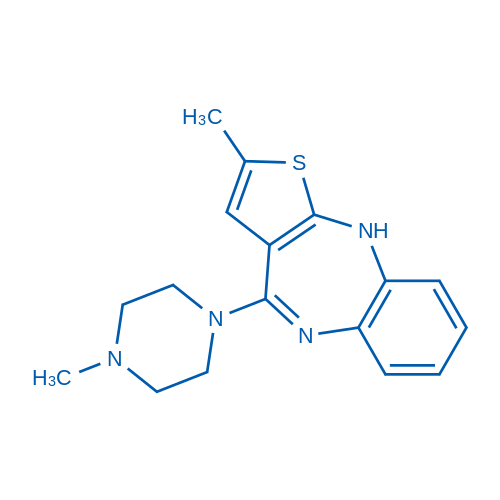
CAS No.: 132539-06-1
Olanzapine/奥氮平 Catalog No. CSN16653
Synonyms: LY170053;LY-170052;Zyprexa
Olanzapine is a multiple-target antagonist for 5-HT2A receptors and D2 dopamine receptors, as well as muscarinic M3 receptor.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01170117 Anorexia Nervosa Not Applicable Completed - United States, Maryland ... more >> Johns Hopkins Baltimore, Maryland, United States, 21287 United States, New York New York State Psychiatric Institute New York, New York, United States, 10032 Weill Cornell Medical Center White Plains, New York, United States, 10605 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 Canada, Ontario Centre for Addiction and Mental Health Toronto, Ontario, Canada, M5T1R8 Collapse << NCT00402324 Bipolar I Disorder Phase 4 Completed - United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hialeah, Florida, United States, 33016 Collapse << NCT00510211 - - Completed - France ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Paris, France, 75674 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Homburg, Germany, D-61350 Greece For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Thessaloniki, Greece, 54630 Collapse << - 更多
- 参考文献
- [1] Cipriani A, Rendell JM, et al. Olanzapine in long-term treatment for bipolar disorder. Cochrane Database Syst Rev. 2009 Jan 21;(1):CD004367.
- [2] Bymaster FP, Rasmussen K, et al. In vitro and in vivo biochemistry of olanzapine: a novel, atypical antipsychotic drug. J Clin Psychiatry. 1997;58 Suppl 10:28-36.
- [3] Li XM, Perry KW, Wong DT, Bymaster FP. Olanzapine increases in vivo dopamine and norepinephrine release in rat prefrontal cortex, nucleus accumbens and striatum. Psychopharmacology (Berl). 1998;136(2):153-161.
- [5] Bymaster FP, Rasmussen K, Calligaro DO, et al. In vitro and in vivo biochemistry of olanzapine: a novel, atypical antipsychotic drug. J Clin Psychiatry. 1997;58 Suppl 10:28-36.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 132539-06-1 | 储存条件 |
|
|||||
| 分子式 | C17H20N4S | 运输 | 蓝冰 | |||||
| 分子量 | 312.43 | 别名 | LY170053;LY-170052;Zyprexa;Zolafren;奥氮平 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01170117 | Anorexia Nervosa | Not Applicable | Completed | - | United States, Maryland ... more >> Johns Hopkins Baltimore, Maryland, United States, 21287 United States, New York New York State Psychiatric Institute New York, New York, United States, 10032 Weill Cornell Medical Center White Plains, New York, United States, 10605 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 Canada, Ontario Centre for Addiction and Mental Health Toronto, Ontario, Canada, M5T1R8 Collapse << |
| NCT00402324 | Bipolar I Disorder | Phase 4 | Completed | - | United States, Florida ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hialeah, Florida, United States, 33016 Collapse << |
| NCT00510211 | - | - | Completed | - | France ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Paris, France, 75674 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bad Homburg, Germany, D-61350 Greece For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Thessaloniki, Greece, 54630 Collapse << |
| NCT00885690 | Schizophrenia | Not Applicable | Terminated(Due to recruitment ... more >>problems) Collapse << | - | Denmark ... more >> Aalborg Psychiatric Hospital Aalborg, Denmark, 9000 Sweden Universitets Allmänna Sjukhuset, Malmø UMAS Malmø, Sweden Collapse << |
| NCT00100776 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT00174265 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00438776 | Pathological Gambling | Phase 3 | Completed | - | United States, Ohio ... more >> University of Cincinnati Medical Center Cincinnati, Ohio, United States, 45267-0559 Collapse << |
| NCT00174265 | - | - | Completed | - | - |
| NCT00418171 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> Pfizer Investigational Site San Bernardino, California, United States United States, Hawaii Pfizer Investigational Site Aiea, Hawaii, United States United States, Illinois Pfizer Investigational Site Schaumburg, Illinois, United States United States, Oregon Pfizer Investigational Site Grants Pass, Oregon, United States United States, West Virginia Pfizer Investigational Site Whitehall, West Virginia, United States Collapse << |
| NCT00159783 | - | - | Completed | - | - |
| NCT00402324 | - | - | Completed | - | - |
| NCT00159783 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT01996488 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd. Village Bhat, Gandhinagar, Gujarat, India, 382428 Collapse << |
| NCT02287259 | Bipolar Disorder | Phase 4 | Recruiting | October 2019 | Japan ... more >> Chiba University Hospital Recruiting Chiba-shi, Japan Contact: Tadashi Hasegawa t0715.hasegawa@gmail.com Collapse << |
| NCT01996501 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd. Village Bhat, Gandhinagar, Gujarat, India, 382428 Collapse << |
| NCT00934908 | Bipolar Disorder ... more >> Schizophrenia Collapse << | Not Applicable | Withdrawn | - | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT00179062 | Schizophrenia ... more >> Schizoaffective Disorder Depressive Disorder, Major Bipolar Disorder Collapse << | Not Applicable | Completed | - | United States, Tennessee ... more >> Psychiatric Hospital at Vanderbilt Nashville, Tennessee, United States, 37212 Collapse << |
| NCT03578081 | Malignant Neoplasm | Phase 3 | Recruiting | March 2020 | - |
| NCT00203307 | - | - | Terminated(Study terminated by... more >> sponsor) Collapse << | - | - |
| NCT02635984 | Complications of Bone Marrow T... more >>ransplant Hematologic Neoplasms Collapse << | Phase 3 | Completed | - | United States, Georgia ... more >> Augusta University Medical Center Augusta, Georgia, United States, 30912 Collapse << |
| NCT00212836 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00203307 | Migraine | Not Applicable | Terminated(Study terminated by... more >> sponsor) Collapse << | - | United States, Pennsylvania ... more >> Jefferson Headache Center Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02635984 | - | - | Completed | - | - |
| NCT00287820 | Schizophrenia ... more >> Diabetes Metabolic Syndrome Hyperglycemia Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Manhattan Psychaitric Center New York, New York, United States, 10035 Collapse << |
| NCT00143182 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT00190749 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Diego, California, United States, 92161 Collapse << |
| NCT00852956 | Healthy | Phase 1 | Completed | - | Romania ... more >> IFE Human Pharmacology Arad, Romania IFE Human Pharmacology Timisoara, Romania Collapse << |
| NCT00129220 | Bipolar Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 470-1168 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Akita, Japan, 010-1654 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 283-0062 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 807-8555 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gunma, Japan, 370-2455 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 004-0841 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 663-8501 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 236-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kumamoto, Japan, 861-0002 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 384-8540 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 700-8558 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okinawa, Japan, 904-2222 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 561-0803 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saga, Japan, 842-0192 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 343-0032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 160-0023 Collapse << |
| NCT00485680 | Bipolar Disorder | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China Collapse << |
| NCT00183612 | Bipolar Disorder ... more >> Psychotic Disorders Collapse << | Not Applicable | Terminated | - | United States, Massachusetts ... more >> McLean Hospital Belmont, Massachusetts, United States, 02478 Cambridge Health Alliance Medford, Massachusetts, United States, 02155 Collapse << |
| NCT00833300 | Delirium Agit... more >>ation Collapse << | Not Applicable | Terminated | - | Canada, Nova Scotia ... more >> Halifax Infirmary; Queen Elizabeth II Health Sciences Centre Halifax, Nova Scotia, Canada Victoria General Hospital; Queen Elizabeth II Health Sciences Centre Halifax, Nova Scotia, Canada Collapse << |
| NCT00970281 | Schizophrenia | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 470-1168 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gunma, Japan, 3703603 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 0788208 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 234-0051 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kumamoto, Japan, 8660895 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nara, Japan, 634-8522 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okinawa, Japan, 9012111 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 593 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 343-0851 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 120-0005 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yamaguchi, Japan, 579-6613 Collapse << |
| NCT00190749 | - | - | Completed | - | - |
| NCT01148264 | Emesis | Phase 2 | Terminated(poor enrolment) | - | Germany ... more >> Martin-Luther-University Halle-Wittenberg, Department for Oncology and Hematology, Ernst-Grube-Strasse 40 Halle (Saale), Sachsen-Anhalt, Germany, 06097 Collapse << |
| NCT00970281 | - | - | Completed | - | - |
| NCT01282632 | Subjects Had Unipolar, Non-psy... more >>chotic Major Depression Collapse << | Phase 1 Phase 2 | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT00129220 | - | - | Completed | - | - |
| NCT03219710 | Chemotherapy-induced Nausea an... more >>d Vomiting Collapse << | Phase 3 | Recruiting | June 2019 | India ... more >> Dr Bra Irch, Aiims, New Delhi Recruiting New Delhi, Delhi, India, 110029 Contact: Ramavath Devendra Naik, MD 9968721210 devaiims2012@gmail.com Contact: Sameer Bakhshi, MD 01129575253 ext 5253 sambakh@hotmail.com Collapse << |
| NCT01123408 | Schizophrenia | Not Applicable | Completed | - | United States, New York ... more >> Nathan Kline Institute Orangeburg, New York, United States, 10962 Collapse << |
| NCT02006862 | - | - | Completed | - | Greece ... more >> Agia Olga Konstantopouleion Hospital Athens, Greece Collapse << |
| NCT00303602 | Schizophrenia ... more >> Schizoaffective Disorder Bipolar Disorder Collapse << | Phase 4 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Escondido, California, United States, 92025 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Garden Grove, California, United States, 92845 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. National City, California, United States, 91950 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Orange, California, United States, 92868 United States, Georgia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Atlanta, Georgia, United States, 30308 United States, Nevada For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Las Vegas, Nevada, United States, 89102 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Staten Island, New York, United States, 10312 Canada, British Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Penticton, British Columbia, Canada, V2A 4M4 Canada, Manitoba For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Winnipeg, Manitoba, Canada, R3E 3N4 Canada, Nova Scotia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dartmouth, Nova Scotia, Canada, B2Y 3Z9 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Halifax, Nova Scotia, Canada, B3H 2E2 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sydney, Nova Scotia, Canada, B1S 2E8 Canada, Ontario For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chatham, Ontario, Canada, N7L 1B7 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kingston, Ontario, Canada, K7L 4X3 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sudbury, Ontario, Canada, P3C 1T4 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Windsor, Ontario, Canada, N9C 3Z4 Canada, Quebec For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sherbrooke, Quebec, Canada, J1G 1W4 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mexico City, Mexico, 01030 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Amersfoort, Netherlands, 3816 CP For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rotterdam, Netherlands, 3015 GD Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico, 00918 Collapse << |
| NCT00266630 | Manic or Mixed Episode Associa... more >>ted With Bipolar I Disorder Collapse << | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Akita, Japan, 010-1654 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 292-0061 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 812-8582 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gunma, Japan, 371-8511 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 005-0004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 663-8501 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nara, Japan, 634-8522 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 710-0055 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okinawa, Japan, 900-0005 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 343-0032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 151-0053 Collapse << |
| NCT01920802 | Healthy | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00113594 | Schizophrenia ... more >> Bipolar Disorder Collapse << | Phase 3 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Cerritos, California, United States For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician National City, California, United States For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Poway, California, United States United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Fort Lauderdale, Florida, United States United States, Idaho For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Eagle, Idaho, United States United States, Louisiana For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Lake Charles, Louisiana, United States United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Glen Burnie, Maryland, United States United States, New Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Albuquerque, New Mexico, United States United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician New York, New York, United States For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Olean, New York, United States United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Cincinnati, Ohio, United States United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Houston, Texas, United States United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Portsmouth, Virginia, United States United States, Washington For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Kirkland, Washington, United States United States, Wisconsin For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician West Allis, Wisconsin, United States Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician San Juan, Puerto Rico Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician Moscow, Russian Federation Collapse << |
| NCT00266630 | - | - | Completed | - | - |
| NCT00303602 | - | - | Completed | - | - |
| NCT03571126 | Chemotherapy-induced Nausea an... more >>d Vomiting Lung Cancer Collapse << | Phase 4 | Not yet recruiting | June 1, 2020 | - |
| NCT00640510 | - | - | Completed | - | - |
| NCT00640510 | Schizophrenia | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukushima, Japan, 966-0902 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gunma, Japan, 377-0055 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kagoshima, Japan, 899-5652 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 234-0051 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kumamoto, Japan, 861-0002 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 716-0061 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okinawa, Japan, 904-0011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saga, Japan, 842-0192 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 187-8551 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tottori, Japan, 682-0023 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yamagata, Japan, 999-2221 Collapse << |
| NCT00169065 | Schizophrenia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Commonwealth Research Center Jamaica Plain, Massachusetts, United States, 02130 Collapse << |
| NCT00512291 | Advanced Cancer | Not Applicable | Completed | - | United States, Texas ... more >> U.T.M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02643355 | Substance Withdrawal Syndrome | Not Applicable | Recruiting | November 2017 | United States, Minnesota ... more >> Hennepin County Medical Center Recruiting Minneapolis, Minnesota, United States, 55415 Contact: Audrey Hendrickson, MPH 612-873-9528 ED.Research.Associate.Program@hcmed.org Collapse << |
| NCT01920802 | - | - | Completed | - | - |
| NCT01864551 | Bipolar Disorder | Phase 4 | Completed | - | Taiwan ... more >> Tri-Service General Hospital Taipei, Taiwan, 114 Collapse << |
| NCT01687478 | Treatment Resistant Depression | Phase 3 | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baoding, China, 071000 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100088 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kunming, China, 650032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210029 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wuhan, China, 430022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710061 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xinxiang, China, 453002 Collapse << |
| NCT01496183 | Healthy | Not Applicable | Completed | - | United States, New York ... more >> Department of Psychiatry, The State University of New York at Buffalo Buffalo, New York, United States, 14215 Collapse << |
| NCT01496183 | - | - | Completed | - | - |
| NCT00746785 | Alcohol Dependence | Phase 3 | Completed | - | United States, New Mexico ... more >> The Mind Research Network Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT01687478 | - | - | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | - |
| NCT00699946 | Delirium | Not Applicable | Completed | - | United States, Massachusetts ... more >> New England Baptist Hospital Boston, Massachusetts, United States, 02120 Collapse << |
| NCT00512070 | Schizophrenia ... more >> Schizoaffective Disorder Bipolar Disorder Obesity Metabolic Syndrome Collapse << | Not Applicable | Active, not recruiting | December 2021 | United States, Washington ... more >> VA Puget Sound Health Care System Tacoma, Washington, United States, 98493 Collapse << |
| NCT00741026 | Insulin Resistance ... more >> Diabetes Mellitus Collapse << | Not Applicable | Completed | - | United States, Pennsylvania ... more >> Penn State College of Medicine, Penn State Milton S. Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT00746785 | - | - | Completed | - | - |
| NCT00672464 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Withdrawn(lack of funding) | February 2010 | United States, North Carolina ... more >> John Umstead Hospital Butner, North Carolina, United States, 27509 Duke University Medical Center Durham, North Carolina, United States, 27705 Collapse << |
| NCT00741026 | - | - | Completed | - | - |
| NCT00592930 | Anorexia Nervosa | Phase 4 | Completed | - | United States, New York ... more >> Schneider Children's Hospital New Hyde Park, New York, United States, 11040 Collapse << |
| NCT02380118 | Acute Agitation, Behavioural E... more >>mergency Collapse << | Phase 4 | Recruiting | March 2022 | Hong Kong ... more >> Pamela Youde Nethersole Eastern Hospital Recruiting Hong Kong, Hong Kong Contact: Antony CP Wong Prince of Wales Hospital Recruiting Hong Kong, Hong Kong Contact: Colin A. Graham Queen Mary Hospital Recruiting Hong Kong, Hong Kong Contact: Ling Pong Leung Ruttonjee Hospital Recruiting Hong Kong, Hong Kong Contact: Tong Shun Chung Tuen Mun Hospital Recruiting Hong Kong, Hong Kong Contact: Chun Tat Lui United Christian Hospital Recruiting Hong Kong, Hong Kong Contact: Hiu Fung Lam Collapse << |
| NCT00194064 | Bipolar Disorder | Phase 3 | Terminated(Slow enrollment) | - | United States, Ohio ... more >> University Hospitals of Cleveland Cleveland, Ohio, United States, 44140 Collapse << |
| NCT01609153 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Recruiting | April 2020 | Germany ... more >> RWTH Aachen Recruiting Aachen, Germany, 52074 Contact: Tanja Veselinovic, Dr. med. +49-(0)241-8080 ext 0 tveselinovic@ukaachen.de Contact: Marc Augustin, Dr. med. +49-(0)241-8080 ext 166 maugustin@ukaachen.de Rheinhessen Fachklinik Alzey Recruiting Alzey, Germany, 55232 Contact: Anke Brockhaus-Dumke, PD Dr. med. +49-(0)6731 ext 501212 a.brockhaus-dumke@rfk.landeskrankenhaus.de Contact: Siamak Roushanai, Dr. med. +49-(0)6731 ext 501593 s.roushanai@rfk.landeskrankenhaus.de Rhein-Mosel-Fachklinik Andernach Recruiting Andernach, Germany, 56626 Contact: Andreas Konrad, PD Dr. med. +49-(0)2632-407 ext 5534 a.konrad@rmf.landeskrankenhaus.de Contact: Stefan Elsner, Dr. med. +49-(0)2632-407 ext 5412 s.elsner@rmf.landeskrankenhaus.de Bezirkskliniken Schwaben, Bezirkskrankenhaus Augsburg Recruiting Augsburg, Germany, 89156 Contact: Susanne Baumgärtner +49-0821-4803 ext 1021 wissenschaftsabteilung@bkh-augsburg.de Contact: Marianne Bärhold, Dr. +49-0821-4803 ext 3770 wissenschaftsabteilung@bkh-augsburg.de Charite-Universitätsmedizin Berlin Withdrawn Berlin, Germany, 10117 LWL-Klinik Dortmund Recruiting Dortmund, Germany, 44281 Contact: Hans-Jörg Assion, Prof. Dr. +49-(0)231-45092673 hans-joerg.assion@wkp-wl.org Contact: Gerhard Reymann, PD Dr. gerhard.reymann@lwl.org LVR-Klinikum Düsseldorf Recruiting Düsseldorf, Germany, 40629 Contact: Christian Schmidt-KraepelinCordes, Dr. +49-(0)211-922 ext 2783 christian.schmidt-kraepelin@lvr.de Contact: Joachim Cordes, PD Dr. +49-(0)211-922 ext 3402 joachim.cordes@lvr.de Zentrum für Seelische Gesundheit Kreiskliniken Darmstadt-Dieburg Standort: Groß-Umstadt Recruiting Groß-Umstadt, Germany, 64823 Contact: Thomas Wobrock, Prof. MD +49-(0)6078-79 ext 2901 t.wobrock@kreiskliniken-dadi.de Contact: Jens Edinger j.edinger@kreiskliniken-dadi.de Universitätsmedizin Göttingen Recruiting Göttingen, Germany, 37075 Contact: Bernhard Kis, Dr. +49-(0)551-3910114 Bernhard.Kis@med.uni-goettingen.de Contact: David Zilles, Dr. dzilles@gwdg.de Klinik für Psychiatrie, Psychotherapie und Psychosomatik am Bezirkskrankehaus Günzburg Suspended Günzburg, Germany, 89312 Klinik für Psychiatrie, Sozialpsychiatrie und Psychotherapie Medizinische Hochschule Hannover (MHH) Recruiting Hannover, Germany, 30625 Contact: Kai Kahl, Prof. Dr. 0049511532 ext 2495 kahl.kai@mh-hannover.de Contact: Helge Frieling, Prof. Dr. 0049511532 ext 2495 frieling.helge@mh-hannover.de Universitätsklinikum Heidelberg Withdrawn Heidelberg, Germany, 69115 LVR-Klinikum Köln Recruiting Köln, Germany, 51109 Contact: Dirk Reske, Dr. +49-(0)2218993 ext 797 dirk.reske@lvr.de Contact: Ulrike Reinholz, Dr. +49-(0)2218993 ext 0 ulrike.reinholz@lvr.de LVR-Klinik Langenfeld Recruiting Langenfeld, Germany, 40764 Contact: Andrea Neff, Dr. +49-(0)2173102 ext 0 andrea.neff@lvr.de Contact: Martina Pellio-Blume, Dr. +49-(0)2173102 ext 0 martina.pellio-blume@lvr.de Universitätsklinikum Leipzig, Klinik und Poliklinik für Psychiatrie und Psychotherapie Recruiting Leipzig, Germany, 04103 Contact: Michael Kluge, Dr 00493419724 ext 673 michael.kluge@medizin.uni-leipzig.de Contact: Jens Dietzel, Dr. 00493419724 ext 530 jens.dietzel@medizin.uni-leipzig.de Universitätsmedizin Mainz Klinik für Psychiatrie und Psychotherapie Withdrawn Mainz, Germany, 55131 Zentralinstitut für Seelische Gesundheit Recruiting Mannheim, Germany, 68159 Contact: Gerhard Gründer, Prof. MD +49-(0)621-1703 ext 2911 Gerhard.Gruender@zi-mannheim.de Contact: Mathias Zink, Prof. MD +49-(0)621-1703 ext 2911 Mathias.Zink@zi-mannheim.de LMU München Recruiting München, Germany, 80336 Contact: Peter Falkai, Prof. Dr. +49-(0)89-5160 ext 5501 Peter.Falkai@med.uni-muenchen.de Contact: Berend Malchow, Dr. +49-(0)89-4400 ext 55331 Berend.Malchow@med.uni-muenchen.de TU München Suspended München, Germany, 81675 Bezirksklinikum Regensburg, Klinik für Psychiatrie und Psychotherapie Recruiting Regensburg, Germany, 93053 Contact: Berthold Langguth, PD Dr. med. +49-(0)941941 ext 2099 berthold.langguth@medbo.de Contact: Elmar Frank, Dr. +49-(0)941941 ext 0 elmar.frank@medbo.de Universitätsklinikum Würzburg Withdrawn Würzburg, Germany, 97080 Collapse << |
| NCT00797277 | Schizophrenia ... more >> Schizoaffective Disorder Agitation Collapse << | Phase 3 | Completed | - | Taiwan ... more >> Department of Psychiatry, National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT00088465 | - | - | Completed | - | - |
| NCT00088465 | Schizophrenic Disorders ... more >> Schizoaffective Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT00260962 | Anorexia Nervosa | Phase 2 | Completed | - | Canada, Ontario ... more >> Ottawa Hospital Ottawa, Ontario, Canada, K1Y 4E9 Collapse << |
| NCT00260962 | - | - | Completed | - | - |
| NCT00797277 | - | - | Completed | - | - |
| NCT00320489 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00320489 | - | - | Completed | - | - |
| NCT00510146 | Depression, Bipolar | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100088 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410008 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chengdu, China, 610041 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510370 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310003 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kunming, China, 650032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210029 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wu Han, China, 430060 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710032 Japan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 731-0501 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seongnam-Si, Korea, Republic of, 463-707 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00618748 | Bipolar I Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 470-1168 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 270-1694 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 731-0501 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Iwate, Japan, 023-0801 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 231-0027 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 616-8421 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 332-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yamaguchi, Japan, 755-8505 Collapse << |
| NCT01303601 | Bipolar Disorder ... more >> Depression, Bipolar Collapse << | Phase 4 | Completed | - | China, Liaoning ... more >> Department of Psychiatry, the First Hospital of China Medical University Shenyang, Liaoning, China, 110001 Collapse << |
| NCT00618748 | - | - | Completed | - | - |
| NCT00489593 | Advanced Cancer ... more >> Weight Loss Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00910988 | - | - | Completed | - | - |
| NCT00910988 | Hyperglycemia ... more >> Hyperlipidemia Collapse << | Not Applicable | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00510146 | - | - | Completed | - | - |
| NCT00734435 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Collapse << | Phase 2 | Terminated(Sponsor Decision- F... more >>inancial Considerations) Collapse << | - | United States, California ... more >> Synergy Research National City, California, United States, 91950 UCI Medical Center Orange, California, United States, 92868 CNRI San Diego San Diego, California, United States, 92126 United States, Florida Florida Clincal Research Center Bradenton, Florida, United States, 34208 Florida Clinical Research Center Maitland, Florida, United States, 34208 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Illinois American Medical Research Oak Brook, Illinois, United States, 60523 United States, Indiana Larue D. Carter Hospital Indianapolis, Indiana, United States, 46222 United States, New York Brooklyn Medical Institute Brooklyn, New York, United States, 11223 Collapse << |
| NCT00186017 | Bipolar Disorder | Phase 4 | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00186017 | - | - | Completed | - | - |
| NCT03478605 | Emesis Vomiti... more >>ng Nausea Post Chemotherapy Nausea Chemotherapy-induced Nausea and Vomiting Collapse << | Phase 2 | Recruiting | June 1, 2019 | Russian Federation ... more >> N.N. Blokhin Cancer Research Center Recruiting Moscow, Russian Federation, 115478 Contact: Alexey A Rumyantsev, MD +79100022255 alexeymma@gmail.com Principal Investigator: Alexey A Rumyantsev, MD Principal Investigator: Ilya A Pokataev, MD Collapse << |
| NCT00636896 | Weight Gain | Not Applicable | Completed | - | United States, North Dakota ... more >> Neuropsychiatric Research Institute Fargo, North Dakota, United States, 58103 Collapse << |
| NCT00287352 | Psychotic Disorder ... more >> Schizophreniform Disorder Schizophrenia Schizoaffective Disorder Mood Disorders With Psychotic Features Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00273624 | Therapy-resistant Depression | Phase 3 | Terminated | - | Germany ... more >> Dept. of Psychiatry, University of Freiburg Freiburg, Germany, 79104 Collapse << |
| NCT00568672 | Relapse Rate of a Major Depres... more >>sive Episode Safety of Olanzapine in Subjects With Major Depression Collapse << | Phase 3 | Withdrawn(Withdrawn because of... more >> personal ressources before inclusion of the first patient) Collapse << | April 2008 | Germany ... more >> Departement of Psychiatry and Psychotherapy, Charite, University Medicine Berlin, Campus Benjamin Franklin Berlin, Germany, 14050 Collapse << |
| NCT00977301 | HIV Infections | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud Universtity Nijmegen Medical Centre Nijmegen, Netherlands Collapse << |
| NCT00678457 | Alcohol Dependence | Phase 2 | Completed | - | United States, Virginia ... more >> UVA CARE Charlottesville, Virginia, United States, 22911 UVA CARE Richmond Richmond, Virginia, United States, 23294 Collapse << |
| NCT02290470 | Nausea Vomiti... more >>ng Collapse << | Phase 2 | Unknown | November 2015 | Italy ... more >> Istituto Nazionale dei Tumori Recruiting Milan, Italy, 20133 Principal Investigator: Luigi Celio Collapse << |
| NCT02755116 | Postoperative Nausea and Vomit... more >>ing Collapse << | Phase 2 | Recruiting | December 2020 | United States, New York ... more >> Icahn School of Medicine at Mount Sinai Recruiting New York, New York, United States, 10029 Principal Investigator: Jaime Hyman, MD Collapse << |
| NCT01411930 | Diabetes | Phase 4 | Completed | - | United States, California ... more >> VA San Diego Healthcare System San Diego, California, United States, 92161 Collapse << |
| NCT02447835 | Normal Non-fluency | Phase 1 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01170117 | - | - | Completed | - | - |
| NCT02704962 | Schizophrenia | Phase 4 | Completed | - | Taiwan ... more >> Kai-Suan Psychiatric Hospital Kaohsiung, Taiwan, 802 Kai-Suan Psychiatric Hospital Kaohsiung, Taiwan Collapse << |
| NCT03216941 | Psychomotor Agitation ... more >> Psychiatric Disorder Emergency Psychiatric Tranquilizers Collapse << | Phase 4 | Completed | - | Argentina ... more >> hospital Larcade San Miguel, Buenos Aires, Argentina, 1405 Collapse << |
| NCT00275301 | Borderline Personality Disorde... more >>r Collapse << | Not Applicable | Completed | - | United States, Minnesota ... more >> University of Minnesota, Dept of Psychiatry Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT00275301 | - | - | Completed | - | - |
| NCT02918825 | Schizophrenia | Not Applicable | Completed | - | China ... more >> Beijing HuiLongGuan Hospital Beijing, China, 100096 Collapse << |
| NCT03246620 | Acute Agitation ... more >> Behavioural Emergency Collapse << | Phase 4 | Recruiting | June 2022 | Hong Kong ... more >> Queen Elizabeth Hospital Recruiting Hong Kong, Hong Kong Contact: Kin Ling Chan Collapse << |
| NCT02097823 | - | - | Completed | - | - |
| NCT02939287 | Nausea Vomiti... more >>ng Collapse << | Phase 3 | Unknown | November 2018 | - |
| NCT02097823 | Chemotherapy Induced Nausea an... more >>d Vomiting Collapse << | Phase 2 | Completed | - | United States, Indiana ... more >> Riley Hospital for Children at Indiana University Health Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01503437 | Fasting | Phase 1 | Completed | - | India ... more >> Wellquest Clinical Research Mumbai, Maharashtra, India, 400013 Collapse << |
| NCT01503450 | Fed | Phase 1 | Completed | - | India ... more >> Wellquest Clinical Research Mumbai, Maharashtra, India, 400013 Collapse << |
| NCT02861859 | Breast Cancer | Phase 3 | Recruiting | December 2020 | Canada, Ontario ... more >> The Ottawa Hospital Research Institute Cance Center Recruiting Ottawa, Ontario, Canada, K1H 8L6 Contact: Mark J Clemons, MD Collapse << |
| NCT01184443 | Eating Disorder | Phase 3 | Terminated(Poor recruitment) | - | Canada, Ontario ... more >> Children's Hospital of Eastern Ontario Ottawa, Ontario, Canada, K1H 8L1 Collapse << |
| NCT01503398 | Fasting | Phase 1 | Completed | - | India ... more >> Lambda Therapeutic Research Ltd., Ahmedabad, Gujrat, India, 380061 Collapse << |
| NCT00183404 | Autism | Phase 2 Phase 3 | Completed | - | United States, Pennsylvania ... more >> Drexel University College of Medicine at Friends Hospital Philadelphia, Pennsylvania, United States, 19124 Collapse << |
| NCT03304457 | Schizophrenia | Phase 4 | Completed | - | India ... more >> AIIMS Bhubaneshwar, Odisha, India, 751019 Collapse << |
| NCT02435654 | Early-onset Schizophrenia | Phase 4 | Recruiting | December 2018 | China, Yunnan ... more >> Psychiatry Department,First Affiliated Hospial Of Kunming Medical University Recruiting Kunming, Yunnan, China, 650032 Contact: Yang runxu 86-13708764303 707528149@qq.com Contact: Han dong 86-18288928851 372252241@qq.com Collapse << |
| NCT01503424 | Fed | Phase 1 | Completed | - | India ... more >> Lambda Therapeutic Research Ltd., Ahmedabad, Gujrat, India, 380061 Collapse << |
| NCT01625923 | Idiopathic Gastroparesis | Not Applicable | Recruiting | December 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Braden Kuo, MD 617-726-0196 Principal Investigator: Braden Kuo, MD United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Allen Lee, MD 734-936-9454 Principal Investigator: Allen Lee, MD Sub-Investigator: William Hasler, MD Collapse << |
| NCT02970643 | Chemotherapy-induced Nausea an... more >>d Vomiting Collapse << | Not Applicable | Unknown | December 2017 | - |
| NCT03575637 | Gastric Cancer | Phase 2 | Recruiting | June 2021 | China ... more >> Peking University Cancer Hospital & Institute Recruiting Beijing, China Contact: Shen Lin Collapse << |
| NCT03631004 | Postoperative Nausea ... more >> Postoperative Nausea and Vomiting Collapse << | Phase 2 Phase 3 | Not yet recruiting | June 2020 | Brazil ... more >> Cancer Institute of the State of Sao Paulo - ICESP Recruiting Sao Paulo, São Paulo, Brazil, 01403010 Contact: angela m sousa, phD 3893-2000 ext 4144 angela.sousa@hc.fm.usp.br Contact: Thiago R Grigio, MsC 38932000 ext 4144 thiago.grigio@hc.fm.usp.br Principal Investigator: Thiago R Grigio, MD Sub-Investigator: Angela M Sousa, phD Collapse << |
| NCT03118986 | Vomiting in Infants and/or Chi... more >>ldren Nausea Hematopoietic System--Cancer Collapse << | Phase 2 | Recruiting | April 2023 | Canada, Ontario ... more >> Hospital for Sick Children Recruiting Toronto, Ontario, Canada, M5G 1X8 Contact: Lee Dupuis, PhD 416-813-7654 ext 309355 lee.dupuis@sickkids.ca Collapse << |
| NCT03066622 | Headache | Phase 4 | Completed | - | United States, Minnesota ... more >> Regions Hospital Saint Paul, Minnesota, United States, 55101 Collapse << |
| NCT03679182 | Nausea Vomiti... more >>ng End Stage Cancer Collapse << | Phase 2 | Recruiting | September 2021 | Thailand ... more >> Jarin Chindaprasirt Recruiting Khon Kaen, Thailand, 40002 Contact: Jaruda Sringam, Master 6643366592 talnurse8@gmail.com Principal Investigator: Jarin Chindaprasirt, MD Sub-Investigator: Attakorn Raksasataya, MD Sub-Investigator: Suthan Chanthawong, PhD Collapse << |
| NCT02129478 | Chemotherapy-induced Nausea an... more >>d Vomiting Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Children's Hospital, London Health Sciences Centre London, Ontario, Canada, N6A 5W9 Children's Hospital of Eastern Ontario Ottawa, Ontario, Canada, K1H 8L1 The Hospital for Sick Children Toronto, Ontario, Canada, M5G1X8 Collapse << |
| NCT03137121 | Advanced Cancer | Phase 2 Phase 3 | Recruiting | September 30, 2019 | United States, Alabama ... more >> University of Alabama at Birmingham Recruiting Birmingham, Alabama, United States, 35294 United States, Indiana Indiana University Recruiting Indianapolis, Indiana, United States, 46202 Contact: Jessica Sastre, MA 317-278-5615 jsastre@iu.edu United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Mark Manemann 507-284-8759 Manemann.Mark@mayo.edu United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Melissa Haley, CCRP 314-747-8064 mhaley@wustl.edu United States, Wisconsin Hospital Sisters Health System (HSHS) St. Vincent Hospital Recruiting Green Bay, Wisconsin, United States, 54301 Contact: Stuart Mohoney 920-433-8905 Stuart.Mohoney@hshs.org Collapse << |
| NCT01617187 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT01990755 | Anorexia Nervosa ... more >> Bulimia Nervosa Collapse << | Not Applicable | Completed | - | Italy ... more >> Villa Garda Garda, Veneto, Italy, 37016 Collapse << |
| NCT00728195 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT02007928 | Dissociative Disorders ... more >> Schizophrenia Collapse << | Not Applicable | Completed | - | France ... more >> CH de Cannes Cannes, Alpes-maritimes, France Service de psychiatrie de l'enfant et de l'adolescent Nice, Alpes-Maritimes, France, 06200 CH D'antibes Antibes, Alpes-Maritime, France CH Henri laborit Poitiers, Charente Maritime, France, 86328 Fondation Vallée Gentilly, Paris, France Ch le Vinatier Bron, France, 69678 CHRU de Lille Lille, France Centre Hospitalier Spécialisé Esquirol Limoges, France CH St Jean de Dieu Lyon, France, 69355 HCL Lyon, France CHU de Nancy Nancy, France CHU de Nantes Nantes, France AP-HP Paris, France CHU de Toulouse Toulouse, France Collapse << |
| NCT00000373 | Obsessive-Compulsive Disorder | Phase 4 | Completed | - | United States, Florida ... more >> Psychiatric Specialty Clinic, Shands Hospital at the University of Florida Gainesville, Florida, United States, 32608 University of Florida Behavioral Health Mandarin Clinic Jacksonville, Florida, United States, 32257 Collapse << |
| NCT00191399 | Depression, Bipolar | Phase 4 | Completed | - | United States, Washington ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seattle, Washington, United States Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Bayamon, Puerto Rico, 00956 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Caguas, Puerto Rico, 725 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Canovanas, Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hato Rey, Puerto Rico, 00936 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Manati, Puerto Rico, 00674 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Mayaguez, Puerto Rico, 00680 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Rio Piedras, Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician San Juan, Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern time (UTC/GMT - 5 hours, EST) or speak with your personal physician Santurce, Puerto Rico Collapse << |
| NCT00396565 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00396565 | - | - | Completed | - | - |
| NCT00265551 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00932529 | Psychotic Disorders | Phase 4 | Completed | - | Norway ... more >> Haukeland University Hospital, Division of Psychiatry Bergen, Sandviken, Norway, N-5035 Collapse << |
| NCT00490971 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT02538081 | Schizophrenia | Phase 1 Phase 2 | Withdrawn(Withdrawn) | - | United States, Colorado ... more >> VA Eastern Colorado Health Care System, Denver, CO Denver, Colorado, United States, 80220 Collapse << |
| NCT01567124 | Schizophrenia | Phase 4 | Unknown | December 2014 | Australia, Victoria ... more >> Ballarat Health Service Psychiatric Services Recruiting Ballarat, Victoria, Australia Principal Investigator: Abdul Khalid Sub-Investigator: Rajul Tandon Sub-Investigator: Sonia Ghai Sub-Investigator: Arup Dhar Sub-Investigator: Sarah Tremethick Sub-Investigator: Jennifer Grigo Sub-Investigator: Pella Karalis Monash Medical Centre - Monash Health Recruiting Clayton, Victoria, Australia Principal Investigator: David Barton Sub-Investigator: Arup Dhar Sub-Investigator: Sarah Tremethick Sub-Investigator: Jennifer Grigo Sub-Investigator: Krishna Vaddadi Alfred and Baker Medical Unit - Alfred Hospital Recruiting Melbourne, Victoria, Australia Principal Investigator: Gavin Lambert Sub-Investigator: Markus Schlaich Sub-Investigator: Elisabeth Lambert Sub-Investigator: Murray Esler Sub-Investigator: Geoff Head Sub-Investigator: Dagmara Hering Sub-Investigator: Nina Eikelis Sub-Investigator: Carolina Ika Sari Sub-Investigator: Petra Marusic Sub-Investigator: Toni Rice Sub-Investigator: Mariee Grima Sub-Investigator: David Barton Sub-Investigator: Arup Dhar Sub-Investigator: Sarah Tremethick Sub-Investigator: Nora Straznicky Sub-Investigator: John Dixon Sub-Investigator: Jennifer Grigo Sub-Investigator: Donna Vizi Sub-Investigator: Louise Hammond Baker IDI Heart & Diabetes Institute Recruiting Melbourne, Victoria, Australia Principal Investigator: Gavin Lambert Sub-Investigator: Markus Schlaich Sub-Investigator: Elisabeth Lambert Sub-Investigator: Murray Esler Sub-Investigator: Geoff Head Sub-Investigator: Dagmara Hering Sub-Investigator: Nina Eikelis Sub-Investigator: Carolina Ika Sari Sub-Investigator: Petra Marusic Sub-Investigator: Toni Rice Sub-Investigator: Mariee Grima Sub-Investigator: David Barton Sub-Investigator: Arup Dhar Sub-Investigator: Sarah Tremethick Sub-Investigator: Nora Straznicky Sub-Investigator: John Dixon Sub-Investigator: Jennifer Grigo Sub-Investigator: Donna Vizi Sub-Investigator: Louise Hammond Collapse << |
| NCT00490971 | - | - | Completed | - | - |
| NCT01029769 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Collapse << | Not Applicable | Completed | - | Germany ... more >> Psychiatrische Klinik und Poliklinik fuer Psychiatrie und Psychotherapie der Technischen Universitaet Muenchen am Klinikum rechts der Isar Munic, Bavaria, Germany, 81675 Collapse << |
| NCT00981526 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> University of Massachusetts Medical School Worcester, Massachusetts, United States, 01605 Collapse << |
| NCT01495741 | - | - | Completed | - | - |
| NCT03568500 | Schizophrenia ... more >> Schizoaffective Disorder First Episode Psychosis Collapse << | Phase 4 | Recruiting | August 2019 | United Kingdom ... more >> Surrey and Borders Partnership NHS Foundation Trust Recruiting Chertsey, United Kingdom, KT160AE South London and Maudsley NHS Foundation Trust Recruiting London, United Kingdom, SE59RS Northumberland Tyne and Wear NHS Foundation Trust Recruiting Newcastle Upon Tyne, United Kingdom, SR20NB Oxford Health NHS Foundation Trust Recruiting Oxford, United Kingdom, OX42GX Southern Health NHS Foundation Trust Recruiting Southampton, United Kingdom, SO140YG Collapse << |
| NCT01607762 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Merksem, Belgium Collapse << |
| NCT00237913 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00150215 | Schizophrenia | Phase 1 | Completed | - | United States, Texas ... more >> Pfizer Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00981526 | - | - | Completed | - | - |
| NCT00515723 | Schizophrenia ... more >> Schizoaffective Disorder Type 2 Diabetes Mellitus Hyperglycemia Collapse << | Phase 4 | Completed | - | United States, Missouri ... more >> Washington Univeristy School of Medicine St. Louis, Missouri, United States, 63110 Washington University School of Medicine, Psychiatry Dept. St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00534183 | Metabolic Syndrome ... more >> Diabetes Obesity Collapse << | Not Applicable | Completed | - | India ... more >> Central Institute of Psychiatry Ranchi, India, 834009 Collapse << |
| NCT00895921 | Diabetes | Phase 4 | Completed | - | United States, California ... more >> VA San Diego Healthcare System San Diego, California, United States, 92161 Collapse << |
| NCT00183625 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> West Los Angeles Veterans Healthcare Center Los Angeles, California, United States, 90073 United States, New Hampshire Greater Manchester Mental Health Center Manchester, New Hampshire, United States, 03104 Collapse << |
| NCT00351936 | Schizophrenia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Freedom Trail Clinic Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00183625 | - | - | Completed | - | - |
| NCT00351936 | - | - | Completed | - | - |
| NCT00048828 | Schizophrenia | Phase 4 | Completed | - | United States, New York ... more >> Bronx Children's Psychiatric Center Bronx, New York, United States, 10461 Sagamore Children's Psychiatric Center Dix Hills, New York, United States, 11746 Long Island Jewish Medical Center Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00337662 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT00391222 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT01446328 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 4 | Completed | - | Austria ... more >> Medizinische Universität Innsbruck Innsbruck, Austria, 6020 Norway Haukeland University Hospital Bergen, Norway, 5223 Stavanger University Hospital Stavanger, Norway St. Olavs Hospital Trondheim, Norway Collapse << |
| NCT00149292 | Schizophrenia | Phase 2 | Completed | - | Russian Federation ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Village Nikolskoe, Russian Federation Collapse << |
| NCT00391222 | - | - | Completed | - | - |
| NCT01903837 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00159770 | Schizophrenia ... more >> Schizoaffective Disorder Psychotic Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT02161718 | Schizophrenia ... more >> Alcohol Use Disorder Collapse << | Phase 2 | Completed | - | - |
| NCT01207414 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT00419653 | Schizophrenia | Phase 4 | Terminated(Recruitment issues) | - | - |
| NCT00000374 | Schizophrenia | Phase 4 | Completed | - | United States, New York ... more >> Bronx-Lebanon Hospital Center Bronx, New York, United States, 10456 Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |
| NCT01139463 | - | - | Completed | - | Croatia ... more >> Split Clinical Hospital Center Split, Croatia, 21000 Collapse << |
| NCT00337662 | - | - | Completed | - | - |
| NCT00926965 | Tardive Dyskinesia ... more >> Neurocognitive Function Collapse << | Phase 4 | Completed | - | Taiwan ... more >> Yu-Li Veternas Hospital Hualien, Taiwan, 981 Collapse << |
| NCT00349973 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Collapse << | Not Applicable | Completed | - | United States, Maryland ... more >> Maryland Psychiatric Research Center Baltimore, Maryland, United States, 21228 Collapse << |
| NCT00078039 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT01185743 | Schizophrenia ... more >> Diabetes Collapse << | Phase 4 | Completed | - | Austria ... more >> Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT00259870 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT01207414 | - | - | Completed | - | - |
| NCT00188942 | Major Depressive Disorder | Phase 4 | Completed | - | Canada, Ontario ... more >> University Health Network - Toronto General Division Toronto, Ontario, Canada, M5G2C4 University Health Network - Toronto Western Division Toronto, Ontario, Canada, M5T2S8 Collapse << |
| NCT00169702 | Schizophrenia | Not Applicable | Completed | - | Germany ... more >> Heinrich-Heine-University, Rhineland State Clinics Düsseldorf Düsseldorf, Rhineland State, Germany, 40629 Collapse << |
| NCT00056472 | Major Depressive Disorder With... more >> Psychotic Features Collapse << | Phase 3 | Completed | - | United States, Massachusetts ... more >> University of Massachusetts Medical School Worcester, Massachusetts, United States, 01605 United States, New York Cornell University New York, New York, United States, 10021 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 Canada, Ontario University of Toronto Toronto, Ontario, Canada, M5G 2C4 Collapse << |
| NCT00056472 | - | - | Completed | - | - |
| NCT00083668 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00686998 | Schizophrenia | Phase 2 | Completed | - | United States, Maryland ... more >> Research Site Rockville, Maryland, United States Collapse << |
| NCT01893229 | Bipolar Disorder | Phase 4 | Unknown | December 2015 | China, Guangdong ... more >> Guangzhou Psychiatric Hospital Enrolling by invitation Guangzhou, Guangdong, China, 510370 Guangzhou Psychiatric Hospital Recruiting Guangzhou, Guangdong, China, 510370 Contact: Guiyun Xu, M.D xuguiyun2908@hotmail.com Principal Investigator: Guiyun Xu, M.D Collapse << |
| NCT00349973 | - | - | Completed | - | - |
| NCT00212771 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT01043250 | - | - | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT00827918 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00827918 | - | - | Completed | - | - |
| NCT00686998 | - | - | Completed | - | - |
| NCT00191555 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT00212784 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT02087579 | Psychotic Disorders ... more >> Schizophrenia Bipolar Disorder Depressive Disorder Collapse << | Phase 1 | Completed | - | United States, California ... more >> Garden Grove, California, United States United States, Florida Kissimmee, Florida, United States United States, Georgia Atlanta, Georgia, United States United States, Illinois Hoffman Estates, Illinois, United States United States, New York Cedarhurst, New York, United States United States, Texas Austin, Texas, United States Argentina Banfield, Argentina Ciudad Autónoma De Buenos Aires, Argentina Cordoba, Argentina La Plata, Argentina Belgium Aalst, Belgium Antwerpen, Belgium Brussel, Belgium Kortenberg, Belgium Brazil Rio De Janeiro, Brazil Valinhos, Brazil Bulgaria Bourgas, Bulgaria Germany Berlin, Germany Hamburg, Germany Luebeck, Germany Spain Badajoz, Spain Barcelona, Spain Torrevieja, Spain Zamora, Spain Collapse << |
| NCT00053703 | Schizophrenia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Cambridge Health Alliance Medford, Massachusetts, United States, 02155 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27514 United States, Ohio University Hospitals of Cleveland Cleveland, Ohio, United States, 44106 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01420575 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Not Applicable | Terminated(Low enrollment rate... more >>.) Collapse << | - | United States, Nebraska ... more >> Omaha Veterans Affairs Medical Center Omaha, Nebraska, United States, 68105 Collapse << |
| NCT00053703 | - | - | Completed | - | - |
| NCT00006195 | Schizophrenia | Not Applicable | Unknown | - | United States, Missouri ... more >> Washington University Recruiting St. Louis, Missouri, United States, 63110 Contact: John Newcomer, M.D. Collapse << |
| NCT01164059 | Schizophrenia | Phase 4 | Completed | - | Germany ... more >> Universitätsklinikum Aachen Aachen, Germany Krankenhaus Angermünde Angermünde, Germany Karl-Jaspers-Klinik Bad Zwischenahn, Germany Charité - Universitätsmedizin Berlin Berlin, Germany Vivantes Klinikum Neukölln Berlin, Germany LWL-Universitätsklinik Bochum der Ruhr-Universität Bochum, Germany Klinikum Bremen-Ost gGmbH Bremen, Germany Rheinische Kliniken Düsseldorf der Heinrich-Heine-Universität Düsseldorf, Germany Städtisches Krankenhaus Eisenhüttenstadt GmbH Eisenhüttenstadt, Germany Universitätsmedizin Göttingen Göttingen, Germany Universitätsklinikum Hamburg-Eppendorf Hamburg, Germany Medizinische Hochschule Hannover Hannover, Germany Privat-Nerven-Klinik Dr. med. Kurt Fontheim Liebenburg, Germany Dietrich-Bonhoeffer-Klinik Neubrandenburg Neubrandenburg, Germany Ruppiner Kliniken Neuruppin, Germany Ernst von Bergmann Klinikum Potsdam, Germany Immanuel Klinik Rüdersdorf Rüdersdorf, Germany Klinik Taufkirchen Taufkirchen, Germany Collapse << |
| NCT00363181 | - | - | Unknown | - | United States, California ... more >> VA Palo Alto Health Care System Recruiting Palo Alto, California, United States, 94304-1290 Principal Investigator: Steven E Lindley, MD, PhD Collapse << |
| NCT00182013 | Bipolar Spectrum Disorder | Phase 4 | Completed | - | - |
| NCT00182442 | Schizophrenia | Phase 4 | Completed | - | Canada, Ontario ... more >> McMaster University Hamilton, Ontario, Canada Collapse << |
| NCT00290121 | Schizophrenia | Phase 4 | Completed | - | Canada, Quebec ... more >> Centre de recherche Fernand-Seguin Montréal, Quebec, Canada, H1N 3V2 Collapse << |
| NCT00770744 | Schizophrenia | Phase 2 | Completed | - | Czech Republic ... more >> CZ001 Ceske Budejovice, Czech Republic, 37087 CZ005 Litomerice, Czech Republic, 41201 CZ004 Lnare, Czech Republic, 38742 CZ002 Olomouc, Czech Republic, 77111 CZ003 Olomouc, Czech Republic, 77520 CZ006 Praha 8, Czech Republic, 18100 France FR001 Clermont-Ferrand, Cedex 1, France, 63003 FR002 Dole, France, 39100 FR003 Jonzac, France, 17503 Hong Kong HK001 Hong Kong, Hong Kong Indonesia ID001 Bangli, Indonesia, 80613 ID002 Jakarta, Indonesia, 10430 Philippines PH002 Baguio, Philippines, 2600 PH001 Mandaluyong City, Philippines, 1553 Poland PL003 Gdansk, Poland, 80-211 PL002 Lodz, Poland, 92-216 Spain ES001 Barcelona, Spain, 8025 ES002 Salamanca, Spain, 37003 ES004 Zamora, Spain, 49021 Thailand TH001 Bangkok, Thailand, 10330 TH002 Chiang Mai, Thailand, 50200 Collapse << |
| NCT00690235 | Schizophrenia ... more >> Schizoaffective Disorder Diabetes Weight Gain Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> The University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Collapse << |
| NCT00690235 | - | - | Completed | - | - |
| NCT00211562 | Schizophrenia | Phase 3 | Terminated | - | United States, Georgia ... more >> VA Medical Center Augusta, Georgia, United States, 30904 Collapse << |
| NCT00235352 | Schizophrenia | Phase 2 Phase 3 | Completed | - | United States, Massachusetts ... more >> Freedom Trail Clinic Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03730857 | Schizophrenia | Phase 1 | Completed | - | - |
| NCT00312598 | - | - | Completed | - | - |
| NCT00044187 | Schizophrenia ... more >> Psychotic Disorders Bipolar Disorder Collapse << | Phase 4 | Completed | - | United States, California ... more >> El Centro, California, United States United States, Florida Jacksonville, Florida, United States Miami, Florida, United States United States, Idaho Boise, Idaho, United States United States, Illinois Chicago, Illinois, United States United States, Indiana Indianapolis, Indiana, United States Lafayette, Indiana, United States United States, Kansas Prairie Village, Kansas, United States United States, Massachusetts Milford, Massachusetts, United States United States, New Jersey Clementon, New Jersey, United States United States, Ohio Beachwood, Ohio, United States United States, Tennessee Johnson City, Tennessee, United States Memphis, Tennessee, United States United States, Texas Houston, Texas, United States United States, Virginia Richmond, Virginia, United States United States, Washington Bellevue, Washington, United States Collapse << |
| NCT00485823 | Schizophrenic Disorders | Phase 4 | Completed | - | Korea, Republic of ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of Collapse << |
| NCT00191828 | Weight Gain | Phase 4 | Completed | - | Italy ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri 9AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speack with your personal physician. Sesto Fiorentino, Florence, Italy Collapse << |
| NCT00350467 | Acute Schizophrenia | Phase 3 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China China, Guangdong Guangzhou, Guangdong, China China, Hubei Wuhan, Hubei, China China, Jiangsu Nanjing, Jiangsu, China China, Shanghai Shanghai, Shanghai, China China, Zhejiang Suzhou, Zhejiang, China China Xian, China Collapse << |
| NCT00176436 | Schizophrenia | Phase 4 | Completed | - | United States, Maryland ... more >> Maryland Psychiatric Research Center Catonsville, Maryland, United States, 21228 Collapse << |
| NCT00034892 | Schizophrenia ... more >> Psychotic Disorders Mental Health Mental Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT00176436 | - | - | Completed | - | - |
| NCT01234454 | Schizophrenia | Not Applicable | Completed | - | United States, North Carolina ... more >> Unc Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00216632 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 4 | Completed | - | - |
| NCT01088386 | - | - | Enrolling by invitation | May 2021 | United States, Pennsylvania ... more >> United BioSource Corporation Blue Bell, Pennsylvania, United States, 19422 Collapse << |
| NCT00716755 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Delusional Disorder Psychotic Disorder Collapse << | Not Applicable | Completed | - | Canada, Ontario ... more >> Centre for Addiction and Mental Health Toronto, Ontario, Canada, M5T 1R8 Collapse << |
| NCT00378183 | Schizoaffective Disorder ... more >> Schizophrenia Collapse << | Phase 4 | Completed | - | - |
| NCT00845507 | Weight Gain | Phase 4 | Completed | - | United States, Ohio ... more >> University of Cincinnati Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT00223418 | Schizophenia Disorder ... more >> Schizoaffective Disorder Collapse << | Not Applicable | Completed | - | United States, Texas ... more >> The University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00236457 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT00094640 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 1 | Completed | - | Australia, South Australia ... more >> For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Adelaide, South Australia, Australia Belgium For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Kortenberg, Belgium Brazil For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Rio De Janeiro, RJ, Brazil Croatia For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Zagreb, Croatia Czech Republic For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Praha 8, Czech Republic Mexico For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Mexico City, Tlalpan, Mexico Slovakia For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Bratislava, Slovakia Spain For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY (1-877-285-4559) or speak with your personal physician. Barcelona, Spain Collapse << |
| NCT00225498 | Schizophrenia | Not Applicable | Completed | - | United States, New York ... more >> The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00225498 | - | - | Completed | - | - |
| NCT01539733 | Delirium Adva... more >>nced Cancer Collapse << | Not Applicable | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Netherlands, 1081HV Spaarne Hospital Hoofddorp, Netherlands Collapse << |
| NCT00034801 | Depression Sc... more >>hizophrenia Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT00845507 | - | - | Completed | - | - |
| NCT01686815 | - | - | Unknown | December 2015 | United States, Massachusetts ... more >> Freedom Trail Clinic Recruiting Boston, Massachusetts, United States, 02114 Contact: Liana J Petruzzi, BA 617-912-7882 lpetruzzi@partners.org Contact: Leah N Briggs, BS 617-912-7848 lbriggs@partners.org Principal Investigator: David C Henderson, MD Collapse << |
| NCT00036088 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT00205699 | - | - | Completed | - | - |
| NCT00156091 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00034580 | Bipolar Disorder | Phase 4 | Completed | - | - |
| NCT01593774 | Schizophrenia | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Shafa Psychiatric Hospital Rasht, Guilan, Iran, Islamic Republic of, 55599-41939 Collapse << |
| NCT01617187 | - | - | Completed | - | - |
| NCT00205699 | Aggression At... more >>tention Deficit-Hyperactivity Oppositional Defiant Disorder Pervasive Development Disorders Bipolar Disorder Collapse << | Phase 4 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine, Psychiatry Dept. Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00351000 | Schizophrenia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Freedom Trail Clinic Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00428168 | Weight Gain | Phase 2 | Terminated(Interim Analysis re... more >>sult indicated the study will not show a significant benefit of the study medication on the primary endpoint.) Collapse << | - | Canada, Alberta ... more >> Capital Health, Edmonton Mental Health Clinic Edmonton, Alberta, Canada, T5K 2J5 Canada, British Columbia Dr. Alexander McIntyre Penticton, British Columbia, Canada, V2A 4M4 Vancouver Island Health Authority Victoria, British Columbia, Canada, V8R 4Z3 Canada, Manitoba Dr. Ivan Kowalchuk Winnipeg, Manitoba, Canada, R3K 2E2 Canada, Nova Scotia Capital District Health Authority Halifax, Nova Scotia, Canada, B3H 2E2 Canada, Ontario Queen's University Kingston, Ontario, Canada, K7L 5G2 Canada, Quebec Douglas Hospital Research Centre Verdun (Montreal), Quebec, Canada, H4H 1R3 Israel Abarbanel Hospital Bat Yam, Israel Geha Psychiatric Hospital Petach Tikva, Israel, 49100 Lev Hasharon Tirat Hacarmel, Israel Collapse << |
| NCT00351000 | - | - | Completed | - | - |
| NCT01389908 | Schizophrenia ... more >> Schizoaffective Disorders Collapse << | Not Applicable | Unknown | - | Israel ... more >> Shalvata MHC Recruiting Hod hasharon, Israel Contact: Ziv Carmel, Dr. 972-9-7478403 zivca@clalit.org.il Principal Investigator: Ziv Carmel, Dr. Collapse << |
| NCT00221481 | Bipolar Disorder | Phase 4 | Withdrawn(Lack of recruitment) | - | United States, California ... more >> UCLA Neuropsychiatric Institute Los Angeles, California, United States, 90095 Collapse << |
| NCT00844857 | Bipolar Depression | Phase 4 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Diego, California, United States, 92123 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lipetsk, Russian Federation, 399313 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 123367 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saratov, Russian Federation, 410060 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Stavropol, Russian Federation, 355038 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tver, Russian Federation, 170005 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yaroslavl, Russian Federation, 150003 Collapse << |
| NCT00682448 | Metabolic Complications | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT00958568 | Treatment Resistant Depression | Phase 3 | Completed | - | - |
| NCT00071253 | Bipolar Disorder | Phase 4 | Terminated | - | United States, California ... more >> Behavioral and Medical Research, LLC Anaheim, California, United States, 92805 Synergy Clinical Research Chula Vista, California, United States, 91910 United States, Florida Clinical Trial Management Fort Meyers, Florida, United States, 33907 Segal Institute for Clinical Research North Miami, Florida, United States, 33161 United States, Illinois Rush Presbyterian - St. Luke's Chicago, Illinois, United States, 60612 United States, Kentucky University of Louisville Outpatient Psychiatry Louisville, Kentucky, United States, 40202 United States, Mississippi University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 United States, Nebraska Creighton University Department of Psychiatry Omaha, Nebraska, United States, 68131 United States, Nevada Lake Mead Hospital North Las Vegas, Nevada, United States, 89030 United States, New York NYU School of Medicine New York City, New York, United States, 10016 United States, Ohio University Hospital of Cleveland Cleveland, Ohio, United States, 44106 R. Ranjan, MD & Associates, Inc. Lyndhurst, Ohio, United States, 44124 United States, Oklahoma IPS Research Oklahoma City, Oklahoma, United States, 73103 United States, Texas UTMB Dept. of Psychiatry Galveston, Texas, United States, 77555-0197 United States, Wisconsin Zablocki VAMC Milwaukee, Wisconsin, United States, 53295 Collapse << |
| NCT00394095 | Bipolar Disorder ... more >> Weight Gain Collapse << | Phase 4 | Completed | - | United States, Ohio ... more >> University of Cincinnati Medical Center Cincinnati, Ohio, United States, 45267-0559 Collapse << |
| NCT00844857 | - | - | Completed | - | - |
| NCT00394095 | - | - | Completed | - | - |
| NCT00958568 | - | - | Completed | - | - |
| NCT01541371 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00873795 | Major Depressive Disorder | Not Applicable | Completed | - | Taiwan ... more >> Psychiatry Department, Chimei Medical Center Tainan, Taiwan, 700 Collapse << |
| NCT01541371 | - | - | Completed | - | - |
| NCT00616798 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00509067 | Schizophrenia | Phase 2 | Completed | - | United States, District of Col... more >>umbia Washington Veterans Affairs Medical Center Washington, District of Columbia, United States, 20422 Collapse << |
| NCT03510325 | Schizophrenia | Phase 4 | Not yet recruiting | September 1, 2021 | China, Shanghai ... more >> Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine Shanghai, Shanghai, China, 200030 Collapse << |
| NCT00857818 | Schizophrenia ... more >> Schizoaffective Disorder Bipolar I Disorder Metabolic Syndrome Collapse << | Phase 3 | Terminated(Slow Accrual) | - | Canada, Alberta ... more >> Local Institution Calgary, Alberta, Canada, T2N 2T9 Canada, British Columbia Local Institution Pentincton, British Columbia, Canada, V2A 4M4 Local Institution Vancouver, British Columbia, Canada, V6T 2A1 Canada, Ontario Local Institution Hamilton, Ontario, Canada, L8N 3K7 Local Institution London, Ontario, Canada, N6H 4V1 Local Institution Markham, Ontario, Canada, L6B 1A1 Local Institution Mississauga, Ontario, Canada, L5M 4N4 Local Institution Toronto, Ontario, Canada, M5T 1R8 Local Institution Toronto, Ontario, Canada, M5T 2S8 Canada, Quebec Local Institution Montreal, Quebec, Canada, H1N 3M5 Local Institution Montreal, Quebec, Canada, H3A 1A1 Local Institution Montreal, Quebec, Canada, H3M 3A9 Local Institution Montreal, Quebec, Canada, H4H 1R3 Canada Local Institution Quebec, Canada, G1R 2W8 Collapse << |
| NCT00857818 | - | - | Terminated(Slow Accrual) | - | - |
| NCT02640911 | - | - | Recruiting | May 2021 | China, Shanghai ... more >> Shanghai Mental Health Center Recruiting Shanghai, Shanghai, China, 200030 Contact: Huafang Li, PH.D 86-2134773128 lhlh_5@163.com Collapse << |
| NCT02439099 | - | - | Recruiting | December 2018 | Germany ... more >> Department of Psychiatry, Charité - Campus Benjamin Franklin Recruiting Berlin, Germany, 12203 Collapse << |
| NCT00246246 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT00509067 | - | - | Completed | - | - |
| NCT01556763 | - | - | Completed | - | - |
| NCT02455310 | - | - | Active, not recruiting | October 2018 | United States, Iowa ... more >> The University of Iowa College of Public Health Iowa City, Iowa, United States, 52246 Collapse << |
| NCT01151865 | Delirium | Phase 2 Phase 3 | Completed | - | Australia, New South Wales ... more >> Royal North Shore Hospital St Leonards, New South Wales, Australia, 2065 Australia, Queensland Toowoomba Hospital Toowoomba, Queensland, Australia, 4350 Australia, Victoria Northern Hospital Epping, Victoria, Australia, 3076 The Western Hospital Footscray, Victoria, Australia, 3011 Austin Hospital Melbourne, Victoria, Australia, 3084 The Alfred Hospital Prahran, Victoria, Australia, 3181 Australia, Western Australia Royal Perth Hospital Perth, Western Australia, Australia, 6001 Collapse << |
| NCT01323205 | Schizophrenia | Phase 2 | Completed | - | Austria ... more >> Innsbruck, Austria Salzburg, Austria Wien, Austria Belgium Dave, Belgium Duffel, Belgium Kortenberg, Belgium Lede, Belgium Bulgaria Plovdiv, Bulgaria Radnevo N/A, Bulgaria Germany Berlin, Germany Hamburg, Germany Mainz, Germany Mannheim, Germany München, Germany Romania Arad, Romania Brasov, Romania Iasi, Romania Sibiu, Romania Spain Barcelona, Spain Zamora, Spain Collapse << |
| NCT00177567 | Bipolar Disorder | Phase 4 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 University of Pittsubrgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01556763 | Schizophrenia ... more >> Schizoaffective Disorder Central Nervous System Diseases Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Clinical Research Institute Wichita, Kansas, United States, 67211 Collapse << |
| NCT01305707 | Depression | Phase 4 | Terminated(Difficulties in rec... more >>ruiting) Collapse << | - | Spain ... more >> Hospital Universitari de Bellvitge Hospitalet de Llobregat, Barcelona, Spain, 08907 Corporació Sanitària Parc Tauli Sabadell, Barcelona, Spain, 08208 Hospital Universitari de Bellvitge, IDIBELL Barcelona, Spain, 08907 Collapse << |
| NCT03565068 | Schizophrenia | Phase 1 | Suspended(Study is on hold pen... more >>ding evaluation of emerging preclinical findings) Collapse << | October 9, 2019 | United States, California ... more >> California Clinical Trials ( Site 0001) Glendale, California, United States, 91206 Collapse << |
| NCT00854919 | SSRI-Refractory Obsessive-Comp... more >>ulsive Disorder Collapse << | Phase 4 | Completed | - | Japan ... more >> Dept of Neuropsychiatry, Osaka City University, graduate School of Medicine Osaka, Japan, 545-8585 Collapse << |
| NCT02600741 | - | - | Completed | - | - |
| NCT02374567 | Dementia Depr... more >>ession Schizophrenia Psychosomatic Disorders Anxiety Disorders Collapse << | Phase 3 | Terminated | - | Germany ... more >> Bezirkskrankenhaus Augsburg Augsburg, Germany Krankenhaus Hedwigshöhe Berlin, Germany Hannover Medical School Hannover, Germany, 30625 Asklepios Fachklinikum Lübben Lübben, Germany Asklepios Fachklinikum Teupitz Teupitz, Germany Collapse << |
| NCT02307396 | Schizophrenia ... more >> Schizophrenia and Disorders With Psychotic Features Schizoaffective Disorders Collapse << | Phase 4 | Completed | - | Germany ... more >> Psychiatrische Klinik und Poliklinik fuer Psychiatrie und Psychotherapie der Technischen Universitaet Muenchen am Klinikum rechts der Isar Munich, Bavaria, Germany, 81675 Collapse << |
| NCT00433121 | Dementia | Phase 4 | Completed | - | Norway ... more >> Sykehuset Innlandet HF - Sanderud Ottestad, Oppland, Norway, 2312 Collapse << |
| NCT02248987 | - | - | Unknown | - | Korea, Republic of ... more >> Seoul National University Hospital Recruiting Seoul, Korea, Republic of Contact: Jin Woo Park, MD 82-2-3668-7666 parkjinu78@naver.com Contact: Jun Soo Kwon, MD, PhD 82-2-2072-2450 Principal Investigator: Jun Soo Kwon, MD, PhD Sub-Investigator: Euitae Kim, MD, PhD Collapse << |
| NCT00048802 | Bipolar Disorder | Phase 4 | Completed | - | United States, New York ... more >> The Zucker Hillside Hospital, Long Island Jewish Medical Center Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00664976 | Bipolar Disorder | Not Applicable | Completed | - | Norway ... more >> Haukeland University Hospital Bergen, Norway, 5000 St Olavs Hospital, Østmarka sykehus Trondheim, Norway Collapse << |
| NCT00347425 | Schizophrenia and Schizoaffect... more >>ive Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT02431702 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 3 | Active, not recruiting | October 26, 2019 | - |
| NCT03666715 | - | - | Recruiting | August 15, 2019 | Portugal ... more >> Neurobios , Instituto De Neurociências Recruiting Porto, Portugal, Portugal, 4200 - 117 Collapse << |
| NCT02181803 | Schizophrenia | Phase 1 | Completed | - | - |
| NCT00667745 | - | - | Completed | - | - |
| NCT00845988 | Bipolar Disorders ... more >> Metabolic Complication Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-707 Collapse << |
| NCT00399776 | - | - | Completed | - | United States, Nebraska ... more >> Creighton University Psychiatry Research Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00667745 | Bipolar Disorder | Phase 4 | Completed | - | United States, California ... more >> Stanford University Stanford, California, United States, 94035-5723 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 United States, Texas University of Texas Health Science Center San Antonio, Texas, United States, 78229 Collapse << |
| NCT00534222 | - | - | Completed | - | Netherlands ... more >> Research Site Alkmaar, Netherlands Research Site Almelo, Netherlands Research Site Alphen aan de Rigjn, Netherlands Research Site Amersfoort, Netherlands Research Site Amstelveen, Netherlands Research Site Amsterdam, Netherlands Collapse << |
| NCT00272025 | Bipolar Depression | Phase 1 | Terminated(Lundbeck withdrew c... more >>ommittment - expiring patents and prolonged inactivity) Collapse << | - | Canada, Ontario ... more >> Providence Care, Mental Health Services Kingston, Ontario, Canada, K7L 4X3 Collapse << |
| NCT00704860 | Major Depression | Phase 4 | Completed | - | Canada, Ontario ... more >> University of Ottawa Institute of Mental Health Research Ottawa, Ontario, Canada, K1Z 7K4 Collapse << |
| NCT01231074 | Obesity Weigh... more >>t Gain Psychotropic Induced Weight Gain Collapse << | Phase 1 | Unknown | May 2011 | United States, Ohio ... more >> Nationwide Children's Hospital Recruiting Columbus, Ohio, United States, 43205 Contact: Ihuoma Eneli, MD, MS 614-722-4089 ihuoma.eneli@nationwidechildrens.org Contact: Mary Ann Murphy, MD, PhD 614-722-2291 maryann.murphy@nationwidechildrens.org Collapse << |
| NCT02958800 | Dementia Beha... more >>vior Collapse << | Not Applicable | Recruiting | December 2018 | Canada, Ontario ... more >> Blackadar Continuing Care Centre Recruiting Dundas, Ontario, Canada, L9H 3B9 Contact: Henry Siu, MSc, MD, CCFP (COE) (905) 575-1300 siuh3@mcmaster.ca Contact: Jenna Parascandalo 9055259140 ext 21232 parascaj@mcmaster.ca Collapse << |
| NCT00330863 | - | - | Completed | - | - |
| NCT00149734 | Schizophrenia | Phase 3 | Completed | - | United States, Colorado ... more >> Denver VAMC Denver, Colorado, United States, 80220 Collapse << |
| NCT02435095 | Schizophrenia | Phase 4 | Recruiting | April 2019 | Germany ... more >> RWTH University Hospital Aachen Recruiting Aachen, NRW, Germany, 52074 Contact: Sabrina Schaffrath saschaffrath@ukaachen.de Contact: Sarah Lammertz slammertz@ukaachen.de Alexianer Aachen GmbH Not yet recruiting Aachen, Germany, 52062 Zentrum für Neurologie und Seelische Gesundheit im Kapuziner Karree Aachen Not yet recruiting Aachen, Germany, 52062 Contact: Frank Bergmann, Dr. med. LVR Klinik Bonn Recruiting Bonn, Germany, 53111 LVR Klinik Düren Not yet recruiting Düren, Germany, 52353 Klinik und Poliklinik für Psychiatrie und Psychotherapie der Heinrich-Heine-Universität Düsseldorf Recruiting Düsseldorf, Germany, 40629 LVR Klinik Essen Not yet recruiting Essen, Germany, 45147 ViaNobis Gangelt Recruiting Gangelt, Germany, 52538 Klinik Königshof (Abteilung für Allgemeine Psychiatrie) Recruiting Krefeld, Germany, 47807 LVR Klinik Langenfeld Recruiting Langenfeld, Germany, 40764 LVR Klinik Viersen Not yet recruiting Viersen, Germany, 41749 Collapse << |
| NCT01000402 | Bipolar Disorder ... more >> Attention Deficit Disorder With Hyperactivity Collapse << | Phase 4 | Unknown | November 2014 | Brazil ... more >> Hospital de Clinicas de Porto Alegre Recruiting Porto Alegre, RS, Brazil, 90350-903 Contact: Clarissa F Paim +55 51 21018094 cfpaim@hcpa.ufrgs.br Contact: Silza Tramontina, D.Sc. +55 51 21018094 silza.tramontina@terra.com.br Principal Investigator: Luis A Rohde, D.Sc. Sub-Investigator: Silza Tramontina, D.Sc. Sub-Investigator: Cristian P Zeni, M.Sc. Sub-Investigator: Carla R Ketzer, M.Sc. Sub-Investigator: Gabriel F Pheula, MD Sub-Investigator: Fernanda V Krieger, MD Sub-Investigator: Sandra Petresco, M.Sc. Sub-Investigator: Roberta S Coelho, B.Sc. Sub-Investigator: Adriana F Perez, M.Sc. Sub-Investigator: Lanuzia Brum, BA Sub-Investigator: Ana P Guimaraes, M.Sc. Sub-Investigator: Henrique T Ludwig Collapse << |
| NCT00448630 | - | - | Completed | - | Turkey ... more >> Pfizer Investigational Site Kurttepe, Adana, Turkey, 01360 Pfizer Investigational Site Adana, Turkey Pfizer Investigational Site Ankara, Turkey, 06200 Pfizer Investigational Site Ankara, Turkey Pfizer Investigational Site Antakya, Turkey Pfizer Investigational Site Bolu, Turkey Pfizer Investigational Site Denizli, Turkey Pfizer Investigational Site Elazig, Turkey Pfizer Investigational Site Eskisehir, Turkey Pfizer Investigational Site Istanbul, Turkey, 34147 Pfizer Investigational Site Istanbul, Turkey Pfizer Investigational Site Izmir, Turkey Pfizer Investigational Site Kayseri, Turkey Pfizer Investigational Site Konya, Turkey Pfizer Investigational Site Manisa, Turkey Pfizer Investigational Site Samsun, Turkey Pfizer Investigational Site Trabzon, Turkey Pfizer Investigational Site Trakya, Turkey Collapse << |
| NCT00330863 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | United States, California ... more >> University of California, Los Angeles Los Angeles, California, United States, 91344 United States, Georgia Medical College of Georgia, Department of Psychiatry Augusta, Georgia, United States, 30912-3800 United States, Iowa University of Iowa College of Medicine, Psychiatry Research Iowa City, Iowa, United States, 52242 United States, Massachusetts Harvard Medical School -- Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Harvard Medical School -- Dr. John C. Corrigan Community Mental Health Center Fall River, Massachusetts, United States, 02720 United States, Nebraska Creighton University Omaha, Nebraska, United States, 68131 United States, New Mexico University of New Mexico Albuquerque, New Mexico, United States, 87131 United States, New York The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00061789 | Schizophrenia | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Mental Health (NIMH) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02051387 | Schizophrenia | Phase 1 | Completed | - | Germany ... more >> Central Institute of Mental Health Mannheim, BW, Germany, 68159 Dept. of Pharmacology, University of Cologne Cologne, NRW, Germany, 50931 Collapse << |
| NCT00563992 | Bipolar Disorder | Phase 1 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute/Columbia University New York, New York, United States, 10032 Collapse << |
| NCT00189995 | Schizophrenia | Phase 3 | Withdrawn | - | Israel ... more >> Beersheva Mental Health Center Beersheva, Israel Nes Ziona Medical Center Nes Ziona, Israel Collapse << |
| NCT02893371 | - | - | Enrolling by invitation | June 2019 | United States, New Mexico ... more >> Christophe G Lambert Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT00448630 | - | - | Completed | - | - |
| NCT00759993 | Weight Gain O... more >>besity Collapse << | Phase 2 | Terminated(inability to recrui... more >>t and retain) Collapse << | March 2013 | - |
| NCT01003379 | Cognitive Dysfunction ... more >> Schizophrenia Collapse << | Phase 2 | Completed | - | United States, California ... more >> Synergy Clinical Research Center National City, California, United States, 91950-7628 Collaborative Neuroscience Network, Inc LA County Torrance, California, United States, 90502 United States, Georgia Altanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Kansas Clinical Research Institute Wichita, Kansas, United States, 67211 United States, New York Neurobehavioral Reseach, Inc. Cedarhurst, New York, United States, 11516 New York State Pshychiatric Institute, Columbia University New York, New York, United States, 10032 United States, Texas Community Clinical Research, Inc. Austin, Texas, United States, 78754 India Sravani Polyclinic and Mental Health Care Centre Guntur, Andhra Pradesh, India, 522001 Asha Hospital Hyderabaad, Andhra Pradesh, India, 500034 Dept. of Psychiatry, Owaisi Hospital & Research Centre Hyderabaad, Andhra Pradesh, India, 500058 Brain Mind Behaviour Neuroscience Research Institute Visakhapatnam, Andhra Pradesh, India, 530002 Government Hospital for Mental Care Visakhapatnam, Andhra Pradesh, India, 530017 Adhit Khan Neuropsychiatric Centre Mangalore, Karnataka, India, 575002 Dept. of Psychiatry, JSS University Mysore, Karnataka, India, 57004 Mahendru Psychiatric Centre Kanpur, Uttar Pradesh, India, 208005 Dept. of Psychiatry, CSM University Lucknow, Uttar Pradesh, India, 226003 Collapse << |
| NCT01157351 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT01193166 | Schizophrenia | Phase 4 | Withdrawn(This study was stopp... more >>ed due to an internal reconsideration of priorities of the product portfolio.) Collapse << | September 2012 | - |
| NCT00838032 | Acute Schizophrenia | Phase 4 | Unknown | May 2010 | China, Sichuan ... more >> Mental Health Center of Huaxi Hospital affiliated to Sichuan University Recruiting Chengdu, Sichuan, China, 610041 Contact: Bo Zhang, PhD 13808203275 zb_73@126.com Principal Investigator: Bo Zhang, PhD Collapse << |
| NCT02873208 | Schizophrenia | Phase 3 | Enrolling by invitation | September 2019 | - |
| NCT00642369 | - | - | Completed | - | - |
| NCT00642369 | Schizophrenia | Not Applicable | Completed | - | China, Guangdong ... more >> Guangdong Provincial Mental Health Institute Guang Zhou, Guangdong, China, 510120 Collapse << |
| NCT02048241 | Intermittent Explosive Disorde... more >>r Childhood Aggression Oppositional Defiant Disorder Attention Deficit Disorder Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT02048241 | - | - | Completed | - | - |
| NCT01280396 | - | - | Completed | - | Hong Kong ... more >> Western Psychiatric Centre Hong Kong, Hong Kong, 00000 Collapse << |
| NCT00211380 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Not Applicable | Terminated(Recruitment was too... more >> difficult) Collapse << | - | United States, Louisiana ... more >> Psychopharmacology Research Clinic Shreveport, Louisiana, United States, 71103 Collapse << |
| NCT00956189 | Metabolic Syndrome X | Not Applicable | Completed | - | Taiwan ... more >> Yu-Li Hospital Yu-Li, Hualien County, Taiwan, 981 Yu-Li Veterans Hospital Yu-Li, Hualien County, Taiwan, 981 National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT03237052 | Schizophrenia | Not Applicable | Not yet recruiting | December 2020 | - |
| NCT01157351 | - | - | Completed | - | - |
| NCT03557931 | Schizophrenia | Phase 2 | Recruiting | December 2019 | United States, Arkansas ... more >> Woodland Research Northwest, LLC Recruiting Rogers, Arkansas, United States, 72758 United States, California CNS Research Science, Inc. Recruiting Cerritos, California, United States, 90703 Collaborative Neuroscience Network, LLC Recruiting Garden Grove, California, United States, 92845 Synergy East Recruiting Lemon Grove, California, United States, 91945 Pacific Research Partners, LLC Recruiting Oakland, California, United States, 94607 California Neuropsychopharmacology Clinical Research Institute-LA, LLC Recruiting Pico Rivera, California, United States, 90660 Artemis Institute for Clinical Research Recruiting San Diego, California, United States, 92103 Sharp Mesa Vista Hospital Recruiting San Diego, California, United States, 92123 Collaborative Neuroscience Network, LLC Recruiting Torrance, California, United States, 90502 United States, Georgia Radiant Research, Inc. Recruiting Atlanta, Georgia, United States, 30328 Atlanta Center for Medical Research Recruiting Atlanta, Georgia, United States, 30331 United States, Illinois Alam Medical Research Inc. Recruiting Chicago, Illinois, United States, 60612 Uptown Research Institute Recruiting Chicago, Illinois, United States, 60640 United States, Michigan Michigan Clinical Research Institute PC Recruiting Ann Arbor, Michigan, United States, 48105 Cherry Street Services, Inc. Recruiting Grand Rapids, Michigan, United States, 49503 United States, New Jersey Hassman Research Institute Recruiting Berlin, New Jersey, United States, 08009 United States, New Mexico Albuquerque Neuroscience Inc. Recruiting Albuquerque, New Mexico, United States, 87109 United States, New York SPRI Clinical Trials, LLC Recruiting Brooklyn, New York, United States, 11235 CNS Research Science, Inc. Recruiting Jamaica, New York, United States, 11432 Manhattan Psychiatric Center's 125th Street Clinic Recruiting New York, New York, United States, 10035 United States, Ohio Midwest Clinical Research Center Recruiting Dayton, Ohio, United States, 45417 United States, Texas InSite Clinical Research, LLC Recruiting DeSoto, Texas, United States, 75115 Pillar Clinical Research, LLC Recruiting Garland, Texas, United States, 75042 Pillar Clinical Research, LLC Recruiting Richardson, Texas, United States, 75080 Collapse << |
| NCT01765829 | Psychosis Nos/Other | Phase 3 | Unknown | November 2015 | Spain ... more >> Mental Health Unit Villamartín Recruiting Villamartín, Cádiz, Spain, 11650 Contact: José María Mongil San Juan, MD 0034956040209 cheman45@gmail.com Principal Investigator: José María Mongil San Juan, MD Mental Health Unit Baza Recruiting Baza, Granada, Spain, 18800 Contact: Eva Bravo Barba, MD eva.bravo.barba@gmail.com Principal Investigator: Eva Bravo Barba, MD Mental Health Unit Motril Recruiting Motril, Granada, Spain, 18600 Contact: María Luisa Barrigón Estévez, MD 0034958038849 marial.barrigon.sspa@juntadeandalucia.es Principal Investigator: María Luisa Barrigón Estévez, MD Mental Health Unit Andújar Recruiting Andújar, Jaén, Spain, 23740 Contact: Fernando Sarramea Crespo, MD, PhD 0034953021717 fernandosarramea@hotmail.com Principal Investigator: Farnando Sarramea Crespo, MD, PhD Mental Health Unit Martos Recruiting Martos, Jaén, Spain, 23600 Contact: Miguel Ortigosa Luque, MD migueortigosa@hotmail.com Principal Investigator: Miguel Ortigosa Luque, MD Mental Health Unit Miraflores Not yet recruiting Alcobendas, Madrid, Spain, 28100 Contact: María Luisa Zamarro Arraz, MD, PhD 0034916530108 zamarroluisa@yahoo.es Principal Investigator: María Luisa Zamarro Arraz, MD, PhD Hospital Infanta Sofía Not yet recruiting San Sebastian de los Reyes, Madrid, Spain, 28702 Contact: María Rosario Gutiérrez Labrador, MD rosagutierrez@psiquiatra.e.telefonica.net Principal Investigator: María Rosario Guitiérrez Labrador, MD Mental Health Unit Valle del Guadalhorce Not yet recruiting Cártama, Málaga, Spain, 29580 Contact: Daniel Gutiérrez Castillo, MD 0034951033931 dgutierrezcastillo@gmail.com Principal Investigator: Daniel Gutiérrez Castillo, MD Mental Health Unit Las Lagunas Not yet recruiting Fuengiróla, Málaga, Spain, 29650 Contact: María Isabel Osuna Carmona, MD maribelosunac@hotmail.com Principal Investigator: María Isabel Osuna Carmona, MD Fundación Argibide Not yet recruiting Pamplona, Navarra, Spain, 31007 Contact: Ignacio Mata Pastor, MD, PhD 0034948266650 nacho.mata70@gmail.com Principal Investigator: Ignacio Mata Pastor, MD, PhD Hospital El Tomillar Recruiting Dos Hermanas, Seville, Spain, 41700 Contact: Joaquín Carlos Martín Muñoz, MD, PhD 0034955016083 joaquinmartin@hotmail.com Principal Investigator: Joaquín Carlos Martín Muñoz, MD, PhD Hospital Francesc de Borja Recruiting Gandía, Valencia, Spain, 46700 Contact: María Lacruz Silvestre, MD, PhD maria.lacruz.silvestre@gmail.com Principal Investigator: María Lacruz Silvestre, MD, PhD Hospital de Basurto Not yet recruiting Bilbao, Spain, 48013 Contact: Sonia Bustamante Madariaga, MD, Phd 0034944 006137 sonia.bustamantemadariaga@osakidetza.net Principal Investigator: Sonia Bustamante Madariaga, MD, PhD Mental Health Unit Tetuán Not yet recruiting Madrid, Spain, 28003 Contact: Mariano Hernández Monsalve, MD, PhD 0034915347363 hergoico@gmail.com Contact: Ruth Berdún Pe, MD ruthberdun@yahoo.es Principal Investigator: Mariano Hernández Monsalve, MD, PhD Lafora Hospital Not yet recruiting Madrid, Spain, 28049 Contact: Juan Dios Molina Martín, MD, PhD jmolinamar@hotmail.com Principal Investigator: Juan Dios Molina Martín, MD, PhD Hospital Carlos Haya Recruiting Málaga, Spain, 29009 Contact: Fermín Mayoral Cleríes, Md, PhD fermin.mayoral.sspa@juntadeandalucia.es Principal Investigator: Fermín Mayoral Cleríes, MD, PhD Virgen del Rocío Hospital Recruiting Seville, Spain, 41013 Contact: Clara M. Rosso Fernández, MD 0034955013414 claram.rosso.sspa@juntadeandalucia.es Contact: Miguel Ruiz Veguilla, MD, PhD 0034955013414 mruizveguilla@yahoo.com Principal Investigator: Miguel Ruiz Veguilla, MD, PhD Collapse << |
| NCT01845259 | Impaired Glucose Tolerance Ass... more >>ociated With Drugs Collapse << | Phase 2 | Unknown | March 2017 | Denmark ... more >> Psychiatric Centre Rigshospitalet Copenhagen, København Ø, Denmark, 2100 Collapse << |
| NCT00107939 | Bipolar Disorder | Phase 3 | Completed | - | United States, Arkansas ... more >> Investigational Site Little Rock, Arkansas, United States, 72201 United States, California Investigational Site Orange, California, United States, 92868 Investigational Site Pico Rivera, California, United States, 90660 Investigational Site San Diego, California, United States, 92108 Investigational Site San Diego, California, United States, 92126 United States, Florida Investigational Site Boca Raton, Florida, United States, 33432 United States, Georgia Investigational Site Atlanta, Georgia, United States, 30308 United States, Illinois Investigational Site Chicago, Illinois, United States, 60640 Investigational Site Joliet, Illinois, United States, 60435 United States, Indiana Investigational Site Indianapolis, Indiana, United States, 46222 United States, Kansas Investigational Site Topeka, Kansas, United States, 66606 United States, Missouri Investigational Site Kansas City, Missouri, United States, 64133 Investigational Site St. Louis, Missouri, United States, 63104 United States, New York Invetigational Site Brooklyne, New York, United States, 11201 Investigational Site Cedarhurst, New York, United States, 11516 Investigational Site New York, New York, United States, 10003 United States, Ohio Investigational Site Cincinnati, Ohio, United States, 45267 Investigational Site Columbus, Ohio, United States, 43210 United States, Rhode Island Investigational Site Providence, Rhode Island, United States, 02903 United States, Texas Investigational Site Bellaire, Texas, United States, 77401 United States, Washington Investigational Site Bellevue, Washington, United States, 98004 United States, Wisconsin Investigational Site West Allis, Wisconsin, United States, 53227 Collapse << |
| NCT00137020 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 4 | Completed | - | Egypt ... more >> Pfizer Investigational Site Alexandria, Egypt Pfizer Investigational Site Assiut, Egypt Pfizer Investigational Site Cairo, Egypt Pfizer Investigational Site Tanta, Egypt Greece Pfizer Investigational Site Larissa, Mezourlo, Greece, 41110 Pfizer Investigational Site Athens, Greece, 11528 Pfizer Investigational Site Athens, Greece, 12462 Pfizer Investigational Site Athens, Greece, 15126 Jordan Pfizer Investigational Site Jordan, Jordan Kuwait Pfizer Investigational Site Kuwait, Kuwait, 13041 Lebanon Pfizer Investigational Site Beirut, Lebanon Saudi Arabia Pfizer Investigational Site Khobar, Saudi Arabia, 31451 South Africa Pfizer Investigational Site Garankuwa, Gauteng, South Africa, 0208 Pfizer Investigational Site Krugersdorp, Gauteng, South Africa, 1739 Pfizer Investigational Site Noordheuwel, Krugersdorp, Gauteng, South Africa, 1739 Pfizer Investigational Site Bellair, Durban, Kwa-Zulu Natal, South Africa, 4094 Pfizer Investigational Site Pinetown, Durban, Kwa-Zulu Natal, South Africa, 3600 Pfizer Investigational Site Observatory, Cape Town, Western Cape, South Africa, 7925 Turkey Pfizer Investigational Site Ankara, Turkey Pfizer Investigational Site Bursa, Turkey Pfizer Investigational Site Erzurum, Turkey Pfizer Investigational Site Istanbul, Turkey Pfizer Investigational Site Izmir, Turkey, 35340 Pfizer Investigational Site Izmir, Turkey Pfizer Investigational Site Manisa, Turkey Pfizer Investigational Site Sisli, Turkey United Arab Emirates Pfizer Investigational Site Dubai, United Arab Emirates Collapse << |
| NCT00836251 | - | - | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 53110 Collapse << |
| NCT00903916 | - | - | Completed | - | United States, North Carolina ... more >> University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01300364 | Schizophrenia | Phase 4 | Unknown | - | Spain ... more >> Parc Sanitari Sant Joan de Déu Recruiting Sant Boi Llobregat, Barcelona, Spain Contact: Judith Usall 93 6002650 ext 2356 jusall@pssjd.org Collapse << |
| NCT01245348 | - | - | Completed | - | United States, New Jersey ... more >> Medco Health Solutions, Inc. Franklin Lakes, New Jersey, United States, 07417 Collapse << |
| NCT00469664 | Schizophrenia ... more >> Schizoaffective Disorder Cognitive Impairment Collapse << | Phase 4 | Unknown | December 2008 | United States, New York ... more >> Bronx VA Medical Center Bronx, New York, United States, 10468 Mount Sinai Medical Center NY, New York, United States, 10029 Pilgrim Psychiatric Center W. Brentwood, New York, United States, 11717 Collapse << |
| NCT01498770 | - | - | Completed | - | - |
| NCT00552500 | Insulin Resistance ... more >> Body Composition Collapse << | Not Applicable | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00178971 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 3 | Completed | - | United States, Tennessee ... more >> Psychiatric Hospital at Vanderbilt Nashville, Tennessee, United States, 37212 Collapse << |
| NCT00246259 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Psychotic Disorders Collapse << | Phase 4 | Completed | - | Canada, Alberta ... more >> Calgary, Alberta, Canada Edmonton, Alberta, Canada Canada, British Columbia Victoria, British Columbia, Canada Canada, Nova Scotia Dartmouth, Nova Scotia, Canada Canada, Ontario Kingston, Ontario, Canada London, Ontario, Canada Markham, Ontario, Canada Sudbury, Ontario, Canada Canada, Quebec Montreal, Quebec, Canada Verdun, Quebec, Canada Canada Quebec, Canada Collapse << |
| NCT00520923 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT01894984 | - | - | Terminated(The study was termi... more >>nated because of high dropout rate.) Collapse << | - | - |
| NCT02213887 | Psychotic Disorders ... more >> Gastroesophageal Reflux Collapse << | Phase 4 | Active, not recruiting | August 2018 | Canada, British Columbia ... more >> UBC Hospital - Detwiller Pavilion Vancouver, British Columbia, Canada, V6T 2A1 Collapse << |
| NCT02093858 | - | - | Unknown | December 2017 | China, Hunan ... more >> Mental Health Institute Recruiting Changsha, Hunan, China, 410011 Contact: Renrong Wu, PhD 86-15874179855 wurenrong2013@163.com.cn Principal Investigator: Renrong Wu, Doctor Collapse << |
| NCT00246259 | - | - | Completed | - | - |
| NCT00615433 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT02192723 | Schizophrenia | Not Applicable | Completed | - | China ... more >> Beijing HuiLongGuan Hospital Beijing, China, 100096 Collapse << |
| NCT00615433 | - | - | Completed | - | - |
| NCT00014001 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT03201757 | Schizophrenia ... more >> Schizophreniform Disorder Bipolar I Disorder Collapse << | Phase 3 | Enrolling by invitation | January 2021 | - |
| NCT00065273 | Mental Retardation ... more >> Developmental Delay Disorder Collapse << | Phase 3 | Completed | - | United States, Kansas ... more >> University of Kansas Lawrence, Kansas, United States, 66045 Collapse << |
| NCT00018668 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> Department of Veterans Affairs San Diego, California, United States, 92161 Collapse << |
| NCT02850445 | Schizophrenia | Not Applicable | Unknown | - | China, Guangdong ... more >> Guangzhou psychitric hosptial Recruiting Guangzhou, Guangdong, China, 510370 Contact: Yingjun Zheng, Phd,Md +8618998303126 brainzheng@163.com Collapse << |
| NCT00637234 | Schizophrenia | Phase 2 Phase 3 | Completed | - | Germany ... more >> Isar-Amper-Klinikum gemeinnützige GmbH, Klinik Taufkirchen (Vils) Taufkirchen (Vils), Bayern, Germany, 84416 University of Cologne, Dept. of Psychiatry and Psychotherapy Cologne, NRW, Germany, 50924 Collapse << |
| NCT00255879 | Dyskinesia, Drug-Induced | Phase 1 | Completed | - | United States, California ... more >> University of California, San Diego Division of Geriatric Psychiatry San Diego, California, United States, 92161 Collapse << |
| NCT02282085 | Schizophrenia | Phase 4 | Unknown | - | United States, Texas ... more >> UT Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390 Principal Investigator: Matt Byerly, MD Collapse << |
| NCT03629951 | - | - | Not yet recruiting | November 14, 2021 | India ... more >> Panchshil Hospital Not yet recruiting Ahmedabad, India, 380005 Ratandeep Multispeciality Hospital Not yet recruiting Ahmedabad, India, 380008 M S Ramaiah Medical College and Hospital Not yet recruiting Bangalore, India, 560054 Asha hospital Not yet recruiting Hyderabad, India, 500034 Mahendru Psychiatric Centre Not yet recruiting Kanpur, India, 208005 Kasturba Medical College Hospital Not yet recruiting Manipal, India, 576104 Meera Hospital Not yet recruiting Mumbai, India, 421301 Meditrina Institute of Medical Sciences Not yet recruiting Nagpur, India, 440010 GMERS Medical College and Hospital Not yet recruiting Vadodara, India, 390021 Deva Institute of Health Care and Research Pvt Ltd Not yet recruiting Varanasi, India, 221005 Collapse << |
| NCT01894984 | - | - | Terminated(The study was termi... more >>nated because of high dropout rate.) Collapse << | - | - |
| NCT02423096 | - | - | Unknown | December 2016 | Taiwan ... more >> Liang-Jen Wang Recruiting Kaohsiung, Taiwan Collapse << |
| NCT00230828 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT02717195 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00077714 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00439634 | Schizophrenia | Phase 2 | Terminated(Following a pre-spe... more >>cified interim analysis and Data Monitoring Committee recommendation due to insufficient level of efficacy) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada Collapse << |
| NCT00701753 | - | - | Terminated(Insufficient subjec... more >>t recruitment.) Collapse << | - | Canada, British Columbia ... more >> Vancouver Coastal Health Vancouver, British Columbia, Canada Collapse << |
| NCT02529163 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Not Applicable | Active, not recruiting | July 2019 | Canada, Ontario ... more >> Centre for Addiction and Mental Health Toronto, Ontario, Canada, M6J 1H4 Collapse << |
| NCT00193713 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00650793 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00237861 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT02650102 | Schizophrenia | Phase 1 Phase 2 | Unknown | December 2016 | China, Shanxi ... more >> First Clinical Medical College of Shanxi Medical University Recruiting Taiyuan, Shanxi, China, 030001 Contact: Yong Xu, Doctor 18234016125 xuyongsmu@vip.163.com Contact: Sha Liu, Doctor 15803400167 liusha1984114@163.com Collapse << |
| NCT03651674 | Schizophrenia, Bipolar Disorde... more >>r Collapse << | Not Applicable | Not yet recruiting | April 1, 2020 | - |
| NCT00015548 | Alzheimer's Disease | Not Applicable | Completed | - | - |
| NCT00044655 | - | - | Completed | - | - |
| NCT03451734 | - | - | Recruiting | December 31, 2020 | China, Hunan ... more >> Mental Health Institute of 2nd Xiangya Hospital,CSU Recruiting Changsha, Hunan, China, 410011 Contact: Renrong Wu, PhD 15874179855 wurenrong2013@163.com Collapse << |
| NCT01248195 | Schizophrenia ... more >> Schizophreniform Disorder Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT02926859 | Schizophrenia | Phase 2 | Recruiting | December 2018 | Germany ... more >> Dep. of Psychiatry and Psychotherapy, Central Institute of Mental Health Recruiting Mannheim, BW, Germany, 68159 Contact: F. Markus Leweke, MD +49 621 1703 ext 2761 leweke@cimh.de Principal Investigator: F. Markus Leweke, MD Dept. of Psychiatry and Psychotherapy, Ludwig-Maximillians-University Munich Not yet recruiting Munich, BY, Germany, 80336 Contact: Peter Falkai, MD Peter.Falkai@med.uni-muenchen.de Principal Investigator: Peter Falkai, MD Dept. of Psychiatry and Psychotherapy, Charité, Campus Charité-Mitte Recruiting Berlin, B, Germany, 10117 Contact: Henrik Walter, MD, PhD henrik.walter@charite.de Principal Investigator: Henrik Walter, MD Department of Psychiatry Psychotherapy and Psychosomatics, RWTH Aachen Recruiting Aachen, NRW, Germany, 52074 Contact: Tanja Veselinovic, MD tveselinovic@ukaachen.de Principal Investigator: Tanja Veselinovic, MD Dept. of Psychiatry and Psychotherapy Recruiting Cologne, NRW, Germany, 50924 Contact: Frank Jessen, MD frank.jessen@uk-koeln.de Principal Investigator: Frank Jessen, MD Collapse << |
| NCT03019887 | Schizophrenia Relapse | Not Applicable | Active, not recruiting | - | - |
| NCT00044655 | Schizophrenia | Phase 4 | Completed | - | - |
| NCT02462473 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 2 | Terminated(Due to poor enrollm... more >>ent sponsor terminated early after enrolling 9 in Cohort 1 and no enrollment in Cohorts 2 and 3.) Collapse << | - | United States, California ... more >> Torrance, California, United States United States, Florida Kissimmee, Florida, United States United States, Pennsylvania Conshohocken, Pennsylvania, United States Collapse << |
| NCT00210769 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT01052103 | Schizophrenia | Phase 2 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Garden Grove, California, United States, 92845 United States, Illinois For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chicago, Illinois, United States, 60640 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hoffman Estates, Illinois, United States, 60169 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New York, New York, United States, 10035 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jerusalem, Israel, 91060 Italy For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Parma, Italy, 43100 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Torino, Italy, 10126 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08036 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Zamora, Spain, 49021 Collapse << |
| NCT01342120 | - | - | Completed | - | - |
| NCT01129674 | Schizophrenia | Phase 2 Phase 3 | Terminated(The decision to sto... more >>p the trial was based on efficacy results in the overall schizophrenia participant population.) Collapse << | - | - |
| NCT02237417 | Schizophrenia | Phase 4 | Recruiting | July 2018 | United States, Utah ... more >> University of Utah Recruiting Salt Lake City, Utah, United States, 84108 Principal Investigator: Deborah Yurgelun-Todd, PhD Collapse << |
| NCT02949752 | Antipsychotics Weight Gain | Phase 4 | Recruiting | August 2017 | Singapore ... more >> Institute of Mental Health Recruiting Singapore, Singapore, 556121 Contact: Bee Choo Lim 63892000 ext 3618 Bee_Choo_LIM@imh.com.sg Contact: Jaclyn Ong 63892000 ext 2959 jaclyn_yy_ong@imh.com.sg Collapse << |
| NCT00528359 | - | - | Completed | - | Belgium ... more >> Sanatia Hospital Brussels, Belgium, 1030 Collapse << |
| NCT00493233 | Schizophrenia | Not Applicable | Completed | - | Canada, Ontario ... more >> Centre for Addiction and Mental Health Toronto, Ontario, Canada, M5T 1R8 Collapse << |
| NCT02462473 | - | - | Terminated(Due to poor enrollm... more >>ent sponsor terminated early after enrolling 9 in Cohort 1 and no enrollment in Cohorts 2 and 3.) Collapse << | - | - |
| NCT01057849 | Schizophrenia | Not Applicable | Unknown | - | China, Beijing ... more >> Beijing Anding Hospital Recruiting Beijing, Beijing, China, 100000 Contact: Yan Zhou pretty_zhouyan@126.com Principal Investigator: Chuanyue Wang, M.D. Sub-Investigator: Fuchun Zhou, M.D. Beijing Huilongguan Hospital Recruiting Beijing, Beijing, China, 100000 Contact: Song Chen, M.D. asongsi@gmail.com Principal Investigator: Fude Yang, M.D. Sub-Investigator: Yunlong Tan, M.D. Peking University Institute of Mental Health Recruiting Haidian District, Beijing, China, 100191 Contact: Yanbo Yuan, M.D. +86-10-82806157 yuanyb@gmail.com Principal Investigator: Yanbo Yuan, M.D. Sub-Investigator: Qi Liu, M.D. China, Hunan Zhongnan University Xiangya Second Hospital Recruiting Changsha, Hunan, China, 410000 Contact: Maorong Hu, M.D. maron13@126.com Principal Investigator: Jingping Zhao, M.D. Sub-Investigator: Lehua Li, M.D. China, Shanghai Shanghai Mental Health Center Recruiting Shanghai, Shanghai, China, 200030 Contact: Liu, M.D. lnmilk-1982@126.com Principal Investigator: Zheng Lu, M.D. Sub-Investigator: Jianhua Sheng, M.D. China, Sichuan Sichuan University Huaxi Hospital Recruiting Chengdu, Sichuan, China, 610000 Contact: Shanming Liu, M.D. lciauo@sina.com Principal Investigator: Hong Deng, M.D. Sub-Investigator: Shanming Liu, M.D. Collapse << |
| NCT00802100 | - | - | Completed | - | - |
| NCT00806234 | Psychotic Disorders | Phase 4 | Completed | - | United States, Maryland ... more >> University of Maryland Baltimore, Maryland, United States, 21201 Johns Hopkins Hospital Baltimore, Maryland, United States, 21205 United States, New York The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 United States, North Carolina University of North Carolina, Division of Child and Adolescent Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00338949 | Schizophrenia ... more >> Metabolic Syndrome X Insulin Resistance Collapse << | Phase 4 | Completed | - | United States, California ... more >> VA San Diego Healthcare System San Diego, California, United States, 92161 Collapse << |
| NCT00806234 | - | - | Completed | - | - |
| NCT01833897 | Bipolar Disorder | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00802100 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> SHANTI Clinical Trials Colton, California, United States, 92324 Stanford University Palo Alto, California, United States, 94305 United States, Connecticut Yale University New Haven, Connecticut, United States, 06519 United States, Florida University of Miami School of Medicine Miami, Florida, United States, 33316 United States, Georgia Medical College of Georgia Augusta, Georgia, United States, 30912 United States, Maryland Clinical Insights Glen Burnie, Maryland, United States, 21061 United States, Massachusetts University of Massachusetts Worcester, Massachusetts, United States, 01605 United States, Michigan Wayne State University Detroit, Michigan, United States, 48201 United States, Minnesota University of Minnesota School of Medicine Minneapolis, Minnesota, United States, 55454 United States, New York Research Foundation for Mental Hygiene New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center-John Umstead Hospital Butner, North Carolina, United States, 27509 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75235 Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT01833897 | - | - | Completed | - | - |
| NCT02804568 | Schizophrenia | Phase 1 | Completed | - | United States, Arkansas ... more >> Alkermes Investigational Site Little Rock, Arkansas, United States, 72205 United States, New Jersey Alkermes Investigational Site Clementon, New Jersey, United States, 08021 United States, Ohio Alkermes Investigational Site Dayton, Ohio, United States, 45417 Collapse << |
| NCT01592201 | Schizophrenia | Phase 4 | Terminated(This study was earl... more >>y terminated due to insufficient enrollment required for hypothesis testing.) Collapse << | - | Korea, Republic of ... more >> Busan, Korea, Republic of Cheonan City, Korea, Republic of Chuncheon-Si, Korea, Republic of Chungju-Si, Korea, Republic of Dae-Gu, Korea, Republic of Daejeon, Korea, Republic of Goyang-Si, Korea, Republic of Gyeonggi-Do, Korea, Republic of Seoul, Korea, Republic of Collapse << |
| NCT00937755 | - | - | Completed | - | Taiwan ... more >> Taipei Medical University-Municipal Wan-Fang Hospital Taipei, Taiwan, 116 Collapse << |
| NCT00654576 | Schizophrenia | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT00304616 | Schizophrenia | Phase 4 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01272752 | Obesity | Phase 2 | Withdrawn(Study did not get st... more >>arted.) Collapse << | December 2013 | United States, South Dakota ... more >> Avera Research Institute Sioux Falls, South Dakota, United States, 57105 Collapse << |
| NCT02593734 | Schizophrenia ... more >> Mood Disorders, Anxiety Disorder Collapse << | Phase 4 | Completed | - | Ghana ... more >> Mount Horeb Prayer Centre Mamfe, Ghana, 00233 Collapse << |
| NCT01977300 | Bipolar I Disorder | Phase 3 | Completed | - | Canada, British Columbia ... more >> University of British Columbia Vancouver, British Columbia, Canada, V6T 2A1 Collapse << |
| NCT02964923 | - | - | Unknown | December 2017 | China, Shaanxi ... more >> Department of psychiatry,Xijing Hospital Xi'an, Shaanxi, China, 710000 Collapse << |
| NCT02040883 | Schizophrenia | Phase 4 | Unknown | December 2014 | China, Guangdong ... more >> Guangzhou Psychiatric Hospital Not yet recruiting Guangzhou, Guangdong, China, 510170 Contact: qingyun Yin qingyun2000@hotmail.com Collapse << |
| NCT00488891 | - | - | Terminated(The study was termi... more >>nated because it was not enrolling at the expected rate) Collapse << | - | - |
| NCT03230864 | Treatment-resistant Schizophre... more >>nia Collapse << | Phase 3 | Recruiting | September 2020 | - |
| NCT02582736 | - | - | Completed | - | Canada, Ontario ... more >> Women's College Hospital, Women's College Research Institute Toronto, Ontario, Canada, M5G 1N8 Collapse << |
| NCT01272765 | Obesity | Phase 2 | Withdrawn(Study did not get st... more >>arted.) Collapse << | January 2013 | United States, South Dakota ... more >> Avera Research Institute Sioux Falls, South Dakota, United States, 57105 Collapse << |
| NCT00073164 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00089869 | Schizophrenia | Phase 1 Phase 2 | Completed | - | United States, California ... more >> "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Cerritos, California, United States "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician. Chula Vista, California, United States For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY(1-877-285-4559) or speak with your personal physician. Garden Grove, California, United States "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." San Diego, California, United States United States, Connecticut "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." New Haven, Connecticut, United States For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY(1-877-285-4559) or speak with your personal physician. New Haven, Connecticut, United States United States, Georgia "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Atlanta, Georgia, United States United States, Illinois "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Chicago, Illinois, United States United States, Indiana "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Indianapolis, Indiana, United States United States, Iowa "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Iowa City, Iowa, United States United States, Minnesota "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Minneapolis, Minnesota, United States United States, New Hampshire "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Lebanon, New Hampshire, United States United States, New York "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." New York, New York, United States United States, North Carolina "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Butner, North Carolina, United States "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Chapel Hill, North Carolina, United States United States, Oklahoma "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Okalahoma City, Oklahoma, United States United States, Texas "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Austin, Texas, United States For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at 1-877-CTLILLY(1-877-285-4559) or speak with your personal physician. Irving, Texas, United States "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." Irvin, Texas, United States "For additional information regarding investigative sites for this trial, contact the Clinical Trials Support Center at (1-877-285-4559) or speak with your personal physician." San Antonio, Texas, United States Collapse << |
| NCT00483964 | Schizophrenia | Phase 2 | Completed | - | India ... more >> Q.Mundewadi Ayurvedic Research & Charitable Trust Mumbra,Thane, Maharashtra, India, 400612 Collapse << |
| NCT00423878 | - | - | Completed | - | - |
| NCT00205660 | Schizophrenia ... more >> Type 2 Diabetes Mellitus Collapse << | Phase 4 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01016145 | Schizophrenia or Schizoaffecti... more >>ve Disorder Collapse << | Phase 4 | Unknown | July 2010 | Brazil ... more >> Instituto de Psiquiatria - Hospital das Clinicas FMUSP Recruiting Sao Paulo, Brazil, 05403-010 Sub-Investigator: Monica Kayo, MD Collapse << |
| NCT01057329 | - | - | Recruiting | February 2018 | Austria ... more >> Medical University of Vienna Recruiting Vienna, Austria, A-1090 Contact: Andreas Karwautz, Prof 0043-1-40400 ext 3057 andreas.karwautz@meduniwien.ac.at Contact: Julia Huemer, MD 0043-1-40400- ext 2747 julia.huemer@meduniwien.ac.at Sub-Investigator: Julia Huemer, MD Collapse << |
| NCT00508157 | Metabolic Syndrome ... more >> Schizophrenia Collapse << | Phase 4 | Terminated(Slow Accrual) | - | Belgium ... more >> Local Institution Brussels, Belgium, 1160 Czech Republic Local Institution Brno, Czech Republic, 602 00 Local Institution Brno, Czech Republic, 625 00 Local Institution Havirov, Czech Republic, 736 01 Local Institution Hradec Kralove, Czech Republic, 500 05 Local Institution Prague, Czech Republic, 190 00 Local Institution Praha 10, Czech Republic, 100 00 Local Institution Praha 6, Czech Republic, 160 00 Local Institution Prerov, Czech Republic, 750 02 Local Institution Roudnice Nad Labem, Czech Republic, 41301 France Local Institution Fains Veel, France, 55000 Local Institution Lille Cedex, France, 59037 Local Institution Limoges Cedex, France, 87025 Local Institution Montpellier, France, 34295 Local Institution Paris Cedex 14, France, 75674 Local Institution Paris, France, 75013 Local Institution Poitiers, France, 86000 Local Institution Strasbourg, France, 67000 Germany Local Institution Ellwangen, Germany, 73479 Local Institution Werneck, Germany, 97440 Greece Local Institution Chania-Crete, Greece, 73300 Local Institution Corfu, Greece, 49100 Hungary Local Institution Budapest, Hungary, 1125 Local Institution Gyula, Hungary, 5700 Spain Local Institution Oviedo, Asturias, Spain, 33011 Local Institution Barcelona, Spain, 08025 Local Institution Barcelona, Spain, 08036 Local Institution Barcelona, Spain, 08907 Switzerland Local Institution Wetzikon, Switzerland, 8620 Turkey Local Institution Izmir, Turkey, 35370 Collapse << |
| NCT00508157 | - | - | Terminated(Slow Accrual) | - | - |
| NCT00423878 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT01234779 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT02634346 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT00752960 | - | - | Unknown | December 2009 | United States, Connecticut ... more >> Hartford Hospital Institute of Living Recruiting Hartford, Connecticut, United States, 06106 Contact: Steven Woolley, PhD 860-545-7329 Swoolle@harthosp.org Contact: John D. Goethe, MD 860-545-7118 jgoethe@harthosp.org Principal Investigator: John W Goethe, MD United States, Kentucky University of Kentucky Recruiting Lexington, Kentucky, United States, 40508 Contact: Jose de Leon, MD 859-246-7563 jdeleon@uky.edu Principal Investigator: Jose de Leon, MD Collapse << |
| NCT02137616 | Psychotic Disorders | Not Applicable | Unknown | December 2014 | China, Hunan ... more >> the Second Xiangya Hospital Recruiting Changsha, Hunan, China, 410011 Contact: Xiaofeng Guo, dr 073185554052 ext 073185554052 fengcsu@yahoo.com.cn Principal Investigator: xiaofeng Guo, dr Collapse << |
| NCT00177164 | Bipolar I Disorder | Phase 3 | Completed | - | United States, Pennsylvania ... more >> Mayview State Hospital Bridgeville, Pennsylvania, United States, 15017-1599 Dubois Regional Medical Center Dubois, Pennsylvania, United States, 15801 Mon-Yough Community Services, Inc. McKeesport, Pennsylvania, United States, 15132 Western Psychiatric Institute and Clinic Pittsburgh, Pennsylvania, United States, 15213-2593 Collapse << |
| NCT00910780 | Diabetes | Phase 2 Phase 3 | Withdrawn(Study was not funded... more >>.) Collapse << | November 2009 | - |
| NCT00208819 | Senile Dementia, Alzheimer Typ... more >>e Dementia, Alzheimer Type Alzheimer Disease Dementia Collapse << | Phase 4 | Completed | - | United States, Georgia ... more >> Wesley Woods Geriatric Hospital Atlanta, Georgia, United States, 30329 Collapse << |
| NCT00222495 | Psychotic Disorders ... more >> Schizophrenia Schizophreniform Disorders Schizoaffective Disorders Collapse << | Not Applicable | Completed | - | United States, Minnesota ... more >> University of Minnesota Medical Center Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT00231335 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Completed | - | - |
| NCT02634346 | - | - | Completed | - | - |
| NCT03007303 | - | - | Recruiting | July 2020 | China, Liaoning ... more >> Dalian Seventh People's Hospital Recruiting Dalian, Liaoning, China, 116023 Contact: Guanghui Fu, Postgraduate 86-188 4282 1307 fuguanghui1023@126.com Contact: Shoufu Xie, Postgraduate 86-0411 8451 4015 doctorxie1023@126.com Collapse << |
| NCT00245206 | Schizophrenia ... more >> Alzheimer's Disease Dementia Collapse << | Phase 4 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92037 Collapse << |
| NCT00845026 | Schizophrenia | Phase 2 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Escondido, California, United States, 92025 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Garden Grove, California, United States, 92845 United States, District of Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Washington, District of Columbia, United States, 20016 United States, Illinois For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chicago, Illinois, United States, 60640 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hoffman Estates, Illinois, United States, 60194 United States, Indiana For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Indianapolis, Indiana, United States, 46222 United States, Louisiana For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lake Charles, Louisiana, United States, 70601 United States, New Jersey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Willingboro, New Jersey, United States, 08046 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Amityville, New York, United States, 11701 United States, Oklahoma For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Oklahoma City, Oklahoma, United States, 73103 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Desoto, Texas, United States, 75115 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jena, Germany, D-07740 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mannheim, Germany, 68159 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wiesbaden, Germany, D-65199 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guadalajara, Mexico, 44340 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mexico City, Mexico, 14420 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Monterrey, Mexico, 64290 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Khotkovo, Russian Federation, 127025 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lipetsk, Russian Federation, 399007 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 123367 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nizhniy Novgorod, Russian Federation, 603155 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 192019 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Samara, Russian Federation, 443016 Collapse << |
| NCT01485692 | Psychomotor Agitation | Not Applicable | Completed | - | Brazil ... more >> Hospital das Clinicas da Faculdade de Medicina de Ribeirão Preto, USP Ribeirão Preto, São Paulo, Brazil, 14048-900 Collapse << |
| NCT00177164 | - | - | Completed | - | - |
| NCT01786369 | - | - | Completed | - | United States, New York ... more >> Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
