| NCT01555281 | Multiple Myeloma | Phase 1
Phase 2 | Active, not recruiting | December 2019 | Italy
... more >> Istituto Europeo di Oncologia IEO
Milano, Italy, 20141
University of Torino
Torino, Italy, 10127
Switzerland
Kantonsspital Aarau
Aarau, Switzerland, 5001
Kantonsspital Baden
Baden, Switzerland, 5404
Istituto Oncologico Svizzera Italiana IOSI
Bellinzona, Switzerland, 6500
Inselspital Bern
Bern, Switzerland, 3010
Kantonsspital Graubünden
Chur, Switzerland, 7000
Kantonsspital Olten
Olten, Switzerland, 4600
Kantonsspital St. Gallen
St. Gallen, Switzerland, 9007
Regionalspital
Thun, Switzerland, 3600
UniversitätsSpital Zürich
Zürich, Switzerland, 8091 Collapse << |
| NCT00055380 | Cancer | Phase 1 | Completed | - | United States, Maryland
... more >>
National Institute of Dental And Craniofacial Research (NIDCR)
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00053794 | Endometrial Cancer
... more >> Sarcoma Collapse << | Phase 2 | Completed | - | Canada, Alberta
... more >>
Tom Baker Cancer Center - Calgary
Calgary, Alberta, Canada, T2N 4N2
Canada, British Columbia
British Columbia Cancer Agency - Vancouver Island Cancer Centre
Vancouver, British Columbia, Canada, V5Z 4E6
Canada, Ontario
Margaret and Charles Juravinski Cancer Centre
Hamilton, Ontario, Canada, L8V 5C2
Cancer Care Ontario-London Regional Cancer Centre
London, Ontario, Canada, N6A 4L6
Mount Sinai Hospital - Toronto
Toronto, Ontario, Canada, M5G 1X5
Canada, Quebec
Maisonneuve-Rosemont Hospital
Montreal, Quebec, Canada, H1T 2M4 Collapse << |
| NCT00053781 | Melanoma (Skin) | Phase 2 | Completed | - | Canada, Alberta
... more >>
Tom Baker Cancer Center - Calgary
Calgary, Alberta, Canada, T2N 4N2
Canada, Nova Scotia
Nova Scotia Cancer Centre
Halifax, Nova Scotia, Canada, B3H 1V7
Canada, Ontario
Cancer Care Ontario-London Regional Cancer Centre
London, Ontario, Canada, N6A 4L6
Princess Margaret Hospital at University Health Network
Toronto, Ontario, Canada, M5G 2M9
Canada, Quebec
Centre Hospitalier de l'Universite de Montreal
Montreal, Quebec, Canada, H2L-4M1
McGill University
Montreal, Quebec, Canada, H2W 1S6 Collapse << |
| NCT00019656 | Leukemia
Lymp... more >>homa
Myelodysplastic Syndromes
Myelodysplastic/Myeloproliferative Neoplasms
Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Maryland
... more >>
Warren Grant Magnuson Clinical Center - NCI Clinical Studies Support
Bethesda, Maryland, United States, 20892-1182
NCI - Center for Cancer Research
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00054145 | Breast Cancer | Phase 2 | Completed | - | Canada, Ontario
... more >>
Margaret and Charles Juravinski Cancer Centre
Hamilton, Ontario, Canada, L8V 5C2
Cancer Care Ontario-London Regional Cancer Centre
London, Ontario, Canada, N6A 4L6
Ottawa Regional Cancer Centre
Ottawa, Ontario, Canada, K1H 1C4
Princess Margaret Hospital
Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00060437 | Prostate Cancer | Phase 2 | Completed | - | United States, Maryland
... more >>
Warren Grant Magnuson Clinical Center - NCI Clinical Studies Support
Bethesda, Maryland, United States, 20892-1182 Collapse << |
| NCT00005794 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Wisconsin
... more >>
University of Wisconsin Comprehensive Cancer Center
Madison, Wisconsin, United States, 53792-6164 Collapse << |
| NCT00053924 | Pancreatic Cancer | Phase 2 | Completed | - | Canada, Ontario
... more >>
Margaret and Charles Juravinski Cancer Centre
Hamilton, Ontario, Canada, L8V 5C2
Cancer Care Ontario-London Regional Cancer Centre
London, Ontario, Canada, N6A 4L6
Princess Margaret Hospital
Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00064324 | Recurrent Adult Soft Tissue Sa... more >>rcoma
Stage III Adult Soft Tissue Sarcoma
Stage IV Adult Soft Tissue Sarcoma Collapse << | Phase 2 | Completed | - | United States, Minnesota
... more >>
Mayo Clinic
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00431054 | Ovarian Cancer | Phase 1 | Completed | - | United States, Texas
... more >>
UT MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01097018 | Colorectal Cancer | Phase 3 | Completed | - | - |
| NCT00399126 | Neoplasms | Phase 1 | Completed | - | United States, Tennessee
... more >>
AOI Pharmaceuticals Investigative Site
Johnson City, Tennessee, United States, 37604 Collapse << |
| NCT00399087 | Neoplasms | Phase 1 | Completed | - | United States, Tennessee
... more >>
Investigative Site
Johnson City, Tennessee, United States, 37604 Collapse << |
| NCT00062387 | Head and Neck Cancer | Phase 2 | Terminated(Administratively co... more >>mplete.) Collapse << | - | United States, Illinois
... more >>
University of Chicago Cancer Research Center
Chicago, Illinois, United States, 60637-1470
Decatur Memorial Hospital Cancer Care Institute
Decatur, Illinois, United States, 62526
Evanston Northwestern Health Care - Evanston Hospital
Evanston, Illinois, United States, 60201-1781
Ingalls Memorial Hospital
Harvey, Illinois, United States, 60426
LaGrange Memorial Hospital
LaGrange, Illinois, United States, 60525
Cardinal Bernardin Cancer Center at Loyola University Medical Center
Maywood, Illinois, United States, 60153
Oncology/Hematology Associates of Central Illinois, P.C.
Peoria, Illinois, United States, 61602
Central Illinois Hematology Oncology Center
Springfield, Illinois, United States, 62701
United States, Indiana
Fort Wayne Medical Oncology and Hematology, Incorporated
Fort Wayne, Indiana, United States, 46885-5099
CCOP - Northern Indiana CR Consortium
South Bend, Indiana, United States, 46601
United States, Michigan
Oncology Care Associates, P.L.L.C.
Saint Joseph, Michigan, United States, 49085
United States, Wisconsin
Medical College of Wisconsin Cancer Center
Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00301938 | Accelerated Phase Chronic Myel... more >>ogenous Leukemia
Adult Acute Megakaryoblastic Leukemia (M7)
Adult Acute Minimally Differentiated Myeloid Leukemia (M0)
Adult Acute Monoblastic Leukemia (M5a)
Adult Acute Monocytic Leukemia (M5b)
Adult Acute Myeloblastic Leukemia With Maturation (M2)
Adult Acute Myeloblastic Leukemia Without Maturation (M1)
Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities
Adult Acute Myeloid Leukemia With Inv(16)(p13;q22)
Adult Acute Myeloid Leukemia With t(15;17)(q22;q12)
Adult Acute Myeloid Leukemia With t(16;16)(p13;q22)
Adult Acute Myeloid Leukemia With t(8;21)(q22;q22)
Adult Acute Myelomonocytic Leukemia (M4)
Adult Acute Promyelocytic Leukemia (M3)
Adult Erythroleukemia (M6a)
Adult Pure Erythroid Leukemia (M6b)
Blastic Phase Chronic Myelogenous Leukemia
Myelodysplastic/Myeloproliferative Neoplasms
Previously Treated Myelodysplastic Syndromes
Recurrent Adult Acute Lymphoblastic Leukemia
Recurrent Adult Acute Myeloid Leukemia
Relapsing Chronic Myelogenous Leukemia
Secondary Acute Myeloid Leukemia
T-cell Adult Acute Lymphoblastic Leukemia
Untreated Adult Acute Lymphoblastic Leukemia
Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Completed | - | United States, Maryland
... more >>
University of Maryland Greenebaum Cancer Center
Baltimore, Maryland, United States, 21201-1595 Collapse << |
| NCT00415064 | Multiple Myeloma | Phase 1 | Completed | - | United States, Michigan
... more >>
Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00398697 | Neoplasms | Phase 1 | Completed | - | United States, Tennessee
... more >>
Johnson City, Tennessee, United States, 37604 Collapse << |
| NCT02238496 | Brain Tumor, Recurrent
... more >> Glioblastoma
Anaplastic Astrocytoma
Anaplastic Oligodendroglioma
Mixed Glioma Collapse << | Phase 2 | Unknown | December 2016 | United States, New York
... more >>
Columbia University Medical Center
New York City, New York, United States, 10032
Memorial Sloan-Kettering Cancer Center
New York City, New York, United States, 10065 Collapse << |
| NCT00059982 | Pancreatic Cancer | Phase 2 | Completed | - | United States, Illinois
... more >>
CCOP - Carle Cancer Center
Urbana, Illinois, United States, 61801
United States, Indiana
CCOP - Northern Indiana CR Consortium
South Bend, Indiana, United States, 46601
United States, Iowa
CCOP - Iowa Oncology Research Association
Des Moines, Iowa, United States, 50309-1016
United States, Massachusetts
Tufts - New England Medical Center
Boston, Massachusetts, United States, 02111
United States, Minnesota
CCOP - Duluth
Duluth, Minnesota, United States, 55805
Mayo Clinic Cancer Center
Rochester, Minnesota, United States, 55905
CCOP - Metro-Minnesota
Saint Louis Park, Minnesota, United States, 55416
United States, New York
MBCCOP-Our Lady of Mercy Cancer Center
Bronx, New York, United States, 10466
United States, North Dakota
CCOP - Merit Care Hospital
Fargo, North Dakota, United States, 58122
United States, Texas
CCOP - Scott and White Hospital
Temple, Texas, United States, 76508 Collapse << |
| NCT00399152 | Renal Cancer
... more >>GIST Collapse << | Phase 1 | Completed | - | - |
| NCT00398814 | Renal Cancer
... more >>Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT00058214 | Adenocarcinoma of the Prostate... more >>
Recurrent Prostate Cancer
Stage IIB Prostate Cancer
Stage III Prostate Cancer
Stage IV Prostate Cancer Collapse << | Phase 2 | Terminated(Did not meet interi... more >>m stopping criteria for continuation to the second stage.) Collapse << | - | United States, California
... more >>
UC Davis Cancer Center
Sacramento, California, United States, 95817 Collapse << |
| NCT00401388 | Chondrosarcomas
... more >> Alveolar Soft Part Sarcomas
Extra Skeletal Myxoid Chondrosarcomas Collapse << | Phase 2 | Completed | - | - |
| NCT00422656 | Waldenstrom's Macroglobulinemi... more >>a Collapse << | Phase 2 | Completed | - | United States, Massachusetts
... more >>
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00422656 | - | - | Completed | - | - |
| NCT01051557 | - | - | Terminated | - | - |
| NCT00399789 | Non Small Cell Lung Cancer | Phase 1
Phase 2 | Completed | - | United States, Arizona
... more >>
AOI Pharmaceuticals Investigative Site
Tucson, Arizona, United States, 85704
United States, California
AOI Pharmaceuticals Investigative Site
Pomona, California, United States, 91767
United States, Florida
AOI Pharmaceuticals Investigative Site
Aventura, Florida, United States, 33180
AOI Pharmaceuticals Investigative Site
Hollywood, Florida, United States, 33021
AOI Pharmaceuticals Investigative Site
Lakeland, Florida, United States, 33805
AOI Pharmaceuticals Investigative Site
Ormond Beach, Florida, United States, 32174
United States, Georgia
AOI Pharmaceuticals Investigative Site
Lawrenceville, Georgia, United States, 30045
United States, Illinois
AOI Pharmaceuticals Investigative Site
Galesburg, Illinois, United States, 61401
United States, Indiana
AOI Pharmaceuticals Investigative Site
New Albany, Indiana, United States, 47150
United States, Michigan
AOI Pharmaceuticals Investigative Site
Grand Rapids, Michigan, United States, 49546
AOI Pharmaceuticals Investigative Site
Kalamazoo, Michigan, United States, 49048
United States, Montana
AOI Pharmaceuticals Investigative Site
Billings, Montana, United States, 59101
United States, New Mexico
AOI Pharmaceuticals Investigative Site
Albuquerque, New Mexico, United States, 87109
United States, New York
AOI Pharmaceuticals Investigative Site
Albany, New York, United States, 12208
AOI Pharmaceuticals Investigative Site
Armonk, New York, United States, 10504
United States, South Carolina
AOI Pharmaceuticals Investigative Site
Greenville, South Carolina, United States, 29605
United States, Tennessee
AOI Pharmaceuticals Investigative Site
Chattanooga, Tennessee, United States, 37404
AOI Pharmaceuticals Investigative Site
Nashville, Tennessee, United States, 37203
United States, Texas
AOI Pharmaceuticals Investigative Site
Dallas, Texas, United States, 75237
AOI Pharmaceuticals Investigative Site
Dallas, Texas, United States, 75246
AOI Pharmaceuticals Investigative Site
Tyler, Texas, United States, 75702 Collapse << |
| NCT01051557 | Adult Anaplastic Astrocytoma
... more >> Adult Anaplastic Oligodendroglioma
Adult Diffuse Astrocytoma
Adult Giant Cell Glioblastoma
Adult Glioblastoma
Adult Gliosarcoma
Adult Mixed Glioma
Adult Oligodendroglioma
Recurrent Adult Brain Neoplasm Collapse << | Phase 1
Phase 2 | Terminated | - | United States, New Jersey
... more >>
Memorial Sloan Kettering Basking Ridge
Basking Ridge, New Jersey, United States, 07920
United States, New York
Memorial Sloan Kettering Commack
Commack, New York, United States, 11725
Memorial Sloan-Kettering Cancer Center
New York, New York, United States, 10065 Collapse << |
| NCT00058214 | - | - | Terminated(Did not meet interi... more >>m stopping criteria for continuation to the second stage.) Collapse << | - | - |
| NCT00401011 | Multiple Myeloma
... more >> Multiple Myeloma in Relapse Collapse << | Phase 1
Phase 2 | Completed | - | - |
| NCT00391560 | Leukemia | Phase 2 | Completed | - | - |
| NCT00398879 | Colon Cancer | Phase 2 | Completed | - | - |
| NCT00398710 | Waldenström's Macroglobulinemi... more >>a Collapse << | Phase 2 | Completed | - | United States, Massachusetts
... more >>
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00776867 | Solid Tumors | Phase 1 | Completed | - | United States, New York
... more >>
Memorial Sloan-Kettering Cancer Center
New York, New York, United States, 10065 Collapse << |
| NCT00498966 | Kidney Cancer | Phase 2 | Completed | - | United States, Kentucky
... more >>
Investigative Site
Louisville, Kentucky, United States, 40202
United States, New Jersey
Investigative Site
Morristown, New Jersey, United States, 07962
United States, New York
Investigative Site
Armonk, New York, United States, 10504 Collapse << |
| NCT00847366 | Non Small Cell Lung Cancer
... more >> Solid Tumors
Metastatic Breast Cancer
Sarcomas Collapse << | Not Applicable | Completed | - | - |
| NCT01002248 | Multiple Myeloma | Phase 3 | Terminated | - | - |
| NCT00455559 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | - |
| NCT00448721 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, California
... more >>
Investigative Site
Duarte, California, United States, 91010
United States, Massachusetts
Dana Farber Cancer Institute
Boston, Massachusetts, United States, 02115
United States, Pennsylvania
Investigative Site
Philadelphia, Pennsylvania, United States, 19104
United States, Tennessee
Investigative Site
Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00375791 | Multiple Myeloma | Phase 2 | Completed | - | United States, California
... more >>
Investigative Site
Berkeley, California, United States, 94704
Investigative Site
Duarte, California, United States, 91010
United States, Georgia
Investigative Site
Atlanta, Georgia, United States, 30322
United States, Illinois
Investigative Site
Chicago, Illinois, United States, 60611
United States, Massachusetts
Investigative Site
Boston, Massachusetts, United States, 02115
United States, Michigan
Investigative Site
Ann Arbor, Michigan, United States, 48103
United States, Virginia
Investigative Site
Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00873457 | Chronic Lymphocytic Leukemia
... more >> Small Lymphocytic Lymphoma Collapse << | Phase 2 | Completed | - | United States, North Carolina
... more >>
Duke University Medical Center
Durham, North Carolina, United States, 27710 Collapse << |
| NCT00389077 | Tumors
Lympho... more >>mas Collapse << | Phase 2 | Completed | - | - |
| NCT00873457 | - | - | Completed | - | - |
| NCT01224730 | Cancer | Phase 1 | Completed | - | United States, Texas
... more >>
Dallas, Texas, United States, 75230
Houston, Texas, United States, 77030 Collapse << |
| NCT01048580 | Colon Cancer | Phase 1 | Completed | - | - |
| NCT00590954 | Malignant Gliomas
... more >> CNS
Brain Cancer
Cancer Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, New York
... more >>
Memorial Sloan-Kettering Cancer Center
New York, New York, United States, 10065 Collapse << |
| NCT01049841 | Pediatric Solid Tumors | Phase 1 | Completed | - | United States, New York
... more >>
Memorial Sloan-Kettering Cancer Center
New York, New York, United States, 10065 Collapse << |
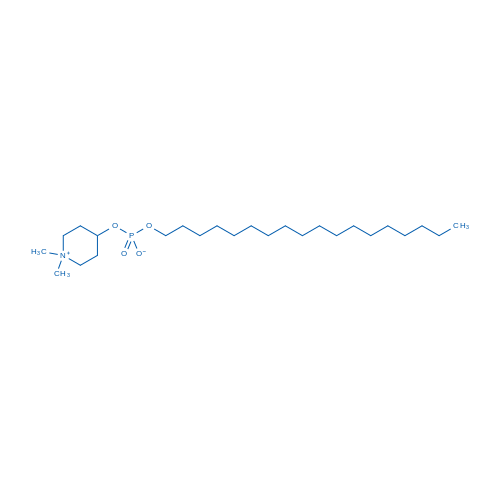
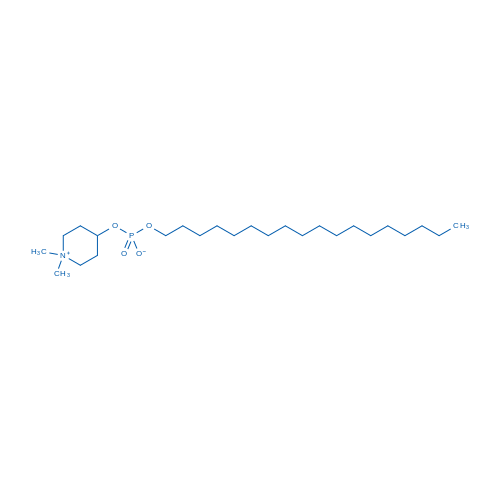

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
