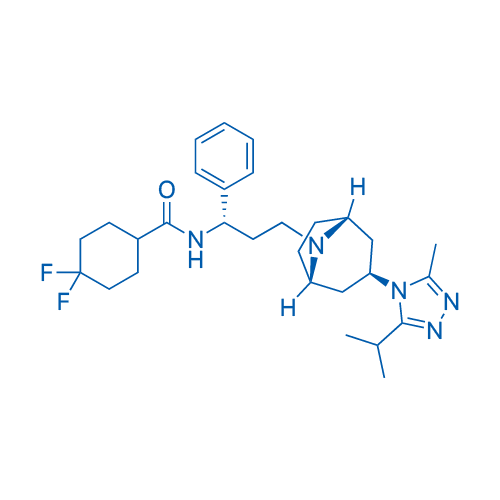
CAS No.: 376348-65-1
Maraviroc/马拉韦罗 Catalog No. CSN11361
Synonyms: UK-427857;Selzentry;Celsentri
Maraviroc is a selective CCR5 antagonist (IC50= 6.4 nM) and displays potent anti-HIV-1 activity.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 CCR5
IC50:6.4nM- 描述
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice: 10 mg/kg[3] (i.p.); 50 mg/kg[3] (p.o., BID) Rat: 1 mg/kg - 5 mg/kg[4] (i.p.)
给药途径 i.p., p.o
动物药代数据 Animal Rats[5] Dogs[5] Dose 1 mg/kg (i.v.)
10 mg/kg (p.o.)0.5 mg/kg (i.v.)
2 mg/kg (p.o.)Administration i.v.
p.o.i.v.
p.o.F 100% (i.v.)
5% (p.o.)100% (i.v.)
40% (p.o.)Vd 6.5 L/kg (i.v.) 4.3 L/kg (i.v.) T1/2 0.9 h (i.v.) 2.3 h (i.v.) Tmax 2 min (p.o.) 0.75 min (p.o.) CL 74 ml/min/kg (i.v.) 21 ml/min/kg (i.v.) Cmax 55 ng/ml (p.o.) 256 ng/ml (p.o.) - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02519777 HIV Infections Phase 4 Active, not recruiting November 30, 2020 - NCT02333045 HIV Not Applicable Terminated(Minimal efficacy of... more >> maraviroc alone was found in preliminary data analysis of another study.) Collapse << - United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Grady Infectious Diseases Clinic (Ponce Clinic) Atlanta, Georgia, United States, 30308 Collapse << NCT00925756 HIV Infections Phase 4 Completed - United States, California ... more >> University Southern California Los Angeles, California, United States, 90033 University California San Diego San Diego, California, United States, 92103 Harbor-UCLA Torrance, California, United States, 90502 Collapse << - 更多
- 参考文献
- [1] Neff CP, Kurisu T, et al. A topical microbicide gel formulation of CCR5 antagonist maraviroc prevents HIV-1 vaginal transmission in humanized RAG-hu mice. PLoS One. 2011;6(6):e20209.
- [2] Dorr P, Westby M, et al. Maraviroc (UK-427,857), a potent, orally bioavailable, and selective small-molecule inhibitor of chemokine receptor CCR5 with broad-spectrum anti-human immunodeficiency virus type 1 activity. Antimicrob Agents Chemother. 2005 Nov;49(11):4721-32.
- [3] Mencarelli A, Graziosi L, et al. CCR5 Antagonism by Maraviroc Reduces the Potential for Gastric Cancer Cell Dissemination. Transl Oncol. 2013 Dec 1;6(6):784-93. eCollection 2013 Dec 1.
- [4] Nayak SU, Cicalese S, et al. Chemokine CCR5 and cocaine interactions in the brain: Cocaine enhances mesolimbic CCR5 mRNA levels and produces place preference and locomotor activation that are reduced by a CCR5 antagonist. Brain Behav Immun. 2020 Jan;83:288-292.
- [5] UK-427857
- [6] Woollard SM, Kanmogne GD. Maraviroc: a review of its use in HIV infection and beyond. Drug Des Devel Ther. 2015 Oct 1;9:5447-68.
- [7] Dorr P, Westby M, Dobbs S, Griffin P, Irvine B, Macartney M, Mori J, Rickett G, Smith-Burchnell C, Napier C, Webster R, Armour D, Price D, Stammen B, Wood A, Perros M. Maraviroc (UK-427,857), a potent, orally bioavailable, and selective small-molecule inhibitor of chemokine receptor CCR5 with broad-spectrum anti-human immunodeficiency virus type 1 activity. Antimicrob Agents Chemother. 2005 Nov;49(11):4721-32.
- [8] Arberas H, Guardo AC, Bargalló ME, Maleno MJ, Calvo M, Blanco JL, García F, Gatell JM, Plana M. In vitro effects of the CCR5 inhibitor maraviroc on human T cell function. J Antimicrob Chemother. 2013 Mar;68(3):577-86.
- [9] Neff CP, Kurisu T, Ndolo T, Fox K, Akkina R. A topical microbicide gel formulation of CCR5 antagonist maraviroc prevents HIV-1 vaginal transmission in humanized RAG-hu mice. PLoS One. 2011;6(6):e20209.
- [10] Ochoa-Callejero L, Pérez-Martínez L, Rubio-Mediavilla S, Oteo JA, Martínez A, Blanco JR. Maraviroc, a CCR5 antagonist, prevents development of hepatocellular carcinoma in a mouse model. PLoS One. 2013;8(1):e53992.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 376348-65-1 | 储存条件 |
|
|||||||||||||
| 分子式 | C29H41F2N5O | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 513.67 | 别名 | UK-427857;Selzentry;Celsentri;UK-427,857;Pfizer Brand of maraviroc | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| CHO cells | - | Function assay | 2 h | Displacement of [128I]RANTES from human CCR5 receptor coexpressed with Galphai6 in CHO cells after 2 hrs by scintillation counting, IC50=1.4 nM | 20137937 |
| HEK293 cells | - | Function assay | - | Inhibition of human MATE1-mediated ASP+ uptake expressed in HEK293 cells after 1.5 mins by fluorescence assay, IC50=17.3 Μm | 23241029 |
| HeLa-P4 cells | - | Function assay | 20 days | Antagonist activity at CCR5 receptor expressed in HeLa-P4 cells co-expressing CD4 assessed as inhibition of infusion to HIV gp120 expressed in CHO-tat10 cells after 20 hrs by cell-cell fusion assay, IC50=0.2 nM | 21128663 |
| HOS cells | - | Function assay | - | Antiviral activity against HIV1 Ba-L infected in HOS cells assessed as inhibition of viral infection, IC50=2 nM | 19664920 |
| human astrocytes | 100 nM | Function assay | 30 to 60 mins | Binding affinity to CCR5/MOR in human astrocytes assessed as inhibition of R5 HIV-1 SF162 infection by measuring Tat protein expression at 100 nM preincubated for 30 to 60 mins followed by viral infection measured after 18 hrs by luciferase reporter gene assay | 23682308 |
| human TZM-bl cells | - | Function assay | 48 h | Antagonist activity against CCR5 receptor in human TZM-bl cells assessed as inhibition of HIV-1 MGC26-induced cell-cell fusion between viral envelope protein expressing human HEK293 cells to TZM-bl cells after 48 hrs by luciferase reporter gene assay, IC50=0.37 nM | 24563723 |
| rhesus monkey Chem-1 cell | - | Function assay | 120 mins | Displacement of [125I]-MIP-1alpha from CCR5 in rhesus monkey Chem-1 cell membranes after 120 mins by liquid scintillation counting analysis, Ki=0.24 nM | 23682308 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02519777 | HIV Infections | Phase 4 | Active, not recruiting | November 30, 2020 | - |
| NCT02333045 | HIV | Not Applicable | Terminated(Minimal efficacy of... more >> maraviroc alone was found in preliminary data analysis of another study.) Collapse << | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Grady Infectious Diseases Clinic (Ponce Clinic) Atlanta, Georgia, United States, 30308 Collapse << |
| NCT00925756 | HIV Infections | Phase 4 | Completed | - | United States, California ... more >> University Southern California Los Angeles, California, United States, 90033 University California San Diego San Diego, California, United States, 92103 Harbor-UCLA Torrance, California, United States, 90502 Collapse << |
| NCT02333045 | - | - | Terminated(Minimal efficacy of... more >> maraviroc alone was found in preliminary data analysis of another study.) Collapse << | - | - |
| NCT00717067 | Human Immunodeficiency Virus (... more >>HIV) Infection Collapse << | Phase 4 | Completed | - | Germany ... more >> Pfizer Investigational Site Berlin, Germany, 10117 Pfizer Investigational Site Muenchen, Germany, 81241 Collapse << |
| NCT00717067 | - | - | Completed | - | - |
| NCT01637233 | - | - | Completed | - | Argentina ... more >> Hospital Ramos Mejía Buenos Aires, Argentina, C1221ADC Fundación IDEAA Buenos Aires, Argentina, C1405CKC CAICI Rosario, Argentina Thailand Chulalongkorn University Hospital Bangkok, Thailand, 10330 United Kingdom Brighton & Sussex University NHS Trust Brighton, Sussex, United Kingdom, BN21ES, Imperial Healthcare, St. Mary's Hospital London, United Kingdom, W2 Collapse << |
| NCT01533272 | HIV Infection | Phase 4 | Completed | - | Spain ... more >> Hospital Clinic de Barcelona Barcelona, Spain, 08036 Collapse << |
| NCT02486510 | HIV-1 Infection | Early Phase 1 | Terminated(Futility criteria) | - | Spain ... more >> Hospital Universitario Ramon Y Cajal Madrid, Spain, 28034 Hospital Universitario La Paz Madrid, Spain, 28046 Collapse << |
| NCT00850395 | - | - | Completed | - | - |
| NCT00795444 | HIV-1 | Phase 2 | Completed | - | Spain ... more >> Hospital Universitario Ramon Y Cajal Madrid, Spain, 28034 Collapse << |
| NCT00850395 | - | - | Completed | - | - |
| NCT00808002 | HIV Infections | Phase 3 | Completed | - | Spain ... more >> Hospital Germans Trias i Pujol Badalona, Barcelona, Spain, 08916 Hospital Clinic i Provincial de Barcelona Barcelona, Spain, 08916 Collapse << |
| NCT00844519 | - | - | Completed | - | - |
| NCT01785810 | Hematologic Malignancy | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00844519 | HIV Infection ... more >> Cardiovascular Disease Inflammation HIV Infections Collapse << | Phase 3 | Completed | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94110 Collapse << |
| NCT00634959 | HIV | Phase 2 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmingham, Alabama, United States, 35294-2050 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02215 Germany Pfizer Investigational Site Frankfurt, Germany, 60596 Pfizer Investigational Site Hamburg, Germany, 20099 Pfizer Investigational Site Koeln, Germany, 50931 United Kingdom Pfizer Investigational Site London, United Kingdom, NW3 2QG Pfizer Investigational Site London, United Kingdom, SW10 9NH Collapse << |
| NCT01585753 | - | - | Completed | - | Argentina ... more >> Hospital General de Agudos "Dr. José María Ramos Mejía" Buenos Aires, Argentina, C1221ADC Fundacion IDEAA Buenos Aires, Argentina, C1405CKC Hospital Italiano de Buenos Aires Buenos Aires, Argentina Australia, New South Wales St. Vincent's Hospital Sydney, New South Wales, Australia, 2010 Germany Johann Wolfgang Goethe-University Hospital, Medical HIVCENTER Frankfurt, Frankfurt am Main, Germany, 60590 Thailand Chulalongkorn University Hospital Bangkok, Thailand, 10330 Collapse << |
| NCT01776996 | - | - | - | - | Argentina ... more >> GSK Investigational Site Rosario, Santa Fe, Argentina, 2000 GSK Investigational Site Buenos Aires, Argentina, 1202 GSK Investigational Site Buenos Aires, Argentina, C1264AAV GSK Investigational Site Buenos Aires, Argentina, C1282AEN Belgium GSK Investigational Site Brussels, Belgium, 1000 GSK Investigational Site Gent, Belgium, 9000 GSK Investigational Site Leuven, Belgium, 3000 Italy GSK Investigational Site Milano, Lombardia, Italy, 20127 GSK Investigational Site Modena, Italy, 41124 Poland GSK Investigational Site Bydgoszcz, Poland, 85-030 Russian Federation GSK Investigational Site Moscow, Russian Federation, 105275 GSK Investigational Site Moscow, Russian Federation, 129110 GSK Investigational Site N.Novgorod, Russian Federation, 603005 GSK Investigational Site Saint-Petersburg, Russian Federation, 190103 GSK Investigational Site Smolensk, Russian Federation, 214006 GSK Investigational Site St. Petersburg, Russian Federation, 196645 South Africa GSK Investigational Site Port Elizabeth, Eastern Cape, South Africa, 6001 GSK Investigational Site Soweto, Gauteng, South Africa, 2013 GSK Investigational Site Bloemfontein, South Africa, 9301 GSK Investigational Site Dundee, South Africa, 3000 GSK Investigational Site Gauteng, South Africa, 2047 GSK Investigational Site Port Elizabeth, South Africa, 6070 GSK Investigational Site Pretoria North, South Africa, 0116 GSK Investigational Site Pretoria, South Africa, 0083 GSK Investigational Site Westdene, South Africa, 2092 Switzerland GSK Investigational Site Bern, Switzerland, CH-3010 GSK Investigational Site Lugano, Switzerland, 6900 GSK Investigational Site Zuerich, Switzerland, 8091 Collapse << |
| NCT03118661 | HIV-1-infection | Phase 1 | Withdrawn(No accrual to the st... more >>udy) Collapse << | September 30, 2025 | - |
| NCT01204905 | HIV Infections | Not Applicable | Completed | - | United States, Maryland ... more >> University of Maryland, Institute of Human Virology Baltimore, Maryland, United States, 21201 Collapse << |
| NCT03274804 | Metastatic Colorectal Cancer ... more >> MSS Collapse << | Phase 1 | Active, not recruiting | November 1, 2019 | Germany ... more >> National Center for Tumor Diseases, University Hospital Heidelberg Heidelberg, Germany Collapse << |
| NCT00478231 | - | - | Completed | - | - |
| NCT00478231 | Acquired Immunodeficiency Synd... more >>rome HIV Infection Collapse << | Phase 3 | Completed | - | Brazil ... more >> Pfizer Investigational Site Salvador, BA, Brazil, 40110-160 Pfizer Investigational Site Brasilia, DF, Brazil, 70351-580 Pfizer Investigational Site Belo Horizonte, MG, Brazil, 30130-100 Pfizer Investigational Site Curitiba, PR, Brazil, 80240-280 Pfizer Investigational Site Nova Iguaçu, RJ, Brazil, 26030-380 Pfizer Investigational Site Porto Alegre, RS, Brazil, 90110-270 Pfizer Investigational Site Florianopolis, SC, Brazil, 88025-301 Pfizer Investigational Site Campinas, SP, Brazil, 13059-900 Pfizer Investigational Site Campinas, SP, Brazil, 13083-887 Pfizer Investigational Site Ribeirao Preto, SP, Brazil, 14048900 Pfizer Investigational Site Santo Andre, SP, Brazil, 09060-650 Pfizer Investigational Site Sao Paulo, SP, Brazil, 01246-900 Pfizer Investigational Site Sao Paulo, SP, Brazil, 01307-001 Pfizer Investigational Site Sao Paulo, SP, Brazil, 04040-002 Pfizer Investigational Site Sao Paulo, SP, Brazil, 04121-000 Pfizer Investigational Site São Paulo, SP, Brazil, 04231-030 Collapse << |
| NCT01013987 | HIV-1 Adults Patients ... more >> AIDS Triple Class Failure Collapse << | Phase 4 | Unknown | - | Argentina ... more >> Fundación Huésped Recruiting Ciudad de Buenos Aires, Argentina, C1202ABB Contact: María C. Magneres, MD 54-11-4981-7777 ext 114 maria.magneres@huesped.org.ar Contact: Patricia Luz Patterson, MD 54-11-4981-7777 ext 114 patricia.patterson@huesped.org.ar Principal Investigator: Alejandro Krolewiecki, MD Collapse << |
| NCT01275625 | HIV | Phase 4 | Completed | - | Russian Federation ... more >> Regional Center on AIDS and Infectious Diseases Prophylaxis and Control Krasnoyarsk, Russian Federation, 660049 Federal scientific and methodological center on AIDS prophylaxis and control Moscow, Russian Federation, 105275 Moscow regional center on AIDS and infectious diseases prophylaxis and control Moscow, Russian Federation, 129110 Regional Center on AIDS and Infectious Diseases Prophylaxis and Control Nizhnij Novgorod, Russian Federation, 603005 Saint-Petersburg Center on AIDS and Infectious Diseases Prophylaxis and Control Saint-Petersburg, Russian Federation, 190103 Federal State Institution Republican clinical infectious hospital of Roszdrav Saint-Petersburg, Russian Federation, 196645 Smolensk Center on AIDS and infectious diseases prophylaxis and control Smolensk, Russian Federation, 214006 Volgograd Regional Center on AIDS and Infectious Diseases Prophylaxis and Control Volgograd, Russian Federation, 400040 Collapse << |
| NCT00988780 | Immune Reconstitution Inflamma... more >>tory Syndrome HIV HIV Infections Collapse << | Not Applicable | Unknown | April 2013 | United States, Maryland ... more >> NIH/NIAD Bethesda, Maryland, United States, 20892 United States, Ohio Center for AIDS Research. Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Pennsylvania Center for Clinical Epidemiology and Biostatistics. University of Pennsylvania School of Medicine Philadelphia, Pennsylvania, United States, 19104 HIV-1 Immunopathogenesis Laboratory. The Wistar Institute Philadelphia, Pennsylvania, United States, 19104 Mexico Hospital General de León Leon, Guanajuato, Mexico, 37230 Hospital Civil de Guadalajara Guadalajara, Jalisco, Mexico, 44280 Hospital General de México Mexico City, Mexico, 06726 Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán Mexico City, Mexico, 14000 Hospital Central Dr. Ignacio Morones Prieto San Luis Potosí, Mexico, 78240 South Africa Clinical HIV Research Unit. Themba Lethu Clinic. Helen Joseph Hospital Johannesburg, Gauteng, South Africa, 2092 Collapse << |
| NCT01680536 | HIV | Phase 4 | Completed | - | United Kingdom ... more >> St Stephen's AIDS Trust London, United Kingdom, SW10 9TH Collapse << |
| NCT01275625 | - | - | Completed | - | - |
| NCT01204905 | - | - | Completed | - | - |
| NCT03129113 | Hepatic Steatosis ... more >> HIV-1-infection Collapse << | Phase 2 Phase 3 | Recruiting | November 2020 | United Kingdom ... more >> Royal Free Hospital Recruiting London, United Kingdom, NW3 2QG Contact: PI St Thomas' Hospital Recruiting London, United Kingdom, SE1 9RT Contact: PI King's College Hospital Recruiting London, United Kingdom, SE5 9RJ Contact: PI UCL Mortimer Market Centre Recruiting London, United Kingdom, WC1E 6JB Contact: PI Collapse << |
| NCT01597648 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 1 | Completed | - | - |
| NCT00427934 | Arthritis, Rheumatoid | Phase 2 | Terminated(See Detailed Descri... more >>ption.) Collapse << | - | - |
| NCT00987948 | - | - | Completed | - | - |
| NCT00987948 | HIV Infections | Phase 2 | Completed | - | United States, Hawaii ... more >> Hawaii Center for AIDS Honolulu, Hawaii, United States, 96816 Collapse << |
| NCT00426660 | HIV Infections | Phase 3 | Completed | - | - |
| NCT01290211 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT01242579 | HIV Infections | Phase 1 | Withdrawn(In order to focus ef... more >>forts on the combination ring formulation, IPM decided not to move forward with this trial.) Collapse << | - | - |
| NCT00771823 | HIV Infections | Phase 1 | Completed | - | United Kingdom ... more >> Chelsea and Westminster Hospital London, United Kingdom, SW10 9TH Collapse << |
| NCT00992654 | - | - | - | - | - |
| NCT00426660 | - | - | Completed | - | - |
| NCT00966329 | HIV HIV Infec... more >>tions Collapse << | Phase 4 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Lluita contra la Sida Foundation, HIV Unit, Irsi Caixa Foundation Badalona, Barcelona, Spain, 08916 Lluita contra la Sida Foundation, HIV Unit Badalona, Barcelona, Spain, 08916 Collapse << |
| NCT00427934 | - | - | Terminated(See Detailed Descri... more >>ption.) Collapse << | - | - |
| NCT01363037 | HIV Infections | Phase 1 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Massachusetts The Fenway Institute/Fenway Community Health Boston, Massachusetts, United States, 02115 United States, Pennsylvania Magee-Womens Hospital of UPMC Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00870363 | - | - | Completed | - | - |
| NCT01140412 | Healthy | Phase 1 | Terminated(The trial premature... more >>ly terminated on November 1, 2010, due to healthy volunteer participants experiencing non-serious fosamprenavir-related skin rash.) Collapse << | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT00870363 | HIV Infections | Phase 4 | Completed | - | United States, California ... more >> CARES Clinic Sacramento, California, United States, 95811 Collapse << |
| NCT01345630 | HIV-1 | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01367210 | HIV Infection | Phase 4 | Terminated(Because a higher ra... more >>te of virological failures in study versus control arm.) Collapse << | - | Italy ... more >> Catholic University of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT00782301 | Hepatitis B H... more >>uman Immunodeficiency Virus Hepatitis C, Chronic Collapse << | Phase 4 | Withdrawn | - | United States, Florida ... more >> Pfizer Investigational Site Clearwater, Florida, United States, 33765 Pfizer Investigational Site Orlando, Florida, United States, 32803 Pfizer Investigational Site Safety Harbor, Florida, United States, 34695 Pfizer Investigational Site Wilton Manors, Florida, United States, 33305 United States, Georgia Pfizer Investigational Site Atlanta, Georgia, United States, 30308 Pfizer Investigational Site Atlanta, Georgia, United States, 30312 United States, Louisiana Pfizer Investigational Site New Orleans, Louisiana, United States, 70112 United States, Massachusetts Pfizer Investigational Site Worcester, Massachusetts, United States, 01605 Pfizer Investigational Site Worcester, Massachusetts, United States, 01655 United States, New York Pfizer Investigational Site Mt. Vernon, New York, United States, 10550 United States, Oklahoma Pfizer Investigational Site Tulsa, Oklahoma, United States, 74135 United States, Texas Pfizer Investigational Site Bellaire, Texas, United States, 77401 Pfizer Investigational Site Conroe, Texas, United States, 77301 Pfizer Investigational Site Dallas, Texas, United States, 75390 Pfizer Investigational Site Stafford, Texas, United States, 77477 Canada, British Columbia Pfizer Investigational Site Vancouver, British Columbia, Canada, V6Z 2C7 Canada, Ontario Pfizer Investigational Site Toronto, Ontario, Canada, M5G 2N2 Poland Pfizer Investigational Site Bydgoszcz, Poland, 85-030 Collapse << |
| NCT01291459 | HIV | Phase 2 | Unknown | December 2015 | France ... more >> Cannes hosipital CAnnes, France, 06400 CHU Clermont-Ferrand, France, 63003 Frejus hospital Frejus, France, 83608 Edourad Herriot hospital Lyon, France, 69003 Croix Rousse hospital Lyon, France, 69004 Ste MArguerite Hospital MArseille, France, 13009 Conception hospital Marseille, France, 13385 Hotel Dieu hospital Nantes, France, 44093 Hopital l'Archet 1 Nice, France, 06202 St Louis Hospital Paris, France, 75010 Pitie Salpetriere Hospital Paris, France, 75013 Nord Hospital St Etienne, France, 42277 Collapse << |
| NCT01749566 | HIV | Phase 1 | Completed | - | United States, Georgia ... more >> Grady Infectious Diseases Program Atlanta, Georgia, United States, 30303 Collapse << |
| NCT01068873 | HIV Infections | Phase 4 | Terminated(Poor enrollment) | - | United States, Pennsylvania ... more >> Temple General Internal Medicine Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT02778204 | HIV Infections | Phase 1 | Recruiting | July 31, 2020 | United States, California ... more >> University of California, UC San Diego CRS- Mother-Child-Adolescent HIV Program Recruiting La Jolla, California, United States, 92093-0672 Contact: Megan Loughran, B.A. 858-534-9218 meloughran@ucsd.edu Usc La Nichd Crs Recruiting Los Angeles, California, United States, 90089 Contact: Eva A. Operskalski, Ph.D. 323-865-1554 eva@usc.edu United States, Colorado Univ. of Colorado Denver NICHD CRS Recruiting Aurora, Colorado, United States, 80045 Contact: Emily Barr, C.P.N.P., C.N.M., M.S.N. 720-777-6752 emily.barr@childrenscolorado.org United States, Illinois Rush Univ. Cook County Hosp. Chicago NICHD CRS Recruiting Chicago, Illinois, United States, 60612 Contact: Maureen McNichols, R.N., M.S.N., C.C.R.C. 312-572-4541 maureen_mcnichols@rush.edu Lurie Children's Hospital of Chicago (LCH) CRS Recruiting Chicago, Illinois, United States, 60614-3393 Contact: Margaret Ann Sanders, M.P.H. 312-227-8275 msanders@luriechildrens.org United States, Massachusetts Boston Medical Center Ped. HIV Program NICHD CRS Recruiting Boston, Massachusetts, United States, 02118 Contact: Debra McLaud, R.N. 617-414-5813 demclaud@bmc.org United States, Tennessee St. Jude Children's Research Hospital CRS Recruiting Memphis, Tennessee, United States, 38105-3678 Contact: Jill Utech, M.S.N. 901-595-5059 jill.utech@stjude.org Kenya Kenya Medical Research Institute / Walter Reed Project Clinical Research Center, Kericho CRS Recruiting Kericho, Kenya, 20200 Contact: Samwel K. Chirchir, R.N., B.Sc. 254-522-030388 Samwel.Chirchir@usamru-k.org South Africa Soweto IMPAACT CRS Recruiting Johannesburg, Gauteng, South Africa, 1862 Contact: Nasreen Abrahams, M.B.A., B.Tech 27-11-9899700 abrahamsn@phru.co.za Umlazi CRS Recruiting Durban, Kwa Zulu Natal, South Africa, 4001 Contact: Vani Chetty 27-31-2601998 Chettyv1@ukzn.ac.za Thailand Siriraj Hospital ,Mahidol University NICHD CRS Recruiting Bangkok, Bangkoknoi, Thailand, 10700 Contact: Watcharee Lermankul 66-2-4197000 ext 5695 watchareeped@gmail.com Uganda MU-JHU Research Collaboration (MUJHU CARE LTD) CRS Recruiting Kampala, Uganda Contact: Carolyne P. Onyango, MB ChB, M.S. 256-414-541044 carolonyango@mujhu.org Collapse << |
| NCT01345630 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00864474 | - | - | Active, not recruiting | December 24, 2018 | - |
| NCT01049204 | HIV Infections | Phase 4 | Terminated(The data from the f... more >>irst 48 patients recruited has now been analysed, and it has been determined that it is sufficient to meet the study objectives.) Collapse << | - | United Kingdom ... more >> St Stephen's Centre London, United Kingdom, SW10 9NH Collapse << |
| NCT00993148 | HIV-1 Infection ... more >> HIV Infections Collapse << | Phase 2 | Completed | - | United States, California ... more >> Quest Clinical Research San Francisco, California, United States United States, Florida University of Miami Miami, Florida, United States United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 CORECenter Chicago, Illinois, United States, 60612 United States, Nebraska University of Nebraska Omaha, Nebraska, United States Collapse << |
| NCT00703586 | HIV Infections | Phase 1 | Completed | - | United States, New York ... more >> Rockefeller University Hospital New York, New York, United States, 10065 Collapse << |
| NCT00981318 | HIV Infections ... more >> Acquired Immunodeficiency Syndrome Collapse << | Phase 4 | Terminated(unable to enroll ex... more >>pected number of subjects) Collapse << | - | United States, Florida ... more >> Barry M. Rodwick, M. D. Safety Harbor, Florida, United States, 34695 Collapse << |
| NCT00827112 | Human Immunodeficiency Virus-1 | Phase 2 | Completed | - | - |
| NCT00993148 | - | - | Completed | - | - |
| NCT01327547 | HIV Coinfection | Phase 4 | Completed | - | - |
| NCT02134717 | Sarcoidosis | Not Applicable | Terminated(Poor recruitment) | - | United States, Pennsylvania ... more >> Dorothy P. and Richard P. Simmons center for Interstitial lung Disease at the University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00735072 | HIV Infection | Phase 4 | Completed | - | United States, California ... more >> University of California San Francisco - San Francisco General Hospital San Francisco, California, United States, 94110 Stanford University Stanford, California, United States, 94305 United States, Illinois Rush University - Stroger Hospital of Cook County Chicago, Illinois, United States, 60612 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 Collapse << |
| NCT01384682 | HIV | Phase 4 | Completed | - | - |
| NCT00827112 | - | - | Completed | - | - |
| NCT00875368 | HIV Infections | Phase 4 | Completed | - | Netherlands ... more >> Onze Lieve Vrouwe Gasthuis Amsterdam, Netherlands, 1091 AC Academisch Medisch Centrum (AMC) Amsterdam, Netherlands, 1105AZ Slotervaartziekenhuis Amsterdam, Netherlands Rijnstate Hospital Arnhem, Netherlands Kennemer Gasthuis Haarlem, Netherlands Leids Universitair Medisch Centrum (LUMC) Leiden, Netherlands Erasmus MC Rotterdam, Netherlands, 3015GJ Maasstad Ziekenhuis Rotterdam, Netherlands Sint Elisabeth Ziekenhuis Tilburg, Netherlands Ùniversity Medical Center Utrecht Utrecht, Netherlands, 3584CX Collapse << |
| NCT02039323 | Healthy Adult Females | Phase 1 | Completed | - | United States, North Carolina ... more >> CTRC University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00735072 | - | - | Completed | - | - |
| NCT02134717 | - | - | Terminated(Poor recruitment) | - | - |
| NCT00665561 | - | - | Active, not recruiting | February 26, 2019 | - |
| NCT01719627 | HIV | Phase 1 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Collapse << |
| NCT00853840 | AIDS | Phase 4 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, 1070 Collapse << |
| NCT01896921 | HIV | Phase 3 | Recruiting | December 2018 | United States, Maryland ... more >> University of Maryland, Institute of Human Virology Recruiting Baltimore, Maryland, United States, 21201 Contact: Gregory Brogden, BS 410-706-1660 gbrogden@ihv.umaryland.edu Contact: Judith Shaw 1-866-448-4448 jshaw@ihv.umaryland.edu Principal Investigator: David Riedel, MD Collapse << |
| NCT03178084 | HIV/AIDS | Phase 3 | Completed | - | - |
| NCT00666705 | Healthy | Phase 4 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02626923 | - | - | Completed | - | - |
| NCT01327547 | - | - | Completed | - | - |
| NCT00709111 | HIV Infections | Not Applicable | Completed | - | - |
| NCT00666705 | - | - | Completed | - | - |
| NCT00719823 | Human Immunodeficiency Virus T... more >>ype 1 Collapse << | Phase 3 | Withdrawn | May 2010 | - |
| NCT00853840 | - | - | Completed | - | - |
| NCT01009034 | - | - | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital Ottawa, Ontario, Canada, K1H 8L6 Canadian Immunodeficiency Research Collaborative Toronto, Ontario, Canada, M5B1L6 Collapse << |
| NCT00884858 | HIV Infections | Phase 4 | Completed | - | Italy ... more >> Servizio Regionale di Immunologia Clinica e Tipizzazione Tissutale, Azienda Ospedaliero-Universitaria Torrette di Ancona, AN, Italy, 60126 Clinica di Malattie Infettive, Policlinico, Universita' degli Studi Bari, BA, Italy, 70124 Clinica di Malattie Infettive e Tropicali, Università degli Studi di Brescia, Spedali civili Brescia, BS, Italy, 25125 Divisione di Malattie Infettive, Ospedale S. Maria Annunziata Antella, FI, Italy, 50011 Clinica di Malattie Infettive, Ospedale San Martino Genova, GE, Italy, 16132 Divisione di Malattie Infettive, Ospedale San Gerardo Monza, MB, Italy, 20052 Polo di Medicina Chirurgia e Odontoiatria, Polo Didattico S. Paolo Milano, MI, Italy, 20124 U.O di Malattie Infettive, Fondazione San Raffaele del Monte Tabor Milano, MI, Italy, 20132 Divisione Clinicizzata di Malatie Infettive, Azienda Ospedaliera-Polo Universitario "Luigi Sacco" Milano, MI, Italy, 20157 I e II Divisione Malattie Infettive, Azienda Ospedaliera-Polo Universitario Luigi Sacco Milano, MI, Italy, 20157 Clinica delle Malattie Infettive, Policlinico Universitario Modena, MO, Italy, 41100 U.O. Malattie Infettive, Ospedale S. Spirito Pescara, PE, Italy, 65100 Clinica delle Malattie Infettive, Policlinico Monteluce Perugia, PG, Italy, 06126 Clinica delle Malattie Infettive, Policlinico "Tor Vergata" Roma, RM, Italy, 00133 III Divisione di Malattie Infettive, I.N.M.I Lazzaro Spallanzani Roma, RM, Italy, 00149 IV Divisione di Malattie Infettive, INMI Lazzaro Spallanzani Roma, RM, Italy, 00149 U.O. Malattie Infettive, Azienda Policlinico Umberto I Roma, RM, Italy, 00161 Istituto Clinica delle Malattie Infettive, Università Cattolica del Sacro Cuore Roma, RM, Italy, 00168 Clinica delle Malattie Infettive ,Ospedale Amedeo di Savoia Torino, TO, Italy, 10149 Divisione Dipartimento Urgenze Infettivologiche ad Alta Complessità e correlate all'AIDS, Ospedale Cotugno Napoli, Italy, 80131 Collapse << |
| NCT01009034 | - | - | Completed | - | - |
| NCT00982878 | HIV Infections | Phase 1 | Completed | - | United Kingdom ... more >> Imperial College Healthcare NHS Trust London, United Kingdom, W2 1NY Collapse << |
| NCT00709111 | - | - | Completed | - | - |
| NCT02346084 | HIV/AIDS | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Magee Women's Hospital of UPMC Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02799888 | Graft-versus-host Disease ... more >> Hematopoietic Stem Cell Transplantation Collapse << | Phase 2 | Unknown | April 2017 | China, Beijing ... more >> Department of Hematopoietic Stem Cell Transplantation Recruiting Beijing, Beijing, China, 100071 Collapse << |
| NCT01276236 | Kaposi's Sarcoma | Not Applicable | Unknown | April 2015 | United States, California ... more >> San Francisco General Hospital, Clinical Trials Unit San Francisco, California, United States, 94110 Collapse << |
| NCT00821535 | Human Immunodeficiency Virus (... more >>HIV) Infection Collapse << | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT01597895 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT01348308 | HIV-1 Infection ... more >> AIDS Collapse << | Phase 3 | Completed | - | France ... more >> Hôpital Henri Mondor Creteil, France, 94010 Collapse << |
| NCT01056874 | Healthy HIV I... more >>nfections Collapse << | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT00791700 | - | - | Active, not recruiting | - | - |
| NCT01420523 | Human Immunodeficiency Virus ... more >> Lipohypertrophy Collapse << | Phase 2 | Terminated | - | France ... more >> Hôpital Pitié Salpétrière Paris, France, 75013 Collapse << |
| NCT01235013 | HIV-1 Infection | Phase 4 | Unknown | - | Spain ... more >> Hospital Clínic i Provincial Not yet recruiting Barcelona, Spain, 08036 Contact: José María Gatell, MD 0034932275400 jmgatell@clinic.ub.es Contact: Judit Pich, Pharmacist 0034932275400 jpich@clinic.ub.es Principal Investigator: José Luis Blanco, MD Collapse << |
| NCT01348763 | HIV | Phase 1 | Completed | - | United Kingdom ... more >> Imperial College Healthcare NHS Trust London, United Kingdom, W2 1NY Collapse << |
| NCT00944541 | HIV Infection ... more >> HIV Infections Collapse << | Phase 2 | Completed | - | France ... more >> French National Agency for Research on AIDS and Viral Hepatits Paris, France, 75013 Collapse << |
| NCT00791700 | Human Immunodeficiency Virus | Phase 2 | Active, not recruiting | July 29, 2019 | - |
| NCT02480894 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> GSK Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT01449006 | - | - | Completed | - | - |
| NCT03402815 | HIV Infection With Other Condi... more >>tions Cardiovascular Risk Factor Atherosclerosis Inflammation Collapse << | Phase 4 | Completed | - | Italy ... more >> Elisabetta Schiaroli Perugia, Italy, 06126 Collapse << |
| NCT00948753 | Graft-versus-host Disease ... more >> Hematopoietic Stem Cell Transplantation Collapse << | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT01894776 | HIV Infection ... more >> HIV-1 Infection Mycobacterium Avium Complex (MAC) Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital -General Campus Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT01449006 | Human Immunodeficiency Virus (... more >>HIV) HIV Associated Neurocognitive Disorders (HAND) Collapse << | Phase 4 | Completed | - | Australia, New South Wales ... more >> St. Vincent's Hospital Sydney, New South Wales, Australia, 2010 Australia, Victoria The Alfred Hospital Melbourne, Victoria, Australia, 3181 Collapse << |
| NCT02625207 | Healthy Subjects | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00775294 | HIV/AIDS | Phase 1 | Completed | - | United States, North Carolina ... more >> UNC Hospitals CTRC Chapel Hill, North Carolina, United States, 27514 Collapse << |
| NCT02990312 | Hiv Kidney Tr... more >>ansplant HIV Reservoir CCR5 Collapse << | Phase 4 | Recruiting | July 2020 | United States, Maryland ... more >> Institute of human virology Recruiting Baltimore, Maryland, United States, 21201 Contact: Jennifer Husson, MD 410-706-6973 Jhusson@ihv.umaryland.edu Contact: Ilise Marrazzo 4107062564 IMarrazzo@ihv.umaryland.edu Collapse << |
| NCT01866267 | Human Immunodeficiency Virus ... more >> AIDS Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> St. Hope Foundation, Inc. Bellaire, Texas, United States, 77401 Collapse << |
| NCT02625207 | - | - | Completed | - | - |
| NCT01627717 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Centre Hospitalier de l'Université de Montréal Montréal, Quebec, Canada, H2W 1T8 Collapse << |
| NCT01133210 | Hypertriglyceridemia | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT02159027 | AIDS Dementia Complex | Phase 2 Phase 3 | Recruiting | December 2018 | United States, Hawaii ... more >> Clint Spencer Clinic, Hawaii Center for AIDS, University of Hawaii Recruiting Honolulu, Hawaii, United States, 96813 Contact: Debra Ogata-Arakaki, R.N. 808-692-1332 ogataara@hawaii.edu Contact: Nancy Hanks, R.N. 808-692-1336 nhanks@hawaii.edu Principal Investigator: Cecilia M. Shikuma, M.D. Sub-Investigator: Dominic Chow, M.D. Ph.D. Puerto Rico Puerto Rico Clinical and Translational Research Consortium Recruiting San Juan, Puerto Rico, 00936 Contact: Valerie E Wojna, MD 7877586250 valerie.wojna1@upr.edu Contact: Miriam Matos, MD 7877590306 ext 221 miriam.matos@upr.edu Collapse << |
| NCT01428986 | - | - | Completed | - | Japan ... more >> National Center for Global Health and Medicine Shinjuku, Tokyo, Japan, 1628655 Collapse << |
| NCT01190293 | HIV Infection | Phase 4 | Completed | - | United Kingdom ... more >> St Stephen's Centre London, United Kingdom, SW109NH Collapse << |
| NCT02167451 | Diagnoses That Require Stem Ce... more >>ll Transplant Graft Versus Host Disease (GVHD) Collapse << | Phase 1 Phase 2 | Recruiting | December 2021 | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Sharon Mitchell 513-636-4174 Sharon.Mitchell@cchmc.org Collapse << |
| NCT03172026 | Stroke | Phase 2 Phase 3 | Recruiting | December 2019 | United States, California ... more >> California Rehabilitation Institute Recruiting Los Angeles, California, United States, 90067 United States, New York Burke Rehabilitation Center Not yet recruiting White Plains, New York, United States, 10605 Collapse << |
| NCT01980329 | Cytochrome P450 CYP3A5 Enzyme ... more >>Polymorphism Healthy Subjects Pharmacokinetics of Maraviroc Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> The Johns Hopkins University School of Medicine Division of Clinical Pharmacology Baltimore, Maryland, United States, 21210 Collapse << |
| NCT02934022 | HIV Infection | Not Applicable | Completed | - | United States, California ... more >> VA Long Beach Healthcare System Long Beach, California, United States, 90822 Collapse << |
| NCT02881762 | Hepatitis C H... more >>uman Immunodeficiency Virus Collapse << | Phase 4 | Recruiting | December 2019 | United States, Maryland ... more >> Institute of Human Virology at the University of Maryland School of Medicine Recruiting Baltimore, Maryland, United States, 21201 Contact: Lydia Tang, MBChB 410-706-6567 lydiatang@ihv.umaryland.edu Collapse << |
| NCT03708861 | HIV Infection | Phase 3 | Withdrawn | December 2017 | Italy ... more >> University of Torino Torino, Italy Collapse << |
| NCT01389063 | Endothelial Dysfunction | Phase 4 | Unknown | October 2014 | Netherlands ... more >> University Medical Center Utrecht Recruiting Utrecht, Netherlands, 3584CX Contact: Steven FL van Lelyveld, MD s.f.l.vanlelyveld@umcutrecht.nl Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| CCR5 | IC50:6.4nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
