| NCT01071278 | - | - | Completed | - | - |
| NCT00533676 | Flushing | Phase 2 | Completed | - | - |
| NCT01450410 | Dyslipidemias | Phase 4 | Terminated(Study terminated (h... more >>alted prematurely) as recomended by the drug supplier and medical agencies) Collapse << | May 2013 | Spain
... more >> Unitat Funcional de Risc Vascular. Servei Medicina Interna. Hospital Universitari de Bellvitge.
L'Hospitalet de Llobregat, Barcelona, Spain, 08907 Collapse << |
| NCT01118598 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | United Kingdom
... more >> Hull & East Yorkshire Hospitals NHS Trust
Hull, United Kingdom, HU3 2RW Collapse << |
| NCT01071278 | - | - | Completed | - | - |
| NCT01391377 | Peripheral Arterial Disease | Not Applicable | Terminated(Tredaptive has been... more >> suspended worldwide) Collapse << | December 2014 | Australia, Victoria
... more >>
Baker IDI Heart and diabetes research institute
Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01071525 | Hypercholesterolemia | Phase 3 | Unknown | October 2010 | Israel
... more >> Rambam Health Care Campus
Not yet recruiting
Haifa, Israel, 31096
Contact: Shadi +972-523591876 s_hamoud@rambam.health.gov.il
Contact: Tony Hayek, Prof +972-523782009 t_hayek@rambam.health.gov.il
Sub-Investigator: Tony Hayek, Prof Collapse << |
| NCT01054508 | Hypercholesterolemia | Phase 4 | Completed | - | United Kingdom
... more >> Manchester Royal Infirmary
Manchester, United Kingdom, M13 9WL Collapse << |
| NCT02153879 | Type 2 Diabetes Mellitus
... more >> Dyslipidemia Collapse << | Phase 4 | Completed | - | Spain
... more >> Hospital Universitari Sant Joan
Reus, Tarragona, Spain, 43204 Collapse << |
| NCT01010516 | Dyslipidemia | Phase 4 | Unknown | - | Greece
... more >> University of Ioannina Medical School
Recruiting
Ioannina, Greece, 45 110
Contact: M S Elisaf, MD +302651007509 egepi@cc.uoi.gr
Principal Investigator: Moses S Elisaf, MD
Sub-Investigator: Evangelos N Liberopoulos, MD
Sub-Investigator: Anastazia Kei, MD Collapse << |
| NCT00533208 | Seasonal Allergies | Phase 2 | Completed | - | - |
| NCT00961636 | - | - | Completed | - | - |
| NCT00111891 | Dyslipidemia | Phase 2 | Completed | - | - |
| NCT00376584 | Hypercholesteremia
... more >> Hyperlipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00961636 | Dyslipidemia | Phase 3 | Completed | - | - |
| NCT01942291 | Hypoalphalipoproteinemia | Phase 4 | Completed | - | Brazil
... more >> University of Campinas
Campinas, SP, Brazil, 13083-887 Collapse << |
| NCT00269204 | Primary Hypercholesterolaemia
... more >>
Mixed Hyperlipidaemia Collapse << | Phase 3 | Completed | - | - |
| NCT00289900 | Mixed Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT01052311 | Coronary Artery Disease
... more >> Dyslipidemia Collapse << | Phase 4 | Terminated(The sponsor decided... more >> to withdraw study drug from market) Collapse << | - | Israel
... more >> Leviev Heart Center, Sheba Medical Center
Tel Hashomer, Israel, 52621 Collapse << |
| NCT01414166 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01414166 | Dyslipidemia | Phase 3 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT00289900 | - | - | Completed | - | - |
| NCT00461630 | Cardiovascular Disease
... more >> Peripheral Arterial Disease
Diabetes Mellitus
Coronary Heart Disease Collapse << | Phase 3 | Completed | - | United Kingdom
... more >> Clinical Trial Service Unit, University of Oxford
Oxford, United Kingdom, OX3 7LF Collapse << |
| NCT00536237 | Flushing | Phase 2 | Completed | - | - |
| NCT00533312 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| NCT00384293 | - | - | Terminated | - | - |
| NCT00378833 | Hypercholesterolemia
... more >> Hyperlipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00461630 | - | - | Completed | - | - |
| NCT00738985 | Cardiovascular Diseases
... more >> Hyperlipidemia Collapse << | Phase 4 | Withdrawn(The study was cancel... more >>led due to budget limitations) Collapse << | - | - |
| NCT00269217 | Primary Hypercholesterolemia
... more >> Mixed Hyperlipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00384293 | Hypercholesterolemia, Familial | Phase 3 | Terminated | - | - |
| NCT00479882 | Primary Hypercholesterolemia
... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00533611 | Flushing | Phase 3 | Completed | - | - |
| NCT00769132 | Hypercholesterolemia | Phase 1 | Completed | - | - |
| NCT00479882 | - | - | Completed | - | - |
| NCT00618995 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT00618995 | - | - | Completed | - | - |
| NCT01335997 | Primary Hypercholesterolemia
... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Terminated(Business Reasons) | - | - |
| NCT01335997 | - | - | Terminated(Business Reasons) | - | - |
| NCT00783601 | Asthma | Phase 2 | Completed | - | - |
| NCT00769132 | - | - | Completed | - | - |
| NCT01321034 | Hypercholesterolemia | Phase 4 | Completed | - | Spain
... more >> Hospital San Jorge
Huesca, Spain
Hospital Universitario Miguel Servet
Zaragoza, Spain, 50009
Hospital Royo Villanova
Zaragoza, Spain Collapse << |
| NCT01012219 | Primary Hypercholesterolemia
... more >> Mixed Hyperlipidemia Collapse << | Phase 1 | Completed | - | - |
| NCT01012219 | - | - | Completed | - | - |
| NCT00479388 | Primary Hypercholesterolemia
... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00536510 | Hypercholesterolemia
... more >> Hyperlipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00536510 | - | - | Completed | - | - |
| NCT01274559 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT00479388 | - | - | Completed | - | - |
| NCT01274559 | Primary Hypercholesterolemia
... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01683656 | HIV
Atheroscl... more >>erosis Collapse << | Phase 4 | Terminated(Withdrawal of IMP f... more >>rom the market. Data on risk-benefit ratio pending.) Collapse << | July 2014 | Switzerland
... more >> University Hospital Berne Inselspital
Berne, BE, Switzerland, 3010
University Hospital Basel
Basel, BS, Switzerland, 4031
University Hospitals Genève
Geneva, GE, Switzerland, 1211
Kantonsspital St Gallen
St Gallen, SG, Switzerland, 9007
EOC Ente Ospedaliero Cantonale, civico
Lugano, TI, Switzerland, 6903
CHUV Cantonal University Hospital Vaud
Lausanne, VD, Switzerland, 1011
University Hospital Zurich
Zurich, ZH, Switzerland Collapse << |
| NCT01228019 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01239992 | - | - | Terminated(negative endpoint s... more >>tudy resulting in withdrawal of study drug) Collapse << | - | - |
| NCT01239992 | Hyperlipoproteinemia
... more >> Metabolic Syndrome Collapse << | Phase 4 | Terminated(negative endpoint s... more >>tudy resulting in withdrawal of study drug) Collapse << | - | Germany
... more >> Medizinische Klinik II, Klinikum der Universitaet Muenchen, Grosshadern
Munich, Germany, 81377 Collapse << |
| NCT01228019 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01451619 | Rosacea | Phase 1 | Completed | - | - |
| NCT01308203 | Coronary Artery Disease
... more >> Dyslipidemias Collapse << | Phase 4 | Terminated(Merck has decided t... more >>o discontinue all studies with extended-release niacin/laropiprant.because the HPS2-THRIVE did not meet its primary endpoint) Collapse << | - | Argentina
... more >> Hospital Italiano de Buenos Aires
Buenos Aires, Argentina, C1181ACH Collapse << |
| NCT01294683 | Primary Hypercholesterolemia
... more >> Dyslipidemia Collapse << | Phase 3 | Terminated | - | - |
| NCT01294683 | - | - | Terminated | - | - |
| NCT00944645 | - | - | Completed | - | - |
| NCT00944645 | Dyslipidemia | Phase 1 | Completed | - | - |
| NCT00485758 | - | - | Completed | - | - |
| NCT00943124 | Dyslipidemia | Phase 1 | Completed | - | - |
| NCT01126073 | Coronary Heart Disease | Phase 4 | Completed | - | Slovenia
... more >> UMC Ljubljana
Ljubljana, Slovenia, 1000 Collapse << |
| NCT00943124 | - | - | Completed | - | - |
| NCT00485758 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00664287 | Dyslipidemia | Phase 3 | Withdrawn | - | - |
| NCT01583647 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs.) Collapse << | - | - |
| NCT01583647 | Hypercholesterolemia, Familial... more >>
Heterozygous Familial Hypercholesterolemia Collapse << | Phase 1 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs.) Collapse << | - | - |
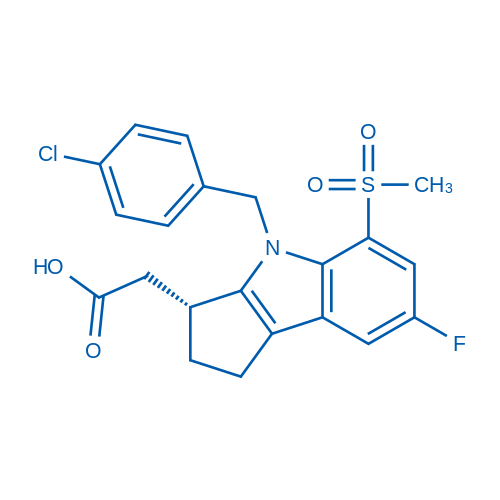

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
