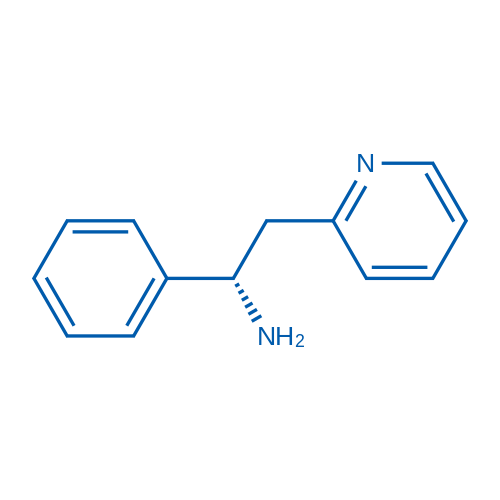
CAS No.: 153322-05-5
Lanicemine/拉尼西明 Catalog No. CSN20030
Synonyms: AZD6765;AR-R 15896AR;ARL-15896AR
Lanicemine is a low-trapping NMDA receptor antagonist. Lanicemine is used the management of severe and treatment-resistant depression. Lanicemine was originally developed as a neuroprotective agent, but was redeveloped as an antidepressant.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02248818 - - Completed - - NCT02248818 Healthy Volunteers Phase 1 Completed - United States, New York ... more >> Clinilabs, Inc. New York, New York, United States, 10019 Collapse << NCT03166501 Depression Po... more >>st Traumatic Stress Disorder Collapse << Phase 1 Not yet recruiting December 31, 2019 - - 更多
- 参考文献
- [1] Agbo F, Bui KH, Zhou D. Population pharmacokinetic analysis of lanicemine (AZD6765), an NMDA channel blocker, in healthy subjects and patients with major depressive disorder. J Clin Pharm Ther. 2017 May 4.
- [2] Sanacora G, Johnson MR, et al. Adjunctive Lanicemine (AZD6765) in Patients with Major Depressive Disorder and History of Inadequate Response to Antidepressants: A Randomized, Placebo-Controlled Study. Neuropsychopharmacology. 2017 Mar;42(4):844-853.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 153322-05-5 | 储存条件 |
|
|
| 分子式 | C13H14N2 | 运输 | 蓝冰 | |
| 分子量 | 198.26 | 别名 | AZD6765;AR-R 15896AR;ARL-15896AR;ARR-15896;FPL-15896;FPL-15896AR ARL15896;FPL 15896AR;拉尼西明 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
