| 647-V cell | - | Growth inhibition assay | - | Inhibition of human 647-V cell growth in a cell viability assay, IC50=6.9 nM | SANGER |
| 697 cell | - | Growth inhibition assay | - | Inhibition of human 697 cell growth in a cell viability assay, IC50=6.13 nM | SANGER |
| A2780 cell | - | Growth inhibition assay | - | Inhibition of human A2780 cell growth in a cell viability assay, IC50=0.594 nM | SANGER |
| A375 cell | - | Growth inhibition assay | - | Inhibition of human A375 cell growth in a cell viability assay, IC50=4.14 nM | SANGER |
| A427 cell | - | Growth inhibition assay | - | Inhibition of human A427 cell growth in a cell viability assay, IC50=6.07 nM | SANGER |
| A549 cell | - | Growth inhibition assay | - | Inhibition of human A549 cell growth in a cell viability assay, IC50=19.6 nM | SANGER |
| A673 cell | - | Growth inhibition assay | - | Inhibition of human A673 cell growth in a cell viability assay, IC50=11.6 nM | SANGER |
| A704 cell | - | Growth inhibition assay | - | Inhibition of human A704 cell growth in a cell viability assay, IC50=27.45 nM | SANGER |
| ABC-1 cell | - | Growth inhibition assay | - | Inhibition of human ABC-1 cell growth in a cell viability assay, IC50=0.365 nM | SANGER |
| AGS cell | - | Growth inhibition assay | - | Inhibition of human AGS cell growth in a cell viability assay, IC50=2.86 nM | SANGER |
| BCPAP cell | - | Growth inhibition assay | - | Inhibition of human BCPAP cell growth in a cell viability assay, IC50=1.34 nM | SANGER |
| BHT-101 cell | - | Growth inhibition assay | - | Inhibition of human BHT-101 cell growth in a cell viability assay, IC50=3.72 nM | SANGER |
| BV-173 cell | - | Growth inhibition assay | - | Inhibition of human BV-173 cell growth in a cell viability assay, IC50=5.39 nM | SANGER |
| C-33-A cell | - | Growth inhibition assay | - | Inhibition of human C-33-A cell growth in a cell viability assay, IC50=7.58 nM | SANGER |
| Ca9-22 cell | - | Growth inhibition assay | - | Inhibition of human Ca9-22 cell growth in a cell viability assay, IC50=4.25 nM | SANGER |
| CAL-51 cell | - | Growth inhibition assay | - | Inhibition of human CAL-51 cell growth in a cell viability assay, IC50=0.126 nM | SANGER |
| CAL-72 cell | - | Growth inhibition assay | - | Inhibition of human CAL-72 cell growth in a cell viability assay, IC50=3.02 nM | SANGER |
| CHL-1 cell | - | Growth inhibition assay | - | Inhibition of human CHL-1 cell growth in a cell viability assay, IC50=2.81 nM | SANGER |
| CHP-212 cell | - | Growth inhibition assay | - | Inhibition of human CHP-212 cell growth in a cell viability assay, IC50=7.34 nM | SANGER |
| COLO-684 cell | - | Growth inhibition assay | - | Inhibition of human COLO-684 cell growth in a cell viability assay, IC50=9.21 nM | SANGER |
| COR-L105 cell | - | Growth inhibition assay | - | Inhibition of human COR-L105 cell growth in a cell viability assay, IC50=6.76 nM | SANGER |
| DMS-273 cell | - | Growth inhibition assay | - | Inhibition of human DMS-273 cell growth in a cell viability assay, IC50=7.06 nM | SANGER |
| ES1 cell | - | Growth inhibition assay | - | Inhibition of human ES1 cell growth in a cell viability assay, IC50=0.749 nM | SANGER |
| ES3 cell | - | Growth inhibition assay | - | Inhibition of human ES3 cell growth in a cell viability assay, IC50=5.79 nM | SANGER |
| ES4 cell | - | Growth inhibition assay | - | Inhibition of human ES4 cell growth in a cell viability assay, IC50=4.39 nM | SANGER |
| ES7 cell | - | Growth inhibition assay | - | Inhibition of human ES7 cell growth in a cell viability assay, IC50=3.59 nM | SANGER |
| ES8 cell | - | Growth inhibition assay | - | Inhibition of human ES8 cell growth in a cell viability assay, IC50=1.32 nM | SANGER |
| EW-1 cell | - | Growth inhibition assay | - | Inhibition of human EW-1 cell growth in a cell viability assay, IC50=5.24 nM | SANGER |
| EW-11 cell | - | Growth inhibition assay | - | Inhibition of human EW-11 cell growth in a cell viability assay, IC50=4.9 nM | SANGER |
| EW-16 cell | - | Growth inhibition assay | - | Inhibition of human EW-16 cell growth in a cell viability assay, IC50=1.61 nM | SANGER |
| EW-24 cell | - | Growth inhibition assay | - | Inhibition of human EW-24 cell growth in a cell viability assay, IC50=3.83 nM | SANGER |
| F-36P cells | - | Proliferation assay | - | Antiproliferative activity against human F-36P cells, IC50=0.28 μM | 21341675 |
| FADU cell | - | Growth inhibition assay | - | Inhibition of human FADU cell growth in a cell viability assay, IC50=5.66 nM | SANGER |
| G-401 cell | - | Growth inhibition assay | - | Inhibition of human G-401 cell growth in a cell viability assay, IC50=5.43 nM | SANGER |
| GAMG cell | - | Growth inhibition assay | - | Inhibition of human GAMG cell growth in a cell viability assay, IC50=2.51 nM | SANGER |
| GB-1 cell | - | Growth inhibition assay | - | Inhibition of human GB-1 cell growth in a cell viability assay, IC50=6.71 nM | SANGER |
| HCE-T cell | - | Growth inhibition assay | - | Inhibition of human HCE-T cell growth in a cell viability assay, IC50=4.45 nM | SANGER |
| HD-MY-Z cell | - | Growth inhibition assay | - | Inhibition of human HD-MY-Z cell growth in a cell viability assay, IC50=5.91 nM | SANGER |
| HEL cell | - | Growth inhibition assay | - | Inhibition of human HEL cell growth in a cell viability assay, IC50=7.08 nM | SANGER |
| H-EMC-SS cell | - | Growth inhibition assay | - | Inhibition of human H-EMC-SS cell growth in a cell viability assay, IC50=3.68 nM | SANGER |
| HOS cell | - | Growth inhibition assay | - | Inhibition of human HOS cell growth in a cell viability assay, IC50=2.53 nM | SANGER |
| HSC-3 cell | - | Growth inhibition assay | - | Inhibition of human HSC-3 cell growth in a cell viability assay, IC50=3.02 nM | SANGER |
| HSC-4 cell | - | Growth inhibition assay | - | Inhibition of human HSC-4 cell growth in a cell viability assay, IC50=4.8 nM | SANGER |
| HT-1080 cell | - | Growth inhibition assay | - | Inhibition of human HT-1080 cell growth in a cell viability assay, IC50=4.99 nM | SANGER |
| HuCCT1 cell | - | Growth inhibition assay | - | Inhibition of human HuCCT1 cell growth in a cell viability assay, IC50=5.52 nM | SANGER |
| human H4 cell | - | Growth inhibition assay | - | Inhibition of human H4 cell growth in a cell viability assay, IC50=7.25 nM | SANGER |
| human JAR cell | - | Growth inhibition assay | - | Inhibition of human JAR cell growth in a cell viability assay, IC50=5.97 nM | SANGER |
| human K5 cell | - | Growth inhibition assay | - | Inhibition of human K5 cell growth in a cell viability assay, IC50=7 nM | SANGER |
| human KU-19-19 cell | - | Growth inhibition assay | - | Inhibition of human KU-19-19 cell growth in a cell viability assay, IC50=7.09 nM | SANGER |
| human NB14 cell | - | Growth inhibition assay | - | Inhibition of human NB14 cell growth in a cell viability assay, IC50=6.64 nM | SANGER |
| human SW982 cell | - | Growth inhibition assay | - | Inhibition of human SW982 cell growth in a cell viability assay, IC50=7.44 nM | SANGER |
| human T47D cell | - | Growth inhibition assay | - | Inhibition of human T47D cell growth in a cell viability assay, IC50=16.79 nM | SANGER |
| HUTU-80 cell | - | Growth inhibition assay | - | Inhibition of human HUTU-80 cell growth in a cell viability assay, IC50=0.791 nM | SANGER |
| K562 cells | - | Proliferation assay | - | Antiproliferative activity against human K562 cells, IC50=18 μM | 21341675 |
| KOSC-2 cell | - | Growth inhibition assay | - | Inhibition of human KOSC-2 cell growth in a cell viability assay, IC50=5.45 nM | SANGER |
| KP-4 cell | - | Growth inhibition assay | - | Inhibition of human KP-4 cell growth in a cell viability assay, IC50=2.17 nM | SANGER |
| MCF7 cell | - | Growth inhibition assay | - | Inhibition of human MCF7 cell growth in a cell viability assay, IC50=31.36 nM | SANGER |
| MC-IXC cell | - | Growth inhibition assay | - | Inhibition of human MC-IXC cell growth in a cell viability assay, IC50=7.17 nM | SANGER |
| MDA-MB-157 cell | - | Growth inhibition assay | - | Inhibition of human MDA-MB-157 cell growth in a cell viability assay, IC50=2.92 nM | SANGER |
| MES-SA/Dx5 cells | - | Cytotoxicity assay | - | Cytotoxicity against human MES-SA/Dx5 cells, IC50=5 nM | 23937981 |
| MHH-ES-1 cell | - | Growth inhibition assay | - | Inhibition of human MHH-ES-1 cell growth in a cell viability assay, IC50=5.69 nM | SANGER |
| MOLT-16 cell | - | Growth inhibition assay | - | Inhibition of human MOLT-16 cell growth in a cell viability assay, IC50=4.53 nM | SANGER |
| NB69 cell | - | Growth inhibition assay | - | Inhibition of human NB69 cell growth in a cell viability assay, IC50=1.22 nM | SANGER |
| NBsusSR cell | - | Growth inhibition assay | - | Inhibition of human NBsusSR cell growth in a cell viability assay, IC50=3.13 nM | SANGER |
| NCI-H1563 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H1563 cell growth in a cell viability assay, IC50=4.13 nM | SANGER |
| NCI-H1703 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H1703 cell growth in a cell viability assay, IC50=3.68 nM | SANGER |
| NCI-H292 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H292 cell growth in a cell viability assay, IC50=0.886 nM | SANGER |
| NCI-SNU-1 cell | - | Growth inhibition assay | - | Inhibition of human NCI-SNU-1 cell growth in a cell viability assay, IC50=4.86 nM | SANGER |
| NOS-1 cell | - | Growth inhibition assay | - | Inhibition of human NOS-1 cell growth in a cell viability assay, 3.66e-05 μM | SANGER |
| NUGC-3 cell | - | Growth inhibition assay | - | Inhibition of human NUGC-3 cell growth in a cell viability assay, IC50=7.33 nM | SANGER |
| OCI-AML2 cells | - | Proliferation assay | - | Antiproliferative activity against human OCI-AML2 cells, IC50=0.35 μM | 21341675 |
| OVCAR-3 cell | - | Growth inhibition assay | - | Inhibition of human OVCAR-3 cell growth in a cell viability assay, IC50=6.2 nM | SANGER |
| OVCAR-4 cell | - | Growth inhibition assay | - | Inhibition of human OVCAR-4 cell growth in a cell viability assay, IC50=5.87 nM | SANGER |
| OVCAR-5 cell | - | Growth inhibition assay | - | Inhibition of human OVCAR-5 cell growth in a cell viability assay, IC50=1.83 nM | SANGER |
| OVCAR-8 cell | - | Growth inhibition assay | - | Inhibition of human OVCAR-8 cell growth in a cell viability assay, IC50=3.99 nM | SANGER |
| PC-14 cell | - | Growth inhibition assay | - | Inhibition of human PC-14 cell growth in a cell viability assay, IC50=24.73 nM | SANGER |
| PC-3 cell | - | Growth inhibition assay | - | Inhibition of human PC-3 cell growth in a cell viability assay, IC50=7.89 nM | SANGER |
| PC3 cells | - | Function assay | - | Inhibition of PDK1-mediated AKT phosphorylation at Thr308 residue in human PC3 cells by ELISA, IC50=0.113 μM | 21341675 |
| PSN1 cell | - | Growth inhibition assay | - | Inhibition of human PSN1 cell growth in a cell viability assay, IC50=3.84 nM | SANGER |
| SF126 cell | - | Growth inhibition assay | - | Inhibition of human SF126 cell growth in a cell viability assay, IC50=1.95 nM | SANGER |
| SK-OV-3 cell | - | Growth inhibition assay | - | Inhibition of human SK-OV-3 cell growth in a cell viability assay, IC50=3.75 nM | SANGER |
| SNU-387 cell | - | Growth inhibition assay | - | Inhibition of human SNU-387 cell growth in a cell viability assay, IC50=9.49 nM | SANGER |
| SW1710 cell | - | Growth inhibition assay | - | Inhibition of human SW1710 cell growth in a cell viability assay, IC50=5.16 nM | SANGER |
| SW1990 cell | - | Growth inhibition assay | - | Inhibition of human SW1990 cell growth in a cell viability assay, IC50=4.32 nM | SANGER |
| SW756 cell | - | Growth inhibition assay | - | Inhibition of human SW756 cell growth in a cell viability assay, IC50=4.13 nM | SANGER |
| T-24 cell | - | Growth inhibition assay | - | Inhibition of human T-24 cell growth in a cell viability assay, IC50=1.55 nM | SANGER |
| TE-8 cell | - | Growth inhibition assay | - | Inhibition of human TE-8 cell growth in a cell viability assay, IC50=5.41 nM | SANGER |
| U251 cell | - | Growth inhibition assay | - | Inhibition of human U251 cell growth in a cell viability assay, IC50=9.86 nM | SANGER |
| UM-UC-3 cell | - | Growth inhibition assay | - | Inhibition of human UM-UC-3 cell growth in a cell viability assay, IC50=7.06 nM | SANGER |
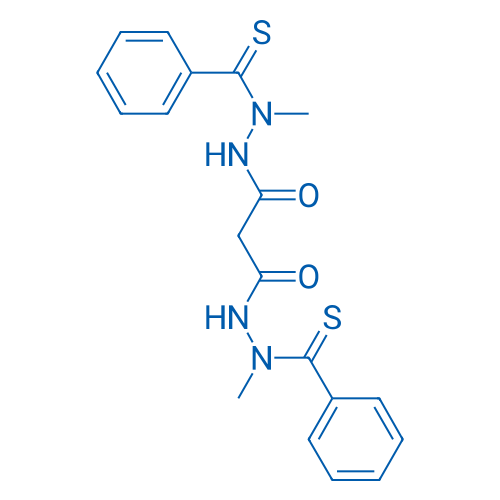
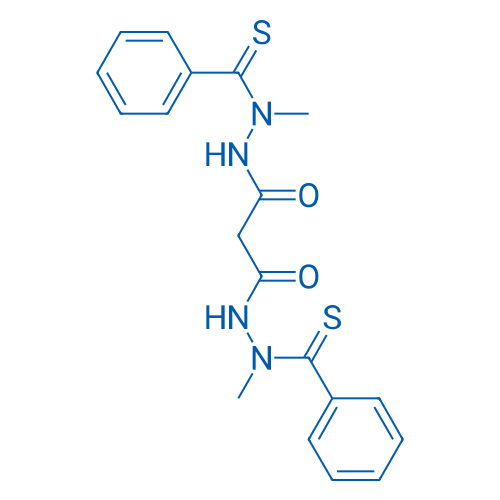

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
