, MD CHU Lyon-Sud Recruiting Pierre-Bénite, France Contact: Emmanuel BACHY, MD CH Annecy Genevois Recruiting Pringy, France Contact: Nicolas DAGUINDAU, MD CHU Pontchaillou Recruiting Rennes, France Contact: Roch HOUOT, MD Centre Henri Becquerel Recruiting Rouen, France Contact: Hervé TILLY, MD IUCT - Oncopole Recruiting Toulouse, France Contact: Loïc YSEBAERT, MD CHRU de Nancy - Hôpital de Brabois Recruiting Vandœuvre-lès-Nancy, France Contact: Lauriane CLEMENT-FILLIATRE, MD Collapse << |
| NCT03703375 | Lymphoma, T-Cell | Phase 3 | Not yet recruiting | December 31, 2021 | Japan ... more >> National Hospital Organization Nagoya Medical Center Not yet recruiting Nagoya, Aichi, Japan National Cancer Center Hospital East Not yet recruiting Kashiwa, Chiba, Japan Kyushu University Hospital Not yet recruiting Higashi-ku, Fukuoka, Japan University of Tsukuba Hospital Not yet recruiting Tsukuba, Ibataki, Japan Tokai University Hospital Not yet recruiting Isehara, Kanagawa, Japan National University Corporation Tohoku University Hospital Not yet recruiting Sendai, Miyagi, Japan Kindai University Hospital Not yet recruiting Osaka-Sayama, Osaka, Japan Saitama Medical University International Medical Center Not yet recruiting Hidaka, Saitama, Japan National Cancer Center Hospital Not yet recruiting Chuo-ku, Tokyo, Japan The Cancer Institute Hospital of Japanese Foundation For Cancer Research Not yet recruiting Koto-ku, Tokyo, Japan Collapse << |
| NCT02551718 | Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00037817 | Advanced Esophageal Cancers ... more >> Primary Small Cell Lung Cancers Non-Small-Cell Lung Carcinoma Pleural Mesotheliomas Cancers of Non-thoracic Origin With Metastases to the Lungs or Pleura Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01482962 | - | - | Completed | - | - |
| NCT01482962 | Relapsed Peripheral T-Cell Lym... more >>phoma Refractory Peripheral T-Cell Lymphoma Collapse << | Phase 3 | Completed | - | - |
| NCT00094978 | Carcinoma, Small Cell ... more >> Carcinoma, Non-Small-Cell Lung Esophageal Neoplasms Mesothelioma Collapse << | Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00299351 | Peripheral T Cell Lymphoma (PT... more >>CL) Collapse << | Phase 2 | Unknown | - | - |
| NCT02512172 | Colorectal Cancer | Phase 1 | Recruiting | January 2019 | United States, Maryland ... more >> Johns Hopkins Recruiting Baltimore, Maryland, United States, 21205 Principal Investigator: Nilofer Azad, MD Collapse << |
| NCT03619278 | HIV Infections | Phase 1 Phase 2 | Not yet recruiting | July 15, 2020 | - |
| NCT00020202 | Extensive Stage Small Cell Lun... more >>g Cancer Stage IIIA Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Recurrent Small Cell Lung Cancer Stage IIIB Non-small Cell Lung Cancer Recurrent Non-small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Surgery Branch Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00079443 | Recurrent Grade 1 Follicular L... more >>ymphoma Recurrent Grade 2 Follicular Lymphoma Recurrent Small Lymphocytic Lymphoma Collapse << | Phase 2 | Terminated | - | United States, Maryland ... more >> University of Maryland Greenebaum Cancer Center Baltimore, Maryland, United States, 21201-1595 Collapse << |
| NCT02223208 | Peripheral T-cell Lymphomas (P... more >>TCL) PTCL-NOS Angioimmunoblastic T-cell Lymphoma (AITL) ALK- Anaplastic Large Cell Lymphoma (ALCL) Collapse << | Phase 1 Phase 2 | Recruiting | March 2025 | Italy ... more >> Ospedale SS. Antonio e Biagio e Cesare Arrigo Recruiting Alessandria, AL, Italy, 15121 Contact: Manuela Zanni, MD 0039 0131206133 manuela.zanni@ospedale.al.it Principal Investigator: Flavia Salvi, MD Ospedali Riuniti Umberto I Not yet recruiting Ancona, AN, Italy, 60126 Contact: Attilio Olivieri, PROF 0039 0715964226 a.olivieri@unipm.it Principal Investigator: Attilio Olivieri, PROF AOU Policlinico di Bari Not yet recruiting Bari, BA, Italy, 70124 Contact: Giorgina Specchia, PROF 0039 0805593471 giorgina.specchia@uniba.it Principal Investigator: Giorgina Specchia, PROF Policlinico S. Orsola Malpighi Recruiting Bologna, BO, Italy, 40138 Contact: Vittorio Stefoni, MD 0039 0516364042 vittorio.stefoni2@unibo.it Principal Investigator: Vittorio Stefoni, MD Spedali Civili Recruiting Brescia, BS, Italy, 26123 Contact: Alessandro Re, MD 0039 0303995438 ematologia@spedalicivili.it Principal Investigator: Alessandro Re, MD Ospedale Businco Recruiting Cagliari, CA, Italy, 09121 Contact: Giuseppina Cabras, MD 0039 0706095171 cabras.giuseppina@tiscali.it Principal Investigator: Giuseppina Cabras, MD Fondazione di Ricerca e Cura "Giovanni Paolo II" Not yet recruiting Campobasso, CB, Italy, 86100 Contact: Sergio Storti, MD 0039 0874312317 sstorti@rm.unicatt.it Principal Investigator: Sergio Storti, MD Azienda Ospedaliera S.Croce e Carle Recruiting Cuneo, CN, Italy, 12100 Contact: Claudia Castellino, MD 0039 0171641070 castellino.c@ospedale.cuneo.it Principal Investigator: Claudia Castellino, MD Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (I.R.S.T.) - Sede di Meldola Not yet recruiting Meldola, FC, Italy, 47014 Contact: Gerardo Musuraca, MD 0039 0543739280 g.musuraca@irst.em.it Principal Investigator: Gerardo Musuraca, MD IRCCS Casa Sollievo Della Sofferenza Not yet recruiting San Giovanni Rotondo, FG, Italy, 71013 Contact: Nicola Cascavilla, MD 0039 0882410566 ematologia@operapadrepio.it Principal Investigator: Nicola Cascavilla, MD IRCCS AOU San Martino - Clinica Ematologica Not yet recruiting Genova, GE, Italy, 16321 Contact: Filippo Ballerini, PROF 0039 0105554336 ballerini@unige.it Principal Investigator: Filippo Ballerini, PROF IRCCS AOU San Martino - UO Ematologia 1 Recruiting Genova, GE, Italy, 16321 Contact: Angela Giovanna Congiu, MD 0039 0105513731 angelagiovanna.congiu@hsanmartino.it Principal Investigator: Angela Giovanna Congiu, MD Istituto Clinico Humanitas Terminated Rozzano, Milano, Italy, 20089 Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico Recruiting Milano, MI, Italy, 20122 Contact: Luca Baldini, MD 0039 02 55033334 luca.baldini@unimi.it Principal Investigator: Luca Baldini, PROF Fondazione IRCCS "Istituto Nazionale dei Tumori" Recruiting Milano, MI, Italy, 20133 Contact: Paolo Corradini, PROF 0039 0223902950 paolo.corradini@unimi.it Principal Investigator: Paolo Corradini, PROF Azienda Ospedaliera Ospedale Niguarda Ca' Granda Recruiting Milano, MI, Italy, 20162 Contact: Chiara Rusconi, MD 0039 02 64447597 chiara.rusconi@ospedaleniguarda.it Principal Investigator: Chiara Rusconi, MD AO Ospedali Riuniti Villa Sofia - Cervello (Presidio Cervello) Not yet recruiting Palermo, PA, Italy, 90146 Contact: Caterina Patti, MD 0039 0917803162 kpatti@ospedaliriunitipalermo.it Principal Investigator: Caterina Patti, MD P.O. di Pescara-Ospedale Civile Spirito Santo Not yet recruiting Pescara, PE, Italy, 65126 Contact: Francesco Angrilli, MD 0039 0854252838 f.angrilli@virgilio.it Principal Investigator: Francesco Angrilli, MD IRCCS Centro di Riferimento Oncologico (CRO) Not yet recruiting Aviano, PN, Italy, 33081 Contact: Michele Spina, MD 0039 043459730 mspina@cro.it Principal Investigator: Michele Spina, MD AOU di Parma Not yet recruiting Parma, PR, Italy, 43100 Contact: Francesca Re, MD 0039 0521 703962 fre@ao.pr.it Principal Investigator: Francesca Re, MD Fondazione IRCCS - Policlinico San Matteo Recruiting Pavia, PV, Italy, 27100 Contact: Luca Arcaini, MD 0039 0382501381 luca.arcaini@unipv.it Principal Investigator: Luca Arcaini, MD IRCCS Centro di Riferimento Oncologico della Basilicata (CROB) Not yet recruiting Rionero in Vulture, PZ, Italy, 85028 Contact: Roberto Guariglia, MD 0039 0972726225 robertoguariglia@libero.it Principal Investigator: Roberto Guariglia, MD A.O. Bianchi - Melacrino - Morelli Not yet recruiting Reggio Calabria, RC, Italy, 89125 Contact: Caterina Stelitano, MD 0039 0965397653 cstelit@libero.it Principal Investigator: Caterina Stelitano, MD IRCCS Arcispedale "Santa Maria Nuova" Not yet recruiting Reggio Emilia, RE, Italy, 42123 Contact: Francesco Merli, MD 0039 0522296618 merli.francesco@asmn.re.it Principal Investigator: Francesco Merli, MD Università di Roma "La Sapienza" Active, not recruiting Roma, RM, Italy, 00161 Università Cattolica del Sacro Cuore - Policlinico "A. Gemelli" Not yet recruiting Roma, RM, Italy, 00168 Contact: Stefan Hohaus, MD 0039 0630154278 stephan.hohaus@rm.unicatt.it Principal Investigator: Stefan Hohaus, MD Ospedale degli Infermi Not yet recruiting Rimini, RN, Italy, 47900 Contact: Annalia Molinari, MD 0039 0541705423 annalia.molinari@auslrn.net Principal Investigator: Annalia Molinari, MD AO Città della Salute e della Scienza - Ematologia 1U Not yet recruiting Torino, TO, Italy, 10126 Contact: Federica Cavallo, MD 0039 0116334198 f.cavallo@unito.it Principal Investigator: Federica Cavallo, MD AO Città della Salute e della Scienza - SC Ematologia Recruiting Torino, TO, Italy, 10126 Contact: Annalisa Chiappella, MD 0039 0116336751 achiappella@cittadellasalute.to.it Principal Investigator: Annalisa Chiappella, MD AOU "Santa Maria della Misericordia" Recruiting Udine, UD, Italy, 33100 Contact: Francesco Zaja, MD 0039 0432559604 zaja.francesco@aoud.sanita.fgv.it Principal Investigator: Francesco Zaja, MD Ospedale Borgo Roma Not yet recruiting Verona, VR, Italy, 37134 Contact: Fabio Benedetti, MD 0039 045 8124647 fabio.benedetti@univr.it Principal Investigator: Fabio Benedetti, MD Azienda Ospedaliero-Universitaria Careggi Not yet recruiting Firenze, Italy, 50134 Contact: Luigi Rigacci, MD 0039 055 7946448 luigi.rigacci@unifi.it Principal Investigator: Luigi Rigacci, MD Istituto Nazionale Tumori IRCCS - Fondazione G. Pascale Recruiting Napoli, Italy, 80131 Contact: Antonio Pinto, MD 0039 0815903381 a.pinto@istitutotumori.na.it Principal Investigator: Antonio Pinto, MD Università del Piemonte Orientale - SCDU Ematologia Recruiting Novara, Italy, 28100 Contact: Gianluca Gaidano, PROF 0039 03213732194 gaidano@med.unipmn.it Principal Investigator: Gianluca Gaidano, MD A.O. di Perugia - Santa Maria della Misericordia Not yet recruiting Perugia, Italy, 06132 Contact: Leonardo Flenghi, MD 0039 075 5784110 flenghi@yahoo.it Principal Investigator: Leonardo Flenghi, MD Ospedale G. Da Saliceto - AUSL di Piacenza Not yet recruiting Piacenza, Italy, 29121 Contact: Annalisa Arcari, MD 0039 0523 303719 a.arcari@ausl.pc.it Principal Investigator: Annalisa Arcari, MD UO Ematologia Ospedale S.Maria delle Croci Recruiting Ravenna, Italy, 48121 Contact: Monica Tani, MD 0039 0544286223 monica.tani@auslromagna.it Principal Investigator: Monica Tani, MD Collapse << |
| NCT00042822 | Leukemia Lymp... more >>homa Myelodysplastic Syndromes Myelodysplastic/Myeloproliferative Neoplasms Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00024180 | Leukemia Lymp... more >>homa Collapse << | Phase 1 | Completed | - | United States, Ohio ... more >> Arthur G. James Cancer Hospital - Ohio State University Columbus, Ohio, United States, 43210-1240 Collapse << |
| NCT00114257 | Chronic Myeloproliferative Dis... more >>orders Leukemia Myelodysplastic Syndromes Myelodysplastic/Myeloproliferative Neoplasms Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> M.D. Anderson Cancer Center at University of Texas Houston, Texas, United States, 77030-4009 Collapse << |
| NCT00077194 | Lymphoma | Phase 2 | Completed | - | United States, District of Col... more >>umbia Howard University Cancer Center at Howard University Hospital Washington, District of Columbia, United States, 20060 United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States, 21231 United States, Michigan Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 United States, Wisconsin University of Wisconsin Comprehensive Cancer Center Madison, Wisconsin, United States, 53792-5256 Collapse << |
| NCT00019318 | Lymphoma | Phase 1 | Completed | - | United States, Maryland ... more >> Medicine Branch Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00106613 | Carcinoma, Renal Cell ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States, 91010 United States, Illinois University of Chicago Chicago, Illinois, United States, 60637 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109-1023 Collapse << |
| NCT00007345 | Cutaneous T Cell Lymphoma ... more >> Peripheral T Cell Lymphoma Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Mayo Clinic Scottsdale Scottsdale, Arizona, United States, 85259 United States, Arkansas University of Arkansas Little Rock, Arkansas, United States, 72205 United States, California City of Hope National Cancer Center Duarte, California, United States, 91010 Mercy General Hospital Sacramento, California, United States, 95819 United States, District of Columbia Georgetown University Washington, District of Columbia, United States, 20007-2197 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Maryland National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 United States, New York North Shore University Hospital Manhasset, New York, United States, 11030 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15261 United States, West Virginia West Virginia University Morgantown, West Virginia, United States, 26506 Australia Royal Adelaide Hospital Adelaide, Australia Peter MacCallum Cancer Centre Melbourne, Australia Sir Charles Gairdner Hospital Perth, Australia Collapse << |
| NCT00066638 | DS Stage II Plasma Cell Myelom... more >>a DS Stage III Plasma Cell Myeloma Refractory Plasma Cell Myeloma Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Montefiore Medical Center - Moses Campus Bronx, New York, United States, 10467-2490 Collapse << |
| NCT00077337 | Colorectal Cancer | Phase 2 | Completed | - | - |
| NCT00431990 | Multiple Myeloma | Phase 1 Phase 2 | Unknown | January 2012 | Australia, Victoria ... more >> Peter MacCallum Cancer Centre Recruiting Melbourne, Victoria, Australia, 8006 Sub-Investigator: Alvin Milner Collapse << |
| NCT00053963 | Blastic Phase Chronic Myelogen... more >>ous Leukemia Childhood Central Nervous System Germ Cell Tumor Childhood Choroid Plexus Tumor Childhood Chronic Myelogenous Leukemia Childhood Craniopharyngioma Childhood Grade I Meningioma Childhood Grade II Meningioma Childhood Grade III Meningioma Childhood High-grade Cerebral Astrocytoma Childhood Infratentorial Ependymoma Childhood Low-grade Cerebral Astrocytoma Childhood Spinal Cord Neoplasm Childhood Supratentorial Ependymoma Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Recurrent Childhood Brain Stem Glioma Recurrent Childhood Cerebellar Astrocytoma Recurrent Childhood Cerebral Astrocytoma Recurrent Childhood Ependymoma Recurrent Childhood Medulloblastoma Recurrent Childhood Supratentorial Primitive Neuroectodermal Tumor Recurrent Childhood Visual Pathway and Hypothalamic Glioma Refractory Chronic Lymphocytic Leukemia Relapsing Chronic Myelogenous Leukemia Unspecified Childhood Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, California ... more >> Children's Oncology Group Arcadia, California, United States, 91006-3776 Collapse << |
| NCT01742793 | Hodgkin's Lymphoma ... more >> Mature T-cell Lymphoma Multiple Myeloma Collapse << | Phase 1 Phase 2 | Terminated | - | United States, Connecticut ... more >> Yale Cancer Center New Haven, Connecticut, United States, 06519 Collapse << |
| NCT00383565 | Recurrent Adult Diffuse Large ... more >>Cell Lymphoma Recurrent Mantle Cell Lymphoma Collapse << | Phase 2 | Terminated | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT03041012 | Hiv | Phase 2 | Recruiting | August 20, 2019 | Denmark ... more >> Department of Infectious Diseases Recruiting Aalborg, Denmark Contact: Henrik Nielsen Dept. of Infectious Diseases, Aarhus University Hospital Recruiting Aarhus, Denmark, 8200 Contact: Jesper D Gunst, MD +45 78452844 jesdam@rm.dk Department of Infectious Diseases Recruiting Hvidovre, Denmark Contact: Thomas Benfield Department of Infectious Diseases Recruiting København, Denmark Contact: Jan Gerstoft Department of Infectious Diseases Recruiting Odense, Denmark Contact: Isik S. Johansen United Kingdom Imperial College Healthcare NHS Trust Recruiting London, United Kingdom Contact: Amin Aamshah Principal Investigator: Sarah Fidler Collapse << |
| NCT00007345 | - | - | Completed | - | - |
| NCT02061449 | Cutaneous T-cell Lymphoma | Phase 1 | Active, not recruiting | March 2019 | United States, New York ... more >> NYU Cancer Institute New York, New York, United States, 10016 Collapse << |
| NCT00383565 | - | - | Terminated | - | - |
| NCT00087295 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Urethral Cancer Collapse << | Phase 2 | Terminated(Permanently Closed ... more >>Due to Poor Accrual) Collapse << | - | - |
| NCT00106301 | Carcinoma, Renal Cell ... more >> Prostatic Neoplasms Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States, 91010-3000 United States, Florida University of Florida Gainesville, Florida, United States, 32610 United States, Illinois University of Chicago Chicago, Illinois, United States, 60637-1470 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 United Kingdom Royal Marsden Hospital Sutton, United Kingdom, SM2 5PT Collapse << |
| NCT00048334 | Neoplasms | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02181218 | Lymphoma, T-Cell, Cutaneous ... more >> Lymphoma, T-Cell, Peripheral Hodgkin Disease Lymphoma, Large B-Cell, Diffuse Collapse << | Phase 1 | Recruiting | June 30, 2020 | United States, California ... more >> University of California - San Francisco Not yet recruiting San Francisco, California, United States, 94143 Contact: Weiyun Ai, M.D., Ph.D. 415-353-4061 weiai@medicine.ucsf.edu Principal Investigator: Weiyun Ai, M.D., Ph.D. United States, Illinois University of Chicago Recruiting Chicago, Illinois, United States, 60637 Contact: Peter Riedell, M.D. 773-753-3288 Principal Investigator: Peter Riedell, M.D. Sub-Investigator: Justin Kline, M.D. Sub-Investigator: Sonali Smith, M.D. Sub-Investigator: Kenneth Cohen, M.D. Sub-Investigator: Michael Bishop, M.D. United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Neha Mehta-Shah, M.D. 314-747-7402 mehta-n@wustl.edu Principal Investigator: Neha Mehta-Shah, M.D. Sub-Investigator: Nancy Bartlett, M.D. Sub-Investigator: Todd Fehniger, M.D., Ph.D. Sub-Investigator: Amanda Cashen, M.D. Sub-Investigator: Brad Kahl, M.D. Sub-Investigator: Peter Riedell, M.D. Collapse << |
| NCT00098527 | Adenocarcinoma of the Esophagu... more >>s Adenocarcinoma of the Stomach Recurrent Esophageal Cancer Recurrent Gastric Cancer Collapse << | Phase 2 | Terminated | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00098397 | Male Breast Cancer ... more >> Recurrent Breast Cancer Stage IV Breast Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02092116 | HIV I Infection | Phase 1 Phase 2 | Completed | - | Denmark ... more >> Aarhus University Hospital, Skejby Sygehus Aarhus N, Denmark, 8200 Collapse << |
| NCT00106418 | Prostate Cancer ... more >> Metastases Collapse << | Phase 2 | Completed | - | - |
| NCT01590732 | Mycosis Fungoides ... more >> Recurrent Anaplastic Large Cell Lymphoma Recurrent Angioimmunoblastic T-Cell Lymphoma Recurrent Enteropathy-Associated T-Cell Lymphoma Recurrent Hepatosplenic T-Cell Lymphoma Recurrent Peripheral T-Cell Lymphoma, Not Otherwise Specified Refractory Anaplastic Large Cell Lymphoma Refractory Angioimmunoblastic T-Cell Lymphoma Refractory Enteropathy-Associated T-Cell Lymphoma Refractory Hepatosplenic T-Cell Lymphoma Refractory Peripheral T-Cell Lymphoma, Not Otherwise Specified Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02092116 | - | - | Completed | - | - |
| NCT02783625 | Lymphoma Rela... more >>psed/Refractory T-cell Lymphomas Collapse << | Phase 1 | Recruiting | May 2019 | United States, California ... more >> Stanford University Medical Center Recruiting Stanford, California, United States, 94305-5408 Contact: Youn Kim, MD 650-498-6000 United States, Massachusetts Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02115 Contact: Eric Jacobsen, MD 617-582-9086 United States, Missouri University of Washington School of Medicine in St. Louis Recruiting Saint Louis, Missouri, United States, 63110 Contact: Neha Mehta-Shah, MD 314-362-5654 Principal Investigator: Neha Mehta-Shah, MD Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Neha Mehta-Shah, MD 314-362-5654 Principal Investigator: Neha Mehta-Shah, MD United States, New Jersey Memoral Sloan Kettering Cancer Center Basking Ridge Recruiting Basking Ridge, New Jersey, United States Contact: Steven Horwitz, MD 212-639-3045 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Steven Horwitz, MD 212-639-3045 United States, New York Memorial Sloan Kettering Cancer Center @ Commack Recruiting Commack, New York, United States, 11725 Contact: Steven Horwitz, MD 212-639-3045 Memoral Sloan Kettering Westchester Recruiting Harrison, New York, United States Contact: Steven Horwitz, MD 212-639-3045 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Steven Horwitz, MD 212-639-3045 Contact: Alison Moskowitz, MD 212-639-4839 Principal Investigator: Steven Horwitz, MD Memorial Sloan Kettering at Mercy Medical Center Recruiting Rockville Centre, New York, United States Contact: Steven Horwitz, MD 212-639-3045 Collapse << |
| NCT00098813 | - | - | Completed | - | - |
| NCT00084682 | - | - | Completed | - | - |
| NCT00062075 | Adult Acute Myeloid Leukemia W... more >>ith 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(15;17)(q22;q12) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Promyelocytic Leukemia (M3) Recurrent Adult Acute Myeloid Leukemia Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637-1470 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00098813 | Recurrent Thyroid Cancer ... more >> Stage IV Follicular Thyroid Cancer Stage IV Papillary Thyroid Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center at Suffolk Commack, New York, United States, 11725 Collapse << |
| NCT00086827 | Recurrent Small Cell Lung Canc... more >>er Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Ohio State University Columbus, Ohio, United States, 43210 Collapse << |
| NCT00106431 | Cutaneous T-cell Lymphoma | Phase 2 | Completed | - | United States, California ... more >> UCLA Jonsson Cancer Center Los Angeles, California, United States, 90095 Stanford Comprehensive Cancer Center Stanford, California, United States, 94305 United States, Massachusetts Boston Medical Center Boston, Massachusetts, United States, 02118 United States, Pennsylvania University of Pennsylvania Abrahamson Cancer Center Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 United States, Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 France Research Site Various Cities, France Germany Research Site Various Cities, Germany Poland Research Site Various Cities, Poland Russian Federation Research Site Various Cities, Russian Federation United Kingdom Research Site Various Cities, United Kingdom Collapse << |
| NCT00084682 | Stage IV Squamous Cell Carcino... more >>ma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Larynx Stage IV Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IV Squamous Cell Carcinoma of the Oropharynx Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Montefiore Medical Center Bronx, New York, United States, 10467-2490 Collapse << |
| NCT00106431 | - | - | Completed | - | - |
| NCT01280526 | Peripheral T Cell Lymphoma | Phase 1 Phase 2 | Completed | - | France ... more >> Hôpital Henri Mondor Créteil, France, 94010 CHU de Dijon Dijon, France, 21000 Hôpital Claude Huriez Lille, France, 59037 Centre Léon Bérard Lyon cedex 8, France, 69373 Hôpital St Louis Paris, France, 75475 Centre Hospitalier Lyon Sud Pierre-Bénite, France, 69495 Centre Henri Becquerel Rouen, France, 76038 Institut Gustave Roussy Villejuif, France, 94805 Collapse << |
| NCT00084461 | Gastrinoma Gl... more >>ucagonoma Insulinoma Metastatic Gastrointestinal Carcinoid Tumor Pancreatic Polypeptide Tumor Pulmonary Carcinoid Tumor Recurrent Gastrointestinal Carcinoid Tumor Recurrent Islet Cell Carcinoma Regional Gastrointestinal Carcinoid Tumor Somatostatinoma Collapse << | Phase 2 | Terminated | - | United States, Ohio ... more >> Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT03161223 | Lymphoma, T-Cell | Phase 1 Phase 2 | Recruiting | June 30, 2020 | United States, New York ... more >> Center for Lymphoid Malignancies at CUMC Recruiting New York, New York, United States, 10019 Contact: Michelle Malanga, MPA Principal Investigator: Owen A O'Connor, M.D., PhD Sub-Investigator: Jennifer E Amengual, M.D. Sub-Investigator: Ahmed Sawas, M.D. Sub-Investigator: Changchun Deng, M.D. Sub-Investigator: Lorenzo Falchi, M.D. Italy University of Bologna Not yet recruiting Bologna, Italy Principal Investigator: Pier Luigi Zinzani, M.D., PhD. Korea, Republic of Samsung Medical Center Not yet recruiting Seoul, Korea, Republic of Principal Investigator: Won Seog Kim, M.D., Ph.D. Collapse << |
| NCT00091195 | Primary Peritoneal Cavity Canc... more >>er Recurrent Ovarian Epithelial Cancer Collapse << | Phase 2 | Terminated(Administratively co... more >>mplete.) Collapse << | - | United States, North Carolina ... more >> High Point Regional Hospital High Point, North Carolina, United States, 27261 Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT01302808 | Lung Cancer M... more >>etastatic Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center - Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT00085527 | Ovarian Cancer | Phase 2 | Withdrawn | - | United States, Illinois ... more >> Cardinal Bernardin Cancer Center at Loyola University Medical Center Maywood, Illinois, United States, 60153 Collapse << |
| NCT00104884 | Malignant Melanoma ... more >> Melanoma Collapse << | Phase 2 | Terminated(The study was termi... more >>nated on May 17, 2006 due to slow accrual.) Collapse << | - | - |
| NCT01933594 | HIV Infections | Phase 1 Phase 2 | Completed | - | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, California UCLA CARE Center CRS Los Angeles, California, United States, 90035 United States, Colorado University of Colorado Hospital CRS Aurora, Colorado, United States, 80045 United States, Massachusetts Massachusetts General Hospital CRS (MGH CRS) Boston, Massachusetts, United States, 02114 United States, New York University of Rochester Adult HIV Therapeutic Strategies Network CRS Rochester, New York, United States, 14642 United States, North Carolina Chapel Hill CRS Chapel Hill, North Carolina, United States, 27599 United States, Ohio Ohio State University CRS Columbus, Ohio, United States, 43210 United States, Pennsylvania Penn Therapeutics, CRS Philadelphia, Pennsylvania, United States, 19104 University of Pittsburgh CRS Pittsburgh, Pennsylvania, United States, 15213 United States, Washington University of Washington AIDS CRS Seattle, Washington, United States, 98104-9929 Collapse << |
| NCT00379639 | - | - | Completed | - | - |
| NCT00112463 | - | - | Completed | - | - |
| NCT00104884 | - | - | Terminated(The study was termi... more >>nated on May 17, 2006 due to slow accrual.) Collapse << | - | - |
| NCT02281279 | B-cell Adult Acute Lymphoblast... more >>ic Leukemia Extranodal Marginal Zone B-cell Lymphoma of Mucosa-associated Lymphoid Tissue Intraocular Lymphoma Nodal Marginal Zone B-cell Lymphoma Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Burkitt Lymphoma Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Adult Diffuse Mixed Cell Lymphoma Recurrent Adult Diffuse Small Cleaved Cell Lymphoma Recurrent Adult Grade III Lymphomatoid Granulomatosis Recurrent Adult Immunoblastic Large Cell Lymphoma Recurrent Adult Lymphoblastic Lymphoma Recurrent Grade 1 Follicular Lymphoma Recurrent Grade 2 Follicular Lymphoma Recurrent Grade 3 Follicular Lymphoma Recurrent Mantle Cell Lymphoma Recurrent Marginal Zone Lymphoma Recurrent Small Lymphocytic Lymphoma Refractory Hairy Cell Leukemia Small Intestine Lymphoma Splenic Marginal Zone Lymphoma Testicular Lymphoma Waldenström Macroglobulinemia Collapse << | Phase 1 Phase 2 | Withdrawn | - | - |
| NCT02341014 | T-cell Lymphomas ... more >> Relapsed or Refractory Collapse << | Phase 1 Phase 2 | Active, not recruiting | January 2019 | United States, Nebraska ... more >> University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-7680 United States, New Jersey Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States, 11570 Collapse << |
| NCT00112463 | Adult Alveolar Soft-part Sarco... more >>ma Adult Angiosarcoma Adult Epithelioid Sarcoma Adult Extraskeletal Chondrosarcoma Adult Extraskeletal Osteosarcoma Adult Fibrosarcoma Adult Leiomyosarcoma Adult Liposarcoma Adult Malignant Fibrous Histiocytoma Adult Malignant Hemangiopericytoma Adult Malignant Mesenchymoma Adult Neurofibrosarcoma Adult Rhabdomyosarcoma Adult Synovial Sarcoma Gastrointestinal Stromal Tumor Metastatic Ewing Sarcoma/Peripheral Primitive Neuroectodermal Tumor Recurrent Adult Soft Tissue Sarcoma Recurrent Ewing Sarcoma/Peripheral Primitive Neuroectodermal Tumor Stage III Adult Soft Tissue Sarcoma Stage IV Adult Soft Tissue Sarcoma Collapse << | Phase 2 | Completed | - | - |
| NCT00085540 | Adult Anaplastic Astrocytoma ... more >> Adult Anaplastic Oligodendroglioma Adult Giant Cell Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> University of California Los Angeles Los Angeles, California, United States, 90095 University of California San Francisco San Francisco, California, United States, 94143 United States, Maryland National Cancer Institute Neuro-Oncology Branch Bethesda, Maryland, United States, 20814 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15232 United States, Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT03742921 | - | - | Not yet recruiting | April 17, 2023 | - |
| NCT00379639 | Pancreatic Cancer | Phase 1 | Completed | - | United States, Tennessee ... more >> Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00085540 | - | - | Completed | - | - |
| NCT01902225 | Lymphoma T-Ce... more >>ll Lymphoma Cutaneous Lymphoma Collapse << | Phase 1 | Recruiting | April 2019 | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center Recruiting San Francisco, California, United States, 94115 Contact: Weiyun Ai, MD, PHD 415-353-4061 weiyun.ai@ucsf.edu Principal Investigator: Weiyun Ai, MD, PhD United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Jonathan Branmmer, MD 614-293-2268 Jonathan.Brammer@osumc.edu Collapse << |
| NCT01897012 | MYC Positive ... more >>Recurrent B-Cell Non-Hodgkin Lymphoma Recurrent Burkitt Lymphoma Recurrent Diffuse Large B-Cell Lymphoma Recurrent Follicular Lymphoma Recurrent High Grade B-Cell Lymphoma With MYC and BCL2 or BCL6 Rearrangements Recurrent Hodgkin Lymphoma Recurrent Mantle Cell Lymphoma Recurrent Mature T- and NK-Cell Non-Hodgkin Lymphoma Refractory B-Cell Non-Hodgkin Lymphoma Refractory Burkitt Lymphoma Refractory Diffuse Large B-Cell Lymphoma Refractory Follicular Lymphoma Refractory High Grade B-Cell Lymphoma With MYC and BCL2 or BCL6 Rearrangements Refractory Hodgkin Lymphoma Refractory Mantle Cell Lymphoma Refractory Mature T-Cell and NK-Cell Non-Hodgkin Lymphoma Collapse << | Phase 1 | Active, not recruiting | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00963274 | Leukemia Lymp... more >>homa Collapse << | Phase 1 | Completed | - | United States, Illinois ... more >> Robert H. Lurie Comprehensive Cancer Center, Northwestern University Chicago, Illinois, United States, 60611 United States, Maryland University of Maryland Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Tennessee Vanderbilt-Ingram Cancer Center, Vanderbilt University Nashville, Tennessee, United States, 37232 United States, Virginia Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01908777 | T Cell Non-Hodgkin Lymphoma | Phase 2 | Recruiting | July 2020 | United States, Florida ... more >> Moffitt Cancer Center Recruiting Tampa, Florida, United States, 33612 Contact: Farhad Khimani, MD Contact 1-888-663-3488 Principal Investigator: Farhad Khimani, MD United States, New Jersey Memoral Sloan Kettering Cancer Center Recruiting Basking Ridge, New Jersey, United States Contact: Steven Horwitz, MD 212-639-3045 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Steve Horwitz, MD 212-639-3045 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Recruiting Commack, New York, United States, 11725 Contact: Steven Horwitz, MD 212-639-3045 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Steven Horwitz, MD 212-639-3045 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 11065 Contact: Steven Horwitz, MD 212-639-3045 Contact: Parastoo Dahi, MD 212-639-5846 Principal Investigator: Steven Horwitz, MD Weill Cornell Medical Center Recruiting New York, New York, United States Contact: Jia Ruan, MD Principal Investigator: Jia Ruan, MD Memorial Sloan Kettering at Mercy Medical Center Recruiting Rockville Centre, New York, United States Contact: Steven Horwitz, MD United States, Washington University of Washington School of Medicine Recruiting Seattle, Washington, United States, 98109 Contact: Andrei Shustov, MD Principal Investigator: Andrei Shustov, MD Collapse << |
| NCT01796002 | Peripheral T-cell Lymphoma | Phase 3 | Active, not recruiting | July 2024 | - |
| NCT02232516 | Adult Nasal Type Extranodal NK... more >>/T-cell Lymphoma Anaplastic Large Cell Lymphoma Angioimmunoblastic T-cell Lymphoma Hepatosplenic T-cell Lymphoma Peripheral T-cell Lymphoma Stage I Cutaneous T-cell Non-Hodgkin Lymphoma Stage IA Mycosis Fungoides/Sezary Syndrome Stage IB Mycosis Fungoides/Sezary Syndrome Stage II Cutaneous T-cell Non-Hodgkin Lymphoma Stage IIA Mycosis Fungoides/Sezary Syndrome Stage IIB Mycosis Fungoides/Sezary Syndrome Stage III Cutaneous T-cell Non-Hodgkin Lymphoma Stage IIIA Mycosis Fungoides/Sezary Syndrome Stage IIIB Mycosis Fungoides/Sezary Syndrome Stage IV Cutaneous T-cell Non-Hodgkin Lymphoma Stage IVA Mycosis Fungoides/Sezary Syndrome Stage IVB Mycosis Fungoides/Sezary Syndrome Collapse << | Phase 2 | Recruiting | August 2020 | United States, California ... more >> City of Hope Recruiting Duarte, California, United States, 91010 Contact: Jasmine Zain, MD 626-256-4673 Principal Investigator: Jasmine Zain, MD United States, Connecticut Yale University Not yet recruiting New Haven, Connecticut, United States, 06520 Contact: Francine M. Foss, MD 203-200-4363 Principal Investigator: Francine M. Foss, MD United States, Illinois Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Adam M. Petrich, MD 312-695-4537 Principal Investigator: Adam M. Petrich, MD United States, New York Weill Cornell Medicine Recruiting New York, New York, United States, 10021 Contact: Jia Ruan, MD 646-962-2064 Principal Investigator: Jia Ruan, MD United States, Washington Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Not yet recruiting Seattle, Washington, United States, 98109 Contact: Andrei R. Shustov 206-288-6739 Principal Investigator: Andrei R. Shustov, MD Collapse << |
| NCT02616874 | HIV | Phase 1 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Clinic Hospital Barcelona, Spain, 08036 Collapse << |
| NCT02616965 | Cutaneous T-cell Lymphoma (CTC... more >>L) Collapse << | Phase 1 | Recruiting | July 2021 | United States, Pennsylvania ... more >> Fox Chase Cancer Center Recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: Stefan K. Barta, MD 215-728-2674 Principal Investigator: Stefan K. Barta, MD Collapse << |
| NCT01947140 | Lymphoid Malignancies ... more >> Multiple Myeloma Lymphoma Hodgkin Lymphoma Non-hodgkin Lymphoma Collapse << | Phase 1 Phase 2 | Recruiting | December 2018 | United States, New York ... more >> Columbia University Medical Center Recruiting New York, New York, United States, 10019 Contact: Renee Lichtenstein 212-326-5720 rl2619@cumc.columbia.edu Principal Investigator: Jennifer E Amengual, MD Collapse << |
| NCT01456039 | Lymphoma, T-cell, Peripheral | Phase 1 Phase 2 | Active, not recruiting | June 30, 2018 | Japan ... more >> Nagoya Daini Red Cross Hospital Nagoya, Aichi, Japan, 466-8650 Nagoya City University Hospital Nagoya, Aichi, Japan, 467-8602 National Cancer Center Hospital East Kashiwa, Chiba, Japan, 277-8577: Ehime University Hospital To-on, Ehime, Japan, 791-0295 Chugoku Central Hospital Fukuyamashi, Hiroshima, Japan, 720-0001 Sapporo Hokuyu Hospital Sapporo, Hokkaido, Japan, 003-0006 Sapporo Medical University Hospital Sapporo, Hokkaido, Japan, 060-8543 Tokai University School of Medicine Isehara, Kanagawa, Japan, 259-1193 Kochi Medical School Hospital Nankoku, Kochi, Japan, 783-8505 Kinki University Hospital Sayama, Osaka, Japan, 589-8511 National Cancer Center Hospital Chuo, Tokyo, Japan, 104-0045 Japanese Red Cross Medical Center Shibuya, Tokyo, Japan, 150-8935 Kyushu University Hospital Fukuoka, Japan, 812-8582 Kumamoto University Hospital Kumamoto, Japan, 860-8556 University hospital, Kyoto prefectural University of Medicine Kyoto, Japan, 602-8566 Tohoku University Hospital Miyagi, Japan, 980-8574 The Cancer Institute Hospital of JFCR Tokyo, Japan, 135-8550 Collapse << |
| NCT02757248 | PTCL CTCL | Phase 1 | Withdrawn(Boehringer Ingelheim... more >> is discontinuing their volasertib development program) Collapse << | - | - |
| NCT01456039 | - | - | Active, not recruiting | - | - |
| NCT01998035 | Lymphoid Malignancies ... more >> Lymphoma Hodgkin Lymphoma Non-hodgkin Lymphoma Collapse << | Phase 1 Phase 2 | Recruiting | February 2019 | United States, New York ... more >> Columbia University Medical Center Recruiting New York, New York, United States, 10019 Contact: Celeste Rojas 212-326-5720 cr2393@cumc.columbia.edu Principal Investigator: Owen A O'Connor, MD, Ph.D. Collapse << |
| NCT01445340 | Mycosis Fungoides ... more >> Cutaneous T-Cell Lymphoma Neoplasms Collapse << | Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00765102 | - | - | Terminated(There was a change ... more >>in the Sponsor's research strategy; safety concerns were not a factor.) Collapse << | - | - |
| NCT02393794 | Triple-Negative Breast Cancer ... more >> Breast Cancer Collapse << | Phase 1 Phase 2 | Recruiting | July 2020 | United States, Kansas ... more >> University of Kansas Cancer Center - Clinical Research Center Recruiting Fairway, Kansas, United States, 66205 Contact: Priyanka Sharma, MD 913-588-6029 psharma2@kumc.edu Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu University of Kansas Cancer Center - West Recruiting Kansas City, Kansas, United States, 66112 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu University of Kansas Cancer Center - Overland Park Recruiting Overland Park, Kansas, United States, 66210 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu University of Kansas Cancer Center - Westwood Recruiting Westwood, Kansas, United States, 66205 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu United States, Missouri University of Kansas Cancer Center - South Recruiting Kansas City, Missouri, United States, 64131 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu University of Kansas Cancer Center - North Recruiting Kansas City, Missouri, United States, 64154 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu University of Kansas Cancer Center - Lee's Summit Recruiting Lee's Summit, Missouri, United States, 64064 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu Collapse << |
| NCT00426764 | - | - | Active, not recruiting | - | - |
| NCT00765102 | Multiple Myeloma | Phase 2 | Terminated(There was a change ... more >>in the Sponsor's research strategy; safety concerns were not a factor.) Collapse << | - | United States, California ... more >> Loma Linda University Cancer Center Loma Linda, California, United States, 92354 Desert Cancer Care, Inc Rancho Mirage, California, United States, 92270 Santa Barbara Hematology Oncology Medical Group, Inc. Santa Barbara, California, United States, 93105 James R Berenson, MD, Inc. West Hollywood, California, United States, 90069 United States, Georgia Georgia Cancer Specialists I, PC Atlanta, Georgia, United States, 30341 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 United States, North Carolina Mecklenburg Medical Group Charlotte, North Carolina, United States, 28204 United States, Texas Baylor Sammons Cancer Center Dallas, Texas, United States, 75246 Dallas Oncology Consultants, P.A. Duncanville, Texas, United States, 75137 Oncology Consultants, P.A Houston, Texas, United States, 77024 United States, Utah Central Utah Clinic, PC Provo, Utah, United States, 84604 United States, Washington Virginia Mason Medical Centre Seattle, Washington, United States, 98101 Collapse << |
| NCT01846390 | Peripheral T-Cell Lymphoma ... more >> Diffuse Large B-Cell Lymphoma Collapse << | Phase 1 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Canada, British Columbia BCCA - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, New Brunswick Regional Health Authority B, Zone 2 Saint John, New Brunswick, Canada, E2L 4L2 Canada, Nova Scotia QEII Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 1V7 Canada, Ontario Odette Cancer Centre Toronto, Ontario, Canada, M4N 3M5 Univ. Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01755975 | Multiple Myeloma ... more >> Non-Hodgkin's Lymphoma Collapse << | Phase 1 Phase 2 | Active, not recruiting | December 2019 | United States, Connecticut ... more >> Saint Francis/Mount Sinai Regional Cancer Center Hartford, Connecticut, United States, 06105 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-7680 United States, New Jersey Memorial Sloan Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Weill Cornell Medical Center New York, New York, United States Memorial Sloan Kettering Cancer Center at Mercy Rockville Centre, New York, United States, 11570 Collapse << |
| NCT00426764 | Peripheral T-cell Lymphoma | Phase 2 | Active, not recruiting | May 17, 2018 | - |
| NCT01324323 | - | - | Completed | - | - |
| NCT01979276 | Multiple Myeloma | Phase 1 Phase 2 | Terminated(this study was term... more >>inated due to losing financial support, and enrollment challenges) Collapse << | - | United States, New York ... more >> Weill Cornell Medical College New York, New York, United States, 10065 Collapse << |
| NCT03141203 | Peripheral T Cell Lymphoma | Phase 1 Phase 2 | Recruiting | February 2021 | United Kingdom ... more >> Queen Elizabeth Hospital Recruiting Birmingham, United Kingdom Principal Investigator: Ram Malladi St James's University Hospital Recruiting Leeds, United Kingdom Principal Investigator: Rod Johnson Leicester Royal Infirmary Recruiting Leicester, United Kingdom Principal Investigator: Simon Wagner Clatterbridge Cancer Centre Recruiting Liverpool, United Kingdom Principal Investigator: Nagesh Kalakonda Guy's Hospital Recruiting London, United Kingdom Principal Investigator: Paul Fields St Bartholomew's Hospital Recruiting London, United Kingdom Principal Investigator: Jeff Davies University College London Hospitals Recruiting London, United Kingdom Principal Investigator: Kate Cwynarski Christie Hospital Recruiting Manchester, United Kingdom Principal Investigator: Kim Linton Nottingham University Hospitals Recruiting Nottingham, United Kingdom Principal Investigator: Chris Fox Churchill Hospital Recruiting Oxford, United Kingdom Principal Investigator: Graham Collins Derriford Hospital Recruiting Plymouth, United Kingdom Principal Investigator: David Lewis Southampton General Hospital Recruiting Southampton, United Kingdom Principal Investigator: Andrew Davies The Royal Marsden NHS Foundation Trust Recruiting Sutton, United Kingdom, SM2 5PT Principal Investigator: Dima El-Sharkawi Collapse << |
| NCT01324323 | Hematologic Malignancy ... more >> Malignant Lymphoma Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Sarasota, Florida, United States, 34232 United States, Tennessee Sarah Canon Research Institute Nashville, Tennessee, United States, 37203 United Kingdom Sarah Cannon Research UK London, United Kingdom, W1G6AD Collapse << |
| NCT01938833 | - | - | Terminated(Closed by Sponsor) | - | - |
| NCT02203578 | Graft Versus Host Disease | Not Applicable | Terminated(slow accrual) | - | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT01938833 | HER2-negative Breast Cancer ... more >> Inflammatory Breast Cancer Male Breast Cancer Recurrent Breast Cancer Stage IV Breast Cancer Collapse << | Phase 1 Phase 2 | Terminated(Closed by Sponsor) | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02850016 | Human Immunodeficiency Virus (... more >>HIV) Collapse << | Phase 2 | Recruiting | December 2019 | United States, New York ... more >> The Rockefeller University Recruiting New York, New York, United States, 10065 Contact: Katelyn Bastert 800-782-2737 rucares@rockefeller.edu Principal Investigator: Marina Caskey, MD Denmark Aarhus University Hospital Recruiting Aarhus, Denmark Contact: Ole Sogaard, MD, PhD +45 7845 2842 olesoega@rm.dk Principal Investigator: Ole Sogaard, MD, PhD Germany University of Cologne Recruiting Cologne, Germany, 50937 Contact: Gisela Kremer, RN +41-221-478-3324 gisela.kremer@uk-koeln.de Principal Investigator: Gerd Kaetkenheuer, MD Collapse << |
| NCT01353664 | Lymphoma Canc... more >>er Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Sarasota, Florida, United States, 34232 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 United Kingdom Sarah Cannon Research UK London, United Kingdom, W1G6AD Collapse << |
| NCT02512497 | Cutaneous T-cell Lymphoma ... more >> T-Prolymphocytic Leukemia T-Large Granulocytic Leukemia T-Lymphoblastic Leukemia/Lymphoma Peripheral T-Cell Lymphoma Collapse << | Phase 1 | Recruiting | October 2019 | United States, Ohio ... more >> The Ohio State University Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Rachel Conley Rachel.Conley@osumc.edu Principal Investigator: Jonathan Brammer, MD Collapse << |
| NCT01979276 | - | - | Terminated(this study was term... more >>inated due to losing financial support, and enrollment challenges) Collapse << | - | - |
| NCT01353664 | - | - | Completed | - | - |
| NCT02203578 | - | - | Terminated(slow accrual) | - | - |
| NCT01324310 | Hematologic Malignancy ... more >> Malignant Lymphoma Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Sarasota, Florida, United States, 34232 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 United Kingdom Sarah Cannon Research UK London, United Kingdom, W1G6AD Collapse << |
| NCT01324310 | - | - | Completed | - | - |
| NCT01822886 | Peripheral T-cell Lymphoma | Phase 2 | Completed | - | Italy ... more >> A.O. SS. Antonio e Biagio e C. Arrigo Alessandria, Italy, 15121 Istituto di Ematologia ed Oncologia Medica A. Seragnoli Policlinico S. Orsola Bologna, Italy, 40138 Fondazione IRCCS Istituto Nazionale dei Tumori Milano, Italy, 20133 A.O. Universitaria Citta' Della Salute E Della Scienza Di Torino Torino, Italy, 10126 Collapse << |
| NCT01913119 | Histologically Proven Extranod... more >>al NKTcell Lymphoma Collapse << | Early Phase 1 | Completed | - | Korea, Republic of ... more >> National Cancer Center Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Korea Cancer Center Hospital Nowon-gu, Seoul, Korea, Republic of, 139-709 Severance Hospital Seodaemun-gu, Seoul, Korea, Republic of, 120-752 Asan Medical Center Songpa-gu, Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01537744 | Solid Tumors ... more >>Virally Mediated Cancers and Liposarcoma Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Sidney Kimmel Cancer Center @ Johns Hopkins Baltimore, Maryland, United States, 21231 Collapse << |
| NCT01638533 | Glioma Lympho... more >>ma Metastatic Malignant Solid Neoplasm Neuroendocrine Neoplasm Recurrent Adult Soft Tissue Sarcoma Recurrent Bladder Carcinoma Recurrent Breast Carcinoma Recurrent Chronic Lymphocytic Leukemia Recurrent Colorectal Carcinoma Recurrent Head and Neck Carcinoma Recurrent Lung Carcinoma Recurrent Malignant Solid Neoplasm Recurrent Melanoma Recurrent Pancreatic Carcinoma Recurrent Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Recurrent Prostate Carcinoma Recurrent Renal Cell Carcinoma Recurrent Thyroid Gland Carcinoma Refractory Chronic Lymphocytic Leukemia Refractory Mature T-Cell and NK-Cell Non-Hodgkin Lymphoma Refractory Primary Cutaneous T-Cell Non-Hodgkin Lymphoma Stage III Breast Cancer AJCC v7 Stage III Colorectal Cancer AJCC v7 Stage III Cutaneous Melanoma AJCC v7 Stage III Lung Cancer AJCC v7 Stage III Pancreatic Cancer AJCC v6 and v7 Stage III Prostate Cancer AJCC v7 Stage III Renal Cell Cancer AJCC v7 Stage III Soft Tissue Sarcoma AJCC v7 Stage IIIA Breast Cancer AJCC v7 Stage IIIA Colorectal Cancer AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Breast Cancer AJCC v7 Stage IIIB Colorectal Cancer AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Breast Cancer AJCC v7 Stage IIIC Colorectal Cancer AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Breast Cancer AJCC v6 and v7 Stage IV Colorectal Cancer AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Stage IV Lung Cancer AJCC v7 Stage IV Pancreatic Cancer AJCC v6 and v7 Stage IV Prostate Cancer AJCC v7 Stage IV Renal Cell Cancer AJCC v7 Stage IV Soft Tissue Sarcoma AJCC v7 Stage IVA Colorectal Cancer AJCC v7 Stage IVB Colorectal Cancer AJCC v7 Unresectable Solid Neoplasm Collapse << | Phase 1 | Recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Suspended Duarte, California, United States, 91010 University of California Davis Comprehensive Cancer Center Suspended Sacramento, California, United States, 95817 City of Hope South Pasadena Recruiting South Pasadena, California, United States, 91030 Contact: Stephen C. Koehler 626-396-2900 skoehler@cohmg.com Principal Investigator: Stephen C. Koehler United States, Georgia Emory University Hospital/Winship Cancer Institute Suspended Atlanta, Georgia, United States, 30322 United States, Illinois University of Chicago Comprehensive Cancer Center Active, not recruiting Chicago, Illinois, United States, 60637 United States, Maryland Johns Hopkins University/Sidney Kimmel Cancer Center Recruiting Baltimore, Maryland, United States, 21287 Contact: Roisin M. Connolly 410-955-8804 jhcccro@jhmi.edu Principal Investigator: Roisin M. Connolly National Cancer Institute Developmental Therapeutics Clinic Suspended Bethesda, Maryland, United States, 20892 National Institutes of Health Clinical Center Active, not recruiting Bethesda, Maryland, United States, 20892 United States, Michigan Wayne State University/Karmanos Cancer Institute Suspended Detroit, Michigan, United States, 48201 United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Paul Haluska 507-284-2511 haluska.paul@mayo.edu Principal Investigator: Paul Haluska United States, Ohio Case Western Reserve University Active, not recruiting Cleveland, Ohio, United States, 44106 United States, Pennsylvania Penn State Milton S Hershey Medical Center Active, not recruiting Hershey, Pennsylvania, United States, 17033-0850 Thomas Jefferson University Hospital Active, not recruiting Philadelphia, Pennsylvania, United States, 19107 United States, Wisconsin University of Wisconsin Hospital and Clinics Recruiting Madison, Wisconsin, United States, 53792 Contact: Roisin M. Connolly 410-614-9217 rconnol2@jhmi.edu Principal Investigator: Roisin M. Connolly Canada, Ontario University Health Network-Princess Margaret Hospital Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Lillian L. Siu 416-946-4501 clinical.trials@uhn.on.ca Principal Investigator: Lillian L. Siu Collapse << |
| NCT03278782 | Lymphoid Haematopoietic ... more >> Malignant Neoplasms Cutaneous T-Cell Lymphoma Refractory Cutaneous T-cell Lymphoma Collapse << | Phase 1 Phase 2 | Recruiting | November 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Clinical Research Operations spiyer@mdanderson.org Collapse << |
| NCT03547700 | Lymphoma, T-Cell, Peripheral | Phase 1 Phase 2 | Recruiting | July 2021 | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Ryan Wilcox, M.D. 734-615-1482 rwilcox@med.umich.edu Contact: Sumana Devata 734-615-7813 sdevata@med.umich.edu Collapse << |
| NCT02296398 | - | - | Completed | - | United States, Illinois ... more >> Northwestern University Dermatology Department Chicago, Illinois, United States, 60611 Collapse << |
| NCT03355768 | Lymphoma, T-Cell, Peripheral | Phase 3 | Not yet recruiting | January 2020 | United States, New York ... more >> Center for Lymphoid Malignancies at CUMC Not yet recruiting New York, New York, United States, 10019 Contact: Michelle Malanga, MPA 212-326-5731 mm4629@cumc.columbia.edu Collapse << |
| NCT01738594 | Recurrent Cutaneous T-cell Non... more >>-Hodgkin Lymphoma Recurrent Mycosis Fungoides/Sezary Syndrome Stage I Cutaneous T-cell Non-Hodgkin Lymphoma Stage IA Mycosis Fungoides/Sezary Syndrome Stage IB Mycosis Fungoides/Sezary Syndrome Stage II Cutaneous T-cell Non-Hodgkin Lymphoma Stage IIA Mycosis Fungoides/Sezary Syndrome Stage IIB Mycosis Fungoides/Sezary Syndrome Stage III Cutaneous T-cell Non-Hodgkin Lymphoma Stage IIIA Mycosis Fungoides/Sezary Syndrome Stage IIIB Mycosis Fungoides/Sezary Syndrome Stage IV Cutaneous T-cell Non-Hodgkin Lymphoma Stage IVA Mycosis Fungoides/Sezary Syndrome Stage IVB Mycosis Fungoides/Sezary Syndrome Collapse << | Phase 1 | Active, not recruiting | January 2020 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT03770000 | T Cell Lymphoma | Phase 1 Phase 2 | Not yet recruiting | November 2020 | United States, Texas ... more >> The University of Texas MD Anderson Cancer Center, Not yet recruiting Houston, Texas, United States, 77030 Contact: Swaminathan P Iyer, MD The University of Texas MD Anderson Cancer Center, Not yet recruiting Houston, Texas, United States, 77030 Contact: Auris Huen, MD Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| HDAC1 | IC50:36nM | |
| HDAC2 | IC50:47nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
