| NCT03176472 | Diabetic Neuropathic Pain | Phase 2 | Not yet recruiting | January 2021 | - |
| NCT02088398 | Healthy | Phase 1 | Completed | - | United States, Wisconsin
... more >>
Covance Clinical Research Unit Incorporated
Madison, Wisconsin, United States, 53704 Collapse << |
| NCT02189343 | Multiple Myeloma | Phase 1 | Active, not recruiting | January 30, 2019 | United States, Texas
... more >>
UT Southwestern Medical Center Simmons Comprehensive Cancer Center
Dallas, Texas, United States, 75390-8852
CTRC at The UT Health Science Center at San Antonio
San Antonio, Texas, United States, 78229
United States, Utah
University of Utah Huntsman Cancer Institute
Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02787369 | Recurrent Chronic Lymphoid Leu... more >>kemia Collapse << | Phase 1 | Active, not recruiting | April 2022 | United States, Massachusetts
... more >>
Dana Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01583283 | Multiple Myeloma | Phase 1
Phase 2 | Active, not recruiting | January 30, 2019 | United States, Massachusetts
... more >>
Dana Farber Cancer Institute
Boston, Massachusetts, United States, 02215
Massachusetts General Hospital
Boston, Massachusetts, United States, 2114
United States, North Carolina
University of North Carolina
Chapel Hill, North Carolina, United States, 27599
United States, Tennessee
Sarah Cannon Research Institute Drug Development Unit
Nashville, Tennessee, United States, 37203
United States, Washington
Fred Hutchinson Cancer Research Center
Seattle, Washington, United States, 98109 Collapse << |
| NCT01323751 | Multiple Myeloma | Phase 1
Phase 2 | Completed | - | United States, Georgia
... more >>
Winship Cancer Institute, Emory University
Atlanta, Georgia, United States, 30322
United States, Massachusetts
Massachusetts General Hospital
Boston, Massachusetts, United States, 02115
United States, New York
Mt. Sinai Medical Center
New York, New York, United States, 10029
United States, Pennsylvania
University of Pennsylvania
Philadelphia, Pennsylvania, United States, 19104
United States, Texas
MD Anderson Cancer Center
Houston, Texas, United States, 77030
United States, Wisconsin
Medical College of Wisconsin - Clinical Cancer Center
Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT01997840 | Multiple Myeloma | Phase 1
Phase 2 | Active, not recruiting | January 27, 2020 | United States, Georgia
... more >>
Winship Cancer Institute of Emory University
Atlanta, Georgia, United States, 30322
United States, Indiana
Horizon Oncology Center
Lafayette, Indiana, United States, 47905
United States, Massachusetts
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02215
United States, Michigan
Univesity of Michigan Comprehensive Cancer Center
Ann Arbor, Michigan, United States, 48109-0922
United States, Missouri
Washington University School of Medicine
Saint Louis, Missouri, United States, 63110
United States, New York
New York Medical College
Hawthorne, New York, United States, 10532
Mt. Sinai Medical Center Division of Hematology/Oncology
New York, New York, United States, 10029
United States, Ohio
Cleveland Clinic
Cleveland, Ohio, United States, 44195
Ohio State University Arthur G. James Cancer Hospital
Columbus, Ohio, United States, 43210-1240
United States, Pennsylvania
University of Pittsburgh Medical Center - Cancer Pavilion
Pittsburgh, Pennsylvania, United States, 15232
United States, Texas
University of Texas Health Science Center at San Antonio
San Antonio, Texas, United States, 78229
United States, Washington
Fred Hutchinson Cancer Research Center
Seattle, Washington, United States, 98109-1023
United States, Wisconsin
Medical College of Wisconsin
Milwaukee, Wisconsin, United States, 53226
Canada, Alberta
Tom Baker Cancer Center
Calgary, Alberta, Canada, T2N 4N2
Cross Cancer Institute
Edmonton, Alberta, Canada, T6G 1Z2
Canada, Ontario
Princess Margaret Cancer Centre
Toronto, Ontario, Canada, M5G 2M9
Greece
Alexandra Hospital, University of Athens
Athens, Greece, 11528
Italy
Azienda Ospedaliera Citta della Salute e della Scienza di Torino
Torino, Italy, O1012 Collapse << |
| NCT02856568 | Non-Resectable Cholangiocarcin... more >>oma
Recurrent Cholangiocarcinoma
Stage III Extrahepatic Bile Duct Cancer
Stage III Intrahepatic Cholangiocarcinoma
Stage IIIA Hilar Cholangiocarcinoma
Stage IIIB Hilar Cholangiocarcinoma
Stage IVA Extrahepatic Bile Duct Cancer
Stage IVA Hilar Cholangiocarcinoma
Stage IVA Intrahepatic Cholangiocarcinoma
Stage IVB Extrahepatic Bile Duct Cancer
Stage IVB Hilar Cholangiocarcinoma
Stage IVB Intrahepatic Cholangiocarcinoma
Unresectable Extrahepatic Bile Duct Carcinoma Collapse << | Phase 1 | Withdrawn(Site dropped study) | October 2021 | United States, Arizona
... more >>
Mayo Clinic in Arizona
Scottsdale, Arizona, United States, 85259
United States, Florida
Mayo Clinic in Florida
Jacksonville, Florida, United States, 32224-9980
United States, Minnesota
Mayo Clinic
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT02632071 | Metastatic Breast Cancer
... more >> Breast Carcinoma Collapse << | Phase 1 | Recruiting | January 2019 | United States, New York
... more >>
Columbia University Medical Center
Recruiting
New York, New York, United States, 10032
Contact: Kevin Kalinsky, MD, MS 212-305-1945 KK2693@cumc.columbia.edu
Principal Investigator: Kevin Kalinsky, MD, MS Collapse << |
| NCT02091063 | Lymphoma
Lymp... more >>hoid Malignancies Collapse << | Phase 1
Phase 2 | Recruiting | December 10, 2019 | United States, Florida
... more >>
Moffit Cancer Center
Recruiting
Tampa, Florida, United States, 33612
Contact: Amanda Cameron 813-745-3304 Amanda.Cameron@moffitt.org
Principal Investigator: Bijal Shah, MD
United States, New York
Columbia University Medical Center
Recruiting
New York, New York, United States, 10019
Contact: Celeste Rojas 212-326-5720 cr2393@cumc.columbia.edu
Contact: Renee Lichtenstein 212-326-5720 rl2619@columbia.edu
Principal Investigator: Jennifer E Amengual, MD Collapse << |
| NCT02661815 | Ovarian Cancer
... more >> Fallopian Tube Cancer
Primary Peritoneal Carcinoma Collapse << | Phase 1 | Active, not recruiting | January 2022 | United States, Massachusetts
... more >>
Massacusetts General Hospital
Boston, Massachusetts, United States, 02114
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
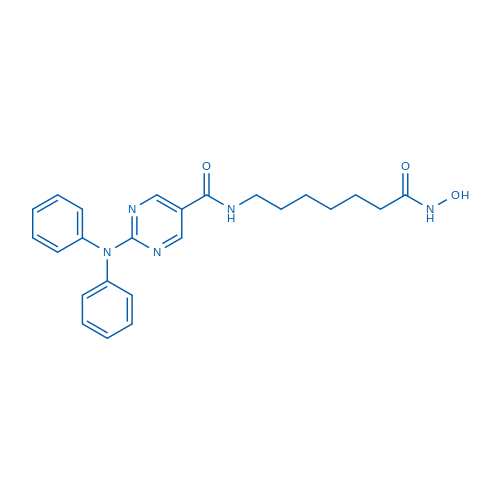

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
