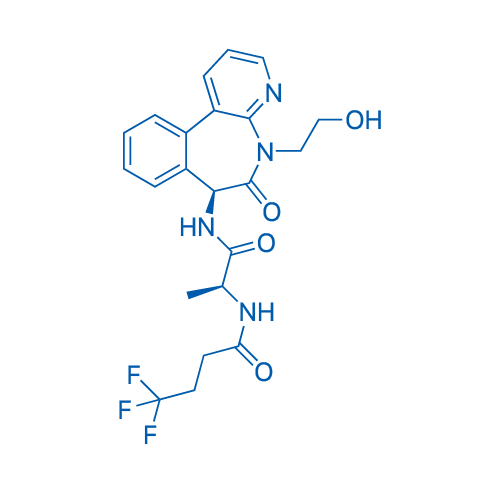
CAS No.: 1421438-81-4
Crenigacestat Catalog No. CSN15691
Synonyms: LY3039478
LY3039478 is a novel, potent Notch inhibitor with an IC50 of 0.41 nM.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Notch
IC50:0.41nM- 描述
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice: 8 mg/kg[3] (p.o.)
给药途径 p.o.
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01695005 Neoplasms Neo... more >>plasm Metastasis Lymphoma Collapse << Phase 1 Completed - United States, Florida ... more >> University of Miami School of Medicine Miami, Florida, United States, 33136 United States, Massachusetts Harvard Medical School Boston, Massachusetts, United States, 02215 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, New York Montefiore Medical Center Bronx, New York, United States, 10461 Columbia University Medical Center New York, New York, United States, 10032 Denmark For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kobenhavn, Denmark, 2100 France For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Paris, France, 75248 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Villejuif, France, 94805 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tübingen, Germany, 72076 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08035 United Kingdom For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. London, United Kingdom, SE1 9RT Collapse << NCT03502577 Recurrent Plasma Cell Myeloma ... more >> Refractory Plasma Cell Myeloma Collapse << Phase 1 Recruiting July 30, 2034 United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Immunotherapy Trials Intake, SCCA 206-606-4668 immunotherapy@seattlecca.org Principal Investigator: Andrew J. Cowan Collapse << NCT02784795 Solid Tumor B... more >>reast Cancer Colon Cancer Cholangiocarcinoma Soft Tissue Sarcoma Collapse << Phase 1 Active, not recruiting January 31, 2019 United States, Florida ... more >> Sylvester Comprehensive Cancer Center Miami, Florida, United States, 33136 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, Texas University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Denmark Rigshospitalet Copenhagen, København Ø, Denmark, 2100 France Institut Bergonie Bordeaux, France, 33076 Centre Leon Berard Lyon Cedex 08, France, 69373 Gustave Roussy Villejuif Cedex, France, 94805 Spain Hospital Universitari Vall d'Hebron Barcelona, Spain, 08035 Fundacion Jimenez Diaz Madrid, Spain, 28040 Hospital Madrid Norte Sanchinarro Madrid, Spain, 28050 Collapse << - 更多
- 参考文献
- [1] Warren J. P. Discovery of a Novel Notch Inhibitor.The 8th SCI-RSC Symposium on Proteinase Inhibitor Design April 15-16, 2013, Basel, Switzerland.
- [2] Mark H. B, Hong G, et al. Novel inhibitor of Notch signaling for the treatment of cancer. Abstract 1131, AACR 104th Annual Meeting 2013.
- [3] Bhagat TD, Zou Y, et al. Notch Pathway Is Activated via Genetic and Epigenetic Alterations and Is a Therapeutic Target in Clear Cell Renal Cancer. J Biol Chem. 2017 Jan 20;292(3):837-846.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1421438-81-4 | 储存条件 |
|
|||||
| 分子式 | C22H23F3N4O4 | 运输 | 蓝冰 | |||||
| 分子量 | 464.44 | 别名 | LY3039478 | |||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
