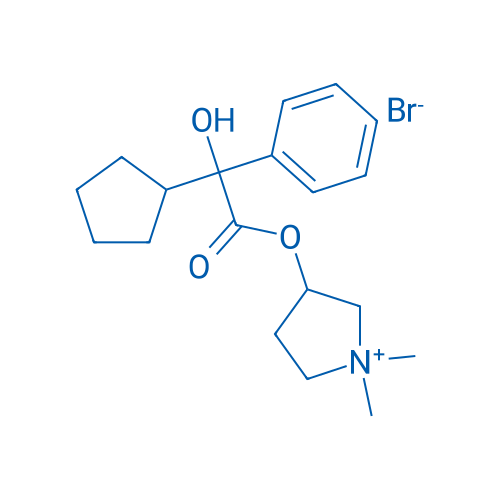
CAS No.: 596-51-0
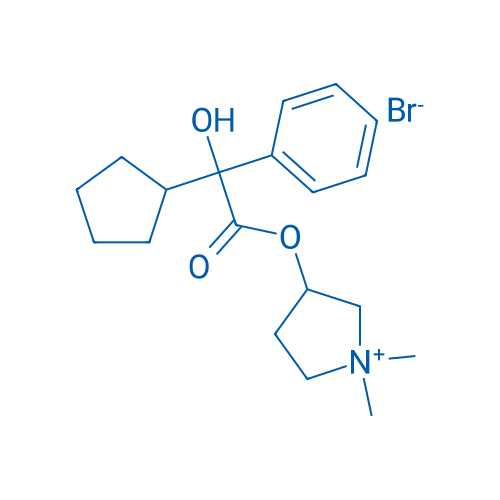
格隆溴铵 Catalog No. CSN11109
Synonyms: Glycopyrronium bromide;Glycopyrrolate bromide;Gastrodyn
Glycopyrrolate is a muscarinic competitive antagonist used as an antispasmodic.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02634983 Chronic Obstructive Pulmonary ... more >>Disease Collapse << Phase 3 Completed - United Kingdom ... more >> Novartis Investigative Site Sheffield, South Yorkshire, United Kingdom, S10 2JF Novartis Investigative Site Bradford, West Yorkshire, United Kingdom, BD9 6RJ Novartis Investigative Site Manchester, United Kingdom, M23 9QZ Collapse << NCT01511627 Hysterectomy Not Applicable Withdrawn(Research question wa... more >>s answered by Acta Anaesthesiol Scand 2012; 56 102-109.) Collapse << - Canada, Saskatchewan ... more >> Saskatoon City Hospital Saskatoon, Saskatchewan, Canada, S7K0M7 Collapse << NCT02325648 - - Completed - Korea, Republic of ... more >> Severance Hospital Seoul, South Korea, Korea, Republic of, 120-752 Collapse << - 更多
- 参考文献
- [1] Kim HC, Lim SM, et al. Effect of glycopyrrolate versus atropine coadministered with neostigmine for reversal of rocuronium on postoperative catheter-related bladder discomfort in patients undergoing transurethral resection of bladder tumor: a prospective randomized study. J Anesth. 2015 Dec;29(6):831-5.
- [2] Blissit KT, Tillery E, et al. Glycopyrrolate for treatment of clozapine-induced sialorrhea in adults. Am J Health Syst Pharm. 2014 Aug 1;71(15):1282-7.
- [3] Haddad EB, Patel H, Keeling JE, Yacoub MH, Barnes PJ, Belvisi MG. Pharmacological characterization of the muscarinic receptor antagonist, glycopyrrolate, in human and guinea-pig airways. Br J Pharmacol. 1999;127(2):413‐420
- [4] Man WH, Colen-de Koning JC, Schulte PF, et al. The Effect of Glycopyrrolate on Nocturnal Sialorrhea in Patients Using Clozapine: A Randomized, Crossover, Double-Blind, Placebo-Controlled Trial. J Clin Psychopharmacol. 2017;37(2):155‐161
- [5] Howard J, Wigley J, Rosen G, D'mello J. Glycopyrrolate: It's time to review. J Clin Anesth. 2017;36:51‐53
- [6] Olsen AK, Sjøgren P. Oral glycopyrrolate alleviates drooling in a patient with tongue cancer. J Pain Symptom Manage. 1999;18(4):300‐302
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 596-51-0 | 储存条件 |
|
|||||
| 分子式 | C19H28BrNO3 | 运输 | 蓝冰 | |||||
| 分子量 | 398.33 | 别名 | Glycopyrronium bromide;Glycopyrrolate bromide;Gastrodyn;AHR 504;NSC 250836;NSC 251251;NSC 251252;GLYCOPYRROLATE;Robinul;Asecryl;NVA-237;格隆溴铵 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02634983 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Novartis Investigative Site Sheffield, South Yorkshire, United Kingdom, S10 2JF Novartis Investigative Site Bradford, West Yorkshire, United Kingdom, BD9 6RJ Novartis Investigative Site Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT01511627 | Hysterectomy | Not Applicable | Withdrawn(Research question wa... more >>s answered by Acta Anaesthesiol Scand 2012; 56 102-109.) Collapse << | - | Canada, Saskatchewan ... more >> Saskatoon City Hospital Saskatoon, Saskatchewan, Canada, S7K0M7 Collapse << |
| NCT02325648 | - | - | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, South Korea, Korea, Republic of, 120-752 Collapse << |
| NCT03735563 | Surfactant Deficiency Syndrome... more >> Neonatal Collapse << | Phase 4 | Not yet recruiting | October 31, 2020 | - |
| NCT01703624 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02446249 | Squamous Cell Head and Neck Ca... more >>ncer Radiation Induced Xerostomia Salivary Hypofunction Collapse << | Phase 1 Phase 2 | Recruiting | November 1, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Patient Recruitment and Public Liaison Office (PRPL) 800-411-1222 ext TTY8664111010 prpl@mail.cc.nih.gov Collapse << |
| NCT03316963 | Snoring | Early Phase 1 | Recruiting | August 2019 | United States, Georgia ... more >> Raj C. Dedhia Recruiting Atlanta, Georgia, United States, 30308 Contact: Raj C Dedhia, MD, MS 404-778-3015 raj.dedhia@emory.edu Collapse << |
| NCT02684331 | - | - | Recruiting | January 2019 | Denmark ... more >> The diabetes outpatient clinic, Slagelse Hospital, Denmark Recruiting Slagelse, Denmark, 4200 Contact: Peter H Gaede, DMSc +45 23959055 phgo@regionsjaelland.dk Contact: Annemie S Bojer, DM +45 22857526 asph@regionsjaelland.dk Principal Investigator: Martin H Soerensen, DM Collapse << |
| NCT01970878 | - | - | Completed | - | - |
| NCT02622243 | Asthma | Phase 4 | Completed | - | Canada, Saskatchewan ... more >> Asthma Research Lab Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT01970878 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT01422304 | Neuromuscular Blockade ... more >> Arthroplasty, Replacement, Hip Arthroplasty, Replacement, Knee Blood Coagulation Antithrombotic Agents Collapse << | Phase 3 | Completed | - | - |
| NCT01422304 | - | - | Completed | - | - |
| NCT03114969 | - | - | Completed | - | Netherlands ... more >> GSK Investigational Site Beek En Donk, Netherlands, 5741 CG GSK Investigational Site Beek, Netherlands, 6191JW GSK Investigational Site Eindhoven, Netherlands, 5623 EJ GSK Investigational Site Hengelo, Netherlands, 7555 DL GSK Investigational Site Hoorn, Netherlands, 1624 NP GSK Investigational Site Kloosterhaar, Netherlands, 7694 AC GSK Investigational Site Nijverdal, Netherlands, 7442 LS GSK Investigational Site Rotterdam, Netherlands, 3051 GV GSK Investigational Site Zutphen, Netherlands, 7207 AE United Kingdom GSK Investigational Site Northwood, Middlesex, United Kingdom, HA6 2RN GSK Investigational Site London, United Kingdom, EC1M 6BQ GSK Investigational Site London, United Kingdom, SW17 0QT GSK Investigational Site Sidcup, Kent, United Kingdom, DA14 6LT Collapse << |
| NCT03357393 | Sedation, Bronchoscopy | Not Applicable | Completed | - | - |
| NCT02996591 | Nerve Block G... more >>eneral Anesthesia Spinal Anesthesia Pain Postoperative Nausea and Vomiting Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Hospital for Special Surgery, New York New York, New York, United States, 10021 Collapse << |
| NCT02410460 | Rhytidoplasty | Not Applicable | Completed | - | United States, Missouri ... more >> Mercy Facial Plastic Surgery Center Springfield, Missouri, United States, 65804 Collapse << |
| NCT00451100 | Anesthesia, General | Phase 3 | Completed | - | - |
| NCT03346070 | Neuromuscular Blockade | Phase 4 | Recruiting | May 31, 2019 | - |
| NCT00451217 | Anesthesia, General | Phase 3 | Completed | - | - |
| NCT02197975 | COPD | Phase 1 | Completed | - | United States, Maryland ... more >> SNBL Clinical Pharmacology Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT02574611 | Gastrointestinal Dysfunction | Phase 1 | Unknown | December 2017 | United States, New York ... more >> James J. Peters VA Medical Center Recruiting Bronx, New York, United States, 10468 Contact: Brian Lyons 718-584-9000 ext 3128 brian.lyons@va.gov Contact: Joshua Hobson 718-584-9000 ext 3129 joshua.hobson@va.gov Principal Investigator: Mark A. Korsten, MD Sub-Investigator: William A. Bauman, MD Sub-Investigator: Miroslav Radulovic, MD Sub-Investigator: Marinella D. Galea, MD Sub-Investigator: Ann M. Spungen, EdD Collapse << |
| NCT02601508 | Neuromuscular Blockade | Not Applicable | Unknown | August 2017 | Korea, Republic of ... more >> Chonnam University Hwasun Hospital Not yet recruiting Hwasun, Korea, Republic of, 519-763 Contact: Chanhong Park, BsC +82-61-379-7598 cnuhpch@naver.com Sub-Investigator: Seongheon Lee, MD, PhD Sub-Investigator: Inje Kim, BsC Sub-Investigator: Shi Young Chung, MD Principal Investigator: Seongwook Jeong, MD, PhD Collapse << |
| NCT03351608 | Neuromuscular Blockade | Phase 4 | Recruiting | September 24, 2020 | United States, California ... more >> Childrens Hospital Los Angeles ( Site 0030) Recruiting Los Angeles, California, United States, 90027 Contact: Study Coordinator 323-361-8649 Lucille Packard Children's Hospital ( Site 0006) Completed Palo Alto, California, United States, 94304 United States, District of Columbia Children's National Medical Center ( Site 0008) Recruiting Washington, District of Columbia, United States, 20010 Contact: Study Coordinator 202-476-2025 United States, Michigan C.S. Mott Children's Hospital/ University of Michigan Medical center ( Site 0014) Recruiting Ann Arbor, Michigan, United States, 48109-4245 Contact: Study Coordinator 734-763-2435 United States, New Jersey Saint Peter's University Hospital [New Brunswick, NJ] ( Site 0009) Completed New Brunswick, New Jersey, United States, 08901 United States, North Carolina Duke University Medical Center ( Site 0019) Recruiting Durham, North Carolina, United States, 27710 Contact: Study Coordinator 919-681-4877 United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC ( Site 0005) Recruiting Pittsburgh, Pennsylvania, United States, 15224 Contact: Study Coordinator 412-692-6390 Collapse << |
| NCT03513757 | Headache Tumo... more >>r Seizure Disorder Neurofibromatoses Hydrocephalus Abdominal Neoplasm Spine Deformity Collapse << | Phase 4 | Recruiting | December 31, 2018 | United States, Wisconsin ... more >> Children's Hospital of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Susan P Taylor, MD, MPH 414-266-3560 sutaylor@mcw.edu Contact: Amy Henry, MD 4142663560 ahenry@mcw.edu Collapse << |
| NCT02861131 | Postoperative Complications ... more >> Neuromuscular Blockade Collapse << | Phase 4 | Active, not recruiting | December 31, 2018 | United States, Oregon ... more >> Oregon Health and Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT01706536 | COPD | Phase 2 | Completed | - | - |
| NCT02347774 | COPD Chronic ... more >>Obstructive Pulmonary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT03574337 | Residual Neuromuscular Blockad... more >>e Collapse << | Phase 4 | Not yet recruiting | March 30, 2019 | United States, Michigan ... more >> Henry Ford Health Systems Not yet recruiting Detroit, Michigan, United States, 48202 Contact: Mary Jarzebowski, MD 313-282-5673 mjarzeb1@hfhs.org Collapse << |
| NCT02347761 | COPD | Phase 3 | Completed | - | - |
| NCT02347774 | - | - | Completed | - | - |
| NCT01706536 | - | - | Completed | - | - |
| NCT02102542 | - | - | Completed | - | Thailand ... more >> Anesthesiology Department Siriraj Hospital Bangkoknoi, Bangkok, Thailand, 10700 Collapse << |
| NCT02347761 | - | - | Completed | - | - |
| NCT02038829 | COPD | Phase 2 | Completed | - | United States, Florida ... more >> Clinical Research of West Florida, Inc. Clearwater, Florida, United States, 33765 United States, North Carolina American Health Research, Inc. Charlotte, North Carolina, United States, 28207 North Carolina Clinical Research Raleigh, North Carolina, United States, 27607 United States, Oregon Clinical Research Institute of Southern Oregon, PC Medford, Oregon, United States, 97504 United States, South Carolina Palmetto Medical Research Associates, LLC Easley, South Carolina, United States, 29640 Greenville Pharmaceutical Research, Inc. Greenville, South Carolina, United States, 29615 Upstate Pharmaceutical Research Greenville, South Carolina, United States, 29615 S. Carolina Spartanburg, South Carolina, United States, 29303 Spartanburg Medical Research Spartanburg, South Carolina, United States, 29303 CU Pharmaceutical Research Union, South Carolina, United States, 29379 Collapse << |
| NCT02038829 | - | - | Completed | - | - |
| NCT02953041 | Asthma | Phase 4 | Completed | - | Canada, Saskatchewan ... more >> Asthma Research Lab, University of Saskatchewan Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT00745095 | Spinal Cord Injury | Phase 4 | Completed | - | United States, New York ... more >> VA Medical Center, Bronx Bronx, New York, United States, 10468 Collapse << |
| NCT03583021 | Gallbladder Diseases | Not Applicable | Recruiting | December 31, 2019 | Korea, Republic of ... more >> Department of Anesthesiology and Pain Medicine, Ajou University School of Medicine Recruiting Suwon, Seoul, Korea, Republic of Contact: Ji Eun Kim 82-31-219-5575 Collapse << |
| NCT00745095 | - | - | Completed | - | - |
| NCT02189304 | COPD | Phase 1 | Completed | - | United States, Maryland ... more >> SNBL Clinical Pharmacology Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT02181023 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | Italy ... more >> Fondazione Salvatore Maugeri Milan, Italy, 20142 Collapse << |
| NCT02403063 | Respiratory Muscles ... more >> Electromyography Collapse << | Phase 4 | Completed | - | Belgium ... more >> OLV Hospital Aalst, Belgium, 9300 Collapse << |
| NCT03346057 | Reversal of Neuromuscular Bloc... more >>kade Collapse << | Phase 4 | Recruiting | December 26, 2019 | - |
| NCT02524145 | Heart Failure | Not Applicable | Completed | - | United States, Texas ... more >> The Institute for Exercise and Environmental Medicine Dallas, Texas, United States, 75231 Collapse << |
| NCT01285492 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Anjo, Aichi, Japan, 446-8602 Novartis Investigative Site Nagoya, Aichi, Japan, 457-8511 Novartis Investigative Site Nishio-city, Aichi, Japan, 445-8510 Novartis Investigative Site Kasuga-city, Fukuoka, Japan, 816-0813 Novartis Investigative Site Kitakyushu, Fukuoka, Japan, 820-0052 Novartis Investigative Site Kurume, Fukuoka, Japan, 830-0011 Novartis Investigative Site Yanagawa, Fukuoka, Japan, 832-0059 Novartis Investigative Site Asahikawa, Hokkaido, Japan, 070-8644 Novartis Investigative Site Obihiro, Hokkaido, Japan, 080-0805 Novartis Investigative Site Sapporo-city, Hokkaido, Japan, 060-8648 Novartis Investigative Site Himeji-city, Hyogo, Japan, 672-8064 Novartis Investigative Site Kanazawa, Ishikawa, Japan, 920-8610 Novartis Investigative Site Takamatsu, Kagawa, Japan, 760-8538 Novartis Investigative Site Kawasaki, Kanagawa, Japan, 210-0852 Novartis Investigative Site Yokohama, Kanagawa, Japan, 236-0051 Novartis Investigative Site Koshi-city, Kumamoto, Japan, 861-1196 Novartis Investigative Site Matsusaka-city, Mie, Japan, 515-8544 Novartis Investigative Site Ueda, Nagano, Japan, 386-8610 Novartis Investigative Site Osaka-city, Osaka, Japan, 545-8586 Novartis Investigative Site Osakasayama, Osaka, Japan, 589-0022 Novartis Investigative Site Takatsuki, Osaka, Japan, 569-1192 Novartis Investigative Site Toyonaka, Osaka, Japan, 560-8552 Novartis Investigative Site Kawaguhi-city, Saitama, Japan, 333-0833 Novartis Investigative Site Hamamatsu, Shizuoka, Japan, 430-8525 Novartis Investigative Site Fuchu, Tokyo, Japan, 183-8524 Novartis Investigative Site Meguro, Tokyo, Japan, 152-8902 Novartis Investigative Site Yamagata city, Yamagata, Japan, 990-8533 Novartis Investigative Site Ube, Yamaguchi, Japan, 755-0241 Novartis Investigative Site Akita, Japan, 010-0933 Novartis Investigative Site Fukuoka, Japan, 811-0213 Novartis Investigative Site Fukuoka, Japan, 812-0033 Novartis Investigative Site Fukuoka, Japan, 815-8588 Novartis Investigative Site Kochi, Japan, 780-8077 Novartis Investigative Site Osaka, Japan, 558-8558 Novartis Investigative Site Wakayama, Japan, 641-8510 Collapse << |
| NCT01285492 | - | - | Completed | - | - |
| NCT01120717 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT03324607 | Copd | Phase 2 Phase 3 | Recruiting | March 2019 | United States, North Carolina ... more >> Duke Asthma, Allergy, and Airway Center Recruiting Durham, North Carolina, United States, 27705 Contact: Erika Coleman 919-479-0861 Collapse << |
| NCT02284412 | Respiratory-Gated Imaging Tech... more >>niques Collapse << | Phase 4 | Completed | - | Belgium ... more >> OLV Hospital Aalst, Belgium, 9300 Collapse << |
| NCT03111121 | Microlaryngoscopy ... more >> Rigid Bronchoscopy Collapse << | Phase 4 | Recruiting | December 31, 2018 | United States, West Virginia ... more >> WVU Healthcare Ruby Memorial Hospital Recruiting Morgantown, West Virginia, United States, 26506 Contact: Pavithra Ranganathan, MD 304-598-4929 ranganathanp@wvuhealthcare.com Contact: Matthew Ellison, MD 304-598-4929 EllisonMa@wvuhealthcare.com Principal Investigator: Pavithra Ranganathan, MD Sub-Investigator: Matthew Ellison, MD Collapse << |
| NCT01768780 | Orthopedic Surgery-lower Leg S... more >>urgery Collapse << | Not Applicable | Unknown | November 2014 | Korea, Republic of ... more >> Severance hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT01120717 | - | - | Completed | - | - |
| NCT01854658 | - | - | Completed | - | - |
| NCT03742141 | Vocal Cord Function in Neck Pr... more >>ocedures Collapse << | Not Applicable | Recruiting | September 26, 2020 | United States, New York ... more >> Mount Sinai Queens Not yet recruiting Long Island City, New York, United States, 11102 Mount Sinai Beth Israel Recruiting New York, New York, United States, 10003 Mount Sinai West Recruiting New York, New York, United States, 10019 Mount Sinai St. Luke's Recruiting New York, New York, United States, 10025 The Mount Sinai Hospital Recruiting New York, New York, United States, 10029 Collapse << |
| NCT01142648 | Anaesthesia | Phase 4 | Completed | - | Finland ... more >> Tampere University Hospital Tampere, Finland Collapse << |
| NCT01587079 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Pearl Investigative Site Glendale, Arizona, United States United States, California Pearl Investigative Site Rancho Mirage, California, United States United States, Connecticut Pearl Investigative Site Waterbury, Connecticut, United States United States, Florida Pearl Investigative Site Clearwater, Florida, United States, 33765 Pearl Investigative Site Clearwater, Florida, United States Pearl Investigative Site Pensacola, Florida, United States Pearl Investigative Site Winter Park, Florida, United States, 32789 United States, Louisiana Pearl Investigative Site Lafayette, Louisiana, United States United States, Massachusetts Pearl Investigative Site North Dartmouth, Massachusetts, United States United States, Minnesota Pearl Investigative Site Fridley, Minnesota, United States United States, Missouri Pearl Investigative Site St. Louis, Missouri, United States United States, New Jersey Pearl Investigative Site Summit, New Jersey, United States United States, Ohio Pearl Investigative Site Cincinnati, Ohio, United States United States, Oregon Pearl Investigative Site Medford, Oregon, United States, 97504 Pearl Investigative Site Medford, Oregon, United States United States, South Carolina Pearl Investigative Site Rock Hill, South Carolina, United States Pearl Investigative Site Spartanburg, South Carolina, United States, 29303 United States, Texas Pearl Investigative Site San Antonio, Texas, United States United States, West Virginia Pearl Investigative Site Morgantown, West Virginia, United States Collapse << |
| NCT02370433 | Spinal Cord Injury ... more >> Neurogenic Bowel Collapse << | Phase 1 | Recruiting | December 2018 | United States, New York ... more >> James J. Peters VA Medical Center Recruiting The Bronx, New York, United States, 10468 Contact: Brian Lyons 718-584-9000 ext 3128 brian.lyons@va.gov Contact: Joshua Hobson 718-584-9000 ext 3129 joshua.hobson@va.gov Principal Investigator: Mark A. Korsten, MD Sub-Investigator: William A. Bauman, MD Sub-Investigator: Miroslav Radulovic, MD Sub-Investigator: Marinella D. Galea, MD Sub-Investigator: Ann M. Spungen, EdD Sub-Investigator: Gregory Schilero, MD Collapse << |
| NCT03513406 | Burns | Phase 3 | Active, not recruiting | March 2021 | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Collapse << |
| NCT01748643 | Obesity Lapar... more >>oscopic Gastric Bypass Surgery Surgical Conditions Respiratory Function Cerebral Tissue Oxygenation Collapse << | Phase 4 | Completed | - | Belgium ... more >> Ziekenhuis Oost-Limburg Genk, Limburg, Belgium, 3600 Collapse << |
| NCT01748643 | - | - | Completed | - | - |
| NCT03250182 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Pearl Investigative Site Clearwater, Florida, United States, 33765 Collapse << |
| NCT01854658 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT03138967 | Malignant Neoplasms of Urinary... more >> Tract Bladder Cancer Collapse << | Phase 4 | Recruiting | September 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Clinical Research Operations CR_Study_Registration@mdanderson.org Collapse << |
| NCT01199237 | - | - | Completed | - | - |
| NCT03432845 | - | - | Not yet recruiting | March 2019 | - |
| NCT00855283 | SCI | Phase 2 | Completed | - | United States, New York ... more >> VA Medical Center, Bronx Bronx, New York, United States, 10468 Collapse << |
| NCT01453530 | Anesthesia Recovery Period | Phase 4 | Unknown | September 2012 | Netherlands ... more >> UMC Sint Radboud Recruiting Nijmegen, Gelderland, Netherlands, 6500 HB Contact: Diana M Peters-Gómez Valdez, MD d.peters-gomezvaldez@anes.umcn.nl Principal Investigator: Gert J. Scheffer, MD PhD Sub-Investigator: Diana M. Peters-Gómez Valdez, MD Collapse << |
| NCT01199237 | Airway Reflexes, Protective ... more >> Recovery After Neuromuscular Block Anesthetic Recovery Collapse << | Phase 4 | Completed | - | United States, California ... more >> UCSF Helen Diller Cancer Center San Francisco, California, United States, 94115 UCSF Moffitt-Long Hospital San Francisco, California, United States, 94143 Collapse << |
| NCT00570778 | - | - | Completed | - | - |
| NCT02825576 | Pulmonary Complications | Phase 4 | Not yet recruiting | July 2020 | - |
| NCT00570778 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | United States, Missouri ... more >> Novartis Investigator Site Saint Louis, Missouri, United States United States, North Carolina Novartis Investigator Site Charlotte, North Carolina, United States Novartis Investigator site Raleigh, North Carolina, United States Belgium Novartis Investigator Site Antwerpen, Belgium Novartis Investigator Site Gent, Belgium Novartis Investigator Site Jambes, Belgium Novartis Investigator Site Saint Vith, Belgium Canada Novartis Investigator Site Moncton, Canada Novartis Investigator Site Montreal, Canada Novartis Investigator site Toronto, Canada Germany Novartis Investigator Site Bad Worishofen, Germany Novartis Investigator Site Berlin, Germany Novartis Investigator Site Frankfurt, Germany Novartis Investigator Site Mainz, Germany Novartis Investigator Site Rudersdorf, Germany Novartis Investigator Site Wiesbaden, Germany Netherlands Novartis Investigator Site Almelo, Netherlands Novartis Investigator Site Breda, Netherlands Novartis Investigator site Eindhoven, Netherlands Novartis investigator site Heerlen, Netherlands Novartis Investigator Site Nijmegen, Netherlands Novartis Investigator Site Veldhoven, Netherlands Collapse << |
| NCT02430272 | Fever | Not Applicable | Completed | - | Korea, Republic of ... more >> Ilsan Paik hospital Goyang-si, Gyeonggi-do, Korea, Republic of, 411-706 Collapse << |
| NCT03168308 | Neuromuscular Blockade | Phase 4 | Recruiting | June 30, 2019 | United States, Texas ... more >> Parkland Health & Hospital System Recruiting Dallas, Texas, United States, 75211 Contact: Tiffany S. Moon, MD Collapse << |
| NCT00558285 | - | - | Completed | - | - |
| NCT02860507 | Neuromuscular Blockade | Phase 4 | Enrolling by invitation | February 2018 | - |
| NCT00558285 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | - |
| NCT01587079 | - | - | Completed | - | - |
| NCT01176903 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 1 Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02867761 | COPD (Chronic Obstructive Pulm... more >>onary Disease) Collapse << | Phase 3 | Recruiting | July 2019 | United States, Alabama ... more >> Universityof Alabama Recruiting Birmingham, Alabama, United States, 35294 Contact: Erica Anderson eanderson@uabmc.edu Principal Investigator: Mark Dransfield United States, California University of California Recruiting Los Angeles, California, United States, 90095 Contact: Miguel Villarreal MiguelVillarreal@mednet.ucla.edu Principal Investigator: Igor Barjaktarevic, MD, PhD University of California Recruiting San Francisco, California, United States, 94143 Contact: Sundos Yassin sundos.yassin@ucsf.edu Principal Investigator: Stephen Lazarus, MD LABIOMED at Harbor-UCLA Medical Center Recruiting Torrance, California, United States, 90502 Contact: Carmen Lopez-Garcia Clopez_garcia@labiomed.org Principal Investigator: William Stringer, MD United States, Illinois University of Illinois Recruiting Chicago, Illinois, United States, 60608 Contact: Lauren Castro lschra3@uic.edu Principal Investigator: Jerry Krishnan, MD, PhD Northwestern University Recruiting Chicago, Illinois, United States, 60657 Contact: Jenny Hixon j-franzen@northwestern.edu Principal Investigator: Ravi Kalhan United States, Iowa University of Iowa Recruiting Iowa City, Iowa, United States, 52242 Contact: Fozia Ghaforr fozia-ghafoor@uiowa.edu Principal Investigator: Alejandro Comellas United States, Maryland Johns Hopkins University Recruiting Baltimore, Maryland, United States, 21224 Contact: Cheryl Clare cdaniel9@jhu.edu Principal Investigator: Bob Wise United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Crystal Cutlip ccutlip@umich.edu Principal Investigator: MeiLan Han United States, Minnesota Minneapolis VA Medical Center Recruiting Minneapolis, Minnesota, United States, 55417 Contact: Allison Aase Allison.Aase@va.gov Principal Investigator: Chris Wendt, MD Minnesota Health partners Recruiting Saint Paul, Minnesota, United States, 55130 Contact: Pamela Neuenfeldt, MPH pamela.j.neuenfeldt@healthpartners.com Principal Investigator: Charlene McEvoy United States, New York Cornell University Recruiting New York, New York, United States, 10021 Contact: Matthew Marcelino mam2225@med.cornell.edu Principal Investigator: Robert kaner, MD United States, North Carolina Duke University Recruiting Durham, North Carolina, United States, 27705 Contact: Erika Coleman erika.coleman@duke.edu Principal Investigator: Neil MacIntyre United States, Pennsylvania Temple University Recruiting Philadelphia, Pennsylvania, United States, 19140 Contact: Dee Fehrle Delores.Fehrle@tuhs.temple.edu Principal Investigator: Gerard Criner, MD University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Elizabeth Stempkowski stempkowskiem@upmc.edu Principal Investigator: Toru Nyunoya, MD United States, Texas Baylor College of Medicine Recruiting Houston, Texas, United States, 77505 Contact: Brock Melissa mbrock@bcm.edu Principal Investigator: Nicola Hanania, MD, MS United States, Utah University of Utah Recruiting Salt Lake City, Utah, United States, 84132 Contact: Newsha Sedghi u0984618@utah.edu Principal Investigator: Kanner Richard Collapse << |
| NCT02119234 | COPD | Phase 1 | Completed | - | Poland ... more >> Medical University in Lodz Lodz, ul. Kopcińskiego 22, Poland, 90-153 Collapse << |
| NCT02370862 | Spinal Cord Injury ... more >> Neurogenic Bowel Collapse << | Early Phase 1 | Completed | - | United States, New York ... more >> James J. Peters VA Medical Center Bronx, New York, United States, 10468 Collapse << |
| NCT03322657 | Neuromuscular Blockade ... more >> Anesthesia Collapse << | Phase 4 | Recruiting | October 30, 2019 | United States, Ohio ... more >> Cleveland Clinic Recruiting Cleveland, Ohio, United States, 44195 Contact: Ehab Farag, MD 216-444-7550 farage@ccf.org Contact: Roberta J Johnson 216-444-9950 johnsor13@ccf.org Collapse << |
| NCT01479764 | - | - | Completed | - | - |
| NCT02296411 | Asthma | Phase 2 | Completed | - | Bulgaria ... more >> Chiesi Clinical Trial Site 0107 Ruse, Bulgaria, 7002 Chiesi Clinical Trial Site 0106 Sevlievo, Bulgaria, 5400 Chiesi Clinical Trial Site 0101 Sofia, Bulgaria, 1000 Chiesi Clinical Trial Site 0109 Sofia, Bulgaria, 1336 Chiesi Clinical Trial Site 0108 Sofia, Bulgaria, 1407 Chiesi Clinical Trial Site 0102 Sofia, Bulgaria, 1431 Chiesi Clinical Trial Site 0103 Sofia, Bulgaria, 1431 Germany Chiesi Clinical Trial Site 0201 Berlin, Germany, D-12165 Chiesi Clinical Trial Site 0203 Leipzig, Germany, 04207 Chiesi Clinical Trial Site 0202 Leipzig, Germany, 04357 Chiesi Clinical Trial Site 0206 Magdeburg, Germany, 39112 Chiesi Clinical Trial Site 0207 Radebeul, Germany, 01445 Chiesi Clinical Trial Site 0208 Witten, Germany, 58452 Italy Chiesi Clinical Trial Site 0306 Brescia, Italy, 25123 Chiesi Clinical Trial Site 0301 Pisa, Italy, 56124 Chiesi Clinical Trial Site 0304 Pisa, Italy, 56124 Chiesi Clinical Trial Site 0303 Pordenone, Italy, 33170 Netherlands Chiesi Clinical Trial Site 0404 Assen, Netherlands, 9401 RK Chiesi Clinical Trial Site 0405 Helmond, Netherlands, 5707 HA Poland Chiesi Clinical Trial Site 0501 Bialystok, Poland, 15-010 Chiesi Clinical Trial Site 0502 Bialystok, Poland, 15-351 Chiesi Clinical Trial Site 0503 Elblag, Poland, 82-300 Chiesi Clinical Trial Site 0507 Krakow, Poland, 30-349 Chiesi Clinical Trial Site 0504 Krakow, Poland, 31-011 Chiesi Clinical Trial Site 0512 Lodz, Poland, 90-141 Chiesi Clinical Trial Site 0510 Lodz, Poland, 90-252 Chiesi Clinical Trial Site 0505 Wroclaw, Poland, 53-310 Chiesi Clinical Trial Site 0506 Zgierz, Poland, 95-100 Collapse << |
| NCT02127866 | Asthma | Phase 2 | Completed | - | - |
| NCT01479764 | Surgical Procedures, Elective | Phase 3 | Completed | - | - |
| NCT02276222 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT02276222 | - | - | Completed | - | - |
| NCT02948582 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | - |
| NCT02375217 | Anesthesia Recovery | Phase 4 | Completed | - | Lebanon ... more >> AUBMC Beirut, Lebanon Collapse << |
| NCT02948582 | - | - | Completed | - | - |
| NCT03229486 | Emergence Delirium | Phase 4 | Recruiting | April 30, 2019 | Korea, Republic of ... more >> Pusan National University Yangsan Hospital Recruiting Yangsan, Gyeongsangnam-do, Korea, Republic of, 50612 Contact: Juyeon Park, MD Collapse << |
| NCT03116997 | Surgery | Phase 3 | Recruiting | April 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: German Echeverry, MD 212-639-8865 United States, New York Memorial Sloan Kettering Cancer Center @ Commack Recruiting Commack, New York, United States, 11725 Contact: German Echeverry, MD 212-639-8865 Memoral Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: German Echeverry, MD 212-639-8865 Memorial Sloan - Kettering Cancer Center Recruiting New York, New York, United States, 10021 Contact: German Echeverry, MD 212-639-8865 Collapse << |
| NCT02040597 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 1 | Completed | - | Poland ... more >> Biovirtus Research Site Nadarzyn, Mokra 7, Poland, 05-830 Medical University in Lodz Lodz, ul. Kopcińskiego 22, Poland, 90-153 Collapse << |
| NCT01176851 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT01911364 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Germany ... more >> Institut für klinische Forschung Hessen, Germany Hungary Csongrád Megyei Mellkasi Betegségek Szakkórháza Szeged, Hungary Italy Azienda Ospedaliera Perugia Perugia, Italy Collapse << |
| NCT02613494 | Sialorrhea | Phase 1 Phase 2 | Completed | - | United Kingdom ... more >> Lancashire Care NHS Foundation Trust Preston, Lancashire, United Kingdom Mersey Care NHS Trust Liverpool, Merseyside, United Kingdom Collapse << |
| NCT01398111 | COPD | Phase 1 | Completed | - | Belgium ... more >> Clinical Pharmacology Unit - SGS Life Science Services Antwerpen, Lange Beeldekensstraat 267, Belgium, 2060 Collapse << |
| NCT01476813 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Ltd. Manchester, United Kingdom Collapse << |
| NCT02951312 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | - |
| NCT00315536 | Urological Surgery | Phase 2 | Completed | - | Belgium ... more >> University Hospital Ghent Ghent, Belgium, 9000 Collapse << |
| NCT00491894 | - | - | Completed | - | - |
| NCT02129660 | Hyperhidrosis | Phase 2 | Completed | - | United States, California ... more >> California Dermatology & Clinical Research Institute Encinitas, California, United States, 92024 Center For Dermatology Clinical Research Fremont, California, United States, 94538 United States, Florida Olympian Clinical Research Tampa, Florida, United States, 33609 Kenneth R. Beer MD West Palm Beach, Florida, United States, 33401 United States, Indiana Shideler Clinical Research Center Carmel, Indiana, United States, 46032 United States, Missouri Saint Louis University Dermatology Saint Louis, Missouri, United States, 63122 United States, Ohio Haber Dermatology and Cosmetic Surgery Beachwood, Ohio, United States, 44122 United States, Tennessee Rivergate Dermatology Clinical Research Center, PLLC Goodlettsville, Tennessee, United States, 37072 United States, Texas DermResearch, Inc Austin, Texas, United States, 78759 United States, Utah Dermatology Research Center, Inc. Salt Lake City, Utah, United States, 84117 United States, Virginia Virginia Clinical Research, Inc. Norfolk, Virginia, United States, 23507 United States, Washington Dermatology Associates Seattle, Washington, United States, 98101 Women's Clinical Research Center Seattle, Washington, United States, 98105 Premier Clinical Research Spokane, Washington, United States, 99204 Canada Innovaderm Research Inc. Montreal, Canada, H2K 4L5 Collapse << |
| NCT00491894 | Cerebral Palsy ... more >> Neurological Conditions Mental Retardation Sialorrhea Collapse << | Phase 3 | Completed | - | United States, Colorado ... more >> Rocky Mountain Pediatrics Lakewood, Colorado, United States, 80214 United States, Georgia Child Neurology Associates, PC Atlanta, Georgia, United States, 30342 United States, New York St. Mary's for Children Bayside, New York, United States, 11360 United States, Ohio Akron's Childrens Akron, Ohio, United States, 44308 Hattie Larltham Center for Children with Disabilities Mantua, Ohio, United States, 44255 United States, Oklahoma The Children's Center Bethany, Oklahoma, United States, 73008 United States, Texas Alamo City Clinical Research San Antonio, Texas, United States, 78258 Collapse << |
| NCT02189577 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | Bulgaria ... more >> Chiesi Clinical Trial Site 1017 Sevlievo, Bulgaria Chiesi Clinical Trial Site 1010 Sofia, Bulgaria Chiesi Clinical Trial Site 1011 Sofia, Bulgaria Chiesi Clinical Trial Site 1014 Sofia, Bulgaria Chiesi Clinical Trial Site 1015 Sofia, Bulgaria Chiesi Clinical Trial Site 1016 Sofia, Bulgaria Chiesi Clinical Trial Site 1012 Stara Zagora, Bulgaria Chiesi Clinical Trial Site 1013 Troyan, Bulgaria Germany Chiesi Clinical Trial Site 2024 Berlin, Germany Chiesi Clinical Trial Site 2028 Berlin, Germany Site 2020 - Pulmonary Research Institute at Hospital Grosshansdorf Großhansdorf, Germany, D-22927 Chiesi Clinical Trial Site 2027 Hamburg, Germany Chiesi Clinical Trial Site 2026 Leipzig, Germany Chiesi Clinical Trial Site 2023 Magdeburg, Germany Chiesi Clinical Trial Site 2021 Radebeul, Germany Chiesi Clinical Trial Site 2022 Witten, Germany Poland Chiesi Clinical Trial Site 3039 Bydgoszcz, Poland Chiesi Clinical Trial Site 3032 Katowice, Poland Chiesi Clinical Trial Site 3035 Kraków, Poland Chiesi Clinical Trial Site 3037 Lubin, Poland Chiesi Clinical Trial Site 3031 Ostrowiec Świętokrzyski, Poland Chiesi Clinical Trial Site 3033 Oświęcim, Poland Chiesi Clinical Trial Site 3038 Rzeszów, Poland Chiesi Clinical Trial Site 3034 Tarnów, Poland Chiesi Clinical Trial Site 3030 Wrocław, Poland Chiesi Clinical Trial Site 3036 Zgierz, Poland United Kingdom Site 4040 - Royal Brompton and Harefield NHS Foundation Trust; South Kensington Campus London, United Kingdom, SW7 2AZ Chiesi Clinical Trial Site 4042 London, United Kingdom Chiesi Clinical Trial Site 4041 Manchester, United Kingdom Collapse << |
| NCT01917331 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Hungary ... more >> Dr Beatrix BALINT Szeged, Hungary, 6722 Collapse << |
| NCT02512302 | COPD | Phase 1 | Completed | - | United Kingdom ... more >> Queen Anne Street Medical Center London, United Kingdom, W1G 8HU Medicines evaluation Unit Ltd. Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02512302 | - | - | Completed | - | - |
| NCT01191398 | Sialorrhea | Not Applicable | Completed | - | United States, Texas ... more >> Children's Medical Center at Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT01191398 | - | - | Completed | - | - |
| NCT02382198 | Sialorrhea Pa... more >>rkinson's Disease Collapse << | Phase 2 | Recruiting | December 2018 | Canada, Ontario ... more >> The Ottawa Hospital - Civic Campus Recruiting Ottawa, Ontario, Canada, K1Y 4E9 Contact: Shawna Reddie 613-798-5555 ext 19369 sreddie@ohri.ca Contact: Tiago Mestre, MSc, MD 613-798-5555 ext 18986 tmestre@toh.ca Principal Investigator: Tiago Mestre, MSc, MD Toronto Western Hospital Recruiting Toronto, Ontario, Canada, M5T 2S8 Contact: Alison Tian 416-603-5800 ext 3415 Alison.Tian@uhnresearch.ca Contact: Susan Fox, MBChB, MRCP, PhD 416-699-9837 Sfox@uhnresearch.ca Principal Investigator: Susan Fox, MBChB, MRCP, PhD Collapse << |
| NCT02129660 | - | - | Completed | - | - |
| NCT02951312 | - | - | Completed | - | - |
| NCT02872935 | Nausea Vomiti... more >>ng Collapse << | Phase 4 | Recruiting | December 31, 2018 | United States, Iowa ... more >> University of Iowa Recruiting Iowa City, Iowa, United States, 52242 Contact: Kokila N Thenuwara, MD 319-541-8634 kokila-thenuwara@uiowa.edu Principal Investigator: Kokila N Thenuwara, MD Collapse << |
| NCT02016885 | Hyperhidrosis | Phase 2 | Completed | - | United States, Arkansas ... more >> Burke Pharmaceutical Research Hot Springs, Arkansas, United States, 71913 Gary M. Petrus, MD, PA Little Rock, Arkansas, United States, 72202 United States, California California Dermatology & Clinical Research Institute Encinitas, California, United States, 92024 Center For Dermatology Clinical Research Fremont, California, United States, 94538 United States, Florida Florida Academic Dermatology Center Miami, Florida, United States, 33136 Kenneth R. Beer, MD, PA West Palm Beach, Florida, United States, 33401 United States, Indiana Shideler Clinical Research Center Carmel, Indiana, United States, 46032 United States, Kansas Cypress Medical Research Center, LLC Wichita, Kansas, United States, 67226 United States, Minnesota Minnesota Clinical Study Center Fridley, Minnesota, United States, 55432 United States, Missouri Saint Louis University Dermatology Saint Louis, Missouri, United States, 63122 United States, Nebraska Skin Specialists, PC Omaha, Nebraska, United States, 68144 United States, New Jersey The Dermatology Group, PC Verona, New Jersey, United States, 07044 United States, Oregon Oregon Medical Research Center Portland, Oregon, United States, 97223 United States, Texas J&S Studies, Inc. College Station, Texas, United States, 77845 Clinical Trials of Texas, Inc. San Antonio, Texas, United States, 78229 United States, Utah Dermatology Research Center, Inc. Salt Lake City, Utah, United States, 84117 United States, Virginia Virginia Clinical Research, Inc. Norfolk, Virginia, United States, 23507 Westend Dermatology Associates Richmond, Virginia, United States, 23233 United States, Washington Dermatology Associates Seattle, Washington, United States, 98101 Women's Clinical Research Center Seattle, Washington, United States, 98105 Premier Clinical Research Spokane, Washington, United States, 99204 Collapse << |
| NCT03081156 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Recruiting | December 31, 2019 | United States, California ... more >> Los Angeles Biomedical Institute, Inc (LABIOMED) Recruiting Torrance, California, United States, 90509 Contact: William Stringer, MD 424-571-7626 wstringer@labiomed.org Contact: Leticia Diaz, RN 310-222-8022 ext 7258 ldiaz@labiomed.org Principal Investigator: William W Stringer, MD Collapse << |
| NCT02016885 | - | - | Completed | - | - |
| NCT03580889 | Lower Extremity Fracture | Phase 4 | Recruiting | September 2018 | Nepal ... more >> Dr. BishnuPokharel Recruiting Dharān Bāzār, Sunsari, Nepal, 977 Contact: Bishnu Pokharel, Dr. 025525555 Bpkihs@bpkihs.edu Principal Investigator: Raju Thapamagar, Dr Collapse << |
| NCT03322098 | Spinal Anesthesia | Not Applicable | Completed | - | Korea, Republic of ... more >> Department of Anesthesiology and Pain Medicine, Anesthesia and Pain Research Institute, Yonsei University College of Medicine Seoul, Korea, Republic of, 03722 Collapse << |
| NCT01588717 | Hyperhydrosis ... more >> Depression Collapse << | Phase 1 | Terminated(PI left institution... more >>) Collapse << | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT03050242 | Cholecystitis, Acute | Not Applicable | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Hospital Seoul, Korea, Republic of, 03181 Collapse << |
| NCT03322150 | Spinal Anesthesia | Not Applicable | Recruiting | October 2018 | Korea, Republic of ... more >> Department of Anesthesiology and Pain Medicine, Anesthesia and Pain Research Institute, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 03722 Contact: Ki-Young Lee, MD Collapse << |
| NCT02228473 | Catheter Site Discomfort ... more >> Complications Anesthesia Urinary Bladder Neoplasms Collapse << | Not Applicable | Unknown | March 2015 | Korea, Republic of ... more >> Seoul National University of Hospital Recruiting Seoul, Korea, Republic of, Korea, Republic of Contact: Hee-Pyoung Park, PhD +82-10-2971-7647 hppark@snu.ac.kr Principal Investigator: Hyun-Chang Kim, MD Principal Investigator: Hee-Pyoung Park, PhD Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
