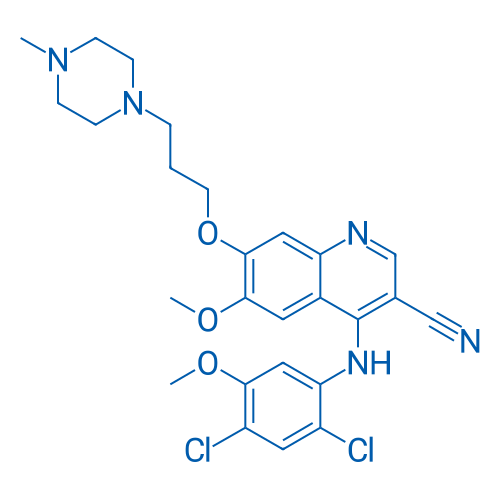
CAS No.: 380843-75-4
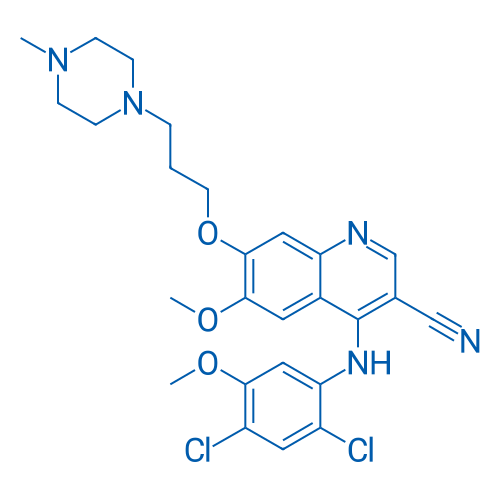
博舒替尼 Catalog No. CSN11935
Synonyms: SKI-606;SK-I606. SK 606;SK-606
Bosutinib is an inhibitor of Src and Abl with IC50 of 1.2 nM and 1 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Abl
IC50:1nMSrc
IC50:1.2nM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02551718 Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << Not Applicable Recruiting - United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << NCT03610971 Chronic Phase Chronic Myeloid ... more >>Leukemia Chronic Myeloid Leukemia, Chronic Phase Collapse << Phase 2 Not yet recruiting January 2022 United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Anthony McLaughlin 813-745-5941 anthony.mclaughlin@moffitt.org Contact: Kendra Sweet, M.D. 813-745-8986 kendra.sweet@moffitt.org Principal Investigator: Kendra Sweet, M.D. Collapse << NCT03746054 Chronic Myeloid Leukemia (CML) Phase 3 Not yet recruiting March 2023 - - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 380843-75-4 | 储存条件 |
|
|||||||||||||
| 分子式 | C26H29Cl2N5O3 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 530.45 | 别名 | SKI-606;SK-I606. SK 606;SK-606;KIN 001-160;Brand name: Bosulif;博舒替尼 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 23132-87 | - | Growth Inhibition Assay | - | IC50=15.8495 μM | SANGER |
| 5637 | - | Growth Inhibition Assay | - | IC50=1.13803 μM | SANGER |
| 647-V | - | Growth Inhibition Assay | - | IC50=29.7003 μM | SANGER |
| 697 | - | Growth Inhibition Assay | - | IC50=0.45633 μM | SANGER |
| 769-P | - | Growth Inhibition Assay | - | IC50=1.69596 μM | SANGER |
| 786-0 | - | Growth Inhibition Assay | - | IC50=9.36026 μM | SANGER |
| 8305C | - | Growth Inhibition Assay | - | IC50=7.32492 μM | SANGER |
| 8505C | - | Growth Inhibition Assay | - | IC50=3.10301 μM | SANGER |
| 8-MG-BA | - | Growth Inhibition Assay | - | IC50=5.3428 μM | SANGER |
| A101D | - | Growth Inhibition Assay | - | IC50=1.93628 μM | SANGER |
| A172 | - | Growth Inhibition Assay | - | IC50=5.54925 μM | SANGER |
| A204 | - | Growth Inhibition Assay | - | IC50=18.9164 μM | SANGER |
| A2058 | - | Growth Inhibition Assay | - | IC50=22.434 μM | SANGER |
| A2780 | - | Growth Inhibition Assay | - | IC50=6.37701 μM | SANGER |
| A375 | - | Growth Inhibition Assay | - | IC50=1.51772 μM | SANGER |
| A388 | - | Growth Inhibition Assay | - | IC50=12.7713 μM | SANGER |
| A3-KAW | - | Growth Inhibition Assay | - | IC50=0.79208 μM | SANGER |
| A427 | - | Growth Inhibition Assay | - | IC50=3.46124 μM | SANGER |
| A431 | - | Growth Inhibition Assay | - | IC50=2.37025 μM | SANGER |
| A498 | - | Growth Inhibition Assay | - | IC50=1.40113 μM | SANGER |
| A4-Fuk | - | Growth Inhibition Assay | - | IC50=1.30704 μM | SANGER |
| A549 | - | Growth Inhibition Assay | - | IC50=11.2478 μM | SANGER |
| A673 | - | Growth Inhibition Assay | - | IC50=15.9636 μM | SANGER |
| A704 | - | Growth Inhibition Assay | - | IC50=7.38278 μM | SANGER |
| ABC-1 | - | Growth Inhibition Assay | - | IC50=4.23333 μM | SANGER |
| ACHN | - | Growth Inhibition Assay | - | IC50=0.54069 μM | SANGER |
| AGS | - | Growth Inhibition Assay | - | IC50=21.8451 μM | SANGER |
| ALL-PO | - | Growth Inhibition Assay | - | IC50=8.66597 μM | SANGER |
| AsPC-1 | - | Growth Inhibition Assay | - | IC50=14.564 μM | SANGER |
| ATN-1 | - | Growth Inhibition Assay | - | IC50=39.7792 μM | SANGER |
| AU565 | - | Growth Inhibition Assay | - | IC50=15.2096 μM | SANGER |
| BALL-1 | - | Growth Inhibition Assay | - | IC50=25.2062 μM | SANGER |
| BB30-HNC | - | Growth Inhibition Assay | - | IC50=1.28714 μM | SANGER |
| BB65-RCC | - | Growth Inhibition Assay | - | IC50=1.89849 μM | SANGER |
| BE-13 | - | Growth Inhibition Assay | - | IC50=0.48572 μM | SANGER |
| Becker | - | Growth Inhibition Assay | - | IC50=26.1574 μM | SANGER |
| BEN | - | Growth Inhibition Assay | - | IC50=11.7402 μM | SANGER |
| BFTC-905 | - | Growth Inhibition Assay | - | IC50=17.4832 μM | SANGER |
| BFTC-909 | - | Growth Inhibition Assay | - | IC50=25.3551 μM | SANGER |
| BHT-101 | - | Growth Inhibition Assay | - | IC50=0.4659 μM | SANGER |
| BHY | - | Growth Inhibition Assay | - | IC50=1.22892 μM | SANGER |
| BPH-1 | - | Growth Inhibition Assay | - | IC50=1.44805 μM | SANGER |
| BT-20 | - | Growth Inhibition Assay | - | IC50=28.3685 μM | SANGER |
| BT-549 | - | Growth Inhibition Assay | - | IC50=3.67854 μM | SANGER |
| BV-173 | - | Growth Inhibition Assay | - | IC50=0.50792 μM | SANGER |
| C2BBe1 | - | Growth Inhibition Assay | - | IC50=2.03372 μM | SANGER |
| C-33-A | - | Growth Inhibition Assay | - | IC50=3.41421 μM | SANGER |
| C8166 | - | Growth Inhibition Assay | - | IC50=38.5267 μM | SANGER |
| Ca9-22 | - | Growth Inhibition Assay | - | IC50=1.5626 μM | SANGER |
| CAKI-1 | - | Growth Inhibition Assay | - | IC50=4.24571 μM | SANGER |
| CAL-120 | - | Growth Inhibition Assay | - | IC50=32.4317 μM | SANGER |
| CAL-12T | - | Growth Inhibition Assay | - | IC50=2.40417 μM | SANGER |
| CAL-27 | - | Growth Inhibition Assay | - | IC50=0.92906 μM | SANGER |
| CAL-33 | - | Growth Inhibition Assay | - | IC50=1.19302 μM | SANGER |
| CAL-39 | - | Growth Inhibition Assay | - | IC50=2.1448 μM | SANGER |
| CAL-54 | - | Growth Inhibition Assay | - | IC50=1.65709 μM | SANGER |
| CAL-72 | - | Growth Inhibition Assay | - | IC50=19.0877 μM | SANGER |
| CaR-1 | - | Growth Inhibition Assay | - | IC50=15.4412 μM | SANGER |
| Ca-Ski | - | Growth Inhibition Assay | - | IC50=5.46115 μM | SANGER |
| ChaGo-K-1 | - | Growth Inhibition Assay | - | IC50=4.59559 μM | SANGER |
| CHL-1 | - | Growth Inhibition Assay | - | IC50=0.55465 μM | SANGER |
| CHP-212 | - | Growth Inhibition Assay | - | IC50=7.51896 μM | SANGER |
| COLO-678 | - | Growth Inhibition Assay | - | IC50=14.7271 μM | SANGER |
| COLO-680N | - | Growth Inhibition Assay | - | IC50=16.5108 μM | SANGER |
| COLO-684 | - | Growth Inhibition Assay | - | IC50=38.7428 μM | SANGER |
| COLO-792 | - | Growth Inhibition Assay | - | IC50=18.7997 μM | SANGER |
| COLO-800 | - | Growth Inhibition Assay | - | IC50=42.8517 μM | SANGER |
| COLO-829 | - | Growth Inhibition Assay | - | IC50=10.8004 μM | SANGER |
| COR-L105 | - | Growth Inhibition Assay | - | IC50=1.00394 μM | SANGER |
| COR-L23 | - | Growth Inhibition Assay | - | IC50=2.79464 μM | SANGER |
| COR-L88 | - | Growth Inhibition Assay | - | IC50=7.29319 μM | SANGER |
| CTB-1 | - | Growth Inhibition Assay | - | IC50=0.5482 μM | SANGER |
| CTV-1 | - | Growth Inhibition Assay | - | IC50=0.16398 μM | SANGER |
| D-336MG | - | Growth Inhibition Assay | - | IC50=17.8294 μM | SANGER |
| D-392MG | - | Growth Inhibition Assay | - | IC50=27.7694 μM | SANGER |
| D-423MG | - | Growth Inhibition Assay | - | IC50=4.53924 μM | SANGER |
| D-502MG | - | Growth Inhibition Assay | - | IC50=7.38473 μM | SANGER |
| D-566MG | - | Growth Inhibition Assay | - | IC50=1.57155 μM | SANGER |
| Daoy | - | Growth Inhibition Assay | - | IC50=9.12028 μM | SANGER |
| DB | - | Growth Inhibition Assay | - | IC50=2.42869 μM | SANGER |
| DBTRG-05MG | - | Growth Inhibition Assay | - | IC50=28.4838 μM | SANGER |
| DEL | - | Growth Inhibition Assay | - | IC50=1.43657 μM | SANGER |
| Detroit562 | - | Growth Inhibition Assay | - | IC50=7.32506 μM | SANGER |
| DJM-1 | - | Growth Inhibition Assay | - | IC50=11.3833 μM | SANGER |
| DK-MG | - | Growth Inhibition Assay | - | IC50=26.0443 μM | SANGER |
| DOHH-2 | - | Growth Inhibition Assay | - | IC50=5.45986 μM | SANGER |
| DOK | - | Growth Inhibition Assay | - | IC50=0.37786 μM | SANGER |
| DoTc2-4510 | - | Growth Inhibition Assay | - | IC50=23.6472 μM | SANGER |
| DSH1 | - | Growth Inhibition Assay | - | IC50=3.64122 μM | SANGER |
| DU-4475 | - | Growth Inhibition Assay | - | IC50=35.1553 μM | SANGER |
| ECC10 | - | Growth Inhibition Assay | - | IC50=8.28397 μM | SANGER |
| EC-GI-10 | - | Growth Inhibition Assay | - | IC50=3.17263 μM | SANGER |
| EFM-19 | - | Growth Inhibition Assay | - | IC50=18.0577 μM | SANGER |
| EFO-27 | - | Growth Inhibition Assay | - | IC50=1.6892 μM | SANGER |
| EGI-1 | - | Growth Inhibition Assay | - | IC50=5.58938 μM | SANGER |
| EKVX | - | Growth Inhibition Assay | - | IC50=6.13501 μM | SANGER |
| EM-2 | - | Growth Inhibition Assay | - | IC50=0.4389 μM | SANGER |
| EoL-1-cell | - | Growth Inhibition Assay | - | IC50=0.41576 μM | SANGER |
| EPLC-272H | - | Growth Inhibition Assay | - | IC50=3.00235 μM | SANGER |
| ES1 | - | Growth Inhibition Assay | - | IC50=0.67498 μM | SANGER |
| ES3 | - | Growth Inhibition Assay | - | IC50=48.3529 μM | SANGER |
| ES4 | - | Growth Inhibition Assay | - | IC50=1.2079 μM | SANGER |
| ES5 | - | Growth Inhibition Assay | - | IC50=1.86747 μM | SANGER |
| ES6 | - | Growth Inhibition Assay | - | IC50=13.2512 μM | SANGER |
| ES7 | - | Growth Inhibition Assay | - | IC50=6.16115 μM | SANGER |
| ES8 | - | Growth Inhibition Assay | - | IC50=6.18082 μM | SANGER |
| ETK-1 | - | Growth Inhibition Assay | - | IC50=7.25514 μM | SANGER |
| EW-1 | - | Growth Inhibition Assay | - | IC50=49.9084 μM | SANGER |
| EW-11 | - | Growth Inhibition Assay | - | IC50=3.14277 μM | SANGER |
| EW-13 | - | Growth Inhibition Assay | - | IC50=22.1022 μM | SANGER |
| EW-16 | - | Growth Inhibition Assay | - | IC50=1.43973 μM | SANGER |
| EW-18 | - | Growth Inhibition Assay | - | IC50=28.2218 μM | SANGER |
| EW-22 | - | Growth Inhibition Assay | - | IC50=14.1502 μM | SANGER |
| EW-24 | - | Growth Inhibition Assay | - | IC50=5.04038 μM | SANGER |
| EW-3 | - | Growth Inhibition Assay | - | IC50=0.55935 μM | SANGER |
| FADU | - | Growth Inhibition Assay | - | IC50=2.32296 μM | SANGER |
| FTC-133 | - | Growth Inhibition Assay | - | IC50=19.9304 μM | SANGER |
| G-361 | - | Growth Inhibition Assay | - | IC50=5.01003 μM | SANGER |
| G-402 | - | Growth Inhibition Assay | - | IC50=43.9006 μM | SANGER |
| GAMG | - | Growth Inhibition Assay | - | IC50=9.65534 μM | SANGER |
| GB-1 | - | Growth Inhibition Assay | - | IC50=35.0729 μM | SANGER |
| GCIY | - | Growth Inhibition Assay | - | IC50=16.1916 μM | SANGER |
| GCT | - | Growth Inhibition Assay | - | IC50=7.82172 μM | SANGER |
| GI-ME-N | - | Growth Inhibition Assay | - | IC50=4.71291 μM | SANGER |
| GMS-10 | - | Growth Inhibition Assay | - | IC50=3.34086 μM | SANGER |
| GOTO | - | Growth Inhibition Assay | - | IC50=21.485 μM | SANGER |
| GP5d | - | Growth Inhibition Assay | - | IC50=4.5012 μM | SANGER |
| GT3TKB | - | Growth Inhibition Assay | - | IC50=5.44212 μM | SANGER |
| H4 | - | Growth Inhibition Assay | - | IC50=1.24255 μM | SANGER |
| H9 | - | Growth Inhibition Assay | - | IC50=0.43464 μM | SANGER |
| HAL-01 | - | Growth Inhibition Assay | - | IC50=1.0881 μM | SANGER |
| HC-1 | - | Growth Inhibition Assay | - | IC50=5.03291 μM | SANGER |
| HCC1187 | - | Growth Inhibition Assay | - | IC50=25.0235 μM | SANGER |
| HCC1395 | - | Growth Inhibition Assay | - | IC50=9.30896 μM | SANGER |
| HCC1806 | - | Growth Inhibition Assay | - | IC50=0.35216 μM | SANGER |
| HCC1937 | - | Growth Inhibition Assay | - | IC50=5.83929 μM | SANGER |
| HCC1954 | - | Growth Inhibition Assay | - | IC50=9.85859 μM | SANGER |
| HCC2218 | - | Growth Inhibition Assay | - | IC50=7.72258 μM | SANGER |
| HCC2998 | - | Growth Inhibition Assay | - | IC50=21.3926 μM | SANGER |
| HCE-T | - | Growth Inhibition Assay | - | IC50=14.8874 μM | SANGER |
| HCT-116 | - | Growth Inhibition Assay | - | IC50=2.22984 μM | SANGER |
| HCT-15 | - | Growth Inhibition Assay | - | IC50=14.8855 μM | SANGER |
| HDLM-2 | - | Growth Inhibition Assay | - | IC50=24.146 μM | SANGER |
| HD-MY-Z | - | Growth Inhibition Assay | - | IC50=1.63584 μM | SANGER |
| HEC-1 | - | Growth Inhibition Assay | - | IC50=18.2918 μM | SANGER |
| HEL | - | Growth Inhibition Assay | - | IC50=12.8212 μM | SANGER |
| H-EMC-SS | - | Growth Inhibition Assay | - | IC50=3.58569 μM | SANGER |
| HGC-27 | - | Growth Inhibition Assay | - | IC50=5.37682 μM | SANGER |
| HL-60 | - | Growth Inhibition Assay | - | IC50=3.2913 μM | SANGER |
| HLE | - | Growth Inhibition Assay | - | IC50=18.8331 μM | SANGER |
| HMV-II | - | Growth Inhibition Assay | - | IC50=14.078 μM | SANGER |
| HN | - | Growth Inhibition Assay | - | IC50=1.89874 μM | SANGER |
| HO-1-N-1 | - | Growth Inhibition Assay | - | IC50=1.26369 μM | SANGER |
| HOP-92 | - | Growth Inhibition Assay | - | IC50=4.81379 μM | SANGER |
| HOS | - | Growth Inhibition Assay | - | IC50=4.29141 μM | SANGER |
| HPAF-II | - | Growth Inhibition Assay | - | IC50=28.779 μM | SANGER |
| Hs-578-T | - | Growth Inhibition Assay | - | IC50=17.8149 μM | SANGER |
| HSC-2 | - | Growth Inhibition Assay | - | IC50=3.22232 μM | SANGER |
| HSC-3 | - | Growth Inhibition Assay | - | IC50=1.11783 μM | SANGER |
| HSC-4 | - | Growth Inhibition Assay | - | IC50=0.17158 μM | SANGER |
| HT-1080 | - | Growth Inhibition Assay | - | IC50=0.55349 μM | SANGER |
| HT-1376 | - | Growth Inhibition Assay | - | IC50=1.6436 μM | SANGER |
| HT-144 | - | Growth Inhibition Assay | - | IC50=5.72685 μM | SANGER |
| HTC-C3 | - | Growth Inhibition Assay | - | IC50=2.26824 μM | SANGER |
| HuCCT1 | - | Growth Inhibition Assay | - | IC50=3.74177 μM | SANGER |
| HuO9 | - | Growth Inhibition Assay | - | IC50=12.0666 μM | SANGER |
| HuP-T3 | - | Growth Inhibition Assay | - | IC50=18.5929 μM | SANGER |
| HuP-T4 | - | Growth Inhibition Assay | - | IC50=5.14334 μM | SANGER |
| IGROV-1 | - | Growth Inhibition Assay | - | IC50=0.48797 μM | SANGER |
| IST-MEL1 | - | Growth Inhibition Assay | - | IC50=11.7319 μM | SANGER |
| J82 | - | Growth Inhibition Assay | - | IC50=29.8013 μM | SANGER |
| JAR | - | Growth Inhibition Assay | - | IC50=1.4936 μM | SANGER |
| JEG-3 | - | Growth Inhibition Assay | - | IC50=16.25 μM | SANGER |
| J-RT3-T3-5 | - | Growth Inhibition Assay | - | IC50=1.51722 μM | SANGER |
| JVM-2 | - | Growth Inhibition Assay | - | IC50=15.2681 μM | SANGER |
| JVM-3 | - | Growth Inhibition Assay | - | IC50=1.05017 μM | SANGER |
| K5 | - | Growth Inhibition Assay | - | IC50=6.0517 μM | SANGER |
| KARPAS-299 | - | Growth Inhibition Assay | - | IC50=39.839 μM | SANGER |
| KARPAS-45 | - | Growth Inhibition Assay | - | IC50=0.92466 μM | SANGER |
| KASUMI-1 | - | Growth Inhibition Assay | - | IC50=0.7233 μM | SANGER |
| KE-37 | - | Growth Inhibition Assay | - | IC50=0.49509 μM | SANGER |
| KG-1 | - | Growth Inhibition Assay | - | IC50=1.41273 μM | SANGER |
| KGN | - | Growth Inhibition Assay | - | IC50=20.4699 μM | SANGER |
| KINGS-1 | - | Growth Inhibition Assay | - | IC50=24.242 μM | SANGER |
| KM12 | - | Growth Inhibition Assay | - | IC50=11.9091 μM | SANGER |
| KMOE-2 | - | Growth Inhibition Assay | - | IC50=8.0407 μM | SANGER |
| KNS-62 | - | Growth Inhibition Assay | - | IC50=17.6629 μM | SANGER |
| KNS-81-FD | - | Growth Inhibition Assay | - | IC50=40.3604 μM | SANGER |
| KOSC-2 | - | Growth Inhibition Assay | - | IC50=1.31775 μM | SANGER |
| KS-1 | - | Growth Inhibition Assay | - | IC50=6.17993 μM | SANGER |
| KU-19-19 | - | Growth Inhibition Assay | - | IC50=7.16476 μM | SANGER |
| KU812 | - | Growth Inhibition Assay | - | IC50=0.34068 μM | SANGER |
| KY821 | - | Growth Inhibition Assay | - | IC50=1.13351 μM | SANGER |
| KYSE-140 | - | Growth Inhibition Assay | - | IC50=1.75655 μM | SANGER |
| KYSE-150 | - | Growth Inhibition Assay | - | IC50=12.7455 μM | SANGER |
| KYSE-180 | - | Growth Inhibition Assay | - | IC50=8.08648 μM | SANGER |
| KYSE-270 | - | Growth Inhibition Assay | - | IC50=4.65126 μM | SANGER |
| KYSE-410 | - | Growth Inhibition Assay | - | IC50=16.029 μM | SANGER |
| KYSE-450 | - | Growth Inhibition Assay | - | IC50=3.96997 μM | SANGER |
| KYSE-520 | - | Growth Inhibition Assay | - | IC50=4.09799 μM | SANGER |
| KYSE-70 | - | Growth Inhibition Assay | - | IC50=26.5317 μM | SANGER |
| L-363 | - | Growth Inhibition Assay | - | IC50=2.43507 μM | SANGER |
| LAMA-84 | - | Growth Inhibition Assay | - | IC50=0.28664 μM | SANGER |
| LB1047-RCC | - | Growth Inhibition Assay | - | IC50=1.23785 μM | SANGER |
| LB2241-RCC | - | Growth Inhibition Assay | - | IC50=0.53718 μM | SANGER |
| LB771-HNC | - | Growth Inhibition Assay | - | IC50=0.83104 μM | SANGER |
| LCLC-97TM1 | - | Growth Inhibition Assay | - | IC50=28.2762 μM | SANGER |
| LoVo | - | Growth Inhibition Assay | - | IC50=1.97679 μM | SANGER |
| LOXIMVI | - | Growth Inhibition Assay | - | IC50=1.74599 μM | SANGER |
| LU-134-A | - | Growth Inhibition Assay | - | IC50=16.9893 μM | SANGER |
| LU-139 | - | Growth Inhibition Assay | - | IC50=16.1823 μM | SANGER |
| LU-65 | - | Growth Inhibition Assay | - | IC50=34.1408 μM | SANGER |
| M14 | - | Growth Inhibition Assay | - | IC50=4.48792 μM | SANGER |
| MC116 | - | Growth Inhibition Assay | - | IC50=30.0733 μM | SANGER |
| MDA-MB-175-VII | - | Growth Inhibition Assay | - | IC50=10.6396 μM | SANGER |
| MDA-MB-231 | - | Growth Inhibition Assay | - | IC50=47.6249 μM | SANGER |
| MDA-MB-361 | - | Growth Inhibition Assay | - | IC50=6.09261 μM | SANGER |
| ME-180 | - | Growth Inhibition Assay | - | IC50=3.26051 μM | SANGER |
| MEG-01 | - | Growth Inhibition Assay | - | IC50=0.51214 μM | SANGER |
| MEL-HO | - | Growth Inhibition Assay | - | IC50=4.61261 μM | SANGER |
| MEL-JUSO | - | Growth Inhibition Assay | - | IC50=2.04464 μM | SANGER |
| Mewo | - | Growth Inhibition Assay | - | IC50=4.06361 μM | SANGER |
| MFM-223 | - | Growth Inhibition Assay | - | IC50=17.1535 μM | SANGER |
| MHH-ES-1 | - | Growth Inhibition Assay | - | IC50=4.62411 μM | SANGER |
| MHH-NB-11 | - | Growth Inhibition Assay | - | IC50=1.31383 μM | SANGER |
| MHH-PREB-1 | - | Growth Inhibition Assay | - | IC50=2.80161 μM | SANGER |
| MIA-PaCa-2 | - | Growth Inhibition Assay | - | IC50=3.9729 μM | SANGER |
| MKN1 | - | Growth Inhibition Assay | - | IC50=4.83914 μM | SANGER |
| MKN45 | - | Growth Inhibition Assay | - | IC50=7.18699 μM | SANGER |
| MKN7 | - | Growth Inhibition Assay | - | IC50=27.7479 μM | SANGER |
| ML-2 | - | Growth Inhibition Assay | - | IC50=4.85626 μM | SANGER |
| MLMA | - | Growth Inhibition Assay | - | IC50=0.90675 μM | SANGER |
| MMAC-SF | - | Growth Inhibition Assay | - | IC50=23.6911 μM | SANGER |
| MN-60 | - | Growth Inhibition Assay | - | IC50=4.10501 μM | SANGER |
| MOLT-13 | - | Growth Inhibition Assay | - | IC50=25.6744 μM | SANGER |
| MOLT-16 | - | Growth Inhibition Assay | - | IC50=2.07058 μM | SANGER |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=3.48199 μM | SANGER |
| Mo-T | - | Growth Inhibition Assay | - | IC50=12.7307 μM | SANGER |
| MS-1 | - | Growth Inhibition Assay | - | IC50=43.0483 μM | SANGER |
| MSTO-211H | - | Growth Inhibition Assay | - | IC50=2.66391 μM | SANGER |
| MV-4-11 | - | Growth Inhibition Assay | - | IC50=1.69856 μM | SANGER |
| MZ2-MEL | - | Growth Inhibition Assay | - | IC50=5.61486 μM | SANGER |
| MZ7-mel | - | Growth Inhibition Assay | - | IC50=10.4753 μM | SANGER |
| NB13 | - | Growth Inhibition Assay | - | IC50=4.37355 μM | SANGER |
| NB14 | - | Growth Inhibition Assay | - | IC50=42.7263 μM | SANGER |
| NB6 | - | Growth Inhibition Assay | - | IC50=31.3629 μM | SANGER |
| NB7 | - | Growth Inhibition Assay | - | IC50=0.08231 μM | SANGER |
| NBsusSR | - | Growth Inhibition Assay | - | IC50=2.09889 μM | SANGER |
| NCI-H1048 | - | Growth Inhibition Assay | - | IC50=12.5506 μM | SANGER |
| NCI-H1155 | - | Growth Inhibition Assay | - | IC50=39.0471 μM | SANGER |
| NCI-H1299 | - | Growth Inhibition Assay | - | IC50=8.59774 μM | SANGER |
| NCI-H1304 | - | Growth Inhibition Assay | - | IC50=33.0354 μM | SANGER |
| NCI-H1395 | - | Growth Inhibition Assay | - | IC50=22.2907 μM | SANGER |
| NCI-H1563 | - | Growth Inhibition Assay | - | IC50=41.5039 μM | SANGER |
| NCI-H1573 | - | Growth Inhibition Assay | - | IC50=49.6861 μM | SANGER |
| NCI-H1581 | - | Growth Inhibition Assay | - | IC50=45.9889 μM | SANGER |
| NCI-H1648 | - | Growth Inhibition Assay | - | IC50=24.4024 μM | SANGER |
| NCI-H1650 | - | Growth Inhibition Assay | - | IC50=3.5729 μM | SANGER |
| NCI-H1666 | - | Growth Inhibition Assay | - | IC50=10.4355 μM | SANGER |
| NCI-H1693 | - | Growth Inhibition Assay | - | IC50=1.99888 μM | SANGER |
| NCI-H1703 | - | Growth Inhibition Assay | - | IC50=3.41812 μM | SANGER |
| NCI-H1755 | - | Growth Inhibition Assay | - | IC50=3.96971 μM | SANGER |
| NCI-H1770 | - | Growth Inhibition Assay | - | IC50=28.3566 μM | SANGER |
| NCI-H1792 | - | Growth Inhibition Assay | - | IC50=3.49802 μM | SANGER |
| NCI-H1793 | - | Growth Inhibition Assay | - | IC50=10.4248 μM | SANGER |
| NCI-H1838 | - | Growth Inhibition Assay | - | IC50=9.90371 μM | SANGER |
| NCI-H2009 | - | Growth Inhibition Assay | - | IC50=3.2901 μM | SANGER |
| NCI-H2029 | - | Growth Inhibition Assay | - | IC50=13.8108 μM | SANGER |
| NCI-H2030 | - | Growth Inhibition Assay | - | IC50=2.18973 μM | SANGER |
| NCI-H209 | - | Growth Inhibition Assay | - | IC50=0.40808 μM | SANGER |
| NCI-H2228 | - | Growth Inhibition Assay | - | IC50=7.41458 μM | SANGER |
| NCI-H2291 | - | Growth Inhibition Assay | - | IC50=44.8017 μM | SANGER |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=6.21816 μM | SANGER |
| NCI-H2342 | - | Growth Inhibition Assay | - | IC50=8.83101 μM | SANGER |
| NCI-H28 | - | Growth Inhibition Assay | - | IC50=17.2066 μM | SANGER |
| NCI-H292 | - | Growth Inhibition Assay | - | IC50=0.44108 μM | SANGER |
| NCI-H358 | - | Growth Inhibition Assay | - | IC50=10.4197 μM | SANGER |
| NCI-H510A | - | Growth Inhibition Assay | - | IC50=23.7939 μM | SANGER |
| NCI-H526 | - | Growth Inhibition Assay | - | IC50=1.99325 μM | SANGER |
| NCI-H630 | - | Growth Inhibition Assay | - | IC50=49.1742 μM | SANGER |
| NCI-H661 | - | Growth Inhibition Assay | - | IC50=6.18081 μM | SANGER |
| NCI-H69 | - | Growth Inhibition Assay | - | IC50=30.6093 μM | SANGER |
| NCI-H720 | - | Growth Inhibition Assay | - | IC50=4.53612 μM | SANGER |
| NCI-H82 | - | Growth Inhibition Assay | - | IC50=18.0183 μM | SANGER |
| NCI-N87 | - | Growth Inhibition Assay | - | IC50=3.30172 μM | SANGER |
| NCI-SNU-1 | - | Growth Inhibition Assay | - | IC50=12.8046 μM | SANGER |
| NCI-SNU-5 | - | Growth Inhibition Assay | - | IC50=8.83001 μM | SANGER |
| NH-12 | - | Growth Inhibition Assay | - | IC50=28.1229 μM | SANGER |
| NKM-1 | - | Growth Inhibition Assay | - | IC50=1.71802 μM | SANGER |
| NMC-G1 | - | Growth Inhibition Assay | - | IC50=6.07797 μM | SANGER |
| no-11 | - | Growth Inhibition Assay | - | IC50=7.1641 μM | SANGER |
| NOS-1 | - | Growth Inhibition Assay | - | IC50=3.64276 μM | SANGER |
| NUGC-3 | - | Growth Inhibition Assay | - | IC50=1.29356 μM | SANGER |
| OAW-42 | - | Growth Inhibition Assay | - | IC50=8.25496 μM | SANGER |
| OC-314 | - | Growth Inhibition Assay | - | IC50=0.86965 μM | SANGER |
| OCI-AML2 | - | Growth Inhibition Assay | - | IC50=47.0918 μM | SANGER |
| OE19 | - | Growth Inhibition Assay | - | IC50=49.2902 μM | SANGER |
| OE33 | - | Growth Inhibition Assay | - | IC50=37.7577 μM | SANGER |
| OMC-1 | - | Growth Inhibition Assay | - | IC50=2.85003 μM | SANGER |
| ONS-76 | - | Growth Inhibition Assay | - | IC50=1.67551 μM | SANGER |
| OS-RC-2 | - | Growth Inhibition Assay | - | IC50=2.76673 μM | SANGER |
| OVCAR-4 | - | Growth Inhibition Assay | - | IC50=2.00435 μM | SANGER |
| OVCAR-5 | - | Growth Inhibition Assay | - | IC50=8.77625 μM | SANGER |
| OVCAR-8 | - | Growth Inhibition Assay | - | IC50=43.6809 μM | SANGER |
| P12-ICHIKAWA | - | Growth Inhibition Assay | - | IC50=22.1603 μM | SANGER |
| P30-OHK | - | Growth Inhibition Assay | - | IC50=3.20117 μM | SANGER |
| PA-1 | - | Growth Inhibition Assay | - | IC50=5.81893 μM | SANGER |
| PC-14 | - | Growth Inhibition Assay | - | IC50=1.29205 μM | SANGER |
| PC-3 | - | Growth Inhibition Assay | - | IC50=45.8023 μM | SANGER |
| QIMR-WIL | - | Growth Inhibition Assay | - | IC50=1.09058 μM | SANGER |
| Ramos-2G6-4C10 | - | Growth Inhibition Assay | - | IC50=4.16739 μM | SANGER |
| RCM-1 | - | Growth Inhibition Assay | - | IC50=9.08145 μM | SANGER |
| RH-1 | - | Growth Inhibition Assay | - | IC50=12.4472 μM | SANGER |
| RMG-I | - | Growth Inhibition Assay | - | IC50=33.4285 μM | SANGER |
| RO82-W-1 | - | Growth Inhibition Assay | - | IC50=4.9298 μM | SANGER |
| RPMI-2650 | - | Growth Inhibition Assay | - | IC50=7.70312 μM | SANGER |
| RPMI-7951 | - | Growth Inhibition Assay | - | IC50=8.84157 μM | SANGER |
| RPMI-8226 | - | Growth Inhibition Assay | - | IC50=2.73164 μM | SANGER |
| RPMI-8866 | - | Growth Inhibition Assay | - | IC50=1.26106 μM | SANGER |
| RS4-11 | - | Growth Inhibition Assay | - | IC50=0.48584 μM | SANGER |
| RT-112 | - | Growth Inhibition Assay | - | IC50=16.4143 μM | SANGER |
| RVH-421 | - | Growth Inhibition Assay | - | IC50=11.152 μM | SANGER |
| RXF393 | - | Growth Inhibition Assay | - | IC50=13.9927 μM | SANGER |
| SAS | - | Growth Inhibition Assay | - | IC50=2.99415 μM | SANGER |
| SBC-1 | - | Growth Inhibition Assay | - | IC50=25.2993 μM | SANGER |
| SBC-5 | - | Growth Inhibition Assay | - | IC50=36.5902 μM | SANGER |
| SCC-15 | - | Growth Inhibition Assay | - | IC50=1.61804 μM | SANGER |
| SCC-25 | - | Growth Inhibition Assay | - | IC50=7.23414 μM | SANGER |
| SCC-4 | - | Growth Inhibition Assay | - | IC50=6.43561 μM | SANGER |
| SCC-9 | - | Growth Inhibition Assay | - | IC50=17.6285 μM | SANGER |
| SCH | - | Growth Inhibition Assay | - | IC50=3.40844 μM | SANGER |
| SF126 | - | Growth Inhibition Assay | - | IC50=17.1701 μM | SANGER |
| SF268 | - | Growth Inhibition Assay | - | IC50=18.6702 μM | SANGER |
| SF295 | - | Growth Inhibition Assay | - | IC50=2.7431 μM | SANGER |
| SF539 | - | Growth Inhibition Assay | - | IC50=15.0643 μM | SANGER |
| SH-4 | - | Growth Inhibition Assay | - | IC50=22.7045 μM | SANGER |
| SK-HEP-1 | - | Growth Inhibition Assay | - | IC50=2.00745 μM | SANGER |
| SK-LU-1 | - | Growth Inhibition Assay | - | IC50=3.18418 μM | SANGER |
| SK-MEL-1 | - | Growth Inhibition Assay | - | IC50=4.18695 μM | SANGER |
| SK-MEL-2 | - | Growth Inhibition Assay | - | IC50=2.89243 μM | SANGER |
| SK-MEL-24 | - | Growth Inhibition Assay | - | IC50=2.78224 μM | SANGER |
| SK-MEL-28 | - | Growth Inhibition Assay | - | IC50=20.3761 μM | SANGER |
| SK-MES-1 | - | Growth Inhibition Assay | - | IC50=3.24829 μM | SANGER |
| SK-N-DZ | - | Growth Inhibition Assay | - | IC50=2.81638 μM | SANGER |
| SK-NEP-1 | - | Growth Inhibition Assay | - | IC50=0.97248 μM | SANGER |
| SK-OV-3 | - | Growth Inhibition Assay | - | IC50=5.91121 μM | SANGER |
| SK-PN-DW | - | Growth Inhibition Assay | - | IC50=43.8776 μM | SANGER |
| SK-UT-1 | - | Growth Inhibition Assay | - | IC50=2.71406 μM | SANGER |
| SN12C | - | Growth Inhibition Assay | - | IC50=4.34244 μM | SANGER |
| SNU-449 | - | Growth Inhibition Assay | - | IC50=7.37651 μM | SANGER |
| SW1116 | - | Growth Inhibition Assay | - | IC50=47.0182 μM | SANGER |
| SW13 | - | Growth Inhibition Assay | - | IC50=0.73798 μM | SANGER |
| SW1573 | - | Growth Inhibition Assay | - | IC50=8.44465 μM | SANGER |
| SW1710 | - | Growth Inhibition Assay | - | IC50=5.69091 μM | SANGER |
| SW1990 | - | Growth Inhibition Assay | - | IC50=25.9655 μM | SANGER |
| SW48 | - | Growth Inhibition Assay | - | IC50=3.25486 μM | SANGER |
| SW620 | - | Growth Inhibition Assay | - | IC50=9.95357 μM | SANGER |
| SW626 | - | Growth Inhibition Assay | - | IC50=13.2485 μM | SANGER |
| SW756 | - | Growth Inhibition Assay | - | IC50=0.14024 μM | SANGER |
| SW780 | - | Growth Inhibition Assay | - | IC50=7.9906 μM | SANGER |
| SW837 | - | Growth Inhibition Assay | - | IC50=29.3385 μM | SANGER |
| SW954 | - | Growth Inhibition Assay | - | IC50=1.55007 μM | SANGER |
| SW982 | - | Growth Inhibition Assay | - | IC50=3.58829 μM | SANGER |
| T-24 | - | Growth Inhibition Assay | - | IC50=3.21814 μM | SANGER |
| T47D | - | Growth Inhibition Assay | - | IC50=6.26457 μM | SANGER |
| TE-1 | - | Growth Inhibition Assay | - | IC50=8.11057 μM | SANGER |
| TE-11 | - | Growth Inhibition Assay | - | IC50=2.38019 μM | SANGER |
| TE-12 | - | Growth Inhibition Assay | - | IC50=5.41585 μM | SANGER |
| TE-8 | - | Growth Inhibition Assay | - | IC50=26.0668 μM | SANGER |
| TE-9 | - | Growth Inhibition Assay | - | IC50=16.062 μM | SANGER |
| TGBC11TKB | - | Growth Inhibition Assay | - | IC50=18.1021 μM | SANGER |
| TGBC24TKB | - | Growth Inhibition Assay | - | IC50=13.0267 μM | SANGER |
| TI-73 | - | Growth Inhibition Assay | - | IC50=1.0165 μM | SANGER |
| TK10 | - | Growth Inhibition Assay | - | IC50=2.13458 μM | SANGER |
| TYK-nu | - | Growth Inhibition Assay | - | IC50=2.30909 μM | SANGER |
| U031 | - | Growth Inhibition Assay | - | IC50=19.4909 μM | SANGER |
| U-266 | - | Growth Inhibition Assay | - | IC50=3.97481 μM | SANGER |
| U-2-OS | - | Growth Inhibition Assay | - | IC50=12.0893 μM | SANGER |
| U-87-MG | - | Growth Inhibition Assay | - | IC50=33.0251 μM | SANGER |
| UM-UC-3 | - | Growth Inhibition Assay | - | IC50=12.8874 μM | SANGER |
| VA-ES-BJ | - | Growth Inhibition Assay | - | IC50=6.97043 μM | SANGER |
| VM-CUB-1 | - | Growth Inhibition Assay | - | IC50=8.28364 μM | SANGER |
| VMRC-RCZ | - | Growth Inhibition Assay | - | IC50=7.59066 μM | SANGER |
| YH-13 | - | Growth Inhibition Assay | - | IC50=33.2535 μM | SANGER |
| YKG-1 | - | Growth Inhibition Assay | - | IC50=3.14706 μM | SANGER |
| ZR-75-30 | - | Growth Inhibition Assay | - | IC50=47.0239 μM | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02551718 | Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << |
| NCT03610971 | Chronic Phase Chronic Myeloid ... more >>Leukemia Chronic Myeloid Leukemia, Chronic Phase Collapse << | Phase 2 | Not yet recruiting | January 2022 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Anthony McLaughlin 813-745-5941 anthony.mclaughlin@moffitt.org Contact: Kendra Sweet, M.D. 813-745-8986 kendra.sweet@moffitt.org Principal Investigator: Kendra Sweet, M.D. Collapse << |
| NCT03746054 | Chronic Myeloid Leukemia (CML) | Phase 3 | Not yet recruiting | March 2023 | - |
| NCT02269267 | Leukemia, Myeloid, Chronic | Phase 2 | Active, not recruiting | December 2021 | United States, California ... more >> Helen Diller Family Comprehensive Cancer Center University of California San Francisco, California, United States, 94143 United States, Florida Moffit Cancer Center Tampa, Florida, United States, 33612 United States, Georgia Winship Cancer Institute of Emory University Atlanta, Georgia, United States, 30322 United States, Illinois The University of Chicago Chicago, Illinois, United States, 60637 The University of Chicago Medicine Comprehensive Cancer Center at Silver Cross New Lenox, Illinois, United States, 60451 United States, Massachusetts Beth Israel Deaconess Medical Center (Satellite site of Dana Farber) Boston, Massachusetts, United States, 02215 Dana Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Michigan Karmanos Cancer Institute of Wayne State University Detroit, Michigan, United States, 48201 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Weill Medical College of Cornell University New York, New York, United States, 10021 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Texas MD Anderson Cancer Center Houston, Texas, United States, 77054 United States, Utah University of Utah Huntsman Cancer Institute Salt Lake City, Utah, United States, 84132-2408 United States, Washington Fred Hutchinson Cancer Research Center Seattle, Washington, United States, 98109-1024 United States, Wisconsin Froedtert Hospital & Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT02767063 | Leukemia, Myeloid, Chronic-Pha... more >>se Collapse << | Phase 1 Phase 2 | Recruiting | June 2021 | France ... more >> Martine GARDEMBAS Recruiting Angers, France Contact: Martine GARDEMBAS 02.41.35.44.75 MaGardembas@chu-angers.fr Pascale CONY.MAKHOUL Recruiting Annecy, France Contact: Pascale CONY.MAKHOUL 04.50.63.64.31 pconymakhoul@ch-annecygenevois.fr Thorsten BRAUN Recruiting Bobigny, France Contact: Thorsten BRAUN 01.48.95.54.58 Thorsten.braun@avc.aphp.fr Etienne Recruiting Bordeaux, France Contact: Gabriel Etienne 05.56.33.04.76 G.Etienne@bordeaux.unicancer.fr CHU Côte de Nacre Not yet recruiting Caen, France Contact: Hyacynthe Johnson-Ansah 02.31.27.25.39 Johnsonansah-a@chu-caen.fr CHU Estaing Not yet recruiting Clermont-Ferrand, France Contact: Marc BERGER mberger@chu-clermontferrand.fr CH Henri Mondor Recruiting Créteil, France Contact: ROY Lydia Rousselot Recruiting Le Chesnay, France Contact: Philippe Rousselot 01.39.63.89.09 phrousselot@ch-versailles.fr CHU Lille Recruiting Lille, France Contact: COITEUX Valérie CHU de LIMOGES Not yet recruiting Limoges, France Contact: Amélie PENOT 05.55.05.66.42 Amelie.penot@chu-limoges.fr Franck NICOLINI Recruiting Lyon, France Contact: Franck NICOLINI 04.72.11.74.01 franck.nicolini@chu-lyon.fr Institut P Calmette Recruiting Marseille, France Contact: CHARBONNIER Aude Viviane DUBRUILLE Recruiting Nantes, France Contact: Viviane DUBRUILLE 02.40.08.32.71 Viviane.dubruille@chu-nantes.fr Hopital l'Archet Not yet recruiting Nice, France Contact: Laurence LEGROS 04.92.03.58.41 legros.l@chu-nice.fr Eric JOURDAN Recruiting Nimes, France Contact: Eric JOURDAN 04.66.68.32.31 eric.jourdan@chu-nimes.fr Hôpital La Source Recruiting Orléans, France Contact: BENBRAHIM Omar Delphine REA _St louis Recruiting Paris, France Contact: Delphine REA 01.42.49.96.49 delphine.rea@aphp.fr Simona LAPUSAN_St Antoine Recruiting Paris, France Contact: Simona LAPUSAN 01.49.28.34.42 Simona.lapusan@sat.aphp.fr Cayssials Recruiting Poitiers, France Contact: Emilie Cayssials : 05.49.44. 44.72 e.cayssials@chu-poitiers.fr CHU de Rennes - Pontchaillou Recruiting Rennes, France Contact: ESCOFFRE-BARBE Martine Hôpital René Huguenin Not yet recruiting Saint-Cloud, France Contact: Sylvie GLAISNER 01.47.11.15.30 Sylvie.glaisner@curie.net Institut Universitaire contre le Cancer Not yet recruiting Toulouse, France Contact: Françoise HUGUET 05.31.15.63.56 Huguet.françoise@iuct-oncopole.fr CHU Tours Not yet recruiting Tours, France Contact: DARTIGEAS Caroline Collapse << |
| NCT00406406 | Healthy | Phase 1 | Completed | - | Netherlands ... more >> Utrecht, Netherlands, 3584 CJ Collapse << |
| NCT00499538 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States, 78217 Collapse << |
| NCT00434486 | Healthy | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Philadelphia, Pennsylvania, United States, 19148 Collapse << |
| NCT02693535 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Advanced Solid Tumors Collapse << | Phase 2 | Recruiting | - | - |
| NCT01001936 | Tumors | Phase 1 | Completed | - | - |
| NCT02921477 | Mild Cognitive Impairment ... more >> Dementia Collapse << | Phase 1 | Unknown | September 2018 | United States, California ... more >> Neurological Associates of West LA Santa Monica, California, United States, 90403 Collapse << |
| NCT02638467 | Leukemia Myel... more >>ogenous Chronic BCR-ABL Positive Collapse << | Phase 2 | Recruiting | January 2019 | Italy ... more >> ASST-Monza Recruiting Monza, Italy/MB, Italy, 20900 Contact: Carlo Gambacorti-Passerini, MD +390392339553 carlo.gambacorti@unimib.it Principal Investigator: Carlo Gambacorti-Passerini, MD Ospedale San Raffaele Recruiting Milano, MI, Italy, 20132 Contact: Fabio Ciceri, MD +390226437703 fabio.ciceri@hsr.it Collapse << |
| NCT00757341 | Healthy Subjects | Phase 1 | Completed | - | United States, Washington ... more >> Tacoma, Washington, United States, 98418 Collapse << |
| NCT00777530 | Breast Cancer ... more >> Tumors Leukemia Collapse << | Phase 1 | Completed | - | Netherlands ... more >> Utrecht, Netherlands, 3584 Collapse << |
| NCT00195260 | Neoplasms | Phase 1 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmington, Alabama, United States, 35233 United States, Arizona Pfizer Investigational Site Scottsdale, Arizona, United States, 85258 United States, California Pfizer Investigational Site Los Angeles, California, United States, 90033 United States, Florida Pfizer Investigational Site Tampa, Florida, United States, 33612 United States, Georgia Pfizer Investigational Site Atlanta, Georgia, United States, 30341 United States, Indiana Pfizer Investigational Site Indianpolis, Indiana, United States, 46202 United States, Maryland Pfizer Investigational Site Baltimore, Maryland, United States, 21287 United States, Michigan Pfizer Investigational Site Detroit, Michigan, United States, 48202 Pfizer Investigational Site Lansing, Michigan, United States, 48910 United States, New York Pfizer Investigational Site New York, New York, United States, 10016 Pfizer Investigational Site New York, New York, United States, 10032 United States, North Carolina Pfizer Investigational Site Charlotte, North Carolina, United States, 28203 Pfizer Investigational Site Charlotte, North Carolina, United States, 28211 Pfizer Investigational Site UNC Chapel Hill, North Carolina, United States, 27514 Pfizer Investigational Site UNC Chapel Hill, North Carolina, United States, 27759 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44106-1736 United States, Texas Pfizer Investigational Site San Antonio, Texas, United States, 78258 Pfizer Investigational Site Tyler, Texas, United States, 75702 United States, Washington Pfizer Investigational Site Seattle, Washington, United States, 98104 Collapse << |
| NCT00319254 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Duarte, California, United States, 91010-3000 Pfizer Investigational Site Santa Monica, California, United States, 90404 United States, Florida Pfizer Investigational Site Tampa, Florida, United States, 33612 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 Australia, New South Wales Pfizer Investigational Site Darlinghurst, New South Wales, Australia, 2010 France Pfizer Investigational Site Dijon, France, 21034 Pfizer Investigational Site Saint-Herblain, France, 44805 Hong Kong Pfizer Investigational Site Pokfulam, Hong Kong Malta Pfizer Investigational Site Floriana, Malta, VLT 14 Poland Pfizer Investigational Site Lodz, Poland, 90-553 Pfizer Investigational Site Wroclaw, Poland, 51-124 Russian Federation Pfizer Investigational Site Moscow, Russian Federation, 115478 Ukraine Pfizer Investigational Site Dnipropetrovsk, Ukraine, 49102 Pfizer Investigational Site Sumy, Ukraine, 40005 Pfizer Investigational Site Uzhgorod, Ukraine, 88014 Collapse << |
| NCT00880009 | Breast Cancer | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | United States, California ... more >> Pfizer Investigational Site Whittier, California, United States, 90603 United States, Illinois Pfizer Investigational Site Joliet, Illinois, United States, 60435 Pfizer Investigational Site Niles, Illinois, United States, 60714 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 United States, Michigan Pfizer Investigational Site Detroit, Michigan, United States, 84202 Belgium Pfizer Investigational Site Wilrijk, Belgium, 2610 China, Beijing Pfizer Investigational Site Beijing, Beijing, China, 100021 Hong Kong Pfizer Investigational Site Hong Kong, Hong Kong Hungary Pfizer Investigational Site Budapest, Hungary, 1122 Poland Pfizer Investigational Site Warszawa, Poland, 04125 Singapore Pfizer Investigational Site Singapore, Singapore, 308433 Collapse << |
| NCT00880009 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00759837 | Breast Cancer ... more >> Leukemia, Myeloid, Chronic Collapse << | Phase 1 | Completed | - | Poland ... more >> Warsaw, Poland, 02-507 Collapse << |
| NCT00914121 | Healthy Subjects | Phase 1 | Completed | - | United States, Washington ... more >> Tacoma, Washington, United States, 98418 Collapse << |
| NCT03297606 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Advanced Solid Tumors Collapse << | Phase 2 | Recruiting | September 2021 | Canada, British Columbia ... more >> BCCA - Vancouver Cancer Centre Recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Daniel John Renouf 604 877-6000 ext 672357 Canada, Ontario London Regional Cancer Program Recruiting London, Ontario, Canada, N6A 5W9 Contact: Stephen Welch 519 685-8640 Ottawa Hospital Research Institute Recruiting Ottawa, Ontario, Canada, K1H 8L6 Contact: John Hilton 613 737-7700 ext 70179 University Health Network Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Lillian Siu 416 946-2911 Canada, Quebec The Jewish General Hospital Recruiting Montreal, Quebec, Canada, H3T 1E2 Contact: Cristiano Ferrario 514 398-8307 Collapse << |
| NCT00319254 | - | - | Completed | - | - |
| NCT03747679 | CML | Phase 1 | Not yet recruiting | December 20, 2018 | Belgium ... more >> Pfizer Clinical Research Unit Not yet recruiting Brussels, Belgium, B-1070 Collapse << |
| NCT02228382 | Previously Treated PH + CML | Phase 4 | Active, not recruiting | September 18, 2021 | - |
| NCT00725426 | Healthy | Phase 1 | Completed | - | - |
| NCT00793546 | Advanced Breast Cancer | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01233882 | Renal Disease, End-Stage ... more >> Renal Insufficiency, Chronic Renal Insufficiency, Acute Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site DeLand, Florida, United States, 32720 Pfizer Investigational Site Gainesville, Florida, United States, 32608 Pfizer Investigational Site Miami, Florida, United States, 33169 Pfizer Investigational Site Orlando, Florida, United States, 32806 United States, Minnesota Pfizer Investigational Site Saint Paul, Minnesota, United States, 55114 Collapse << |
| NCT00195260 | - | - | Completed | - | - |
| NCT00952913 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site Miami, Florida, United States, 33126 Collapse << |
| NCT00811070 | Chronic Myelogenous Leukemia | Phase 2 | Completed | - | Japan ... more >> Tohoku University Hospital Sendai-city, Miyagi, Japan, 980-8574 National Cancer Center Hospital Chuo-ku, Tokyo, Japan, 104-0045 Toyohashi Municipal Hospital Aichi, Japan, 441-8570 Japanese Red Cross Nagoya First Hospital Aichi, Japan, 453-8511 Aichi Cancer Center Aichi, Japan, 464-8681 Akita University Hospital Akita, Japan, 010-8543 Chiba University Hospital Chiba, Japan, 260-8677 National Hospital Organization Kyushu Cancer Center Fukuoka, Japan, 811-1395 Harasanshin Hospital Fukuoka, Japan, 812-0033 Kobe City Medical Center General Hospital Hyogo, Japan, 650-0047 Hospital of Hyogo College of Medicine Hyogo, Japan, 663-8501 Kanazawa University Hospital Ishikawa, Japan, 920-8641 Tokai University Hospital Kanagawa, Japan, 259-1193 Kumamoto University Hospital Kumamoto, Japan, 860-8556 University Hospital,Kyoto Prefectural University of Medicine Kyoto, Japan, 602-8566 Okayama University Hospital Okayama, Japan, 700-8558 Osaka University Hospital Osaka, Japan, 565-0871 Kinki University School of Medicine Osaka, Japan, 589-8511 Saga University Hospital Saga, Japan, 846-8501 Hamamatsu Medical Univ. HP Faculty of Medicine Shizuoka, Japan, 431-3192 Tokyo Metropolitan Cancer&Infectious disease Ctr Komagome Hp Tokyo, Japan, 113-8677 Japanese Red Cross Medical Center Tokyo, Japan, 150-8935 Jikei University Hospital Daisan Tokyo, Japan, 201-8601 Collapse << |
| NCT00793546 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00811070 | - | - | Completed | - | - |
| NCT00721474 | Healthy Subjects | Phase 1 | Completed | - | United States, Washington ... more >> Tacoma, Washington, United States, 98418 Collapse << |
| NCT01233869 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 2 | Completed | - | - |
| NCT01233869 | - | - | Completed | - | - |
| NCT00934674 | Healthy | Phase 1 | Completed | - | United States, Washington ... more >> Tacoma, Washington, United States, 98418 Collapse << |
| NCT00574873 | Chronic Myeloid Leukemia | Phase 3 | Completed | - | - |
| NCT00574873 | - | - | Completed | - | - |
| NCT02501330 | - | - | Recruiting | September 22, 2023 | Japan ... more >> Recruiting Shibuya-ku, Japan, 151-8589 Collapse << |
| NCT02102633 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site DeLand, Florida, United States, 32720 Collapse << |
| NCT01080365 | Healthy | Phase 1 | Completed | - | United States, Washington ... more >> Pfizer Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT02058277 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site DeLand, Florida, United States, 32720 Collapse << |
| NCT00959946 | - | - | Terminated | - | - |
| NCT00959946 | Advanced Breast Cancer (Parts ... more >>1 and 2) Advanced Pancreatic Cancer (Part 1) Advanced Colorectal Cancer (Part 1) Advanced Cholangiocarcinoma (Part 1) Advanced Glioblastoma Multiforme (Part 1) Collapse << | Phase 1 Phase 2 | Terminated | - | United States, Massachusetts ... more >> Pfizer Investigational Site Boston, Massachusetts, United States, 02114 United States, Michigan Pfizer Investigational Site Detroit, Michigan, United States, 84202 Australia, South Australia Pfizer Investigational Site Adelaide, South Australia, Australia, 5037 Belgium Pfizer Investigational Site Edegem, Belgium, 2650 France Pfizer Investigational Site Saint Herblain, France, 44805 Hong Kong Pfizer Investigational Site Hong Kong, Hong Kong Spain Pfizer Investigational Site Madrid, Spain, 28050 Collapse << |
| NCT01374139 | Philadelphia Chromosome Positi... more >>ve (Ph+) Chronic Myeloid Leukemia (CML) Collapse << | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT02192294 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Clinical Ruddington Fields, Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT00261846 | Chronic Myeloid Leukemia | Phase 2 | Completed | - | - |
| NCT01025570 | Pancreatic Cancer | Phase 1 | Terminated(Terminated due to s... more >>low accrual) Collapse << | - | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 United States, Utah Huntsman Cancer Institute, University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT03106779 | Chronic Myelogenous Leukemia | Phase 3 | Recruiting | March 19, 2025 | - |
| NCT00261846 | - | - | Completed | - | - |
| NCT02130557 | Leukemia, Myelogenous, Chronic... more >>, Breakpoint Cluster Region-Abelson Proto-oncogene (BCR-ABL) Positive Collapse << | Phase 3 | Active, not recruiting | April 4, 2020 | - |
| NCT02445742 | Chronic Myeloblastic Leukaemia | Phase 2 | Active, not recruiting | July 2019 | Spain ... more >> C. H. U. de Gran Canaria Dr. Negrín Gran Canaria, Spain C. H. Gregorio Marañón Madrid, Spain C. U. La Paz - H. U. La Paz Madrid, Spain H. Ramón y Cajal Madrid, Spain H. U. de la Princesa Madrid, Spain H. U. Fundación Jiménez Díaz Madrid, Spain Hospital Universitario 12 de Octubre Madrid, Spain C. H. Regional de Málaga , H. General Málaga, Spain H. U. Son Espases Palma de Mallorca, Spain C. Asistencial U. de Salamanca Salamanca, Spain C. H. U. de Santiago Santiago de Compostela, Spain H. Virgen de la Salud Toledo, Spain Clínica Quirón Zaragoza S.A. Zaragoza, Spain Collapse << |
| NCT03205267 | Chronic Myelogenous Leukaemia | Phase 2 | Recruiting | October 2019 | Germany ... more >> University Hospital Bonn Recruiting Bonn, Germany, 53127 Contact: Dominik Wolf, Prof. Dr. +49 228 287 17233 dominik.wolf@ukb.uni-bonn.de Collapse << |
| NCT02546375 | - | - | Completed | - | United Kingdom ... more >> Royal Liverpool Hospital Liverpool, Merseyside, United Kingdom, L7 8XP Hammersmith Hospital London, United Kingdom, W12 0HS Nottingham University Hospital Nottingham, United Kingdom, NG5 1PB Collapse << |
| NCT01331291 | Glioblastoma | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana=Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01903733 | Chronic Myeloid Leukemia | Not Applicable | Active, not recruiting | April 3, 2020 | - |
| NCT02782403 | Leukemia | Phase 1 Phase 2 | Recruiting | March 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact pbose@mdanderson.org Collapse << |
| NCT02130557 | - | - | Active, not recruiting | - | - |
| NCT02311998 | Leukemia | Phase 1 Phase 2 | Recruiting | April 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT01331291 | - | - | Completed | - | - |
| NCT02810990 | Chronic Myeloid Leukemia | Phase 2 | Recruiting | November 2021 | Italy ... more >> S.O.C. di Ematologia - Azienda Ospedaliera - SS. Antonio e Biagio e Cesare Arrigo Not yet recruiting Alessandria, Italy Contact: Pini Principal Investigator: Pini Azienda Ospedaliero - Universitaria Ospedali Riuniti Umberto I - G.M. LANCISI - G. SALESI Recruiting Ancona, Italy Contact: Rupoli Principal Investigator: Rupoli UO Ematologia con trapianto-Università degli Studi di Bari Aldo Moro Not yet recruiting Bari, Italy Contact: Specchia Principal Investigator: Specchia Department of Oncology and Hematology, O.U. of Hematology, S. Orsola-Malpighi University Hospital, Bologna Recruiting Bologna, Italy Contact: Gianantonio Rosti Principal Investigator: Gianantonio Rosti Istituto di Ematologia "Lorenzo e A. Seragnoli" - Policlinico S. Orsola - Malpighi Recruiting Bologna, Italy Contact: Rosti Principal Investigator: Rosti ASL N.8 - Ospedale "A. Businco" - Struttura Complessa di Ematologia e CTMO Recruiting Cagliari, Italy Contact: Usala Principal Investigator: Usala Università di Catania - Cattedra di Ematologia - Ospedale "Ferrarotto" Recruiting Catania, Italy Contact: Stagno Principal Investigator: Stagno S.C. Ematologia ASO S. Croce e Carle Not yet recruiting Cuneo, Italy Contact: Rapezzi Principal Investigator: Rapezzi Arcispedale Sant'Anna Dipartimento di Scienze Mediche Sezione di Ematologia Not yet recruiting Ferrara, Italy Contact: Cavazzini Principal Investigator: Cavazzini Policlinico di Careggi Not yet recruiting Firenze, Italy Contact: Bosi Principal Investigator: Bosi Struttura Complessa di Ematologia Ospedali Riuniti Foggia Not yet recruiting Foggia, Italy Contact: Capalbo Principal Investigator: Capalbo IRCCS_AOU San Martino-IST-Ematologia 1-Monoblocco 11°piano- lato ponente Recruiting Genova, Italy Contact: Gobbi Principal Investigator: Gobbi ASL Le/1 P.O. Vito Fazzi - U.O. di Ematologia ed UTIE Recruiting Lecce, Italy Contact: Di Renzo Principal Investigator: Di Renzo Istituto Scientifico Romagnoli per lo Studio e la Cura dei Tumori- IRST Recruiting Meldola, Italy Contact: Lucchesi Principal Investigator: Lucchesi Policlinico G. Martino Dipartimento di Medicina Interna - U.O. Messina Recruiting Messina, Italy Contact: Musolino Principal Investigator: Musolino Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico UOC Oncoematologia Recruiting Milano, Italy Contact: Iurlo Principal Investigator: Iurlo U.O. Ematologia e Trapianto di MIdollo - Ist.Scientifico Ospedale San Raffaele Recruiting Milano, Italy Contact: Ciceri Principal Investigator: Ciceri Unità Trapianto di Midollo Ist. Nazionale Tumori Recruiting Milano, Italy Contact: Corradini Principal Investigator: Corradini Azienda Ospedaliera "S.Gerardo" Not yet recruiting Monza, Italy Contact: Gambacorti Principal Investigator: Gambacorti Azienda Università degli Studi di Napoli "Federico II" - Facoltà di Medicina e Chirurgia Recruiting Napoli, Italy Contact: Pane Principal Investigator: Pane S.C.D.U. Ematologia - Università del Piemonte Orientale Amedeo Avogadro Recruiting Novara, Italy Contact: Lunghi Principal Investigator: Lunghi Dip. di Scienze Cliniche e Biologiche - Ospedale S. Luigi Gonzaga-Medicina Interna 2 Recruiting Orbassano, Italy Contact: Rege Cambrin Principal Investigator: Rege Cambrin Università degli Studi di Padova - Ematologia ed Immunologia Clinica Not yet recruiting Padova, Italy Contact: Binotto Principal Investigator: Binotto U.O. di Ematologia con trapianto - A.U. Policlinico "Paolo Giaccone" Not yet recruiting Palermo, Italy Contact: Siragusa Principal Investigator: Siragusa Cattedra di Ematologia CTMO Università degli Studi di Parma Recruiting Parma, Italy Contact: Crugnola Principal Investigator: Crugnola S.C. Ematologia - Fondazione IRCCS Policlinico S. Matteo Recruiting Pavia, Italy Contact: Cazzola Principal Investigator: Cazzola Dipartimento di Oncologia ed Ematologia - AUSL Ospedale G. da Saliceto Recruiting Piacenza, Italy Contact: Vallisa Principal Investigator: Vallisa Ematologia - Ospedale San Carlo Not yet recruiting Potenza, Italy Contact: Pizzuti Principal Investigator: Pizzuti Dipartimento Oncologico - Ospedale S.Maria delle Croci Recruiting Ravenna, Italy Contact: Salvucci Principal Investigator: Salvucci Unità Operativa Complessa di Ematologia - Arcispedale S. Maria Nuova Recruiting Reggio Emilia, Italy Contact: Capodanno Principal Investigator: Capodanno Ospedale "Infermi" Recruiting Rimini, Italy Contact: Molinari Principal Investigator: Molinari U.O. di Ematologia - Centro Oncologico Basilicata Not yet recruiting Rionero in Vulture, Italy Contact: Pietrantonio Principal Investigator: Pietrantonio Complesso Ospedaliero S. Giovanni Addolorata Recruiting Roma, Italy Contact: Cedrone Principal Investigator: Cedrone Divisione Ematologia - Università Campus Bio-Medico Not yet recruiting Roma, Italy Contact: De Muro Principal Investigator: De Muro Padiglione Cesalpino - I piano - Divisione di Ematologia - Ospedale S. Camillo Recruiting Roma, Italy Contact: Mancini Principal Investigator: Mancini Università Cattolica del Sacro Cuore - Policlinico A. Gemelli Recruiting Roma, Italy Contact: Sorà Principal Investigator: Sorà UOC Pronto Soccorso - Dipartimento Biotecnologie Cellulari Università di Roma "Sapienza" Not yet recruiting Roma, Italy Contact: Breccia Principal Investigator: Breccia Istituto di Ematologia - IRCCS Ospedale Casa Sollievo della Sofferenza Recruiting S. G. Rotondo, Italy Contact: Cascavilla Principal Investigator: Cascavilla U.O.C. Ematologia e Trapianti - A.O. Senese - Policlinico " Le Scotte" Not yet recruiting Siena, Italy Contact: Bocchia Principal Investigator: Bocchia A.O. Santa Maria - Terni S.C Oncoematologia Not yet recruiting Terni, Italy Contact: Liberati Principal Investigator: Liberati Divisione di Ematologia - "Città della Salute e della Scienza di Torino" Recruiting Torino, Italy Contact: Ferrero Principal Investigator: Ferrero S.C. Ematologia 2 A.O. Città della Salute e della Scienza di Torino San Giovanni Battista Recruiting Torino, Italy Contact: Pregno Principal Investigator: Pregno Clinica Ematologica-Centro Trapianti e Terapie cellulari Not yet recruiting Udine, Italy Contact: Tiribelli Principal Investigator: Tiribelli Medicina Interna I - Ospedale di Circolo Not yet recruiting Varese, Italy Contact: Passamonti Principal Investigator: Passamonti A. O. - Istituti Ospitalieri di Verona- Div. di Ematologia - Policlinico G.B. Rossi Recruiting Verona, Italy Contact: Bonifacio Principal Investigator: Bonifacio Collapse << |
| NCT03023319 | Carcinoma, Non-Small-Cell Lung... more >> Mesothelioma Bladder Cancer Ovarian Cancer Peritoneal Cancer Thymoma Thymus Cancer Uterine Cervical Cancer Collapse << | Phase 1 | Recruiting | July 2020 | United States, Ohio ... more >> UC Health Recruiting Cincinnati, Ohio, United States, 45219 Contact: UCCI Clinical Trials Office 513-584-7698 kastla@ucmail.uc.edu Collapse << |
| NCT02546375 | - | - | Completed | - | - |
| NCT03128411 | Leukemia, Chronic Myelogenous | Phase 2 | Active, not recruiting | November 30, 2020 | Japan ... more >> Fujita Health University Hospital Toyoake-City, Aichi, Japan, 470-1192 Toyohashi Municipal Hospital Toyohashi, Aichi, Japan, 441-8570 Akita University Hospital Akita City, Akita, Japan, 010-8543 Japanese Red Cross Narita Hospital Narita, Chiba, Japan, 286-8523 Ehime University Hospital Toon-shi, Ehime, Japan, 791-0295 Kobe City Medical Center General Hospital Kobe-city, Hyogo, Japan, 650-0047 Ishikawa Prefectural Central Hospital Kanazawa, Ishikawa, Japan, 920-8530 Yokohama City University Medical Center Yokohama, Kanagawa, Japan, 232-0024 National Hospital Organization Osaka Minami Medical Center Kawachinagano, Osaka, Japan, 586-8521 Osaka City University Hospital Osaka-City, Osaka, Japan, 545-8586 Kindai University Hospital Osaka-Sayama, Osaka, Japan, 589-8511 Hamamatsu university school of medicine, university hospital Hamamatsu-shi, Shizuoka, Japan, 4313192 Juntendo University Hospital Bunkyo-ku, Tokyo, Japan, 113-8431 Nippon Medical School Hospital Bunkyo-ku, Tokyo, Japan, 113-8603 Tokyo Metropolitan Cancer and Infectious diseases Center Komagome Hospital Bunkyo-ku, Tokyo, Japan, 113-8677 NTT Medical Center Tokyo Shinagawa-ku, Tokyo, Japan, 141-8625 National Hospital Organization Disaster Medical Center Tachikawa, Tokyo, Japan, 190-0014 Yamagata University Hospital Yamagata-Shi, Yamagata, Japan, 990-9585 Chiba University Hospital Chiba, Japan, 260-8677 National Hospital Organization Kyushu Cancer Center Fukuoka, Japan, 811-1395 Saga University Hospital Saga, Japan, 849-8501 Collapse << |
| NCT02906696 | Chronic Myelogenous Leukemia | Phase 2 | Recruiting | October 2022 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| Abl | IC50:1nM | |
| Src | IC50:1.2nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
