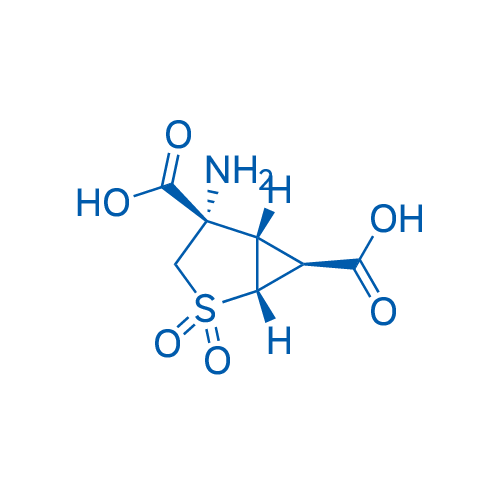
CAS No.: 635318-11-5
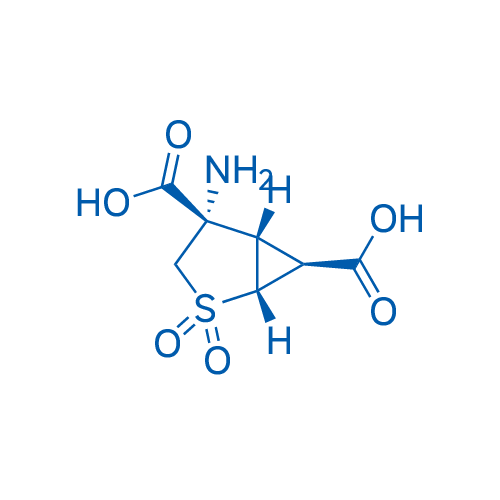
LY404039 Catalog No. CSN12419
Synonyms: LY 404040;LY-404,039;Pomaglumetad
LY404039 is an inhibitor for mGluR1 (Ki=149 nM) and mGluR2 (Ki= 92 nM), which can also inhibit dopamine receptor.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01609218 Healthy Volunteer Study Phase 1 Completed - United Kingdom ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Leeds, West Yorkshire, United Kingdom, LS2 9LH Collapse << NCT01475136 Hepatic Insufficiency Phase 1 Completed - Germany ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Munich, Germany, 81241 Hungary For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Balatonfüred, Hungary, 8230 Collapse << NCT01659177 Healthy Participants Phase 1 Withdrawn(The decision to stop... more >> the trial was based on efficacy results in the overall schizophrenia patient population.) Collapse << September 2012 Switzerland ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Allschwil, Switzerland, CH-4123 Collapse << - 更多
- 参考文献
- [1] Seeman P. An agonist at glutamate and dopamine D2 receptors, LY404039. Neuropharmacology. 2013 Mar;66:87-8.
- [2] Rorick-Kehn LM, Johnson BG, et al. Pharmacological and pharmacokinetic properties of a structurally novel, potent, and selective metabotropic glutamate 2/3 receptor agonist: in vitro characterization of agonist (-)-(1R,4S,5S,6S)-4-amino-2-sulfonylbicyclo[3.1.0] -hexane-4,6-dicarboxylic acid(LY404039).
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 635318-11-5 | 储存条件 |
|
|||||||
| 分子式 | C7H9NO6S | 运输 | 蓝冰 | |||||||
| 分子量 | 235.21 | 别名 | LY 404040;LY-404,039;Pomaglumetad | |||||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
