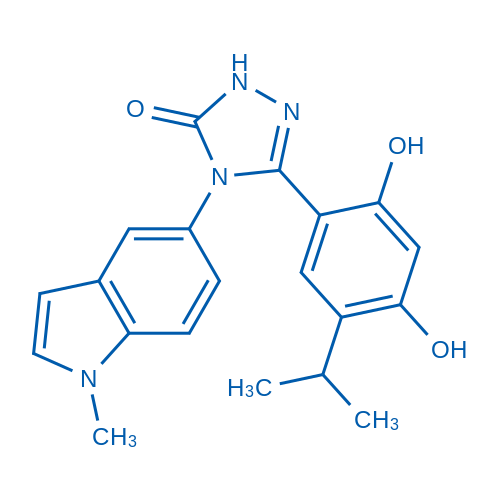
CAS No.: 888216-25-9
Ganetespib Catalog No. CSN15732
Synonyms: STA-9090
Ganetespib is a unique triazolone-containing Hsp90 inhibitor with an IC50 of 4 nM in OSA 8 cells.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 HSP90
IC50:4nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01042379 Breast Neoplasms ... more >> Breast Cancer Breast Tumors Collapse << Phase 2 Recruiting - - NCT01227018 Adenocarcinoma of the Pancreas... more >> Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << Phase 2 Terminated(interim analysis fo... more >>und the study drug to be ineffective) Collapse << - United States, Tennessee ... more >> The Jones Clinic Memphis, Tennessee, United States, 38138 Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << NCT01227018 - - Terminated(interim analysis fo... more >>und the study drug to be ineffective) Collapse << - - - 更多
- 参考文献
- [1] Lin TY, Bear M, et al. The novel HSP90 inhibitor STA-9090 exhibits activity against Kit-dependent and -independent malignant mast cell tumors. Exp Hematol. 2008;36(10):1266-77.
- [2] Shimamura T, Perera SA, et al. Ganetespib (STA-9090), a nongeldanamycin HSP90 inhibitor, has potent antitumor activity in in vitro and in vivo models of non-small cell lung cancer. Clin Cancer Res. 2012 Sep 15;18(18):4973-85.
- [3] Nagaraju GP, Mezina A, et al. Targeting the Janus-activated kinase-2-STAT3 signalling pathway in pancreatic cancer using the HSP90 inhibitor ganetespib. Eur J Cancer. 2016 Jan;52:109-19.
- [4] Zhou D, Liu Y, et al. A rat retinal damage model predicts for potential clinical visual disturbances induced by Hsp90 inhibitors. Toxicol Appl Pharmacol. 2013 Dec 1;273(2):401-9.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 888216-25-9 | 储存条件 |
|
|||||||||||||
| 分子式 | C20H20N4O3 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 364.40 | 别名 | STA-9090 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 35612 | - | Growth Inhibition Assay | - | IC50=38 nM | 24784839 |
| 5637 | - | Growth Inhibition Assay | - | IC50=44 nM | 24784839 |
| 639-V | - | Growth Inhibition Assay | - | IC50=10 nM | 24784839 |
| 647-V | - | Growth Inhibition Assay | - | IC50=27 nM | 24784839 |
| A1847 | 0-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 23900136 |
| A1847 | 10-100 nM | Apoptosis Assay | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 |
| A549 | - | Growth Inhibition Assay | - | IC50=43 nM | 23012248 |
| AC3 | - | Growth Inhibition Assay | - | IC50=25.9 nM | 24166505 |
| BFTC | - | Growth Inhibition Assay | - | IC50=17 nM | 24784839 |
| BT-12 | - | Growth Inhibition Assay | - | IC50=14.3 nM | 23303741 |
| BT-20 | 100/250 nM | Function Assay | 24 h | resulted in a dose-dependent destabilization of EGFR, IGF-IR, MET, and CRAF | 24173541 |
| CAL27 | 10/50 nM | Cytoxicity Assay | 24 h | decreases cell proliferation dose dependently | 25205430 |
| Calu-1 | - | Growth Inhibition Assay | - | IC50=58 nM | 23012248 |
| Calu-6 | - | Growth Inhibition Assay | - | IC50=64 nM | 23012248 |
| CCRF-CEM (1) | - | Growth Inhibition Assay | - | IC50=12.5 nM | 23303741 |
| CCRF-CEM (2) | - | Growth Inhibition Assay | - | IC50=7.2 nM | 23303741 |
| CHLA-10 | - | Growth Inhibition Assay | - | IC50=5.7 nM | 23303741 |
| CHLA-136 | - | Growth Inhibition Assay | - | IC50=23.2 nM | 23303741 |
| CHLA-258 | - | Growth Inhibition Assay | - | IC50=6.4 nM | 23303741 |
| CHLA-266 | - | Growth Inhibition Assay | - | IC50=27.1 nM | 23303741 |
| CHLA-9 | - | Growth Inhibition Assay | - | IC50=4.6 nM | 23303741 |
| CHLA-90 | - | Growth Inhibition Assay | - | IC50=22.3 nM | 23303741 |
| COG-LL-317 | - | Growth Inhibition Assay | - | IC50=4.4 nM | 23303741 |
| COLO-205 | - | Growth Inhibition Assay | - | IC50=14 nM | 24682747 |
| COLO-678 | - | Growth Inhibition Assay | - | IC50=21 nM | 24682747 |
| COR-L23 | - | Growth Inhibition Assay | - | IC50=22 nM | 23012248 |
| Detroit562 | 10/50 nM | Cytoxicity Assay | 24 h | decreases cell proliferation dose dependently | 25205430 |
| DSH1 | - | Growth Inhibition Assay | - | IC50=6 nM | 24784839 |
| FUDA | 10/50 nM | Cytoxicity Assay | 24 h | decreases cell proliferation dose dependently | 25205430 |
| GLC4 | - | Growth Inhibition Assay | - | IC50=20.47 nM | 24166505 |
| GLC4 | 30 nM | Function Assay | 72 h | induces persistent G2/M phase arrest | 24166505 |
| H1173 | - | Growth Inhibition Assay | - | IC50=12.62 nM | 24166505 |
| H128 | - | Growth Inhibition Assay | - | IC50=69.55 nM | 24166505 |
| H1355 | - | Growth Inhibition Assay | - | IC50=5 nM | 23012248 |
| H146 | - | Growth Inhibition Assay | - | IC50=28.51 nM | 24166505 |
| H146 | 30 nM | Function Assay | 72 h | induces persistent G2/M phase arrest | 24166505 |
| H157 | - | Growth Inhibition Assay | - | IC50=7 nM | 23012248 |
| H1734 | - | Growth Inhibition Assay | - | IC50=28 nM | 23012248 |
| H1792 | - | Growth Inhibition Assay | - | IC50=20 nM | 23012248 |
| H187 | - | Growth Inhibition Assay | - | IC50=24.99 nM | 24166505 |
| H2009 | - | Growth Inhibition Assay | - | IC50=19 nM | 23012248 |
| H2030 | - | Growth Inhibition Assay | - | IC50=12 nM | 23012248 |
| H2122 | - | Growth Inhibition Assay | - | IC50=53 nM | 23012248 |
| H2212 | - | Growth Inhibition Assay | - | IC50=17 nM | 23012248 |
| H2228 | 0-1000 nM | Cell Viability Assay | 72 h | IC50=13 nM | 23533265 |
| H23 | - | Growth Inhibition Assay | - | IC50=11 nM | 23012248 |
| H3122 | 0-1000 nM | Cell Viability Assay | 72 h | IC50=10 nM | 23533265 |
| H358 | - | Growth Inhibition Assay | - | IC50=29 nM | 23012248 |
| H441 | - | Growth Inhibition Assay | - | IC50=14 nM | 23012248 |
| H460 | - | Growth Inhibition Assay | - | IC50=8 nM | 23012248 |
| H526 | - | Growth Inhibition Assay | - | IC50=21.64 nM | 24166505 |
| H620 | - | Growth Inhibition Assay | - | IC50=32.67 nM | 24166505 |
| H69 | - | Growth Inhibition Assay | - | IC50=83.36 nM | 24166505 |
| H727 | - | Growth Inhibition Assay | - | IC50=28 nM | 23012248 |
| H792 | - | Growth Inhibition Assay | - | IC50=45.07 nM | 24166505 |
| H82 | - | Growth Inhibition Assay | - | IC50=30.27 nM | 24166505 |
| H82 | 30 nM | Function Assay | 72 h | induces persistent G2/M phase arrest | 24166505 |
| HCC2998 | - | Growth Inhibition Assay | - | IC50=128 nM | 24682747 |
| HCT 116 | - | Growth Inhibition Assay | - | IC50=14 nM | 24682747 |
| HCT-116 | 50nM | Function Assay | 24 h | induced G0/G1 arrest | 25210794 |
| HCT-15 | - | Growth Inhibition Assay | - | IC50=8 nM | 24682747 |
| HL60 | 30/80/150/250 nM | Apoptosis Assay | 24/48/72 h | induces dose dependant induction of apoptosis | 25882550 |
| HOP-62 | - | Growth Inhibition Assay | - | IC50=11 nM | 23012248 |
| HT-1197 | - | Growth Inhibition Assay | - | IC50=53 nM | 24784839 |
| HT-1376 | - | Growth Inhibition Assay | - | IC50=21 nM | 24784839 |
| HT-29 | 50nM | Function Assay | 24 h | induced G0/G1 arrest | 25210794 |
| HuTu-80 | - | Growth Inhibition Assay | - | IC50=13 nM | 24682747 |
| IA-LM | - | Growth Inhibition Assay | - | IC50=10 nM | 23012248 |
| J82 | - | Growth Inhibition Assay | - | IC50=18 nM | 24784839 |
| K008 | - | Cell Viability Assay | - | IC50=60 nM | 23418523 |
| K008 | 250 nM | Function Assay | 24 h | induces G2 arrest | 23418523 |
| K008 | 100 nM | Apoptosis Assay | 72 h | significantly induces apoptosis | 23418523 |
| K028 | - | Cell Viability Assay | - | IC50=84 nM | 23418523 |
| K028 | 250 nM | Function Assay | 24 h | induces G2 arrest | 23418523 |
| K028 | 100 nM | Apoptosis Assay | 72 h | significantly induces apoptosis | 23418523 |
| K029 | - | Cell Viability Assay | - | IC50=46 nM | 23418523 |
| K029 | 250 nM | Function Assay | 24 h | induces G1 arrest | 23418523 |
| K029 | 100 nM | Apoptosis Assay | 72 h | significantly induces apoptosis | 23418523 |
| K033 | - | Cell Viability Assay | - | IC50=75.5 nM | 23418523 |
| K033 | 250 nM | Function Assay | 24 h | induces a modest increase in G1 population | 23418523 |
| K033 | 100 nM | Apoptosis Assay | 72 h | significantly induces apoptosis | 23418523 |
| Karpas-299 | - | Growth Inhibition Assay | - | IC50=9.6 nM | 23303741 |
| Kasumi-1 | - | Growth Inhibition Assay | - | IC50=5.8 nM | 23303741 |
| KU-19-19 | - | Growth Inhibition Assay | - | IC50=36 nM | 24784839 |
| LB831-BLC | - | Growth Inhibition Assay | - | IC50=34 nM | 24784839 |
| LNCaP | - | Growth Inhibition Assay | - | IC50=8 nM | 23152004 |
| LoVo | - | Growth Inhibition Assay | - | IC50=22 nM | 24682747 |
| LS-1034 | - | Growth Inhibition Assay | - | IC50=31 nM | 24682747 |
| LS-123 | - | Growth Inhibition Assay | - | IC50=73 nM | 24682747 |
| LS-411 N | - | Growth Inhibition Assay | - | IC50=5 nM | 24682747 |
| M23 | - | Cell Viability Assay | - | IC50=37.5 nM | 23418523 |
| M23 | 250 nM | Function Assay | 24 h | induces G1 and G2/M arrest | 23418523 |
| M23 | 100 nM | Apoptosis Assay | 72 h | significantly induces apoptosis | 23418523 |
| MDA-MB-231 | 100 nM | Function Assay | 30 min | inhibits accumulation of HIF-1α | 24248265 |
| MDA-MB-231 | 100 nM | Function Assay | 24 h | inhibits the migratory and invasive capacity | 24173541 |
| MDA-MB-435 | 100 nM | Function Assay | 30 min | inhibits accumulation of HIF-1α | 24248265 |
| MGC-803 | 0.1-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 25590805 |
| MGC-803 | 0.1-1000 nM | Function Assay | 24 h | induces G2/M cell-cycle arrest | 25590805 |
| MGH-U3 | - | Growth Inhibition Assay | - | IC50=53 nM | 24784839 |
| MKN-28 | 0.1-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 25590805 |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=10.6 nM | 23303741 |
| MV411 | 30/80/150/250 nM | Apoptosis Assay | 24/48/72 h | induces dose dependant induction of apoptosis | 25882550 |
| N592 | - | Growth Inhibition Assay | - | IC50=14.12 nM | 24166505 |
| NALM-6 | - | Growth Inhibition Assay | - | IC50=11.7 nM | 23303741 |
| NB-1643 | - | Growth Inhibition Assay | - | IC50=7.4 nM | 23303741 |
| NB-EBc1 | - | Growth Inhibition Assay | - | IC50=16.8 nM | 23303741 |
| NCI-H1975 | - | Growth Inhibition Assay | 48 h | IC50=16 nM | 22144665 |
| NCI-H1975 | - | Growth Inhibition Assay | 72 h | IC50=8 nM | 22144665 |
| NCI-H747 | - | Growth Inhibition Assay | - | IC50=17 nM | 24682747 |
| OVCAR-5 | 0-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 23900136 |
| OVCAR-5 | 10-100 nM | Apoptosis Assay | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 |
| OVCAR-8 | 0-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 23900136 |
| OVCAR-8 | 10-100 nM | Apoptosis Assay | 24/48/72 h | induces apoptosis time and dose dependently | 23900136 |
| Ramos-RA1 | - | Growth Inhibition Assay | - | IC50=7.4 nM | 23303741 |
| RD | - | Growth Inhibition Assay | - | IC50=8 nM | 23303741 |
| Rh18 | - | Growth Inhibition Assay | - | IC50=6.2 nM | 23303741 |
| Rh30 | - | Growth Inhibition Assay | - | IC50=5.6 nM | 23303741 |
| Rh41 | - | Growth Inhibition Assay | - | IC50=10.4 nM | 23303741 |
| RKO | - | Growth Inhibition Assay | - | IC50=4 nM | 24682747 |
| RS4;11 | - | Growth Inhibition Assay | - | IC50=13.5 nM | 23303741 |
| RT112 | - | Growth Inhibition Assay | - | IC50=9 nM | 24784839 |
| RT4 | - | Growth Inhibition Assay | - | IC50=1733 nM | 24784839 |
| SCaBER | - | Growth Inhibition Assay | - | IC50=10 nM | 24784839 |
| SCC25 | 10/50 nM | Cytoxicity Assay | 24 h | decreases cell proliferation dose dependently | 25205430 |
| SGC-7901 | 0.1-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 25590805 |
| SJ-GBM2 | - | Growth Inhibition Assay | - | IC50=12.9 nM | 23303741 |
| SK-CO-1 | - | Growth Inhibition Assay | - | IC50=81 nM | 24682747 |
| SK-LU-1 | - | Growth Inhibition Assay | - | IC50=18 nM | 23012248 |
| SKOV-3 | 0-1000 nM | Cell Viability Assay | 72 h | inhibits cell viability dose dependently | 23900136 |
| SNU-C2B | - | Growth Inhibition Assay | - | IC50=45 nM | 24682747 |
| SW-1710 | - | Growth Inhibition Assay | - | IC50=6 nM | 24784839 |
| SW620 | - | Growth Inhibition Assay | - | IC50=8 nM | 24682747 |
| SW780 | - | Growth Inhibition Assay | - | IC50=3451 nM | 24784839 |
| T24 | - | Growth Inhibition Assay | - | IC50=7 nM | 24784839 |
| TC-71 | - | Growth Inhibition Assay | - | IC50=4.5 nM | 23303741 |
| TCCSUP | - | Growth Inhibition Assay | - | IC50=142 nM | 24784839 |
| UM-UC3 | - | Growth Inhibition Assay | - | IC50=33 nM | 24784839 |
| VCaP | - | Growth Inhibition Assay | - | IC50=7 nM | 23152004 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01042379 | Breast Neoplasms ... more >> Breast Cancer Breast Tumors Collapse << | Phase 2 | Recruiting | - | - |
| NCT01227018 | Adenocarcinoma of the Pancreas... more >> Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Terminated(interim analysis fo... more >>und the study drug to be ineffective) Collapse << | - | United States, Tennessee ... more >> The Jones Clinic Memphis, Tennessee, United States, 38138 Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << |
| NCT01227018 | - | - | Terminated(interim analysis fo... more >>und the study drug to be ineffective) Collapse << | - | - |
| NCT00964873 | Acute Myeloid Leukemia ... more >> Acute Lymphoblastic Leukemia Blast-phase Chronic Myelogenous Leukemia AML ALL CML Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Tampa, Florida, United States, 33612 United States, Maryland Baltimore, Maryland, United States, 21231 United States, North Carolina Durham, North Carolina, United States, 27710 Collapse << |
| NCT01236144 | Acute Myeloid Leukaemia ... more >> High Risk Myelodysplastic Syndrome Collapse << | Phase 1 Phase 2 | Completed | - | United Kingdom ... more >> Aberdeen Royal Infirmary Aberdeen, United Kingdom Belfast City Hospital Belfast, United Kingdom Birmingham Heartlands Hospital Birmingham, United Kingdom Addenbrooke's Hospital Cambridge, United Kingdom University Hospital of Wales Cardiff, United Kingdom Castle Hill Hospital Hull, United Kingdom St James's University Hospital Leeds, United Kingdom Royal Liverpool University Hospital Liverpool, United Kingdom St Bartholomew's Hospital London, United Kingdom Christie Hospital Manchester, United Kingdom Manchester Royal Infirmary Manchester, United Kingdom Freeman Hospital Newcastle, United Kingdom Nottingham University Hospital Nottingham, United Kingdom Collapse << |
| NCT01270880 | - | - | Completed | - | - |
| NCT02637375 | Breast Cancer | Not Applicable | Withdrawn(Study never opened (... more >>terminated as study drug no longer available)) Collapse << | December 2018 | - |
| NCT01031225 | Non Small Cell Lung Cancer | Phase 2 | Completed | - | - |
| NCT01270880 | Adenocarcinoma of the Prostate... more >> Hormone-resistant Prostate Cancer Recurrent Prostate Cancer Stage IV Prostate Cancer Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Hospital Baltimore, Maryland, United States, 21231 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, New Jersey University of Medicine nd Denistry of New Jersey Piscataway, New Jersey, United States, 08854 United States, Wisconsin University of Wisconsin Cancer Center Riverview Wisconsin Rapids, Wisconsin, United States, 54494 Collapse << |
| NCT00858572 | AML CML ... more >> MDS Myeloproliferative Disorders Collapse << | Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02115 United States, New York New York, New York, United States, 10065 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT01111838 | Colon Cancer ... more >>Rectal Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01111838 | - | - | Completed | - | - |
| NCT01273896 | - | - | Completed | - | - |
| NCT02012192 | Epithelial Ovarian Cancer ... more >> Fallopian Tube Cancer Primary Peritoneal Cancer Collapse << | Phase 1 Phase 2 | Terminated(production of IMP h... more >>as stopped) Collapse << | - | Austria ... more >> Medical University Innsbruck, Department for Gynaecology and Obstetrics Innsbruck, Austria, 6020 Belgium Katholieke Universiteit Leuven, Dept. of Gynaecologic Oncology Leuven, Belgium, 3000 France Centre de lutte contre le cancer Francois Baclesse Caen, France, 14076 Centre Anticancereux Léon Bérard Lyon, France, 69373 Assistance Publique - Hôpitaux de Paris Medical Oncology Department Paris, France, 45004 Germany Universitätsmedizin Berlin Charité, Dept. for Gynecology Berlin, Germany, 10117 University Hospital Carl Gustav Carus Dresden, Department of Gynaecology and Obstetrics Dresden, Germany, 01069 Kliniken Essen Mitte, Evang. Huyssens-Stiftung / Knappschaft GmbH Department of Gynaecologic Oncology Essen, Germany, 45136 Universitätsklinikum Hamburg-Eppendorf Dept. of Gynecology and Gynecologic Oncology Hamburg, Germany, 20246 Otto-von-Guericke-Universität Magdeburg Magdeburg, Germany, 39106 Collapse << |
| NCT01273896 | Breast Cancer | Phase 2 | Completed | - | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan-Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memorial Sloan-Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT00688116 | Solid Tumors | Phase 1 | Unknown | November 2014 | United States, California ... more >> UCLA Los Angeles, California, United States United States, Massachusetts Massachusetts General Hospital Cancer Center Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center (BIDMC) - Cancer Center Boston, Massachusetts, United States, 02115 Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Michigan Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 Collapse << |
| NCT01173523 | - | - | Completed | - | - |
| NCT01348126 | Non-small Cell Lung Cancer Sta... more >>ge IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Metastatic Collapse << | Phase 2 Phase 3 | Terminated | - | - |
| NCT01368003 | Adenocarcinoma of the Prostate | Phase 2 | Withdrawn(Loss of funding.) | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01167114 | Esophagogastric Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01551693 | Melanoma | Phase 2 | Terminated(due to weak accrual... more >>) Collapse << | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01173523 | Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massacusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00687934 | Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> Premiere Oncology Santa Monica, California, United States, 90404-2111 United States, Ohio US Oncology Dayton Oncology and Hematology, P.A Kettering, Ohio, United States, 45409 Collapse << |
| NCT01551693 | - | - | Terminated(due to weak accrual... more >>) Collapse << | - | - |
| NCT01665937 | Hepatocellular Carcinoma | Phase 1 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01590160 | Lung Cancer - Malignant Pleura... more >>l Mesothelioma Collapse << | Phase 1 Phase 2 | Active, not recruiting | November 2018 | United Kingdom ... more >> UCL Cancer Trials Centre London, United Kingdom, W1T 4TJ Collapse << |
| NCT01039519 | Gastrointestinal Stromal Tumor | Phase 2 | Completed | - | United States, California ... more >> UCLA Medical Center Los Angeles, California, United States, 90095 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Oregon Oregon Health and Science University-Knight Cancer Institute Portland, Oregon, United States, 97239 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT01677455 | Breast Cancer ... more >> HER-2 Positive Breast Cancer Triple Negative Breast Cancer ER/Progressive Response (PR) + Refractory to Prior Hormonal Treatment Collapse << | Phase 2 | Completed | - | - |
| NCT02261805 | Cancer Small ... more >>Cell Lung Cancer Collapse << | Phase 1 Phase 2 | Terminated(Company terminated ... more >>drug support) Collapse << | - | United States, District of Col... more >>umbia Georgetown Lombardi Comprehensive Cancer Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT01039519 | - | - | Completed | - | - |
| NCT02261805 | - | - | Terminated(Company terminated ... more >>drug support) Collapse << | - | - |
| NCT02389751 | Gastroesophageal Junction Aden... more >>ocarcinoma Malignant Neoplasm of the Cervical Esophagus Malignant Neoplasm of the Thoracic Esophagus Stage IIA Esophageal Cancer AJCC v7 Stage IIB Esophageal Cancer AJCC v7 Stage IIIA Esophageal Cancer AJCC v7 Stage IIIB Esophageal Cancer AJCC v7 Stage IIIC Esophageal Cancer AJCC v7 Collapse << | Phase 1 | Active, not recruiting | April 10, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01183364 | Solid Tumor Malignancies | Phase 1 | Completed | - | United States, Georgia ... more >> Emory University School of Medicine Atlanta, Georgia, United States, 30322 Collapse << |
| NCT02060253 | HER2-positive Breast Cancer ... more >> Male Breast Cancer Recurrent Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Stage IV Breast Cancer Collapse << | Phase 1 | Completed | - | United States, New York ... more >> NYU Cancer Institute New York, New York, United States, 10016 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01200238 | Ocular Melanoma | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01798485 | Non-Small-Cell Lung Adenocarci... more >>noma Non-small Cell Lung Cancer Stage IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Metastatic Collapse << | Phase 3 | Terminated(The study was stopp... more >>ed after the first Interim Analysis due to futility.) Collapse << | - | - |
| NCT01962948 | Recurrent Fallopian Tube Cance... more >>r Recurrent Ovarian Epithelial Cancer Recurrent Primary Peritoneal Cavity Cancer Collapse << | Phase 1 Phase 2 | Active, not recruiting | July 7, 2019 | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT01562015 | Non Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Florida ... more >> Synta Pharmaceuticals Investigative Site Tampa, Florida, United States, 33612 United States, Ohio Synta Pharmaceuticals Investigative Site Cleveland, Ohio, United States, 44195 Canada, Ontario Synta Pharmaceuticals Investigative Site Hamilton, Ontario, Canada, ON L8V 5C2 Synta Pharmaceuticals Investigative Site Ottawa, Ontario, Canada, K1H 8L6 Synta Pharmaceuticals Investigative Site Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01579994 | Advanced Lung Cancer | Phase 1 | Active, not recruiting | April 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States Memoral Sloan Kettering Cancer Center at Phelps Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01200238 | - | - | Completed | - | - |
| NCT01798485 | - | - | Terminated(The study was stopp... more >>ed after the first Interim Analysis due to futility.) Collapse << | - | - |
| NCT02334319 | Stage I Hypopharyngeal Squamou... more >>s Cell Carcinoma Stage I Laryngeal Squamous Cell Carcinoma Stage I Oral Cavity Squamous Cell Carcinoma Stage I Oropharyngeal Squamous Cell Carcinoma Stage II Hypopharyngeal Squamous Cell Carcinoma Stage II Laryngeal Squamous Cell Carcinoma Stage II Oral Cavity Squamous Cell Carcinoma Stage II Oropharyngeal Squamous Cell Carcinoma Stage III Hypopharyngeal Squamous Cell Carcinoma Stage III Laryngeal Squamous Cell Carcinoma Stage III Oral Cavity Squamous Cell Carcinoma Stage III Oropharyngeal Squamous Cell Carcinoma Stage IVA Hypopharyngeal Squamous Cell Carcinoma Stage IVA Laryngeal Squamous Cell Carcinoma Stage IVA Oral Cavity Squamous Cell Carcinoma Stage IVA Oropharyngeal Squamous Cell Carcinoma Collapse << | Phase 1 | Terminated(Lack of funding) | - | United States, Georgia ... more >> Emory University Hospital Midtown Atlanta, Georgia, United States, 30308 Emory University/Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01485835 | Multiple Myeloma | Phase 1 | Completed | - | United States, Colorado ... more >> Colorado Blood Cancer Institute Denver, Colorado, United States, 80218 United States, Georgia Emory University Hospital Midtown Atlanta, Georgia, United States, 30308 Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Michigan Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Missouri Siteman Cancer Center at Washington University Saint Louis, Missouri, United States, 63110 United States, North Carolina Levine Cancer Institute Charlotte, North Carolina, United States, 28204 United States, Texas The Center for Cancer and Blood Disorders Fort Worth, Texas, United States, 76104 United States, Virginia Virginia Cancer Specialists Fairfax, Virginia, United States, 22031 Collapse << |
| NCT02008877 | Malignant Peripheral Nerve She... more >>ath Tumors (MPNST) Sarcoma Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Sarcoma Oncology Center Santa Monica, California, United States, 90403 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 United States, Maryland National Cancer Institute Bethesda, Maryland, United States, 20892 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Missouri Washington University Saint Louis, Missouri, United States, 63130 United States, Utah Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT01554969 | Rectal Cancer | Phase 1 | Completed | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT02192541 | Neoplasms | Phase 1 | Terminated(Drug supplier suspe... more >>nded further clinical development of Ganetespib) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02192541 | - | - | Terminated(Drug supplier suspe... more >>nded further clinical development of Ganetespib) Collapse << | - | - |
| NCT01560416 | Breast Cancer | Phase 2 | Active, not recruiting | December 2018 | United States, Massachusetts ... more >> DFCI at Faulkner Hospital Boston, Massachusetts, United States, 02130 Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, New Hampshire New Hampshire Oncology and Hematology, P.A. Concord, New Hampshire, United States, 03301 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| HSP90 | IC50:4nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
