| NCT00734032 | Atherosclerosis | Phase 2 | Completed | - | Japan
... more >> GSK Investigational Site
Fukuoka, Japan, 812-0025
GSK Investigational Site
Fukuoka, Japan, 818-0036
GSK Investigational Site
Fukuoka, Japan, 819-1102
GSK Investigational Site
Tokyo, Japan, 105-0004
GSK Investigational Site
Tokyo, Japan, 154-0024
GSK Investigational Site
Tokyo, Japan, 160-0017
GSK Investigational Site
Tokyo, Japan, 174-0051 Collapse << |
| NCT01916720 | Atherosclerosis | Phase 2 | Completed | - | - |
| NCT01067339 | Endothelial Dysfunction | Phase 3 | Completed | - | United States, Minnesota
... more >>
Mayo Clinic
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00734032 | - | - | Completed | - | - |
| NCT01636271 | - | - | Completed | - | - |
| NCT01067339 | - | - | Completed | - | - |
| NCT01000727 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT00743860 | Atherosclerosis | Phase 1 | Completed | - | United States, Washington
... more >>
GSK Investigational Site
Tacoma, Washington, United States, 98418 Collapse << |
| NCT01000727 | - | - | Completed | - | - |
| NCT00799903 | Atherosclerosis | Phase 3 | Completed | - | - |
| NCT00411073 | Atherosclerosis | Phase 1 | Completed | - | United States, Indiana
... more >>
GSK Investigational Site
Evansville, Indiana, United States, 47714 Collapse << |
| NCT00622830 | Atherosclerosis | Phase 1 | Completed | - | Japan
... more >> GSK Investigational Site
Ibaraki, Japan, 305-0856 Collapse << |
| NCT00799903 | - | - | Completed | - | - |
| NCT01711723 | Atherosclerosis | Phase 1 | Completed | - | United States, Florida
... more >>
GSK Investigational Site
Orlando, Florida, United States, 32809
United States, Minnesota
GSK Investigational Site
Minneapolis, Minnesota, United States, 55404 Collapse << |
| NCT01154114 | Atherosclerosis | Phase 1 | Completed | - | United States, Florida
... more >>
GSK Investigational Site
Miami, Florida, United States, 33143
GSK Investigational Site
Orlando, Florida, United States, 32809 Collapse << |
| NCT01506895 | Retinopathy, Diabetic | Phase 2 | Completed | - | Australia, New South Wales
... more >>
GSK Investigational Site
Parramatta, New South Wales, Australia, 2150
GSK Investigational Site
Sydney, New South Wales, Australia, 2000
Australia, Victoria
GSK Investigational Site
East Melbourne, Victoria, Australia, 3002
Australia, Western Australia
GSK Investigational Site
Nedlands, Western Australia, Australia, 6009
Denmark
GSK Investigational Site
Glostrup, Denmark
Germany
GSK Investigational Site
Freiburg, Baden-Wuerttemberg, Germany, 79106
GSK Investigational Site
Ulm, Baden-Wuerttemberg, Germany, 89075
GSK Investigational Site
Koeln, Nordrhein-Westfalen, Germany, 51109
GSK Investigational Site
Muenster, Nordrhein-Westfalen, Germany, 48145
GSK Investigational Site
Leipzig, Sachsen, Germany, 04103
Italy
GSK Investigational Site
Milano, Lombardia, Italy, 20132
GSK Investigational Site
Milano, Lombardia, Italy, 20157
GSK Investigational Site
Torino, Piemonte, Italy, 10122
GSK Investigational Site
Padova, Veneto, Italy, 35128
Netherlands
GSK Investigational Site
Amsterdam, Netherlands, 1105 AZ
GSK Investigational Site
Rotterdam, Netherlands, 3011 BH Collapse << |
| NCT00551317 | Atherosclerosis | Phase 1 | Completed | - | United States, Hawaii
... more >>
GSK Investigational Site
Honolulu, Hawaii, United States, 96813 Collapse << |
| NCT01852565 | Atherosclerosis | Phase 1 | Completed | - | United States, Maryland
... more >>
GSK Investigational Site
Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00704431 | Atherosclerosis | Phase 1 | Completed | - | United States, Pennsylvania
... more >>
GSK Investigational Site
King of Prussia, Pennsylvania, United States, 19406 Collapse << |
| NCT01751074 | Atherosclerosis | Phase 1 | Completed | - | United States, California
... more >>
GSK Investigational Site
Glendale, California, United States, 91206 Collapse << |
| NCT01873339 | Atherosclerosis | Phase 1 | Completed | - | United States, Maryland
... more >>
GSK Investigational Site
Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02000804 | Atherosclerosis | Phase 1 | Completed | - | China
... more >> GSK Investigational Site
Shanghai, China, 200030 Collapse << |
| NCT02058641 | Atherosclerosis | Phase 1 | Completed | - | United Kingdom
... more >> GSK Investigational Site
Cambridge, United Kingdom, CB2 0GG Collapse << |
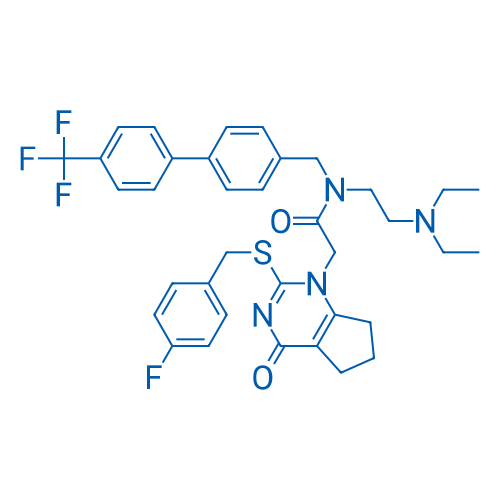

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
