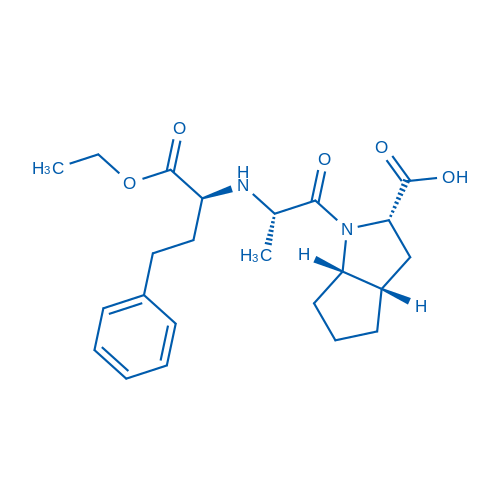
CAS No.: 87333-19-5
Ramipril/雷米普利 Catalog No. CSN13066
Synonyms: HOE-498;Altace;Carasel
Ramipril is an angiotensin-converting enzyme (ACE) inhibitor with IC50 of 5 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00732069 End Stage Renal Failure on Dia... more >>lysis Complication of Hemodialysis Collapse << Phase 2 Completed - United States, Tennessee ... more >> Vanderbilt University Medical Center Nashville, Tennessee, United States, 37323 Collapse << NCT00732069 - - Completed - - NCT01225445 Biopsy-confirmed IgA Nephropat... more >>hy Proteinuria Less Than 0.5 g Per Day Normal Blood Pressure Serum Creatinine Below 120 Umol/l Collapse << Phase 3 Completed - Hong Kong ... more >> Department of Medicine & Therapeutics, Prince of Wales Hospital Shatin, Hong Kong Collapse << - 更多
- 参考文献
- [1] Kohlstedt K, Brandes RP, et al. Angiotensin-converting enzyme is involved in outside-in signaling in endothelial cells. Circ Res. 2004 Jan 9;94(1):60-7. Epub 2003 Nov 13.
- [2] Stevens BR, Phillips MI, et al. Ramipril inhibition of rabbit (Oryctolagus cuniculus) small intestinal brush border membrane angiotensin converting enzyme. Comp Biochem Physiol C. 1988;91(2):493-7.
- [3] Wei Q, Liu H, Liu M, Yang C, Yang J, Liu Z, Yang P. Ramipril attenuates left ventricular remodeling by regulating the expression of activin A-follistatin in a rat model of heart failure. Sci Rep. 2016 Sep 19;6:33677. doi: 10.1038/srep33677. PMID: 27642098; PMCID: PMC5027547.
- [4] Raasch W, Jöhren O, Schwartz S, Gieselberg A, Dominiak P. Combined blockade of AT1-receptors and ACE synergistically potentiates antihypertensive effects in SHR. J Hypertens. 2004 Mar;22(3):611-8. doi: 10.1097/00004872-200403000-00025. PMID: 15076168.
- [5] Kohlstedt K, Brandes RP, Müller-Esterl W, Busse R, Fleming I. Angiotensin-converting enzyme is involved in outside-in signaling in endothelial cells. Circ Res. 2004 Jan 9;94(1):60-7. doi: 10.1161/01.RES.0000107195.13573.E4. Epub 2003 Nov 13. PMID: 14615289.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 87333-19-5 | 储存条件 |
|
|||||||
| 分子式 | C23H32N2O5 | 运输 | 蓝冰 | |||||||
| 分子量 | 416.51 | 别名 | HOE-498;Altace;Carasel;Zabien; Triatec ; Vesdil;雷米普利 | |||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00732069 | End Stage Renal Failure on Dia... more >>lysis Complication of Hemodialysis Collapse << | Phase 2 | Completed | - | United States, Tennessee ... more >> Vanderbilt University Medical Center Nashville, Tennessee, United States, 37323 Collapse << |
| NCT00732069 | - | - | Completed | - | - |
| NCT01225445 | Biopsy-confirmed IgA Nephropat... more >>hy Proteinuria Less Than 0.5 g Per Day Normal Blood Pressure Serum Creatinine Below 120 Umol/l Collapse << | Phase 3 | Completed | - | Hong Kong ... more >> Department of Medicine & Therapeutics, Prince of Wales Hospital Shatin, Hong Kong Collapse << |
| NCT00674596 | Diabetes Prot... more >>einuria Renin Angiotensin System Collapse << | Phase 4 | Completed | - | Turkey ... more >> Gulhane School of Medicine Ankara, Turkey, 06018 Collapse << |
| NCT01504828 | Left Ventricular Function Syst... more >>olic Dysfunction Left Ventricular Function Diastolic Dysfunction Ageing Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Campus for Ageing and Vitality Newcastle upon Tyne, United Kingdom, NE4 6BE Collapse << |
| NCT00005928 | Atherosclerosis ... more >> Coronary Disease Vasculitis Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Heart, Lung and Blood Institute (NHLBI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03593317 | Arrhythmogenic Right Ventricul... more >>ar Dysplasia Collapse << | Phase 3 | Not yet recruiting | January 2023 | France ... more >> Hôpital Cardiologique Louis Pradel Not yet recruiting Bron, France Contact: Philippe Chevalier Collapse << |
| NCT00437463 | Glomerulonephritis | Phase 3 | Completed | - | Hong Kong ... more >> Sanofi-Aventis Hong Kong, Hong Kong Collapse << |
| NCT01004705 | Elevated LDL Cholesterol | Phase 2 | Terminated | - | United States, New York ... more >> Mount Sinai Medical Center New York, New York, United States, NY 10029-6574 Collapse << |
| NCT02525081 | Microvascular Angina | Phase 4 | Completed | - | Denmark ... more >> Bispebjerg Hospital Copenhagen NV, Denmark, 2400 Collapse << |
| NCT00367939 | Hypertension | Phase 3 | Completed | - | Germany ... more >> Investigative Centers, Germany Collapse << |
| NCT00219089 | Hypertension, Diabetes Mellitu... more >>s Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers, Germany Collapse << |
| NCT00268619 | Myocardial Ischemia | Phase 3 | Completed | - | - |
| NCT03201185 | Transcatheter Aortic Valve Rep... more >>lacemen Angiotensin-Converting Enzyme Inhibitors Collapse << | Phase 4 | Recruiting | February 2023 | Spain ... more >> Hospital Clínico Universitario de Valladolid Recruiting Valladolid, Spain, 47005 Contact: Luis Varela-Falcón, Dr 983420000 ext 86571 lhvf86@hotmail.com Collapse << |
| NCT00366119 | Essential Hypertension | Phase 4 | Unknown | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, 28 Yeongeon-dong, Jongno-gu, Korea, Republic of, 110-744 Collapse << |
| NCT01004705 | - | - | Terminated | - | - |
| NCT01302899 | Non-diabetic Nephropathy | Phase 2 | Terminated(In consequence of t... more >>ermination of ALTITUDE. A number of studies were continued in consultation with the Altitude Data Monitoring Committee.) Collapse << | - | Netherlands ... more >> Novartis Investigative Site Groningen, Netherlands Novartis Investigative Site Leeuwarden, Netherlands Collapse << |
| NCT02924727 | Acute Myocardial Infarction | Phase 3 | Recruiting | April 27, 2020 | - |
| NCT00219063 | Hypertension | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States Collapse << |
| NCT00681226 | Peripheral Arterial Disease | Phase 4 | Completed | - | Australia, Victoria ... more >> Baker Heart Research Institute Melbourne, Victoria, Australia, 8008 Collapse << |
| NCT00240422 | Diabetes Mellitus, Type 2 ... more >> Hypertension Collapse << | Phase 4 | Completed | - | France ... more >> Boehringer Ingelheim Investigational Site Lyon, France Boehringer Ingelheim Investigational Site Montpellier, France Germany Friedrich-Alexander-Universität Erlangen, Germany, 91054 Boehringer Ingelheim Investigational Site Nürnberg, Germany, 90402 Universität Erlangen-Nürnberg Nürnberg, Germany, 90471 Spain Edificio de Medicina Comunitaria Madrid, Spain, 28041 Collapse << |
| NCT00841880 | Hypertension | Phase 4 | Completed | - | Taiwan ... more >> Sanofi-Aventis Administrative Office Taipei, Taiwan Collapse << |
| NCT00274599 | Hypertension | Phase 4 | Completed | - | - |
| NCT01302899 | - | - | Terminated(In consequence of t... more >>ermination of ALTITUDE. A number of studies were continued in consultation with the Altitude Data Monitoring Committee.) Collapse << | - | - |
| NCT00281593 | Hypertension | Phase 3 | Completed | - | - |
| NCT01078363 | Cardiac Allograft Vasculopathy | Not Applicable | Completed | - | United States, California ... more >> VA Palo Alto Health Care System Palo Alto, California, United States, 94304 Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT01922141 | Hypertension | Phase 4 | Withdrawn | June 2018 | - |
| NCT01078363 | - | - | Completed | - | - |
| NCT01362218 | Elevated LDL Cholesterol | Phase 2 | Terminated(Per sponsor's decis... more >>ion) Collapse << | - | Spain ... more >> Hospital Clinic Barcelona, Spain Collapse << |
| NCT00274612 | Hypertension | Phase 4 | Completed | - | - |
| NCT00770497 | Inflammation ... more >>Hypertension Collapse << | Phase 2 | Completed | - | Germany ... more >> Deggingen, Baden-Württemberg, Germany Rottweil, Baden-Württemberg, Germany Spaichingen, Baden-Württemberg, Germany Weilersbach, Bayern, Germany Hannover, Niedersachsen, Germany Essen, Nordrhein-Westfalen, Germany Köln, Nordrhein-Westfalen, Germany Werne, Nordrhein-Westfalen, Germany Mainz, Rheinland-Pfalz, Germany Schauenburg, Rheinland-Pfalz, Germany Dresden, Sachsen, Germany Blankenhain, Thüringen, Germany Berlin, Germany Collapse << |
| NCT00095654 | Impaired Glucose Tolerance ... more >> Cardiovascular Disease Glucose Metabolism Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT00355589 | Hypertension ... more >>Blood Pressure, High Collapse << | Phase 3 | Completed | - | - |
| NCT00153101 | Cardiovascular Diseases | Phase 4 | Completed | - | - |
| NCT00140647 | Atherosclerosis ... more >> Impaired Glucose Tolerance Isolated Impaired Fasting Glucose Cardiovascular Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00356395 | Hypertension | Phase 4 | Completed | - | - |
| NCT00208221 | Hypertension ... more >>Type 2 Diabetes Albuminuria Collapse << | Phase 3 | Terminated(Not enough recruitm... more >>ent) Collapse << | - | Canada, Quebec ... more >> Institut de Recherches Cliniques de Montreal Montreal, Quebec, Canada, J4X 1J3 Collapse << |
| NCT00980785 | Alzheimer's Disease ... more >> Hypertension Collapse << | Phase 4 | Completed | - | United States, Wisconsin ... more >> Wisconisn Alzheimer's Disease Research Center Madison, Wisconsin, United States, 53705 Collapse << |
| NCT00044265 | Hypertension | Phase 4 | Completed | - | - |
| NCT00325806 | Hypertension | Phase 4 | Completed | - | - |
| NCT00168467 | Peripheral Arterial Disease | Phase 4 | Completed | - | Australia, Victoria ... more >> Alfred Hospital Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01703234 | Proteinuria D... more >>iabetic Nephropathy Chronic Kidney Disease Collapse << | Phase 4 | Completed | - | Turkey ... more >> Gulhane School of Medicine Ankara, Turkey Collapse << |
| NCT01485978 | Renal Insufficiency, Chronic | Phase 3 | Active, not recruiting | August 2019 | Germany ... more >> University Medical Center Goettingen Goettingen, Germany, 37075 Collapse << |
| NCT00095290 | Albuminuria | Phase 4 | Completed | - | - |
| NCT00702936 | Acute Coronary Syndrome ... more >> Myocardial Infarction Coronary Disease Collapse << | Phase 4 | Unknown | November 2008 | Italy ... more >> Catholic University of the Sacred Heart Recruiting Rome, Italy, 00168 Contact: Italo Porto, MD, PhD i.porto@doctors.org.uk Contact: Luca Di Vito, MD divitoluca@yahoo.it Principal Investigator: Italo Porto, MD, PhD Sub-Investigator: Luca Di Vito, MD Collapse << |
| NCT00702260 | Hypertension | Not Applicable | Completed | - | United States, Texas ... more >> CEDRA Clinical Research, LLC Austin, Texas, United States, 78759 Collapse << |
| NCT01005290 | - | - | Terminated | - | - |
| NCT00368277 | Hypertension | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT00368277 | - | - | Completed | - | - |
| NCT01829880 | - | - | Unknown | August 2013 | Slovenia ... more >> University Clinic of Respiratory and Allergic Diseases Golnik Recruiting Golnik, Slovenia, 4204 Contact: Mitja Lainscak, MD, PhD (0) 4 256 9483 ext +386 mitja.lainscak@guest.arnes.si Contact: Katja Trobec, MPharm (0) 4 256 9360 ext +386 katja.trobec@klinika-golnik.si Principal Investigator: Mitja Lainscak, MD, PhD Sub-Investigator: Katja Trobec, MPharm Collapse << |
| NCT00923156 | Heart Failure | Phase 2 | Completed | - | Germany ... more >> Novartis Investigator Site Bad Krozingen, Germany, 79189 Novartis Investigator Site Berlin, Germany, 12207 Novartis Investigator Site Berlin, Germany, 13353 Novartis Investigator Site Göttingen, Germany, 37057 Novartis Investigator Site Jena, Germany, 07747 Novartis Investigator Site München, Germany, 80336 Poland Novartis Investigator Site Krakow, Poland, 31-501 Novartis Investigator Site Lublin, Poland, 20-090 Novartis Investigator Site Poznan, Poland, 61-848 Novartis Investigator Site Warszawa, Poland, 02-507 Novartis Investigator Site Wroclaw, Poland, 50-981 Russian Federation Novartis Investigator Site Moscow, Russian Federation, 117292 Novartis Investigator Site Moscow, Russian Federation, 119620 Novartis Investigator Site Moscow, Russian Federation, 121309 Novartis Investigator Site Moscow, Russian Federation, 121552 Novartis Investigative Site Moscow, Russian Federation Collapse << |
| NCT00702091 | Hypertension | Not Applicable | Completed | - | United States, Texas ... more >> CEDRA Clinical Research, LLC Austin, Texas, United States, 78759 Collapse << |
| NCT00923156 | - | - | Completed | - | - |
| NCT01284621 | - | - | Completed | - | - |
| NCT01038895 | Hypertension ... more >>Type 2 Diabetes Collapse << | Phase 4 | Unknown | June 2011 | Italy ... more >> University of Pavia Recruiting Pavia, PV, Italy, 27100 Contact: Amedeo Mugellini, MD +39 0382 526217 amedeo.mugellini@unipv.it Principal Investigator: Amedeo Mugellini, MD Collapse << |
| NCT00153101 | - | - | Completed | - | - |
| NCT01284621 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1245.45.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT00502242 | Kidney Transplant | Phase 4 | Completed | - | - |
| NCT01005290 | Hypertension | Phase 2 | Terminated | - | United States, New York ... more >> Mount Sinai Medical Center New York, New York, United States, NY 10029-6574 Collapse << |
| NCT00507845 | Hypertension | Phase 4 | Completed | - | Argentina ... more >> Sanofi-aventis administrative office Buenos Aires, Argentina Collapse << |
| NCT00946621 | Hypertension | Phase 1 | Completed | - | - |
| NCT00760214 | - | - | Completed | - | - |
| NCT00946465 | Hypertension | Phase 1 | Completed | - | - |
| NCT00273533 | Arthritis, Rheumatoid | Phase 2 Phase 3 | Completed | - | - |
| NCT00502242 | - | - | Completed | - | - |
| NCT00829530 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, H2X 2H9 Anapharm Inc. Sainte-Foy, Quebec, Canada, G1V 2K8 Collapse << |
| NCT00760214 | Hypertension | Phase 3 | Completed | - | - |
| NCT00829530 | - | - | Completed | - | - |
| NCT00829452 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, H2X 2H9 Anapharm Inc. Sainte-Foy, Quebec, Canada, G1V 2K8 Collapse << |
| NCT00828321 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, H2X 2H9 Anapharm Inc. Sainte-Foy, Quebec, Canada, G1V 2K8 Collapse << |
| NCT00828321 | - | - | Completed | - | - |
| NCT00829452 | - | - | Completed | - | - |
| NCT00517322 | Left Atrial Volume ... more >> Hypertensive Heart Disease Antihypertensive Drugs Diastolic Function Renin Angiotensin System Collapse << | Phase 4 | Unknown | August 2010 | Italy ... more >> University of Insubria-Department of Clinical Medicine Recruiting Varese, Italy, 21100 Contact: anna maria grandi, MD +39+332 278594 amgrandi@libero.it Contact: anna maria grandi, MD +39 + 332 278594 amgrandi@libero.it Sub-Investigator: andrea maria maresca, MD Sub-Investigator: andrea bertolini, MD Sub-Investigator: monica gianni, MD Sub-Investigator: eleonora nicolini, MD Collapse << |
| NCT00480805 | Type 2 Diabetes Mellitus ... more >> Hypertension Collapse << | Phase 3 | Completed | - | - |
| NCT00819845 | Duchenne Muscular Dystrophy ... more >> Becker Muscular Dystrophy Collapse << | Phase 4 | Unknown | December 2016 | Italy ... more >> Unione Italiana lotta Distrofia Muscolare Recruiting Rome, Italy, 00167 Contact: Vincenzo Giglio, MD, PhD 39-6-6604881 giglio.echo@libero.it Contact: Fortunato Mangiola, MD 39-6-6604881 fortunato.mangiola@uildmlazio.org Principal Investigator: Vincenzo Giglio, MD, PhD Collapse << |
| NCT02214992 | Healthy | Phase 1 | Completed | - | - |
| NCT00738660 | Diabetic Nephropathy | Phase 3 | Completed | - | India ... more >> Post Graduate Institute of Medical Education and Research Chandigarh, India, 1600012 Collapse << |
| NCT02214979 | Healthy | Phase 1 | Completed | - | - |
| NCT00657865 | Healthy | Phase 4 | Completed | - | Austria ... more >> Medical University Vienna, Department of Clinical Pharacology Vienna, Austria, 1090 Collapse << |
| NCT02632747 | Diabetes Mellitus, Type 1 ... more >> Diabetes Mellitus, Type 2 Obesity Collapse << | Phase 2 | Recruiting | September 9, 2019 | Canada, Ontario ... more >> Toronto General Hospital Recruiting Toronto, Ontario, Canada, M5G 2N2 Collapse << |
| NCT02791958 | Hypertension | Phase 2 | Active, not recruiting | October 2017 | - |
| NCT00542269 | Hypertension With Metabolic Sy... more >>ndrome Collapse << | Phase 4 | Terminated(Early termination o... more >>f the study due to slow recruitment.) Collapse << | - | United Kingdom ... more >> Addenbrookes Hospital Cambridge, United Kingdom Collapse << |
| NCT00939588 | Hypertension | Phase 2 | Completed | - | Russian Federation ... more >> Novartis Investigative Site Moscow, Russian Federation Collapse << |
| NCT01230034 | Hypertension ... more >>Type 2 Diabetes Mellitus Microalbuminuria Collapse << | Phase 3 | Unknown | July 2011 | Italy ... more >> University of Pavia Recruiting Pavia, Italy, 27100 Contact: Giuseppe Derosa, MD +39 0382 526217 giuseppe.derosa@unipv.it Sub-Investigator: Giuseppe Derosa, MD Collapse << |
| NCT02901977 | Hypertension | Phase 4 | Completed | - | Sweden ... more >> Karolinska Institutet, Daprtment of Clinical Sciences, Danderyd Hospital, Cardiovascular Research Laboratory Stockholm, Sweden, SE-182 88 Collapse << |
| NCT03099213 | Coronary Artery Disease | Phase 4 | Completed | - | Indonesia ... more >> Sanofi Administrative Office Bali, Indonesia Collapse << |
| NCT01042392 | Essential Hypertension | Phase 4 | Completed | - | - |
| NCT00542269 | - | - | Terminated(Early termination o... more >>f the study due to slow recruitment.) Collapse << | - | - |
| NCT02499822 | Hypertension ... more >>High Blood Pressure Variability Collapse << | Phase 4 | Recruiting | December 2018 | China ... more >> Centre for Epidemiological Studies and Clinical Trials, Ruijin Hospital Shanghai Institute of Hypertension, Shanghai Jiaotong University School of Medicine Recruiting Shanghai, China Contact: Jiguang Wang, MD jiguangwang@aim.com Greece Hypertension Center, Third University Department of Medicine, Sotiria Hospital Not yet recruiting Athens, Greece Contact: George S Stergiou, MD gstergi@med.uoa.gr Italy Istituto Auxologico Italiano Recruiting Milan, Italy Contact: Gianfranco Parati, MD 390261911 ext 2890 gianfranco.parati@unimib.it Collapse << |
| NCT02215005 | Healthy | Phase 1 | Completed | - | - |
| NCT01037530 | Intermittent Claudicants | Phase 4 | Completed | - | United Kingdom ... more >> Hull and East Yorkshire Hospitals Hull, Yorkshire, United Kingdom, HU3 2JZ Collapse << |
| NCT00389519 | Hypertension | Phase 3 | Terminated(A planned interim a... more >>nalysis was performed after approx. 240 subjects completed the trial. The study was stopped, as permitted by protocol, after the analysis.) Collapse << | - | - |
| NCT01042392 | - | - | Completed | - | - |
| NCT00863915 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> CEDRA Clinical Research, LLC Austin, Texas, United States, 78759 Collapse << |
| NCT00389519 | - | - | Terminated(A planned interim a... more >>nalysis was performed after approx. 240 subjects completed the trial. The study was stopped, as permitted by protocol, after the analysis.) Collapse << | - | - |
| NCT02214966 | Healthy | Phase 1 | Completed | - | - |
| NCT00864448 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> CEDRA Clinical Research, LLC Austin, Texas, United States, 78759 Collapse << |
| NCT00864162 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> CEDRA Clinical Research, LLC Austin, Texas, United States, 78759 Collapse << |
| NCT00772577 | Hypertension | Phase 4 | Completed | - | United States, California ... more >> Investigative Site Beverly Hills, California, United States Investigative Site Santa Ana, California, United States United States, Georgia Investigative Site Conyers, Georgia, United States United States, Kentucky Investigative Site Lexington, Kentucky, United States United States, South Carolina Investigative Site Columbia, South Carolina, United States United States, Texas Investigative Site Houston, Texas, United States Collapse << |
| NCT00631917 | Hypertension | Phase 4 | Completed | - | United States, Missouri ... more >> Investigative Site Kansas City, Missouri, United States Argentina Investigative Site Investigative Site, Argentina Colombia Investigative Site Investigative Site, Colombia France Investigative Site Investigative Site, France Germany Investigative Site Investigative Site, Germany India Investigative Site investigative Site, India Spain Investigative Site Investigative Site, Spain Collapse << |
| NCT00473174 | Hypertension | Phase 4 | Completed | - | Spain ... more >> Hospital Clínico Universitario de Santiago Santiago de Compostela, Spain, 15701 Collapse << |
| NCT00772577 | - | - | Completed | - | - |
| NCT00879476 | Healthy | Not Applicable | Completed | - | India ... more >> Ranbaxy Clinical Pharmacology Unit, Ranbaxy Laboratories Limited Noida, Uttar Pradesh, India, 201 301 Collapse << |
| NCT00574834 | - | - | Terminated(Lack of funding) | - | - |
| NCT00574834 | Metabolic Syndrome | Early Phase 1 | Terminated(Lack of funding) | - | United States, Maryland ... more >> University of Maryland, Baltimore Baltimore, Maryland, United States, 21201 Collapse << |
| NCT00879788 | Healthy | Not Applicable | Completed | - | India ... more >> Clinical Pharmacology Unit, Majeedia Hospital (2nd Floor) New Delhi, India, 110 062 Collapse << |
| NCT00529451 | Hypertension | Phase 3 | Completed | - | China ... more >> Novartis Investigative Sites China, China India Novartis Investigative Sites India, India Thailand Novartis Investigative Sites Thailand, Thailand Collapse << |
| NCT00631917 | - | - | Completed | - | - |
| NCT01402479 | Migraine With Hypertension | Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01053910 | Atherosclerosis | Phase 4 | Completed | - | Bangladesh ... more >> Sanofi-Aventis Administrative Office Dhaka, Bangladesh Collapse << |
| NCT02729441 | Diabetes Micr... more >>oalbuminuria Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N3M5 Collapse << |
| NCT01856361 | Oligospermia ... more >>Teratospermia Asthenozoospermia Male Infertility Collapse << | Not Applicable | Terminated(We did not accrue a... more >>s much as anticipated and the PI (Dabaja) is no longer at Weill Cornell.) Collapse << | - | United States, New York ... more >> Weill Cornell Medical College, Department of Urology New York, New York, United States, 10065 Collapse << |
| NCT01856361 | - | - | Terminated(We did not accrue a... more >>s much as anticipated and the PI (Dabaja) is no longer at Weill Cornell.) Collapse << | - | - |
| NCT01164761 | Healthy | Phase 1 | Completed | - | India ... more >> Lambda Therapeutic Research Limited Ahmedabad, Gujrat, India, 380054 Collapse << |
| NCT03715998 | Myocardial Infarction | Phase 2 | Not yet recruiting | September 1, 2020 | - |
| NCT03440177 | - | - | Recruiting | December 2026 | Norway ... more >> St. Olavs University Hospital Recruiting Trondheim, Norway Contact: Magnus Strømmen, MSc Collapse << |
| NCT03414723 | Diabetes Mellitus ... more >> Healthy Subjects Collapse << | Phase 1 | Completed | - | Germany ... more >> Investigational Site Number 2760001 Berlin, Germany, 14050 Collapse << |
| NCT02842424 | Peripheral Arterial Disease | Phase 4 | Recruiting | March 31, 2020 | United States, Nebraska ... more >> VA Medical Center Recruiting Omaha, Nebraska, United States, 68105 Contact: Holly DeSpiegelaere 402-995-4171 Holly.DeSpiegelaere@va.gov Collapse << |
| NCT01164774 | Healthy | Phase 1 | Completed | - | India ... more >> Lambda Therapeutic Research Limited Ahmedabad, Gujrat, India, 380054 Collapse << |
| NCT01743014 | Diabetes Type 2 ... more >> Diabetic Nephropathy Vascular Disease Collapse << | Phase 4 | Unknown | July 2015 | Greece ... more >> AHEPA University Hospital Recruiting Thessaloniki, Greece, 546 36 Contact: Fotios S Iliadis, Lecturer of Internal Medicine +302310993587 iliadis@med.auth.gr Contact: Vaia F Bougatsa, Resident of Internal Medicine +306944334265 vaiabou@yahoo.gr Aristotle University of Thessaloniki/ AHEPA University Hospital Recruiting Thessaloniki, Greece Principal Investigator: Vaia F Bougatsa, MD Collapse << |
| NCT00529451 | - | - | Completed | - | - |
| NCT03475186 | Glioblastoma ... more >>Radiotherapy; Complications Cognitive Decline Chemoradiation Collapse << | Phase 2 | Recruiting | August 2021 | United States, North Carolina ... more >> Wake Forest Baptist Medical Center Recruiting Winston-Salem, North Carolina, United States, 27157 Contact: Karen Craver, MT, MHA 336-713-4394 kcraver@wakehealth.edu Contact: Magan Lane 336-716-5992 mlane@wakehealth.edu Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
