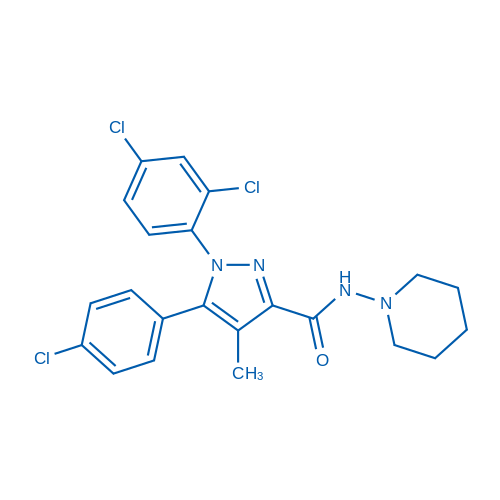
CAS No.: 168273-06-1
Rimonabant/利莫那班 Catalog No. CSN19427
Synonyms: SR141716;利莫那班
Rimonabant is a selective central cannabinoid (CB1) receptor inverse agonist with Ki of 1.8 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Cannabinoid Receptor 1
IC50:13.6nM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03204305 Cannabis Use Disorder ... more >> Cannabis Dependence, Continuous Collapse << Early Phase 1 Recruiting May 31, 2020 United States, Maryland ... more >> Johns Hopkins Bayview Medical Center Recruiting Baltimore, Maryland, United States, 21224 Contact: Alisha Eversole 410-550-5254 aeverso1@jhmi.edu Contact: Elise Weerts, Ph.D. 410-550-2781 eweerts@jhmi.edu Collapse << NCT00628290 Schizophrenia Phase 2 Completed - Germany ... more >> University of Cologne, Dept. of Psychiatry and Psychotherapy Cologne, NRW, Germany, 50924 Collapse << NCT00636766 Carotid Atherosclerosis ... more >> Stroke Type 2 Diabetes Metabolic Syndrome Collapse << Not Applicable Completed - Greece ... more >> General Hospital of Thessaloniki "Hippokratio" Thessaloniki, Greece Collapse << - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 168273-06-1 | 储存条件 |
|
|||||||||||||
| 分子式 | C22H21Cl3N4O | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 463.79 | 别名 | SR141716;利莫那班 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| CHO cells | - | Function assay | - | Ability to displace [3H]-SR- 141716A binding to human CB1 receptor expressed in CHO cell membranes, Ki=8.9 nM | 10465552 |
| CHO cells | - | Function assay | - | Antagonistic activity towards cannabinoid receptor 1 expressed as [3H]Arachidonic acid release in CHO cells, Kd=2.51 nM | 14736243 |
| CHOK1 cells | - | Function assay | - | Antagonist activity at human CB1 receptor expressed in CHOK1 cells by luciferase assay, IC50=0.12 μM | 18243711 |
| CHOK1 cells | - | Function assay | - | Antagonist activity at human CB1 receptor expressed in CHOK1 cells assessed as inhibition of CP-55940-induced response after 10 mins by GTPgamma[35S] binding assay, Ki=0.0016 μM | 19351113 |
| COS7 cells | - | Function assay | - | Displacement of [3H]CP-55940 from human CB1 receptor expressed in COS7 cells, Ki=0.0051 μM | 20015647 |
| HEK cells | - | Function assay | - | Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK cells, Ki=0.008 μM | 18579386 |
| HEK cells | - | Function assay | - | Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK cells, Ki=0.79 μM | 18579386 |
| HEK293 cells | - | Function assay | - | Immediate antagonist/Inverse agonist activity at hemagglutinin-tagged human CB1 receptor expressed in HEK293 cells assessed as reversal of 1 uM CP55,940-induced inhibition of 5 uM forskolin-induced cAMP accumulation by kinetic cAMP assay | 26203658 |
| HEK293 cells | - | Function assay | - | Displacement of [3H]CP-55940 from human recombinant CB2R expressed in HEK293 cells, IC50=1.6311 μM | 19530697 |
| N1E-115 cells | - | Function assay | - | Inverse agonist activity at CB1 receptor in mouse N1E-115 cells assessed as inhibition of WIN 55,212-2-induced ERK1/2 phosphorylation at 1 uM treated 5 mins prior to WIN 55,212-2 challenge by Western blot analysis | 23357307 |
| RD-HGA16 cells | - | Function assay | - | Antagonist activity at CB1 receptor (unknown origin) expressed in RD-HGA16 cells assessed as inhibition of CP55940-induced calcium mobilization by fluorometric imaging plate reader analysis | 24944734 |
| SF9 cells | - | Function assay | - | Inverse agonist at human CB1 receptor expressed in SF9 cells assessed as decrease in GTPgammaS level, IC50=1.35 nM | 18448340 |
| SF9 cells | - | Function assay | - | Antagonist activity at human CB2 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding, Ki=0.815 μM | 18448340 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03204305 | Cannabis Use Disorder ... more >> Cannabis Dependence, Continuous Collapse << | Early Phase 1 | Recruiting | May 31, 2020 | United States, Maryland ... more >> Johns Hopkins Bayview Medical Center Recruiting Baltimore, Maryland, United States, 21224 Contact: Alisha Eversole 410-550-5254 aeverso1@jhmi.edu Contact: Elise Weerts, Ph.D. 410-550-2781 eweerts@jhmi.edu Collapse << |
| NCT00628290 | Schizophrenia | Phase 2 | Completed | - | Germany ... more >> University of Cologne, Dept. of Psychiatry and Psychotherapy Cologne, NRW, Germany, 50924 Collapse << |
| NCT00636766 | Carotid Atherosclerosis ... more >> Stroke Type 2 Diabetes Metabolic Syndrome Collapse << | Not Applicable | Completed | - | Greece ... more >> General Hospital of Thessaloniki "Hippokratio" Thessaloniki, Greece Collapse << |
| NCT00734123 | Atherosclerosis ... more >> Cardiovascular Diseases Collapse << | Phase 4 | Unknown | May 2013 | Spain ... more >> Hospital Arnau de Vilanova Recruiting Lleida, Catalunya, Spain, 25260 Contact: Blai Coll, MD,PhD +34 619633249 bcoll@arnau.scs.es Sub-Investigator: Blai Coll, MD,PhD Sub-Investigator: Merce Borras, MD,PhD Collapse << |
| NCT00029848 | Obesity Diabe... more >>tes Mellitus, Non-Insulin-Dependent Obesity in Diabetes Collapse << | Phase 3 | Completed | - | - |
| NCT00029861 | Obesity Weigh... more >>t Loss Collapse << | Phase 3 | Completed | - | - |
| NCT00029835 | Obesity Dysli... more >>pidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00656487 | Cannabis Dependence ... more >> Cannabis Withdrawal Collapse << | Phase 2 | Completed | - | United States, California ... more >> The Scripps Research Institute La Jolla, California, United States, 92037 Collapse << |
| NCT00656487 | - | - | Completed | - | - |
| NCT01041170 | Cannabis Depe... more >>ndence Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> University of Maryland, Baltimore Baltimore, Maryland, United States, 21201-1595 National Institute on Drug Abuse, Biomedical Research Center (BRC) Baltimore, Maryland, United States, 21224 Collapse << |
| NCT00434096 | Obesity | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT00257257 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Argentina Sanofi-Aventis Administrative Office Buenos Aires, Argentina Chile Sanofi-Aventis Administrative Office Santiago, Chile Germany Sanofi-Aventis Administrative Office Berlin, Germany Hungary Sanofi-Aventis Administrative Office Budapest, Hungary Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Poland Sanofi-Aventis Administrative Office Warszawa, Poland Collapse << |
| NCT00288236 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Argentina Sanofi-Aventis Buenos Aires, Argentina Australia Sanofi-Aventis Lane Cove, Australia Canada Sanofi-Aventis Laval, Canada Chile Sanofi-Aventis Providencia, Chile France Sanofi-Aventis Paris, France Germany Sanofi-Aventis Berlin, Germany Italy Sanofi-Aventis Milano, Italy Netherlands Sanofi-Aventis Gouda, Netherlands Russian Federation Sanofi-Aventis Moscow, Russian Federation South Africa Sanofi-Aventis Midrand, South Africa United Kingdom Sanofi-Aventis Guildford, United Kingdom Collapse << |
| NCT00124332 | Coronary Atherosclerosis | Phase 3 | Completed | - | United States, Ohio ... more >> The Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Australia sanofi-aventis, Australia Macquarie Park, Australia Belgium sanofi-aventis, Belgium Diegem, Belgium Canada Sanofi-Aventis Administrative Office Laval, Canada France sanofi-aventis, France Paris, France Italy sanofi-aventis Italy Milano, Italy Netherlands sanofi-aventis, Netherlands Gouda, Netherlands Poland sanofi-aventis Poland Warszawa, Poland Spain sanofi-aventis Spain Barcelona, Spain Collapse << |
| NCT00325546 | Obesity | Phase 3 | Completed | - | China ... more >> Sanofi-Aventis Shangai, China, 200040 Korea, Republic of Sanofi-Aventis Seoul, Korea, Republic of, 135-755 Taiwan Sanofi-Aventis Taipei, Taiwan, 105 Collapse << |
| NCT00481923 | Obesity | Phase 3 | Completed | - | United Kingdom ... more >> Sanofi-Aventis Administrative Office Guilford, United Kingdom Collapse << |
| NCT00750347 | Pain | Phase 1 | Completed | - | France ... more >> CIC-CPC Clermont-Ferrand, France, 63000 CHU Clermont-Ferrand Clermont-Ferrand, France, 63003 Collapse << |
| NCT00239967 | Dyslipidemia ... more >>Obesity Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Australia Sanofi-aventis Administrative Office Macquarie Park, Australia Brazil Sanofi-aventis Administrative Office Sao Paulo, Brazil Canada Sanofi-aventis Administrative Office Laval, Canada Denmark Sanofi-aventis Administrative Office Horsholm, Denmark Hong Kong Sanofi-aventis Administrative Office Causeway Bay, Hong Kong Italy Sanofi-aventis Administrative Office Milano, Italy Korea, Republic of Sanofi-aventis Administrative Office Seoul, Korea, Republic of Malaysia Sanofi-aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-aventis Administrative Office Mexico, Mexico Singapore Sanofi-aventis Administrative Office Singapore, Singapore South Africa Sanofi-aventis Administrative Office Midrand, South Africa Sweden Sanofi-aventis Administrative Office Bromma, Sweden Taiwan Sanofi-aventis Administrative Office Taipei, Taiwan Collapse << |
| NCT00458718 | Smoking Cessation | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00408148 | Obesity | Phase 3 | Terminated(Company decision ha... more >>s been taken in light of recent demands by certain national health authorities) Collapse << | - | Australia ... more >> Sanofi-Aventis Administrative Office North Ryde, Australia Finland Sanofi-Aventis Administrative Office Helsinki, Finland France Sanofi-Aventis Administrative Office Paris, France United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00386061 | Obesity | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Belgium Sanofi-Aventis Brussels, Belgium Finland Sanofi-Aventis Helsinki, Finland France Sanofi-Aventis Paris, France Germany Sanofi-Aventis Berlin, Germany Netherlands Sanofi-Aventis Gouda, Netherlands Sweden Sanofi-Aventis Bromma, Sweden Collapse << |
| NCT00481975 | Obesity Eatin... more >>g Disorders Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Finland Sanofi-Aventis Administrative Office Helsinki, Finland France Sanofi-Aventis Administrative Office Paris, France Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Portugal Sanofi-Aventis Administrative Office Porto Salvo, Portugal Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Switzerland Sanofi-Aventis Administrative Office Geneva, Switzerland Collapse << |
| NCT00459173 | Maintenance of Smoking Cessati... more >>on Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Australia, New South Wales Sanofi-Aventis Cove, New South Wales, Australia Canada, Quebec Sanofi-Aventis Laval, Quebec, Canada Collapse << |
| NCT00459004 | Obesity | Phase 2 | Completed | - | Japan ... more >> Sanofi-Aventis Tokyo, Japan Collapse << |
| NCT00412698 | Obesity Dysli... more >>pidemias Collapse << | Phase 3 | Terminated(Company decision ha... more >>s been taken in light of recent demands by certain national health authorities) Collapse << | - | Czech Republic ... more >> Sanofi-Aventis Administrative Office Prague, Czech Republic Finland Sanofi-Aventis Administrative Office Helsinki, Finland Germany Sanofi-Aventis Administrative Office Berlin, Germany Greece Sanofi-Aventis Administrative Office Athens, Greece Hungary Sanofi-Aventis Administrative Office Budapest, Hungary Ireland Sanofi-Aventis Administrative Office Dublin, Ireland Italy Sanofi-Aventis Administrative Office Milan, Italy Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Norway Sanofi-Aventis Administrative Office Lysaker, Norway Portugal Sanofi-Aventis Administrative Office Porto-Salvo, Portugal Slovakia Sanofi-Aventis Administrative Office Bratislava, Slovakia Sweden Sanofi-Aventis Administrative Office Stockholm, Sweden Switzerland Sanofi-Aventis Administrative Office Meyrin, Switzerland Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00458081 | Obesity Micro... more >>albuminuria Diabetes Mellitus, Type 2 Dyslipidemia Collapse << | Phase 3 | Terminated(Company decision ha... more >>s been taken in light of recent demands by certain national health authorities) Collapse << | - | Spain ... more >> Sanofi-Aventis Administrative Office Barcelona, Spain Collapse << |
| NCT00678483 | Obesity Weigh... more >>t Loss Collapse << | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | Croatia ... more >> Sanofi-Aventis Administrative Office Zagreb, Croatia Finland Sanofi-Aventis Administrative Office Helsinki, Finland Hungary Sanofi- Aventis Administrative Office Budapest, Hungary Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Collapse << |
| NCT00405808 | Obesity | Phase 3 | Terminated(EMEA recommendation... more >> to suspend Acomplia marketing authorisation) Collapse << | - | - |
| NCT00358228 | Smoking Cessation | Phase 3 | Completed | - | United States, Pennsylvania ... more >> Sanofi-Aventis Administrative Office Malvern, Pennsylvania, United States, 19355 Collapse << |
| NCT00547118 | - | - | Terminated(Withdrawn due to me... more >>dication withdrawal from the EMEA) Collapse << | - | - |
| NCT00464165 | Smoking Cessation | Phase 3 | Completed | - | Belgium ... more >> Sanofi-Aventis Administrative Office Diegem, Belgium Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark France Sanofi-Aventis Administrative Office Paris, France Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Switzerland Sanofi-Aventis Administrative Office Geneva, Switzerland United Kingdom Sanofi-Aventis Administrative Office Guildford, Surrey, United Kingdom Collapse << |
| NCT00546325 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Australia ... more >> Sanofi-Aventis Administrative Office North Ryde, Australia Collapse << |
| NCT00075205 | Healthy Alcoh... more >>ol Drinking Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00449605 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Chile Sanofi-Aventis Administrative Office Santiago, Chile Sanofi-aventis adminsitrative office Santiago, Chile Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark Finland Sanofi-Aventis Administrative Office Helsinki, Finland Hungary Sanofi-Aventis Administrative Office Budapest, Hungary India Sanofi-Aventis Administrative Office Mumbai, India Italy Sanofi-Aventis Administrative Office Milan, Italy Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Puerto Rico Sanofi-Aventis Administrative Office Puerto Rico, Puerto Rico Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Collapse << |
| NCT00547118 | Schizophrenia ... more >> Schizoaffective Disorder Obesity Hypertension Smoking Collapse << | Phase 2 | Terminated(Withdrawn due to me... more >>dication withdrawal from the EMEA) Collapse << | - | United States, Maryland ... more >> Baltimore VA Medical Center Baltimore, Maryland, United States, 21201 Keypoint Health System Baltimore, Maryland, United States, 21201 Sheppard Pratt Health System Baltimore, Maryland, United States, 21201 Maryland Psychiatric Research Center (MPRC) Outpatient Research Program (ORP); the MPRC Treatment Research Program (TRP) Catonsville, Maryland, United States, 21228 Collapse << |
| NCT00464256 | Smoking Cessation | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00478595 | Obesity Diabe... more >>tes Mellitus Type 2 Collapse << | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT00478972 | Obesity Diabe... more >>tes Mellitus Type 2 Collapse << | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT00525681 | Renal Transplantation | Phase 4 | Completed | - | Norway ... more >> Rikshospitalet, Section of Nephrology Oslo, Norway, 0027 Collapse << |
| NCT00603109 | Prader-willi Syndrome | Phase 3 | Unknown | August 2008 | United States, New York ... more >> New York Presbyterian Hospital-Weill Cornell Medical College Recruiting New york, New York, United States, 10065 Contact: Roja Motaghedi, MD 212-746-3462 rom9027@med.cornell.edu Contact: Elizabeth G Diaz, CPNP 212-746-3462 egl2002@med.cornell.edu Principal Investigator: Roja Motaghedi, MD Sub-Investigator: Elizabeth G Diaz, CPNP Collapse << |
| NCT00228176 | Carotid Artery Plaque ... more >> Arteriosclerosis Obesity Metabolic Syndrome X Collapse << | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada France Sanofi-Aventis Administrative Office Paris, France Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Spain Sanofi-Aventis Administrative Office Barcelona, Spain United Kingdom Sanofi-Aventis Administrative Office Guildford Surrey, United Kingdom Collapse << |
| NCT00754689 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(Company decision tak... more >>en in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00263042 | Cardiovascular Disease | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | - |
| NCT00325650 | Prediabetic State | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00299325 | Metabolic Syndrome | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada Denmark Sanofi-Aventis Administrative Office Hoersholm, Denmark Finland Sanofi-Aventis Administrative Office Helsinki, Finland France Sanofi-Aventis Administrative Office Paris, France Italy Sanofi-Aventis Administrative Office Milan, Italy Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Stockholm, Sweden United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00577148 | Fatty Liver | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | - |
| NCT00584389 | Obesity | Phase 4 | Terminated(Suspension of licen... more >>ce for rimonabant by European Medicines Agency) Collapse << | May 2010 | United Kingdom ... more >> Royal Surrey County Hospital Guildford, Surrey, United Kingdom, GU2 7XX Collapse << |
| NCT00690456 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Indonesia Sanofi-Aventis Administrative Office Jakarta, Indonesia Lithuania Sanofi-Aventis Administrative Office Vilnius, Lithuania Malaysia Sanofi-Aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Slovakia Sanofi-Aventis Administrative Office Brastislava, Slovakia Taiwan Sanofi-Aventis Administrative Office Taipei, Taiwan Thailand Sanofi-Aventis Administrative Office Bangkok, Thailand Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT00576667 | Fatty Liver | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | - |
| 靶点 | Description | IC50 |
|---|---|---|
| Cannabinoid Receptor 1 | IC50:13.6nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
