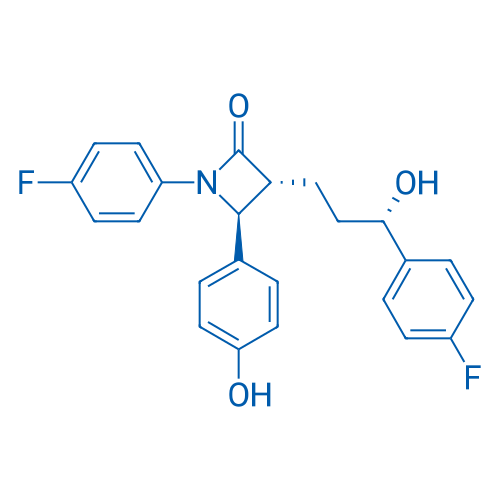
CAS No.: 163222-33-1
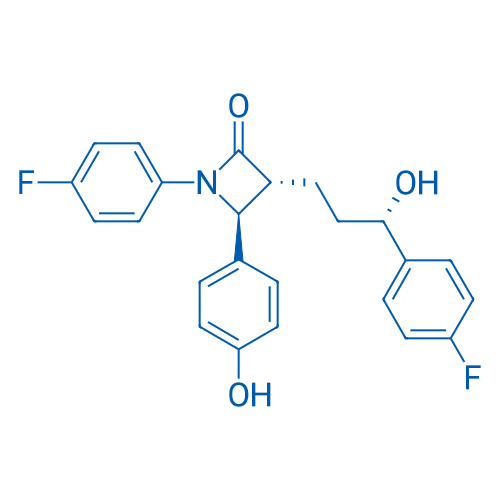
Ezetimibe/依泽替米贝 Catalog No. CSN12575
Synonyms: SCH 58235;Ezetrol;Zetia
Ezetimibe is a cholesterol-absorption inhibitor, reduces levels of low-density lipoprotein (LDL) cholesterol when added to statin treatment. It's also a Niemann-Pick C1-like1 (NPC1L1) inhibitor and Nrf2 activator.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00101439 Hypercholesterolemia Phase 3 Completed - - NCT00101439 - - Completed - - NCT00863265 - - Completed - - - 更多
- 参考文献
- [1] Clader JW. The discovery of ezetimibe: a view from outside the receptor. J Med Chem. 2004 Jan 1;47(1):1-9.
- [2] van Heek M, Farley C, et al. Ezetimibe selectively inhibits intestinal cholesterol absorption in rodents in the presence and absence of exocrine pancreatic function. Br J Pharmacol. 2001 Sep;134(2):409-17.
- [3] Lee DH, Han DH, Nam KT, Park JS, Kim SH, Lee M, Kim G, Min BS, Cha BS, Lee YS, Sung SH, Jeong H, Ji HW, Lee MJ, Lee JS, Lee HY, Chun Y, Kim J, Komatsu M, Lee YH, Bae SH. Ezetimibe, an NPC1L1 inhibitor, is a potent Nrf2 activator that protects mice from diet-induced nonalcoholic steatohepatitis. Free Radic Biol Med. 2016 Oct;99:520-532
- [4] Chang E, Kim L, Park SE, Rhee EJ, Lee WY, Oh KW, Park SW, Park CY. Ezetimibe improves hepatic steatosis in relation to autophagy in obese and diabetic rats. World J Gastroenterol. 2015 Jul 7;21(25):7754-63
- [5] Zhang Y, Liu J, Li S, Xu RX, Sun J, Li JJ. Impact of currently prescribed lipid-lowering drugs on plasma PCSK9 concentration: single or in combination study in rats. Lipids Health Dis. 2014 Feb 18;13:35
- [6] Kiourtzidis M, Kühn J, Schutkowski A, Baur AC, Hirche F, Stangl GI. Inhibition of Niemann-Pick C1-like protein 1 by ezetimibe reduces uptake of deuterium-labeled vitamin D in mice. J Steroid Biochem Mol Biol. 2020 Mar;197:105504
- [7] Tie C, Gao K, Zhang N, Zhang S, Shen J, Xie X, Wang JA. Ezetimibe Attenuates Atherosclerosis Associated with Lipid Reduction and Inflammation Inhibition. PLoS One. 2015 Nov 10;10(11):e0142430
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 163222-33-1 | 储存条件 |
|
|||||||||||||
| 分子式 | C24H21F2NO3 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 409.43 | 别名 | SCH 58235;Ezetrol;Zetia;Ezetrol,;依泽替米贝 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00101439 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00101439 | - | - | Completed | - | - |
| NCT00863265 | - | - | Completed | - | - |
| NCT00863265 | Hypercholesterolemia ... more >> Coronary Heart Disease Collapse << | Early Phase 1 | Completed | - | United States, Utah ... more >> Center for Advance Nutrition at Utah State University Logan, Utah, United States, 84322-4715 Collapse << |
| NCT00677248 | Hypercholesterolemia | Phase 2 | Completed | - | Sweden ... more >> Karo Bio AB HUddinge, Sweden Collapse << |
| NCT00651391 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 3 | Terminated(Slow enrollment) | - | - |
| NCT00905905 | Myocardial Infarction | Phase 4 | Completed | - | Brazil ... more >> Hospital de Base do Distrito Federal Brasilia, DF, Brazil, 70673103 Collapse << |
| NCT00699023 | Postprandial Lipemia ... more >> Type 2 Diabetes Collapse << | Phase 4 | Completed | - | Italy ... more >> Department of Clinical and Experimental Medicine, Federico II University Hospital, Naples, Italy, 80131 Collapse << |
| NCT00299884 | - | - | Completed | - | Canada, Ontario ... more >> Vascular Disease Prevention and Research Centre, Hotel Dieu Hospital Kingston, Ontario, Canada, K7L 5G2 Collapse << |
| NCT00753883 | Hyperlipidemia | Phase 4 | Completed | - | - |
| NCT00423579 | Hypercholesterolemia ... more >> Coronary Disease Collapse << | Phase 4 | Completed | - | - |
| NCT00423579 | - | - | Completed | - | - |
| NCT03051100 | Hypercholesterolemia | Phase 2 | Completed | - | United States, Illinois ... more >> PMG Research of Christie Clinic Champaign, Illinois, United States, 61820 United States, North Carolina PMG Research of Cary Cary, North Carolina, United States, 27518 PMG Research of Charlotte Charlotte, North Carolina, United States, 28209 Sensenbrenner Primary Care Charlotte, North Carolina, United States, 28277 PMG Research of Hickory Hickory, North Carolina, United States, 28601 PMG Research of Raleigh Raleigh, North Carolina, United States, 27609 PMG Research of Rocky Mount Rocky Mount, North Carolina, United States, 27804 PMG Research Salisbury Salisbury, North Carolina, United States, 28144 PMG Research of Wilmington Wilmington, North Carolina, United States, 28401 United States, South Carolina PMG Research of Charleston Mount Pleasant, South Carolina, United States, 29464 United States, Virginia Hampton Roads Center for Clinical Research Virginia Beach, Virginia, United States, 23451 Collapse << |
| NCT00909389 | - | - | Completed | - | - |
| NCT00909389 | - | - | Completed | - | - |
| NCT00481351 | Coronary Heart Disease | Phase 4 | Completed | - | - |
| NCT01603758 | Coronary Heart Disease ... more >> Cardiovascular Disease Dyslipidemia Disorder of Cholesterol Metabolism Collapse << | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00481351 | - | - | Completed | - | - |
| NCT00268697 | Hypercholesterolemia | Phase 3 | Completed | - | Estonia ... more >> Tallinn, Estonia Tartu, Estonia Latvia Riga, Latvia Russian Federation Moscow, Russian Federation Saratov, Russian Federation Smolensk, Russian Federation St. Petersburg, Russian Federation Tyumen, Russian Federation Serbia Kragujevac, Serbia Collapse << |
| NCT01370590 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01370590 | - | - | Completed | - | - |
| NCT01370603 | - | - | Completed | - | - |
| NCT00705081 | - | - | Completed | - | - |
| NCT00705081 | - | - | Completed | - | - |
| NCT00819403 | Metabolic Syndrome | Phase 4 | Completed | - | United States, Maryland ... more >> VA Maryland Health Care System Baltimore, Maryland, United States, 21201 University of Maryland Medical Center Baltimore, Maryland, United States, 21202 Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01375764 | - | - | Completed | - | - |
| NCT00819403 | - | - | Completed | - | - |
| NCT01035320 | Diabetes Mellitus ... more >> Renal Impairment Collapse << | Phase 4 | Completed | - | Sweden ... more >> Department of Medicine, Clinical Pharmacology Unit, Karolinska University Hospital (Solna) Stockholm, Sweden, 171 76 Collapse << |
| NCT02127320 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, Korea, Republic of Collapse << |
| NCT00650689 | Hypercholesterolemia ... more >> Atherosclerosis Coronary Artery Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01375764 | Hyperlipidemia | Phase 2 | Completed | - | - |
| NCT00701727 | Hypercholesterolemia | Phase 4 | Completed | - | United States, Illinois ... more >> Radiant Research Chicago, Illinois, United States, 60610 Collapse << |
| NCT00701727 | - | - | Completed | - | - |
| NCT03771053 | Coronary Heart Disease | Not Applicable | Recruiting | December 31, 2019 | China, Beijing ... more >> Chinese People's Liberation Army General Hospital Recruiting Peking, Beijing, China, 100853 Contact: Geng Qian, MD 086-01055499312 qiangeng9396@263.net Contact: Yun-dai Chen, MD 086-01055499309 cyundai@126.com Collapse << |
| NCT00474123 | Stable Angina | Not Applicable | Completed | - | Brazil ... more >> Heart Institute (InCor) HOSPITAL DAS CLINICAS DA FACULDADE DE MEDICINA DA UNIVERSIDADE DE SAO PAULO (HCFMUSP) Sao Paulo, Brazil, 05403-000 Collapse << |
| NCT00474123 | - | - | Completed | - | - |
| NCT03337308 | Hyperlipidemias | Phase 3 | Completed | - | United States, Iowa ... more >> PMG Research of McFarland Ames, Iowa, United States, 50010 United States, New Hampshire Foundation Cardiology Nashua, New Hampshire, United States, 03060 United States, North Carolina PMG Research of Piedmont Healthcare Statesville, North Carolina, United States, 28625 PMG Research of Wilmington Wilmington, North Carolina, United States, 28401 United States, Tennessee PMG Research of Knoxville Knoxville, Tennessee, United States, 37912 Collapse << |
| NCT00652847 | Hypercholesterolemia | Phase 4 | Completed | - | - |
| NCT01941836 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| NCT00651144 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 1 | Completed | - | - |
| NCT00705211 | - | - | Completed | - | - |
| NCT00783263 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00783263 | - | - | Completed | - | - |
| NCT00932620 | - | - | Completed | - | - |
| NCT00932620 | Hypercholesterolemia | Phase 4 | Completed | - | Greece ... more >> University Hospital of Ioannina Ioannina, Greece, 45 110 Collapse << |
| NCT00653835 | Hypercholesterolaemia ... more >> Atherosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01370603 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00651963 | Lipid Metabolism Disorder | Phase 4 | Completed | - | - |
| NCT00650819 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01597700 | Hypercholesterolaemia | Phase 1 | Completed | - | India ... more >> GSK Investigational Site Electronics City, Bengalore, India, 560100 Collapse << |
| NCT00704444 | - | - | Completed | - | - |
| NCT00653796 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT00965055 | High Cholesterol ... more >> Coronary Artery Disease Collapse << | Phase 3 | Terminated(The study was termi... more >>nated due to inability to recruit subjects. A total of 2/100 anticipated were radomized.) Collapse << | - | United States, California ... more >> UCSD Medical Center in Hillcrest Clinical Trials Facility San Diego, California, United States, 92103 Collapse << |
| NCT01241097 | Endothelial Function | Not Applicable | Unknown | December 2011 | Brazil ... more >> Escola Bahiana de Medicina e Saúde Púiblica Recruiting Salvador, Bahia, Brazil, 40000 Contact: Marilia G Rodrigues, MD 55-71-3359485 mgaleffi@cardiol.br Contact: Paulo Roberto P Lima, student 55-71-99184765 prpassoslima@hotmail.com Sub-Investigator: Carolina Garcez, student Collapse << |
| NCT00817843 | Metabolic Syndrome | Phase 4 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, Netherlands, 1005 AZ Vascular Research Center Hoorn Hoorn, Netherlands, 1624 NP Department of Vascular Medicine UMC Utrecht Utrecht, Netherlands, 3584 CX Tweesteden Ziekenhuis Waalwijk, Netherlands, 5141 BM Spain Hospital Arnau de Vilanova Lleida, Spain, E-25198 Collapse << |
| NCT00814723 | Hypercholesterolemia | Phase 4 | Completed | - | Austria ... more >> Depart. of Internal Medicine, Medical University of Graz Graz, Austria, 8036 Collapse << |
| NCT00651404 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 3 | Completed | - | - |
| NCT00650663 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT00129402 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00129402 | - | - | Completed | - | - |
| NCT00652301 | Cholesterol | Phase 3 | Completed | - | - |
| NCT03044665 | Cardiovascular Diseases ... more >> Cerebrovascular Disease Peripheral Atherosclerotic Disease Collapse << | Not Applicable | Recruiting | February 15, 2022 | Korea, Republic of ... more >> Division of Cardiology, Yonsei Cardiovascular Hospital, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 03722 Contact: Yang-Soo Jang, MD, PhD Collapse << |
| NCT03434613 | Nonalcoholic Fatty Liver Disea... more >>se Dyslipidemias Collapse << | Phase 4 | Not yet recruiting | September 1, 2019 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Not yet recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT02089867 | Coronary Artery Disease | Phase 2 Phase 3 | Completed | - | Brazil ... more >> Jose Rocha Faria Neto Curitiba, Paraná, Brazil, 80215-901 Collapse << |
| NCT01611883 | Hypercholesterolemia | Phase 4 | Completed | - | - |
| NCT01611883 | - | - | Completed | - | - |
| NCT00651014 | Hypercholesterolemia ... more >> Atherosclerosis Collapse << | Phase 4 | Terminated(slow subject recrui... more >>tment and lack of medical and scientific merit due to change in new standard of therapy during that same period.) Collapse << | - | - |
| NCT03001076 | Hypercholesterolemia ... more >> Atherosclerosis Statin Adverse Reaction Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Georgetown, Texas, United States, 78626 Collapse << |
| NCT02288338 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> ChongKunDang Seoul, Korea, Republic of Collapse << |
| NCT01103648 | Prediabetes H... more >>ypercholesterolemia Inflammation Cardiovascular Risk Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital do Rim e Hipertensao da UNIFESP Sao Paulo, Brazil, 04038-002 Collapse << |
| NCT01086020 | Coronary Artery Disease | Phase 4 | Unknown | December 2012 | China, Shanghai ... more >> Ruijin Hospital, Recruiting Shanghai, Shanghai, China, 200025 Contact: Ruiyan Zhang, MD 862164370045 ext 665215 zhangruiyan@263.net Contact: Xin Chen, MD 862164370045 ext 665380 rjchenxin@yahoo.com.cn Principal Investigator: Ruiyan Zhang, MD Collapse << |
| NCT00319449 | Hypercholesterolemia ... more >> Coronary Arteriosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT00817843 | - | - | Completed | - | - |
| NCT01584206 | Sitosterolemia | Not Applicable | Unknown | December 2016 | Canada, Manitoba ... more >> Richardson Centre for Functional Foods and Nutraceuticals Winnipeg, Manitoba, Canada, R3T 2N2 Collapse << |
| NCT01490229 | Coronary Artery Disease | Phase 4 | Unknown | December 2016 | Italy ... more >> San Raffaele Pisana Recruiting Rome, Italy, 00100 Contact: Giuseppe Marazzi, MD +39 335 8381320 giuseppe.marazzi@yahoo.com Collapse << |
| NCT00319449 | - | - | Completed | - | - |
| NCT00653913 | Hypercholesterolemia | Phase 1 | Completed | - | - |
| NCT00772551 | HIV | Phase 1 | Completed | - | United Kingdom ... more >> Chelsea and Westminster Hospital NHS Foundation Trust London, United Kingdom, SW10 9NH Collapse << |
| NCT00653276 | Cholesterol | Phase 1 | Completed | - | - |
| NCT01950884 | •Non-alcoholic Steatohepatitis... more >> (NASH) Collapse << | Phase 4 | Unknown | December 2014 | Italy ... more >> Dipartimento Biomedico di Medicina Interna e Specialistica Di.Bi.M.I.S. Palermo, Italy, 90127 Collapse << |
| NCT00202904 | Hypercholesterolemia ... more >> Coronary Heart Disease Collapse << | Phase 4 | Completed | - | - |
| NCT00738985 | Cardiovascular Diseases ... more >> Hyperlipidemia Collapse << | Phase 4 | Withdrawn(The study was cancel... more >>led due to budget limitations) Collapse << | - | - |
| NCT01766713 | - | - | Completed | - | - |
| NCT03217409 | Diabetes Mellitus and Hypercho... more >>lesterolemia Collapse << | Phase 4 | Recruiting | July 2018 | Korea, Republic of ... more >> Soon Chun Hyang University Hospital Cheonan Recruiting Cheonan, Korea, Republic of Soon Chun Hyang University Hospital Gumi Recruiting Gumi, Korea, Republic of Soon Chun Hyang University Hospital Bucheon Recruiting Gyeonggi-do, Korea, Republic of Soon Chun Hyang University Hospital Seoul Recruiting Seoul, Korea, Republic of Collapse << |
| NCT01766713 | Non Alcoholic Steatohepatitis | Phase 2 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 Collapse << |
| NCT01008345 | Coronary Artery Disease ... more >> Diabetes Collapse << | Phase 4 | Completed | - | Lebanon ... more >> Hotel Dieu de France Hospital Achrafieh, Beirut, Lebanon, 0000 Collapse << |
| NCT00653445 | Hypercholesterolemia ... more >> Coronary Heart Disease Atherosclerosis Collapse << | Phase 3 | Completed | - | - |
| NCT00867165 | Primary Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT02103049 | Dyslipidemia ... more >>Transplants Collapse << | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00621101 | Pharmacokinetics ... more >> Drug Interactions Hypercholesterolemia Immunosuppression Collapse << | Phase 1 | Completed | - | Germany ... more >> Department of Clinical Pharmacology Greifswald, Germany, 17487 Collapse << |
| NCT00654095 | - | - | Completed | - | - |
| NCT03277079 | Coronary Artery Disease | Phase 4 | Not yet recruiting | March 31, 2018 | Italy ... more >> Sapienza University Rome, Italy, 00161 Collapse << |
| NCT00653523 | - | - | Completed | - | - |
| NCT01385631 | ST-Segment Elevation Myocardia... more >>l Infarction Collapse << | Phase 4 | Completed | - | Denmark ... more >> Department of Cardiology, Odense University Hospital Odense C, Denmark, 5000 Collapse << |
| NCT00908011 | Hypercholesterolemia ... more >> HIV Infections Collapse << | Not Applicable | Completed | - | Canada, British Columbia ... more >> St. Paul's Hospital HIV Immunodeficiency/Metabolic Clinic Vancouver, British Columbia, Canada, V6Z 1Y6 Collapse << |
| NCT00867165 | - | - | Completed | - | - |
| NCT00654095 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00908011 | - | - | Completed | - | - |
| NCT00704535 | - | - | Completed | - | - |
| NCT00653523 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00621699 | Pharmacokinetics ... more >> Drug Interactions Hypercholesterolemia Immunosuppression Collapse << | Phase 1 | Completed | - | Germany ... more >> Department of Clinical Pharmacology Greifswald, Germany, 17487 Collapse << |
| NCT02304926 | Dyslipidemia | Not Applicable | Completed | - | - |
| NCT02289430 | Healthy | Phase 1 | Completed | - | - |
| NCT03169985 | Coronary Artery Disease | Phase 4 | Recruiting | December 31, 2023 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of, 06351 Contact: Joo-Yong Hahn, MD, PhD 82-2-3410-6653 ichjy1@gmail.com Contact: Joo Myung Lee, MD, MPH 82-2-3410-1246 drone80@hanmail.net Collapse << |
| NCT02304926 | - | - | Completed | - | - |
| NCT02244944 | Nonalcoholic Fatty Liver Disea... more >>se (NAFLD) Collapse << | Phase 2 | Terminated(Failure to recruit) | - | United States, Kentucky ... more >> University of Kentucky Medical Center Lexington, Kentucky, United States, 40536 Collapse << |
| NCT00634140 | Chronic Acalculous Cholecystit... more >>is Collapse << | Not Applicable | Withdrawn(Study was never init... more >>iated due to lack of funding support.) Collapse << | August 2009 | United States, Indiana ... more >> Indiana University Hospital Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00651274 | Atherosclerosis ... more >> Hypercholesterolemia Coronary Heart Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01807078 | Coronary Artery Disease | Phase 4 | Unknown | December 2017 | Italy ... more >> Sapienza University Not yet recruiting Rome, Lazio, Italy, 00161 Contact: Francesco Pelliccia, MD +39064997 ext 123 f.pelliccia@mclink.it Contact: Giuseppe Marazzi, MD +39064997 ext 123 md4151@mclink.it Principal Investigator: Francesco Pelliccia, MD Collapse << |
| NCT02588235 | Coronary Artery Disease | Phase 4 | Unknown | November 2017 | China, Shaanxi ... more >> Xijing Hospital, Fourth Military Medical University Recruiting Xi'an, Shaanxi, China, 710032 Contact: Dongdong Sun, M.D., Ph.D. Collapse << |
| NCT00704535 | - | - | Completed | - | - |
| NCT02244944 | - | - | Terminated(Failure to recruit) | - | - |
| NCT02126137 | Chronic Hepatitis C | Phase 1 | Unknown | March 2016 | Chile ... more >> Department of Gastroenterology, Pontificia Universidad Católica de Chile Recruiting Santiago, Metropolitan, Chile, 8330024 Contact: Pilar Labbé, RN +56223543820 pililabbe@gmail.com Principal Investigator: Alejandro Soza, MD Collapse << |
| NCT00762229 | Hypercholesterolemia | Not Applicable | Completed | - | United States, New York ... more >> Bronx VA Medical Center Bronx, New York, United States, 10468 Collapse << |
| NCT02768545 | Hepatitis C | Phase 4 | Unknown | June 2016 | Chile ... more >> Departamento de Gastroenterología, Pontificia Universidad Católica de Chile Recruiting Santiago, RM, Chile, 833-0024 Contact: Alejandro Soza, MD 56-22-6397780 asoza@med.puc.cl Contact: Hugo Monrroy, MD 56-22-6397780 hmonrroy@gmail.com Collapse << |
| NCT00794677 | Hypercholesterolemia | Phase 4 | Completed | - | United States, California ... more >> Cholesterol Research Center Berkeley, California, United States, 94705 Collapse << |
| NCT00794677 | - | - | Completed | - | - |
| NCT02029625 | Dyslipidemia | Phase 1 | Completed | - | Korea, Republic of ... more >> Kyungpook National Hospital Jung-gu, Daegu, Korea, Republic of, 700-721 Collapse << |
| NCT02548936 | Atherosclerosis | Early Phase 1 | Unknown | December 2016 | China, Beijing ... more >> Peking Union Medical College Hospital Beijing, Beijing, China, 100730 Collapse << |
| NCT00762229 | - | - | Completed | - | - |
| NCT01849068 | Metabolic Syndrome X | Phase 3 | Completed | - | Canada ... more >> Laval University Quebec, Canada, G1V 0A6 Collapse << |
| NCT01849068 | - | - | Completed | - | - |
| NCT03099278 | Chronic Hepatitis D | Phase 2 | Recruiting | October 2017 | Pakistan ... more >> Ziauddin University Hospital Recruiting Karachi, Sindh, Pakistan, 75600 Contact: Zaigham Abbas Collapse << |
| NCT02772640 | Hypercholesterolemia | Phase 4 | Unknown | December 2016 | Poland ... more >> Cardiology Department, Dr. A. Jurasz University Hospital Recruiting Bydgoszcz, Kujawsko-Pomorskie, Poland, 85-094 Contact: Karolina Obońska, MD, PhD +48 52 5854023 kalaobonska@op.pl Collapse << |
| NCT03105310 | Hepatitis D, Chronic | Phase 2 | Active, not recruiting | June 2017 | - |
| NCT02971033 | Chronic Hepatitis C | Phase 2 | Recruiting | June 30, 2020 | United States, Illinois ... more >> Edward Hines Jr. VA Hospital, Hines, IL Recruiting Hines, Illinois, United States, 60141-5000 Contact: Susan L Uprichard, PhD 708-202-8387 ext 25848 Susan.Uprichard@va.gov Principal Investigator: Susan L. Uprichard, PhD Collapse << |
| NCT03531905 | Diabetes Diab... more >>etes Mellitus, Type 2 Cholesterolemia Collapse << | Phase 2 | Recruiting | May 2019 | United States, California ... more >> Clinical Trials Research Recruiting Lincoln, California, United States, 95648 United States, Florida FInlay Medical Research Recruiting Miami, Florida, United States, 33126 United States, Kentucky L-MARC Research Center Recruiting Louisville, Kentucky, United States, 40213 United States, Virginia Hampton Roads Center for Clinical Research Recruiting Suffolk, Virginia, United States, 23435 Collapse << |
| NCT02682680 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | December 2017 | Canada, Alberta ... more >> LMC Calgary Not yet recruiting Calgary, Alberta, Canada Contact: Akshay Jain, MD 403-288-3224 calgaryresearch@lmc.ca Canada, Ontario LMC Barrie Recruiting Barrie, Ontario, Canada Contact: Hani Alasaad, MBBS 705-737-0830 barrieresearch@lmc.ca LMC Brampton Recruiting Brampton, Ontario, Canada, L6S 0C9 Contact: Harpreet Bajaj, MBBS, MPH 905-595-0560 bramptonresearch@lmc.ca LMC Etobicoke Recruiting Etobicoke, Ontario, Canada Contact: Hasnain Khandwala, MBBS 416-645-1035 etobicokeresearch@lmc.ca LMC Markham Recruiting Markham, Ontario, Canada Contact: Nikhil Gupta, MBBS, MPH 905-294-0800 markhamresearch@lmc.ca LMC Oakville Recruiting Oakville, Ontario, Canada Contact: David Twum-Barima, MB ChB, MSc 905-337-0040 oakvilleresearch@lmc.ca LMC Thornhill Recruiting Thornhill, Ontario, Canada Contact: Ronald Goldenberg, MD 905-763-8660 thornhillresearch@lmc.ca LMC Bayview Recruiting Toronto, Ontario, Canada Contact: Ronnie Aronson, MD 416-645-2929 torontoresearch@lmc.ca Collapse << |
| NCT01384058 | Diabetes Mellitus Type 2 ... more >> Hypercholesterolemia Collapse << | Phase 4 | Completed | - | Germany ... more >> Institut für Stoffwechselforschung Frankfurt, Germany, 60322 Stephan Jacob, MD Villingen-.Schwenningen, Germany, 78048 Collapse << |
| NCT02890992 | Hypercholesterolaemia | Phase 2 | Active, not recruiting | February 22, 2019 | United States, Missouri ... more >> Investigational Site Number 8400002 Saint Louis, Missouri, United States, 63110 United States, North Carolina Investigational Site Number 8400005 Charlotte, North Carolina, United States, 28204 United States, Ohio Investigational Site Number 8400001 Cincinnati, Ohio, United States, 45229 Canada Investigational Site Number 1240001 Quebec, Canada, G1V 4W2 Czechia Investigational Site Number 2030001 Brno, Czechia, 62500 Investigational Site Number 2030003 Praha 5 - Motol, Czechia, 15006 Investigational Site Number 2030002 Zlin, Czechia, 76275 France Investigational Site Number 2500001 Bron Cedex, France, 69677 Netherlands Investigational Site Number 5280001 Amsterdam, Netherlands, 1105 AZ Norway Investigational Site Number 5780001 Oslo, Norway, 0373 Russian Federation Investigational Site Number 6430001 Kemerovo, Russian Federation, 650002 Investigational Site Number 6430004 Saint-Petersburg, Russian Federation, 194100 South Africa Investigational Site Number 7100001 Parow, South Africa, 7500 Spain Investigational Site Number 7240004 A Coruna, Spain, 15001 Investigational Site Number 7240001 Madrid, Spain, 28009 Sweden Investigational Site Number 7520001 Stockholm, Sweden, 171 76 Collapse << |
| NCT00079638 | Dyslipidemia ... more >>Coronary Heart Disease Atherosclerosis Stroke Diabetes Collapse << | Phase 4 | Completed | - | - |
| NCT02755194 | - | - | Recruiting | September 2019 | Canada, Ontario ... more >> The Hospital for Sick Children Recruiting Toronto, Ontario, Canada, M5G1X8 Contact: Shinya Ito, MD, FRCPC 416-813-7562 shinya.ito@sickkids.ca Collapse << |
| NCT00120289 | Cardiovascular Diseases ... more >> Heart Diseases Cerebrovascular Accident Coronary Disease Atherosclerosis Myocardial Infarction Collapse << | Phase 3 | Terminated(AIM-HIGH was stoppe... more >>d on the recommendation of the DSMB because of lack of efficacy of niacin in preventing primary outcome events.) Collapse << | - | - |
| NCT01660945 | Hypercholesterolemia | Phase 4 | Completed | - | Korea, Republic of ... more >> Gil Medical Center Incheon, Korea, Republic of, 405-760 Collapse << |
| NCT03214211 | - | - | Not yet recruiting | June 30, 2022 | Korea, Republic of ... more >> Chonnam national university hospital Not yet recruiting Gwangju, Korea, Republic of Contact: Myung-Ho Jeong, Ph.D myungho@chollian.net Collapse << |
| NCT00701883 | Hyperlipidemia | Phase 2 | Completed | - | - |
| NCT00120289 | - | - | Terminated(AIM-HIGH was stoppe... more >>d on the recommendation of the DSMB because of lack of efficacy of niacin in preventing primary outcome events.) Collapse << | - | - |
| NCT03449784 | - | - | Recruiting | May 1, 2041 | France ... more >> Uhmontpellier Recruiting Montpellier, France, 34295 Contact: cyril BREUKER c-breuker@chu-montpellier.fr Collapse << |
| NCT01252875 | Ischemic Stroke ... more >> Transient Ischemic Attack Atherosclerotic Stenosis Collapse << | Not Applicable | Recruiting | December 2019 | France ... more >> BICHAT HOSPITAL Departement of Neurology Recruiting Paris, France, 75018 Contact: Pierre AMARENCO, Pr, MD, PhD +33(0)1 40 25 87 26 Pierre.amarenco@bch.aphp.fr Principal Investigator: Pierre AMARENCO, Pr, MD, PhD BICHAT HOSPITAL Department of neurology and stroke center Recruiting Paris, France, 75018 Contact: Pierre Amarenco, MD +33 (0) 1 40 25 87 26 ext 87 24 pierre.amarenco@bch.aphp.fr Collapse << |
| NCT01308203 | Coronary Artery Disease ... more >> Dyslipidemias Collapse << | Phase 4 | Terminated(Merck has decided t... more >>o discontinue all studies with extended-release niacin/laropiprant.because the HPS2-THRIVE did not meet its primary endpoint) Collapse << | - | Argentina ... more >> Hospital Italiano de Buenos Aires Buenos Aires, Argentina, C1181ACH Collapse << |
| NCT01274559 | Primary Hypercholesterolemia ... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01274559 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT03310671 | - | - | Recruiting | July 18, 2018 | Spain ... more >> Fernando Civeira Murillo Recruiting Zaragoza, Spain, 50009 Contact: Victoria MARCO BENEDÍ, RD 976765500 ext 2895 vmarcobenedi@gmail.com Collapse << |
| NCT02941848 | Primary Hypercholesterolemia | Phase 1 | Completed | - | - |
| NCT00679237 | Coronary Heart Disease | Not Applicable | Active, not recruiting | December 2024 | Norway ... more >> Sorlandet Hospital Arendal, Norway Collapse << |
| NCT02642159 | Dyslipidemia | Phase 4 | Completed | - | - |
| NCT02055976 | Hypercholesterolemia | Phase 2 | Completed | - | Japan ... more >> Maebashi Hirosegawa Clinic Maebashi, Gunma, Japan, 371-0022 Yokohama Minoru Clinic Yokohama, Kanagawa, Japan, 232-0064 Heishinkai Medical Group Incorporated OCROM Clinic Suita, Osaka, Japan, 565-0853 Meiwa Hospital Chiyoda-ku, Tokyo, Japan, 101-0041 Tokyo-Eki Center-building Clinic Chuo-ku, Tokyo, Japan, 103-0027 Heishinkai Medical Group Incorporated ToCROM Clinic Shinjuku-ku, Tokyo, Japan, 160-0022 Clinical Research Hospital Tokyo Shinjuku-ku, Tokyo, Japan, 162-0053 Oda Clinic Shinjuku-ku, Tokyo, Japan, 169-0072 Sekino Hospital Toshima-ku, Tokyo, Japan, 171-0014 Collapse << |
| NCT00655265 | Familial Hypercholesterolemia | Phase 4 | Completed | - | France ... more >> Service d'Endocrinologie, Metabolisme Prevention des Maladies Cardio-Vasculaires Paris Cedex, France Germany Kardiologie Marburg, Germany Netherlands Academic Medical Centre Amsterdam, Netherlands University Medical Centre Nijmegen, Netherlands University Medical Centre Utrecht, Netherlands TweeSteden Ziekenhius Waalwijk, Netherlands Sweden Centre for Metabolism and Endocrinology Huddinge, Sweden United Kingdom Department of Medicine, University of Manchester Manchester, United Kingdom Collapse << |
| NCT02642159 | - | - | Completed | - | - |
| NCT01477944 | - | - | Completed | - | - |
| NCT02942602 | Hyperlipidemia | Not Applicable | Completed | - | Korea, Republic of ... more >> Division of Cardiology, Department of Internal Medicine, Seoul, Korea, Republic of, 03722 Collapse << |
| NCT00359281 | Healthy | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT03433755 | Primary Hypercholesterolemia ... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Not yet recruiting | August 25, 2020 | - |
| NCT01516879 | - | - | Completed | - | - |
| NCT01948648 | Sitosterolemia | Not Applicable | Recruiting | December 31, 2019 | Canada, Manitoba ... more >> Richardson Center for Functional Foods and Nutraceuticals Recruiting Winnipeg, Manitoba, Canada Contact: Semone B Myrie, PhD 204-272-1555 myrie@cc.umanitoba.ca Contact: Peter J Jones, PhD 204-474-8883 Peter.Jones@umanitoba.ca Sub-Investigator: Semone B Myrie, PhD Principal Investigator: Peter J Jones, PhD Sub-Investigator: David Mymim, PhD Sub-Investigator: Taback Shayne, PHD Collapse << |
| NCT00090168 | Hypercholesterolemia ... more >> Atherosclerotic Disease Coronary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01516879 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01420328 | Inflammation | Phase 3 | Unknown | December 2013 | United States, New York ... more >> Diabetes Endocrinology Center of WNY Buffalo, New York, United States, 14209 Collapse << |
| NCT03549260 | LDL Cholesterol | Phase 2 | Active, not recruiting | November 30, 2018 | United States, Indiana ... more >> Midwest Institute For Clinical Research Indianapolis, Indiana, United States, 46260 United States, Kentucky Louisville Metabolic and Atherosclerosis Research Center Louisville, Kentucky, United States, 40213 United States, Ohio Sterling Research Group Cincinnati, Ohio, United States, 45219 The Lindner Research Center Cincinnati, Ohio, United States, 45219 Metabolic & Atherosclerosis Research Center (MARC) Cincinnati, Ohio, United States, 45227 Collapse << |
| NCT00952770 | Coronary Artery Disease | Phase 4 | Unknown | - | Korea, Republic of ... more >> Chungnam National University Hospital Recruiting Daejeon, Korea, Republic of Contact: Jae-Hyeong Park, MD, PhD 82-42-280-7167 jaehpark@cnuh.co.kr Collapse << |
| NCT01070966 | - | - | Completed | - | - |
| NCT00724477 | - | - | Completed | - | - |
| NCT00359281 | - | - | Completed | - | - |
| NCT02205775 | Stable Coronary Artery Disease... more >> Undergoing PCI Collapse << | Phase 3 | Terminated(difficult recruitme... more >>nt) Collapse << | - | Italy ... more >> SS. Annunziata Hospital Chieti, CH, Italy, 66100 Fondazione IRCCS Policlinico S. Matteo Pavia, PV, Italy, 27100 A.O. S. Anna e S. Sebastiano - II Università di Napoli Caserta, Italy, 81100 Azienda ASL 6 - P. Ospedaliero Livorno Livorno, Italy, 57100 Ospedale Civile G. Fornaroli Magenta, Italy, 20013 Azienda Ospedaliera - Ospedale San Paolo Milano, Italy, 20142 Collapse << |
| NCT01070966 | - | - | Completed | - | - |
| NCT01112423 | Dyslipidemia | Phase 2 | Completed | - | United States, Arkansas ... more >> Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, Kansas Pra International Lenexa, Kansas, United States, 66219 United States, Ohio Sterling Research Grp, Ltd. Cincinnati, Ohio, United States, 45219 United States, Texas Cetero Research - San Antonio San Antonio, Texas, United States, 78229 United States, Virginia National Clinical Research - Norfolk, Inc. Norfolk, Virginia, United States, 23502 National Clinical Research - Richmond, Inc. Richmond, Virginia, United States, 23294 Australia, Queensland Local Institution Brisbane, Queensland, Australia, 4021 Canada, Manitoba Local Institution Winnipeg, Manitoba, Canada, R2V 4W3 Canada, Newfoundland and Labrador Local Institution Mount Pearl, Newfoundland and Labrador, Canada, A1N 1W7 Canada, Ontario Local Institution London, Ontario, Canada, N6A 5R8 Canada, Quebec Local Institution Drummondville, Quebec, Canada, J2B 7T1 Local Institution Montreal, Quebec, Canada, H3J 2V5 Collapse << |
| NCT00724477 | - | - | Completed | - | - |
| NCT00548145 | - | - | Terminated | - | - |
| NCT00548145 | Alzheimer Disease ... more >> Hypercholesterolemia Collapse << | Not Applicable | Terminated | - | Japan ... more >> Osaka University Hospital Suita, Osaka, Japan, 565-0871 Collapse << |
| NCT00738972 | Hypertension | Phase 3 | Terminated(Study terminated ea... more >>rly due to sample size, not possible to perform further statistical analysis.) Collapse << | - | - |
| NCT00738972 | - | - | Terminated(Study terminated ea... more >>rly due to sample size, not possible to perform further statistical analysis.) Collapse << | - | - |
| NCT00092807 | Lipid Metabolism, Inborn Error... more >>s Heart Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00559962 | Hyperlipidemia | Phase 2 | Completed | - | United States, California ... more >> Scripps Clinic San Diego, California, United States, 92128 Radiant Research Santa Rosa, California, United States, 95405 United States, District of Columbia MedStar Research Institute Washington, District of Columbia, United States, 20003 United States, Illinois Radiant Research Chicago, Illinois, United States, 60610 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 United States, Kentucky LMARC Louisville, Kentucky, United States, 40213 United States, Maine Maine Research Associates Auburn, Maine, United States, 04210 United States, Maryland Health Trends Research Baltimore, Maryland, United States, 21209 Johns Hopkins Baltimore, Maryland, United States, 21287 United States, Missouri Washington Univ. School of Medicine Saint Louis, Missouri, United States, 63110 United States, Ohio Sterling Research Group Cincinnati, Ohio, United States, 45219 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 Clinical Trial Network Houston, Texas, United States, 77074 dgd Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT00092820 | Lipid Metabolism, Inborn Error... more >>s Heart Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00132717 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT02767362 | Endometrial Cancer | Early Phase 1 | Recruiting | November 2020 | United States, North Carolina ... more >> University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Leslie Clark, MD 919-843-7572 leslie_clark@med.unc.edu Principal Investigator: Leslie Clark, MD Collapse << |
| NCT01890889 | Cholesterol H... more >>yperlipidemia Hypercholesteremia Collapse << | Not Applicable | Unknown | December 2013 | United States, Texas ... more >> Integrative Health Technologies Recruiting San Antonio, Texas, United States, 78209 Contact: Patricia l Keith, BBA 210-824-4200 hmrcenterstudy@gmail.com Contact: Mike E Gale, BS 210-824-4200 hmrcenterstudy@gmail.com Sub-Investigator: Patricia L Keith, BBA Sub-Investigator: Samuel C Keith, BBA Sub-Investigator: Joel A Michalek, PhD Sub-Investigator: Harry A Croft, MD Collapse << |
| NCT01218204 | Dyslipidaemias | Phase 2 | Completed | - | - |
| NCT01218204 | - | - | Completed | - | - |
| NCT00687076 | Peripheral Arterial Disease | Phase 4 | Completed | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00559962 | - | - | Completed | - | - |
| NCT00449410 | Atrial Fibrillation ... more >> Neuropsychology Magnetic Resonance Imaging Inflammation Hemostasis Collapse << | Phase 4 | Completed | - | Netherlands ... more >> Gheorghe AM Pop Nijmegen, Netherlands, 6500 HB Collapse << |
| NCT03110432 | - | - | Recruiting | February 16, 2021 | Germany ... more >> Medizinische Klinik und Poliklinik III Recruiting Dresden, Germany Contact: Andreas Birkenfeld, MD Collapse << |
| NCT01890967 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| NCT00687076 | - | - | Completed | - | - |
| NCT00442897 | Hypercholesterolemia | Phase 4 | Completed | - | - |
| NCT01890967 | - | - | Completed | - | - |
| NCT02260648 | Hypercholesterolemia | Phase 3 | Terminated(Study termination d... more >>ue to insufficient efficacy.) Collapse << | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 650-0021 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ibaragi, Japan, 311-3516 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ibaraki, Japan, 311-4153 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 6150035 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 530-0001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 350-1305 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shizuoka, Japan, 424-0855 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 103-0028 Collapse << |
| NCT01587235 | Dyslipidemia | Phase 4 | Withdrawn | May 2014 | - |
| NCT01043380 | Hypercholesterolemia ... more >> Coronary Artery Disease Collapse << | Phase 4 | Completed | - | Japan ... more >> Kumamoto University Kumamoto, Japan, 8608556 Collapse << |
| NCT03204799 | - | - | Recruiting | December 16, 2018 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT00461630 | Cardiovascular Disease ... more >> Peripheral Arterial Disease Diabetes Mellitus Coronary Heart Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Clinical Trial Service Unit, University of Oxford Oxford, United Kingdom, OX3 7LF Collapse << |
| NCT02260648 | - | - | Terminated(Study termination d... more >>ue to insufficient efficacy.) Collapse << | - | - |
| NCT00461630 | - | - | Completed | - | - |
| NCT00433823 | Coronary Heart Disease ... more >> Diabetes Mellitus Non-Coronary Atherosclerotic Disease Hypercholesterolemia Collapse << | Phase 4 | Unknown | - | Turkey ... more >> Bolu Izzet Baysal School of Medicine Recruiting Bolu, Turkey, 14280 Contact: Mustafa Kanat, MD +905385448782 mustafa.kanat@gmail.com Collapse << |
| NCT00462748 | - | - | Completed | - | - |
| NCT02460159 | Hypercholesterolemia ... more >> Heterozygous Familial Hypercholesterolemia Collapse << | Phase 3 | Completed | - | - |
| NCT00408057 | - | - | Completed | - | Canada, Ontario ... more >> St. MIchael's Hospital Toronto, Ontario, Canada, m5g 2p9 Collapse << |
| NCT00442897 | - | - | Completed | - | - |
| NCT03532854 | Healthy Male Volunteers | Phase 1 | Not yet recruiting | September 18, 2018 | Korea, Republic of ... more >> LG chem Not yet recruiting Seoul, Gangseo-Gu, Korea, Republic of, 07795 Contact: Subin Lee 02-6987-4153 leesubin@lgchem.com Collapse << |
| NCT00462748 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00738296 | Cardiovascular Diseases | Phase 4 | Completed | - | - |
| NCT02460159 | - | - | Completed | - | - |
| NCT00110435 | Type 2 Diabetes Mellitus ... more >> Hypercholesterolemia Collapse << | Phase 3 | Completed | - | - |
| NCT03355027 | Atherosclerosis ... more >> Cardiovascular Diseases Collapse << | Not Applicable | Recruiting | May 8, 2019 | United Kingdom ... more >> Addenbrooke's Hospital Recruiting Cambridge, Cambridgeshire, United Kingdom, CB2 0QQ Contact: Paul Cacciottolo 01223256653 pjc99@medschl.cam.ac.uk Principal Investigator: Joseph Cheriyan, MBChB, MA, FRCP Collapse << |
| NCT00496730 | - | - | Completed | - | - |
| NCT02550288 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01763866 | Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT02550288 | - | - | Completed | - | - |
| NCT00651560 | Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT00090298 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT03750760 | Myocardial Infarction ... more >> Dyslipidemias Collapse << | Phase 4 | Not yet recruiting | July 2019 | United Kingdom ... more >> Royal Devon & Exeter Hospital Not yet recruiting Exeter, Devon, United Kingdom, EX2 5DW Contact: Andrew Sharp, MBChB, MRCP, MD The Royal Bournemouth General Hospital Not yet recruiting Bournemouth, Dorset, United Kingdom, BH7 7DW Contact: Terry Levy, BSc, MB ChB, FRCP East Sussex Healthcare NHS Trust Not yet recruiting Saint Leonards-on-Sea, East Sussex, United Kingdom, TN37 7PT Contact: Robert Gerber, MBBS Basildon Hospital Not yet recruiting Basildon, Essex, United Kingdom, SS16 5NL Contact: Rajesh Aggarwal, MB ChB, FRCP (London), MD St Mary's Hospital Not yet recruiting London, Greater London, United Kingdom, W2 1NY Contact: Jamil Mayet, MBChB, MD, FESC, FACC, FRC Queen Alexandra Hospital Not yet recruiting Portsmouth, Hampshire, United Kingdom, PO6 3LY Contact: Huw Griffiths, MB BS (Hons), MD, FRCP Southampton General Hospital Not yet recruiting Southampton, Hampshire, United Kingdom, SO16 6YD Contact: Nick Curzen, BM(Hons), PhD, FRCP Northwick Park Hospital Not yet recruiting Harrow, Middlesex, United Kingdom, HA1 3UJ Contact: Ahmed Elghamaz, MB BCh MRCP Hull Royal Infirmary Not yet recruiting Hull, North Humberside, United Kingdom, HU3 2JZ Contact: Richard Oliver, MB BS, RFCP DM, FESC Queens Medical Centre Not yet recruiting Nottingham, Nottinghamshire, United Kingdom, NG7 2UH Contact: Shahnaz Jamil-Copley, MBChB, MRCP, PhD St Peters Hospital Not yet recruiting Chertsey, Surrey, United Kingdom, KT16 0PZ Contact: Sumeet Sharma, MD, MRCP, CCT East Surrey Hospital Not yet recruiting Redhill, Surrey, United Kingdom, RH1 5RH Contact: Rashid Iqbal, MSc, MRCP, MRCPI Freeman Hospital Not yet recruiting Newcastle Upon Tyne, Tyne And Wear, United Kingdom, NE7 7DN Contact: Vijay Kunadian, MBBS, MD, FRCP, FACC, FESC Queen Elizabeth Medical Centre Not yet recruiting Birmingham, West Midlands, United Kingdom, B15 2TH Contact: John Townend, MBChB, MD, FRCP, FESC City Hospital Not yet recruiting Birmingham, West Midlands, United Kingdom, B18 7QH Contact: Derek Connolly, MBChB, PhD, FRCP Worchestershire Royal Hospital Not yet recruiting Worcester, Worcestershire, United Kingdom, WR5 1DD Contact: Jasper Trevelyan, MB ChB Northern General Hospital Not yet recruiting Sheffield, Yorkshire, United Kingdom, S5 7AU Contact: Robert Storey, BM, DM, FRCP, FESC Collapse << |
| NCT00541697 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00440154 | Hypercholesterolemia | Phase 2 | Completed | - | Chile ... more >> Sanofi-Aventis Administrative Office Santiago, Chile Hungary Sanofi-Aventis Administrative Office Budapest, Hungary Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey Collapse << |
| NCT00496730 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00587678 | Peripheral Artery Disease | Not Applicable | Completed | - | United States, Virginia ... more >> University of Virgina Health System Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00587678 | - | - | Completed | - | - |
| NCT02741245 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01520077 | Alopecia Areata | Phase 1 | Completed | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT02741245 | - | - | Completed | - | - |
| NCT02445352 | Primary Hypercholesterolemia | Phase 3 | Completed | - | Korea, Republic of ... more >> Severance Hospital Seodaemun-gu, Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00092716 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00092586 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT03543774 | Hypercholesterolemia ... more >> Chronic Kidney Diseases Collapse << | Phase 4 | Recruiting | September 15, 2019 | Vietnam ... more >> Hue University of Medicine and Pharmacy Recruiting Hue, Thua Thien Hue, Vietnam, 0234 Contact: Nguyen Thanh Thao 84.234.3822873 hcmp@huemed-univ.edu.vn Contact: Nguyen Thanh Thao 84.234.3822873 thaonrad@gmail.com Principal Investigator: Duong Thi Ngoc Lan, Master Sub-Investigator: Le Van An, A/Professor Collapse << |
| NCT01763866 | - | - | Completed | - | - |
| NCT00413972 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00413972 | - | - | Completed | - | - |
| NCT02227784 | Hyperlipidemia | Phase 3 | Terminated(Study termination d... more >>ue to program termination.) Collapse << | - | - |
| NCT00157911 | Hypercholerolemia | Phase 3 | Completed | - | - |
| NCT00092898 | Lipid Metabolism, Inborn Error... more >>s Heart Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02451098 | Hyperlipidemia | Phase 3 | Completed | - | Korea, Republic of ... more >> The catholic university of korea seoul st. Mary's hospital Seoul, Korea, Republic of Collapse << |
| NCT00092664 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00092599 | Hypercholesterolemia ... more >> Coronary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01077830 | - | - | Completed | - | - |
| NCT00092612 | Hypercholesterolemia ... more >> Coronary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00092625 | Hypercholesterolemia ... more >> Coronary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00092638 | Hypercholesterolemia ... more >> Coronary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02227784 | - | - | Terminated(Study termination d... more >>ue to program termination.) Collapse << | - | - |
| NCT00093899 | Hyperlipidemia ... more >> Hypercholesterolemia Hypertriglyceridemia Collapse << | Phase 3 | Completed | - | - |
| NCT00566267 | Hyperlipidemia | Phase 2 Phase 3 | Completed | - | - |
| NCT00762164 | Hypercholesterolemia | Phase 4 | Completed | - | United States, New York ... more >> Bronx VA Medical Center Bronx, New York, United States, 10468 Collapse << |
| NCT00762164 | - | - | Completed | - | - |
| NCT01414192 | - | - | Completed | - | - |
| NCT01414192 | - | - | Completed | - | - |
| NCT01857843 | Acute Coronary Syndrome | Phase 4 | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT01730040 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00092677 | Aortic Stenosis | Phase 3 | Completed | - | - |
| NCT01730040 | - | - | Completed | - | - |
| NCT02547402 | Acute Kidney Injury | Phase 1 | Completed | - | United States, Michigan ... more >> Jasper Clinical Research & Development, Inc. Kalamazoo, Michigan, United States, 49007 Collapse << |
| NCT01154036 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT03768427 | Hypercholesterolemia | Phase 3 | Not yet recruiting | January 29, 2021 | - |
| NCT00092677 | - | - | Completed | - | - |
| NCT01154036 | - | - | Completed | - | - |
| NCT02730689 | Healthy Volunteers | Phase 1 | Completed | - | - |
| NCT00092560 | Hypercholesterolemia ... more >> Hypertriglyceridemia Collapse << | Phase 3 | Completed | - | - |
| NCT02534376 | Prostate Cancer | Early Phase 1 | Recruiting | February 2020 | United States, California ... more >> Cedars Sinai Medical Center Recruiting Los Angeles, California, United States, 90048 Contact: Jenny Park, MPH 310-423-8762 jenny.park@cshs.org Principal Investigator: Hyung L Kim, MD Collapse << |
| NCT02501200 | Healthy | Phase 1 | Completed | - | - |
| NCT00395603 | Cardiovascular Diseases ... more >> Diabetes Mellitus Collapse << | Phase 3 | Terminated(This study was term... more >>inated early due to poor recruitment.) Collapse << | - | - |
| NCT01077830 | - | - | Completed | - | - |
| NCT02461004 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> The Korea University Anam Hospital Seoul, Korea, Republic of Collapse << |
| NCT02584504 | Hypercholesterolemia | Phase 3 | Completed | - | Japan ... more >> Investigational Site Number 392028 Ageo-Shi, Japan Investigational Site Number 392007 Chuo-Ku, Japan Investigational Site Number 392029 Chuo-Ku, Japan Investigational Site Number 392014 Fukui-Shi, Japan Investigational Site Number 392023 Hachioji-Shi, Japan Investigational Site Number 392013 Itoshima-Shi, Japan Investigational Site Number 392010 Kanazawa-Shi, Japan Investigational Site Number 392024 Kasuga-Shi, Japan Investigational Site Number 392004 Kawanishi-Shi, Japan Investigational Site Number 392015 Kitakyushu-Shi, Japan Investigational Site Number 392005 Komatsu-Shi, Japan Investigational Site Number 392032 Matsudo-Shi, Japan Investigational Site Number 392017 Matsumoto-Shi, Japan Investigational Site Number 392003 Mito-Shi, Japan Investigational Site Number 392018 Morioka-Shi, Japan Investigational Site Number 392009 Moriya-Shi, Japan Investigational Site Number 392006 Nagoya-Shi, Japan Investigational Site Number 392011 Nagoya-Shi, Japan Investigational Site Number 392019 Nagoya-Shi, Japan Investigational Site Number 392025 Nagoya-Shi, Japan Investigational Site Number 392027 Osaka-Shi, Japan Investigational Site Number 392030 Sakura-Shi, Japan Investigational Site Number 392016 Shinagawa-Ku, Japan Investigational Site Number 392001 Shinjuku-Ku, Japan Investigational Site Number 392008 Shinjuku-Ku, Japan Investigational Site Number 392012 Shizuoka-Shi, Japan Investigational Site Number 392002 Suita-Shi, Japan Investigational Site Number 392031 Suita-Shi, Japan Investigational Site Number 392020 Toyonaka-Shi, Japan Investigational Site Number 392022 Yao-Shi, Japan Collapse << |
| NCT01047683 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT01047501 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT02584504 | - | - | Completed | - | - |
| NCT01492361 | Cardiovascular Diseases | Phase 3 | Completed | - | - |
| NCT02378064 | Inflammation Plaque, Atheroscl... more >>erotic Coronary Disease Renin-Angiotensin System Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02023879 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT03430284 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 31, 2020 | China, Shandong ... more >> Shandong Provincial Hospital Not yet recruiting Jinan, Shandong, China, 250021 Contact: Shanshan Shao +8653168776094 shaoshanshan11@126.com Shandong Provincial Hospital Recruiting Jinan, Shandong, China Collapse << |
| NCT03510884 | Hypercholesterolaemia | Phase 3 | Recruiting | March 2022 | - |
| NCT01666067 | Hypercholesterolemia | Phase 4 | Completed | - | Korea, Republic of ... more >> Gil Medical Center Incheon, Korea, Republic of, 405-760 Collapse << |
| NCT02023879 | - | - | Completed | - | - |
| NCT01266876 | - | - | Completed | - | - |
| NCT02984982 | Hypercholesterolemia ... more >> Acute Coronary Syndrome Collapse << | Phase 4 | Completed | - | Japan ... more >> Investigational Site Number 392026 Bunkyoku-ku, Japan Investigational Site Number 392022 Chiyoda-ku, Japan Investigational Site Number 392032 Fujisawa-shi, Japan Investigational Site Number 392004 Fukui-shi, Japan Investigational Site Number 392007 Fukuoka-shi, Japan Investigational Site Number 392048 Fukuoka-shi, Japan Investigational Site Number 392008 Gifu-shi, Japan Investigational Site Number 392039 Hiroshima-shi, Japan Investigational Site Number 392028 Isehara-shi, Japan Investigational Site Number 392036 Itabashi-ku, Japan Investigational Site Number 392037 Itabashi-ku, Japan Investigational Site Number 392024 Izumisano-shi, Japan Investigational Site Number 392013 Izunokuni-shi, Japan Investigational Site Number 392020 Kawaguchi-shi, Japan Investigational Site Number 392041 Kisarazu-shi, Japan Investigational Site Number 392009 Kitakyushu-shi, Japan Investigational Site Number 392034 Kitakyushu-shi, Japan Investigational Site Number 392044 Kochi-shi, Japan Investigational Site Number 392002 Kumamoto-shi, Japan Investigational Site Number 392011 Kumamoto-shi, Japan Investigational Site Number 392003 Kurashiki-shi, Japan Investigational Site Number 392018 Matsuyama-shi, Japan Investigational Site Number 392021 Morioka-shi, Japan Investigational Site Number 392017 Nagakute-shi, Aichi, Japan Investigational Site Number 392047 Nagaoka-shi, Japan Investigational Site Number 392038 Nagoya-shi, Japan Investigational Site Number 392042 Nagoya-shi, Japan Investigational Site Number 392033 Okayama-shi, Japan Investigational Site Number 392006 Osaka-shi, Japan Investigational Site Number 392010 Osaka-shi, Japan Investigational Site Number 392045 Osaka-shi, Japan Investigational Site Number 392046 Osaka-shi, Japan Investigational Site Number 392015 Sagamihara-shi, Japan Investigational Site Number 392019 Sakai-shi, Japan Investigational Site Number 392035 Sapporo-shi, Japan Investigational Site Number 392001 Tenri-shi, Japan Investigational Site Number 392016 Toyoake-shi, Japan Investigational Site Number 392025 Tsukuba-shi, Japan Investigational Site Number 392040 Tsukuba-shi, Japan Investigational Site Number 392005 Wakayama-shi, Japan Investigational Site Number 392027 Yokohama-shi, Japan Investigational Site Number 392043 Yokohama-shi, Japan Collapse << |
| NCT03510715 | Hypercholesterolemia | Phase 3 | Recruiting | June 2020 | United States, California ... more >> Investigational Site Number 8400004 Recruiting Los Angeles, California, United States, 90027 Austria Investigational Site Number 0400001 Recruiting Wien, Austria, 1090 Brazil Investigational Site Number 0760001 Recruiting Sao Paulo, Brazil, 05403-000 Canada Investigational Site Number 1240002 Recruiting Montreal, Canada, H3T 1C5 Investigational Site Number 1240001 Recruiting Quebec, Canada, G1V 4W2 Denmark Investigational Site Number 2080001 Recruiting Viborg, Denmark, 8800 France Investigational Site Number 2500001 Recruiting Bron Cedex, France, 69677 Italy Investigational Site Number 3800001 Recruiting Palermo, Italy, 90127 Mexico Investigational Site Number 4840006 Recruiting Oaxaca, Mexico, 68000 Netherlands Investigational Site Number 5280001 Recruiting Amsterdam, Netherlands, 1105 AZ Norway Investigational Site Number 5780001 Recruiting Oslo, Norway, 0373 Russian Federation Investigational Site Number 6430002 Recruiting Kemerovo, Russian Federation, 650002 Slovenia Investigational Site Number 7050001 Recruiting Ljubljana, Slovenia, 1000 Spain Investigational Site Number 7240001 Recruiting A Coruna, Spain, 15001 Taiwan Investigational Site Number 1580001 Recruiting Taipei, Taiwan, 112 Turkey Investigational Site Number 7920002 Recruiting Ankara, Turkey, 06500 Investigational Site Number 7920001 Recruiting Izmir, Turkey, 35040 Collapse << |
| NCT01266876 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
