CAS No.: 54024-22-5
Desogestrel/去氧孕烯 Catalog No. CSN10779
Synonyms: Org-2969;DESOGESTREL;Cerazette
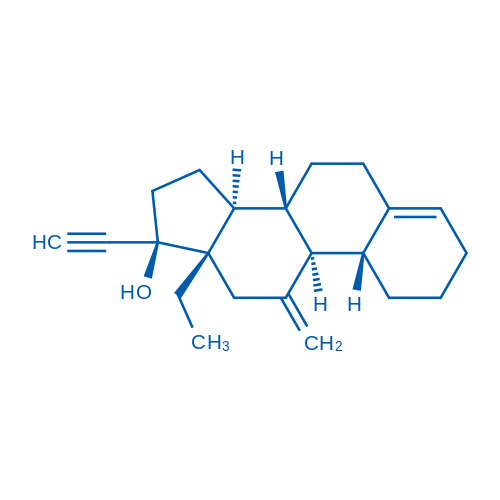
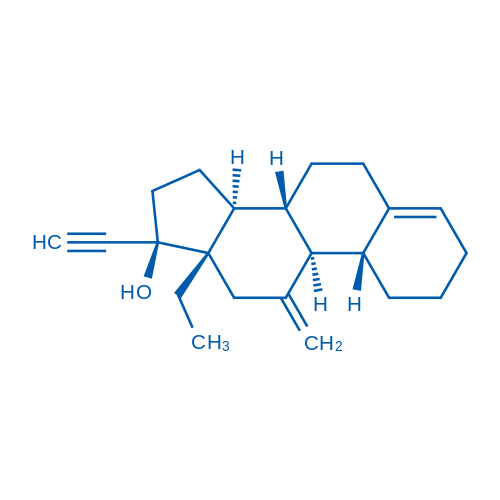
Desogestrel is a synthetic progestational hormone with low androgenic potency that is used as contraceptives.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00269620 Contraception Phase 4 Completed - United States, California ... more >> UCLA-Harbor Los Angeles, California, United States United States, Illinois University of Chicago Chicago, Illinois, United States United States, Maryland Johns Hopkins Baltimore, Maryland, United States United States, Massachusetts Boston University Boston, Massachusetts, United States United States, New York Columbia University New York, New York, United States United States, Oregon Oregon Health Science University Portland, Oregon, United States United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States Magee-Womens Hospital Pittsburgh, Pennsylvania, United States, 15213 United States, Virginia Eastern Virginia Medical School Norfolk, Virginia, United States United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States Collapse << NCT00042289 HIV Infections Phase 4 Recruiting September 30, 2019 - NCT01903031 HIV-1 Infection Phase 2 Completed - - - 更多
- 参考文献
- [1] Gassen DT, Herter LD, et al. Evaluation of hormonal suppression with two contraceptive regimens using ethinyl estradiol and desogestrel. Arch Gynecol Obstet. 2013 Feb;287(2):289-94.
- [2] Verhoeven CH, Krebbers SF, et al. In vitro and in vivo metabolism of desogestrel in several species. Drug Metab Dispos. 1998 Sep;26(9):927-36.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 54024-22-5 | 储存条件 |
|
|||||||
| 分子式 | C22H30O | 运输 | 蓝冰 | |||||||
| 分子量 | 310.47 | 别名 | Org-2969;DESOGESTREL;Cerazette;Desogen;去氧孕烯 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00269620 | Contraception | Phase 4 | Completed | - | United States, California ... more >> UCLA-Harbor Los Angeles, California, United States United States, Illinois University of Chicago Chicago, Illinois, United States United States, Maryland Johns Hopkins Baltimore, Maryland, United States United States, Massachusetts Boston University Boston, Massachusetts, United States United States, New York Columbia University New York, New York, United States United States, Oregon Oregon Health Science University Portland, Oregon, United States United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States Magee-Womens Hospital Pittsburgh, Pennsylvania, United States, 15213 United States, Virginia Eastern Virginia Medical School Norfolk, Virginia, United States United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States Collapse << |
| NCT00042289 | HIV Infections | Phase 4 | Recruiting | September 30, 2019 | - |
| NCT01903031 | HIV-1 Infection | Phase 2 | Completed | - | - |
| NCT00475553 | Breakthrough Bleeding ... more >> Breakthrough Spotting Collapse << | Not Applicable | Completed | - | United States, Texas ... more >> Scott & White Hospital and Clinic Temple, Texas, United States, 76508 Collapse << |
| NCT00403793 | Contraception | Phase 2 | Completed | - | - |
| NCT01388582 | Underweight | Phase 4 | Completed | - | Brazil ... more >> University of Campinas Campinas, SP, Brazil, 13083-888 Collapse << |
| NCT01903031 | - | - | Completed | - | - |
| NCT01044056 | Contraception | Phase 4 | Completed | - | - |
| NCT01044056 | - | - | Completed | - | - |
| NCT03660046 | Hormonal Contraception | Phase 4 | Recruiting | September 2023 | United States, Georgia ... more >> Grady Health System Recruiting Atlanta, Georgia, United States, 30303 Contact: Lisa Haddad, MD, MS, MPH 404-778-1385 lisa.haddad@emory.edu Principal Investigator: Lisa Haddad, MD, MS, MPH The Emory Clinic, Bldg A., 2nd Floor, 1365 Clifton Road, NE Recruiting Atlanta, Georgia, United States, 30322 Contact: Lisa Haddad, MD, MS,MPH 404-778-1385 lisa.haddad@emory.edu Principal Investigator: Lisa Haddad, MD, MS,MPH Atlanta Women's Center Recruiting Atlanta, Georgia, United States, 30342 Contact: Lisa Haddad, MD, MS, MPH 404-778-1385 lisa.haddad@emory.edu Principal Investigator: Lisa Haddad, MD, MS, MPH Collapse << |
| NCT02668783 | Moderate to Severe Primary Dys... more >>menorrhea Collapse << | Phase 3 | Terminated(Study terminated by... more >> Sponsor as a result of a business decision to discontinue the development program for MK-8342B for reasons unrelated to safety or efficacy.) Collapse << | - | - |
| NCT02616146 | Contraception | Phase 3 | Terminated | - | - |
| NCT02668783 | - | - | Terminated(Study terminated by... more >> Sponsor as a result of a business decision to discontinue the development program for MK-8342B for reasons unrelated to safety or efficacy.) Collapse << | - | - |
| NCT00635570 | Birth Control Compliance | Phase 4 | Completed | - | United States, Illinois ... more >> University of Chicago, Section of Family Planning Chicago, Illinois, United States, 60637 University of Chicago Chicago, Illinois, United States, 60637 University of Illinois Chicago, Illinois, United States Collapse << |
| NCT02038335 | - | - | Active, not recruiting | April 2019 | Zimbabwe ... more >> UZ UCSF Harare, Zimbabwe Collapse << |
| NCT01448291 | Vaginosis, Bacterial | Phase 4 | Unknown | March 2013 | United States, Virginia ... more >> Clinical Research Center at Eastern Virginia Medical School Recruiting Norfolk, Virginia, United States, 23507 Contact: Julia Caul 757-446-5808 Collapse << |
| NCT02082652 | HIV Contracep... more >>tion Collapse << | Phase 4 | Completed | - | Uganda ... more >> Infectious Disease Institute Kampala, Uganda Collapse << |
| NCT01873170 | - | - | Active, not recruiting | June 2019 | United States, Pennsylvania ... more >> Center for Family Planning Research Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01670656 | Dysmenorrhea | Phase 2 | Completed | - | - |
| NCT01670656 | - | - | Completed | - | - |
| NCT01298128 | Infertility I... more >>n-vitro Fertilization Collapse << | Not Applicable | Terminated | - | - |
| NCT03120728 | Emergency Contraception ... more >> Healthy, Reproductive Age Women Collapse << | Phase 4 | Recruiting | July 1, 2018 | United States, California ... more >> Keck Medicine of USC--Downtown LA Recruiting Los Angeles, California, United States, 90017 Contact: Blanca Ovalle 323-409-3104 obgynresearch@med.usc.edu Principal Investigator: Diana Crabtree Sokol, MD LAC+USC Medical Center Recruiting Los Angeles, California, United States, 90033 Contact: Blanca Ovalle 323-409-3104 obgynresearch@med.usc.edu Principal Investigator: Diana Crabtree Sokol, MD Collapse << |
| NCT00565240 | Ovarian Follicle | Not Applicable | Completed | - | Canada, Saskatchewan ... more >> Royal University Hospital Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT00635570 | - | - | Completed | - | - |
| NCT02852265 | - | - | Completed | - | - |
| NCT03197077 | Infertility, Female ... more >> Polycystic Ovary Syndrome Collapse << | Phase 3 | Recruiting | July 1, 2019 | Belgium ... more >> UZ Brussel Recruiting Brussels, Belgium, 1090 Contact: Elsie Nulens +3224776648 elsie.nulens@uzbrussel.be Collapse << |
| NCT03307863 | Contraception ... more >> Drug Interactions Collapse << | Phase 4 | Recruiting | July 2019 | Brazil ... more >> Hospital das Clínicas de Ribeirão Preto da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo Recruiting Ribeirão Preto, Sao Paulo, Brazil, 14049-900 Contact: Carolina S Vieira, MD +551636022818 carol.sales@usp.br Contact: Leticia S Ferreira, MD +553491924258 lelezinhasanchez1@yahoo.com.br Principal Investigator: Carolina S Vieira, MD Collapse << |
| NCT00710606 | Pharmacokinetics | Not Applicable | Completed | - | United States, New York ... more >> Division of Family Planning and Preventive Services New York, New York, United States, 10032 Collapse << |
| NCT01298128 | - | - | Terminated | - | - |
| NCT01490190 | Contraception | Phase 4 | Completed | - | - |
| NCT02852265 | Contraception | Phase 4 | Completed | - | United States, California ... more >> University of California, Davis Sacramento, California, United States, 95817 Collapse << |
| NCT00828542 | Breastfeeding ... more >> Contraception Collapse << | Not Applicable | Completed | - | Brazil ... more >> University of Sao Paulo Ribeirao Preto, Sao Paulo, Brazil, 14049-900 Collapse << |
| NCT01490190 | - | - | Completed | - | - |
| NCT03336346 | - | - | Recruiting | December 2018 | Botswana ... more >> Botswana-UPenn Partnership Recruiting Gaborone, Botswana Contact: Chelsea Morroni, MD 26776524112 chelseaamorroni@gmail.com Principal Investigator: Chelsea Morroni, MD Collapse << |
| NCT01356927 | - | - | Completed | - | United States, Massachusetts ... more >> Boston University Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT00710606 | - | - | Completed | - | - |
| NCT02367833 | Bone; Disorder, Development an... more >>d Growth Collapse << | Phase 4 | Active, not recruiting | December 2018 | United States, Pennsylvania ... more >> Women's Health and Exercise Laboratories, The Pennsylvania State University University Park, Pennsylvania, United States, 16802 Collapse << |
| NCT01920022 | Early Pregnancy Termination ... more >> Medical Abortion Postabortion Contraception Collapse << | Phase 4 | Completed | - | Sweden ... more >> Sahlgrenska Universitetssjukhuset/Östra Göteborg, Sweden Universitetssjukhuset Linköping, Sweden Karolinska Universitetssjukhuset Stockholm, Sweden, 17176 Danderyds Hospital Stockholm, Sweden Södersjukhuset Stockholm, Sweden Universitetssjukhuset i Örebro Örebro, Sweden United Kingdom Chalmers Sexual and Reproductive Health Service Edinburgh, United Kingdom, EH3 9ES Collapse << |
| NCT00828542 | - | - | Completed | - | - |
| NCT00725413 | Contraception | Phase 4 | Completed | - | - |
| NCT00847587 | - | - | Completed | - | - |
| NCT01666912 | Adolescence C... more >>ontraception Postpartum Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> University of North Carolina Hospitals Chapel Hill, North Carolina, United States, 27514 Collapse << |
| NCT03589040 | HIV Contracep... more >>tion Collapse << | Phase 2 | Not yet recruiting | June 2020 | Uganda ... more >> Infectious Diseases Institue Not yet recruiting Kampala, Uganda, 256 Contact: Stephen Research Manager 0392878652 sokoboi@idi.co.ug Principal Investigator: Mohammed Lamorde, PhD Sub-Investigator: Kimberley Scarsi, PharmD,MSc Sub-Investigator: Catherine Chappell, MD, MSc Sub-Investigator: Saye Khoo, PhD Sub-Investigator: Pauline Byakika, PhD Sub-Investigator: Kristin Darin, PharmD Sub-Investigator: Shadia Nakalema, MBChB, DTM&H Sub-Investigator: Stephen Walimbwa, MD Sub-Investigator: Julian Kaboggoza, BPharm Collapse << |
| NCT03353012 | Breastfeeding ... more >> Post Partum Collapse << | Phase 4 | Active, not recruiting | August 1, 2018 | Thailand ... more >> King Chulalongkorn Memorial Hospital Pathum Wan, Bangkok, Thailand, 10330 Collapse << |
| NCT00778999 | - | - | Completed | - | - |
| NCT01666912 | - | - | Completed | - | - |
| NCT00778999 | Infertility | Phase 4 | Completed | - | - |
| NCT02469454 | Contraception ... more >> Newborn Child Development Growth Collapse << | Phase 4 | Completed | - | Brazil ... more >> Clinical Hospital of Sao Paulo University Ribeirao Preto, SP, Brazil, 14048900 Collapse << |
| NCT00847587 | Complications; Contraceptive ... more >> Female Lactation Collapse << | Phase 4 | Completed | - | United States, Utah ... more >> University of Utah Hospital Salt Lake City, Utah, United States, 84105 Collapse << |
| NCT02480647 | Endometriosis | Phase 4 | Completed | - | Brazil ... more >> Nelsilene Mota Carvalho Campinas, São Paulo, Brazil, 13083887 Collapse << |
| NCT02657148 | - | - | Completed | - | United States, Pennsylvania ... more >> Magee-Womens Hospital of UPMC Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01796613 | Contraception | Phase 2 Phase 3 | Completed | - | Rwanda ... more >> Rinda Ubuzima Kigali, Rwanda Collapse << |
| NCT03282799 | HIV/AIDS Cont... more >>raception Collapse << | Phase 2 | Not yet recruiting | July 2022 | Uganda ... more >> Infectious Disease Institute Not yet recruiting Kampala, Uganda Contact: Mohammed Lamorde, MRCP PhD +256414307291 mlamorde@idi.co.ug Contact: Shadia Nakalema, MBChB, DTMH +256414307238 snakalema@idi.co.ug Principal Investigator: Catherine A Chappell, MD, MSc Principal Investigator: Mohammed Lamorde, MRCP PhD Principal Investigator: Kim Scarsi, PharmD, MSc Collapse << |
| NCT02866643 | Postpartum Contraception ... more >> Family Planning Collapse << | Phase 3 | Recruiting | December 2020 | United States, California ... more >> Obstetrics--Lucile Packard Children's Hospital Recruiting Stanford, California, United States, 94305 Contact: Klaira M Lerma, MPH 650-721-1562 familyplanningresearch@stanford.edu Principal Investigator: Kate A Shaw, MD, MS Collapse << |
| NCT03656289 | Contraception | Phase 1 | Not yet recruiting | January 1, 2020 | United States, Colorado ... more >> University of Colorado Denver Not yet recruiting Aurora, Colorado, United States, 80045 Contact: Eva Dindinger, BA 303-724-8482 Eva.Dindinger@ucdenver.edu Contact: Rebecca Seale, MA 303-724-2013 rebecca.seale@ucdenver.edu Principal Investigator: Aaron Lazorwitz, MD Collapse << |
| NCT03585504 | - | - | Completed | - | - |
| NCT00620035 | Contraception | Phase 3 | Completed | - | - |
| NCT00620035 | - | - | Completed | - | - |
| NCT02416687 | Contraception | Phase 4 | Completed | - | Brazil ... more >> Hospital das Clinica de Ribeirao Preto Ribeirão Preto, SP, Brazil, 14049-900 Collapse << |
| NCT02529683 | Hormonal Contraception | Not Applicable | Completed | - | - |
| NCT00620464 | Contraception | Phase 3 | Completed | - | - |
| NCT00651807 | Benign Prostatic Hyperplasia (... more >>BPH) Collapse << | Phase 2 | Terminated(Business Reasons) | - | - |
| NCT02445989 | Bacterial Vaginosis ... more >> HIV Collapse << | Phase 4 | Completed | - | Kenya ... more >> Thika Clinic Thika, Kenya Collapse << |
| NCT00967746 | Contraception | Phase 2 | Completed | - | - |
| NCT03585504 | Contraceptive Usage | Phase 3 | Completed | - | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT01277211 | Contraception | Phase 3 | Completed | - | - |
| NCT01767285 | Continuation Rate of Contracep... more >>tive Implant Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27701 Collapse << |
| NCT02037919 | Contraception ... more >> Abortion, Therapeutic Collapse << | Not Applicable | Active, not recruiting | December 2017 | United States, Illinois ... more >> Family Planning Associates Medical Grooup, LTD Chicago, Illinois, United States, 60661 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00620464 | - | - | Completed | - | - |
| NCT02267616 | Contraception | Phase 4 | Recruiting | December 2020 | United States, Indiana ... more >> Indiana University Recruiting Indianapolis, Indiana, United States, 46202 Contact: Sandra L Guelker, BA 317-948-0796 sguelker@iu.edu Contact: Shannon Barnes, RN MSN 317.948.7624 shanbarn@iupui.edu Principal Investigator: Jeffrey F Peipert, MD PhD United States, Missouri Washington University in St. Louis School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Lisa J Muench 314-362-4317 lisajmuench@wustl.edu Principal Investigator: Colleen McNicholas, MD Collapse << |
| NCT02275546 | Contraception: Optional Applic... more >>ator for Insertion of Vaginal Ring Collapse << | Phase 3 | Completed | - | - |
| NCT01277211 | - | - | Completed | - | - |
| NCT03118297 | Contraception ... more >> Bleeding Collapse << | Phase 3 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine in St. Louis Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT03118297 | - | - | Completed | - | - |
| NCT02367846 | Other Disorders of Bone Develo... more >>pment and Growth Collapse << | Phase 4 | Active, not recruiting | December 2018 | United States, Pennsylvania ... more >> Women's Health and Exercise Laboratories, The Pennsylvania State University University Park, Pennsylvania, United States, 16802 Collapse << |
| NCT00826839 | Infertility | Phase 4 | Withdrawn(poor enrollment) | - | United States, New York ... more >> The Ronald O. Perlman and Claudia Cohen Center for Reproductive Medicine at Weill Cornell Medical College New York, New York, United States, 10021 Collapse << |
| NCT01767285 | - | - | Completed | - | - |
| NCT01292785 | - | - | Completed | - | Austria ... more >> Department of Psychiatry and Psychotherapy, Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT00374972 | - | - | Withdrawn(difficulty in recrui... more >>tment. lack of sponsorship) Collapse << | - | - |
| NCT01310647 | Ovarian Diseases | Phase 2 | Completed | - | Spain ... more >> La Fe University Hospital. Department of Obstetrics and Gynecology Valencia, Spain, 46026 Collapse << |
| NCT00844012 | Endometriosis ... more >> Chronic Pelvic Pain Collapse << | Phase 4 | Unknown | July 2010 | Italy ... more >> University of Catanzaro, Italy Not yet recruiting Catanzaro, Italy, 88100 Contact: Fulvio Zullo, MD +3909613697180 zullo@unicz.it Principal Investigator: Stefano Palomba, MD Principal Investigator: Fulvio Zullo, MD Collapse << |
| NCT01638767 | Infertility | Phase 4 | Terminated(Difficulty to recru... more >>it patient due to lack of eligible patient) Collapse << | - | Canada, Quebec ... more >> Clinique Ovo Montreal, Quebec, Canada, H4P 2S4 Collapse << |
| NCT02594462 | Sickle Cell Disease | Phase 4 | Active, not recruiting | June 2019 | Brazil ... more >> Bahiana School of Medicne and Public Health Salvador, Bahia, Brazil University of Sao Paulo Ribeirao Preto, Sao Paulo, Brazil, 14049-900 Collapse << |
| NCT01178125 | Contraception ... more >> Female Contraception Collapse << | Phase 3 | Completed | - | - |
| NCT01185678 | - | - | Completed | - | Poland ... more >> Many Locations, Poland Collapse << |
| NCT01384331 | Menstrual Problem | Phase 4 | Unknown | April 2013 | Australia, New South Wales ... more >> Sydney centre for Reproductive Health Research, FPNSW Not yet recruiting Sydney, New South Wales, Australia, 2131 Principal Investigator: Edith Weisberg, MB BS MM Collapse << |
| NCT00946192 | Exercise-related Amenorrhea | Phase 3 | Recruiting | December 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Madhu Misra, M.D. 617-724-5602 mmisra@partners.org Contact: Alexander Toth, B.A. 617 724 6046 attoth@MGH.HARVARD.EDU Principal Investigator: Madhu Misra, M.D. Collapse << |
| NCT01087879 | Contraception | Not Applicable | Completed | - | Finland ... more >> Department of Obstetrics and Gynaecology, University of Oulu Oulu, Finland, 90014 Collapse << |
| NCT00326404 | Contraception | Phase 3 | Unknown | May 2009 | Canada, Ontario ... more >> The Hospital for Sick Children Recruiting Toronto, Ontario, Canada, M5G 1X8 Contact: Lisa Allen, MD 416 813 6188 lisa.allen@sickkids.ca Principal Investigator: Lisa Allen, MD Principal Investigator: Sari Kives, MD Sub-Investigator: Rachel Spitzer, MD Sub-Investigator: Karen Leslie, MD Collapse << |
| NCT00204438 | Contraception | Not Applicable | Completed | - | Canada, Saskatchewan ... more >> Ob-Gyn Royal University Hospital Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT01252186 | Hemostasis Or... more >>al Contraceptive Collapse << | Phase 2 | Completed | - | United States, California ... more >> Teva Investigational Site San Diego, California, United States, 92103 Teva Investigational Site San Diego, California, United States, 92108 Teva Investigational Site San Diego, California, United States, 92123 United States, District of Columbia Teva Investigational Site Washington, District of Columbia, United States, 20036 United States, Florida Teva Investigational Site Miami, Florida, United States, 33186 Teva Investigational Site West Palm Beach, Florida, United States, 33409 United States, Georgia Teva Investigational Site Sandy Springs, Georgia, United States, 30328 United States, New Jersey Teva Investigational Site Edison, New Jersey, United States, 08817 Teva Investigational Site Plainsboro, New Jersey, United States, 08536 United States, New Mexico Teva Investigational Site Albuquerque, New Mexico, United States, 87102 United States, New York Teva Investigational Site Port Jefferson, New York, United States, 11777 Teva Investigational Site Rochester, New York, United States, 14609 United States, North Carolina Teva Investigational Site Winston Salem, North Carolina, United States, 27103 United States, Pennsylvania Teva Investigational Site Philadelphia, Pennsylvania, United States, 19114 Teva Investigational Site Pittsburgh, Pennsylvania, United States, 15206 Teva Investigational Site Uniontown, Pennsylvania, United States, 15401 United States, Texas Teva Investigational Site Dallas, Texas, United States, 75234 Teva Investigational Site Houston, Texas, United States, 77054 Teva Investigational Site San Antonio, Texas, United States, 78258 United States, Virginia Teva Investigational Site Richmond, Virginia, United States, 23233 United States, Washington Teva Investigational Site Seattle, Washington, United States, 98105 Italy Teva Investigational Site Cagliari, Italy, 09124 Teva Investigational Site Modena, Italy, 41100 Teva Investigational Site Pavia, Italy, 27100 Collapse << |
| NCT01291004 | Follicle Development ... more >> Ovarian Follicle Follicle Count Follicle Size Oral Contraceptive Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Teva Investigational Site Philadelphia, Pennsylvania, United States, 19114 United States, Washington Teva Investigational Site Seattle, Washington, United States, 98105 Collapse << |
| NCT01252186 | - | - | Completed | - | - |
| NCT03353857 | Biological Availability | Phase 1 | Active, not recruiting | December 18, 2018 | Germany ... more >> CRS Clinical-Research-Services Mannheim GmbH Mannheim, Baden-Württemberg, Germany, 68167 CRS Clinical-Research-Services Mönchengladbach GmbH Mönchengladbach, Nordrhein-Westfalen, Germany, 41061 Collapse << |
| NCT01005056 | - | - | Completed | - | - |
| NCT01388491 | Hemostasis Or... more >>al Contraceptive Collapse << | Phase 2 | Completed | - | Germany ... more >> Teva Investigational Site Essen, Germany, 45127 Teva Investigational Site Frankfurt am Main, Germany, 60439 Teva Investigational Site Frankfurt, Germany Teva Investigational Site Hamburg, Germany, 22149 Teva Investigational Site Hamburg, Germany, 22159 Teva Investigational Site Magdeburg, Germany, 39112 Teva Investigational Site Muehlheim am Main, Germany, 63165 Israel Teva Investigational Site Givataim, Israel Teva Investigational Site Haifa, Israel Teva Investigational Site Modi'in, Israel Teva Investigational Site Or-Yehuda, Israel Teva Investigational Site RishonLe'zio, Israel Teva Investigational Site Tel-Aviv, Israel Italy Teva Investigational Site Bari, Italy, 70124 Teva Investigational Site Brescia, Italy, 25123 Teva Investigational Site Cagliari, Italy, 09124 Teva Investigational Site Catania, Italy, 95123 Teva Investigational Site Modena, Italy, 41124 Teva Investigational Site Napoli, Italy, 80131 Teva Investigational Site Padova, Italy, 35128 Teva Investigational Site Pavia, Italy, 27100 Teva Investigational Site Pisa, Italy, 56126 Teva Investigational Site Siena, Italy, 53100 Spain Teva Investigational Site Alicante, Spain Teva Investigational Site Barcelona, Spain Teva Investigational Site Gava', Barcelona, Spain Teva Investigational Site Guadalajara, Spain Teva Investigational Site Lugo, Spain Teva Investigational Site Madrid, Spain Teva Investigational Site Vitoria-Gasteiz, Spain Collapse << |
| NCT00185419 | Contraception | Phase 3 | Completed | - | China ... more >> ask Contact, China Collapse << |
| NCT01388491 | - | - | Completed | - | - |
| NCT00612508 | - | - | Completed | - | - |
| NCT01178125 | - | - | Completed | - | - |
| NCT00544882 | Healthy | Phase 3 | Completed | - | United States, New Jersey ... more >> Duramed Investigational Site Lawrenceville, New Jersey, United States, 08648 United States, Ohio Duramed Investigational Site Columbus, Ohio, United States, 43213 United States, Pennsylvania Duramed Investigational Site Philadelphia, Pennsylvania, United States, 19114 United States, Washington Duramed Investigational Site Seattle, Washington, United States, 98105 Collapse << |
| NCT00612508 | Contraceptive Usage ... more >> Vaginal Epithelial Disruption Collapse << | Not Applicable | Completed | - | United States, Oregon ... more >> Oregon Health and Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT01076582 | Oral Contraceptive | Phase 3 | Completed | - | - |
| NCT00544882 | - | - | Completed | - | - |
| NCT01256619 | Women With PMS | Phase 2 | Unknown | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences-BASIR center Recruiting Tehran, Iran, Islamic Republic of, 61-14185 Contact: abbas noroozi, PhD 009802166581560 noroozia@tums.ac.ir Principal Investigator: Abbas noroozi, PhD Collapse << |
| NCT02275546 | - | - | Completed | - | - |
| NCT00563472 | Healthy | Phase 2 | Completed | - | Netherlands ... more >> Dinox Groningen, Netherlands, 9713 GZ Collapse << |
| NCT02579590 | - | - | Recruiting | March 2018 | Egypt ... more >> Assiut university Recruiting Assiut, Egypt, 71111 Contact: MOhammed k Ali, MD m_khairy2001@yahoo.com Collapse << |
| NCT01500473 | Congenital Central Hypoventila... more >>tion Syndrome Collapse << | Not Applicable | Unknown | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Children's Hospital Los Angeles Not yet recruiting Los Angeles, California, United States, 90027 Contact: Thomas Keens, MD 323-361-2101 Collapse << |
| NCT01482338 | Premenstrual Syndrome | Phase 4 | Completed | - | Thailand ... more >> Family Unit, King Chulalongkorn Memorial Hospital Bangkok, Thailand, 10330 Collapse << |
| NCT01438736 | Bleeding | Phase 4 | Unknown | - | Finland ... more >> VL Medi Oy Not yet recruiting Helsinki, Finland, 00101 Principal Investigator: Dan Apter, docent Collapse << |
| NCT01588873 | Adverse Effect of Oral Contrac... more >>eptives, Sequela Collapse << | Phase 4 | Recruiting | December 2019 | Finland ... more >> Department of Obstetrics and Gynaecology, University Hospital of Oulu Recruiting Oulu, Finland, 90029 Contact: Laure C Morin-Papunen, PhD +358 8 3154109 lmp@cc.oulu.fi Contact: Juha S Tapanainen, PhD +358 8 3153172 juha.tapanainen@oulu.fi Principal Investigator: Anni S Rantala, med student Sub-Investigator: Johanna Puurunen, MD Collapse << |
| NCT01738724 | Uterine Leiomyoma | Phase 4 | Unknown | December 2013 | - |
| NCT01466673 | - | - | Completed | - | - |
| NCT03354494 | - | - | Completed | - | - |
| NCT02103764 | Dysfunctional Uterine Bleeding | Phase 3 | Active, not recruiting | December 2018 | Thailand ... more >> Faculty of Medicine Siriraj Hospital, Mahidol University Bangkok, Thailand, 10700 Collapse << |
| NCT02757287 | Infertility | Phase 4 | Withdrawn(Lack of financial su... more >>pport) Collapse << | December 1, 2018 | Spain ... more >> Hospital Quiron Dexeus Barcelona, Spain, 08028 Collapse << |
| NCT01466673 | Acne Vulgaris | Phase 4 | Completed | - | Thailand ... more >> Department of Obstetrics and Gynecology, Faculty of Medicine, Chulalongkorn University Bangkok, Thailand Department of Obstetrics and Gynecology, Faculty of Medicine, Siriraj Hospital, Mahidol University Bangkok, Thailand Department of Obstetrics and Gynecology, Faculty of Medicine, Chiang Mai University Chiang Mai, Thailand Collapse << |
| NCT01559480 | Endometriosis | Not Applicable | Completed | - | Thailand ... more >> Faculty of medicine Siriraj hospital, Mahidol university Bangkok noi, Bangkok, Thailand, 10700 Siriraj hospital Mahidol university Bangkok noi, Bangkok, Thailand, 10700 Collapse << |
| NCT02398903 | - | - | Recruiting | December 2018 | France ... more >> Hôpital Ambroise Paré Recruiting Boulogne-Billancourt, France Pitié Salpetriere Hospital Recruiting Paris, France, 75013 Contact: Anne Bachelot, MD, PhD anne.bachelot@psl.aphp.fr Collapse << |
| NCT01243697 | Ondine Syndrome | Phase 2 Phase 3 | Completed | - | France ... more >> Pitie salpetriere hospital Paris, France, 75013 Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
