CAS No.: 388082-77-7
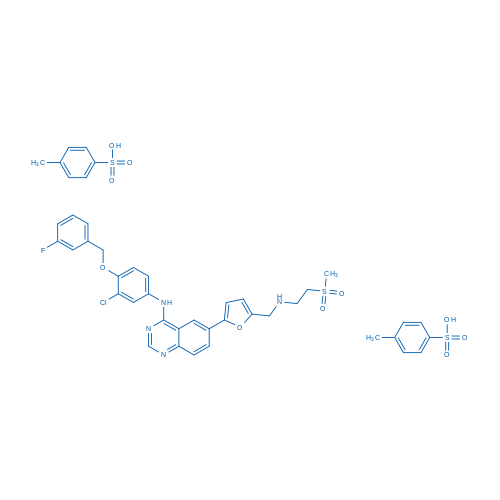
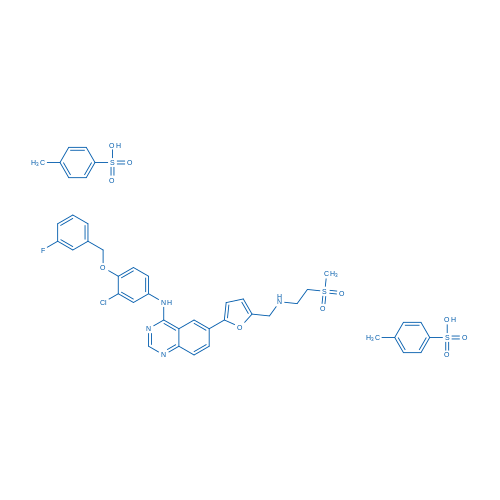
二甲苯磺酸拉帕替尼 Catalog No. CSN11269
Synonyms: GW572016 ditosylate;GW2016 ditosylate;GSK-572016
Lapatinib ditosylate is a potent EGFR and ErbB2 inhibitor with IC50 of 10.8 and 9.2 nM, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 EGFR
IC50:10.8nMHER2
IC50:9.2nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice[2]: 2 mg/kg - 100 mg/kg (p.o.), 10 mg/kg (i.v.), rat[2]: 2 mg/kg - 10 mg/kg (p.o.), 10 mg/kg (i.v.)
给药途径 p.o., i.v.
动物药代数据 Animal Mice[2] Rats[2] Dogs[2] Dose 10 mg/kg 10 mg/kg 10 mg/kg Administration i.v. i.v. i.v. AUC∞ 3469 ng·h/ml 3596 ± 924 ng·h/ml 14087 ± 3094 ng·h/ml T1/2 5.69 h 12.3 ± 8.24 h 5.85 ± 0.17 h Tmax 0.5 h 0.08 h 0.08 h CL 48.0 ml/min/kg 48.8 ± 14.3 ml/min/kg 12.2 ± 2.83 ml/min/kg Cmax 942 ng/ml 4557 ± 1210 ng/ml 3793 ± 167 ng/ml Vss 9.55 L/kg 6.16 ± 1.95 L/kg 5.70 ± 1.03 L/kg - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00849329 Neoplasms, Breast Phase 1 Completed - United States, South Carolina ... more >> GSK Investigational Site Greenville, South Carolina, United States, 29605 Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 135-710 GSK Investigational Site Songpa-gu, Seoul, Korea, Republic of, 138-736 Spain GSK Investigational Site Hospitalet de Llobregat (Barcelona), Spain, 08907 Collapse << NCT00486954 Neoplasms, Gastrointestinal Tr... more >>act Collapse << Phase 3 Completed - China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China, 510060 China GSK Investigational Site Beijing, China, 100021 GSK Investigational Site Beijing, China, 100071 GSK Investigational Site Shanghai, China, 200032 Japan GSK Investigational Site Tokyo, Japan, 113-8677 Korea, Republic of GSK Investigational Site Hwasun, Korea, Republic of, 519-809 GSK Investigational Site Seongnam-si Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 120-752 GSK Investigational Site Seoul, Korea, Republic of, 135-710 Taiwan GSK Investigational Site Kaohsiung, Taiwan, 807 GSK Investigational Site Niaosong Township, Kaohsiung, Taiwan, 833 GSK Investigational Site Tainan County, Taiwan, 736 GSK Investigational Site Tainan, Taiwan, 704 GSK Investigational Site Taipei, Taiwan, 100 GSK Investigational Site Taipei, Taiwan, 112 GSK Investigational Site Tau-Yuan County, Taiwan, 333 Collapse << NCT00486954 - - Completed - - - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 388082-77-7 | 储存条件 |
|
|||||||||||
| 分子式 | C43H42ClFN4O10S3 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 925.46 | 别名 | GW572016 ditosylate;GW2016 ditosylate;GSK-572016;GW-2016;Lapatinib;Brand name: Tykerb. CAT#100490B;二甲苯磺酸拉帕替尼 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 22Rv1 cell | - | Cytotoxicity assay | 4 days | Cytotoxicity against human 22Rv1 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=6.06 μM | 22169601 |
| A253 cell | - | Growth inhibition assay | - | Inhibition of human A253 cell growth in a cell viability assay, IC50=2.0483 μM | SANGER |
| A388 cell | - | Growth inhibition assay | - | Inhibition of human A388 cell growth in a cell viability assay, IC50=2.0483 μM | SANGER |
| A431 cells | - | Cytotoxicity assay | - | Cytotoxicity against human A431 cells after 72 hrs by SRB assay, IC50=0.104 μM | 19888761 |
| A431 cells | - | Function assay | - | Inhibition of EGFR intracellular phosphorylation in human A431 cells by ELISA, IC50=0.052 μM | 20346655 |
| BB30-HNC cell | - | Growth inhibition assay | - | Inhibition of human BB30-HNC cell growth in a cell viability assay, IC50=1.97335 μM | SANGER |
| BT474 cell | - | Growth inhibition assay | - | Inhibition of BT474 cell growth after 72 hrs, IC50=0.08 μM | 16777410 |
| BT474 cell line | - | Proliferation assay | - | Antiproliferative activity against human BT474 cell line, IC50=0.025 μM | 16483772 |
| BXF T24 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human BXF T24 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=9.65 μM | 22169601 |
| CAL27 cells | - | Cytotoxicity assay | - | Cytotoxicity against human CAL27 cells overexpressing EGFR by resazurin dye reduction assay, IC50=0.007 μM. | 21080629 |
| CAL27 cells | - | Function assay | 16 h | Inhibition of EGF-induced EGFR phosphorylation in human CAL27 cells overexpressing EGFR after 16 hrs by Western blot, IC50=0.032 μM | 21080629 |
| CXF 269L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human CXF 269L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=8.36 μM | 22169601 |
| DIFI cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human DIFI cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=0.235 μM | 22169601 |
| DSH1 cell | - | Growth inhibition assay | - | Inhibition of human DSH1 cell growth in a cell viability assay, IC50=0.09396 μM | SANGER |
| DU145 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human DU145 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=2.99 μM | 22169601 |
| ECC12 cell | - | Growth inhibition assay | - | Inhibition of human ECC12 cell growth in a cell viability assay, IC50=0.09231 μM | SANGER |
| EKVX cell | - | Growth inhibition assay | - | Inhibition of human EKVX cell growth in a cell viability assay, IC50=0.44874 μM | SANGER |
| GXF251L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human GXF251L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=1.48 μM | 22169601 |
| HCC2218 cell | - | Growth inhibition assay | - | Inhibition of human HCC2218 cell growth in a cell viability assay, IC50=0.05326 μM | SANGER |
| Hep3B2 cells | - | Proliferation assay | - | Antiproliferative activity against human Hep3B2 cells after hrs by ATP content assay, IC50=5.49 μM | 20143778 |
| HepG2 cells | - | Proliferation assay | - | Antiproliferative activity against human HepG2 cells after hrs by ATP content assay, IC50=6.27 μM | 20143778 |
| HFF cell | - | Growth inhibition assay | - | Inhibition of HFF cell growth, IC50=9.9 μM | 16777410 |
| HN5 cell | - | Growth inhibition assay | - | Inhibition of HN5 cell growth after 72 hrs, IC50=0.12 μM | 16777410 |
| HN5 cell line | - | Proliferation assay | - | Antiproliferative activity against human HN5 cell line, IC50=0.025 μM | 16483772 |
| HT-29 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human HT-29 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=4.62 μM | 22169601 |
| LB2241-RCC cell | - | Growth inhibition assay | - | Inhibition of human LB2241-RCC cell growth in a cell viability assay, IC50=1.15403 μM | SANGER |
| LB996-RCC cell | - | Growth inhibition assay | - | Inhibition of human LB996-RCC cell growth in a cell viability assay, IC50=1.36228 μM | SANGER |
| LC-1F cell | - | Growth inhibition assay | - | Inhibition of human LC-1F cell growth in a cell viability assay, IC50=1.38244 μM | SANGER |
| LIXF 575L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LIXF 575L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=7.18 μM | 22169601 |
| LNCAP cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LNCAP cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=3.68 μM | 22169601 |
| LS-513 cell | - | Growth inhibition assay | - | Inhibition of human LS-513 cell growth in a cell viability assay, IC50=3.40041 μM | SANGER |
| LXFA 289L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LXFA 289L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=5.79 μM | 22169601 |
| LXFA 526L | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LXFA 526L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=4.21 μM | 22169601 |
| LXFA 629L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LXFA 629L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=2.87 μM | 22169601 |
| LXFL 1121L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LXFL 1121L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=7.73 μM | 22169601 |
| LXFL 529L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human LXFL 529L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=3.51 μM | 22169601 |
| MCF7 cells | - | Proliferation assay | - | Antiproliferative activity against human MCF7 cells after hrs by ATP content assay, IC50=6.6 μM | 20143778 |
| MCF7 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human MCF7 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=4.83 μM | 22169601 |
| MDA231 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human MDA231 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=7.7 μM | 22169601 |
| MDA-MB-231 cells | - | Proliferation assay | - | Antiproliferative activity against human MDA-MB-231 cells after hrs by ATP content assay, IC50=5.4 μM | 20143778 |
| MIAPaCa cells | - | Function assay | - | Inhibition of EGFR phosphorylation in human MIAPaCa cells by ELISA, IC50=0.433 μM | 20817523 |
| MIAPaCa cells | - | Function assay | - | Inhibition of ERBb2 phosphorylation in human MIAPaCa cells by ELISA, IC50=0.14 μM | 20817523 |
| N87 cell | - | Growth inhibition assay | - | Inhibition of N87 cell growth after 72 hrs, IC50=0.08 μM | 16777410 |
| N87 cells | - | Function assay | - | Inhibition of ErbB2 intracellular phosphorylation in human N87 cells by ELISA, IC50=0.1 μM | 20346655 |
| NCI-H1648 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H1648 cell growth in a cell viability assay, IC50=0.02544 μM | SANGER |
| NIH/3T3 cells | - | Proliferation assay | 72 h | Antiproliferative activity against mouse NIH/3T3 cells after 72 hrs by MTT assay, IC50=4.3 μM | 22595177 |
| NMC-G1 cell | - | Growth inhibition assay | - | Inhibition of human NMC-G1 cell growth in a cell viability assay, IC50=3.54501 μM | SANGER |
| NTERA-S-cl-D1 cell | - | Growth inhibition assay | - | Inhibition of human NTERA-S-cl-D1 cell growth in a cell viability assay, IC50=6.26561 μM | SANGER |
| OCUB-M cell | - | Growth inhibition assay | - | Inhibition of human OCUB-M cell growth in a cell viability assay, IC50=0.0574 μM | SANGER |
| OS-RC-2 cell | - | Growth inhibition assay | - | Inhibition of human OS-RC-2 cell growth in a cell viability assay, IC50=1.99199 μM | SANGER |
| OVCAR-4 cell | - | Growth inhibition assay | - | Inhibition of human OVCAR-4 cell growth in a cell viability assay, IC50=9.11675 μM | SANGER |
| OVXF 899L | - | Cytotoxicity assay | 4 days | Cytotoxicity against human OVXF 899L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=3.35 μM | 22169601 |
| PANC1 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human PANC1 cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=8.12 μM | 22169601 |
| PAXF 546L cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human PAXF 546L cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=6.12 μM | 22169601 |
| PC3M cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human PC3M cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=4.55 μM | 22169601 |
| RKO cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human RKO cells after 4 days by propidium iodide staining-based fluorometric analysis, IC50=5.35 μM | 22169601 |
| RL95-2 cell | - | Growth inhibition assay | - | Inhibition of human RL95-2 cell growth in a cell viability assay, IC50=3.1567 μM | SANGER |
| SKBR3 cells | - | Cytotoxicity assay | - | Cytotoxicity against human SKBR3 cells after 72 hrs by SRB assay, IC50=0.017 μM | 19028425 |
| SKBR3 cells | - | Cytotoxicity assay | - | Cytotoxicity against human SKBR3 cells after 72 hrs by SRB assay, IC50=0.029 μM | 19888761 |
| SK-BR-3 cells | - | Proliferation assay | - | Antiproliferative activity against human SK-BR-3 cells after hrs by ATP content assay, IC50=0.04 μM | 20143778 |
| SK-BR-3 cells | - | Proliferation assay | - | Antiproliferative activity against ErbB2 overexpressing human SK-BR-3 cells by MTT assay, IC50=0.032 μM | 21570843 |
| SKHEP1 cells | - | Proliferation assay | - | Antiproliferative activity against human SKHEP1 cells after hrs by ATP content assay, IC50=5.3 μM | 20143778 |
| SKOV3 cells | - | Cytotoxicity assay | - | Cytotoxicity against human SKOV3 cells overexpressing HER2 by resazurin dye reduction assay, IC50=0.003 μM. | 21080629 |
| SW954 cell | - | Growth inhibition assay | - | Inhibition of human SW954 cell growth in a cell viability assay, IC50=5.39245 μM | SANGER |
| SW962 cell | - | Growth inhibition assay | - | Inhibition of human SW962 cell growth in a cell viability assay, IC50=5.39245 μM | SANGER |
| TE-1 cell | - | Growth inhibition assay | - | Inhibition of human TE-1 cell growth in a cell viability assay, IC50=5.02159 μM | SANGER |
| TE-12 cell | - | Growth inhibition assay | - | Inhibition of human TE-12 cell growth in a cell viability assay, IC50=0.72258 μM | SANGER |
| TE-5 cell | - | Growth inhibition assay | - | Inhibition of human TE-5 cell growth in a cell viability assay, IC50=0.24654 μM | SANGER |
| TE-6 cell | - | Growth inhibition assay | - | Inhibition of human TE-6 cell growth in a cell viability assay, IC50=0.49057 μM | SANGER |
| TE-8 cell | - | Growth inhibition assay | - | Inhibition of human TE-8 cell growth in a cell viability assay, IC50=4.0373 μM | SANGER |
| TE-9 cell | - | Growth inhibition assay | - | Inhibition of human TE-9 cell growth in a cell viability assay, IC50=1.55201 μM | SANGER |
| TK10 cell | - | Growth inhibition assay | - | Inhibition of human TK10 cell growth in a cell viability assay, IC50=4.16522 μM | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00849329 | Neoplasms, Breast | Phase 1 | Completed | - | United States, South Carolina ... more >> GSK Investigational Site Greenville, South Carolina, United States, 29605 Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 135-710 GSK Investigational Site Songpa-gu, Seoul, Korea, Republic of, 138-736 Spain GSK Investigational Site Hospitalet de Llobregat (Barcelona), Spain, 08907 Collapse << |
| NCT00486954 | Neoplasms, Gastrointestinal Tr... more >>act Collapse << | Phase 3 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China, 510060 China GSK Investigational Site Beijing, China, 100021 GSK Investigational Site Beijing, China, 100071 GSK Investigational Site Shanghai, China, 200032 Japan GSK Investigational Site Tokyo, Japan, 113-8677 Korea, Republic of GSK Investigational Site Hwasun, Korea, Republic of, 519-809 GSK Investigational Site Seongnam-si Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 120-752 GSK Investigational Site Seoul, Korea, Republic of, 135-710 Taiwan GSK Investigational Site Kaohsiung, Taiwan, 807 GSK Investigational Site Niaosong Township, Kaohsiung, Taiwan, 833 GSK Investigational Site Tainan County, Taiwan, 736 GSK Investigational Site Tainan, Taiwan, 704 GSK Investigational Site Taipei, Taiwan, 100 GSK Investigational Site Taipei, Taiwan, 112 GSK Investigational Site Tau-Yuan County, Taiwan, 333 Collapse << |
| NCT00486954 | - | - | Completed | - | - |
| NCT02362958 | HER2 Positive Breast Cancer | Phase 2 | Recruiting | December 2020 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510080 Contact: Zhong-Yu Yuan, M.D. 86-20-87343794 Collapse << |
| NCT01218529 | Brain Metastases ... more >> Lung Tumors Breast Tumors Collapse << | Phase 2 | Completed | - | Greece ... more >> General Hospital of Athens 'Hippokratio', 2nd Dept of Internal Medicine Athens, Greece, 11527 Sotiria General Hospital, 3rd Dept of Medicine, Oncology Unit Athens, Greece, 11527 General Peripheral Hospital of Athens "Alexandra" Athens, Greece, 11528 Agii Anargiri Cancer Hospital, 3rd Dept of Medical Oncology Athens, Greece, 14564 Hygeia Hospital, 1st Dept of Medical Oncology Athens, Greece, 15123 Hygeia Hospital, 2nd Dept of Medical Oncology Athens, Greece, 15123 Metropolitan Hospital, 1st Dept of Medical Oncology Athens, Greece, 18547 Metropolitan Hospital, 2nd Dept of Medical Oncology Athens, Greece, 18547 Chania General Hospital, Oncology Section Chania, Greece, 73100 Ioannina University Hospital, Dept of Medical Oncology Ioannina, Greece, 45110 University Hospital of Larissa, Dept of Internal Medicine, Oncology Section Larissa, Greece, 44110 Rio University Hospital, Dept of Medical Oncology Patras, Greece, 26500 Papageorgiou General Hospital, Dept of Medical Oncology Thessaloniki, Greece, 56429 Collapse << |
| NCT00632489 | - | - | Completed | - | - |
| NCT00632489 | Breast Cancer | Phase 1 | Completed | - | United States, Tennessee ... more >> Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00352443 | Lymphoma Unsp... more >>ecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, California ... more >> USC/Norris Comprehensive Cancer Center and Hospital Los Angeles, California, United States, 90089-9181 University of California Davis Cancer Center Sacramento, California, United States, 95817 United States, Colorado University of Colorado Cancer Center at UC Health Sciences Center Aurora, Colorado, United States, 80045 United States, Kentucky Lucille P. Markey Cancer Center at University of Kentucky Lexington, Kentucky, United States, 40536-0093 United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109-0942 United States, Washington University Cancer Center at University of Washington Medical Center Seattle, Washington, United States, 98195-6043 Collapse << |
| NCT01497626 | Advanced Solid Tumors | Phase 1 | Terminated(Drug support withdr... more >>awn) Collapse << | - | United States, District of Col... more >>umbia Georgetown Lombardi Comprehensive Cancer Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT00962312 | Pancreatic Cancer | Phase 2 | Completed | - | Ireland ... more >> Cork University Hospital Cork, Ireland Mercy University Hospital Cork, Ireland Adelaide and Meath Hospital, Dublin Incorporating the National Children's Hospital Dublin, Ireland, 24 St. Vincent's University Hospital Dublin, Ireland, 4 Mater Misericordiae University Hospital Dublin, Ireland, 7 Mater Private Hospital Dublin, Ireland, 7 St. James's Hospital Dublin, Ireland, 8 Beaumont Hospital Dublin, Ireland, 9 University College Hospital Galway, Ireland Collapse << |
| NCT01730677 | Metastatic Breast Cancer | Phase 2 | Unknown | December 2017 | Korea, Republic of ... more >> National Cancer Center Recruiting Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Contact: Jungsil Ro +82-31-920-1610 jungsro@ncc.re.kr Contact: Inhae Park +82-31-920-1680 parkih@ncc.re.kr Principal Investigator: Jungsil Ro Collapse << |
| NCT01985893 | Metastatic Breast Cancer | Phase 2 | Withdrawn(Due to missing patie... more >>nt recruitment, the financial support was stopped.) Collapse << | - | Germany ... more >> Charite Campus Mitte II. Medizinische Klinik Hämatologie und Onkologie Berlin, Germany, 10117 Gynäkologische Praxis Dr. Jörg Schilling Berlin, Germany, 10317 MediOnko Institut GbR Dr. Klare Berlin, Germany, 10367 Gynäkologische Praxis Dr. Ruhmland Berlin, Germany, 12683 Gynäkologische Gemeinschaftspraxis Morack/Letschert Berlin, Germany, 13156 Gynäkologische Praxis Dr. Jungberg Chemnitz, Germany, 09117 UK, Frauenheilk. u. Geburtsklinik Prof. Dr. med. Pauline Wimberger Dresden, Germany, 01307 Kliniken Essen-Mitte PD Dr. Kümmel Essen, Germany, 45136 Gynäkologische Praxis Dr. Heinrich Fürstenwalde, Germany, 15517 Gynäkologische Praxis Dr. Busch Mühlhausen, Germany, 99974 Gemeinschaftspraxis "Gynäkologie Arabella" Dr. Prechtl München, Germany, 81925 Praxis für Innere Medizin Dr. Uhlig Naunhof, Germany, 14683 Gynäkologische Praxis Dr. Guth Plauen, Germany, 08525 Gynäkologische Praxis Dr. Dietz Salzgitter, Germany, 38226 g.sund Kompetenzzentrum Dr. Hielscher Stralsund, Germany, 18435 Asklepios Kliniken Weißenfels Dr. Lampe Weißenfels, Germany, 06667 Gynäkologische Praxis Dr. Guido Augustinus Süttmann Wunstorf, Germany, 31515 Collapse << |
| NCT00903656 | Metastatic Breast Cancer | Phase 2 | Terminated(Caelyx was not avai... more >>lable anymore) Collapse << | - | Austria ... more >> Uniklinik Salzburg Salzburg, Austria, 5020 Collapse << |
| NCT00753207 | Breast Cancer | Phase 1 | Completed | - | Ireland ... more >> The Adelaide and Meath Hospital, Dublin Incorporating the National Childresn's Hospital Dublin, Ireland, 24 St Vincent's University Hospital Dublin, Ireland, 4 St James's Hospital Dublin, Ireland, 8 Collapse << |
| NCT01847599 | Metastatic Breast Cancer ... more >> Colorectal Cancer Collapse << | Not Applicable | Terminated(interim analysis d... more >>iscontinuation for efficacy of the intervention) Collapse << | - | France ... more >> Centre Jean Perrin Clermont Ferrand, France, 63011 Collapse << |
| NCT00095563 | - | - | Completed | - | - |
| NCT00939523 | Pituitary Adenomas ... more >> Prolactinomas Collapse << | Phase 2 | Recruiting | December 2019 | United States, California ... more >> Cedars-Sinai Medical Center Recruiting Los Angeles, California, United States, 90048 Contact: Vivian Hwe 424-315-4489 vivian.hwe@cshs.org Principal Investigator: Odelia Cooper, MD United States, Maryland Johns Hopkins University Recruiting Baltimore, Maryland, United States, 21287 Contact: Roberto Salvatori, MD 410-955-3921 salvator@jhmi.edu Principal Investigator: Roberto Salvatori, MD United States, Massachusetts Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Karen Liebert, BSN, RN 617-726-7473 kpulaski@partners.org Principal Investigator: Janet Lo, MD Collapse << |
| NCT00118157 | Estrogen Receptor Positive ... more >> Male Breast Carcinoma Progesterone Receptor Positive Recurrent Breast Carcinoma Stage IIIB Breast Cancer Stage IIIC Breast Cancer Stage IV Breast Cancer Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> Harper University Hospital - DMC Detroit, Michigan, United States, 48201 Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 Collapse << |
| NCT00095563 | High-grade Salivary Gland Carc... more >>inoma High-grade Salivary Gland Mucoepidermoid Carcinoma Low-grade Salivary Gland Carcinoma Low-grade Salivary Gland Mucoepidermoid Carcinoma Recurrent Adenoid Cystic Carcinoma of the Oral Cavity Recurrent Salivary Gland Cancer Salivary Gland Acinic Cell Tumor Salivary Gland Adenocarcinoma Salivary Gland Adenoid Cystic Carcinoma Salivary Gland Malignant Mixed Cell Type Tumor Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Phase 2 Consortium Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01873833 | HER2-positive Breast Cancer ... more >> Recurrent Breast Cancer Stage IV Breast Cancer Collapse << | Phase 2 | Active, not recruiting | July 27, 2020 | United States, California ... more >> USC Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 Collapse << |
| NCT00118157 | - | - | Completed | - | - |
| NCT00101036 | - | - | Completed | - | - |
| NCT01138384 | Breast Cancer | Phase 1 Phase 2 | Completed | - | Canada, British Columbia ... more >> BCCA - Cancer Centre for the Southern Interior Kelowna, British Columbia, Canada, V1Y 5L3 BCCA - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Cancer Centre of Southeastern Ontario at Kingston Kingston, Ontario, Canada, K7L 5P9 London Regional Cancer Program London, Ontario, Canada, N6A 4L6 Canada, Quebec McGill University - Dept. Oncology Montreal, Quebec, Canada, H2W 1S6 Collapse << |
| NCT00320385 | Neoplasms, Breast | Phase 3 | Completed | - | - |
| NCT00320385 | - | - | Completed | - | - |
| NCT00095667 | Recurrent Prostate Cancer ... more >> Stage IV Prostate Cancer Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Phase 2 Consortium Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00101036 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Recurrent Extrahepatic Bile Duct Cancer Recurrent Gallbladder Cancer Unresectable Extrahepatic Bile Duct Cancer Unresectable Gallbladder Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> UC Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 Collapse << |
| NCT00113373 | - | - | Completed | - | - |
| NCT01262469 | Metastatic Breast Cancer ... more >> 70 Years Old Patients and Over After One Line of Chemotherapy With Trastuzumab Collapse << | Phase 2 | Completed | - | France ... more >> Centre Paul Papin Angers, France, 49933 Centre Hospitalier de Beauvais Beauvais, France, 60021 Clinique Tivoli Bordeaux, France, 33000 Ch Fleyriat Bourg-en-Bresse, France, 01012 Institut Cancérologie- CENTRE HOSPITALIER BREST Brest, France, 29200 Centre Francois Baclesse Caen, France, 14076 Centre Hospitalier de Lagny-Sur-Marne Lagny-sur-Marne, France, 77405 Centre Oscar Lambret Lille, France, 59020 Institut Paoli Calmettes Marseille, France, 13273 Centre Rene Gauducheau Nantes, France, 44805 Centre Antoine Lacassagne Nice, France, 06189 Centre Hospitalier Orleans La Source Orléans, France, 45100 Institut Curie Paris, France, 75005 Hegp-Hopital Broussais Paris, France, 75015 Polyclinique de Courlancy Reims, France, 51100 Centre Hospitalier de Roanne Roanne, France, 42328 Centre Henri Becquerel Rouen, France, 76038 Centre Rene Huguenin Saint-Cloud, France, 92210 Centre Paul Strauss Strasbourg, France, 67065 Institut Claudius Regaud Toulouse, France, 31052 Collapse << |
| NCT01275859 | Breast Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Nedical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT00949455 | Bladder Cancer | Phase 2 Phase 3 | Unknown | - | United Kingdom ... more >> Barts and the London NHS Trust London, England, United Kingdom, EC1M 6BQ NHS Grampian - Aberdeen Royal Infirmary Aberdeen, United Kingdom Basildon and Thurrock University Hospital NHS Trust - Basildon Hospital Basildon, United Kingdom University Hospitals Birmingham NHS Foundation Trust - Birmingham University Hospital Birmingham, United Kingdom Royal Bournemouth and Christchurch NHS Foundation Trust - Royal Bournemouth Hospital Bournemouth, United Kingdom University Hospitals Bristol NHS Trust - Bristol University Hospital Bristol, United Kingdom Cambridge University Hospitals NHS Trust - Addenbrooke's Hospital Cambridge, United Kingdom Mid Essex NHS Trust - Broomfield Hospital Chelmsford, United Kingdom Colchester University Hospitals NHS Trust Colchester, United Kingdom University Hospitals Coventry & Warwickshire NHS Trust Coventry, United Kingdom Derby Hospitals NHS Trust - Royal Derby Hospital Derby, United Kingdom NHS Greater Glasgow and Clyde - The Beatson Glasgow, United Kingdom Calderdale and Huddersfield NHS Trust - Huddersfield Royal Infirmary Huddersfield, United Kingdom Ipswich Hospital NHS Trust Ipswich, United Kingdom University Hospitals of Leicester NHS Trust Leicester, United Kingdom Clatterbridge Centre for Oncology NHS Trust Liverpool, United Kingdom Guys & St Thomas' Hospital NHS Trust - Guys Hospital London, United Kingdom Imperial Healthcare NHS Trust London, United Kingdom Royal Marsden NHS Trust London, United Kingdom South Tees NHS Trust - James Cook University Hospital Middlesborough, United Kingdom Newcastle Upon Tyne Hospitals NHS Trust Newcastle, United Kingdom Northampton General Hospitals NHS Trust Northampton, United Kingdom Nottingham University Hospitals NHS Trust Nottingham, United Kingdom Sherwood Forest Hospitals NHS Trust - Kings Mill Hospital Nottingham, United Kingdom Portsmouth Hospitals NHS Trust - Queen Alexandra Hospital Portsmouth, United Kingdom Barking, Havering and Redbridge NHS Trust - Queens Hospital Romford, United Kingdom Taunton and Somerset NHS Trust - Musgrove Park Hospital Taunton, United Kingdom Collapse << |
| NCT02238509 | Metastatic Breast Cancer | Phase 2 | Unknown | October 2017 | Italy ... more >> A.O.U. Ospedali Riuniti Umberto I Not yet recruiting Ancona, Italy, 60020 Contact: Stefano Cascinu, MD Principal Investigator: Stefano Cascinu, MD Centro di Riferimento Oncologico Recruiting Aviano, Italy, 33081 Contact: Simon Spazzapan, MD Principal Investigator: Simon Spazzapan, Md Policlinico S. Orsola Malpighi Not yet recruiting Bologna, Italy, MD Contact: Claudio Zamagni, MD Principal Investigator: Claudio Zamagni, MD Ospedale Centrale di Bolzano Not yet recruiting Bolzano, Italy, 39100 Contact: Elisabetta Cretella, MD Principal Investigator: Elisabetta Cretella, MD Presidio Ospedaliero `Antonio Perrino Recruiting Brindisi, Italy, 72100 Contact: Saverio Cinieri, MD Principal Investigator: Saverio Cinieri, MD A.O.R.N.A.S. Garibaldi Nesima di Catania Recruiting Catania, Italy, 95122 Contact: Roberto Bordonaro, MD Principal Investigator: Roberto Bordonaro, MD Humanitas Centro Catanese di Oncologia Not yet recruiting Catania, Italy, 95126 Contact: Michele Caruso, MD Principal Investigator: Michele Caruso, MD Azienda Ospedaliera S. Anna Not yet recruiting Como, Italy, 22100 Contact: Monica Giordano, MD Principal Investigator: Monica Giordano, MD Ospedale `F. Spaziani` Not yet recruiting Frosinone, Italy, 03100 Contact: Teresa Gamucci, MD Principal Investigator: Teresa Gamucci, MD I.R.C.C.S. A.O.U. San Martino Not yet recruiting Genova, Italy, 16132 Contact: Lucia Del Mastro, MD Principal Investigator: Lucia Del Mastro, MD Ospedale Civile di Guastalla Not yet recruiting Guastalla, Italy, 42016 Contact: Laura Scaltriti, Md Principal Investigator: Laura Scaltriti, Md Ospedale Vito Fazzi Recruiting Lecce, Italy, 73100 Contact: Mariangela Ciccarese, MD Principal Investigator: Mariangela Ciccarese, MD Ospedale di Lugo - AUSL della Romagna Not yet recruiting Lugo, Italy, 48022 Contact: Michele Gianni Turolla, MD Principal Investigator: Michele Gianni Turolla, Md Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori Not yet recruiting Meldola, Italy, 47014 Contact: Dino Amadori, MD Principal Investigator: Dino Amadori, MD Ospedale Niguarda Ca' Granda Not yet recruiting Milano, Italy, 20162 Contact: Salvatore Siena, Md Principal Investigator: Salvatore Siena, Md A.O. San Gerardo Recruiting Monza, Italy, 20900 Contact: Marina Elena Cazzaniga, MD 039-233.3683 ricercaindipendente.monza@gmail.com Principal Investigator: Marina Elena Cazzaniga, MD AORN "A. Cardarelli" Recruiting Naples, Italy, 80131 Contact: Nando Riccardi, MD 08174721740 nando.riccardi@alice.it Istituto Nazionale Tumori - IRCCS "Fondazione G.Pascale" Recruiting Napoli, Italy, 8011 Contact: Michele De Laurentiis, MD Principal Investigator: Michele De Laurentiis, MD Policlinico SUN Not yet recruiting Napoli, Italy, 80131 Contact: Michele Orditura, MD Principal Investigator: Michele Orditura, MD Università degli Studi di Napoli "Federico II" Recruiting Napoli, Italy, 80131 Contact: Grazia Arpino, MD Principal Investigator: Grazia Arpino, MD A.R.N.A.S. Ospedale Civico e Benfratelli Recruiting Palermo, Italy, 90127 Contact: Vita Leonardi, MD Principal Investigator: Vita Leonardi, MD Ospedale S. Maria della Misericordia Recruiting Perugia, Italy, 06156 Contact: Anna Maria Mosconi, MD Principal Investigator: Anna Maria Mosconi, MD Azienda Ospedaliera Santa Maria degli Angeli Not yet recruiting Pordenone, Italy, 33170 Contact: Silvana Saracchini, MD Principal Investigator: Silvana Saracchini, MD Ospedale di Ravenna Not yet recruiting Ravenna, Italy, 48100 Contact: Claudio Dazzi, MD Principal Investigator: Dazzi Claudio, MD Arcispedale S. Maria Nuova Azienda Ospedaliera di Reggio Emilia Not yet recruiting Reggio Emilia, Italy, 42123 Contact: Corrado Boni, MD Principal Investigator: Corrado Boni, MD Ospedale Infermi di Rimini Not yet recruiting Rimini, Italy, 47923 Contact: Lorenzo Gianni, MD Principal Investigator: Lorenzo Gianni, MD Policlinico Universitario Campus Biomedico Recruiting Roma, Italy, 00128 Contact: Giuseppe Tonini, Md Principal Investigator: Giuseppe Tonini, MD Istituto Regina Elena per lo studio e la cura dei tumori Recruiting Roma, Italy, 00144 Contact: Francesco Cognetti, Md Principal Investigator: Cognetti Francesco, MD Ospedale G. Da Procida Not yet recruiting Salerno, Italy, 84126 Contact: Maria Luisa Barzelloni, MD Principal Investigator: Maria Luisa Barzelloni, MD Ospedale Civile di Sassari SS Annunaziata Recruiting Sassari, Italy, 07100 Contact: Nina Olmeo, MD Principal Investigator: Nina Olmeo, MD Ospedale `SS. Trinità` Recruiting Sora, Italy, 03039 Contact: Teresa Gamucci, MD Principal Investigator: Teresa Gamucci, MD Azienda Ospedaliera S.Maria di Terni Recruiting Terni, Italy, 05100 Contact: Fauso Roila, Md Principal Investigator: Fausto Roila, MD A.O.U. San Giovanni Battista di Torino Not yet recruiting Torino, Italy, 10126 Contact: Mario Airoldi, MD Principal Investigator: Mario Airoldi, MD A.O.U. Santa Maria della Misericordia di Udine Recruiting Udine, Italy, 33100 Contact: fabio Puglisi, MD Principal Investigator: Fabio Puglisi, MD A.O. Ospedale di Circolo e Fondazione Macchi Not yet recruiting Varese, Italy, 21100 Contact: Giovanni Giardina, MD Principal Investigator: Giovanni Giardina, MD Ospedale Sacro Cuore Don Calabria Not yet recruiting Verona, Italy, 37024 Contact: Stefania Gori, MD Principal Investigator: Stefania Gori, MD Collapse << |
| NCT00113373 | Primary Peritoneal Cavity Canc... more >>er Recurrent Ovarian Epithelial Cancer Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Gynecologic Oncology Group Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT01128543 | Cancer | Phase 2 | Completed | - | Turkey ... more >> GSK Investigational Site Ankara, Turkey, 06100 GSK Investigational Site Ankara, Turkey, 06500 GSK Investigational Site Ankara, Turkey, 06590 GSK Investigational Site Diskapi / Ankara, Turkey, 337088 GSK Investigational Site Diyarbakir, Turkey, 21280 GSK Investigational Site Istanbul, Turkey, 34390 GSK Investigational Site Istanbul, Turkey, 34668 GSK Investigational Site Kayseri, Turkey, 38039 Collapse << |
| NCT00258050 | Neoplasms, Breast | Phase 1 | Completed | - | United States, New Hampshire ... more >> GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00107536 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Recurrent Extrahepatic Bile Duct Cancer Recurrent Gallbladder Cancer Unresectable Extrahepatic Bile Duct Cancer Unresectable Gallbladder Cancer Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Arthur G. James Cancer Hospital and Solove Research Institute at Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00424164 | Breast Cancer | Phase 1 | Unknown | - | France ... more >> Centre Regional Rene Gauducheau Dijon, France, 21079 Collapse << |
| NCT00804310 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Terminated(Lack of funding) | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010-3000 USC/Norris Comprehensive Cancer Center and Hospital Los Angeles, California, United States, 90089-9181 University of California Davis Cancer Center Sacramento, California, United States, 95817 United States, Louisiana Feist-Weiller Cancer Center at Louisiana State University Health Sciences Shreveport, Louisiana, United States, 71130-3932 Collapse << |
| NCT01128543 | - | - | Completed | - | - |
| NCT03085368 | HER2-positive Breast Cancer | Phase 2 Phase 3 | Recruiting | March 1, 2021 | China ... more >> PUMCH Recruiting Beijing, China, 100730 Contact: Sun Qiang, M.D. 0086-13001289600 1210548954@qq.com Contact: Wang Xuefei Collapse << |
| NCT01087983 | Advanced Cancers | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00684983 | HER2/Neu Positive ... more >> Recurrent Breast Carcinoma Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00107536 | - | - | Completed | - | - |
| NCT00684983 | - | - | Active, not recruiting | - | - |
| NCT00371488 | Neoplasms, Breast | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Saitama, Japan, 350-1298 GSK Investigational Site Tokyo, Japan, 135-8550 Collapse << |
| NCT00470847 | - | - | Completed | - | - |
| NCT00544804 | Breast Cancer ... more >> Metastatic Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 Collapse << |
| NCT00470847 | Breast Cancer ... more >> Brain Metastases Collapse << | Phase 1 | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, Massachusetts Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01272141 | Breast Neoplasms ... more >> Breast Cancer Cancer of the Breast Collapse << | Phase 2 | Terminated(Slow accrual, Fundi... more >>ng stopped) Collapse << | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory Midtown (Crawford Long Hospital) Atlanta, Georgia, United States, 30308 Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30329 Collapse << |
| NCT01044433 | Head and Neck Cancer ... more >> Metastatic Squamous Neck Cancer With Occult Primary Squamous Cell Carcinoma Recurrent Metastatic Squamous Neck Cancer With Occult Primary Recurrent Salivary Gland Cancer Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage IV Salivary Gland Cancer Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Larynx Stage IV Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IV Squamous Cell Carcinoma of the Oropharynx Stage IV Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IV Verrucous Carcinoma of the Larynx Stage IV Verrucous Carcinoma of the Oral Cavity Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center of The University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00299286 | Breast Cancer | Phase 2 | Completed | - | United Kingdom ... more >> Royal Marsden NHS Foundation Trust London, United Kingdom Collapse << |
| NCT00313599 | Bladder Cancer ... more >> Brain and Central Nervous System Tumors Breast Cancer Esophageal Cancer Extragonadal Germ Cell Tumor Gastric Cancer Lung Cancer Ovarian Cancer Prostate Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 Collapse << |
| NCT01272141 | - | - | Terminated(Slow accrual, Fundi... more >>ng stopped) Collapse << | - | - |
| NCT00444535 | Neoplasms, Breast | Phase 2 | Active, not recruiting | December 31, 2019 | United States, Arizona ... more >> Novartis Investigative Site Tucson, Arizona, United States, 85724 United States, California Novartis Investigative Site San Francisco, California, United States, 94115 United States, Florida Novartis Investigative Site Hollywood, Florida, United States, 33021 Novartis Investigative Site Tampa, Florida, United States, 33612 United States, New York Novartis Investigative Site Basking Ridge, New York, United States, 07920 Novartis Investigative Site Commack, New York, United States, 11725 Novartis Investigative Site New York, New York, United States, 10065 Novartis Investigative Site Rockville Centre, New York, United States, 11570 Novartis Investigative Site Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT00096447 | Recurrent Endometrial Carcinom... more >>a Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Gynecologic Oncology Group Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT03075995 | Breast Cancer | Not Applicable | Recruiting | April 1, 2019 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510000 Contact: Fei Xu, Degree 87342693 xufei@sysucc.org.cn Collapse << |
| NCT00759642 | - | - | Active, not recruiting | - | - |
| NCT00759642 | Metastatic Breast Cancer | Phase 2 | Active, not recruiting | December 2017 | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Cotton-O-Neil Cancer Center (Stormont Vail Health Care) Topeka, Kansas, United States, 66606 United States, Missouri Truman Medical Center Kansas City, Missouri, United States, 64108 Collapse << |
| NCT01591577 | Newly Diagnosed Glioblastoma M... more >>ultiforme Collapse << | Phase 2 | Recruiting | December 7, 2019 | United States, California ... more >> UCLA Recruiting Los Angeles, California, United States, 90095 Principal Investigator: Pioanh Nghiemphu, MD Collapse << |
| NCT00096447 | - | - | Completed | - | - |
| NCT00952692 | Metastatic Breast Cancer | Phase 1 Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT01118975 | Breast Cancer ... more >> Neoplasm Metastasis Collapse << | Phase 1 Phase 2 | Terminated(Lost sponsorship fo... more >>r study drug) Collapse << | - | United States, Maryland ... more >> University of Maryland Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT01118975 | - | - | Terminated(Lost sponsorship fo... more >>r study drug) Collapse << | - | - |
| NCT01700010 | Urothelial Cancer ... more >> Bladder Cancer Collapse << | Phase 2 | Withdrawn(Funding issues.) | January 2019 | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02342587 | HER2-positive Refractory Advan... more >>ced Cancer Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01622868 | ERBB2 Gene Amplification ... more >> HER2 Positive Breast Carcinoma Invasive Breast Carcinoma Metastatic Malignant Neoplasm in the Brain Recurrent Breast Carcinoma Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 2 | Recruiting | - | - |
| NCT00462956 | Neoplasms, Breast | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Ehime, Japan, 791-0280 GSK Investigational Site Fukuoka, Japan, 802-0077 GSK Investigational Site Kanagawa, Japan, 241-0815 GSK Investigational Site Saitama, Japan, 350-1298 GSK Investigational Site Tochigi, Japan, 320-0834 GSK Investigational Site Tokyo, Japan, 104-0045 GSK Investigational Site Tokyo, Japan, 113-8677 GSK Investigational Site Tokyo, Japan, 135-8550 Collapse << |
| NCT00444535 | - | - | Active, not recruiting | - | - |
| NCT01044485 | Metastatic Breast Cancer | Phase 1 Phase 2 | Completed | - | France ... more >> Centre Georges François Leclerc Dijon, Bourgogne, France, 21000 Collapse << |
| NCT00996762 | Neoplasms, Breast | Phase 1 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Scottsdale, Arizona, United States, 85259 GSK Investigational Site Tucson, Arizona, United States, 85724 United States, District of Columbia GSK Investigational Site Washington, District of Columbia, United States, 20007 United States, Florida GSK Investigational Site Fort Myers, Florida, United States, 33905 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30341 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48202 United States, Tennessee GSK Investigational Site Memphis, Tennessee, United States, 38120 GSK Investigational Site Nashville, Tennessee, United States, 37203 Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00721630 | Breast Cancer | Phase 2 | Completed | - | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan-Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memorial Sloan-Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01133912 | Breast Cancer | Phase 1 | Completed | - | Korea, Republic of ... more >> National Cancer Center Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Collapse << |
| NCT00169533 | Neoplasms, Breast | Phase 1 | Completed | - | United States, Indiana ... more >> GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48201 United States, New Hampshire GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, New Mexico GSK Investigational Site Albuquerque, New Mexico, United States, 87131 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599 GSK Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44106 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37203 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78229 Canada, Alberta GSK Investigational Site Edmonton, Alberta, Canada, T6G 1Z2 Israel GSK Investigational Site Tel Aviv, Israel, 64239 Collapse << |
| NCT00721630 | - | - | Completed | - | - |
| NCT01264081 | Malignant Melanoma | Phase 2 | Terminated(Protocol would not ... more >>be able to reach stated accrual.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01557764 | Breast Neoplasms | Phase 2 | Withdrawn | March 2016 | - |
| NCT00225758 | - | - | Terminated(slow accrual) | - | - |
| NCT00225758 | Metastatic Breast Cancer | Phase 2 | Terminated(slow accrual) | - | United States, Colorado ... more >> University of Colorado Cancer Center Aurora, Colorado, United States, 80045 United States, New Hampshire Norris Cotton Cancer Center Lebanon, New Hampshire, United States, 03756 United States, New York North Shore University Hospital Lake Success, New York, United States, 11042 Collapse << |
| NCT00881621 | Pancreas Cancer | Phase 2 | Terminated(slow enrollment) | - | United States, District of Col... more >>umbia Georgetown University Medical Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT01499160 | Breast Neoplasms ... more >> Endocrine Breast Diseases Neoplasm Metastasis Collapse << | Phase 2 | Terminated(low accrual) | - | United States, Maryland ... more >> University of Maryland Marlene & Stewart Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT01264081 | - | - | Terminated(Protocol would not ... more >>be able to reach stated accrual.) Collapse << | - | - |
| NCT00114283 | Recurrent Salivary Gland Cance... more >>r Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Nasopharynx Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Nasopharynx Stage IVA Salivary Gland Cancer Stage IVA Squamous Cell Carcinoma of the Larynx Stage IVA Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVA Squamous Cell Carcinoma of the Oropharynx Stage IVA Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVA Verrucous Carcinoma of the Larynx Stage IVA Verrucous Carcinoma of the Oral Cavity Stage IVB Salivary Gland Cancer Stage IVB Squamous Cell Carcinoma of the Larynx Stage IVB Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVB Squamous Cell Carcinoma of the Oropharynx Stage IVB Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVB Verrucous Carcinoma of the Larynx Stage IVB Verrucous Carcinoma of the Oral Cavity Stage IVC Salivary Gland Cancer Stage IVC Squamous Cell Carcinoma of the Larynx Stage IVC Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVC Squamous Cell Carcinoma of the Oropharynx Stage IVC Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVC Verrucous Carcinoma of the Larynx Stage IVC Verrucous Carcinoma of the Oral Cavity Tongue Cancer Collapse << | Phase 2 | Completed | - | United States, Virginia ... more >> University of Virginia Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00984425 | Solid Tumors | Phase 1 | Completed | - | Italy ... more >> Istituto Clinco Humanitas Rozzano, Milano, Italy, 20089 Collapse << |
| NCT00528281 | Lung Cancer, Non-Small Cell | Phase 1 | Completed | - | Germany ... more >> GSK Investigational Site Heidelberg, Baden-Wuerttemberg, Germany, 69126 GSK Investigational Site Grosshansdorf, Schleswig-Holstein, Germany, 22927 Italy GSK Investigational Site Orbassano (TO), Piemonte, Italy, 10043 Poland GSK Investigational Site Poznan, Poland, 60-569 United Kingdom GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE7 7DN Collapse << |
| NCT00470704 | Breast Cancer | Phase 2 | Active, not recruiting | December 2019 | United States, Alabama ... more >> University fo Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Illinois University of Chicago Chicago, Illinois, United States, 60452 United States, Massachusetts Dana-Farber at Faulkner Hospital Boston, Massachusetts, United States, 02130 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37232 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00881621 | - | - | Terminated(slow enrollment) | - | - |
| NCT01427322 | Epithelial Cancer | Early Phase 1 | Terminated(Slow accrual) | - | United States, Virginia ... more >> Hunter Holmes McGuire VA Medical Center Richmond, Virginia, United States, 23249 Massey Cancer Center, Virginia Commonwealth University Richmond, Virginia, United States, 23298-0037 Collapse << |
| NCT01499160 | - | - | Terminated(low accrual) | - | - |
| NCT01245660 | Bladder Carcinoma ... more >> Infiltrative Bladder Carcinoma Cystectomy Collapse << | Early Phase 1 | Terminated(The rythm of enroll... more >>ment was not compatible with the objective of recruitement in the research.) Collapse << | - | France ... more >> CHU Bordeaux - Hôpital Saint André - Department of Medical Oncology Bordeaux, France, 33075 Collapse << |
| NCT00359190 | Neoplasms, Breast | Phase 1 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33136-1002 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Pennsylvania GSK Investigational Site Allentown, Pennsylvania, United States, 18104 GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37203 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75235 United States, Virginia GSK Investigational Site Roanoke, Virginia, United States, 24018 United States, Wisconsin GSK Investigational Site Milwaukee, Wisconsin, United States, 53226 Israel GSK Investigational Site Ramat Gan, Israel, 52621 GSK Investigational Site Zrifin, Israel, 70300 Collapse << |
| NCT00820924 | Neoplasms, Breast | Phase 2 | Terminated | - | Italy ... more >> GSK Investigational Site Rimini, Emilia-Romagna, Italy, 47900 GSK Investigational Site Udine, Friuli-Venezia-Giulia, Italy, 33100 GSK Investigational Site Bergamo, Lombardia, Italy, 24128 GSK Investigational Site Milano, Lombardia, Italy, 20121 GSK Investigational Site Rozzano (MI), Lombardia, Italy, 20089 GSK Investigational Site Livorno, Toscana, Italy, 57124 GSK Investigational Site Prato (PO), Toscana, Italy, 59100 GSK Investigational Site Perugia, Umbria, Italy, 06156 GSK Investigational Site Ancona, Italy, 60020 United Kingdom GSK Investigational Site London, United Kingdom, W6 8RF Collapse << |
| NCT00103194 | Recurrent Prostate Cancer ... more >> Stage I Prostate Cancer Stage IIA Prostate Cancer Stage IIB Prostate Cancer Stage III Prostate Cancer Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> ECOG-ACRIN Cancer Research Group Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT00555152 | Ductal Breast Carcinoma In Sit... more >>u HER2/Neu Positive Collapse << | Not Applicable | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Cancer Center Birmingham, Alabama, United States, 35233 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00103194 | - | - | Completed | - | - |
| NCT00206427 | Breast Cancer | Phase 2 | Completed | - | United States, California ... more >> USC Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 United States, New Mexico University of New Mexico Albuquerque, New Mexico, United States, 87131 United States, Texas Baylor Breast Center Houston, Texas, United States, 77030 Collapse << |
| NCT02158507 | Metastatic Triple Negative Bre... more >>ast Cancer Collapse << | Not Applicable | Active, not recruiting | September 1, 2020 | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 Collapse << |
| NCT00614978 | Metastatic Breast Cancer ... more >> Brain Metastases HER2 Positive Collapse << | Phase 1 | Completed | - | Belgium ... more >> Jules Bordet Institute Brussels, Belgium, 1000 Collapse << |
| NCT00496366 | Breast Cancer ... more >> Metastatic Breast Cancer Advanced Breast Cancer HER2/Neu-positive Breast Cancer Collapse << | Phase 2 | Terminated | - | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey at Hamilton Hamilton, New Jersey, United States, 08690 Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT00496366 | - | - | Terminated | - | - |
| NCT00555152 | - | - | Completed | - | - |
| NCT00973739 | Neurofibromatosis 2 ... more >> Vestibular Schwannoma Collapse << | Phase 2 | Completed | - | United States, New York ... more >> New York University School of Medicine New York, New York, United States, 10016 Collapse << |
| NCT00206427 | - | - | Completed | - | - |
| NCT01868503 | - | - | Terminated(Protocol modificati... more >>on) Collapse << | - | - |
| NCT01868503 | Male Breast Cancer ... more >> Recurrent Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Collapse << | Phase 2 | Terminated(Protocol modificati... more >>on) Collapse << | - | United States, California ... more >> Stanford University Cancer Institute Stanford, California, United States, 94305 Collapse << |
| NCT00912275 | Metastatic Breast Cancer | Phase 1 Phase 2 | Unknown | December 2013 | Taiwan ... more >> Department of Oncology,National Taiwan University Hospital Recruiting Taipei, Taiwan, 100 Contact: Yen-Shen Lu, M.D.,Ph.D 886-2-23123456 ext 7787 yslu@ntu.edu.tw Contact: Ching-Hung Lin, M.D. 886-2-23123456 ext 7513 chinghlin@ntu.edu.tw Principal Investigator: Yen-Shen Lu, M.D.,Ph.D Sub-Investigator: Ching-Hung Lin, M.D. Sub-Investigator: Ching-Jin Chung, M.D.,Ph.D Sub-Investigator: Chiun-Sheng Huang, M.D.,Ph.D Collapse << |
| NCT00973739 | - | - | Completed | - | - |
| NCT01328054 | Cancer | Phase 4 | Completed | - | United States, Michigan ... more >> GSK Investigational Site Detroit, Michigan, United States, 48202 United States, New Hampshire GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Utah GSK Investigational Site Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00650910 | Neoplasms, Breast | Phase 1 | Completed | - | United States, New Mexico ... more >> GSK Investigational Site Albuquerque, New Mexico, United States, 87131 Canada, Alberta GSK Investigational Site Edmonton, Alberta, Canada, T6G 1Z2 Korea, Republic of GSK Investigational Site Seodaemun-gu, Seoul, Korea, Republic of, 120-752 GSK Investigational Site Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01934894 | Metastatic Breast Cancer With ... more >>Intracranial Metastases Collapse << | Phase 2 | Terminated(Study was terminate... more >>d due to lack of significant signal of efficacy) Collapse << | - | United States, Florida ... more >> Florida Cancer Specialists - South Fort Myers, Florida, United States, 33916 Florida Cancer Specialists-North Saint Petersburg, Florida, United States, 33705 United States, Ohio Oncology Hematology Care Inc. Cincinnati, Ohio, United States, 45242 United States, Tennessee Tennessee Oncology Nashville, Tennessee, United States, 37203 Collapse << |
| NCT01589861 | Breast Cancer | Phase 1 Phase 2 | Suspended(Data analysis) | January 2018 | France ... more >> Institut Paoli-Calmettes Marseille, France, 13008 Collapse << |
| NCT01245205 | HER2-positive Breast Cancer ... more >> Male Breast Cancer Recurrent Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Stage IV Breast Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, South Dakota ... more >> Sanford Cancer Center-Oncology Clinic Sioux Falls, South Dakota, United States, 57104 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01434303 | HER2/Neu Positive ... more >> Invasive Breast Carcinoma Recurrent Breast Carcinoma Stage IV Breast Cancer AJCC v6 and v7 Collapse << | Phase 1 | Active, not recruiting | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00857714 | Ductal Carcinoma in Situ | Not Applicable | Terminated(Terminated due to l... more >>ow accrual) Collapse << | - | United States, Indiana ... more >> Indiana University Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01328054 | - | - | Completed | - | - |
| NCT01934894 | - | - | Terminated(Study was terminate... more >>d due to lack of significant signal of efficacy) Collapse << | - | - |
| NCT01705340 | Adenocarcinoma of the Gastroes... more >>ophageal Junction HER2-positive Breast Cancer Male Breast Cancer Recurrent Breast Cancer Recurrent Esophageal Cancer Recurrent Gastric Cancer Stage IIIC Breast Cancer Stage IIIC Esophageal Cancer Stage IIIC Gastric Cancer Stage IV Breast Cancer Stage IV Esophageal Cancer Stage IV Gastric Cancer Collapse << | Phase 1 | Terminated | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01290354 | Cancer | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, EC1A 7BE GSK Investigational Site London, United Kingdom, W12 0NN GSK Investigational Site London, United Kingdom, W6 8RF Collapse << |
| NCT00246753 | Prostate Cancer | Phase 2 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Duke Comprehensive Cancer Center Durham, North Carolina, United States, 27710 Rex Cancer Center at Rex Hospital Raleigh, North Carolina, United States, 27607 Collapse << |
| NCT00857714 | - | - | Terminated(Terminated due to l... more >>ow accrual) Collapse << | - | - |
| NCT00246753 | - | - | Completed | - | - |
| NCT01454479 | Recurrent Endometrial Cancer | Phase 1 | Unknown | April 2014 | - |
| NCT02015169 | HER2-positive Gastric Cancer P... more >>atients With Liver Metastasis Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Korea, Republic of, 135-720 Collapse << |
| NCT00694252 | Metastatic Breast Cancer | Phase 2 | Completed | - | Greece ... more >> University Hospital of Crete Heraklion, Greece, 71110 Collapse << |
| NCT01283789 | Metastatic Breast Cancer | Phase 2 | Active, not recruiting | April 2017 | United States, Kansas ... more >> University of Kansas Medical Centner Kansas City, Kansas, United States, 66160 Collapse << |
| NCT02131506 | HER-2 Positive Breast Cancer ... more >> Malignant Neoplasm of Breast Collapse << | Phase 1 | Terminated(Safety reasons) | - | Italy ... more >> UO Oncologia medica, IRCCS IRST Meldola, FC, Italy, 47014 Collapse << |
| NCT01281163 | Estrogen Receptor Negative ... more >> Estrogen Receptor Positive HER2/Neu Positive Progesterone Receptor Negative Progesterone Receptor Positive Recurrent Breast Carcinoma Stage IV Breast Cancer Collapse << | Phase 1 | Terminated | - | United States, Connecticut ... more >> Yale University New Haven, Connecticut, United States, 06520 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Tennessee Vanderbilt University/Ingram Cancer Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00863122 | - | - | Completed | - | - |
| NCT01947023 | BRAF V600E Mutation Present ... more >> BRAF V600K Mutation Present Metastatic Thyroid Gland Carcinoma Unresectable Thyroid Gland Carcinoma Collapse << | Phase 1 | Active, not recruiting | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00863122 | Vestibular Schwannoma ... more >> NF2 Neurofibromatosis 2 Acoustic Neuroma Auditory Tumor Collapse << | Early Phase 1 | Completed | - | United States, California ... more >> House Reserach Institute Los Angeles, California, United States, 90057 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Missouri Washington University Medical Center Saint Louis, Missouri, United States, 63110 United States, New York New York University Medical Center New York, New York, United States, 10016 Weil Cornell Medical College, New York Presbyterian Hospital New York, New York, United States, 10065 United States, Ohio Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT02101905 | Anaplastic Astrocytoma ... more >> Anaplastic Ependymoma Anaplastic Oligodendroglioma Gliosarcoma Mixed Glioma Recurrent Adult Brain Neoplasm Recurrent Glioblastoma Collapse << | Phase 1 | Recruiting | - | United States, California ... more >> UCLA / Jonsson Comprehensive Cancer Center Suspended Los Angeles, California, United States, 90095 United States, Maryland Johns Hopkins University/Sidney Kimmel Cancer Center Suspended Baltimore, Maryland, United States, 21287 United States, Massachusetts Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Site Public Contact 877-442-3324 Principal Investigator: Eudocia Q. Lee United States, Michigan Henry Ford Hospital Recruiting Detroit, Michigan, United States, 48202 Contact: Site Public Contact 313-916-1784 Principal Investigator: Tobias Walbert United States, New Jersey Memorial Sloan Kettering Basking Ridge Suspended Basking Ridge, New Jersey, United States, 07920 Memorial Sloan Kettering Monmouth Suspended Middletown, New Jersey, United States, 07748 United States, New York Memorial Sloan Kettering Commack Suspended Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Suspended Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center Suspended New York, New York, United States, 10065 United States, North Carolina Wake Forest University Health Sciences Recruiting Winston-Salem, North Carolina, United States, 27157 Contact: Site Public Contact 336-713-6771 Principal Investigator: Glenn J. Lesser United States, Ohio Cleveland Clinic Foundation Suspended Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pittsburgh Cancer Institute (UPCI) Recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: Site Public Contact 412-647-8073 Principal Investigator: Frank S. Lieberman Collapse << |
| NCT00975988 | - | - | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 134-701 Collapse << |
| NCT01184482 | Colorectal Cancer ... more >> Lung Cancer Head and Neck Cancer Collapse << | Phase 1 | Completed | - | United States, District of Col... more >>umbia Georgetown University Medical Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT00075270 | - | - | Completed | - | - |
| NCT00741260 | Breast Cancer | Phase 1 Phase 2 | Completed | - | - |
| NCT00741260 | - | - | Completed | - | - |
| NCT02139358 | - | - | Active, not recruiting | - | - |
| NCT01912963 | - | - | Terminated(The study terminate... more >>d early due to weak accrual.) Collapse << | - | - |
| NCT01912963 | HER2-positive Breast Cancer ... more >> Metastatic Breast Cancer Collapse << | Phase 2 | Terminated(The study terminate... more >>d early due to weak accrual.) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT02029001 | Malignant Solid Neoplasms | Phase 2 | Recruiting | October 2022 | France ... more >> Institut Bergonié Recruiting Bordeaux, France, 33076 Contact: Antoine ITALIANO +335 56 33 33 33 a.italiano@bordeaux.unicancer.fr Principal Investigator: Antoine ITALIANO Sub-Investigator: Delphine GARBAY Sub-Investigator: Maud TOULMONDE Sub-Investigator: Sophie COUSIN Sub-Investigator: Sylvestre LE MOULEC Sub-Investigator: Mathilde CABART Sub-Investigator: Guilhem ROUBAUD Centre Jean Perrin Not yet recruiting Clermont-Ferrand, France, 63011 Contact: Xavier DURANDO 04 73 27 81 31 xavier.durando@cjp.fr Principal Investigator: Xavier DURANDO Sub-Investigator: Pierre DALLOZ Sub-Investigator: Anne-Françoise DILLIES-LEGRAIN Sub-Investigator: Pascale DUBRAY-LONGERAS Sub-Investigator: Hakim MAHAMMEDI Sub-Investigator: Marie-Ange MOURET-REYNIER Sub-Investigator: Isabelle VAN PRAAGH DOREAU Institut Daniel Hollard Not yet recruiting Grenoble, France, 38028 Contact: Cécile LEYRONNAS 04 76 70 73 33 c.leyronnas@ghm-grenoble.fr Principal Investigator: Cécile LEYRONNAS, MD Sub-Investigator: Claire GARNIER-TIXIDRE, MD Sub-Investigator: Laurence LANCRY-LECOMTE, MD Sub-Investigator: Jérôme LONG, MD Sub-Investigator: Christine REBISCHUNG, MD Centre Leon Berard Recruiting Lyon, France, 69373 Contact: Jean-Yves BLAY +33478785126 jean-yves.blay@lyon.unicancer.fr Principal Investigator: Jean-Yves BLAY, MD Sub-Investigator: Virginie AVRILLON Sub-Investigator: Thomas BACHELOT Sub-Investigator: Christophe BERGERON Sub-Investigator: Pierre BIRON Sub-Investigator: Helen BOYLE Sub-Investigator: Philippe CASSIER Sub-Investigator: Patrick COMBEMALE Sub-Investigator: Maria CHELGHOUM Sub-Investigator: Françoise DESSEIGNE Sub-Investigator: Cécile FAURE-CONTER Sub-Investigator: Jérome FAYETTE Sub-Investigator: Aude FLECHON Sub-Investigator: Christelle DE LA FOUCHARDIERE Sub-Investigator: Didier FRAPPAZ Sub-Investigator: Pierre GUIBERT Sub-Investigator: Pierre-Etienne HEUDEL Sub-Investigator: Eve-Marie NEIDHARDT Sub-Investigator: Maurice PEROL Sub-Investigator: Isabelle RAY-COQUARD Sub-Investigator: Paul REBATTU Sub-Investigator: Pierre SAINTIGNY Sub-Investigator: Catherine TERRET Sub-Investigator: Olivier TREDAN Sub-Investigator: Olivia BALLY Sub-Investigator: Stéphane DEPIL Sub-Investigator: Olfa DERBEL Sub-Investigator: Nathalie MARQUES Sub-Investigator: Philippe TOUSSAINT Sub-Investigator: Mehdi BRAHMI Sub-Investigator: Matthieu SARABI Sub-Investigator: Alice BONNEVILLE-LEVARD Sub-Investigator: Nadège CORRADINI Sub-Investigator: Louis DOUBLET Sub-Investigator: Armelle DUFRESNE Sub-Investigator: Lauriane EBERST Sub-Investigator: Sylvie NEGRIER Sub-Investigator: Katell MICHAUX Sub-Investigator: Anne-Sophie MICHALLET Sub-Investigator: Guillaume MAURICEAU Sub-Investigator: Juliette REURE Sub-Investigator: Pauline ROCHEFORT Hopital Edouard Herriot Not yet recruiting Lyon, France, 69437 Contact: Julien FORESTIER, MD +33472119738 julien.forestier@chu-lyon.fr Principal Investigator: Julien FORESTIER Sub-Investigator: Catherine LOMBARD BOHAS Sub-Investigator: Thomas WALTER Sub-Investigator: Yann MOLIN Sub-Investigator: Noura BEN SLAMA Institut Paoli Calmettes Recruiting Marseille, France, 13273 Contact: François BERTUCCI +334 91 22 35 37 bertuccif@ipc.unicancer.fr Principal Investigator: François BERTUCCI Sub-Investigator: Jean-Marc EXTRA Sub-Investigator: Anthony GONCALVES Sub-Investigator: Gwenaêlle GRAVIS-MESCAM Sub-Investigator: Anne MADROSZYK-FLANDIN Sub-Investigator: Jean-Luc RAOUL Sub-Investigator: Delphine PERROT Sub-Investigator: Magali PROVENSAL Sub-Investigator: Carole TARPIN Sub-Investigator: Frédéric VIRET Sub-Investigator: Louis TASSY Sub-Investigator: Maria Antonietta CAPPIELLO Sub-Investigator: Slimane DERMECHE Sub-Investigator: Simon LAUNAY Sub-Investigator: Frederique ROUSSEAU Sub-Investigator: Nicolas MEYER Sub-Investigator: Loïc MOUREY Sub-Investigator: Elisabeth MOYAL Sub-Investigator: Roxana Maria RADUT Sub-Investigator: Henri ROCHE Sub-Investigator: Thibaud VALENTIN Sub-Investigator: Sarah ZAHI Institut Curie Recruiting Paris, France, 75248 Contact: Christophe LE TOURNEAU, MD +33144324672 Christophe.letourneau@curie.fr Principal Investigator: Christophe LE TOURNEAU, MD Sub-Investigator: Philippe BEUZEBOC Sub-Investigator: François-Clément BIDARD Sub-Investigator: Bruno BUECHER Sub-Investigator: Paul-Henri COTTU Sub-Investigator: Catherine DANIEL Sub-Investigator: Véronique DIERAS Sub-Investigator: Valérie LAURENCE Sub-Investigator: Alain LIVARTOWSKI Sub-Investigator: Jean-Yves PIERGA Sub-Investigator: Marie ALT Sub-Investigator: Delphine LOIRAT Sub-Investigator: Marie-Paule SABLIN Sub-Investigator: Mustapha ZOUBIR Sub-Investigator: Francesco RICCI Sub-Investigator: Emanuela ROMANO Sub-Investigator: Olivier COLLARD Centre Hospitalier Lyon Sud Recruiting Pierre-Bénite, France, 69495 Contact: Benoit YOU, MD +33478864319 benoit.you@chu-lyon.fr Principal Investigator: Benoit YOU, MD Sub-Investigator: Nathalie BONNIN Sub-Investigator: Gilles FREYER Sub-Investigator: Salima HAMIZI Sub-Investigator: Sophie TARTAS Sub-Investigator: Véronique TRILLET-LENOIR Sub-Investigator: Marion CHAUVENET Sub-Investigator: Denis MAILLET Sub-Investigator: Julien PERON Sub-Investigator: Claire FALANDRY Sub-Investigator: Olivia LE SAUX Institut de cancérologie LUCIEN NEUWIRTH Not yet recruiting Saint-Priest-en-Jarez, France, 42270 Contact: Olivier COLLARD, MD 04 77 91 70 34 olivier.collard@icloire.fr Principal Investigator: Olivier COLLARD, MD Sub-Investigator: Claire BOSACKI, MD Sub-Investigator: Pierre FOURNEL, MD Sub-Investigator: Aline GUILLOT, MD Sub-Investigator: Jean-Philippe JACQUIN, MD Sub-Investigator: Thierry MURON, MD Sub-Investigator: Léa SABAN-ROCHE, MD Institut Claudius Regaud Recruiting Toulouse, France, 31059 Contact: Carlos-Alberto GOMEZ-ROCA 05 31 15 51 01 Gomez-roca.carlos@iuct-oncopole.fr Principal Investigator: Carlos-Alberto GOMEZ-ROCA Sub-Investigator: Christine CHEVREAU Sub-Investigator: Ewa-Anna COTTURA Sub-Investigator: Florence DALENC Sub-Investigator: Jean-Pierre DELORD Sub-Investigator: Marion DESLANDRES Sub-Investigator: Cécile GANDY Sub-Investigator: Laurence GLADIEFF Sub-Investigator: Eleonora MAIO D'ESPOSITO Sub-Investigator: Nicolas MEYER Sub-Investigator: Loïc MOUREY Sub-Investigator: Elisabeth MOYAL Sub-Investigator: Henri ROCHE Sub-Investigator: Thibaud VALENTIN Sub-Investigator: Sarah ZAHI Sub-Investigator: Roxana Maria RADUT Collapse << |
| NCT02396108 | Breast Cancer | Phase 1 Phase 2 | Recruiting | March 2019 | Singapore ... more >> National University Hospital Recruiting Singapore, Singapore, 119228 Contact: Soo Chin Lee (65) 6779 5555 Collapse << |
| NCT02394496 | Metastatic Breast Cancer | Phase 3 | Unknown | January 2017 | - |
| NCT02139358 | Breast Cancer | Phase 1 Phase 2 | Active, not recruiting | December 2019 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01479179 | Breast Cancer | Phase 1 Phase 2 | Withdrawn(Drug sponsor decisio... more >>n to discontinue study.) Collapse << | - | - |
| NCT02559778 | Neuroblastoma | Phase 2 | Recruiting | September 2026 | - |
| NCT02768415 | Breast Cancer | Phase 2 | Active, not recruiting | - | China, Beijing ... more >> Cancer Institute and Hospital, Chinese Academy of Medical Sciences Beijing, Beijing, China Collapse << |
| NCT00706030 | Breast Cancer ... more >> Advanced Malignant Solid Tumors Collapse << | Phase 1 Phase 2 | Completed | - | - |
| NCT01306045 | Carcinoma, Non-Small-Cell Lung... more >> Carcinoma, Small Cell Lung Carcinoma, Thymic Collapse << | Phase 2 | Recruiting | December 30, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office (888) NCI-1937 Collapse << |
| NCT00051103 | Metastatic Breast Cancer ... more >> Neoplasms, Breast Collapse << | Phase 2 | Completed | - | - |
| NCT01058044 | Cancer | Not Applicable | Completed | - | France ... more >> Centre Jean Perrin Clermont-Ferrand, France, 63011 Collapse << |
| NCT00795678 | - | - | Completed | - | United States, New York ... more >> Memorial Sloan Kettering New York, New York, United States, 10065 United States, Ohio Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01426880 | Tubular Breast Cancer Stage II... more >> Mucinous Breast Cancer Stage II Breast Cancer Female NOS Invasive Ductal Breast Cancer Tubular Breast Cancer Stage III HER-2 Positive Breast Cancer Inflammatory Breast Cancer Stage IV Inflammatory Breast Cancer Collapse << | Phase 2 Phase 3 | Completed | - | Germany ... more >> Praxis Dr. Heinrich Fürstenwalde, Brandenburg, Germany, 15517 Luisenkrankenhaus Düsseldorf, Nordrhein-Westfalen, Germany, 40235 NCT Heidelberg Heidelberg, Germany Collapse << |
| NCT03523585 | Breast Cancer | Phase 3 | Recruiting | February 2022 | United States, California ... more >> Cancer Research Collaboration Recruiting Santa Ana, California, United States, 92705 Contact: Principal Investigator 562-981-6101 wademsmith@breastlink.com United States, District of Columbia Washington Cancer Institute Recruiting Washington, District of Columbia, United States, 20010 Contact: Principal Investigator 202-877-9386 christopher.gallagher@medstar.net United States, Georgia Piedmont Cancer Institute, PC Recruiting Atlanta, Georgia, United States, 30318 Contact: Principal Investigator 717-724-6780 rsinha@piedmontcancerinstitute.com United States, New York North Shore Hematology Oncology Associates, PC Recruiting East Setauket, New York, United States, 11733 Contact: Principal Investigator 631-675-5111 researchpi@nycancer.com United States, Ohio Dayton Physicians, LLC Recruiting Kettering, Ohio, United States, 45409 Contact: Principal Investigator 937-771-2422 cbane@precisioncancerresearch.com United States, Utah Northern Utah Associates Recruiting Ogden, Utah, United States, 84405 Contact: Principal Investigator 801-387-7166 nua2@outlook.com United States, Washington MultiCare Health System Institute for Research and Innovation Recruiting Tacoma, Washington, United States, 98405 Contact: Principal Investigator 253-403-3321 anu.belur@multicare.org Japan NHO Shikoku Cancer Center Recruiting Ehime, Japan, 791-0280 Contact: See Central Contact NHO Kyushu Cancer Center Recruiting Fukuoka, Japan, 811-1395 Contact: See Central Contact Hiroshima City Hiroshima Citizens Hospital Recruiting Hiroshima, Japan, 730-8518 Contact: See Central Contact NHO Hokkaido Cancer Center Recruiting Hokkaido, Japan, 003-0804 Contact: See Central Contact Hyōgo College of Medicine Hospital Recruiting Hyōgo, Japan, 663-8501 Contact: See Central Contact Kanagawa Cancer Center Recruiting Kanagawa, Japan, 241-8515 Contact: See Central Contact Tohoku University Hospital Recruiting Miyagi, Japan, 980-8574 Contact: See Central Contact Niigata Cancer Center Hospital Recruiting Niigata, Japan, 951-8566 Contact: See Central Contact Okayama University Hospital Recruiting Okayama, Japan, 700-8558 Contact: See Central Contact NHO Osaka National Hospital Recruiting Osaka, Japan, 540-0006 Contact: See Central Contact Osaka International Cancer Institute Recruiting Osaka, Japan, 541-8567 Contact: See Central Contact Saitama Cancer Center Recruiting Saitama, Japan, 362-0806 Contact: See Central Contact The Cancer Institute Hospital of JFCR Recruiting Tokyo, Japan, 135-8550 Contact: See Central Contact Tokyo Medical University Hospital Recruiting Tokyo, Japan, 160-0023 Contact: See Central Contact Collapse << |
| NCT00706030 | - | - | Completed | - | - |
| NCT00495872 | Solid Tumors | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01304784 | HER-2 Gene Amplification | Phase 1 | Completed | - | United States, Colorado ... more >> Rocky Mountain Cancer Centers Denver, Colorado, United States, 80218 United States, Georgia Georgia Cancer Specialists Atlanta, Georgia, United States, 30342 United States, Indiana Central Indiana Cancer Centers Indianapolis, Indiana, United States, 46151 Horizon Oncology Research, Inc Lafayette, Indiana, United States, 47908 United States, Nevada Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89135 United States, New York New York Oncology/Hematology Albany, New York, United States, 12206 United States, Ohio Innovation Center - Kettering Medical Center Health Network Kettering, Ohio, United States, 45429 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, South Carolina GHS Institute of Transitional Oncology Research Greenville, South Carolina, United States, 29605 United States, Texas Texas Oncology Cancer Center Austin, Texas, United States, 78731 Texas Oncology PA North/Sammans Cancer Center Dallas, Texas, United States, 75246 Texas Oncology - Tyler Tyler, Texas, United States, 75702 United States, Virginia Virginia Oncology Associates Norfolk, Virginia, United States, 23502 United States, Washington Evergreen Hematology and Oncology Spokane, Washington, United States, 99218 Northwest Cancer Specialists-Vancouver Cancer Center Vancouver, Washington, United States, 98684 Collapse << |
| NCT00897351 | - | - | Completed | - | - |
| NCT01190241 | Advanced Solid Tumors ... more >> Inoperable Metastasis Collapse << | Not Applicable | Unknown | - | Netherlands ... more >> Medical Center Alkmaar Alkmaar, Netherlands, 1815 JD VU University Medical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT01441596 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | - |
| NCT01612351 | Head and Neck Cancer ... more >> Squamous Cell Carcinoma of the Head and Neck Collapse << | Phase 2 | Active, not recruiting | November 2031 | United States, North Carolina ... more >> The University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00387205 | Carcinoma, Renal Cell | Phase 1 | Completed | - | - |
| NCT01441596 | - | - | Completed | - | - |
| NCT01077648 | - | - | Completed | - | - |
| NCT03052634 | Advanced Breast Cancer | Phase 1 Phase 2 | Recruiting | December 2020 | China, Beijing ... more >> Cancer Hospital Chinese Academy of Medical Sciences Recruiting Beijing, Beijing, China, 100021 Collapse << |
| NCT01462604 | - | - | Completed | - | Hong Kong ... more >> GSK Investigational Site Pokfulam, Hong Kong GSK Investigational Site Shatin, Hong Kong Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 120-752 Pakistan GSK Investigational Site Karachi, Pakistan GSK Investigational Site Lahore, Pakistan, 54600 Singapore GSK Investigational Site Singapore, Singapore, 258500 GSK Investigational Site Singapore, Singapore, 308433 Collapse << |
| NCT03418558 | Metastatic Colorectal Cancer | Phase 2 | Recruiting | June 2019 | Italy ... more >> Fondazione del Piemonte per l'Oncologia Completed Candiolo, Italy, 10060 Grande Ospedale Metropolitano Niguarda Recruiting Milano, Italy Contact: Salvatore Siena, MD salvatore.siena@ospedaleniguarda.it IOV - Istituto Oncologico Veneto Completed Padova, Italy Collapse << |
| NCT01612351 | - | - | Active, not recruiting | - | - |
| NCT03500380 | Breast Neoplasms ... more >> Breast Diseases Capecitabine HER2-positive Breast Cancer HER2 Positive Breast Carcinoma Collapse << | Phase 2 | Recruiting | December 2020 | China, Beijing ... more >> Cancer Hospital Chinese Academy of Medical Sciences Recruiting Beijing, Beijing, China, 100021 Contact: Binghe Xu China, Liaoning The First Hospital of China Medical University Recruiting Shengyang, Liaoning, China Contact: Yuee teng Liaoning Cancer Hospital & Institute Recruiting Shenyang, Liaoning, China Contact: Tao sun China Peking Union Medical College Hospital Recruiting Beijing, China Contact: Qiang Sun Peking University People's Hospital Recruiting Beijing, China Contact: Shu Wang The First Hospital of Jilin University Recruiting Changchun, China Contact: Zhimin Fan Hunan Cancer Hospital Recruiting Changsha, China Contact: Quchang Ouyang Affiliated Hospital of Chengde Medical University Recruiting Chengde, China Contact: Qingshan Li Sichuan Cancer Hospital Recruiting Chengdu, China Contact: Hui Li West China Hospital Recruiting Chengdu, China Contact: Jing Jing The Second Hospital of Dalian Medical University Recruiting Dalian, China Contact: Yang Zhang Fujian Cancer Hospital Recruiting Fuzhou, China Contact: Jian Liu Sun Yat-Sen Memorial Hospital Recruiting Guangzhou, China Contact: Herui Yao The Affiliated Tumor Hospital of Guangzhou Medical University Recruiting Guangzhou, China Contact: Hongsheng Li Guizhou Cancer Hospital Recruiting Guiyang, China Contact: Li Ran The First Affiliated Hospital of Medical School of Zhejiang University Recruiting Hangzhou, China Contact: Peifen Fu The Second Affiliated Hospital of Zhejiang University School of Medicine Recruiting Hangzhou, China Contact: Yongchuan Deng Zhejiang Cancer Hospital Recruiting Hangzhou, China Contact: Xiaojia Wang Harbin Medical University Cancer Hospital Recruiting Harbin, China Contact: Qingyuan Zhang Anhui Cancer Hospital Recruiting Hefei, China Contact: Changlu Hu Anhui Province Hospital Recruiting Hefei, China Contact: Yueyin Pan The First Affiliated Hospital of Anhui Medical University Recruiting Hefei, China Contact: Kangsheng Gu Qilu Hospital of Shandong University Recruiting Jinan, China Contact: Xiuwen Wang Shandong Cancer Hospital Recruiting Jinan, China Contact: Yongsheng Wang Linyi Cancer Hospital Recruiting Linyi, China Contact: Jingfen Wang The First Affiliated Hospital of Henan University of Science & Technology Recruiting Luoyang, China Contact: Xinshuai Wang Jiangxi Cancer Hospital Recruiting Nanchang, China Contact: Yudong Wu Jiangsu Cancer Hospital Recruiting Nanjing, China Contact: Jifeng Feng Jiangsu Province Hospital Recruiting Nanjing, China Contact: Yongmei Yin The Affiliated Tumor Hospital of Guangxi Medical University Recruiting Nanning, China Contact: Weimin Xie Yunnan Cancer Hospital Recruiting Nanning, China Contact: Dequan Liu Hospital of Qingdao University Recruiting Qingdao, China Contact: Haibo Wang Fudan University Shanghai Cancer Center Recruiting Shanghai, China Contact: Zhimin Shao The Fourth Hospital of Hebei Medical University Recruiting Shijiazhuang, China Contact: Yunjiang Liu Hubei Cancer Hospital Recruiting Wuhan, China Contact: Xinhong Wu Wuhan Union Hospital Recruiting Wuhan, China Contact: Jing Cheng The First Affiliated Hospital of the Fourth Military University of P.L.A. Recruiting Xi'an, China Contact: Yan Xue The First Affiliated Hospital of Xi'an Jiaotong University Recruiting Xi'an, China Contact: Jin Yang The Affiliated Hospital of Xuzhou Medical University Recruiting Xuzhou, China Contact: Zhengqiu Zhu Xuzhou Central Hospital Recruiting Xuzhou, China Contact: Yong Liu Yantai Yuhuangding Hospital Recruiting Yantai, China Contact: Liangming Zhang Henan Cancer Hospital Recruiting Zhengzhou, China Contact: Min Yan The First Affiliated Hospital of Zhengzhou University Recruiting Zhengzhou, China Contact: Yuanting Gu Collapse << |
| NCT01891357 | Carcinoma, Ductal, Breast | Phase 2 | Unknown | December 2016 | Germany ... more >> Klinikum Esslingen Recruiting Esslingen, Baden-Württemberg, Germany, 73730 Contact: Thorsten Kühn, MD +49 711 3103 ext 3051 Contact: Angelika Jursik +49 711 3103 ext 3064 angelikajursik@gmx.de Principal Investigator: Thorsten Kühn, MD SLK Kliniken Recruiting Heilbronn, Baden-Württemberg, Germany, 74078 Contact: Reinhard Hackenberg, MD +49 7131 4934 ext 00 Principal Investigator: Reinhard Hackenberg, MD Klinikum Frankfurt Höchst Recruiting Frankfurt (Main), Hessen, Germany, 65929 Contact: Volker Möbus, MD +49 69 3106 ext 2339 volker.moebus@klinikumfrankfurt.de Principal Investigator: Volker Möbus, MD Niels-Stensen-Kliniken Recruiting Georgsmarienhütte, Niedersachsen, Germany, 49124 Contact: Albert von der Assen, MD +49 541 502 ext 2275 Principal Investigator: Albert von der Assen, MD Klinikum Westfalen GmbH - Knappschaftskrankenhaus Recruiting Dortmund, Nordrhein-Westfalen, Germany, 44309 Contact: Markus Skrobol, MD Contact: Anna Moik +49 231 9221 ext 078 studienzentrale@klinikum-westfalen.de Principal Investigator: Markus Skrobol, MD Bethesda Krankenhaus, Senologie Recruiting Duisburg, Nordrhein-Westfalen, Germany, 47053 Contact: Björn Wieland Lisboa, MD +49 203 6008 ext 1270 senologie@bethesda.de Principal Investigator: Björn Wieland Lisboa, MD St. Barbara-KIinik Recruiting Hamm, Nordrhein-Westfalen, Germany, 59073 Contact: Herrmann Wiebringhaus, MD Contact: Scharnewski Christiane +49 2381 68123 ext 52 sscharnewski@barbaraklinik.de Principal Investigator: Hermann Wiebringhaus, MD Krankenhaus Köln Holweide, Brustzentrum Recruiting Köln, Nordrhein-Westfalen, Germany, 51067 Contact: Mathias Warm, MD +49 221 8907 ext 6707 warmm@kliniken-koeln.de Principal Investigator: Mathias Warm, MD Klinikum Chemnitz gGmbH Recruiting Chemnitz, Sachsen, Germany, 09116 Contact: Nikos Fersis, MD +49 371 333 ext 22200 Principal Investigator: Nikos Fersis, MD Praxis für gynäkologische Onkologie am Brustzentrum City Recruiting Berlin, Germany, 10713 Contact: Lidia Perlova-Griff, MD Contact: Michaela Platzer +49 30 8272 ext 29840 michaela.platzer@sankt-gertrauden.de Principal Investigator: Lidia Perlova-Griff, MD Collapse << |
| NCT01248897 | - | - | Completed | - | Hong Kong ... more >> GSK Investigational Site Hong Kong, Hong Kong GSK Investigational Site Pokfulam, Hong Kong GSK Investigational Site Tuen Mun, Hong Kong GSK Investigational Site Wanchai, Hong Kong Korea, Republic of GSK Investigational Site Busan, Korea, Republic of, 602-030 GSK Investigational Site Busan, Korea, Republic of, 602-715 GSK Investigational Site Busan, Korea, Republic of, 602-739 GSK Investigational Site Gangwon-do, Korea, Republic of, 210-852 GSK Investigational Site Incheon, Korea, Republic of, 400-711 GSK Investigational Site Kyunggi-do, Korea, Republic of, 410-769 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 120-752 GSK Investigational Site Seoul, Korea, Republic of, 136-705 GSK Investigational Site Seoul, Korea, Republic of, 137-701 GSK Investigational Site Songpa-gu, Seoul, Korea, Republic of, 138-736 Philippines GSK Investigational Site Pasay City, Philippines, 1300 Singapore GSK Investigational Site Singapore, Singapore, 169610 Collapse << |
| NCT00709878 | - | - | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00709878 | - | - | Completed | - | - |
| NCT00543504 | Advanced Cancer | Phase 1 | Active, not recruiting | October 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01068704 | Breast Cancer | Phase 2 | Completed | - | United States, Texas ... more >> Texas Oncology-Abilene Abilene, Texas, United States, 79606 Texas Oncology-Beaumont Beaumont, Texas, United States, 77702 Us Oncology Central Pharmacy Fort Worth, Texas, United States, 76177 United States, Washington Yakima Valley Memorial Hospital/North Star Lodge Yakima, Washington, United States, 98902 Argentina Local Institution Rosario, Santa Fe, Argentina, S2000DSK Local Institution La Rioja, Argentina, 5300 Mexico Local Institution Colima, Mexico, 28030 Peru Local Institution Lima, Peru, 34 Collapse << |
| NCT00567554 | Breast Cancer | Phase 3 | Completed | - | Germany ... more >> Universitätsfrauenklinik Frankfurt / Main Frankfurt / Main, Germany Collapse << |
| NCT00436566 | - | - | Completed | - | - |
| NCT02056080 | - | - | Unknown | October 2014 | Spain ... more >> Recruiting Barcelona, Spain Recruiting Córdoba, Spain Recruiting Girona, Spain Recruiting Jaen, Spain Recruiting Madrid, Spain Recruiting Navarra, Spain Recruiting Salamanca, Spain Recruiting Santiago de Compostela, Spain Recruiting Sevilla, Spain Recruiting Valencia, Spain Recruiting Zaragoza, Spain Collapse << |
| NCT00688194 | Breast Cancer | Phase 3 | Unknown | - | Italy ... more >> Federico II University Medical School Recruiting Naples, Italy, 80131 Contact: Contact Person 39-081-746-3660 Collapse << |
| NCT01300962 | Metastatic Breast Cancer | Phase 1 | Active, not recruiting | December 2018 | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 Duke Comprehensive Cancer Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00898573 | - | - | Withdrawn | - | United States, Ohio ... more >> Geauga Regional Hospital Cleveland, Ohio, United States, 44024 Lake/University Ireland Cancer Center Cleveland, Ohio, United States, 44060 Ireland Cancer Center at University Hospitals Case Medical Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106-5065 University Suburban Health Center Cleveland, Ohio, United States, 44121 UHHS Chagrin Highlands Medical Center Cleveland, Ohio, United States, 44122 Southwest General Health Center Cleveland, Ohio, United States, 44130 UHHS Westlake Medical Center Cleveland, Ohio, United States, 44145 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Mercy Cancer Center at Mercy Medical Center Cleveland, Ohio, United States, 44708 Collapse << |
| NCT01526369 | Metastatic Breast Cancer | Phase 3 | Active, not recruiting | December 2018 | - |
| NCT00295243 | Advanced Solid Malignancies | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00436566 | Breast Cancer ... more >> Cardiac Toxicity Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Cancer Research Consortium Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00482391 | Breast Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber/Harvard Cancer Center at Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT01172223 | Breast Cancer | Phase 1 Phase 2 | Completed | - | Germany ... more >> Sana Klinikum Lichtenberg Berlin, Germany Collapse << |
| NCT01462552 | - | - | Completed | - | - |
| NCT03273595 | Neoplasms, Breast | Phase 2 | Recruiting | December 2019 | China, Shanxi ... more >> Xijing Hospital Recruiting Xi'an, Shanxi, China, 710032 Contact: Juliang Zhang, Doctor +862984775271 zhangjl@fmmu.edu.cn Principal Investigator: Juliang Zhang, Doctor Collapse << |
| NCT00482391 | - | - | Completed | - | - |
| NCT01471847 | Locally Advance Breast Cancer ... more >>(LABC) Metastatic Breast Cancer (MBC) Collapse << | Phase 1 | Completed | - | Spain ... more >> Novartis Investigative Site Madrid, Spain, 28050 United Kingdom Novartis Investigative Site Leicester, United Kingdom, LE1 5WW Novartis Investigative Site Manchester, United Kingdom, M20 2BX Collapse << |
| NCT00450203 | Oesophagogastric Cancer | Phase 2 Phase 3 | Unknown | December 2017 | United Kingdom ... more >> Royal Bournemouth Hospital Recruiting Bournemouth, England, United Kingdom, BH7 7DW Contact: Tom Geldart 44-1202-726-088 Bradford Royal Infirmary Active, not recruiting Bradford, England, United Kingdom, BD9 6RJ Bristol Haematology and Oncology Centre Recruiting Bristol, England, United Kingdom, BS2 8ED Contact: Stephen J. Falk, MD 44-117-928-3074 stephen.falk@ubht.nhs.uk Addenbrooke's Hospital Active, not recruiting Cambridge, England, United Kingdom, CB2 2QQ Cumberland Infirmary Active, not recruiting Carlisle, England, United Kingdom, CA2 7HY Doncaster Royal Infirmary Recruiting Doncaster, England, United Kingdom, DN2 5LT Contact: Jonathan Wadsley 44-1302-366-666 St. Luke's Cancer Centre at Royal Surrey County Hospital Recruiting Guildford, England, United Kingdom, GU2 7XX Contact: Gary W. Middleton 44-1483-570-122 gmiddleton@royalsurrey.nhs.uk Huddersfield Royal Infirmary Recruiting Huddersfield, West Yorks, England, United Kingdom, HD3 3EA Contact: Jo Dent 44-1484-342-000 Leeds Cancer Centre at St. James's University Hospital Recruiting Leeds, England, United Kingdom, LS9 7TF Contact: Matthew T. Seymour, MA, MD, FRCP 44-113-206-6400 Lincoln County Hospital Active, not recruiting Lincoln, England, United Kingdom, LN2 5QY Aintree University Hospital Recruiting Liverpool, England, United Kingdom, L9 7AL Contact: David Smith, MD 44-151-525-5980 Saint Bartholomew's Hospital Recruiting London, England, United Kingdom, EC1A 7BE Contact: Sarah Slater, MD 44-20-7601-8391 St. George's Hospital Active, not recruiting London, England, United Kingdom, SW17 0QT St. Mary's Hospital Active, not recruiting London, England, United Kingdom, W2 1NY Mid Kent Oncology Centre at Maidstone Hospital Recruiting Maidstone, England, United Kingdom, ME16 9QQ Contact: Justin Waters, MD 44-1622-729-000 Christie Hospital Recruiting Manchester, England, United Kingdom, M20 4BX Contact: Was Mansoor, MD 44-845-226-3000 Clatterbridge Centre for Oncology Recruiting Merseyside, England, United Kingdom, CH63 4JY Contact: David Smith, MD 44-151-334-1155 david.smith@ccotrust.nhs.uk Northern Centre for Cancer Treatment at Newcastle General Hospital Recruiting Newcastle-Upon-Tyne, England, United Kingdom, NE4 6BE Contact: Fareeda Coxon, MD 44-191-256-3551 fareeda.coxon@nuth.nhs.uk Derriford Hospital Active, not recruiting Plymouth, England, United Kingdom, PL6 8DH Dorset Cancer Centre Active, not recruiting Poole Dorset, England, United Kingdom, BH15 2JB Berkshire Cancer Centre at Royal Berkshire Hospital Recruiting Reading, England, United Kingdom, RG1 5AN Contact: Joss Adams, MD 44-118-322-7878 Rochdale Infirmary Active, not recruiting Rochdale, England, United Kingdom, 0L12 0NB Salisbury District Hospital Recruiting Salisbury, England, United Kingdom, SP2 8BJ Contact: Tim J. Iveson, MD 44-1722-336-262 ext. 4688 Wexham Park Hospital Recruiting Slough, Berkshire, England, United Kingdom, SL2 4HL Contact: Marcia Hall, MD 44-1753-634-364 marcia.hall@nhs.net.uk Southampton General Hospital Recruiting Southampton, England, United Kingdom, SO16 6YD Contact: Tim J. Iveson, MD 44-23-8079-6802 t.iveson@soton.ac.uk Royal Marsden - Surrey Recruiting Sutton, England, United Kingdom, SM2 5PT Contact: David Cunningham, MD 44-20-8661-3279 david.cunningham@rmh.nhs.uk Aberdeen Royal Infirmary Recruiting Aberdeen, Scotland, United Kingdom, AB25 2ZN Contact: Russell Petty, MD 44-84-5456-6000 Velindre Cancer Center at Velindre Hospital Recruiting Cardiff, Wales, United Kingdom, CF14 2TL Contact: Tom Crosby, MD 44-29-2031-6292 Basingstoke and North Hampshire Hospital Recruiting Basingstoke, United Kingdom Principal Investigator: Charlotte Rees Birmingham Heartlands Hospital Recruiting Birmingham, United Kingdom Principal Investigator: Jo Dent Castle Hill Hospital Recruiting Cottingham, United Kingdom Principal Investigator: Mohan Hingorani University Hospitals Coventry and Warwickshire Recruiting Coventry, United Kingdom Principal Investigator: Sharmila Sothi Beatson West of Scotland Cancer Centre Recruiting Glasgow, United Kingdom Principal Investigator: Janet Graham St James Hospital Recruiting Leeds, United Kingdom Principal Investigator: Matt Seymour Leicester Royal Infirmary Recruiting Leicester, United Kingdom Principal Investigator: Anne Thomas Norfolk and Norwich University Hospital Recruiting Norwich, United Kingdom Principal Investigator: Tom Roques Churchill Hospital Recruiting Oxford, United Kingdom Principal Investigator: Kinnari Patel Queens Hospital Recruiting Romford, United Kingdom Principal Investigator: Sherif Raouf Weston Park Recruiting Sheffield, United Kingdom Principal Investigator: Suzanne Darby Great Western Hospital Recruiting Swindon, United Kingdom Principal Investigator: Claire Blesing Musgrove Park Hospital Recruiting Taunton, United Kingdom Principal Investigator: Emma Cattell Collapse << |
| NCT00807859 | Breast Cancer ... more >> Breast Neoplasms Breast Tumors Cancer Locally Recurrent and Metastatic Breast Cancer Metastases Metastatic Cancer Oncology Solid Tumors Tumors Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Research Site Tucson, Arizona, United States, 85724 United States, Florida Research Site Boca Raton, Florida, United States, 33428 United States, Iowa Research Site Iowa City, Iowa, United States, 52242 United States, Massachusetts Research Site Boston, Massachusetts, United States, 02111 United States, Minnesota Research Site Minneapolis, Minnesota, United States, 55407 United States, New Hampshire Research Site Lebanon, New Hampshire, United States, 03756-0001 Research Site Lebanon, New Hampshire, United States, 03756 United States, New Mexico Research Site Albuquerque, New Mexico, United States, 87131 United States, New York Research Site Great Neck, New York, United States, 11021 Research Site New City, New York, United States, 10956 Research Site New York, New York, United States, 10032 Research Site Nyack, New York, United States, 10960 United States, Ohio Research Site Middletown, Ohio, United States, 45042 Belgium Research Site Leuven, Belgium, 3000 Research Site Liege, Belgium, 4000 Research Site Wilrijk, Belgium, 2610 France Research Site Bordeaux, France, 33075 Research Site Caen Cedex 05, France, 14076 Research Site La Roche Sur Yon Cedex 9, France, 85925 Research Site Marseille, France, 13009 Research Site Nantes Cedex 2, France, 44202 Research Site Pierre Bénite Cedex, France, 69495 Research Site Toulouse, France, 31052 Collapse << |
| NCT00498953 | Head and Neck Cancer | Phase 1 Phase 2 | Completed | - | Belgium ... more >> Institut Jules Bordet Brussels, Belgium, 1000 Collapse << |
| NCT03084939 | Breast Cancer | Phase 3 | Active, not recruiting | October 5, 2019 | China ... more >> Beijing Hospital Beijing City, China, 100006 The Affiliated Hospital of Military Medical Sciences(The 307th Hospital of Chinese PLA) Beijing, China, 100071 Beijing Cancer Hospital Beijing, China, 100142 Jilin Cancer Hospital Changchun, China, 130012 the First Hospital of Jilin University Changchun, China, 130021 Changzhou First People's Hospital Changzhou, China, 213003 West China Hospital, Sichuan University Chengdu, China, 610041 Fujian Medical University Union Hospital Fujian, China, 350001 Zhejiang Cancer Hospital; Zhejiang Cancer Hospital cancer department Hangzhou City, China, 310022 The Second Affiliated Hospital of Zhejiang University College Hangzhou, China, 310009 Harbin Medical University Cancer Hospital Harbin, China, 150081 Jiangsu Cancer Hospital Nanjing, China, 210009 Jiangsu Province Hospital Nanjing, China, 210036 Ruijin Hospital, Shanghai Jiao Tong University School of Medicine Shanghai, China, 200025 Fudan University Shanghai Cancer Center Shanghai, China, 200032 Liaoning cancer Hospital & Institute Shenyang, China, 110042 Tianjin Cancer Hospital Tianjin, China, 300060 First Affiliated Hospital of Medical College of Xi'an Jiaotong University Xi'an, China, 710061 Collapse << |
| NCT00078572 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT01361945 | Breast Cancer ... more >> Metastatic Breast Cancer HER-2 Positive Breast Cancer ER Positive Breast Cancer Collapse << | Phase 1 Phase 2 | Withdrawn(Closed due to no enr... more >>ollment) Collapse << | - | - |
| NCT00044343 | Colorectal Cancer | Phase 2 | Completed | - | - |
| NCT00486668 | Invasive Breast Cancer | Phase 3 | Unknown | March 2017 | - |
| NCT00281658 | Neoplasms, Breast | Phase 3 | Active, not recruiting | December 31, 2020 | - |
| NCT01104571 | Breast Cancer | Phase 3 | Active, not recruiting | September 2025 | United Kingdom ... more >> Wythenshawe Hospital Manchester, England, United Kingdom, M23 9LJ Collapse << |
| NCT00281658 | - | - | Active, not recruiting | - | - |
| NCT00524303 | - | - | Completed | - | - |
| NCT00524303 | Neoplasms, Breast | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Fountain Valley, California, United States, 92708 GSK Investigational Site Los Angeles, California, United States, 90057 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80220 United States, Florida GSK Investigational Site Hudson, Florida, United States, 34667 GSK Investigational Site Miami, Florida, United States, 33176 GSK Investigational Site Pembroke Pines, Florida, United States, 33028 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46219 United States, Nevada GSK Investigational Site Henderson, Nevada, United States, 89052 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19107 United States, Texas GSK Investigational Site Austin, Texas, United States, 78731 GSK Investigational Site Beaumont, Texas, United States, 77702-1449 GSK Investigational Site Bedford, Texas, United States, 76022 GSK Investigational Site Dallas, Texas, United States, 75231 GSK Investigational Site Dallas, Texas, United States, 75246 GSK Investigational Site Dallas, Texas, United States, 75320-2510 GSK Investigational Site El Paso, Texas, United States, 79915 GSK Investigational Site Houston, Texas, United States, 77024 GSK Investigational Site Lewisville, Texas, United States, 75067 GSK Investigational Site Sugar Land, Texas, United States, 77479 GSK Investigational Site Tyler, Texas, United States, 75702 United States, Virginia GSK Investigational Site Norfolk, Virginia, United States, 23502 United States, Washington GSK Investigational Site Seattle, Washington, United States, 98117 GSK Investigational Site Yakima, Washington, United States, 98902 Collapse << |
| NCT02213042 | Neoplasms, Breast | Phase 2 | Active, not recruiting | March 4, 2019 | - |
| NCT01205217 | Neoplasms, Breast | Phase 2 | Withdrawn(Data presented at SA... more >>BCS 2010 showing that dual blockade is superior to monotherapy) Collapse << | December 2010 | Austria ... more >> GSK Investigational Site Graz, Austria, A-8036 Hungary GSK Investigational Site Budapest, Hungary, 1122 GSK Investigational Site Gyula, Hungary, 5700 Norway GSK Investigational Site Oslo, Norway, 0310 GSK Investigational Site Oslo, Norway, 0407 Spain GSK Investigational Site Castellón, Spain, 12002 Collapse << |
| NCT00769470 | Breast Cancer | Phase 2 | Completed | - | United States, California ... more >> Central Hematology Oncology Medical Group, Incorporated - Alhambra Alhambra, California, United States, 91801 Comprehensive Blood and Cancer Center Bakersfield, California, United States, 93309-0633 St. Jude Heritage Medical Group at Virginia K. Crosson Cancer Center Fullerton, California, United States, 92835 North Valley Hematology-Oncology Medical Group Northridge, California, United States, 91328 Wilshire Oncology Medical Group, Incorporated - Pomona Pomona, California, United States, 91767-3021 Sansum Medical Clinic Santa Barbara, California, United States, 93105 Central Coast Medical Oncology Corporation Santa Maria, California, United States, 93454 Santa Barbara Hematology Oncology - Solvang Solvang, California, United States, 93463 Cancer Care Associates Medical Group, Incorporated - Redondo Beach Torrance, California, United States, 90505 United States, Florida Cancer Institute of Florida, PA - Orlando Orlando, Florida, United States, 32804 Florida Hospital Cancer Institute Orlando, Florida, United States, 32804 Hematology and Oncology Consultants, PA - Orlando Orlando, Florida, United States, 32804 United States, Indiana Providence Medical Group Haute Terre, Indiana, United States, 47802 United States, Nevada Comprehensive Cancer Centers of Nevada - Henderson Henderson, Nevada, United States, 89052 United States, New Mexico New Mexico Cancer Center Albuquerque, New Mexico, United States, 87109 Collapse << |
| NCT01973660 | Breast Cancer | Phase 2 | Completed | - | Spain ... more >> Hospital de Torrevieja Torrevieja, Alicante, Spain Institut Català d'Oncologia Hospitalet Hospitalet de Llobregat, Barcelona, Spain Hospital Mutua de Terrassa Terrassa, Barcelona, Spain Consorcio Hospitalario Provincial de Castellón Castelló de la Plana, Castellón, Spain Hospital Clínico Universitario de Santiago de Compostela Santiago de Compostela, Galicia, Spain Hospital Son Llàtzer Palma de Mallorca, Islas Baleares, Spain Hospital Universitario Son Espases Palma de Mallorca, Islas Baleares, Spain Hospital Universitario de Fuenlabrada Fuenlabrada, Madrid, Spain Hospital Universitario Virgen de la Arrixaca El Palmar, Murcia, Spain Hospital Universitario Sant Joan de Reus Reus, Tarragona, Spain Hospital Luis Alcanyís de Xàtiva Xàtiva, Valencia, Spain, 46800 Hospital Universitario Infanta Cristina Badajoz, Spain, 06071 Vall d'Hebron University Hospital Barcelona, Spain, 08035 Hospital Clínic de Barcelona Barcelona, Spain Instituto Dexeus Barcelona, Spain Hospital San Pedro de Alcántara Cáceres, Spain, 10003 Hospital Universitario Arnau de Vilanova de Lleida Lleida, Spain Hospital Universitario 12 de Octubre Madrid, Spain, 28041 Hospital Clínico San Carlos Madrid, Spain Hospital Universitario Ramón y Cajal Madrid, Spain Hospital Clínico Universitario de Valencia Valencia, Spain Hospital Universitario Arnau de Vilanova de Valencia Valencia, Spain Collapse << |
| NCT00388076 | Neoplasms, Breast | Phase 1 | Completed | - | United States, New Jersey ... more >> GSK Investigational Site New Brunswick, New Jersey, United States, 08901 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44106 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00450892 | Breast Cancer | Phase 1 Phase 2 | Completed | - | Belgium ... more >> C.H.U. Sart-Tilman Liege, Belgium Clinique Sainte Elisabeth Namur, Belgium France Institut Bergonie Bordeaux, France Institut Bergonié Bordeaux, France CHRU de Limoges Limoges, France Centre Leon Berard Lyon, France CRLC Val D'Aurelle Montpellier, France Centre Henri Becquerel Rouen, France Hopital Rene Huguenin - Institut Curie Saint-Cloud, France Centre Paul Strauss Strasbourg, France Institut Gustave Roussy Villejuif, France Slovenia The Institute Of Oncology Ljubljana, Slovenia Switzerland Hôpitaux universitaires de Genève - HUG - site de Cluse-Roseraie Geneve, Switzerland Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland United Kingdom Western General Hospital Edinburgh, United Kingdom Collapse << |
| NCT00316407 | Ovarian Epithelial Cancer Stag... more >>e III Stage IV Ovarian Cancer Stage IV Breast Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, New Mexico ... more >> University of New Mexico Cancer Center Albuquerque, New Mexico, United States, 87131 United States, Washington Swedish Medical Center Cancer Institute Seattle, Washington, United States, 98104 Collapse << |
| NCT00404066 | - | - | Completed | - | - |
| NCT00062686 | Neoplasms, Breast | Phase 2 | Completed | - | Argentina ... more >> GSK Investigational Site Buenos Aires, Argentina, C1455BWU France GSK Investigational Site Villejuif Cedex, France, 94805 Germany GSK Investigational Site Augsburg, Bayern, Germany, 86150 United Kingdom GSK Investigational Site Sutton, Surrey, United Kingdom, SM2 5PT GSK Investigational Site Bebington, Wirral, United Kingdom, CH63 4JY GSK Investigational Site London, United Kingdom, SW3 6JJ GSK Investigational Site Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00404066 | Breast Cancer ... more >> Metastatic Breast Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> Santa Clara Valley Medical Center San Jose, California, United States, 95128 Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00379509 | Breast Cancer | Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Collapse << |
| NCT00437073 | Neoplasms, Breast | Phase 2 | Terminated(Lapatinib-topotecan... more >> arm enrollment closed early per protocol amendment 2. Then enrollment into remaining arm terminated due to operational issues.) Collapse << | - | - |
| NCT00756470 | Breast Cancer | Phase 2 | Terminated(Slow accrual.) | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01711658 | Non-HPV Locally Advanced Head ... more >>and Neck Cancer Collapse << | Phase 2 | Active, not recruiting | March 31, 2023 | United States, Alabama ... more >> University of Alabama at Birmingham Comprehensive Cancer Center Birmingham, Alabama, United States, 35294 United States, California University of California, San Diego La Jolla, California, United States, 92093 Sutter General Hospital Sacramento, California, United States, 95816 University of California San Francisco San Francisco, California, United States, 94143 United States, Connecticut Yale University New Haven, Connecticut, United States, 06520 United States, Georgia Emory University Atlanta, Georgia, United States, 30308 United States, Kentucky James Graham Brown Cancer Center at University of Louisville Louisville, Kentucky, United States, 40202 United States, Ohio University Hospitals of Cleveland Cleveland, Ohio, United States, 44106 Ohio State University Medical Center Columbus, Ohio, United States, 43210 United States, Oklahoma University of Oklahoma Health Sciences Center Oklahoma City, Oklahoma, United States, 73190 United States, Pennsylvania Fox Chase Cancer Center Buckingham Furlong, Pennsylvania, United States, 18925 United States, Texas University of Texas Southwestern Medical School Dallas, Texas, United States, 75390 University of Texas - MD Anderson Cancer Center Houston, Texas, United States, 77030-4009 United States, Wisconsin University of Wisconsin Comprehensive Cancer Center Madison, Wisconsin, United States, 53792 Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Canada, Quebec McGill Cancer Centre at McGill University Montreal, Quebec, Canada, H2W 1S6 Collapse << |
| NCT00756470 | - | - | Terminated(Slow accrual.) | - | - |
| NCT00422903 | Neoplasms, Breast | Phase 2 | Completed | - | Italy ... more >> GSK Investigational Site Carpi (MO), Emilia-Romagna, Italy, 41012 GSK Investigational Site Forlì, Emilia-Romagna, Italy, 47100 GSK Investigational Site Modena, Emilia-Romagna, Italy, 41100 GSK Investigational Site Piacenza, Emilia-Romagna, Italy, 29100 GSK Investigational Site Rimini, Emilia-Romagna, Italy, 47900 GSK Investigational Site Treviglio (BG), Lombardia, Italy, 24047 GSK Investigational Site Brindisi, Puglia, Italy, 72100 GSK Investigational Site Pisa, Toscana, Italy, 56126 GSK Investigational Site Chieti, Italy, 66100 GSK Investigational Site Cremona, Italy, 26100 GSK Investigational Site Perugia, Italy, 06156 GSK Investigational Site Reggio Emilia, Italy, 42100 GSK Investigational Site Varese, Italy, 21100 Spain GSK Investigational Site Badalona, Spain, 08916 Collapse << |
| NCT00437073 | - | - | Terminated(Lapatinib-topotecan... more >> arm enrollment closed early per protocol amendment 2. Then enrollment into remaining arm terminated due to operational issues.) Collapse << | - | - |
| NCT00347919 | - | - | Completed | - | - |
| NCT00347919 | Neoplasms, Breast | Phase 2 | Completed | - | - |
| NCT00841828 | - | - | Completed | - | - |
| NCT00841828 | Breast Cancer | Phase 2 Phase 3 | Completed | - | Spain ... more >> Hospital Central de Asturias Oviedo, Asturias, Spain, 33006 Hospital Germans Trias i Pujol Badalona, Barcelona, Spain, 08916 Hospital General de Granollers Granollers, Barcelona, Spain, 08400 Xarxa Asistencial de Manresa Manresa, Barcelona, Spain, 08243 Corporación Sanitaria Parc Taulí Sabadell, Barcelona, Spain, 08208 Hospital del Espíritu Santo Santa Coloma De Gramenet, Barcelona, Spain, 08923 Consorci Sanitari de Terrassa Terrassa, Barcelona, Spain, 08221 Hospital Mutua de Terrassa Terrassa, Barcelona, Spain, 08221 Onkologikoa Donostia-San Sebastián, Guipuzcoa, Spain, 20012 Hospital Xeral Cíes Vigo, Pontevedra, Spain, 36204 Hospital Universitario de Canarias La Laguna, Santa Cruz De Tenerife, Spain, 38320 Complejo Hospitalario Universitario A Coruña A Coruña, Spain, 15006 Centro Oncológico de Galicia A Coruña, Spain, 15009 Hospital del Mar Barcelona, Spain, 08003 Hospital Clinic i Provincial Barcelona, Spain, 08036 Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08041 Hospital General Yagüe Burgos, Spain, 09005 Complejo Hospitalario San Pedro de Alcántara Cáceres, Spain, 10003 Complejo Hospitalario Universitario Reina Sofía Córdoba, Spain, 14004 Complejo Hospitalario de Jaén Jaén, Spain, 23007 Hospital Universitario de la Princesa Madrid, Spain, 28006 Hospital Clínico Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Virgen de la Salud Toledo, Spain, 45004 Hospital Universitario La Fe Valencia, Spain, 46009 Hospital Clinico Universitario de Valencia Valencia, Spain, 46010 Hospital Universitario Miguel Servet Zaragoza, Spain, 50009 Collapse << |
| NCT00085020 | Breast Cancer | Phase 1 | Completed | - | United States, California ... more >> Jonsson Comprehensive Cancer Center, UCLA Los Angeles, California, United States, 90095-1781 Collapse << |
| NCT00338247 | - | - | - | - | - |
| NCT00667251 | Breast Cancer | Phase 3 | Active, not recruiting | December 31, 2020 | - |
| NCT00422903 | - | - | Completed | - | - |
| NCT00439179 | Metastatic Pancreatic Cancer | Phase 1 | Completed | - | United States, Rhode Island ... more >> brown University oncology group Providence, Rhode Island, United States, 02912 Collapse << |
| NCT01005641 | Breast Cancer | Phase 2 | Withdrawn(Study not started fo... more >>r administrative reasons) Collapse << | March 2012 | Italy ... more >> Istituto Nazionale dei Tumori , Divisione di Oncologia Medica B Napoli, Italy, 80131 Istituto Nazionale dei Tumori, Divisione di Oncologia Medica C Napoli, Italy, 80131 Ospedale S. Luca ASL SA 3 Vallo della Lucania, Italy Collapse << |
| NCT00107003 | Glioma Brain ... more >>Tumor Glioblastoma Multiforme GBM Gliosarcoma GS Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00667251 | - | - | Active, not recruiting | - | - |
| NCT01137994 | Neoplasms, Breast | Phase 2 | Withdrawn | March 2018 | - |
| NCT00439179 | - | - | Completed | - | - |
| NCT00272987 | Neoplasms, Breast | Phase 3 | Active, not recruiting | December 31, 2018 | - |
| NCT00075270 | Neoplasms, Breast | Phase 3 | Completed | - | - |
| NCT01160094 | - | - | Completed | - | Colombia ... more >> Novartis Investigative Site Bogota, Colombia Novartis Investigative Site Bogotá, Colombia Novartis Investigative Site Bucaramanga, Colombia Novartis Investigative Site Medellin, Colombia Novartis Investigative Site Pasto, Colombia Novartis Investigative Site Pereira, Colombia Saudi Arabia Novartis Investigative Site Dammam, Saudi Arabia, Dammam 31444 Novartis Investigative Site Jeddah, Saudi Arabia, 21499 Novartis Investigative Site Jeddah, Saudi Arabia, jEDDAH 21423 Novartis Investigative Site Jeddah, Saudi Arabia Novartis Investigative Site Riyadh, Saudi Arabia Collapse << |
| NCT02073487 | Breast Cancer | Phase 2 | Active, not recruiting | June 2019 | United States, Texas ... more >> Houston Methodist Hospital Houston, Texas, United States, 77030 Houston Methodist Hospital Willowbrook Houston, Texas, United States, 77070 Houston Methodist Hospital Sugar Land Sugar Land, Texas, United States, 77479 Collapse << |
| NCT00089999 | Neoplasms, Breast | Phase 2 | Completed | - | - |
| NCT01666431 | Squamous Cell Carcinoma | Phase 2 | Terminated | - | Switzerland ... more >> University Hospital Zurich, Division of Dermatology Zurich, ZH, Switzerland, 8091 Collapse << |
| NCT00089999 | - | - | Completed | - | - |
| NCT00770809 | HER2/Neu Positive ... more >> Male Breast Carcinoma Stage IIA Breast Cancer AJCC v6 and v7 Stage IIB Breast Cancer AJCC v6 and v7 Stage IIIA Breast Cancer AJCC v7 Stage IIIB Breast Cancer AJCC v7 Stage IIIC Breast Cancer AJCC v7 Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT03225937 | Metastatic Colorectal Cancer | Phase 2 | Active, not recruiting | June 2019 | Italy ... more >> Fondazione del Piemonte per l'Oncologia - IRCCS Candiolo, Please Select, Italy, 10060 AOU Policlinico S. Orsola Malpighi Bologna, Italy Grande Ospedale Metropolitano Niguarda Milano, Italy, 20162 Seconda Università di Napoli Napoli, Italy Istituto Oncologico Veneto - IRCCS Padova, Italy Campus Biomedico Roma, Italy AOU Città della Salute e della Scienza di Torino Torino, Italy Collapse << |
| NCT00272987 | - | - | Active, not recruiting | - | - |
| NCT01827163 | HER2-Positive Early Stage Brea... more >>st Cancer Collapse << | Phase 2 | Active, not recruiting | April 2019 | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan-Kettering at Mercy Medical Center Rockville Centre, New York, United States Memorial Sloan-Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01769508 | HER2 Positive Esophagogastric ... more >>Cancer Collapse << | Phase 2 | Terminated(Study closed due to... more >> slow accrual.) Collapse << | - | United States, Florida ... more >> Florida Cancer Specialists - South Fort Myers, Florida, United States, 33916 Florida Hospital Cancer Institute Orlando, Florida, United States, 32804 Woodlands Medical Specialists Pensacola, Florida, United States, 32503 Florida Cancer Specialists-North St. Petersburg, Florida, United States, 33705 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Michigan Grand Rapids Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, Tennessee Chattanooga Oncology and Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology Nashville, Tennessee, United States, 37203 Collapse << |
| NCT01769508 | - | - | Terminated(Study closed due to... more >> slow accrual.) Collapse << | - | - |
| NCT02422199 | HER2 Positive Metastatic Breas... more >>t Cancer Collapse << | Phase 1 Phase 2 | Active, not recruiting | December 2018 | China, Beijing ... more >> Cancer Institute and Hospital, Chinese Academy of Medical Sciences Beijing, Beijing, China, 100021 China 307 Hospital Affiliated to Academy Military Medical Science Beijing, China Collapse << |
| NCT00770809 | - | - | Active, not recruiting | - | - |
| NCT00826267 | - | - | Completed | - | - |
| NCT00826267 | Breast Neoplasms | Phase 2 | Completed | - | United States, Texas ... more >> 1200.44.01001 Boehringer Ingelheim Investigational Site Houston, Texas, United States Brazil 1200.44.12008 Boehringer Ingelheim Investigational Site Brasilia, Brazil 1200.44.12011 Boehringer Ingelheim Investigational Site Cachoeiro de Itapemirim, Brazil 1200.44.12012 Boehringer Ingelheim Investigational Site Campo Grande, Brazil 1200.44.12009 Boehringer Ingelheim Investigational Site Caxias do Sul, Brazil 1200.44.12010 Boehringer Ingelheim Investigational Site Goiania, Brazil 1200.44.12005 Boehringer Ingelheim Investigational Site Ijui, Brazil 1200.44.12007 Boehringer Ingelheim Investigational Site Natal, Brazil 1200.44.12004 Boehringer Ingelheim Investigational Site Novo Hamburgo, Brazil 1200.44.12001 Boehringer Ingelheim Investigational Site Porto Alegre, Brazil 1200.44.12013 Boehringer Ingelheim Investigational Site Porto Alegre, Brazil Colombia 1200.44.14002 Boehringer Ingelheim Investigational Site Bogotá, Colombia 1200.44.14001 Boehringer Ingelheim Investigational Site Cali, Colombia Peru 1200.44.19005 Boehringer Ingelheim Investigational Site Cercado, Peru 1200.44.19001 Boehringer Ingelheim Investigational Site Lima, Peru 1200.44.19004 Boehringer Ingelheim Investigational Site Lima, Peru 1200.44.19003 Boehringer Ingelheim Investigational Site San Isidro, Peru Collapse << |
| NCT00820222 | Metastases, Brain | Phase 3 | Completed | - | - |
| NCT00429299 | - | - | Completed | - | - |
| NCT00820222 | - | - | Completed | - | - |
| NCT00479856 | Relapsed Breast Cancer ... more >> Neoplasms, Breast Collapse << | Phase 2 | Terminated(Study was terminate... more >>d due to difficulty in identifying eligible subjects) Collapse << | - | - |
| NCT00790816 | Neoplasms, Breast | Phase 1 | Completed | - | - |
| NCT00367471 | Neoplasms, Breast | Phase 1 | Active, not recruiting | December 31, 2018 | United States, California ... more >> Novartis Investigative Site La Jolla, California, United States, 92093-0987 United States, Indiana Novartis Investigative Site Indianapolis, Indiana, United States, 46202 United States, North Carolina Novartis Investigative Site Greensboro, North Carolina, United States, 27403 Collapse << |
| NCT00429299 | Neoplasms, Breast | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Berlin, Germany, 13125 Italy GSK Investigational Site Carpi (MO), Emilia-Romagna, Italy, 41012 GSK Investigational Site Forlì, Emilia-Romagna, Italy, 47100 GSK Investigational Site Modena, Emilia-Romagna, Italy, 41100 GSK Investigational Site Parma, Emilia-Romagna, Italy, 43100 GSK Investigational Site Piacenza, Emilia-Romagna, Italy, 29100 GSK Investigational Site Rimini, Emilia-Romagna, Italy, 47900 GSK Investigational Site Treviglio (BG), Lombardia, Italy, 24047 GSK Investigational Site Candiolo (TO), Piemonte, Italy, 10060 GSK Investigational Site Brindisi, Puglia, Italy, 72100 GSK Investigational Site Pisa, Toscana, Italy, 56126 GSK Investigational Site Cremona, Italy, 26100 GSK Investigational Site Pavia, Italy, 27100 GSK Investigational Site Perugia, Italy, 06156 GSK Investigational Site Reggio Emilia, Italy, 42100 GSK Investigational Site Varese, Italy, 21100 Poland GSK Investigational Site Warszawa, Poland, 00-909 Collapse << |
| NCT00662636 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 United States, Florida Mayo Clinic in Florida Jacksonville, Florida, United States, 32224-9980 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00479856 | - | - | Terminated(Study was terminate... more >>d due to difficulty in identifying eligible subjects) Collapse << | - | - |
| NCT00490139 | Neoplasms, Breast | Phase 3 | Active, not recruiting | July 1, 2021 | - |
| NCT00424255 | - | - | Completed | - | - |
| NCT00424255 | Neoplasms, Head and Neck | Phase 3 | Completed | - | - |
| NCT02142036 | Metastatic Cancer | Phase 2 | Active, not recruiting | January 2022 | Norway ... more >> Akershus University Hospital Lillestrøm, Norway, 1478 The Norwegian Radium Hospital Oslo, Norway, 0379 Collapse << |
| NCT02551718 | Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << |
| NCT03262935 | Metastatic Breast Cancer | Phase 3 | Recruiting | May 2021 | - |
| NCT01494662 | Breast Cancer | Phase 2 | Recruiting | December 2023 | United States, California ... more >> University of California, San Francisco Medical Center Active, not recruiting San Francisco, California, United States, 94115 United States, District of Columbia MedStar Georgetown Univeristy Hospital Active, not recruiting Washington, District of Columbia, United States, 20007 United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Active, not recruiting Baltimore, Maryland, United States, 21231 United States, Massachusetts Beth Israel Deaconess Medical Center Active, not recruiting Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Rachel Freedman, MD Principal Investigator: Rachel Freedman, MD, MPH Massachusetts General Hosptial Recruiting Boston, Massachusetts, United States, 02215 Contact: Beverly Moy, MD Principal Investigator: Beverly Moy, MD United States, Michigan University of Michigan Comprehensive Cancer Center Active, not recruiting Ann Arbor, Michigan, United States, 48109 United States, North Carolina University of North Carolina at Chapel Hill - Lineberger Comprehensive Cancer Center Active, not recruiting Chapel Hill, North Carolina, United States, 27599 Duke University Medical Center Active, not recruiting Durham, North Carolina, United States, 27710 United States, Pennsylvania UPMC Cancer Centers - Magee-Womens Hospital of UPMC Active, not recruiting Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Baylor College of Medicine Lester and Sue Smith Breast Center Active, not recruiting Houston, Texas, United States, 77030 MD Anderson Cancer Center Active, not recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT02208583 | Hormone Refractory Prostate Ca... more >>ncer Collapse << | Not Applicable | Unknown | December 2016 | China, Tianjin ... more >> Department of Interventional Oncology, Tianjin Medical University Cancer Institute and Hospital Recruiting Tianjin, Tianjin, China, 300060 Contact: Wang HaiTao, Ph.D +86-18630955984 peterrock2000@126.com Principal Investigator: Wang HaiTao, Ph.D Collapse << |
| NCT01636908 | Cancer Advanc... more >>ed Solid Tumors Collapse << | Not Applicable | Active, not recruiting | - | Netherlands ... more >> VUMedical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT00490139 | - | - | Active, not recruiting | - | - |
| NCT02836847 | Cholangiocarcinoma of the Extr... more >>ahepatic Bile Duct Gallbladder Cancer Collapse << | Phase 2 | Recruiting | December 2020 | China, Shanghai ... more >> Xinhua Hospital Recruiting Shanghai, Shanghai, China, 200092 Contact: yingbin liu 13918803900 laoniulyb@163.com Collapse << |
| NCT01419197 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT02645149 | Melanoma | Phase 4 | Not yet recruiting | August 2021 | Australia, New South Wales ... more >> Royal Prince Alfred Hospital Not yet recruiting Camperdown, New South Wales, Australia, 2050 Contact: Maria Gonzalez 612 9911 7200 maria.gonzalez@melanoma.org.au Principal Investigator: John Thompson Westmead Hospital Westmead, New South Wales, Australia, 2145 Melanoma Institute Australia Not yet recruiting Wollstonecraft, New South Wales, Australia, 2065 Contact: Maria Gonzalez +612 9911 7200 maria.gonzalez@melanoma.org.au Contact: Alex Menzies +612 9911 7200 info@melanoma.org.au Sub-Investigator: Georgina Long Principal Investigator: Alex Menzies Collapse << |
| NCT01594177 | Unilateral HER2 Positive Breas... more >>t Cancer Collapse << | Phase 2 | Completed | - | Germany ... more >> Rotkreuzklinikum München München, Germany, 80634 Collapse << |
| NCT01419197 | - | - | Completed | - | - |
| NCT01098500 | - | - | Completed | - | - |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01098500 | - | - | Completed | - | - |
| NCT03768375 | Cholangiocarcinoma of the Extr... more >>ahepatic Bile Duct Gallbladder Cancer Collapse << | Phase 2 | Recruiting | December 30, 2021 | China, Shanghai ... more >> Xinhua Hospital Recruiting Shanghai, Shanghai, China, 200092 Contact: Yingbin Liu, PHD 13918803900 laoniulyb@163.com Principal Investigator: yingbin liu, PHD Collapse << |
| NCT01466504 | - | - | Terminated(Accrual did not mee... more >>t expectations. Over nearly 2 years just 4 subjects were treated on study. The goal of 30 subjects was not attainable.) Collapse << | - | United States, New Hampshire ... more >> Dartmouth-Hitchcock Medical Center, Norris Cotton Cancer Center Lebanon, New Hampshire, United States, 03756 United States, Vermont White River Junction VA Medical Center White River Junction, Vermont, United States, 05009 Collapse << |
| NCT03755141 | HER2-positive Breast Cancer ... more >> Metastatic Breast Cancer Collapse << | Phase 2 | Not yet recruiting | June 30, 2022 | - |
| NCT01431196 | Stage II Breast Cancer ... more >> Stage III Breast Cancer Collapse << | Phase 2 | Completed | - | Spain ... more >> Clínica Universidad de Navarra Pamplona, Navarra, Spain, 31008 Collapse << |
| NCT01152853 | Advanced Gastric Cancer ... more >> HER2 Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01565200 | HER-2 Positive Breast Cancer | Phase 2 | Active, not recruiting | December 2019 | Belgium ... more >> Universitair Ziekenhuis Antwerpen (UZA) Antwerpen, Edegem, Belgium, 2650 Institut Jules Bordet Brussels, Belgium, 1000 Netherlands Vrije Universiteit Amsterdam (VUMC) Amsterdam, Netherlands, 1081HV University Medical Center Groningen (UMCG) Groningen, Netherlands, 9700RB Radboud University Medical Centre Nijmegen (UMCN) Nijmegen, Netherlands, 6525 GA Collapse << |
| NCT01771458 | Reccurent/Metastatic Solid Tum... more >>or Disease Collapse << | Phase 2 | Unknown | December 2016 | France ... more >> Centre régional de lutte contre le cancer de Bourgogne Georges François Leclerc Dijon, France, 21079 Centre Leon Berard Lyon, France, 69373 Institut Paoli Calmettes Marseille, France, 13009 Insitut Curie Paris, France, 75248 Institut Curie Hopital Rene Huguenin Saint-cloud, France, 92210 Institut de cancérologie de l'Ouest Centre René Gauducheau Saint-herblain, France, 44000 Institut Claudius Régaud Toulouse, France, 31052 Centre Alexis Vautrin Vandoeuvre Les Nancy, France, 54500 Collapse << |
| NCT01064349 | - | - | Completed | - | Philippines ... more >> GSK Investigational Site Pasay City, Philippines, 1300 GSK Investigational Site Quezon City, Philippines, 1101 GSK Investigational Site Quezon City, Philippines, 1102 Singapore GSK Investigational Site Singapore, Singapore, 119074 Thailand GSK Investigational Site Bangkok, Thailand, 10110 GSK Investigational Site Bangkok, Thailand, 10310 GSK Investigational Site Bangkok, Thailand, 10330 GSK Investigational Site Bangkok, Thailand, 10400 GSK Investigational Site Bangkok, Thailand, 10700 GSK Investigational Site Chiangmai, Thailand, 50200 Collapse << |
| NCT00363194 | Carcinoma, Renal Cell | Phase 1 | Completed | - | United States, Indiana ... more >> GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48201 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| EGFR | IC50:10.8nM | |
| HER2 | IC50:9.2nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
