| NCT01974544 | Type 2 Diabetes | Not Applicable | Unknown | December 2016 | Chile
... more >> Pontificia Universidad Católica de Chile
Recruiting
Santiago, Región Metropolitana, Chile, 8330033
Contact: Castillo Alejandra, Nurse 56223543527 macastia@uc.cl
Contact: Vega Andrea, Nurse 56223543879 abvega@uc.cl
Principal Investigator: Boza Camilo, MD surgeon
Sub-Investigator: Valderas Juan Patricio, MD
Sub-Investigator: Arrese Marco, MD Internist
Sub-Investigator: Muñoz Rodrigo, MD Collapse << |
| NCT03323788 | - | - | Recruiting | August 1, 2018 | United States, Arizona
... more >>
The University of Arizona
Recruiting
Tucson, Arizona, United States, 85724
Contact: Oscar Parra, MADM 520-626-6485 oscardp@email.arizona.edu
Principal Investigator: Lawrence Mandarino, PhD Collapse << |
| NCT00004266 | Diabetic Nephropathy | Phase 3 | Completed | - | United States, Minnesota
... more >>
Hennepin County Medical Center
Minneapolis, Minnesota, United States, 55415 Collapse << |
| NCT03031821 | Prostate Cancer
... more >> Metabolic Syndrome Collapse << | Phase 3 | Not yet recruiting | June 1, 2023 | Canada, British Columbia
... more >>
BC Cancer Agency - Vancouver Cancer Centre
Not yet recruiting
Vancouver, British Columbia, Canada, V5Z 4E6
Contact: Bernie Eigl, MD 604-877-6000 bernie.eigl@bccancer.bc.ca
Principal Investigator: Bernie Eigl, MD Collapse << |
| NCT03723395 | Drug-drug Interaction | Phase 1 | Recruiting | November 2018 | United States, Florida
... more >>
Covance Clinical Research Unit
Recruiting
Daytona Beach, Florida, United States, 32117
Contact: Michelle Saint Pierre 386-366-6428 michelle.saintpierre@covance.com
Principal Investigator: Hugh Coleman
United States, Texas
Covance Clinical Research Unit
Recruiting
Dallas, Texas, United States, 75247
Contact: Iona Tihina 469-513-6716 Ioana.Tihina@covance.com
Principal Investigator: Jennifer Zon Collapse << |
| NCT01148004 | HIV
Fenofibra... more >>te
Protease Inhibitors
Hypertriglyceridemia
Glucuronosyltransferase Collapse << | Phase 1 | Completed | - | United States, Maryland
... more >>
National Institutes of Health Clinical Center, 9000 Rockville Pike
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00000461 | Cardiovascular Diseases
... more >> Coronary Disease
Heart Diseases
Myocardial Ischemia Collapse << | Phase 2 | Completed | - | - |
| NCT00005676 | - | - | Completed | - | - |
| NCT00243672 | Atherosclerosis | Phase 4 | Withdrawn(The study chair chan... more >>ged his employment, the realization of the study was not possible) Collapse << | - | - |
| NCT03624959 | Healthy Volunteer | Phase 1 | Recruiting | April 15, 2019 | United States, Texas
... more >>
PPD Phase 1 Clinic
Recruiting
Austin, Texas, United States, 78744 Collapse << |
| NCT00039663 | HIV Infections | Phase 1 | Completed | - | United States, Maryland
... more >>
National Institutes of Health Clinical Center (CC)
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00283335 | Coronary Heart Disease | Phase 3 | Completed | - | United States, Minnesota
... more >>
Minneapolis VA Health Care System, Minneapolis, MN
Minneapolis, Minnesota, United States, 55417 Collapse << |
| NCT00108511 | Hypertriglyceridemia | Phase 1 | Completed | - | United States, Indiana
... more >>
Roudebush VA Medical Center
Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01836198 | Healthy Volunteers | Phase 1 | Completed | - | United States, Indiana
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Evansville, Indiana, United States, 47710 Collapse << |
| NCT01913379 | Healthy Subjects
... more >> Pharmacokinetics
Drug-Drug Interaction Collapse << | Phase 1 | Completed | - | France
... more >> SGS
Paris, France, 75015 Collapse << |
| NCT01797198 | Drug-Drug Interaction (DDI)
... more >> Healthy Subjects Collapse << | Phase 1 | Completed | - | United Kingdom
... more >> PAREXEL Early Phase Clinical Unit
Harrow, United Kingdom, HA1 3U Collapse << |
| NCT01301742 | Healthy | Phase 1 | Completed | - | Germany
... more >> 1245.58.1 Boehringer Ingelheim Investigational Site
Biberach, Germany Collapse << |
| NCT01340846 | Cancer | Phase 1 | Completed | - | United States, Arizona
... more >>
GSK Investigational Site
Scottsdale, Arizona, United States, 85259
United States, Florida
GSK Investigational Site
Sarasota, Florida, United States, 34232
United States, Michigan
GSK Investigational Site
Detroit, Michigan, United States, 48201
United States, Ohio
GSK Investigational Site
Columbus, Ohio, United States, 43210
United States, Oklahoma
GSK Investigational Site
Oklahoma City, Oklahoma, United States, 73104
United States, South Carolina
GSK Investigational Site
Greenville, South Carolina, United States, 29605
United States, Tennessee
GSK Investigational Site
Nashville, Tennessee, United States, 37203
United States, Utah
GSK Investigational Site
Salt Lake City, Utah, United States, 84112
Australia, Western Australia
GSK Investigational Site
Nedlands, Western Australia, Australia, 6009
United Kingdom
GSK Investigational Site
Headington, United Kingdom, OX3 7LJ
GSK Investigational Site
London, United Kingdom, W1G 6AD Collapse << |
| NCT01376232 | Anaemia | Phase 1 | Completed | - | India
... more >> GSK Investigational Site
Electronics City, Bengalore, India, 560100 Collapse << |
| NCT01301742 | - | - | Completed | - | - |
| NCT02230033 | Healthy | Phase 1 | Completed | - | United States, Arizona
... more >>
Tempe, Arizona, United States Collapse << |
| NCT01736254 | Healthy Volunteers | Phase 1 | Completed | - | United States, Florida
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Daytona Beach, Florida, United States, 32117 Collapse << |
| NCT00800475 | Bioavailability | Phase 1 | Completed | - | - |
| NCT02770222 | Healthy Subjects | Phase 1 | Completed | - | Germany
... more >> Investigator Site
Kiel, Germany, 24105 Collapse << |
| NCT00566865 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT00966966 | Healthy | Phase 1 | Completed | - | Netherlands
... more >> Pfizer Investigational Site
Utrecht, Netherlands, 3584 CJ Collapse << |
| NCT02045056 | Preclinical Alzheimer's Diseas... more >>e Collapse << | Early Phase 1 | Active, not recruiting | June 2019 | United States, Kentucky
... more >>
University of Kentucky
Lexington, Kentucky, United States, 40536-0230 Collapse << |
| NCT00474201 | Healthy Volunteers | Not Applicable | Completed | - | United States, Maryland
... more >>
National Institutes of Health Clinical Center, 9000 Rockville Pike
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01876810 | Nicotine Dependence | Phase 2 | Completed | - | Canada, Ontario
... more >>
Centre for Addiction and Mental Health
Toronto, Ontario, Canada, M5S 1S8
Centre of Addiction and Mental Health
Toronto, Ontario, Canada, M5S 2S1 Collapse << |
| NCT01736254 | - | - | Completed | - | - |
| NCT03539432 | Alcohol Use Disorder | Phase 2 | Active, not recruiting | December 30, 2018 | United States, New Mexico
... more >>
The Mind Research Network
Albuquerque, New Mexico, United States, 87106 Collapse << |
| NCT01385020 | Healthy Subjects | Phase 4 | Completed | - | Taiwan
... more >> National Taiwan University Hospital
Taipei, Taiwan Collapse << |
| NCT01083212 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Sweden
... more >> Reserach Site
Linkoping, Sweden
Research Site
Lulea, Sweden
Research Site
Uppsala, Sweden Collapse << |
| NCT00474201 | - | - | Completed | - | - |
| NCT01876810 | - | - | Completed | - | - |
| NCT02638597 | Smoking Cessation | Phase 2 | Unknown | March 2017 | United States, Texas
... more >>
Parkland Memorial Hospital
Recruiting
Dallas, Texas, United States, 75235
Contact: Adriane Dela Cruz 214-648-7312 Collapse << |
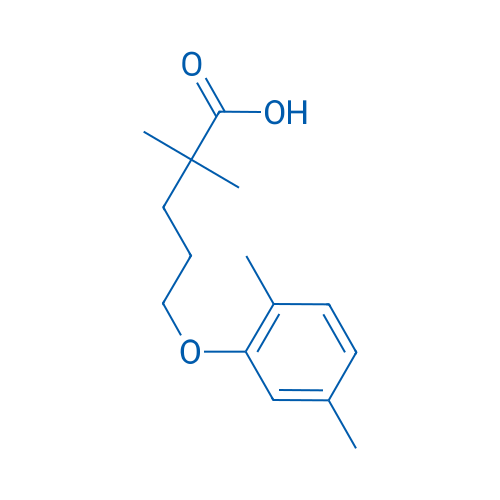

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
