CAS No.: 446-72-0
染料木素 Catalog No. CSN18651
Synonyms: NPI 031L;CI-75610;NSC 36586
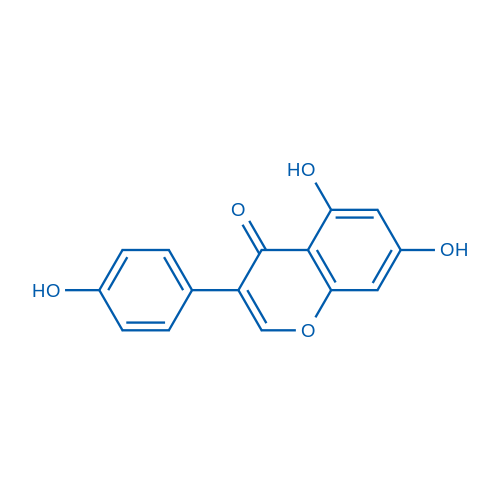
Genistein has multiple biological functions. It can inhibit several tyrosine kinases (IC50 of 0.6 μM for EGFR), inhibits DNA topoisomerase-II and activation of PPARs. Genistein is a phytoestrogen which has selective estrogen receptor modulator properties.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 PTK
IC50:12μM/19μM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01036321 Prostate Cancer Phase 2 Completed - United States, Florida ... more >> University of Florida & Shands Medical Center - Jacksonville Jacksonville, Florida, United States, 32209 H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 James A. Haley VA Hospital Tampa, Florida, United States, 33612 Collapse << NCT01036321 - - Completed - - NCT01232751 Postmenopause Not Applicable Completed - Netherlands ... more >> Wageningen University Wageningen, Gelderland, Netherlands, 6703 HD Collapse << - 更多
- 参考文献
- [1] Jalili C, Ahmadi S, et al. Effect of Genistein on reproductive parameter and serum nitric oxide levels in morphine-treated mice. Int J Reprod Biomed (Yazd). 2016 Feb;14(2):95-102.
- [2] Akiyama T, Ishida J, et al. Genistein, a specific inhibitor of tyrosine-specific protein kinases. J Biol Chem. 1987 Apr 25;262(12):5592-5.
- [3] Mukund V, Mukund D, Sharma V, Mannarapu M, Alam A. Genistein: Its role in metabolic diseases and cancer. Crit Rev Oncol Hematol. 2017 Nov;119:13-22
- [4] Jaiswal N, Akhtar J, Singh SP; Badruddeen, Ahsan F. An Overview on Genistein and its Various Formulations. Drug Res (Stuttg). 2019 Jun;69(6):305-313
- [5] Ganai AA, Farooqi H. Bioactivity of genistein: A review of in vitro and in vivo studies. Biomed Pharmacother. 2015 Dec;76:30-8
- [6] Rahman Mazumder MA, Hongsprabhas P. Genistein as antioxidant and antibrowning agents in in vivo and in vitro: A review. Biomed Pharmacother. 2016 Aug;82:379-92
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 446-72-0 | 储存条件 |
|
|||||||
| 分子式 | C15H10O5 | 运输 | 蓝冰 | |||||||
| 分子量 | 270.24 | 别名 | NPI 031L;CI-75610;NSC 36586;BIO-300;G-2535;PTI-G-4660;SIPI-9764-I;染料木素 | |||||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 293T cells | - | Function assay | - | Displacement of [3H]17beta-estradiol from recombinant human ERbeta expressed in 293T cells, IC50=0.0013 μM | 16219463 |
| 3T3 cells | - | Cytotoxic assay | - | Cytotoxic effect on 3T3 cells, IC50=24 μM | 8201603 |
| A431 cells | 50 to 75 uM | Function assay | 36 h | Cell cycle arrest in human A431 cells assessed as decrease in accumulation at G0/G1 phase at 50 to 75 uM after 36 hrs by propidium iodide staining-based flow cytometric analysis | 24456004 |
| ANN-1 cells | - | Cytotoxic assay | - | Cytotoxic effect on v-abl transformed murine ANN-1 cells, IC50=8 μM | 8201603 |
| HEK293 cells | - | Function assay | - | Antagonist activity at human recombinant LXRbeta expressed in HEK293 cells by luciferase reporter gene assay, IC50=22 μM | 18343126 |
| HEK293 cells | - | Function assay | - | Antagonist activity at human recombinant LXRalpha expressed in HEK293 cells by luciferase reporter gene assay, IC50=31 μM | 18343126 |
| HeLa cells | - | Function assay | - | Activation of estrogen response element in HeLa cells stably transfected with human Estrogen receptor beta, EC50=0.0041 μM | 11906280 |
| human A549 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human A549 cells after 72 hrs by SRB assay, IC50=43.09 μM | 21129977 |
| human AGS cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human AGS cells after 72 hrs by MTT assay, IC50=41.67 μM | 21129977 |
| human BT20 cell | - | Proliferation assay | - | Antiproliferative activity against human BT20 cell line by MTT assay, IC50=46 μM | 16789737 |
| human BxPC3 cell | - | Proliferation assay | - | Antiproliferative activity against human BxPC3 cell line by MTT assay, IC50=30 μM | 16789737 |
| human DU145 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human DU145 cells after 72 hrs by MTT assay, IC50=47.29 μM | 21129977 |
| human HCT116 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human HCT116 cells after 72 hrs by MTT assay, IC50=34.9 μM | 21129977 |
| human HL60 cells | - | Function assay | 24 h | Antileukemic activity against human HL60 cells after 24 hrs by clonogenic assay, LC50=6.3 μM | 18163589 |
| human Ishikawa cells | - | Function assay | 4 days | Estrogenic activity in human Ishikawa cells assessed as induction of alkaline phosphatase activity after 4 days by para-nitrophenol release assay, IC50=0.51 μM | 12502307 |
| human LNCAP cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human LNCAP cells after 72 hrs by SRB assay, IC50=30.65 μM | 21129977 |
| human LoVo cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human LoVo cells after 72 hrs by MTT assay, IC50=15.88 μM | 21129977 |
| human MCF7 cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50=1 μM | 20813524 |
| human MCF7 cells | - | Proliferation assay | - | Antiproliferative activity against estrogen receptor expressing human MCF7 cells, GI50=10 μM | 19818612 |
| human MCF7 cells | - | Function assay | 6 h | Inhibition of phorbol ester-induced ornithine decarboxylase in human MCF7 cells after 6 hrs, IC50=26 μM | 10075742 |
| human MCF7 cells | 0.1-20 μM | Function assay | 4 days | Stimulation of growth in human MCF7 cells at 0.1 to 20 uM after 4 days by MTT assay | 25078314 |
| human MCF7 cells | 20-100 μM | Growth inhibition assay | 4 days | Growth inhibition in human MCF7 cells at 20 to 100 uM after 4 days by MTT assay | 25078314 |
| human MDA-kb2 cells | - | Function assay | - | Agonist activity at androgen receptor in human MDA-kb2 cells assessed as stimulation of luciferase activity by luciferase reporter gene assay | 19592245 |
| human MOLT3 cells | - | Function assay | 24 h | Antileukemic activity against human MOLT3 cells after 24 hrs by clonogenic assay, LC50=7.5 μM | 18163589 |
| human MOLT3 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human MOLT3 cells after 48 hrs | 18163589 |
| human PC3 cells | - | Proliferation assay | 48 h | Antiproliferative activity against androgen receptor-deficient human PC3 cells incubated with 0.6 nM of estradiol and 0.7 nM of testosterone after 48 hrs by sulforhodamine B assay, GI50=42.658 μM | 21513275 |
| human Raji cells | - | Function assay | 24 h | Antileukemic activity against human Raji cells after 24 hrs by clonogenic assay, LC50=13.7 μM | 18163589 |
| human SKBR3 cells | 50 to 75 uM | Function assay | 36 h | Cell cycle arrest in human SKBR3 cells assessed as decrease in accumulation at G0/G1 phase at 50 to 75 uM after 36 hrs by propidium iodide staining-based flow cytometric analysis | 24456004 |
| human T47D cells | - | Function assay | - | Estrogenic activity in human T47D cells assessed as drug level causing stimulation of cell proliferation equivalent to 10 pM estradiol by alamar blue assay | 15787436 |
| human U937 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human U937 cells after 72 hrs by WST-8 assay, IC50=48 μM | 17158054 |
| HUVE12 cells | - | Function assay | - | Reversal of hydrogen peroxide-induced LDH activity in human HUVE12 cells | 18068980 |
| K562 cells | - | Growth inhibition assay | 5 days | Growth inhibition of K562 cells by XTT assay after 5 days, IC50=17.56 μM | 17411092 |
| MDCK cells | - | Function assay | - | Inhibition of BCRP expressed in MDCK cells using Hoechst 33342 staining, IC50=6.9 μM | 21354800 |
| mouse L1210 cells | - | Function assay | 24 h | Antileukemic activity against mouse L1210 cells after 24 hrs by clonogenic assay, LC50=6.5 μM | 18163589 |
| mouse RAW264.7 cells | - | Function assay | 24 h | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced TNFalpha production treated 2 hrs before LPS challenge measured after 24 hrs by ELISA, IC50=19.1 μM | 21288727 |
| NHEM cells | - | Function assay | 72 h | Inhibition of melanin synthesis in NHEM cells assessed as [14C]thiouracil incorporation after 72 hrs by liquid scintillation counting, IC50=29.2 μM | 19132934 |
| NIH3T3 cells | - | Function assay | - | Agonist activity at CFTR-deltaF508 mutant expressed in NIH3T3 cells assessed as increase in forskolin-stimulated current, EC50=4.4 μM | 18595696 |
| RAW 264 cells | - | Function assay | 24 h | Inhibition of 1 ug/ml LPS-stimulated TNFalpha accumulation in RAW 264 cells after 24 hrs, IC50=18.1 μM | 17320246 |
| WiDr human colon cells | - | Cytotoxic assay | - | Cytotoxic effect on WiDr human colon cells, IC50=27.7 μM | 8201603 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01036321 | Prostate Cancer | Phase 2 | Completed | - | United States, Florida ... more >> University of Florida & Shands Medical Center - Jacksonville Jacksonville, Florida, United States, 32209 H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 James A. Haley VA Hospital Tampa, Florida, United States, 33612 Collapse << |
| NCT01036321 | - | - | Completed | - | - |
| NCT01232751 | Postmenopause | Not Applicable | Completed | - | Netherlands ... more >> Wageningen University Wageningen, Gelderland, Netherlands, 6703 HD Collapse << |
| NCT00042380 | Depression | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Mental Health (NIMH) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02309801 | Healthy | Phase 1 | Completed | - | Spain ... more >> Parc de Salut Mar. Barcelona, Spain, 08003 Collapse << |
| NCT02026518 | Irritable Bowel Disease | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Gastrointestinal Clinics of Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of Collapse << |
| NCT00078923 | Prostate Cancer | Phase 2 | Completed | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 Collapse << |
| NCT03008785 | Bloodpressure | Not Applicable | Completed | - | Brazil ... more >> Guilherme Morais Puga Uberlandia, Minas Gerais, Brazil, 38400-678 Collapse << |
| NCT00925639 | Menopause | Phase 3 | Unknown | August 2010 | Brazil ... more >> Ambulatório de Ginecologia Endócrina Recruiting São Paulo, Brazil, 04039-060 Contact: Maria Cecília Santos, Nurse 5511-55496174 mc.santos@hotmail.com Principal Investigator: Regiane HB Rabelo, M.D. Collapse << |
| NCT03167827 | Pelvic Floor | Phase 4 | Completed | - | - |
| NCT01497977 | Low Serum Lipid Levels | Phase 4 | Completed | - | Serbia ... more >> Ultramedica Clinic, American Medical Academy Belgrade, Serbia, 11000 Collapse << |
| NCT00741208 | Asthma | Not Applicable | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT01288859 | Healthy | Not Applicable | Completed | - | Italy ... more >> Department of Food Science Portici, Italy, 80055 Collapse << |
| NCT01288859 | - | - | Completed | - | - |
| NCT00491595 | Drug Toxicity | Phase 1 | Completed | - | - |
| NCT01463436 | Cardiovascular Disease ... more >> Osteoporosis Collapse << | Phase 3 | Completed | - | Indonesia ... more >> Trisakti University Jakarta, DKI Jakarta, Indonesia Collapse << |
| NCT00277446 | Asthma | Not Applicable | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT02567799 | Carcinoma, Non-Small-Cell Lung | Phase 1 Phase 2 | Recruiting | December 2019 | United States, Louisiana ... more >> Ochsner Clinic Foundation Withdrawn New Orleans, Louisiana, United States, 70121 United States, Maryland University of Maryland School of Medicine Recruiting Baltimore, Maryland, United States, 21201 Contact: Caitlin Eggleston, BS 410-369-5351 caitlineggleston@umm.edu Principal Investigator: Charles B. Simone, II, M.D. Sub-Investigator: Kenneth D. Miller, M.D. Sub-Investigator: Pranshu Mohindra, M.D. Sub-Investigator: Petr Hausner, M.D. Sub-Investigator: Katherine Scilla, M.D. Sub-Investigator: Christian Rolfo, M.D. Sub-Investigator: Isabelle Choi, M.D. United States, Michigan Henry Ford Hospital Recruiting Detroit, Michigan, United States, 48202-2689 Contact: Bonita Braxton, RN 313-916-3938 BBRAXTO1@hfhs.org Principal Investigator: Benjamin Movsas, M.D. Principal Investigator: Ding Wang, M.D. Sub-Investigator: Thomas Doyle, M.D. Sub-Investigator: Munther Ajlouni, M.D. Sub-Investigator: Igor Rybkin, M.D. Sub-Investigator: David Spizarny, M.D. Sub-Investigator: Stephen Brown, PhD United States, Wisconsin Medical College of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Igli Arapi, MS 414-805-8773 iarapi@mcw.edu Principal Investigator: Elizabeth M. Gore, M.D. Sub-Investigator: Smitha Menon, M.D. Sub-Investigator: Johnathan Thompson, M.D. Sub-Investigator: Lindsay Puckett, M.D. Zablocki VA Medical Center Recruiting Milwaukee, Wisconsin, United States, 53295 Contact: Shelley Dufek 414-384-2000 ext 42593 Shelley.Dufek@va.gov Principal Investigator: Elizabeth M Gore, MD Sub-Investigator: Adam Currey, MD Sub-Investigator: John Longo, MD Sub-Investigator: Hina Saeed, MD Sub-Investigator: Yee C Cheng, MD Sub-Investigator: Prabhas Mittal, MD Sub-Investigator: Deepak Kilari, MD Sub-Investigator: Lindsay Puckett, MD Collapse << |
| NCT02174666 | Osteopenia Os... more >>teoporosis Collapse << | Not Applicable | Unknown | September 2016 | Denmark ... more >> Aarhus University Hospital Aarhus, Central Jutland Region, Denmark, 8000 Collapse << |
| NCT00661856 | Osteoporosis | Not Applicable | Completed | - | United States, Nebraska ... more >> Creighton University Medical Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT03561662 | Menopause | Not Applicable | Completed | - | United Kingdom ... more >> Human Intervention Studies Unit, Ulster University Coleraine, Co.Londonderry, United Kingdom, BT52 1SA Collapse << |
| NCT00277446 | - | - | Completed | - | - |
| NCT01302639 | Obesity, Insulin Sensitivity, ... more >>Type 2 Diabetes Mellitus Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Maastricht University Maastricht, Netherlands, 6200 MD Collapse << |
| NCT02336087 | Pancreatic Adenocarcinoma ... more >> Unresectable Pancreatic Carcinoma Collapse << | Phase 1 | Recruiting | August 2019 | United States, California ... more >> City of Hope Medical Center Recruiting Duarte, California, United States, 91010 Contact: Vincent Chung 626-471-9200 vchung@coh.org Principal Investigator: Vincent Chung City of Hope Rancho Cucamonga Recruiting Rancho Cucamonga, California, United States, 91730 Contact: Valerie Estala 626-256-4673 ext 81699 Sub-Investigator: Behnam Ebrahimi, MD City of Hope South Pasadena Recruiting South Pasadena, California, United States, 91030 Contact: Odessa Rodriguez 626-256-4673 ext 81409 orodriguez@coh.org Sub-Investigator: Christina Yeon, MD City of Hope West Covina Recruiting West Covina, California, United States, 91790 Contact: Meizi Zheng 626-256-4673 ext 81336 Sub-Investigator: Gargi Upadhyaya, MD Collapse << |
| NCT00924339 | Familial Hypercholesterolemia | Not Applicable | Unknown | September 2011 | Austria ... more >> Division of Nutrition and Metabolism, Department of Pediatrics, Medical University of Vienna Not yet recruiting Vienna, Austria, 1090 Contact: Alexandra Kreißl, Mag.rer.nat 0034140400 ext 2051 alexandra.kreissl@meduniwien.ac.at Principal Investigator: Alexandra Kreißl, Mag.rer.nat Collapse << |
| NCT00000613 | Bone Diseases ... more >> Cardiovascular Diseases Coronary Disease Depression Heart Diseases Myocardial Ischemia Osteoporosis Postmenopause Collapse << | Phase 2 | Completed | - | - |
| NCT01556737 | Postmenopause | Not Applicable | Completed | - | Netherlands ... more >> Wageningen University Wageningen, Gelderland, Netherlands, 6700 EV Collapse << |
| NCT01182246 | Adenocarcinoma | Phase 1 Phase 2 | Unknown | March 2016 | Sweden ... more >> Dept. of Clinical Science, Intervention and Technology, Div. of surgery, Karolinska University Hospital, Huddinge Recruiting Stockholm, Sweden, SE-141 86 Contact: Matthias Löhr +46 8 585 89591 matthias.loehr@ki.se Principal Investigator: Mattias Löhr, MD, PhD,Prof. Dept. of Oncology-Pathology, Karolinska University Hospital, Solna Recruiting Stockholm, Sweden, SE-17176 Contact: Jan-Erik Frödin +46 8 517 733 89 jan-erik.frodin@karolinksa.se Principal Investigator: Jan-Erik Frödin, MD Collapse << |
| NCT00004858 | Leukemia Lymp... more >>homa Collapse << | Phase 1 | Unknown | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027-0700 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010-2970 United States, Minnesota Parker Hughes Institute St. Paul, Minnesota, United States, 55113 Collapse << |
| NCT02075112 | Cancer of Head and Neck | Phase 1 | Active, not recruiting | July 2022 | United States, Georgia ... more >> Emory University Hospital Midtown Atlanta, Georgia, United States, 30308 Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT00698984 | Osteoporosis | Not Applicable | Completed | - | United States, Nebraska ... more >> Creighton University Medical Center - Osteoporosis Research Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00269555 | Prostate Cancer | Not Applicable | Completed | - | United States, California ... more >> University of California, Davis Sacramento, California, United States, 95817 Collapse << |
| NCT00376948 | - | - | Completed | - | - |
| NCT00244907 | Osteoporosis ... more >>Osteopenia Collapse << | Phase 1 | Completed | - | United States, Indiana ... more >> Purdue University West Lafayette, Indiana, United States, 47907 Collapse << |
| NCT00355953 | Menopause Ost... more >>eopenia Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT00951912 | - | - | Completed | - | - |
| NCT00287690 | Coronary Artery Disease | Phase 2 Phase 3 | Completed | - | United Kingdom ... more >> Royal Brompton & Harefield NHS Trust London, United Kingdom, SW3 6NP Collapse << |
| NCT00376948 | Pancreatic Cancer | Phase 2 | Completed | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 United States, Texas M. D. Anderson Cancer Center at University of Texas Houston, Texas, United States, 77030-4009 Collapse << |
| NCT00584532 | Prostate Cancer | Phase 2 Phase 3 | Completed | - | - |
| NCT01553773 | Atrophy | Phase 2 | Completed | - | Brazil ... more >> Department of Gynecology - UNIFESP Sao Paulo, Brazil, 04021003 Collapse << |
| NCT00005827 | Prostate Cancer | Phase 1 | Withdrawn | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center, UNC Chapel Hill, North Carolina, United States, 27599-7295 Collapse << |
| NCT00290758 | - | - | Completed | - | - |
| NCT00951912 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | China, Guangdong ... more >> The First Affiliated Hospital, Sun Yat-sen University Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT00546039 | Prostatic Neoplasms | Phase 2 | Unknown | January 2009 | Norway ... more >> Aker University Hospital Oslo, Norway, 0514 Collapse << |
| NCT00453960 | Endometrial Hyperplasia | Phase 2 | Completed | - | Italy ... more >> Menopause Centre, Department of Gynecology and Obstetrics, Policlinico Universitario "G.Martino" Messina, Italy, 98100 Collapse << |
| NCT00001696 | Cancer | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00118040 | Recurrent Bladder Cancer ... more >> Stage I Bladder Cancer Stage II Bladder Cancer Stage III Bladder Cancer Collapse << | Phase 2 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Chemoprevention Consortium Madison, Wisconsin, United States, 53706-1969 Collapse << |
| NCT00118040 | - | - | Completed | - | - |
| NCT00244933 | Breast Cancer | Phase 2 | Completed | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 Collapse << |
| NCT00290758 | Breast Cancer | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00244933 | - | - | Completed | - | - |
| NCT00099008 | Breast Cancer ... more >> Endometrial Cancer Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Collapse << |
| NCT00276835 | Kidney Cancer ... more >> Melanoma (Skin) Collapse << | Early Phase 1 | Completed | - | United States, Illinois ... more >> Robert H. Lurie Comprehensive Cancer Center at Northwestern University Chicago, Illinois, United States, 60611-3013 Collapse << |
| NCT00626769 | - | - | Completed | - | - |
| NCT00016744 | Cystic Fibrosis | Phase 1 Phase 2 | Completed | - | - |
| NCT01628471 | Non Small Cell Lung Cancer | Phase 1 Phase 2 | Completed | - | Canada, Quebec ... more >> Hôpital Notre Dame du CHUM Montreal, Quebec, Canada, H2L 4M1 Collapse << |
| NCT00058266 | Prostate Cancer | Phase 2 | Terminated(Data Monitoring Com... more >>mittee cited poor accrual.) Collapse << | - | United States, Illinois ... more >> Robert H. Lurie Comprehensive Cancer Center at Northwestern University Chicago, Illinois, United States, 60611-3013 Veterans Affairs Medical Center - Lakeside Chicago Chicago, Illinois, United States, 60611 Evanston Northwestern Healthcare - Evanston Hospital Evanston, Illinois, United States, 60201-1781 Ingalls Cancer Care Center at Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 United States, Washington University of Washington School of Medicine Seattle, Washington, United States, 98195 Collapse << |
| NCT01664650 | Metabolic Syndrome | Phase 2 Phase 3 | Completed | - | Italy ... more >> University of Magnia Graecia Catanzaro, Italy University of Messina Messina, Italy, 98123 University of Palermo Palermo, Italy, 90129 Collapse << |
| NCT00590538 | - | - | Terminated(12/15/2008 Voluntar... more >>ily placed on inactive status-requested by the PI) Collapse << | - | - |
| NCT01538316 | Primary Prevention of Prostate... more >> Cancer Collapse << | Not Applicable | Unknown | April 2014 | Germany ... more >> Institute of Nutritional Medicine, University of Hohenheim Recruiting Stuttgart, Germany, 70599 Contact: Antje Damms Machado, dipl. troph. antje.machado@uni-hohenheim.de Principal Investigator: Stephan C Bischoff, MD, Prof. Department of Urology, University Hospital Tübingen Recruiting Tübingen, Germany, 72076 Contact: Stefan Aufderklamm, MD Principal Investigator: Arnulf Stenzl, MD, Prof. Collapse << |
| NCT00590538 | Cystic Fibrosis | Phase 1 Phase 2 | Terminated(12/15/2008 Voluntar... more >>ily placed on inactive status-requested by the PI) Collapse << | - | United States, Pennsylvania ... more >> The Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00541710 | Metabolic Syndrome | Phase 2 Phase 3 | Completed | - | Italy ... more >> University of Magnia Graecia Catanzaro, Italy Azienda Policlinico Universitario, G. Martino Messina, Italy, 090 University of Palermo Palermo, Italy, 90129 Collapse << |
| NCT03040531 | Osteoporosis, Steroid Induced | Phase 2 Phase 3 | Recruiting | December 30, 2018 | Italy ... more >> University of Messina Recruiting Messina, Italy, 98123 Principal Investigator: Francesco Squadrito, MD University Hospital Recruiting Messina, Italy, 98125 Contact: Francesco Squadrito, MD +390902213648 fsquadrito@unime.it Principal Investigator: Marco Atteritano, MD Sub-Investigator: Alessandra Bitto, MD/PhD Collapse << |
| NCT01489813 | Bladder Cancer | Phase 2 | Recruiting | May 2022 | United States, Georgia ... more >> Emory University Department of Urology Recruiting Atlanta, Georgia, United States, 30322 Contact: Ella Anastasiades, BA 404-778-4538 ella.anastasiades@emory.edu Contact: Mersiha Torlak, MPH 404-778-4823 mersiha.torlak@emory.edu Emory Saint Joseph's Hospital Recruiting Atlanta, Georgia, United States, 30342 Contact: Ella Anastasiades, BA 404-778-4538 ella.anastasiades@emory.edu Contact: Mersiha Torlak, MPH 404-778-4823 mersiha.torlak@emory.edu Collapse << |
| NCT00769990 | Breast Cancer ... more >> Kidney Cancer Lung Cancer Melanoma Metastatic Cancer Pain Prostate Cancer Collapse << | Phase 1 Phase 2 | Withdrawn(No patient accrual.) | - | - |
| NCT01325311 | Prostate Adenocarcinoma ... more >> Stage I Prostate Cancer Stage IIA Prostate Cancer Stage IIB Prostate Cancer Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Cancer Center Birmingham, Alabama, United States, 35233 United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Hospital Baltimore, Maryland, United States, 21231 Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 United States, Massachusetts Lahey Hospital and Medical Center Burlington, Massachusetts, United States, 01805 United States, Minnesota Minneapolis Veterans Medical Center Minneapolis, Minnesota, United States, 55417 University of Minnesota Medical Center-Fairview Minneapolis, Minnesota, United States, 55455 United States, New York University of Rochester Rochester, New York, United States, 14642 United States, Oklahoma University of Oklahoma Health Sciences Center Oklahoma City, Oklahoma, United States, 73104 United States, South Carolina Carolina Urologic Research Center Myrtle Beach, South Carolina, United States, 29572 United States, Texas Urology San Antonio Research PA San Antonio, Texas, United States, 78229 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT02624388 | Lymphoma Chil... more >>dhood Lymphoma Solid Tumor Childhood Solid Tumor Neuroblastoma Ewing Sarcoma Hodgkin Lymphoma Non-Hodgkin Lymphoma Rhabdomyosarcoma Soft Tissue Sarcoma Medulloblastoma Germ Cell Tumor Wilms Tumor Brain Neoplasms Medulloblastoma, Childhood Neuroectodermal Tumors, Primitive Collapse << | Phase 2 | Recruiting | June 2019 | United States, Virginia ... more >> University of Virginia Recruiting Charlottesville, Virginia, United States, 22908 Contact: Cynthia Fischer, C.C.R.C. 434-243-0910 crb3y@virginia.edu Contact: Candace Hudspeth, C.C.R.C. 434-982-1091 ckh3k@virginia.edu Principal Investigator: William C Petersen, Jr., M.D. Collapse << |
| NCT01325311 | - | - | Completed | - | - |
| NCT02766478 | Prostate Cancer | Phase 2 | Recruiting | October 2019 | United States, Georgia ... more >> Emory University Recruiting Atlanta, Georgia, United States, 30322 Contact: Elizabeth Ivie, RD, LD, CDE 404-727-1399 elizabeth.a.ivie@emory.edu Collapse << |
| NCT02499861 | Cancer | Phase 1 Phase 2 | Completed | - | Canada, Quebec ... more >> St. Justine's Hospital Montreal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT01126879 | Adenocarcinoma of the Prostate... more >> Recurrent Prostate Cancer Stage I Prostate Cancer Stage II Prostate Cancer Stage III Prostate Cancer Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT02796794 | Sepsis | Phase 4 | Unknown | September 2016 | Turkey ... more >> Erciyes University Medical School Recruiting Kayseri, Turkey, 38039 Contact: Kudret Dogru, MD +90 352 207 6666 ext 20000 kdogru@erciyes.edu.tr Contact: Emine Alp, MD +90 352 207 6666 ext 20000 ealp@erciyes.edu.tr Collapse << |
| NCT01982578 | Alzheimer's Disease | Not Applicable | Unknown | September 2018 | Spain ... more >> Hospital de La Ribera Recruiting Alzira, Valencia, Spain, 46600 Contact: Francisco Tarazona, MD +34 962 458 100 FJTarazona@Hospital-Ribera.com Hospital Clínico Universitario de Valencia Recruiting Valencia, Spain, 46010 Contact: José Miguel Lainez, M.D. 963 86 26 00 jlaineza@medynet.com Universitat de València Recruiting Valencia, Spain, 46010 Contact: Jose Viña, MD PhD (hon) 0034 963864646 ext 64650 jose.vina@uv.es Principal Investigator: Jose Viña, MD PhD (hon) Collapse << |
| NCT00882765 | Pancreatic Cancer | Phase 2 | Withdrawn(Study has been close... more >>d due to no accrual.) Collapse << | - | United States, California ... more >> UCLA Los Angeles, California, United States, 90095 Collapse << |
| NCT01985763 | Colon Cancer ... more >>Rectal Cancer Colorectal Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| PTK | IC50:12μM/19μM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
