CAS No.: 41432-97-7
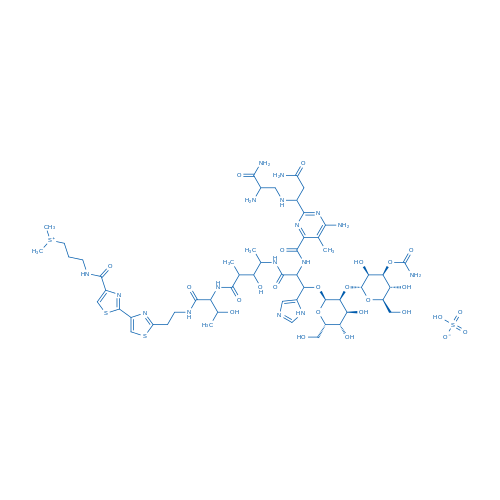
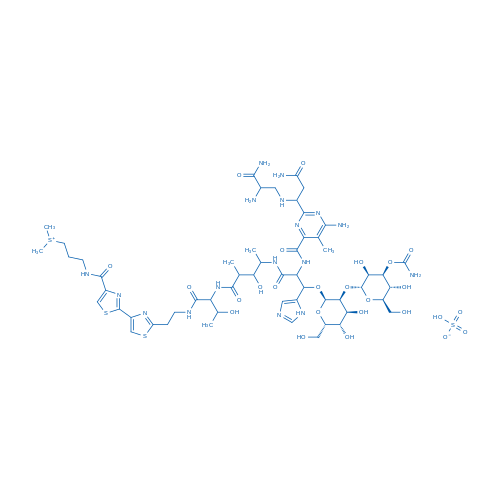
Bleomycin A2/博莱霉素A2 Catalog No. CSN10472
Synonyms: NSC 125066;Bleo;Blexane
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Rat: 1.5 mg/kg[4] (i.p.), 0.35 mg/kg[4] (s.c.); 3.5 mg/kg - 4 mg/kg[5] (intra-tracheal instillation)
给药途径 i.p., s.c., p.o., intra-tracheal instillation
动物药代数据 Animal Mice[3] Dose 0.33 mg/kg Administration i.v. MRT 2.83 ± 0.14 h Vd 2.87 ± 0.08 ml T1/2 3.38 ± 0.18 h AUC0→inf 14.12 ± 0.48 µg·h/ml AUClast 11.34 ± 0.41 µg·h/ml KE 0.205 ± 0.015 h-1 AUMC 32.13 ± 1.09 µg.h2/ml CLtotal 0.587 ± 0.01 ml/h - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01435018 HIV-1 Infection Phase 3 Active, not recruiting March 13, 2020 Brazil ... more >> Instituto de Pesquisa Clinica Evandro Chagas (12101) Rio de Janeiro, Brazil, 21045 Kenya Moi University International Clnical Trials Unit Eldoret, Kenya, 30100 KMRI / Walter Reed Project Clinical Research Center Kericho, Kenya Kenya Medical Research Institute/Center for Disease Control (KEMRI/CDC) CRS (31460) Kisumu, Kenya, 40100 Malawi Univ. of Malawi, John Hopkins Project Blantyre, Malawi South Africa Family Clinical Research Unit (FAM-CUR) CRS (8950) Cape Town, West Cape, South Africa, 7505 Durban Adult HIV CRS (11201) Durban, South Africa, 4013 SF University of Witwatersrand Johannesburg, South Africa Uganda Uganda Cancer Institute ACTG CRS Kampala, Uganda Zimbabwe UZ-Parirenyatwa CRS (30313) Harare, Zimbabwe Collapse << NCT03159897 Hodgkin Lymphoma Phase 3 Recruiting January 2023 - NCT02292979 Hodgkin Lymphoma Phase 2 Active, not recruiting August 2022 - - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 41432-97-7 | 储存条件 |
|
|||||
| 分子式 | C55H85N17O25S4 | 运输 | 蓝冰 | |||||
| 分子量 | 1512.62 | 别名 | NSC 125066;Bleo;Blexane;博莱霉素A2;硫酸博来霉素 | |||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 231-H2N | 0–200 μg/mL | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 22819196 |
| A431 | 50-200 IU | Growth Inhibition Assay | 48 h | inhibits cell growth dose dependently | 25101299 |
| A459 | 10 mU | Apoptosis Assay | 48 h | induces apoptosis | 23902766 |
| A549 | 1 µg/mL | Function Assay | 0-48 h | increases the expression of OPN mRNA | 24963635 |
| A549 | 200 μM | Growth Inhibition Assay | 24 h | decreases cell viability significantly | 23805793 |
| A549 | 200/400 μM | Function Assay | - | results in a dose-responsive increase in ROS | 22773697 |
| A549 | 0-400 μM | Function Assay | 2-48 h | causes more mtDNA than nDNA damage | 22773697 |
| A549 | 100 μM | Function Assay | 0–48 h | causes caspase-3 activation | 22773697 |
| A549 | 100 μM | Function Assay | 0–48 h | causes PKD1 cleavage | 22773697 |
| A549 | 100 μM | Function Assay | 0–48 h | causes mitochondrial localization of Bax | 22773697 |
| A549 | 100 μM | Function Assay | 0–48 h | decreases MMP and ATP levels | 22773697 |
| A-549 | 0/40/80 μg/ml | Function Assay | 3 h | induces a concentration-dependent increase in the MN frequency | 21599612 |
| AY-27 | 50-200 IU | Growth Inhibition Assay | 48 h | inhibits cell growth dose dependently | 25101299 |
| BMG-1 | 40/80 μg/m | Growth Inhibition Assay | 3 h | inhibits cell growth dose dependently | 23906726 |
| C18-4 | 0-100 μM | Growth Inhibition Assay | - | inhibits cell growth dose dependently | 24571982 |
| CHO | 2.5 μg/ml | Function Assay | 18 h/6 d | induces persistence of chromosome damage | 22230195 |
| F98 | 0–1000 μm | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 26088711 |
| hBMSC | 0/40/80 μg/ml | Function Assay | 3 h | induces a concentration-dependent increase in the MN frequency | 21599612 |
| HCT116 | - | Cytotoxicity Assay | 72 h | IC50=11.34% | 25276792 |
| HCT116 | 0-100 μg/ml | Growth Inhibition Assay | 48 h | inhibits cell growth dose dependently | 23518201 |
| HCT116 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | inhibits cell growth dose dependently | 21081487 |
| HCT-116 | 3/7.5/15 μg/ml | Apoptosis Assay | 48 h | reduces the level of BLH at high concentration | 23708668 |
| HCT116 sAPE1+Apn1 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | significantly inhibits cell growth dose dependently | 21081487 |
| HCT116 sLUC+Apn1 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | inhibits cell growth dose dependently | 21081487 |
| HDFn | - | Cytotoxicity Assay | 72 h | IC50=9.31% | 25276792 |
| HeLa | - | Growth Inhibition Assay | - | IC50=10.2 μM | 24732397 |
| HeLa | 3/7.5/15 μg/ml | Apoptosis Assay | 48 h | reduces the level of BLH at high concentration | 23708668 |
| HeLa | 30/70 μg/ml | Function Assay | 24 h | increases the mRNA levels of p53,ATM,p21 and GADD45 | 22487937 |
| HeLa | 30/70 μg/ml | Cytotoxicity Assay | 24 h | induces highly significant levels of fragmented DNA release into the cytoplasm | 22487937 |
| HT1080 | 1 μM | Growth Inhibition Assay | 24 h | leads to a G2/M delay | 25518961 |
| HT1080 | 1 μM | Function Assay | 24 h | induces a potent global DNA damage response | 25518961 |
| HT-29 | - | Cytotoxicity Assay | 72 h | IC50=11.49% | 25276792 |
| IMR-32 | 7.5−120 µg/ml | Growth Inhibition Assay | 12−48 h | IC50=60 µg/ml at the end of 24 hrs of incubation | 25536345 |
| Jurkat | 20 μm | Growth Inhibition Assay | 24 h | arrests the cell cycle at the G2 phase | 24916893 |
| Jurkat | 0-100 μg/ml | Growth Inhibition Assay | 24 h | arrests the cell cycle at the G2 phase | 22223332 |
| Jurkat | 30 μg/ml | Function Assay | 24 h | increases the protein levels of cyclins D3, A and B1 | 22223332 |
| Jurkat | 0-100 μg/ml | Growth Inhibition Assay | 24 h | inhibits cell proliferation in a dose-dependent manner with an IC50 value of 100μg/mL | 22223332 |
| Jurkat | 30 μg/ml | Function Assay | 24 h | increases the effect of pycnidione on phosphorylation levels of histone H2A.X | 22223332 |
| MCF-7 | 0.25 μg/ml. | PDT/PCI Treatment | 1-4 h | shows significant (P < 0.05) cytotoxic effect | 22591284 |
| MCF-7 | 0-2 μg/ml. | Growth Inhibition Assay | 24/48 h | EC50=1.2 μg/mL | 22591284 |
| MCF-7 | 30/70 μg/ml | Function Assay | 24 h | increases the mRNA levels of p53,ATM,p21 and GADD45 | 22487937 |
| MCF-7 | 30/70 μg/ml | Cytotoxicity Assay | 24 h | induces highly significant levels of fragmented DNA release into the cytoplasm | 22487937 |
| MCF-7 | 0-400 μM | Growth Inhibition Assay | 72 h | LC50=151.8 μM | 21703291 |
| MCF-7/Adr | 0-400 μM | Growth Inhibition Assay | 72 h | LC50=58.5 μM | 21703291 |
| MCF-7/Her-18 | 0.42-1700 μg/mL | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 22621404 |
| MDA-MB-231 | 0.25 μg/ml. | PDT/PCI Treatment | 1-4 h | shows significant (P < 0.05) cytotoxic effect | 22591284 |
| MDA-MB-235 | 0.25 μg/ml. | PDT/PCI Treatment | 1-4 h | shows significant (P < 0.05) cytotoxic effect | 22591284 |
| MDA-MB-468 | 0–200 μg/mL | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 22819196 |
| MLE12 | 1 µg/mL | Function Assay | 0-48 h | increases the expression of OPN mRNA | 24963635 |
| MMP1 | 10 mU | Apoptosis Assay | 48 h | induces apoptosis | 23902766 |
| MOCK | 10 mU | Apoptosis Assay | 48 h | induces apoptosis | 23902766 |
| NCCIT | 120 μg/ml | Function Assay | 72 h | significantly increases 8-isoprostane levels | 22825355 |
| NCCIT | 120 μg/ml | Function Assay | 72 h | significantly increases TBARS levels | 22825355 |
| NCCIT | 120 μg/ml | Function Assay | 72 h | significantly decreases GSH levels | 22825355 |
| NCCIT | 120 μg/ml | Function Assay | 72 h | significantly increases Protein carbonyl levels | 22825355 |
| NCCIT | 120 μg/ml | Function Assay | 72 h | significantly increases LPO levels | 22825355 |
| NCCIT | 20–140 μg/ml | Growth Inhibition Assay | 72 h | LD50=120 μg/ml | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly increases caspase-3 activity | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly increases caspase-8 activity | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly increases caspase-9 activity | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly increases BAX level | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly decreases the Bcl-2 content | 22562160 |
| NCCIT | - | Function Assay | 72 h | significantly decreases the Cyt-c level | 22562160 |
| NCI-H23 | 0/40/80 μg/ml | Function Assay | 3 h | induces a concentration-dependent increase in the MN frequency | 21599612 |
| NCM460 | 0-100 μg/ml | Growth Inhibition Assay | 48 h | inhibits cell growth dose dependently | 23518201 |
| NT2 | - | Growth Inhibition Assay | 24 h | LD50=400 µg/ml | 23386420 |
| NT2 | - | Growth Inhibition Assay | 48 h | LD50=100 µg/ml | 23386420 |
| NT2 | - | Growth Inhibition Assay | 72 h | LD50=20 µg/ml | 23386420 |
| NT2 | 400 µg/ml | Apoptosis Assay | 24 h | induces significant increases in caspase-3,8,9 activity | 23386420 |
| NT2 | 100-600 μg/ml | Growth Inhibition Assay | 24 h | IC50=400 µg/ml | 22469952 |
| NT2 | 400 μg/ml | Function Assay | 24 h | significantly increases caspase-3,8,9 activity | 22469952 |
| NT2 | 400 μg/ml | Function Assay | 24 h | significantly increases BAX level | 22469952 |
| NT2 | 400 μg/ml | Function Assay | 24 h | significantly decreases Bcl-2 level | 22469952 |
| NTera-2 | 120 μg/ml | Function Assay | 72 h | significantly increases 8-isoprostane levels | 22825355 |
| NTera-2 | 120 μg/ml | Function Assay | 72 h | significantly increases TBARS levels | 22825355 |
| NTera-2 | 120 μg/ml | Function Assay | 72 h | significantly increases Protein carbonyl levels | 22825355 |
| NTera-2 | 120 μg/ml | Function Assay | 72 h | significantly increases LPO levels | 22825355 |
| PBL | 0/40/80 μg/ml | Function Assay | 3 h | induces a concentration-dependent increase in the MN frequency | 21599612 |
| PMCs | 0.1–2 μg/ml | Function Assay | 24 h | causes a dose-dependent increase in the protein level of collagen-I | 25595642 |
| PMCs | 0.2 μg/ml | Function Assay | 24–72 h | induces increases in vimentin and α-SMA protein | 25595642 |
| PMCs | 0.2 μg/ml | Function Assay | 72 h | induces decreases in cytokeratin-8 and E-cadherin protein | 25595642 |
| PMCs | 0.2 μg/ml | Function Assay | 12/24/48 h | increases cell migration | 25595642 |
| pol β −/− | 0.25-200 μg/ml | Growth Inhibition Assay | 24 h | decreases cell viability with increasing concentrations | 21251944 |
| pol β −/− | 0-2.5 μg/ml | Function Assay | 2 h | causes DNA damage | 21251944 |
| pol β −/− | 0-5 μg/ml | Function Assay | 48 h | causes micronucleus dose and time dependently | 21251944 |
| pol β +/+ | 0.25-200 μg/ml | Growth Inhibition Assay | 24 h | decreases cell viability with increasing concentrations | 21251944 |
| pol β +/+ | 0-2.5 μg/ml | Function Assay | 2 h | causes DNA damage | 21251944 |
| pol β +/+ | 0-5 μg/ml | Function Assay | 48 h | causes micronucleus dose and time dependently | 21251944 |
| POLK CD | - | Growth Inhibition Assay | - | IC50=5.59 ± 1.00 μM | 26031400 |
| POLK KO | - | Growth Inhibition Assay | - | IC50=4.03 ± 1.92 μM | 26031400 |
| POLK WT | - | Growth Inhibition Assay | - | IC50=11.7 ± 1.7 μM | 26031400 |
| RLE/Abca3 | 0.4 μM | Function Assay | 144 h | induces a morphological change from epithelial-like morphology to a wide, spread-out morphology | 25760538 |
| T24 | 50-200 IU | Growth Inhibition Assay | 48 h | inhibits cell growth dose dependently | 25101299 |
| THP-1 | - | Cytotoxicity Assay | 72 h | IC50=4.77% | 25276792 |
| TK6 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | inhibits cell growth dose dependently | 21081487 |
| TK6 sAPE1+Apn1 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | significantly inhibits cell growth dose dependently | 21081487 |
| TK6 sLUC+Apn1 | 25-150 μg/ml | Growth Inhibition Assay | 96 h | inhibits cell growth dose dependently | 21081487 |
| U2OS EGFPnls | 0-2 μg/ml | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 21811007 |
| U2OS KuEnls | 0-2 μg/ml | Growth Inhibition Assay | 24 h | inhibits cell growth dose dependently | 21811007 |
| WI-38 | 0/40/80 μg/ml | Function Assay | 3 h | induces a concentration-dependent increase in the MN frequency | 21599612 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01435018 | HIV-1 Infection | Phase 3 | Active, not recruiting | March 13, 2020 | Brazil ... more >> Instituto de Pesquisa Clinica Evandro Chagas (12101) Rio de Janeiro, Brazil, 21045 Kenya Moi University International Clnical Trials Unit Eldoret, Kenya, 30100 KMRI / Walter Reed Project Clinical Research Center Kericho, Kenya Kenya Medical Research Institute/Center for Disease Control (KEMRI/CDC) CRS (31460) Kisumu, Kenya, 40100 Malawi Univ. of Malawi, John Hopkins Project Blantyre, Malawi South Africa Family Clinical Research Unit (FAM-CUR) CRS (8950) Cape Town, West Cape, South Africa, 7505 Durban Adult HIV CRS (11201) Durban, South Africa, 4013 SF University of Witwatersrand Johannesburg, South Africa Uganda Uganda Cancer Institute ACTG CRS Kampala, Uganda Zimbabwe UZ-Parirenyatwa CRS (30313) Harare, Zimbabwe Collapse << |
| NCT03159897 | Hodgkin Lymphoma | Phase 3 | Recruiting | January 2023 | - |
| NCT02292979 | Hodgkin Lymphoma | Phase 2 | Active, not recruiting | August 2022 | - |
| NCT00000658 | Lymphoma, Non-Hodgkin ... more >> HIV Infections Collapse << | Phase 3 | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States, 900331079 UCLA CARE Center CRS Los Angeles, California, United States, 90095 Ucsf Aids Crs San Francisco, California, United States, 941102859 United States, Colorado University of Colorado Hospital CRS Aurora, Colorado, United States, 80262 United States, Illinois Northwestern University CRS Chicago, Illinois, United States, 60611 Rush Univ. Med. Ctr. ACTG CRS Chicago, Illinois, United States, 60612 United States, Indiana Indiana Univ. School of Medicine, Infectious Disease Research Clinic Indianapolis, Indiana, United States, 462025250 United States, Maryland Johns Hopkins Adult AIDS CRS Baltimore, Maryland, United States United States, Massachusetts Beth Israel Deaconess Med. Ctr., ACTG CRS Boston, Massachusetts, United States, 02118 Beth Israel Deaconess - East Campus A0102 CRS Boston, Massachusetts, United States, 02215 Bmc Actg Crs Boston, Massachusetts, United States United States, Missouri Washington U CRS St. Louis, Missouri, United States United States, New York SUNY - Buffalo, Erie County Medical Ctr. Buffalo, New York, United States, 14215 Beth Israel Med. Ctr. (Mt. Sinai) New York, New York, United States, 02215 NY Univ. HIV/AIDS CRS New York, New York, United States, 10016 Memorial Sloan-Kettering Cancer Ctr. New York, New York, United States, 10021 Univ. of Rochester ACTG CRS Rochester, New York, United States, 14642 United States, North Carolina Unc Aids Crs Chapel Hill, North Carolina, United States, 275997215 United States, Ohio Case CRS Cleveland, Ohio, United States The Ohio State Univ. AIDS CRS Columbus, Ohio, United States, 432101228 United States, Pennsylvania Hosp. of the Univ. of Pennsylvania CRS Philadelphia, Pennsylvania, United States, 19104 Pitt CRS Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01578967 | Hodgkin Lymphoma, Adult | Not Applicable | Active, not recruiting | August 10, 2021 | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, North Carolina University of North Carolina at Chapel Hill, Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 Levine Cancer Istitute, Carolinas Health Care system Charlotte, North Carolina, United States, 28204 Rex Cancer Center Raleigh, North Carolina, United States, 27607 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37240 Collapse << |
| NCT01578967 | - | - | Active, not recruiting | - | - |
| NCT00000689 | Lymphoma, Non-Hodgkin ... more >> HIV Infections Collapse << | Phase 1 | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States, 90033 Collapse << |
| NCT02782754 | Intracranial Germinoma | Phase 2 | Unknown | - | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Ki Woong Sung 82-2-3410-3529 kwsped@skku.edu Principal Investigator: Ki Woong Sung Collapse << |
| NCT01026220 | Childhood Nodular Lymphocyte P... more >>redominant Hodgkin Lymphoma Stage III Childhood Hodgkin Lymphoma Stage IV Childhood Hodgkin Lymphoma Collapse << | Phase 3 | Completed | - | - |
| NCT00352027 | - | - | Active, not recruiting | - | - |
| NCT00104676 | Extragonadal Germ Cell Tumor ... more >> Teratoma Testicular Germ Cell Tumor Collapse << | Phase 3 | Unknown | - | United States, Texas ... more >> M. D. Anderson Cancer Center at University of Texas Recruiting Houston, Texas, United States, 77030-4009 Contact: Clinical Trials Office - M. D. Anderson Cancer Center at the U 713-792-3245 France Centre Paul Papin Recruiting Angers, France, 49100 Contact: Remy Delva 33-49-800-918-507 Institut Bergonie Recruiting Bordeaux, France, 33076 Contact: Nguyen Binh Bui, MD 33-5-5633-3813 C.H.U. de Brest Recruiting Brest, France, 29609 Contact: Jean-Pierre Malhaire, MD 33-298-223-333 ext. 233-95 Centre Regional Francois Baclesse Recruiting Caen, France, 14076 Contact: Emmanuel Sevin, MD 33-2-3145-5000 e.sevin@baclesse.fr CHU de Grenoble - Hopital de la Tronche Recruiting Grenoble, France, 38043 Contact: Francois Ringeisen 33-4-7676-5537 Centre Oscar Lambret Recruiting Lille, France, 59020 Contact: Armelle Caty, MD 33-32-029-5959 acaty@o-lambret.fr Centre Leon Berard Recruiting Lyon, France, 69008 Contact: Aude Flechon 33-4-78-78-26-45 Marseille Institute of Cancer - Institut J. Paoli and I. Calmettes Recruiting Marseille, France, 13273 Contact: Gwenaelle Gravis, MD 33-4-9122-3740 gravisg@marseille.fnclcc.fr Hopital Notre-Dame de Bon Secours Recruiting Metz, France, 57038 Contact: Christian Platini, MD 33-3-8755-3554 cplatini@chr-metz-thionville.fr Centre Regional de Lutte Contre le Cancer - Centre Val d'Aurelle Recruiting Montpellier, France, 34298 Contact: Stephane Culine, MD 33-4-6761-3755 stculine@valdorel.fnclcc.fr CRLCC Nantes - Atlantique Recruiting Nantes-Saint Herblain, France, 44805 Contact: Frederic Rolland, MD 33-2-40-67-99-76 F-rolland@nantes.fnclcc.fr Centre Antoine Lacassagne Recruiting Nice, France, 06189 Contact: Antoine Thyss, MD 33-04-9203-1538 antoine.thyss@cal.nice.fnclcc.fr Hopital Europeen Georges Pompidou Recruiting Paris, France, 75015 Contact: Stephane Oudard, MD, PhD 33-1-56-093-476 stephane.oudard@hop.egp.ap-hop-paris.fr Hopital Tenon Recruiting Paris, France, 75970 Contact: Jean-Pierre Lotz, MD 33-1-5601-6058 jean-pierre.lotz@tnn.ap-hop-paris.fr Institut Jean Godinot Recruiting Reims, France, 51056 Contact: Jean-Christophe Eymard, MD 33-03-2650-4444 jc.eymard@reims.fnclcc.fr Centre Eugene Marquis Recruiting Rennes, France, 35042 Contact: Pierre Kerbrat, MD, PhD 33-299-253-280 kerbrat@rennes.fnclcc.fr Centre Hospitalier de Rodez Recruiting Rodez, France, 12027 Contact: Laurent Mosser 33-05-6575-1212 Centre Henri Becquerel Recruiting Rouen, France, 76038 Contact: Paule Chinet-Charrot 33-2-3208-2222 Institut Claudius Regaud Recruiting Toulouse, France, 31052 Contact: Christine Chevreau-Dalbianco, MD 33-56-142-4114 chevreau.christine@claudiusregaud.fr Centre Hospitalier Universitaire Bretonneau de Tours Recruiting Tours, France, 37044 Contact: Claude Linassier, MD, PhD 33-2-4747-3827 linassier@med.univ-tours.fr Centre Alexis Vautrin Recruiting Vandoeuvre-les-Nancy, France, 54511 Contact: Lionnel Geoffrois, MD 33-3-8359-8400 Institut Gustave Roussy Recruiting Villejuif, France, F-94805 Contact: Karim Fizazi, MD, PhD 33-1-4211-4559 fizazi@igr.fr Slovakia National Cancer Institute - Bratislava Recruiting Bratislava, Slovakia, 833 10 Contact: Maria Reckova, MD 421-2-5937-8366 maria.reckova@nou.sk Collapse << |
| NCT00352027 | Hodgkin's Lymphoma | Phase 2 | Active, not recruiting | November 2022 | United States, California ... more >> Stanford University Medical Center Palo Alto, California, United States, 94304 Rady Children's Hospital San Diego San Diego, California, United States, 92123 United States, Maine Maine Children's Medical Center Portland, Maine, United States, 04102-3175 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Tennessee St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT00736320 | Hodgkin Lymphoma | Phase 3 | Active, not recruiting | May 2020 | Germany ... more >> 1st Dept. of Medicine, Cologne University Hospital Cologne, Germany Collapse << |
| NCT00005867 | Lymphoma | Phase 3 | Completed | - | United Kingdom ... more >> Stoke Mandeville Hospital Aylesbury-Buckinghamshire, England, United Kingdom, HP21 8AL Horton Hospital Banbury, England, United Kingdom, OX16 9AL Basildon University Hospital Basildon, England, United Kingdom, SS16 5NL Birmingham Heartlands Hospital Birmingham, England, United Kingdom, B9 5SS Bradford Hospitals NHS Trust Bradford, England, United Kingdom, BD9 6RJ Bristol Haematology and Oncology Centre Bristol, England, United Kingdom, BS2 8ED Addenbrooke's Hospital at Cambridge University Hospitals NHS Foundation Trust Cambridge, England, United Kingdom, CB2 2QQ Cheltenham General Hospital Cheltenham, England, United Kingdom, GL53 7AN Countess of Chester Hospital NHS Foundation Trust Chester, England, United Kingdom, CH2 1UL Saint Richards Hospital Chichester, England, United Kingdom, P019 4SE Essex County Hospital Colchester, England, United Kingdom, C03 3NB Walsgrave Hospital Coventry, England, United Kingdom, CV2 2DX Russells Hall Hospital Dudley, England, United Kingdom, DY1 2HQ Chase Farm Hospital Enfield, England, United Kingdom, EN 28 JL Medway Maritime Hospital Gillingham Kent, England, United Kingdom, ME7 5NY Hull Royal Infirmary Hull, England, United Kingdom, HU3 2KZ Hinchingbrooke Hospital Huntingdon, England, United Kingdom, PE18 6NT Queen Elizabeth Hospital King's Lynn, England, United Kingdom, PE30 4ET Leicester Royal Infirmary Leicester, England, United Kingdom, LE1 5WW Royal Liverpool and Broadgreen Hospitals NHS Trust Liverpool, England, United Kingdom, L7 8XP Aintree University Hospital Liverpool, England, United Kingdom, L9 7AL Saint Bartholomew's Hospital London, England, United Kingdom, EC1A 7BE St. Thomas' Hospital London, England, United Kingdom, SE1 7EH St. George's Hospital London, England, United Kingdom, SW17 0QT Middlesex Hospital London, England, United Kingdom, WC1E 6HX Clatterbridge Centre for Oncology NHS Trust Merseyside, England, United Kingdom, CH63 4JY Norfolk and Norwich University Hospital Norwich, England, United Kingdom, NR4 7UY Nottingham City Hospital NHS Trust Nottingham, England, United Kingdom, NG5 1PB Oxford Radcliffe Hospital Oxford, England, United Kingdom, 0X3 9DU Pontefract General Infirmary Pontefract West Yorkshire, England, United Kingdom, WF8 1PL Oldchurch Hospital Romford, England, United Kingdom, RM7 OBE Scunthorpe General Hospital Scunthorpe, England, United Kingdom, DN15 7BH Cancer Research Centre at Weston Park Hospital Sheffield, England, United Kingdom, S1O 2SJ Southampton University Hospital NHS Trust Southampton, England, United Kingdom, SO16 6YD University Hospital of North Staffordshire Stoke-On-Trent Staffs, England, United Kingdom, ST4 6QG East Surrey Hospital Surrey, England, United Kingdom, RH1 5RH Royal Marsden NHS Foundation Trust - Surrey Sutton, England, United Kingdom, SM2 5PT Sandwell General Hospital West Bromwich, England, United Kingdom, B71 4HJ Cancer Care Centre at York Hospital York, England, United Kingdom, Y031 8HE Centre for Cancer Research and Cell Biology at Belfast City Hospital Belfast, Northern Ireland, United Kingdom, BT9 7AB Aberdeen Royal Infirmary Aberdeen, Scotland, United Kingdom, AB25 2ZN Pinderfields Hospital NHS Trust Wakefield, Scotland, United Kingdom, WF1 4DG Ysbyty Gwynedd Bangor, Wales, United Kingdom, LL57 2PW University Hospital of Wales Cardiff, Wales, United Kingdom, CF14 4XN Glan Clywd District General Hospital Rhyl, Denbighshire, Wales, United Kingdom, LL 18 5UJ Mount Vernon Cancer Centre at Mount Vernon Hospital Rhyl, Denbighshire, Wales, United Kingdom, LL 18 5UJ Collapse << |
| NCT00038558 | Hodgkin Disease | Phase 3 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00225173 | Hodgkin Disease | Phase 2 | Terminated | - | United States, California ... more >> Stanford University Medical Center Stanford, California, United States, 94305 Collapse << |
| NCT01026220 | - | - | Completed | - | - |
| NCT00429754 | Nausea Vomiti... more >>ng Collapse << | Phase 4 | Withdrawn(change of study popu... more >>lation and chemotherapeutic regimen) Collapse << | - | - |
| NCT02758717 | Ann Arbor Stage IB Hodgkin Lym... more >>phoma Ann Arbor Stage II Hodgkin Lymphoma Ann Arbor Stage IIA Hodgkin Lymphoma Ann Arbor Stage IIB Hodgkin Lymphoma Ann Arbor Stage III Hodgkin Lymphoma Ann Arbor Stage IIIA Hodgkin Lymphoma Ann Arbor Stage IIIB Hodgkin Lymphoma Ann Arbor Stage IV Hodgkin Lymphoma Ann Arbor Stage IVA Hodgkin Lymphoma Ann Arbor Stage IVB Hodgkin Lymphoma Classic Hodgkin Lymphoma Collapse << | Phase 2 | Recruiting | May 13, 2024 | United States, California ... more >> Stanford Cancer Institute Palo Alto Recruiting Palo Alto, California, United States, 94304 Contact: Ann Moffat 650-721-4096 amoffat@stanford.edu Principal Investigator: Ranjana H. Advani United States, District of Columbia MedStar Georgetown University Hospital Recruiting Washington, District of Columbia, United States, 20007 Contact: Pari Ramzi 202-784-0038 ramzip1@georgetown.edu Principal Investigator: Bruce D. Cheson United States, Illinois University of Chicago Comprehensive Cancer Center Withdrawn Chicago, Illinois, United States, 60637 United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Mayo Clinic Clinical Trials Office 507-538-7623 clinicaltrials@mayo.edu Principal Investigator: Stephen M. Ansell United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Kelly Hoye 314-362-4206 khoye@wustl.edu Principal Investigator: Nancy L. Bartlett United States, New Jersey Hackensack University Medical Center Recruiting Hackensack, New Jersey, United States, 07601 Contact: Kara M. Yannotti 551-996-5900 Kara.Yannotti@HackensackMeridian.org Principal Investigator: Tatyana A. Feldman United States, New York Laura and Isaac Perlmutter Cancer Center at NYU Langone Recruiting New York, New York, United States, 10016 Contact: Mohammad Maher Abdul-Hay 212-731-5670 Maher.Abdulhay@nyumc.org Principal Investigator: Catherine S. Diefenbach United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Rashmeet Reen 614-688-9283 rashmeet.reen@osumc.edu Principal Investigator: Kristie A. Blum United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Emily Wesson 713-794-5656 etwesson@mdanderson.org Principal Investigator: Michelle A. Fanale Collapse << |
| NCT00848289 | - | - | Recruiting | August 2019 | United States, Texas ... more >> Baylor College of Medicine Terminated Houston, Texas, United States, 77030 MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Xifeng Wu, MD 713-792-3020 xwu@mdanderson.org Collapse << |
| NCT00000626 | HIV Infections ... more >> Hodgkin's Disease Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama Therapeutics CRS Birmingham, Alabama, United States, 35294 United States, California USC CRS Los Angeles, California, United States, 900331079 United States, Illinois Northwestern University CRS Chicago, Illinois, United States, 60611 United States, Indiana Indiana Univ. School of Medicine, Infectious Disease Research Clinic Indianapolis, Indiana, United States, 462025250 United States, Missouri St. Louis ConnectCare, Infectious Diseases Clinic St Louis, Missouri, United States, 63112 Washington U CRS St. Louis, Missouri, United States United States, New York SUNY - Buffalo, Erie County Medical Ctr. Buffalo, New York, United States United States, Ohio The Ohio State Univ. AIDS CRS Columbus, Ohio, United States, 432101228 Collapse << |
| NCT01468740 | HIV-associated Hodgkin Lymphom... more >>a Collapse << | Phase 2 | Unknown | July 2012 | Germany ... more >> Vivantes Auguste Victoria Klinikum Recruiting Berlin, Germany, 12157 Contact: Markus Müller, MD 0049 30 130 20 2321 Markus.Mueller2@vivantes.de Contact: Marcel Berger, MD 0049 30 130 20 2321 Marcel.Berger@vivantes.de Sub-Investigator: Markus Müller, MD Sub-Investigator: Marcel Berger, MD Principal Investigator: Keikawus Arasteh, MD Ärzteforum Seestrasse Recruiting Berlin, Germany, 13347 Contact: Jan Siehl, MD 0049 30 455095-0 jan.siehl@aerzteforum-seestrasse.de Principal Investigator: Jan Siehl, MD Universiy of Bonn Recruiting Bonn, Germany, 53127 Contact: Juergen Rockstroh, MD 0049 228 287 16558 Rockstroh@uni-bonn.de; Principal Investigator: Jürgen Rockstroh, MD University of Cologne Recruiting Cologne, Germany, 50924 Contact: Christoph Wyen, MD 0049 221 478 88835 christoph.wyen@uk-koeln.de Contact: Gerd Fätkenheuer, MD 0049 221 478 4886 g.faetkenheuer@uni-koeln.de Sub-Investigator: Christoph Wyen, MD Principal Investigator: Gerd Fätkenheuer, MD University of Frankfurt Recruiting Frankfurt, Germany, 60590 Contact: Timo Wolf, MD 0049 69 6301 5452 Timo.Wolf@kgu.de Principal Investigator: Timo Wolf, MD Asklepios Klinikum St. Georg Recruiting Hamburg, Germany, 20099 Contact: Maike Nickelsen, MD 0049 40 18 18 85 20 05 m.nickelsen@asklepios.com Principal Investigator: Maike Nickelsen, MD Infektionsmedizinisches Zentrum Hamburg Recruiting Hamburg, Germany, 20146 Contact: Christian Hoffmann, MD 0049 40 4132420 hoffmann@ich-hamburg.de; Principal Investigator: Christian Hoffmann, MD Harlaching Hospital Recruiting Munich, Germany, 81545 Contact: Marcus Hentrich, MD 0049 89 6210 2663 or 2731 marcus.hentrich@klinikum-muenchen.de Principal Investigator: Marcus Hentrich, MD Collapse << |
| NCT00301782 | Extragonadal Germ Cell Tumor ... more >> Teratoma Testicular Germ Cell Tumor Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> Queen Elizabeth Hospital at University Hospital of Birmingham NHS Trust Birmingham, England, United Kingdom, B15 2TH Bristol Haematology and Oncology Centre Bristol, England, United Kingdom, BS2 8ED Addenbrooke's Hospital Cambridge, England, United Kingdom, CB2 2QQ Gloucestershire Oncology Centre at Cheltenham General Hospital Cheltenham, England, United Kingdom, GL53 7AN Walsgrave Hospital Coventry, England, United Kingdom, CV2 2DX Royal Devon and Exeter Hospital Exeter, England, United Kingdom, EX2 5DW Leeds Cancer Centre at St. James's University Hospital Leeds, England, United Kingdom, LS9 7TF Leicester Royal Infirmary Leicester, England, United Kingdom, LE1 5WW Saint Bartholomew's Hospital London, England, United Kingdom, EC1A 7BE University College of London Hospitals London, England, United Kingdom, WIT 3AA Christie Hospital Manchester, England, United Kingdom, M20 4BX Clatterbridge Centre for Oncology Merseyside, England, United Kingdom, CH63 4JY Nottingham City Hospital Nottingham, England, United Kingdom, NG5 1PB Rosemere Cancer Centre at Royal Preston Hospital Preston, England, United Kingdom, PR2 9HT Berkshire Cancer Centre at Royal Berkshire Hospital Reading, England, United Kingdom, RG1 5AN Southampton General Hospital Southampton, England, United Kingdom, SO16 6YD Royal Marsden - Surrey Sutton, England, United Kingdom, SM2 5PT Beatson West of Scotland Cancer Centre Glasgow, Scotland, United Kingdom, G11 6NT Raigmore Hospital Inverness, Scotland, United Kingdom, 1V2 3UJ Velindre Cancer Center at Velindre Hospital Cardiff, Wales, United Kingdom, CF14 2TL Collapse << |
| NCT01251107 | Hodgkin Lymphoma | Phase 3 | Completed | - | Italy ... more >> Fondazione IRCCS Istituto Nazionale di Tumori di Milano Milano, Italy Collapse << |
| NCT00901303 | Hodgkin's Lymphoma | Not Applicable | Terminated(The study was termi... more >>nated due to lack of accrual.) Collapse << | - | United States, New York ... more >> Weill Cornell Medical College New York, New York, United States, 10021 Collapse << |
| NCT00005578 | Cardiac Toxicity ... more >> Lymphoma Collapse << | Phase 3 | Completed | - | - |
| NCT01118026 | Lymphoma | Phase 2 | Active, not recruiting | - | - |
| NCT01390584 | Lymphoma | Phase 2 | Terminated(slow accrual) | - | United States, California ... more >> Stanford Cancer Center Stanford, California, United States, 94305-5824 United States, Pennsylvania McGlinn Family Regional Cancer Center at Reading Hospital and Medical Center Reading, Pennsylvania, United States, 19612-6052 Collapse << |
| NCT00901303 | - | - | Terminated(The study was termi... more >>nated due to lack of accrual.) Collapse << | - | - |
| NCT00369681 | Lymphoma | Phase 2 | Completed | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010-3000 United States, Iowa Holden Comprehensive Cancer Center at University of Iowa Iowa City, Iowa, United States, 52242-1009 United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States, 21231-2410 United States, Minnesota Mayo Clinic Cancer Center Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00369681 | - | - | Completed | - | - |
| NCT00002827 | Cardiac Toxicity ... more >> Lymphoma Collapse << | Phase 3 | Completed | - | - |
| NCT00135499 | Lymphoma, Large-Cell, Diffuse | Phase 3 | Terminated(Recruitment too low... more >>) Collapse << | - | Belgium ... more >> Groupe d'Etude des Lymphomes de l'adulte Yvoir, Belgium France Hôpital Henri Mondor Créteil, France, 94010 Hôpital Saint Louis Paris, France, 75010 Service d'Hématologie - Centre Hospitalier Lyon-Sud Pierre-Bénite cedex, France, 69495 Centre Hospitalier Robert Debré Reims, France, 51092 Centre Henri Becquerel Rouen, France, 76038 Institut Gustave Roussy Villejuif, France Switzerland Schweirische Arbeitsgruppe fur klinische Krebsforschung Lausanne, Switzerland Collapse << |
| NCT03142061 | Advanced Inoperable Vulva Carc... more >>inoma Collapse << | Not Applicable | Recruiting | December 31, 2019 | Germany ... more >> Department of Women's Health Recruiting Tübingen, Germany, 72076 Contact: Harald Seeger, Prof. Dr. Collapse << |
| NCT00822120 | Lymphoma Nonn... more >>eoplastic Condition Collapse << | Phase 2 | Active, not recruiting | April 2020 | - |
| NCT00053352 | Childhood Embryonal Tumor ... more >> Childhood Extracranial Germ Cell Tumor Childhood Extragonadal Germ Cell Tumor Childhood Malignant Ovarian Germ Cell Tumor Childhood Malignant Testicular Germ Cell Tumor Childhood Teratoma Ovarian Embryonal Carcinoma Ovarian Yolk Sac Tumor Stage II Malignant Testicular Germ Cell Tumor Stage IIA Ovarian Germ Cell Tumor Stage IIB Ovarian Germ Cell Tumor Stage IIC Ovarian Germ Cell Tumor Stage III Malignant Testicular Germ Cell Tumor Stage IIIA Ovarian Germ Cell Tumor Stage IIIB Ovarian Germ Cell Tumor Stage IIIC Ovarian Germ Cell Tumor Testicular Choriocarcinoma and Yolk Sac Tumor Testicular Embryonal Carcinoma Collapse << | Phase 3 | Completed | - | - |
| NCT03755804 | Hodgkin Lymphoma | Phase 2 | Recruiting | July 1, 2028 | United States, Tennessee ... more >> St. Jude Children's Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Jamie Flerlage, MD, MS 866-278-5833 referralinfo@stjude.org Collapse << |
| NCT00822120 | - | - | Active, not recruiting | - | - |
| NCT02298283 | Hodgkin Lymphoma | Phase 2 | Recruiting | February 2021 | France ... more >> CH Victor Dupouy Recruiting Argenteuil, France, 95100 Contact: Driss CHAOUI, MD driss.chaoui@ch-argenteuil.fr Principal Investigator: Driss CHAOUI, MD Polyclinique Bordeaux Nord Recruiting Bordeaux, France, 33300 Contact: Olivier FITOUSSI, MD o.fitoussi@bordeauxnord.com Contact: Olivier Principal Investigator: FITOUSSI, MD Centre François Baclesse Recruiting Caen, France, 14076 Contact: Christophe FRUCHART, MD c.fruchart@baclesse.unicancer.fr Principal Investigator: Christophe FRUCHART, MD CH de Chambéry Recruiting Chambéry, France, 73011 Contact: Gian Mathéo PICA, MD gian-matteo.pica@ch-chambery.fr Principal Investigator: Gian Mathéo PICA, MD CH Sud Francilien Recruiting Corbeil-Essonnes, France, 91108 Contact: Bertrand JOLY, MD bertrand.joly@ch-sud-francilien.fr Principal Investigator: Bertrand JOLY, MD Hôpital Henri Mondor Recruiting Creteil, France, 94010 Contact: Corinne HAIOUN, Prof corinne.haioun@hmn.aphp.fr Principal Investigator: Corinne HAIOUN, Prof CHU de Dijon - Hôpital le Bocage Recruiting Dijon, France, 21034 Contact: Olivier CASASNOVAS, MD olivier.casasnovas@chu-dijon.fr Principal Investigator: Olivier CASASNOVAS, MD Hôpital André Mignot Recruiting Le Chesnay, France, 78157 Contact: Hassan FARHAT, MD hfarhat@ch-versailles.fr Principal Investigator: Hassan FARHAT, MD Clinique Victor Hugo Recruiting Le Mans, France, 72000 Contact: Katell LE DÛ, MD k.ledu@cjb72.org Principal Investigator: Katell LE DÛ, MD CHRU Lille - Hôpital Claude Huriez Recruiting Lille, France, 59037 Contact: Franck MORSCHHAUSER, Prof franck.morschhauser@chru-lille.fr Principal Investigator: Franck MORSCHHAUSER, Prof CHU de Limoges Recruiting Limoges, France, 87042 Contact: Mohamed TOUATI, MD mohamed.touati@chu-limoges.fr Principal Investigator: Mohamed TOUATI, MD Centre Léon Bérard Recruiting Lyon, France, 69008 Contact: Emmanuelle NICOLAS- VIRELIZIER, MD emmanuelle.nicolas@lyon.unicancer.fr Principal Investigator: Emmanuelle NICOLAS- VIRELIZIER, MD Institut Paoli Calmette Recruiting Marseille, France, 13273 Contact: Réda BOUABDALLAH, MD bouabdallahr@ipc.unicancer.fr Principal Investigator: Réda BOUABDALLAH, MD Hôpital de la Conception Recruiting Marseille, France, 13385 Contact: Régis COSTELLO, Prof regis.costello@free.fr Principal Investigator: Régis COSTELLO, Prof CHU Montpellier - Saint ELOI Recruiting Montpellier, France, 34295 Contact: Philippe QUITTET, MD p-quittet@chu-montpellier.fr Principal Investigator: Philippe QUITTET, MD CHU de Nantes Recruiting Nantes, France, 44093 Contact: Thomas GASTINNE, MD thomas.gastinne@chu-nantes.fr Principal Investigator: Thomas GASTINNE, MD Hôpital Saint Louis Recruiting Paris cedex 10, France, 75475 Contact: Pauline BRICE, MD pauline.brice@sls.aphp.fr Principal Investigator: Pauline BRICE, MD Hôpital Cochin Recruiting Paris, France, 75004 Contact: Bénédicte DEAU-FISCHER, MD benedicte.deau-fischer@cch.aphp.fr Principal Investigator: Bénédicte DEAU-FISCHER, MD Hôpital de la Pitié Salpétrière Recruiting Paris, France, 75651 Contact: Jean GABARRE, MD jean.gabarre@psl.aphp.fr Principal Investigator: Jean GABARRE, MD CH Perpignan Recruiting Perpignan, France, 66046 Contact: Laurence SANHES, MD laurence.sanhes@ch-perpignan.fr Principal Investigator: Laurence SANHES, MD Hôpital Haut Lévêque Recruiting Pessac, France, 33604 Contact: Krimo BOUABDALLAH, MD krimo.bouabdallah@chu-bordeaux.fr Principal Investigator: Krimo BOUABDALLAH, MD CHU Lyon Sud Recruiting Pierre Bénite Cedex, France, 69495 Contact: Gilles SALLES, Prof gilles.salles@chu-lyon.fr Principal Investigator: Gilles SALLES, Prof CHU Robert Debre Recruiting Reims, France, 51092 Contact: Alain DELMER, MD adelmer@chu-reims.fr Principal Investigator: Alain DELMER, MD CHU Pontchaillou Recruiting Rennes, France, 35033 Contact: Thierry LAMY de la CHAPELLE, Prof thierry.lamy.de.la.chapelle@chu-rennes.fr Principal Investigator: Thierry LAMY de la CHAPELLE, Prof Centre Henri Becquerel Recruiting Rouen, France, 76000 Contact: Aspasia STAMATOULLAS, MD aspasia.stamatoullas@chb.unicancer.fr Principal Investigator: Aspasia STAMATOULLAS, MD CHU de Strasbourg Recruiting Strasbourg, France, 67098 Contact: Luc-Matthieu FORNECKER, MD luc-matthieu.fornecker@chru-strasbourg.fr Principal Investigator: Luc-Matthieu FORNECKER, MD I.U.C.T Oncopole Recruiting Toulouse, France, 31059 Contact: Cécile BOREL, MD borel.c@chu-toulouse.fr Principal Investigator: Cécile BOREL, MD CHU Bretonneau Recruiting Tours, France, 37044 Contact: Marjan ERTAULT, MD m.ertault@chu-tours.fr Principal Investigator: Marjan ERTAULT, MD CHU de Brabois Recruiting Vandœuvre-lès-Nancy, France, 54511 Contact: Serge BOLOGNA, MD s.bologna@chu-nancy.fr Principal Investigator: Serge BOLOGNA, MD Gustave Roussy Cancer Campus Recruiting Villejuif, France, 94805 Contact: Vincent RIBRAG, MD vincent.ribrag@gustaveroussy.fr Principal Investigator: Vincent RIBRAG, MD Collapse << |
| NCT00846742 | Hodgkin Lymphoma | Phase 2 | Recruiting | December 2021 | United States, California ... more >> Packard Children's Hospital, Stanford University Recruiting Palo Alto, California, United States, 94304 Contact: Michael Link, MD 650-495-8815 mlink@stanford.edu Principal Investigator: Michael Link, MD Rady Children's Hospital- San Diego Recruiting San Diego, California, United States, 92123 Contact: Dennis Kuo, MD 858-966-5811 dkuo@rchsd.org Principal Investigator: Dennis Kuo, MD United States, Maine Maine Children's Cancer Program (MCCP) Recruiting Scarborough, Maine, United States, 04074 Contact: Eric Larsen, MD 207-396-7565 larsee1@mmc.org Principal Investigator: Eric Larsen, MD United States, Massachusetts Massachusetts General Hospital Cancer Center Withdrawn Boston, Massachusetts, United States, 02114 Dana-Farber Harvard Cancer Center Recruiting Boston, Massachusetts, United States, 02115 Contact: Amy Billett, MD 617-632-5640 amy_billett@dfci.harvard.edu Principal Investigator: Amy Billett, MD United States, Tennessee St. Jude Children's Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Monika Metzger, MD 866-278-5833 referralinfo@stjude.org Principal Investigator: Monika Metzger, MD Canada, Ontario The Hospital for Sick Children Withdrawn Toronto, Ontario, Canada, M5G1X8 Collapse << |
| NCT00136565 | Peripheral T-cell Lymphoma | Phase 2 | Completed | - | Belgium ... more >> Groupe d'Etude des Lymphomes de l'Adulte Yvoir, Belgium France Service d'Hématologie - Centre Hospitalier Lyon-Sud Pierre-Bénite cedex, France, 69495 Centre Hospitalier Robert Debré Reims, France, 51092 Centre Henri Becquerel Rouen, France, 76038 Institut Gustave Roussy Villejuif, France Collapse << |
| NCT00654732 | Classic Hodgkin Lymphoma ... more >> Lugano Classification Stage III Hodgkin Lymphoma AJCC v8 Lugano Classification Stage IV Hodgkin Lymphoma AJCC v8 Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> University of Miami Miller School of Medicine-Sylvester Cancer Center Miami, Florida, United States, 33136 United States, Illinois Rush University Medical Center Chicago, Illinois, United States, 60612 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01726374 | Stage I Testicular Non-Seminom... more >>atous Germ Cell Tumor Collapse << | Phase 3 | Active, not recruiting | August 2019 | United Kingdom ... more >> Guy's Hospital London, England, United Kingdom, SE1 9RT Northampton General Hospital NHS Trust Northampton, England, United Kingdom, NN6 8BJ Velindre Cancer Center at Velindre Hospital Cardiff, Wales, United Kingdom, CF14 2TL Aberdeen Royal Infirmary Aberdeen, United Kingdom Ysbyty Gwynedd Bangor, United Kingdom Queen Elizabeth Hospital Birmingham, United Kingdom Royal Sussex County Hospital Brighton, United Kingdom Bristol Haematology and Oncology Centre Bristol, United Kingdom Queen's Hospital Burton-on-Trent, United Kingdom Addenbrooke's Hospital Cambridge, United Kingdom Cheltenham General Hospital Cheltenham, United Kingdom Gloucestershire Royal Hospital Cheltenham, United Kingdom University Hospitals Coventry and Warwickshire NHS Trust Coventry, United Kingdom Royal Derby Hospital Derby, United Kingdom Western General Hospital Edinburgh, United Kingdom Royal Devon and Exeter Hospital Exeter, United Kingdom Beatson West of Scotland Cancer Centre Glasgow, United Kingdom Royal Surrey County Hospital Guildford, United Kingdom Castle Hill Hospital Hull, United Kingdom Ipswich Hospital Ipswich, United Kingdom St James's University Hospital Leeds, United Kingdom Leicester Royal Infirmary Leicester, United Kingdom Lincoln County Hospital Lincoln, United Kingdom Clatterbridge Centre for Oncology Liverpool, United Kingdom Royal Liverpool University Hospital Liverpool, United Kingdom St Bartholomew's Hospital London, United Kingdom University College Hospital London, United Kingdom Maidstone Hospital Maidstone, United Kingdom James Cook University Hospital Middlesbrough, United Kingdom Norfolk and Norwich University Hospital Norwich, United Kingdom Nottingham City Hospital Nottingham, United Kingdom Churchill Hospital Oxford, United Kingdom Weston Park Hospital Sheffield, United Kingdom Southampton General Hospital Southampton, United Kingdom Royal Marsden Hospital Sutton, United Kingdom Collapse << |
| NCT00453232 | Extragonadal Germ Cell Tumor ... more >> Teratoma Testicular Germ Cell Tumor Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> Addenbrooke's Hospital Cambridge, England, United Kingdom, CB2 2QQ Leeds Cancer Centre at St. James's University Hospital Leeds, England, United Kingdom, LS9 7TF Saint Bartholomew's Hospital London, England, United Kingdom, EC1A 7BE Northern Centre for Cancer Treatment at Newcastle General Hospital Newcastle-Upon-Tyne, England, United Kingdom, NE4 6BE Churchill Hospital Oxford, England, United Kingdom, OX3 7LJ Edinburgh Cancer Centre at Western General Hospital Edinburgh, Scotland, United Kingdom, EH4 2XU Beatson West of Scotland Cancer Centre Glasgow, Scotland, United Kingdom, G11 6NT Collapse << |
| NCT00053352 | - | - | Completed | - | - |
| NCT02582697 | Germ Cell Tumor | Phase 3 | Recruiting | July 2023 | Australia, New South Wales ... more >> Calvary Mater Newcastle Recruiting Newcastle, New South Wales, Australia, 2298 Contact: Louise Plowman Louise.Plowman@calvarymater.org.au Contact: Girish Mallesara girish.mallesara@calvarymater.org.au Principal Investigator: Girish Mallesara Royal North Shore Hospital Recruiting St Leonards, New South Wales, Australia, 2065 Contact: Susan Kirby-Lewis Susan.KirbyLewis@health.nsw.gov.au Contact: Alexander Guminski aguminski@nsccahs.health.nsw.gov.au Principal Investigator: Alexander Guminski Prince of Wales Hospital Recruiting Sydney, New South Wales, Australia, 2031 Contact: Daisy Buchanan Daisy.buchanan@sesiahs.health.nsw.gov.au Contact: Julie Howard Julie.howard@sesiahs.health.nsw.gov.au Principal Investigator: Elizabeth Hovey Chris O'Brien Lifehouse Recruiting Sydney, New South Wales, Australia, 2050 Contact: Melissa Quaggiott melissa.mcmahon@lh.org.au Contact: Peter Grimison peter.grimison@lh.org.au Principal Investigator: Peter Grimison Macquarie Cancer Clinical Trials Recruiting Sydney, New South Wales, Australia, 2109 Contact: Louise Francisco louise.francisco@mq.edu.au Contact: Radhika Butala radhika.butala@mq.edu.au Principal Investigator: Howard Gurney Concord Repatriation General Hospital Recruiting Sydney, New South Wales, Australia, 2139 Contact: Kathy Hall Kathy.Hall@sswahs.nsw.gov.au Contact: Martin Stockler martin.stockler@sydney.edu.au Principal Investigator: Martin Stockler Westmead Hospital Recruiting Sydney, New South Wales, Australia, 2145 Contact: Vicky Wegener vicky.wegener@sydney.edu.au Contact: Howard Gurney howard_gurney@wmi.usyd.edu.au Principal Investigator: Howard Gurney Nepean Hospital Recruiting Sydney, New South Wales, Australia, 2751 Contact: Jeremy Jones jeremy.jones@swahs.health.nsw.gov.au Contact: Amanda Stevanovic amanda.stevanovic@swahs.health.nsw.gov.au Principal Investigator: Amanda Stevanovic Tweed Hospital Recruiting Tweed Heads, New South Wales, Australia, 2485 Contact: Charmayne Chorlton Charmayne.Chorlton@ncahs.health.nsw.gov.au Contact: Ehtesham Abdi eaabdi@mac.com Principal Investigator: Ehtesham Abdi SAN Clinical Trials Unit Recruiting Wahroonga, New South Wales, Australia, 2076 Contact: James McQuilan James.McQuillan@sah.org.au Contact: Gavin Marx gmarx@nhog.com.au Principal Investigator: Gavin Marx Australia, Queensland Royal Brisbane & Women's Hospital Recruiting Brisbane, Queensland, Australia, 4029 Contact: Natasha Roberts natasha.roberts@health.qld.gov.au Contact: David Wyld david.wyld@health.qld.gov.au Principal Investigator: David Wyld Princess Alexandra Recruiting Woolloongabba, Queensland, Australia, 4102 Contact: Paul Baxter Paul.Baxter@health.qld.gov.au Contact: Euan Walpole Euan.Walpole@health.qld.gov.au Principal Investigator: Euan Walpole Australia, South Australia Royal Adelaide Hospital Recruiting Adelaide, South Australia, Australia, 5000 Contact: Hazel Bourke hazel.bourke@health.sa.gov.au Contact: Thean Hsiang Tan hsiang.tan@health.sa.gov.au Principal Investigator: Thean Tan Flinders Medical Centre Recruiting Bedford Park, South Australia, Australia, 5042 Contact: Alex Scott-Hoy Alex.Scott-Hoy@health.sa.gov.au Contact: Ganessan Kichenadasse Ganessan.Kichenadasse@health.sa.gov.au Principal Investigator: Ganessan Kichenadasse Australia, Tasmania Royal Hobart Hospital Recruiting Hobart, Tasmania, Australia, 7000 Contact: Lesley Oliver lesley.oliver@dhhs.tas.gov.au Contact: David Boadle david.boadle@dhhs.tas.gov.au Principal Investigator: David Boadle Australia, Victoria Box Hill Hospital Recruiting Box Hill, Victoria, Australia, 3128 Contact: Lauren Mitchell lauren.mitchell@monash.edu Contact: Philip Parente Phillip.parente@monash.edu Principal Investigator: Philip Parente Peter MacCallum Cancer Centre Recruiting East Melbourne, Victoria, Australia, 3002 Contact: Jennifer Petersen jennifer.petersen@petermac.org Contact: Guy Toner guy.toner@petermac.org Principal Investigator: Guy Toner Austin Health Recruiting Heidelberg, Victoria, Australia, 3084 Contact: Jaren Caine Jaren.Caine@austin.org.au Contact: Andrew Weickhardt Andrew.Weickhardt@ludwig.edu.au Principal Investigator: Andrew Weickhardt Sunshine Hospital Recruiting St Albans, Victoria, Australia, 3021 Contact: Jessica Tanner Jessica.Tanner@wh.org.au Contact: Shirley Wong shirleys.wong@mh.org.au Principal Investigator: Shirley Wong Border Medical Oncology Recruiting Wodonga, Victoria, Australia, 3690 Contact: Lauren Callow lcallow@bordermedonc.com.au Contact: Craig Underhill cunderhill@bordermedonc.com.au Principal Investigator: Craig Underhill Australia, Western Australia Fiona Stanley Hospital Recruiting Murdoch, Western Australia, Australia, 6847 Contact: Jaye Harding jaye.harding@health.wa.gov.au Contact: Simon Troon simon.troon@health.wa.gov.au Principal Investigator: Simon Troon New Zealand Auckland Hospital Recruiting Grafton, Auckland, New Zealand, 1142 Contact: Andrew Conley andrewcon@adhb.govt.nz Contact: Fritha Hanning FrithaH@adhb.govt.nz Principal Investigator: Fritha Hanning Palmerston North Hospital Recruiting Roslyn, Palmerston North, New Zealand, 4442 Contact: Sarah Holwell Sarah.Holwell@midcentraldhb.govt.nz Contact: Gary Forgeson Garry.Forgeson@midcentraldhb.govt.nz Principal Investigator: Gary Forgeson Christchurch Hospital Recruiting Christchurch, New Zealand, 8011 Contact: Elizabeth Thompson liz.thompson@cdhb.health.nz Contact: Mark Jeffrey Mark.jeffery@cdhb.health.nz Principal Investigator: Mark Jeffrey Dunedin Hospital Recruiting Dunedin, New Zealand, 9054 Contact: Rachel McLay-Barnes ONCRESEARCH@southerndhb.govt.nz Contact: David Perez david.perez@southerndhb.govt.nz Principal Investigator: David Perez Collapse << |
| NCT02104986 | Extra Cranial Non Seminomateou... more >>s Malignant Germ Cell Tumour Collapse << | Phase 2 | Recruiting | May 2024 | France ... more >> CHU Not yet recruiting Amiens, France, 80054 Principal Investigator: Catherine DEVOLDERE CHU Recruiting Angers, France, 49033 Principal Investigator: Isabelle PELLIER CHRU Not yet recruiting Besançon, France, 25030 Principal Investigator: Véronique LAITHIER CHU Recruiting Bordeaux, France, 33076 Principal Investigator: Cécile VERITE Chu Morvan Not yet recruiting Brest, France, 26609 Principal Investigator: Philippe LEMOINE CHU Not yet recruiting Caen, France, 14033 Principal Investigator: Odile MINCKES Chu Estaing Not yet recruiting Clermont-ferrand, France, 63003 Principal Investigator: Eric DORE CHU Not yet recruiting Dijon, France, 21079 Principal Investigator: GERARD COUILLAULT Centre Oscar Lambret Recruiting Lille, France, 59000 Principal Investigator: HELENE SUDOUR CHU Not yet recruiting Limoges, France, 87042 Principal Investigator: CHRISTOPHE PIGUET Hopital de La Timone Recruiting Marseille, France, 13385 Principal Investigator: ANGELIQUE ROME CHU Not yet recruiting Montpellier, France, 34295 Principal Investigator: NICOLAS KALFA CHU Recruiting Nancy, France, 54511 Principal Investigator: PASCAL CHASTAGNER CHU Recruiting Nantes, France, 44093 Principal Investigator: ESTELLE THEBAUD CHU Recruiting Nice, France, 06202 Principal Investigator: MARILYNE POIREE Institut Curie Not yet recruiting Paris, France, 75005 Principal Investigator: DANIEL ORBACH Hopital Trousseau Not yet recruiting Paris, France, 75012 Principal Investigator: SYLVIE FASOLA CHU Not yet recruiting Poitiers, France, 86021 Principal Investigator: FREDERIC MILLOT CHU Not yet recruiting Reims, France, 51092 Principal Investigator: MARTINE MUNZER CHU Not yet recruiting Rennes, France, 35203 Principal Investigator: VIRGINIE GANDEMER CHU Recruiting Rouen, France, 76031 Principal Investigator: CECILE DUMESNIL DE MARICOURT Hopital Felix Guyon Not yet recruiting Saint Denis, France, 97405 Principal Investigator: MATHILDE JEHANNE CHRU Recruiting Saint Etienne, France, 42055 Principal Investigator: CLAIRE BERGER CHRU Not yet recruiting Strasbourg, France, 67098 Principal Investigator: PATRICK LUTZ CHU Not yet recruiting Toulouse, France, 31059 Principal Investigator: MARIE PIERRE CASTEIX CHRU Not yet recruiting Tours, France, 37044 Principal Investigator: ODILE LEJARS Institut Gustave Roussy Not yet recruiting Villejuif, France, 94805 Principal Investigator: VERONIQUE MINARD Collapse << |
| NCT02161692 | Testicular Neoplasms ... more >> Germ Cell Tumors Collapse << | Phase 2 | Completed | - | Italy ... more >> Istituto Nazionale dei Tumori Milano, Italy, 20133 Collapse << |
| NCT00722865 | - | - | Terminated(slow enrollment) | - | - |
| NCT01712490 | - | - | Active, not recruiting | - | - |
| NCT01322100 | Brain Metastases ... more >> CNS Metastases Collapse << | Phase 1 | Terminated(Due to slow patient... more >> recruitment) Collapse << | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark, 2100 Herlev Hospital Herlev, Denmark, 2730 Collapse << |
| NCT00722865 | Hodgkin Lymphoma | Phase 2 | Terminated(slow enrollment) | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT02352259 | Liver Metastases | Phase 2 | Unknown | December 2016 | Slovenia ... more >> Faculty of Electrical Engineering, University of Ljubljana, Slovenia Recruiting Ljubljana, Slovenia, 1000 Sub-Investigator: Damijan Miklavcic, PhD Sub-Investigator: Tomaz Jarm, PhD Sub-Investigator: Denis Pavliha, PhD Sub-Investigator: Bor Kos, PhD Sub-Investigator: Marija Marcan, Msc Institute of Oncology Ljubljana Recruiting Ljubljana, Slovenia, 1000 Contact: Violeta Kaluza +386 1 5879 122 vkaluza@onko-i.si Principal Investigator: Ibrahim Edhemovic, MD, PhD Sub-Investigator: Gregor Sersa, PhD Sub-Investigator: Marko Snoj, MD, PhD Sub-Investigator: Janja Ocvirk, MD, PhD Sub-Investigator: Maja Music, MD, PhD Sub-Investigator: Gorana Gasljevic, MD, PhD Sub-Investigator: Maja Cemazar, PhD Sub-Investigator: Tjasa Pecnik, BSc Sub-Investigator: Masa Bosnjak, MPharm University Medical Centre Ljubljana, Ljubljana, Slovenia Recruiting Ljubljana, Slovenia, 1000 Principal Investigator: Blaz Trotovsek, MD, PhD Sub-Investigator: Mihajlo Djokic, MD Sub-Investigator: Valentin Sojar, MD Sub-Investigator: Vojka Gorjup, MD Sub-Investigator: Miha Petric, MD University Medical Centre Maribor, Maribor, Slovenia Recruiting Maribor, Slovenia, 2000 Principal Investigator: Stojan Potrc, MD, PhD Sub-Investigator: Arpad Ivanecz, MD, PhD Collapse << |
| NCT00000987 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 1 | Completed | - | United States, California ... more >> UCLA CARE Center CRS Los Angeles, California, United States, 90095 Collapse << |
| NCT01712490 | Hodgkin Lymphoma | Phase 3 | Active, not recruiting | March 1, 2020 | - |
| NCT00992030 | Hodgkin Lymphoma | Phase 3 | Active, not recruiting | December 2019 | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Italy Ospedali Riuniti Umberto I Ancona, Italy, 60020 Ospedali Riuniti Bergamo, Italy, 24100 Policlinico S. Orsola Malpighi Bologna, Italy, 40138 Ospedale Roberto Binaghi Cagliari, Italy, 09126 Azienda Ospedaliera Vittorio Emanuele Ferrarotto Catania, Italy, 95124 Fondazione IRCCS Istituto Nazionale Tumori Milano, Italy, 20133 Azienda Ospedaliero Universitaria S. Luigi Gonzaga Orbassano, Italy, 10043 Gianpietro Semenzato Padova, Italy, 35128 Ospedali Riuniti Villa Sofia Cervello Palermo, Italy, 90146 Ospedale San Carlo Potenza, Italy, 85100 Collapse << |
| NCT00918593 | Ulcerated Cutaneous Metastases | Phase 2 | Withdrawn(No recruitment of pa... more >>tients) Collapse << | February 2014 | Denmark ... more >> Copenhagen University Hospital at Herlev Herlev, Copenhagen, Denmark, 2730 Collapse << |
| NCT02166463 | Ann Arbor Stage IIB Hodgkin Ly... more >>mphoma Ann Arbor Stage IIIB Hodgkin Lymphoma Ann Arbor Stage IV Hodgkin Lymphoma Ann Arbor Stage IVA Hodgkin Lymphoma Ann Arbor Stage IVB Hodgkin Lymphoma Childhood Hodgkin Lymphoma Classic Hodgkin Lymphoma Collapse << | Phase 3 | Recruiting | - | - |
| NCT02549742 | Head Neck Cancer | Phase 2 | Recruiting | September 2019 | Denmark ... more >> Department of Otorhinolaryngology, Rigshospitalet, Copenhagen University Hospital Recruiting Copenhagen, Denmark, 2100 Contact: Christina C. Plaschke, MD +45 29 25 92 45 caroline@dadlnet.dk Contact: Irene Wessel, MD +45 35 45 83 22 irene.wessel.01@regionh.dk Collapse << |
| NCT00504504 | Lymphoma | Phase 2 | Completed | - | United States, Texas ... more >> UT MD . Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT03199300 | - | - | Recruiting | January 2021 | Netherlands ... more >> University Medical Center Groningen Recruiting Groningen, Netherlands, 9713 GZ Contact: J.A. Gietema, MD, PhD +31 50 3612821 j.a.gietema@umcg.nl Contact: L.C. Steggink, MD +31 50 3612821 l.c.steggink@umcg.nl Principal Investigator: J.A. Gietema, MD, PhD Collapse << |
| NCT00744653 | Breast Cancer | Phase 2 | Terminated(Primary endpoint me... more >>asure not suitable for evaluation) Collapse << | - | Denmark ... more >> Herlev Hospital, Herlev Ringvej 75 Herlev, Denmark, DK-2730 Collapse << |
| NCT01941901 | Cutaneous Metastases | Phase 2 | Unknown | April 2018 | Denmark ... more >> Department of Oncology, Copenhagen University Hospital, Herlev Recruiting Herlev, Denmark, 2730 Contact: Julie Gehl 004538683868 julie.gehl@regionh.dk Principal Investigator: Julie Gehl Collapse << |
| NCT00744653 | - | - | Terminated(Primary endpoint me... more >>asure not suitable for evaluation) Collapse << | - | - |
| NCT00504504 | - | - | Completed | - | - |
| NCT00003811 | Childhood Germ Cell Tumor ... more >> Drug/Agent Toxicity by Tissue/Organ Extragonadal Germ Cell Tumor Ovarian Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT01067690 | Kaposi's Sarcoma | Phase 2 | Completed | - | Italy ... more >> Dermatologic Unit, Ospedale Maggiore Policlinico, Milan, Italy Milan, Italy, 20100 Collapse << |
| NCT00004010 | Lymphoma | Phase 2 | Completed | - | - |
| NCT01264952 | Liver Metastases | Phase 1 Phase 2 | Completed | - | Slovenia ... more >> Institute of Oncology Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02291133 | Liver Cancer | Phase 1 Phase 2 | Unknown | July 2017 | Slovenia ... more >> University Medical Centre Ljubljana, Ljubljana, Slovenia Recruiting Ljubljana, Slovenia, 1000 Collapse << |
| NCT00000954 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 1 | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States, 90033 UCLA CARE Center CRS Los Angeles, California, United States, 90095 Ucsf Aids Crs San Francisco, California, United States United States, Colorado University of Colorado Hospital CRS Aurora, Colorado, United States, 80262 United States, Illinois Northwestern University CRS Chicago, Illinois, United States, 60611 Rush Univ. Med. Ctr. ACTG CRS Chicago, Illinois, United States United States, Indiana Indiana Univ. School of Medicine, Infectious Disease Research Clinic Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins Adult AIDS CRS Baltimore, Maryland, United States United States, Massachusetts Bmc Actg Crs Boston, Massachusetts, United States, 02118 United States, Missouri Washington U CRS St. Louis, Missouri, United States United States, New York SUNY - Buffalo, Erie County Medical Ctr. Buffalo, New York, United States, 14215 Memorial Sloan-Kettering Cancer Ctr. New York, New York, United States, 10021 United States, North Carolina Unc Aids Crs Chapel Hill, North Carolina, United States, 27599 United States, Ohio The Ohio State Univ. AIDS CRS Columbus, Ohio, United States, 43210 Collapse << |
| NCT01264952 | - | - | Completed | - | - |
| NCT00079105 | Lymphoma | Phase 2 | Completed | - | - |
| NCT00276718 | Brain and Central Nervous Syst... more >>em Tumors Childhood Germ Cell Tumor Extragonadal Germ Cell Tumor Ovarian Cancer Collapse << | Not Applicable | Unknown | - | Ireland ... more >> Our Lady's Hospital for Sick Children Dublin, Ireland, 12 United Kingdom Birmingham Children's Hospital Birmingham, England, United Kingdom, B4 6NH Institute of Child Health at University of Bristol Bristol, England, United Kingdom, BS2 8AE Bristol Royal Hospital for Children Bristol, England, United Kingdom, BS2 8BJ Addenbrooke's Hospital at Cambridge University Hospitals NHS Foundation Trust Cambridge, England, United Kingdom, CB2 2QQ Leeds Cancer Centre at St. James's University Hospital Leeds, England, United Kingdom, LS9 7TF Leicester Royal Infirmary Leicester, England, United Kingdom, LE1 5WW Royal Liverpool Children's Hospital, Alder Hey Liverpool, England, United Kingdom, L12 2AP Royal London Hospital London, England, United Kingdom, E1 1BB Great Ormond Street Hospital for Children NHS Trust London, England, United Kingdom, WC1N 3JH Central Manchester and Manchester Children's University Hospitals NHS Trust Manchester, England, United Kingdom, M27 4HA Sir James Spence Institute of Child Health Newcastle-Upon-Tyne, England, United Kingdom, NE1 4LP Queen's Medical Centre Nottingham, England, United Kingdom, NG7 2UH Oxford Radcliffe Hospital Oxford, England, United Kingdom, 0X3 9DU Children's Hospital - Sheffield Sheffield, England, United Kingdom, S10 2TH Southampton General Hospital Southampton, England, United Kingdom, SO16 6YD Royal Marsden NHS Foundation Trust - Surrey Sutton, England, United Kingdom, SM2 5PT Royal Belfast Hospital for Sick Children Belfast, Northern Ireland, United Kingdom, BT12 6BE Royal Aberdeen Children's Hospital Aberdeen, Scotland, United Kingdom, AB25 2ZG Royal Hospital for Sick Children Edinburgh, Scotland, United Kingdom, EH9 1LF Royal Hospital for Sick Children Glasgow, Scotland, United Kingdom, G3 8SJ Childrens Hospital for Wales Cardiff, Wales, United Kingdom, CF14 4XW Collapse << |
| NCT03040180 | Locally Advanced Rectal Cancer... more >> Electrochemotherapy Neoadjuvant Therapy Down Staging Collapse << | Phase 2 | Recruiting | December 2018 | Denmark ... more >> Department of Oncology Recruiting Herlev, Capitol Region, Denmark, 2730 Contact: Julie Gehl, MD, DMSc +45 38683868 karen.julie.gehl@regionh.dk Department of Surgery Recruiting Roskilde, Denmark, 4000 Contact: Ismail Gögenur, MD, DMSc +45 26336426 igo@regionsjaelland.dk Contact: Rasmus P Vogelsang, MD +45 27351103 Collapse << |
| NCT00066482 | Childhood Germ Cell Tumor ... more >> Extragonadal Germ Cell Tumor Collapse << | Not Applicable | Completed | - | - |
| NCT02429700 | Ovarian Sex Cord Stromal Tumor... more >> Ovarian Neoplasms Ovarian Cancer Collapse << | Phase 3 | Recruiting | May 2024 | China, Shandong ... more >> Qilu Hospital of Shandong University Recruiting Jinan, Shandong, China Contact: Beihua Kong, MD. PhD. Collapse << |
| NCT00000681 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 1 | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States, 900331079 UCLA CARE Center CRS Los Angeles, California, United States, 90095 United States, Massachusetts Beth Israel Deaconess Med. Ctr., ACTG CRS Boston, Massachusetts, United States, 02215 United States, New York SUNY - Buffalo, Erie County Medical Ctr. Buffalo, New York, United States, 14215 United States, Pennsylvania Pitt CRS Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02429687 | Ovarian Germ Cell Cancer ... more >> Ovarian Neoplasms Ovarian Cancer Collapse << | Phase 3 | Recruiting | May 2024 | China, Shandong ... more >> Qilu Hospital of Shandong University Recruiting Jinan, Shandong, China, 250012 Contact: Beihua Kong, MD. PhD. +8618560081888 kongbeihua@sdu.edu.cn Collapse << |
| NCT00983944 | Lymphoma | Phase 2 | Withdrawn(Inadequate Accrual) | - | - |
| NCT00002093 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 3 | Completed | - | United States, Arizona ... more >> Univ of Arizona / Arizona Cancer Ctr Tucson, Arizona, United States, 85724 United States, California Kenneth Norris Jr Cancer Hosp Los Angeles, California, United States, 90033 Desert Hosp Comprehensive Cancer Ctr Palm Springs, California, United States, 92262 Saint Francis Mem Hosp San Francisco, California, United States, 94109 Davies Med Ctr San Francisco, California, United States, 94114 United States, Colorado Denver Gen Hosp Denver, Colorado, United States, 802044507 United States, District of Columbia George Washington Univ Med Ctr Washington, District of Columbia, United States, 20037 United States, Florida Univ of Miami Dept of Medicine Miami, Florida, United States, 33136 United States, Illinois Northwestern Univ Med School Chicago, Illinois, United States, 60611 United States, Massachusetts New England Deaconess Hosp Boston, Massachusetts, United States, 02215 United States, New York New York Univ Med Ctr New York, New York, United States, 10016 United States, Oregon Kaiser Permanente Med Ctr Portland, Oregon, United States, 97227 United States, Texas Dr Edward Stool Houston, Texas, United States, 77004 Collapse << |
| NCT00165438 | - | - | Completed | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT03067181 | Adult Germ Cell Tumor ... more >> Childhood Extracranial Germ Cell Tumor Childhood Germ Cell Tumor Extragonadal Embryonal Carcinoma Grade 2 Immature Ovarian Teratoma Grade 3 Immature Ovarian Teratoma Malignant Germ Cell Tumor Stage I Ovarian Choriocarcinoma Stage I Ovarian Embryonal Carcinoma Stage I Ovarian Teratoma Stage I Ovarian Yolk Sac Tumor Stage I Testicular Choriocarcinoma AJCC v6 and v7 Stage I Testicular Embryonal Carcinoma AJCC v6 and v7 Stage I Testicular Yolk Sac Tumor AJCC v6 and v7 Stage II Ovarian Choriocarcinoma Stage II Ovarian Embryonal Carcinoma Stage II Ovarian Yolk Sac Tumor Stage II Testicular Choriocarcinoma AJCC v6 and v7 Stage II Testicular Embryonal Carcinoma AJCC v6 and v7 Stage II Testicular Yolk Sac Tumor AJCC v6 and v7 Stage III Ovarian Choriocarcinoma Stage III Ovarian Embryonal Carcinoma Stage III Ovarian Yolk Sac Tumor Stage III Testicular Choriocarcinoma AJCC v6 and v7 Stage III Testicular Embryonal Carcinoma AJCC v6 and v7 Stage III Testicular Yolk Sac Tumor AJCC v6 and v7 Stage IV Ovarian Choriocarcinoma Stage IV Ovarian Embryonal Carcinoma Stage IV Ovarian Yolk Sac Tumor Testicular Mixed Choriocarcinoma and Embryonal Carcinoma Testicular Mixed Choriocarcinoma and Teratoma Testicular Mixed Choriocarcinoma and Yolk Sac Tumor Collapse << | Phase 3 | Recruiting | June 30, 2027 | - |
| NCT00002318 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 3 | Unknown | - | United States, California ... more >> East Bay AIDS Ctr Berkeley, California, United States, 94705 Pacific Oaks Med Group Beverly Hills, California, United States, 90211 Hematology - Oncology Med Group of San Fernando Valley Encino, California, United States, 91436 Dr Becky Miller Los Angeles, California, United States, 90048 Apogee Med Group San Diego, California, United States, 92103 UCSF - San Francisco Gen Hosp San Francisco, California, United States, 94110 Kaiser Permanente Med Ctr San Francisco, California, United States, 94115 UCSF San Francisco, California, United States, 94117 San Francisco Veterans Administration Med Ctr San Francisco, California, United States, 94121 UCSF San Francisco, California, United States, 941430324 Pacific Oaks Med Group Sherman Oaks, California, United States, 91403 United States, District of Columbia Dr Mahmoud Mustafa Washington, District of Columbia, United States, 20037 United States, Florida Univ of Miami School of Medicine Miami, Florida, United States, 33136 H Lee Moffit Cancer Ctr and Research Institute Tampa, Florida, United States, 33612 United States, Georgia American Med Research Institute Atlanta, Georgia, United States, 30329 Infectious Disease Rsch Consortium of GA / SE Clin Resources Atlanta, Georgia, United States, 30345 United States, Illinois Northwestern Med Faculty Foundation Chicago, Illinois, United States, 60611 Rush Presbyterian Med College Chicago, Illinois, United States, 60612 Illinois Masonic Med Ctr / The Cancer Ctr Chicago, Illinois, United States, 60657 United States, Michigan Henry Ford Hosp Detroit, Michigan, United States, 48202 United States, Missouri Washington Univ St Louis, Missouri, United States, 63108 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Saint Vincent's Hosp and Med Ctr New York, New York, United States, 10011 New York Univ Med Ctr New York, New York, United States, 10016 Saint Luke's - Roosevelt Hosp Ctr New York, New York, United States, 10023 United States, Pennsylvania Graduate Hosp / Tuttleman Cancer Ctr Philadelphia, Pennsylvania, United States, 19146 United States, Texas Comprehensive Care Ctr Dallas, Texas, United States, 75235 Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00324298 | Drug/Agent Toxicity by Tissue/... more >>Organ Testicular Germ Cell Tumor Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Basildon University Hospital Basildon, England, United Kingdom, SS16 5NL Addenbrooke's Hospital Cambridge, England, United Kingdom, CB2 2QQ Essex County Hospital Colchester, England, United Kingdom, C03 3NB Ipswich Hospital Ipswich, England, United Kingdom, IP4 5PD Leeds Cancer Centre at St. James's University Hospital Leeds, England, United Kingdom, LS9 7TF Saint Bartholomew's Hospital London, England, United Kingdom, EC1A 7BE University College of London Hospitals London, England, United Kingdom, WIT 3AA Norfolk and Norwich University Hospital Norwich, England, United Kingdom, NR4 7UY Royal Marsden - Surrey Sutton, England, United Kingdom, SM2 5PT Southend University Hospital NHS Foundation Trust Westcliff-On-Sea, England, United Kingdom, SS0 0RY Collapse << |
| NCT00797472 | Hodgkin's Disease | Phase 2 | Unknown | April 2014 | United States, New Jersey ... more >> Raritan Bay Medical Center Not yet recruiting Englishtown, New Jersey, United States, 07726 Contact: Prem A Nandiwada, M.D. 908-941-5480 rgreywal@americanscitech.com Contact: Ratna Grewal, M.D. 908-941-5480 rgreywal@americanscitech.com Collapse << |
| NCT00002105 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 3 | Completed | - | United States, Massachusetts ... more >> Beth Israel Hosp Boston, Massachusetts, United States, 02215 United States, New York Mount Sinai Med Ctr New York, New York, United States, 10029 United States, Texas Twelve Oaks Hosp Houston, Texas, United States, 77074 United States, Washington Virginia Mason Research Center / Clinical Trial Unit Seattle, Washington, United States, 98101 Collapse << |
| NCT02341989 | Testicular Neoplasms ... more >> Seminoma Collapse << | Phase 3 | Recruiting | December 2020 | Norway ... more >> Institutt for kreftforskning og molekylær medisin, St Olavs Hospital Recruiting Trondheim, Norway Contact: Torgrim Tandstad, md phd torgrim.tandstad@ntnu.no Collapse << |
| NCT00993512 | Head and Neck Neoplasms ... more >> Skin Neoplasms Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> University College London Hospital London, United Kingdom, NW1 2PG Collapse << |
| NCT00132613 | Malignant Pericardial Effusion | Phase 3 | Completed | - | Japan ... more >> Aichi Cancer Center Hospital Nagoya,Chikusa-ku,Kanokoden,1-1, Aichi, Japan, 464-8681 Aichi Cancer Center,Aichi Hospital Okazaki,Kake-machi,Kuriyado,18, Aichi, Japan, 444-0011 National Cancer Center Hospital East Kashiwa,Kashiwanoha,6-5-1, Chiba, Japan, 277-8577 National Hospital Organization Shikoku Cancer Center Matsuyama,Horinouchi,13, Ehime, Japan, 790-0007 Kyushu University Hospital Fukuoka,Higashi-ku,Maidashi,3-1-1, Fukuoka, Japan, 812-8582 Gifu Municipal Hospital Gifu,Kashima-cho,7-1, Gifu, Japan, 500-8323 Gunma Prefectural Cancer Center Ota,Takabayashi-nishi-cho,617-1, Gunma, Japan, 373-8550 National Nishigunma Hospital Shibukawa,Kanai,2854, Gunma, Japan, 377-8511 National Hospital Organization, Dohoku National Hospital Asahikawa,Hanasaki,7-4048, Hokkaido, Japan, 070-8644 National Hospital Organization Hokkaido Cancer Center Sapporo,Shiroishi-ku,Kikusui,4-2-3-54, Hokkaido, Japan, 003-0804 Kobe City General Hospital Kobe,Chuo-ku,Minatojimanakamachi,4-6, Hyogo, Japan, 650-0046 Hyogo College of Medicine Nishinomiya,Mukogawa-cho,1-1, Hyogo, Japan, 663-8501 Ibaraki Kenritsu Chuo Hospital & Cancer Center Nishi-ibarakigun,Tomobemachi,Koibuchi,6528, Ibaraki, Japan, 309-1793 Kanagawa Cancer Center Yokohama,Asahi-ku,Nakao,1-1-2, Kanagawa, Japan, 241-0815 Yokohama Mucipical Citizen's Hospital Yokohama,Hodogaya-ku,Okazawa-cho,56, Kanagawa, Japan, 240-8555 Kumamoto Regional Medical Center Hospital Kumamoto,Honjo,5-16-10, Kumamoto, Japan, 860-0811 Tohoku University Hospital Sendai,Aoba-ku,Seiryo-machi,1-1, Miyagi, Japan, 980-8574 Niigata Cancer Center Hospital Niigata,Kawagishi-cho,2-15-3, Niigata, Japan, 951-8566 Osaka Prefectural Medical Center for Respiratory and Allergic Disease Habikino,Habikino,3-7-1, Osaka, Japan, 583-8588 Rinku General Medical Center Izumisano,rinku-ohrai-kita,2-23, Osaka, Japan, 598-0048 Graduate School of Medicine, Osaka City University Osaka,Abeno-ku,Asahi-machi,1-5-7, Osaka, Japan, 545-0051 Osaka Medical Center for Cancer and Cardiovascular Diseases Osaka,Higashinari-ku,Nakamichi,1-3-3, Osaka, Japan, 537-8511 Kinki University School of Medicine Osaka-Sayama,Ohno-higashi,377-2, Osaka, Japan, 589-8511 National Hospital Organization Kinki-Chuo Chest Medical Center Sakai,Nagasone,1180, Osaka, Japan, 591-8555 National Hospital Organization Toneyama National Hospital Toyonaka,Toneyama,5-1-1, Osaka, Japan, 560-8552 Saitama Cancer Center Kita-adachi,Ina,Komuro,818, Saitama, Japan, 362-0806 Tochigi Cancer Center Utsunomiya,Yohnan,4-9-13, Tochigi, Japan, 320-0834 National Cancer Center Hospital Chuo-ku,Tsukiji, 5-1-1, Tokyo, Japan, 104-0045 Cancer Institute Hospital Koto-ku,Ariake,3-10-6, Tokyo, Japan, 135-8550 International Medical Center of Japan Shinjuku-ku,Toyama,1-21-1, Tokyo, Japan, 162-8655 Yamagata Prefectural Central Hospital Yamagata,Aoyagi,1800, Yamagata, Japan, 990-2292 Collapse << |
| NCT01873326 | Germ Cell Tumors | Phase 2 | Active, not recruiting | June 2019 | United States, California ... more >> University of Southern California Los Angeles, California, United States, 90033 Stanford University Medical Center Stanford, California, United States, 94305-5408 United States, Illinois University of Chicago Chicago, Illinois, United States United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New Jersey Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States Memorial Sloan Kettering Monmouth Middletown, New Jersey, United States, 07748 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27514 United States, Pennsylvania University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01404936 | - | - | Completed | - | - |
| NCT00006035 | Melanoma (Skin) | Phase 1 | Unknown | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00001059 | Sarcoma, Kaposi ... more >> HIV Infections Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama Therapeutics CRS Birmingham, Alabama, United States, 35294 United States, California USC CRS Los Angeles, California, United States, 90033 UCLA CARE Center CRS Los Angeles, California, United States, 90095 Ucsf Aids Crs San Francisco, California, United States United States, Colorado University of Colorado Hospital CRS Aurora, Colorado, United States, 80262 United States, Florida Univ. of Miami AIDS CRS Miami, Florida, United States, 33136 United States, Hawaii Queens Med. Ctr. Honolulu, Hawaii, United States, 96813 Univ. of Hawaii at Manoa, Leahi Hosp. Honolulu, Hawaii, United States, 96816 United States, Illinois Northwestern University CRS Chicago, Illinois, United States, 60611 Rush Univ. Med. Ctr. ACTG CRS Chicago, Illinois, United States, 60612 United States, Indiana Indiana Univ. School of Medicine, Infectious Disease Research Clinic Indianapolis, Indiana, United States, 46202 United States, Massachusetts Bmc Actg Crs Boston, Massachusetts, United States, 02118 Beth Israel Deaconess - East Campus A0102 CRS Boston, Massachusetts, United States, 02215 Massachusetts General Hospital ACTG CRS Boston, Massachusetts, United States United States, Missouri Washington U CRS St. Louis, Missouri, United States, 63110 United States, New York SUNY - Buffalo, Erie County Medical Ctr. Buffalo, New York, United States, 14215 Memorial Sloan-Kettering Cancer Ctr. New York, New York, United States, 10021 Collapse << |
| NCT00027521 | Pancreatic Cancer | Phase 1 | Withdrawn(Program terminated) | - | - |
| NCT00002872 | Metastatic Cancer | Phase 3 | Completed | - | - |
| NCT01606566 | Recurrent Head and Neck Cancer... more >> Carcinoma, Squamous Cell of Head and Neck Collapse << | Phase 2 | Terminated(Business reasons) | - | France ... more >> CHU de Nantes Hôtel Dieu Nantes, Nantes Cedex 1, France, 44093 Centre Alexis Vautrin (CAV)-Nancy Université Nancy, France Germany Universitätsklinikum Schleswig-Holstein Lübeck, Schleswig-Holstein, Germany, 23538 Charité Comprehensive Cancer Center Berlin, Germany, 10117 Universitätsklinikum Essen Essen, Germany, 45147 Ludwig Maximilian University Munich München, Germany Lithuania Institute of Oncology, Vilnius University Vilnius, Lithuania, 08660 Netherlands The Netherlands Cancer Institute, Antoni van Leeuwenhoek Hospital Amsterdam, Netherlands Poland Szpital Specjalistyczny w Brzozowie Brzozów, Poland United Kingdom University College London Hospital London, United Kingdom Collapse << |
| NCT01404936 | Lymphoma | Phase 2 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00006916 | - | - | Terminated(Low accrual) | - | - |
| NCT02883023 | Capillary Malformations ... more >> Vascular Malformations Collapse << | Phase 2 | Recruiting | March 2018 | Netherlands ... more >> Academic Medical Center (AMC) Recruiting Amsterdam, Noord-Holland, Netherlands, 1105AZ Contact: Sophie E Horbach, MD 0031205666679 s.e.horbach@amc.uva.nl Contact: Albert Wolkerstorfer, MD PhD a.wolkerstorfer@amc.uva.nl Sub-Investigator: Sophie E Horbach, MD Principal Investigator: Chantal M van der Horst, MD PhD Sub-Investigator: Albert Wolkerstorfer, MD PhD Sub-Investigator: Martijn de Bruin, MSc PhD Collapse << |
| NCT03225781 | Pancreatic Cancer | Not Applicable | Recruiting | June 15, 2020 | Italy ... more >> University of Verona Hospital Recruiting Verona, Italy, 37134 Contact: Salvatore Paiella, MD +390458126009 salvatore.paiella@aovr.veneto.it Principal Investigator: Salvatore Paiella, MD Sub-Investigator: Luca Landoni, MD Sub-Investigator: Alessandro Esposito, MD Sub-Investigator: Luca Casetti, MD Principal Investigator: Claudio Bassi, MD, FRCS, FACS, FEBS Collapse << |
| NCT01347294 | Venous Malformation | Phase 4 | Recruiting | December 2018 | Norway ... more >> Oslo Universitetssykehus Rikshospitalet Recruiting Oslo, Norway, 0227 Contact: Rune Andersen, MD +4723073992 randerse@ous-hf.no Collapse << |
| NCT03649113 | Hemangioma Liver | Phase 1 | Not yet recruiting | September 15, 2020 | - |
| NCT00006916 | Brain and Central Nervous Syst... more >>em Tumors Collapse << | Phase 2 | Terminated(Low accrual) | - | United States, Arizona ... more >> Foundation for Cancer Research and Education Phoenix, Arizona, United States, 85013 United States, Kentucky Markey Cancer Center at University of Kentucky Chandler Medical Center Lexington, Kentucky, United States, 40536-0293 United States, Nebraska Methodist Hospital Cancer Center at Nebraska Methodist Hospital - Omaha Omaha, Nebraska, United States, 68114-4199 United States, New Jersey South Jersey Regional Cancer Center Millville, New Jersey, United States, 08332 Fox Chase Virtua Health Cancer Program at Virtua Memorial Hospital Mount Holly, New Jersey, United States, 08060 United States, Ohio Cancer Treatment Center Wooster, Ohio, United States, 44691 United States, Oklahoma St. John Health System Tulsa, Oklahoma, United States, 74104 United States, Utah Cottonwood Hospital Medical Center Murray, Utah, United States, 84107 Utah Valley Regional Medical Center - Provo Provo, Utah, United States, 84604 Dixie Regional Medical Center Saint George, Utah, United States, 84770 LDS Hospital Salt Lake City, Utah, United States, 84143 United States, Wisconsin CCOP - Marshfield Clinic Research Foundation Marshfield, Wisconsin, United States, 54449 Medical College of Wisconsin Cancer Center Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT03628417 | Cutaneous Melanoma | Not Applicable | Enrolling by invitation | March 31, 2019 | - |
| NCT01042522 | Ovarian Granulosa Cell Tumor ... more >> Ovarian Gynandroblastoma Ovarian Sertoli-Leydig Cell Tumor Ovarian Sex Cord Tumor With Annular Tubules Ovarian Sex Cord-Stromal Tumor Ovarian Sex Cord-Stromal Tumor of Mixed or Unclassified Cell Types Ovarian Steroid Cell Tumor Collapse << | Phase 2 | Recruiting | - | - |
| NCT00198328 | Head Neck Cancer | Phase 3 | Withdrawn(Enrollment was stopp... more >>ed based on the Data Monitoring Committee's recommendation.) Collapse << | - | United States, California ... more >> Inovio Biomedical Corporation San Diego, California, United States, 92121 Collapse << |
| NCT03596918 | AIDS-Related Kaposi Sarcoma ... more >> Human Immunodeficiency Virus 1 Positive Collapse << | Phase 1 | Recruiting | March 31, 2020 | Tanzania ... more >> Bugando Medical Centre Recruiting Mwanza, Tanzania Contact: Nestory Masalu 255-783-0-00004 nmasalu2000@yahoo.com Principal Investigator: Nestory Masalu, MD MMed Collapse << |
| NCT03477448 | Warts | Phase 4 | Not yet recruiting | April 1, 2020 | - |
| NCT00003110 | Lymphoma | Phase 2 | Completed | - | United States, Ohio ... more >> Ireland Cancer Center at University Hospitals Case Medical Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106-5065 Collapse << |
| NCT00198263 | Head and Neck Cancer | Phase 4 | Completed | - | United States, California ... more >> Inovio Biomedical Corporation San Diego, California, United States, 92121 Collapse << |
| NCT00198276 | Cancer | Phase 4 | Completed | - | United States, California ... more >> Inovio Biomedical Corporation San Diego, California, United States, 92121 Collapse << |
| NCT00198315 | Head and Neck Cancer | Phase 3 | Withdrawn(Enrollment was stopp... more >>ed based on the Data Monitoring Committee's recommendation.) Collapse << | - | United States, California ... more >> Inovio Biomedical Corporation San Diego, California, United States, 92121 Collapse << |
| NCT01872923 | Cutaneous or Sub-cutaneous Mal... more >>ignancies Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> University College London Hospital London, United Kingdom Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
