| NCT02372604 | Chronic Spontaneous Urticaria | Phase 3 | Completed | - | - |
| NCT01567501 | Fed | Phase 1 | Completed | - | India
... more >> Accutest Research Lab (I) Pvt. Ltd.
Ahmedabad, Gujarat, India, 380054 Collapse << |
| NCT03555890 | Rhinitis | Phase 1 | Completed | - | Japan
... more >> GSK Investigational Site
Fukuoka, Japan, 812-0025 Collapse << |
| NCT02394119 | Nephrotic Syndrome | Phase 2 | Recruiting | May 2019 | Italy
... more >> IRCCS Istituto Giannina Gaslini
Recruiting
Genoa, Italy/GE, Italy, 16147
Contact: Gian Marco Ghiggeri, MD 0039 010 5636 2419 gmarcoghiggeri@ospedale-gaslini.ge.it
Principal Investigator: Gian Marco Ghiggeri, MD
Sub-Investigator: Alice Bonanni, MD Collapse << |
| NCT02394106 | Nephrotic Syndrome | Phase 2 | Recruiting | March 2018 | Italy
... more >> IRCCS Istituto Giannina Gaslini
Recruiting
Genoa, Italy/GE, Italy, 16147
Contact: Gian Marco Ghiggeri, MD 0039 010 5636 2419 gmarcoghiggeri@ospedale-gaslini.ge.it
Principal Investigator: Gian Marco Ghiggeri, MD
Sub-Investigator: Alice Bonanni, MD Collapse << |
| NCT02771249 | Healthy Volunteers | Phase 1 | Recruiting | December 31, 2019 | United States, Maryland
... more >>
National Institutes of Health Clinical Center
Recruiting
Bethesda, Maryland, United States, 20892
Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT00557284 | - | - | Completed | - | - |
| NCT00001910 | - | - | Completed | - | United States, Maryland
... more >>
National Institute of Allergy and Infectious Diseases (NIAID)
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03590912 | Otitis Media With Effusion | Phase 4 | Not yet recruiting | September 2019 | Nepal
... more >> Lumbini Medical College
Tansen, Palpa, Nepal, 32500 Collapse << |
| NCT00557284 | Atopic Dermatitis | Phase 4 | Completed | - | United States, Colorado
... more >>
1st Allergy & Clinical Research Centers
Centennial, Colorado, United States, 80112
1st Allergy & Clinical Research Centers
Thornton, Colorado, United States, 80229 Collapse << |
| NCT02873364 | Urticaria | Phase 3 | Unknown | January 2017 | Iran, Islamic Republic of
... more >>
Shiraz University of Medical Sciences
Recruiting
Shiraz, Fars, Iran, Islamic Republic of
Contact: Keramatallah Jahanshahi, MD +989177416523 kjahanshahi91@gmail.com Collapse << |
| NCT03523221 | Mild -Moderate Anaphylaxis - S... more >>ymptoms That Affect the Skin With Cardiovascular, Respiratory or Gastrointestinal Involvement, But no Signs of Hypotension ,Laryng Collapse << | Phase 1 | Recruiting | August 2020 | Qatar
... more >> Hamad Medical Corporation
Recruiting
Doha, Qatar, 3050
Contact: Rafah Sayyed, MD +97455747045 ralsayyed@hamad.qa Collapse << |
| NCT03296358 | Urticaria
All... more >>ergy Collapse << | Not Applicable | Recruiting | October 31, 2019 | Thailand
... more >> Chulalongkorn Hospital
Recruiting
Pathum Wan, Bangkok, Thailand, 10600
Contact: Pakhawadee Palungwachira, Bachelor 66897704758 zaphyr_s@yahoo.com
Principal Investigator: Pakhawadee Palungwachira, Bachelor
Sub-Investigator: Katsara Vilaisri, Bachelor
Sub-Investigator: Klongwong Musikatavorn, Bachelor Collapse << |
| NCT01451996 | - | - | Completed | - | - |
| NCT01451996 | Healthy | Phase 4 | Completed | - | United States, Illinois
... more >>
University of Chicago
Chicago, Illinois, United States, 60637 Collapse << |
| NCT02613910 | Pemphigus | Phase 3 | Terminated(Novartis has acquir... more >>ed the rights to ofatumumab and terminated the OPV116910 and OPV117059 studies. This decision is not linked to any safety consideration.) Collapse << | - | United States, California
... more >>
GSK Investigational Site
Los Angeles, California, United States, 90045
United States, Michigan
GSK Investigational Site
Ann Arbor, Michigan, United States, 48103 Collapse << |
| NCT02646904 | Allergic Rhinitis | Phase 4 | Completed | - | Italy
... more >> Institute of Biomedicine and Molecular Immunology (IBIM), National Research Council
Palermo, Sicily, Italy, 90146
Institute of Biomedicine and Molecular Immunology, IBIM
Palermo, Italy, 90146 Collapse << |
| NCT02613910 | - | - | Terminated(Novartis has acquir... more >>ed the rights to ofatumumab and terminated the OPV116910 and OPV117059 studies. This decision is not linked to any safety consideration.) Collapse << | - | - |
| NCT01985789 | Asthma | Phase 4 | Completed | - | Canada, Saskatchewan
... more >>
University of Saskatchewan
Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT02583594 | Progressive Multiple Sclerosis | Phase 1 | Active, not recruiting | April 2021 | Spain
... more >> Investigational Site Number 724001
Barcelona, Spain, 08035 Collapse << |
| NCT01902836 | Childhood Atopic Dermatitis | Phase 2 | Completed | - | Mexico
... more >> Hospital Infantil de México, Fedérico Gómez
México, DF, Mexico, 06720 Collapse << |
| NCT01981499 | Migraine | Phase 1 | Terminated(The trial was prema... more >>turely terminated on 01April2014 due to safety concerns.) Collapse << | - | United States, Minnesota
... more >>
Pfizer Investigational Site
St. Paul, Minnesota, United States, 55114 Collapse << |
| NCT02024152 | Acute Urticaria | Phase 1 | Completed | - | Canada, Quebec
... more >> Algorithme Pharma
Montreal, Quebec, Canada Collapse << |
| NCT01940393 | Urticaria | Phase 4 | Completed | - | Colombia
... more >> Medellin
Medellin, Antioquia, Colombia Collapse << |
| NCT01241214 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | United States, Texas
... more >>
Clinical Investigative Site 7905
Austin, Texas, United States, 78731
Clinical Investigative Site 7907
Kerrville, Texas, United States, 78028
Clinical Investigative Site 7903
New Braunfels, Texas, United States, 78130
Clinical Investigative Site 7901
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7902
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7904
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7906
San Antonio, Texas, United States, 78229 Collapse << |
| NCT02535416 | Healthy | Phase 1 | Completed | - | Australia, Queensland
... more >>
QPharm, Pty Limited, Royal Brisbane Hospital
Herston, Queensland, Australia, 4029 Collapse << |
| NCT02535416 | - | - | Completed | - | - |
| NCT00561717 | Seasonal Allergic Rhinitis | Phase 4 | Completed | - | Canada, Ontario
... more >>
Kingston, Ontario, Canada, K7L 2V7 Collapse << |
| NCT00517946 | - | - | Completed | - | United Kingdom
... more >> GSK Investigational Site
London, United Kingdom, W1G 8HU Collapse << |
| NCT01134328 | - | - | Completed | - | - |
| NCT01008592 | - | - | Terminated(Mediators of intere... more >>st were not consistently detectable with the analytical methods employed.) Collapse << | - | United States, North Carolina
... more >>
Wake Forest University Health Sciences, Department of Dermatology
Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT01318681 | Seasonal Allergic Rhinitis
... more >> Driving Ability
Cognition Collapse << | Not Applicable | Completed | - | Netherlands
... more >> Maastricht University
Maastricht, Limburg, Netherlands, 62ooMD Collapse << |
| NCT00533637 | Allergic Rhinitis | Phase 2 | Completed | - | Sweden
... more >> Department of Otorhinolaryngology, Lund University Hospital
Lund, Sweden, SE-221 85 Collapse << |
| NCT01484119 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | United States, Texas
... more >>
Clinical Investigative Site 7905
Austin, Texas, United States, 78731
Clinical Investigative Site 7907
Kerrville, Texas, United States, 78028
Clinical Investigative Site 7903
New Braunfels, Texas, United States, 78130
Clinical Investigative Site 7901
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7902
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7904
San Antonio, Texas, United States, 78229
Clinical Investigative Site 7906
San Antonio, Texas, United States, 78229 Collapse << |
| NCT02935699 | Acute Urticaria | Phase 3 | Recruiting | April 2018 | United States, Texas
... more >>
City Doc Urgent Care center
Recruiting
Dallas, Texas, United States, 75209
Contact: Brook Jackson
Canada, Ontario
Ottawa Hospital
Recruiting
Ottawa, Ontario, Canada, K1Y 4E9
Contact: Cathy Clement Collapse << |
| NCT01068223 | Histamine Induced Itch | Phase 1 | Completed | - | United Kingdom
... more >>
Harrow, United Kingdom Collapse << |
| NCT00610584 | Seasonal Allergic Rhinitis | Not Applicable | Completed | - | Germany
... more >> Charité - Institute for Social Medicine
Berlin, Germany, 10098 Collapse << |
| NCT00291642 | Rhinitis
Alle... more >>rgic
Seasonal Collapse << | Phase 2 | Completed | - | Canada, Ontario
... more >>
Kingston, Ontario, Canada Collapse << |
| NCT00974571 | - | - | Completed | - | - |
| NCT01134328 | Allergic Conjunctivitis | Phase 2 | Completed | - | United States, Massachusetts
... more >>
Ora, Inc
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT01495338 | Normal Ocular Health | Phase 1 | Completed | - | United States, Massachusetts
... more >>
Andover Eye Associates
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT03167216 | Chronic Prostatitis With Chron... more >>ic Pelvic Pain Syndrome Collapse << | Early Phase 1 | Recruiting | June 30, 2021 | United States, Illinois
... more >>
Northwestern Medical Faculty Foundation
Recruiting
Chicago, Illinois, United States, 60611
Principal Investigator: Praveen Thumbikat, PhD Collapse << |
| NCT01825655 | Pruritus | Phase 4 | Terminated(Unable to recruit p... more >>atients) Collapse << | - | United States, Virginia
... more >>
Children's Hospital of Richmond at VCU
Richmond, Virginia, United States, 23298 Collapse << |
| NCT00974571 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT01825655 | - | - | Terminated(Unable to recruit p... more >>atients) Collapse << | - | - |
| NCT00474890 | Atopy
Rhiniti... more >>s Collapse << | Not Applicable | Completed | - | Germany
... more >> Fraunhofer Institute for Toxicology and Experimental Medicine
Hannover, Germany, 30625 Collapse << |
| NCT02451579 | Actinic Keratoses | Not Applicable | Completed | - | United States, California
... more >>
Cosmetic Laser Dermatology
San Diego, California, United States, 92121 Collapse << |
| NCT00851344 | Rhinitis, Allergic, Seasonal | Phase 2 | Completed | - | Germany
... more >> GSK Investigational Site
Hannover, Niedersachsen, Germany, 30625 Collapse << |
| NCT02447393 | Dermatitis | Phase 1 | Completed | - | Japan
... more >> GSK Investigational Site
Kagoshima, Japan, 890-0081 Collapse << |
| NCT00972504 | Rhinitis, Allergic, Seasonal | Phase 2 | Completed | - | Germany
... more >> GSK Investigational Site
Hannover, Niedersachsen, Germany, 30625 Collapse << |
| NCT00291642 | - | - | Completed | - | - |
| NCT00972504 | - | - | Completed | - | - |
| NCT00257595 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT00420082 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | Austria
... more >> Allergy Center Vienna West
Vienna, Austria, A-1150 Collapse << |
| NCT02543346 | Allergic Rhinitis | Phase 4 | Completed | - | United States, Texas
... more >>
Biogenics Research Chamber
San Antonio, Texas, United States, 78229
Canada, Ontario
Kingston General Hospital
Kingston, Ontario, Canada, K7L 2V7 Collapse << |
| NCT00240032 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT01103050 | Allergic Rhinitis | Phase 2 | Completed | - | Canada
... more >> Novartis Investigative Site
Ontario, Canada Collapse << |
| NCT00544388 | Rhinitis, Allergic, Seasonal | Phase 3 | Completed | - | - |
| NCT02173249 | Healthy | Phase 1 | Completed | - | United States, Massachusetts
... more >>
Andover Eye Associates
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00253058 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT03192488 | Exercise Intolerance at Altitu... more >>de Collapse << | Phase 4 | Recruiting | May 31, 2018 | United States, Indiana
... more >>
Indiana University
Recruiting
Bloomington, Indiana, United States, 47401
Contact: Robert Chapman, PhD 812-856-2452 rfchapma@indiana.edu Collapse << |
| NCT00257569 | Dermatitis, Atopic | Phase 3 | Completed | - | - |
| NCT01651871 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | - |
| NCT00334698 | Seasonal Allergic Rhinitis | Not Applicable | Completed | - | Germany
... more >> Fraunhofer-Institute of Toxicology and Experimental Medicine
Hannover, Germany, 30623 Collapse << |
| NCT01881113 | Allergic Conjunctivitis | Phase 3 | Completed | - | United States, Massachusetts
... more >>
Ora, Inc.
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT01322282 | Allergy | Phase 1 | Completed | - | Canada, Quebec
... more >> Algorithme Pharma Inc.
Mount-Royal, Quebec, Canada, H3P 3P1 Collapse << |
| NCT01748344 | Seasonal Allergic Rhinitis | Phase 2 | Completed | - | Austria
... more >> Vienna Clinical Site
Vienna, Austria Collapse << |
| NCT01881113 | - | - | Completed | - | - |
| NCT01127620 | Perennial Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT01551056 | Allergic Conjunctivitis | Phase 3 | Completed | - | United States, Massachusetts
... more >>
Ora, Inc.
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT01685242 | - | - | Completed | - | - |
| NCT01551056 | - | - | Completed | - | - |
| NCT01685242 | Allergic Conjunctivitis | Phase 3 | Completed | - | United States, Massachusetts
... more >>
Andover Eye Associates
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00490204 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | Japan
... more >> GSK Investigational Site
Chiba, Japan, 277-0882
GSK Investigational Site
Fukuoka, Japan, 807-0856
GSK Investigational Site
Fukuoka, Japan, 811-1201
GSK Investigational Site
Fukuoka, Japan, 819-0002
GSK Investigational Site
Hokkaido, Japan, 001-0923
GSK Investigational Site
Hokkaido, Japan, 007-0840
GSK Investigational Site
Hokkaido, Japan, 053-0833
GSK Investigational Site
Hokkaido, Japan, 061-1133
GSK Investigational Site
Hokkaido, Japan, 061-1448
GSK Investigational Site
Hokkaido, Japan, 062-0034
GSK Investigational Site
Kanagawa, Japan, 212-0027
GSK Investigational Site
Kanagawa, Japan, 213-0011
GSK Investigational Site
Kanagawa, Japan, 216-0002
GSK Investigational Site
Kanagawa, Japan, 222-0011
GSK Investigational Site
Kanagawa, Japan, 224-0003
GSK Investigational Site
Kanagawa, Japan, 232-0056
GSK Investigational Site
Kumamoto, Japan, 862-0952
GSK Investigational Site
Kumamoto, Japan, 862-0962
GSK Investigational Site
Oita, Japan, 870-0021
GSK Investigational Site
Saitama, Japan, 333-0861
GSK Investigational Site
Saitama, Japan, 336-0022
GSK Investigational Site
Saitama, Japan, 350-1205
GSK Investigational Site
Saitama, Japan, 355-0062
GSK Investigational Site
Shizuoka, Japan, 420-0803
GSK Investigational Site
Shizuoka, Japan, 422-8066
GSK Investigational Site
Shizuoka, Japan, 436-0058
GSK Investigational Site
Tokyo, Japan, 157-0067
GSK Investigational Site
Tokyo, Japan, 170-0005 Collapse << |
| NCT03047278 | Neuropathic Pain
... more >> Type 2 Diabetes Mellitus
Diabetic Neuropathy, Painful Collapse << | Phase 4 | Enrolling by invitation | July 1, 2019 | - |
| NCT00257582 | Pruritus | Phase 3 | Completed | - | - |
| NCT00751218 | Urticaria | Phase 4 | Completed | - | - |
| NCT00637455 | Seasonal Allergic Rhinitis | Phase 4 | Completed | - | United States, New Jersey
... more >>
Sanofi-Aventis
Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00504933 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT02756624 | Healthy Subjects | Phase 3 | Completed | - | United States, Massachusetts
... more >>
Ora Clinical Site Network
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT02756624 | - | - | Completed | - | - |
| NCT00881127 | Allergies | Phase 1 | Completed | - | - |
| NCT00150761 | Anti-allergic Agents | Phase 4 | Completed | - | - |
| NCT01622283 | Rhinitis, Allergic, Perennial ... more >>and Seasonal Collapse << | Phase 1 | Completed | - | Japan
... more >> GSK Investigational Site
Kagoshima, Japan, 890-0081 Collapse << |
| NCT00488176 | Seasonal Allergic Rhinitis | Phase 4 | Unknown | December 2013 | Poland
... more >> Department of Pediatrics and Allergy, Medical University of Lodz Lodz, Poland
Recruiting
Lodz, Poland, 93-513
Contact: Violetta Ścibiorek, MD 00 48 42 689 59 72 alergol@kopernik.lodz.pl
Principal Investigator: Iwona Stelmach, MD, PhD, Prof Collapse << |
| NCT00751166 | Urticaria | Phase 4 | Terminated(Study could not be ... more >>re-supplied with study medication in a timely manner.) Collapse << | - | - |
| NCT01332188 | Allergic Conjunctivitis | Phase 2
Phase 3 | Completed | - | United States, Massachusetts
... more >>
Ora, Inc.
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00375713 | - | - | Completed | - | - |
| NCT00881634 | Allergies | Phase 1 | Completed | - | - |
| NCT01332188 | - | - | Completed | - | - |
| NCT00779116 | Allergies | Phase 4 | Completed | - | - |
| NCT00649857 | Healthy | Phase 1 | Completed | - | United States, Texas
... more >>
Novum Pharmaceutical Research Services
Houston, Texas, United States, 77042 Collapse << |
| NCT00779116 | - | - | Completed | - | - |
| NCT00780403 | Allergies | Phase 4 | Completed | - | - |
| NCT00826943 | Allergic Rhinitis | Phase 4 | Completed | - | United States, Tennessee
... more >>
Vanderbilt University Asthma, Sinus, and Allergy Clinic
Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00780403 | - | - | Completed | - | - |
| NCT00794495 | Seasonal Allergic Rhinitis
... more >> Perennial Allergic Rhinitis Collapse << | Phase 4 | Completed | - | - |
| NCT00375713 | Dermatitis
Ec... more >>zema Collapse << | Phase 3 | Completed | - | Korea, Republic of
... more >>
Gyeunggi-do, Korea, Republic of
Kyeonggi-Do, Korea, Republic of
Seoul, Korea, Republic of Collapse << |
| NCT00650065 | Healthy | Phase 1 | Completed | - | United States, Texas
... more >>
Novum Pharmaceutical Research Services
Houston, Texas, United States, 77042 Collapse << |
| NCT00794846 | Seasonal Allergic Rhinitis
... more >> Perennial Allergic Rhinitis Collapse << | Phase 4 | Completed | - | - |
| NCT00639587 | Rhinitis, Allergic, Perennial | Phase 3 | Completed | - | - |
| NCT02132169 | Atopic Disease (Including Alle... more >>rgic Conjunctivitis) Collapse << | Phase 3 | Completed | - | United States, Massachusetts
... more >>
Ora, Inc.
Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00826943 | - | - | Completed | - | - |
| NCT00794599 | Seasonal Allergic Rhinitis
... more >> Perennial Allergic Rhinitis Collapse << | Phase 4 | Completed | - | - |
| NCT01916226 | Rhinitis, Allergic, Perennial ... more >>and Seasonal Collapse << | Phase 4 | Completed | - | - |
| NCT00794378 | Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00794794 | Allergic Rhinitis | Phase 3 | Completed | - | - |
| NCT00776022 | Healthy | Not Applicable | Completed | - | United States, Maryland
... more >>
Bioanalytical Systems, Inc
Baltimore, Maryland, United States, 21201 Collapse << |
| NCT00776139 | Healthy | Not Applicable | Completed | - | United States, Maryland
... more >>
Bioanalytical Systems, Inc.
Baltimore, Maryland, United States, 21201 Collapse << |
| NCT00864279 | Healthy | Phase 1 | Completed | - | United States, North Carolina
... more >>
AAIPharma Inc.- AAI Clinic
Morrisville, North Carolina, United States, 27560 Collapse << |
| NCT00863902 | Healthy | Phase 1 | Completed | - | United States, North Carolina
... more >>
AAIPharma Inc.- AAI Clinic
Morrisville, North Carolina, United States, 27560 Collapse << |
| NCT01781507 | Allergic Rhinitis
... more >> Asthma Collapse << | Phase 4 | Completed | - | United States, California
... more >>
California Allergy and Asthma Medical Group
Los Angeles, California, United States, 90025 Collapse << |
| NCT02132169 | - | - | Completed | - | - |
| NCT03340740 | Allergic Rhinitis
... more >> Asthma Collapse << | Phase 4 | Recruiting | December 31, 2018 | United States, New York
... more >>
Jacobi Medical Center
Recruiting
Bronx, New York, United States, 10461
Contact: Stephen Blumberg, MD 718-918-5826 stephen.blumberg@nychhc.org
Contact: Katherine Huston, MD 718-918-5875 hustonk@nychhc.org Collapse << |
| NCT02932774 | Allergic Rhinitis
... more >> Hay Fever
SAR
Rhinitis, Allergic, Seasonal Collapse << | Phase 4 | Completed | - | - |
| NCT01064102 | Healthy | Phase 1 | Completed | - | - |
| NCT02865018 | Neuromyelitis Optica | Phase 1
Phase 2 | Completed | - | United States, New York
... more >>
Icahn School of Medicine at Mount Sinai
New York, New York, United States, 10029 Collapse << |
| NCT01916226 | - | - | Completed | - | - |
| NCT01064115 | Healthy | Phase 1 | Completed | - | - |
| NCT03772158 | Allergy | Phase 1 | Recruiting | February 3, 2019 | Canada, Quebec
... more >> Algorithme (An altascience Company)
Recruiting
Mount-Royal, Quebec, Canada, H3P 3P1
Contact: Bich-Chau Le +1 (514) 858-6077 ext ext. 3236 Bich-Chau@altasciences.com
Principal Investigator: Eric Sicard, MD Collapse << |
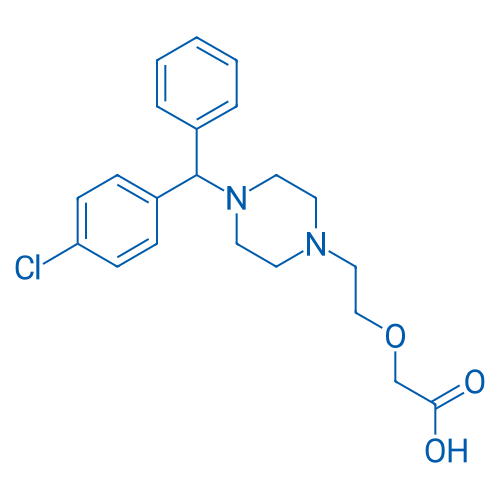

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
