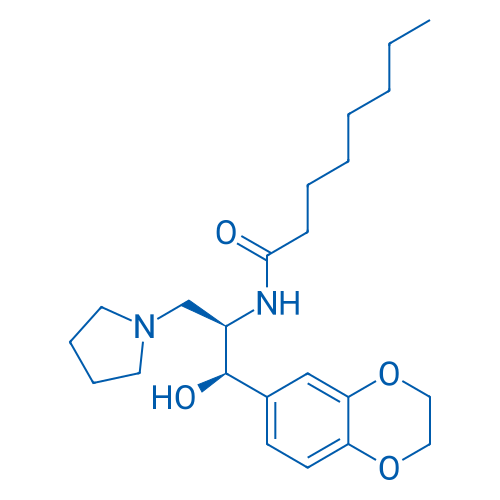
CAS No.: 491833-29-5
Eliglustat/依利格鲁司特 Catalog No. CSN13621
Synonyms: Genz 99067;Genz-112638;UNIIN0493335P3
Eliglustat is a specific and potent inhibitor of glucosylceramide synthase.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01074944 Gaucher Disease Phase 3 Completed - - NCT00358150 Gaucher Disease, Type 1 ... more >> Cerebroside Lipidosis Syndrome Glucocerebrosidase Deficiency Disease Glucosylceramide Beta-Glucosidase Deficiency Disease Gaucher Disease, Non-Neuronopathic Form Collapse << Phase 2 Completed - United States, New York ... more >> New York University New York, New York, United States New York, New York, United States Argentina Aprillus Asistencia e Investigación Buenos Aires, Argentina Hospital de Oncologia Maria Curie Buenos Aires, Argentina IMAI Buenos Aires, Argentina Instituto Argentino de Diagnostico y Tratamiento (IADT) Buenos Aires, Argentina Buenos Aires, Argentina Hospital Ramos Mejia Ciudad Autonoma de Buenos Aires, Argentina Israel Rambam Medical Center Haifa, Israel Haifa, Israel Sha'are Zedek Medical Centre Jerusalem, Israel Jerusalem, Israel Italy Universita degli Studi di Milano Milano, Italy Mexico Instituto Mexicano del Seguro Social D.f., Mexico Mexico City, Mexico Russian Federation Hematology Research Center of Ministry of Healthcare of the Russian Federation Moscow, Russian Federation Moscow, Russian Federation Collapse << NCT00943111 - - Completed - - - 更多
- 参考文献
- [1] Shayman JA. ELIGLUSTAT TARTRATE: Glucosylceramide Synthase Inhibitor Treatment of Type 1 Gaucher Disease. Drugs Future. 2010 Aug 1;35(8):613-620.
- [2] Yew NS, Zhao H, et al. Increased hepatic insulin action in diet-induced obese mice following inhibition of glucosylceramide synthase. PLoS One. 2010 Jun 21;5(6):e11239.
- [3] Almeida-Calpe A, López de Frutos L, Medrano-Engay B, García-García CB, Ribate MP, Giraldo P. Metabolizing profile of the cytochrome pathway CYP2D6, CYP3A4 and the ABCB 1 transporter in Spanish patients affected by Gaucher disease. Chem Biol Interact. 2021 Aug 25;345:109527
- [4] LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012–. Eliglustat. 2018 Mar 5
- [5] Balwani M, Burrow TA, Charrow J, Goker-Alpan O, Kaplan P, Kishnani PS, Mistry P, Ruskin J, Weinreb N. Recommendations for the use of eliglustat in the treatment of adults with Gaucher disease type 1 in the United States. Mol Genet Metab. 2016 Feb;117(2):95-103
- [6] Dagher R, Watzinger M, Chevalier G, Thirion-Delalande C, Gervais F, Forster R. Carcinogenicity testing of eliglustat in mice and rats. Regul Toxicol Pharmacol. 2015 Oct;73(1):401-12
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 491833-29-5 | 储存条件 |
|
|||||
| 分子式 | C23H36N2O4 | 运输 | 蓝冰 | |||||
| 分子量 | 404.54 | 别名 | Genz 99067;Genz-112638;UNIIN0493335P3;eliglustat hemitartrate;trade name Cerdelga;依利格鲁司特 | |||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
