CAS No.: 796073-54-6
DM3 Catalog No. CSN20041
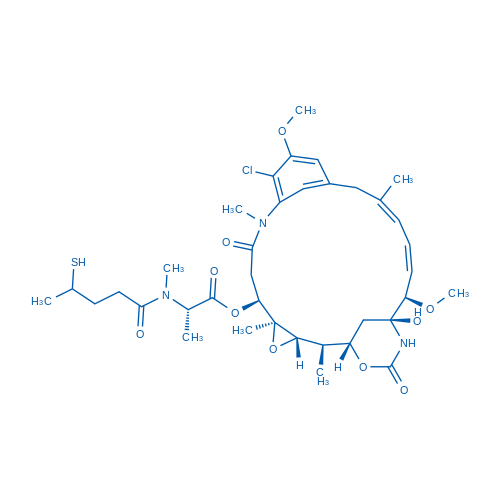
Synonyms: Maytansinoid DM3
DM3 is a tubulin inhibitor, inhibits the assemblyof microtubules by binding to tubulin, with a linker structure can create an antibody-drug conjugate (ADC).
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03577483 Parkinson Disease ... more >> MSA - Multiple System Atrophy Collapse << Not Applicable Recruiting June 1, 2019 France ... more >> Centre Hospitalier Universitaire de Bordeaux Recruiting Bordeaux, France, 33000 Contact: Wassilios MEISSNER, MD, PhD +335 57 82 12 53 wassilios.meissner@chu-bordeaux.fr Contact: Sandrine VILLARS +335 57 82 12 53 sandrine.villars@chu-bordeaux.fr Centre Hospitalier Universitaire de Toulouse Not yet recruiting Toulouse, France, 31000 Contact: Anne PAVY-LE-TRAON, MD,PhD +335 61 77 95 19 pavy-letraon.a@chu-toulouse.fr Collapse << NCT00593957 Rett Syndrome Phase 2 Terminated(Study changed to a ... more >>placebo controlled trial of dextromethorphan) Collapse << - United States, Maryland ... more >> Kennedy Krieger Institute/Johns Hopkins Medical Institutions Baltimore, Maryland, United States, 21205 Collapse << NCT02835053 - - Completed - Canada, Ontario ... more >> Ottawa Hospital Research Institute Ottawa, Ontario, Canada, K1Y 4E9 Collapse << - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 796073-54-6 | 储存条件 |
|
|
| 分子式 | C37H52ClN3O10S | 运输 | 蓝冰 | |
| 分子量 | 766.34 | 别名 | Maytansinoid DM3 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
