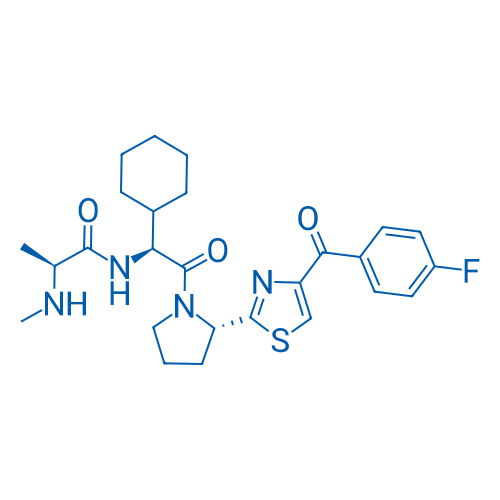
CAS No.: 1005342-46-0
LCL161 Catalog No. CSN17158
Synonyms: NVP-LCL161
LCL161 is an IAP inhibitor, inhibits XIAP activity in HEK293 cell with IC50 of 35 nM, also inhibits cIAP1 activity in MDA-MB-231 cell with IC50 of 0.4 nM.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02098161 Leukemia Phase 2 Recruiting December 2020 United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << NCT03111992 Multiple Myeloma Phase 1 Recruiting May 27, 2020 United States, Arizona ... more >> Novartis Investigative Site Recruiting Phoenix, Arizona, United States, 85054 United States, Tennessee Novartis Investigative Site Recruiting Nashville, Tennessee, United States, 37203 Contact: Kimberly Cox 615-329-7413 Kimberly.Cox3@scresearch.net Principal Investigator: Jesus G. Berdeja Canada, Quebec Novartis Investigative Site Recruiting Montreal, Quebec, Canada, H3T 1E2 Germany Novartis Investigative Site Recruiting Heidelberg, Germany, 69120 Novartis Investigative Site Recruiting Kiel, Germany, 24105 Italy Novartis Investigative Site Recruiting Milano, MI, Italy, 20133 Spain Novartis Investigative Site Recruiting Salamanca, Castilla Y Leon, Spain, 37007 Novartis Investigative Site Recruiting Madrid, Spain, 28006 Switzerland Novartis Investigative Site Recruiting Locarno, Switzerland, 6600 Collapse << NCT02890069 Colorectal Cancer, Non-small C... more >>ell Lung Carcinoma (Adenocarcinoma), Triple Negative Breast Cancer, Renal Cell Carcinoma Collapse << Phase 1 Recruiting December 19, 2019 - - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1005342-46-0 | 储存条件 |
|
|||||||
| 分子式 | C26H33FN4O3S | 运输 | 蓝冰 | |||||||
| 分子量 | 500.63 | 别名 | NVP-LCL161 | |||||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
