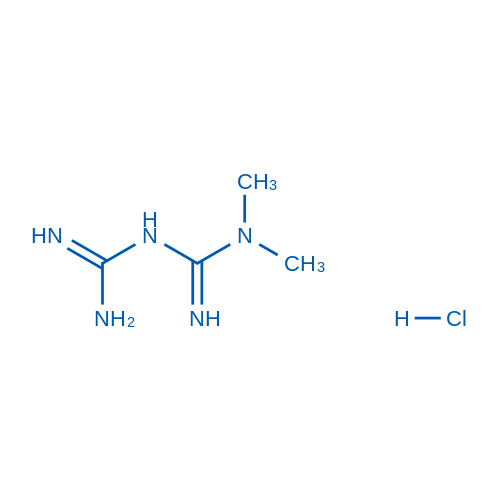
CAS No.: 1115-70-4
盐酸二甲双胍 Catalog No. CSN16694
Synonyms: 1,1-Dimethylbiguanide hydrochloride;1,1-Dimethylbiguanide HCl;Metformin (hydrochloride)
Metformin HCl, a widely used anti-diabetic drug has potential efficacy as an anti-cancer drug, decreasing hyperglycemia in hepatocytes primarily by suppressing glucose production by the liver (hepatic gluconeogenesis).
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02435277 Type 2 Diabetes Mellitus Phase 2 Completed - United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 United States, Florida Palm Beach Research Palm Beach, Florida, United States, 33409 Meridien Research Tampa, Florida, United States, 33606 United States, Georgia River Birch Research Alliance Blue Ridge, Georgia, United States, 3051313 Meridian Research Savannah, Georgia, United States, 31406 United States, Ohio Streling Research Group Cincinnati, Ohio, United States, 45219 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Meharry Medical College Nashville, Tennessee, United States, 37208 Vanderbilt University Medical Center, Diabetes, Endocrinology, and Metabolism Nashville, Tennessee, United States, 37232 Collapse << NCT03056274 Intrahepatic Cholestases ... more >> Pregnancy Complications Collapse << Phase 2 Phase 3 Not yet recruiting September 2019 - NCT02115464 Lung Cancer Phase 2 Recruiting December 2020 Canada, Alberta ... more >> Cross Cancer Institute Recruiting Edmonton, Alberta, Canada Principal Investigator: Wilson Roa Canada, Manitoba CancerCare Manitoba Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Principal Investigator: Naseer Ahmed Canada, Ontario Juravinski Cancer Centre Recruiting Hamilton, Ontario, Canada Principal Investigator: Theos Tsakiridis Cancer Centre of Southeastern Ontario at Kingston General Hospital Recruiting Kingston, Ontario, Canada Principal Investigator: Andrew Robinson Grand River Regional Cancer Centre Active, not recruiting Kitchener, Ontario, Canada Walker Family Cancer Centre - Niagara Health System Recruiting St. Catharines, Ontario, Canada Principal Investigator: Theos Tsakiridis Canada, Quebec Montreal General Hospital - McGill Recruiting Montreal, Quebec, Canada Principal Investigator: Bassam Abdulkarim Collapse << - 更多
- 参考文献
- [1] Dos Santos CC, Gopal B, Verma S. Metformin: An Old Dog with a New Trick?. Cell Metab. 2018;28(3):334-336。
- [2] Qin N, Li CB, et al. Synthesis and biological activity of novel tiliroside derivants. Eur J Med Chem. 2011;46(10):5189-95.
- [3] Polianskyte-Prause Z, Tolvanen TA, et al. Metformin increases glucose uptake and acts renoprotectively by reducing SHIP2 activity. FASEB J. 2019;33(2):2858-2869.
- [4] Takiyama Y, Harumi T, et al. Tubular injury in a rat model of type 2 diabetes is prevented by metformin: a possible role of HIF-1α expression and oxygen metabolism. Diabetes. 2011;60(3):981-92.
- [5] Luo T, Nocon A, et al. AMPK Activation by Metformin Suppresses Abnormal Extracellular Matrix Remodeling in Adipose Tissue and Ameliorates Insulin Resistance in Obesity. Diabetes. 2016;65(8):2295-310.
- [6] Sesen J, Dahan P, et al. Metformin inhibits growth of human glioblastoma cells and enhances therapeutic response. PLoS One. 2015;10(4):e0123721.
- [7] Jenkins AJ, Welsh P, Petrie JR. Metformin, lipids and atherosclerosis prevention. Curr Opin Lipidol. 2018;29(4):346-353.
- [8] Rangarajan S, Bone NB, et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med. 2018;24(8):1121-1127.
- [9] D'Amato G, Caringella AM, et al. Mild ovarian stimulation with letrozole plus fixed dose human menopausal gonadotropin prior to IVF/ICSI for infertile non-obese women with polycystic ovarian syndrome being pre-treated with metformin: a pilot study. Reprod Biol Endocrinol. 2018;16(1):89.
- [10] Yan J, Yao B, et al. Liraglutide, Sitagliptin, and Insulin Glargine Added to Metformin: The Effect on Body Weight and Intrahepatic Lipid in Patients With Type 2 Diabetes Mellitus and Nonalcoholic Fatty Liver Disease. Hepatology. 2018.
- [11] Rotermund C, Machetanz G, Fitzgerald JC. The Therapeutic Potential of Metformin in Neurodegenerative Diseases. Front Endocrinol (Lausanne). 2018.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1115-70-4 | 储存条件 |
|
|||||||||
| 分子式 | C4H12ClN5 | 运输 | 蓝冰 | |||||||||
| 分子量 | 165.62 | 别名 | 1,1-Dimethylbiguanide hydrochloride;1,1-Dimethylbiguanide HCl;Metformin (hydrochloride);Metformin Hydrochloride;ADX-155;EFB-0027;EX-404;La-6023;SMP-862;Fortamet;盐酸二甲双胍 | |||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| human HepG2 cells | - | Function assay | 24 h | Increase in glucose consumption in insulin-resistant human HepG2 cells after 24 hrs, EC50=0.27 μM. | 21856048 |
| human HepG2 cells | 1 mM | Function assay | 24 h | Reduction of glucose consumption in insulin-resistant human HepG2 cells at 1 mM after 24 hrs by glucose oxidase method in presence of 22.2 mM of glucose | 23025244 |
| human HepG2 cells | 1 mM | Function assay | 24 h | Activation of AMPK in human HepG2 cells assessed as reduction of gluconeogenesis at 1 mM after 24 hrs by enzymatic colorimetric assay | 26471090 |
| human MDA-MB-231 cells | 1 to 20 mM | Function assay | 24 h | Antiproliferative activity against human MDA-MB-231 cells at 1 to 20 mM after 24 hrs by MTT assay. | 22459208 |
| mouse 3T3L1 cells | 1 mM | Function assay | - | Induction of AMPK phosphorylation in mouse 3T3L1 cells at 1 mM by Western blot analysis | 25216379 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02435277 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 United States, Florida Palm Beach Research Palm Beach, Florida, United States, 33409 Meridien Research Tampa, Florida, United States, 33606 United States, Georgia River Birch Research Alliance Blue Ridge, Georgia, United States, 3051313 Meridian Research Savannah, Georgia, United States, 31406 United States, Ohio Streling Research Group Cincinnati, Ohio, United States, 45219 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Meharry Medical College Nashville, Tennessee, United States, 37208 Vanderbilt University Medical Center, Diabetes, Endocrinology, and Metabolism Nashville, Tennessee, United States, 37232 Collapse << |
| NCT03056274 | Intrahepatic Cholestases ... more >> Pregnancy Complications Collapse << | Phase 2 Phase 3 | Not yet recruiting | September 2019 | - |
| NCT02115464 | Lung Cancer | Phase 2 | Recruiting | December 2020 | Canada, Alberta ... more >> Cross Cancer Institute Recruiting Edmonton, Alberta, Canada Principal Investigator: Wilson Roa Canada, Manitoba CancerCare Manitoba Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Principal Investigator: Naseer Ahmed Canada, Ontario Juravinski Cancer Centre Recruiting Hamilton, Ontario, Canada Principal Investigator: Theos Tsakiridis Cancer Centre of Southeastern Ontario at Kingston General Hospital Recruiting Kingston, Ontario, Canada Principal Investigator: Andrew Robinson Grand River Regional Cancer Centre Active, not recruiting Kitchener, Ontario, Canada Walker Family Cancer Centre - Niagara Health System Recruiting St. Catharines, Ontario, Canada Principal Investigator: Theos Tsakiridis Canada, Quebec Montreal General Hospital - McGill Recruiting Montreal, Quebec, Canada Principal Investigator: Bassam Abdulkarim Collapse << |
| NCT01440127 | Colon Cancer | Phase 1 | Terminated(Unable to accrue al... more >>l planned subjects in a timely fashion, but data collected will still be analyzed.) Collapse << | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT02435277 | - | - | Completed | - | - |
| NCT03106870 | Gestational Diabetes Mellitus ... more >>in Pregnancy Collapse << | Not Applicable | Completed | - | - |
| NCT03106870 | - | - | Completed | - | - |
| NCT02437656 | Rectal Cancer | Phase 2 | Completed | - | France ... more >> Centre Marie Curie Arras, France, 62000 Centre Pierre Curie Beuvry, France, 62880 Centre Léonard de Vinci - SARL du pont Saint Vaast Douai, France, 59500 Institut André Dutreix Dunkerque, France, 59240 Centre Hospitalier Lens, France, 62300 Clinique du Bois - Centre Bourgogne Lille, France, 59000 Centre Oscar Lambret Lille, France, 59020 Centre Galilée - Hôpital Privé La Louvière Lille, France, 59045 Centre Gray Maubeuge, France, 59600 Centre Joliot-Curie St Martin-Boulogne, France, 62200 Clinique des Dentellières Valenciennes, France, 59300 Collapse << |
| NCT01231074 | Obesity Weigh... more >>t Gain Psychotropic Induced Weight Gain Collapse << | Phase 1 | Unknown | May 2011 | United States, Ohio ... more >> Nationwide Children's Hospital Recruiting Columbus, Ohio, United States, 43205 Contact: Ihuoma Eneli, MD, MS 614-722-4089 ihuoma.eneli@nationwidechildrens.org Contact: Mary Ann Murphy, MD, PhD 614-722-2291 maryann.murphy@nationwidechildrens.org Collapse << |
| NCT02378441 | Healthy | Phase 1 | Completed | - | - |
| NCT01526616 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | - |
| NCT03774108 | PreDiabetes H... more >>IV-1-infection Collapse << | Phase 4 | Not yet recruiting | March 8, 2019 | Mexico ... more >> Hospital Civil de Guadalajara Not yet recruiting Guadalajara, Jalisco, Mexico, 44280 Contact: Juan Luis Soto-Mancilla, MD +523339422441 ext 41014 Collapse << |
| NCT01954732 | Stage IA Pancreatic Cancer ... more >> Stage IB Pancreatic Cancer Stage IIA Pancreatic Cancer Stage IIB Pancreatic Cancer Collapse << | Phase 1 | Withdrawn(Slow accrual) | - | - |
| NCT03629639 | Psoriasis Vulgaris With Metabo... more >>lic Disorders Collapse << | Phase 4 | Not yet recruiting | December 1, 2020 | - |
| NCT03359681 | Colon Cancer | Phase 2 | Not yet recruiting | December 2018 | Denmark ... more >> Department of Surgery, Slagelse Hospital Not yet recruiting Slagelse, Denmark, 4200 Contact: Emilie P Colov, MD +45 61335122 eco@regionsjaelland.dk Collapse << |
| NCT01546558 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Florida ... more >> SeaView Research Inc. Miami, Florida, United States, 33126 Collapse << |
| NCT02733679 | Ataxia-Telangiectasia | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital Dundee, Angus, United Kingdom, DD19SY Collapse << |
| NCT03117517 | Polycystic Ovarian Syndrome | Early Phase 1 | Completed | - | Pakistan ... more >> Mardan Medical Complex Mardan, Khyber Pukhtunkhwah, Pakistan, 25000 Collapse << |
| NCT02042495 | Endometrial Cancer | Phase 2 | Withdrawn(Did not obtain Healt... more >>h Canda approval for drug use in reasonable time and moved onward to new trial) Collapse << | - | Canada, Quebec ... more >> Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT03765359 | Diabetic Pregnancy ... more >> Insulin Resistance Type1diabetes Collapse << | Phase 4 | Not yet recruiting | December 31, 2021 | - |
| NCT03710343 | Cerebral Palsy ... more >> Spastic Spastic Cerebral Palsy Cerebral Palsy, Spastic Cerebral Palsy, Mixed Collapse << | Phase 2 | Not yet recruiting | February 1, 2022 | Canada, Ontario ... more >> Holland Bloorview Kids Rehabilitation Hospital Not yet recruiting Toronto, Ontario, Canada, M4G1R8 Contact: Daniel Warner, BScH 416-425-6220 ext 6482 dwarner@hollandbloorview.ca Principal Investigator: Darcy Fehlings, MD, MSc The Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT03583385 | Healthy | Phase 1 | Completed | - | Indonesia ... more >> PT Equilab International Jakarta, Indonesia, 12430 Collapse << |
| NCT02064881 | Type 2 Diabetes | Phase 2 Phase 3 | Recruiting | August 2020 | Mexico ... more >> Instituto Nacional de Ciencias Médicas Y Nutrición Salvador Zubirán Recruiting México, Distrito Federal, Mexico, 14000 Contact: Francisco Gómez, Doctor 55133891 gomezperezfco@gmail.com Contact: Paloma Almeda, Doctor 56554523 palomaalmeda@yahoo.com Principal Investigator: Francisco J Gómez, Doctor Collapse << |
| NCT01905046 | Atypical Ductal Breast Hyperpl... more >>asia BRCA1 Mutation Carrier BRCA2 Mutation Carrier Ductal Breast Carcinoma in Situ Lobular Breast Carcinoma in Situ Collapse << | Phase 3 | Recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Suspended Duarte, California, United States, 91010 Los Angeles County-USC Medical Center Suspended Los Angeles, California, United States, 90033 USC / Norris Comprehensive Cancer Center Suspended Los Angeles, California, United States, 90033 City of Hope South Pasadena Suspended South Pasadena, California, United States, 91030 United States, Kansas University of Kansas Cancer Center Recruiting Kansas City, Kansas, United States, 66160 Contact: Site Public Contact 913-945-7552 ctnursenav@kumc.edu Principal Investigator: Carol J. Fabian United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Site Public Contact 855-776-0015 Principal Investigator: Sandhya Pruthi United States, New York Roswell Park Cancer Institute Suspended Buffalo, New York, United States, 14263 Mount Sinai Hospital Recruiting New York, New York, United States, 10029 Contact: Site Public Contact 212-824-7309 CCTO@mssm.edu Principal Investigator: Sarah P. Cate Columbia University/Herbert Irving Cancer Center Suspended New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center Suspended Durham, North Carolina, United States, 27710 United States, Ohio The Christ Hospital Recruiting Cincinnati, Ohio, United States, 45219 Contact: Site Public Contact 513-585-0844 Principal Investigator: Jennifer B. Manders Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Lisa D. Yee United States, Oklahoma University of Oklahoma Health Sciences Center Recruiting Oklahoma City, Oklahoma, United States, 73104 Contact: Site Public Contact 405-271-8777 ou-clinical-trials@ouhsc.edu Principal Investigator: William C. Dooley United States, South Carolina Greenville Health System Cancer Institute-Easley Recruiting Easley, South Carolina, United States, 29640 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Andrews Suspended Greenville, South Carolina, United States, 29601 Greenville Health System Cancer Institute-Butternut Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Faris Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Memorial Hospital Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Eastside Recruiting Greenville, South Carolina, United States, 29615 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Greer Recruiting Greer, South Carolina, United States, 29650 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Seneca Recruiting Seneca, South Carolina, United States, 29672 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Spartanburg Recruiting Spartanburg, South Carolina, United States, 29307 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Therese B. Bevers Doctor's Hospital of Laredo Active, not recruiting Laredo, Texas, United States, 78041 United States, Wisconsin University of Wisconsin Hospital and Clinics Recruiting Madison, Wisconsin, United States, 53792 Contact: Site Public Contact 800-622-8922 Principal Investigator: Lee G. Wilke Collapse << |
| NCT02035787 | Complex Atypical Hyperplasia ... more >> Endometrial Cancer Collapse << | Not Applicable | Recruiting | March 2020 | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Jennifer Gruhn 919-966-4432 jennifer_gruhn@med.unc.edu Principal Investigator: Allison Staley, MD Principal Investigator: Victoria Bae-Jump, MD United States, Ohio TriHealth Recruiting Cincinnati, Ohio, United States, 45220 Principal Investigator: Kevin Schuler, MD Collapse << |
| NCT01422746 | Obesity Hyper... more >>androgenemia Polycystic Ovary Syndrome Collapse << | Not Applicable | Recruiting | December 2019 | United States, Virginia ... more >> University of Virginia Center for Research in Reproduction Recruiting Charlottesville, Virginia, United States, 22908 Contact: Deborah Sanderson 434-243-6911 pcos@virginia.edu Principal Investigator: Christine Burt Solorzano, MD Sub-Investigator: John C. Marshall, MD, PhD Collapse << |
| NCT01386671 | Type 2 Diabetes | Phase 3 | Completed | - | Mexico ... more >> Unidad Antidiabética Integral Mexico City, AA, Mexico, 06600 Paracelsus S.A. de C.V. Mexico, city, AA, Mexico, 03800 Unidad de Investigacion en Epidemiologia Clinica. UMAE Hospital de Especialidades Centro Medico Nacional Siglo XXI. Instituto Mexicano del Seguro Social Mexico City, Distrito Federal, Mexico, 06720 Instituto de terapéutica experimental y clínica (INTEC) Guadalajara, Jalisco, Mexico, 44600 Collapse << |
| NCT01409395 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Ucsf Ctsi Crc San Francisco, California, United States, 94158 Collapse << |
| NCT01561482 | Prostate Carcinoma | Phase 2 | Withdrawn(Study closed due to ... more >>slow/low enrollment; no subjects were enrolled.) Collapse << | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Ben Taub General Hospital Houston, Texas, United States, 77030 Michael E. Debakey Veterans Affairs Medical Center Houston, Texas, United States, 77030 Collapse << |
| NCT02815397 | - | - | Terminated(Poor accrual) | - | - |
| NCT02308228 | Aging | Not Applicable | Active, not recruiting | January 2019 | United States, Alabama ... more >> University of Alabama Birmingham Birmingham, Alabama, United States, 35294 United States, Kentucky University of Kentucky Lexington, Kentucky, United States, 40536 Collapse << |
| NCT02815397 | Diffuse Large B-Cell Lymphoma | Phase 2 | Terminated(Poor accrual) | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT02072382 | Hypertension | Phase 4 | Unknown | December 2015 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Recruiting Porto Alegre, RS, Brazil Contact: Corrêa Junior Vicente (51)99839395 vicentecorreajunior@terra.com.br Sub-Investigator: Correa Junior Vicente Collapse << |
| NCT02109549 | - | - | Completed | - | Netherlands ... more >> The Netherlands Cancer Institute (NKI) Amsterdam, Netherlands, 1066 CX AMC Amsterdam, Netherlands, 1105 AZ MAASTRO clinic Maastricht, Netherlands, 6229 ET Collapse << |
| NCT00187798 | Other Conditions That May Be A... more >> Focus of Clinical Attention Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California San Francsico San Francisco, California, United States, 94143 Collapse << |
| NCT02581137 | Erythroplakia ... more >> Hyperplasia Mild Dysplasia Moderate Dysplasia Oral Cavity Carcinoma Oral Leukoplakia Severe Dysplasia Collapse << | Phase 2 | Active, not recruiting | - | United States, California ... more >> University of California San Diego San Diego, California, United States, 92103 United States, Minnesota University of Minnesota/Masonic Cancer Center Minneapolis, Minnesota, United States, 55455 Canada, British Columbia BC Cancer Research Centre Vancouver, British Columbia, Canada, V5Z 1L3 University of British Columbia Hospital Vancouver, British Columbia, Canada, V6T 2B5 Collapse << |
| NCT02420652 | Recurrent Prostate Carcinoma ... more >> Stage I Prostate Cancer Stage IIA Prostate Cancer Stage IIB Prostate Cancer Stage III Prostate Cancer Collapse << | Phase 2 | Active, not recruiting | January 2019 | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT02659306 | HIV Infection | Phase 1 | Active, not recruiting | December 2018 | Canada, Quebec ... more >> Chronic Viral Illness Service, McGill University Health Centre Montreal, Quebec, Canada, H4A 3J1 Collapse << |
| NCT01685762 | Endometrial Hyperplasia ... more >> Endometrial Hyperplasia Without Atypia Collapse << | Early Phase 1 | Active, not recruiting | December 2018 | United States, North Carolina ... more >> The University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Southern Pines Women's Health Center Southern Pines, North Carolina, United States, 28388 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02593097 | Migraine | Phase 3 | Active, not recruiting | December 2018 | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 Collapse << |
| NCT03183752 | Thyroid Nodule ... more >> Insulin Resistance Collapse << | Not Applicable | Recruiting | July 10, 2018 | Brazil ... more >> Patricia Santos Recruiting Rio de Janeiro, Brazil, 21941913 Contact: Patricia Santos 55 21 981669799 patriciaborges.endocrino@gmail.com Collapse << |
| NCT01266486 | Breast Cancer | Phase 2 | Completed | - | United Kingdom ... more >> Mount Vernon Centre for Cancer Treatment, Rickmansworth Road Northwood, Middlesex, United Kingdom, HA6 2RN Dept Oncology, Churchill Hospital, Old Road, Headington Oxford, Oxfordshire, United Kingdom, OX3 7LJ Surgery and Molecular Oncology Ninewells Hospital Dundee, Scotland, United Kingdom, DD1 9SY Collapse << |
| NCT01884558 | Pharmacokinetics of Isavuconaz... more >>ole Pharmacokinetics of Metformin Healthy Subjects Collapse << | Phase 1 | Completed | - | United States, California ... more >> California Clinical Trials Medical Group Glendale, California, United States, 91026 Collapse << |
| NCT01243385 | Prostate Cancer | Phase 2 | Active, not recruiting | December 2032 | Switzerland ... more >> Kantonsspital Aarau Aarau, Switzerland, CH-5001 Universitaetsspital-Basel Basel, Switzerland, CH-4031 Inselspital Bern Bern, Switzerland, CH-3010 Kantonsspital Graubuenden Chur, Switzerland, CH-7000 Kantonsspital Luzern Luzerne, Switzerland, CH-6000 Kantonsspital - St. Gallen St. Gallen, Switzerland, CH-9007 Kantonsspital Winterthur Winterthur, Switzerland, CH-8401 Onkozentrum Zurich, Switzerland, 8038 UniversitaetsSpital Zuerich Zurich, Switzerland, CH-8091 Collapse << |
| NCT02432287 | Aging | Phase 4 | Completed | - | United States, New York ... more >> Einstein College of Medicine of Yeshiva University Bronx, New York, United States, 10461 Collapse << |
| NCT02432287 | - | - | Completed | - | - |
| NCT02339168 | Prostate Cancer | Phase 1 | Active, not recruiting | December 2019 | United States, California ... more >> University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 Collapse << |
| NCT02198417 | Polycystic Ovary Syndrome | Not Applicable | Active, not recruiting | December 2020 | - |
| NCT03684707 | Oral Cancer | Phase 4 | Recruiting | September 15, 2019 | Egypt ... more >> Noha Nasr Recruiting Cairo, New Cairo, Egypt, 11835 Contact: Noha N. El-Zalabany, Masters 1005365769 ext 02 noha_nasr84@yahoo.com Contact: Noha N. El-Zalabany, Masters 01005365769 ext 02 noha_nasr84@yahoo.com Collapse << |
| NCT03335423 | Metformin Org... more >>anic Cation Transporter 1 Codeine Collapse << | Phase 1 | Not yet recruiting | July 31, 2021 | - |
| NCT03510390 | Head and Neck Cancer ... more >> Oral Cavity Squamous Cell Carcinoma Collapse << | Not Applicable | Recruiting | September 30, 2019 | Switzerland ... more >> Inselspital, Bern University Hospital Recruiting Bern, Switzerland, 3010 Contact: Olgun Elicin, MD +41316322431 olgun.elicin@insel.ch Contact: Simon Mueller, MD +41316322931 simon.mueller@insel.ch Collapse << |
| NCT03593668 | Obesity; Drug | Phase 4 | Recruiting | March 2019 | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Recruiting Nanjing, Jiangsu, China, 210008 Contact: Dalong Zhu, MD,PhD 86-25-83-105302 zhudldr@gmail.com Contact: Yan Bi, MD,PhD 86-25-83-105302 biyan@nju.edu.cn Principal Investigator: Dalong Zhu, MD,PhD Sub-Investigator: Yan Bi, MD,PhD Collapse << |
| NCT02285855 | Lung Cancer | Phase 2 | Active, not recruiting | February 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01681693 | Healthy Type ... more >>2 Diabetes Mellitus Collapse << | Phase 1 | Completed | - | United States, California ... more >> San Francisco General Hospital San Francisco, California, United States, 94110 Collapse << |
| NCT01334684 | Type 2 Diabetes | Not Applicable | Unknown | September 2013 | Italy ... more >> Casa Sollievo Della Sofferenza IRCCS Not yet recruiting San Giovanni Rotondo, Foggia, Italy, 71013 Collapse << |
| NCT02153450 | Acinar Cell Adenocarcinoma of ... more >>the Pancreas Duct Cell Adenocarcinoma of the Pancreas Recurrent Pancreatic Cancer Stage I-III Pancreatic Cancer Collapse << | Early Phase 1 | Recruiting | December 2018 | United States, Ohio ... more >> University Hospitals Cleveland Medical Center, Seidman Cancer Center, Case Comprehensive Cancer Center Recruiting Cleveland, Ohio, United States, 44106-5065 Contact: Jennifer A. Dorth 800-641-2422 jennifer.dorth@uhhospitals.org Principal Investigator: Jennifer A. Dorth Collapse << |
| NCT02915198 | Prediabetic State ... more >> Atherosclerosis Metformin Collapse << | Phase 4 | Not yet recruiting | June 30, 2024 | United States, Arizona ... more >> Phoenix VA Health Care System, Phoenix, AZ Not yet recruiting Phoenix, Arizona, United States, 85012 Contact: Peter Reaven, MD 602-277-5551 ext 6875 Peter.Reaven@va.gov United States, California VA Loma Linda Healthcare System, Loma Linda, CA Not yet recruiting Loma Linda, California, United States, 92357 Contact: Alan Jacobson, MD 909-583-6050 alan.jacobson@va.gov VA Long Beach Healthcare System, Long Beach, CA Not yet recruiting Long Beach, California, United States, 90822 Contact: Anthony Vo, MD 562-826-3497 Anthony.Vo@va.gov VA Palo Alto Health Care System, Palo Alto, CA Not yet recruiting Palo Alto, California, United States, 94304-1290 Contact: Arlina Ahluwalia, MD 650-493-5000 ext 61755 Arlina.Ahluwalia@va.gov Contact: Paul Heidenreich, MD 6504935000 ext 64069 Paul.Heidenreich@va.gov VA Greater Los Angeles Healthcare System, West Los Angeles, CA Not yet recruiting West Los Angeles, California, United States, 90073 Contact: Tannaz Moin, MD 310-478-3711 ext 48380 Tannaz.Moin@va.gov; tmoin@mednet.ucla.edu United States, Colorado Rocky Mountain Regional VA Medical Center, Aurora, CO Not yet recruiting Aurora, Colorado, United States, 80045 Contact: Allan V Prochazka, MD MSc 303-399-8020 ext 2144 Allan.Prochazka@va.gov Study Chair: Gregory G. Schwartz, PhD MD United States, Florida North Florida/South Georgia Veterans Health System, Gainesville, FL Not yet recruiting Gainesville, Florida, United States, 32608 Contact: Carsten Schmalfuss, MD 352-376-1611 ext 6052 Carsten.Schmalfuss@va.gov Miami VA Healthcare System, Miami, FL Not yet recruiting Miami, Florida, United States, 33125 Contact: Ana Palacio, MD 305-926-3780 Ana.Palacio@va.gov United States, Oregon VA Portland Health Care System, Portland, OR Not yet recruiting Portland, Oregon, United States, 97239 Contact: North Noelck, MD 503-220-8262 ext 56426 North.Noelck2@va.gov Contact: Linda Humphrey, MD 5032208262 ext 57176 Linda.Humphrey@va.gov United States, Utah VA Salt Lake City Health Care System, Salt Lake City, UT Not yet recruiting Salt Lake City, Utah, United States, 84148 Contact: Charles Lui, MD 801-582-1565 Charles.Lui@va.gov United States, Virginia Salem VA Medical Center, Salem, VA Not yet recruiting Salem, Virginia, United States, 24153 Contact: Ali Iranmanesh, MD 540-983-1071 Ali.Iranmanesh@va.gov Collapse << |
| NCT01510522 | Diabetes Mellitus Type 2 | Phase 4 | Withdrawn | - | - |
| NCT01613755 | Diabetes | Phase 4 | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Centre Nijmegen, Gelderland, Netherlands, 6500 HB Collapse << |
| NCT01160042 | Healthy | Phase 1 | Completed | - | India ... more >> Vimta Labs Ltd. Hyderabad, Andhra pradesh, India, 500051 Collapse << |
| NCT01910246 | Insulin Resistance | Phase 4 | Withdrawn(No funding) | - | United States, California ... more >> UCSF, Radiology and Biomedical Imaging San Francisco, California, United States, 94143 Collapse << |
| NCT03039075 | Diabetes Mellitus | Phase 4 | Recruiting | December 2018 | China, Chongqing ... more >> The First Affiliated Hospital of Chongqing Medical University Recruiting Chongqing, Chongqing, China, 400016 Contact: Qifu Li, PhD +86 23 89011552 liqifu@yeah.net Collapse << |
| NCT02903511 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 2 | Recruiting | June 2020 | United States, Colorado ... more >> University of Colorado Denver, Anschutz Medical Campus Recruiting Aurora, Colorado, United States, 80045 Principal Investigator: Godela Brosnahan, MD Sub-Investigator: Michel Chonchol, MD Sub-Investigator: Berenice Gitomer, PhD Collapse << |
| NCT01160068 | Healthy | Phase 1 | Completed | - | India ... more >> Vimta Labs Ltd. Hyderabad, Andhra pradesh, India, 500051 Collapse << |
| NCT03086733 | Non Small Cell Lung Cancer ... more >> Lung Cancer Collapse << | Phase 2 | Recruiting | December 2020 | Canada, Ontario ... more >> Toronto General Hospital Recruiting Toronto, Ontario, Canada, M5G 2C4 Contact: Alexandra Salvarrey, MD MSc 1-416-340-4800 ext 6992 alexandra.salvarrey@uhn.on.ca Contact: Jennifer Lister, BSc 1-416-340-4800 ext 4857 jennifer.lister@uhn.on.ca Principal Investigator: Thomas Waddell, MD FRCSC Princess Margaret Hospital Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Alexandra Salvarrey, MD MSc 1-416-946-4501 ext 5444 alexandra.salvarrey@uhn.on.ca Contact: Jennifer Lister, BSc 1-416-340-4800 ext 4857 jennifer.lister@uhn.on.ca Principal Investigator: Natasha Leighl, MD MSc FRCPC Collapse << |
| NCT03086005 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Songpa-gu, Korea, Republic of, 05505 Collapse << |
| NCT03467763 | Metformin Adverse Reaction ... more >> Tolerance Collapse << | Phase 4 | Recruiting | July 1, 2019 | United States, New York ... more >> Weill Cornell Medical College Recruiting New York, New York, United States, 10065 Contact: James H Flory, MD 646-962-9309 jaf9052@med.cornell.edu Contact: Leon Igel, MD 6469622111 lei9004@med.cornell.edu Collapse << |
| NCT01442870 | Cancer | Phase 1 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01717482 | Lung Neoplasms ... more >> Carcinoma, Non-Small-Cell Lung Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01750567 | Relapsed Chronic Lymphocytic L... more >>eukemia Collapse << | Phase 2 | Recruiting | January 2019 | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Sami Malek, MD 734-936-5310 smalek@med.umich.edu Contact: Erlene Seymour, MD 734-647-2892 ekuizon@med.umich.edu Principal Investigator: Sami Malek, MD Sub-Investigator: Daniel Lebovic, MD Sub-Investigator: Erlene Seymour, MD Collapse << |
| NCT01681680 | Healthy | Phase 1 | Completed | - | - |
| NCT01793948 | Breast Cancer ... more >> Obesity Collapse << | Early Phase 1 | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT02176161 | Prostate Cancer ... more >> Prostate Cancer Recurrent Collapse << | Phase 2 | Recruiting | June 2020 | United States, New York ... more >> Winthrop University Hospital Recruiting Mineola, New York, United States, 11501 Principal Investigator: Aaron E Katz, MD Collapse << |
| NCT01718340 | Abortion, Habitual | Phase 4 | Unknown | June 2014 | Egypt ... more >> Women's Health Hospital Recruiting Assiut, Egypt Contact: Hassan S Kamel, M D hkamelhkamel@yahoo.com Principal Investigator: Alaa mM Ismail, M D Collapse << |
| NCT02026869 | PCOS | Early Phase 1 | Unknown | February 2014 | Egypt ... more >> Women's Health Hospital Assiut, Egypt, 71516 Collapse << |
| NCT01997775 | Non-small Cell Lung Cancer | Phase 2 | Terminated(Difficult enrollmen... more >>t) Collapse << | - | Taiwan ... more >> National Cheng-Kung Uni. Hosp. Tainan, Taiwan, 704 Collapse << |
| NCT03017833 | Solid Tumor A... more >>dvanced Cancer Collapse << | Phase 1 | Recruiting | March 2023 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Clinical Research Operations CR_Study_Registration@mdanderson.org Collapse << |
| NCT01876992 | Diabetes Pre-... more >>diabetes Obesity Collapse << | Not Applicable | Terminated(All investigators/c... more >>o-investigators relocating to other institutions.) Collapse << | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01876992 | - | - | Terminated(All investigators/c... more >>o-investigators relocating to other institutions.) Collapse << | - | - |
| NCT02086526 | Polycystic Ovary Syndrome | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03618472 | Metformin CA ... more >>Endometrium Collapse << | Not Applicable | Recruiting | November 30, 2019 | Thailand ... more >> Rajavithi Hospital Recruiting Bangkok, Thailand, 10400 Contact: charuwan manmee, Phd 023548108 ext 2803 charuwan_manmee@yahoo.com Principal Investigator: kittisak Petchsila, MD Collapse << |
| NCT03477162 | Thoracic Neoplasm | Early Phase 1 | Recruiting | December 31, 2018 | United States, New Hampshire ... more >> Dartmouth Hitchcock Medical Center Recruiting Lebanon, New Hampshire, United States, 03756 Contact: Kayla Fay 603-650-8537 Kayla.A.Fay@hitchcock.org Contact: Research Nurse 800-639-6918 cancer.research.nurse@dartmouth.edu Collapse << |
| NCT02274948 | Pediatric Obesity | Phase 4 | Completed | - | Sri Lanka ... more >> Lions Club of Negombo Host Negombo, Western Province, Sri Lanka Collapse << |
| NCT02274948 | - | - | Completed | - | - |
| NCT03555565 | - | - | Active, not recruiting | October 31, 2019 | Japan ... more >> Takeda Selected Site Tokyo, Japan Collapse << |
| NCT03593707 | Healthy Volunteers | Phase 1 | Suspended(Sponsor elected to v... more >>oluntarily pause study.) Collapse << | February 12, 2019 | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT01873859 | - | - | Completed | - | - |
| NCT03445702 | Diabetes Mellitus, Type 2 ... more >> Metformin Adverse Reaction Collapse << | Early Phase 1 | Recruiting | December 2019 | United States, Missouri ... more >> Saint Louis University Recruiting Saint Louis, Missouri, United States, 63110 Contact: Linda Eggemeyer-Sharpe, RN 314-977-9350 linda.eggemeyersharpe@health.slu.edu Collapse << |
| NCT02354027 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01312467 | Adenomatous Polyp ... more >> Colorectal Cancer Obesity Collapse << | Phase 2 | Completed | - | United States, California ... more >> Veterans Administration Long Beach Medical Center Long Beach, California, United States, 90822 University of California Medical Center At Irvine-Orange Campus Orange, California, United States, 92868 Kaiser Permanente - Sacramento Sacramento, California, United States, 95825 Canada, Quebec Jewish General Hospital Montreal, Quebec, Canada, H3T 1E2 Collapse << |
| NCT01312467 | - | - | Completed | - | - |
| NCT01873859 | Diabetes Mellitus ... more >> Lactic Acidosis Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Cardiovascular research center, Modarres hospital. Tehran, Iran, Islamic Republic of Collapse << |
| NCT03118128 | Acute Lymphoblastic Leukemia | Not Applicable | Completed | - | - |
| NCT01677260 | Metformin XR BE Study in Healt... more >>hy Volunteers With Single and Multiple Dose Collapse << | Not Applicable | Completed | - | Indonesia ... more >> PT Equilab International Jakarta, Indonesia, 12430 Collapse << |
| NCT00986401 | - | - | Completed | - | - |
| NCT02895750 | Renal Insufficiency, Chronic | Phase 2 | Recruiting | August 30, 2020 | France ... more >> CHU Amiens Picardie Recruiting Amiens, France, 80054 Contact: Jean-Daniel LALAU, MD, PhD +33 3 22 45 58 95 lalau.jean-daniel@chu-amiens.fr Collapse << |
| NCT03772964 | Inflammatory Response | Phase 1 Phase 2 | Not yet recruiting | September 2020 | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Not yet recruiting Pittsburgh, Pennsylvania, United States, 15209 Contact: Katherine M Reitz, MD reitzkm2@upmc.edu Contact: Brian Zuckerbraun, MD zuckerbraunbs@upmc.edu Collapse << |
| NCT02597400 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, New Jersey ... more >> Frontage Clinical Services Inc. Hackensack, New Jersey, United States, 07601 Collapse << |
| NCT02741960 | Systemic Lupus Erythematosus | Phase 4 | Active, not recruiting | March 2019 | China, Shanghai ... more >> Ethics Committee of Renji Hospital, Shanghai Jiaotong University School of Medicine Shanghai, Shanghai, China, 200000 Collapse << |
| NCT02496741 | Glioma Cholan... more >>giocarcinoma Chondrosarcoma Collapse << | Phase 1 Phase 2 | Unknown | December 2016 | Netherlands ... more >> VU University Medical Center Not yet recruiting Amsterdam, Noord-Holland, Netherlands, 1081 HZ Contact: Myra E Van Linde, M.D. +31(20)4441412 m.vanlinde@vumc.nl Academic Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1105AZ Contact: Hanneke W Wilmink, M.D., Ph.D. +31(20)5662895 j.w.wilmink@amc.uva.nl Contact: Remco J Molenaar, M.Sc. +31(20)5668587 r.j.molenaar@amc.nl Principal Investigator: Hanneke W Wilmink, M.D., Ph.D. Leiden University Medical Center Not yet recruiting Leiden, Zuid-Holland, Netherlands, 2333 ZA Contact: Hans J Gelderblom, M.D., Ph.D. +31(71)5269111 A.J.Gelderblom@lumc.nl Collapse << |
| NCT00986401 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT03590262 | Type 1 Diabetes Mellitus ... more >> Metformin Collapse << | Phase 4 | Recruiting | September 30, 2019 | China, Guangdong ... more >> Shenzhen People's Hospital Recruiting Shenzhen, Guangdong, China, 518020 Contact: Xiuzhen Zhang, MD 0086-755-22943422 876538754@qq.com Collapse << |
| NCT02972723 | Chronic Hepatitis C Infection | Phase 4 | Completed | - | United Kingdom ... more >> Secondary care Hepatitis clinic at Nottingham University Hospital Nottingham, United Kingdom, NG7 2UH Collapse << |
| NCT03722290 | Fragile X Syndrome | Phase 2 | Recruiting | September 1, 2019 | Canada, Quebec ... more >> Université de Sherbrooke Recruiting Sherbrooke, Quebec, Canada, J1H 5H3 Contact: Artuela Çaku, MD 819-346-1110 ext 71035 Artuela.s.çaku@usherbrooke.ca Collapse << |
| NCT03733132 | Insulin Resistance ... more >> Obesity Obesity, Abdominal Collapse << | Phase 2 | Not yet recruiting | May 31, 2020 | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03361878 | Diabetes Mellitus, Type 2 ... more >> Metformin Adverse Reaction Collapse << | Phase 4 | Completed | - | - |
| NCT02684578 | Age-Related Macular Degenerati... more >>on Macular Degeneration, Age-Related Dry Macular Degeneration Geographic Atrophy Collapse << | Phase 2 | Recruiting | April 2019 | United States, California ... more >> University of California, Davis Recruiting Davis, California, United States, 95616 Contact: Angela Beliveau, MPH 916-734-6814 University of California San Francisco Recruiting San Francisco, California, United States, 94143 Contact: Catherine Psaras, BA 415-206-3123 eyestudy@ucsf.edu Principal Investigator: Jay M Stewart, MD Collapse << |
| NCT03685409 | Oral Cancer | Phase 3 | Recruiting | September 30, 2020 | Egypt ... more >> Faculty of Dentistry- Cairo University Recruiting Cairo, Egypt Contact: Fatheya M Zahran, Professor 00201060775113 Fatheyazahran@gmail.com Contact: Gihan G Madkour, As.Prof. 00201121001005 gihanemadkour@gmail.com Collapse << |
| NCT02978547 | Resectable Pancreatic Ductal A... more >>denocarcinoma Collapse << | Phase 2 | Not yet recruiting | January 2021 | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Collapse << |
| NCT00648492 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT02438020 | Acanthosis Nigricans | Phase 4 | Unknown | October 2016 | Mexico ... more >> Hospital Central Dr.Ignacio Morones Prieto Active, not recruiting San Luis Potosi, SLP, Mexico, 78210 Collapse << |
| NCT00649350 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT02084056 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.11.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02084056 | - | - | Completed | - | - |
| NCT01710553 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01005160 | Healthy Male Volunteers | Phase 1 | Completed | - | - |
| NCT01357148 | - | - | Terminated(Completion of MK-04... more >>31A-235 was rendered unnecessary, as the local oversight authority accepted in its stead the results of another study [MK-0431-234].) Collapse << | - | - |
| NCT01664845 | Chronic Hepatitis c ... more >> Insulin Resistance Collapse << | Phase 2 Phase 3 | Unknown | August 2014 | Taiwan ... more >> Kaohsiung Veterans General Hospital Recruiting Kaohsiung, Taiwan, 813 Contact: Wei-Lun Tsai, MD 886-7-3422121 ext 2075 tsaiwl@yahoo.com.tw Principal Investigator: Wei-Lun Tsai, MD Collapse << |
| NCT01247870 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> North Tees and Hartlepool NHS Trust Hartlepool, Cleveland, United Kingdom, TS24 9AH University Hospitals of Morecambe Bay NHS Trust Lancaster, Cumbria, United Kingdom, LA9 7RG East Sussex Healthcare NHS Trust Hastings, East Sussex, United Kingdom, TN37 7PT Blackpool Teaching Hospitals NHS Trust Blackpool, Lancashire, United Kingdom, FY3 8NR Lancashire Teaching Hospitals NHS Trust Preston, Lancashire, United Kingdom, PR2 9HT Sherwood Forest Hospitals NHS Trust Sutton-in-Ashfield, Nottinghamshire, United Kingdom, NG17 4JL Chelsea and Westminster Hospital London, United Kingdom, SW10 9NH St George's Hospital London, United Kingdom, SW17 0QT Freeman Hospital Newcastle, United Kingdom, NE7 7DN Collapse << |
| NCT03479476 | Fragile X Syndrome ... more >> Fragile X Mental Retardation Syndrome Mental Retardation, X Linked Genetic Diseases, X-Linked Trinucleotide Repeat Expansion Fra(X) Syndrome Intellectual Disability FXS Neurobehavioral Manifestations Sex Chromosome Disorders Collapse << | Phase 2 Phase 3 | Recruiting | May 2021 | United States, California ... more >> UC Davis MIND Institute Recruiting Sacramento, California, United States, 95817 Contact: Laura A Potter, BA 916-703-0471 Lapotter@ucdavis.edu Principal Investigator: Randi J Hagerman, MD Collapse << |
| NCT02773927 | Metabolic Syndrome | Phase 3 | Completed | - | Mexico ... more >> Universidad de Guadalajara Guadalajara, Jalisco, México, Mexico, 44100 Collapse << |
| NCT01357148 | - | - | Terminated(Completion of MK-04... more >>31A-235 was rendered unnecessary, as the local oversight authority accepted in its stead the results of another study [MK-0431-234].) Collapse << | - | - |
| NCT02461667 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT01234649 | Gestational Diabetes Mellitus ... more >> Type 2 Diabetes Mellitus Metabolic Syndrome Impaired Glucose Tolerance Disorder of Glucose Regulation Collapse << | Phase 3 | Active, not recruiting | August 2019 | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70815 Collapse << |
| NCT02948283 | Anemia Fatigu... more >>e Fever Lymphadenopathy Lymphocytosis Night Sweats Recurrent Chronic Lymphocytic Leukemia Recurrent Plasma Cell Myeloma Refractory Chronic Lymphocytic Leukemia Refractory Plasma Cell Myeloma Splenomegaly Thrombocytopenia Weight Loss Collapse << | Phase 1 | Active, not recruiting | October 2019 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << |
| NCT01342744 | Metabolic Syndrome | Phase 4 | Unknown | March 2012 | Thailand ... more >> Siriraj Hospital Recruiting Bangkoknoi, Bangkok, Thailand, 10700 Contact: Suchada Indhavivadhana, M.D. 0-2419-4657 sisto@mahidol.ac.th Collapse << |
| NCT02752113 | Diabetes Mellitus Type 2 | Phase 3 | Recruiting | January 2019 | Germany ... more >> Clinical Research Center, Dept of Nephrology and Hypertenison, University of Erlangen/Nürnberg Recruiting Erlangen, Bavaria, Germany, 91054 Contact: Christian Ott, MD +49-9131-85 ext 36245 christian.ott@uk-erlangen.de Contact: Roland Schmieder, Prof. MD +49 9131 85 ext 36245 roland.schmieder@uk-erlangen.de Principal Investigator: Roland Schmieder, Prof. MD Sub-Investigator: Christian Ott, MD Collapse << |
| NCT03416127 | Diabetes Mellitus, Type 2 | Phase 2 | Not yet recruiting | May 1, 2019 | Mexico ... more >> Intstituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Not yet recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: MANUEL GONZALEZ, PhD +523310585200 ext 34212 uiec@prodigy.net.mx Contact: KARINA PEREZ RUBIO, PhD +523310585200 ext 34212 karina2410@hotmail.com Collapse << |
| NCT02524821 | Healthy Overweight Obese | Not Applicable | Completed | - | Canada, Quebec ... more >> Quebec City, Quebec, Canada Collapse << |
| NCT02546050 | Healthy | Phase 1 | Completed | - | Denmark ... more >> The Novo Nordisk Foundation Center of Basic Metabolic Research, Section for Metabolic Genetics, University of Copenhagen Copenhagen, Copenhagen Ø, Denmark, 2100 Collapse << |
| NCT01237522 | Metformin Org... more >>anic Cation Transporter 2 Polymorphism,Single Nucleotide Collapse << | Phase 4 | Completed | - | Denmark ... more >> Institute of Public Health, Clinical Pharmacology, University of Southern Denmark Odense, Denmark, 5000 Collapse << |
| NCT02931253 | Atrial Fibrillation | Phase 2 | Terminated(Recruitment issue E... more >>nrollment expectation not met) Collapse << | - | United States, Pennsylvania ... more >> The Guthrie Clinic Sayre, Pennsylvania, United States, 18840 Collapse << |
| NCT01666665 | Insulin Resistance ... more >> Hypermetabolism Hyperglycemia Collapse << | Phase 2 Phase 3 | Recruiting | August 2020 | United States, Texas ... more >> Shriners Hospitals for Children Recruiting Galveston, Texas, United States, 77551 Contact: Cathy Reed, BSN 409-770-6987 ca2reed@utmb.edu Contact: Deb Benjamin, MSN 409-770-6731 dbenjami@utmb.edu Principal Investigator: David N Herndon, MD Sub-Investigator: Oscar Suman, PhD Collapse << |
| NCT01980823 | Breast Cancer ... more >> Breast Tumors Cancer of Breast Collapse << | Early Phase 1 | Recruiting | April 2018 | United States, New York ... more >> Columbia University Medical Center - Herbert Irving Cancer Center Recruiting Manhattan, New York, United States, 10032 Contact: Dan Otap 212-342-0937 do2267@cumc.columbia.edu Principal Investigator: Kevin Kalinsky, MD Collapse << |
| NCT01879293 | Left Ventriclar Mass | Phase 4 | Completed | - | China, Hubei ... more >> Wuhan General Hospital Wuhan, Hubei, China, 430070 Collapse << |
| NCT02823691 | Neuroendocrine Tumors | Early Phase 1 | Recruiting | April 2022 | Italy ... more >> NationalCIMilan Recruiting Milan, Italy, 20133 Contact: Sara Pusceddu, MD +390223903251 sara.pusceddu@istitutotumori.mi.it Collapse << |
| NCT02028767 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> 1276.24.001 Boehringer Ingelheim Investigational Site Toronto, Ontario, Canada Collapse << |
| NCT02028767 | - | - | Completed | - | - |
| NCT00723307 | Coronary Artery Disease | Phase 4 | Completed | - | United Kingdom ... more >> Glasgow Clinical Research Facility, NHS Greater Glasgow and Clyde Glasgow, United Kingdom, G11 6NT Collapse << |
| NCT01540487 | Healthy | Phase 1 | Completed | - | China ... more >> 1288.5.86001 Boehringer Ingelheim Investigational Site Beijing, China Collapse << |
| NCT01813929 | Type 1 Diabetes | Phase 4 | Active, not recruiting | December 2018 | United States, Colorado ... more >> University of Colorado Denver Aurora, Colorado, United States, 80045 Collapse << |
| NCT01483560 | Diabetes Mellitus, Type 1 | Phase 3 | Completed | - | Australia ... more >> Royal Melbourne Hospital Melbourne, Australia St Vincent's Hospital Melbourne, Australia Royal Prince Albert Hospital Sydney, Australia Canada St Joseph's Health Care Ontario, Canada Ottawa Hospital Riverside Campus Ottawa, Canada Denmark Steno Diabetes Centre Gentofte, Denmark Netherlands Maastricht University Medical Centre Maastricht, Netherlands United Kingdom Aberdeen Royal Infirmary Aberdeen, United Kingdom Ayr Hospital Ayr, United Kingdom, KA6 6DX University Hospitals Bristol Bristol, United Kingdom, BS2 8HW Diabetes Support Unit, Ninewells Hospital and Medical School Dundee, United Kingdom University Hospital North Durham Durham, United Kingdom Edinburgh Royal Infirmary Edinburgh, United Kingdom Edinburgh Western Infirmary Edinburgh, United Kingdom Peninsula NIHR Clinical Research Facility, Royal Devon and Exeter NHS Foundation Trust Exeter, United Kingdom Stobhill Hospital, Diabetes Clinic Glasgow, United Kingdom Gloucestershire Royal Hospital Gloucester, United Kingdom, GL1 3NN Michael White Diabetes Centre, Hull Royal Infirmary Hull, United Kingdom Clinical Sciences Centre, University Hospital Liverpool, United Kingdom Clinical Investigation Unit, International Centre for Circulatory Health, Imperial College Healthcare NHS Trust London, United Kingdom Wellcome Trust Clinical Research Facility, Manchester Royal Infirmary Manchester, United Kingdom Newcastle NIHR Clinical Research Facility, Royal Victoria Hospital Newcastle, United Kingdom Diabetes Clinical Research Centre, Plymouth Plymouth, United Kingdom Salford Royal NHS Foundation Trust Salford, United Kingdom Collapse << |
| NCT02125305 | Healthy Elderly | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul, Korea, Republic of Collapse << |
| NCT02071368 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01540487 | - | - | Completed | - | - |
| NCT01756105 | Gestational Diabetes ... more >> Metformin Treatment Oxidative Stress Collapse << | Phase 2 | Terminated | - | France ... more >> Hôpitaux Civils de Colmar Colmar, France, 68024 CHU Dijon, Dijon, France, 21030 Centre hospitalier de Mulhouse Mulhouse, France, 68070 CHU de Reims Reims, France, 51092 Hôpitaux Universitaires de Strasbourg Strasbourg, France, 67091 Collapse << |
| NCT02633488 | Metabolic Syndrome ... more >> Insulin Sensitivity Collapse << | Not Applicable | Completed | - | United States, Virginia ... more >> University of Virginia Charlottesville, Virginia, United States, 22903 Collapse << |
| NCT01831674 | Fasting | Phase 1 | Completed | - | India ... more >> Veeda Clinical Research Pvt. Ltd. Ahmedabad, Gujarat, India, 380 015 Collapse << |
| NCT02523898 | Polycystic Ovary Syndrome ... more >> Hyperandrogenism Collapse << | Phase 2 | Enrolling by invitation | August 2020 | - |
| NCT03507413 | Abdominal Aortic Aneurysm | Phase 2 Phase 3 | Not yet recruiting | November 15, 2022 | Austria ... more >> Medical University of Vienna, Department of Surgery, Division of Vascular Surgery Not yet recruiting Vienna, Austria, 1090 Contact: Wolf Eilenberg, MD +43140400 ext 56200 wolf.eilenberg@meduniwien.ac.at Collapse << |
| NCT03525028 | Uveitis Metfo... more >>rmin Glucocorticoid Collapse << | Not Applicable | Not yet recruiting | January 1, 2023 | China, Tianjin ... more >> Tianjin Medical University Eye Hospital Recruiting Tianjin, Tianjin, China, 300000 Collapse << |
| NCT03331861 | Heart Failure | Phase 2 | Not yet recruiting | December 2020 | - |
| NCT02644954 | Psoriasis | Phase 3 | Unknown | - | - |
| NCT03686657 | Type 2 Diabetes ... more >> High Blood Pressure Arthritis Obesity Collapse << | Phase 1 Phase 2 | Not yet recruiting | August 31, 2021 | United States, New York ... more >> Albany Medical College Not yet recruiting Albany, New York, United States, 12206 Contact: Kevin Fiesthamel, MS 518-264-4472 feistht@amc.edu Contact: Spencer Phelps, CCRC 518-264-4474 phelpss2@amc.edu Principal Investigator: Robert Busch, MD Sub-Investigator: Grimm Loretta, FNP-C Collapse << |
| NCT02672488 | Hepatocellular Carcinoma | Phase 2 | Unknown | December 2018 | China ... more >> Tianjin Medical University Cancer Institute and Hospital Recruiting Tianjin, China, 300060 Contact: TI ZHANG, MD +862223340123 ext 3092 zhangti@tjmuch.com Contact: Huikai Li, MD +862223340123 ext 3091 lihuikai@tjmuch.com Collapse << |
| NCT02571608 | Diabetes Mellitus Type 2 | Phase 4 | Not yet recruiting | June 2020 | - |
| NCT03229057 | PCOS | Phase 3 | Recruiting | April 30, 2022 | United States, Pennsylvania ... more >> University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Anuja Dokras Collapse << |
| NCT02810132 | Heart Failure, Systolic | Phase 2 | Completed | - | Denmark ... more >> Aarhus University Hospital, Department of Cardiology, Palle Juul-Jensens Boulevard 99 Aarhus N, Denmark, 8200 Collapse << |
| NCT01996696 | Prostatic Neoplasm | Phase 2 | Recruiting | September 2021 | Canada, Alberta ... more >> Cross Cancer Institute Recruiting Edmonton, Alberta, Canada, T6G 1Z2 Contact: Nawaid Usmani, MD 780-432-8518 Nawaid.usmani@albertahealthservices.ca Canada, Manitoba Prairie Mountain Health - Western Manitoba Cancer Centre Not yet recruiting Brandon, Manitoba, Canada, R7A2B3 Contact: Joelle Dumontier 204-578-2108 jdumontier@pmh-mb.ca Contact: William Hunter, MD 204-578-2208 whunter@cancercare.mb.ca Principal Investigator: William Hunter, MD CancerCare Manitoba Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Contact: Julian Kim, MD 204-787-4760 jkim7@cancercare.mb.ca Collapse << |
| NCT00941382 | Obesity | Phase 3 | Unknown | September 2009 | Mexico ... more >> Centro Universitario de Ciencias de la Salud Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03452306 | Bioequivalence | Phase 1 | Completed | - | Russian Federation ... more >> Yarosslavl Clinical Hospital #3 Yaroslavl, Russian Federation, 150007 Collapse << |
| NCT03458208 | Bioequivalence | Phase 1 | Completed | - | Russian Federation ... more >> Yarosslavl Clinical Hospital #3 Yaroslavl, Russian Federation, 150007 Collapse << |
| NCT02360618 | Bladder Cancer | Phase 2 | Unknown | January 2018 | Canada, Ontario ... more >> London Health Sciences Center; Victoria Hospital Recruiting London, Ontario, Canada, N6A 5W9 Contact: Jonathan Izawa, MD Collapse << |
| NCT02065752 | Healthy Volunteers | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01831661 | Fasting | Phase 1 | Completed | - | India ... more >> Veeda Clinical Research Pvt. Ltd. Ahmedabad, Gujarat, India, 380 015 Collapse << |
| NCT03031821 | Prostate Cancer ... more >> Metabolic Syndrome Collapse << | Phase 3 | Not yet recruiting | June 1, 2023 | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Cancer Centre Not yet recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Bernie Eigl, MD 604-877-6000 bernie.eigl@bccancer.bc.ca Principal Investigator: Bernie Eigl, MD Collapse << |
| NCT01154348 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Dennis Ruff, MD San Antonio, Texas, United States, 78209 Collapse << |
| NCT00864669 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT02319200 | Hepatocellular Carcinoma ... more >> Hepatitis C, Chronic Cirrhosis Collapse << | Phase 3 | Terminated(Decision of investi... more >>gator) Collapse << | - | France ... more >> Roulot Dominique Bobigny, France, 93009 Collapse << |
| NCT01340300 | Colorectal Cancer ... more >> Breast Cancer Collapse << | Phase 2 | Completed | - | United States, Connecticut ... more >> Yale School of Medicine New Haven, Connecticut, United States, 06520 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01305551 | Type 2 Diabetes | Phase 1 | Withdrawn | - | Brazil ... more >> Local Institution Campinas, Sao Paulo, Brazil, 13073 Collapse << |
| NCT03137186 | Prostate Cancer | Phase 2 | Recruiting | January 2020 | Egypt ... more >> Mansoura university Recruiting Mansourah, Egypt, 35516 Contact: Reham Alghandour 00201002682875 Rema-200@hotmail.com Contact: Ahmed Elshaal 00201001855490 Elshalam@hotmail.com Collapse << |
| NCT00865748 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT00865839 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT03048500 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage III Non-Small Cell Lung Cancer Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 2 | Recruiting | February 2021 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Young K. Chae 312-926-6895 Principal Investigator: Young K. Chae, MD, MPH, MBA Sub-Investigator: Sachin Pai, MD Sub-Investigator: Nisha Mohindra, MD Sub-Investigator: Victoria Villaflor, MD Sub-Investigator: Ricardo Costa, MD Sub-Investigator: Valerie Nelson, MD Sub-Investigator: Timothy Taxter, MD Northwestern University- Lake Forest Hospital Recruiting Lake Forest, Illinois, United States, 60045 Contact: Valerie Nelson, MD 847-582-2134 Principal Investigator: Valerie Nelson, MD Collapse << |
| NCT01340300 | - | - | Completed | - | - |
| NCT01579812 | Ovarian, Fallopian Tube, and P... more >>rimary Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01855763 | Gestational Diabetes ... more >> Type 2 Diabetes Pregnancy Collapse << | Phase 2 Phase 3 | Unknown | December 2013 | - |
| NCT02472353 | Breast Cancer ... more >> Breast Tumors Collapse << | Phase 2 | Completed | - | United States, South Dakota ... more >> Avera Cancer Institute Sioux Falls, South Dakota, United States, 57105 Collapse << |
| NCT01816659 | Colorectal Carcinoma | Phase 1 | Terminated(Slow Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02274090 | Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT01579812 | - | - | Completed | - | - |
| NCT02325401 | Head and Neck Cancer | Phase 1 | Active, not recruiting | March 2021 | United States, Ohio ... more >> University of Cincinnati Cancer Institute Cincinnati, Ohio, United States, 45267 Collapse << |
| NCT00588172 | Type 2 Diabetes | Phase 4 | Withdrawn(No funding) | June 2011 | - |
| NCT01711320 | Healthy | Phase 1 | Completed | - | China, Shaanxi ... more >> Department of pharmacy, Xijing Hospital, Fourth Military Medical University Xi'an, Shaanxi, China Collapse << |
| NCT01654640 | Schizophrenia | Phase 4 | Terminated(Failure to recruit ... more >>the planned number of participants) Collapse << | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT00834587 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> PRACS Institute Ltd. East Grand Forks, Minnesota, United States, 56721 United States, North Dakota PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT03109873 | Larynx Lip ... more >> Oral Cavity Pharynx Collapse << | Early Phase 1 | Active, not recruiting | June 2020 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT03310749 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Mexico ... more >> Unidad de Farmacología Clínica de la Facultad de Medicina de la Universidad Nacional Autónoma de México Nezahualcóyotl, Estado de México, Mexico, 57740 Collapse << |
| NCT01055691 | Healthy Volunteers | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT01383356 | - | - | Completed | - | - |
| NCT00834587 | - | - | Completed | - | - |
| NCT00835497 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> PRACS Institute Ltd. East Grand Forks, Minnesota, United States, 56721 United States, North Dakota PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00835497 | - | - | Completed | - | - |
| NCT01394887 | Glucose Intolerance ... more >> Inflammation Diabetes Collapse << | Phase 2 Phase 3 | Completed | - | Mexico ... more >> Endocrine Outpatient Clinic of the Hospital de Pediatria del CMN "Siglo XXI" Mexico City, DF, Mexico, 06720 Collapse << |
| NCT01383356 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Canada, Ontario ... more >> 1288.19.1 Boehringer Ingelheim Investigational Site Toronto, Ontario, Canada Collapse << |
| NCT02336087 | Pancreatic Adenocarcinoma ... more >> Unresectable Pancreatic Carcinoma Collapse << | Phase 1 | Recruiting | August 2019 | United States, California ... more >> City of Hope Medical Center Recruiting Duarte, California, United States, 91010 Contact: Vincent Chung 626-471-9200 vchung@coh.org Principal Investigator: Vincent Chung City of Hope Rancho Cucamonga Recruiting Rancho Cucamonga, California, United States, 91730 Contact: Valerie Estala 626-256-4673 ext 81699 Sub-Investigator: Behnam Ebrahimi, MD City of Hope South Pasadena Recruiting South Pasadena, California, United States, 91030 Contact: Odessa Rodriguez 626-256-4673 ext 81409 orodriguez@coh.org Sub-Investigator: Christina Yeon, MD City of Hope West Covina Recruiting West Covina, California, United States, 91790 Contact: Meizi Zheng 626-256-4673 ext 81336 Sub-Investigator: Gargi Upadhyaya, MD Collapse << |
| NCT00678080 | Pregnancy Complications | Not Applicable | Completed | - | United States, Texas ... more >> Valley Baptist Hospital Brownsville, Texas, United States, 78520 Lyndon B Johnson Hospital Houston, Texas, United States, 77026 Memorial Hermann Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT00678080 | - | - | Completed | - | - |
| NCT03398824 | Fanconi Anemia | Phase 2 | Recruiting | March 2021 | United States, Massachusetts ... more >> Boston Children's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Ashley E Kuniholm 617-355-6513 ashley.kuniholm@childrens.harvard.edu Contact: Jacob Cotton 617-919-4227 jacob.cotton@childrens.harvard.edu Principal Investigator: Akiko Shimamura, MD, PhD Sub-Investigator: Elissa Furutani, MD Sub-Investigator: Erica Esrick, MD Collapse << |
| NCT03467971 | Healthy | Phase 1 | Completed | - | Mexico ... more >> CECYPE Mexico City, Mexico Collapse << |
| NCT02019979 | Non-small Cell Lung Cancer Sta... more >>ge IIIB/IV Non-small Cell Lung Cancer Metastatic Nonsquamous Nonsmall Cell Neoplasm of Lung Collapse << | Phase 2 | Terminated(PI left the institu... more >>tion) Collapse << | - | United States, New York ... more >> Beth Israel Medical Center New York, New York, United States, 10003 Beth Israel Comprehensive Cancer Center New York, New York, United States, 10011 St.Luke's-Roosevelt Hospital Center New York, New York, United States, 10019 Mount Sinai Ichan School of Medicine New York, New York, United States, 10029 Collapse << |
| NCT02019979 | - | - | Terminated(PI left the institu... more >>tion) Collapse << | - | - |
| NCT01438723 | Cardiovascular Disease ... more >> Ischemic Heart Disease Collapse << | Phase 4 | Completed | - | Netherlands ... more >> RUNMC Nijmegen, Gelderland, Netherlands Collapse << |
| NCT01273584 | Pregnancy Complications ... more >> Obesity Collapse << | Phase 2 Phase 3 | Completed | - | United Kingdom ... more >> Medway Hospital NHS Trust Gillingham, Kent, United Kingdom, ME7 5NY Epsom and St Helier University Hospitals NHS Trust Carshalton, Surrey, United Kingdom, SM5 1AA Kings College, London London, United Kingdom, SE5 8RX Collapse << |
| NCT01929512 | Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01573949 | Diabetes Hear... more >>t Failure Collapse << | Not Applicable | Completed | - | United States, California ... more >> Ahmanson-UCLA Cardiomyopathy Center Los Angeles, California, United States, 90095 Collapse << |
| NCT02273050 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Changchun, China Research Site Chuangchun, China Research Site Fuzhou, China Research Site Guiyang, China Research Site Ha'er bin, China Research Site Hangzhou, China Research Site Hefei, China Research Site Jinan, China Research Site Nanchang, China Research Site Nanjing, China Research Site Shanghai, China Research Site Shijiazhuang, China Research Site Siping, China Research Site Tianjin, China Research Site Wuxi, China Research Site Yueyang, China Collapse << |
| NCT02505516 | Bladder Cancer ... more >> Diabetes Collapse << | Early Phase 1 | Completed | - | Ghana ... more >> Universty of Capecoast Cape Coast, Ghana Collapse << |
| NCT03107884 | Atrophy Insul... more >>in Resistance Collapse << | Early Phase 1 | Recruiting | July 1, 2021 | United States, Utah ... more >> University of Utah Recruiting Salt Lake City, Utah, United States, 84108 Contact: Amy Rogers 801-585-5961 amy.rogers@hsc.utah.edu Collapse << |
| NCT02273050 | - | - | Completed | - | - |
| NCT02577315 | Healthy | Phase 1 | Completed | - | Russian Federation ... more >> 1276.27.001 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation Collapse << |
| NCT02949700 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 1 Phase 2 | Recruiting | December 2021 | United States, Texas ... more >> Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Michael E. DeBakey Veterans Affairs Medical Center Recruiting Houston, Texas, United States, 77030 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Harris Health System - Smith Clinic Recruiting Houston, Texas, United States, 77054 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Collapse << |
| NCT02577315 | - | - | Completed | - | - |
| NCT01405118 | Rheumatoid Arthritis | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT01336322 | Pre-diabetes | Phase 2 | Recruiting | December 2018 | Italy ... more >> Department of Endocrinology and Metabolism, University of Pisa Recruiting Pisa, Italy, 56124 Contact: Stefano Del Prato, MD +39 050 995103 stefano.delprato@med.unipi.it Sub-Investigator: Graziano Di Cianni, MD Collapse << |
| NCT02498522 | Polycystic Ovary Syndrome | Early Phase 1 | Completed | - | Egypt ... more >> Faculty of Medicine, Ain Shams University Cairo, Al Qahirah, Egypt Collapse << |
| NCT02498522 | - | - | Completed | - | - |
| NCT01589445 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Bangladesh ... more >> Bangladesh Institute of Research and Rehabilitation in Diabetes, Endocrine and Metabolic Disorders (BIRDEM) Dhaka, Bangladesh, 1000 Collapse << |
| NCT01589445 | - | - | Completed | - | - |
| NCT01632020 | Colorectal Neoplasms | Phase 2 | Terminated(Inadequate accrual) | - | United States, Arkansas ... more >> Central Arkansas Veterans Heathcare System Little Rock, Arkansas, United States, 72205 University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT01632020 | - | - | Terminated(Inadequate accrual) | - | - |
| NCT03076281 | Larynx LIP ... more >> Oral Cavity Pharynx Collapse << | Phase 2 | Recruiting | August 2020 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Jennifer Johnson, MD, PhD 215-955-8874 jennifer.johnson@jefferson.edu Collapse << |
| NCT02796144 | Schizophrenia ... more >> Schizoaffective Disorder Overweight Collapse << | Phase 4 | Recruiting | March 2020 | United States, New York ... more >> New York State Psychiatric Institute (NYSPI), Columbia University Not yet recruiting New York, New York, United States, 11032 Contact: Scott Stroup, MD, MPH 646-774-8435 stroups@nyspi.columbia.edu United States, North Carolina University of North Carolina at Chapel Hill Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Lars F Jarskog, MD 919-843-7683 lars_jarskog@med.unc.edu Collapse << |
| NCT02050009 | Ovarian Papillary Serous Carci... more >>noma Ovarian Serous Cystadenocarcinoma Recurrent Fallopian Tube Cancer Recurrent Ovarian Epithelial Cancer Recurrent Ovarian Germ Cell Tumor Recurrent Primary Peritoneal Cavity Cancer Collapse << | Phase 1 | Withdrawn(study was not initia... more >>ted) Collapse << | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT03054519 | Peripheral Artery Disease | Phase 3 | Recruiting | September 30, 2023 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu United States, Louisiana Tulane University Recruiting New Orleans, Louisiana, United States, 70112 Contact: Lydia Bazzano, MD 504-842-4747 lbazzano@tulane.edu Contact: Shannon Terry 504-988-7249 sterry3@tulane.edu Ochsner Medical Center Recruiting New Orleans, Louisiana, United States, 70121 Contact: Lydia Bazzano, MD 504-842-4747 lbazzano@tulane.edu Contact: Grace Gartel 504-897-5844 ggartel@tulane.edu Collapse << |
| NCT02221180 | Bioequivalence | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT02179788 | Low Milk Supply ... more >> Pre-diabetes Insulin Resistance Suppressed Lactation Collapse << | Phase 1 Phase 2 | Completed | - | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 Collapse << |
| NCT00728351 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02844803 | Diabetes Mellitus | Early Phase 1 | Unknown | - | Korea, Republic of ... more >> Dongguk University Ilsan Hospital Recruiting Goyang, Gyeonggi, Korea, Republic of, 410-773 Contact: Namyi Gu, MD, PhD +82-31-961-8440 namyi.gu@gmail.com Collapse << |
| NCT00930579 | Breast Cancer | Phase 2 | Unknown | March 2014 | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT03686722 | Diabetes Mellitus, Type 2 ... more >> Hepatitis C Drug Interactions Collapse << | Phase 1 | Completed | - | Egypt ... more >> Drug research centre Cairo, Egypt Collapse << |
| NCT01911468 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02778776 | Metabolic Syndrome | Phase 3 | Completed | - | Mexico ... more >> Universidad de Guadalajara Guadalajara, Jalisco, México, Mexico, 44100 Collapse << |
| NCT02325245 | Pre-frail Elderly | Phase 3 | Unknown | March 2017 | Indonesia ... more >> Cipto Mangunkusumo General Hospital Recruiting Central Jakarta, Jakarta, Indonesia, 10430 Contact: Purwita Laksmi, MD +62-21-3918301 pwlaksmi@yahoo.com Collapse << |
| NCT02670018 | Healthy | Phase 1 | Unknown | March 2016 | Korea, Republic of ... more >> Yonsei University College of Medicine Seoul, Korea, Republic of Collapse << |
| NCT00820573 | Diabetes | Phase 4 | Completed | - | United States, Texas ... more >> Texas Diabetes Institute San Antonio, Texas, United States, 78207 Collapse << |
| NCT00856284 | - | - | Completed | - | - |
| NCT02745886 | Overweight Subjects ... more >> Metformin Aging Collapse << | Phase 4 | Unknown | December 2016 | China, Hubei ... more >> Zhang Jiajia Recruiting Wuhan, Hubei, China, 430070 Contact: Guangda Xiang, MD, PhD 02750772981 Guangda64@hotmail.com Collapse << |
| NCT02394652 | Uterine Cervical Neoplasms ... more >> Squamous Cell Carcinoma Adenocarcinoma Carcinoma, Adenosquamous Collapse << | Phase 2 | Recruiting | December 2019 | Canada, Alberta ... more >> Tom Baker Cancer Centre Not yet recruiting Calgary, Alberta, Canada, T2N 4N2 Contact: Corinne Doll, M.D. 403-521-3685 Cross Cancer Institute Not yet recruiting Edmonton, Alberta, Canada, T6G 1Z2 Contact: Ericka Wiebe, M.D. 480-432-8755 Canada, Ontario Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Kathy Han, M.D., M.Sc. 416-946-2919 Canada, Quebec Hôpital Maisonneuve-Rosemont Not yet recruiting Montréal, Quebec, Canada, H1T 2M4 Contact: Eve-Lyne Marchand, M.D., Ph.D. 514-252-3425 Centre Hospitalier De L'Université de Montréal Not yet recruiting Montréal, Quebec, Canada, H2L 4M1 Contact: Maroie Barkati, M.D. 514-890-8000 ext 28820 Collapse << |
| NCT00856284 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02990728 | Endometrial Cancer | Phase 2 | Enrolling by invitation | March 2020 | - |
| NCT00820573 | - | - | Completed | - | - |
| NCT01089179 | Type 2 Diabetes | Phase 1 | Completed | - | India ... more >> Torrent Research Centre Bhat, Gujarat, India, 382 428 Collapse << |
| NCT00332488 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00332488 | - | - | Completed | - | - |
| NCT03451006 | Aging Inflamm... more >>ation Frailty Collapse << | Phase 2 | Recruiting | April 30, 2020 | United States, Minnesota ... more >> Mayo Clinic in Rochester Recruiting Rochester, Minnesota, United States, 55905 Contact: Mandeep Singh, MD, MPH 507-255-5891 singh.mandeep@mayo.edu Contact: James Kirkland, MD, PhD 5072933346 kirkland.james@mayo.edu Collapse << |
| NCT03467945 | Healthy | Phase 1 | Completed | - | Germany ... more >> Please Contact the Merck KGaA Communication Center Darmstadt, Germany, 64293 Collapse << |
| NCT01595880 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01765023 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Pusan Paik Hospital Busan, Korea, Republic of, 614-735 Collapse << |
| NCT00835861 | Non Insulin Dependent Diabetes... more >> Pregnancy Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> University of North Carolina Hospitals Obstetric Clinics Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01541956 | Type 2 Diabetes | Phase 4 | Completed | - | China ... more >> Novartis Investigative Site Beijing, China, 100028 Collapse << |
| NCT03486626 | Polycystic Ovary Syndrome ... more >> Anovulatory Collapse << | Phase 4 | Active, not recruiting | October 2018 | Egypt ... more >> Ain Shams University Maternity Hospital Cairo, Egypt, 1156 Collapse << |
| NCT03004612 | Prediabetic State ... more >> Insulin Resistance Collapse << | Phase 4 | Active, not recruiting | August 2018 | Mexico ... more >> Universidad de Guanajuato León, Guanajuato, Mexico, 37670 Collapse << |
| NCT01941953 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | Brazil ... more >> Instituto Do Cancer Do Estado de São Paulo Sao Paulo, Brazil, 01246-000 Collapse << |
| NCT00835861 | - | - | Completed | - | - |
| NCT01662674 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT02794792 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Russian Federation ... more >> Site RU70011 Moscow, Russian Federation, 117036 Site RU70005 Moscow, Russian Federation, 119034 Site RU70003 Moscow, Russian Federation, 121374 Site RU70009 Moscow, Russian Federation, 125315 Site RU70010 Nizhniy Novgorod, Russian Federation, 603018 Site RU70006 Samara, Russian Federation, 443067 Site RU70004 Saratov, Russian Federation, 410012 Site RU70008 St. Petersburg, Russian Federation, 191119 Site RU70014 St. Petersburg, Russian Federation, 194354 Site RU70007 St. Petersburg, Russian Federation, 197022 Site RU70002 St. Petersburg, Russian Federation, 197706 Site RU70015 Volgograd, Russian Federation, 400001 Site RU70001 Yaroslavl, Russian Federation, 150003 Site RU70013 Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT01796028 | Prostatic Neoplasms | Phase 2 | Completed | - | France ... more >> Centre ANTOINE LACASSAGNE Nice, France, 06189 Collapse << |
| NCT03757923 | Menstrual Irregularities ... more >> Hyperandrogenism Collapse << | Phase 1 Phase 2 | Recruiting | January 2019 | Pakistan ... more >> Dow University Hospital OJHA Recruiting Karachi, Sindh, Pakistan, 75270 Collapse << |
| NCT01528046 | Solid Tumors ... more >>Primary Brain Tumors Collapse << | Phase 1 | Recruiting | December 2020 | United States, Connecticut ... more >> Connecticut Children's Medical Center Recruiting Hartford, Connecticut, United States, 06106 Contact: Robin Arens 860-545-9637 Rarens@ccmckids.org Principal Investigator: Michael Isakoff, M.D. Sub-Investigator: Donna Boruchov, M.D. Sub-Investigator: Kerry Moss, M.D. Sub-Investigator: Andrea Orsey, M.D. Sub-Investigator: Nehal Parikh, M.D. Sub-Investigator: Arnold Altman, M.D. United States, Delaware Nemours/Alfred I. duPont Hospital for Children, Delaware Recruiting Wilmington, Delaware, United States, 19803 Contact: Debra J. Bertz 302-651-5757 debra.bertz@nemours.org Principal Investigator: Edward A. Kolb, M.D. Sub-Investigator: Andrew Walter, M.D. Sub-Investigator: Jonathan Powell, M.D. Sub-Investigator: Emi Caywood, M.D. Sub-Investigator: Robin Miller, M.D. Sub-Investigator: Gregory Griffin, M.D. United States, Florida University of Florida Terminated Gainesville, Florida, United States, 32611 Nemours Children's Clinic Recruiting Jacksonville, Florida, United States, 32207 Contact: Ingrid Ingram 904-697-3985 iingram@nemours.org Principal Investigator: Scott Bradfield, M.D. Sub-Investigator: Eric Sandler, M.D. Sub-Investigator: Michael Joyce, M.D. Sub-Investigator: Cynthia Gauger, M.D. Sub-Investigator: Manisha Bansal, M.D. Sub-Investigator: Paul Pitel, M.D. Holtz Children's Hospital at the University of Miami Recruiting Miami, Florida, United States, 33124 Contact: Myriam Zayas 305-243-7846 MZayas2@med.miami.edu Principal Investigator: John Goldberg, M.D. Sub-Investigator: Cristina Fernandes, M.D. Sub-Investigator: Joanna Davis, M.D. Sub-Investigator: Antonello Podda, M.D. Sub-Investigator: Martin Andreansky, M.D. Sub-Investigator: Julio Barredo, M.D. Sub-Investigator: Ofelia Alvarez, M.D. Johns Hopkins All Children's Hospital Recruiting Saint Petersburg, Florida, United States, 33701 Contact: Ashley Repp, RN 727-767-4784 Ashley.Repp@allkids.org Contact: Frances Hamblin 727-767-2423 frances.hamblin@allkids.org Principal Investigator: Damon Reed, M.D. Sub-Investigator: Irmel Ayala, M.D. Sub-Investigator: Gregory Hale, M.D. Sub-Investigator: Nanette Grana, M.D. Sub-Investigator: Stacie Stapleton, M.D. Sub-Investigator: Kelly Sawczyn, M.D. Sub-Investigator: Jennifer Mayer, M.D. Sub-Investigator: Jody Kerr, M.D. Tampa General Hospital Recruiting Tampa, Florida, United States, 33606 Contact: Denise Fife 813-844-7829 dafife@tgh.org Principal Investigator: Cameron Tebbi, M.D. H. Lee Moffitt Cancer Center and Research Institute Recruiting Tampa, Florida, United States, 33612 Contact: Tiffany Smith 813-745-6250 tiffany.smith@moffitt.org Principal Investigator: Damon Reed, M.D. United States, Kentucky University of Kentucky Recruiting Lexington, Kentucky, United States, 40536 Contact: Tammy Taylor 859-323-6975 tammy.taylor@uky.edu Principal Investigator: Lars Wagner, M.D. United States, Maryland Johns Hopkins Sidney Kimmel Comprehensive Cancer Center Recruiting Baltimore, Maryland, United States, 21231 Contact: David Loeb, M.D. 410-502-7247 loebda@jhmi.edu Principal Investigator: David Loeb, M.D. United States, New York The Children's Hospital at Montefiore Recruiting Bronx, New York, United States, 10467 Contact: Noam Zeffren 718-741-2356 nzeffren@montefiore.org Principal Investigator: Jonathan Gill, M.D. United States, North Carolina University of North Carolina at Chapel Hill Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Sharon Guerry 919-966-1178 Contact: Patrick Thompson, M.D 919-966-1178 patom@email.unc.edu Principal Investigator: Patrick Thompson, M.D. United States, Ohio Nationwide Children's Hospital Recruiting Columbus, Ohio, United States, 43205 Contact: Amy Yekisa 614-722-6570 Amy.Yekisa@nationwidechildrens.org Principal Investigator: Bhuvana Setty, M.D. United States, Utah Primary Children's Medical Center/Utah Recruiting Salt Lake City, Utah, United States, 84113 Contact: Melissa Bolton 801-213-3909 melissa.bolton@hsc.utah.edu Principal Investigator: Holly Spraker-Perlman, M.D. Sub-Investigator: Richard Lemons, M.D., Ph.D. Sub-Investigator: Jennifer Wright, M.D. Collapse << |
| NCT03192293 | Breast Cancer | Phase 2 | Recruiting | January 20, 2019 | Singapore ... more >> National University Hospital Singapore Recruiting Singapore, Singapore, 119228 Collapse << |
| NCT01192139 | Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT02417090 | - | - | Enrolling by invitation | December 2019 | - |
| NCT02438540 | Insulin Sensitivity ... more >> Obesity Collapse << | Not Applicable | Completed | - | - |
| NCT01215032 | - | - | Terminated(slow accrual, compe... more >>ting clinical trials) Collapse << | - | - |
| NCT02438540 | - | - | Completed | - | - |
| NCT01529593 | Advanced Cancers | Phase 1 | Recruiting | March 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT01228240 | Diabetes Mellitus, Type 2 AND ... more >>Metformin Collapse << | Not Applicable | Completed | - | - |
| NCT01192139 | - | - | Completed | - | - |
| NCT01604213 | Type 2 Diabetes Mellitus ... more >> Ischemic Heart Disease Collapse << | Phase 4 | Completed | - | Israel ... more >> Sheba Medical Center, Cardiac Rehabilitation Institute Tel Hashomer, Israel, 52621 Collapse << |
| NCT03713801 | Aging Vaccine... more >> Response Impaired Collapse << | Phase 1 | Recruiting | December 31, 2019 | United States, Texas ... more >> UT Health San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Grace Lee, PhD 210-450-8097 leeg3@uthscsa.edu Collapse << |
| NCT01122316 | Heart Failure ... more >> Diabetes Collapse << | Not Applicable | Completed | - | United States, California ... more >> Ahmanson-UCLA Cardiomyopathy Center Los Angeles, California, United States, 90095-7368 Collapse << |
| NCT01215032 | Prostate Cancer | Phase 2 | Terminated(slow accrual, compe... more >>ting clinical trials) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00778349 | Healthy | Not Applicable | Completed | - | - |
| NCT01719614 | Healthy Participants | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT01929863 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT01573377 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Not Applicable | Completed | - | China, Chongqing ... more >> The First Affiliated Hospital of Chongqing Medical University Chongqing, Chongqing, China, 400016 Collapse << |
| NCT01677897 | Metastatic Prostate Cancer | Phase 2 | Unknown | April 2015 | Switzerland ... more >> Kantonsspital Graubünden Recruiting Chur, Graubünden, Switzerland, 7000 Principal Investigator: michael mark, md Collapse << |
| NCT02981329 | Sickle Cell Anemia ... more >> Sickle Cell Disease Hemoglobin Disorder Hemoglobin Disease; Sickle-Cell, Thalassemia Collapse << | Early Phase 1 | Recruiting | November 2022 | United States, Texas ... more >> Texas Children's Hospital Recruiting Houston, Texas, United States, 77030 Contact: Vivien Sheehan, MD 832-824-4459 vxsheeha@texaschildrens.org Contact: Bodgan Dinu brdinu@texaschildrens.org The University of Texas Health Science Center at Houston Recruiting Houston, Texas, United States, 77030 Contact: Harinder Juneja, MD Collapse << |
| NCT01068743 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT01441869 | Bioequivalence, Log-transforme... more >>d AUCss and Cmax,ss Values for Saxagliptin and Metformin Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01068743 | - | - | Completed | - | - |
| NCT01929863 | - | - | Completed | - | - |
| NCT02230995 | - | - | Completed | - | - |
| NCT02751307 | Metabolic Syndrome | Phase 4 | Completed | - | - |
| NCT01310231 | Metastatic Breast Cancer | Phase 2 | Completed | - | Canada, Ontario ... more >> London Regional Cancer Program London, Ontario, Canada St. Michael's Hospital Toronto, Ontario, Canada, M5B 1N9 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Windsor Regional Cancer Centre Windsor, Ontario, Canada, N8W 2X3 Collapse << |
| NCT02145559 | Breast Neoplasms ... more >> Lung Neoplasms Cancer of Liver Lymphoma Cancer of Kidney Collapse << | Phase 1 | Active, not recruiting | July 2019 | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT02230995 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach an der Riss, Germany Collapse << |
| NCT00865241 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT01725672 | Diabetes Mellitus, Type 2 | Phase 1 | Terminated(Due to changes in E... more >>MA guidelines on modified release dosage forms (Feb 2013; EMA/CHMP/EWP/280/96). The Part B formulation would not meet the new guidelines.) Collapse << | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT00864734 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT00865436 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT01808690 | Type 1 Diabetes | Phase 3 | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado and University of Colorado Denver Health Sciences Center Aurora, Colorado, United States, 80045 Collapse << |
| NCT01992211 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02280057 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Gynecology Dept. Herning Hospital Herning, Denmark, 7400 Collapse << |
| NCT03271866 | Schizophrenia | Not Applicable | Recruiting | March 1, 2019 | China, Hunan ... more >> The Second Xiangya Hospital of Central South University Recruiting Changsha, Hunan, China, 410011 Contact: Tiannan Shao, M.D., Ph.D. +86 13341318057 shaostndoc@163.com Contact: Renrong Wu, M.D., Ph.D. wurenrong2013@163.com Principal Investigator: Dongyu Kang, M.D. Collapse << |
| NCT01037660 | Cardiovascular Disease | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT02084082 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.9.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02084082 | - | - | Completed | - | - |
| NCT02121509 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.10.1 Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT02121509 | - | - | Completed | - | - |
| NCT00892073 | Hypothalamic Obesity | Phase 2 | Completed | - | Canada, Ontario ... more >> The Hospital for Sick Children Toronto, Ontario, Canada Collapse << |
| NCT01756417 | Healthy | Phase 1 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States Collapse << |
| NCT00159536 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Norway ... more >> University hospital of Bergen Bergen, Norway Central Hospital of Northern Norway Bodø, Norway Buskerud Hospital Drammen, Norway Haugesund Hospital Haugesund, Norway Ringerike Hospital Hønefoss, Norway Elvebredden Gynekologpraksis Kristiansand, Norway Lillehammer Hospital Lillehammer, Norway Stavanger University Hospital Stavanger, Norway Departments of Obstetrics and Gynecology and Endocrinology, St. Olav's Hospital Trondheim, Norway Ålesund Hospital Ålesund, Norway Collapse << |
| NCT01718093 | Type 1 Diabetes | Phase 4 | Completed | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT02212756 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju, Jeollabuk-do, Korea, Republic of, 561-712 Collapse << |
| NCT01523639 | Colorectal Cancer ... more >> Steatohepatitis Collapse << | Phase 2 | Terminated(Prematurely due to ... more >>slow recruitment (07/08/2013). Newly defined study end=LPLV=05/11/2013. ABCSG guaranteed completed treatment period for ethical reasons.) Collapse << | May 2014 | Austria ... more >> Medical University Graz, Oncology Graz, Styria, Austria, 8036 Medical University Innsbruck, Internal Medicine Innsbruck, Tyrol, Austria, 6020 Hospital St. Vinzenz Zams, Tyrol, Austria, 6511 Hospital BHS Ried Ried, Upper Austria, Austria, 4910 KH BHB Vienna Vienna, Austria, 1021 Med. Univ. Vienna, General Hospital Vienna Vienna, Austria, 1090 KH St. Josef KH Vienna, Austria, 1130 Collapse << |
| NCT03764605 | ADPKD | Phase 3 | Not yet recruiting | January 30, 2022 | Italy ... more >> AOUC "Policlinico" Bari, Italy, 70124 AOUConsorziale Policlinico Di Bari Not yet recruiting Bari, Italy, 70124 Contact: Loreto Gesualdo +390805594040 trialnefrobari@gmail.com Contact: Giovanni Piscopo +393299664404 giopiscopo@gmail.com Collapse << |
| NCT02853630 | TYpe 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | December 2018 | India ... more >> Jnana Sanjeevini Medical Centre Bangalore, Karnataka, India, 560 078 Vikas Pai Research Foundation Pune, Maharashtra, India, 411 005 Singhvi Health Centre Chennai, Tamil Nadu, India, 600 019 Dr V.Seshiah Diabetes research Institute, Dr V.Balaji Diabetes care centre Chennai, Tamil Nadu, India, 600029 Arthur Asirvatham Hospital Madurai, Tamil Nadu, India, 625 020 Ramana Maharishi Rangammal Hospital Tiruvannamalai, Tamil Nadu, India, 606 603 Collapse << |
| NCT01864174 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT00994812 | Polycystic Ovary Syndrome ... more >> Miscarriage Infertility Toxemia Gestational Diabetes Collapse << | Phase 3 | Completed | - | Finland ... more >> University Hospital Of Helsinki Helsinki, Finland University Hospital of Kuopio Kuopio, Finland University Hopsital of Oulu Oulu, Finland, 90029 University Hospital of Tampere Tampere, Finland University Hospital of Turku Turku, Finland Collapse << |
| NCT02712619 | Healthy | Phase 1 | Completed | - | - |
| NCT00845936 | Drug Induced Weight Gain | Phase 4 | Unknown | May 2010 | Israel ... more >> Beer-Yaacov MHC Not yet recruiting Beer Yaacov, Israel, 70350 Contact: Pazit Leibovich, MD 972-9284065 pazit.leib@gmail.com Sub-Investigator: Pazit Leibovich, MD Collapse << |
| NCT03309007 | PreDiabetes A... more >>ging Collapse << | Phase 3 | Recruiting | July 31, 2018 | United States, New Mexico ... more >> University of New Mexico Clincal & Translational Science Center Recruiting Albuquerque, New Mexico, United States, 87131 Contact: Mark R Burge, MD 505-272-4658 mburge@salud.unm.edu Collapse << |
| NCT02266472 | - | - | Completed | - | - |
| NCT01864174 | - | - | Completed | - | - |
| NCT02372656 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT00754403 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT03651531 | Gestational Diabetes | Phase 3 | Not yet recruiting | July 2020 | - |
| NCT02266472 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT00660907 | - | - | Completed | - | - |
| NCT01551615 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT00660907 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00650234 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT01205672 | Endometrial Cancer | Early Phase 1 | Completed | - | United States, Texas ... more >> Lyndon B. Johnson General Hospital Houston, Texas, United States, 77026 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00736385 | - | - | Terminated | - | - |
| NCT00736385 | Fatty Liver | Phase 4 | Terminated | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT01459445 | Polycystic Ovary Syndrome ... more >> Endothelial Dysfunction Collapse << | Not Applicable | Unknown | January 2012 | Romania ... more >> Clinic of Endocrinology Cluj-Napoca, Cluj, Romania, 400349 Collapse << |
| NCT02306070 | HIV Infection ... more >> Hepatitis C Collapse << | Phase 2 | Withdrawn(insufficient funding... more >>) Collapse << | April 3, 2018 | Canada, Ontario ... more >> The Ottawa Hospital, General Campus Ottawa, Ontario, Canada Collapse << |
| NCT01400191 | Pharmacogenetics of Metformin | Phase 4 | Completed | - | Denmark ... more >> Clinical pharmacology, Institute og public health, University of Southern Denmark Odense, Denmark, 5000 Collapse << |
| NCT02765347 | Diabetes Mellitus, Type 1 | Phase 4 | Completed | - | China, Guangdong ... more >> The Third Affliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT00648518 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01840007 | Metastatic Melanoma (Stage III... more >>C Non-résécable or no Surgically Curable or Stage IV With Classification AJCC) Collapse << | Phase 2 | Unknown | February 2014 | France ... more >> Bahadoran Recruiting Nice, Alpes-Maritimes, France, 06200 Contact: BAHADORAN Philipp, PhD +33492036484 bahadoran.p@chu-nice.fr Principal Investigator: BAHADORAN Philippe, Phd MORTIER Laurent Recruiting Lille, Nord, France, 59000 Contact: MORTIER Laurent, PhD +33 3.20.44.41.93 Laurent.mortier@chru-lille.fr Sub-Investigator: MORTIER Laurent, PhD Collapse << |
| NCT03393208 | Healthy | Phase 1 | Completed | - | China ... more >> Xuanwu Hospital Beijing, China, 100053 Collapse << |
| NCT00650312 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT02073227 | Healthy Volunteers | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT02745912 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT02548741 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | August 2017 | - |
| NCT00649948 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT01638676 | Melanoma | Phase 1 Phase 2 | Recruiting | June 2019 | United States, Kentucky ... more >> James Graham Brown Cancer Center-University of Louisville Recruiting Louisville, Kentucky, United States, 40202 Contact: Jason A Chesney, MD PhD 502-562-3429 jason.chesney@louisville.edu Contact: Sarah Lush, RN 502-540-1537 s.lush@louisville.edu Collapse << |
| NCT01093794 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02884089 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> Dallas Clinical Research Unit Dallas, Texas, United States, 75247 Collapse << |
| NCT02793505 | - | - | Completed | - | - |
| NCT02026310 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | China, Chongqing ... more >> The First Affiliated Hospital of Chongqing Medical University Chongqing,, Chongqing, China, 400016 Collapse << |
| NCT02980276 | Gestational Diabetes | Phase 3 | Recruiting | August 2020 | Ireland ... more >> Cork University Hospital Not yet recruiting Cork, Ireland Principal Investigator: Prof. Louise Kenny University Hospital Galway Recruiting Galway, Ireland Principal Investigator: Prof. Fidelma Dunne Collapse << |
| NCT01093794 | - | - | Completed | - | - |
| NCT01935804 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 2 | Completed | - | Saudi Arabia ... more >> Center of Excellence for Osteoporosis Research, King Abdulaziz University Jeddah, Makkah, Saudi Arabia, 21589 Collapse << |
| NCT02064374 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT00830076 | - | - | Completed | - | - |
| NCT01844531 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.6.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02884089 | - | - | Completed | - | - |
| NCT02802215 | Polycystic Ovary Syndrome ... more >> Diabetes, Gestational Collapse << | Phase 2 Phase 3 | Unknown | September 2016 | Egypt ... more >> Ain Shams maternity hospital Recruiting Cairo, Egypt Principal Investigator: Ahmed E Elbohoty, Ph.D. Collapse << |
| NCT00830076 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT02186847 | Adenosquamous Lung Carcinoma ... more >> Bronchioloalveolar Carcinoma Large Cell Lung Carcinoma Lung Adenocarcinoma Non-Small Cell Lung Carcinoma Recurrent Non-Small Cell Lung Carcinoma Squamous Cell Lung Carcinoma Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Collapse << | Phase 2 | Active, not recruiting | March 2023 | - |
| NCT00546741 | Healthy Male and Female Subjec... more >>ts Collapse << | Phase 1 | Completed | - | United States, New Jersey ... more >> Bristol-Myers Squibb Clinical Pharmacology Unit Hamilton, New Jersey, United States, 08690 Collapse << |
| NCT01844531 | - | - | Completed | - | - |
| NCT01068730 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT02360059 | Breast Cancer | Phase 2 | Terminated(Low Accrual) | - | United States, Texas ... more >> Lyndon B. Johnson General Hospital Houston, Texas, United States, 77026 Collapse << |
| NCT01099618 | Ketosis Prone Diabetes ... more >> Diabetes Ketoacidosis Hyperglycemia Collapse << | Phase 4 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Collapse << |
| NCT01068730 | - | - | Completed | - | - |
| NCT01099618 | - | - | Completed | - | - |
| NCT03655301 | Healthy Volunteers | Phase 1 | Active, not recruiting | January 18, 2019 | United States, Texas ... more >> Pharmaceutical Product Development, LLC Austin, Texas, United States, 78744 Collapse << |
| NCT02394158 | Gestational Diabetes Mellitus | Phase 4 | Unknown | July 2017 | United Kingdom ... more >> Imperial College NHS Trust Recruiting London, United Kingdom, W2 1PG Contact: Stephen Robinson, FRCP, MD 0203 312 1253 Contact: Rochan Agha-Jaffar, BMBS, MRCP 0207 594 6140 r.agha-jaffar@imperial.ac.uk London North West Healthcare Trust Recruiting London, United Kingdom Contact: Mushtaqur Rahman, FRCP, PhD Contact: Rochan Agha-Jaffar, BMBS, MRCP 0207 594 6140 r.agha-jaffar@imperial.ac.uk Collapse << |
| NCT01799057 | Peripheral Arterial Disease ... more >> Intermittent Claudication Collapse << | Phase 4 | Terminated(The trial has been ... more >>terminated due to difficulties with recruitment.) Collapse << | - | Australia, Victoria ... more >> Baker IDI Heart and Diabetes Institute Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT02360059 | - | - | Terminated(Low Accrual) | - | - |
| NCT01196546 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Thailand ... more >> BMA Medical College and Vajira Hospital Bangkok, Thailand King Chulalongkorn Memorial Hospital Bangkok, Thailand Phamongkutklao Hospital Bangkok, Thailand Police General Hospital Bangkok, Thailand Siriraj Hospital Bangkok, Thailand Khon Kaen Hospital Khon Kaen, Thailand Fort Suranaree Hospital Nakhon Ratchasima, Thailand Maharat Nakhon Ratchasima Hospital Nakhon Ratchasima, Thailand Collapse << |
| NCT01273571 | Healthy | Phase 1 | Completed | - | - |
| NCT01593137 | Type 2 Diabetes | Phase 4 | Withdrawn(Lack of patients wit... more >>h the criteria established in the protocol.) Collapse << | December 2014 | Spain ... more >> Hospital Clínico San Carlos Madrid, Spain, 28040 Collapse << |
| NCT02696941 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> University of Oxford Oxford, United Kingdom Collapse << |
| NCT01929811 | pCR Rate BCT ... more >>Rate Safety Collapse << | Phase 2 | Recruiting | September 2020 | China, Shandong ... more >> Linyi People's Hospital Not yet recruiting Linyi, Shandong, China, 276003 Contact: Hong Liang Principal Investigator: Hong Liang China, Shanghai Ruijin Hospital, Shanghai Jiaotong University School of Medicine Recruiting Shanghai, Shanghai, China, 200025 Contact: Xiaosong Chen Principal Investigator: Kunwei Shen, Dr Collapse << |
| NCT01068717 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT02951260 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT01068717 | - | - | Completed | - | - |
| NCT00918138 | - | - | Completed | - | - |
| NCT01755494 | Type 2 Diabetes Mellitus(T2DM) | Phase 1 | Completed | - | China ... more >> Research Site Beijing, China Collapse << |
| NCT02986659 | Coronary Artery Disease ... more >> Mild Cognitive Impairment Obesity, Abdominal Hypertension Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> Wake Forest Health Sciences Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT00918138 | Type 2 Diabetes | Phase 3 | Completed | - | United States, Arizona ... more >> Dedicated Phase I, Inc. Phoenix, Arizona, United States, 85013 United States, California Pacific Sleep Medicine Services (Avastra Clinical Trials) Redlands, California, United States, 92373 Orange County Research Center Tustin, California, United States, 92780 United States, Florida Clinical Research Of South Florida Coral Gables, Florida, United States, 33134 Palm Springs Research Institute Hialeah, Florida, United States, 33012 Healthcare Clinical Data, Inc. North Miami, Florida, United States, 33161 United States, Georgia River Birch Research Alliance, Llc Blue Ridge, Georgia, United States, 30513 United States, Michigan Jasper Clinic, Inc. Kalamazoo, Michigan, United States, 49007 United States, New York Clinilabs, Inc. New York, New York, United States, 10019 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 United States, Texas Clinical Trials Of Texas Inc. San Antonio, Texas, United States, 78229 United States, Utah Advanced Clinical Research West Jordan, Utah, United States, 84088 Argentina Local Institution Capital Federal, Buenos Aires, Argentina, 1429 Local Institution Buenos Aires, Argentina, 1425 Israel Local Institution Beer-Sheva, Israel, 84101 Local Institution Holon, Israel, 58100 Local Institution Kfar-Saba, Israel, 44281 Local Institution Zefat, Israel, 13100 Mexico Local Institution Monterrey, Nuevo Leon, Mexico, 64460 Local Institution Durango, Mexico, 34000 Local Institution Durango, Mexico, 34075 Collapse << |
| NCT01384279 | Obesity | Not Applicable | Completed | - | Taiwan ... more >> Beitou Armed Forces Hospital Taipei, Taiwan, 11243 Collapse << |
| NCT01544751 | Nonalcoholic Fatty Liver Disea... more >>se (NAFLD) Collapse << | Not Applicable | Unknown | June 2015 | Italy ... more >> Endocrinology Unit Recruiting Catanzaro, Italy, 88100 Contact: Angela Mazza, Post doc +03909613647110 angela_mazza@libero.it Contact: Barbara Fruci, Doctor +03909613647110 barbara.fruci@yahoo.it Principal Investigator: Antonino Belfiore, Professor Collapse << |
| NCT02250794 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | December 2015 | - |
| NCT02065687 | Endometrial Adenocarcinoma ... more >> Endometrial Clear Cell Adenocarcinoma Endometrial Serous Adenocarcinoma Endometrial Undifferentiated Carcinoma Recurrent Uterine Corpus Carcinoma Stage III Uterine Corpus Cancer AJCC v7 Stage IIIA Uterine Corpus Cancer AJCC v7 Stage IIIB Uterine Corpus Cancer AJCC v7 Stage IIIC Uterine Corpus Cancer AJCC v7 Stage IV Uterine Corpus Cancer AJCC v7 Stage IVA Uterine Corpus Cancer AJCC v7 Stage IVB Uterine Corpus Cancer AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | - | - |
| NCT01968317 | Endometrial Atypical Hyperplas... more >>ia Endometrial Adenocarcinoma Collapse << | Phase 2 | Completed | - | China, Shanghai ... more >> Obstetrics and Gynecology Hospital, Fudan University Shanghai, Shanghai, China, 200011 Fudan University Shanghai Cancer Center Shanghai, Shanghai, China, 200032 Shanghai Changning Maternity & Infant Health Hospital Shanghai, Shanghai, China Shanghai Sixth People's Hospital Shanghai, Shanghai, China China, Zhejiang Zhejiang Cancer Hospital Hangzhou, Zhejiang, China Collapse << |
| NCT02056600 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT03717701 | Pre-Eclampsia | Not Applicable | Not yet recruiting | January 1, 2021 | - |
| NCT02027103 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Diabetes Clinic, Vali-Asr Hospital, Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT00105066 | Obesity Hyper... more >>tension Hypercholesterolemia Hyperglycemia Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute on Aging (NIA), Harbor Hospital Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02519543 | Bipolar Disorder | Phase 3 | Recruiting | September 2019 | United States, Pennsylvania ... more >> Western Psychiatric Institute and Clinic, University of Pittsburgh Medical Center, University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213-2593 Contact: Roy Chengappa, MD FRCPC chengappakn@upmc.edu Contact: Patricia Schlicht, BN 412-246-5258 schlpx@UPMC.EDU Principal Investigator: Roy Chengappa, MD FRCPC Canada, Nova Scotia Nova Scotia Health Authority - Dept. of Psychiatry Recruiting Halifax, Nova Scotia, Canada, B3H 2E2 Contact: Julie S Garnham, BN (902) 473 7144 jgarnham@dal.ca Principal Investigator: Cynthia Calkin, MD FRCPC Collapse << |
| NCT01077479 | Metabolic Syndrome ... more >> Hypercholesterolemia Collapse << | Phase 2 | Completed | - | Australia, New South Wales ... more >> Westmead Hospital Sydney, New South Wales, Australia, 2145 Collapse << |
| NCT00105066 | - | - | Completed | - | - |
| NCT02605772 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | March 2017 | - |
| NCT01316042 | Premature Pubarche | Not Applicable | Terminated(poor recruitment at... more >> collaborating sites) Collapse << | - | United States, Indiana ... more >> Riley Hospital for Children, Indiana University School of Medicine Indianapolis, Indiana, United States, 46202 United States, Pennsylvania Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Children's Hospital of Pittsburgh at the University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT01316042 | - | - | Terminated(poor recruitment at... more >> collaborating sites) Collapse << | - | - |
| NCT01302743 | Diabetes | Not Applicable | Terminated(lack of recruitment... more >>) Collapse << | - | United States, Nevada ... more >> Mike O'Callaghan Federal Hospital Nellis Air Force Base, Nevada, United States, 89191 Collapse << |
| NCT03236740 | PCOS | Phase 4 | Completed | - | India ... more >> SCB Medical College Cuttack, Orissa, India, 753007 Collapse << |
| NCT01302743 | - | - | Terminated(lack of recruitment... more >>) Collapse << | - | - |
| NCT03200015 | Diffuse Large B Cell Lymphoma | Phase 2 | Active, not recruiting | January 27, 2019 | Mexico ... more >> Hospital Universitario Dr. José Eleuterio González Monterrey, Nuevo Leon, Mexico, 64610 Collapse << |
| NCT00842400 | Obesity | Phase 1 | Completed | - | United States, Arizona ... more >> MDS Pharma Services Phoenix, Arizona, United States, 85283 Collapse << |
| NCT01070160 | - | - | Completed | - | United States, Connecticut ... more >> Yale-New Haven Hospital Women's Center New Haven, Connecticut, United States, 06520-8063 Collapse << |
| NCT00969566 | Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, 463-707 Collapse << |
| NCT02099838 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Hubei ... more >> Tongji Hospital Wuhan, Hubei, China, 430030 Collapse << |
| NCT02099838 | - | - | Completed | - | - |
| NCT01216397 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.6.1 Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT01300637 | Obesity Metab... more >>olic Syndrome Schizophrenia Collapse << | Not Applicable | Unknown | July 2011 | Taiwan ... more >> Taipei Medical University-WanFang Hospital Recruiting Taipei, Taiwan, 116 Contact: Chun-Hsin Chen, MD 886-2-29307930 ext 53961 chunhsin57@yahoo.com.tw Contact: Mong-Liang Lu, MD 886-2-29307930 ext 53961 mongliang@hotmail.com Collapse << |
| NCT03490019 | Bardet-Biedl Syndrome ... more >> Visual Impairment Collapse << | Phase 2 | Not yet recruiting | August 2020 | - |
| NCT01216397 | - | - | Completed | - | - |
| NCT02275845 | Gestational Diabetes Mellitus | Phase 3 | Unknown | September 2017 | Netherlands ... more >> Maasstad Ziekenhuis Recruiting Rotterdam, Netherlands Contact: Huguette Brink, Drs +31 10 291 2889 brinkH@maasstadziekenhuis.nl Contact: Wetenschapsbureau +31 10 291 3357 wetenschapsbureau@maasstadziekenhuis.nl Collapse << |
| NCT02226510 | Left Ventricular Hypertrophy ... more >> Insulin Resistance Coronary Artery Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital & Medical School Dundee, Angus, United Kingdom, DD1 9SY Collapse << |
| NCT00159575 | Polycystic Ovary Syndrome | Phase 4 | Terminated(Slow patient recrui... more >>tment and expiry of study medication(aug2009)) Collapse << | - | Norway ... more >> Sigrun Kjøtrød Trondheim, Norway, 7006 Fertility UNit- -St Olavs Hospital Trondheim, Norway, 7046 Collapse << |
| NCT03629340 | Pulmonary Hypertension ... more >> Heart Failure Collapse << | Phase 2 | Not yet recruiting | December 2021 | - |
| NCT02278965 | Stage 0 Breast Carcinoma ... more >> Breast Neoplasms Stage I Breast Carcinoma Stage II Breast Carcinoma Stage III Breast Carcinoma Collapse << | Phase 1 | Active, not recruiting | October 2018 | United States, New York ... more >> Columbia University New York, New York, United States, 10032 Collapse << |
| NCT03270553 | Healthy Volunteers | Phase 1 | Completed | - | United States, Nebraska ... more >> Clinical Site Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT00965991 | Gestational Diabetes | Not Applicable | Completed | - | United States, New Mexico ... more >> University of New Mexico Diabetes in Pregnancy Clinic Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT01697566 | Gynecology | Phase 3 | Active, not recruiting | May 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01666730 | Acinar Cell Adenocarcinoma of ... more >>the Pancreas Duct Cell Adenocarcinoma of the Pancreas Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Active, not recruiting | November 2018 | United States, Ohio ... more >> University Hospitals Cleveland Medical Center, Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT03029390 | Prediabetes I... more >>mpaired Fasting Glucose Impaired Glucose Tolerance Collapse << | Phase 4 | Active, not recruiting | May 2018 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00679679 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Argentina ... more >> Hospital Privado de Córdoba Cordoba, Argentina, X5016KEH Collapse << |
| NCT01565096 | Diabetes Mellitus Type II | Phase 4 | Unknown | - | Germany ... more >> ikfe GmbH Recruiting Mainz, Germany, 55116 Contact: Thomas Forst, Prof., MD +49 6131 576 36 16 thomasf@ikfe.de Contact: Swantje Anders +49 6131 327 90 22 s.anders@ikfe-cro.de Collapse << |
| NCT01561976 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT02217670 | Comparative Bioavailability | Phase 1 | Withdrawn(Commercial reasons) | - | Jordan ... more >> International Pharmaceutical Research Center Amman, Jordan Collapse << |
| NCT02151461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 United States, Florida Palm Beach Research Palm Beach, Florida, United States, 33409 Meridien Research Tampa, Florida, United States, 33606 United States, Georgia River Birch Research Alliance Blue Ridge, Georgia, United States, 30513 Meridian Research Savannah, Georgia, United States, 31406 United States, Ohio Streling Research Group Cincinnati, Ohio, United States, 45219 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Meharry Medical College Nashville, Tennessee, United States, 37208 Vanderbilt University Medical Center, Diabetes, Endocrinology, and Metabolism Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02018731 | Becker's Muscular Dystrophy (B... more >>MD) Collapse << | Phase 2 | Completed | - | Switzerland ... more >> University Children's Hospital Basel, BS, Switzerland, 4031 Collapse << |
| NCT01349387 | Type 2 Diabetes | Not Applicable | Completed | - | France ... more >> Centre Hospitalier Sud Francilien Corbeil Essonnes, France, 91106 Collapse << |
| NCT02151461 | - | - | Completed | - | - |
| NCT01561976 | - | - | Completed | - | - |
| NCT01911247 | Endometrial Cancer | Early Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT03690778 | Diabetes Hype... more >>rlipidemias Collapse << | Phase 1 | Recruiting | March 2019 | Korea, Republic of ... more >> Inha University Hospital Recruiting Incheon, Korea, Republic of Contact: Sang-Heon Cho, MD., Ph D Collapse << |
| NCT02755844 | Recurrent Endometrial Cancer | Phase 1 Phase 2 | Not yet recruiting | November 2019 | France ... more >> Centre Hospitalier Lyon Sud Not yet recruiting Pierre Benite, France, 69495 Contact: Benoit YOU Contact: Marine DUPUIS Principal Investigator: Benoit YOU, MD Collapse << |
| NCT02437812 | Epithelial Ovarian Carcinoma | Phase 2 | Recruiting | April 2021 | United States, California ... more >> Gynecologic Oncology Associates Recruiting Newport Beach, California, United States, 92663 Contact: Katrina Lopez, CCRC 949-642-5165 ext 259 Katrinal@gynoncology.com Collapse << |
| NCT02982798 | Diabetes Hype... more >>rlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inha University Hospital Incheon, Korea, Republic of Collapse << |
| NCT02102932 | Healthy | Phase 1 | Completed | - | China ... more >> 1276.23.86002 Boehringer Ingelheim Investigational Site Beijing, China Collapse << |
| NCT03151005 | Polycystic Ovary Syndrome (PCO... more >>S) Overweight Obesity Collapse << | Phase 4 | Recruiting | December 31, 2018 | China, Chongqing ... more >> The Second Affiliated Hospital, Third Military Medical University Recruiting Chongqing, Chongqing, China, 400037 Principal Investigator: Min Long, MD Collapse << |
| NCT02102932 | - | - | Completed | - | - |
| NCT02167620 | Diabetes Mellitus, Type 2 ... more >> Schizophrenia Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Center for Addiction and Mental Health Toronto, Ontario, Canada Collapse << |
| NCT02647827 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Hyperandrogenism Collapse << | Phase 2 | Recruiting | December 2019 | China ... more >> Peking University Recruiting Beijing, China, 100871 Contact: Jie Qiao, Professor +8615611908108 jie.qiao@263.net Contact: Haolin Zhang, PhD +8613911533466 zoe@bjmu.edu.cn Sweden Karolinska University Hospital Recruiting Stockholm, Sweden, 17176 Contact: Berit Legerstam, Study nurse +46851773782 berit.legerstam@karolinska.se Contact: Liselotte Blomberg, Study nurse +46851773782 liselott.blomberg@karolinska.se Principal Investigator: Elisabet Stener-Victorin, PhD Sub-Investigator: Angelica Linden-Hirschberg, Professor Sub-Investigator: Sebastian Gidlöf, PhD Collapse << |
| NCT03258723 | Diabetes | Phase 4 | Not yet recruiting | June 2021 | United States, New York ... more >> Flatbush Family Health Center at NYU Langone Not yet recruiting Brooklyn, New York, United States, 11203 Contact: Paula Pollard-Thomas, MD 347-380-0377 Paula.Pollard-Thomas@nyumc.org Contact: La Sharra Bennett 718-630-2197 ext 4040 Internal Medicine Associates at Mt. Sinai Not yet recruiting New York, New York, United States, 10029 Contact: Victoria L Mayer, MD 212-659-9194 victoria.mayer@mountsinai.org Contact: Carol Horowitz, MD 212-659-8551 carol.horowitz@mountsinai.org Barbados University of the West Indies, Cavehill and Barbados Ministry of Health Polyclinics Not yet recruiting Bridgetown, Barbados Contact: Joseph Herbert, MD 2462347715 familymedicinebarbados@gmail.com Contact: Natasha Sobers, MD 2462400313 natasha.sobers@cavehill.uwi.edu Puerto Rico Internal Medicine Clinic at the University of Puerto Rico Hospital Not yet recruiting Carolina, Puerto Rico, 00984 Contact: Elsie Cruz, MD elsie.cruz1@upr.edu Contact: Enid J Garcia-Rivera, MD (787) 701-1121 enid.garcia3@upr.edu Trinidad and Tobago Southwest Regional Health Authority, La Romaine Health Center Not yet recruiting San Fernando, Trinidad and Tobago Contact: Albert Persaud, MD 1-868-225-1802 albertpersaud@swrha.co.tt Contact: Kavita Dharamraj, MD 1-868-730-3418 kavitadharamraj@swrha.co.tt Collapse << |
| NCT00941239 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Pharmacology and toxicology department, UANL Monterrey, Nuevo León, Mexico, 64460 Collapse << |
| NCT03570632 | Diabetes Mellitus, Type 1 ... more >> Pregnancy in Diabetes Collapse << | Phase 4 | Not yet recruiting | December 2020 | United States, Pennsylvania ... more >> Magee Womens Hospital of UPMC Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02375139 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01710540 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01973933 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul, Korea, Republic of Collapse << |
| NCT01762046 | Diabetes Mellitus, Type 2 | Phase 1 | Active, not recruiting | October 2017 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Brigham and Women's Hospital Boston, Massachusetts, United States, 02116 Joslin Diabetes Center Boston, Massachusetts, United States, 02116 Collapse << |
| NCT01728740 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01342614 | Diabetes Mellitus ... more >> Essential Hypertension Collapse << | Not Applicable | Completed | - | - |
| NCT02694289 | Type 2 Diabetes Mellitus | Not Applicable | Withdrawn(failure to enroll pa... more >>rticipants) Collapse << | - | - |
| NCT01842620 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01087983 | Advanced Cancers | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01842620 | - | - | Completed | - | - |
| NCT01866462 | Type 2 Diabetes | Early Phase 1 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT01240785 | Gestational Diabetes | Phase 4 | Completed | - | - |
| NCT03566810 | Healthy | Phase 1 | Recruiting | November 8, 2018 | Germany ... more >> Please Contact the Merck KGaA Communication Center Recruiting Darmstadt, Germany, 64293 Contact +49 6151 72 5200 service@merckgroup.com Collapse << |
| NCT03465345 | Prostate Cancer | Phase 1 | Withdrawn(Study was stopped to... more >> pursue a new treatment plan with a new study.) Collapse << | - | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT00940472 | Diabetes Type 2 | Phase 2 Phase 3 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud, Universidad de Guadalajara Guadalajara, Jal, Mexico, 44340 Collapse << |
| NCT02515773 | Bipolar Disorder | Phase 4 | Recruiting | October 2020 | United States, Kentucky ... more >> The Children's Home of Northern Kentucky Recruiting Covington, Kentucky, United States, 41011 Contact: Joe Reiman 859-292-4122 jrieman@chnk.org United States, New Jersey Jersey Shore Medical Center Not yet recruiting Neptune City, New Jersey, United States, 07753 Contact: Nicholas Remo 732-776-3498 NRemo@meridianhealth.com United States, New York South Oaks Recruiting Amityville, New York, United States, 11701 Contact: Patricia Flaherty, LCSW-R,ACSW, 631-608-5222 pflaherty@northwell.edu SUNY Downstate/ Kings County Hospital Not yet recruiting Brooklyn, New York, United States, 11203 Contact: Cathryn A. Galanter, M.D. 718-245-2502 cathryn.galanter@downstate.edu Maimonides Not yet recruiting Brooklyn, New York, United States, 11219 Contact: George Alvarado, M.D. 718-283-8215 GAlvarado@maimonidesmed.org NYCCC Not yet recruiting Brooklyn, New York, United States, 11233 Contact: Eva Sheridan 718-470-4391 eschenk@northwell.edu Northwell Zucker Long Island Jewish Hospital Recruiting Glen Oaks, New York, United States, 11004 Contact: Arielle Carmel 718-470-8021 acarmel@northwell.edu Mount Sinai Recruiting New York, New York, United States, 10029 Contact: Julia G Jones 212-241-3692 julia.george-jones@mssm.edu LIJ Zucker Hillside Hospital Recruiting New York, New York, United States, 11004 Contact: Eva Shenck 718-470-4391 eschenk@northwell.edu Child Center of New York, Recruiting Queens, New York, United States, 11373 Contact: Isaiah Mui 718-358-8288 ext 221 isaiahmui@childcenterny.org Contact: Alla Weinstein 718.358.8288 allaweinstein@childcenterny.org NorthShore Child and Family Guidance Recruiting Roslyn Heights, New York, United States, 11577 Contact: Esther Gonzalez 516-626-1971 EGonzalez@northshorechildguidance.org StonyBrook Recruiting Stony Brook, New York, United States, 11794 Contact: Gabrielle A Carlson, MD 631-632-8840 Gabrielle.Carlson@stonybrookmedicine.edu United States, Ohio Lighthouse Youth Services Recruiting Cincinnati, Ohio, United States, 45206 Contact: Jody Harding 513-487-6778 jarding@lys.org Contact: Shannon Kiniyalocts 513-487-7706 skiniyalocts@lys.org Central Clinic Recruiting Cincinnati, Ohio, United States, 45219 Contact: Maria Piombo, Ed.D.,LPCC-S 513-558-5890 piombom@ucmail.uc.edu Children's Home Recruiting Cincinnati, Ohio, United States, 45227 Contact: Jessica Aritonovich 513-272-2800 ext 4352 jaritonovich@thechildrenshomecinti.org Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Elizabeth Picard, MS 513-803-8898 elizabeth.picard@cchmc.org Contact: Shelley Randall 513-803-2342 Shelley.randall@cchmc.org Resident Mood Medication Clinic Recruiting Cincinnati, Ohio, United States, 45229 Contact: Christy Klein, MPH 513-558-5746 kleinci@ucmail.uc.edu St. Aloysius Recruiting Cincinnati, Ohio, United States, 45237 Contact: Morrisa Gray, RN, BSN 513-482-7257 mgray@stalsorphanage.org Talbert House Recruiting Cincinnati, Ohio, United States, 45238 Contact: Sharon Stanford, MD 513-265-6090 sharon.stanford@talberthouse.com St. Joseph's Orphanage Recruiting Cincinnati, Ohio, United States, 45239 Contact: Tyann Godwin, RN 513-741-3100 ext 2166 tyann.godwin@sjokids.org Child Focus Recruiting Cincinnati, Ohio, United States, 45244 Contact: Jessica Bowlin JBowlin@child-focus.org NECCO Recruiting Cincinnati, Ohio, United States, 45246 Contact: Jessica Parks, MSM, LISW-S 513-771-9600 jparks@necco.org University Hospital Medical Center Cleveland Recruiting Cleveland, Ohio, United States, 44106 Contact: Elizabeth Deyling Elizabeth.Deyling@UHhospitals.org Nationwide Children's Hospital Columbus Recruiting Columbus, Ohio, United States, 43205 Contact: Raymond Troy raymond.troy@nationwidechildrens.org Ohio State University Recruiting Columbus, Ohio, United States, 43210 Contact: Ghada Lteif, MD 614-293-8237 ghada.lteif@osumc.edu South Community Recruiting Dayton, Ohio, United States Contact: Mary Day, RN 937-245-6121 mday@southcommunity.com Butler Behavioral Health Services Recruiting Hamilton, Ohio, United States, 45011 Contact: Randy Allman, LISW-S 513-881-7189 ext 3134 Rallman@bbhs.org TCN Family Solutions Recruiting Xenia, Ohio, United States, 45385 Contact: Carmel Flores 937-427-3837 cflores@tcn.org United States, Texas Seton Family of Hospitals Not yet recruiting Austin, Texas, United States, 78712 Contact: Ashleigh Smith AJSmith2@seton.org Collapse << |
| NCT01089192 | Type 2 Diabetes | Phase 1 | Completed | - | India ... more >> Torrent Research Centre Bhat, Gujarat, India, 382 428 Collapse << |
| NCT01240785 | - | - | Completed | - | - |
| NCT02037672 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT03693560 | Coronary Artery Disease ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Active, not recruiting | February 2019 | Egypt ... more >> Faculty of Pharmacy - Damanhour University. Damanhūr, Egypt Collapse << |
| NCT01089205 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02220218 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT02526615 | Insulin Sensitivity ... more >> Type 2 Diabetes Collapse << | Phase 4 | Completed | - | Finland ... more >> Turku PET Centre Turku, Finland, 20520 Turku university hospital, PET center Turku, Finland, 20520 Turku PET Centre (Turku University Hospital) Turku, Finland, 20521 Collapse << |
| NCT02960711 | Metabolic Syndrome | Phase 3 | Unknown | July 2018 | Italy ... more >> Fondazione IRCCS Istituto Nazionale Tumori Recruiting Milan, Italy, 20133 Contact: Franco Berrino, MD +390223903515 Franco.berrino@istitutotumori.mi.it Contact: Patrizia Pasanisi, MD +390223903513 trialcenter@istitutotumori.mi.it Collapse << |
| NCT01747083 | Type 2 Diabetes | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT00859898 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00570739 | Type 2 Diabetes Mellitus ... more >> Hypercholesterolemia Pre-diabetes Collapse << | Phase 3 | Completed | - | - |
| NCT00859898 | - | - | Completed | - | - |
| NCT00286442 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00899470 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Mds Pharma Services Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT00899470 | - | - | Completed | - | - |
| NCT00570739 | - | - | Completed | - | - |
| NCT01302145 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Hungary ... more >> Balatonfured, Hungary, 8230 Budapest, Hungary, 1083 Netherlands Groningen, Netherlands, 9470 AE Poland Warsaw, Poland, 02-097 Slovakia Bratislava, Slovakia, 83305 Collapse << |
| NCT02028689 | Healthy | Phase 1 | Completed | - | United States, Michigan ... more >> Kalamazoo, Michigan, United States, 49007 Collapse << |
| NCT01206647 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | February 2011 | Germany ... more >> Diabetologische Schwerpunktpraxis Dr. Lorra / Dr. Bonnermann Bochum, Germany, 44869 Zentrum für klinische Studien Dresden, GWT-TUD GmbH Dresden, Germany, 01307 Gemeinschaftspraxis Partner der Gesundheit Essen, Germany, 45355 IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Zentrum für Diabetes und Gefäßerkrankungen Münster, Germany, 48145 Diabetes Zentrum Neuwied Neuwied, Germany, 56564 ikfe Studiencenter Potsdam GmbH Potsdam, Germany, 14469 Collapse << |
| NCT02941445 | Polycystic Ovary Syndrome ... more >> Body Weight Collapse << | Phase 4 | Completed | - | - |
| NCT00915772 | - | - | Completed | - | - |
| NCT03349684 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | September 11, 2019 | - |
| NCT01341886 | Malignant Neoplasm of Thyroid ... more >>Stage I Malignant Neoplasm of Thyroid Stage II Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Mashad University of Medical Sciences Mashad, Khorasan Razavi, Iran, Islamic Republic of Collapse << |
| NCT02508168 | Healthy Volunteers | Phase 1 | Completed | - | - |
| NCT01881828 | Type 1 Diabetes | Phase 3 | Unknown | December 2014 | United States, Florida ... more >> Jaeb Center for Health Research Tampa, Florida, United States, 33647 Collapse << |
| NCT00915772 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01877564 | Adenocarcinoma of the Endometr... more >>ium Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT00286442 | - | - | Completed | - | - |
| NCT00391261 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Bipolar Disorder Autism Spectrum Disorders Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> Cambridge Health Alliance Medford, Massachusetts, United States, 02155 Collapse << |
| NCT03071705 | Non-Small Cell Adenocarcinoma ... more >> Tyrosine Kinase Mutation EGFR Gene Mutation Collapse << | Not Applicable | Recruiting | December 2017 | Mexico ... more >> Instituto National de Cancerologia Recruiting Mexico, Mexico, 14080 Contact: Oscar Gerardo MD Arrieta, Oncology 015556280400 ext 71101 ogar@servidor.unam.mx Collapse << |
| NCT01877564 | - | - | Completed | - | - |
| NCT03710460 | Obesity | Phase 4 | Recruiting | June 30, 2019 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: Manuel Gonzalez, PhD +52-10-58-52-00 ext 34212 uiec@prodigy.net.mx Contact: Esperanza Martinez, PhD +52-10-58-52-00 ext 34212 esperanzamartnezabundi@yahoo.com Collapse << |
| NCT01780051 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Korea, Republic of ... more >> Chounbuk National University Hospital Jeonju, Jeonbuk, Korea, Republic of Collapse << |
| NCT02851095 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT00466622 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Norway ... more >> Dept. of Obstetrics and Gynecology, National Center for fetal Medicine, University Hospital of Trondheim Trondheim, Norway, 7006 Collapse << |
| NCT01438814 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02865668 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT03475524 | Melasma | Phase 4 | Not yet recruiting | August 2019 | Egypt ... more >> Assuit University Not yet recruiting Assiut, Egypt, 71111 Contact: Radwa Bakr 01119988115 Contact: sahar Ismail saharsotohy@yahoo.com Collapse << |
| NCT00778791 | Healthy | Not Applicable | Completed | - | United States, Missouri ... more >> Gateway medical research St. Charles, Missouri, United States, 63301 Collapse << |
| NCT01438814 | - | - | Completed | - | - |
| NCT00473876 | Congestive Heart Failure ... more >> Insulin Resistance Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Medicine and Therapeutics, Ninewells Hospital Dundee, Scotland, United Kingdom, DD1 9SY Collapse << |
| NCT00682448 | Metabolic Complications | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT00648869 | Healthy | Not Applicable | Withdrawn(Drug was not sent) | November 2008 | Israel ... more >> Diabetes Unit, Hadassah Hebew University Hospital Jerusalem, Israel, 91120 Collapse << |
| NCT00546728 | Impaired Glucose Tolerance | Phase 4 | Completed | - | United States, Minnesota ... more >> International Diabetes Center at Park Nicollet St. Louis Park, Minnesota, United States, 55416 St. Paul Heart Clinic St. Paul, Minnesota, United States, 55102 Collapse << |
| NCT02819869 | Hepatocellular Carcinoma | Phase 2 | Terminated(insufficient for th... more >>e fund) Collapse << | - | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT02143050 | Melanoma | Phase 1 Phase 2 | Recruiting | June 2020 | United States, Kentucky ... more >> James Graham Brown Cancer Center-Universityof Louisville Recruiting Louisville, Kentucky, United States, 40202 Contact: Jason A Chesney, MD, PhD 502-562-3429 jason.chesney@louisville.edu Contact: Christina LaDuke 502-217-5205 christy.laduke@louisville.edu Sub-Investigator: Donald M Miller, MD, PhD Sub-Investigator: Kelly M McMasters, MD, PhD Sub-Investigator: Sucheta Telang, MD James Graham Brown Cancer Center Recruiting Louisville, Kentucky, United States, 40202 Contact: Karen Carter, BSN 502-562-3690 kmcart05@louisville.edu Contact: Christina LaDuke, BS 502-217-5205 christi.laduke@louisville.edu Collapse << |
| NCT02640534 | Cancer of the Prostate ... more >> Prostate Cancer Collapse << | Phase 2 | Recruiting | June 2026 | Switzerland ... more >> Kantonsspital Aarau Recruiting Aarau, Switzerland, 5001 Contact: Tobias Wehrhahn, Dr. med. +41 62 838 60 50 tobias.wehrhahn@ksa.ch Principal Investigator: Tobias Wehrhahn, Dr. med. Universitaetsspital Basel Recruiting Basel, Switzerland, 4031 Contact: Sacha Rothschild, MD +41 61 265 25 25 sacha.rothschild@usb.ch Principal Investigator: Sacha Rothschild, MD Istituto Oncologico della Svizzera Italiana (IOSI) Recruiting Bellinzona, Switzerland, 6500 Contact: Ricardo Pereira Mestre, MD +41 91 811 84 46 ricardo.pereiramestre@eoc.ch Principal Investigator: Ricardo Pereira Mestre, MD Kantonsspital Graubuenden Recruiting Chur, Switzerland, 7000 Contact: Richard Cathomas, MD +41 81 256 66 95 richard.cathomas@ksgr.ch Principal Investigator: Richard Cathomas, MD Spital Thurgau AG Recruiting Frauenfeld, Switzerland, CH-8500 Contact: Regina Woelkly, MD +41 52 723 76 91 regina.woelkly@stgag.ch Principal Investigator: Regina Woelkly, MD Hôpitaux Universitaires de Genève Recruiting Genève 14, Switzerland, 1211 Contact: Anna Patrikidou, MD +41 79 553 60 99 anna.patrikidou@hcuge.ch Principal Investigator: Anna Patrikidou, MD CCAC Lausanne Recruiting Lausanne, Switzerland, 1004 Contact: Pierre Bohanes, MD +41 (21) 646 79 94 pbohanes@bluewin.ch Principal Investigator: Pierre Bohanes, MD Luzerner Kantonsspital Recruiting Luzern 16, Switzerland, 6000 Contact: Beat Müller, MD +41 41 205 58 60 beat.mueller@luks.ch Principal Investigator: Beat Müller, MD Hôpital du Valais Recruiting Martigny, Switzerland, 1920 Contact: Sandro Anchisi, MD +41 27 603 90 00 sandro.anchisi@hopitalvs.ch Principal Investigator: Sandro Anchisi, MD Kantonsspital Olten Recruiting Olten, Switzerland, CH-4600 Contact: Walter Mingrone, MD 41-62-311-4241 wmingrone_ol@spital.ktso.ch Principal Investigator: Walter Mingrone, MD Hôpital du Valais Recruiting Sion, Switzerland, 1951 Contact: Sandro Anchisi, MD +41 27 603 45 00 sandro.anchisi@hopitalvs.ch Principal Investigator: Sandro Anchisi, MD Bürgerspital Solothurn Recruiting Solothurn, Switzerland, CH-4500 Contact: Philippe von Burg, MD +41 32 627 47 00 philippe.vonburg@spital.so.ch Principal Investigator: Philippe von Burg, MD Kantonsspital St. Gallen Recruiting St. Gallen, Switzerland, 9007 Contact: Christian Rothermundt, MD +41 71 494 11 63 christian.rothermundt@kssg.ch Principal Investigator: Christian Rothermundt, MD Kantonsspital Winterthur Recruiting Winterthur, Switzerland, 8401 Contact: Natalie Fischer, MD +41 052 266 40 87 natalie.fischer@ksw.ch Principal Investigator: Natalie Fischer, MD Stadtspital Triemli Recruiting Zürich, Switzerland, 8063 Contact: Aleksandra Marek, Dr. med. +41 (44) 466 11 11 aleksandra.marek@triemli.zuerich.ch Principal Investigator: Aleksandra Marek, Dr. med. UniversitätsSpital Zürich Recruiting Zürich, Switzerland, 8091 Contact: Thomas Hermanns, MD +41 44 255 11 11 thomas.hermanns@usz.ch Principal Investigator: Thomas Hermanns, MD Collapse << |
| NCT00473876 | - | - | Completed | - | - |
| NCT00546728 | - | - | Completed | - | - |
| NCT02580331 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT02125227 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of, 561712 Collapse << |
| NCT02945813 | Prostate Cancer | Phase 2 | Recruiting | December 2030 | France ... more >> Centre Hospitalier Régional Universitaire (CHRU) Jean Minjoz Recruiting Besançon, France, 25030 Contact: Bernardino De Bari, MD +33 381 23 24 28 bdebari@chu-besancon.fr Principal Investigator: Bernardino De Bari Clinique Pasteur - Centre finistérien de radiothérapie et d'oncologie Recruiting Brest, France, 29220 Contact: Ali Hasbini, MD +33 298 31 32 00 alihasbini@oncologie-brest.fr Principal Investigator: Ali Hasbini, MD Centre de lutte contre le cancer Léon Bérard Recruiting Lyon, France, 69008 Contact: Pascal Pommer, MD +33 478 78 51 66 pascal.pommier@lyon.unicancer.fr Principal Investigator: Pascal Pommer, MD Hôpital Saint-Louis Recruiting Paris, France, 75010 Contact: Christophe Hennequin, Prof +33 142 49 90 24 christophe.hennequin2@aphp.fr Principal Investigator: Christophe Hennequin, Prof CHU de Poitiers - La Miletrie Recruiting Poitiers Cedex, France, 86021 Contact: Stéphane Guerif, MD +33 549 44 29 61 stephane.guerif@chu-poitiers.fr Principal Investigator: Stéphane Guerif, MD Institut de Cancérologie de L'Ouest René Gauducheau Recruiting Saint Herblain cedex, France, 44800 Contact: Stéphane Supiot, MD +33 240 679 913 stephane.supiot@ico.unicancer.fr Principal Investigator: Stéphane Supiot, MD Institut de Cancérologie de la Loire Lucien Neuwirth Recruiting Saint Priest en Jarez, France, 42270 Contact: Nicolas Magne, Prof +33 477 91 71 04 nicolas.magne@icloire.fr Principal Investigator: Nicolas Magne, Prof Clinique Pasteur - Oncorad Recruiting Toulouse Cedex 3, France, 31076 Contact: Igor Latorzeff, MD +33 567 20 44 00 ilatorzeff@clinique-pasteur.com Principal Investigator: Igor Latorzeff, MD Germany Universitätsmedizin Berlin Recruiting Berlin, Germany, 13353 Contact: Pirus Ghadjar, MD +49 450 657055 pirus.ghadjar@charite.de Principal Investigator: Pirus Ghadjar, MD Klinikum der Universität München Not yet recruiting München, Germany, 81377 Contact: Claus Belka, Prof +49 89 4400 74520 claus.belka@med.uni-muenchen.de Principal Investigator: Claus Belka, Prof Universitätsklinikum Rostock Recruiting Rostock, Germany, 18059 Contact: Guido Hildebrandt, Prof +49 381 494 90 00 guido.hildebrandt@uni-rostock.de Principal Investigator: Guido Hildebrandt, Prof Universitätsklinikum Würzburg Recruiting Würzburg, Germany, 97080 Contact: Michael Flentje, Prof +49 931 201 288 91 flentje_m@ukw.de Principal Investigator: Michael Flentje, Prof Switzerland Universitätsspital Basel Recruiting Basel, Switzerland, 4031 Contact: Alexandros Papachristofilou, MD +41 61 265 49 46 alexandros.papachristofilou@usb.ch Principal Investigator: Alexandros Papachristofilou, MD Istituto Oncologico della Svizzera Italiana IOSI - Ospedale San Giovanni Recruiting Bellinzona, Switzerland, 6500 Contact: Che Ngwa Azinwi, MD +41 091 811 89 32 ngwache.azinwi@eoc.ch Principal Investigator: Che Ngwa Azinwi, MD Inselspital Bern Recruiting Bern, Switzerland, 3010 Contact: Daniel Aebersold, Prof +41 31 632 26 32 daniel.aebersold@insel.ch Principal Investigator: Daniel Aebersold, Prof Kantonsspital Graubuenden Recruiting Chur, Switzerland, 7000 Contact: Daniel R. Zwahlen, MD +41 81 256 64 95 daniel.zwahlen@ksgr.ch Principal Investigator: Daniel R. Zwahlen, MD HFR - Hôpital cantonal Recruiting Fribourg, Switzerland, 1708 Contact: David Lauffer, MD +41 26 306 00 00 David.Lauffer@h-fr.ch Principal Investigator: David Lauffer, MD Hôpitaux Universitaires Genève HUG Recruiting Geneva, Switzerland, 1211 Contact: Thomas Zilli, MD +41 22 372 70 90 thomas.zilli@hcuge.ch Principal Investigator: Thomas Zilli, MD Clinique de Genolier Recruiting Genolier, Switzerland, 1272 Contact: Cristina Picardi, MD +41 22 366 94 59 cpicardi@genolier.net Principal Investigator: Cristina Picardi, MD Spital Thurgau Recruiting Münsterlingen, Switzerland, CH-8596 Contact: Christiane Reuter, MD +41 71 686 23 33 christiane.reuter@stgag.ch Principal Investigator: Christiane Reuter, MD Hopital de Sion Recruiting Sion, Switzerland, 1951 Contact: Kaouthar Khanfir, MD +41 27 603 87 72 kaouthar.khanfir@hopitalvs.ch Principal Investigator: Kaouthar Khanfir, MD Kantonsspital St. Gallen Recruiting St. Gallen, Switzerland, 9007 Contact: Paul Martin Putora, MD +41 71 494 22 68 paulmartin.putora@kssg.ch Principal Investigator: Paul Martin Putora, MD Kantonsspital Winterthur KSW Recruiting Winterthur, Switzerland, 8401 Contact: Binaya Kumar Shrestha, MD +41 52 266 55 05 binayakumar.shrestha@ksw.ch Principal Investigator: Binaya Kumar Shrestha, MD Klinik Hirslanden Recruiting Zurich, Switzerland, 8032 Contact: Marcin Sumila, MD +41 44 387 25 51 marcin.sumila@hirslanden.ch Principal Investigator: Marcin Sumila, MD Stadtspital Triemli Recruiting Zurich, Switzerland, 8063 Contact: Helmut Kranzbühler, MD +41 44 466 13 67 helmut.kranzbuehler@triemli.zuerich.ch Principal Investigator: Helmut Kranzbühler, MD UniversitätsSpital Zürich Recruiting Zurich, Switzerland, 8091 Contact: Matthias Guckenberger, Prof +41 44 255 29 30 Matthias.Guckenberger@usz.ch Principal Investigator: Matthias Guckenberger, Prof Collapse << |
| NCT02506790 | Breast Cancer | Phase 2 | Recruiting | August 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting Saint - Petersburg, Russian Federation, 191124 Contact: Tatiana Y Semiglazova, MD, PhD, DSc +79219468072 tsemiglazova@mail.ru Sub-Investigator: Petr V Krivorotko, MD, PhD, DSc Principal Investigator: Tatiana Y Semiglazova, MD, PhD, DSc Collapse << |
| NCT00809536 | Generalized Anxiety Disorder | Phase 1 | Terminated(Please Detailed Des... more >>cription for termination reason.) Collapse << | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT00659568 | Breast Cancer ... more >> Endometrial Cancer Kidney Cancer Lung Cancer Lymphoma Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> London Regional Cancer Program at London Health Sciences Centre London, Ontario, Canada, N6A 4L6 Collapse << |
| NCT02722239 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Russian Federation ... more >> Research Site Moscow, Russian Federation, 123423 Collapse << |
| NCT02722239 | - | - | Completed | - | - |
| NCT01521624 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT01915550 | Diabetes Mellitus | Phase 3 | Completed | - | Egypt ... more >> Ain Shams University Maternity Hospital Cairo, Egypt, 19931 Collapse << |
| NCT00778141 | Healthy | Not Applicable | Completed | - | United States, Missouri ... more >> Gateway medical research St. Charles, Missouri, United States, 63301 Collapse << |
| NCT02104804 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Changchun, China Research Site Fuzhou, China Research Site Guangzhou, China Research Site Ha'er bing, China Research Site Hefei, China Research Site Nanchang, China Research Site Nanjing, China Research Site Shanghai, China Research Site ShiJiazhuang, China Research Site Shiyan, China Collapse << |
| NCT01999686 | Polycystic Ovary Syndrome (PCO... more >>S) Insulin Resistance Collapse << | Phase 3 | Recruiting | October 2018 | Indonesia ... more >> Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Lambung Mangkurat, Ulin Banjarmasin Hospital Recruiting Banjarmasin, Indonesia Contact: Iwan D Putra, dr., SpOG-K +62811503472 i_darmaputra@yahoo.com Principal Investigator: Iwan D Putra, dr., SpOG-K Sub-Investigator: Hardyan Sauqi, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Udayana, Sanglah Hospital Recruiting Denpasar, Indonesia Contact: Putu Doster Mahayasa, dr., SpOG-K +628123812259 dmahayasa@gmail.com Principal Investigator: Putu Doster Mahayasa, dr., SpOG-K Sub-Investigator: Anom Suardika, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Hasanuddin, Dr. Wahidin Sudirohusodo Hospital Recruiting Makasar, Indonesia Contact: Nusratuddin Abdullah, Dr., dr., SpOG-K, MARS +6281342752561 nusraya@yahoo.com Principal Investigator: Nusratuddin Abdullah, Dr., dr., SpOG-K, MARS Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Brawijaya, Dr. Saiful Anwar Hospital Recruiting Malang, Indonesia Contact: Arsana Wiyasa IW, Dr., dr., SpOG-K +62811343876 abiyasa9@yahoo.com Principal Investigator: Arsana Wiyasa IW, Dr., dr., SpOG-K Sub-Investigator: Sutrisno Sutrisno, dr., SpOG-K Sub-Investigator: Widjajanto Ngartjono, dr., SpOG-K Sub-Investigator: Pande M Dwijayasa, dr., SpOG-K Sub-Investigator: Hermawan Wibisono, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Sam Ratulangi, Prof. Dr. Kandou Hospital Recruiting Manado, Indonesia Contact: Eddy Suparman, Prof., Dr., dr., SpOG-K +62811431600 obsgyn_manado@yahoo.com.sg Principal Investigator: Eddy Suparman, Prof., Dr., dr., SpOG-K Sub-Investigator: Freddy W Wagey, Dr., dr., SpOG-K Sub-Investigator: Maria F Loho, dr., SpOG-K Sub-Investigator: Juneke J Kaeng, dr., SpOG-K Sub-Investigator: Maya E Mewengkang, dr., SpOG Sub-Investigator: Linda M Mamengko, dr., SpOG-K Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, Diponegoro University, Dr. Kariadi Hospital Recruiting Semarang, Indonesia Contact: Syarief Taufik, dr., SpOG-K +628112715580 tofik_obg@yahoo.com Principal Investigator: Syarief Taufik, dr., SpOG-K Sub-Investigator: Inu Mulyantoro, dr., SpOG-K Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Airlangga, Dr. Soetomo Hospital. Recruiting Surabaya, Indonesia Contact: Soehartono Ds, Prof., dr., SpOG-K +62811347720 batabsby@yahoo.com Principal Investigator: Soehartono Ds, Prof., dr., SpOG-K Sub-Investigator: Budi Santoso, Prof., Dr., dr., SpOG-K Sub-Investigator: Relly Y Primariawan, SpOG-K Collapse << |
| NCT02383563 | HIV Coronary ... more >>Artery Disease Collapse << | Phase 2 | Unknown | February 2016 | United States, Hawaii ... more >> Hawaii Center for AIDS Recruiting Honolulu, Hawaii, United States, 96813 Contact: Cecilia Shikuma, MD 808-692-1328 shikuma@hawaii.edu Contact: Debra Ogata-Arakaki, RN 808 692-1332 ogataara@hawaii.edu Collapse << |
| NCT01478308 | Prostate Cancer | Phase 2 | Withdrawn(No accrual in initia... more >>l period, PI decided to close study.) Collapse << | - | - |
| NCT02984722 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Italy ... more >> Catholic university of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT02984475 | Beta Thalassemia Major Anemia | Phase 4 | Recruiting | April 2019 | Egypt ... more >> Abo El Reesh Hospital Recruiting Cairo., Egypt Contact: Mona Gaber, Bachelor. 0223648368 mona.sobhy@pharma.cu.edu.eg El Demerdash (Ain Shams Teaching Hospital). Completed Cairo., Egypt Collapse << |
| NCT01210911 | Locally Advanced Pancreatic Ca... more >>ncer Metastatic Pancreatic Cancer Collapse << | Phase 2 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, Netherlands, 1105AZ Collapse << |
| NCT01329016 | Gestational Diabetes Mellitus | Phase 1 Phase 2 | Unknown | June 2015 | United States, Indiana ... more >> Indiana University School of Medicine Indianapolis, Indiana, United States, 46202 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT02104804 | - | - | Completed | - | - |
| NCT01046032 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | United Kingdom ... more >> IVF Hammersmith London, United Kingdom Nuture IVF Unit Nottingham, United Kingdom Oxford Fertility Unit Oxford, United Kingdom, OX39DU Collapse << |
| NCT01710527 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01367054 | Nutritional and Metabolic Dise... more >>ases Collapse << | Not Applicable | Unknown | December 2011 | Brazil ... more >> Biocinese Toledo, Pr, Brazil, 85903-590 Collapse << |
| NCT01710527 | - | - | Completed | - | - |
| NCT01733836 | Prostate Cancer | Phase 2 | Withdrawn(The reason for this ... more >>decision is that this study is being merged with a larger multi-centre study using the same agent in the same population.) Collapse << | October 2014 | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01525823 | Healthy Volunteers | Phase 1 | Completed | - | Australia, Victoria ... more >> Local Institution Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT02040246 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Shanghai ... more >> Renji Hospital Shanghai, Shanghai, China, 200127 China Renji Hospital Shanghai, China, 200127 Collapse << |
| NCT03329651 | Impaired Glucose Tolerance ... more >> Diabetes Mellitus, Type 2 Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT01593371 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT02039245 | Healthy Volunteers | Phase 1 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States Collapse << |
| NCT01857609 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul,, Korea, Republic of Collapse << |
| NCT01292993 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Lexicon Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT02797340 | Heart Failure, Systolic | Phase 4 | Active, not recruiting | April 30, 2018 | Denmark ... more >> Aarhus University Hospital, Department of Cardiology, Palle Juul-Jensens Boulevard 99 Aarhus N, Denmark, 8200 Collapse << |
| NCT00720161 | Spina Bifida ... more >>Neuromuscular Diseases Collapse << | Not Applicable | Completed | - | Belgium ... more >> UZLeuven Leuven, Brabant, Belgium, 3000 Collapse << |
| NCT00960076 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01002807 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT00988078 | Polycystic Ovary Syndrome | Not Applicable | Unknown | December 2013 | Brazil ... more >> Federal University of Sao Paulo Recruiting Sao Paulo, Brazil Contact: Margareth Iwata megiwata@yahoo.com Collapse << |
| NCT01560481 | Healthy | Phase 1 | Completed | - | Spain ... more >> Centre d'Investigació de Medicaments Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT02183571 | Healthy | Phase 1 | Completed | - | - |
| NCT02188654 | Psoriatic Arthritis | Not Applicable | Completed | - | Egypt ... more >> University of Alexandria Alexandria, Egypt, 00203 Collapse << |
| NCT00960076 | - | - | Completed | - | - |
| NCT00661362 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03558867 | Pre Diabetes ... more >>Type 2 Diabetes Mellitus Collapse << | Not Applicable | Recruiting | December 23, 2020 | Australia, New South Wales ... more >> Garvan Institute of Medical Research Recruiting Sydney, New South Wales, Australia, 2010 Contact: Jerry R Greenfield, MD, PhD +61292958217 j.greenfield@garvan.org.au Contact: Dorit Samocha-Bonet, PhD +61292958309 d.samochabonet@garvan.org.au Principal Investigator: Dorit Samocha-Bonet, PhD Principal Investigator: Jerry R Greenfield, MD, PhD Collapse << |
| NCT01357876 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, Cambridgeshire, United Kingdom, CB2 2GG Collapse << |
| NCT00661362 | - | - | Completed | - | - |
| NCT00961480 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01443221 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01961388 | - | - | Withdrawn | June 2017 | - |
| NCT01454622 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT02022007 | Polycystic Ovary Syndrome ... more >> Disorder of Glucose Regulation Collapse << | Phase 3 | Completed | - | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70817 Collapse << |
| NCT01800227 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd., Village Bhat, Gandhinagar, Gujarat, India Collapse << |
| NCT02007278 | - | - | Completed | - | - |
| NCT00897390 | - | - | Completed | - | - |
| NCT01800240 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd., Village Bhat, Gandhinagar, Gujarat, India Collapse << |
| NCT01930864 | Colorectal Neoplasms ... more >> Adenocarcinoma Collapse << | Phase 2 | Recruiting | December 1, 2020 | Brazil ... more >> Barretos Cancer Hospital Recruiting Barretos, SP, Brazil, 14784-400 Contact: Joao Paulo SN Lima, MD 551733216600 ext 6829 joao.lima@accamargo.org.br Contact: Arinilda C Bragagnoli, MD 551733216600 ext 6829 aryscampos@yahoo.com.br Principal Investigator: Arinilda C Bragagnoli, MD State University of Campinas Not yet recruiting Campinas, São Paulo, Brazil, 1300000 Contact: Lígia T Macedo, MD ligaimed@gmail.com Principal Investigator: Ligia T Macedo, MD Collapse << |
| NCT00897390 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Mds Pharma Services Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT02483299 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02022007 | - | - | Completed | - | - |
| NCT01463228 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT00667498 | Pediatric Obesity ... more >> Insulin Resistance Hyperinsulinemia Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale Center for Clinical Investigation (YCCI) New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02007278 | Type 2 Diabetes ... more >> Hypoglycemia Collapse << | Phase 4 | Completed | - | Colombia ... more >> Novartis Investigative Site Medellín, Antioquia, Colombia Novartis Investigative Site Manizales, Caldas, Colombia, 1700 Novartis Investigative Site Bogotá, Cundinamarca, Colombia Novartis Investigative Site Chía, Cundinamarca, Colombia, 11001000 Novartis Investigative Site Cali, Valle del Cauca, Colombia, 760001 Novartis Investigative Site Bogotá, Colombia, 00000 Novartis Investigative Site Cali, Colombia Novartis Investigative Site Monteria, Colombia Collapse << |
| NCT02440893 | - | - | Completed | - | - |
| NCT01778244 | Dyslipidemia | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT01518712 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT03627039 | - | - | Not yet recruiting | August 1, 2020 | - |
| NCT01426399 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT01280409 | Diabetes, Gestational | Phase 4 | Completed | - | United States, Texas ... more >> Memorial Hermann Hospital, Texas Medical Center Houston, Texas, United States, 77030 University of Texas Health Science Center at Houston, Professional Building Houston, Texas, United States, 77030 Collapse << |
| NCT01901224 | Peripheral Artery Disease | Phase 4 | Terminated(study was not funde... more >>d) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01901224 | - | - | Terminated(study was not funde... more >>d) Collapse << | - | - |
| NCT02881528 | Prediabetic State | Phase 4 | Withdrawn(Poor Recruitment pri... more >>or to end of funding timelines.) Collapse << | - | United Kingdom ... more >> NHS Ayrshire & Arran Kilmarnock, Ayrshire, United Kingdom, KA2 0BE NHS Grampian Aberdeen, Grampian, United Kingdom, AB25 2ZG NHS Lanarkshire Wishaw, Lanarkshire, United Kingdom, ML2 0DP NHS Tayside Dundee, Tayside, United Kingdom, DD1 9SY NHS Greater Glasgow & Clyde Glasgow, United Kingdom, G3 8SJ Collapse << |
| NCT01177709 | Schizophrenia ... more >> Obesity Collapse << | Not Applicable | Terminated(PI moved to differe... more >>nt institution. Decided to concentrate on different studies.) Collapse << | - | United States, New York ... more >> Manhatan Psychiatric Center New York, New York, United States, 10035 Collapse << |
| NCT01280409 | - | - | Completed | - | - |
| NCT02312661 | Epithelial Ovarian Cancer | Phase 1 | Completed | - | Netherlands ... more >> University Mecdical Center Groningen Groningen, Netherlands, 9713 GZ Collapse << |
| NCT01177709 | - | - | Terminated(PI moved to differe... more >>nt institution. Decided to concentrate on different studies.) Collapse << | - | - |
| NCT01882907 | Type 2 Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Busan Saint Mary's Medical Center Busan, Korea, Republic of Dong-AUniversity Medical Center Busan, Korea, Republic of Inje University Baik Hospital Busan, Korea, Republic of Kosin University Hospital Busan, Korea, Republic of Pusan National University Hospital Busan, Korea, Republic of Changwon Fatima Hospital Changwon, Korea, Republic of Daegu Catholic University Medical Center Daegu, Korea, Republic of Keimyung University Dongsan Medical Center Daegu, Korea, Republic of Kyungbuk National Universtiy Hospital Daegu, Korea, Republic of Chonnam National University Hospital Gwangju, Korea, Republic of Chosun University Hospital Gwangju, Korea, Republic of Chonbuk National University Hospital Jeonju, Korea, Republic of Gyeongsang National University Hospital Jinju, Korea, Republic of Masan Samsung Medical Center Masan, Korea, Republic of Ulsan University Hospital Ulsan, Korea, Republic of Collapse << |
| NCT01882907 | - | - | Completed | - | - |
| NCT01963663 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Diabetes Clinic, Vali-Asr Hospital, Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT02473094 | Rectal Neoplasms ... more >> Adenocarcinoma Carcinoma Collapse << | Phase 2 | Terminated(Slow Accrual) | - | Brazil ... more >> Clinical Oncology Department - General Hospital - State University of Campinas (UNICAMP) Campinas, São Paulo, Brazil, 13083888 Collapse << |
| NCT03138356 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Harrow, United Kingdom, HA1 3UJ Collapse << |
| NCT03355469 | Metabolic Syndrome | Phase 4 | Recruiting | January 31, 2022 | United States, Virginia ... more >> University of Virginia Recruiting Charlottesville, Virginia, United States, 22904 Contact: Stephanie Miller, M.S.Ed. 434-243-8677 slm4ps@virginia.edu Sub-Investigator: Emily Heiston, M.S.Ed Sub-Investigator: Monique Francois, PhD Principal Investigator: Steven K Malin, PhD Collapse << |
| NCT01871558 | - | - | Completed | - | - |
| NCT00743002 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> Artesia, California, United States Los Angeles, California, United States Santa Ana, California, United States Torrence, California, United States Walnut Creek, California, United States United States, Florida Deland, Florida, United States United States, Illinois Bloomingdale, Illinois, United States United States, Michigan Grand Rapids, Michigan, United States United States, Missouri St. Louis, Missouri, United States United States, Nevada Las Vegas, Nevada, United States United States, New Jersey Clifton, New Jersey, United States United States, North Carolina Asheboro, North Carolina, United States Wilmington, North Carolina, United States United States, Pennsylvania Bensalem, Pennsylvania, United States United States, Tennessee Athens, Tennessee, United States United States, Texas Corpus Christi, Texas, United States Dallas, Texas, United States Houston, Texas, United States Richmond, Texas, United States San Antonio, Texas, United States Collapse << |
| NCT01871558 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 3 | Completed | - | France ... more >> Novartis Investigative Site Antibes, France, 06600 Novartis Investigative Site Auxerre, France, 89000 Novartis Investigative Site Bar le Duc, France, 55012 Novartis Investigative Site Bondy, France, 93143 Novartis Investigative Site Brest, France, 29200 Novartis Investigative Site Caen, France, 14000 Novartis Investigative Site Corbeil Essonnes, France, 91100 Novartis Investigative Site Fleury sur Orne, France, 14123 Novartis Investigative Site Maisons Laffitte, France, 78600 Novartis Investigative Site Menton, France, 06500 Novartis Investigative Site Montpellier, France, 34090 Novartis Investigative Site Paris, France, 75010 Novartis Investigative Site Rennes, France, 35203 Novartis Investigative Site Saint Nazaire, France, 44600 Novartis Investigative Site Sanary Sur Mer, France, 83110 Novartis Investigative Site Strsbourg, France, 67000 Novartis Investigative Site Toulouse, France, 31300 Novartis Investigative Site Valence, France, 26000 Novartis Investigative Site Valenciennes, France, 59300 Collapse << |
| NCT02744131 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | December 2019 | India ... more >> KAR clinic and hospital pvt ltd Recruiting Bhubaneswar, Odisha, India, 751001 Contact: Sujata Kar, M.D +919437824163 suju2463@gmail.com Contact: Suneeta Panigrahi, B.A womenwing@karhospitals.com Collapse << |
| NCT00909506 | Breast Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02846506 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT02851212 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01890122 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01508195 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT01508182 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01365091 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Brazil ... more >> Local Institution Campinas, Sao Paulo, Brazil, 13073 Collapse << |
| NCT01890122 | - | - | Completed | - | - |
| NCT01970046 | Type 2 Diabetes | Phase 3 | Unknown | January 2015 | China ... more >> Chinese PLA General Hospital Recruiting Beijing, China Contact: Changyu Pan, M.D. panchy301@aliyun.com Principal Investigator: Changyu Pan, M.D. Collapse << |
| NCT03756623 | Healthy | Not Applicable | Recruiting | June 30, 2019 | China, Jiangsu ... more >> The Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School Recruiting Nanjing, Jiangsu, China, 210008 Contact: Mingming Zhang, MD,PhD 18551831115 doczmm@126.com Collapse << |
| NCT01101438 | Breast Cancer | Phase 3 | Active, not recruiting | February 2022 | - |
| NCT01365091 | - | - | Completed | - | - |
| NCT01087866 | Gestational Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | Finland ... more >> Oulu University Hospital Oulu, Finland, 90029 Collapse << |
| NCT01233206 | Infertility P... more >>olycystic Ovary Syndrome (PCOS) Collapse << | Phase 4 | Completed | - | Italy ... more >> Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT03297879 | Type2 Diabetes | Phase 4 | Completed | - | - |
| NCT02045290 | Type 1 Diabetes | Phase 3 | Completed | - | United States, Florida ... more >> Jaeb Center for Health Research Tampa, Florida, United States, 33647 Collapse << |
| NCT01791647 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Italy ... more >> Catholic University of Sacred Heart Recruiting Rome, Italy, 00168 Contact: Maurizio Guido, Medical Doctor 063057794 Principal Investigator: Maurizio Guido, Medical Doctor Collapse << |
| NCT03316690 | Impaired Glucose Tolerance ... more >> Diabetes Mellitus, Type 2 Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT00337610 | - | - | Completed | - | - |
| NCT01766778 | Type-2 Diabetes Mellitus | Phase 4 | Completed | - | Hong Kong ... more >> Novartis Investigative Site Hong Kong SAR, Hong Kong Novartis Investigative Site HongKong, Hong Kong Novartis Investigative Site Tuen Mun, Hong Kong Collapse << |
| NCT01353391 | Type 2 Diabetes | Phase 3 | Active, not recruiting | June 2019 | Canada, Ontario ... more >> The Centre for Mother, Infant, and Child Research, Sunnybrook Research Institute Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT00770653 | Diabetes Mellitus ... more >> Dyslipidemias Collapse << | Phase 3 | Completed | - | - |
| NCT00770653 | - | - | Completed | - | - |
| NCT01766778 | - | - | Completed | - | - |
| NCT00875394 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00875394 | - | - | Completed | - | - |
| NCT00005669 | Hyperinsulinemia ... more >> Obesity Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00337610 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 3 | Completed | - | - |
| NCT00005669 | - | - | Completed | - | - |
| NCT02551874 | - | - | Completed | - | - |
| NCT02551874 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01489644 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Novo Nordisk Investigational Site Miami, Florida, United States, 33169 United States, Washington Novo Nordisk Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT00835991 | - | - | Completed | - | - |
| NCT03078725 | Gestational Diabetes Mellitus,... more >> Class A2 Collapse << | Phase 4 | Withdrawn(Research was competi... more >>ng with other projects in the department, and no patients were recruited.) Collapse << | May 10, 2018 | - |
| NCT01509001 | Type 2 Diabetes | Phase 4 | Completed | - | Brazil ... more >> Clinical Hospital of São Paulo Medical School são Paulo, SP, Brazil Collapse << |
| NCT00836472 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT00836472 | - | - | Completed | - | - |
| NCT00835991 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT01910441 | - | - | Terminated(The study was termi... more >>nated due to the unavailability of Continuous Glucose Monitoring sensors (CGMS) which were required to assess the primary end-point.) Collapse << | - | - |
| NCT00468039 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, Texas ... more >> Diabetes and Glandular Disease Research Associates San Antonio, Texas, United States, 78229 Collapse << |
| NCT00961857 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01910441 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(The study was termi... more >>nated due to the unavailability of Continuous Glucose Monitoring sensors (CGMS) which were required to assess the primary end-point.) Collapse << | - | - |
| NCT01622257 | PCOS | Phase 2 | Completed | - | Egypt ... more >> Ain shams university Cairo, Egypt Collapse << |
| NCT01622257 | - | - | Completed | - | - |
| NCT02244567 | Polycystic Ovarian Syndrome | Phase 1 | Recruiting | June 2019 | Egypt ... more >> Nesreen Abdel Fattah Abdullah Shehata Recruiting Cairo, Egypt Contact: Nesreen A Shehata, MD 00201227866337 nesoomar@yahoo.com Collapse << |
| NCT02294006 | Well Differentiated Pancreatic... more >> Endocrine Tumor Collapse << | Phase 2 | Unknown | June 2017 | Italy ... more >> Fondazione IRCCS Istituto Tumori Milano Recruiting Milano, Italy, 20133 Contact: Sara Pusceddu, MD sara.pusceddu@istitutotumori.mi.it Collapse << |
| NCT01021579 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Zainabieh Hospital Shiraz, Fars, Iran, Islamic Republic of, 7173646199 Collapse << |
| NCT01008046 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals,OB/GYN department Mansoura, Dakahlia Governorate, Egypt Collapse << |
| NCT03380091 | PCOS Depressi... more >>on Vitamin D Deficiency Insulin Resistance Collapse << | Phase 4 | Recruiting | April 2019 | United States, California ... more >> UCSF Center for Reproductive Health Recruiting San Francisco, California, United States, 94158 Contact: Nik Lenhart 415-885-3598 Nik.Lenhart@ucsf.edu Collapse << |
| NCT01811953 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.8.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01845077 | - | - | Completed | - | - |
| NCT02048761 | Intrabony Defects in Chronic P... more >>eriodontitis Collapse << | Phase 2 | Unknown | February 2014 | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT01845077 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> 1288.8.1 Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT02173639 | Healthy | Phase 1 | Completed | - | - |
| NCT03513874 | Type 1 Diabetes Mellitus ... more >> Autoimmune Diabetes Collapse << | Phase 4 | Recruiting | March 1, 2019 | China, Hunan ... more >> Institute of Metabolism and Endocrinology, Second Xiangya Hospital, Central South University Recruiting Changsha, Hunan, China, 410011 Contact: Zhiguang Zhou, MD/PhD 86-731-85292154 zhouzg@hotmail.com Collapse << |
| NCT01211197 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Germany ... more >> 1276.5.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01917656 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01211197 | - | - | Completed | - | - |
| NCT02083692 | Head and Neck Squamous Cell Ca... more >>ncer Collapse << | Early Phase 1 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT01811953 | - | - | Completed | - | - |
| NCT01917656 | - | - | Completed | - | - |
| NCT01587378 | Pregnancy Pol... more >>ycystic Ovary Syndrome Collapse << | Not Applicable | Completed | - | Iceland ... more >> Landspital University Hospital Reykjavik, Iceland, 101 Norway Haukeland University Hospital Bergen, Norway, 5000 Nordlandssykehuset HF Bodø, Norway, 1480 Sykehuset Innlandet Brumunddal, Norway, 2380 Vestre Viken Hospital Trust Drammen, Norway Vestre Viken Ringerike sykehus Honefoss, Norway, 3511 Sykehuset Telemark Skien, Norway, 3710 St Olavs Hospital Trondheim, Norway, 7006 Sentralsykehuset i Vestfold Tønsberg, Norway, 3116 Sykehuset Ålesund Ålesund, Norway, 6026 Sweden Karolinska Universitetssjukhus Stockholm, Sweden, 17176 Norrlands universitetssjukhus Umeå, Sweden, 901 85 Uppsala University Hospital Uppsala, Sweden, 75185 Collapse << |
| NCT01725490 | Familial Adenomatous Polyposis | Phase 2 | Unknown | September 2015 | Korea, Republic of ... more >> Department of Internal Medicine, Yonsei University College of Medicine Not yet recruiting Seoul, Korea, Republic of, 120-752 Contact: TAE IL KIM, MD 82-2-2228-1965 taeilkim@yuhs.ac Department of Internal Medicine, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 120-752 Contact: TAE IL KIM, MD 82-2-2228-1965 taeilkim@yuhs.ac Collapse << |
| NCT01525225 | Type 2 Diabetes Mellitus | Phase 1 | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | United States, Arkansas ... more >> Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 United States, Kentucky Kosair Charities Pediatric Clinical Research Unit Louisville, Kentucky, United States, 40202 United States, Missouri The Children'S Mercy Hospital And Clinics Kansas City, Missouri, United States, 64108 United States, Ohio Promedica Toledo Children'S Hospital Toledo, Ohio, United States, 43606 Promedica Toledo Childrens Hospital Toledo, Ohio, United States, 43606 United States, Pennsylvania Childrens Hospital Of Pittsburgh Of Upmc Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT01525225 | - | - | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | - |
| NCT01975220 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> 1276.13.1 Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT01187849 | Patients With Glucocorticoid T... more >>reatment Collapse << | Phase 4 | Completed | - | Switzerland ... more >> University Hospital Basel Basel, Basel-Stadt, Switzerland, 4031 Collapse << |
| NCT03189732 | Diabetes Mellitus, Type 2 | Not Applicable | Active, not recruiting | December 2017 | Czechia ... more >> Third Faculty of Medicine, Charles University Prague, Czechia, 100 00 Collapse << |
| NCT02106923 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT01975220 | - | - | Completed | - | - |
| NCT00881725 | Prostate Cancer | Phase 2 | Terminated(Slow Accrual) | - | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G2M9 Collapse << |
| NCT00690456 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Indonesia Sanofi-Aventis Administrative Office Jakarta, Indonesia Lithuania Sanofi-Aventis Administrative Office Vilnius, Lithuania Malaysia Sanofi-Aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Slovakia Sanofi-Aventis Administrative Office Brastislava, Slovakia Taiwan Sanofi-Aventis Administrative Office Taipei, Taiwan Thailand Sanofi-Aventis Administrative Office Bangkok, Thailand Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT01578551 | Carcinoma, Non-Small-Cell Lung... more >> Lung Adenocarcinoma Collapse << | Phase 2 | Active, not recruiting | November 2019 | United States, Maryland ... more >> Johns Hopkins Bayview Medical Center Baltimore, Maryland, United States, 21224 Johns Hopkins University SKCCC Baltimore, Maryland, United States, 21231 Collapse << |
| NCT00499707 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02106923 | - | - | Completed | - | - |
| NCT03088631 | Intracytoplasmatic Sperm Injec... more >>tion Collapse << | Phase 2 | Recruiting | May 2019 | Egypt ... more >> Women Health Hospital - Assiut university Recruiting Assiut, Egypt, 71111 Contact: Mohammed K ALi, MD +201005537951 m_khairy2001@yahoo.com Collapse << |
| NCT00959101 | Diabetes Heal... more >>thy Collapse << | Phase 1 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100032 Collapse << |
| NCT01590797 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01167738 | Pancreatic Cancer | Phase 2 | Terminated(concern of detrimen... more >>tal effect) Collapse << | - | Italy ... more >> Istituto Scientifico H. San Raffaele Milan, Italy, 20132 Collapse << |
| NCT01797523 | Endometrial Cancer | Phase 2 | Active, not recruiting | October 2020 | United States, Florida ... more >> Sacred Heart Health Systems Pensacola, Florida, United States, 32504 United States, New Jersey MD Anderson Cooper Cancer Center Voorhees, New Jersey, United States, 08043 United States, Texas Memorial City Medical Center Houston, Texas, United States, 77024 Lyndon B Johnson General Hospital Houston, Texas, United States, 77026 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 The Woman's Hospital of Texas Houston, Texas, United States, 77054 Collapse << |
| NCT01590797 | - | - | Completed | - | - |
| NCT02183506 | Healthy | Phase 1 | Completed | - | - |
| NCT01690091 | Insulin Resistance ... more >> Chronic Heart Failure Collapse << | Phase 2 Phase 3 | Completed | - | Czechia ... more >> Diabetes Center, Institute of Clinical and Experimental Medicine Prague, Czechia Collapse << |
| NCT01550900 | Colorectum | Not Applicable | Withdrawn | - | - |
| NCT01885013 | Human Epidermal Growth Factor ... more >>2 Negative Carcinoma of Breast Collapse << | Phase 2 | Completed | - | Italy ... more >> P.O. M. Bufalini Cesena, FC, Italy UO Oncologia Medica IRCCS IRST Meldola (FC), FC, Italy, 47014 Ospedale Civile degli Infermi Faenza, RA, Italy Ospedale Umberto I Lugo, RA, Italy Ospedale Civile Santa Maria delle Croci Ravenna, RA, Italy ULSS n.8 Asolo Ospedale di Castelfranco Asolo, Italy Centro di Riferimento Oncologico CRO Aviano, Italy Ospedale S.Martino Belluno, Italy Azienda Ospedaliera "Antonio Cardarelli" Campobasso, Italy P.O. "SS. Annunziata" Chieti, Italy Presidio Ospedaliero E. Profili Fabriano, Italy E.O. Galliera Genova, Italy Ospedale di Guastalla Guastalla, Italy Azienda per i Servizi Sanitari n.5 "Bassa Friulana" Latisana, Italy Presidio Ospedaliero "Vito Fazzi" Lecce, Italy ULSS n.13 di Mirano Mirano, Italy Arcispedale S. Maria Nuova Modena, Italy Azienda Ospedaliera S. Salvatore di Pesaro Pesaro, Italy Ospedale S. Spirito Pescara, Italy Ospedale Civile di Piacenza Piacenza, Italy Azienda Ospedaliera Santa Maria degli Angeli Pordenone, Italy Arcispedale S. Maria Nuova Reggio Emilia, Italy Ospedale Civile degli Infermi Rimini, Italy IRCCS Centro di riferimento Oncologico di Basilicata di Rionero in Vulture Rionero in Vulture, Italy Ospedale Nuovo Regina Margherita Roma, Italy Collapse << |
| NCT00855166 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Bulgaria ... more >> Research Site Blagoevgrad, Bulgaria Research Site Sofia, Bulgaria Czech Republic Research Site Beroun, Czech Republic Research Site Brno, Czech Republic Research Site Praha, Czech Republic Research Site Semily, Czech Republic Research Site Slany, Czech Republic Hungary Research Site Balatonfured, Hungary Research Site Budapest, Hungary Research Site Csongrad, Hungary Research Site Kecskemet, Hungary Research Site TAT, Hungary Poland Research Site Elblag, Poland Research Site Krakow, Poland Research Site Torun, Poland Sweden Research Site Goteborg, Sweden Research Site Jarfalla, Sweden Research Site Lund, Sweden Research Site Malmo, Sweden Research Site Stockholm, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT00855166 | - | - | Completed | - | - |
| NCT01333852 | Head and Neck Neoplasms ... more >> Squamous Cell Carcinoma Collapse << | Phase 2 | Terminated(Low accrual) | - | Brazil ... more >> Barretos Cancer Hospital Barretos, SP, Brazil, 14784400 Collapse << |
| NCT03243851 | Glioblastoma | Phase 2 | Recruiting | December 31, 2018 | Korea, Republic of ... more >> National Cancer Center Korea Recruiting Ilsan, Gyeonggi-Do, Korea, Republic of, 10408 Contact: Heon Yoo, MD., PhD. +82-31-920-1249 heonyoo@ncc.re.kr Seoul National University Bundang Hospital Recruiting Seongnam-si, Gyeonggi-Do, Korea, Republic of, 13620 Contact: Yu Jung Kim, MD., PhD. +82-31-787-8210 cong1005@gmail.com Ajou University Hospital Recruiting Suwon-si, Gyeonggi-Do, Korea, Republic of, 16499 Contact: Se Hyuk Kim, MD., PhD. +82-31-219-3559 nsksh@ajou.ac.kr Chonnam National University Hwasun Hospital Recruiting Hwasun, Jeollanam-Do, Korea, Republic of, 58128 Contact: Shin Jung, MD., PhD. +82-2-379-8954 sjung@chonnam.ac.kr Incheon St. Mary's Hospital Recruiting Incheon, Korea, Republic of, 21431 Contact: Dong Sup Chung, MD., PhD. +82-32-280-5876 dschung@catholic.ac.kr Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Contact: Tae Min Kim, MD., PhD. +82-2-2072-1672 gabriel9@snu.ac.kr Severance Hospital Recruiting Seoul, Korea, Republic of, 03722 Contact: Jong Hee Chang, MD., PhD. +82-2-2228-2162 changjh@yuhs.ac Konkuk University Hospital Recruiting Seoul, Korea, Republic of, 05030 Contact: Young Cho Koh, MD., PhD. +82-2-2030-7622 kohyc@kuh.ac.kr Asan Medical Center Recruiting Seoul, Korea, Republic of, 05505 Contact: Jeong Hoon Kim, MD., PhD. +82-2-3010-3559 jhkim1@amc.seoul.kr Samsung Medical Center Recruiting Seoul, Korea, Republic of, 06351 Contact: Do Hyun Nam, MD., PhD. +82-2-3410-3497 nsnam@skku.edu Seoul St Mary's Hospital Recruiting Seoul, Korea, Republic of, 06591 Contact: Yong-Kil Hong, MD., PhD. +82-2-2258-2101 hongyk@catholic.ac.kr Saint Vincent's Hospital, Korea Recruiting Suwon-si, Korea, Republic of, 16247 Contact: Seung Ho Yang, MD., PhD. +82-31-249-8304 72ysh@catholic.ac.kr Collapse << |
| NCT03047837 | Tertiary Prevention in Colon C... more >>ancer Collapse << | Phase 2 | Recruiting | March 15, 2020 | Italy ... more >> Medical Oncology Ente Ospedaliero Ospedali Galliera Recruiting Genova, Italy, 16128 Contact: Andrea De Censi, MD +39010 5634501 andrea.decensi@galliera.it Contact: Marilena Petrera, PhD 00390105634580 marilena.petrera@galliera.it Collapse << |
| NCT03108963 | PCOS | Phase 2 | Completed | - | Egypt ... more >> Zagazig University Zagazig, Egypt Collapse << |
| NCT01429818 | Endothelial Dysfunction ... more >> Diabetes Mellitus Collapse << | Phase 4 | Completed | - | Mexico ... more >> Unidad PET Ciclotrón. Facultad de Medicina. Universidad Nacional Autónoma de México Mexico city, Distrito Federal, Mexico, 04510 Collapse << |
| NCT02910817 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Assiut University Assiut, Egypt, 71515 Collapse << |
| NCT02514629 | Hypogonadism ... more >>Obesity Collapse << | Phase 3 | Completed | - | Spain ... more >> Hospital Uiversitario Virgen de la Victoria Malaga, Spain, 29010 Collapse << |
| NCT03600363 | Lymphoma, Large B-Cell, Diffus... more >>e Stage III Follicular Lymphoma Collapse << | Phase 2 | Not yet recruiting | December 2020 | - |
| NCT01627067 | Breast Cancer | Phase 2 | Active, not recruiting | September 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02791490 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03603912 | Atrial Fibrillation | Phase 4 | Recruiting | June 30, 2022 | United States, Ohio ... more >> Cleveland Clinic Recruiting Cleveland, Ohio, United States, 44195 Contact: Mina K Chung, MD 216-444-2290 chungm@ccf.org Contact: Niraj Varma, MD 216-444-2142 varman@ccf.org Collapse << |
| NCT01192152 | Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT03238495 | HER2-positive Breast Cancer | Phase 2 | Recruiting | September 1, 2019 | United States, Kansas ... more >> The University of Kansas Cancer Center, West Clinic Recruiting Kansas City, Kansas, United States, 66112 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Westwood Campus Recruiting Kansas City, Kansas, United States, 66205 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Overland Park Clinic Recruiting Overland Park, Kansas, United States, 66210 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu United States, Missouri The University of Kansas Cancer Center, South Clinic Recruiting Kansas City, Missouri, United States, 64131 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, North Clinic Recruiting Kansas City, Missouri, United States, 64154 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Lee's Summit Clinic Recruiting Lee's Summit, Missouri, United States, 64064 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu Collapse << |
| NCT01966978 | Diabetes Mellitus, Type 2 ... more >> Diabetes Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> UT Southwestern Medical Center Dallas, Texas, United States, 02720 Collapse << |
| NCT03709147 | Advanced LKB1-inactive Lung Ad... more >>enocarcinoma Collapse << | Phase 2 | Not yet recruiting | September 10, 2023 | Italy ... more >> Marina Chiara Garassino Not yet recruiting Milan, Italy, 20133 Contact: MARINA CHIARA GARASSINO, MD 0223903813 ext +39 marina.garassino@istitutotumori.mi.it Collapse << |
| NCT01192152 | - | - | Completed | - | - |
| NCT01427595 | Polycystic Ovary Syndrome ... more >> Hyperandrogenism Collapse << | Not Applicable | Active, not recruiting | December 2019 | United States, Virginia ... more >> Center for Research in Reproduction, University of Virginia Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT03311308 | Advanced Melanoma | Phase 1 | Recruiting | December 2028 | United States, Pennsylvania ... more >> Univ of Pittsburgh Medical Center Hillman Cancer Center Recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: Amy Rose, RN 412-647-8587 Collapse << |
| NCT00063232 | Hepatitis | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03312582 | Peri-Implantitis | Phase 4 | Completed | - | - |
| NCT02186483 | Hyperlipidemia ... more >> Diabetes Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center(SMC) Seoul, Gangnam-Gu, Korea, Republic of, 135-710 Collapse << |
| NCT00063232 | - | - | Completed | - | - |
| NCT00670800 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | United States, Michigan ... more >> University of Michigan, Michigan Clinical Research Unit Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02438397 | Type 2 Diabetes | Phase 4 | Unknown | October 2016 | China, Shanghai ... more >> the 6th affliliated hospital of Shanghai Jiaotong university Recruiting Shanghai, Shanghai, China, 200233 Collapse << |
| NCT00670800 | - | - | Completed | - | - |
| NCT03184493 | Liver Cancer | Phase 3 | Recruiting | June 2021 | China, Guangxi ... more >> Affiliated Tumor Hospital of Guangxi Medical University Recruiting Nanjing, Guangxi, China, 530021 Contact: Bang-de Xiang, PhD 7715330855 xiangbangde@163.com Collapse << |
| NCT00772174 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02827903 | Type 2 Diabetes ... more >> Dyslipidemia Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Buchoen St. mary's Hospital Bucheon, Korea, Republic of SeJong Hostpital Bucheon, Korea, Republic of SoonChunHyang University Bucheon Hospital Bucheon, Korea, Republic of InJe University Busan paik Hospital Busan, Korea, Republic of Inje University Haeundae paik hospital Busan, Korea, Republic of Daegu Catholic University Medical Center Daegu, Korea, Republic of Youngnam University Hostpital Daegu, Korea, Republic of Daeheon Eulji Medical Center Daejeon, Korea, Republic of KonYang University Hostpital Daejeon, Korea, Republic of Chosun University Hospital Gwangju, Korea, Republic of BunDang Jesang Hospital(DMC) Gyeonggi-do, Korea, Republic of Hallym University Sacred Heart hospital Gyeonggi-do, Korea, Republic of HanYang University Guri Hospital Gyeonggi-do, Korea, Republic of Inje University Ilsan paik hospital Gyeonggi-do, Korea, Republic of Myoung Ji Hospital Gyeonggi-do, Korea, Republic of Gachon Gil Hospital Incheon, Korea, Republic of Inchoen St. mary's Hospital Incheon, Korea, Republic of Eulji Hospital Nowon Seoul, Korea, Republic of Hallym University Kangnam Sacred Heart hospital Seoul, Korea, Republic of KangBuk SamSung Hospital Seoul, Korea, Republic of Kangdong Sacred Heart Hospital Seoul, Korea, Republic of KonKuk University Medical Center Seoul, Korea, Republic of Korea University Anam Hospital Seoul, Korea, Republic of KyungHee University Hospital at Gangdong Seoul, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of Seoul National University Hospital(Famlily medicine) Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Severance hospital Seoul, Korea, Republic of SoonChunHayng University Hospital Seoul Seoul, Korea, Republic of The catholic University of Korea YEOUIDO St mary's Hospital Seoul, Korea, Republic of Korea University Guro hospital Soeul, Korea, Republic of Ajou University hospital Suwon, Korea, Republic of Wonju Severance Christian hospital Wŏnju, Korea, Republic of Collapse << |
| NCT02077803 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01208740 | Infertility P... more >>COS Prognosis for Poor Response Collapse << | Phase 4 | Terminated(Concerns about safe... more >>ty at the first interim analysis) Collapse << | - | Italy ... more >> Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT02495103 | Renal Cell Carcinoma ... more >> Hereditary Leiomyomatosis and Renal Cell Cancer Papillary Renal Cell Carcinoma, Sporadic Collapse << | Phase 1 Phase 2 | Recruiting | May 1, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00953355 | Polycystic Ovary Syndrome ... more >> Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy Collapse << |
| NCT02185066 | Healthy Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Busan Paik Hospital Busan, Korea, Republic of Collapse << |
| NCT01555164 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01864681 | NSCLC EGFR Ge... more >>ne Amplification Advanced Cancer Stage IIIB NSCLC Stage IV NSCLC Collapse << | Phase 2 | Completed | - | China, Chongqing ... more >> Department of Respiratory Diseases, Daping Hospital, Third Military Medical University Chongqing, Chongqing, China, 400042 Collapse << |
| NCT01528254 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | April 18, 2019 | - |
| NCT01555164 | - | - | Completed | - | - |
| NCT02049814 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Anhui ... more >> Hefei, Anhui, China Maanshan, Anhui, China China, Beijing Beijing, Beijing, China China, Guangdong Guangzhou, Guangdong, China Shenzhen, Guangdong, China Taishan, Guangdong, China China, Hunan Zhuzhou, Hunan, China China, Jiangsu Nanjing, Jiangsu, China Xuzhou, Jiangsu, China China, Jilin Changchun, Jilin, China Jilin, Jilin, China China, Liaoning Shenyang, Liaoning, China China, Shandong Qingdao, Shandong, China China, Shanghai Shanghai, Shanghai, China China, Shanxi Yan An, Shanxi, China Yanan, Shanxi, China China, Tianjin Tianjin, Tianjin, China China, Zhejiang Wenzhou, Zhejiang, China Collapse << |
| NCT02477969 | Type 2 Diabetes Mellitus | Phase 3 | Unknown | June 2017 | China, Shanghai ... more >> Shanghai sixth People's Hospital Shanghai, Shanghai, China, 200233 Collapse << |
| NCT00528879 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00134303 | Obesity | Phase 4 | Unknown | July 2015 | Belgium ... more >> University Hospital Ghent Recruiting Ghent, Belgium, 9000 Contact: Hans Van Vlierberghe, MD, PhD + 32 9 332.23.70 hans.vanvlierberghe@UGent.be Collapse << |
| NCT03010683 | Type 2 Diabetes Mellitus ... more >> Coronary Artery Disease Collapse << | Not Applicable | Recruiting | December 2017 | Greece ... more >> ''Attikon'' University General Hospital Recruiting Haidari, Athens, Attiki, Greece, 12462 Principal Investigator: Ignatios Ikonomidis, MD Collapse << |
| NCT00993187 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01423487 | Weight Gain A... more >>menorrhea Collapse << | Not Applicable | Withdrawn(Difficult to obtain ... more >>informed consent) Collapse << | August 2013 | China, Hunan ... more >> Mental Health Institute of The Second Xiangya Hospital Changsha, Hunan, China, 410011 Collapse << |
| NCT00501904 | Polycystic Ovary Syndrome | Phase 4 | Unknown | December 2016 | Italy ... more >> Pugliese Hospital Recruiting Catanzaro, Catanzaro, CZ, Italy, Italy, 88100 Contact: Ingrid Tomaino, MD +39-0961965097 angela.falbo@libero.it Principal Investigator: Stefano Palomba, MD Collapse << |
| NCT00525330 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> San Diego, California, United States Collapse << |
| NCT00528879 | - | - | Completed | - | - |
| NCT02570672 | Frailty | Phase 2 | Recruiting | October 2018 | United States, Texas ... more >> University of Texas Health Science Center at San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Alicia Conde 210-617-5197 Collapse << |
| NCT02276274 | Clinical Pharmacology | Phase 3 | Completed | - | Japan ... more >> Fukuoka-shi, Fukuoka, Japan Collapse << |
| NCT00993187 | - | - | Completed | - | - |
| NCT00145379 | Type 1 Diabetes Mellitus | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Center Odense, Funen, Denmark, 5000 Collapse << |
| NCT00247117 | Liver Diseases | Not Applicable | Unknown | - | Israel ... more >> Institute of Endocrinology, Kaplan Medical Center Recruiting Rehovot, Israel, 76100 Collapse << |
| NCT02276274 | - | - | Completed | - | - |
| NCT01240759 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Texas ... more >> Juno Research, LLC Houston, Texas, United States, 77074 Collapse << |
| NCT00865033 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01438190 | Infertility P... more >>COS Collapse << | Phase 4 | Unknown | December 2014 | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT00754689 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(Company decision tak... more >>en in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT02689843 | Polycystic Ovary Syndrome | Early Phase 1 | Completed | - | Iran, Islamic Republic of ... more >> Shahid Motahhari Clinic, Shiraz University of Medical Sciences Shiraz, Fars, Iran, Islamic Republic of Collapse << |
| NCT02614339 | Non-DM Stage III Colorectal Ca... more >>ncer Collapse << | Phase 3 | Not yet recruiting | November 2021 | - |
| NCT01195259 | - | - | Completed | - | - |
| NCT00359112 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 3 | Completed | - | - |
| NCT01477060 | Metastatic Breast Cancer | Phase 2 | Terminated(The study was stopp... more >>ed due to insufficient accrual) Collapse << | - | Italy ... more >> Cliniche Gavazzeni S.p.A. - Humanitas Gavazzeni Bergamo, BG, Italy, 24125 Fondazione Poliambulanza Brescia, BS, Italy, 25124 Azienda Ospedaliera San Gerardo Monza, MB, Italy, 20052 Azienda Ospedaliera "G. Salvini" - P.O. Garbagnate Milanese Garbagnate Milanese, MI, Italy, 20020 Ospedale Civile Di Legnano Legnano, MI, Italy, 20025 IRCCS Fondazione San Raffaele Monte Tabor Milano, MI, Italy, 20132 IRCCS Istituto Nazionale dei Tumori Milano, MI, Italy, 20133 Azienda Ospedaliera Ospedale Ca' Granda Milano, MI, Italy, 20162 Fondazione Salvatore Maugeri Clinica del Lavoro e della Riabilitazione - Reparto Riabilitazione Oncologica Pavia, PV, Italy, 27100 Fondazione Salvatore Maugeri Clinica del Lavoro e della Riabilitazione - U.O. Oncologia Pavia, PV, Italy, 27100 Azienda Ospedaliera della Valtellina e della Valchiavenna - P.O. Sondrio Sondrio, SO, Italy, 23100 Collapse << |
| NCT01135433 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Chubu, Japan Chugoku, Japan Hokkaido, Japan Kansai, Japan Kantou, Japan Kyushu, Japan Touhoku, Japan Collapse << |
| NCT01037842 | Type 2 Diabetes | Phase 3 | Completed | - | Korea, Republic of ... more >> Pusan National University College of Medicine Busan, Korea, Republic of Kyungpook National University School of Medicine Daegu, Korea, Republic of Chungnam National University College of Medicine Daejeon, Korea, Republic of Chonnam National University College of Medicine Gwangju, Korea, Republic of Chonbuk National University College of Medicine Jeonju, Korea, Republic of Eulji University School of Medicine Seoul, Korea, Republic of Hallym University College of Medicine Seoul, Korea, Republic of Korea University College of Medicine Seoul, Korea, Republic of Seoul St. Mary's Hospital Seoul, Korea, Republic of SungKyunKwan University School of Medicine Seoul, Korea, Republic of The Catholic University of Korea Seoul, Korea, Republic of Yonsei University College of Medicine Seoul, Korea, Republic of Uijeongbu St. Mary Hospital Uijeongbu, Korea, Republic of Collapse << |
| NCT02457286 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Collapse << | Phase 1 | Withdrawn(patients do not want... more >> to participate) Collapse << | - | United States, New York ... more >> North Shore Hospital Manhasset, New York, United States, 11030 Collapse << |
| NCT01459809 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00666458 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00857870 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> GWT -TUD, Center for Clinical Studies Dresden, Germany, 01307 Collapse << |
| NCT00666458 | - | - | Completed | - | - |
| NCT00396357 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative Site Investigative Centers, Germany Collapse << |
| NCT02744976 | Coronary Artery Disease ... more >> Atherosclerosis Prediabetic State Collapse << | Phase 4 | Recruiting | December 2020 | Latvia ... more >> Pauls Stradins Clinical University hospital Recruiting Riga, Latvia Collapse << |
| NCT02813863 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02911792 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | April 2019 | United States, Florida ... more >> Sanford Burnham Prebys Medical Discovery Institute Not yet recruiting Orlando, Florida, United States, 32827 Contact: Richard Pratley, MD 407-745-2145 rpratley@SBPdiscovery.org United States, Illinois Northwestern Medical School Not yet recruiting Chicago, Illinois, United States, 60611 Contact: Mark Molitch, MD 312-503-4130 molitch@northwestern.edu The University of Chicago Not yet recruiting Chicago, Illinois, United States, 60637 Contact: George Bakris, MD 773-702-7936 gbakris@medicine.bsd.uchicago.edu United States, Texas The University of Texas Health Science Center at San Antonio Not yet recruiting San Antonio, Texas, United States, 78229 Contact: Ralph A DeFronzo, MD 210-567-6691 defronzo@uthscsa.edu Contact: Monica Palomo, BS 210-567-6710 palomom@uthscsa.edu Sub-Investigator: Ralph A DeFronzo, MD Principal Investigator: Eugenio Cersosimo, MD University Health Systems Texas Diabetic Institute Recruiting San Antonio, Texas, United States Contact: Eugenio Cersosimo, MD 210-358-7200 cersosimo@uthscsa.edu United States, Washington Sacred Heart Medical Center Not yet recruiting Spokane, Washington, United States, 99204 Contact: Katherine Tuttle, MD 509-340-0930 Australia, Victoria Baker Medical Research Institute and Alfred Hospital Not yet recruiting Melbourne, Victoria, Australia Contact: Mark Cooper, MD 61385321362 mark.cooper@bakeridi.edu.au Collapse << |
| NCT01984489 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT01089166 | Type 2 Diabetes | Phase 1 | Completed | - | India ... more >> Torrent Research Centre Bhat, Gujarat, India, 382 428 Collapse << |
| NCT02866786 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | India ... more >> Department of Obstetrics & Gynecology Cuttack, Odisha, India, 753007 Collapse << |
| NCT00376038 | Type 2 Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | Mexico ... more >> GSK Investigational Site Mexico, D.F., Mexico, 14050 Collapse << |
| NCT02611518 | Healthy | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany Collapse << |
| NCT01890629 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Severance hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02039258 | Healthy Volunteers | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT02172248 | Healthy | Phase 1 | Completed | - | - |
| NCT03670043 | Type2 Diabetes Mellitus | Phase 4 | Not yet recruiting | November 2020 | - |
| NCT02140788 | Schizophrenia ... more >> Schizo-affective Disorder Collapse << | Phase 4 | Terminated(PI left institution... more >>) Collapse << | - | - |
| NCT01809327 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01809327 | - | - | Completed | - | - |
| NCT02140788 | - | - | Terminated(PI left institution... more >>) Collapse << | - | - |
| NCT00819741 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Fujian ... more >> Novo Nordisk Investigational Site Fuzhou, Fujian, China, 350001 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210009 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210012 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210029 Novo Nordisk Investigational Site Suzhou, Jiangsu, China, 215004 Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200001 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200025 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200092 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200120 China, Zhejiang Novo Nordisk Investigational Site Hangzhou, Zhejiang, China, 310003 Novo Nordisk Investigational Site Wenzhou, Zhejiang, China, 325000 China Novo Nordisk Investigational Site Hangzhou, China, 310016 Novo Nordisk Investigational Site Nantong, China, 226001 Novo Nordisk Investigational Site Shanghai, China, 200040 Collapse << |
| NCT02637037 | Bioequivalence ... more >> Fixed Dose Combination Tablets Healthy Male and Female Subjects Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02637037 | - | - | Completed | - | - |
| NCT01318109 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | - |
| NCT01677299 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> Celerion, Inc. Tempe, Arizona, United States, 85283 United States, Nebraska Celerion, Inc. Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT01318109 | - | - | Completed | - | - |
| NCT02909933 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00819741 | - | - | Completed | - | - |
| NCT01677299 | - | - | Completed | - | - |
| NCT02190838 | Melanoma | Phase 2 | Unknown | December 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Ambulatory Chemotherapy Department Recruiting St. Petersburg, Russian Federation, 197758 Contact: Elena V. Tkachenko, MD +78124399575 elenatkachen@yandex.ru Principal Investigator: Elena V. Tkachenko, MD N.N. Petrov Research Institute of Oncology Chemotherapy and Innovative Technologies Department Recruiting St. Petersburg, Russian Federation, 197758 Contact: Aleksei V. Novik, MD, PhD +78124399505 anovik@list.ru Principal Investigator: Svetlana A. Protsenko, MD, PhD, DSc N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting St.Petersburg, Russian Federation, 191124 Contact: Tatiana Y. Semiglazova, MD, PhD, DSc tsemiglazova@mail.ru Principal Investigator: Tatiana V Semiglazova, MD, PhD, DSc Collapse << |
| NCT02431676 | Breast Cancer ... more >> Prostate Cancer Lung Cancer Colon Cancer Melanoma of Skin Endometrial Cancer Liver Cancer Pancreatic Cancer Rectal Cancer Kidney Cancer Other Solid Malignant Tumors Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, Maryland ... more >> Johns Hopkins ProHealth Baltimore, Maryland, United States, 21207 Collapse << |
| NCT00612144 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals, Co., LTD Seoul, Korea, Republic of Collapse << |
| NCT01352026 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 2 | Withdrawn(Several therapeutic ... more >>have been developed making more difficult the eligibility of patients - Recruitment of new centers wasn't possible due to competing trials) Collapse << | - | France ... more >> CHU de Nantes Nantes, France, 44093 CHU de Bordeaux Pessac, France, 33604 Collapse << |
| NCT01457911 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational Site Number 156001 Beijing, China, 100044 Investigational Site Number 156016 Beijing, China, 100088 Investigational Site Number 156003 Beijing, China, 100730 Investigational Site Number 156018 Beijing, China, 100730 Investigational Site Number 156002 Beijing, China, 100853 Investigational Site Number 156019 Changchun, China, 130041 Investigational Site Number 156017 Chengdu, China, 610083 Investigational Site Number 156014 Guangzhou, China, 510080 Investigational Site Number 156022 Guangzhou, China, 510150 Investigational Site Number 156011 Hangzhou, China, 310003 Investigational Site Number 156012 Hefei, China, 230001 Investigational Site Number 156020 Jinan, China, 250012 Investigational Site Number 156023 Jinan, China, 250021 Investigational Site Number 156010 Shanghai, China, 200040 Investigational Site Number 156006 Shanghai, China, 200072 Investigational Site Number 156009 Shanghai, China, 200080 Investigational Site Number 156021 Shenyang, China, 110022 Investigational Site Number 156005 Shijiazhuang, China, 050051 Investigational Site Number 156004 Taiyuan, China, 030001 Investigational Site Number 156024 Tianjin, China, 300052 Investigational Site Number 156008 Wuhan, China, 430022 Investigational Site Number 156015 Xiamen, China, 361003 Collapse << |
| NCT02109679 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1307.7.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02506777 | Breast Cancer | Phase 2 | Recruiting | August 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting Saint - Petersburg, Russian Federation, 191124 Contact: Tatiana Y Semiglazova, MD, PhD, DSc +79219468072 tsemiglazova@mail.ru Contact: Michael A Osipov, MD +79052075653 Ocipovmixail@mail.ru Principal Investigator: Tatiana Y Semiglazova, MD, PhD, DSc Sub-Investigator: Petr V Krivorotko, MD, PhD, DSc Collapse << |
| NCT02167061 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT00762957 | Diabetes Mellitus | Phase 3 | Terminated(Hepatic Safety Sign... more >>al Identified.) Collapse << | - | - |
| NCT00617240 | Weight Gain | Phase 2 | Completed | - | United States, North Carolina ... more >> University of North Carolina, Department of Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT03499704 | Diabetes Mellitus, Type 2 | Phase 4 | Not yet recruiting | December 31, 2020 | - |
| NCT01693289 | Focus of Study | Phase 1 | Completed | - | Egypt ... more >> Zagazig University Zagazig, Egypt Collapse << |
| NCT01437813 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Investigación Farmacológica Y Biofarmaceutica, S.A. de C.V. México, Mexico, 14610 Collapse << |
| NCT00617240 | - | - | Completed | - | - |
| NCT00948402 | Polycystic Ovarian Syndrome ... more >> Insulin Sensitivity Collapse << | Phase 3 | Completed | - | - |
| NCT00449605 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Chile Sanofi-Aventis Administrative Office Santiago, Chile Sanofi-aventis adminsitrative office Santiago, Chile Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark Finland Sanofi-Aventis Administrative Office Helsinki, Finland Hungary Sanofi-Aventis Administrative Office Budapest, Hungary India Sanofi-Aventis Administrative Office Mumbai, India Italy Sanofi-Aventis Administrative Office Milan, Italy Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Puerto Rico Sanofi-Aventis Administrative Office Puerto Rico, Puerto Rico Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Collapse << |
| NCT02429258 | - | - | Completed | - | - |
| NCT00969631 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals, OB/GYN department Mansoura, Dakahlia Governorate, Egypt Collapse << |
| NCT02429258 | Type II Diabetes | Phase 4 | Completed | - | - |
| NCT02471404 | Type 2 Diabetes Mellitus ... more >> Inadequate Glycaemic Control Collapse << | Phase 4 | Completed | - | - |
| NCT02488564 | HER-2 Positive Breast Cancer | Phase 2 | Active, not recruiting | October 2019 | Italy ... more >> IRST Oncologia medica Meldola, FC, Italy, 47014 Oncologia Medica AOU Ferrara Ferrara, FE, Italy, 44124 Oncologia Medica, Policlinico di Modena Modena, MO, Italy, 41100 Oncologia Medica Ospedale Guglielmo da Saliceto Parma, PR, Italy, 29121 Oncologia , IRCCS azienda ospedaliera S.Maria Nuova Reggio Emilia, RE, Italy, 42123 U.O Oncologia AUSLdella Romagna Rimini, RN, Italy, 47923 Day Hospital Oncologico, Ospedale Guastalla Guastalla, Italy, 42016 Oncologia Medica AUSL Imola Imola, Italy, 40133 Oncologia Medica AOU Parma Parma, Italy, 43100 Collapse << |
| NCT00727857 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03253562 | Reducing the Elevated Blood Pr... more >>essure for Diabetic Hypertensive Patients Collapse << | Phase 4 | Completed | - | Egypt ... more >> National institute of diabetes and endocrinology Cairo, Egypt, 11311 Collapse << |
| NCT00883259 | Obesity Polyc... more >>ystic Ovary Syndrome Gestational Diabetes Collapse << | Phase 4 | Unknown | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 "Pugliese" Hospital Not yet recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39-0961-883234 angela.falbo@libero.it University of Catanzaro, Italy Catanzaro, Italy, 88100 Collapse << |
| NCT00727857 | - | - | Completed | - | - |
| NCT01702298 | - | - | Completed | - | - |
| NCT01702298 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01856907 | Disorder of Glucose Regulation | Phase 4 | Completed | - | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70817 Collapse << |
| NCT01675310 | Pregestational Obesity (BMI > ... more >>27kg/m2) History of Polycystic Ovary Syndrome Collapse << | Phase 4 | Completed | - | Mexico ... more >> National Institute of Perinatology Mexico, Mexico, 11000 Collapse << |
| NCT00390273 | Hyperlactatemia | Phase 1 Phase 2 | Completed | - | France ... more >> Centre Hospitalier Universitaire de Grenoble Grenoble cedex 9, France, 38043 Collapse << |
| NCT01856907 | - | - | Completed | - | - |
| NCT02552355 | Physical Activity | Phase 2 | Completed | - | United States, Colorado ... more >> Colorado State University, Dept. of Health and Exercise Science Fort Collins, Colorado, United States, 80523-1582 Collapse << |
| NCT03151772 | Glioblastoma | Early Phase 1 | Recruiting | June 2019 | Sweden ... more >> Sahlgrenska University Hospital Recruiting Göteborg, Sweden Contact: Louise Carstam, MD Collapse << |
| NCT02234440 | NASH Related Cirrhosis | Not Applicable | Completed | - | India ... more >> Institute of Liver & Biliary Sciences New Delhi, Delhi, India, 110070 Collapse << |
| NCT02798172 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT01969318 | Type 2 Diabetes | Phase 2 | Completed | - | China ... more >> Chinese PLA General Hospital Beijing, China Collapse << |
| NCT01545388 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03651895 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Early Phase 1 | Not yet recruiting | August 31, 2020 | - |
| NCT03336840 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | June 2019 | China, Jiangsu ... more >> Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Recruiting Nanjing, Jiangsu, China, 210008 Contact: Dalong Zhu, MD,PhD 86-25-83-105302 zhudldr@gmail.com Collapse << |
| NCT00560690 | Hepatitis C | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14114 Collapse << |
| NCT00367055 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01775813 | Obesity Insul... more >>in Resistance Gonadal Dysfunction Type 2 Diabetes Collapse << | Phase 4 | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado Aurora, Colorado, United States, 80045 Collapse << |
| NCT03618654 | Head and Neck Squamous Cell Ca... more >>rcinoma Oral Cavity Squamous Cell Carcinoma Oropharyngeal Squamous Cell Carcinoma Collapse << | Phase 1 | Active, not recruiting | January 2021 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02565862 | Hepatitis C D... more >>iabetes Mellitus Insulin Resistance Collapse << | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud University Medical Center Nijmegen, Netherlands Collapse << |
| NCT00118937 | Diabetes Mellitus, Type 1 | Phase 4 | Completed | - | - |
| NCT01941199 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00367055 | - | - | Completed | - | - |
| NCT01545388 | - | - | Completed | - | - |
| NCT02223026 | Healthy | Phase 1 | Completed | - | - |
| NCT01995032 | Duchenne's Muscular Dystrophy ... more >>(DMD) Collapse << | Phase 3 | Completed | - | Switzerland ... more >> University Children's Hospital Basel, BS, Switzerland, 4031 Collapse << |
| NCT03246451 | Glucose Metabolism Disorders | Not Applicable | Completed | - | Denmark ... more >> Center for Diabetes Research Gentofte, Copenhagen, Denmark, 2900 Collapse << |
| NCT01206153 | Amenorrhea | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT02338193 | Diabetes Prevention in Women A... more >>fter GDM Who Are at High-risk Collapse << | Phase 3 | Active, not recruiting | March 2019 | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70815 Collapse << |
| NCT02609815 | Diabetes | Not Applicable | Recruiting | December 31, 2019 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Tae Jung Oh, MD, PHD 82-31-787-7078 ohtjmd@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT02484209 | Diabetes Mellitus | Phase 3 | Withdrawn(Inclusion criteria t... more >>oo strict making it difficult to recruit in primary care) Collapse << | November 2016 | - |
| NCT01841697 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03216278 | Healthy Subjects in Fasted and... more >> Fed State Collapse << | Phase 1 | Completed | - | Brazil ... more >> Research Site Aparecida de Goiania, Brazil, 74935-530 Collapse << |
| NCT01841697 | - | - | Completed | - | - |
| NCT00942123 | - | - | Completed | - | Italy ... more >> Ospedale Maggiore Policlinico Milano, MI, Italy, 20122 Collapse << |
| NCT00757588 | - | - | Completed | - | - |
| NCT02283554 | Periodontitis | Phase 2 | Completed | - | - |
| NCT00757588 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03204058 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT02497638 | Prostate Cancer | Phase 2 | Not yet recruiting | December 2022 | Canada, Ontario ... more >> Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Principal Investigator: Robert Hamilton, MD, MPH, FRCSC Principal Investigator: Neil Fleshner, MD, MPH, FRCSC Collapse << |
| NCT00900146 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Terminated(Numerically modest ... more >>lowering of HbA1c with canakinumab in combination with metformin was inadequate to continue patients with T2DM into Period IV of this study.) Collapse << | - | - |
| NCT00885378 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01612858 | Lipodystrophy ... more >> HIV Infection Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01612858 | - | - | Completed | - | - |
| NCT00543361 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT01535677 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT00900146 | - | - | Terminated(Numerically modest ... more >>lowering of HbA1c with canakinumab in combination with metformin was inadequate to continue patients with T2DM into Period IV of this study.) Collapse << | - | - |
| NCT02063802 | Obesity | Phase 2 | Unknown | May 2014 | Mexico ... more >> General Hospital of Mexico Recruiting Mexico, Distrito Federal, Mexico, 06726 Contact: Nayely G Garibay, Pediatrician +52 (55) 59-48-26-37 gngaribay@hotmail.com Contact: Mayra A Bustos, Doctor +52 7773406509 mayibe2609@gmail.com Collapse << |
| NCT01410604 | Obesity | Phase 4 | Completed | - | Mexico ... more >> Hospital Infantil de México Federico Gómez México, Distrito Federal, Mexico, 06720 Hospital Regional de Alta Especialidad del Bajío León, Guanajuato, Mexico, 37670 Collapse << |
| NCT01410604 | - | - | Completed | - | - |
| NCT01658514 | Renal Insufficiency | Phase 2 | Completed | - | - |
| NCT00414245 | Gestational Diabetes ... more >> Diabetes Mellitus Type 2 Collapse << | Phase 3 | Unknown | - | Israel ... more >> Division of Obstetrics and Gynecology, Soroka University Medical Center Not yet recruiting Beer-Sheva, Israel Contact: Boaz Sheizaf, MD 972-54-2401191 bsheizaf@bgu.ac.il Principal Investigator: Boaz Sheizaf, MD Collapse << |
| NCT01658514 | - | - | Completed | - | - |
| NCT01474018 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, Texas ... more >> UT Southwestern Medical Center Dallas, Texas, United States, 75390 UT Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT01204580 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Indonesia ... more >> Sanofi-Aventis Administrative Office Jakarta, Indonesia Collapse << |
| NCT01286233 | - | - | Unknown | September 2016 | - |
| NCT03352869 | Polycystic Ovary Syndrome ... more >> Overweight and Obesity Disorder of Glucose Regulation Collapse << | Phase 4 | Recruiting | July 31, 2018 | China ... more >> Renji Hospital Department of Endocrinology and Metabolism Recruiting Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Contact: Jing Ma, MD +86-15800983436 cherry1996@live.cn Collapse << |
| NCT00975065 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals Seoul, Korea, Republic of Collapse << |
| NCT03088670 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Russian Federation ... more >> Non-state healthcare Organization "Road Clinical Hospital at the station Chelyabinsk Open Joint Stock Company" Russian Railways Chelyabinsk, Russian Federation, 454048 State Budget Healthcare Institution of Moscow "Endocrinology Dispensary" Moscow, Russian Federation, 119034 SEI HPE "First Moscow State Medical University n.a. I.M. Sechenov of Ministry of Health and Social Development of the Russian Federation", Chair of faculty therapy #2 based at SHI of Moscow "City clinical hospital #61" Moscow, Russian Federation, 119048 State Budget Educational Institution higher professional education "1st Moscow State Medical University n.a. I.M. Sechenov of the Ministry of Healthcare of Russian Federation" Moscow, Russian Federation, 119435 Federal State Institution "Clinical Hospital № 1" of the Office of the President of the Russian Federation Moscow, Russian Federation, 121352 State Budget Healthcare Institution of Moscow city "City Clinical Hospital # 71 by the Department of Healthcare in Moscow" Moscow, Russian Federation, 121374 State Budget Educational Institution of higher professional education "1st Moscow State Medical University n.a. I.M. Sechenov of the Ministry of Healthcare of Russian Federation" based on Endocrinology department of Hospital № 67 n.a L A Vorohobova Moscow, Russian Federation, 123423 SEI APE "Russian Medical Academy of Postgraduate Education of Roszdrav", Chair of endocrinology and diabetology with course of endocrine surgery based at FSI "Central Clinical hospital of civil aviation" Moscow, Russian Federation, 125367 State budgetary institution "Nizhny Novgorod Regional Clinical Hospital n.a Semashko Nizhny Novgorod, Russian Federation, 603126 State Health Care Institution "Perm Regional Hospital for War Veterans" Perm, Russian Federation, 614097 State Educational Institution of Higher Professional Education "Perm State Medical Academy n.a. academician EA Wagner's "Ministry of Health of the Russian Federation on the basis of Department of Endocrinology Regional Clinical Hospital Perm, Russian Federation, 614990 SHI "Republic Hospital n.a. V.A. Baranov" Petrozavodsk, Russian Federation, 185019 State budgetary institution of Ryazan region "City Clinical Hospital № 11" Ryazan, Russian Federation, 390037 St. Petersburg State healthcare institution "City Outpatient Clinic #37" Saint Petersburg, Russian Federation, 191119 FSHI "Clinical Hospital #122 n.a. L.G. Sokolov of FMBA of Russia", Internal medicine department Saint Petersburg, Russian Federation, 194291 State Health Care Institution Leningrad Regional Hospital Saint Petersburg, Russian Federation, 194291 Autonomous Noncommercial Organization "Medical Centre "XXI сentury" Saint Petersburg, Russian Federation, 194354 St. Petersburg State healthcare institution "City Hospital of Saint Martyr Elizaveta" Saint Petersburg, Russian Federation, 195257 FSMEI HPE "Military Medical Academy n.a. S.M. Kirov" of the Ministry of Defence of the Russian Federation, 1st Internal medicine Chair of advanced training Saint Petersburg, Russian Federation, 198013 "Diabetes Center "LLC Samara, Russian Federation, 443041 State Budget Educational Institution higher professional education "Saratov State Medical University n.a. V.I. Razumovsky" of the Ministry of Healthcare of Russian Federation based on Clinical Hospital n.a S.R. Mirotvortsev Saratov, Russian Federation, , 410054 Municipal health care institution "City polyclinic №20 Saratov, Russian Federation, 410018 SBEI HPE "Smolensk State Medical Academy" of the Ministry of Health and Social Development of the Russian Federation, Center of clinical research of diagnostic and medicinal products, Chair of clinical pharmacology Smolensk, Russian Federation, 214019 State budget educational institution of higher education "Smolensk State Medical Academy," of Ministry of Health and Social Development of the Russian Federation on the basis of sanatorium SSMA Smolensk, Russian Federation, 214019 State autonomous healthcare institution of Yaroslavl Region "Сlinical hospital for emergency medical care n. a. N.V. Solovyov" Yaroslavl, Russian Federation, 150003 Municipal clinical health institution Health Service Novo-Yaroslavl Oil Refinery Yaroslavl, Russian Federation, 150023 SHI of Yaroslavl region "Regional Clinical Hospital", Endocrinology department Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT00968812 | - | - | Completed | - | - |
| NCT02695810 | Prediabetic State ... more >> Obesity Collapse << | Phase 2 | Recruiting | February 2019 | Denmark ... more >> Steno Diabetes Center A/S Recruiting Gentofte, Denmark, 2820 Contact: Kristine Færch, PhD +4530791461 krif@steno.dk Contact: Marit E Jørgensen, PhD +4530756008 maej@steno.dk Sub-Investigator: Hanan Amadid, MD Sub-Investigator: Lea B Nielsen, MSc Collapse << |
| NCT01606007 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01606007 | - | - | Completed | - | - |
| NCT01274130 | Healthy Adult | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00451568 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Odense University Hospital Odense, Denmark Collapse << |
| NCT00968812 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01267448 | Diabetes Mellitus Type 2 ... more >> Severe Hyperglycemia - Blood Glucose Level >300mg/dl. Collapse << | Phase 4 | Unknown | December 2012 | United States, Illinois ... more >> John Stroger Hospital of Cook County Not yet recruiting Chicago, Illinois, United States, 60612 Contact: Ambika Babu, M.D. 312-864-0543 Ambika_Babu@rush.edu Contact: Leon Fogelfeld, M.D. 312-864-0539 lfogelfe@cchil.org Principal Investigator: Ambika Babu, M.D. John Stroger Hospital of Cook County Recruiting Chicago, Illinois, United States, 60612 Contact: Ambika Babu, M.D. 312-864-0543 aamblee@hotmail.com Contact: Leon Fogelfeld, M.D. 312-864-0539 lfogelfe@cchil.org Principal Investigator: Ambika Babu, M.D. Collapse << |
| NCT01717911 | Type 2 Diabetes | Phase 4 | Unknown | December 2018 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD 886-2-28757515 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT01013051 | - | - | Completed | - | Canada, Alberta ... more >> University of Alberta Hospital Clinical Investigation Unity Edmonton, Alberta, Canada, T6G2B7 Collapse << |
| NCT02202161 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21225 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT00292799 | Anovulation O... more >>besity Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Sheffield Teaching Hospitals NHS Trust, Jessop Wing Sheffield, United Kingdom, S10 2SF Collapse << |
| NCT00770445 | Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Lichtenfels, Bavaria, Germany Frankfurt, Hessen, Germany Kassel, Hessen, Germany Wiesbaden, Hessen, Germany Bad Oeynhausen, North Rhine-Westphalia, Germany Dinslaken, North Rhine-Westphalia, Germany Duisburg, North Rhine-Westphalia, Germany Essen, North Rhine-Westphalia, Germany Münster, North Rhine-Westphalia, Germany Wuppertal, North Rhine-Westphalia, Germany Mainz, Rheinland-Pfalz, Germany Diez, Rhineland-Palatinate, Germany Landau, Rhineland-Palatinate, Germany Dresden, Saxony, Germany Leipzig, Saxony, Germany Jena, Thüringen, Germany Berlin, Germany Hamburg, Germany Collapse << |
| NCT02562664 | Reproductive Endocrinology ... more >> Polycystic Ovarian Syndrome Collapse << | Phase 2 | Completed | - | - |
| NCT02202161 | - | - | Completed | - | - |
| NCT02531308 | - | - | Terminated(poor accrual) | - | - |
| NCT02531308 | Diffuse Large B-Cell Lymphoma | Phase 2 | Terminated(poor accrual) | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT01998113 | - | - | Completed | - | Spain ... more >> Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT01704261 | - | - | Completed | - | - |
| NCT01704261 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00382096 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers, Germany Collapse << |
| NCT03204071 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT02434744 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Alabama ... more >> Achieve Clinical Research, LLC Birmingham, Alabama, United States, 35216 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 National Research Institute Los Angeles, California, United States, 90057 Infosphere Clinical Research, Inc West Hills, California, United States, 91307 United States, Florida Clinical Research of South Florida Coral Gables, Florida, United States, 33134 Med Research of Florida, LLC Miami, Florida, United States, 33186 United States, North Carolina High Point Clinical Trials Center High Point, North Carolina, United States, 27265 United States, South Carolina Coastal Carolina Research Center Mt. Pleasant, South Carolina, United States, 29464 United States, Texas Clinical Trial Network Houston, Texas, United States, 77074 United States, Virginia Clinical Research Associates of Tidewater Norfolk, Virginia, United States, 23507 Collapse << |
| NCT01245166 | The Objectives of the Study is... more >> to Evaluate the Efficacy and Safety of Acarmet (Metformin HCl 500 mg Plus Acarbose 50 mg Tablets) Thrice Daily Versus Acarbose 50 mg Thrice Daily Over 16 Weeks in Subjects With Type 2 Diabetes Mellitus. Collapse << | Phase 3 | Unknown | December 2011 | Taiwan ... more >> Taichung Veterans General Hospital Recruiting Taichung, Taiwan, 40705 Contact: Elaine Liu, Bachelor 886-2-2778-5188 ext 335 elaine@lotuspharm.com Collapse << |
| NCT00712673 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Australia sanofi-aventis Australia & New Zealand administrative office Macquarie Park, Australia Canada Sanofi-Aventis Administrative Office Laval, Canada Chile Sanofi-Aventis Administrative Office Santiago, Chile Croatia Sanofi-Aventis Administrative Office Zagreb, Croatia Czech Republic Sanofi-Aventis Administrative Office Praha, Czech Republic Germany Sanofi-Aventis Administrative Office Berlin, Germany Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Morocco Sanofi-Aventis Administrative Office Casablanca, Morocco Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation South Africa Sanofi-Aventis Administrative Office Midrand, South Africa Spain Sanofi-Aventis Administrative Office Barcelona, Spain Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Venezuela Sanofi-Aventis Administrative Office Caracas, Venezuela Collapse << |
| NCT01699932 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Lebanon ... more >> Investigational Site Number 422-002 Beirut, Lebanon Investigational Site Number 422-001 Hazmieh, Lebanon Russian Federation Investigational Site Number 643001 Samara, Russian Federation Investigational Site Number 643002 St-Petersburg, Russian Federation Investigational Site Number 643-03 St.-Petersburg, Russian Federation Ukraine Investigational Site Number 804003 Chernivtsi, Ukraine, 58022 Investigational Site Number 804008 Donetsk, Ukraine, 83003 Investigational Site Number 804004 Donetsk, Ukraine, 83059 Investigational Site Number 804001 Donetsk, Ukraine, 83099 Investigational Site Number 804010 Odessa, Ukraine Investigational Site Number 804006 Poltava, Ukraine, 36011 Investigational Site Number 804007 Vinnytsya, Ukraine, 21010 Investigational Site Number 804002 Vinnytsya, Ukraine, 21029 Collapse << |
| NCT03267576 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Mexico ... more >> Consultorio Privado Guadalajara, Mexico, 44150 Investigación Clínica Especializada Guadalajara, Mexico, 44600 Consultorio Privado en Unidad de Patología Clínica Guadalajara, Mexico, 44650 Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán Mexico City, Mexico, 14080 Hospital Universitario 'Dr. Jose Eleuterio Gonzalez' Monterrey, Mexico, 64460 Collapse << |
| NCT02133118 | - | - | Completed | - | Netherlands ... more >> Isala Zwolle, Overijssel, Netherlands, 8000 AB Collapse << |
| NCT01590771 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01023581 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01023581 | - | - | Completed | - | - |
| NCT00712673 | - | - | Completed | - | - |
| NCT01871415 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | China ... more >> Shanghai, China, 200003 ShenYang, China, 110004 Shiyan, China, 442000 Suzhou, China, 215004 Korea, Republic of Gyeonggi-do, Korea, Republic of, 463-712 Incheon, Korea, Republic of, 405-760 Seoul, Korea, Republic of, 150-950 Collapse << |
| NCT00241605 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02249910 | Diabetes Heal... more >>thy Collapse << | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany, 14050 Collapse << |
| NCT01136512 | - | - | Completed | - | United States, New York ... more >> Staten Island University Hospital Staten Island, New York, United States, 10305 Collapse << |
| NCT01590771 | - | - | Completed | - | - |
| NCT00437333 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT02614859 | Cancer of Prostate | Phase 2 | Recruiting | April 2022 | United States, Maryland ... more >> National Cancer Institute Recruiting Bethesda, Maryland, United States, 20892-9760 Contact: Marijo Bilusic, MD marijo.bilusic@nih.gov United States, Pennsylvania Fox Chase Cancer Center - Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT00701090 | Type 2 Diabetes Mellitus, Non ... more >>Insulin Dependent Diabetes Mellitus, Non-Insulin-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT01825798 | Overweight Au... more >>tism Spectrum Disorder Collapse << | Phase 3 | Completed | - | United States, Ohio ... more >> Ohio State University/Nationwide Children's Hospital Columbus, Ohio, United States, 43210 United States, Pennsylvania University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15203 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37212 Canada, Ontario Holland Bloorview Kids Rehabilitation Hospital Toronto, Ontario, Canada, M4G 1R8 Collapse << |
| NCT01825798 | - | - | Completed | - | - |
| NCT00303537 | Fatty Liver | Phase 2 Phase 3 | Unknown | June 2008 | Norway ... more >> Haukeland Universitetssykehus Bergen, Norway Aker University Hospital Oslo, Norway Akershus University Hospital Oslo, Norway Universitetssykehuset i Nord-Norge Tromsø, Norway Collapse << |
| NCT00701090 | - | - | Completed | - | - |
| NCT00131664 | - | - | Completed | - | - |
| NCT03189407 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | - |
| NCT00131664 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Canada, Ontario ... more >> Canadian Heart Research Centre Toronto, Ontario, Canada, m5b 2p9 Collapse << |
| NCT01524705 | Type 2 Diabetes | Phase 4 | Completed | - | United States, California ... more >> So Calif. Permanente Medical Group San Diego, California, United States, 92109 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Georgia Atlanta Diabetes Associates Atlanta, Georgia, United States, 30309 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Minnesota International Diabetes Center Minneapolis, Minnesota, United States, 55416 United States, Missouri Washington University St. Louis, Missouri, United States, 63110 United States, New York Kaledia Health of Western New York Buffalo, New York, United States, 14209 United States, North Carolina Diabetes Care Center Durham, North Carolina, United States, 27713 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239 United States, Vermont University of Vermont Colchester, Vermont, United States, 05446 United States, Washington University of Washington Seattle, Washington, United States, 98105 Washington State University Spokane, College of Pharmacy Spokane WA 99202 USA Spokane, Washington, United States, 99202 Collapse << |
| NCT01947153 | Healthy | Phase 1 | Completed | - | Russian Federation ... more >> 1288.21.1 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation Collapse << |
| NCT02719756 | Diabetes Mellitus ... more >> Hypoglycemic Episodes Collapse << | Phase 4 | Unknown | December 2016 | Russian Federation ... more >> Research Clinical Centre of the Russian Railways, JSC Not yet recruiting Moscow, Russian Federation, 125993 Contact: Alexander Ametov, prof., MD (499)1510951 ext 8107 alexander.ametov@gmail.ru Contact: Viktoria Blagova, PhD cand. (499)6130363 ext 8107 vika-blagova@mail.ru Collapse << |
| NCT01336673 | - | - | Terminated(The study was sched... more >>uled for completion February 2013. Because of difficulties encountered in completing the trial the study was terminated by sponsor June 2013.) Collapse << | - | Thailand ... more >> Faculty of Medicine Vajira Hospital Bangkok, Thailand Collapse << |
| NCT01034111 | - | - | Completed | - | - |
| NCT01947153 | - | - | Completed | - | - |
| NCT01965756 | Alzheimer's Disease ... more >> Vascular Dementia Dementia Memory Impairment Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania, Penn Memory Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01034111 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01965756 | - | - | Completed | - | - |
| NCT00327015 | Diabetes | Phase 3 | Completed | - | - |
| NCT02087826 | Genetics Meta... more >>bolism Type 2 Diabetes Collapse << | Not Applicable | Recruiting | January 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Varinderpal Kaur 617-643-5423 sigma2mgh@partners.org Principal Investigator: Jose C Florez, MD, PhD Sub-Investigator: Jennifer Todd, MD Sub-Investigator: Jamie L Garry, MS, RD Collapse << |
| NCT01505426 | Type II Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Busan, Korea, Republic of Daegu, Korea, Republic of Daejeon, Korea, Republic of Gwangju, Korea, Republic of Incheon, Korea, Republic of Jeonju, Korea, Republic of Seoul, Korea, Republic of Wonju, Korea, Republic of Taiwan Kaohsiung, Taiwan Taichung, Taiwan Tainan, Taiwan Taipei, Taiwan Collapse << |
| NCT00519480 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33169 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 Argentina GSK Investigational Site Buenos Aires, Argentina, 1425 Germany GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT02111967 | - | - | Completed | - | Switzerland ... more >> Pharmaceutical Care Research Group Basel, Switzerland, 4056 Collapse << |
| NCT02342925 | Healthy Volunteer | Phase 1 | Completed | - | United Kingdom ... more >> Leeds, United Kingdom, LS2 9LH Collapse << |
| NCT01666171 | - | - | Unknown | - | - |
| NCT00575588 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00327015 | - | - | Completed | - | - |
| NCT02231931 | Healthy | Phase 1 | Completed | - | Germany ... more >> 352.2082.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01677923 | Obesity | Phase 3 | Completed | - | Lithuania ... more >> Rasa Verkauskiene Kaunas, Eiveniu str. 2, Lithuania, LT50009 Collapse << |
| NCT01691846 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01512979 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT00575588 | - | - | Completed | - | - |
| NCT00383578 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Investigative Centers Nurnberg, Germany Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT03259490 | Healthy | Phase 1 | Completed | - | Germany ... more >> Humanpharmakologisches Zentrum Biberach Biberach, Germany, 88397 Collapse << |
| NCT01679574 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Assiut University Assiut, Egypt, 11111 Collapse << |
| NCT01117103 | - | - | Completed | - | Singapore ... more >> Kevin Tan Clinic For Diabetes, Thyroid & Hormones Pte Ltd Mount Elizabeth, Singapore Collapse << |
| NCT00922194 | - | - | Completed | - | - |
| NCT01512979 | - | - | Completed | - | - |
| NCT00922194 | Obesity Type ... more >>2 Diabetes Mellitus Collapse << | Not Applicable | Completed | - | India ... more >> Sircar Diabetes Clinic Lucknow, UP, India, 226006 Collapse << |
| NCT00151411 | Polycystic Ovary Syndrome | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Penn State Milton S. Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT00885378 | - | - | Completed | - | - |
| NCT03129113 | Hepatic Steatosis ... more >> HIV-1-infection Collapse << | Phase 2 Phase 3 | Recruiting | November 2020 | United Kingdom ... more >> Royal Free Hospital Recruiting London, United Kingdom, NW3 2QG Contact: PI St Thomas' Hospital Recruiting London, United Kingdom, SE1 9RT Contact: PI King's College Hospital Recruiting London, United Kingdom, SE5 9RJ Contact: PI UCL Mortimer Market Centre Recruiting London, United Kingdom, WC1E 6JB Contact: PI Collapse << |
| NCT00151411 | - | - | Completed | - | - |
| NCT01582243 | - | - | Completed | - | - |
| NCT03608358 | Type 2 Diabetes Mellitus | Phase 3 | Not yet recruiting | March 1, 2021 | China ... more >> Research Site Not yet recruiting Baotou, China, 014016 Research Site Not yet recruiting Beijing, China, 100039 Research Site Not yet recruiting Beijing, China, 100044 Research Site Not yet recruiting Beijing, China, 100101 Research Site Not yet recruiting Beijing, China, 100144 Research Site Not yet recruiting Beijing, China, 101199 Research Site Not yet recruiting Beijing, China, 101200 Research Site Not yet recruiting Changchun, China, 130021 Research Site Not yet recruiting Chenzhou, China Research Site Withdrawn Chongqing, China, 400014 Research Site Not yet recruiting Fuzhou, China, 350025 Research Site Withdrawn Guangzhou, China, 510000 Research Site Withdrawn Guangzhou, China, 510080 Research Site Not yet recruiting Guangzhou, China, 510515 Research Site Not yet recruiting Haerbin, China, 150001 Research Site Not yet recruiting Hefei, China, 230001 Research Site Not yet recruiting Hefei, China Research Site Not yet recruiting Hengyang, China, CN-421001 Research Site Not yet recruiting Hohhot, China, 010017 Research Site Not yet recruiting Huizhou, China, 516001 Research Site Not yet recruiting Jinan, China, 250001 Research Site Withdrawn Jinzhou, China, 121004 Research Site Withdrawn Nanchang, China, 330006 Research Site Not yet recruiting Nanjing, China, 210012 Research Site Not yet recruiting Nanjing, China, 210029 Research Site Not yet recruiting Nantong, China, 226001 Research Site Not yet recruiting Qingdao, China, 266003 Research Site Not yet recruiting Shanghai, China, 200240 Research Site Not yet recruiting Shantou, China, 515065 Research Site Not yet recruiting Shengyang, China, 110004 Research Site Not yet recruiting Tangshan, China Research Site Not yet recruiting Tianjin, China, 300211 Research Site Not yet recruiting Wuhan, China, 430033 Research Site Not yet recruiting Wuhan, China, CN-430022 Research Site Not yet recruiting Wulumuqi, China, 830054 Research Site Not yet recruiting Xian, China, 710061 Research Site Withdrawn Xining, China, 810001 Research Site Not yet recruiting Yancheng, China, 224005 Research Site Withdrawn Yinchuan, China, 750004 Research Site Not yet recruiting Yueyang, China, 414000 Research Site Not yet recruiting Zhuzhou, China, 412007 Thailand Research Site Not yet recruiting Bangkok, Thailand, 10400 Vietnam Research Site Not yet recruiting Ho Chi Minh, Vietnam, 10000 Research Site Not yet recruiting Ho Chi Minh, Vietnam Collapse << |
| NCT01582243 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Taiwan ... more >> Novartis Investigative Site Changhua, Taiwan, 500 Collapse << |
| NCT00618072 | - | - | Completed | - | - |
| NCT00618072 | Hyperinsulinemia ... more >> Insulin Resistance Obesity Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Albert Einstein College of Medicine Bronx, New York, United States, 10461 Collapse << |
| NCT03629054 | Healthy | Phase 1 | Completed | - | Germany ... more >> Humanpharmakologisches Zentrum Biberach Biberach, Germany, 88397 Collapse << |
| NCT02395237 | Healthy | Phase 1 | Unknown | May 2015 | Jordan ... more >> Arab Pharmaceutical industry Consulting/ Pharmaceutical Research Unit Amman, Jordan, 00962 Collapse << |
| NCT01490658 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Novo Nordisk Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00684528 | Diabetes Type 2 | Phase 3 | Unknown | December 2012 | Israel ... more >> Clalit Health services center Recruiting Tel Aviv, Israel, 62098 Contact: Zinger 972-5062-63652 Principal Investigator: Zinger Joel, MD Collapse << |
| NCT00889876 | Cardiovascular Diseases | Not Applicable | Unknown | December 2012 | Sweden ... more >> Karolinska Institutet, Karolinska University Hospital Huddinge Recruiting Stockholm, Sweden, 141 86 Contact: Maria Westerståhl, PhD +468 585 873 96 maria.westerstahl@ki.se Contact: Pernilla Hedvall, PhDstudent +468 585 873 96 pernilla.hedvall@ki.se Principal Investigator: Claude Marcus, Professor Collapse << |
| NCT03068065 | Non-Alcoholic Fatty Liver Dise... more >>ase Type2 Diabetes Collapse << | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT01171456 | Gestational Diabetes Mellitus | Not Applicable | Withdrawn(Insufficient funding... more >> for enrollment of patients. The invesitgators realized the study could not be performed.) Collapse << | - | United States, Utah ... more >> Intermountain Medical Center Murray, Utah, United States, 84107 Collapse << |
| NCT01076088 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02535975 | Mild Graves' Ophthalmopathy | Not Applicable | Unknown | June 2017 | Korea, Republic of ... more >> Endocrinology, Internal Medicine, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 03722 Contact: Eun Jig Lee, MD +82-2-2228-1983 ejlee432@yuhs.ac Collapse << |
| NCT01434186 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States United States, Florida Research Site Tallahassee, Florida, United States United States, Michigan Research Site Dearborn, Michigan, United States United States, Minnesota Research Site St. Paul, Minnesota, United States United States, New York Research Site Mineola, New York, United States United States, Ohio Research Site Cleveland, Ohio, United States United States, Tennessee Research Site Memphis, Tennessee, United States Belgium Research Site Namur, Belgium Canada, Alberta Research Site Calgary, Alberta, Canada India Research Site Bangalore, India Mexico Research Site Aguascalientes, Mexico Research Site Meridas, Mexico Research Site Monterrey, Mexico Research Site Veracruz, Mexico Taiwan Research Site Taichung, Taiwan United Kingdom Research Site Leicester, United Kingdom Collapse << |
| NCT02644577 | Bipolar Disorder | Phase 4 | Completed | - | China, Liaoning ... more >> Department of Psychiatry, the First Hospital of China Medical University Shenyang, Liaoning, China, 110001 Collapse << |
| NCT01076088 | - | - | Completed | - | - |
| NCT01107808 | Insulin Resistance ... more >> Insulin Sensitivity Obesity Vitamin D Deficiency Collapse << | Not Applicable | Withdrawn(Poor recruitment to ... more >>the study) Collapse << | March 2011 | - |
| NCT00924573 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT01434186 | - | - | Completed | - | - |
| NCT00501787 | Polycystic Ovary Syndrome | Phase 4 | Withdrawn | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT00683657 | - | - | Completed | - | - |
| NCT00399204 | Type 2 Diabetes | Phase 4 | Unknown | - | India ... more >> PGIMER Recruiting Chandigarh, India, 160012 Contact: Samir Malhotra, MD, DM samirmalhotra345@yahoo.com Sub-Investigator: Samir Malhotra, MD,DM Collapse << |
| NCT00683657 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03202563 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | October 30, 2018 | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 06351 Collapse << |
| NCT02091336 | Gestational Diabetes | Phase 4 | Unknown | December 2016 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Recruiting Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT00669864 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT01834274 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT00396227 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT01834274 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT00767351 | - | - | Completed | - | - |
| NCT02682121 | Prediabetes | Phase 1 | Completed | - | United States, Utah ... more >> University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00669864 | - | - | Completed | - | - |
| NCT00971243 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00971243 | - | - | Completed | - | - |
| NCT01001962 | Hypertension ... more >>Type II Diabetes Collapse << | Phase 4 | Not yet recruiting | January 2020 | Greece ... more >> Hypertension 24h ABPM center Papageorgiou Hospital Not yet recruiting Thessaloniki, Greece Contact: VASILEIOS KOTSIS, PROF Principal Investigator: VASILEIOS KOTSIS, PROF Collapse << |
| NCT03018665 | Diabetes Mellitus, Type 2 | Phase 4 | Not yet recruiting | - | China, Jilin ... more >> First Hospital of Jilin University Not yet recruiting Changchun, Jilin, China, 130021 Contact: Guixia Wang, PhD gwang168@jlu.edu.cn Collapse << |
| NCT01459094 | Healthy | Phase 1 | Completed | - | - |
| NCT00279045 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02574845 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT03115112 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02956044 | Type2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Indiana ... more >> Clinical Research Site Evansville, Indiana, United States, 47710 Collapse << |
| NCT00775164 | Obesity Insul... more >>in Resistance Collapse << | Phase 4 | Withdrawn(Inadequate enrollmen... more >>t) Collapse << | February 2011 | - |
| NCT02574845 | - | - | Completed | - | - |
| NCT01117584 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01729403 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | Germany ... more >> Neuss, Germany, 41460 Collapse << |
| NCT00451399 | Weight Gain | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT03317028 | T2DM With Inadequate Glycemic ... more >>Control Collapse << | Phase 2 | Recruiting | March 31, 2021 | United States, Florida ... more >> Clinical Research of South Florida Recruiting Coral Gables, Florida, United States, 33134 Contact: Rosen Jeffrey, MD 305-445-5637 jrosen@crsouthflorida.com Principal Investigator: Rosen Jeffrey, MD The Community Research of South Florida Recruiting Hialeah, Florida, United States, 33016 Contact: Carpio Jose, MD 305-441-6611 jcarpio@thecommunityresearch.com Principal Investigator: Carpio Jose, MD Taiwan Kaohsiung Veterans General Hospital Recruiting Kaohsiung, Taiwan, 81362 Contact: Chih-Hsun Chu, MD +886-7-3422-121 ext 8089 chchu@vghks.gov.tw Principal Investigator: Chih-Hsun Chu, MD Kaohsiung Chang Gung Memorial Hospital Recruiting Kaohsiung, Taiwan, 833 Contact: Jung-Fu Chen, MD +886-7-733-7166 0722cjf@cgmh.org.tw Principal Investigator: Jung-Fu Chen, MD Chung Shan Medical University Hospital Recruiting Taichung, Taiwan, 402 Contact: Chien-Ning Huang, MD +886-4-2473-9595 ext 11600 cshy049@csh.org.tw Principal Investigator: Chien-Ning Huang, MD China Medical University Hospital Recruiting Taichung, Taiwan, 404 Contact: Ching-Chu Chen, MD +886-4-2206-2121 ext 3489 d1918@mail.cmuh.org.tw Principal Investigator: Ching-Chu Chen, MD Taichung Veterans General Hospital Recruiting Taichung, Taiwan, 40705 Contact: I-Te Lee, MD, PhD +886-933-553-211 itlee@vghtc.gov.tw Principal Investigator: I-Te Lee, MD, PhD Chi-Mei Medical Center Recruiting Tainan, Taiwan, 710 Contact: Kai-Jen Tien, MD +886-6-2816-31 tkzzkimo@gmail.com Principal Investigator: Kai-Jen Tien, MD National Taiwan University Hospital Recruiting Taipei, Taiwan, 10048 Contact: Chih-Yuan Wang, MD, PhD +886-938-036-246 cyw1965@gmail.com Principal Investigator: Chih-Yuan Wang, MD, PhD Taipei Veterans general Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD +886-2-2875-7516 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Tri-Service General Hospital Recruiting Taipei, Taiwan, 114 Contact: Chang-Hsun Hsieh, MD +886-2-8792-7182 10324@yahoo.com.tw Principal Investigator: Chang-Hsun Hsieh, MD Chang Gung Memorial Hospital_Linkou Not yet recruiting Taoyuan, Taiwan, 333 Contact: Yu-Yao Huang, MD, PhD +886-975-368-137 yyh@cgmh.org.tw Principal Investigator: Yu-Yao Huang, MD, PhD Collapse << |
| NCT01518101 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Berlin, Germany, 10115 Novartis Investigative Site Berlin, Germany, 13055 Novartis Investigative Site Dortmund, Germany, 44137 Novartis Investigative Site Falkensee, Germany, 14612 Novartis Investigative Site Meissen, Germany, 01662 Novartis Investigative Site Neunkirchen, Germany, 57290 Novartis Investigative Site Saarlouis, Germany, 66740 Novartis Investigative Site Völlkingen, Germany, 66333 Collapse << |
| NCT02960659 | Type 2 Diabetes | Phase 1 | Recruiting | August 31, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00882882 | Type II Diabetes | Phase 1 | Completed | - | - |
| NCT01708902 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02821910 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01708902 | - | - | Completed | - | - |
| NCT02050074 | Type 2 Diabetes | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Division, Department of Medicine, Gentofte Hospital Hellerup, Copenhagen, Denmark, 2900 Collapse << |
| NCT00871936 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Burbank, California, United States, 91505 Los Angeles, California, United States, 90036 Los Angeles, California, United States, 90057 United States, Florida West Palm Beach, Florida, United States, 33401 United States, Illinois Addison, Illinois, United States, 60101 United States, Maine Auburn, Maine, United States, 04210 United States, North Carolina Charlotte, North Carolina, United States, 28211 Hickory, North Carolina, United States, 28602 Winston-Salem, North Carolina, United States, 27103 United States, South Carolina Greenville, South Carolina, United States, 29615 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT01318135 | - | - | Completed | - | - |
| NCT03349775 | Obesity | Early Phase 1 | Recruiting | September 1, 2024 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Elizabeth Liu, BS 617-724-3064 ELIU9@MGH.HARVARD.EDU Collapse << |
| NCT02080377 | Pregnancy Ges... more >>tational Diabetes Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Princess Royal Infirmary Glasgow, Lanarkshire, United Kingdom, G31 2ER Simpson Centre for Reproductive Health, Royal Infirmary Hospital, Edinburgh Edinburgh, Lothian, United Kingdom, EH16 4SA Western General Hospital Edinburgh, Lothian, United Kingdom, EH4 2XU St Johns Hospital Livingston, West Lothian, United Kingdom Queen Elizabeth Hospital Glasgow, United Kingdom Collapse << |
| NCT00501020 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01973231 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT03179254 | Diabetes Type 2 | Phase 4 | Completed | - | - |
| NCT01318135 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | - |
| NCT00934570 | Obesity Type ... more >>2 Diabetes Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Children's Hospital, London Health Sciences Centre London, Ontario, Canada, N6A 5W9 Collapse << |
| NCT00649454 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01973231 | - | - | Completed | - | - |
| NCT00648505 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01899430 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00134290 | Body Weight I... more >>nsulin Resistance Collapse << | Phase 4 | Completed | - | Belgium ... more >> University Hospital Ghent Ghent, Belgium, 9000 Collapse << |
| NCT01892254 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Copenhagen University Hospital Gentofte Hellerup, Select A State, Denmark, 2900 Collapse << |
| NCT02088918 | Healthy Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Chungnam University Hospital Daejeon, Korea, Republic of Collapse << |
| NCT02343926 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Russian Federation ... more >> Investigational Site 03 Vladimir, Vladimirskaya Oblast, Russian Federation, 600023 Collapse << |
| NCT00362323 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT02366377 | Type 2 Diabetes Mellitus | Phase 1 | Unknown | - | - |
| NCT01065766 | - | - | Completed | - | - |
| NCT01926769 | Previously Treated Advanced Co... more >>lorectal Cancer Collapse << | Phase 2 | Terminated(Slow accrual rate) | - | Korea, Republic of ... more >> Gachon University Gil Medical Center Incheon, Korea, Republic of Collapse << |
| NCT00941161 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico Collapse << |
| NCT01065766 | - | - | Completed | - | - |
| NCT03092635 | Breast Cancer | Phase 1 | Recruiting | December 1, 2019 | United States, South Carolina ... more >> Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Vistea Crawford 843-792-9321 hcc-clinical-trials@musc.edu Collapse << |
| NCT03141073 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 2019 | China ... more >> Hua Medicine Limited Recruiting Shanghai, China Contact: YI ZHANG, MD,PhD +86-021-58869997 yzhang@huamedicine.com Collapse << |
| NCT01787396 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> LG Life Sciences Seoul, Korea, Republic of, 110-062 Collapse << |
| NCT03194009 | Diabetes Mellitus, Type 2 ... more >> Prediabetic State Collapse << | Not Applicable | Recruiting | December 31, 2022 | Mexico ... more >> Centros de Salud. Secretaría de Salud Ciudad de México Recruiting Mexico City, Mexico, 06820 Contact: Alberto Gallardo, MD +52 (55) 50381700 ext 1597 albgallardo@yahoo.com.mx Contact: Carmen Vanegas, MD +52(55) 50381700 ext 1067 carmenvanegas_5@hotmail.com Collapse << |
| NCT01463774 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT02022254 | Diabetes Diab... more >>etes Mellitus, Type 2 Healthy Collapse << | Phase 1 | Completed | - | Germany ... more >> Novo Nordisk Investigational Site Berlin, Germany, 10117 Collapse << |
| NCT00911313 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals,OB/GYN department Mansoura, Dakahlia, Egypt, 35511 Collapse << |
| NCT00490178 | Diabetes Mellitus, Type 2 ... more >> Dyslipidemia Collapse << | Phase 3 | Completed | - | France ... more >> Site 3 Marseille, France Site 1 Nantes, France Site 2 Tours, France Collapse << |
| NCT02355145 | - | - | Completed | - | Bulgaria ... more >> Research Site Pleven, Bulgaria Research Site Sofia, Bulgaria Research Site Stara Zagora, Bulgaria Collapse << |
| NCT02188186 | Type 2 Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, 463-707 Collapse << |
| NCT02629627 | Metabolic Syndrome | Phase 2 | Completed | - | - |
| NCT03122041 | PCOS | Phase 4 | Completed | - | - |
| NCT02008682 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100071 Novo Nordisk Investigational Site Beijing, Beijing, China, 100700 Novo Nordisk Investigational Site Beijing, Beijing, China, 100853 China, Chongqing Novo Nordisk Investigational Site ChongQing, Chongqing, China, 404000 China, Hebei Novo Nordisk Investigational Site Cangzhou, Hebei, China, 061000 Novo Nordisk Investigational Site Hengshui, Hebei, China, 053000 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050000 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050051 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210011 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210012 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210029 Novo Nordisk Investigational Site Suzhou, Jiangsu, China, 215004 Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 Novo Nordisk Investigational Site Zhenjiang, Jiangsu, China, 212001 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Jilin Novo Nordisk Investigational Site Changchun, Jilin, China, 130041 China, Liaoning Novo Nordisk Investigational Site Dalian, Liaoning, China, 116033 Novo Nordisk Investigational Site Shenyang, Liaoning, China, 110021 China, Shandong Novo Nordisk Investigational Site Qingdao, Shandong, China, 266003 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200080 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200092 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200120 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200240 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 201199 China Novo Nordisk Investigational Site Beijing, China, 101200 Collapse << |
| NCT02926950 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | March 2019 | - |
| NCT02187250 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00657943 | Type 2 Diabetes ... more >> Atherosclerosis Arteriosclerosis Collapse << | Phase 4 | Completed | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark, 2100 Bispebjerg Hospital Copenhagen, Denmark, 2400 Frederiksberg Hospital Frederiksberg, Denmark, 2000 Gentofte Sygehus Gentofte, Denmark, 2820 Steno Diabetes Center Gentofte, Denmark, 2820 Herlev Hospital Herlev, Denmark, 2830 Hillerod Sygehus Hillerod, Denmark, 3400 Hvidovre Hospital Hvidovre, Denmark, 2650 Køge Sygehus Koge, Denmark, 4600 Collapse << |
| NCT01358994 | - | - | Completed | - | Sweden ... more >> Dept of Nephrology Malmö, Sweden, SE 20502 Collapse << |
| NCT00643851 | - | - | Completed | - | - |
| NCT01472367 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | October 1, 2019 | - |
| NCT00643851 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00834743 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Biovail Contract Research Toronto, Ontario, Canada, M1L4S4 Collapse << |
| NCT02008682 | - | - | Completed | - | - |
| NCT00834743 | - | - | Completed | - | - |
| NCT00834613 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Biovail Contract Research Toronto, Ontario, Canada, M1L4S4 Collapse << |
| NCT01296412 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00834613 | - | - | Completed | - | - |
| NCT01594697 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Italy ... more >> Catholic University of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT01296412 | - | - | Completed | - | - |
| NCT03076112 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 30, 2019 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Kyoung Min Kim, MD, PHD 82-31-787-7047 kyoungmin02@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT01832181 | - | - | Recruiting | July 2021 | Canada, Ontario ... more >> Dr. Denice Feig Recruiting Toronto, Ontario, Canada Collapse << |
| NCT01638988 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 3 | Withdrawn(Previous studies hav... more >>e been done regarding same condition) Collapse << | December 2015 | Canada, Quebec ... more >> Clinique Ovo Montréal, Quebec, Canada, H4P 2S4 Collapse << |
| NCT01672788 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.7.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01563120 | Gestational Diabetes Mellitus | Phase 4 | Completed | - | Israel ... more >> Dep. OB/GYN, Emek Medical Center Afula, Israel Collapse << |
| NCT01672788 | - | - | Completed | - | - |
| NCT00099866 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Novartis investigative Site Investigative Sites, Germany Collapse << |
| NCT01006590 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01650181 | Liver Disease | Phase 4 | Completed | - | Mexico ... more >> Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán Mexico City, Mexico, 14000 Collapse << |
| NCT00778622 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Beijing ... more >> Local Institution Beijing, Beijing, China, 100028 Local Institution Beijing, Beijing, China, 100034 Local Institution Beijing, Beijing, China, 100044 Local Institution Beijing, Beijing, China, 100088 Local Institution Beijing, Beijing, China, 100730 Local Institution Beijing, Beijing, China, 101100 Local Institution Beijing, Beijing, China, 200016 China, Guangdong Local Institution Guangdong Province, Guangdong, China, 510180 Local Institution Guangdong Province, Guangdong, China, 528000 Local Institution Guangdong, Guangdong, China, 510180 China, Shanghai Local Institution Shanghai, Shanghai, China, 200003 Local Institution Shanghai, Shanghai, China, 200092 Local Institution Shanghai, Shanghai, China, 201100 Local Institution Shanghai, Shanghai, China, 201200 Collapse << |
| NCT01760447 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | September 13, 2019 | - |
| NCT01006590 | - | - | Completed | - | - |
| NCT00283816 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, New York ... more >> University of Rochester Medical Center Rochester, New York, United States, 14642 Collapse << |
| NCT00283816 | - | - | Completed | - | - |
| NCT02516085 | Duchenne Muscular Dystrophy | Phase 1 | Completed | - | - |
| NCT00681460 | Diabetes, Gestational ... more >> Insulin Resistance Collapse << | Not Applicable | Completed | - | Poland ... more >> Div of Obstetrics and Women's Diseases, Dept of Obstetrics and Gynecology, K Marcinkowski Univ of Med Sciences Poznan, Poland, 60-535 Collapse << |
| NCT00778622 | - | - | Completed | - | - |
| NCT01581814 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Italy ... more >> Policlinico A. Gemelli Rome, Italy, 00168 Collapse << |
| NCT00682890 | Polycystic Ovary Syndrome ... more >> PCOS Insulin Sensitivity Collapse << | Phase 4 | Terminated(Lack of recruitment... more >>) Collapse << | - | United States, Virginia ... more >> General Clinical Research Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT00325910 | Diabetes Mellitus, Type 2 ... more >> Heart Failure, Congestive Collapse << | Phase 3 | Terminated(Insufficient study ... more >>participants) Collapse << | - | Canada, Alberta ... more >> Misericordia Hospital Edmonton, Alberta, Canada, T5R 4H5 University of Alberta Hospital Edmonton, Alberta, Canada, T6G 2B7 Collapse << |
| NCT01107717 | Diabetes | Not Applicable | Recruiting | December 2024 | United States, Texas ... more >> Texas Diabetes Institute Recruiting San Antonio, Texas, United States, 78229-3900 Contact: Muhammad Abdul-Ghani, MD, PhD 210-567-2391 ABDULGHANI@UTHSCSA.EDU Contact: Curtiss Puckett, PA (210) 358 7200 Curtiss.Puckett@uhs-sa.com Principal Investigator: Ralph DeFronzo, MD Collapse << |
| NCT02252081 | Chronic Kidney Disease ... more >> Cardiovascular Disease Metabolic Syndrome Collapse << | Phase 2 | Recruiting | December 31, 2019 | United States, Tennessee ... more >> Tennessee Valley Healthcare System Nashville Campus, Nashville, TN Recruiting Nashville, Tennessee, United States, 37212-2637 Contact: Adriana M Hung, MD MPH 615-873-6731 Adriana.Hung@va.gov Contact: Cindy A Booker (615) 343-5828 cindy.a.booker@vanderbilt.edu Principal Investigator: Adriana M Hung, MD MPH Collapse << |
| NCT00682890 | - | - | Terminated(Lack of recruitment... more >>) Collapse << | - | - |
| NCT00707031 | - | - | Completed | - | - |
| NCT01341717 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Jothydev's Diabetes and Research Center Thiruvananthapuram, Kerala, India, 695032 Collapse << |
| NCT02221414 | Healthy | Phase 1 | Completed | - | - |
| NCT01514149 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 2 | Terminated(Business decision) | - | - |
| NCT00707031 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00745433 | - | - | Completed | - | Belgium ... more >> Brussels, Belgium, 1070 Collapse << |
| NCT02221401 | Healthy | Phase 1 | Completed | - | - |
| NCT01514149 | - | - | Terminated(Business decision) | - | - |
| NCT02220647 | Healthy | Phase 1 | Completed | - | - |
| NCT00400231 | Metabolic Syndrome x | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00015691 | HIV Infections ... more >> Lipodystrophy Hyperinsulinemia Collapse << | Not Applicable | Completed | - | - |
| NCT01106625 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01106625 | - | - | Completed | - | - |
| NCT01115140 | Pregnancy Pol... more >>ycystic Ovary Syndrome Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT00755287 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01755156 | - | - | Completed | - | - |
| NCT01755156 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00361868 | Dyslipidemia ... more >>Glucose Metabolism Disorder Collapse << | Phase 3 | Terminated(The study was disco... more >>ntinued prematurely at the end of March 2007 due to slow enrolment.) Collapse << | - | - |
| NCT00970593 | Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site Miami Gardens, Florida, United States, 33169 Collapse << |
| NCT02243176 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China ... more >> Research Site Shanghai, China Collapse << |
| NCT02100475 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02243176 | - | - | Completed | - | - |
| NCT02100475 | - | - | Completed | - | - |
| NCT00413179 | Polycystic Ovary Syndrome ... more >> Anovulation Oligoovulation Infertility Hyperandrogenism Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Wiford Hall Medical Center Lackland AFB, Texas, United States, 78236 Collapse << |
| NCT03169959 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT01232036 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT03363815 | Heathy Volunteer | Phase 1 | Not yet recruiting | February 23, 2018 | United States, Florida ... more >> Covance Clinical Research Unit Not yet recruiting Daytona Beach, Florida, United States, 32117 Collapse << |
| NCT00708578 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Collapse << |
| NCT02946632 | Diabetes Mellitus, Type II | Phase 3 | Not yet recruiting | December 2019 | Korea, Republic of ... more >> Korea University Anam Hospital Seoul, Korea, Republic of Collapse << |
| NCT01043965 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | - |
| NCT01763346 | Prediabetes T... more >>ype 2 Diabetes Obesity Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of Southern California Los Angeles, California, United States, 91011 Collapse << |
| NCT00607867 | Diabetes Mellitus ... more >> Diet Collapse << | Not Applicable | Terminated(The funding ended b... more >>efore the study was completed.) Collapse << | - | United States, Minnesota ... more >> VA Medical Center, Minneapolis Minneapolis, Minnesota, United States, 55417 Collapse << |
| NCT00607867 | - | - | Terminated(The funding ended b... more >>efore the study was completed.) Collapse << | - | - |
| NCT00638716 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Quebec ... more >> ConjuChem Biotechnologies Inc. Montreal, Quebec, Canada, H2X 3Y8 Collapse << |
| NCT00638716 | - | - | Completed | - | - |
| NCT00699036 | Nonalcoholic Steatohepatitis ... more >> Nonalcoholic Fatty Liver Disease Collapse << | Phase 2 | Unknown | August 2009 | United States, Texas ... more >> Brooke Army Medical Center Recruiting San Antonio, Texas, United States, 78234 Contact: Dawn M Torres, MD 210-916-5649 Principal Investigator: Dawn M Torres, MD Sub-Investigator: Stephen A Harrison, MD Collapse << |
| NCT02532855 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00941148 | Type 2 Diabetic Patients ... more >> Insufficient Metabolic Control OAD Treatment Collapse << | Phase 4 | Completed | - | Germany ... more >> ikfe GmbH, Clinic Department Mainz, RLP, Germany, 55116 Collapse << |
| NCT02192424 | Type 2 Diabetes | Phase 3 | Recruiting | September 2020 | Canada, Ontario ... more >> Mount Sinai Hospital Recruiting Toronto, Ontario, Canada, M5G1X5 Principal Investigator: Ravi Retnakaran, MD Collapse << |
| NCT00428311 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Spain ... more >> Department of Endocrinology, Hospital Ramón y Cajal Madrid, Spain, E-28034 Collapse << |
| NCT02565368 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT01889706 | - | - | Unknown | February 2014 | Poland ... more >> Department of Internal Medicine and Diabetology, Raszeja Hospital Recruiting Poznan, Great Poland, Poland, 60-834 Contact: Agnieszka Zawada, MD aga.zawada@gmail.com Collapse << |
| NCT02419612 | Diabetes | Phase 3 | Active, not recruiting | August 15, 2019 | - |
| NCT01159600 | - | - | Completed | - | - |
| NCT01046500 | Metabolic Syndrome | Phase 2 | Unknown | December 2014 | Italy ... more >> University Hospital Recruiting Messina, Italy Contact: Rosario D'Anna, associate professor +39 090 2217324 rosariodanna@tin.it Principal Investigator: Rosario D'Anna, associate professor Collapse << |
| NCT02419612 | - | - | Active, not recruiting | - | - |
| NCT02532855 | - | - | Completed | - | - |
| NCT00763451 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Chile Sanofi-Aventis Administrative Office Santiago, Chile Colombia Sanofi-Aventis Administrative Office Santafe de Bogota, Colombia Estonia Sanofi-Aventis Administrative Office Tallinn, Estonia Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Lithuania Sanofi-Aventis Administrative Office Vilnius, Lithuania Malaysia Sanofi-Aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Slovakia Sanofi-Aventis Administrative Office Brastislava, Slovakia Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT01159600 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02205528 | Diabetes Diab... more >>etes Mellitus Type 2 Collapse << | Phase 2 | Completed | - | United States, California ... more >> Novo Nordisk Investigational Site Chino, California, United States, 91710 Novo Nordisk Investigational Site Hawaiian Gardens, California, United States, 90716 Novo Nordisk Investigational Site Los Angeles, California, United States, 90057 Novo Nordisk Investigational Site Spring Valley, California, United States, 91978 United States, Florida Novo Nordisk Investigational Site Hialeah, Florida, United States, 33012 Novo Nordisk Investigational Site Port Orange, Florida, United States, 32127 Novo Nordisk Investigational Site Sanford, Florida, United States, 32771 United States, Illinois Novo Nordisk Investigational Site Chicago, Illinois, United States, 60607 Novo Nordisk Investigational Site Evanston, Illinois, United States, 60201 United States, Maryland Novo Nordisk Investigational Site Oxon Hill, Maryland, United States, 20745 United States, New Jersey Novo Nordisk Investigational Site Berlin, New Jersey, United States, 08009 United States, New Mexico Novo Nordisk Investigational Site Albuquerque, New Mexico, United States, 87102 United States, North Carolina Novo Nordisk Investigational Site Greensboro, North Carolina, United States, 27408 United States, Ohio Novo Nordisk Investigational Site Cincinnati, Ohio, United States, 45227 United States, Oklahoma Novo Nordisk Investigational Site Tulsa, Oklahoma, United States, 74136 United States, Oregon Novo Nordisk Investigational Site Eugene, Oregon, United States, 97404 United States, Tennessee Novo Nordisk Investigational Site Knoxville, Tennessee, United States, 37920 United States, Texas Novo Nordisk Investigational Site Corpus Christi, Texas, United States, 78404 Novo Nordisk Investigational Site Houston, Texas, United States, 77074 United States, Virginia Novo Nordisk Investigational Site Manassas, Virginia, United States, 20110 Collapse << |
| NCT01779375 | Prediabetes T... more >>ype 2 Diabetes Collapse << | Phase 3 | Completed | - | United States, Colorado ... more >> Childrens Hospital Colorado Denver, Colorado, United States, 80045 United States, Connecticut Yale School of Medicine Pediatric Obesity and Type 2 Diabetes Clinic New Haven, Connecticut, United States, 06511 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT00763451 | - | - | Completed | - | - |
| NCT00162240 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00362765 | Type 2 Diabetes ... more >> Dyslipidemia Collapse << | Phase 2 Phase 3 | Terminated(The study was prema... more >>turely terminated, due to difficulties in the recruitment of T2DM patients who are not under statin therapy at inclusion.) Collapse << | - | Finland ... more >> Site 2 Turku, Finland Ireland Site 3 Dublin, Ireland Italy Site 1 Pisa, Italy Collapse << |
| NCT00491725 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT00885352 | - | - | Completed | - | - |
| NCT00995345 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT01779375 | - | - | Completed | - | - |
| NCT02719132 | Diabetes Mellitus Type 2 | Phase 4 | Withdrawn | November 2017 | - |
| NCT00282945 | Type 2 Diabetes Mellitus | Not Applicable | Terminated(See statement in De... more >>tailed Description.) Collapse << | - | Israel ... more >> Pfizer Investigational Site Holon, Israel, 58100 Pfizer Investigational Site Jerusalem, Israel, 91120 Collapse << |
| NCT00885352 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00995345 | - | - | Completed | - | - |
| NCT01907854 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT00399711 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03377335 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | January 2019 | Italy ... more >> University Hospital of Palermo Palermo, Italy, 90127 Collapse << |
| NCT02393573 | Pre-Diabetes ... more >>Major Lung or Abdominal Surgery Insulin Resistance Collapse << | Not Applicable | Terminated(Poor recruitment) | - | Canada, Quebec ... more >> Montreal General Hospital Montreal, Quebec, Canada, H3G 1A4 Collapse << |
| NCT00672464 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Withdrawn(lack of funding) | February 2010 | United States, North Carolina ... more >> John Umstead Hospital Butner, North Carolina, United States, 27509 Duke University Medical Center Durham, North Carolina, United States, 27705 Collapse << |
| NCT01437800 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Investigacion Farmacologica Y Biofarmaceutica, S.A. de C.V. México, Mexico Collapse << |
| NCT01907854 | - | - | Completed | - | - |
| NCT01596504 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Investigational Site Number 276008 Berlin, Germany, 10117 Investigational Site Number 276006 Berlin, Germany, 14050 Investigational Site Number 276004 Kiel, Germany, 24105 Investigational Site Number 276002 Mainz, Germany, 55116 Investigational Site Number 276005 Mönchengladbach, Germany, 41061 Investigational Site Number 276007 München, Germany, 80636 Investigational Site Number 276003 Neu-Ulm, Germany, 89231 Investigational Site Number 276001 Neuss, Germany, 41460 Collapse << |
| NCT03196362 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | December 31, 2018 | China, Guangdong ... more >> endocrinology department of the first affiliated hospital of Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510080 Contact: Yanbing Li, MD,PhD 8602087334331 easd04lyb@126.com Principal Investigator: Yanbing Li, MD,PhD Collapse << |
| NCT01519674 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01596504 | - | - | Completed | - | - |
| NCT01758380 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT00344851 | Polycystic Ovary Syndrome | Phase 2 | Completed | - | United States, Louisiana ... more >> Facility: Metabolic Center of Louisiana Research Foundation Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT00975052 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01519674 | - | - | Completed | - | - |
| NCT01324180 | Acute Lymphoblastic Leukemia | Phase 1 | Completed | - | United States, Florida ... more >> Holtz Children's Hospital University of Miami Miller School of Medicine Miami, Florida, United States, 33136 Arnold Palmer Hospital for Children Orlando, Florida, United States, 32806 All Children's Hospital Saint Petersburg, Florida, United States, 33701 United States, New York Montefiore Medical Center, The Children's Hospital at Montefiore The Bronx, New York, United States, 10467 United States, Ohio Nationwide Children's Hospital Columbus, Ohio, United States, 43205 Collapse << |
| NCT01582230 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China, Beijing ... more >> Novartis Investigative Site Beijing, Beijing, China, 100730 China, Hunan Novartis Investigative Site Changsha City, Hunan, China, 410011 Novartis Investigative Site Changsha, Hunan, China, 410003 China, Jiangsu Novartis Investigative Site Nanjing, Jiangsu, China, 210006 Novartis Investigative Site Wu Xi, Jiangsu, China, 214023 China, Jiangxi Novartis Investigative Site Nanchang, Jiangxi, China, 330006 China, Liaoning Novartis Investigative Site Shenyang, Liaoning, China, 110003 China, Shandong Novartis Investigative Site Jinan, Shandong, China, 250031 China, Shanxi Novartis Investigative Site Xi'an, Shanxi, China, 710032 China, Sichuan Novartis Investigative Site Chengdu, Sichuan, China, 610072 China Novartis Investigative Site Beijing, China Novartis Investigative Site Shanghai, China, 200025 Novartis Investigative Site Tianjin, China, 300052 Philippines Novartis Investigative Site Metro Manila, Philippines, 1500 Novartis Investigative Site Pasay City, Philippines, 1300 Novartis Investigative Site Quezon City, Philippines, 1102 Singapore Novartis Investigative Site Singapore, Singapore, 169608 Novartis Investigative Site Singapore, Singapore, 768825 Thailand Novartis Investigative Site Bangkok, Thailand, 10330 Novartis Investigative Site Bangkok, Thailand, 10400 Novartis Investigative Site Khon Kaen, Thailand, 40002 Collapse << |
| NCT01381900 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | China ... more >> Baotou, China Beijing, China Changchun, China Changsha, China Chengdu, China Chongqing, China Guangzhou, China Hangzhou, China Harbin, China Nanchang, China Nanjing, China Nanning, China Shanghai, China Shenyang, China Shiyan, China Siping, China Su Zhou, China Tianjin, China Wuxi, China Xi'An, China Malaysia Kota Bharu, Malaysia Pulau Pinang, Malaysia Selangor, Malaysia Vietnam Hanoi, Vietnam Ho Chi Minh, Vietnam Collapse << |
| NCT02184832 | Metformin Response ... more >> Fasting Glucose Tryptophan Concentration Metformin Concentration Diet Tolerability Collapse << | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01095666 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02341703 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Early Phase 1 | Completed | - | Egypt ... more >> faculty of medicine, Cairo University Cairo, Egypt, 002 Collapse << |
| NCT01381900 | - | - | Completed | - | - |
| NCT01217892 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01217892 | - | - | Completed | - | - |
| NCT01095666 | - | - | Completed | - | - |
| NCT00353691 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00121667 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02947620 | Type II Diabetes ... more >> Dyslipidemia Collapse << | Phase 3 | Unknown | - | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Daewoong Clinical Collapse << |
| NCT02630706 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01340664 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00121667 | - | - | Completed | - | - |
| NCT02280486 | Type 2 Diabetes | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT01340664 | - | - | Completed | - | - |
| NCT02630706 | - | - | Completed | - | - |
| NCT02831361 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01720290 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100191 China Novo Nordisk Investigational Site Beijing, China, 100029 Novo Nordisk Investigational Site Shanghai, China Collapse << |
| NCT01497522 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Kitakyushu, Fukuoka, Japan, 800-0296 Novartis Investigative Site Kurume, Fukuoka, Japan, 830-8543 Novartis Investigative Site Ohkawa-city, Fukuoka, Japan, 831-0016 Novartis Investigative Site Kawasaki-city, Kanagawa, Japan, 210-0014 Novartis Investigative Site Yokohama, Kanagawa, Japan, 221-0802 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Takatsuki, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-0031 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Kiyose, Tokyo, Japan, 204-0021 Novartis Investigative Site Minato-ku, Tokyo, Japan, 105-7390 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Novartis Investigative Site Fukuoka, Japan, 810-0001 Novartis Investigative Site Fukuoka, Japan, 819-0168 Novartis Investigative Site Kyoto, Japan, 607-8062 Collapse << |
| NCT00792935 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00792935 | - | - | Completed | - | - |
| NCT01719003 | Diabetes Mellitus, Type 2 ... more >> Hyperglycemia Collapse << | Phase 3 | Completed | - | - |
| NCT01422876 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02409238 | Mild Cognitive Impairment ... more >> Insulin Resistance Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Unknown | - | Singapore ... more >> SingHealth Polyclinics - Marine Parade Polyclinic Recruiting Singapore, Singapore, 440080 Contact: WEE KIEN HAN ANDREW, MCI (65) 63450049 andrew.wee@singhealth.com.sg Contact: NG CHIAT ENG (65)90718977 ng.chiat.eng@singhealth.com.sg Collapse << |
| NCT01422876 | - | - | Completed | - | - |
| NCT02681094 | Type 2 Diabetes Mellitus ... more >> Inadequate Glycaemic Control Collapse << | Phase 3 | Completed | - | - |
| NCT01719003 | - | - | Completed | - | - |
| NCT02681094 | - | - | Completed | - | - |
| NCT00348725 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 3 | Completed | - | France ... more >> Site 24 Anzin, France Site 23 Bachant, France Site 21 Baune, France Site 32 Bersee, France Site 34 Briollay, France Site 27 Denain, France Site 28 Denain, France Site 5 Le Temple de Bretagne, France Site 29 Nantes, France Site 2 Nantes, France Site 8 Nantes, France Site 9 Nantes, France Site 1 Nort sur Erdre, France Site 13 Parcay les Pins, France Site 26 Quarouble, France Site 7 Saint Aignan le Grand, France Site 4 Saint Herblain, France Site 30 Saint-Amand, France Site 11 Sautron, France Site 17 Segre, France Site 10 St Etienne de Montluc, France Site 33 Thiant, France Site 12 Thouars, France Site 25 Vieux Conde, France Site 20 Vihiers, France Collapse << |
| NCT00814294 | Diabetes | Phase 2 Phase 3 | Unknown | October 2009 | - |
| NCT01399645 | Nonalcoholic Fatty Liver Disea... more >>se Nonalcoholic Steatohepatitis Type 2 Diabetes Collapse << | Phase 2 | Completed | - | Canada, Quebec ... more >> Centre hospitalier de l'Université de Montréal Montréal, Quebec, Canada, H2W 1T7 Collapse << |
| NCT00349128 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT00349635 | Obesity | Phase 2 | Completed | - | Finland ... more >> Site 1 Helsinki, Finland Site 3 Kuopio, Finland Site 2 Oulu, Finland Collapse << |
| NCT01465152 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Spain ... more >> Novo Nordisk Investigational Site Barcelona, Spain, 08020 Novo Nordisk Investigational Site Benamargosa, Spain, 29718 Novo Nordisk Investigational Site Burlada, Spain, 31600 Novo Nordisk Investigational Site Camas, Spain, 41900 Novo Nordisk Investigational Site Fuenlabrada, Spain, 28942 Novo Nordisk Investigational Site Gijón, Spain, 33212 Novo Nordisk Investigational Site Granada, Spain, 18012 Novo Nordisk Investigational Site Lugo, Spain, 27004 Novo Nordisk Investigational Site Madrid, Spain, 28021 Novo Nordisk Investigational Site Madrid, Spain, 28030 Novo Nordisk Investigational Site Madrid, Spain, 28035 Novo Nordisk Investigational Site Portugalete, Spain, 48920 Novo Nordisk Investigational Site San Adria del Besos, Spain, 08930 Novo Nordisk Investigational Site Santander, Spain, 39009 Novo Nordisk Investigational Site Santander, Spain, 39011 Novo Nordisk Investigational Site Tegueste, Spain, 38280 Novo Nordisk Investigational Site Valencia, Spain, 46021 Novo Nordisk Investigational Site Zaragoza, Spain, 50007 Collapse << |
| NCT01389778 | Polycystic Ovary Syndrome | Not Applicable | Active, not recruiting | October 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01850615 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03610412 | Diabetes Mellitus, Type 2 | Phase 2 | Not yet recruiting | December 2018 | - |
| NCT01850615 | - | - | Completed | - | - |
| NCT00266253 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT03514108 | Heart Failure ... more >> Diabetes Collapse << | Phase 4 | Recruiting | September 1, 2023 | Denmark ... more >> Sygehus Sønderjylland, Aabenraa Not yet recruiting Aabenraa, Denmark Contact: Bartlomiej Jonczy Aalborg University Hospital Not yet recruiting Aalborg, Denmark Contact: Søren Vraa Aarhus University Hospital Recruiting Aarhus, Denmark Contact: Henrik Wiggers +45 40136627 henrikwiggers@dadlnet.dk Sub-Investigator: Anders Jorsal Amager Hospital Not yet recruiting Copenhagen, Denmark Contact: Margrethe Ege Olsen Bispebjerg Hospital Not yet recruiting Copenhagen, Denmark Contact: Olav Wendelbo Nielsen Gentofte Hospital Recruiting Copenhagen, Denmark Contact: Morten Schou Glostrup Hospital Not yet recruiting Copenhagen, Denmark Contact: Morten Petersen Herlev Hospital Recruiting Copenhagen, Denmark Contact: Morten Schou Hvidovre Hospital Not yet recruiting Copenhagen, Denmark Contact: Jens Hove Rigshospitalet Recruiting Copenhagen, Denmark Contact: Lars Køber Principal Investigator: Finn Gustafsson Sydvestjysk Sygehus, Esbjerg Not yet recruiting Esbjerg, Denmark Contact: Kirsten Vilain Mikkelsen Herning Hospital Not yet recruiting Herning, Denmark Contact: Morten Bøttcher Regionshospital Nordjylland, Hjørring Not yet recruiting Hjørring, Denmark Contact: Gitte Nielsen Holbæk Hospital Not yet recruiting Holbæk, Denmark Contact: Ilan Raymond Horsens Hospital Not yet recruiting Horsens, Denmark Contact: Karen Kaae Dodt Kolding Hospital Not yet recruiting Kolding, Denmark Contact: Monica Petronela Poenaru Nykøbing Falster Hospital Not yet recruiting Nykøbing Falster, Denmark Contact: Lisbeth Tingsted Odense University Hospital Not yet recruiting Odense, Denmark Contact: Mikael Kjær Poulsen Sjællands Universitetshospital, Roskilde Not yet recruiting Roskilde, Denmark Contact: Nina Pernille Gardshodn Buch Silkeborg Hospital Not yet recruiting Silkeborg, Denmark Contact: Anne Sejr Knudsen Slagelse Sygehus Not yet recruiting Slagelse, Denmark Contact: Henrik Ryde Vejle Hospital Not yet recruiting Vejle, Denmark Contact: Vibeke Brogaard Viborg Hospital Not yet recruiting Viborg, Denmark Contact: Anne Pauline Schrøder Collapse << |
| NCT00813995 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00118950 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Denmark ... more >> Steno Diabetes Center Gentofte, Denmark, 2820 Collapse << |
| NCT01430351 | Brain Cancer | Phase 1 | Active, not recruiting | September 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01822548 | Type 2 Diabetes | Phase 3 | Completed | - | Italy ... more >> Azienda Opedaliera-Universitaria Parma, Italy, 43126 Azienda Ospedaliera-Universitaria Parma, Italy, 43126 Collapse << |
| NCT01822548 | - | - | Completed | - | - |
| NCT00813995 | - | - | Completed | - | - |
| NCT01490918 | Type-II Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> MedicalExcellence Seoul, Korea, Republic of, 137-701 Collapse << |
| NCT01757587 | Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> CH Sud Francilien Evry, France, 91000 Collapse << |
| NCT00396513 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Poland ... more >> Division of Infertility and Reproductive Endocrinology, Department of Gynecology and Obstetrics Recruiting Poznan, Poland, 60-184 Collapse << |
| NCT00815399 | Type 2 Diabetes | Phase 4 | Completed | - | Italy ... more >> Department of Geriatrics and Metabolic Diseases Naples, Italy, 80138 Collapse << |
| NCT00856986 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02526524 | - | - | Completed | - | - |
| NCT02875821 | Type 2 Diabetes With Non-alcoh... more >>olic Fatty Liver (NAFLD) Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes Center Seoul, Korea, Republic of, 03722 Collapse << |
| NCT01709305 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT00856986 | - | - | Completed | - | - |
| NCT02526524 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01709305 | - | - | Completed | - | - |
| NCT00798161 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT03566225 | Pioglitazone | Early Phase 1 | Not yet recruiting | December 25, 2018 | - |
| NCT01099137 | Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Korea, Republic of Collapse << |
| NCT01106690 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00251953 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT03489668 | PCOS | Not Applicable | Recruiting | March 31, 2020 | Poland ... more >> Division of Infertility and Reproductive Endocrinology, Department of Gynecology and Obsterics Recruiting Poznan, Poland Contact: Beata Banaszewska, MD PhD bbeata48@gmail.com Contact: Mateusz Trzciński, MD mtrzcinski13@gmail.com Sub-Investigator: Antoni J Duleba, Prof Sub-Investigator: Leszek A Pawelczyk, Prof Sub-Investigator: Robert Z Spaczyński, Prof Sub-Investigator: Piotr Piekarski, MD Sub-Investigator: Jeff Chang, Prof Collapse << |
| NCT00798161 | - | - | Completed | - | - |
| NCT01106690 | - | - | Completed | - | - |
| NCT00169624 | Coronary and Peripheral Endoth... more >>elial Dysfunction Collapse << | Phase 3 | Terminated | - | France ... more >> Laurent SEBBAG Lyon, France, 69677 Collapse << |
| NCT00502229 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Phase 4 | Withdrawn | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT01972724 | Type II Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Chagwon, Korea, Republic of Daegu, Korea, Republic of Daejeon, Korea, Republic of Gwangju, Korea, Republic of Gyeonggi-do, Korea, Republic of Jeonju, Korea, Republic of Seoul, Korea, Republic of Ulsan, Korea, Republic of Wonju, Korea, Republic of Collapse << |
| NCT01972724 | - | - | Completed | - | - |
| NCT00343395 | Coronary Artery Disease | Phase 4 | Terminated(FDA concerns regard... more >>ing Avandamet) Collapse << | - | United States, Utah ... more >> Intermountain Medical Center Murray, Utah, United States, 84157 Collapse << |
| NCT01909141 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Early Phase 1 | Completed | - | Egypt ... more >> Cairo University Cairo, Egypt Collapse << |
| NCT01909141 | - | - | Completed | - | - |
| NCT03689374 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | November 13, 2020 | - |
| NCT00519142 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02069379 | Depression In... more >>sulin Resistance Metabolic Syndrome Collapse << | Phase 4 | Completed | - | United States, Michigan ... more >> University of Michigan Medical School Ann Arbor, Michigan, United States, 48103 Collapse << |
| NCT00698789 | Type 2 Diabetes | Phase 2 | Terminated(Study ended after d... more >>iabetes development plan review. .) Collapse << | - | United States, Arizona ... more >> Phoenix, Arizona, United States, 85029 Tucson, Arizona, United States, 85705 United States, California Chula Vista, California, United States, 91911 Los Angeles, California, United States Mission Viejo, California, United States, 92691 Orange, California, United States, 92869 Paramount, California, United States, 90723 San Diego, California, United States, 92117 Santa Ana, California, United States, 92701 Tustin, California, United States, 92780 United States, Colorado Colorado Springs, Colorado, United States, 80909 United States, Florida St. Petersburg, Florida, United States, 33709 United States, Kansas Shawnee Mission, Kansas, United States, 66216 United States, Louisiana Shreveport, Louisiana, United States, 71101 United States, Michigan Dearborn, Michigan, United States, 48126 United States, Missouri St. Louis, Missouri, United States, 63110 United States, Nebraska Omaha, Nebraska, United States, 68154 United States, New Jersey Elizabeth, New Jersey, United States, 07202 United States, North Carolina Charlotte, North Carolina, United States, 28211 United States, Oklahoma Oklahoma City, Oklahoma, United States, 73103 United States, Rhode Island East Providence, Rhode Island, United States, 02914 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT00976937 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00532935 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00370617 | Chronic Hepatitis C ... more >> Insulin Resistance Collapse << | Phase 4 | Unknown | - | Italy ... more >> Division of Gastroenterology, University of Torino, Ospedale San Giovanni Battista Recruiting Torino, Italy, 10126 Contact: Mario Rizzetto, MD +39-011-6336397 mrizzetto@molinette.piemonte.it Contact: Elisabetta Bugianesi, MD +39-011-6336397 ebugianesi@yahoo.it Sub-Investigator: Elisabetta Bugianesi, MD Collapse << |
| NCT02069379 | - | - | Completed | - | - |
| NCT02150824 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Germany ... more >> 1307.4.49003 Boehringer Ingelheim Investigational Site Berlin, Germany 1307.4.49002 Boehringer Ingelheim Investigational Site Mainz, Germany 1307.4.49001 Boehringer Ingelheim Investigational Site Neuss, Germany Collapse << |
| NCT01529541 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Hospital Seoul, Korea, Republic of Collapse << |
| NCT00976937 | - | - | Completed | - | - |
| NCT00532935 | - | - | Completed | - | - |
| NCT01471275 | Type 2 Diabetes Mellitus ;Obes... more >>ity,High Triglycerides;TCM Collapse << | Not Applicable | Unknown | December 2013 | China, Beijing ... more >> Guang'anmen Hospital of China Academy ofChinese Medical Sciences Recruiting Beijing, Beijing, China Contact: Zhou Qiang, doctor 15101016416 15101016416@126.com Principal Investigator: Tong Xianlin, professor Sub-Investigator: Lian Fengmei, professor Sub-Investigator: Zhou Qiang, doctor China, Hebei Baoding Hospital of TCM Recruiting Baoding, Hebei, China Contact: Li Liping lilipingm@163.com Principal Investigator: Li Liping, professor China, Jilin Affiliated Hospital of Changchun University of TCM Recruiting Changchun, Jilin, China Contact: Piao Chunli, professor piaochunli981027@yahoo.com.cn Principal Investigator: Piao Chunli, professor Sub-Investigator: Wang Yanyan China, Qinghai Qinghai Provence Hospital of TCM Recruiting Xining, Qinghai, China Contact: Pu Weirong puweirong123@163.com Principal Investigator: Ba Zhuoma, professor Sub-Investigator: Pu Weirong China, Tianjin Tianjin Dagang Hospital Recruiting Dagang, Tianjin, China Contact: Jin Chuan 58023442@qq.com Principal Investigator: Guo Hailong Sub-Investigator: Jin Chuan Tianjin University of Traditional Chinese Medicine, First Affiliated Hospital Recruiting Tianjin, Tianjin, China Contact: Wu Shentao, professor 13752262891 wushentao@yahoo.com.cn Principal Investigator: Wu Shentao, professor Sub-Investigator: Wang Bin, doctor China, Yichang Yichang City Yiling Hospital Recruiting Yichang, Yichang, China Contact: Yang Xingzhong clcy0401@163.com Principal Investigator: Yang Xingzhong China, Zhejiang Hangzhou Hospital of TCM Recruiting Hangzhou, Zhejiang, China Contact: Hong Yuzhi, professor hyzhcy@yahoo.cn Principal Investigator: Hong Yuzhi, professor Sub-Investigator: Wei Yan Collapse << |
| NCT03136484 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00614120 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00095030 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00754988 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00614120 | - | - | Completed | - | - |
| NCT00700817 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00319293 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | - |
| NCT01128621 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT00700817 | - | - | Completed | - | - |
| NCT02129985 | Type 2 Diabetes | Phase 4 | Unknown | September 2015 | China, Shanghai ... more >> Xiaolong Zhao Recruiting Jingan, Shanghai, China, 20041 Contact: xiaolong zhao, MD. 86-18918067241 xiaolongzhao@163.com Principal Investigator: xiaolong zhao, MD. Collapse << |
| NCT01895569 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | June 2016 | Italy ... more >> IRCCS Policlinico S. Matteo Foundation Recruiting Pavia, Italy, 27100 Contact: Giuseppe Derosa, MD, PhD giuseppe.derosa@unipv.it Contact: Pamela Maffioli, MD pamelamaffioli@hotmail.it Principal Investigator: Giuseppe Derosa, MD, PhD Sub-Investigator: Pamela Maffioli, MD Collapse << |
| NCT00102466 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Investigative Centers Nurnberg, Germany Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT01128621 | - | - | Completed | - | - |
| NCT01257334 | Type 2 Diabetes | Phase 3 | Unknown | July 2012 | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT02036515 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00978263 | Diabetes | Not Applicable | Completed | - | China, Chongqing ... more >> the First Affiliated Hospital of Chongqing Medical University Chongqing, Chongqing, China, 400016 Collapse << |
| NCT00703755 | Patients With Metabolic Syndro... more >>me Collapse << | Phase 2 | Completed | - | - |
| NCT01686126 | Complex Endometrial Hyperplasi... more >>a With Atypia Grade 1 Endometrial Endometrioid Adenocarcinoma Collapse << | Phase 2 | Recruiting | December 2020 | Australia, New South Wales ... more >> Chris O'Brien Lifehouse Recruiting Camperdown, New South Wales, Australia, 2050 Principal Investigator: Jonathan Carter Sub-Investigator: Selvan Pather John Hunter Hospital Not yet recruiting New Lambton, New South Wales, Australia, 2305 Principal Investigator: Geoffrey Otton Australia, Queensland The Wesley Hospital Recruiting Auchenflower, Queensland, Australia, 4066 Principal Investigator: Jim Nicklin Sub-Investigator: Andrea Garrett Brisbane Private Hospital Withdrawn Brisbane, Queensland, Australia, 4000 Royal Brisbane and Women's Hospital Recruiting Brisbane, Queensland, Australia, 4029 Principal Investigator: Andreas Obermair Sub-Investigator: James Nicklin Sub-Investigator: Russell Land Sub-Investigator: Andrea Garrett Greenslopes Private Hospital Recruiting Greenslopes, Queensland, Australia, 4120 Principal Investigator: Andreas Obermair Sub-Investigator: Russell Land Sub-Investigator: Naven Chetty Mater Health Services, Brisbane Recruiting South Brisbane, Queensland, Australia, 4101 Principal Investigator: Lewis Perrin Sub-Investigator: Naven Chetty Sub-Investigator: Alex Crandon Mater Private Hospital Recruiting South Brisbane, Queensland, Australia, 4101 Principal Investigator: Lewis Perrin Sub-Investigator: Naven Chetty Gold Coast Hospital Recruiting Southport, Queensland, Australia, 4215 Principal Investigator: Marcelo Nascimento Townsville Hospital Recruiting Townsville, Queensland, Australia, 4810 Principal Investigator: Andreas Obermair Sub-Investigator: James Nicklin Sub-Investigator: Lewis Perrin Sub-Investigator: Russell Land Sub-Investigator: Andrea Garrett Sub-Investigator: Naven Chetty John Flynn Hospital Not yet recruiting Tugun, Queensland, Australia, 4224 Principal Investigator: Marcelo Nascimento Sub-Investigator: Alex Crandon Australia, South Australia Royal Adelaide Hospital Active, not recruiting Adelaide, South Australia, Australia, 5000 Queen Elizabeth Hospital Not yet recruiting Woodville, South Australia, Australia, 5011 Principal Investigator: John Miller Australia, Victoria Royal Women's Hospital Recruiting Carlton, Victoria, Australia, 3053 Principal Investigator: Orla McNally Sub-Investigator: Michael Quinn Sub-Investigator: Deborah Neesham Sub-Investigator: David Wrede Monash Medical Centre Active, not recruiting Clayton, Victoria, Australia, 3168 Australia, Western Australia King Edward Memorial Hospital for Women Recruiting Perth, Western Australia, Australia, 6008 Principal Investigator: Yee Leung Sub-Investigator: Stuart Salfinger Sub-Investigator: Jason Tan St John of God Hospital Recruiting Subiaco, Western Australia, Australia, 6904 Principal Investigator: Stuart Salfinger Sub-Investigator: Jason Tan New Zealand Christchurch Women's Hospital Recruiting Christchurch, New Zealand Contact: Dianne Harker dianne.harker@otago.ac.nz Principal Investigator: Peter Sykes Sub-Investigator: Bryony Simcock Wellington Hospital Recruiting Wellington, New Zealand Contact: Cecile Bergzoll cecile.bergzoll@ccdhb.org.nz Principal Investigator: Cecile Bergzoll Collapse << |
| NCT02036515 | - | - | Completed | - | - |
| NCT01547104 | Diabetes Mellitus Type 2 | Phase 4 | Unknown | October 2012 | Germany ... more >> ikfe GmbH Recruiting Mainz, Rhineland-Palatinate, Germany, 55116 Contact: Thomas Forst, MD, PhD + 49 6131 57636 ext 16 ThomasF@ikfe.de Contact: Daniela Sachsenheimer, MD + 49 6131 57636 ext 46 DanielaS@ikfe.de Sub-Investigator: Daniela Sachsenheimer, MD Sub-Investigator: Stephanie Helleberg, MD Sub-Investigator: Stefan Diessel Sub-Investigator: Michael Mitry, MD Collapse << |
| NCT00617058 | Weight Gain | Phase 2 | Terminated(Terminated in lieu ... more >>of similar,competing large, multi-site study.) Collapse << | - | United States, North Carolina ... more >> University of North Carolina, Department of Psychiatry Chapel HIll, North Carolina, United States, 27599 Collapse << |
| NCT02685774 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT00617058 | - | - | Terminated(Terminated in lieu ... more >>of similar,competing large, multi-site study.) Collapse << | - | - |
| NCT00138515 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT00740051 | - | - | Completed | - | - |
| NCT01319994 | Glucocorticoid Treatment | Phase 2 Phase 3 | Unknown | January 2015 | United Kingdom ... more >> Barts and the London Recruiting London, United Kingdom Collapse << |
| NCT00740051 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT03135301 | Infertility, Female | Phase 3 | Recruiting | March 2019 | Egypt ... more >> Assiut Faculty of Medicine Recruiting Assiut, Egypt Contact: Ahmed Abbas, MD 0100 bmr90@hotmail.com Collapse << |
| NCT01208012 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Germany ... more >> IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Collapse << |
| NCT00538486 | Hypertension ... more >>Obesity Collapse << | Phase 4 | Completed | - | China ... more >> The third hospital affiliated to the Third Military Medical University Chongqing, China, 400042 Collapse << |
| NCT02521753 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Not Applicable | Recruiting | December 2018 | Mexico ... more >> Hospital de Gineco Obstetricia Centro Médico "La Raza" Recruiting Mexico City, D.f., Mexico, 2990 Contact: Victor S Vital-Reyes, MD, PhD 52 55 57245900 ext 23719 vitalito23@hotmail.com Collapse << |
| NCT00676338 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00318656 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 4 | Completed | - | - |
| NCT00947557 | Diabetes Mellitus, Type II | Phase 3 | Terminated | - | - |
| NCT00676338 | - | - | Completed | - | - |
| NCT00814372 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(MBX-102 did not mee... more >>t the target efficacy profile of HbA1c change.) Collapse << | - | United States, California ... more >> Providence Clinical Research Burbank, California, United States, 91505 American Institute of Research Los Angeles, California, United States, 90017 United States, Nevada Nevada Alliance Against Diabetes Las Vegas, Nevada, United States, 89101 United States, North Carolina Piedmont Medical Research Associates Winston-Salem, North Carolina, United States, 27103 United States, Texas DGD Research, Inc. San Antonio, Texas, United States, 78229 India Kamineni Hospitals Pvt, Ltd. Hyderabad, Andhra Pradesh, India, 50008 Bangalore Diabetes Centre Bangalore, India Diacon Hospital Diabetes & Research Centre Bangalore, India M. S. Ramaiah Memorial Hospital Bangalore, India St. John's Medical College & Hospital Bangalore, India Dr. V. Seshiah Diabetes Care & Research Institute Chennai, India Mediciti Hospital Hyderabad, India Medwin Hospital Hyderabad, India Diabetes Thyroid Hormone Research Institute Pvt. Ltd. Indore, India Fortis Hospital Jaipur, India Bharti Research Institute of Diabetes & Endocrinology Karnal, India Amrita Institute of Medical Sciences Kochi, India Diabetes Action Centre Mumbai, India, 400067 BYL Nair Hospital Mumbai, India PD Hindhuja National Hospital Mumbai, India Gandhi Endocrinology & Diabetes Centre Nagpur, India All India Institute of Medical Sciences New Delhi, India Deenanath Mangeshkar Hospital & Research Centre Pune, India Diabetes Care & Research Centre Pune, India Health & Research Centre Trivendrum, India Christian Medical College Vellore, India Collapse << |
| NCT01444248 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals Seoul, Korea, Republic of Collapse << |
| NCT01241669 | Healthy Subjects | Phase 1 | Completed | - | United States, Kansas ... more >> Quintiles Phase I Services Overland Park, Kansas, United States, 661200 Collapse << |
| NCT01068652 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Algeria ... more >> Novo Nordisk Investigational Site Ain Témouchent, Algeria, 46000 Novo Nordisk Investigational Site Alger, Algeria, 16000 Novo Nordisk Investigational Site Algiers, Algeria, 16000 Novo Nordisk Investigational Site Constantine, Algeria, 25000 Novo Nordisk Investigational Site Oran, Algeria, 31000 Novo Nordisk Investigational Site Setif, Algeria, 19000 Novo Nordisk Investigational Site Sidi Bel Abbes, Algeria, 22000 Egypt Novo Nordisk Investigational Site Alexandria, Egypt, 21131 Morocco Novo Nordisk Investigational Site Casablanca, Morocco, 20000 South Africa Novo Nordisk Investigational Site Port Elizabeth, Eastern Cape, South Africa, 6014 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1812 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0002 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0083 Novo Nordisk Investigational Site Vaderbijlpark, Gauteng, South Africa, 1900 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4170 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4450 Novo Nordisk Investigational Site Kimberly, Northern Cape, South Africa, 8301 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7945 Novo Nordisk Investigational Site Sandton, South Africa, 2146 Tunisia Novo Nordisk Investigational Site Tunisia, Tunisia, 1053 Collapse << |
| NCT00110851 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Connecticut ... more >> Phoenix Internal Medicine Associates Waterbury, Connecticut, United States, 06708 United States, Florida University Clinical Research-DeLand DeLand, Florida, United States, 32720 Center for Diabetes and Endocrine Care Hollywood, Florida, United States, 33021 Genesis Research International Longwood, Florida, United States, 32779 Baptist Diabetes Associates Miami, Florida, United States, 33156 Andres Patron, DO PA Pembroke Pines, Florida, United States, 33028 CLIRECO, Inc. Tamarac, Florida, United States, 33321 Endocrine Clinical Research Winter Park, Florida, United States, 32789, United States, Kansas PRN of Kansas Wichita, Kansas, United States, 67203 United States, North Carolina Medical Research Associates of Charlotte Charlotte, North Carolina, United States, 28211 Neem Research Group of Charlotte Charlotte, North Carolina, United States, 28262 Unifour Medical Research Associates Hickory, North Carolina, United States, 28601 Neem Research Group of Raleigh Raleigh, North Carolina, United States, 27609 Piedmont Medical Research Associates Winston-Salem, North Carolina, United States, 27103 United States, Oklahoma COR Clinical Research, LLC Oklahoma City, Oklahoma, United States, 73103 United States, Oregon Clinical Research Institute of Southern Oregon, PC Medford, Oregon, United States, 97504 United States, Pennsylvania Camp Hill Clinical Research Center Camp Hill, Pennsylvania, United States, 17011 United States, South Carolina Neem Research Group Columbia, South Carolina, United States, 29201 Palmetto Medical Research Associates Mt. Pleasant, South Carolina, United States, 29464 United States, Texas Oaks Medical Center Spring, Texas, United States, 77386 Collapse << |
| NCT01971034 | Pancreatic Adenocarcinoma Adva... more >>nced or Metastatic Collapse << | Phase 2 | Completed | - | Brazil ... more >> ICESP Sao Paulo, SP, Brazil, 01246000 Collapse << |
| NCT02497313 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for Diabetes Research, Herlev-Gentofte Hospital Hellerup, Denmark, 2900 Collapse << |
| NCT01068652 | - | - | Completed | - | - |
| NCT02588859 | - | - | Completed | - | India ... more >> Research Site Nellure, Andhra Pradesh, India Research Site Vishakhapatnam, Andhra Pradesh, India Research Site Guwahati, Assam, India Research Site New Delhi, Delhi, India Research Site Ahmedabad, Gujarat, India Research Site Rajkot, Gujarat, India Research Site Vadodara, Gujarat, India Research Site Karnal, Haryana, India Research Site Yamunanagar, Haryana, India Research Site Bangalore, Karnataka, India Research Site Trivandrum, Kerala, India Research Site Cochin, Kochi, India Research Site Bhopal, Madhya Pradesh, India Research Site Indore, Madhya Pradesh, India Research Site Mumbai, Maharashtra, India Research Site Pune, Maharashtra, India Research Site Amritsar, Punjab, India Research Site Chandigarh, Punjab, India Research Site Ludhiana, Punjab, India Research Site Jaipur, Rajasthan, India Research Site Udaipur, Rajasthan, India Research Site Madurai, Tamiladu, India Research Site Chennai, Tamilnadu, India Research Site Hyderabad, Telangana, India Research Site Hyderebad, Telangana, India Research Site Allahabad, Uttar Pradesh, India Research Site Kanpur, Uttar Pradesh, India Research Site Lucknow, Uttar Pradesh, India Research Site Noida, Uttar Pradesh, India Research Site Kolkata, West Bengal, India Research Site new Delhi, India Collapse << |
| NCT00795808 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | New Zealand ... more >> University of Auckland Auckland, New Zealand Collapse << |
| NCT01020123 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00318656 | - | - | Completed | - | - |
| NCT01768559 | - | - | Completed | - | - |
| NCT00386100 | - | - | Completed | - | - |
| NCT01020123 | - | - | Completed | - | - |
| NCT01768559 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00386100 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01678820 | - | - | Terminated(Merck terminated th... more >>e study for business reasons in November 2013.) Collapse << | - | - |
| NCT00437970 | Diabetes Mellitus, Type 2 | Phase 4 | Withdrawn(Unable to secure sup... more >>ply of the study medication) Collapse << | February 2009 | Australia, Northern Territory ... more >> Menzies School of Health Research Darwin, Northern Territory, Australia, 0810 Collapse << |
| NCT00558077 | Polycystic Ovary Syndrome ... more >> Infertility Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Catanzaro/Italy, Italy, 88100 Collapse << |
| NCT01481116 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT03066830 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | May 2019 | - |
| NCT01678820 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(Merck terminated th... more >>e study for business reasons in November 2013.) Collapse << | - | - |
| NCT03125694 | Uncontrolled Type 2 Diabetes M... more >>ellitus Collapse << | Phase 3 | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of Collapse << |
| NCT01106131 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Hospital Seoul, Korea, Republic of Collapse << |
| NCT01481116 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT02147925 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 4 | Completed | - | China, Guangdong ... more >> The Third Affiliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT02305381 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02931591 | Small for Gestational Age | Not Applicable | Not yet recruiting | June 2020 | - |
| NCT03761134 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | September 2020 | China ... more >> Investigational Site Number 1560013 Recruiting Shanghai, China Collapse << |
| NCT02131272 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated | - | - |
| NCT02305381 | - | - | Completed | - | - |
| NCT01392677 | Type 2 Diabetes Mellitus ... more >> High HbA1c Level Inadequate Glycaemic Control Collapse << | Phase 3 | Completed | - | - |
| NCT02131272 | - | - | Terminated | - | - |
| NCT01012037 | - | - | Completed | - | - |
| NCT02769481 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | December 2019 | - |
| NCT01392677 | - | - | Completed | - | - |
| NCT00240370 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03118713 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | January 2019 | Korea, Republic of ... more >> Site 13 Busan, Korea, Republic of Site 02 Deagu, Korea, Republic of Site 11 Guri-si, Korea, Republic of Site 01 Seoul, Korea, Republic of Site 05 Seoul, Korea, Republic of Site 07 Seoul, Korea, Republic of Site 09 Seoul, Korea, Republic of Site 12 Suwon-si, Korea, Republic of Collapse << |
| NCT01426802 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Egypt ... more >> Investigative site Cairo, Egypt Collapse << |
| NCT01012037 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01169779 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational Site Number 156011 Beijing, China, 100034 Investigational Site Number 156012 Beijing, China, 100101 Investigational Site Number 156019 Beijing, China, 100191 Investigational Site Number 156002 Beijing, China, 100700 Investigational Site Number 156003 Beijing, China, 100730 Investigational Site Number 156009 Beijing, China, 100730 Investigational Site Number 156001 Beijing, China, 100853 Investigational Site Number 156036 Changchun, China, 130041 Investigational Site Number 156016 Changsha, China, 410008 Investigational Site Number 156015 Changsha, China, 410011 Investigational Site Number 156006 Chengdu, China, 610041 Investigational Site Number 156032 Chengdu, China, 610072 Investigational Site Number 156010 Dalian, China, 116027 Investigational Site Number 156004 Guangzhou, China, 510080 Investigational Site Number 156008 Guangzhou, China, 510080 Investigational Site Number 156025 Guangzhou, China, 510630 Investigational Site Number 156031 Haikou, China, 57028 Investigational Site Number 156014 Harbin, China, 150001 Investigational Site Number 156029 Hefei, China, 230022 Investigational Site Number 156013 Qingdao, China, 266003 Investigational Site Number 156007 Shanghai, China, 200003 Investigational Site Number 156030 Shanghai, China, 200065 Investigational Site Number 156020 Shenyang, China, 110004 Investigational Site Number 156035 Suzhou, China, 215004 Investigational Site Number 156033 Taiyuan, China, 030001 Investigational Site Number 156037 Tianjin, China, 300052 Investigational Site Number 156022 Xi'An, China, 710032 Investigational Site Number 156023 Xi'An, China, 710061 Hong Kong Investigational Site Number 344001 Hong Kong, Hong Kong Investigational Site Number 344003 Hong Kong, Hong Kong Investigational Site Number 344002 Shatin, Nt, Hong Kong Malaysia Investigational Site Number 458001 Kelantan, Malaysia, 16150 Investigational Site Number 458003 Kuala Lumpur, Malaysia, 59100 Investigational Site Number 458002 Putrajaya, Malaysia, 62250 Thailand Investigational Site Number 764002 Bangkok, Thailand, 10400 Collapse << |
| NCT01233622 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03770728 | Type 2 Diabetes Mellitus | Phase 3 | Not yet recruiting | April 2021 | - |
| NCT01123980 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01169779 | - | - | Completed | - | - |
| NCT03155087 | - | - | Completed | - | - |
| NCT00486187 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Canada, Ontario ... more >> Partners Research Brampton, Ontario, Canada, L6V 1B4 Collapse << |
| NCT00379769 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00118963 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Denmark ... more >> Steno Diabetes Center Gentofte, Denmark, 2820 Collapse << |
| NCT01123980 | - | - | Completed | - | - |
| NCT00934817 | Healthy | Phase 1 | Completed | - | - |
| NCT01294553 | - | - | Completed | - | - |
| NCT01294553 | - | - | Completed | - | - |
| NCT01819129 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03684642 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 2020 | United States, Alabama ... more >> Investigational Site Number 8400038 Recruiting Birmingham, Alabama, United States, 35211 United States, Arizona Investigational Site Number 8400035 Recruiting Chandler, Arizona, United States, 85224 Investigational Site Number 8400005 Recruiting Glendale, Arizona, United States, 85306 United States, California Investigational Site Number 8400040 Recruiting Tustin, California, United States, 92780 United States, Florida Investigational Site Number 8400041 Recruiting Pembroke Pines, Florida, United States, 33026 United States, Georgia Investigational Site Number 8400025 Recruiting Snellville, Georgia, United States, 30078 United States, Kentucky Investigational Site Number 8400044 Recruiting Lexington, Kentucky, United States, 40503 United States, New Jersey Investigational Site Number 8400001 Recruiting Bridgeton, New Jersey, United States, 08302 United States, North Carolina Investigational Site Number 8400036 Recruiting Morehead City, North Carolina, United States, 28557 United States, Utah Investigational Site Number 8400037 Recruiting Layton, Utah, United States, 84041 Collapse << |
| NCT01819129 | - | - | Completed | - | - |
| NCT01310452 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | - | China ... more >> Zhong Shan Hospital, Fudan University Shang hai, China Collapse << |
| NCT00995787 | Type II Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site San Diego, California, United States Collapse << |
| NCT00568984 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100034 Novo Nordisk Investigational Site Beijing, Beijing, China, 100044 Novo Nordisk Investigational Site Beijing, Beijing, China, 100730 China, Tianjin Novo Nordisk Investigational Site Tianjin, Tianjin, China, 300052 Malaysia Novo Nordisk Investigational Site Cheras, Malaysia, 56000 Novo Nordisk Investigational Site Ipoh, Malaysia, 30450 Novo Nordisk Investigational Site Kota Bharu, Kelantan, Malaysia, 16150 Philippines Novo Nordisk Investigational Site Cebu City, Philippines, 6000 Novo Nordisk Investigational Site Manila, Philippines, 1003 Novo Nordisk Investigational Site Marikina City, Philippines, 1800 Novo Nordisk Investigational Site Pampanga, Philippines, 2000 Novo Nordisk Investigational Site Quezon City, Philippines, 1100 Novo Nordisk Investigational Site Quezon, Philippines, 4301 Thailand Novo Nordisk Investigational Site Bangkok, Thailand, 10300 Novo Nordisk Investigational Site Bangkok, Thailand, 10330 Collapse << |
| NCT00463502 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03606694 | Type 2 Diabetes Mellitus | Phase 2 | Not yet recruiting | September 30, 2020 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00379769 | - | - | Completed | - | - |
| NCT01867502 | Diabetes Mellitus, Type 2 ... more >> Hypoglycemic Agents Diabetic Blood Glucose Monitoring Exercise Collapse << | Phase 4 | Unknown | August 2015 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre Recruiting Porto Alegre, Rio Grande do Sul, Brazil, 90035-903 Contact: Aline Fofonka, MSc 55-5192339191 alinefofonka@hotmail.com Principal Investigator: Beatriz D Schaan, PhD Collapse << |
| NCT01874080 | Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT00261352 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT00744367 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT02854527 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Pharma GmbH & Co KG Biberach, Germany, 88397 Collapse << |
| NCT02854527 | - | - | Completed | - | - |
| NCT00018616 | Diabetes | Phase 2 | Completed | - | United States, Connecticut ... more >> VA Connecticut Health Care System (West Haven) West Haven, Connecticut, United States, 06516 Collapse << |
| NCT00592969 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Argentina ... more >> Novo Nordisk Investigational Site Mariano Acosta s/n, Argentina, 5016 Brazil Novo Nordisk Investigational Site Santa Efigenia, Brazil, 30150-320 Collapse << |
| NCT00317928 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | - |
| NCT00432276 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02949193 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Medical Center Seoul, Korea, Republic of, 110-746 Collapse << |
| NCT00960661 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00432276 | - | - | Completed | - | - |
| NCT00960661 | - | - | Completed | - | - |
| NCT00698230 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00108615 | Glucose Metabolism Disorders ... more >> Diabetes Collapse << | Phase 4 | Completed | - | United States, Arkansas ... more >> Central Arkansas Veterans HCS Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT02512523 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | - | Korea, Republic of ... more >> HANDOK Inc. Recruiting Seoul, Korea, Republic of Contact: Sejin Kim, CRM SeJin.Kim@handok.com Contact: Sojung Park, CPL SoJung.Park@handok.com Collapse << |
| NCT03604419 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 2 | Recruiting | June 1, 2019 | United States, Florida ... more >> Clinical Pharmacology of Miami Recruiting Miami, Florida, United States, 33014 Contact: KENNETH Lasseter, MD Collapse << |
| NCT01289119 | - | - | Completed | - | - |
| NCT01289119 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00817778 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00324363 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Guangzhou, China Research Site Nanjing, China Research Site Shanghai, China Research Site Sichuan, China India Research Site Chennai, India Research Site Mumbai, India Research Site Pune, India Korea, Republic of Research Site Seoul, Korea, Republic of Research Site Sungnam City, Korea, Republic of Taiwan Research Site Chiayi, Taiwan Research Site Taichung, Taiwan Research Site Tainan, Taiwan Research Site Taipei, Taiwan Collapse << |
| NCT00817778 | - | - | Completed | - | - |
| NCT00139477 | Obesity | Not Applicable | Completed | - | United States, Florida ... more >> Nemours Children's Clinic Jacksonville, Florida, United States, 32207 Collapse << |
| NCT00774553 | Type 2 Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States India Research site Ahmedabad, India Collapse << |
| NCT00139477 | - | - | Completed | - | - |
| NCT00603239 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Alabama ... more >> Research Site Birmingham, Alabama, United States United States, California Research Site Concord, California, United States Research Site Fresno, California, United States United States, Colorado Research Site Denver, Colorado, United States United States, Nevada Research Site Las Vegas, Nevada, United States United States, Ohio Research Site Mogadore, Ohio, United States United States, Oregon Research Site Corvallis, Oregon, United States Canada, British Columbia Research Site New Westminster, British Columbia, Canada Canada, Manitoba Research Site Winnipeg, Manitoba, Canada Canada, Ontario Research Site Ajax, Ontario, Canada Research Site Cambridge, Ontario, Canada Research Site Windsor, Ontario, Canada Mexico Research Site Mexico City, Distrito Federal, Mexico Research Site Celaya, Guanajuato, Mexico Research Site Monterrey, Nuevo Leon, Mexico Research Site Chihuahua, Mexico Research Site Mexico City, Mexico Romania Research Site Baia Mare, Romania Research Site Brasov, Romania Research Site Bucuresti, Romania Research Site Dolj, Romania Research Site Iasi, Romania Research Site Suceava, Romania South Africa Research Site Johannesburg, South Africa Research Site Pretoria, South Africa Collapse << |
| NCT00603239 | - | - | Completed | - | - |
| NCT02288273 | Type 2 Diabetes | Phase 4 | Completed | - | - |
| NCT02288273 | - | - | Completed | - | - |
| NCT00351884 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative Site Investigative Centers, Germany Collapse << |
| NCT01336751 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States Collapse << |
| NCT02025907 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01126580 | - | - | Completed | - | - |
| NCT01376557 | - | - | Completed | - | - |
| NCT02025907 | - | - | Completed | - | - |
| NCT01126580 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01376557 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01496430 | - | - | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT00541437 | - | - | Completed | - | Taiwan ... more >> National Taiwan university hospital Taipei, Taiwan Collapse << |
| NCT00299871 | Type 2 Diabetes | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Canada Sanofi-Aventis Administrative Office Laval, Canada Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT00627445 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT01496430 | Hypertension ... more >>Diabetes Collapse << | Phase 3 | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT00627445 | - | - | Completed | - | - |
| NCT00469092 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT02467920 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Hubei ... more >> Tongji Hospital, Tongji Medical College, Huazhong University of Science and technology Wuhan, Hubei, China, 430000 Collapse << |
| NCT01664247 | - | - | Completed | - | - |
| NCT00420511 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Ontario ... more >> Leadership Sinai Centre for Diabetes Toronto, Ontario, Canada, M5T 3L9 Collapse << |
| NCT03030300 | Type2 Diabetes | Phase 4 | Completed | - | China, Shandong ... more >> Shandong Provincial Hospital Jinan, Shandong, China, 250001 Qianfoshan Hospital Jinan, Shandong, China, 250012 Qilu Hospital Jinan, Shandong, China, 250014 Collapse << |
| NCT01664247 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00420511 | - | - | Completed | - | - |
| NCT00044447 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Dallas Diabetes & Endocrine Center Dallas, Texas, United States, 75230 Collapse << |
| NCT02693392 | Type-2 Diabetes Mellitus | Not Applicable | Completed | - | India ... more >> Department of Medicine and Department of Pharmacology, AIIMS Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT01779362 | Prediabetes T... more >>ype 2 Diabetes Collapse << | Phase 3 | Active, not recruiting | August 2019 | United States, Illinois ... more >> Jesse Brown VA Medical Center Chicago, Illinois, United States, 60612 University of Chicago Chicago, Illinois, United States, 60637 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Washington VA Puget Sound Health Care System Seattle, Washington, United States, 98108 Collapse << |
| NCT00039013 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00046462 | Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00449930 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00402909 | Type 2 Diabetes | Phase 4 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07974 Collapse << |
| NCT02338921 | Type 2 Diabetes | Phase 4 | Recruiting | December 2018 | Korea, Republic of ... more >> Soo Lim Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Dong Hwa Lee, MD Collapse << |
| NCT00097500 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Finland ... more >> Research Site Helsinki, Finland Netherlands Research Site Amsterdam, Netherlands Sweden Research Site Goteborg, Sweden Collapse << |
| NCT00237237 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers Nurnberg, Germany Collapse << |
| NCT00449930 | - | - | Completed | - | - |
| NCT00384215 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00097500 | - | - | Completed | - | - |
| NCT00934323 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT03351478 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | May 2019 | - |
| NCT03766256 | Gestational Diabetes | Not Applicable | Not yet recruiting | December 31, 2019 | United States, California ... more >> Toluca Lake Health Center Burbank, California, United States, 91505 100 Medical Plaza Primary Care Suites 455 & 490 Los Angeles, California, United States, 90024 Internal Medicine Women's Health Clinic Suites 250 & 290 Los Angeles, California, United States, 90024 UCLA Internal Medicine Family Westwood Suite 465 Los Angeles, California, United States, 90024 UCLA Internal Medicine Geriatrics Suite 365 & 420 Los Angeles, California, United States, 90024 CPN Brentwood Los Angeles, California, United States, 90049 CPN West Washington Internal Medicine Los Angeles, California, United States, 90066 Pacific Palisades Pacific Palisades, California, United States, 90272 12th Street Clinic Santa Monica, California, United States, 90401 Wilshire Office Santa Monica, California, United States, 90401 CPN Santa Monica 15th Street Family Medicine Practice Santa Monica, California, United States, 90404 CPN/Santa Monica Parkside Santa Monica, California, United States, 90404 Santa Moica Bay Physicians/20th Street 10th Floor Santa Monica, California, United States, 90404 Santa Monica Bay Physicians Plaza Santa Monica, California, United States, 90404 Santa Monica Bay Physicians/20th Street 3rd Floor Santa Monica, California, United States, 90404 Santa Monica Internal Medicine Santa Monica, California, United States, 90404 UCLA Family Health Clinic Santa Monica, California, United States, 90404 Santa Moica Bay Physicians Ocean Park Santa Monica, California, United States, 90405 Jack H. Skirball Health Center Woodland Hills, California, United States, 91364 CPN Woodland Hills Practice Woodland Hills, California, United States, 91367 Collapse << |
| NCT00283049 | - | - | Terminated(Due to technical is... more >>sues relating to the Electronic diary data.) Collapse << | - | - |
| NCT01778049 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01101204 | Diabetes Dysl... more >>ipidemia Inflammation Cytokines Collapse << | Not Applicable | Unknown | December 2012 | Poland ... more >> Department of Clinical Pharmacology Not yet recruiting Katowice, Poland, 40752 Contact: Krzysztof Labuzek, MD, PhD +48 32 252 39 02 lbuldak@gmail.com Sub-Investigator: Łukasz Bułdak, MD Principal Investigator: Bogusław Okopień, MD, PhD Sub-Investigator: Krzysztof Labuzek, MD, PhD Collapse << |
| NCT00283049 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(Due to technical is... more >>sues relating to the Electronic diary data.) Collapse << | - | United States, New Jersey ... more >> sanofi-aventis, US Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT01619059 | - | - | Completed | - | - |
| NCT02033876 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00044668 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Hungary ... more >> Diagnostic Units Hungary Kft. Budapest, Hungary, H 1036 Peterfy Teaching Hospital Budapest, Hungary, H 1076 Uzsoki Street Municipal Hospital Budapest, Hungary, H 1145 Collapse << |
| NCT00839527 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01619059 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00998686 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | November 2011 | - |
| NCT00839527 | - | - | Completed | - | - |
| NCT00622089 | Type 2 Diabetes | Phase 2 Phase 3 | Terminated(Program terminated) | December 2008 | - |
| NCT02635386 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Phase 3 | Recruiting | August 2019 | United States, Louisiana ... more >> Woman's Hospital Recruiting Baton Rouge, Louisiana, United States, 70815 Contact: Karen Elkind-Hirsch, PhD. 225-231-5278 Contact: Ericka Seidemann, MS 225-231-5275 ericka.seidemann@womans.org Principal Investigator: Karen Elkind-Hirsch, Ph.D. Sub-Investigator: Renee Harris, M.D. Collapse << |
| NCT00348712 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT00837590 | Pre-diabetes ... more >>Obesity Collapse << | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana Clinical Research Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01376323 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Anniston, Alabama, United States, 36207 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 GSK Investigational Site Miramar, Florida, United States, 33025 France GSK Investigational Site Nantes cedex 01, France, 44093 GSK Investigational Site Pierre-Bénite Cedex, France, 69495 GSK Investigational Site Rennes Cedex, France, 35046 GSK Investigational Site Rueil-Malmaison, France, 92502 Spain GSK Investigational Site Alicante, Spain, 03114 GSK Investigational Site Badalona, Spain, 08916 GSK Investigational Site Granada, Spain, 18004 GSK Investigational Site Madrid, Spain, 28046 United Kingdom GSK Investigational Site Edinburgh, Midlothian, United Kingdom, EH4 2XU GSK Investigational Site Cambridge, United Kingdom, CB2 0GG GSK Investigational Site Coventry, United Kingdom, CV2 2DX GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT00701935 | Diabetes Mellitus | Phase 2 | Terminated(Enrollment was much... more >> slower than anticipated, leading to a decision to terminate the study early for enrollment futility.) Collapse << | - | United States, Arizona ... more >> Research Site Temple, Arizona, United States United States, Colorado Research Site Colorado Springs, Colorado, United States United States, Hawaii Research Site Honolulu, Hawaii, United States United States, Nebraska Research Site Bellevue, Nebraska, United States United States, Nevada Research Site Las Vegas, Nevada, United States Canada, Alberta Research Site Calgary, Alberta, Canada Canada, British Columbia Research Site Vancouver, British Columbia, Canada Research Site Victoria, British Columbia, Canada Canada, Manitoba Research Site Winnipeg, Manitoba, Canada Canada, New Brunswick Research Site Saint John, New Brunswick, Canada Canada, Ontario Research Site Brampton, Ontario, Canada Research Site London, Ontario, Canada Research Site Ottawa, Ontario, Canada Research Site Toronto, Ontario, Canada Canada, Quebec Research Site Chicoutimi, Quebec, Canada Research Site Pointe-Claire, Quebec, Canada Canada Research Site Quebec, Canada Collapse << |
| NCT00701935 | - | - | Terminated(Enrollment was much... more >> slower than anticipated, leading to a decision to terminate the study early for enrollment futility.) Collapse << | - | - |
| NCT00961909 | Diabetes Mellitus Type 2 | Phase 1 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Chula Vista, California, United States, 91911 United States, Florida Ft. Myers, Florida, United States, 33901 Miramar, Florida, United States, 33025 United States, Texas San Antonio, Texas, United States, 78209 San Antonio, Texas, United States, 78229-4801 Collapse << |
| NCT02373865 | Diabetes Mellitus Type 2 | Phase 4 | Terminated(Premature terminati... more >>on due to insufficient patient recruitement..) Collapse << | - | Germany ... more >> GWT-TUD GmbH / Studienzentrum Hanefeld Dresden, Germany, 01307 Collapse << |
| NCT02004886 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01175473 | - | - | Completed | - | - |
| NCT01175473 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Sanofi-Aventis Investigational Site Number 276006 Berlin, Germany, 14050 Sanofi-Aventis Investigational Site Number 276004 Kiel, Germany, 24105 Sanofi-Aventis Investigational Site Number 276002 Mainz, Germany, 55116 Sanofi-Aventis Investigational Site Number 276003 Mannheim, Germany, 68167 Sanofi-Aventis Investigational Site Number 276005 Mönchengladbach, Germany, 41061 Sanofi-Aventis Investigational Site Number 276007 München, Germany, 80636 Sanofi-Aventis Investigational Site Number 276001 Neuss, Germany, 41460 Collapse << |
| NCT02786823 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | India ... more >> All India Institute of Medical Sciences, Bhubaneswar Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT02004886 | - | - | Completed | - | - |
| NCT02549924 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(no financial suppor... more >>t) Collapse << | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03353948 | Polycystic Ovary Syndrome ... more >> Obesity Infertility, Female Collapse << | Phase 4 | Completed | - | - |
| NCT01376323 | - | - | Completed | - | - |
| NCT01471808 | Diabetes Mellitus, Type 2 | Phase 4 | Enrolling by invitation | December 2021 | - |
| NCT02564926 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Research Site Ansan-si, Korea, Republic of, 15355 Research Site Changwon-si, Korea, Republic of, 51353 Research Site Daegu, Korea, Republic of, 41931 Research Site Daejeon-si, Korea, Republic of, 35233 Research Site Goyang-si, Korea, Republic of, 10326 Research Site Incheon, Korea, Republic of, 21565 Research Site Seongnam-si, Korea, Republic of, 13620 Research Site Seoul, Korea, Republic of, 01830 Research Site Seoul, Korea, Republic of, 04401 Research Site Seoul, Korea, Republic of, 05278 Research Site Seoul, Korea, Republic of, 06273 Research Site Suwon-si, Korea, Republic of, 16499 Research Site Uijeongbu-si, Korea, Republic of, 11765 Research Site Wonju-si, Korea, Republic of, 220-701 Collapse << |
| NCT00333151 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01332071 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Brazil ... more >> GSK Investigational Site Goiania, Goiás, Brazil Collapse << |
| NCT01332071 | - | - | Completed | - | - |
| NCT03268005 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | January 29, 2019 | - |
| NCT02235298 | Type 2 Diabetes | Phase 4 | Recruiting | December 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Gianluca Iacobellis, MD PhD giacobellis@med.miami.edu Principal Investigator: Gianluca Iacobellis, MD PhD University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Gianluca Iacobellis, MD PhD giacobellis@med.miami.edu Collapse << |
| NCT01059825 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00437554 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Korea, Republic of ... more >> Handok Seoul, Korea, Republic of Collapse << |
| NCT02981121 | PreDiabetes | Phase 4 | Recruiting | November 2020 | Korea, Republic of ... more >> Kyunghee University Medical Center Recruiting Seoul, Korea, Republic of, 02447 Contact: Jeong-taek Woo, MD, PhD +82-2-958-8128 jtwoomd@khmc.or.kr Contact: Sang Youl Rhee, MD, PhD +82-2-958-8220 bard95@hanmail.net Principal Investigator: Jeong-taek Woo, MD, PhD Sub-Investigator: Suk Chon, MD, PhD Sub-Investigator: Sang Youl Rhee, MD, PhD Collapse << |
| NCT03757910 | Alzheimer Disease | Early Phase 1 | Not yet recruiting | August 2019 | United States, New York ... more >> Columbia University Medical Center Not yet recruiting New York, New York, United States, 10032 Contact: Jose A Luchsinger, MD 212-305-4730 jal94@cumc.columbia.edu Principal Investigator: Jose A Luchsinger, MD Sub-Investigator: F X Pi-Sunyer, MD Albert Einstein College of Medicine Not yet recruiting New York, New York, United States, 10461 Principal Investigator: Jill Crandall, MD Collapse << |
| NCT00850239 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | December 2010 | - |
| NCT02443922 | Type 2 Diabetes | Not Applicable | Unknown | December 2015 | - |
| NCT01059825 | - | - | Completed | - | - |
| NCT01066715 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03770052 | Type 2 Diabetes Mellitus ... more >> Inadequate Glucose Control Collapse << | Phase 4 | Not yet recruiting | December 31, 2020 | Korea, Republic of ... more >> Pusan National University Hospital Busan, Korea, Republic of Collapse << |
| NCT01933256 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 Collapse << |
| NCT02621489 | Atherosclerosis ... more >> Diabetes Restenosis Collapse << | Phase 4 | Recruiting | December 2019 | Sweden ... more >> Dept of clinical science and education Karolinska Institutet Södersjukhuset Recruiting Stockholm, Other, Sweden, 11883 Contact: Thomas Nyström, MD, PhD +4686163211 thomas.nystrom@sodersjukhuset.se Contact: Nils Witt, Md, PhD +4686161000 Sub-Investigator: Nils Witt, MD, PhD Sub-Investigator: Irene Santos, MD Collapse << |
| NCT01075282 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01269996 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Hong Kong ... more >> Department of Medicine & Therapeutics, Prince of Wales Hospital, The Chinese University of Hong Kong Hong Kong, Hong Kong Collapse << |
| NCT00991055 | Type 2 Diabetes | Phase 4 | Unknown | October 2010 | Taiwan ... more >> National Cheng Kung University Hospital Recruiting Tainan, Taiwan Contact: Ta-Jen Wu, Dr 886-6-2353535 ext 5387 djwu@mail.ncku.edu.tw Principal Investigator: Ta-Jen Wu, Dr. Collapse << |
| NCT03603080 | Obesity | Phase 4 | Not yet recruiting | November 1, 2019 | United States, Connecticut ... more >> Hartford Hospital Not yet recruiting Hartford, Connecticut, United States, 06102 Contact: Pavlos K Papasavas, MD 860-246-2071 pavlos.papasavas@hhchealth.org Contact: Darren Tishler, MD 8605244566 darren.tishler@hhchealth.org Sub-Investigator: Pavlos K Papasavas, MD Sub-Investigator: Darren Tishler, MD Principal Investigator: Devika Umashanker, MD Collapse << |
| NCT00783744 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Austria ... more >> Sanofi aventis administrative office Vienna, Austria Finland Sanofi-aventis administrative office Helsinki, Finland France Sanofi-aventis administrative office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Switzerland Sanofi-Aventis Administrative Office Geneva, Switzerland United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT01075282 | - | - | Completed | - | - |
| NCT00474838 | Type 2 Diabetes Mellitus ... more >> Pancreatic Beta Cell Function Glucotoxicity Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Hanyang University Medical Center Kuri, Kyunggi-do, Korea, Republic of, 471-020 The Catholic University of Korea Bucheon St.Mary's Hospital Bucheon, Korea, Republic of Inha University Hospital In Cheon, Korea, Republic of, 400-711 Jeju National University Hospital Jeju-do, Korea, Republic of Kyunghee University Medical Center Seoul, Korea, Republic of, 130-702 Korea University Guro Hospital Seoul, Korea, Republic of, 152-730 Kyung Hee University East Weast Neo Medicalcenter Seoul, Korea, Republic of Ajou University Medical Center Suwon, Korea, Republic of, 443-721 Collapse << |
| NCT03746106 | Vitamin B1 Deficiency ... more >> Thiamine Deficiency Collapse << | Phase 4 | Not yet recruiting | October 2019 | - |
| NCT00358124 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT02554877 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02304081 | Type2 Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Profil Mainz GmbH & Co. KG Mainz, Germany, 55116 Collapse << |
| NCT01144728 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Kazakhstan ... more >> Sanofi-Aventis Administrative Office Almaty, Kazakhstan Collapse << |
| NCT01003483 | Obesity | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Research and Clinical Center for Infertility Yazd, Iran, Islamic Republic of, 8916877391 Collapse << |
| NCT01805830 | Type 2 Diabetes | Phase 3 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals CO. LTD Seoul, Korea, Republic of Collapse << |
| NCT02554877 | - | - | Completed | - | - |
| NCT00546442 | Chronic Hepatitis C | Phase 4 | Completed | - | Spain ... more >> Hospital Germans Trias i Pujol Badalona, Barcelona, Spain, 08916 Hospital Parc Taulí Sabadell, Barcelona, Spain, 08208 Hospital de Donostia San Sebastian, Guipuzcoa, Spain, 20014 Fundación Hospital de Alcorcón Alcorcón, Madrid, Spain, 28922 Hospital del Mar Barcelona, Spain, 08003 Hospital General de Ciudad Real Ciudad Real, Spain, 13005 Hospital Reina Sofía Córdoba, Spain, 14004 Hospital Clínico Universitario San Cecilio Granada, Spain, 18003 Hospital Virgen de las Nieves Granada, Spain, 18004 Hospital de Leon Leon, Spain, 24071 Hospital de la Princesa Madrid, Spain, 28006 Hospital Gregorio Marañón Madrid, Spain, 28007 Hospital Carlos III Madrid, Spain, 28029 Hospital Puerta de Hierro Madrid, Spain, 28035 Hospital La Paz Madrid, Spain, 28046 Hospital Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Virgen Macarena Sevilla, Spain, 41009 Hospital Universitario de Valme Sevilla, Spain, 41014 Hospital General Universitario Valencia, Spain, 46014 Collapse << |
| NCT01341795 | - | - | Unknown | - | Korea, Republic of ... more >> Ajou University School of Medicine Recruiting Suwon, Gyeonggi, Korea, Republic of, 433-721 Contact: Doo-Yeoun Cho, MD +82-31-219-4271 dooycho@ajou.ac.kr Sub-Investigator: Young-Sang Kim, MD Collapse << |
| NCT01364675 | Pre-diabetes ... more >>Pre-hypertension Collapse << | Not Applicable | Unknown | December 2016 | Thailand ... more >> Ubon ratchathani Public Health Office Not yet recruiting Muang District, Ubon ratchathani, Thailand, 34000 Principal Investigator: Phisitt Vejakama, M.D. Collapse << |
| NCT02480465 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | March 2017 | Korea, Republic of ... more >> Korea University Anam Hospital Recruiting Seoul, Korea, Republic of Contact: Dong-sub Choi Collapse << |
| NCT03421119 | Diabetes Mellitus, Type 2 | Phase 3 | Not yet recruiting | December 1, 2019 | - |
| NCT01968265 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Alberta ... more >> Isis Investigative Site Red Deer, Alberta, Canada Canada, British Columbia Isis Investigative Site Penticton, British Columbia, Canada Canada, Ontario Isis Investigative Site Cornwall, Ontario, Canada Isis Investigative Site Courtice, Ontario, Canada Isis Investigative Site Sudbury, Ontario, Canada Canada, Quebec Isis Investigative Site Montreal, Quebec, Canada, H2R1V6 Romania Isis Investigative Site Cluj Napoca, Cluj, Romania, 400349 Isis Investigative Site Bucharest, Romania, 010507 Isis Investigative Site Bucharest, Romania, 011794 Isis Investigative site Bucharest, Romania, 022441 South Africa Isis Investigative Site Bloemfontein, Free State, South Africa, 9300 Isis Investigative Site Benoni, Gauteng, South Africa, 1501 Isis Investigative Site Soweto, Gauteng, South Africa, 1818 Isis Investigative Site Somerset West, Western Cape, South Africa, 7130 Collapse << |
| NCT00282451 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Egypt ... more >> Novo Nordisk Investigational Site Aghakhan, Egypt Novo Nordisk Investigational Site Giza, Egypt Saudi Arabia Novo Nordisk Investigational Site Jeddah, Saudi Arabia, 80215 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 245 Collapse << |
| NCT00263965 | Type 2 Diabetes | Phase 2 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | Finland ... more >> Research Site Helsinki, Finland Italy Research Site Pisa, Italy United Kingdom Research Site Oxford, United Kingdom Collapse << |
| NCT01648582 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100730 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chengdu, China, 610041 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chongqing, China, 400030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dalian, China, 116011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guiyang, China, 550004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hefei, China, 230022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanchang, China, 330006 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210006 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanning, China, 530021 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shenyang, China, 110004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shijiazhuang, China, 050051 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wu Han, China, 430022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yangzhou, China, 225001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Zhenjiang, China, 212000 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bucheon,, Korea, Republic of, 420-717 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyunggi-Do, Korea, Republic of, 425-020 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 137-701 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wonju-Si, Korea, Republic of, 220-701 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chihuahua, Mexico, 31238 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Monterrey, Mexico, 64460 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chelyabinsk, Russian Federation, 454047 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 127018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 192012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sankt-Petersburg, Russian Federation, 190000 Collapse << |
| NCT00619697 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01648582 | - | - | Completed | - | - |
| NCT01464437 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03503942 | Pre-diabetes | Not Applicable | Recruiting | December 31, 2022 | Singapore ... more >> Singapore General Hospital Recruiting Singapore, Singapore, 169608 Contact: Yong Mong Bee 6321 3753 ext +65 bee.yong.mong@singhealth.com.sg Collapse << |
| NCT02027753 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Collapse << |
| NCT01918865 | Type 2 Diabetes Mellitus ... more >> Obese Collapse << | Phase 2 | Completed | - | Argentina ... more >> Isis Investigative Site Mar del Plata, Buenos Aires, Argentina, B7600FZN Isis Investigative Site Rosario, Santa Fe, Argentina, S2000CVD Canada, Alberta Isis Investigational Site Edmonton, Alberta, Canada, T6G 2E1 Canada, British Columbia Isis Investigational Site Kelowna, British Columbia, Canada, V1Y 3G8 Isis Investigational Site Vancouver, British Columbia, Canada, V6J 1S3 Canada, Ontario Isis Investigational Site Etobicoke, Ontario, Canada, M9R 4E1 Isis Investigational Site Toronto, Ontario, Canada, M4G 3E8 Isis Investigative Site Toronto, Ontario, Canada, M9V 4B4 Canada, Saskatchewan Isis Investigational Site Saskatoon, Saskatchewan, Canada, S7K 3H3 South Africa Isis Investigational Site Centurion, Gauteng, South Africa, 0154 Isis Investigational Site Mamelodi, Gauteng, South Africa, 0122 Isis Investigational Site Pretoria, Gauteng, South Africa, 0001 Isis Investigational Site Port Elizabeth, Korsten, South Africa Isis Investigational Site Durban, KwaZulu-Natal, South Africa Isis Investigational Site Middelburg, Mpumalanga, South Africa Isis Investigational Site Limpopo, Thabazimbi, South Africa, 0380 Isis Investigational Site Cape Town, Western Cape, South Africa, 7570 Isis Investigational Site Cape Town, Western Cape, South Africa Isis Investigational Site Benoni, South Africa, 1500 Isis Investigational Site Bloemfontein, South Africa, 9301 Isis Investigational Site Pretoria, South Africa, 0184 Collapse << |
| NCT00881530 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00313001 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Novo Nordisk Investigational Site Plainsboro, New Jersey, United States, 08536 Collapse << |
| NCT00881530 | - | - | Completed | - | - |
| NCT02683226 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00396071 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Diabetes Zentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT01804842 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | - |
| NCT01725126 | Obesity | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Kansas GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT00111631 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT02613897 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Texas ... more >> The University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT03527537 | Gestational Diabetes | Phase 4 | Not yet recruiting | June 1, 2028 | United States, Wisconsin ... more >> Medical College of Wisconsin Not yet recruiting Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT01804842 | - | - | Completed | - | - |
| NCT02648854 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Deokjin-gu, Jeonju-si, Jeollabuk-do, Korea, Republic of, 561-712 Collapse << |
| NCT01819272 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00328172 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01819272 | - | - | Completed | - | - |
| NCT00909480 | - | - | Completed | - | - |
| NCT01131182 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 4 | Completed | - | - |
| NCT00328172 | - | - | Completed | - | - |
| NCT01131182 | - | - | Completed | - | - |
| NCT01388361 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01725126 | - | - | Completed | - | - |
| NCT00909480 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT00378729 | Polycystic Ovary Syndrome | Phase 4 | Unknown | October 2011 | France ... more >> Hopital Antoine Beclere Recruiting Clamart, France, 92170 Contact: Hervé FERNANDES, MD,PhD +33(0)- 1 45 37 44 69 herve.fernandez@abc.aphp.fr Principal Investigator: Hervé FERNANDEZ, MD Collapse << |
| NCT01388361 | - | - | Completed | - | - |
| NCT00972283 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02737657 | - | - | Completed | - | Kuwait ... more >> Kuwait City, Kuwait Lebanon Amioun, Lebanon Beirut, Lebanon Nabatieh, Lebanon Saida, Lebanon Taalabya, Lebanon Tripoli, Lebanon United Arab Emirates Al Ain, United Arab Emirates Dubai, United Arab Emirates Collapse << |
| NCT00434954 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Germany ... more >> Research Site Bad Mergentheim, Germany Research Site Berlin, Germany Research Site Bosenheim, Germany Research Site Burghausen, Germany Research Site Datteln, Germany Research Site Dresden, Germany Research Site Essen, Germany Research Site Friedrichsthal, Germany Research Site Hildesheim, Germany Research Site Hirschhorn, Germany Research Site Hohenmolsen, Germany Research Site Jena, Germany Research Site Lehrte, Germany Research Site Leipzig, Germany Research Site Ludwigsburg, Germany Research Site Mannheim, Germany Research Site Marburg, Germany Research Site Marktheidenfeld, Germany Research Site Meissen, Germany Research Site Munchen, Germany Research Site Offenbach, Germany Research Site Oschatz, Germany Research Site Pohlheim, Germany Research Site Regensburg, Germany Research Site Riesa, Germany Research Site Rodgau, Germany Research Site Roding, Germany Research Site Rosenheim, Germany Research Site Schluchtern, Germany Research Site Schwedt/Oder, Germany Research Site Sinsheim, Germany Research Site Speyer, Germany Research Site Unterhaching, Germany Research Site Volklingen, Germany Research Site Wallerfing, Germany Research Site Wangen, Germany Research Site Warburg, Germany Research Site Wiesbaden, Germany Research Site Wolfsburg, Germany Collapse << |
| NCT00972283 | - | - | Completed | - | - |
| NCT00851903 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Vienna, Austria Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Colombia Sanofi-Aventis Administrative Office Bogota, Colombia Egypt Sanofi-Aventis Administrative Office Cairo, Egypt Greece Sanofi-Aventis Administrative Office Kallithea, Greece Hong Kong Sanofi-Aventis Administrative Office Hong Kong, Hong Kong India Sanofi-Aventis Administrative Office Mumbai, India Israel Sanofi-Aventis Administrative Office Natanya, Israel Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Lebanon Sanofi-Aventis Administrative Office Beirut, Lebanon Mexico Sanofi-Aventis Administrative Office Col. Coyoacan, Mexico Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Portugal Sanofi-Aventis Administrative Office Porto Salvo, Portugal Spain Sanofi-Aventis Administrative Office Barcelona, Spain United Kingdom Sanofi-Aventis Administrative Office Guildford Surrey, United Kingdom Collapse << |
| NCT00434954 | - | - | Completed | - | - |
| NCT01947790 | LVM Type 2 Di... more >>abetic Patients With IHD Endothelial Function Collapse << | Phase 4 | Completed | - | China, Hubei ... more >> Wuhan City, Hubei, China, 43000 Xiang Guangda Wuhan, Hubei, China, 430070 Collapse << |
| NCT01215331 | Gestational Diabetes Mellitus | Phase 3 | Completed | - | Canada, Quebec ... more >> Centre de recherche clinique du CHUS Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT00035568 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 4 | Completed | - | United States, Arkansas ... more >> Local Institution Little Rock, Arkansas, United States United States, California Local Institution Los Angeles, California, United States United States, Illinois Local Institution Chicago, Illinois, United States United States, Indiana Local Institution Indianapolis, Indiana, United States United States, New York Local Institution Brooklyn, New York, United States Local Institution Rochester, New York, United States United States, Pennsylvania Local Institution Pittsburgh, Pennsylvania, United States United States, Texas Local Institution Dallas, Texas, United States Local Institution San Antonio, Texas, United States Collapse << |
| NCT00851903 | - | - | Completed | - | - |
| NCT02911948 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Japan ... more >> Novo Nordisk Investigational Site Asahikawa-shi, Hokkaido, Japan, 078-8211 Novo Nordisk Investigational Site Bunkyo-ku, Tokyo, Japan, 113 8431 Novo Nordisk Investigational Site Chigasaki-shi, Kanagawa, Japan, 253-0044 Novo Nordisk Investigational Site Chitose, Hokkaido, Japan, 066-0032 Novo Nordisk Investigational Site Chiyoda-ku, Tokyo, Japan, 101-0024 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0027 Novo Nordisk Investigational Site Fujisawa-shi, Kanagawa, Japan, 251-0041 Novo Nordisk Investigational Site Fukui-shi, Fukui, Japan, 918-8503 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 810-0001 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 819-0006 Novo Nordisk Investigational Site Fukuoka, Japan, 830 8522 Novo Nordisk Investigational Site Kashiwara-shi, Osaka, Japan, 582 0005 Novo Nordisk Investigational Site Kawagoe-shi, Saitama, Japan, 350 0851 Novo Nordisk Investigational Site Kawaguchi-shi, Saitama, Japan, 332-0012 Novo Nordisk Investigational Site Koriyama-shi, Fukushima, Japan, 963 8851 Novo Nordisk Investigational Site Kumamoto-shi, Kumamoto, Japan, 862-0965 Novo Nordisk Investigational Site Kumamoto-shi,Kumamoto, Japan, 862 0976 Novo Nordisk Investigational Site Matsumoto-shi, Nagano, Japan, 390-8621 Novo Nordisk Investigational Site Miyazaki-shi, Japan, 880 0034 Novo Nordisk Investigational Site Naka-shi, Ibaraki, Japan, 311 0113 Novo Nordisk Investigational Site Nakagami, Okinawa, Japan, 901-2393 Novo Nordisk Investigational Site Neyagawa-shi, Osaka, Japan Novo Nordisk Investigational Site Niigata-shi, Niigata, Japan, 950 1104 Novo Nordisk Investigational Site Okawa-shi, Fukuoka, Japan, 831 0016 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 536-0001 Novo Nordisk Investigational Site Ota-ku, Tokyo, Japan, 1430015 Novo Nordisk Investigational Site Oyama-shi, Tochigi, Japan, 323 0022 Novo Nordisk Investigational Site Saitama-shi, Saitama, Japan, 336-0963 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060 0062 Novo Nordisk Investigational Site Sendai-shi, Japan, 980 0021 Novo Nordisk Investigational Site Shimotsuke-shi, Tochigi, Japan, 329 0433 Novo Nordisk Investigational Site Takatsuki-shi, Osaka, Japan, 569 1096 Novo Nordisk Investigational Site Tokyo, Japan, 103-0028 Novo Nordisk Investigational Site Tokyo, Japan, 181-0013 Novo Nordisk Investigational Site Tomigusuku-shi, Okinawa, Japan, 901-0243 Novo Nordisk Investigational Site Tomigusuku-shi, Okinawa, Japan, 901-0244 Novo Nordisk Investigational Site Yamaguchi-shi, Yamaguchi, Japan, 754-0002 Novo Nordisk Investigational Site Yokohama-shi, Japan, 235 0045 Collapse << |
| NCT03175120 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | April 8, 2019 | - |
| NCT00637273 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00637273 | - | - | Completed | - | - |
| NCT02231021 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | November 2018 | Korea, Republic of ... more >> Seoul St Mary's Hospital, The Catholic University of Korea Recruiting Seoul, Korea, Republic of, 137-701 Principal Investigator: Kun-Ho Yoon, MD, PhD Sub-Investigator: Yoon-Hee Choi, MD, PhD Sub-Investigator: Hye-Kyung Yang, MD Collapse << |
| NCT02503943 | Type 2 Diabetes ... more >> Overweight Obesity Masked Hypertension Collapse << | Phase 4 | Unknown | September 2016 | - |
| NCT03700801 | Type 2 Diabetes Mellitus | Phase 4 | Not yet recruiting | March 2019 | - |
| NCT00605475 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Arkansas ... more >> Arkansas Research Medical Testing Little Rock, Arkansas, United States, 72202 United States, Florida Allied Research International - Cetero Research Miami Miami, Florida, United States, 33169 Elite Research Institute Miami Miami, Florida, United States, 33169 United States, Maryland International Research Center Towson MD, Maryland, United States, 21286 United States, Oregon Covance Clinical Research Unit Inc Portland, Oregon, United States, 97239 United States, Washington Charles River Clinical Services NW Tacoma, Washington, United States, 98418 Germany Novartis Investigator Site Berlin, Germany Novartis Investigator Site Kiel, Germany Novartis Investigator Site Moenchengladbach, Germany Novartis Investigator Site Munich, Germany Novartis Investigative Site Neuss, Germany Russian Federation Novartis Investigative Site Moscow, Russian Federation Novartis Investigative Site St. Petersberg, Russian Federation Collapse << |
| NCT03222765 | PreDiabetes | Not Applicable | Recruiting | December 31, 2019 | Spain ... more >> Centro de Salud Jose Marva Recruiting Madrid, Spain, 28030 Contact: Esther Sanchez, MD +34629 Contact: Ana M Santos Collapse << |
| NCT01425580 | Congestive Heart Failure ... more >> Type 2 Diabetes Mellitus Collapse << | Phase 2 | Completed | - | Sweden ... more >> Karolinska Institutet, Department of Clinical Science and Education, Södersjukhuset Stockholm, Sweden, 118 83 Karolinska Institutet, Division of Internal Medicine Södersjukhuset AB Stockholm, Sweden, 118 83 Collapse << |
| NCT00933309 | Breast Cancer | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00494884 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Novartis Investigative Site Bochum, Germany Collapse << |
| NCT01618162 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01589653 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Egypt ... more >> Novo Nordisk Investigational Site Alexandria, Egypt, 21131 Novo Nordisk Investigational Site Beni-Suef, Egypt, 62511 Novo Nordisk Investigational Site Cairo, Egypt, 11562 Indonesia Novo Nordisk Investigational Site Bandung, Indonesia, 40161 Novo Nordisk Investigational Site Malang, Indonesia, 65111 Morocco Novo Nordisk Investigational Site Casablanca, Morocco, 20100 Novo Nordisk Investigational Site Marrakech, Morocco, 40000 Saudi Arabia Novo Nordisk Investigational Site Jeddah, Saudi Arabia, 80215 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 11211 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 11426 Novo Nordisk Investigational Site Taif, Saudi Arabia, 21944 Tunisia Novo Nordisk Investigational Site Tunis, Tunisia, 1007 Novo Nordisk Investigational Site Tunis, Tunisia, 1008 Vietnam Novo Nordisk Investigational Site Ha Noi City, Vietnam Novo Nordisk Investigational Site Ho Chi Minh City, Vietnam Collapse << |
| NCT00494663 | Type 2 Diabetes | Phase 2 | Terminated(Program Terminated) | December 2008 | - |
| NCT01618162 | - | - | Completed | - | - |
| NCT00605475 | - | - | Completed | - | - |
| NCT02315287 | Type 2 Diabetes | Phase 4 | Recruiting | December 2018 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Eu Jeong Ku, MD 82-31-787-6232 eujeong.ku@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT01589653 | - | - | Completed | - | - |
| NCT01602003 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> LG Life Sciences Seoul, Korea, Republic of, 110-062 Collapse << |
| NCT02014740 | Type 2 Diabetes ... more >> Overweight Obesity Collapse << | Phase 4 | Completed | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT02014740 | - | - | Completed | - | - |
| NCT01778049 | - | - | Completed | - | - |
| NCT01140438 | Diabetes | Not Applicable | Active, not recruiting | December 2018 | Norway ... more >> Valdemar Grill Trondheim, Norway, 7006 Collapse << |
| NCT00996658 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00996658 | - | - | Completed | - | - |
| NCT00373178 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Greece ... more >> AHEPA University Hospital of Thessaloniki Thessaloniki, Greece Aristotle University of Thessaloniki Thessaloniki, Greece Collapse << |
| NCT01482286 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT00717457 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00622284 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00870194 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Argentina ... more >> Research Site Buenos Aires, Argentina Research Site Morón, Argentina Australia Research Site Adelaide, Australia Research Site Geelong, Australia Research Site Melbourne, Australia Germany Research Site Aschaffenburg, Germany Research Site Asslar, Germany Research Site Beckum-Neubeckum, Germany Research Site Berlin, Germany Research Site Bosenheim, Germany Research Site Essen, Germany Research Site Falkensee, Germany Research Site Furth im Wald, Germany Research Site Grevenbroich, Germany Research Site Hamburg-Othmarschen, Germany Research Site Hohenmolsen, Germany Research Site Leipzig, Germany Research Site Neuwied, Germany Research Site Pohlheim, Germany Research Site Speyer, Germany Greece Research Site Athens, Greece Research Site Thessaloniki, Greece India Research Site Ahmedabad, India Research Site Bangalore, India Research Site Coimbatore, India Research Site Indore, India Research Site Jaipur, India Korea, Republic of Research Site Daegu, Korea, Republic of Research Site Gwangju, Korea, Republic of Research Site Seoul, Korea, Republic of Research Site Ulsan, Korea, Republic of Mexico Research Site Coatzacoalcos, Mexico Research Site Guadalajara, Mexico Research Site Merida, Mexico Research Site Monterrey, Mexico Research Site Tampico, Mexico Collapse << |
| NCT02284893 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02142309 | Type 2 Diabetes | Phase 4 | Unknown | January 2017 | Italy ... more >> Department of Geriatrics and Metabolic Diseases Active, not recruiting Naples, Italy, 80138 Katherine Esposito Recruiting Naples, Italy, 80138 Contact: Maria Ida Maiorino, MD mariaida.maiorino@unina2.it Sub-Investigator: Giuseppe Bellastella, MD Collapse << |
| NCT00870194 | - | - | Completed | - | - |
| NCT00622284 | - | - | Completed | - | - |
| NCT02002221 | - | - | Completed | - | - |
| NCT00086515 | Diabetes Mellitus, Type II | Phase 3 | Completed | - | - |
| NCT02284893 | - | - | Completed | - | - |
| NCT00984867 | - | - | Completed | - | - |
| NCT02002221 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 4 | Completed | - | Japan ... more >> Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 810-8798 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 815-0071 Novartis Investigative Site Fukutsu-city, Fukuoka, Japan, 811-3217 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 800-0295 Novartis Investigative Site Koriyama-city, Fukushima, Japan, 963-8851 Novartis Investigative Site Mito-city, Ibaraki, Japan, 311-4153 Novartis Investigative Site Ibusuki-city, Kagoshima, Japan, 891-0401 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0802 Novartis Investigative Site Kumamoto-city, Kumamoto, Japan, 861-8039 Novartis Investigative Site Kumamoto-city, Kumamoto, Japan, 861-8520 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8533 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Sendai-city, Miyagi, Japan, 980-0021 Novartis Investigative Site Hirakata-City, Osaka, Japan, 573-0153 Novartis Investigative Site Izumisano-city, Osaka, Japan, 598-8577 Novartis Investigative Site Osaka-city, Osaka, Japan, 530-0001 Novartis Investigative Site Osaka-city, Osaka, Japan, 534-0021 Novartis Investigative Site Takatsuki-city, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Saitama-city, Saitama, Japan, 336-0963 Novartis Investigative Site Sayama-city, Saitama, Japan, 350-1305 Novartis Investigative Site Tokorozawa-city, Saitama, Japan, 359-1161 Novartis Investigative Site Adachi-ku, Tokyo, Japan, 123-0845 Novartis Investigative Site Chiyoda-ku, Tokyo, Japan, 101-0024 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 103-0027 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 103-0028 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 104-0061 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0041 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Ota-ku, Tokyo, Japan, 144-0051 Novartis Investigative Site Suginami-ku, Tokyo, Japan, 166-0004 Collapse << |
| NCT00362440 | HIV Lipodystrophy | Phase 2 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00362440 | - | - | Completed | - | - |
| NCT00184574 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02502344 | Insulin Resistance | Phase 4 | Unknown | - | China, Beijing ... more >> BEI JING Tsinghua Changguang Hospital Recruiting Beijing, Beijing, China, 102218 Contact: jian Z xiao, doctor 0086-010-56118899 ext 13683685711 xiaojzh858@sina.com Collapse << |
| NCT02501850 | Diabetes Mellitus Type 2 | Phase 4 | Unknown | October 2015 | Spain ... more >> Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Contact: Andreea Ciudin, PhD +34932744736 aciudin@vhebron.net Contact: Cristina Hernandez, MD, PhD +34 932746591 cristina.hernandez@vhir.org Principal Investigator: Cristina Hernandez, MD, PhD Sub-Investigator: Andreea Ciudin, MD Sub-Investigator: Olga Simó-Servat, MD Collapse << |
| NCT00081328 | Diabetes Mellitus, Type II | Phase 3 | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Colorado University of Colorado Health Sciences Center, The Children's Hospital Denver, Colorado, United States, 80262 United States, Connecticut Yale University New Haven, Connecticut, United States, 06520 United States, Maryland George Washington University Biostatistics Center Rockville, Maryland, United States, 20852 United States, Massachusetts Massachusetts General Hospital Diabetes Center Boston, Massachusetts, United States, 02114 Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Saint Louis University Health Sciences Center St Louis, Missouri, United States, 63104 Washington University Department of Pediatrics St. Louis, Missouri, United States, 63110 United States, New York Columbia University Medical Center New York City, New York, United States, 10032 State University of New York Upstate Medical University Syracuse, New York, United States, 13210 United States, Ohio Case Western Reserve Cleveland, Ohio, United States, 44106 United States, Oklahoma University of Oklahoma Oklahoma City, Oklahoma, United States, 93104 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT01370707 | Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Bucheon St. Mary's Hospital Bucheon, Korea, Republic of Seoul National University Bundang Hospital Bundang, Korea, Republic of Chonbuk National University Hospital Chonju, Korea, Republic of Kyungpook University Hospital Daegu, Korea, Republic of Yeungnam University Medical Center Daegu, Korea, Republic of National Health Insurance Coporation Ilsan Hospital Ilsan, Korea, Republic of Inha University Hospital Incheon, Korea, Republic of Inje University Pusan Baik Hospital Pusan, Korea, Republic of Pusan National University Hospital Pusan, Korea, Republic of Asan Medical Center Seoul, Korea, Republic of Eulji Medical Center Seoul, Korea, Republic of Hallym University Medical Center(Gangdong) Seoul, Korea, Republic of Hallym University Medical Center(Gangnam) Seoul, Korea, Republic of Kangbuk Samsung Hospital Seoul, Korea, Republic of Korea University Guro Hospital Seoul, Korea, Republic of Kyung Hee University Hospital Seoul, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of Seoul St. Mary's hospital Seoul, Korea, Republic of Severance Hospital Seoul, Korea, Republic of Ajou University Hospital Suwon, Korea, Republic of Uijeongbu St. Mary's Hospital Uijeongbu, Korea, Republic of Collapse << |
| NCT01232491 | - | - | Completed | - | - |
| NCT02623998 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | December 2018 | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2T 5C7 Canada, Manitoba Health Science Centre Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario McMaster University Hamilton, Ontario, Canada St. Joseph's Hospital London, Ontario, Canada, N6A 4V2 LMC Markham, Ontario, Canada Canada, Quebec Univeristy of Montreal Montreal, Quebec, Canada Collapse << |
| NCT01232491 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT00068861 | Polycystic Ovary Syndrome ... more >> Infertility Pregnancy Collapse << | Phase 3 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35249 United States, California University of California at San Diego La Jolla, California, United States, 92037 Stanford University Stanford, California, United States, 94305 United States, Colorado University of Colorado Aurora, Colorado, United States, 80045 United States, Michigan Wayne State Detroit, Michigan, United States, 48201 United States, New Jersey University of Medicine and Dentistry New Jersey Newark, New Jersey, United States, 07103 United States, Pennsylvania Penn State Hershey, Pennsylvania, United States, 17033 University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Baylor College of Medicine Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT01771185 | Type 2 Diabetes | Not Applicable | Unknown | December 2013 | - |
| NCT03000218 | Overweight Ab... more >>normal Liver Function Tests Collapse << | Phase 1 | Unknown | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seoul, Korea, Republic of Contact: Jae-Yong Chung, MD, PhD +82-031-787-3955 Principal Investigator: Jae-Yong Chung, MD, PhD Collapse << |
| NCT02584075 | Coronary Endothelial Function | Phase 4 | Unknown | January 2018 | - |
| NCT01624259 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00081328 | - | - | Completed | - | - |
| NCT01624259 | - | - | Completed | - | - |
| NCT01404676 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Division of Endocrinology and Metabolism, Department of Internal Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01215968 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United Kingdom ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Glasgow, United Kingdom Collapse << |
| NCT01786707 | - | - | Completed | - | - |
| NCT00443755 | Type 2 Diabetes ... more >> Insulin Resistance Metabolic Syndrome Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01307306 | Burns | Not Applicable | Recruiting | September 2019 | Canada, Ontario ... more >> Ross Tilley Burn Centre, Sunnybrook Health Sciences Centre Recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Marc G Jeschke, MD PhD 416-480-6703 marc.jeschke@sunnybrook.ca Contact: Marjorie Burnett, HonsBSc 416-480-6100 ext 88021 marjorie.burnett@sunnybrook.ca Sub-Investigator: Robert Cartotto, MD Sub-Investigator: Shahriar Shahrokhi, MD Sub-Investigator: Alison Cheung, PhD Collapse << |
| NCT01332370 | - | - | Completed | - | - |
| NCT01215968 | - | - | Completed | - | - |
| NCT03738449 | Type 2 Diabetes Mellitus | Phase 1 | Not yet recruiting | December 26, 2018 | Korea, Republic of ... more >> Severance Hospital Not yet recruiting Seoul, Korea, Republic of, 03722 Contact: Min Soo Park, Ph.D. M.D +82-2-2228-0270 minspark@yuhs.ac Principal Investigator: Minsoo Park, Ph.D. M.D Collapse << |
| NCT01786707 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Diabetes Research Institute, University of Miami Miller School of Medicine Miami, Florida, United States, 33136 Collapse << |
| NCT02088034 | Diabetes | Phase 4 | Unknown | February 2016 | United States, Pennsylvania ... more >> Temple University - Center for Obesity Research and Education Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT00506298 | Type 2 Diabetes | Phase 2 | Completed | - | Canada, British Columbia ... more >> Chilliwack, British Columbia, Canada Vancouver, British Columbia, Canada Canada, Manitoba Winnipeg, Manitoba, Canada Canada, Newfoundland and Labrador Bay Roberts, Newfoundland and Labrador, Canada Holyroad, Newfoundland and Labrador, Canada St. Johns, Newfoundland and Labrador, Canada Canada, Ontario Aylmer, Ontario, Canada Burlington, Ontario, Canada Collingwood, Ontario, Canada Corunna, Ontario, Canada Robarts Research Institute London, Ontario, Canada London, Ontario, Canada Newmarket, Ontario, Canada Sarnia, Ontario, Canada Sudbury, Ontario, Canada Toronto, Ontario, Canada Canada, Quebec Montreal, Quebec, Canada Canada, Saskatchewan Saskatoon, Saskatchewan, Canada Collapse << |
| NCT00443755 | - | - | Completed | - | - |
| NCT01958671 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00518115 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT03751657 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | November 7, 2019 | - |
| NCT01958671 | - | - | Completed | - | - |
| NCT01624116 | Diabetes, Type 2 | Not Applicable | Completed | - | Pakistan ... more >> Endocrinology Unit & Diabetes Management Centre, Services Hospital. Lahore, Pakistan Collapse << |
| NCT00518115 | - | - | Completed | - | - |
| NCT00510705 | Severe Coronary Artery Disease... more >> Obesity Impaired Glucose Tolerance Collapse << | Not Applicable | Terminated | - | Germany ... more >> University of Leipzig - Heart Center Leipzig, Germany, 04289 Collapse << |
| NCT01076075 | - | - | Completed | - | - |
| NCT00951899 | - | - | Completed | - | - |
| NCT00951899 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01076075 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00751114 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01090752 | - | - | Completed | - | - |
| NCT01090752 | Diabetes Hype... more >>rtension Collapse << | Phase 4 | Completed | - | Switzerland ... more >> Pechère-Bertschi Antoinette Geneva, Switzerland, 1211 Collapse << |
| NCT00984867 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01111955 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> Farid Marquez, Md Hialeah, Florida, United States, 33012 United States, Washington Capital Clinical Reserch Center Olympia, Washington, United States, 98502 United States, Wisconsin Aurora Advanced Healthcare Milwaukee, Wisconsin, United States, 53209 Australia, New South Wales Local Institution Blacktown, New South Wales, Australia, 2148 Australia, Victoria Local Institution Box Hill, Victoria, Australia, 3128 Australia, Western Australia Local Institution Freemantle, Western Australia, Australia, 6959 Canada, British Columbia Local Institution Surrey, British Columbia, Canada, V4A 2H9 Canada, Manitoba Local Institution Winnipeg, Manitoba, Canada, R3E 3P4 Canada, New Brunswick Local Institution Bathurst, New Brunswick, Canada, E2A 4X7 Canada, Newfoundland and Labrador Local Institution St. John'S, Newfoundland and Labrador, Canada, A1A 3R5 Canada, Ontario Local Institution Thornhill, Ontario, Canada, L4J 8L7 Canada, Quebec Local Institution Mirabel, Quebec, Canada, J7J 2K8 Canada, Saskatchewan Local Institution Saskatoon, Saskatchewan, Canada, S7K 7H9 Canada Local Institution Quebec, Canada, G1N 4V3 Collapse << |
| NCT01751321 | Type 2 Diabetes | Not Applicable | Unknown | October 2013 | - |
| NCT02384109 | Prediabetic State | Not Applicable | Active, not recruiting | June 2019 | United States, California ... more >> Toluca Lake Health Center Burbank, California, United States, 91505 100 Medical Plaza Primary Care Suites 455 & 490 Los Angeles, California, United States, 90024 Internal Medicine Women's Health Clinic Suites 250 & 290 Los Angeles, California, United States, 90024 UCLA Internal Medicine Family Westwood Suite 465 Los Angeles, California, United States, 90024 UCLA Internal Medicine Geriatrics Suite 365 & 420 Los Angeles, California, United States, 90024 CPN Brentwood Los Angeles, California, United States, 90049 CPN West Washington Internal Medicine Los Angeles, California, United States, 90066 Pacific Palisades Pacific Palisades, California, United States, 90272 12th Street Clinic Santa Monica, California, United States, 90401 Wilshire Office Santa Monica, California, United States, 90401 CPN Santa Monica 15th Street Family Medicine Practice Santa Monica, California, United States, 90404 CPN/Santa Monica Parkside Santa Monica, California, United States, 90404 Santa Moica Bay Physicians/20th Street 10th Floor Santa Monica, California, United States, 90404 Santa Monica Bay Physicians Plaza Santa Monica, California, United States, 90404 Santa Monica Bay Physicians/20th Street 3rd Floor Santa Monica, California, United States, 90404 Santa Monica Internal Medicine Santa Monica, California, United States, 90404 UCLA Family Health Clinic Santa Monica, California, United States, 90404 Santa Moica Bay Physicians Ocean Park Santa Monica, California, United States, 90405 Jack H. Skirball Health Center Woodland Hills, California, United States, 91364 CPN Woodland Hills Practice Woodland Hills, California, United States, 91367 Collapse << |
| NCT00715624 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Canada Sanofi-Aventis Administrative Office Laval, Canada Chile Sanofi-Aventis Administrative Office Santiago, Chile Egypt Sanofi-Aventis Administrative Office Cairo, Egypt France Sanofi-Aventis Administrative Office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany India Sanofi-Aventis Administrative Office Mumbai, India Italy Sanofi-Aventis Administrative Office Milano, Italy Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Puerto Rico Sanofi-Aventis Administrative Office Puerto Rico, Puerto Rico Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey United Kingdom Sanofi-Aventis Administrative Office Guildford Surrey, United Kingdom Collapse << |
| NCT00751114 | - | - | Completed | - | - |
| NCT00714233 | - | - | Completed | - | - |
| NCT00219440 | Diabetes Mellitus | Phase 4 | Completed | - | United States, Louisiana ... more >> Pennington Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT00331851 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00715624 | - | - | Completed | - | - |
| NCT00714233 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | - |
| NCT00510588 | Stable Coronary Artery Disease... more >> Obesity Impaired Glucose Tolerance Collapse << | Not Applicable | Completed | - | Germany ... more >> University of Leipzig - heart center Leipzig, Germany, 04289 Collapse << |
| NCT01452451 | Diabetes Mellitus | Phase 2 | Completed | - | United States, Ohio ... more >> Ohio, Ohio, United States Collapse << |
| NCT00082407 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02803918 | Type 2 Diabetes Mellitus | Phase 1 | Recruiting | January 27, 2020 | United States, Louisiana ... more >> Investigational Site Number 840002 Recruiting Baton Rouge, Louisiana, United States, 70808 Investigational Site Number 840006 Recruiting New Orleans, Louisiana, United States, 70115-6969 United States, Pennsylvania Investigational Site Number 840009 Recruiting Philadelphia, Pennsylvania, United States, 19104 United States, Texas Investigational Site Number 840007 Recruiting Lufkin, Texas, United States, 75904 Mauritius Investigational Site Number 480001 Recruiting Phoenix, Mauritius Mexico Investigational Site Number 484001 Recruiting Monterrey, Mexico, 64460 Investigational Site Number 484002 Recruiting Puebla, Mexico, 72190 South Africa Investigational Site Number 710001 Recruiting Cape Town, South Africa, 7530 Spain Investigational Site Number 724002 Recruiting Barcelona, Spain, 08009 Investigational Site Number 724001 Recruiting Vitoria, Spain, 01009 Collapse << |
| NCT03196154 | - | - | Completed | - | Malaysia ... more >> Clinical Research Centre, Sarawak General Hospital Kuching, Sarawak, Malaysia, 93586 Collapse << |
| NCT00082407 | - | - | Completed | - | - |
| NCT01128153 | - | - | Completed | - | - |
| NCT01128153 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02446834 | Polycystic Ovary Syndrome | Not Applicable | Unknown | August 2017 | China, Shanghai ... more >> Renji Hospital Department of Endocrinology and Metabolism Recruiting Shanghai, Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Contact: Jing Ma, MD +86-15800983436 cherry1996@live.cn Collapse << |
| NCT00598793 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00371306 | Gestational Diabetes ... more >> Type 2 Diabetes Pregnancy Collapse << | Not Applicable | Unknown | September 2008 | United States, Tennessee ... more >> Regional Obstetrical Consultants Recruiting Chattanooga, Tennessee, United States, 37403 Collapse << |
| NCT01765894 | - | - | Completed | - | Denmark ... more >> XLab, Center for Healthy Aging, Department of Biomedical Sciences, University of Copenhagen Copenhagen, Denmark, 2200 Collapse << |
| NCT02964247 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02763007 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | September 2020 | Korea, Republic of ... more >> Seoul St Mary's Hospital, The Catholic University of Korea Recruiting Seoul, Korea, Republic of, 137-701 Contact: YOON HEE CHOI, MD Principal Investigator: Kun-Ho Yoon, MD, PhD Sub-Investigator: Yoon-Hee Choi, MD, PhD Sub-Investigator: Eun-Young Lee, MD, PhD Collapse << |
| NCT02919345 | Diabetes Mellitus, Type 2 ... more >> Coronary Artery Disease Carotid Artery Diseases Collapse << | Phase 4 | Unknown | October 2017 | Brazil ... more >> State University of Campinas Campinas, Sao Paulo, Brazil, 13083-887 Collapse << |
| NCT00838903 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00082381 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00838903 | - | - | Completed | - | - |
| NCT00849056 | - | - | Completed | - | - |
| NCT00849056 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00082381 | - | - | Completed | - | - |
| NCT01775176 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT02627027 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT00647023 | Polycystic Ovary Syndrome | Not Applicable | Terminated | - | - |
| NCT00035542 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT03364335 | Obesity | Phase 2 | Completed | - | United States, Alabama ... more >> AMR Mobile, Alabama, United States, 36608 United States, California Catalina Research Institute Montclair, California, United States, 91763 Northern California Research Sacramento, California, United States, 95821 United States, Idaho ACR Meridian, Idaho, United States, 83642 United States, Illinois Synexus Chicago, Illinois, United States, 60602 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Clinical Trials Management Metairie, Louisiana, United States, 70006 United States, Missouri Synexus Saint Louis, Missouri, United States, 63141 United States, Nevada AMR Las Vegas, Nevada, United States, 89119 United States, Ohio Rapid Medical Research Cleveland, Ohio, United States, 44122 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Premier Clarksville, Tennessee, United States, 37040 United States, Texas Synexus Dallas, Texas, United States, 75234 Synexus San Antonio, Texas, United States, 78229 United States, Utah ACR Jordan, Utah, United States, 84088 Collapse << |
| NCT03364335 | - | - | Completed | - | - |
| NCT02365233 | - | - | Terminated(IRB withheld the da... more >>ta due to inadequate supporting documentation) Collapse << | - | - |
| NCT02365233 | Type II Diabetes ... more >> Nonalcoholic Fatty Liver Collapse << | Phase 4 | Terminated(IRB withheld the da... more >>ta due to inadequate supporting documentation) Collapse << | - | United States, Texas ... more >> University of Texas Medical Branch -Galveston Galveston, Texas, United States, 77555 Collapse << |
| NCT00427154 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT03642600 | PCOS Obesity ... more >> Metabolic Syndrome Collapse << | Phase 4 | Not yet recruiting | December 30, 2019 | - |
| NCT00806234 | Psychotic Disorders | Phase 4 | Completed | - | United States, Maryland ... more >> University of Maryland Baltimore, Maryland, United States, 21201 Johns Hopkins Hospital Baltimore, Maryland, United States, 21205 United States, New York The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 United States, North Carolina University of North Carolina, Division of Child and Adolescent Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01089790 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT01258075 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | June 2019 | - |
| NCT01302379 | Breast Neoplasms | Not Applicable | Unknown | December 2016 | United States, California ... more >> Moores UCSD Cancer Center La Jolla, California, United States, 92093 Collapse << |
| NCT01717313 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01717313 | - | - | Completed | - | - |
| NCT00479466 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Sufficient data reg... more >>arding the dose-response to MK-0893 had been obtained from the first cohort of the study to assess the safety and efficacy) Collapse << | - | - |
| NCT00806234 | - | - | Completed | - | - |
| NCT01794143 | Type 2 Diabetes ... more >> Comparative Effectiveness of Glycemia-lowering Medications Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT00479466 | - | - | Terminated(Sufficient data reg... more >>arding the dose-response to MK-0893 had been obtained from the first cohort of the study to assess the safety and efficacy) Collapse << | - | - |
| NCT01792830 | Poor Glycemic Control | Phase 3 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory Midtown Hospital Atlanta, Georgia, United States, 30308 Emory University Hospital Atlanta, Georgia, United States, 30326 Collapse << |
| NCT02099110 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02099110 | - | - | Completed | - | - |
| NCT00970528 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Wonju-si, Gang''weondo, Korea, Republic of, 220-701 Busan, Korea, Korea, Republic of, 602-714 Gyeonggi-do, Korea, Korea, Republic of, 463-500 Gyeonggi-do, Korea, Korea, Republic of, 471-701 Pusan, Korea, Korea, Republic of, 602-739 Seoul, Korea, Korea, Republic of, 150-713 Daegu, Korea, Republic of, 705-717 Jeonju-si, Korea, Republic of, 456-712 Seongbuk-gu Seoul, Korea, Republic of, 136-705 Seoul, Korea, Republic of, 134-701 Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01792830 | - | - | Completed | - | - |
| NCT01526317 | Pharmacokinetics and Drug Inte... more >>raction of Crestor and Glucodown SR Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Severance Hospital, Yonsei University Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00482729 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02920801 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Shanghai ... more >> Fang Li Shanghai, Shanghai, China, 200032 Collapse << |
| NCT00482729 | - | - | Completed | - | - |
| NCT02058160 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02787551 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | November 20, 2018 | - |
| NCT02058160 | - | - | Completed | - | - |
| NCT00366301 | Type 2 Diabetes | Phase 4 | Terminated(Interim analyses de... more >>monstrated futility. Thus, recruitment curtailed 10/08.) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Collapse << |
| NCT02128932 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00366301 | - | - | Terminated(Interim analyses de... more >>monstrated futility. Thus, recruitment curtailed 10/08.) Collapse << | - | - |
| NCT00631033 | Obesity | Phase 2 | Completed | - | Netherlands ... more >> Rijnstate Hospital Arnhem, Netherlands Collapse << |
| NCT00099320 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03207698 | Furcation Defects | Phase 2 Phase 3 | Completed | - | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT01485614 | Diabetes Mellitus ... more >> Type 2 Diabetes Collapse << | Phase 3 | Active, not recruiting | October 17, 2019 | - |
| NCT00948324 | Type 2 Diabetes Mellitus | Phase 4 | Suspended | December 2021 | - |
| NCT02128932 | - | - | Completed | - | - |
| NCT00834652 | Depression | Phase 4 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT03230786 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Czechia ... more >> Vdoviak Miroslav MUDr. - Interní Ambulance Karlovy Vary, Czechia, 360 17 General University Hospital Prague 2, Czechia, 128 00 Institute for Clinical and experimental Medicine Praha 4, Czechia, 140 21 Interní a Diabetologická ambulance, Clintrial s.r.o Praha, Czechia, 100 00 Endokrinologicky Ústav Praha, Czechia, 113 94 Diabet2 s.r.o Praha, Czechia Interní a Diabetologická ambulance Uherské Hradiště, Czechia, 68 601 Diabetologická a Obezitologická ambulance České Budějovice, Czechia Denmark Bispebjerg Hospital, Endokrinologisk Afdeling Bispebjerg, Copenhagen NV, Denmark, 2400 Bioclinica Ballerup, Copenhagen, Denmark, 2750 Bioclinica Aalborg, Jutland, Denmark, 9000 Aarhus University Hospital, Endocrinlogy Department Aarhus, Jutland, Denmark, 8000 Sydvestjysk Sygehus Esbjerg, Jutland, Denmark, 6700 Bioclinica Vejle, Jutland, Denmark, 7100 Holbæk Sygehus Holbæk, Denmark, 4300 Moldova, Republic of Rtl Sm Srl/Scr Chisinau, Moldova, Republic of, MD2025 Rtl Sm Srl Chisinau, Moldova, Republic of, MD2025 Poland Centrum Badań Klinicznych PI-House sp. z o.o. Gdańsk, Poland, 80-546 Specjalistyczna Praktyka Lekarska Piotr Kubalski Grudziądz, Poland, 86-302 Medyczne Centrum Diabetologiczno Endokrynologiczno Metaboliczno Kraków, Poland, 31-261 Prywatny Gabinet Lekarski M. Horodecki (budynek Medicus) Opole, Poland, 45-367 Centrum Medyczne Hygea Tychy, Poland, 43-100 Centrum Medyczne AMED Warsaw, Poland, 01-518 Prywatny Gabinet Lekarski i Wizyty Lekarskie Jan Ruxer Łódź, Poland, 90-074 Romania S.C. Policlinica CCBR S.R.L. Bucharest, Romania, 030463 Institutului Național de Diabet, Nutriție și Boli Metabolice "Prof.Dr. N.C. Paulescu" Bucharest, Romania S.C Nicodiab S.R.L. Bucharest, Romania S.C. Sfinx Medica S.R.L. Constanţa, Romania, 900412 S.C. Mediab S.R.L. Targu Mures, Romania S.C. Mediab S.R.L. Târgu-Mureş, Romania United Kingdom St. Pancras London, United Kingdom, WC1X 8QD Collapse << |
| NCT00513630 | Type 2 Diabetes ... more >> Coronary Artery Disease Collapse << | Phase 4 | Completed | - | China ... more >> Shanghai Jiao Tong University School of Medicine Shanghai, China, 200025 Collapse << |
| NCT01357135 | - | - | Completed | - | - |
| NCT02160743 | Food Effect Study of CJ-30056 ... more >>20mg/500mg Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Busan Paik Hospital Busan, Korea, Republic of, 614-735 Collapse << |
| NCT00834652 | - | - | Completed | - | - |
| NCT01646320 | - | - | Completed | - | - |
| NCT01357135 | - | - | Completed | - | - |
| NCT01646320 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03283657 | PreDiabetes D... more >>iabetes Mellitus Diabetes Mellitus, Type 2 Body Weight Body Weight Changes Collapse << | Not Applicable | Enrolling by invitation | March 31, 2019 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00483392 | Insulin Resistance | Not Applicable | Completed | - | Slovenia ... more >> Department of Endocrinology, Diabetes and Metabolic diseases Ljubljana, Slovenia, 1525 Collapse << |
| NCT00641056 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00232583 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, Texas ... more >> University of Texas Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT00067951 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00641056 | - | - | Completed | - | - |
| NCT01064687 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00232583 | - | - | Completed | - | - |
| NCT03593135 | Randomized Controlled Trial ... more >> Diabetes Mellitus Collapse << | Not Applicable | Completed | - | Pakistan ... more >> Shaikh Zayed Medical Complex Lahore, Punjab, Pakistan, 54000 Collapse << |
| NCT00562172 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-aventis Seoul, Korea, Republic of Collapse << |
| NCT01238978 | Type 2 Diabetes Mellitus ... more >> Elderly Collapse << | Phase 4 | Completed | - | France ... more >> Novartis Investigative Site Armentieres, France, 59208 Novartis Investigative Site Brest, France, 29200 Novartis Investigative Site Caen, France, 14000 Novartis Investigative Site Creil, France, 60100 Novartis Investigative Site #2 Paris, France, 75012 Novartis Investigative Site Paris, France, 75020 Novartis Investigative Site Pau, France, 64000 Novartis Investigative Site St Jean de la Ruelle, France, 45140 Novartis Investigative Site Strasbourg, France, 67000 Novartis Investigative Site Strasbourg, France Novartis Investigative Site Toulouse, France, 31200 Novartis Investigative Site Tours, France, 37000 Novartis Investigative Site Venissieux, France, 69200 Novartis Investigative Site #2 Venissieux, France Novartis Investigative Site Versailles, France, 78000 Collapse << |
| NCT00086515 | - | - | Completed | - | - |
| NCT01755468 | Type 2 Diabetes | Phase 3 | Completed | - | Canada, Ontario ... more >> Mount Sinai Hospital Toronto, Ontario, Canada, M5G1X5 Collapse << |
| NCT01006603 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01064687 | - | - | Completed | - | - |
| NCT01006603 | - | - | Completed | - | - |
| NCT01340768 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00138567 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Investigative Site East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative site Investigative Centers, Germany Collapse << |
| NCT00332085 | Type 2 Diabetes | Phase 1 | Terminated(Issues w/recruitmen... more >>t) Collapse << | - | United States, Washington ... more >> Bastyr University Campus SPR Kenmore, Washington, United States, 98036 Bastyr Center for Natural Health Seattle, Washington, United States Collapse << |
| NCT01814748 | - | - | Completed | - | - |
| NCT01814748 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01340768 | - | - | Completed | - | - |
| NCT00309608 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01167881 | - | - | Completed | - | - |
| NCT00099892 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT01167881 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00375388 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Netherlands ... more >> Bethesda General Hospital and Bethesda Diabetes Center Hoogeveen, Drenthe, Netherlands, 7909 AA Collapse << |
| NCT00309608 | - | - | Completed | - | - |
| NCT01984606 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn | February 2017 | - |
| NCT00545584 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00308373 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00755846 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00541450 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02229396 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00541450 | - | - | Completed | - | - |
| NCT03430856 | Type2 Diabetes Mellitus | Phase 2 Phase 3 | Recruiting | September 2020 | India ... more >> Diacon Hospital Recruiting Bangalore, Karnataka, India, 560010 Contact: Aravind Aravind, MBBS, DNB 8028085305 draravind@hotmail.com Contact: Geethanjali Rangaswamy 9008465877 geethanjali.kst@gmail.com Collapse << |
| NCT00395343 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00395343 | - | - | Completed | - | - |
| NCT01278160 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100029 Collapse << |
| NCT02229396 | - | - | Completed | - | - |
| NCT00755846 | - | - | Completed | - | - |
| NCT01144975 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Mexico ... more >> Mexico City, Mexico Collapse << |
| NCT01278160 | - | - | Completed | - | - |
| NCT01789957 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00099619 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Australia, New South Wales ... more >> Research Site Westmead, New South Wales, Australia Australia, South Australia Research Site Daw Park, South Australia, Australia Research Site Fullarton, South Australia, Australia Australia, Victoria Research Site Box Hill, Victoria, Australia Research Site East Ringwood, Victoria, Australia Greece Research Site Athens, Greece Research Site Piraeus, Greece Research Site Thessaloniki, Greece Hungary Research Site Budapest, Hungary Research Site Gyula, Hungary Research Site Pecs, Hungary Research Site Veszprem, Hungary Research Site Zalaegerszeg, Hungary Italy Research Site Bari, Italy Research Site Bergamo, Italy Research Site Catania, Italy Research Site Milan, Italy Research Site Perugia, Italy Research Site Rome, Italy Mexico Research Site Guadalajara, Jalisco, Mexico Research Site Zapopan, Jalisco, Mexico Research Site Monterrey, N.l., Mexico Research Site Mexico City, Mexico Poland Research Site Bydgoszcz, Poland Research Site Gdansk, Poland Research Site Lublin, Poland Collapse << |
| NCT03642717 | - | - | Recruiting | March 31, 2020 | Korea, Republic of ... more >> Hyewon Medical Foundation Sejong Hospital Recruiting Bucheon, Korea, Republic of, 14754 Inje University Busan Paik Hospital Recruiting Busan, Korea, Republic of, 47392 Keimyung University Dongsan Hospital Recruiting Daegu, Korea, Republic of, 41931 Inha University Hospital Recruiting Incheon, Korea, Republic of, 22332 Seoul Medical Center Recruiting Seoul, Korea, Republic of, 02053 Hongik Hospital Recruiting Seoul, Korea, Republic of, 07937 Ajou University Hospital Recruiting Suwon, Korea, Republic of, 16499 Yonsei University Wonju Severance Christian Hospital Recruiting Wonju, Korea, Republic of, 26426 Collapse << |
| NCT01071850 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01193296 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> Investigative Site Bondy, France Investigative Site Corbeil, France Investigative Site Marseille, France Investigative Site Nancy, France Investigative Site Paris, France Investigative Site Toulouse, France Investigative Site Venissieux, France Collapse << |
| NCT02969798 | Diabetes Mellitus, Type 2 ... more >> Impaired Glucose Tolerance (IGT) Impaired Fasting Glucose (IFG) Collapse << | Not Applicable | Recruiting | July 2020 | United States, Texas ... more >> The University of Texas Health Science Center at San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Ralph A DeFronzo, MD 210-567-6691 defronzo@uthscsa.edu Contact: Monica Palomo, BS 210-567-6710 palomom@uthscsa.edu Principal Investigator: Ralph A DeFronzo, MD Sub-Investigator: Eugenio Cersosimo, MD Collapse << |
| NCT02020616 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Terminated(Lack of Efficacy) | - | United States, California ... more >> Orange County Research Center Orange, California, United States, 92868 United States, Florida Miami Research Associates Miami, Florida, United States, 33143 Compass Research Orlando, Florida, United States, 32806 United States, New York Clinilabs, Inc (New York) New York, New York, United States, 10019 United States, Texas Dallas Diabetes Endocrine Center Dallas, Texas, United States, 75230 Collapse << |
| NCT02712632 | - | - | Recruiting | January 2019 | United Kingdom ... more >> University of Dundee Recruiting Dundee, Tayside, United Kingdom, DD1 9SY Contact: Louise Greig, RGN, MBA 01382 383208 louise.greig@nhs.net Collapse << |
| NCT00699322 | Type 2 Diabetes | Phase 4 | Unknown | June 2010 | Korea, Republic of ... more >> Kangnam St. Mary's hospital Recruiting Seoul, Korea, Republic of, 137-701 Sub-Investigator: Seung-Hwan Lee, M.D. Sub-Investigator: Eun-Sook Kim, M.D. Principal Investigator: Kun-Ho Yoon, M.D., Ph.D. Collapse << |
| NCT02020616 | - | - | Terminated(Lack of Efficacy) | - | - |
| NCT00316758 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT02686177 | Type 2 Diabetes | Phase 4 | Recruiting | June 18, 2019 | France ... more >> Nutrition department, pole ENDO - Assistance Publique Hôpitaux Marseille Recruiting Marseille, France, 13005 Contact: Anne DUTOUR, MD, PhD 33 4 91 38 29 63 anne.dutour@ap-hm.fr Collapse << |
| NCT00135330 | - | - | Completed | - | - |
| NCT00135330 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Phoenix, Arizona, United States United States, California Research Site San Francisco, California, United States United States, Florida Research Site Jacksonville, Florida, United States Research Site Orlando, Florida, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Hawaii Research Site Honolulu, Hawaii, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Massachusetts Research Site Boston, Massachusetts, United States Research Site Pittsfield, Massachusetts, United States United States, Minnesota Research Site Minneapolis, Minnesota, United States Research Site Rochester, Minnesota, United States United States, New York Research Site Bronx, New York, United States Research Site Syracuse, New York, United States United States, Ohio Research Site Cincinatti, Ohio, United States United States, Texas Research Site League City, Texas, United States Research Site New Braunfels, Texas, United States Research Site San Antonio, Texas, United States United States, Washington Research Site Renton, Washington, United States Research Site Spokane, Washington, United States Collapse << |
| NCT01734785 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01339143 | Diabetes Mellitus ... more >> Oxidative Stress Collapse << | Phase 4 | Unknown | May 2013 | Korea, Republic of ... more >> Korea University Anam Hospital Recruiting Seoul, Korea, Republic of Collapse << |
| NCT01734785 | - | - | Completed | - | - |
| NCT00494312 | Diabetes Mellitus ... more >> Liver Collapse << | Phase 4 | Completed | - | - |
| NCT01356381 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United Kingdom ... more >> Novartis Investigative Site Newcastle Upon Tyne, United Kingdom, NE4 5PL Collapse << |
| NCT02682563 | Diabetes Mellitus, Type 2 ... more >> Diabetic Nephropathies Collapse << | Phase 4 | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Noord-Holland, Netherlands, 1081 HV Collapse << |
| NCT00817271 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT01278485 | - | - | Completed | - | - |
| NCT01278485 | - | - | Completed | - | - |
| NCT00069836 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01674348 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(Due to non-recruitm... more >>ent, the study is being halted. There are no major safety or tolerability concerns in the study conducted so far.) Collapse << | - | Canada, Ontario ... more >> LMC Endocrinolgy Centres Ltd Toronto, Ontario, Canada, M4G 3E8 Collapse << |
| NCT00618007 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01576328 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | United States, California ... more >> SC Clinical Research Garden Grove, California, United States, 92844 United States, Florida Diabetes Research Institute Miami, Florida, United States, 33136 Compass Research Orlando, Florida, United States, 32806 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States Tulane University Medical Center New Orleans, Louisiana, United States United States, Montana Big Sky Clinical Research Butte, Montana, United States, 59701 United States, Nevada Desert Endocrinology Clinical Research Center-Henderson Henderson, Nevada, United States, 89052 Alliance Against Diabetes/AAD Clinical Research Las Vegas, Nevada, United States, 89101 United States, New Hampshire Active Practices and Research Newington, New Hampshire, United States, 03801 United States, Ohio The Carl and Edyth Lindner Center for Research and Education at The Christ Hospital Cincinnati, Ohio, United States, 45219 Providence Health Partners - Center for Clinical Reseach Dayton, Ohio, United States, 45439 United States, Texas Dallas Diabetes and Endocrine Center Dallas, Texas, United States, 75230 West Houston Clinical Research Services Houston, Texas, United States Paragon Research Center San Antonio, Texas, United States, 78205 United States, Utah Wasatch Clinical Research Salt Lake City, Utah, United States United States, Virginia National Clinical Research - Norfolk, Inc Norfolk, Virginia, United States, 23502 National Clinical Research - Richmond, Inc. Richmond, Virginia, United States, 23294 United States, Washington Capital Clinical Research Center Olympia, Washington, United States, 98502 Collapse << |
| NCT02401880 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Profil Mainz GmbH & Co. KG Mainz, Germany, 55116 Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT00819884 | Type II Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00363948 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Chubu Region Chubu, Japan Chugoku Region Chugoku, Japan Hokkaido Region Hokkaido, Japan Hokuriku Region Hokuriku, Japan Kanto Region Kanto, Japan Kinki Region Kinki, Japan Kyushu Region Kyushu, Japan Tohoku Region Tohoku, Japan Collapse << |
| NCT01017302 | Diabetes Mellitus Type 2 | Phase 1 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States, 78229 Collapse << |
| NCT00679939 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01455922 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT00679939 | - | - | Completed | - | - |
| NCT01455883 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT00174642 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01455870 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02653599 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01455909 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT01045707 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02462369 | Microalbuminuria ... more >> Microalbuminuria /Creatinine Ratios ACR Collapse << | Phase 4 | Unknown | October 2017 | - |
| NCT03029702 | Gestational Diabetes ... more >> Diabetes, Gestational Pregnancy in Diabetes Collapse << | Phase 4 | Recruiting | March 30, 2021 | United States, Pennsylvania ... more >> Magee Womens Hospital of UPMC Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Maisa Feghali, MD 412-641-4874 feghalim@upmc.edu Principal Investigator: Maisa Feghali, MD Collapse << |
| NCT02111096 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(The overall benefit... more >>-risk profile did not support continued development of LY2409021 for type 2 diabetes.) Collapse << | - | - |
| NCT01045707 | - | - | Completed | - | - |
| NCT02887625 | Type 2 Diabetes | Not Applicable | Unknown | May 2017 | Qatar ... more >> Hamad General Hospital Recruiting Doha, Qatar Contact: Osama Mujahed, MD 66703172 pvedromigahid@gmail.com Contact: Ayman Mujahed, MD 44391208 ymndodo20@gmail.com Collapse << |
| NCT02111096 | - | - | Terminated(The overall benefit... more >>-risk profile did not support continued development of LY2409021 for type 2 diabetes.) Collapse << | - | - |
| NCT00097279 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01885260 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Alabama ... more >> Isis Investigational Site Birmingham, Alabama, United States, 35216 Isis Investigational Site Muscle Shoals, Alabama, United States, 35662 United States, California Isis Investigational Site Anaheim, California, United States, 92801 Isis Investigational Site Chino, California, United States, 91710 Isis Investigational Site Los Angeles, California, United States, 90057 United States, Florida Isis Investigational Site Deland, Florida, United States, 32720 Isis Investigational Site Ft. Lauderdale, Florida, United States, 33306 Isis Investigational Site Miami, Florida, United States, 33143 United States, Georgia Isis Investigational Site Marietta, Georgia, United States, 30066 United States, Nevada Isis Investigational Site Las Vegas, Nevada, United States, 89101 United States, Ohio Isis Investigational Site Cincinnati, Ohio, United States, 45245 United States, Oklahoma Isis Investigational Site Oklahoma City, Oklahoma, United States, 73103 United States, Oregon Isis Investigational Site Eugene, Oregon, United States, 97404 United States, Texas Isis Investigational Site Corpus Christi, Texas, United States, 78404 Isis Investigational Site Katy, Texas, United States, 77450 United States, Utah Isis Investigational Site Salt Lake City, Utah, United States, 84107 United States, Washington Isis Investigational Site Renton, Washington, United States, 98057 Isis Investigational Site Spokane, Washington, United States, 99202 South Africa Isis Investigational Site Lenasia South, Gauteng, South Africa, 1829 Isis Investigational Site Soweto, Gauteng, South Africa, 1818 Isis Investigational Site Durban, KwaZulu-Natal, South Africa, 4091 Isis Investigational Site Cape Town, Western Cape, South Africa, 7700 Isis Investigational Site Parow, Cape Town, Western Cape, South Africa, 7500 Isis Investigational Site Western Cape, South Africa, 7130 Collapse << |
| NCT02567994 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02824003 | Type 2 Diabetes | Phase 2 | Completed | - | Austria ... more >> Ionis Investigator Site Vienna, Austria, A-1130 Collapse << |
| NCT02583919 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> Ionis Investigational Site Greenbrae, California, United States, 94904 Ionis Investigational Site Huntington Park, California, United States, 90255 Ionis Investigational Site La Mesa, California, United States, 91942 Ionis Investigational Site Los Angeles, California, United States, 90057 Isis Investigational Site Spring Valley, California, United States, 91978 United States, Florida Ionis Investigational Site Port Orange, Florida, United States, 32127 United States, Georgia Ionis Investigational Site Marietta, Georgia, United States, 30060 United States, Maryland Ionis Investigational Site Oxon Hill, Maryland, United States, 20745 United States, New York Ionis Investigational Site New York, New York, United States, 10016 United States, North Carolina Ionis Investigational Site Greensboro, North Carolina, United States, 27410 Ionis Investigational Site Morehead City, North Carolina, United States, 28557 United States, Ohio Ionis Investigational Site Cincinnati, Ohio, United States, 45219 Ionis Investigational Site Cincinnati, Ohio, United States, 45245 United States, Oregon Ionis Investigational Site Eugene, Oregon, United States, 97404 United States, South Carolina Ionis Investigational Site Spartanburg, South Carolina, United States, 29303 United States, Texas Ionis Investigational Site Houston, Texas, United States, 77036 Ionis Investigational Site Houston, Texas, United States, 77074 Ionis Investigational Site Katy, Texas, United States, 77450 United States, Washington Ionis Investigational Site Olympia, Washington, United States, 98502 Collapse << |
| NCT02475070 | Type 2 Diabetes | Phase 4 | Completed | - | Sweden ... more >> Lund University Lund, Sweden, 22184 Collapse << |
| NCT01933672 | - | - | Completed | - | - |
| NCT02325960 | Type 2 Diabetes | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT00942318 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> University Hospital Toulouse Toulouse, France, 31059 Collapse << |
| NCT01933672 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Florida ... more >> Avail Clinical Research, LLC DeLand, Florida, United States, 32720 Miami Research Associates, Inc. South Miami, Florida, United States, 33143 MRA Clinical Research, LLC South Miami, Florida, United States, 33143 United States, North Carolina High Point Clinical Trials Center, LLC High Point, North Carolina, United States, 27265 United States, Ohio Community Research Cincinnati, Ohio, United States, 45255 United States, Texas Clinical Trials of Texas, Inc San Antonio, Texas, United States, 78229 Diabetes and Glandular Disease Clinic San Antonio, Texas, United States, 78229 United States, Wisconsin Spaulding Clinical Research, LLC West Bend, Wisconsin, United States, 53095 Collapse << |
| NCT00032487 | - | - | Completed | - | - |
| NCT00032487 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02303405 | Diabetes Mellitus Type 2 With ... more >>Hyperglycemia Collapse << | Phase 2 | Terminated(Investigator decisi... more >>on) Collapse << | - | United States, California ... more >> Charles Drew University of Medicine and Science Los Angeles, California, United States, 90059 Collapse << |
| NCT00823680 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United States, California ... more >> Chula Vista, California, United States, 91911 United States, Florida Miami, Florida, United States, 330014 Austria Graz, Austria, 8036 Germany München, Germany, 80636 Neuss, Germany, 41460 Collapse << |
| NCT00506194 | Type 2 Diabetes | Not Applicable | Completed | - | Taiwan ... more >> Division of Endocrinology and Metabolism, Department of Medicine Taipei, Taiwan, 112 Collapse << |
| NCT03495102 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | October 7, 2019 | - |
| NCT02546609 | NAFLD | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 University of California San Diego San Diego, California, United States, 92103 United States, Colorado Rocky Mountain Research Wheat Ridge, Colorado, United States, 80033 United States, Georgia Atlanta Gastroenterology Associates Atlanta, Georgia, United States, 30312 GI Specialists of Georgia Marietta, Georgia, United States, 30060 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, North Carolina University of North Carolina Chapel Hill Chapel Hill, North Carolina, United States, 27599 United States, Ohio Sterling Research Cincinnati, Ohio, United States, 45246 United States, Tennessee Premier Clinical Research Clarksville, Tennessee, United States, 37043 Gastro One Germantown, Tennessee, United States, 38138 Quality Medical Research Nashville, Tennessee, United States, 37211 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT00461643 | Anovulatory Infertility Relate... more >>d to Polycystic Ovary Syndrome Collapse << | Phase 4 | Unknown | - | Italy ... more >> "Pugliese" Hospital Recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39 0961 883234 angela.falbo@libero.it Principal Investigator: Stefano Palomba, MD "Pugliese" Hospital Recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39 0961 883234 zullo@unicz.it Principal Investigator: Stefano Palomba, MD Principal Investigator: Francesco Orio, MD Principal Investigator: Achille Tolino, MD Principal Investigator: Pasquale Mastrantonio, MD Principal Investigator: Brian Dale, MD Principal Investigator: Ettore Cittadini, MD Principal Investigator: Andrea Riccardo Genazzani, MD Pugliese-Ciaccio Hospital Recruiting Catanzaro, Italy, 88100 Contact: , MD Collapse << |
| NCT00633425 | Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> Sanofi-Aventis Paris, France Collapse << |
| NCT01219582 | - | - | Completed | - | India ... more >> Many Locations, India Collapse << |
| NCT02256189 | Type 2 Diabetes | Phase 4 | Completed | - | Sweden ... more >> Skane University Hospital Malmö Malmö, Sweden, 20502 Collapse << |
| NCT00818077 | - | - | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT00543751 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT02546609 | - | - | Completed | - | - |
| NCT01595789 | Coronary Artery Disease ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Denmark ... more >> Copenhagen University Hospital, Bispebjerg Copenhagen, Bispebjerg, Denmark, 2400 Collapse << |
| NCT00543738 | Diabetes Mellitus | Phase 3 | Terminated | - | - |
| NCT02492763 | Type II Diabetes Mellitus | Phase 2 | Terminated | - | - |
| NCT00780715 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital & Medical School Dundee, United Kingdom, DD1 9SY Collapse << |
| NCT02492763 | - | - | Terminated | - | - |
| NCT00123110 | Insulin Resistance ... more >> Postmenopause Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Clinical and Translational Research Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01990469 | T2DM | Phase 3 | Completed | - | - |
| NCT02496000 | Diabetes Type 2 | Phase 2 | Completed | - | - |
| NCT00780715 | - | - | Completed | - | - |
| NCT02496000 | - | - | Completed | - | - |
| NCT02449187 | Diabetes Hype... more >>rlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inha University Hospital Incheon, Korea, Republic of Collapse << |
| NCT00347100 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China ... more >> Sanofi-Aventis Beijing, China Collapse << |
| NCT03239119 | Type2 Diabetes Mellitus | Phase 3 | Not yet recruiting | January 15, 2019 | - |
| NCT01025765 | Chronic Hepatitis C | Phase 4 | Unknown | December 2012 | Taiwan ... more >> National Taiwan University Hospital Department of Internal Medicine, Taipei, Taiwan, 10002 Collapse << |
| NCT02726490 | Gestational Diabetes | Not Applicable | Terminated(Lack of Recruitment... more >>) Collapse << | - | United States, Texas ... more >> TTUHSC El Paso El Paso, Texas, United States, 79905 Collapse << |
| NCT00964184 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Fortis Flt Lt Rajan Dhall Hospital New Delhi, Delhi, India, 110070 Collapse << |
| NCT02042664 | Obesity Diabe... more >>tes Collapse << | Phase 3 | Completed | - | France ... more >> Assistance Publique Hopitaux de Marseille Marseille, France, 13354 Collapse << |
| NCT00541775 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03130426 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 2019 | Canada, Alberta ... more >> University of Calgary Recruiting Calgary, Alberta, Canada, T2T 5C7 Contact: Ron Sigal, MD 403-955-8107 rsigal@ucalgary.ca Canada, Ontario LMC Recruiting Burlington, Ontario, Canada, M4G 3E8 Contact: Mohammed Azharuddin, MD Joanne Liutkus Recruiting Cambridge, Ontario, Canada, N1R 7L6 Contact: Danielle Gelinas 519-624-8977 McMaster University Recruiting Hamilton, Ontario, Canada, L8N 3Z5 Contact: Natalia McInnes, MD 905-521-2100 ext 73794 natalia.mcinnes@mcmaster.ca Contact: Ada Smith 905-521-2100 ext 22205 smithad@mcmaster.ca St. Joseph's Hospital Recruiting London, Ontario, Canada, N6A 4V2 Contact: Irene Hramiak, MD 519-646-6353 Irene.Hramiak@sjhc.london.on.ca Western University Recruiting London, Ontario, Canada, N6G 2M1 Contact: Stewart Harris, MD 519-661-2111 Stewart.Harris@schulich.uwo.ca The Ottawa Hospital Recruiting Ottawa, Ontario, Canada, K1Y 4E9 Contact: Heather Lochnan, MD hlochnan@toh.ca Canada, Quebec McGill University Recruiting Montreal, Quebec, Canada, H4A 3J1 Contact: Angie Lombardo 514-934-1934 ext 36492 angie.lombardo@muhc.mcgill.ca Collapse << |
| NCT00971659 | Type 2 Diabetes | Phase 1 | Completed | - | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT02274740 | Type II Diabetes Mellitus | Phase 2 | Terminated(Sponsor decision no... more >>t related to safety reasons) Collapse << | - | - |
| NCT00541775 | - | - | Completed | - | - |
| NCT01937598 | Type 2 Diabetes | Phase 3 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Collapse << |
| NCT02561130 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | October 2018 | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2T 5C7 Canada, Manitoba Health Science Centre Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario McMaster University Hamilton, Ontario, Canada, L8N 3Z5 St. Joseph's Hospital London, Ontario, Canada, N6A 4V2 Western University London, Ontario, Canada, N6G 2M1 LMC Toronto, Ontario, Canada, M4G 3E8 St. Michaels's Hospital Toronto, Ontario, Canada, M5C 2T2 Collapse << |
| NCT03378297 | Ovarian Cancer | Early Phase 1 | Recruiting | June 1, 2020 | Norway ... more >> Helse Bergen HF, Haukeland University Hospital Recruiting Bergen, Norway, 5021 Contact: Liv Cecilie V Thomsen, MD, PhD +47 55975000 Liv.Vestrheim@uib.no Contact: Line Bjørge, MD, PhD +47 55975000 line.bjorge@uib.no Principal Investigator: Line Bjørge, MD, PhD Sub-Investigator: Liv Cecilie V Thomsen, MD, PhD Sub-Investigator: Kathrine Woie, MD, PhD Sub-Investigator: Bjørn Tore Gjertsen, MD, PhD Oslo University Hospital Recruiting Oslo, Norway, 0424 Contact: Erik Rokkones, MD, PhD +47 915 02770 erk@ous-hf.no Contact: Olysea Solheim, MD, PhD +47 915 02770 olsolh@ous-hf.no Principal Investigator: Erik Rokkones, MD, PhD Sub-Investigator: Olysea Solheim, MD, PhD Helse Stavanger HF, Stavanger University Hospital Recruiting Stavanger, Norway, 4068 Contact: Bent Fiane, MD +47 51518000 bent.eltvedt.fiane@sus.no Contact: Cecilie F Torkildsen, MD +47 51518000 cecilie.torkildsen@gmail.com Principal Investigator: Bent Fiane, MD Sub-Investigator: Cecilie F Torkildsen, MD Sub-Investigator: Ragnar K Sande, MD, PhD Collapse << |
| NCT01937598 | - | - | Completed | - | - |
| NCT00673465 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT00673465 | - | - | Completed | - | - |
| NCT02587741 | Diabetic Retinopathy | Early Phase 1 | Recruiting | July 2025 | China, Guangdong ... more >> the third affiliated hospital of Sun yet-san university Recruiting Guangzhou, Guangdong, China, 510000 Contact: Zhu Bilian, master +8613580364394 bilianzhu@qq.com Contact: Tang Xixiang, master +8613570434387 txx_8711@126.com Collapse << |
| NCT00374907 | Type 2 Diabetes | Phase 3 | Completed | - | United States, California ... more >> Va San Diego Healthcare System San Diego, California, United States, 92161 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 United States, Texas Diabetes & Glandular Disease Research Assoc,, Inc. San Antonio, Texas, United States, 78229 Collapse << |
| NCT00374907 | - | - | Completed | - | - |
| NCT01965509 | Type 2 Diabetes | Phase 2 | Completed | - | China ... more >> China-Japan Friendship Hospital Beijing, China Collapse << |
| NCT03617458 | Pulmonary Artery Hypertension | Phase 2 | Recruiting | December 2022 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << |
| NCT03072485 | Aging | Phase 1 | Recruiting | December 31, 2017 | United States, California ... more >> Stanford Dermatology Recruiting Redwood City, California, United States, 94603 Contact: Anne Chang, MD 650-721-7151 alschang@stanford.edu Contact: Michaella Montana, BS 650 721 7159 mmontana@stanford.edu Collapse << |
| NCT01477853 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(The study was termi... more >>nated early by the Sponsor for business reasons.) Collapse << | - | - |
| NCT01046422 | Diabetes Type 2 | Phase 2 | Completed | - | United States, California ... more >> Marina Raikhel, Md Lomita, California, United States, 90717 United States, Nevada Nevada Alliance Against Diabetes Las Vegas, Nevada, United States, 89101 Australia, Queensland Local Institution Caboolture, Queensland, Australia, 4510 Local Institution Meadowbrook, Queensland, Australia, 4131 Australia, South Australia Local Institution Daw Park, South Australia, Australia, 5041 Australia, Victoria Local Institution Geelong, Victoria, Australia, 3220 Australia, Western Australia Local Institution Nedlands, Western Australia, Australia, 6009 Canada, Manitoba Local Institution Winnipeg, Manitoba, Canada, R3P 1R9 Canada, Newfoundland and Labrador Local Institution Mount Pearl, Newfoundland and Labrador, Canada, A1N 1W7 Canada, Ontario Local Institution Brampton, Ontario, Canada, L6T 3J1 Local Institution Toronto, Ontario, Canada, M4G 3E8 Canada, Prince Edward Island Local Institution Charlottetown, Prince Edward Island, Canada, C1A 5Y9 Canada, Quebec Local Institution Drummondville, Quebec, Canada, J2B 7T1 Local Institution Lachine, Quebec, Canada, H8S 2E4 Local Institution Laval, Quebec, Canada, H7T 2P5 Local Institution Montreal, Quebec, Canada, H3J 2V5 Korea, Republic of Local Institution Incheon, Korea, Republic of, 405-760 Local Institution Suwon, Korea, Republic of, 443-721 Collapse << |
| NCT01477853 | - | - | Terminated(The study was termi... more >>nated early by the Sponsor for business reasons.) Collapse << | - | - |
| NCT00694759 | Polycystic Ovary Syndrome | Not Applicable | Unknown | July 2011 | United States, Oregon ... more >> Oregon Health & Science University Recruiting Portland, Oregon, United States, 97239 Principal Investigator: Bethany J. Klopfenstein, MD Collapse << |
| NCT02426294 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | May 2016 | Korea, Republic of ... more >> Pusan National University Hospital Recruiting Busan, Korea, Republic of, 602-739 Contact: Sang Soo Kim, MD., PhD. +82-51-240-7228 drsskim7@gmail.com Contact: Jong Ho Kim, MD. +82-51-240-7983 bedaya@hanmail.net Collapse << |
| NCT01224366 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00709917 | - | - | Completed | - | Belgium ... more >> Brussels, Belgium, 1070 Luxembourg Luxembourg, Luxembourg Collapse << |
| NCT02291510 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT01035528 | Coronary Artery Disease ... more >> Type 2 Diabetes Collapse << | Phase 4 | Unknown | August 2010 | Germany ... more >> Staedt. Klinikum Muenchen Bogenhausen Munich, Bavaria, Germany, 81925 Collapse << |
| NCT00150410 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00545584 | - | - | Completed | - | - |
| NCT00601250 | - | - | Completed | - | - |
| NCT00601250 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01195090 | Type 2 Diabetes | Phase 4 | Completed | - | Taiwan ... more >> Division of Endocrinology and Metabolism, Department of Internal Medicine, Mackay Memorial Hospital Taipei, Taiwan, 10449 Collapse << |
| NCT02291510 | - | - | Completed | - | - |
| NCT00471523 | Infertility P... more >>olycystic Ovary Syndrome Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese Hospital" Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT00361738 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Argentina ... more >> Sanofi-Aventis Administrative Office San Isidro, Argentina Australia, New South Wales Sanofi-Aventis Administrative Office Cove, New South Wales, Australia Belgium Sanofi-Aventis Administrative Office Diegem, Belgium Chile Sanofi-Aventis Administrative Office Santiago, Chile Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark France Sanofi-Aventis Administrative Office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Poland Sanofi-Aventis Administrative Office Warszawa, Poland South Africa Sanofi-Aventis Administrative Office Midrand, South Africa Collapse << |
| NCT03060980 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Decision by Sponsor... more >>) Collapse << | - | - |
| NCT00602472 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00602472 | - | - | Completed | - | - |
| NCT03061214 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | April 17, 2019 | - |
| NCT01033773 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, District of Col... more >>umbia Washington Hospital Center Washington, District of Columbia, United States, 20010 Collapse << |
| NCT00686634 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, California ... more >> Charles Drew University of Medicine and Science Los Angeles, California, United States, 90059 Collapse << |
| NCT03053518 | Type2 Diabetes | Not Applicable | Recruiting | December 2019 | United States, New York ... more >> New York University School of Medicine Recruiting New York, New York, United States, 10016 Contact: Ram Jagannathan Ram.Jagannathan@nyumc.org Principal Investigator: Mary Sevick, MD Collapse << |
| NCT01195090 | - | - | Completed | - | - |
| NCT00686634 | - | - | Completed | - | - |
| NCT00694057 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Anaheim, California, United States, 92805 Beverly Hills, California, United States, 90211 Buena Park, California, United States, 90620 Burbank, California, United States, 91505 Carmichael, California, United States, 95608 Escondido, California, United States, 92025 Fresno, California, United States, 93726 Huntington Park, California, United States, 90255 Long Beach,, California, United States, 90806 Los Angeles, California, United States, 90057 National City, California, United States, 91950 Roseville, California, United States, 95661 United States, Illinois Chicago, Illinois, United States, 60616 United States, Louisiana Baton Rouge, Louisiana, United States, 70808 United States, Missouri St. Louis, Missouri, United States, 63141 United States, Nevada Las Vegas, Nevada, United States, 89106 Las Vegas, Nevada, United States, 89148 United States, Texas El Paso, Texas, United States, 79905 San Antonio, Texas, United States, 78229 United States, Utah Salt Lake City, Utah, United States, 84107 United States, Wisconsin Kenosha, Wisconsin, United States, 53142 Collapse << |
| NCT03646799 | Type II Diabetes | Phase 1 | Recruiting | September 14, 2018 | Korea, Republic of ... more >> Yonsei University Severance Hospital Recruiting Soeul, Korea, Republic of Contact: Min Soo Park, Ph.D. M.D +82 2 2228 0400 minspark@yuhs.ac Principal Investigator: Min Soo Park, Ph.D. M.D Collapse << |
| NCT02941367 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Investigational Site Number 356005 Ahmedabad, India, 380007 Investigational Site Number 356002 Bangalore, India, 560043 Investigational Site Number 356015 Bangalore, India, 560055 Investigational Site Number 356008 Bangalore, India, 560060 Investigational Site Number 356009 Hyderabad, India, 500001 Investigational Site Number 356018 Hyderabad, India, 500004 Investigational Site Number 356010 Hyderabad, India, 500072 Investigational Site Number 356003 Hyderabad, India, 500095 Investigational Site Number 356007 Jaipur, India Investigational Site Number 356019 Madurai, India, 625020 Investigational Site Number 356014 Mumbai, India, 400007 Investigational Site Number 356022 Nagpur, India, 440010 Israel Investigational Site Number 376001 Haifa, Israel, 31096 Investigational Site Number 376002 Safed, Israel, 13100 Kuwait Investigational Site Number 414001 Kuwait, Kuwait Turkey Investigational Site Number 792002 Zonguldak, Turkey Collapse << |
| NCT02638805 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01162772 | - | - | Unknown | - | Austria ... more >> Medical University of Viemma Recruiting Vienna, Austria, 1090 Contact: Jeanette Strametz-Juranek, MD 0043140400 ext 4618 jeanette.strametz-juranek@meduniwien.ac.at Contact: Alexandra Kautzky-Willer, MD 0140400 ext 4316 alexandra.kautzky-willer@meduniwien.ac.at Principal Investigator: Jeanette Strametz-Juranek, MD Principal Investigator: Alexandra Kautzky-Willer, MD Sub-Investigator: Alia Sabri, MD Sub-Investigator: Lana Kosi, MD Collapse << |
| NCT02856113 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | June 11, 2021 | - |
| NCT02201381 | Cancer Overal... more >>l Survival Collapse << | Phase 3 | Recruiting | October 22, 2023 | United Kingdom ... more >> Care Oncology Clinic Recruiting London, United Kingdom, W1G 9PP Contact: Gregory Stoloff +44 207 580 3266 info@careoncologyclinic.com Contact: Robin Bannister, PhD +44 207 153 6577 robin.bannister@seekacure.com Collapse << |
| NCT02882477 | Diabetes Mellitus ... more >> Iron Metabolism Disorders Gastroduodenal Ulcer Optic Atrophy Sensorineural Hearing Loss Platelet Dysfunction Collapse << | Phase 2 Phase 3 | Unknown | December 2018 | Israel ... more >> Hadassah medical center Recruiting Jerusalem, Israel Contact: David Zangen, Professor 0097507874488 zanegnd@hadassah.org.il Contact: Ulla Najwa Abdulhag, MD 0097505172866 najwa.ped@gmail.com Collapse << |
| NCT00360698 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-aventis Bridgewater, New Jersey, United States, 08807 Russian Federation Sanofi-Aventis Moscow, Russian Federation United Kingdom Sanofi-aventis Guildford, United Kingdom Collapse << |
| NCT00360698 | - | - | Completed | - | - |
| NCT01028391 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02152371 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01028391 | - | - | Completed | - | - |
| NCT02006836 | Diabetes | Not Applicable | Completed | - | China, Hubei ... more >> Tongji Hospital Wuhan, Hubei, China, 430030 Collapse << |
| NCT00767000 | - | - | Terminated | - | - |
| NCT01237314 | - | - | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT00121641 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00121641 | - | - | Completed | - | - |
| NCT02006836 | - | - | Completed | - | - |
| NCT02152371 | - | - | Completed | - | - |
| NCT00653185 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01364350 | - | - | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Colorado University of Colorado Denver Denver, Colorado, United States, 80262 United States, Connecticut Yale University New Haven, Connecticut, United States, 06519 United States, Maryland George Washington University Biostatistics Center Rockville, Maryland, United States, 20852 United States, Massachusetts Massachusetts General Hospital Diabetes Center Boston, Massachusetts, United States, 02114 Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Saint Louis University St Louis, Missouri, United States, 63104 Washington University St Louis, Missouri, United States, 63110 United States, New York Columbia University Medical Center New York City, New York, United States, 10032 State University of New York Upstate Medical University Syracuse, New York, United States, 13214 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Oklahoma University of Oklahoma Health Sciences Center Oklahoma City, Oklahoma, United States, 73117 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00767000 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated | - | - |
| NCT03235050 | Diabetes Mellitus, Type 2 | Phase 2 | Active, not recruiting | March 9, 2020 | - |
| NCT02183935 | Obesity Diabe... more >>tes Metabolic Syndrome Collapse << | Phase 3 | Unknown | December 2016 | Slovenia ... more >> UMC Ljubljana Recruiting Ljubljana, Slovenia Contact: Tadej Battelino, PhD 00386 01 522 9235 tadej.battelino@mf.uni-lj.si Principal Investigator: Tadej Battelino, MD, PhD Sub-Investigator: Rok Orel, MD, PhD Sub-Investigator: Matjaž Homan, MD, PhD Sub-Investigator: Primož Kotnik, MD, PhD Collapse << |
| NCT00313313 | Diabetes | Phase 3 | Completed | - | - |
| NCT00313313 | - | - | Completed | - | - |
| NCT00901979 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00675857 | Diabetes Mellitus, Type 2 ... more >> Metabolic Syndrome X Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Calgary, Alberta, Canada, T3B 0M3 Canada, Manitoba Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario Etobicoke, Ontario, Canada, M9R 4E1 London, Ontario, Canada, N6A 4V2 Oakville, Ontario, Canada, L6H 3P1 Ottawa, Ontario, Canada, K1H 1A2 Ottawa, Ontario, Canada, K1K 4L2 Thornhill, Ontario, Canada, L4J 8L7 Toronto, Ontario, Canada, M4R 2G4 Toronto, Ontario, Canada, M5C 2T2 Canada, Quebec Chicoutimi, Quebec, Canada, G7H 5H6 Greenfield Park, Quebec, Canada, J4V 2H1 Laval, Quebec, Canada, H7T 2P5 Montreal, Quebec, Canada, H1T 2M4 Montreal, Quebec, Canada, H2W 1T7 Montréal, Quebec, Canada, H3A 1A1 Montréal, Quebec, Canada, H3H 2L9 Montréal, Quebec, Canada, H3N 1S4 Québec, Quebec, Canada, G1V 4G5 Sherbrooke, Quebec, Canada, J1G 1B8 Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT02246257 | Rheumatoid Arthritis ... more >> Cardiovascular Diseases Collapse << | Not Applicable | Recruiting | September 2020 | Denmark ... more >> Department of Rheumathology, Frederiksberg and Bispebjerg univeristy Hospital Recruiting Frederiksberg, Region Of Copenhagen, Denmark, 2000 Contact: Annemarie Lyng Svesson, MD, PhD 004528555126 lyng.annemarie@gmail.com Contact: Torkell J Ellingsen, MD, PhD, Professor 004565413523 torkell.ellingsen@rsyd.dk Collapse << |
| NCT00853151 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | - |
| NCT00280046 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Russian Federation ... more >> Novo Nordisk Investigational Site Arkhangelsk, Russian Federation, 163045 Novo Nordisk Investigational Site Belgorod, Russian Federation, 308007 Novo Nordisk Investigational Site Ekaterinburg, Russian Federation, 620102 Novo Nordisk Investigational Site Kazan, Russian Federation, 420064 Novo Nordisk Investigational Site Khabarovsk, Russian Federation, 680067 Novo Nordisk Investigational Site Nizhniy Novgorod, Russian Federation, 603126 Novo Nordisk Investigational Site Novosibirsk, Russian Federation, 630090 Novo Nordisk Investigational Site Omsk, Russian Federation, 644070 Novo Nordisk Investigational Site Saint-Petersburg, Russian Federation, 194291 Novo Nordisk Investigational Site Vladivostok, Russian Federation, 690105 Novo Nordisk Investigational Site Voronezh, Russian Federation, 394053 Collapse << |
| NCT00934219 | Hypertriglyceridemia | Phase 4 | Unknown | December 2012 | United States, Ohio ... more >> Jewish Hospital Cholesterol Center Recruiting Cincinnati, Ohio, United States, 45229 Collapse << |
| NCT00853151 | - | - | Completed | - | - |
| NCT03763240 | Blood Glucose, High | Not Applicable | Recruiting | December 1, 2020 | Sweden ... more >> Gothia Forum Recruiting Gothenburg, Sweden, 41345 Contact: Helen Svanström, Nurse 031-3439919 rekrytering.ctc.su@vgregion.se Collapse << |
| NCT00032474 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> University of Texas Health Sciences Center San Antonio, Texas, United States, 78229 Collapse << |
| NCT01068665 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02695121 | - | - | Recruiting | April 30, 2024 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States Netherlands Research Site Recruiting Utrecht, Netherlands United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT00511667 | - | - | Completed | - | - |
| NCT00511667 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT01076647 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01068665 | - | - | Completed | - | - |
| NCT01076647 | - | - | Completed | - | - |
| NCT00500955 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01068678 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01068678 | - | - | Completed | - | - |
| NCT01608724 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 4 | Completed | - | China ... more >> Research Site Baotou, China Research Site Beijing, China Research Site Benxi, China Research Site Changchun, China Research Site Chengdu, China Research Site Chongqing, China Research Site Dalian, China Research Site Dongguan, China Research Site Foshan, China Research Site Fuzhou, China Research Site Guangzhou, China Research Site Guiyang, China Research Site Haikou, China Research Site Hangzhou, China Research Site Hhht, China Research Site Hubei, China Research Site Jimo, China Research Site Jinan, China Research Site Kunming, China Research Site Linyi, China Research Site Nanjing, China Research Site Nantong, China Research Site Qingdao, China Research Site Shanghai, China Research Site Shantou, China Research Site Shenyang, China Research Site Shiyan, China Research Site Suzhou, China Research Site Taiyuan, China Research Site Tianjin, China Research Site Urumqi, China Research Site Wenzhou, China Research Site Wuhan, China Research Site Wuxi, China Research Site Xian, China Research Site Zhanjiang, China Research Site Zhengzhou, China Collapse << |
| NCT00274274 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Germany ... more >> Düsseldorf, Germany Collapse << |
| NCT01046110 | - | - | Completed | - | - |
| NCT01326026 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01046110 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01608724 | - | - | Completed | - | - |
| NCT00871572 | - | - | Completed | - | - |
| NCT01365507 | - | - | Completed | - | - |
| NCT01159249 | Type 2 Diabetes | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Hunabashi, Chiba, Japan, 274-0805 Novartis Investigative Site Chikushino, Fukuoka, Japan, 818-0036 Novartis Investigative Site Itoshima-shi, Fukuoka, Japan, 819-1102 Novartis Investigative Site Koriyama, Fukushima, Japan, 963-8851 Novartis Investigative Site Kobe, Hyogo, Japan, 658-0064 Novartis Investigative Site Kawasaki, Kanagawa, Japan, 210-0852 Novartis Investigative Site Kawasaki, Kanagawa, Japan, 212-0024 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0077 Novartis Investigative Site Yokohama, Kanagawa, Japan, 221-0065 Novartis Investigative Site Izumisano, Osaka, Japan, 598-0048 Novartis Investigative Site Hannou, Saitama, Japan, 357-0024 Novartis Investigative Site Hiki-Gun, Saitama, Japan, 355-0328 Novartis Investigative Site Kawaguchi, Saitama, Japan, 332-0012 Novartis Investigative Site Koshigaya city, Saitama, Japan, 343-0826 Novartis Investigative Site Tokorozawa, Saitama, Japan, 359-1161 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Katsushika-ku, Tokyo, Japan, 125-0041 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Novartis Investigative Site Fukuoka, Japan, 807-0857 Novartis Investigative Site Fukuoka, Japan, 816-0094 Novartis Investigative Site Fukuoka, Japan, 819-0168 Collapse << |
| NCT01365507 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00620282 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | United States, Minnesota ... more >> Novo Nordisk Investigational Site Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01059812 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01326026 | - | - | Completed | - | - |
| NCT01068860 | Type 2 Diabetes Mellitus ... more >> Impaired Glucose Tolerance Collapse << | Phase 2 | Completed | - | - |
| NCT00871572 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Buena Park, California, United States, 90620 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Huntington Park, California, United States, 90255 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Westlake Village, California, United States, 91361 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fort Lauderdale, Florida, United States, 33306 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jupiter, Florida, United States, 33458 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New Port Richey, Florida, United States, 34652 United States, Idaho For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Idaho Falls, Idaho, United States, 83404 United States, Kansas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Topeka, Kansas, United States, 66606 United States, Kentucky For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Madisonville, Kentucky, United States, 42431 United States, Nevada For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Las Vegas, Nevada, United States, 89101 United States, South Dakota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rapid City, South Dakota, United States, 57702 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Norfolk, Virginia, United States, 23502 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aschaffenburg, Germany, 63739 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55116 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Munchen, Germany, 80336 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saarlouis, Germany, 66740 Lithuania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kaunas, Lithuania, LT-51270 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vilnius, Lithuania, LT-08661 Collapse << |
| NCT00789750 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01068860 | - | - | Completed | - | - |
| NCT00620282 | - | - | Completed | - | - |
| NCT01059812 | - | - | Completed | - | - |
| NCT00789750 | - | - | Completed | - | - |
| NCT00482950 | Type 2 Diabetes Mellitus | Phase 2 | Unknown | February 2008 | - |
| NCT00766441 | Diabetes Hypo... more >>glycemia Collapse << | Phase 4 | Terminated(inadequate recruitm... more >>ent) Collapse << | - | United Kingdom ... more >> Wellcome Trust Clinical Research Facility Manchester, United Kingdom, M13 9NT Collapse << |
| NCT01970033 | Type 2 Diabetes | Phase 3 | Unknown | January 2015 | China ... more >> Chinese PLA General Hospital Recruiting Beijing, China Contact: Changyu Pan, M.D. panchy301@aliyun.com Principal Investigator: Changyu Pan, M.D. Collapse << |
| NCT00952198 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research Chula Vista, California, United States, 91911 United States, Texas Cetero San Antonio, Texas, United States, 78229 Collapse << |
| NCT03164187 | - | - | Completed | - | Azerbaijan ... more >> Azerbaijan Republic Ministry of Health Republic Endocrinology Center Baku, Azerbaijan, AZ 1/122 Azer-Turk Med clinic Baku, Azerbaijan, AZ 1025 Hospital of oil workers, polyclinics and private clinics Baku, Azerbaijan, AZ 1025 Medical Clinic of Azerbaijan Medical University Baku, Azerbaijan, AZ-1078 Collapse << |
| NCT00747383 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United Kingdom ... more >> Royal Bournemouth Hospital Bournemouth, Dorset, United Kingdom, BH7 7DW Collapse << |
| NCT00479986 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 4 | Completed | - | Germany ... more >> Dr. Michael Morcos Heidelberg, Germany IKFE Mainz, Germany, 55116 Collapse << |
| NCT00679237 | Coronary Heart Disease | Not Applicable | Active, not recruiting | December 2024 | Norway ... more >> Sorlandet Hospital Arendal, Norway Collapse << |
| NCT00658021 | Type 2 Diabetes | Phase 3 | Recruiting | March 6, 2023 | - |
| NCT01285076 | - | - | Completed | - | Taiwan ... more >> Merck Sharp & Dohme (I.A.) Corp. Taipei, Taiwan, 106 Collapse << |
| NCT00862875 | Type 2 Diabetes | Phase 4 | Completed | - | Canada, Quebec ... more >> Institut de Recherches Cliniques de Montréal Montréal, Quebec, Canada, H2W 1R7 Collapse << |
| NCT02695173 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT01755442 | Diabetes Mellitus | Phase 1 | Terminated(Amgen determined no... more >> further need for this study.) Collapse << | - | United States, California ... more >> Research Site San Diego, California, United States, 92161 Collapse << |
| NCT01495013 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Bucheon, Korea, Republic of, 134 727 GSK Investigational Site Busan, Korea, Republic of, 602-739 GSK Investigational Site Kangwondo, Korea, Republic of, 220-701 GSK Investigational Site Seocho-ku, Seoul, Korea, Republic of, 137-701 GSK Investigational Site Seongnam-si Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 110-746 GSK Investigational Site Seoul, Korea, Republic of, 135-710 GSK Investigational Site Seoul, Korea, Republic of, 138-736 GSK Investigational Site Seoul, Korea, Republic of, 139-872 GSK Investigational Site Seoul, Korea, Republic of, 152-703 GSK Investigational Site Suwon, Korea, Republic of, 463442 Malaysia GSK Investigational Site Ipoh, Malaysia, 30450 GSK Investigational Site Johor Bahru, Malaysia, 80100 GSK Investigational Site Kuala Lumpur, Malaysia, 59100 GSK Investigational Site Kubang Kerian, Malaysia, 16150 Mexico GSK Investigational Site Cuernavaca, Mexico, 62250 GSK Investigational Site Del. Cuauhtémoc, Mexico, 06700 GSK Investigational Site Gadalajara, Jalisco, Mexico, C.P. 44130 GSK Investigational Site Guadalajara Jalisco, Mexico, C.P. 44210 GSK Investigational Site Mexico City, Mexico, 06700 GSK Investigational Site Mexico, D.F., Mexico, 11650 Philippines GSK Investigational Site Cebu City, Philippines, 6000 GSK Investigational Site Davao City, Philippines, 8000 GSK Investigational Site Manila, Philippines, 1008 GSK Investigational Site Pasig City, Philippines, 1600 GSK Investigational Site Sta. Cruz, Manila, Philippines, 1012 Russian Federation GSK Investigational Site Moscow, Russian Federation, 109240 GSK Investigational Site Saratov, Russian Federation, 410054 GSK Investigational Site St. Petersburg, Russian Federation, 191119 Thailand GSK Investigational Site Chiangrai, Thailand, 57000 GSK Investigational Site Nakhon Ratchasima, Thailand, 30000 GSK Investigational Site Rajathevee, Thailand, 10400 Collapse << |
| NCT00062764 | - | - | Completed | - | - |
| NCT01285076 | - | - | Completed | - | - |
| NCT00062764 | Hepatitis | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01494987 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01451398 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02695082 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT02695095 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT00778765 | Healthy | Not Applicable | Completed | - | Canada, Quebec ... more >> MDS Pharma Services Saint-Laurent,, Quebec, Canada Collapse << |
| NCT00445627 | - | - | Recruiting | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT01451398 | - | - | Completed | - | - |
| NCT02768948 | Diabetic Nephropathy | Not Applicable | Recruiting | July 2019 | France ... more >> CHU de Nice Recruiting Nice, France, 06000 Contact: Guillaume FAVRE, PH favre.g@chu-nice.fr Collapse << |
| NCT01494987 | - | - | Completed | - | - |
| NCT00763347 | Diabetes Mellitus | Phase 2 | Terminated(Voluntarily termina... more >>ted based on preliminary non-clinical findings.) Collapse << | - | - |
| NCT00795600 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Spain ... more >> Málaga, Spain, 29009 Collapse << |
| NCT00272064 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Italy ... more >> Sanofi-Aventis Administrative Office Milan, Italy Collapse << |
| NCT02871882 | Type 2 Diabetes Mellitus ... more >> Obese Overweight Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00673309 | Burns | Phase 2 Phase 3 | Completed | - | United States, Texas ... more >> University of Texas Medical Branch Galveston, Texas, United States, 77550 Collapse << |
| NCT00058981 | Diabetes, Autoimmune | Phase 2 | Terminated | - | United States, Alabama ... more >> University of Alabama at Birmingham Endocrinology Department Birmingham, Alabama, United States, 35294 United States, Colorado University of Colorado Hospital Endocrinology Practice Aurora, Colorado, United States, 80010 United States, Kentucky University of Kentucky Department of Internal Medicine Lexington, Kentucky, United States, 40536 United States, Missouri Washington University School of Medicine Endocrinology/Metabolic Dept Saint Louis, Missouri, United States, 63110 United States, New York North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States, 11042 United States, Texas Diabetes & Glandular Disease Research Associates San Antonio, Texas, United States, 78229-4801 United States, Washington DVA Puget Sound Health Care System Endocrinology (III) Department Seattle, Washington, United States, 98108 Collapse << |
| NCT01699737 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00795600 | - | - | Completed | - | - |
| NCT02449603 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Shaanxi ... more >> Xijing Hospital, Fourth Military Medical university Xi'an, Shaanxi, China, 710032 Collapse << |
| NCT00528372 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01700075 | Myocardial Infarction ... more >> Coronary Artery Diseases Diabetes Mellitus, Type 2 Atherosclerosis of Femoral Artery Collapse << | Phase 4 | Completed | - | Kazakhstan ... more >> Scientific Research Institute of Cardiology and Internal Diseases Almaty, Kazakhstan, 050000 Collapse << |
| NCT00873821 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | - |
| NCT03406377 | Type 2 Diabetes Mellitus | Phase 2 | Recruiting | June 2019 | United States, California ... more >> National Research Institute - Huntington Park Recruiting Huntington Park, California, United States, 90255 Contact: Stanley Hsin-Wei Hsia National Research Institute - Wilshire Recruiting Los Angeles, California, United States, 90057 Contact: Juan Pablo Frias United States, Florida Clinical Pharmacology of Miami, LLC Recruiting Hialeah, Florida, United States, 33014 Contact: Kenneth Lasseter, MD Collapse << |
| NCT00528372 | - | - | Completed | - | - |
| NCT00873821 | - | - | Completed | - | - |
| NCT01923350 | Weight Reduction | Not Applicable | Completed | - | United States, Maryland ... more >> Social & Scientific Systems Inc. Silver Spring, Maryland, United States, 20910 Collapse << |
| NCT01647308 | Type 2 Diabetes | Phase 2 | Terminated(The study was termi... more >>nated for business planning purposes. Assessments of APOCIIIRx in this population will be explored in a larger efficacy study) Collapse << | - | United States, California ... more >> Profil Institute for Clinical Research Chula Vista, California, United States, 91911 Collapse << |
| NCT01301456 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Metabolic Diseases Endocrine System Diseases Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 Comprehensive Phase One (A Division of Comprehensive NeuroScience, Inc.) Miramar, Florida, United States, 33025 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Nebraska ICON Clinical Pharmacology, LLC Omaha, Nebraska, United States, 68154 United States, Pennsylvania CRI Worldwide, LLC Philadelphia, Pennsylvania, United States, 19139 United States, Texas Healthcare Discoveries LLC d/b/a ICON Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT00300911 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Turkey ... more >> Baskent University Ankara Hospital Ankara, Turkey, 06490 Collapse << |
| NCT00603291 | Obesity | Phase 3 | Completed | - | United States, California ... more >> Arena Pharmaceuticals, Inc. San Diego, California, United States, 92121 Collapse << |
| NCT03526289 | Type 2 Diabetes Mellitus ... more >> Overweight and Obesity Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for diabetes research Hellerup, Denmark, DK-2900 Collapse << |
| NCT03433248 | Type2 Diabetes | Phase 4 | Recruiting | December 1, 2020 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1081HV Contact: Michaël JB van Baar, MD +31 20 444 4709 m.vanbaar@vumc.nl Contact: Daniël H van Raalte, MD PhD +31 20 444 0534 d.vanraalte@vumc.nl Principal Investigator: Mark HH Kramer, MD PhD Principal Investigator: Daniël H van Raalte, MD PhD Collapse << |
| NCT03061409 | Nutrient Deficiency | Not Applicable | Recruiting | July 2018 | United States, Massachusetts ... more >> Jean Mayer USDA Human Nutrition Research Center on Aging, Tufts University Recruiting Boston, Massachusetts, United States, 02111 Contact: Kimberly Dupiton, BA 617-556-3012 Kimberly.Dupiton@tufts.edu Collapse << |
| NCT00603291 | - | - | Completed | - | - |
| NCT01301456 | - | - | Completed | - | - |
| NCT02367066 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT02049034 | Type 2 Diabetes | Phase 4 | Completed | - | United Kingdom ... more >> University of Surrey FHMS Diabetes and Metabolic Medicine Guildford, Surrey, United Kingdom, GU2 7WG Collapse << |
| NCT02367066 | - | - | Completed | - | - |
| NCT00454233 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | Russian Federation ... more >> Ekaterinburg, Russian Federation 4 Sites Moscow, Russian Federation N. Novgorod, Russian Federation Petrozavodsk, Russian Federation Samara, Russian Federation 10 Sites St. Petersburg, Russian Federation Collapse << |
| NCT00377676 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03538743 | Type 2 Diabetes Mellitus | Phase 1 | Recruiting | April 20, 2019 | United States, California ... more >> Anaheim Clinical Trials, LLC Recruiting Anaheim, California, United States, 92801 United States, Florida Qps-Mra, Llc Recruiting South Miami, Florida, United States, 33143 Qps-Mra,Llc Recruiting South Miami, Florida, United States, 33143 United States, Kansas Vince & Associates Clinical Research, Inc. Recruiting Overland Park, Kansas, United States, 66212 Vince and Associates Clinical Research, Inc. Recruiting Overland Park, Kansas, United States, 66212 Collapse << |
| NCT03624803 | - | - | Not yet recruiting | May 31, 2019 | - |
| NCT00377676 | - | - | Completed | - | - |
| NCT03098654 | Human Immunodeficiency Virus ... more >> Diabetes Mellitus, Type 2 Hypertension Collapse << | Not Applicable | Recruiting | June 30, 2021 | Tanzania ... more >> Muhimbili University of Health and Allied Sciences Recruiting Dar Es Salaam, Tanzania Contact: Jessie Mbwambo, MD jmbwambo@gmail.com Collapse << |
| NCT02072096 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(The trial was termi... more >>nated per protocol because of lack of feasibility.) Collapse << | - | - |
| NCT03168880 | Triple Negative Breast Cancer | Phase 3 | Recruiting | November 30, 2024 | India ... more >> Tata memorial Centre Recruiting Mumbai, Maharashtra, India, 400012 Contact: Rohini Hawaldar 24168601 rwhawaldar@gmail.com Collapse << |
| NCT02072096 | - | - | Terminated(The trial was termi... more >>nated per protocol because of lack of feasibility.) Collapse << | - | - |
| NCT03462069 | Diabetes Mellitus | Phase 2 | Recruiting | August 7, 2019 | Germany ... more >> Investigational Site Number 2760001 Recruiting Berlin, Germany, 13353 Collapse << |
| NCT00722371 | - | - | Completed | - | - |
| NCT00722371 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00631007 | - | - | Completed | - | - |
| NCT00631007 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00155545 | Leg Length Discrepancy ... more >> Scoliosis Collapse << | Phase 1 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT00913367 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> HeeYoung Lee Seoul, Korea, Republic of Collapse << |
| NCT01922492 | Partial Pancreatectomy Due to ... more >>Benign Pancreatic Neoplasm Collapse << | Not Applicable | Unknown | December 2017 | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT00887094 | Diabetes Mellitus, Type 2 ... more >> Treatment Exercise Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital de Clinicas de Porto Alegre Porto Alegre, RS, Brazil, 90035-003 Collapse << |
| NCT00794963 | Metabolic Syndrome | Not Applicable | Terminated(Difficulty recruiti... more >>ng) Collapse << | - | United States, New York ... more >> Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00502710 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT00147836 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | China, Guangdong ... more >> the first Affiliated Hospital of Sun Yat-Sen university Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT00794963 | - | - | Terminated(Difficulty recruiti... more >>ng) Collapse << | - | - |
| NCT01697592 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01697592 | - | - | Completed | - | - |
| NCT00316082 | Diabetes | Phase 3 | Completed | - | - |
| NCT00295633 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00295633 | - | - | Completed | - | - |
| NCT01204294 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> 1218.78.008 Boehringer Ingelheim Investigational Site Akishima, Tokyo, Japan 1218.78.030 Boehringer Ingelheim Investigational Site Amagasaki, Hyogo, Japan 1218.78.017 Boehringer Ingelheim Investigational Site Annaka, Gunma, Japan 1218.78.006 Boehringer Ingelheim Investigational Site Aomori, Aomori, Japan 1218.78.007 Boehringer Ingelheim Investigational Site Aomori, Aomori, Japan 1218.78.009 Boehringer Ingelheim Investigational Site Chuo-ku, Tokyo, Japan 1218.78.013 Boehringer Ingelheim Investigational Site Chuo-ku, Tokyo, Japan 1218.78.032 Boehringer Ingelheim Investigational Site Fukuoka, Fukuoka, Japan 1218.78.040 Boehringer Ingelheim Investigational Site Fukuoka, Fukuoka, Japan 1218.78.037 Boehringer Ingelheim Investigational Site Higashi Osaka, Osaka, Japan 1218.78.043 Boehringer Ingelheim Investigational Site Hitachinaka, Ibaraki, Japan 1218.78.019 Boehringer Ingelheim Investigational Site Isesaki, Gunma, Japan 1218.78.036 Boehringer Ingelheim Investigational Site Kashiwara, Osaka, Japan 1218.78.026 Boehringer Ingelheim Investigational Site Kasugai, Aichi, Japan 1218.78.038 Boehringer Ingelheim Investigational Site Kawachinagano, Osaka, Japan 1218.78.021 Boehringer Ingelheim Investigational Site Kitaazumi-gun, Nagano, Japan 1218.78.022 Boehringer Ingelheim Investigational Site Matsumoto, Nagano, Japan 1218.78.033 Boehringer Ingelheim Investigational Site Matsumoto, Nagano, Japan 1218.78.012 Boehringer Ingelheim Investigational Site Meguro-ku, Tokyo, Japan 1218.78.041 Boehringer Ingelheim Investigational Site Meguro-ku, Tokyo, Japan 1218.78.034 Boehringer Ingelheim Investigational Site Morioka, Iwate, Japan 1218.78.035 Boehringer Ingelheim Investigational Site Morioka, Iwate, Japan 1218.78.018 Boehringer Ingelheim Investigational Site Moriya, Ibaraki, Japan 1218.78.024 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.025 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.027 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.028 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.031 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.039 Boehringer Ingelheim Investigational Site Oita, Oita, Japan 1218.78.004 Boehringer Ingelheim Investigational Site Okinawa, Okinawa, Japan 1218.78.016 Boehringer Ingelheim Investigational Site Sagae, Yamagata, Japan 1218.78.001 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.020 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.042 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.005 Boehringer Ingelheim Investigational Site Shimajiri-gun, Okinawa, Japan 1218.78.014 Boehringer Ingelheim Investigational Site Shinjuku-ku, Tokyo, Japan 1218.78.002 Boehringer Ingelheim Investigational Site Shinjyuku-ku. Tokyo, Japan 1218.78.011 Boehringer Ingelheim Investigational Site Shizuoka, Shizuoka, Japan 1218.78.003 Boehringer Ingelheim Investigational Site Suita, Osaka, Japan 1218.78.023 Boehringer Ingelheim Investigational Site Tokorozawa, Saitama, Japan 1218.78.029 Boehringer Ingelheim Investigational Site Tokorozawa, Saitama, Japan 1218.78.010 Boehringer Ingelheim Investigational Site Tsuchiura, Ibaraki, Japan 1218.78.015 Boehringer Ingelheim Investigational Site Yamagata, Yamagata, Japan Collapse << |
| NCT01204775 | Type 2 Diabetes | Phase 3 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States Research Site San Diego, California, United States United States, Florida Research Site Hollywood, Florida, United States Research Site Tallahassee, Florida, United States Research Site Tampa, Florida, United States United States, Michigan Research Site Dearborn, Michigan, United States United States, New York Research Site Buffalo, New York, United States United States, Tennessee Research Site Memphis, Tennessee, United States United States, Virginia Research Site Richmond, Virginia, United States Russian Federation Research Site Moscow, Russian Federation Research Site Novosibirsk, Russian Federation Research Site Saint Petersburg, Russian Federation South Africa Research Site Cape Town, South Africa Research Site Pretoria, South Africa Taiwan Research Site Taichung, Taiwan Research Site Taipei, Taiwan Collapse << |
| NCT00316082 | - | - | Completed | - | - |
| NCT01429506 | Diabetes Mellitus ... more >> Obesity Collapse << | Not Applicable | Completed | - | India ... more >> PGIMER Chandigarh, India, 160012 Collapse << |
| NCT01204775 | - | - | Completed | - | - |
| NCT03711682 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | El Salvador ... more >> Unidad Comunitaria de Salud Familiar Intermedia de Comasagua (UCSFI-Comasagua) Comasagua, La Libertad, El Salvador, 0504 Collapse << |
| NCT02823808 | Diabetes | Phase 4 | Recruiting | December 2021 | Korea, Republic of ... more >> Kyunghee University Medical Center Recruiting Seoul, Korea, Republic of Contact: Sang Youl Rhee Collapse << |
| NCT02738879 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02009488 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> San Diego, La Jolla, California, United States United States, Florida Gainesville, Florida, United States Collapse << |
| NCT02052011 | - | - | Completed | - | - |
| NCT02052011 | Microvascular Angina | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale New Haven Hospital New Haven, Connecticut, United States, 06510 Collapse << |
| NCT01204294 | - | - | Completed | - | - |
| NCT00765817 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00765817 | - | - | Completed | - | - |
| NCT01244646 | - | - | Terminated | - | Netherlands ... more >> Research Site Alkmaar, Netherlands Research Site Den Haag, Netherlands Research Site Edam, Netherlands Research Site Gorinchem, Netherlands Research Site Heerhugowaard, Netherlands Research Site Heiloo, Netherlands Research Site Hoogvliet, Netherlands Research Site Lichtenvoorde, Netherlands Research Site Limmen, Netherlands Research Site Maasbracht, Netherlands Research Site Noord-Scharwoude, Netherlands Research Site Poortvliet, Netherlands Research Site Rotterdam, Netherlands Research Site Spijkenisse, Netherlands Research Site Tuitjenhorn, Netherlands Research Site Vaassen, Netherlands Research Site Valkenswaard, Netherlands Research Site Voerendaal, Netherlands Research Site Volendam, Netherlands Research Site Wamel, Netherlands Research Site Zuid-Scharwoude, Netherlands Research Site Zutphen, Netherlands Collapse << |
| NCT00393705 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Croatia ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Zagreb, Croatia, HR-10000 Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bialystok, Poland, 15-276 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rzeszow, Poland, 35-0723 Romania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bucharest, Romania, 020475 South Africa For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kempton Park, South Africa, 1619 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kenilworth, South Africa, 7708 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kuilsriver, South Africa, 7580 Turkey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Istanbul, Turkey, 34662 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Konya, Turkey, 42075 Collapse << |
| NCT00425009 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | China ... more >> Department of Endocrinology, Xinhua Hospital Affiliated to Shanghai Jiaotong University School of Medicine Shanghai, China, 200092 Collapse << |
| NCT01483781 | Diabetes Mellitus, Type 2 ... more >> Plasma Volume Collapse << | Phase 1 | Completed | - | Germany ... more >> Neuss, Germany Collapse << |
| NCT00393705 | - | - | Completed | - | - |
| NCT00086502 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00086502 | - | - | Completed | - | - |
| NCT00636766 | Carotid Atherosclerosis ... more >> Stroke Type 2 Diabetes Metabolic Syndrome Collapse << | Not Applicable | Completed | - | Greece ... more >> General Hospital of Thessaloniki "Hippokratio" Thessaloniki, Greece Collapse << |
| NCT01283308 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | India ... more >> Madras Diabetes Research Foundation Chennai, Tamil Nadu, India, 600 086 Collapse << |
| NCT02708758 | Gestational Diabetes | Not Applicable | Recruiting | December 2019 | Mexico ... more >> Instituto Nacional de Perinatología Isidro Espinosa de los Reyes Recruiting Mexico City, Mexico, 11000 Contact: Nayeli Martínez-Cruz, MD +525555209900 ext 299 nayemc_21@hotmail.com Contact: Enrique Reyes-Muñoz, MD, PhD. +525555209900 ext 299 dr.enriquereyes@gmail.com Collapse << |
| NCT01845831 | - | - | Completed | - | - |
| NCT00806520 | - | - | Completed | - | United States, Massachusetts ... more >> Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Minnesota International Diabetes Center Minneapolis, Minnesota, United States, 55416 United States, Nevada Palm Medical Group Las Vegas, Nevada, United States, 89148 United States, North Carolina University of North Carolina Diabetes Care Center Durham, North Carolina, United States, 27713 United States, Washington Rockwood Clinic Spokane, Washington, United States, 99202 Collapse << |
| NCT03489317 | - | - | Recruiting | December 2021 | Hong Kong ... more >> Department of Microbiology, Chinese University of Hong Kong Recruiting Sha Tin, Hong Kong Contact: Yun Kit Yeoh 85235053333 yeohyunkit@cuhk.edu.hk Lek Yuen Health Centre Recruiting Sha Tin, Hong Kong Contact: Martin Wong 85222528782 wong_martin@cuhk.edu.hk Collapse << |
| NCT01845831 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory University Hospital Atlanta, Georgia, United States, 30324 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Pennsylvania Temple University Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT02973321 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00696657 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT01011868 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01011868 | - | - | Completed | - | - |
| NCT00696657 | - | - | Completed | - | - |
| NCT00106704 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00106704 | - | - | Completed | - | - |
| NCT02946996 | Prostate Cancer | Phase 2 | Recruiting | March 1, 2020 | United States, South Carolina ... more >> Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Mike Wheeler 843-792-9321 hcc-clinical-trials@musc.edu Collapse << |
| NCT01084005 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01084005 | - | - | Completed | - | - |
| NCT01944618 | - | - | Terminated() | - | Australia, Victoria ... more >> Local Institution Clayton, Victoria, Australia, VIC 3168 Collapse << |
| NCT00094770 | - | - | Completed | - | - |
| NCT01638260 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(Terminated because ... more >>of insufficient number of subjects included.) Collapse << | - | Netherlands ... more >> Maastricht University Medical Center Maastricht, Netherlands, 6229HX Collapse << |
| NCT03041129 | - | - | Recruiting | June 1, 2022 | United States, Colorado ... more >> University of Colorado Anshutz Medical Campus/Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Yesenia Garcia 720-777-6984 yesenia.garciareyes@childrenscolorado.org Collapse << |
| NCT00094770 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02879409 | Diabetes Mellitus Type 2 | Not Applicable | Recruiting | August 2019 | Qatar ... more >> Hamad Medical Corporation Recruiting Doha, Qatar, 3050 Contact: Ibrahim Janahi, MD 4439-2434 ext 00974 Imohd@hamad.qa Principal Investigator: Ibrahim Janahi, MD Sub-Investigator: Mashhood Siddique, MD Sub-Investigator: Hamda Ali, MD Collapse << |
| NCT00465335 | - | - | Completed | - | - |
| NCT00664846 | Stroke | Not Applicable | Completed | - | - |
| NCT00732121 | Type 2 Diabetes | Phase 4 | Unknown | August 2010 | United States, Vermont ... more >> University of Vermont South Burlington, Vermont, United States, 05403 Collapse << |
| NCT01156597 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Florida ... more >> Diabetes Research Institute Miami, Florida, United States, 33136 Collapse << |
| NCT01156597 | - | - | Completed | - | - |
| NCT00341406 | - | - | Completed | - | United States, Maryland ... more >> National Institute of Diabetes and Digestive and Kidney Disorders (NIDDK), 9000 Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01203111 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Algeria ... more >> Administrative office Algiers, Algeria Brazil Administrative office Sao Paulo, Brazil Israel Administrative office Natanya, Israel Lebanon Administrative office Beirut, Lebanon Mexico Administrative office Col. Coyoacan, Mexico Morocco Administrative office Casablanca, Morocco Peru Administrative office Lima, Peru Saudi Arabia Administrative office Jeddah, Saudi Arabia United Arab Emirates Administrative office Dubaï, United Arab Emirates Venezuela Administrative office Caracas, Venezuela Collapse << |
| NCT03419624 | Obesity Diabe... more >>tes Mellitus, Type 2 Collapse << | Phase 3 | Recruiting | June 15, 2019 | Germany ... more >> Diabeteszentrum Oldenburg Recruiting Oldenburg, Lower Saxony, Germany, 23758 Contact: Thomas Schaum, MD +494361513622 info@diabeteszentrum-oldenburg.de Principal Investigator: Thomas Schaum, MD University Medical Center Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Contact: Anne Lautenbach, MD +4915222816188 a.lautenbach@uke.de Contact: Hilko TD Wittmann h.wittmann@uke.de Diabetologische Schwerpunktpraxis Harburg Recruiting Hamburg, Germany, 21073 Contact: Bjoern Paschen, MD +4940557753300 info@diabetologie-harburg.de Principal Investigator: Bjoern Paschen, MD Gemeinschaftspraxis für Innere Medizin und Diabetologie Recruiting Hamburg, Germany, 22607 Contact: Dominik Dahl, MD +4940896295 Principal Investigator: Dominik Dahl, MD Collapse << |
| NCT01175486 | Diabetes | Phase 4 | Unknown | December 2015 | Taiwan ... more >> Taipei Veterans General Hospital, Taiwan Recruiting Taipei, Taiwan Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT01072578 | Healthy Volunteers | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT00353834 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, Massachusetts ... more >> Joslin Diabetes Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01623024 | Non.Alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 3 | Unknown | - | Italy ... more >> Sezione di Gastroenterologia, Di.Bi.M.I.S. AOUP Paolo Giaccone, University of Palermo, Italy Not yet recruiting Palermo, Italy, 90127 Contact: Antonio Craxì, Professor 00390916552280 antonio.craxi@unipa.it Principal Investigator: Antonio Craxì, Professor Collapse << |
| NCT02686476 | Non Alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Completed | - | India ... more >> Division Of Endocrinology , Medanta The Medicity Sec 38 Gurgaon, Haryana, India, 122001 Collapse << |
| NCT03703115 | Infertility P... more >>olycystic Ovary Syndrome Collapse << | Not Applicable | Recruiting | September 1, 2019 | Egypt ... more >> KasrELAiniH Recruiting Cairo, Egypt Contact: Abdelmaguid Ramzy, MD 00223682030 amramzy@gmail.com Contact: Amira Dieb 00223682030 amirasaied2026@cu.edu.gov Principal Investigator: Ramzy, MD Collapse << |
| NCT02830191 | - | - | Completed | - | - |
| NCT00656864 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, Vermont ... more >> University of Vermont South Burlington, Vermont, United States, 05403 Collapse << |
| NCT00353834 | - | - | Completed | - | - |
| NCT00451620 | Type 2 Diabetes | Phase 2 | Completed | - | Canada, British Columbia ... more >> University of British Columbia Vancouver, British Columbia, Canada Collapse << |
| NCT03503045 | - | - | Completed | - | Turkey ... more >> Gulhane School of Medicine Ankara, Turkey, 06018 Collapse << |
| NCT02051842 | NAFLD Pre-dia... more >>betes Collapse << | Phase 4 | Unknown | January 2017 | Mexico ... more >> Instituto Nacional de Ciencias Médicas y Nutrición "Salvador Zubirán" Recruiting Mexico City, Mexico, 14000 Contact: Aldo Torre, M.D, M.Sc. 54870900 ext 2711 detoal@yahoo.com Principal Investigator: Aldo Torre, MD MSci Collapse << |
| NCT02420392 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00696982 | Diabetes Mellitus ... more >> Arterial Stiffness Collapse << | Not Applicable | Unknown | June 2009 | Israel ... more >> Department of Internal Medicine A , Research & Development unit Assaf Harofeh Medical Center, Zerifin, affiliated to Sackler School of Medicine, Tel Aviv, Israel Recruiting Zrifin, Israel, 70300 Contact: Shlomit Koren, MD 972-546-888=385 shlomitks@gmail.com Contact: Shai Efrati, MD 972-577-346-364 efratishai@013.net Principal Investigator: Shlomit Koren, MD Collapse << |
| NCT03023137 | Recurrent Miscarriage | Not Applicable | Completed | - | Brazil ... more >> Hospital Federal dos Servidores do Estado, Ministry of Health Rio de Janeiro, RJ, Brazil, 20221-903 Collapse << |
| NCT03249896 | Gestational Diabetes ... more >> Pregnancy Complications Weight Gain Collapse << | Not Applicable | Recruiting | April 2019 | Singapore ... more >> National University Hospital Recruiting Singapore, Singapore, 119228 Contact: E Shyong Tai, PhD +65 67724371 e_shyong_tai@nuhs.edu.sg Contact: Tong Wei Yew, MRCP +65 67723943 tong_wei_yew@nuhs.edu.sg Collapse << |
| NCT00832390 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT03732690 | Type 2 Diabetes | Not Applicable | Not yet recruiting | December 30, 2020 | France ... more >> Hôpital PITIE SALPETRIERE - APHP Not yet recruiting Paris, France, 75013 Contact: Karine CLEMENT, MD, PhD Collapse << |
| NCT02925559 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | December 2017 | Brazil ... more >> Centro de Diabetes Curitiba Curitiba, Parana, Brazil, 80810040 Collapse << |
| NCT02444156 | Diabetes Mellitus, Type 2 ... more >> Glucose Intolerance Hyperglycemia Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Leicester Diabetes Centre Leicester, United Kingdom, LE5 4PW Collapse << |
| NCT00832390 | - | - | Completed | - | - |
| NCT01472432 | Chronic Foot Ulcers | Phase 4 | Completed | - | Italy ... more >> Second university of Naples Naples, Italy, I-80100 Collapse << |
| NCT03620773 | Type 2 Diabetes Mellitus ... more >> Obesity Bariatric Surgery Candidate Nephropathy Diabetic Kidney Disease Diabetes Mellitus, Type 2 Diabetes Mellitus Diabetes Complications Weight Loss Diabetic Nephropathies Adolescent Obesity Pediatric Obesity Collapse << | Phase 1 Phase 2 | Recruiting | August 2023 | United States, Colorado ... more >> Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Petter Bjornstad, MD 720-777-4659 petter.bjornstad@childrenscolorado.org Principal Investigator: Petter Bjornstad, M.D. Sub-Investigator: Kristin Nadeau, M.D. Collapse << |
| NCT02627898 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02176681 | Haemodialyzed, Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> CH d'Amiens Amiens, France, 80054 CH de Besançon Besancon, France, 25030 AURAL Colmar Colmar, France, 68000 Hospices civils de Colmar Colmar, France, 68024 CH de Dijon Dijon, France, 21079 AURAL Mulhouse Mulhouse, France, 68070 CH de Mulhouse Mulhouse, France, 68070 CH de Nancy Nancy, France, 54000 AURAL Clinique Sainte Anne Strasbourg, France, 67000 Clinique Sainte Anne Strasbourg, France, 67000 Hôpitaux Universitaires de Strasbourg Strasbourg, France, 67091 AURAL Strasbourg Strasbourg, France, 67200 CH de Valenciennes Valenciennes, France, 59300 Collapse << |
| NCT01472432 | - | - | Completed | - | - |
| NCT03023137 | - | - | Completed | - | - |
| NCT02941770 | Overweight | Not Applicable | Recruiting | February 2019 | Portugal ... more >> Hospital de Santa Maria Recruiting Lisbon, Portugal, 1649-035 Contact: António Videira-Silva, MSc 969172368 antonioascenso@campus.ul.pt Collapse << |
| NCT02076880 | Severe Obesity ... more >> Type 2 Diabetes Collapse << | Not Applicable | Unknown | November 2017 | France ... more >> CHU Montpellier Recruiting Montpellier, France, 34295 Contact: GALTIER Florence f-galtier@chu-montpellier.fr Principal Investigator: GALTIER Florence, MD Collapse << |
| NCT01919489 | Type 2 Diabetes | Phase 4 | Recruiting | June 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States Contact: Gianluca Iacobellis, MD GIacobellis@med.miami.edu Principal Investigator: Gianluca Iacobellis, MD United States, Georgia Grady Memorial Hospital Recruiting Atlanta, Georgia, United States, 30303 Contact: Guillermo E Umpierrez, MD 404-778-1665 geumpie@emory.edu Contact: Francisco J Pasquel, MD 404 778 1697 fpasque@emory.edu Principal Investigator: Guillermo Umpierrez, MD Sub-Investigator: Francisco J Pasquel, MD Sub-Investigator: Priya Vellanki, MD Emory University Hospital Recruiting Atlanta, Georgia, United States, 30324 Contact: Francisco Pasquel, MD fpasque@emory.edu Sub-Investigator: Francisco Pasquel, MD Emory Universtiy Hospital at MIdtown Recruiting Atlanta, Georgia, United States Contact: Francisco Pasquel, MD Sub-Investigator: Rodolfo J Galindo, MD United States, New York State University of NY at Buffalo Recruiting New York, New York, United States Contact: Ajay Chaudhuri, MD AChaudhuri@KaleidaHealth.Org Principal Investigator: Ajay Chaudhuri, MD Collapse << |
| NCT00658164 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Phase 3 | Unknown | - | Italy ... more >> U.O. Medicina Interna 1/B Recruiting Milan, Italy, 20122 Contact: Silvia Fargion, Prof. 39-02-5503-3301 silvia.fargion@unimi.it Principal Investigator: Silvia Fargion, Prof. Sub-Investigator: Luca Valenti, Md Collapse << |
| NCT01849289 | - | - | Completed | - | - |
| NCT01849289 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00965549 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03429543 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | January 31, 2022 | - |
| NCT01122641 | Insulin Resistance ... more >> Microvascular Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Diabetes and Vascular Research Department Exeter, Devon, United Kingdom, EX2 5AX Collapse << |
| NCT01018017 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Neuss, Nordrhein-Westfalen, Germany, 41460 Collapse << |
| NCT01039532 | - | - | Completed | - | India ... more >> AnilBhansali Chandigarh, India, 160012 Collapse << |
| NCT00353587 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | - |
| NCT00736879 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01022658 | Type 2 Diabetes | Not Applicable | Completed | - | France ... more >> Jean Verdier hospital, Department of Endocrinology-Diabetology-Nutrition Bondy, France, 93140 Collapse << |
| NCT02830048 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United Kingdom ... more >> Clinical Research Unit, OCDEM, Churchill Hospital Oxford, Oxfordshire, United Kingdom, OX3 7LE Collapse << |
| NCT00736879 | - | - | Completed | - | - |
| NCT00982644 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00982644 | - | - | Completed | - | - |
| NCT02922179 | - | - | Unknown | January 2017 | - |
| NCT01514942 | PCOS | Phase 4 | Completed | - | Italy ... more >> Istituto di Patologia Ostetrica e Ginecologica Catania, Italy Collapse << |
| NCT03444142 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 30, 2018 | Mexico ... more >> Instituto de terapeutica Experimental y Clinica. Centro universitario de Ciencias de la Salud. Universidad de Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03739268 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | September 1, 2020 | Denmark ... more >> Steno Diabetes Center Copenhagen, Gentofte Hospital Recruiting Hellerup, Denmark, 2900 Contact: Henriette H Nerild, M.D. +45 3867 4264 henriette.holst.nerild@regionh.dk Collapse << |
| NCT03074630 | Diabetes Mellitus, Type 2 ... more >> Hypercholesterolemia Collapse << | Phase 4 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, North Holland, Netherlands, 1105AZ Collapse << |
| NCT01379690 | - | - | Completed | - | Singapore ... more >> Changi General Hospital Singapore, Singapore, 529889 Collapse << |
| NCT03705195 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | June 20, 2019 | United Kingdom ... more >> Ninewells Hospital and Medical School Recruiting Dundee, United Kingdom, DD1 9SY Contact: Ruth LM Cordiner, MBChB, MRCP 0044 1382 386 715 ext 00441382386715 r.cordiner@dundee.ac.uk Principal Investigator: Ruth LM Cordiner Collapse << |
| NCT03652987 | - | - | Not yet recruiting | September 2023 | - |
| NCT02024477 | Type 2 Diabetes | Phase 4 | Active, not recruiting | January 2018 | United States, District of Col... more >>umbia Medical Faculty Associates Inc Washington, District of Columbia, United States, 20037 Collapse << |
| NCT00036504 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, California ... more >> LaJolla, California, United States Walnut Creek, California, United States United States, Connecticut New Britain, Connecticut, United States United States, Florida Longwood, Florida, United States United States, Georgia Atlanta, Georgia, United States Fayetteville, Georgia, United States United States, Idaho Boise, Idaho, United States United States, Illinois Springfield, Illinois, United States United States, New Jersey Mt. Laurel, New Jersey, United States New Brunswick, New Jersey, United States United States, New York Syracuse, New York, United States United States, Tennessee Chattanooga, Tennessee, United States United States, Texas Dallas, Texas, United States Collapse << |
| NCT03766308 | Type 2 Diabetes Mellitus | Not Applicable | Active, not recruiting | May 31, 2019 | China, Hubei ... more >> Tongji Hospital, Tongji Medical College, Huazhong University of Science and technology Wuhan, Hubei, China Collapse << |
| NCT02826525 | T2DM With NAFLD | Phase 1 Phase 2 | Active, not recruiting | December 28, 2020 | - |
| NCT01986855 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01055223 | - | - | Completed | - | - |
| NCT01055223 | - | - | Completed | - | - |
| NCT03376490 | - | - | Completed | - | Singapore ... more >> SingHealth Polyclinics - Marine Parade Polyclinic Singapore, Singapore, 440080 Collapse << |
| NCT02608905 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | May 2019 | United States, Texas ... more >> Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Mandeep Bajaj, MD 713-798-1712 bajaj@bcm.edu Collapse << |
| NCT01606397 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Singapore ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Singapore, Singapore United Kingdom For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Plymouth, United Kingdom Collapse << |
| NCT03536364 | Obesity Pre-D... more >>iabetes Collapse << | Not Applicable | Completed | - | United States, New York ... more >> Comprehensive Weight Control Center New York, New York, United States, 10065 Collapse << |
| NCT01986855 | - | - | Completed | - | - |
| NCT01272232 | Metabolism and Nutrition Disor... more >>der Obesity Collapse << | Phase 3 | Completed | - | - |
| NCT00659711 | Type 2 Diabetes | Not Applicable | Unknown | December 2013 | United States, New York ... more >> 115 Flint Road Buffalo, New York, United States, 14221 Collapse << |
| NCT03735108 | - | - | Recruiting | June 30, 2019 | China ... more >> Shanghai First Maternity and Infant Hospital Recruiting Shanghai, China Contact: Shihua Bao 86-021-20261599 baoshihua@tongji.edu.cn Collapse << |
| NCT01272232 | - | - | Completed | - | - |
| NCT00642278 | - | - | Completed | - | - |
| NCT01869959 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cypress, California, United States, 90630 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tustin, California, United States, 92780 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Deland, Florida, United States, 32720 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miramar, Florida, United States, 33025 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45212 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97239 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 Collapse << |
| NCT00642278 | Diabetes Mellitus, Type II ... more >> Diabetes Mellitus, Non Insulin Dependent Collapse << | Phase 2 | Completed | - | - |
| NCT00546325 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Australia ... more >> Sanofi-Aventis Administrative Office North Ryde, Australia Collapse << |
| NCT02235194 | Type 2 Diabetes, ... more >> Prediabetes Collapse << | Not Applicable | Completed | - | Germany ... more >> Stoffwechselzentrum Rhein Pfalz Mannheim, Germany, 68163 Collapse << |
| NCT01869959 | - | - | Completed | - | - |
| NCT02197520 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research Inc Chula Vista, California, United States, 91911 Collapse << |
| NCT02197520 | - | - | Completed | - | - |
| NCT03409523 | - | - | Enrolling by invitation | December 2018 | United States, New York ... more >> New York University School of Medicine New York, New York, United States, 10016 Collapse << |
| NCT02798757 | Diabetes | Phase 4 | Recruiting | May 2018 | Greece ... more >> University of Ioannina Recruiting Ioánnina, Greece, 45500 Contact: Vasilis Tsimihodimos, Assistant Professor +306971705609 vtsimi@uoi.gr Collapse << |
| NCT01357889 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01357889 | - | - | Completed | - | - |
| NCT01936025 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT03021187 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00502281 | Polycystic Ovary Syndrome | Phase 4 | Suspended | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT02119819 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00375492 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Peoria, Arizona, United States United States, Florida Research Site Jacksonville, Florida, United States Research Site Orlando, Florida, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Minnesota Research Site Minneapolis, Minnesota, United States United States, Missouri Research Site St. Louis, Missouri, United States United States, South Carolina Research Site Spartanburg, South Carolina, United States United States, Texas Research Site San Antonio, Texas, United States United States, Washington Research Site Renton, Washington, United States Collapse << |
| NCT00044694 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 2 | Completed | - | - |
| NCT00800176 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT03244579 | Gestational Diabetes Mellitus ... more >>With Baby Delivered Collapse << | Not Applicable | Recruiting | October 20, 2019 | Jordan ... more >> Sabika Allehdan Recruiting Amman, Jordan, 009626 Contact: Sabika Allehdan, MSc 799232789 sabika_1986@Yahoo.com Collapse << |
| NCT01986881 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | September 11, 2019 | - |
| NCT00482742 | Catheterization, Peripheral | Not Applicable | Terminated(product development... more >> was cancelled) Collapse << | - | Croatia ... more >> University Hospital Split Split, Croatia, 21000 Clinical Hospital Centre Zagreb Zagreb, Croatia, 10000 Israel Rabin Campus, Belinson Hospital Petah-Tikva, Israel, 49100 Collapse << |
| NCT01512108 | - | - | Completed | - | - |
| NCT01512108 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Japan ... more >> Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0002 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0027 Novo Nordisk Investigational Site Ehime, Japan, Japan, 790 0067 Novo Nordisk Investigational Site Fukuoka, Japan, Japan, 810 8798 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 815 8555 Novo Nordisk Investigational Site Fukuoka, Japan, 812 0025 Novo Nordisk Investigational Site Fukuoka, Japan, 818 8502 Novo Nordisk Investigational Site Fukuoka, Japan, 820 8505 Novo Nordisk Investigational Site Kamagaya-shi, Chiba, Japan, 273 0121 Novo Nordisk Investigational Site Kashiwara-shi, Osaka, Japan, 582 0005 Novo Nordisk Investigational Site Kawagoe-shi, Saitama, Japan, 350 0851 Novo Nordisk Investigational Site Kitakyushu-shi, Fukuoka, Japan, 800 0252 Novo Nordisk Investigational Site Koriyama-shi, Fukushima, Japan, 963 8851 Novo Nordisk Investigational Site Kumamoto-shi,Kumamoto, Japan, 862 0976 Novo Nordisk Investigational Site Kurume-shi, Fukuoka, Japan, 839 0863 Novo Nordisk Investigational Site Miyazaki-shi, Japan, 880 0034 Novo Nordisk Investigational Site Naka-shi, Ibaraki, Japan, 311 0113 Novo Nordisk Investigational Site Niigata-shi, Niigata, Japan, 950 1104 Novo Nordisk Investigational Site Oita, Japan, 870 8511 Novo Nordisk Investigational Site Okawa-shi, Fukuoka, Japan, 831 0016 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 532 0003 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 545 8586 Novo Nordisk Investigational Site Ota-ku, Tokyo, Japan, 144 0035 Novo Nordisk Investigational Site Oyama-shi, Tochigi, Japan, 323 0022 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060-0001 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 062 0007 Novo Nordisk Investigational Site Shimotsuke-shi, Tochigi, Japan, 329 0433 Novo Nordisk Investigational Site Shizuoka-shi, Japan, 424 0853 Novo Nordisk Investigational Site Tagajo-shi, Japan, 985 0852 Novo Nordisk Investigational Site Takatsuki-shi, Osaka, Japan, 569 1096 Novo Nordisk Investigational Site Tochigi, Japan, 329 0498 Novo Nordisk Investigational Site Tokyo, Japan, 104 0044 Novo Nordisk Investigational Site Tokyo, Japan, 113 0031 Novo Nordisk Investigational Site Tokyo, Japan, 125 0054 Novo Nordisk Investigational Site Yamaguchi, Japan, 755 0067 Novo Nordisk Investigational Site Yokohama-shi, Japan, 235 0045 Collapse << |
| NCT00375492 | - | - | Completed | - | - |
| NCT01618214 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Anhui ... more >> Novo Nordisk Investigational Site Hefei, Anhui, China, 230001 Novo Nordisk Investigational Site Hefei, Anhui, China, 230022 China, Beijing Novo Nordisk Investigational Site Beijing, Beijing, China, 100029 Novo Nordisk Investigational Site Beijing, Beijing, China, 100088 Novo Nordisk Investigational Site Beijing, Beijing, China, 100700 Novo Nordisk Investigational Site Beijing, Beijing, China, 100730 China, Chongqing Novo Nordisk Investigational Site Chongqing, Chongqing, China, 400010 Novo Nordisk Investigational Site ChongQing, Chongqing, China, 404000 China, Guangdong Novo Nordisk Investigational Site Guangzhou, Guangdong, China, 510080 China, Hebei Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050051 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050082 China, Hubei Novo Nordisk Investigational Site Wuhan, Hubei, China, 430034 China, Inner Mongolia Novo Nordisk Investigational Site Tongliao, Inner Mongolia, China, 028007 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210011 Novo Nordisk Investigational Site Yangzhou, Jiangsu, China, 225001 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Jilin Novo Nordisk Investigational Site Changchun, Jilin, China, 130041 Novo Nordisk Investigational Site Siping, Jilin, China, 136000 China, Shaanxi Novo Nordisk Investigational Site Xi'an, Shaanxi, China, 710032 China, Shandong Novo Nordisk Investigational Site Jinan, Shandong, China, 250013 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 China, Zhejiang Novo Nordisk Investigational Site Hangzhou, Zhejiang, China, 310003 Collapse << |
| NCT03278470 | Rheumatoid Arthritis | Phase 1 | Completed | - | Korea, Republic of ... more >> The catholic university of korea seoul ST. mary's hospital Soeul, Korea, Republic of Collapse << |
| NCT01336023 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01618214 | - | - | Completed | - | - |
| NCT00251940 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT02371187 | Physical Activity | Phase 2 | Completed | - | United States, Colorado ... more >> Colorado State University, Dept. of Health and Exercise Science Fort Collins, Colorado, United States, 80523-1582 Collapse << |
| NCT00736099 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT03086330 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01628445 | Type 2 Diabetes | Phase 3 | Terminated(Error made by local... more >> pharmacy caused mixed randomization of 20 participants) Collapse << | - | Canada, Manitoba ... more >> Winnipeg Regional Health Authority Health Sciences Centre Winnipeg Diabetes Research Group Winnipeg, Manitoba, Canada, R3E3P4 Collapse << |
| NCT00736099 | - | - | Completed | - | - |
| NCT02964572 | Type2 Diabetes Mellitus | Not Applicable | Recruiting | October 2018 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT02240680 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01336023 | - | - | Completed | - | - |
| NCT02240680 | - | - | Completed | - | - |
| NCT01570751 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01098539 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01570751 | - | - | Completed | - | - |
| NCT00156897 | Non-Insulin-Dependent Diabetes... more >> Mellitus Obesity Collapse << | Phase 2 | Completed | - | Denmark ... more >> Aalborg Sygehus Nord Aalborg, Denmark Aarhus University Hospital Aarhus, Denmark, DK-8000 Bispebjerg Hospital Copenhagen, Denmark, DK-2400 Gentofte Hospital Hellerup, Denmark, DK-2900 Hvidovre Hospital Hvidovre, Denmark, DK-2650 Odense University Hospital Odense, Denmark Finland Lääkärikeskus Minerva Eura, Finland, 27510 Obesity Research Unit Helsinki, Finland, 00100 Suomen Terveystalo Jyvaskyla, Finland, 40100 Oy Foodfiles Limited Kuopio, Finland Oulun Diakonissalaitos Oulu, Finland, 90100 University of Oulu Oulu, Finland Turku University Hospital Turku, Finland, 20520 Netherlands Allevon Den Bosch Ziekenhuis, Netherlands Allevon DN de Bilt, Netherlands Zeikenhuisgroep Twente Hengelo, Netherlands Nederlandse Obesitas Klinik Hilversum, Netherlands Sint Franciscus Gasthuis Rotterdam, Netherlands Sweden Sahlgrenska University Hospital Goteborg, Sweden Linkoping University Hospital Linkoping, Sweden Karolinsaka University Hospital Stockholm, Sweden Norrlands Universitetssjukhus Umea, Sweden Samariterhemmets Sjukhus Uppsala, Sweden United Kingdom Aberdeen Royal Infirmary Aberdeen, United Kingdom Royal United Hospital Bath, United Kingdom Clinical Research Centre Edgbaston Birmingham, United Kingdom Walsgrave Hospital Coventry, United Kingdom Glasgow Royal Infirmary Glasgow, United Kingdom Clinical Research Centre, Crosby Liverpool, United Kingdom Liverpool University Hospital Liverpool, United Kingdom Queen Mary's School of Medicine & Dentistry London, United Kingdom, E1 2AA Hammersmith Hospital London, United Kingdom Luton & Dunstable Hospital Luton, United Kingdom Clinical Research Centre Manchester, United Kingdom James Cook University Hospital Middlesborough, United Kingdom Royal Shrewsbury Hospital Shrewsbury, United Kingdom Clinical Research Centre Wigan, United Kingdom Collapse << |
| NCT01098539 | - | - | Completed | - | - |
| NCT00087516 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01096940 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00802100 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> SHANTI Clinical Trials Colton, California, United States, 92324 Stanford University Palo Alto, California, United States, 94305 United States, Connecticut Yale University New Haven, Connecticut, United States, 06519 United States, Florida University of Miami School of Medicine Miami, Florida, United States, 33316 United States, Georgia Medical College of Georgia Augusta, Georgia, United States, 30912 United States, Maryland Clinical Insights Glen Burnie, Maryland, United States, 21061 United States, Massachusetts University of Massachusetts Worcester, Massachusetts, United States, 01605 United States, Michigan Wayne State University Detroit, Michigan, United States, 48201 United States, Minnesota University of Minnesota School of Medicine Minneapolis, Minnesota, United States, 55454 United States, New York Research Foundation for Mental Hygiene New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center-John Umstead Hospital Butner, North Carolina, United States, 27509 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75235 Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00087516 | - | - | Completed | - | - |
| NCT03225209 | Type 2 Diabetes Mellitus | Phase 2 | Active, not recruiting | August 15, 2018 | United States, North Carolina ... more >> Department of Veteran Affairs, Greenville Health Care Center Greenville, North Carolina, United States, 27834 Collapse << |
| NCT01175824 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Argentina ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mar Del Plata, Argentina, B7600FZN For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ramos Mejia, Argentina, B1704ETD Brazil For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Porto Alegre, Brazil, 90035-170 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. São Paulo, Brazil, 01244-030 China For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changzhou, China, 213003 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150086 Egypt For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alexandria, Egypt For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cairo, Egypt, 11562 India For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jaipur, India, 302018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vishakhapatnam, India, 530002 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daegu, Korea, Republic of, 700-712 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Goyang-Si, Korea, Republic of, 410-719 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyunggi-Do, Korea, Republic of, 425-020 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 134-090 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Málaga, Spain, 29010 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Boi De Llobregat, Spain, 08830 Turkey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Antalya, Turkey, 07070 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Istanbul, Turkey, 34865 Collapse << |
| NCT02106104 | Type 2 Diabetes | Phase 4 | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT00802100 | - | - | Completed | - | - |
| NCT01175824 | - | - | Completed | - | - |
| NCT01664624 | - | - | Completed | - | - |
| NCT01338870 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01103609 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT02043054 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> University Hospitals of Leicester NHS Trust, Diabetes Research Centre Leicester, Leicestershire, United Kingdom, LE5 4PW Collapse << |
| NCT01119846 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Phoenix, Arizona, United States, 85013 United States, California GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio GSK Investigational Site Columbus, Ohio, United States, 43212 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 United States, Washington GSK Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT01664624 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Chula Vista, California, United States United States, Florida Orlando, Florida, United States Collapse << |
| NCT01338870 | - | - | Completed | - | - |
| NCT00997152 | Type II Diabetes Mellitus | Phase 2 | Terminated | - | - |
| NCT01336738 | Diabetes, Type 2 | Phase 2 | Completed | - | - |
| NCT03747107 | - | - | Recruiting | March 30, 2020 | United Kingdom ... more >> NHS Tayside Recruiting Dundee, Scotland, United Kingdom, DD2 4BF Contact: Tobias Dreischulte, PhD ++44(0)7540736211 t.dreischulte@dundee.ac.uk Collapse << |
| NCT03626909 | - | - | Recruiting | January 13, 2021 | Guatemala ... more >> Hospital Obras Sociales Recruiting San Lucas Tolimán, Solala, Guatemala Contact: Sean Duffy, MD +502 4607-0561 Sean.Duffy@fammed.wisc.edu Contact: , MD Collapse << |
| NCT02456428 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02588729 | Gestational Diabetes Mellitus | Not Applicable | Completed | - | Norway ... more >> Oslo university hospital Oslo, Norway, 4950 Collapse << |
| NCT03015519 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(The study was stoppe... more >>d as the asset will be withdrawn from the market prior to study end.) Collapse << | April 20, 2020 | United States, Illinois ... more >> GSK Investigational Site Chicago, Illinois, United States, 60634 United States, North Carolina GSK Investigational Site Morehead City, North Carolina, United States, 28557 United States, Texas GSK Investigational Site El Paso, Texas, United States, 79935 Collapse << |
| NCT02475499 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02476760 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00690755 | - | - | Completed | - | - |
| NCT01119846 | - | - | Completed | - | - |
| NCT00690755 | - | - | Completed | - | United States, Florida ... more >> VA Medical Center Tampa, Florida, United States, 33612 Collapse << |
| NCT01916694 | Gestational Diabetes Mellitus ... more >> Pregnancy Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> John Radcliffe Hospital Oxford, Oxfordshire, United Kingdom, OX3 9DU Collapse << |
| NCT00580710 | - | - | Recruiting | August 2020 | United States, Connecticut ... more >> Yale University School of Medicine Recruiting New Haven, Connecticut, United States, 06520 Principal Investigator: Robert Sherwin, M.D. Collapse << |
| NCT01974544 | Type 2 Diabetes | Not Applicable | Unknown | December 2016 | Chile ... more >> Pontificia Universidad Católica de Chile Recruiting Santiago, Región Metropolitana, Chile, 8330033 Contact: Castillo Alejandra, Nurse 56223543527 macastia@uc.cl Contact: Vega Andrea, Nurse 56223543879 abvega@uc.cl Principal Investigator: Boza Camilo, MD surgeon Sub-Investigator: Valderas Juan Patricio, MD Sub-Investigator: Arrese Marco, MD Internist Sub-Investigator: Muñoz Rodrigo, MD Collapse << |
| NCT00000620 | Atherosclerosis ... more >> Cardiovascular Diseases Hypercholesterolemia Hypertension Diabetes Mellitus, Type 2 Diabetes Mellitus Coronary Disease Collapse << | Phase 3 | Completed | - | United States, Minnesota ... more >> Minneapolis Medical Research Foundation Minneapolis, Minnesota, United States, 55404 United States, New York Columbia University New York, New York, United States, 10027 United States, North Carolina Wake Forest University Winston-Salem, North Carolina, United States, 27106 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Tennessee Veterans Affairs Memphis, Tennessee, United States, 38104 United States, Washington University of Washington Seattle, Washington, United States, 98195 Canada, Ontario McMaster University Hamilton, Ontario, Canada Collapse << |
| NCT03426228 | - | - | Recruiting | December 31, 2019 | United Arab Emirates ... more >> Fakih IVF Recruiting Dubai, United Arab Emirates, 72960 Contact: Ayla Coussa, Dietitian 00971 4 349 7600 ext 162 a.coussa@warwick.ac.uk Contact: Michael Fakih, MD 00971 4 349 7600 ext 117 mfakih@fakihivf.com Collapse << |
| NCT02259972 | Healthy | Phase 1 | Completed | - | - |
| NCT02033499 | - | - | Completed | - | - |
| NCT01868646 | Type II Diabetes Mellitus | Phase 4 | Completed | - | Russian Federation ... more >> Municipal budgetary authority "Khimki Central Clinical Hospital" Moscow region, Russian Federation, 141400 State Healthcare Institution of Moscow "Central research institute of gastroenterology" of Department of health care of Moscow Moscow, Russian Federation, 111123 State Educational Institution of Higher Professional Education "Moscow State Medical Academy named after I.M. Sechenov" Moscow, Russian Federation, 119991 Nonstate Health Care Institution "Central Clinical Hospital №2 named after N.A. Semashko of Public Limited Company "Russian Railways" Moscow, Russian Federation, 129128 Nizhny Novgorod regional State Budgetary Health Institution " Nizhny Novgorod regional Clinical Hospital named after N.A. Semashko " Nizhny Novgorod Region, Russian Federation, 603126 State Budgetary Educational Institution of High Professional Training "Rostov State Medical University" of Ministry of Health of Russian Federation, Department of Endocrinology Rostov-on-Don, Russian Federation, 344022 Co.Ltd " Diabet Center" Samara, Russian Federation, 443067 The State Budgetary Educational Institution of Higher Professional Education "Smolensk National Research Medical University" Ministry of Health of the Russian Federation Smolensk, Russian Federation, 214019 St. Petersburg State Budgetary Health Care Institution "City Polyclinic №6" St. Petersburg, Russian Federation, 191482 St. Petersburg State Budgetary Health Care Institution "Saint Venerable Martyr Elizaveta Municipal Hospital" St. Petersburg, Russian Federation, 195257 State Budgetary Educational Institution of High Professional Training "St. Petersburg State Medical University named after academician I.P. Pavlov" of Ministry of Health of Russian Federation, Therapy Faculty Board St. Petersburg, Russian Federation, 197022 St. Petersburg Sate Budgetary Institution "Consultative-Diagnostic Polyclinic №1 of Coastal Area" St. Petersburg, Russian Federation, 197183 St. Petersburg State Budgetary Health Care Institution "Consultative-Diagnostic Center № 85", Diabetes Center №2 St. Petersburg, Russian Federation, 198260 St. Petersburg State Budgetary Health Care Institution "City Polyclinic №77 of Nevsky District", The City Diabetes Center №4 St.Petersburg, Russian Federation, 192177 Vladimir region State budgetary institution of health care "Regional clinical hospital" Vladimir, Russian Federation, 600023 Independent Health Care Institution of Voronezh Region "Voronezh Regional Clinical Consultative-Diagnostic Center" Voronezh, Russian Federation, 394018 State Budgetary Health Care Institution of Yaroslavl Region "Regional Сlinical Hospital" Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT02138097 | - | - | Completed | - | United States, Massachusetts ... more >> 1218.161.1 Boehringer Ingelheim Investigational Site Boston, Massachusetts, United States Collapse << |
| NCT02033499 | DIABETES MELLITUS, NONINSULIN-... more >>DEPENDENT, 2 (Disorder) Collapse << | Not Applicable | Completed | - | United States, North Carolina ... more >> The University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01923181 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT02430870 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Anaheim, California, United States Chula Vista, California, United States Collapse << |
| NCT02138097 | - | - | Completed | - | - |
| NCT00000620 | - | - | Completed | - | - |
| NCT03732209 | Type 2 Diabetes | Phase 1 Phase 2 | Not yet recruiting | July 31, 2020 | United States, Virginia ... more >> Virginia Tech Carilion Research Institute Roanoke, Virginia, United States, 24016 Collapse << |
| NCT00661921 | Diabetes Mellitus | Phase 2 | Withdrawn(Program Development ... more >>discontinued) Collapse << | October 2009 | - |
| NCT02430870 | - | - | Completed | - | - |
| NCT00392678 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | United States, Connecticut ... more >> Chapel Medical Group New Haven, Connecticut, United States, 06511 United States, District of Columbia MedStar Research Institute Washington, District of Columbia, United States, 20003-4393 United States, Florida Endocrine Clinical Research Winter Park, Florida, United States, 32746 United States, Georgia Kaiser Permanente Atlanta, Georgia, United States, 30084 Emory School of Medicine Atlanta, Georgia, United States, 30303 United States, Illinois University of Illinois at Chicago Chicago, Illinois, United States, 60612 United States, Louisiana Tulane University New Orleans, Louisiana, United States, 70112 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68105 United States, New York Kaleida Health Center Buffalo, New York, United States, 14226 North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States, 11042 Columbia University New York, New York, United States, 10032 University of Rochester Medical Center Rochester, New York, United States, 14642 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Texas University of Texas Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT00328627 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00392678 | - | - | Completed | - | - |
| NCT00799643 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham, Alabama, United States United States, California University of California, San Diego San Diego, California, United States United States, Connecticut Chapel Medical Group New Haven, Connecticut, United States United States, Georgia Emory University School of Medicine Atlanta, Georgia, United States Kaiser Permanente Tucker, Georgia, United States United States, Indiana Indiana University Indianapolis, Indiana, United States United States, Louisiana Tulane University Health Sciences Center New Orleans, Louisiana, United States United States, Maryland Medstar Research Institute Hyattsville, Maryland, United States Dr. Rudo, Westminster, MD Westminster, Maryland, United States, 21157 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48106 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States United States, New York North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States Columbia University New York, New York, United States Lang Medical Center Queens, New York, United States Albert Einstein College of Medicine The Bronx, New York, United States United States, North Carolina Carolina's Health Care Charlotte, North Carolina, United States University of North Carolina Durham, North Carolina, United States United States, Texas University of Texas Southwestern Dallas, Texas, United States Scott and White Temple, Texas, United States Collapse << |
| NCT00799643 | - | - | Completed | - | - |
| NCT02282423 | Diabetes Obes... more >>ity Collapse << | Not Applicable | Terminated(PI job change) | - | United States, Arizona ... more >> Mayo Clinic Scottsdale, Arizona, United States, 85259 Collapse << |
| NCT01703208 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(The study was termi... more >>nated for business reasons and not due to any safety or efficacy concerns related to omarigliptin) Collapse << | - | - |
| NCT00571519 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(DSPD focussing on S... more >>tudy 301 to confirm clinical profile before proceeding.) Collapse << | - | United States, Alabama ... more >> Huntsville, Alabama, United States, 35801 Huntsville, Alabama, United States, 35802 United States, California San Diego, California, United States, 92128 San Francisco, California, United States, 94115 United States, Connecticut Ridgefield, Connecticut, United States, 06877 United States, Florida Fort Lauderdale, Florida, United States, 33308 Pembroke Pines, Florida, United States, 33026 United States, Mississippi Port Gibson, Mississippi, United States, 39150 United States, Nevada Las Vegas, Nevada, United States, 89119 United States, New Mexico Albuquerque, New Mexico, United States, 87109 United States, Ohio Kettering, Ohio, United States, 45429 United States, Pennsylvania Fleetwood, Pennsylvania, United States, 19522 Harrisburg, Pennsylvania, United States, 17112 United States, South Carolina Simpsonville, South Carolina, United States, 29681 United States, Texas Daingerfield, Texas, United States, 75638 Dallas, Texas, United States, 75216 Plano, Texas, United States, 75093 San Antonio, Texas, United States, 78205 Collapse << |
| NCT01106651 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00935805 | - | - | Unknown | July 2012 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT01106651 | - | - | Completed | - | - |
| NCT01221519 | Type 2 Diabetes Mellitus ... more >> High Blood Sugar Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Research Site St. Paul, Minnesota, United States Collapse << |
| NCT00263276 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00328627 | - | - | Completed | - | - |
| NCT03359837 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | November 7, 2019 | China ... more >> Recruiting Not Listed, China Collapse << |
| NCT03568630 | - | - | Recruiting | July 2023 | United States, Nebraska ... more >> Nebraska Medical Center Recruiting Omaha, Nebraska, United States, 68198 Contact: Christina Hoy, DNP 402-559-1577 christina.hoy@unmc.edu Collapse << |
| NCT02954692 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Turkey ... more >> Turkey, Turkey Collapse << |
| NCT01798706 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03685357 | - | - | Recruiting | January 2020 | Egypt ... more >> AinShams University Recruiting Cairo, Egypt Principal Investigator: Afnan Elgnainy Collapse << |
| NCT01798706 | - | - | Completed | - | - |
| NCT01767389 | - | - | Completed | - | - |
| NCT03049228 | - | - | Enrolling by invitation | April 2, 2019 | Netherlands ... more >> Human Biology Maastricht, Limburg, Netherlands, 6200MD Collapse << |
| NCT02157974 | Hepatic Steatosis ... more >> Polycystic Ovarian Syndrome Obesity Collapse << | Phase 2 Phase 3 | Recruiting | December 2018 | United States, Colorado ... more >> University of Colorado Anshutz Medical Campus/Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Yesenia Garcia 720-777-6984 yesenia.garciareyes@childrenscolorado.org Collapse << |
| NCT00940797 | Diabetes Type 2 | Phase 2 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara, México Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT01922791 | Pregnancy Obe... more >>sity Gestational Diabetes Mellitus Collapse << | Not Applicable | Active, not recruiting | January 2020 | Finland ... more >> University of Turku Turku, Finland, 20014 Turku University Hospital Turku, Finland, 20521 Collapse << |
| NCT03552991 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03491241 | Pre-diabetes ... more >>Obesity Collapse << | Not Applicable | Completed | - | Italy ... more >> Raffaele Marfella Naples, Italy, 80138 Collapse << |
| NCT01703208 | - | - | Terminated(The study was termi... more >>nated for business reasons and not due to any safety or efficacy concerns related to omarigliptin) Collapse << | - | - |
| NCT02272556 | Hyperglycemia | Early Phase 1 | Recruiting | November 2019 | United States, Connecticut ... more >> Yale University Recruiting New Haven, Connecticut, United States, 06519 Contact: Janice Hwang, MD 203-785-6222 janice.hwang@yale.edu Collapse << |
| NCT03585777 | - | - | Not yet recruiting | March 2019 | Egypt ... more >> Fayoum university Hospital Recruiting Fayoum, Egypt Contact: Ahmed Hammad, MD Collapse << |
| NCT01077323 | - | - | Completed | - | - |
| NCT01213563 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United Kingdom ... more >> Imperial College London, Charing Cross Hospital London, United Kingdom, W68NA Collapse << |
| NCT02368704 | - | - | Unknown | November 2016 | France ... more >> Saint-Louis Hospital Paris, France, 75010 Collapse << |
| NCT00015626 | Insulin Resistance ... more >> Diabetes Mellitus Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Cornell University New York, New York, United States, 10021 Collapse << |
| NCT01077323 | - | - | Completed | - | United States, Massachusetts ... more >> Research Site Waltham, Massachusetts, United States Collapse << |
| NCT02219750 | Diabetes | Phase 4 | Completed | - | Taiwan ... more >> Mackay Memerial Hospital Taipei, Taiwan Collapse << |
| NCT02269098 | - | - | Completed | - | - |
| NCT00894322 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Nebraska ... more >> Research Site Lincoln, Nebraska, United States Collapse << |
| NCT02269098 | Diabetes Mellitus, Type 2 ... more >> Medication Adherence HYPERGLYCEMIA Collapse << | Not Applicable | Completed | - | United States, District of Col... more >>umbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 Collapse << |
| NCT01289990 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01042379 | Breast Neoplasms ... more >> Breast Cancer Breast Tumors Collapse << | Phase 2 | Recruiting | - | - |
| NCT00894322 | - | - | Completed | - | - |
| NCT01421459 | - | - | Completed | - | - |
| NCT01417897 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | May 2012 | Germany ... more >> ife GmbH, Clinic Mainz, Rhineland-Palatinate, Germany, 55116 Collapse << |
| NCT01121315 | - | - | Completed | - | Sweden ... more >> Research Site Uppsala, Sweden Collapse << |
| NCT02029846 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(No sufficient numbe... more >>rs of eligible participants) Collapse << | - | United States, Maryland ... more >> Bay West Endocrinology Associates Baltimore, Maryland, United States, 21204 Collapse << |
| NCT02453685 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Australia, New South Wales ... more >> Novo Nordisk Investigational Site Coffs Harbour, New South Wales, Australia, 2450 Bulgaria Novo Nordisk Investigational Site Petrich, Bulgaria, 2850 Hungary Novo Nordisk Investigational Site Budapest, Hungary, 1042 India Novo Nordisk Investigational Site New Delhi, India, 110001 Korea, Republic of Novo Nordisk Investigational Site Seoul, Korea, Republic of, 138-736 Serbia Novo Nordisk Investigational Site Belgrade, Serbia, 11080 Thailand Novo Nordisk Investigational Site Bangkok, Thailand, 10330 Turkey Novo Nordisk Investigational Site Istanbul, Turkey, 34752 United Arab Emirates Novo Nordisk Investigational Site Ras Al Khaimah, United Arab Emirates, 4727 Collapse << |
| NCT02805283 | - | - | Completed | - | United States, Minnesota ... more >> Reasearch Site Eden Prairie, Minnesota, United States Collapse << |
| NCT02029846 | - | - | Terminated(No sufficient numbe... more >>rs of eligible participants) Collapse << | - | - |
| NCT01940302 | Type 2 Diabetes | Early Phase 1 | Completed | - | China, Guangdong ... more >> Shaoguan University Shaoguan, Guangdong, China, 512005 Collapse << |
| NCT01935466 | - | - | Recruiting | July 2021 | India ... more >> Deptt of Endocrinology Recruiting Chandigarh, India, 160012 Contact: Ashu Rastogi, MD, DM 919781001046 ashuendo@gmail.com Principal Investigator: Ashu Rastogi, MD, DM Collapse << |
| NCT01421459 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00481429 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United Kingdom ... more >> Addenbrooke's Hospital Cambridge, United Kingdom, CB2 2GG Ealing Hospital London, United Kingdom, UB1 3HW Imperial College London - Hammersmith Campus London, United Kingdom, W12 ONN Charing Cross Hospital London, United Kingdom, W6 8RF Collapse << |
| NCT01270191 | Type 2 Diabetes | Phase 4 | Unknown | December 2015 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD 886-2-28757515 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT02417012 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for Physical Activity Research, Copenhagen University Hospital Copenhagen, Denmark, 2100 Collapse << |
| NCT00790348 | - | - | Unknown | - | United States, Michigan ... more >> Wayne State University Recruiting Detroit, Michigan, United States, 48201 Contact: Berhane Seyoum, MD 313-745-4008 bseyoum@med.wayne.edu Principal Investigator: Berhane Seyoum, MD, MPH Collapse << |
| NCT01759823 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | India ... more >> Postgraduate Institute of Medical Education and Research Chandigarh, UT, India, 160012 Collapse << |
| NCT03172494 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Recruiting | July 29, 2019 | - |
| NCT03511118 | - | - | Recruiting | September 22, 2019 | United States, North Carolina ... more >> Duke University Maternal and Fetal Medicine Recruiting Durham, North Carolina, United States, 27705 Contact: Kristin Weaver 919-613-9630 obresearch@duke.edu Principal Investigator: Brenna Hughes, MD United States, Washington University of Washington Recruiting Seattle, Washington, United States, 98195-7630 Contact: Maggie Leahy 540-525-0121 maleahy@u.washington.edu Principal Investigator: Mary Hébert, Pharm.D Collapse << |
| NCT02060383 | Cushing's Disease ... more >> Acromegaly Collapse << | Phase 4 | Completed | - | - |
| NCT01289990 | - | - | Completed | - | - |
| NCT01785043 | DIABETES Mellitus Type 2 Not W... more >>ell Controlled Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Clínic de Barcelona Barcelona, Spain, 08036 Collapse << |
| NCT01095822 | Diabetes Mellitus | Phase 4 | Unknown | March 2011 | United States, Maryland ... more >> Dr. Pokov's Polyclinic. 6821 Reisterstown Road Suite 206 Baltimore, Maryland, United States, 21215 Collapse << |
| NCT02917629 | Oral Cavity Neoplasm ... more >> Oropharyngeal Neoplasm Stage 0 Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage 0 Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage I Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage I Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage II Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage II Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage III Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage III Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVA Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVA Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVB Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVB Oropharyngeal Squamous Cell Carcinoma AJCC v7 Collapse << | Phase 2 | Recruiting | March 31, 2019 | United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Recruiting Baltimore, Maryland, United States, 21287 Contact: Wayne M. Koch 410-995-4906 wkoch@jhmi.edu Principal Investigator: Wayne M. Koch United States, Minnesota University of Minnesota/Masonic Cancer Center Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Frank G. Ondrey 612-625-3200 ondre002@umn.edu Principal Investigator: Frank G. Ondrey United States, New York University of Rochester Recruiting Rochester, New York, United States, 14642 Contact: Shawn D. Newlands 585-273-1943 Shawn_Newlands@URMC.Rochester.edu Principal Investigator: Shawn D. Newlands United States, Wisconsin University of Wisconsin Hospital and Clinics Recruiting Madison, Wisconsin, United States, 53792 Contact: Aaron M. Wieland 608-265-8207 wieland@surgery.wisc.edu Principal Investigator: Aaron M. Wieland Collapse << |
| NCT01584232 | - | - | Completed | - | - |
| NCT01584232 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 448-0852 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 286-0048 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ehime, Japan, 790-0024 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 819-0168 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 0530018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kagoshima, Japan, 8910401 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 247-0055 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kumamoto, Japan, 860-0811 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 606-8397 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 385-0022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagasaki, Japan, 857-1195 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ooita, Japan, 8700039 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 598-0048 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 103-0028 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yamaguchi, Japan, 754-0002 Collapse << |
| NCT02244164 | Type 2 Diabetes ... more >> Incretinomimetics Pancreas Collapse << | Not Applicable | Recruiting | - | Belgium ... more >> Gastroenterology Department Erasme Hospital Recruiting Brussels, Belgium, 1070 Contact: Vincent Huberty, MD +3225553715 vincent.huberty@erasme.ulb.ac.be Collapse << |
| NCT01586897 | Diabetes Mellitus, Type 2 ... more >> Hypertension Hyperlipidemia Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03173391 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 31, 2019 | China, Shanghai ... more >> Hua Medicine Limited Recruiting Shanghai, Shanghai, China, 201203 Contact: YI ZHANG, PhD +86-021-58863005 yzhang@huamedicine.com Principal Investigator: Dalong Zhu, PhD Collapse << |
| NCT00819325 | Coronary Artery Disease ... more >> Angina Pectoris Type 2 Diabetes Mellitus Percutaneous Coronary Intervention Collapse << | Phase 4 | Completed | - | Canada, Nova Scotia ... more >> Queen Elizabeth II Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 3A7 Collapse << |
| NCT02110979 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Illinois ... more >> EPI-Q Inc Oak Brook, Illinois, United States, 60523 EPI-Q Oak Brook, Illinois, United States, 60523 Collapse << |
| NCT00990444 | Diabetes Mellitus | Phase 1 Phase 2 | Suspended | December 2010 | Sweden ... more >> Karolinska Trial Alliance, Phase I Unit Stockholm, Solna, Stockholm, Sweden, 17176 Collapse << |
| NCT02298192 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01586897 | - | - | Completed | - | - |
| NCT01781754 | - | - | Unknown | January 2014 | Israel ... more >> Tel Aviv Sourasky Medical Center Recruiting Tel-Aviv, Israel, 64239 Contact: Nachum Vaisman, MD 972-3-6974807 vaisman@tasmc.health.gov.il Principal Investigator: Nachum Vaisman, MD Collapse << |
| NCT02298192 | - | - | Completed | - | - |
| NCT00691314 | Stroke Transi... more >>ent Ischemic Attack Atherosclerosis Stenosis Stents Collapse << | Not Applicable | Completed | - | China, Beijing ... more >> Beijing Tiantan Hospital Beijing, Beijing, China, 100050 Xuanwu Hospital Capital Medical University Beijing, Beijing, China, 100053 Peking Union Medical College Hospital Beijing, Beijing, China, 100730 China, Chongqing Southwest Hospital Chongqing, Chongqing, China, 400038 Daping Hospital Chongqing, Chongqing, China, 400042 China, Jiangsu Nanjing General Hospital of Nanjing Military Command Nanjing, Jiangsu, China, 210002 China, Jilin Jilin University Changchun, Jilin, China, 130021 China, Shanghai Huashan Hospital Fudan University Shanghai, Shanghai, China, 200040 China, Sichuan West China Center of Medical Sciences Chengdu, Sichuan, China, 610041 China, Tianjin The General Hospital Under Tianjin Medical Sciences University Tianjin, Tianjin, China, 300052 Collapse << |
| NCT00133718 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Norway ... more >> Asker and Baerum Hospital, Medical Department RUD, Norway, 1309 Collapse << |
| NCT03659461 | Type 2 Diabetes Mellitus ... more >> PreDiabetes Collapse << | Not Applicable | Recruiting | January 2020 | Malaysia ... more >> Hospital Sultan Ismail Recruiting Johor Bahru, Johor, Malaysia, 81100 Contact: Chong Shiau Chin, MPHARM +6073565000 shiauchin83@hotmail.com Contact +60361455555 Collapse << |
| NCT03596450 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | November 19, 2021 | United States, California ... more >> Novo Nordisk Investigational Site Not yet recruiting Monterey, California, United States, 93940 Novo Nordisk Investigational Site Not yet recruiting San Jose, California, United States, 95148 Novo Nordisk Investigational Site Not yet recruiting Tulare, California, United States, 93274 United States, Connecticut Novo Nordisk Investigational Site Not yet recruiting Milford, Connecticut, United States, 06460 United States, Georgia Novo Nordisk Investigational Site Not yet recruiting Lilburn, Georgia, United States, 30047 Novo Nordisk Investigational Site Not yet recruiting Sandersville, Georgia, United States, 31082 Novo Nordisk Investigational Site Not yet recruiting Savannah, Georgia, United States, 31406 Novo Nordisk Investigational Site Recruiting Statesboro, Georgia, United States, 30461 Novo Nordisk Investigational Site Not yet recruiting Stockbridge, Georgia, United States, 30281 Novo Nordisk Investigational Site Not yet recruiting Swainsboro, Georgia, United States, 30401 United States, Kentucky Novo Nordisk Investigational Site Not yet recruiting Covington, Kentucky, United States, 41011 Novo Nordisk Investigational Site Not yet recruiting Owensboro, Kentucky, United States, 42303 United States, Virginia Novo Nordisk Investigational Site Not yet recruiting Chatham, Virginia, United States, 24531 Novo Nordisk Investigational Site Not yet recruiting Colonial Heights, Virginia, United States, 23834 Novo Nordisk Investigational Site Not yet recruiting Falls Church, Virginia, United States, 22044 Novo Nordisk Investigational Site Not yet recruiting Lynchburg, Virginia, United States, 24501 Novo Nordisk Investigational Site Not yet recruiting Warrenton, Virginia, United States, 20186 Collapse << |
| NCT02616666 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | December 31, 2021 | - |
| NCT00802971 | Hypoglycemia | Not Applicable | Completed | - | - |
| NCT02582242 | Diabetes Type... more >> 2 Diabetes Mellitus Collapse << | Phase 4 | Completed | - | - |
| NCT01065298 | - | - | Completed | - | India ... more >> Post Graduate Institute of Medical Education and Research Chandigarh, Chandigarh UT, India Collapse << |
| NCT01083212 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Sweden ... more >> Reserach Site Linkoping, Sweden Research Site Lulea, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT02582242 | - | - | Completed | - | - |
| NCT03640221 | Hypertension ... more >>Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Not yet recruiting | August 31, 2020 | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States, 90048 Collapse << |
| NCT01336738 | - | - | Completed | - | - |
| NCT00929539 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01475461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01103622 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT01298518 | Type 2 Diabetes Patients | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site DeLand, Florida, United States, 32720 Pfizer Investigational Site Miami Gardens, Florida, United States, 33169 Collapse << |
| NCT00241423 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, District of Col... more >>umbia Research Site Washington, District of Columbia, United States United States, Texas Research Site San Antonio, Texas, United States Collapse << |
| NCT01298518 | - | - | Completed | - | - |
| NCT02000700 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Arkansas ... more >> Little Rock, Arkansas, United States United States, California Los Angeles, California, United States United States, Colorado Aurora, Colorado, United States United States, Connecticut New Haven, Connecticut, United States United States, Florida Jacksonville, Florida, United States United States, Indiana Indianapolis, Indiana, United States United States, Kansas Lenexa, Kansas, United States United States, Maryland Baltimore, Maryland, United States United States, Missouri Kansas City, Missouri, United States United States, Ohio Toledo, Ohio, United States United States, Pennsylvania Pittsburgh, Pennsylvania, United States United States, Texas Houston, Texas, United States United States, Utah Salt Lake City, Utah, United States Brazil Campinas, Brazil Curitiba, Brazil Ribeirão Preto, Brazil Sao Paulo, Brazil Collapse << |
| NCT01475461 | - | - | Completed | - | - |
| NCT00942188 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Alabama ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mobile, Alabama, United States, 36689 United States, California For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Anaheim, California, United States, 92801 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chula Vista, California, United States, 91911 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. La Mesa, California, United States, 91942 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Walnut Creek, California, United States, 94598 United States, District of Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Washington, District of Columbia, United States, 20003 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miami Gardens, Florida, United States, 33169 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miami, Florida, United States, 33169 United States, Idaho For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Boise, Idaho, United States, 83702 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21287 United States, Michigan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dearborn, Michigan, United States, 48126 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Philadelphia, Pennsylvania, United States, 19107 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Arlington, Texas, United States, 76014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dallas, Texas, United States, 75230 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tomball, Texas, United States, 77375 United States, Utah For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Salt Lake City, Utah, United States, 84107 United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Richmond, Virginia, United States, 23294 Collapse << |
| NCT01105429 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute For Clinical Research, Inc. (Picr) Chula Vista, California, United States, 91911 United States, Florida Clinical Pharmacology Of Miami Inc. Miami, Florida, United States, 33014 Elite Research Institute Miami, Florida, United States, 33169 Collapse << |
| NCT01232946 | Type 2 Diabetes | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana Clinical Research Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01249677 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> Department of Medicine I; University Hospital St. Josef Hospital Bochum, Germany Collapse << |
| NCT00942188 | - | - | Completed | - | - |
| NCT02286128 | - | - | Completed | - | Thailand ... more >> Mattabhorn Phornputkul Chiang Mai, Thailand, 50200 Collapse << |
| NCT00924534 | Type 2 Diabetes | Phase 2 | Completed | - | Bulgaria ... more >> Site Reference ID/Investigator# 54183 Dimitrovgrad, Bulgaria, 6400 Site Reference ID/Investigator# 54182 Pleven, Bulgaria, 5800 Site Reference ID/Investigator# 44722 Plovdiv, Bulgaria, 4000 Site Reference ID/Investigator# 44723 Sofia, Bulgaria, 1407 Poland Site Reference ID/Investigator# 44725 Lubin, Poland, 59-301 Site Reference ID/Investigator# 44724 Pulawy, Poland, 24-100 Site Reference ID/Investigator# 54185 Radzymin, Poland, 05-250 Site Reference ID/Investigator# 44727 Ruda Slaska, Poland Site Reference ID/Investigator# 54184 Wroclaw, Poland, 50-349 South Africa Site Reference ID/Investigator# 44728 Cape Town, South Africa, 7130 Site Reference ID/Investigator# 44730 Cape Town, South Africa, 7937 Site Reference ID/Investigator# 44729 Johannesburg, South Africa, 2198 Collapse << |
| NCT02081118 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Florida ... more >> Hanmi pharmaceutical Miami, Florida, United States Collapse << |
| NCT02405260 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Arizona ... more >> Mesa, Arizona, United States, 85213 Tuscon, Arizona, United States, 85741 United States, California Canoga Park, California, United States, 91303 Huntington Park, California, United States, 90255 Los Angeles, California, United States, 90056 West Hills, California, United States, 91307 United States, Colorado Centennial, Colorado, United States, 80112 Littleton, Colorado, United States, 80127 United States, Connecticut Waterbury, Connecticut, United States, 06708 United States, Florida West Palm Beach, Florida, United States, 33409 United States, Illinois Gurnee, Illinois, United States, 60031 United States, Indiana Evansville, Indiana, United States, 47725 United States, Minnesota Rochester, Minnesota, United States, 55905 United States, North Carolina Greensboro, North Carolina, United States, 27408 United States, South Carolina Moncks Corner, South Carolina, United States, 29461 United States, Texas San Antonio, Texas, United States, 78209 San Antonio, Texas, United States, 78229 Schertz, Texas, United States, 78154 United States, Virginia Norfolk, Virginia, United States, 23507 United States, Washington Richland, Washington, United States, 99352 Collapse << |
| NCT01889667 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United States, California ... more >> Orange County Research Center Tustin, California, United States, 92780 Collapse << |
| NCT01290575 | Type 2 Diabetes | Phase 1 | Completed | - | United States, Arizona ... more >> Dedicated Phase I, Inc. Phoenix, Arizona, United States, 85013 United States, Arkansas Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, Florida Clinical Pharmacology Of Miami Inc. Miami, Florida, United States, 33014 Mra Clinical Research Miami, Florida, United States, 33143 United States, Texas Cetero Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT01665352 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Site 18 Chula Vista, California, United States, 91911 Site 1 Los Angeles, California, United States, 90057 United States, Colorado Site 13 Denver, Colorado, United States, 80220 United States, Florida Site 4 Coral Gables, Florida, United States, 33134 Site 5 Port Orange, Florida, United States, 32127 United States, Hawaii Site 3 Honolulu, Hawaii, United States, 96814 United States, Indiana Site 17 Indianapolis, Indiana, United States, 46254 United States, Kentucky Site 7 Louisville, Kentucky, United States, 40213 United States, Maine Site 8 Auburn, Maine, United States, 04210 United States, Nevada Site 9 Las Vegas, Nevada, United States, 89101 United States, New Jersey Site 26 Trenton, New Jersey, United States, 08611 United States, New York Site 12 Rochester, New York, United States, 14609 United States, Ohio Site 14 Cincinnati, Ohio, United States, 45219 United States, South Carolina Site 6 Mount Pleasant, South Carolina, United States, 29464 United States, Texas Site 16 Dallas, Texas, United States, 75230 Site 10 Houston, Texas, United States, 77072 Site 2 Houston, Texas, United States, 77074 Site 11 Katy, Texas, United States, 77450 United States, Virginia Site 15 Richmond, Virginia, United States, 23294 Collapse << |
| NCT00359762 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02206607 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, North Carolina ... more >> High Point Clinical Trials Center High Point, North Carolina, United States, 27265 United States, Texas Clinical Trials of Texas, Inc. San Antonio, Texas, United States, 78229 Collapse << |
| NCT01024244 | - | - | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | - |
| NCT01095991 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Germany ... more >> Research Site Neuss, Germany Collapse << |
| NCT02206607 | - | - | Completed | - | - |
| NCT00359762 | - | - | Completed | - | - |
| NCT01082120 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT01024244 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | United States, Kansas ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Topeka, Kansas, United States, 66606 United States, New Jersey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Toms River, New Jersey, United States, 08753 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Syracuse, New York, United States, 13210 Australia, New South Wales For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. St. Leonards, New South Wales, Australia, 2065 Australia, South Australia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daw Park, South Australia, Australia, 5041 Australia, Victoria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Box Hill, Victoria, Australia, 3128 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Manati, Puerto Rico, 00674 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico, 00907 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Arkhangelsk, Russian Federation, 163045 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 119435 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rostov-On-Don, Russian Federation, 344022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 194354 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alcira, Spain, 46600 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alicante, Spain, 03114 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dos Hermanas, Spain, 41014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Santa Cruz De Tenerife, Spain, 38320 Collapse << |
| NCT01889667 | - | - | Completed | - | - |
| NCT01029795 | - | - | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | - |
| NCT01029795 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | Australia, South Australia ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Elizabeth Vale, South Australia, Australia, 5112 Australia, Victoria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. East Ringwood, Victoria, Australia, 3135 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Heidelberg, Victoria, Australia, 3081 Austria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vienna, Austria, 1090 Czech Republic For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Holesov, Czech Republic, 769 01 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Prague, Czech Republic, 140 59 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55116 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Neuss, Germany, D-41460 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jerusalem, Israel, 91120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nahariya, Israel, 22100 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rostov-On-Don, Russian Federation, 344022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 191126 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cordoba, Spain, 14004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dos Hermanas, Spain, 41014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Teruel, Spain, 44002 Collapse << |
| NCT01003184 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Ireland ... more >> Research Site Dublin, Ireland United Kingdom Research Site Addlestone, England, United Kingdom Research Site Bath, England, United Kingdom Research Site Birmingham, England, United Kingdom Research Site Blackburn, England, United Kingdom Research Site Bournemouth, England, United Kingdom Research Site Chippenham, England, United Kingdom Research Site Derby, England, United Kingdom Research Site Exeter, England, United Kingdom Research Site High Wycombe, England, United Kingdom Research Site Hull, England, United Kingdom Research Site Leicester, England, United Kingdom Research Site Liverpool, England, United Kingdom Research Site London, England, United Kingdom Research Site Manchester, England, United Kingdom Research Site Merseyside, England, United Kingdom Research Site Middlesborough, England, United Kingdom Research Site Newcastle, England, United Kingdom Research Site Northampton, England, United Kingdom Research Site Oldham, England, United Kingdom Research Site Plymouth, England, United Kingdom Research Site Portsmouth, England, United Kingdom Research Site Sheffield, England, United Kingdom Research Site Stevenage, England, United Kingdom Research Site Suffolk, England, United Kingdom Research Site Wakefield, England, United Kingdom Research Site Wiltshire, England, United Kingdom Research Site Aberdeen, Scotland, United Kingdom Research Site Dundee, Scotland, United Kingdom Research Site Carmathen, Wales, United Kingdom Research Site Swansea, Wales, United Kingdom Research Site Wrexham, Wales, United Kingdom Research Site Leytonstone, United Kingdom Research Site Livingston, United Kingdom Collapse << |
| NCT01003184 | - | - | Completed | - | - |
| NCT02653209 | Type 2 Diabetes | Phase 4 | Recruiting | December 2019 | United Kingdom ... more >> Exeter Clinical Research Facility Recruiting Exeter, United Kingdom, EX2 5DW Contact: Catherine Angwin 01392408185 C.Angwin@exeter.ac.uk Contact: Andrew Hattersley +441392 408231 A.T.Hattersley@exeter.ac.uk Principal Investigator: Andrew Hattersley Collapse << |
| NCT00203632 | Diabetes Mellitus | Phase 3 | Terminated(Terminated at 50% e... more >>nrollment due to recent concerns about rosiglitazone) Collapse << | - | Canada, Alberta ... more >> Foothills Medical Centre Calgary, Alberta, Canada, T2N 2T9 Collapse << |
| NCT01408095 | Diabetes Mellitus, Type 2 | Phase 2 | Withdrawn(Trial terminated no ... more >>patients were screened or enrolled) Collapse << | September 2012 | - |
| NCT03167918 | Type 2 Diabetes Mellitus ... more >> Pulmonary Function Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT02097342 | Type 2 Diabetes Mellitus ... more >> Insulin Sensitivity/Resistance Collapse << | Phase 4 | Unknown | January 2015 | India ... more >> Postgraduate Institute of Medical Education & Research Recruiting Chandigarh, India, 160012 Contact: Anil Bhansali, MD DM anilbhansaliendocrine@gmail.com Collapse << |
| NCT03737240 | Diabetes Mellitus | Phase 3 | Not yet recruiting | December 2021 | United States, Georgia ... more >> Grady Health System Not yet recruiting Atlanta, Georgia, United States, 30303 Contact: Rodolfo Galindo, MD 404-251-8961 rodolfo.galindo@emory.edu Emory Clinic Not yet recruiting Atlanta, Georgia, United States, 30322 Contact: Rodolfo Galindo, MD 404-251-8961 rodolfo.galindo@emory.edu Collapse << |
| NCT01811485 | - | - | Completed | - | - |
| NCT02145611 | Type 2 Diabetes Mellitus ... more >> Hypertension Endothelial Dysfunction Collapse << | Phase 4 | Completed | - | Brazil ... more >> Fundação Faculdade Regional de Medicina de São José do Rio Preto São José do Rio Preto, São Paulo, Brazil, 15090-000 Collapse << |
| NCT02470039 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Germany ... more >> Novo Nordisk Investigational Site Mainz, Germany, 55116 Novo Nordisk Investigational Site Neuss, Germany, 41460 Collapse << |
| NCT01784380 | - | - | Completed | - | China, Guangdong ... more >> Elecsys2010,French KONTRON EUB-5500 color Doppler ultrasonic diagnostic apparatus ZHONGSHAN City, Guangdong, China, 528400 Reproductive Center of Bo Ai Hospital, Zhongshan Zhongshan, Guangdong, China, 528403 Collapse << |
| NCT01087567 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Ohio ... more >> Ohio University Athens, Ohio, United States, 45701 Collapse << |
| NCT01811485 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Chikushino-city, Fukuoka, Japan, 818-0083 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 810-0014 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 819-0006 Novartis Investigative Site Iizuka-city, Fukuoka, Japan, 820-8505 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 800-0296 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 807-0857 Novartis Investigative Site Koga-city, Ibaraki, Japan, 306-0232 Novartis Investigative Site Kawasaki-city, Kanagawa, Japan, 210-0852 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0802 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 231-0023 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8533 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8660 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 607-8062 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Sakai-city, Osaka, Japan, 590-0064 Novartis Investigative Site Takatsuki-city, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Tokorozawa-city, Saitama, Japan, 359-1161 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-0031 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji-city, Tokyo, Japan, 192-0918 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Katsushika-ku, Tokyo, Japan, 124-0024 Novartis Investigative Site Kiyose-city, Tokyo, Japan, 204-0021 Novartis Investigative Site Minato-ku, Tokyo, Japan, 105-7390 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Shibuya-ku, Tokyo, Japan, 150-0002 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Collapse << |
| NCT00883766 | Polycystic Ovary Syndrome ... more >> In Vitro Fertilization Collapse << | Not Applicable | Terminated | - | Spain ... more >> Institut Universitari Dexeus Barcelona, Spain, 08028 Collapse << |
| NCT00162305 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> Local Institution Fort Lauderdale, Florida, United States, 33301 Local Institution Miami, Florida, United States, 33169 Local Institution Orlando, Florida, United States, 32809 United States, Louisiana Local Institution New Orleans, Louisiana, United States, 70119 United States, Texas Local Institution San Antonio, Texas, United States, 78229-3894 Collapse << |
| NCT02420262 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03038789 | - | - | Recruiting | January 31, 2020 | Korea, Republic of ... more >> Research Site Recruiting Busan, Korea, Republic of Research Site Recruiting ChungCheong, Korea, Republic of Research Site Recruiting DaeGu, Korea, Republic of Research Site Recruiting DaeJeon, Korea, Republic of Research Site Recruiting GyeongGi, Korea, Republic of Research Site Active, not recruiting GyeongGi, Korea, Republic of Research Site Recruiting Seoul, Korea, Republic of Research Site Withdrawn Seoul, Korea, Republic of Research Site Recruiting WonJu, Korea, Republic of Collapse << |
| NCT00464594 | Diabetes Mellitus | Not Applicable | Unknown | - | Japan ... more >> Tokyo Medical and Dental University Recruiting Tokyo, Japan, 113-8519 Contact 03-3813-6111 Collapse << |
| NCT02420262 | - | - | Completed | - | - |
| NCT01465113 | Short Segment Barrett's Esopha... more >>gus Long Segment Barrett's Esophagus Collapse << | Early Phase 1 | Completed | - | United States, Ohio ... more >> University Hospitals Ahuja Medical Center Beachwood, Ohio, United States, 44122 Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02061969 | Diabetes | Phase 4 | Completed | - | United States, Georgia ... more >> Crestvew Nursing Home Atlanta, Georgia, United States, 30315 Budd Terrace Nursing Home Atlanta, Georgia, United States, 30322 Wesley Woods Nursing Home Atlanta, Georgia, United States, 30322 VA Nursing Home Decatur, Georgia, United States, 30033 Collapse << |
| NCT02061969 | - | - | Completed | - | - |
| NCT01754142 | - | - | Completed | - | Korea, Republic of ... more >> Research Site Seoul, Korea, Republic of Collapse << |
| NCT00063635 | Fatty Liver | Phase 3 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94143 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 United States, Missouri St. Louis University St. Louis, Missouri, United States, 63110 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44109 United States, Texas Texas Children's Hospital Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT00135096 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT01117350 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03674385 | Clomiphene Citrate Resistant P... more >>olycystic Ovary Syndrome Collapse << | Phase 2 | Recruiting | January 20, 2019 | Egypt ... more >> Medical Center of Infertility Recruiting Beni-suef, Egypt, 62521 Contact: eman megahed, M.D-PHD 01095115944 dr_emanmojahed@hotmail.com Collapse << |
| NCT01117350 | - | - | Completed | - | - |
| NCT00680745 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00481663 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02597049 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00063635 | - | - | Completed | - | - |
| NCT00269061 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 1 | Completed | - | United States, Massachusetts ... more >> GSK Investigational Site Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT02053116 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(On 25April2014, stu... more >>dy was terminated before any dosing due to an AE of safety concern that occurred in protocol B1731003 with the same drug.) Collapse << | - | United States, Florida ... more >> Pfizer Investigational Site DeLand, Florida, United States, 32720 Collapse << |
| NCT00680745 | - | - | Completed | - | - |
| NCT01426737 | Type 2 Diabetes | Phase 4 | Unknown | January 2013 | Switzerland ... more >> Universitiy Hospital Recruiting Zurich, Switzerland Collapse << |
| NCT02607410 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Brazil ... more >> University of Sao Paulo General Hospital Sao Paulo, Brazil, 05403-000 Collapse << |
| NCT03772262 | Interaction Drug Food | Early Phase 1 | Recruiting | March 31, 2019 | United States, Washington ... more >> Washington State University Recruiting Spokane, Washington, United States, 99202 Contact: Malathi Jandhyala 509-335-3668 irb@wsu.edu Collapse << |
| NCT02597049 | - | - | Completed | - | - |
| NCT01095653 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02290184 | Insulin Resistance, ... more >> Metabolic Syndrome Type 2 Diabetes Collapse << | Not Applicable | Unknown | October 2016 | United States, California ... more >> San Francisco / Daly City Communities San Francisco, California, United States, 94115 Collapse << |
| NCT02846233 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | December 2018 | United States, California ... more >> UCSF Fresno Recruiting Fresno, California, United States, 93701 Contact: SOE NAING snaing@fresno.ucsf.edu Collapse << |
| NCT01952145 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02743260 | Healthy | Phase 4 | Unknown | December 2017 | Germany ... more >> Department of Pharmacology I, University Hospital Cologne Cologne, NRW, Germany, 50931 Collapse << |
| NCT02526810 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | September 2016 | China, Guangdong ... more >> Endocrinology department of the Third Affiliated Hospital of Sun Yet-san University Recruiting Guangzhou City, Guangdong, China, 510630 Contact: Zeng Longyi, Professor 0086-020-85252160 zssynfmk@163.com Contact: Lin Shuo, Doctor 0086-020-85253408 littltpig@yeah.net Principal Investigator: Zeng Longyi, Professor Sub-Investigator: lin Shuo, Doctor Collapse << |
| NCT01095653 | - | - | Completed | - | - |
| NCT01217073 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01217073 | - | - | Completed | - | - |
| NCT01181674 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Canada, Ontario ... more >> McMaster University Medical Centre, Diabetes Care and Research Program Hamilton, Ontario, Canada, L8N 3Z5 Collapse << |
| NCT02091362 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01952145 | - | - | Completed | - | - |
| NCT00290940 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01901757 | Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT02897349 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | January 30, 2019 | China ... more >> China-Japan Friendship Hospital Beijing, China, 100029 Beijing Tongren Hospital Beijing, China, 100730 The General Hospital of Chinese People's Armed Police Forces Beijing, China, 100854 First Hospital of Jilin University Changchun, China, 130021 Fuzhou General Hospital of Nanjing Militray Command Fuzhou, China, 350025 Third Affiliated Hospital of Guangzhou Medical University Guangzhou, China, 510150 The Affiliated Hospital of Guizhou Medical University Guiyang, China, 550004 Hangzhou Second People's Hospital Hangzhou, China, 310015 General Hospital of Jinan Military Area Jinan, China, 250000 Nanjing First Hospital Nanjing, China, 210006 The affiliated hospital of medicalcollege qingdao university Qingdao, China, 266005 Centre Hospital of Putuo District, Shanghai Shanghai, China, 200062 Shanghai Tenth People's Hospital Shanghai, China, 200072 Yangpu Hospital, Tongji University Shanghai, China, 200090 Second Hospital Affiliated to Shantou Medical University Shantou, China, 515041 Shengjing Hospital of China Medical University Shengyang, China, 110072 Siping Central People's Hospital Siping, China, 136000 The First Affiliated Hospital of Soochow University Suzhou, China, 215006 Nankai University Affiliated Hospital Tianjin, China, 300000 The 2nd Hospital of Tianjin Medical University Tianjin, China, 300000 Tianjin Medical University General Hospital Tianjin, China, 30052 Renmin Hospital of Wuhan University Wuhan, China, 430060 First Affiliated Hospital of Xiamen University Xiamen, China, 361003 the first people hospital of Yue Yang Yueyang, China, 414000 Affiliated Hospital of Jiangsu University Zhenjiang, China, 212013 Collapse << |
| NCT02091362 | - | - | Completed | - | - |
| NCT02229240 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(Study was cancelled ... more >>prior to enrolling any patients) Collapse << | December 2015 | - |
| NCT01089569 | Type 2 Diabetes | Not Applicable | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT02229227 | - | - | Completed | - | - |
| NCT03226210 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Jiangsu ... more >> Nanjing First Hostital Nanjing, Jiangsu, China, 210000 Collapse << |
| NCT01089569 | - | - | Completed | - | - |
| NCT02229227 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02836704 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China ... more >> China, China Collapse << |
| NCT02635646 | - | - | Completed | - | - |
| NCT02304536 | Infertility | Phase 3 | Recruiting | - | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Cairo University Hospitals Recruiting Cairo, Egypt Contact: AbdelGany MA Hassan, MRCOG, MD 00217801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT01398592 | Diabetes Mellitus Type 2 | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Oeynhausen, Germany, 32549 Novartis Investigative Site Berlin, Germany, 12347 Novartis Investigative Site Berlin, Germany, 13597 Novartis Investigative Site Berlin, Germany, 14089 Novartis Investigative Site Celle, Germany, 29221 Novartis Investigative Site Hannover, Germany, 30165 Novartis Investigative Site Hannover, Germany, 30167 Novartis Investigative Site Kassel, Germany, 34117 Novartis Investigative Site Kirchhain, Germany, 35274 Novartis Investigative Site Lehrte, Germany, 31275 Novartis Investigative Site Mülheim, Germany, 45468 Novartis Investigative Site Oberhausen, Germany, 46049 Novartis Investigative Site Osnabrück, Germany, 49080 Novartis Investigative Site Potsdam, Germany, 14469 Novartis Investigative Site Stuttgart, Germany, 70378 Collapse << |
| NCT02635646 | Prediabetes | Not Applicable | Completed | - | Mexico ... more >> Universidad de Guanajuato León, Guanajuato, Mexico, 37670 Collapse << |
| NCT01191268 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00789191 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03323294 | - | - | Recruiting | October 2019 | United States, Arkansas ... more >> Arkansas Children's Hospital Recruiting Little Rock, Arkansas, United States, 72202 Contact: Leanna Delhey, BS, MPH 501-364-4519 delheylm@archildrens.org Principal Investigator: Eugenia Carvalho, PhD Sub-Investigator: Shannon Rose, PhD Sub-Investigator: Elisabet Borsheim, PhD Sub-Investigator: Jon Oden, MD Sub-Investigator: Shipra Bansal, MD Sub-Investigator: Emir Tas, MD Sub-Investigator: Stepan Melnyk, MD, PhD Sub-Investigator: Judith Weber, PhD, RD Collapse << |
| NCT00804986 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01191268 | - | - | Completed | - | - |
| NCT01960205 | Prediabetes | Phase 4 | Unknown | - | China, Shandong ... more >> Shandong Provincial Hospital Recruiting Jinan, Shandong, China, 250021 Contact: Xinli Zhou, MD, PhD 15168889976 zhouxinli0301@163.com Principal Investigator: Xinli Zhou, MD,PhD Collapse << |
| NCT00789191 | - | - | Completed | - | - |
| NCT00776607 | Latent Autoimmune Diabetes in ... more >>Adults LADA Collapse << | Not Applicable | Unknown | April 2011 | United Kingdom ... more >> Diabetes Unit Not yet recruiting Cardiff, Wales, United Kingdom, CF64 2XX Contact: Julia Platts 02920 711 711 ext 5149 Julia.Platts@CardiffandVale.wales.nhs.uk Principal Investigator: Julia Platts Clinical Research Unit Recruiting Swansea, Wales, United Kingdom, SA6 6NL Contact: Lucy Barlow +44 (0) 1792 703722 ext 3722 Lucy.Barlow@swansea-tr.wales.nhs.uk Principal Investigator: Jeffrey Stephens Collapse << |
| NCT02463357 | Mountain Sickness | Phase 4 | Active, not recruiting | November 30, 2018 | United States, Michigan ... more >> Alma College Alma, Michigan, United States, 48801 Collapse << |
| NCT00763815 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00763815 | - | - | Completed | - | - |
| NCT01342042 | Type 2 Diabetes | Phase 2 | Unknown | December 2011 | China, Beijing ... more >> Peking University First Hospital Recruiting Peking, Beijing, China, 010 Contact: Cui Yi Min, MD 86-10-88325988 Principal Investigator: Cui Yi Min, MD Collapse << |
| NCT03748810 | - | - | Recruiting | December 31, 2018 | Korea, Republic of ... more >> Chungbuk National University Hospital Recruiting Cheonju, Chungcheongbuk-do, Korea, Republic of, 28644 Contact: Eu Jeong Ku, MD 82-10-3462-8863 eujeong.ku@gmail.com Collapse << |
| NCT00804986 | - | - | Completed | - | - |
| NCT00760344 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02411825 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Germany ... more >> Investigational Site Number 276001 Berlin, Germany, 10117 Collapse << |
| NCT03006172 | Breast Cancer ... more >> Solid Tumor Collapse << | Phase 1 | Recruiting | February 29, 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital. Recruiting Boston, Massachusetts, United States, 02114 Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 United States, New York Columbia University Medical Center Recruiting New York, New York, United States, 10032 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 United States, Tennessee Sarah Cannon Research Institute Recruiting Nashville, Tennessee, United States, 37203 Canada, Ontario Philippe Bedard Clinic Toronto Recruiting Toronto, Ontario, Canada, M5G 1L7 France Institut Bergonie Recruiting Bordeaux, France, 33076 Institut Gustave Roussy Recruiting Villejuif, France, 94805 Spain Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Hospital Clinico Universitario de Valencia Recruiting Valencia, Spain, 46010 United Kingdom Barts and the London NHS Trust. Recruiting London, United Kingdom, EC1A 7BE Royal Marsden Hospital - London Recruiting London, United Kingdom, SW3 6JJ Royal Marsden Hospital - Surrey Recruiting Surrey, United Kingdom, SM2 5PT Collapse << |
| NCT03383068 | PCOS Impaired... more >> Glucose Tolerance Collapse << | Phase 4 | Not yet recruiting | June 30, 2020 | China, Shanghai ... more >> Renji Hospital Department of Endocrinology and Metabolism Not yet recruiting Shanghai, Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Collapse << |
| NCT02694263 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | September 2018 | United Kingdom ... more >> University Hospitals of Leicester NHS Trust Recruiting Leicester, Leicestershire, United Kingdom, LE5 4PW Contact: Emer Brady, PhD 0116248 ext 8959 emer.brady@uhl-tr.nhs.uk Contact: Natasha Wileman, MSc 0116258 ext 8929 natasha.wileman@uhl-tr.nhs.uk University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, West Midlands, United Kingdom, B15 2TH Contact: Wasim Hanif 0121371 ext 4404 wasim.hanif@uhb.nhs.uk Contact: Deborah Smith 0121371 ext 4707 deborah.smith@uhb.nhs.uk Collapse << |
| NCT01392573 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03359590 | Pharmacological Action | Phase 2 | Enrolling by invitation | July 30, 2019 | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany Collapse << |
| NCT00667732 | Type 2 Diabetes | Phase 4 | Completed | - | United States, District of Col... more >>umbia Medstar Research Institute Washington DC, District of Columbia, United States, 20003 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT01392573 | - | - | Completed | - | - |
| NCT03717194 | Type2 Diabetes ... more >> Heart Failure Collapse << | Phase 3 | Not yet recruiting | October 30, 2020 | Korea, Republic of ... more >> Seoul National University Bundang Hospita; Not yet recruiting Seongnam-si, Gyeonggi-do, Korea, Republic of, 13620 Collapse << |
| NCT01963130 | - | - | Completed | - | Turkey ... more >> Bezmialem VU Istanbul, Turkey, 34093 Collapse << |
| NCT02292290 | Type 2 Diabetes | Phase 4 | Completed | - | United Kingdom ... more >> University Hospitals of Leicester Leicester, Leicestershire, United Kingdom University Hospital Birmingham Birmingham, West Midlands, United Kingdom Collapse << |
| NCT00622323 | - | - | Completed | - | Philippines ... more >> Research Site Paranaque City, Philippines Research Site Pasig City, Philippines Research Site Taytay, Philippines Collapse << |
| NCT03278158 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | December 2018 | Denmark ... more >> Odense University Hospital Recruiting Odense, Denmark, 5000 Contact: Henning Beck-Nielsen, Dr henning.beck-nielsen@rsyd.dk Collapse << |
| NCT00667732 | - | - | Completed | - | - |
| NCT01503008 | Type 2 Diabetes Mellitus | Not Applicable | Withdrawn | August 2012 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 United States, Massachusetts Center for Connected Health, Partners Healthcare Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00121966 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Center Odense, Denmark, 5000 Collapse << |
| NCT02597101 | Type 2 Diabetes Mellitus ... more >> Insulin Resistance Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, Pennsylvania ... more >> Allegheny Health Network Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT00268476 | Prostate Cancer | Phase 2 Phase 3 | Recruiting | September 2024 | - |
| NCT01658501 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02586129 | Dyslipidemia ... more >>Type II Diabetes Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul National Universitiy Bundang Hospital Seongnam-Si, Gyeonggi-Do, Korea, Republic of, 463-707 Collapse << |
| NCT00004992 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Glucose Intolerance Collapse << | Phase 3 | Completed | - | United States, Maryland ... more >> George Washington University Biostatistics Center, Suite 750, 6110 Executive Blvd Rockville, Maryland, United States, 20852 Collapse << |
| NCT00868790 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(Upon interim analys... more >>is, sufficient data was accrued to assess study hypotheses.) Collapse << | - | - |
| NCT01009580 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00868790 | - | - | Terminated(Upon interim analys... more >>is, sufficient data was accrued to assess study hypotheses.) Collapse << | - | - |
| NCT02749032 | Type 2 Diabetes | Phase 1 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Niedersachsen, Germany, 37431 Collapse << |
| NCT00672386 | Diabetes Mellitus, Type 2 ... more >> Diabetes Mellitus Endocrine System Diseases Nutritional and Metabolic Diseases Collapse << | Phase 2 | Completed | - | - |
| NCT01009580 | - | - | Completed | - | - |
| NCT01488552 | Stage IV Pancreatic Cancer | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> TGen Clinical Research Services (TCRS) Scottsdale, Arizona, United States, 85258 United States, California Disney Family Cancer Center Burbank, California, United States, 91505 United States, Minnesota Virginia Piper Cancer Institute (VPCI) Minneapolis, Minnesota, United States, 55407 United States, Washington Virginia Mason Medical Center Seattle, Washington, United States, 98101 Evergreen Hematology and Oncology Spokane, Washington, United States, 99218 Collapse << |
| NCT01513590 | - | - | Completed | - | - |
| NCT03199053 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 20, 2021 | - |
| NCT00005654 | Hyperinsulinism ... more >> Polycystic Ovary Syndrome Collapse << | Not Applicable | Completed | - | - |
| NCT00005104 | Polycystic Ovary Syndrome ... more >> Hyperinsulinism Collapse << | Not Applicable | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294-3300 United States, Louisiana Louisiana State University School of Medicine Shreveport, Louisiana, United States, 71130-3932 United States, Missouri Washington University - St. Louis St. Louis, Missouri, United States, 63110 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908 Medical College of Virginia School of Medicine Richmond, Virginia, United States, 23298-0230 Italy University Alma Mater Bologna, Italy, 40100 Collapse << |
| NCT01513590 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01125150 | - | - | Completed | - | United States, Michigan ... more >> Department of Dermatology, NEW CENTER ONE Detroit, Michigan, United States, 48202 Collapse << |
| NCT00159211 | Type 2 Diabetes | Not Applicable | Terminated(inclusion was finis... more >>hed) Collapse << | - | France ... more >> Sce de Diabétologie, hôpital de la Pitié-salpêtrière, 83bld de l'hôpital Paris, France, 75013 Collapse << |
| NCT01351753 | Obesity Metab... more >>olic Syndrome X Collapse << | Phase 2 | Suspended(Change of staff) | October 2019 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52241 Collapse << |
| NCT01241448 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01318564 | - | - | Completed | - | United States, Texas ... more >> Kelsey Seybold Clinics Houston, Texas, United States, 77025 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00449553 | - | - | Completed | - | Denmark ... more >> Multiple, Denmark Iceland Multiple, Iceland Norway Multiple, Norway Sweden Multiple, Sweden Collapse << |
| NCT00035984 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT01884051 | - | - | Recruiting | July 2022 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00182494 | Diabetes Mellitus ... more >> Schizophrenia Collapse << | Phase 4 | Unknown | - | Canada, Ontario ... more >> McMaster University Recruiting Hamilton, Ontario, Canada Contact: Lakshmi P Voruganti, MD 905-522-1155 ext 6355 vorugl@mcmaster.ca Contact: Susan Strong, MSc 905 522 1155 strongs@mcmaster.ca Principal Investigator: Lakshmi P Voruganti, MD Collapse << |
| NCT00641407 | Type 2 Diabetes Mellitus ... more >> Hypoglycemia Collapse << | Phase 4 | Completed | - | Italy ... more >> Department of Geriatrics and Metabolic Disease Naples, Italy, 80138 Collapse << |
| NCT01241448 | - | - | Completed | - | - |
| NCT01368081 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT03375580 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Not Applicable | Recruiting | December 31, 2019 | China, Guangdong ... more >> First Affiliated Hospital of Guangdong Pharmaceutical University Recruiting Guangzhou, Guangdong, China, 510080 Contact: Guobiao Huang 86-20-39352064 153706227@qq.com Collapse << |
| NCT01827280 | 1- Microvascular Function ... more >> 2-oxidative Stress 3-inflammation Collapse << | Phase 4 | Completed | - | Brazil ... more >> Laboratory for Clinical and Experimental Research on Vascular Biology Rio de Janeiro, Brazil, 20550-900 Collapse << |
| NCT00713830 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Bulgaria Sanofi-Aventis Administrative Office Sofia, Bulgaria Czech Republic Sanofi-Aventis Administrative Office Praha, Czech Republic Egypt Sanofi-Aventis Administrative Office Cairo, Egypt Germany Sanofi-Aventis Administrative Office Berlin, Germany India Sanofi-Aventis Administrative Office Mumbai, India Israel Sanofi-Aventis Administrative Office Natanya, Israel Japan Sanofi-Aventis Administrative Office Tokyo, Japan Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Taiwan Sanofi-Aventis Administrative Office Taipei, Taiwan Thailand Sanofi-Aventis Administrative Office Bangkok, Thailand Tunisia Sanofi-Aventis Administrative Office Megrine, Tunisia Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey Collapse << |
| NCT00909597 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01368081 | - | - | Completed | - | - |
| NCT00518882 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01656057 | To Assess the Impact of Bile A... more >>cids on Human Glukagon-like-peptide-1 Secretion Collapse << | Not Applicable | Completed | - | Denmark ... more >> University Hospital of Copenhagen, Gentofte Hospital, Diabetic Research Division Copenhagen, Hellerup, Denmark, 2900 Collapse << |
| NCT00357370 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | - |
| NCT00097877 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01420692 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Turkey ... more >> Ankara University Medical Faculty Dept. of Endocrinology and Metabolism Ankara, Turkey, 06100 Collapse << |
| NCT00357370 | - | - | Completed | - | - |
| NCT00713830 | - | - | Completed | - | - |
| NCT00541567 | Obesity and Type 2 Diabetes | Phase 2 Phase 3 | Withdrawn | June 2010 | - |
| NCT03529123 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | August 30, 2019 | India ... more >> Investigational Site Number 01 Recruiting Bangalore, India, 560092 Investigational Site Number 012 Recruiting Belgaum, India, 590010 Investigational Site Number 017 Recruiting Coimbatore, India, 641009 Investigational Site Number 06 Recruiting Hyderabad, India, 500063 Investigational Site Number 07 Recruiting Hyderabad, India, 500072 Investigational Site Number 08 Recruiting Jaipur, India Investigational Site Number 04 Recruiting Kolkata, India, 700107 Investigational Site Number 018 Recruiting Lucknow, India Investigational Site Number 014 Recruiting Madurai, India, 625020 Investigational Site Number 05 Recruiting Nasik, India, 422002 Investigational Site Number 010 Recruiting New Delhi, India, 110029 Investigational Site Number 016 Recruiting Pune, India, 411040 Collapse << |
| NCT00518882 | - | - | Completed | - | - |
| NCT01069965 | Diabetes Mellitus | Phase 2 | Terminated(Funding support wit... more >>hdrawn.) Collapse << | - | - |
| NCT01680978 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United Kingdom ... more >> Oxford, United Kingdom, OX3 9DU Collapse << |
| NCT03204799 | - | - | Recruiting | December 16, 2018 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT01306214 | Diabetes Mellitus, Type 2 ... more >> Obesity Collapse << | Phase 3 | Completed | - | - |
| NCT01306214 | - | - | Completed | - | - |
| NCT01956305 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Worldwide Clinical Trials San Antonio, Texas, United States, 78217 Collapse << |
| NCT01903356 | - | - | Completed | - | Korea, Republic of ... more >> One or multiple Investigational sites, Korea, Republic of Collapse << |
| NCT00417729 | Diabetes Mellitus | Phase 4 | Completed | - | Taiwan ... more >> Taichung Veterans General Hospital Taichung, Taiwan Collapse << |
| NCT00842140 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Brazil ... more >> University of Sao Paulo Ribeirao Preto, Sao Paulo, Brazil, 14049-900 Collapse << |
| NCT00116922 | Impaired Glucose Tolerance ... more >> Diabetes Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> UWO Research Park London, Ontario, Canada, N6G 4X8 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 Collapse << |
| NCT03655535 | Type2 Diabetes Mellitus | Phase 2 | Recruiting | August 30, 2019 | United States, California ... more >> Coastal Metabolic Research Center, Inc. Recruiting Ventura, California, United States, 93003 Contact: Ronald Chochinov, MD 805-658-8460 rchochinov@yahoo.com Contact: Sagrario Olmos-Garcia, MD sagy68@gmail.com United States, New Mexico Albuquerque Clinical Trials Recruiting Albuquerque, New Mexico, United States, 87102 Contact: Ellen Kim, MD 505-224-7407 ekim@abqct.com Contact: Jose Atencio JAtencio@abqct.com United States, South Carolina Coastal Carolina Research Center Recruiting Mount Pleasant, South Carolina, United States, 29464 Contact: Nathan Morton 843-352-6977 nmorton@coastalcarolinaresearch.com Contact: Yvonne Davis, MD ydavis@coastalcarolinaresearch.com United States, Utah Advanced Research Institute Recruiting Ogden, Utah, United States, 84405 Contact: Jack Wahlen, MD 801-409-2040 jwahlen@advresearch.org Contact: Jonathan Winkfield, PA-C jon@advresearch.org Collapse << |
| NCT01859793 | Diabetes Athe... more >>rosclerosis Collapse << | Phase 4 | Completed | - | United States, Wisconsin ... more >> Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT01859793 | - | - | Completed | - | - |
| NCT00441363 | Type 2 Diabetes | Phase 3 | Terminated(Failure to Recruit ... more >>in a Timely manner) Collapse << | - | - |
| NCT00103857 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01206712 | - | - | Completed | - | Germany ... more >> IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Collapse << |
| NCT01516476 | Diabetes Mellitus Type 2 | Phase 2 | Terminated | - | United States, Ohio ... more >> Cincinnati, Ohio, United States, 45227 Collapse << |
| NCT00789035 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00103857 | - | - | Completed | - | - |
| NCT01798875 | PCOS | Phase 4 | Unknown | December 2015 | Poland ... more >> Department of Clinical and Experimental Endocrinology, Medical University of Gdansk Recruiting Gdansk, Poland Contact: Dominik Rachon, MD, PhD +48583491947 drachon@gumed.edu.pl Principal Investigator: Monika Kuligowska-Jakubowska, MD Principal Investigator: Jolanta Dardzinska, MD, PhD Sub-Investigator: Agnieszka Kowalewska-Wlas, MD, PhD Principal Investigator: Aleksandra Rutkowska, MSc, PhD Principal Investigator: Justyna Breska-Kruszewska, MSc Collapse << |
| NCT01215097 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | China ... more >> 1218.65.86007 Boehringer Ingelheim Investigational Site Beijing, China 1218.65.86011 Boehringer Ingelheim Investigational Site Chongqing, China 1218.65.86008 Boehringer Ingelheim Investigational Site Dalian, China 1218.65.86010 Boehringer Ingelheim Investigational Site Fuzhou, China 1218.65.86014 Boehringer Ingelheim Investigational Site Hangzhou, China 1218.65.86005 Boehringer Ingelheim Investigational Site Hefei, China 1218.65.86006 Boehringer Ingelheim Investigational Site Hefei, China 1218.65.86012 Boehringer Ingelheim Investigational Site Nanjing, China 1218.65.86001 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86002 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86003 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86004 Boehringer Ingelheim Investigational Site Suzhou, China 1218.65.86015 Boehringer Ingelheim Investigational Site Wenzhou, China 1218.65.86009 Boehringer Ingelheim Investigational Site Wuhan, China 1218.65.86013 Boehringer Ingelheim Investigational Site Yangzhou, China Malaysia 1218.65.60002 Boehringer Ingelheim Investigational Site Johor Bahru,, Malaysia 1218.65.60001 Boehringer Ingelheim Investigational Site Kelantan, Malaysia Philippines 1218.65.63001 Boehringer Ingelheim Investigational Site Marikina, Philippines 1218.65.63002 Boehringer Ingelheim Investigational Site San Juan, Philippines Collapse << |
| NCT00789035 | - | - | Completed | - | - |
| NCT01169090 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00441363 | - | - | Terminated(Failure to Recruit ... more >>in a Timely manner) Collapse << | - | - |
| NCT01863667 | - | - | Terminated | - | - |
| NCT01863667 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | - | - |
| NCT01215097 | - | - | Completed | - | - |
| NCT00401973 | Schizophrenia ... more >> Schizoaffective Disorders Collapse << | Phase 3 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. National City, California, United States, 91950 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Canton, Ohio, United States, 44708 United States, Tennessee For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Johnson City, Tennessee, United States, 37614 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Memphis, Tennessee, United States, 38105 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Desoto, Texas, United States, 75115 Brazil For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rio De Janeiro, Brazil, 21020-130 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Salvador, Brazil, 40301500 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beer Yaacov, Israel, 70350 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 143-711 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mexico City, Mexico, 14050 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bayamon, Puerto Rico, 00961 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ponce, Puerto Rico, 00717 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico, 00907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 117603 Collapse << |
| NCT00729326 | - | - | Completed | - | - |
| NCT00401973 | - | - | Completed | - | - |
| NCT00484419 | - | - | Completed | - | - |
| NCT00729326 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00484419 | Type 2 Diabetes ... more >> Hyperlipidemia Collapse << | Phase 3 | Completed | - | United States, California ... more >> La Mesa, California, United States Los Angeles, California, United States Los Gatos, California, United States San Antonio, California, United States United States, Florida Chiefland, Florida, United States United States, Indiana Gary, Indiana, United States United States, Maryland Baltimore, Maryland, United States United States, Michigan Dearborn, Michigan, United States W. Bloomfield, Michigan, United States United States, Nevada Las Vegas, Nevada, United States United States, New York Yonkers, New York, United States United States, North Carolina Lexington, North Carolina, United States Winston-Salem, North Carolina, United States United States, Ohio Munroe Falls, Ohio, United States Zaneville, Ohio, United States United States, Oregon Portland, Oregon, United States United States, Pennsylvania Jersey Shore, Pennsylvania, United States United States, South Carolina Clemson, South Carolina, United States United States, Tennessee Harriman, Tennessee, United States United States, Texas Dallas, Texas, United States Collapse << |
| NCT03307252 | Healthy | Phase 1 | Completed | - | Germany ... more >> CTC North GmbH & Co. KG, Hamburg Hamburg, Germany, 20251 Collapse << |
| NCT00577590 | Diabetes Mellitus, Type II | Not Applicable | Completed | - | United States, Missouri ... more >> Washington University in St. Louis Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00577590 | - | - | Completed | - | - |
| NCT02648204 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01915264 | - | - | Withdrawn | March 2017 | - |
| NCT00674466 | - | - | Completed | - | - |
| NCT00674466 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Quebec ... more >> Maggie Wang Montreal, Quebec, Canada, H2X 3Y8 Collapse << |
| NCT02648204 | - | - | Completed | - | - |
| NCT02229383 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00807092 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100730 Collapse << |
| NCT02229383 | - | - | Completed | - | - |
| NCT00396851 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | - | India ... more >> PGIMER Not yet recruiting Chandigarh, India, 160012 Contact: Samir Malhotra samirmalhotra345@yahoo.com Sub-Investigator: Promila Pandhi, MD,DM Collapse << |
| NCT00807092 | - | - | Completed | - | - |
| NCT01930188 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01517412 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02177643 | Diabetes-Related Complications... more >> Diabetes Mellitus, Type 2 Insulin Resistance Collapse << | Phase 2 | Unknown | - | Brazil ... more >> Universidade Estadual de Feira de Santana Recruiting Feira de Santana, Bahia, Brazil Contact: Dra. Ana Mayra A. Oliveira, PhD +55 (075) 3625-4027 anamayra@uol.com.br Principal Investigator: Dra. Ana Mayra A. Oliveira, PhD Centro de Estudos em Diabetes e Hipertensão Recruiting Fortaleza, Ceará, Brazil Contact: Dra. Adriana C. Forti, PhD +55 (085) 3105-8300 cedh@cedh.med.br Principal Investigator: Dra. Adriana C. Forti, PhD Universidade Estadual de Campinas Recruiting Campinas, São Paulo, Brazil Contact: Dra. Maria Cândida R. Parisi, PhD +55 (019) 98196-2242 emaildacandida@uol.com.br Contact: Dr. Daniel Minuti, MD Msc +55 (019) 98128-2535 danielminutti@yahoo.com.br Sub-Investigator: Dr. Daniel Minuti, MD Msc Collapse << |
| NCT01691989 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01517412 | - | - | Completed | - | - |
| NCT01612741 | - | - | Completed | - | India ... more >> Many Locations, India Collapse << |
| NCT00749190 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01517373 | - | - | Completed | - | - |
| NCT00460941 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01517373 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00473525 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00611884 | - | - | Completed | - | - |
| NCT00423501 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01106677 | - | - | Completed | - | - |
| NCT00749190 | - | - | Completed | - | - |
| NCT03208309 | Complications of Diabetes Mell... more >>itus Diabetes Mellitus Diabetes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Brazil ... more >> Universidade Estadual de Feira de Santana Feira de Santana, Bahia, Brazil Centro de Estudos em Diabetes e Hipertensão Fortaleza, Ceará, Brazil Universidade Estadual de Campinas Campinas, São Paulo, Brazil Collapse << |
| NCT01106677 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00611884 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT01649297 | - | - | Completed | - | - |
| NCT00833027 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02452632 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Site KR00002 Busan, Korea, Republic of Site KR00003 Busan, Korea, Republic of Site KR00007 Daegu, Korea, Republic of Site KR00011 Daegu, Korea, Republic of Site KR00019 Dong Gu Gwangju, Korea, Republic of Site KR00008 Gangwon-do, Korea, Republic of Site KR00018 Gyeonggi-do, Korea, Republic of Site KR00022 Gyeonggi-do, Korea, Republic of Site KR00014 Incheon, Korea, Republic of Site KR00020 Jeollabuk-do, Korea, Republic of Site KR00021 Kyungsangnam-do, Korea, Republic of Site KR00001 Seoul, Korea, Republic of Site KR00004 Seoul, Korea, Republic of Site KR00005 Seoul, Korea, Republic of Site KR00006 Seoul, Korea, Republic of Site KR00009 Seoul, Korea, Republic of Site KR00010 Seoul, Korea, Republic of Site KR00012 Seoul, Korea, Republic of Site KR00013 Seoul, Korea, Republic of Site KR00016 Seoul, Korea, Republic of Site KR00017 Seoul, Korea, Republic of Site KR00015 Suwon, Korea, Republic of Collapse << |
| NCT01649297 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00833027 | - | - | Completed | - | - |
| NCT01541215 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02973100 | - | - | Completed | - | - |
| NCT00469092 | - | - | Completed | - | - |
| NCT01541215 | - | - | Completed | - | - |
| NCT01486966 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Terminated(Trial terminated pr... more >>ematurely due to slow recruitment.) Collapse << | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100034 China, Chongqing Novo Nordisk Investigational Site Chongqing, Chongqing, China, 400016 China, Jiangsu Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200003 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 Collapse << |
| NCT02973100 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00624364 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | South Africa ... more >> Novo Nordisk Investigational Site Bloemfontein, Free State, South Africa, 9301 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1827 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1829 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 2001 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0116 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4001 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4091 Novo Nordisk Investigational Site Pietermaritzburg, KwaZulu-Natal, South Africa, 3235 Novo Nordisk Investigational Site Brits, North West, South Africa, 0250 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7708 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7800 Novo Nordisk Investigational Site Benoni, South Africa, 1500 Novo Nordisk Investigational Site Bloemfontein, South Africa, 27 51 Novo Nordisk Investigational Site Kimberley, South Africa, 8301 Novo Nordisk Investigational Site Richards Bay, South Africa, 3900 Collapse << |
| NCT03714594 | Glucose, Low Blood | Phase 2 | Recruiting | September 2021 | Italy ... more >> Department of Endocrinology and Metabolism, University of Pisa Recruiting Pisa, Italy, 56124 Contact: Stefano Del Prato, MD +39 050 995103 stefano.delprato@med.unipi.it Sub-Investigator: Giuseppe Daniele, MD Collapse << |
| NCT00613951 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Finland ... more >> Novo Nordisk Investigational Site Helsinki, Finland, 00260 Novo Nordisk Investigational Site Kuopio, Finland, 70210 Novo Nordisk Investigational Site Lahti, Finland, 15110 Novo Nordisk Investigational Site Pori, Finland, FI-28100 France Novo Nordisk Investigational Site Bar-Le-Duc, France, 55000 Novo Nordisk Investigational Site GRENOBLE cedex, France, 38043 Novo Nordisk Investigational Site Hayange, France, 57700 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Nanterre, France, 92014 Novo Nordisk Investigational Site NEVERS cedex, France, 58033 Novo Nordisk Investigational Site Pointe à Pitre, France, 97159 Germany Novo Nordisk Investigational Site Berlin, Germany, 12163 Novo Nordisk Investigational Site Pirna, Germany, 01796 Novo Nordisk Investigational Site Riesa, Germany, 01587 Novo Nordisk Investigational Site Saarbrücken, Germany, 66121 Novo Nordisk Investigational Site St. Ingbert, Germany, 66386 Novo Nordisk Investigational Site Völklingen, Germany, 66333 Novo Nordisk Investigational Site Wangen, Germany, 88239 Poland Novo Nordisk Investigational Site Bydgoszcz, Poland, 85-822 Novo Nordisk Investigational Site Gniewkowo, Poland, 88-140 Novo Nordisk Investigational Site Nysa, Poland, 48-300 Novo Nordisk Investigational Site Plock, Poland, 09-400 Novo Nordisk Investigational Site Szczecin, Poland, 70-483 Novo Nordisk Investigational Site Tychy, Poland, 43-100 Novo Nordisk Investigational Site Warszawa, Poland, 02-507 Novo Nordisk Investigational Site Wroclaw, Poland, 50-127 Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Barcelona, Spain, 08035 Novo Nordisk Investigational Site Granada, Spain, 18012 Novo Nordisk Investigational Site Madrid, Spain, 28034 Novo Nordisk Investigational Site San Juan, Spain, 03550 Collapse << |
| NCT00613951 | - | - | Completed | - | - |
| NCT01486966 | - | - | Terminated(Trial terminated pr... more >>ematurely due to slow recruitment.) Collapse << | - | - |
| NCT00231647 | Obesity Diabe... more >>tes Mellitus, Type 2 Diabetes Mellitus, Adult-Onset Collapse << | Phase 2 | Completed | - | - |
| NCT02647320 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00653302 | Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> Sanofi-aventis administrative office Paris, France Collapse << |
| NCT00614055 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | France ... more >> Novo Nordisk Investigational Site Alès, France, 30100 Novo Nordisk Investigational Site Brest, France, 29609 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Le Creusot, France, 71200 Novo Nordisk Investigational Site Mont de Marsan, France, 40024 Novo Nordisk Investigational Site Venissieux, France, 69200 Germany Novo Nordisk Investigational Site Bad Kreuznach, Germany, 55545 Novo Nordisk Investigational Site Bad Mergentheim, Germany, 97980 Novo Nordisk Investigational Site Dormagen, Germany, 41539 Novo Nordisk Investigational Site Hohenmölsen, Germany, 06679 Novo Nordisk Investigational Site Neuss, Germany, 41460 Novo Nordisk Investigational Site Neuwied, Germany, 56564 Norway Novo Nordisk Investigational Site Elverum, Norway, 2408 Novo Nordisk Investigational Site Hamar, Norway, 2317 Novo Nordisk Investigational Site Kongsvinger, Norway, 2212 Novo Nordisk Investigational Site Oslo, Norway, 0586 Novo Nordisk Investigational Site Stavanger, Norway, 4011 Novo Nordisk Investigational Site Tromsø, Norway, 9038 Romania Novo Nordisk Investigational Site Bucharest, Romania, 011234 Novo Nordisk Investigational Site Bucharest, Romania, 020042 Novo Nordisk Investigational Site Bucharest, Romania, 020475 Novo Nordisk Investigational Site Bucharest, Romania, 020992 Novo Nordisk Investigational Site Bucharest, Romania Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Partida de Bacarot, Spain, 03114 Novo Nordisk Investigational Site Sevilla, Spain, 41010 Novo Nordisk Investigational Site Valencia, Spain, 46014 Novo Nordisk Investigational Site Valladolid, Spain, 47011 Collapse << |
| NCT00631488 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00614055 | - | - | Completed | - | - |
| NCT00631488 | - | - | Completed | - | - |
| NCT00823992 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT02647320 | - | - | Completed | - | - |
| NCT00147719 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02053272 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Romania ... more >> Diabol SRL Brasov, Romania, 500365 Nicodiab SRL Bucuresti, Romania, 010507 Spitalul Universitar de Urgenta Militar Central "Dr. Carol Davila" Bucuresti, Romania, 010825 Institutul National de Diabet Bucuresti, Romania, 020475 Centrul Medical 'Sanatatea Ta' SRL Bucuresti, Romania, 020614 ArtMedical Clinic Bucuresti, Romania, 021107 Societatea Civila Medicala dr. Paveliu Bucuresti, Romania, 050538 Consultmed SRL Iasi, Romania, 700547 Cabinet Medical Individual Diabet Nutritie si Boli Metabolice Maramures, Romania, 430123 Grandmed SRL Oradea, Romania, 410159 Spitalul Judetean de Urgente Satu Mare Satu Mare, Romania, 440055 Spitalul Clinic Judetean de Urgenta Sibiu Sibiu, Romania, 550245 Gagiu D. Remus Cabinet Medical Individual Targoviste, Romania, 130083 Mediab SRL Diabet Zaharat Targu Mures, Romania, 540142 United Kingdom Avondale Surgery Chesterfield, Derbyshire, United Kingdom, S40 4TF Hull and East Yorkshire Hospitals NHS Trust Hull, East Yorkshire, United Kingdom, HU3 2JZ Salford Royal NHS Foundation Trust Salford, Greater Manchester, United Kingdom, M6 8HD University Hospitals of Leicester NHS Trust Leicester, Leicestershire, United Kingdom, LE5 4QF Aintree University Hospitals NHS Foundation Trust Liverpool, Merseyside, United Kingdom, L9 7AL Strensall Medical Practice York, North Yorkshire, United Kingdom, YO32 5UA Churchill Hospital Oxford, Oxfordshire, United Kingdom, OX3 9DU Hathaway Surgery Chippenham, Wiltshire, United Kingdom, SN14 6GT Avenue Surgery Warminster, Wiltshire, United Kingdom, BA12 9AA St Chad's Surgery Bath, United Kingdom, BA3 2UH Barts and The London NHS Trust London, United Kingdom, E1 1BB Collapse << |
| NCT01051011 | Diabetes Mellitus Type 2 | Phase 3 | Terminated(high discontinuatio... more >>n rates mainly due to GI tolerability and implementation of risk mitigation plan to address hypersensitivity reactions) Collapse << | - | - |
| NCT00944177 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St, Charles, Missouri, United States Collapse << |
| NCT02872818 | Apoptotic Signal Pathways in E... more >>ndometrial Hyperplasia Collapse << | Phase 4 | Completed | - | Turkey ... more >> Kayseri Training and Research Hospital Kayseri, Turkey, 38000 Collapse << |
| NCT01951183 | Diabetes Mellitus Type 2 | Phase 2 | Withdrawn(Cancelled due to Spo... more >>nsor's decision) Collapse << | December 2013 | - |
| NCT00770835 | Diabetes Mellitus | Phase 4 | Completed | - | Italy ... more >> Padova, Italy Pisa, Italy Collapse << |
| NCT00753896 | Type 2 Diabetes | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Mesa, Arizona, United States Research Site Tempe, Arizona, United States United States, California Research Site Concord, California, United States Research Site Fresno, California, United States Research Site La Mesa, California, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Idaho Research Site Idaho Falls, Idaho, United States United States, Kentucky Research Site Bowling Green, Kentucky, United States United States, Oregon Research Site Corvallis, Oregon, United States United States, Tennessee Research Site Chattanooga, Tennessee, United States Research Site Memphis, Tennessee, United States Canada, British Columbia Research Site New Westminister, British Columbia, Canada Canada, Ontario Research Site Ajax, Ontario, Canada Research Site Cambridge, Ontario, Canada Research Site Windsor, Ontario, Canada Mexico Research Site Chihuahua, Chiuahua, Mexico Research Site Monterrey, Nuevo Leon, Mexico Research Site Distrito Federal, Mexico Romania Research Site Baia Mare, Romania Research Site Brasov, Romania Research Site Bucharesti, Romania Research Site Craiova, Romania Research Site Iasi, Romania Research Site Suceava, Romania South Africa Research Site Johannesburg, South Africa Research Site Pretoria, South Africa Collapse << |
| NCT00106340 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers Nurnberg, Germany Collapse << |
| NCT00944346 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St, Charles, Missouri, United States Collapse << |
| NCT00753896 | - | - | Completed | - | - |
| NCT02053103 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(On 25April2014, stu... more >>dy was terminated before any dosing due to an AE of safety concern that occurred in protocol B1731003 with the same drug.) Collapse << | - | United States, Florida ... more >> Pfizer Investigational Site Orlando, Florida, United States, 32809 Collapse << |
| NCT02033889 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00209482 | Obesity | Phase 2 Phase 3 | Unknown | - | United States, California ... more >> University of California, Los Angeles Los Angeles, California, United States, 90095 University of California, San Francisco San Francisco, California, United States, 94143 Stanford University Stanford, California, United States, 94305 United States, Massachusetts Children's Hospital, Boston Boston, Massachusetts, United States, 02115 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00279240 | Diabetes | Phase 1 | Completed | - | India ... more >> Diabetes Research Centre & MV Hospital for Diabetes Chennai, Tamilnadu, India, 600013 Collapse << |
| NCT00975286 | - | - | Completed | - | - |
| NCT01892020 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT00214591 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT02033889 | - | - | Completed | - | - |
| NCT01892020 | - | - | Completed | - | - |
| NCT00975286 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00318461 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03452267 | Insulin Sensitivity | Phase 2 Phase 3 | Not yet recruiting | June 2020 | - |
| NCT00038727 | Diabetes Mellitus ... more >> Cancer CVD Collapse << | Phase 3 | Active, not recruiting | January 2025 | United States, Maryland ... more >> George Washington University Rockville, Maryland, United States, 20852 Collapse << |
| NCT00544518 | Type 2 Diabetes | Phase 2 | Unknown | October 2008 | China, Shanxi ... more >> XiJing hospital of Fourth Military Medical University Xi'an, Shanxi, China, 710032 Collapse << |
| NCT00700856 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 4 | Unknown | December 2018 | - |
| NCT01868542 | - | - | Completed | - | - |
| NCT00038727 | - | - | Active, not recruiting | - | - |
| NCT00231660 | Obesity Diabe... more >>tes Mellitus, Type 2 Diabetes Mellitus, Adult-Onset Collapse << | Phase 3 | Completed | - | - |
| NCT00318461 | - | - | Completed | - | - |
| NCT00822211 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> China-Japan Friendship Hospital Beijing, China General Hospital of Beijing Military Region of PLA Beijing, China Peking Union Medical College Hospital Beijing, China Peking University First Hospital Beijing, China The General Hospital of the PLA Beijing, China The General Hospital of the Second Artilleryman of PLA Beijing, China The Affiliated Union Hospital of Fujian Medical University Fuzhou, China The Fuzhou General Hospital of the PLA Nanjing Military Area Fuzhou, China Guangdong Provincial People's Hospital Guangzhou, China Second Affiliated Hospital of Nanchang University Nanchang, China The Jiangxi Provincial People's Hospital Nanchang, China Te Affiliated Drum Tower of Nanjing University Medical School Nanjing, China Tongji Hospital of Tongji University Nanjing, China Shanghai Changzheng Hospital Shanghai, China The Second Affiliated Hospital of China Medical University Shenyang, China The Second Affiliated Hospital of Tianjin Medical University Tianjin, China First Affiliated Hospital of 4th Military Medical University Xi'an, China Collapse << |
| NCT00265746 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | - | Poland ... more >> Department of Endocrinology and Isotope Therapy, Polish Mother’s Memorial Hospital, Research Institute, Lodz Recruiting Lodz, Poland Contact: Katarzyna Cypryk, M D, PhD , Habilitation 0048422711159 kcypryk@mp.pl Principal Investigator: Katarzyna Cypryk, PhD, MD , Sub-Investigator: Marcin Kosiński, MD Collapse << |
| NCT03259789 | Type2 Diabetes Mellitus | Phase 3 | Active, not recruiting | January 2019 | - |
| NCT02135549 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, New York ... more >> Accumed Garden City, New York, United States, 11530 Collapse << |
| NCT01868542 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 301-721 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 301-723 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 302-120 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 302-718 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 330-721 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 361-711 Collapse << |
| NCT00399360 | HIV Infections ... more >> Metabolic Syndrome X Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03279263 | Type 2 Diabetes Mellitus | Phase 2 | Recruiting | September 2019 | United States, California ... more >> National Research Institute Recruiting Los Angeles, California, United States, 90057 Contact: Juan Frias, MD Collapse << |
| NCT01511198 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00399360 | - | - | Completed | - | - |
| NCT02932475 | Diabetes Preg... more >>nancy Collapse << | Phase 3 | Recruiting | June 30, 2021 | United States, Alabama ... more >> University of Alabama Not yet recruiting Birmingham, Alabama, United States, 35294 Contact: Lori Harper, MD 205-934-5611 lmharper@uabmc.edu United States, California University of California - San Diego Not yet recruiting San Diego, California, United States, 92093 Contact: Gladys Ramos, MD 619-543-6906 gramos@ucsd.edu United States, Mississippi University of Mississippi Not yet recruiting Jackson, Mississippi, United States, 38677 Contact: Kedra Wallace, MD 601-984-5396 kwallace2@umc.edu United States, New York Columbia University Not yet recruiting New York, New York, United States, 10027 Contact: Ronald Wapner, MD 212-305-1521 rw2191@columbia.edu United States, North Carolina University of North Carolina Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Kim Boggess, MD 919-966-1601 kboggess@med.unc.edu Contact: Karen Dorman 984-974-9012 kdorman@med.unc.edu United States, Ohio Ohio State University Not yet recruiting Columbus, Ohio, United States, 43210 Contact: Mark Landon, MD 614-293-8513 mark.landon@osumc.edu United States, Pennsylvania University of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Celeste Durswald, MD 215-615-3817 celeste.durnwald@uphs.upenn.edu United States, Texas University of Texas Medical Branch Not yet recruiting Galveston, Texas, United States, 77555 Contact: Gayle Olson, MD 409-747-4905 golson@utmb.edu University of Texas - Houston Not yet recruiting Houston, Texas, United States, 77030 Contact: Jeffie Refuerzo 713-500-6416 jerrie.s.refuerzo@uth.tmc.edu United States, Utah University of Utah Not yet recruiting Salt Lake City, Utah, United States, 84112 Contact: Michael Varner 801-581-4173 michael.varner@hsc.utah.edu Collapse << |
| NCT01999218 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03444584 | Type 2 Diabetes Mellitus | Phase 2 | Active, not recruiting | December 10, 2018 | Germany ... more >> Research Site Magdeburg, Germany, 39120 Research Site Mannheim, Germany, 68167 Research Site München, Germany, 81241 Hungary Research Site Balatonfüred, Hungary, 8230 Research Site Miskolc, Hungary, 3529 Research Site Szeged, Hungary, 6720 United Kingdom Research Site Liverpool, United Kingdom, L7 8XP Research Site Manchester, United Kingdom, M13 9NQ Research Site Rotherham, United Kingdom, S65 1DA Research Site Swansea, United Kingdom, SA2 8PP Collapse << |
| NCT03165877 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT02989649 | - | - | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns) Collapse << | - | China, Beijing ... more >> Beijing, Beijing, China China, Hubei Wuhan, Hubei, China China, Liaoning Dalian, Liaoning, China Shenyang, Liaoning, China China, Shandong Jinan, Shandong, China Qingdao, Shandong, China Tai'an, Shandong, China Taian, Shandong, China China, Tianjin Tianjin, Tianjin, China Hong Kong Sai Ying Pun, Hong Kong Sha Tin, Hong Kong Collapse << |
| NCT01557504 | - | - | Completed | - | - |
| NCT01427920 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01999218 | - | - | Completed | - | - |
| NCT01557504 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT02491333 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 3 | Unknown | December 2017 | China, Guangdong ... more >> The First Affiliated Hospital of Guangzhou Medical University Recruiting Guangzhou, Guangdong, China, 510120 Contact: Wanting Wu, Master 15818153730 Contact: Meifang Li, Master 13544426679 Principal Investigator: Hongxia Ma, PhD Collapse << |
| NCT00469586 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT01303055 | Diabetes Mellitus, Type 2 | Not Applicable | Unknown | December 2014 | Japan ... more >> Dept. of Intern. Med., School of Dentistry, Aichi Gakuin University Recruiting Nagoya, Aichi, Japan, 464-8650 Contact: Takahiro Tosaki, MD, PhD +81-52-759-2111 nrd49075@nifty.com Principal Investigator: Takahiro Tosaki, MD, PhD Sub-Investigator: Keiko Naruse, MD, PhD Tosaki Clinic for Diabetes and Endocrinology Not yet recruiting Nagoya, Aichi, Japan, 468-0009 Contact: Takahiro Tosaki, MD, PhD nrd49075@nifty.com Principal Investigator: Takahiro Tosaki, MD, PhD Diabetes Clinic, Okazaki East Hospital Recruiting Okazaki, Aichi, Japan, 444-0008 Contact: Takahiro Tosaki, MD, PhD +81-564-22-6616 nrd49075@nifty.com Sub-Investigator: Chizuko Suzuki, MD, PhD Sub-Investigator: Maiko Mizutani, MD, PhD Principal Investigator: Takahiro Tosaki, MD, PhD Diabetes Center, Yokkaichi Social Insurance Hospital Recruiting Yokkaichi, Mie, Japan, 510-0016 Contact: Takahiro Tosaki, MD, PhD +81-59-331-2000 nrd49075@nifty.com Sub-Investigator: Miho Miyoshi, MD Sub-Investigator: Tomoki Majima, MD Principal Investigator: Takahiro Tosaki, MD, PhD Collapse << |
| NCT01632163 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01427920 | - | - | Completed | - | - |
| NCT02076347 | Type 2 Diabetes ... more >> Vitamin B12 Deficiency Neuropathy Anemia Collapse << | Not Applicable | Completed | - | United States, Ohio ... more >> The Ohio State University General Internal Medicine Columbus, Ohio, United States, 43221 Collapse << |
| NCT01084486 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Phase 1 Phase 2 | Completed | - | Israel ... more >> Wolfson Medical Center Holon, Israel, 58100 Collapse << |
| NCT02149459 | Brain Neoplasms | Phase 1 | Recruiting | July 2018 | Israel ... more >> Sheba Medical Center Recruiting Ramat Gan, Israel, 52621 Collapse << |
| NCT01733459 | Polycystic Ovary Syndrome (PCO... more >>S) Collapse << | Phase 3 | Completed | - | Indonesia ... more >> Department of Obstetrics and Gynecology, Faculty of Medicine, University of Padjadjaran, Hasan Sadikin Hospital Bandung, West Java, Indonesia, 40161 Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Indonesia, dr. Cipto Mangunkusumo Hospital Jakarta, Indonesia, 10430 Collapse << |
| NCT01210001 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00536211 | Metabolic Syndrome X ... more >> Insulin Resistance Hypertension Hypercholesterolemia Obesity Collapse << | Not Applicable | Withdrawn(protocol not approve... more >>d by VA R&D) Collapse << | December 2011 | United States, Missouri ... more >> Harry S. Truman Memorial Veterans' Hospital Columbia, Missouri, United States, 65201 Collapse << |
| NCT01210001 | - | - | Completed | - | - |
| NCT02752412 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02730741 | Antidiarrhoea | Phase 2 | Recruiting | - | France ... more >> BIOSE Recruiting Aurillac, France Contact: NIVOLIEZ Mr Adrien +330471465101 a.nivoliez@biose.com Principal Investigator: Pr. Jacques MOREAU Collapse << |
| NCT00734474 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT01462266 | - | - | Completed | - | - |
| NCT01462266 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00734474 | - | - | Completed | - | - |
| NCT02501616 | Endothelial Function ... more >> Type 2 Diabetes Collapse << | Phase 4 | Unknown | - | Korea, Republic of ... more >> Boramae medical center Recruiting Seoul, Korea, Republic of, 156-707 Contact: Bokyung Koo bokyungkoomd@gmail.com Principal Investigator: Min Kyong Moon Collapse << |
| NCT00184626 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novo Nordisk Investigational Site Istanbul, Turkey, 111 Collapse << |
| NCT01476475 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01549964 | - | - | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01549964 | Glycemic Control | Phase 3 | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01511172 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00191464 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Hampshire ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lebanon, New Hampshire, United States, 03756 Australia, New South Wales For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. St Leonards, New South Wales, Australia, 2065 Greece For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Thessaloniki, Greece, 56429 India For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mumbai, Maharstra, India, 400 007 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Den Helder, Netherlands, 1783 GZ Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Olesnica, Poland, 56-400 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ponce, Puerto Rico, 00731 Collapse << |
| NCT01137812 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01476475 | - | - | Completed | - | - |
| NCT01137812 | - | - | Completed | - | - |
| NCT03194945 | Type 2 Diabetes Mellitus | Phase 4 | Not yet recruiting | December 31, 2020 | China, Guangdong ... more >> endocrinology department of the first affiliated hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT03538704 | Endometrial Cancer ... more >> Fertility Collapse << | Not Applicable | Recruiting | December 31, 2020 | China, Beijing ... more >> Peking University People's Hospital Recruiting Beijing, Beijing, China, 100044 Contact: Jianliu Wang, Professor 010-88324381 wangjianliu1203@163.com Collapse << |
| NCT03332771 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | August 6, 2019 | - |
| NCT03061981 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Maryland ... more >> Early Phase Clinical Unit Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00683735 | Diabetes | Phase 2 Phase 3 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Collapse << |
| NCT02302326 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | December 2017 | Spain ... more >> Antonio Hernández Recruiting Valencia, Spain, 46017 Contact: Antonio Hernández, Phd, MD 34961622492 hernandez_antmij@gva.es Contact: Celia Bañuls, PhD 34961622757 celia.banuls@uv.es Collapse << |
| NCT00350779 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02924064 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational center Beijing, China Collapse << |
| NCT00350779 | - | - | Completed | - | - |
| NCT02730377 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Active, not recruiting | August 12, 2019 | - |
| NCT02058147 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00860288 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT01617434 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01682759 | - | - | Completed | - | - |
| NCT02058147 | - | - | Completed | - | - |
| NCT01617434 | - | - | Completed | - | - |
| NCT01682759 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03639545 | Vascular Stiffness ... more >> Hypoglycemic Agents Diabetes Complications Diabetes Mellitus, Type 1 Collapse << | Phase 4 | Recruiting | January 30, 2019 | Slovenia ... more >> University Medical Centre Ljubljana Recruiting Ljubljana, Slovenia, SI-1000 Contact: Mojca Lunder, MD, PhD +386 1 5223140 mojca.lunder@kclj.si Contact: Andrej Janez, Prof +386 1 5223564 andrej.janez@kclj.si Principal Investigator: Miodrag Janic, MD, PhD Principal Investigator: Miso Sabovic, Prof Collapse << |
| NCT01956929 | Insulin Resistance ... more >> Prediabetic State Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01510522 | Diabetes Mellitus Type 2 | Phase 4 | Withdrawn | - | - |
| NCT03718715 | - | - | Not yet recruiting | March 2021 | - |
| NCT03685409 | Oral Cancer | Phase 3 | Recruiting | September 30, 2020 | Egypt ... more >> Faculty of Dentistry- Cairo University Recruiting Cairo, Egypt Contact: Fatheya M Zahran, Professor 00201060775113 Fatheyazahran@gmail.com Contact: Gihan G Madkour, As.Prof. 00201121001005 gihanemadkour@gmail.com Collapse << |
| NCT02252965 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Germany ... more >> Please contact the Merck KGaA Communication Center Darmstadt, Germany Collapse << |
| NCT02252965 | - | - | Completed | - | - |
| NCT02435277 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 United States, Florida Palm Beach Research Palm Beach, Florida, United States, 33409 Meridien Research Tampa, Florida, United States, 33606 United States, Georgia River Birch Research Alliance Blue Ridge, Georgia, United States, 3051313 Meridian Research Savannah, Georgia, United States, 31406 United States, Ohio Streling Research Group Cincinnati, Ohio, United States, 45219 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Meharry Medical College Nashville, Tennessee, United States, 37208 Vanderbilt University Medical Center, Diabetes, Endocrinology, and Metabolism Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01529177 | Infertility | Phase 4 | Unknown | February 2017 | Egypt ... more >> The Egyptian IVF-ET center Recruiting Cairo, Egypt, 11431 Contact: Ragaa mansour 20225254955 ragaa.mansour@gmail.com Principal Investigator: Ragaa mansour, MD,PhD Collapse << |
| NCT02435277 | - | - | Completed | - | - |
| NCT01279512 | PCO | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Royan Institute Tehran, Iran, Islamic Republic of Collapse << |
| NCT01487993 | Obesity Insul... more >>in Resistance Collapse << | Phase 3 | Completed | - | Netherlands ... more >> Jeroen Bosch Hospital 's Hertogenbosch, Netherlands St. Antonius Hospital Nieuwegein, Netherlands, 3430EM Collapse << |
| NCT02176161 | Prostate Cancer ... more >> Prostate Cancer Recurrent Collapse << | Phase 2 | Recruiting | June 2020 | United States, New York ... more >> Winthrop University Hospital Recruiting Mineola, New York, United States, 11501 Principal Investigator: Aaron E Katz, MD Collapse << |
| NCT03629639 | Psoriasis Vulgaris With Metabo... more >>lic Disorders Collapse << | Phase 4 | Not yet recruiting | December 1, 2020 | - |
| NCT02437656 | Rectal Cancer | Phase 2 | Completed | - | France ... more >> Centre Marie Curie Arras, France, 62000 Centre Pierre Curie Beuvry, France, 62880 Centre Léonard de Vinci - SARL du pont Saint Vaast Douai, France, 59500 Institut André Dutreix Dunkerque, France, 59240 Centre Hospitalier Lens, France, 62300 Clinique du Bois - Centre Bourgogne Lille, France, 59000 Centre Oscar Lambret Lille, France, 59020 Centre Galilée - Hôpital Privé La Louvière Lille, France, 59045 Centre Gray Maubeuge, France, 59600 Centre Joliot-Curie St Martin-Boulogne, France, 62200 Clinique des Dentellières Valenciennes, France, 59300 Collapse << |
| NCT01231074 | Obesity Weigh... more >>t Gain Psychotropic Induced Weight Gain Collapse << | Phase 1 | Unknown | May 2011 | United States, Ohio ... more >> Nationwide Children's Hospital Recruiting Columbus, Ohio, United States, 43205 Contact: Ihuoma Eneli, MD, MS 614-722-4089 ihuoma.eneli@nationwidechildrens.org Contact: Mary Ann Murphy, MD, PhD 614-722-2291 maryann.murphy@nationwidechildrens.org Collapse << |
| NCT01677260 | Metformin XR BE Study in Healt... more >>hy Volunteers With Single and Multiple Dose Collapse << | Not Applicable | Completed | - | Indonesia ... more >> PT Equilab International Jakarta, Indonesia, 12430 Collapse << |
| NCT02733679 | Ataxia-Telangiectasia | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital Dundee, Angus, United Kingdom, DD19SY Collapse << |
| NCT01526616 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | - |
| NCT03710343 | Cerebral Palsy ... more >> Spastic Spastic Cerebral Palsy Cerebral Palsy, Spastic Cerebral Palsy, Mixed Collapse << | Phase 2 | Not yet recruiting | February 1, 2022 | Canada, Ontario ... more >> Holland Bloorview Kids Rehabilitation Hospital Not yet recruiting Toronto, Ontario, Canada, M4G1R8 Contact: Daniel Warner, BScH 416-425-6220 ext 6482 dwarner@hollandbloorview.ca Principal Investigator: Darcy Fehlings, MD, MSc The Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT02659306 | HIV Infection | Phase 1 | Active, not recruiting | December 2018 | Canada, Quebec ... more >> Chronic Viral Illness Service, McGill University Health Centre Montreal, Quebec, Canada, H4A 3J1 Collapse << |
| NCT01440127 | Colon Cancer | Phase 1 | Terminated(Unable to accrue al... more >>l planned subjects in a timely fashion, but data collected will still be analyzed.) Collapse << | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01546558 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Florida ... more >> SeaView Research Inc. Miami, Florida, United States, 33126 Collapse << |
| NCT02378441 | Healthy | Phase 1 | Completed | - | - |
| NCT03117517 | Polycystic Ovarian Syndrome | Early Phase 1 | Completed | - | Pakistan ... more >> Mardan Medical Complex Mardan, Khyber Pukhtunkhwah, Pakistan, 25000 Collapse << |
| NCT02042495 | Endometrial Cancer | Phase 2 | Withdrawn(Did not obtain Healt... more >>h Canda approval for drug use in reasonable time and moved onward to new trial) Collapse << | - | Canada, Quebec ... more >> Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT01312467 | Adenomatous Polyp ... more >> Colorectal Cancer Obesity Collapse << | Phase 2 | Completed | - | United States, California ... more >> Veterans Administration Long Beach Medical Center Long Beach, California, United States, 90822 University of California Medical Center At Irvine-Orange Campus Orange, California, United States, 92868 Kaiser Permanente - Sacramento Sacramento, California, United States, 95825 Canada, Quebec Jewish General Hospital Montreal, Quebec, Canada, H3T 1E2 Collapse << |
| NCT01312467 | - | - | Completed | - | - |
| NCT00570739 | Type 2 Diabetes Mellitus ... more >> Hypercholesterolemia Pre-diabetes Collapse << | Phase 3 | Completed | - | - |
| NCT03684707 | Oral Cancer | Phase 4 | Recruiting | September 15, 2019 | Egypt ... more >> Noha Nasr Recruiting Cairo, New Cairo, Egypt, 11835 Contact: Noha N. El-Zalabany, Masters 1005365769 ext 02 noha_nasr84@yahoo.com Contact: Noha N. El-Zalabany, Masters 01005365769 ext 02 noha_nasr84@yahoo.com Collapse << |
| NCT00865839 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT02035787 | Complex Atypical Hyperplasia ... more >> Endometrial Cancer Collapse << | Not Applicable | Recruiting | March 2020 | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Jennifer Gruhn 919-966-4432 jennifer_gruhn@med.unc.edu Principal Investigator: Allison Staley, MD Principal Investigator: Victoria Bae-Jump, MD United States, Ohio TriHealth Recruiting Cincinnati, Ohio, United States, 45220 Principal Investigator: Kevin Schuler, MD Collapse << |
| NCT00864669 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT01422746 | Obesity Hyper... more >>androgenemia Polycystic Ovary Syndrome Collapse << | Not Applicable | Recruiting | December 2019 | United States, Virginia ... more >> University of Virginia Center for Research in Reproduction Recruiting Charlottesville, Virginia, United States, 22908 Contact: Deborah Sanderson 434-243-6911 pcos@virginia.edu Principal Investigator: Christine Burt Solorzano, MD Sub-Investigator: John C. Marshall, MD, PhD Collapse << |
| NCT00865748 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT00570739 | - | - | Completed | - | - |
| NCT02308228 | Aging | Not Applicable | Active, not recruiting | January 2019 | United States, Alabama ... more >> University of Alabama Birmingham Birmingham, Alabama, United States, 35294 United States, Kentucky University of Kentucky Lexington, Kentucky, United States, 40536 Collapse << |
| NCT01409395 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Ucsf Ctsi Crc San Francisco, California, United States, 94158 Collapse << |
| NCT01561482 | Prostate Carcinoma | Phase 2 | Withdrawn(Study closed due to ... more >>slow/low enrollment; no subjects were enrolled.) Collapse << | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Ben Taub General Hospital Houston, Texas, United States, 77030 Michael E. Debakey Veterans Affairs Medical Center Houston, Texas, United States, 77030 Collapse << |
| NCT02815397 | Diffuse Large B-Cell Lymphoma | Phase 2 | Terminated(Poor accrual) | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT02815397 | - | - | Terminated(Poor accrual) | - | - |
| NCT02072382 | Hypertension | Phase 4 | Unknown | December 2015 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Recruiting Porto Alegre, RS, Brazil Contact: Corrêa Junior Vicente (51)99839395 vicentecorreajunior@terra.com.br Sub-Investigator: Correa Junior Vicente Collapse << |
| NCT03467763 | Metformin Adverse Reaction ... more >> Tolerance Collapse << | Phase 4 | Recruiting | July 1, 2019 | United States, New York ... more >> Weill Cornell Medical College Recruiting New York, New York, United States, 10065 Contact: James H Flory, MD 646-962-9309 jaf9052@med.cornell.edu Contact: Leon Igel, MD 6469622111 lei9004@med.cornell.edu Collapse << |
| NCT03183752 | Thyroid Nodule ... more >> Insulin Resistance Collapse << | Not Applicable | Recruiting | July 10, 2018 | Brazil ... more >> Patricia Santos Recruiting Rio de Janeiro, Brazil, 21941913 Contact: Patricia Santos 55 21 981669799 patriciaborges.endocrino@gmail.com Collapse << |
| NCT02593097 | Migraine | Phase 3 | Active, not recruiting | December 2018 | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 Collapse << |
| NCT02109549 | - | - | Completed | - | Netherlands ... more >> The Netherlands Cancer Institute (NKI) Amsterdam, Netherlands, 1066 CX AMC Amsterdam, Netherlands, 1105 AZ MAASTRO clinic Maastricht, Netherlands, 6229 ET Collapse << |
| NCT01266486 | Breast Cancer | Phase 2 | Completed | - | United Kingdom ... more >> Mount Vernon Centre for Cancer Treatment, Rickmansworth Road Northwood, Middlesex, United Kingdom, HA6 2RN Dept Oncology, Churchill Hospital, Old Road, Headington Oxford, Oxfordshire, United Kingdom, OX3 7LJ Surgery and Molecular Oncology Ninewells Hospital Dundee, Scotland, United Kingdom, DD1 9SY Collapse << |
| NCT01910246 | Insulin Resistance | Phase 4 | Withdrawn(No funding) | - | United States, California ... more >> UCSF, Radiology and Biomedical Imaging San Francisco, California, United States, 94143 Collapse << |
| NCT01884558 | Pharmacokinetics of Isavuconaz... more >>ole Pharmacokinetics of Metformin Healthy Subjects Collapse << | Phase 1 | Completed | - | United States, California ... more >> California Clinical Trials Medical Group Glendale, California, United States, 91026 Collapse << |
| NCT01305551 | Type 2 Diabetes | Phase 1 | Withdrawn | - | Brazil ... more >> Local Institution Campinas, Sao Paulo, Brazil, 13073 Collapse << |
| NCT01685762 | Endometrial Hyperplasia ... more >> Endometrial Hyperplasia Without Atypia Collapse << | Early Phase 1 | Active, not recruiting | December 2018 | United States, North Carolina ... more >> The University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Southern Pines Women's Health Center Southern Pines, North Carolina, United States, 28388 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02198417 | Polycystic Ovary Syndrome | Not Applicable | Active, not recruiting | December 2020 | - |
| NCT03335423 | Metformin Org... more >>anic Cation Transporter 1 Codeine Collapse << | Phase 1 | Not yet recruiting | July 31, 2021 | - |
| NCT02432287 | - | - | Completed | - | - |
| NCT02432287 | Aging | Phase 4 | Completed | - | United States, New York ... more >> Einstein College of Medicine of Yeshiva University Bronx, New York, United States, 10461 Collapse << |
| NCT00865852 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT02285855 | Lung Cancer | Phase 2 | Active, not recruiting | February 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00865072 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT03510390 | Head and Neck Cancer ... more >> Oral Cavity Squamous Cell Carcinoma Collapse << | Not Applicable | Recruiting | September 30, 2019 | Switzerland ... more >> Inselspital, Bern University Hospital Recruiting Bern, Switzerland, 3010 Contact: Olgun Elicin, MD +41316322431 olgun.elicin@insel.ch Contact: Simon Mueller, MD +41316322931 simon.mueller@insel.ch Collapse << |
| NCT03618472 | Metformin CA ... more >>Endometrium Collapse << | Not Applicable | Recruiting | November 30, 2019 | Thailand ... more >> Rajavithi Hospital Recruiting Bangkok, Thailand, 10400 Contact: charuwan manmee, Phd 023548108 ext 2803 charuwan_manmee@yahoo.com Principal Investigator: kittisak Petchsila, MD Collapse << |
| NCT01334684 | Type 2 Diabetes | Not Applicable | Unknown | September 2013 | Italy ... more >> Casa Sollievo Della Sofferenza IRCCS Not yet recruiting San Giovanni Rotondo, Foggia, Italy, 71013 Collapse << |
| NCT01681693 | Healthy Type ... more >>2 Diabetes Mellitus Collapse << | Phase 1 | Completed | - | United States, California ... more >> San Francisco General Hospital San Francisco, California, United States, 94110 Collapse << |
| NCT02947503 | Gestational Diabetes ... more >> Insulin Resistance Collapse << | Phase 3 | Not yet recruiting | December 2040 | - |
| NCT01486043 | - | - | Terminated(The research projec... more >>t was terminated due to lower than projected patient recruitment within the period of time allowed for the study.) Collapse << | - | - |
| NCT03086733 | Non Small Cell Lung Cancer ... more >> Lung Cancer Collapse << | Phase 2 | Recruiting | December 2020 | Canada, Ontario ... more >> Toronto General Hospital Recruiting Toronto, Ontario, Canada, M5G 2C4 Contact: Alexandra Salvarrey, MD MSc 1-416-340-4800 ext 6992 alexandra.salvarrey@uhn.on.ca Contact: Jennifer Lister, BSc 1-416-340-4800 ext 4857 jennifer.lister@uhn.on.ca Principal Investigator: Thomas Waddell, MD FRCSC Princess Margaret Hospital Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Alexandra Salvarrey, MD MSc 1-416-946-4501 ext 5444 alexandra.salvarrey@uhn.on.ca Contact: Jennifer Lister, BSc 1-416-340-4800 ext 4857 jennifer.lister@uhn.on.ca Principal Investigator: Natasha Leighl, MD MSc FRCPC Collapse << |
| NCT02903511 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 2 | Recruiting | June 2020 | United States, Colorado ... more >> University of Colorado Denver, Anschutz Medical Campus Recruiting Aurora, Colorado, United States, 80045 Principal Investigator: Godela Brosnahan, MD Sub-Investigator: Michel Chonchol, MD Sub-Investigator: Berenice Gitomer, PhD Collapse << |
| NCT01681680 | Healthy | Phase 1 | Completed | - | - |
| NCT01442870 | Cancer | Phase 1 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01613755 | Diabetes | Phase 4 | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Centre Nijmegen, Gelderland, Netherlands, 6500 HB Collapse << |
| NCT03086005 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Songpa-gu, Korea, Republic of, 05505 Collapse << |
| NCT02915198 | Prediabetic State ... more >> Atherosclerosis Metformin Collapse << | Phase 4 | Not yet recruiting | June 30, 2024 | United States, Arizona ... more >> Phoenix VA Health Care System, Phoenix, AZ Not yet recruiting Phoenix, Arizona, United States, 85012 Contact: Peter Reaven, MD 602-277-5551 ext 6875 Peter.Reaven@va.gov United States, California VA Loma Linda Healthcare System, Loma Linda, CA Not yet recruiting Loma Linda, California, United States, 92357 Contact: Alan Jacobson, MD 909-583-6050 alan.jacobson@va.gov VA Long Beach Healthcare System, Long Beach, CA Not yet recruiting Long Beach, California, United States, 90822 Contact: Anthony Vo, MD 562-826-3497 Anthony.Vo@va.gov VA Palo Alto Health Care System, Palo Alto, CA Not yet recruiting Palo Alto, California, United States, 94304-1290 Contact: Arlina Ahluwalia, MD 650-493-5000 ext 61755 Arlina.Ahluwalia@va.gov Contact: Paul Heidenreich, MD 6504935000 ext 64069 Paul.Heidenreich@va.gov VA Greater Los Angeles Healthcare System, West Los Angeles, CA Not yet recruiting West Los Angeles, California, United States, 90073 Contact: Tannaz Moin, MD 310-478-3711 ext 48380 Tannaz.Moin@va.gov; tmoin@mednet.ucla.edu United States, Colorado Rocky Mountain Regional VA Medical Center, Aurora, CO Not yet recruiting Aurora, Colorado, United States, 80045 Contact: Allan V Prochazka, MD MSc 303-399-8020 ext 2144 Allan.Prochazka@va.gov Study Chair: Gregory G. Schwartz, PhD MD United States, Florida North Florida/South Georgia Veterans Health System, Gainesville, FL Not yet recruiting Gainesville, Florida, United States, 32608 Contact: Carsten Schmalfuss, MD 352-376-1611 ext 6052 Carsten.Schmalfuss@va.gov Miami VA Healthcare System, Miami, FL Not yet recruiting Miami, Florida, United States, 33125 Contact: Ana Palacio, MD 305-926-3780 Ana.Palacio@va.gov United States, Oregon VA Portland Health Care System, Portland, OR Not yet recruiting Portland, Oregon, United States, 97239 Contact: North Noelck, MD 503-220-8262 ext 56426 North.Noelck2@va.gov Contact: Linda Humphrey, MD 5032208262 ext 57176 Linda.Humphrey@va.gov United States, Utah VA Salt Lake City Health Care System, Salt Lake City, UT Not yet recruiting Salt Lake City, Utah, United States, 84148 Contact: Charles Lui, MD 801-582-1565 Charles.Lui@va.gov United States, Virginia Salem VA Medical Center, Salem, VA Not yet recruiting Salem, Virginia, United States, 24153 Contact: Ali Iranmanesh, MD 540-983-1071 Ali.Iranmanesh@va.gov Collapse << |
| NCT01750567 | Relapsed Chronic Lymphocytic L... more >>eukemia Collapse << | Phase 2 | Recruiting | January 2019 | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Sami Malek, MD 734-936-5310 smalek@med.umich.edu Contact: Erlene Seymour, MD 734-647-2892 ekuizon@med.umich.edu Principal Investigator: Sami Malek, MD Sub-Investigator: Daniel Lebovic, MD Sub-Investigator: Erlene Seymour, MD Collapse << |
| NCT01357148 | - | - | Terminated(Completion of MK-04... more >>31A-235 was rendered unnecessary, as the local oversight authority accepted in its stead the results of another study [MK-0431-234].) Collapse << | - | - |
| NCT01486043 | Acute Lymphoblastic Leukemia ... more >> Hyperglycemia Insulin Resistance Diabetes Mellitus Collapse << | Not Applicable | Terminated(The research projec... more >>t was terminated due to lower than projected patient recruitment within the period of time allowed for the study.) Collapse << | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Collapse << |
| NCT01718340 | Abortion, Habitual | Phase 4 | Unknown | June 2014 | Egypt ... more >> Women's Health Hospital Recruiting Assiut, Egypt Contact: Hassan S Kamel, M D hkamelhkamel@yahoo.com Principal Investigator: Alaa mM Ismail, M D Collapse << |
| NCT03772964 | Inflammatory Response | Phase 1 Phase 2 | Not yet recruiting | September 2020 | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Not yet recruiting Pittsburgh, Pennsylvania, United States, 15209 Contact: Katherine M Reitz, MD reitzkm2@upmc.edu Contact: Brian Zuckerbraun, MD zuckerbraunbs@upmc.edu Collapse << |
| NCT02026869 | PCOS | Early Phase 1 | Unknown | February 2014 | Egypt ... more >> Women's Health Hospital Assiut, Egypt, 71516 Collapse << |
| NCT01717482 | Lung Neoplasms ... more >> Carcinoma, Non-Small-Cell Lung Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03017833 | Solid Tumor A... more >>dvanced Cancer Collapse << | Phase 1 | Recruiting | March 2023 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Clinical Research Operations CR_Study_Registration@mdanderson.org Collapse << |
| NCT01357148 | - | - | Terminated(Completion of MK-04... more >>31A-235 was rendered unnecessary, as the local oversight authority accepted in its stead the results of another study [MK-0431-234].) Collapse << | - | - |
| NCT01876992 | - | - | Terminated(All investigators/c... more >>o-investigators relocating to other institutions.) Collapse << | - | - |
| NCT01876992 | Diabetes Pre-... more >>diabetes Obesity Collapse << | Not Applicable | Terminated(All investigators/c... more >>o-investigators relocating to other institutions.) Collapse << | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21287 Collapse << |
| NCT03477162 | Thoracic Neoplasm | Early Phase 1 | Recruiting | December 31, 2018 | United States, New Hampshire ... more >> Dartmouth Hitchcock Medical Center Recruiting Lebanon, New Hampshire, United States, 03756 Contact: Kayla Fay 603-650-8537 Kayla.A.Fay@hitchcock.org Contact: Research Nurse 800-639-6918 cancer.research.nurse@dartmouth.edu Collapse << |
| NCT02086526 | Polycystic Ovary Syndrome | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03733132 | Insulin Resistance ... more >> Obesity Obesity, Abdominal Collapse << | Phase 2 | Not yet recruiting | May 31, 2020 | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03118128 | Acute Lymphoblastic Leukemia | Not Applicable | Completed | - | - |
| NCT02274948 | - | - | Completed | - | - |
| NCT02274948 | Pediatric Obesity | Phase 4 | Completed | - | Sri Lanka ... more >> Lions Club of Negombo Host Negombo, Western Province, Sri Lanka Collapse << |
| NCT02354027 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT03593707 | Healthy Volunteers | Phase 1 | Suspended(Sponsor elected to v... more >>oluntarily pause study.) Collapse << | February 12, 2019 | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT03445702 | Diabetes Mellitus, Type 2 ... more >> Metformin Adverse Reaction Collapse << | Early Phase 1 | Recruiting | December 2019 | United States, Missouri ... more >> Saint Louis University Recruiting Saint Louis, Missouri, United States, 63110 Contact: Linda Eggemeyer-Sharpe, RN 314-977-9350 linda.eggemeyersharpe@health.slu.edu Collapse << |
| NCT01873859 | Diabetes Mellitus ... more >> Lactic Acidosis Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Cardiovascular research center, Modarres hospital. Tehran, Iran, Islamic Republic of Collapse << |
| NCT02741960 | Systemic Lupus Erythematosus | Phase 4 | Active, not recruiting | March 2019 | China, Shanghai ... more >> Ethics Committee of Renji Hospital, Shanghai Jiaotong University School of Medicine Shanghai, Shanghai, China, 200000 Collapse << |
| NCT03590262 | Type 1 Diabetes Mellitus ... more >> Metformin Collapse << | Phase 4 | Recruiting | September 30, 2019 | China, Guangdong ... more >> Shenzhen People's Hospital Recruiting Shenzhen, Guangdong, China, 518020 Contact: Xiuzhen Zhang, MD 0086-755-22943422 876538754@qq.com Collapse << |
| NCT02895750 | Renal Insufficiency, Chronic | Phase 2 | Recruiting | August 30, 2020 | France ... more >> CHU Amiens Picardie Recruiting Amiens, France, 80054 Contact: Jean-Daniel LALAU, MD, PhD +33 3 22 45 58 95 lalau.jean-daniel@chu-amiens.fr Collapse << |
| NCT01747083 | Type 2 Diabetes | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT00187798 | Other Conditions That May Be A... more >> Focus of Clinical Attention Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California San Francsico San Francisco, California, United States, 94143 Collapse << |
| NCT01997775 | Non-small Cell Lung Cancer | Phase 2 | Terminated(Difficult enrollmen... more >>t) Collapse << | - | Taiwan ... more >> National Cheng-Kung Uni. Hosp. Tainan, Taiwan, 704 Collapse << |
| NCT02496741 | Glioma Cholan... more >>giocarcinoma Chondrosarcoma Collapse << | Phase 1 Phase 2 | Unknown | December 2016 | Netherlands ... more >> VU University Medical Center Not yet recruiting Amsterdam, Noord-Holland, Netherlands, 1081 HZ Contact: Myra E Van Linde, M.D. +31(20)4441412 m.vanlinde@vumc.nl Academic Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1105AZ Contact: Hanneke W Wilmink, M.D., Ph.D. +31(20)5662895 j.w.wilmink@amc.uva.nl Contact: Remco J Molenaar, M.Sc. +31(20)5668587 r.j.molenaar@amc.nl Principal Investigator: Hanneke W Wilmink, M.D., Ph.D. Leiden University Medical Center Not yet recruiting Leiden, Zuid-Holland, Netherlands, 2333 ZA Contact: Hans J Gelderblom, M.D., Ph.D. +31(71)5269111 A.J.Gelderblom@lumc.nl Collapse << |
| NCT02597400 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, New Jersey ... more >> Frontage Clinical Services Inc. Hackensack, New Jersey, United States, 07601 Collapse << |
| NCT03361878 | Diabetes Mellitus, Type 2 ... more >> Metformin Adverse Reaction Collapse << | Phase 4 | Completed | - | - |
| NCT02684578 | Age-Related Macular Degenerati... more >>on Macular Degeneration, Age-Related Dry Macular Degeneration Geographic Atrophy Collapse << | Phase 2 | Recruiting | April 2019 | United States, California ... more >> University of California, Davis Recruiting Davis, California, United States, 95616 Contact: Angela Beliveau, MPH 916-734-6814 University of California San Francisco Recruiting San Francisco, California, United States, 94143 Contact: Catherine Psaras, BA 415-206-3123 eyestudy@ucsf.edu Principal Investigator: Jay M Stewart, MD Collapse << |
| NCT02972723 | Chronic Hepatitis C Infection | Phase 4 | Completed | - | United Kingdom ... more >> Secondary care Hepatitis clinic at Nottingham University Hospital Nottingham, United Kingdom, NG7 2UH Collapse << |
| NCT02056600 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01595880 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT03722290 | Fragile X Syndrome | Phase 2 | Recruiting | September 1, 2019 | Canada, Quebec ... more >> Université de Sherbrooke Recruiting Sherbrooke, Quebec, Canada, J1H 5H3 Contact: Artuela Çaku, MD 819-346-1110 ext 71035 Artuela.s.çaku@usherbrooke.ca Collapse << |
| NCT02978547 | Resectable Pancreatic Ductal A... more >>denocarcinoma Collapse << | Phase 2 | Not yet recruiting | January 2021 | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Collapse << |
| NCT01873859 | - | - | Completed | - | - |
| NCT03355014 | Healthy Male Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT03398824 | Fanconi Anemia | Phase 2 | Recruiting | March 2021 | United States, Massachusetts ... more >> Boston Children's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Ashley E Kuniholm 617-355-6513 ashley.kuniholm@childrens.harvard.edu Contact: Jacob Cotton 617-919-4227 jacob.cotton@childrens.harvard.edu Principal Investigator: Akiko Shimamura, MD, PhD Sub-Investigator: Elissa Furutani, MD Sub-Investigator: Erica Esrick, MD Collapse << |
| NCT02670018 | Healthy | Phase 1 | Unknown | March 2016 | Korea, Republic of ... more >> Yonsei University College of Medicine Seoul, Korea, Republic of Collapse << |
| NCT01662674 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT00736385 | Fatty Liver | Phase 4 | Terminated | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00941382 | Obesity | Phase 3 | Unknown | September 2009 | Mexico ... more >> Centro Universitario de Ciencias de la Salud Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00984490 | - | - | Terminated(slow accrual) | - | - |
| NCT01459445 | Polycystic Ovary Syndrome ... more >> Endothelial Dysfunction Collapse << | Not Applicable | Unknown | January 2012 | Romania ... more >> Clinic of Endocrinology Cluj-Napoca, Cluj, Romania, 400349 Collapse << |
| NCT00984490 | Breast Cancer | Not Applicable | Terminated(slow accrual) | - | United States, Georgia ... more >> Emory University Hospital Atlanta, Georgia, United States, 30322 United States, Tennessee Vanderbilt-Ingram Cancer Center - Cool Springs Nashville, Tennessee, United States, 37064 Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << |
| NCT02306070 | HIV Infection ... more >> Hepatitis C Collapse << | Phase 2 | Withdrawn(insufficient funding... more >>) Collapse << | April 3, 2018 | Canada, Ontario ... more >> The Ottawa Hospital, General Campus Ottawa, Ontario, Canada Collapse << |
| NCT02745912 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT00736385 | - | - | Terminated | - | - |
| NCT02765347 | Diabetes Mellitus, Type 1 | Phase 4 | Completed | - | China, Guangdong ... more >> The Third Affliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT01400191 | Pharmacogenetics of Metformin | Phase 4 | Completed | - | Denmark ... more >> Clinical pharmacology, Institute og public health, University of Southern Denmark Odense, Denmark, 5000 Collapse << |
| NCT03393208 | Healthy | Phase 1 | Completed | - | China ... more >> Xuanwu Hospital Beijing, China, 100053 Collapse << |
| NCT01840007 | Metastatic Melanoma (Stage III... more >>C Non-résécable or no Surgically Curable or Stage IV With Classification AJCC) Collapse << | Phase 2 | Unknown | February 2014 | France ... more >> Bahadoran Recruiting Nice, Alpes-Maritimes, France, 06200 Contact: BAHADORAN Philipp, PhD +33492036484 bahadoran.p@chu-nice.fr Principal Investigator: BAHADORAN Philippe, Phd MORTIER Laurent Recruiting Lille, Nord, France, 59000 Contact: MORTIER Laurent, PhD +33 3.20.44.41.93 Laurent.mortier@chru-lille.fr Sub-Investigator: MORTIER Laurent, PhD Collapse << |
| NCT02438020 | Acanthosis Nigricans | Phase 4 | Unknown | October 2016 | Mexico ... more >> Hospital Central Dr.Ignacio Morones Prieto Active, not recruiting San Luis Potosi, SLP, Mexico, 78210 Collapse << |
| NCT01710553 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01205672 | Endometrial Cancer | Early Phase 1 | Completed | - | United States, Texas ... more >> Lyndon B. Johnson General Hospital Houston, Texas, United States, 77026 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01664845 | Chronic Hepatitis c ... more >> Insulin Resistance Collapse << | Phase 2 Phase 3 | Unknown | August 2014 | Taiwan ... more >> Kaohsiung Veterans General Hospital Recruiting Kaohsiung, Taiwan, 813 Contact: Wei-Lun Tsai, MD 886-7-3422121 ext 2075 tsaiwl@yahoo.com.tw Principal Investigator: Wei-Lun Tsai, MD Collapse << |
| NCT02461667 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT02084056 | - | - | Completed | - | - |
| NCT02122185 | Brenner Tumor ... more >> Malignant Ascites Malignant Pleural Effusion Ovarian Clear Cell Cystadenocarcinoma Ovarian Endometrioid Adenocarcinoma Ovarian Mixed Epithelial Carcinoma Ovarian Serous Cystadenocarcinoma Ovarian Undifferentiated Adenocarcinoma Recurrent Fallopian Tube Cancer Recurrent Ovarian Epithelial Cancer Recurrent Ovarian Germ Cell Tumor Recurrent Primary Peritoneal Cavity Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Epithelial Cancer Stage IIIA Ovarian Germ Cell Tumor Stage IIIA Primary Peritoneal Cavity Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Epithelial Cancer Stage IIIB Ovarian Germ Cell Tumor Stage IIIB Primary Peritoneal Cavity Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Epithelial Cancer Stage IIIC Ovarian Germ Cell Tumor Stage IIIC Primary Peritoneal Cavity Cancer Stage IV Fallopian Tube Cancer Stage IV Ovarian Epithelial Cancer Stage IV Ovarian Germ Cell Tumor Stage IV Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Active, not recruiting | February 25, 2020 | United States, Alabama ... more >> Mitchell Cancer Institute - University of South Alabama Mobile, Alabama, United States, 36604 United States, California City of Hope Duarte, California, United States, 91010 United States, Illinois NCH Medical Group- Northwest Community Hospital Arlington Heights, Illinois, United States, 60005 Rush University Medical Center Chicago, Illinois, United States, 60612 University of Chicago Chicago, Illinois, United States, 60637 Decatur Memorial Hospital Decatur, Illinois, United States, 62526 NorthShore University HealthSystem Evanston, Illinois, United States, 60201 Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT02084056 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.11.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01005160 | Healthy Male Volunteers | Phase 1 | Completed | - | - |
| NCT03479476 | Fragile X Syndrome ... more >> Fragile X Mental Retardation Syndrome Mental Retardation, X Linked Genetic Diseases, X-Linked Trinucleotide Repeat Expansion Fra(X) Syndrome Intellectual Disability FXS Neurobehavioral Manifestations Sex Chromosome Disorders Collapse << | Phase 2 Phase 3 | Recruiting | May 2021 | United States, California ... more >> UC Davis MIND Institute Recruiting Sacramento, California, United States, 95817 Contact: Laura A Potter, BA 916-703-0471 Lapotter@ucdavis.edu Principal Investigator: Randi J Hagerman, MD Collapse << |
| NCT01849276 | Adult Acute Megakaryoblastic L... more >>eukemia (M7) Adult Acute Minimally Differentiated Myeloid Leukemia (M0) Adult Acute Monoblastic Leukemia (M5a) Adult Acute Monocytic Leukemia (M5b) Adult Acute Myeloblastic Leukemia With Maturation (M2) Adult Acute Myeloblastic Leukemia Without Maturation (M1) Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Myelomonocytic Leukemia (M4) Adult Erythroleukemia (M6a) Adult Pure Erythroid Leukemia (M6b) Blastic Phase Chronic Myelogenous Leukemia Recurrent Adult Acute Myeloid Leukemia Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Terminated(Slow accrual) | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT01247870 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> North Tees and Hartlepool NHS Trust Hartlepool, Cleveland, United Kingdom, TS24 9AH University Hospitals of Morecambe Bay NHS Trust Lancaster, Cumbria, United Kingdom, LA9 7RG East Sussex Healthcare NHS Trust Hastings, East Sussex, United Kingdom, TN37 7PT Blackpool Teaching Hospitals NHS Trust Blackpool, Lancashire, United Kingdom, FY3 8NR Lancashire Teaching Hospitals NHS Trust Preston, Lancashire, United Kingdom, PR2 9HT Sherwood Forest Hospitals NHS Trust Sutton-in-Ashfield, Nottinghamshire, United Kingdom, NG17 4JL Chelsea and Westminster Hospital London, United Kingdom, SW10 9NH St George's Hospital London, United Kingdom, SW17 0QT Freeman Hospital Newcastle, United Kingdom, NE7 7DN Collapse << |
| NCT01849276 | - | - | Terminated(Slow accrual) | - | - |
| NCT02773927 | Metabolic Syndrome | Phase 3 | Completed | - | Mexico ... more >> Universidad de Guadalajara Guadalajara, Jalisco, México, Mexico, 44100 Collapse << |
| NCT02420652 | Recurrent Prostate Carcinoma ... more >> Stage I Prostate Cancer Stage IIA Prostate Cancer Stage IIB Prostate Cancer Stage III Prostate Cancer Collapse << | Phase 2 | Active, not recruiting | January 2019 | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT00612144 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals, Co., LTD Seoul, Korea, Republic of Collapse << |
| NCT01864096 | Prostate Cancer | Phase 3 | Recruiting | August 2021 | Canada, Manitoba ... more >> Manitoba Cancer Care Centre Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Principal Investigator: Darrel Drachenberg, MD Canada, Nova Scotia CDHA - Victoria Site Recruiting Halifax, Nova Scotia, Canada, B2H 1Y6 Principal Investigator: Ricardo Rendon, MD Canada, Ontario McMaster Institute of Urology-St .Joseph's Healthcare Recruiting Hamilton, Ontario, Canada, L8N 4A6 Principal Investigator: Bobby Shayegan, MD Centre for Appled Urologic Research, Kingston General Hospital Recruiting Kingston, Ontario, Canada, K7L 3J7 Principal Investigator: Michael Leveridge, MD London Health Sciences Centre-Victoria Hospital Recruiting London, Ontario, Canada, N6A 5W9 Principal Investigator: Jonathan Izawa, MD Ottawa Hospital Research Institute (The Ottawa Hospital) Recruiting Ottawa, Ontario, Canada, K1H 8L6 Principal Investigator: Rodney Breau, MD Sunnybrook Research Institute Recruiting Toronto, Ontario, Canada, M4N 3M5 Principal Investigator: Laurence Klotz, MD Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Principal Investigator: Neil Fleshner, MD Principal Investigator: Anthony Joshua, MD Canada, Quebec Centre L'Hopitalie de l'Universite de Montreal Recruiting Montreal, Quebec, Canada, H2L 4M1 Principal Investigator: Fred Saad, MD MUHC - Montreal General Hospital Recruiting Montreal, Quebec, Canada Principal Investigator: Simon Tanguay Centre de Recherche du CHUS Recruiting Sherbrooke, Quebec, Canada, J1J 3H5 Contact: Elsie Morneau, BSN 819-346-1110 ext 12827 emorneau.chus@ssss.gouv.qc.ca Principal Investigator: Patrick Richard, MD Canada Alberta Urology Institute Recruiting Edmonton, Canada Contact: Adrian Fairey, FRCSC, MD, MSc. Collapse << |
| NCT02546050 | Healthy | Phase 1 | Completed | - | Denmark ... more >> The Novo Nordisk Foundation Center of Basic Metabolic Research, Section for Metabolic Genetics, University of Copenhagen Copenhagen, Copenhagen Ø, Denmark, 2100 Collapse << |
| NCT01234649 | Gestational Diabetes Mellitus ... more >> Type 2 Diabetes Mellitus Metabolic Syndrome Impaired Glucose Tolerance Disorder of Glucose Regulation Collapse << | Phase 3 | Active, not recruiting | August 2019 | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70815 Collapse << |
| NCT01342744 | Metabolic Syndrome | Phase 4 | Unknown | March 2012 | Thailand ... more >> Siriraj Hospital Recruiting Bangkoknoi, Bangkok, Thailand, 10700 Contact: Suchada Indhavivadhana, M.D. 0-2419-4657 sisto@mahidol.ac.th Collapse << |
| NCT02524821 | Healthy Overweight Obese | Not Applicable | Completed | - | Canada, Quebec ... more >> Quebec City, Quebec, Canada Collapse << |
| NCT03416127 | Diabetes Mellitus, Type 2 | Phase 2 | Not yet recruiting | May 1, 2019 | Mexico ... more >> Intstituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Not yet recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: MANUEL GONZALEZ, PhD +523310585200 ext 34212 uiec@prodigy.net.mx Contact: KARINA PEREZ RUBIO, PhD +523310585200 ext 34212 karina2410@hotmail.com Collapse << |
| NCT01666665 | Insulin Resistance ... more >> Hypermetabolism Hyperglycemia Collapse << | Phase 2 Phase 3 | Recruiting | August 2020 | United States, Texas ... more >> Shriners Hospitals for Children Recruiting Galveston, Texas, United States, 77551 Contact: Cathy Reed, BSN 409-770-6987 ca2reed@utmb.edu Contact: Deb Benjamin, MSN 409-770-6731 dbenjami@utmb.edu Principal Investigator: David N Herndon, MD Sub-Investigator: Oscar Suman, PhD Collapse << |
| NCT02752113 | Diabetes Mellitus Type 2 | Phase 3 | Recruiting | January 2019 | Germany ... more >> Clinical Research Center, Dept of Nephrology and Hypertenison, University of Erlangen/Nürnberg Recruiting Erlangen, Bavaria, Germany, 91054 Contact: Christian Ott, MD +49-9131-85 ext 36245 christian.ott@uk-erlangen.de Contact: Roland Schmieder, Prof. MD +49 9131 85 ext 36245 roland.schmieder@uk-erlangen.de Principal Investigator: Roland Schmieder, Prof. MD Sub-Investigator: Christian Ott, MD Collapse << |
| NCT01245166 | The Objectives of the Study is... more >> to Evaluate the Efficacy and Safety of Acarmet (Metformin HCl 500 mg Plus Acarbose 50 mg Tablets) Thrice Daily Versus Acarbose 50 mg Thrice Daily Over 16 Weeks in Subjects With Type 2 Diabetes Mellitus. Collapse << | Phase 3 | Unknown | December 2011 | Taiwan ... more >> Taichung Veterans General Hospital Recruiting Taichung, Taiwan, 40705 Contact: Elaine Liu, Bachelor 886-2-2778-5188 ext 335 elaine@lotuspharm.com Collapse << |
| NCT02931253 | Atrial Fibrillation | Phase 2 | Terminated(Recruitment issue E... more >>nrollment expectation not met) Collapse << | - | United States, Pennsylvania ... more >> The Guthrie Clinic Sayre, Pennsylvania, United States, 18840 Collapse << |
| NCT00723307 | Coronary Artery Disease | Phase 4 | Completed | - | United Kingdom ... more >> Glasgow Clinical Research Facility, NHS Greater Glasgow and Clyde Glasgow, United Kingdom, G11 6NT Collapse << |
| NCT01980823 | Breast Cancer ... more >> Breast Tumors Cancer of Breast Collapse << | Early Phase 1 | Recruiting | April 2018 | United States, New York ... more >> Columbia University Medical Center - Herbert Irving Cancer Center Recruiting Manhattan, New York, United States, 10032 Contact: Dan Otap 212-342-0937 do2267@cumc.columbia.edu Principal Investigator: Kevin Kalinsky, MD Collapse << |
| NCT01879293 | Left Ventriclar Mass | Phase 4 | Completed | - | China, Hubei ... more >> Wuhan General Hospital Wuhan, Hubei, China, 430070 Collapse << |
| NCT02823691 | Neuroendocrine Tumors | Early Phase 1 | Recruiting | April 2022 | Italy ... more >> NationalCIMilan Recruiting Milan, Italy, 20133 Contact: Sara Pusceddu, MD +390223903251 sara.pusceddu@istitutotumori.mi.it Collapse << |
| NCT01386671 | Type 2 Diabetes | Phase 3 | Completed | - | Mexico ... more >> Unidad Antidiabética Integral Mexico City, AA, Mexico, 06600 Paracelsus S.A. de C.V. Mexico, city, AA, Mexico, 03800 Unidad de Investigacion en Epidemiologia Clinica. UMAE Hospital de Especialidades Centro Medico Nacional Siglo XXI. Instituto Mexicano del Seguro Social Mexico City, Distrito Federal, Mexico, 06720 Instituto de terapéutica experimental y clínica (INTEC) Guadalajara, Jalisco, Mexico, 44600 Collapse << |
| NCT00865033 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02028767 | - | - | Completed | - | - |
| NCT01813929 | Type 1 Diabetes | Phase 4 | Active, not recruiting | December 2018 | United States, Colorado ... more >> University of Colorado Denver Aurora, Colorado, United States, 80045 Collapse << |
| NCT02656017 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 2 | Recruiting | December 30, 2020 | United States, Maryland ... more >> University of Maryland Medical Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Charalett Diggs, RN 410-328-0207 cdiggs@som.umaryland.edu Contact: Pushpa Dhillipkannah, MBBS 410.328.0207 pdhillipkannah@som.umaryland.edu Principal Investigator: Terry Watnick, MD Sub-Investigator: Stephen Seliger, MD, MS United States, Massachusetts Tufts Medical Center Recruiting Boston, Massachusetts, United States, 02111 Contact: Peggy Healy 617-636-8117 mhealy2@tuftsmedicalcenter.org Contact: Carly Tucker 617-636-7914 Ctucker@tuftsmedicalcenter.org Principal Investigator: Ronald Perrone, MD Sub-Investigator: Dana Miskulin, MD Collapse << |
| NCT02028767 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> 1276.24.001 Boehringer Ingelheim Investigational Site Toronto, Ontario, Canada Collapse << |
| NCT02125305 | Healthy Elderly | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul, Korea, Republic of Collapse << |
| NCT01237522 | Metformin Org... more >>anic Cation Transporter 2 Polymorphism,Single Nucleotide Collapse << | Phase 4 | Completed | - | Denmark ... more >> Institute of Public Health, Clinical Pharmacology, University of Southern Denmark Odense, Denmark, 5000 Collapse << |
| NCT02633488 | Metabolic Syndrome ... more >> Insulin Sensitivity Collapse << | Not Applicable | Completed | - | United States, Virginia ... more >> University of Virginia Charlottesville, Virginia, United States, 22903 Collapse << |
| NCT01483560 | Diabetes Mellitus, Type 1 | Phase 3 | Completed | - | Australia ... more >> Royal Melbourne Hospital Melbourne, Australia St Vincent's Hospital Melbourne, Australia Royal Prince Albert Hospital Sydney, Australia Canada St Joseph's Health Care Ontario, Canada Ottawa Hospital Riverside Campus Ottawa, Canada Denmark Steno Diabetes Centre Gentofte, Denmark Netherlands Maastricht University Medical Centre Maastricht, Netherlands United Kingdom Aberdeen Royal Infirmary Aberdeen, United Kingdom Ayr Hospital Ayr, United Kingdom, KA6 6DX University Hospitals Bristol Bristol, United Kingdom, BS2 8HW Diabetes Support Unit, Ninewells Hospital and Medical School Dundee, United Kingdom University Hospital North Durham Durham, United Kingdom Edinburgh Royal Infirmary Edinburgh, United Kingdom Edinburgh Western Infirmary Edinburgh, United Kingdom Peninsula NIHR Clinical Research Facility, Royal Devon and Exeter NHS Foundation Trust Exeter, United Kingdom Stobhill Hospital, Diabetes Clinic Glasgow, United Kingdom Gloucestershire Royal Hospital Gloucester, United Kingdom, GL1 3NN Michael White Diabetes Centre, Hull Royal Infirmary Hull, United Kingdom Clinical Sciences Centre, University Hospital Liverpool, United Kingdom Clinical Investigation Unit, International Centre for Circulatory Health, Imperial College Healthcare NHS Trust London, United Kingdom Wellcome Trust Clinical Research Facility, Manchester Royal Infirmary Manchester, United Kingdom Newcastle NIHR Clinical Research Facility, Royal Victoria Hospital Newcastle, United Kingdom Diabetes Clinical Research Centre, Plymouth Plymouth, United Kingdom Salford Royal NHS Foundation Trust Salford, United Kingdom Collapse << |
| NCT01540487 | - | - | Completed | - | - |
| NCT00778791 | Healthy | Not Applicable | Completed | - | United States, Missouri ... more >> Gateway medical research St. Charles, Missouri, United States, 63301 Collapse << |
| NCT01540487 | Healthy | Phase 1 | Completed | - | China ... more >> 1288.5.86001 Boehringer Ingelheim Investigational Site Beijing, China Collapse << |
| NCT01756105 | Gestational Diabetes ... more >> Metformin Treatment Oxidative Stress Collapse << | Phase 2 | Terminated | - | France ... more >> Hôpitaux Civils de Colmar Colmar, France, 68024 CHU Dijon, Dijon, France, 21030 Centre hospitalier de Mulhouse Mulhouse, France, 68070 CHU de Reims Reims, France, 51092 Hôpitaux Universitaires de Strasbourg Strasbourg, France, 67091 Collapse << |
| NCT03507413 | Abdominal Aortic Aneurysm | Phase 2 Phase 3 | Not yet recruiting | November 15, 2022 | Austria ... more >> Medical University of Vienna, Department of Surgery, Division of Vascular Surgery Not yet recruiting Vienna, Austria, 1090 Contact: Wolf Eilenberg, MD +43140400 ext 56200 wolf.eilenberg@meduniwien.ac.at Collapse << |
| NCT02523898 | Polycystic Ovary Syndrome ... more >> Hyperandrogenism Collapse << | Phase 2 | Enrolling by invitation | August 2020 | - |
| NCT03525028 | Uveitis Metfo... more >>rmin Glucocorticoid Collapse << | Not Applicable | Not yet recruiting | January 1, 2023 | China, Tianjin ... more >> Tianjin Medical University Eye Hospital Recruiting Tianjin, Tianjin, China, 300000 Collapse << |
| NCT01780051 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Korea, Republic of ... more >> Chounbuk National University Hospital Jeonju, Jeonbuk, Korea, Republic of Collapse << |
| NCT02644954 | Psoriasis | Phase 3 | Unknown | - | - |
| NCT02360618 | Bladder Cancer | Phase 2 | Unknown | January 2018 | Canada, Ontario ... more >> London Health Sciences Center; Victoria Hospital Recruiting London, Ontario, Canada, N6A 5W9 Contact: Jonathan Izawa, MD Collapse << |
| NCT02672488 | Hepatocellular Carcinoma | Phase 2 | Unknown | December 2018 | China ... more >> Tianjin Medical University Cancer Institute and Hospital Recruiting Tianjin, China, 300060 Contact: TI ZHANG, MD +862223340123 ext 3092 zhangti@tjmuch.com Contact: Huikai Li, MD +862223340123 ext 3091 lihuikai@tjmuch.com Collapse << |
| NCT03229057 | PCOS | Phase 3 | Recruiting | April 30, 2022 | United States, Pennsylvania ... more >> University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Anuja Dokras Collapse << |
| NCT02810132 | Heart Failure, Systolic | Phase 2 | Completed | - | Denmark ... more >> Aarhus University Hospital, Department of Cardiology, Palle Juul-Jensens Boulevard 99 Aarhus N, Denmark, 8200 Collapse << |
| NCT02073227 | Healthy Volunteers | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01460797 | Obesity | Not Applicable | Completed | - | Brazil ... more >> School of Health Sciences, University of Brasília Brasília, DF, Brazil, 70910-900 Collapse << |
| NCT02571608 | Diabetes Mellitus Type 2 | Phase 4 | Not yet recruiting | June 2020 | - |
| NCT03107884 | Atrophy Insul... more >>in Resistance Collapse << | Early Phase 1 | Recruiting | July 1, 2021 | United States, Utah ... more >> University of Utah Recruiting Salt Lake City, Utah, United States, 84108 Contact: Amy Rogers 801-585-5961 amy.rogers@hsc.utah.edu Collapse << |
| NCT03137186 | Prostate Cancer | Phase 2 | Recruiting | January 2020 | Egypt ... more >> Mansoura university Recruiting Mansourah, Egypt, 35516 Contact: Reham Alghandour 00201002682875 Rema-200@hotmail.com Contact: Ahmed Elshaal 00201001855490 Elshalam@hotmail.com Collapse << |
| NCT01996696 | Prostatic Neoplasm | Phase 2 | Recruiting | September 2021 | Canada, Alberta ... more >> Cross Cancer Institute Recruiting Edmonton, Alberta, Canada, T6G 1Z2 Contact: Nawaid Usmani, MD 780-432-8518 Nawaid.usmani@albertahealthservices.ca Canada, Manitoba Prairie Mountain Health - Western Manitoba Cancer Centre Not yet recruiting Brandon, Manitoba, Canada, R7A2B3 Contact: Joelle Dumontier 204-578-2108 jdumontier@pmh-mb.ca Contact: William Hunter, MD 204-578-2208 whunter@cancercare.mb.ca Principal Investigator: William Hunter, MD CancerCare Manitoba Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Contact: Julian Kim, MD 204-787-4760 jkim7@cancercare.mb.ca Collapse << |
| NCT02064881 | Type 2 Diabetes | Phase 2 Phase 3 | Recruiting | August 2020 | Mexico ... more >> Instituto Nacional de Ciencias Médicas Y Nutrición Salvador Zubirán Recruiting México, Distrito Federal, Mexico, 14000 Contact: Francisco Gómez, Doctor 55133891 gomezperezfco@gmail.com Contact: Paloma Almeda, Doctor 56554523 palomaalmeda@yahoo.com Principal Investigator: Francisco J Gómez, Doctor Collapse << |
| NCT03452306 | Bioequivalence | Phase 1 | Completed | - | Russian Federation ... more >> Yarosslavl Clinical Hospital #3 Yaroslavl, Russian Federation, 150007 Collapse << |
| NCT01154348 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Dennis Ruff, MD San Antonio, Texas, United States, 78209 Collapse << |
| NCT03031821 | Prostate Cancer ... more >> Metabolic Syndrome Collapse << | Phase 3 | Not yet recruiting | June 1, 2023 | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Cancer Centre Not yet recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Bernie Eigl, MD 604-877-6000 bernie.eigl@bccancer.bc.ca Principal Investigator: Bernie Eigl, MD Collapse << |
| NCT01340300 | Colorectal Cancer ... more >> Breast Cancer Collapse << | Phase 2 | Completed | - | United States, Connecticut ... more >> Yale School of Medicine New Haven, Connecticut, United States, 06520 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT03458208 | Bioequivalence | Phase 1 | Completed | - | Russian Federation ... more >> Yarosslavl Clinical Hospital #3 Yaroslavl, Russian Federation, 150007 Collapse << |
| NCT01579812 | - | - | Completed | - | - |
| NCT01579812 | Ovarian, Fallopian Tube, and P... more >>rimary Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01340300 | - | - | Completed | - | - |
| NCT01855763 | Gestational Diabetes ... more >> Type 2 Diabetes Pregnancy Collapse << | Phase 2 Phase 3 | Unknown | December 2013 | - |
| NCT02472353 | Breast Cancer ... more >> Breast Tumors Collapse << | Phase 2 | Completed | - | United States, South Dakota ... more >> Avera Cancer Institute Sioux Falls, South Dakota, United States, 57105 Collapse << |
| NCT02319200 | Hepatocellular Carcinoma ... more >> Hepatitis C, Chronic Cirrhosis Collapse << | Phase 3 | Terminated(Decision of investi... more >>gator) Collapse << | - | France ... more >> Roulot Dominique Bobigny, France, 93009 Collapse << |
| NCT02325401 | Head and Neck Cancer | Phase 1 | Active, not recruiting | March 2021 | United States, Ohio ... more >> University of Cincinnati Cancer Institute Cincinnati, Ohio, United States, 45267 Collapse << |
| NCT02071368 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT00588172 | Type 2 Diabetes | Phase 4 | Withdrawn(No funding) | June 2011 | - |
| NCT01816659 | Colorectal Carcinoma | Phase 1 | Terminated(Slow Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02274090 | Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT01654640 | Schizophrenia | Phase 4 | Terminated(Failure to recruit ... more >>the planned number of participants) Collapse << | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT01055691 | Healthy Volunteers | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT03310749 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Mexico ... more >> Unidad de Farmacología Clínica de la Facultad de Medicina de la Universidad Nacional Autónoma de México Nezahualcóyotl, Estado de México, Mexico, 57740 Collapse << |
| NCT03583385 | Healthy | Phase 1 | Completed | - | Indonesia ... more >> PT Equilab International Jakarta, Indonesia, 12430 Collapse << |
| NCT01394887 | Glucose Intolerance ... more >> Inflammation Diabetes Collapse << | Phase 2 Phase 3 | Completed | - | Mexico ... more >> Endocrine Outpatient Clinic of the Hospital de Pediatria del CMN "Siglo XXI" Mexico City, DF, Mexico, 06720 Collapse << |
| NCT03467971 | Healthy | Phase 1 | Completed | - | Mexico ... more >> CECYPE Mexico City, Mexico Collapse << |
| NCT02019979 | Non-small Cell Lung Cancer Sta... more >>ge IIIB/IV Non-small Cell Lung Cancer Metastatic Nonsquamous Nonsmall Cell Neoplasm of Lung Collapse << | Phase 2 | Terminated(PI left the institu... more >>tion) Collapse << | - | United States, New York ... more >> Beth Israel Medical Center New York, New York, United States, 10003 Beth Israel Comprehensive Cancer Center New York, New York, United States, 10011 St.Luke's-Roosevelt Hospital Center New York, New York, United States, 10019 Mount Sinai Ichan School of Medicine New York, New York, United States, 10029 Collapse << |
| NCT01711320 | Healthy | Phase 1 | Completed | - | China, Shaanxi ... more >> Department of pharmacy, Xijing Hospital, Fourth Military Medical University Xi'an, Shaanxi, China Collapse << |
| NCT02019979 | - | - | Terminated(PI left the institu... more >>tion) Collapse << | - | - |
| NCT01273584 | Pregnancy Complications ... more >> Obesity Collapse << | Phase 2 Phase 3 | Completed | - | United Kingdom ... more >> Medway Hospital NHS Trust Gillingham, Kent, United Kingdom, ME7 5NY Epsom and St Helier University Hospitals NHS Trust Carshalton, Surrey, United Kingdom, SM5 1AA Kings College, London London, United Kingdom, SE5 8RX Collapse << |
| NCT02005419 | Stage IA Pancreatic Adenocarci... more >>noma Stage IB Pancreatic Adenocarcinoma Stage IIA Pancreatic Adenocarcinoma Stage IIB Pancreatic Adenocarcinoma Collapse << | Phase 2 | Unknown | June 2017 | China ... more >> Department of Pancreatic and Hepatobiliary Surgery, Fudan University Shanghai Cancer Center; Pancreatic Cancer Institute, Fudan University Recruiting Shanghai, China, 200032 Contact: Xianjun Yu, M.D., Ph.D. +86-21-6417-5590 yuxianjun88@hotmail.com Contact: Liang Liu, M.D., Ph.D. +86-21-6417-5590 liuliang.zlhospital@gmail.com Principal Investigator: Xianjun Yu, M.D., Ph.D. Collapse << |
| NCT01438723 | Cardiovascular Disease ... more >> Ischemic Heart Disease Collapse << | Phase 4 | Completed | - | Netherlands ... more >> RUNMC Nijmegen, Gelderland, Netherlands Collapse << |
| NCT01573949 | Diabetes Hear... more >>t Failure Collapse << | Not Applicable | Completed | - | United States, California ... more >> Ahmanson-UCLA Cardiomyopathy Center Los Angeles, California, United States, 90095 Collapse << |
| NCT00678080 | Pregnancy Complications | Not Applicable | Completed | - | United States, Texas ... more >> Valley Baptist Hospital Brownsville, Texas, United States, 78520 Lyndon B Johnson Hospital Houston, Texas, United States, 77026 Memorial Hermann Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT01929512 | Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT02949700 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 1 Phase 2 | Recruiting | December 2021 | United States, Texas ... more >> Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Michael E. DeBakey Veterans Affairs Medical Center Recruiting Houston, Texas, United States, 77030 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Harris Health System - Smith Clinic Recruiting Houston, Texas, United States, 77054 Contact: Vlad Sandulache, MD, PhD 713-798-7218 vlad.sandulache@bcm.edu Collapse << |
| NCT00678080 | - | - | Completed | - | - |
| NCT02505516 | Bladder Cancer ... more >> Diabetes Collapse << | Early Phase 1 | Completed | - | Ghana ... more >> Universty of Capecoast Cape Coast, Ghana Collapse << |
| NCT01405118 | Rheumatoid Arthritis | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT02273050 | - | - | Completed | - | - |
| NCT02577315 | - | - | Completed | - | - |
| NCT02273050 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Changchun, China Research Site Chuangchun, China Research Site Fuzhou, China Research Site Guiyang, China Research Site Ha'er bin, China Research Site Hangzhou, China Research Site Hefei, China Research Site Jinan, China Research Site Nanchang, China Research Site Nanjing, China Research Site Shanghai, China Research Site Shijiazhuang, China Research Site Siping, China Research Site Tianjin, China Research Site Wuxi, China Research Site Yueyang, China Collapse << |
| NCT02577315 | Healthy | Phase 1 | Completed | - | Russian Federation ... more >> 1276.27.001 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation Collapse << |
| NCT01336322 | Pre-diabetes | Phase 2 | Recruiting | December 2018 | Italy ... more >> Department of Endocrinology and Metabolism, University of Pisa Recruiting Pisa, Italy, 56124 Contact: Stefano Del Prato, MD +39 050 995103 stefano.delprato@med.unipi.it Sub-Investigator: Graziano Di Cianni, MD Collapse << |
| NCT01710540 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01973933 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul, Korea, Republic of Collapse << |
| NCT02755844 | Recurrent Endometrial Cancer | Phase 1 Phase 2 | Not yet recruiting | November 2019 | France ... more >> Centre Hospitalier Lyon Sud Not yet recruiting Pierre Benite, France, 69495 Contact: Benoit YOU Contact: Marine DUPUIS Principal Investigator: Benoit YOU, MD Collapse << |
| NCT03555565 | - | - | Active, not recruiting | October 31, 2019 | Japan ... more >> Takeda Selected Site Tokyo, Japan Collapse << |
| NCT01638676 | Melanoma | Phase 1 Phase 2 | Recruiting | June 2019 | United States, Kentucky ... more >> James Graham Brown Cancer Center-University of Louisville Recruiting Louisville, Kentucky, United States, 40202 Contact: Jason A Chesney, MD PhD 502-562-3429 jason.chesney@louisville.edu Contact: Sarah Lush, RN 502-540-1537 s.lush@louisville.edu Collapse << |
| NCT00969631 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals, OB/GYN department Mansoura, Dakahlia Governorate, Egypt Collapse << |
| NCT01087983 | Advanced Cancers | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01866462 | Type 2 Diabetes | Early Phase 1 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT01240785 | - | - | Completed | - | - |
| NCT01240785 | Gestational Diabetes | Phase 4 | Completed | - | - |
| NCT02694289 | Type 2 Diabetes Mellitus | Not Applicable | Withdrawn(failure to enroll pa... more >>rticipants) Collapse << | - | - |
| NCT01831661 | Fasting | Phase 1 | Completed | - | India ... more >> Veeda Clinical Research Pvt. Ltd. Ahmedabad, Gujarat, India, 380 015 Collapse << |
| NCT02037672 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT03465345 | Prostate Cancer | Phase 1 | Withdrawn(Study was stopped to... more >> pursue a new treatment plan with a new study.) Collapse << | - | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT02515773 | Bipolar Disorder | Phase 4 | Recruiting | October 2020 | United States, Kentucky ... more >> The Children's Home of Northern Kentucky Recruiting Covington, Kentucky, United States, 41011 Contact: Joe Reiman 859-292-4122 jrieman@chnk.org United States, New Jersey Jersey Shore Medical Center Not yet recruiting Neptune City, New Jersey, United States, 07753 Contact: Nicholas Remo 732-776-3498 NRemo@meridianhealth.com United States, New York South Oaks Recruiting Amityville, New York, United States, 11701 Contact: Patricia Flaherty, LCSW-R,ACSW, 631-608-5222 pflaherty@northwell.edu SUNY Downstate/ Kings County Hospital Not yet recruiting Brooklyn, New York, United States, 11203 Contact: Cathryn A. Galanter, M.D. 718-245-2502 cathryn.galanter@downstate.edu Maimonides Not yet recruiting Brooklyn, New York, United States, 11219 Contact: George Alvarado, M.D. 718-283-8215 GAlvarado@maimonidesmed.org NYCCC Not yet recruiting Brooklyn, New York, United States, 11233 Contact: Eva Sheridan 718-470-4391 eschenk@northwell.edu Northwell Zucker Long Island Jewish Hospital Recruiting Glen Oaks, New York, United States, 11004 Contact: Arielle Carmel 718-470-8021 acarmel@northwell.edu Mount Sinai Recruiting New York, New York, United States, 10029 Contact: Julia G Jones 212-241-3692 julia.george-jones@mssm.edu LIJ Zucker Hillside Hospital Recruiting New York, New York, United States, 11004 Contact: Eva Shenck 718-470-4391 eschenk@northwell.edu Child Center of New York, Recruiting Queens, New York, United States, 11373 Contact: Isaiah Mui 718-358-8288 ext 221 isaiahmui@childcenterny.org Contact: Alla Weinstein 718.358.8288 allaweinstein@childcenterny.org NorthShore Child and Family Guidance Recruiting Roslyn Heights, New York, United States, 11577 Contact: Esther Gonzalez 516-626-1971 EGonzalez@northshorechildguidance.org StonyBrook Recruiting Stony Brook, New York, United States, 11794 Contact: Gabrielle A Carlson, MD 631-632-8840 Gabrielle.Carlson@stonybrookmedicine.edu United States, Ohio Lighthouse Youth Services Recruiting Cincinnati, Ohio, United States, 45206 Contact: Jody Harding 513-487-6778 jarding@lys.org Contact: Shannon Kiniyalocts 513-487-7706 skiniyalocts@lys.org Central Clinic Recruiting Cincinnati, Ohio, United States, 45219 Contact: Maria Piombo, Ed.D.,LPCC-S 513-558-5890 piombom@ucmail.uc.edu Children's Home Recruiting Cincinnati, Ohio, United States, 45227 Contact: Jessica Aritonovich 513-272-2800 ext 4352 jaritonovich@thechildrenshomecinti.org Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Elizabeth Picard, MS 513-803-8898 elizabeth.picard@cchmc.org Contact: Shelley Randall 513-803-2342 Shelley.randall@cchmc.org Resident Mood Medication Clinic Recruiting Cincinnati, Ohio, United States, 45229 Contact: Christy Klein, MPH 513-558-5746 kleinci@ucmail.uc.edu St. Aloysius Recruiting Cincinnati, Ohio, United States, 45237 Contact: Morrisa Gray, RN, BSN 513-482-7257 mgray@stalsorphanage.org Talbert House Recruiting Cincinnati, Ohio, United States, 45238 Contact: Sharon Stanford, MD 513-265-6090 sharon.stanford@talberthouse.com St. Joseph's Orphanage Recruiting Cincinnati, Ohio, United States, 45239 Contact: Tyann Godwin, RN 513-741-3100 ext 2166 tyann.godwin@sjokids.org Child Focus Recruiting Cincinnati, Ohio, United States, 45244 Contact: Jessica Bowlin JBowlin@child-focus.org NECCO Recruiting Cincinnati, Ohio, United States, 45246 Contact: Jessica Parks, MSM, LISW-S 513-771-9600 jparks@necco.org University Hospital Medical Center Cleveland Recruiting Cleveland, Ohio, United States, 44106 Contact: Elizabeth Deyling Elizabeth.Deyling@UHhospitals.org Nationwide Children's Hospital Columbus Recruiting Columbus, Ohio, United States, 43205 Contact: Raymond Troy raymond.troy@nationwidechildrens.org Ohio State University Recruiting Columbus, Ohio, United States, 43210 Contact: Ghada Lteif, MD 614-293-8237 ghada.lteif@osumc.edu South Community Recruiting Dayton, Ohio, United States Contact: Mary Day, RN 937-245-6121 mday@southcommunity.com Butler Behavioral Health Services Recruiting Hamilton, Ohio, United States, 45011 Contact: Randy Allman, LISW-S 513-881-7189 ext 3134 Rallman@bbhs.org TCN Family Solutions Recruiting Xenia, Ohio, United States, 45385 Contact: Carmel Flores 937-427-3837 cflores@tcn.org United States, Texas Seton Family of Hospitals Not yet recruiting Austin, Texas, United States, 78712 Contact: Ashleigh Smith AJSmith2@seton.org Collapse << |
| NCT03693560 | Coronary Artery Disease ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Active, not recruiting | February 2019 | Egypt ... more >> Faculty of Pharmacy - Damanhour University. Damanhūr, Egypt Collapse << |
| NCT02220218 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT03054519 | Peripheral Artery Disease | Phase 3 | Recruiting | September 30, 2023 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu United States, Louisiana Tulane University Recruiting New Orleans, Louisiana, United States, 70112 Contact: Lydia Bazzano, MD 504-842-4747 lbazzano@tulane.edu Contact: Shannon Terry 504-988-7249 sterry3@tulane.edu Ochsner Medical Center Recruiting New Orleans, Louisiana, United States, 70121 Contact: Lydia Bazzano, MD 504-842-4747 lbazzano@tulane.edu Contact: Grace Gartel 504-897-5844 ggartel@tulane.edu Collapse << |
| NCT00728351 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00660907 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02221180 | Bioequivalence | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT02844803 | Diabetes Mellitus | Early Phase 1 | Unknown | - | Korea, Republic of ... more >> Dongguk University Ilsan Hospital Recruiting Goyang, Gyeonggi, Korea, Republic of, 410-773 Contact: Namyi Gu, MD, PhD +82-31-961-8440 namyi.gu@gmail.com Collapse << |
| NCT03331861 | Heart Failure | Phase 2 | Not yet recruiting | December 2020 | - |
| NCT03686722 | Diabetes Mellitus, Type 2 ... more >> Hepatitis C Drug Interactions Collapse << | Phase 1 | Completed | - | Egypt ... more >> Drug research centre Cairo, Egypt Collapse << |
| NCT00930579 | Breast Cancer | Phase 2 | Unknown | March 2014 | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT01911468 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02778776 | Metabolic Syndrome | Phase 3 | Completed | - | Mexico ... more >> Universidad de Guadalajara Guadalajara, Jalisco, México, Mexico, 44100 Collapse << |
| NCT00820573 | Diabetes | Phase 4 | Completed | - | United States, Texas ... more >> Texas Diabetes Institute San Antonio, Texas, United States, 78207 Collapse << |
| NCT00856284 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02325245 | Pre-frail Elderly | Phase 3 | Unknown | March 2017 | Indonesia ... more >> Cipto Mangunkusumo General Hospital Recruiting Central Jakarta, Jakarta, Indonesia, 10430 Contact: Purwita Laksmi, MD +62-21-3918301 pwlaksmi@yahoo.com Collapse << |
| NCT00820573 | - | - | Completed | - | - |
| NCT02548741 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | August 2017 | - |
| NCT02745886 | Overweight Subjects ... more >> Metformin Aging Collapse << | Phase 4 | Unknown | December 2016 | China, Hubei ... more >> Zhang Jiajia Recruiting Wuhan, Hubei, China, 430070 Contact: Guangda Xiang, MD, PhD 02750772981 Guangda64@hotmail.com Collapse << |
| NCT00332488 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00856284 | - | - | Completed | - | - |
| NCT02394652 | Uterine Cervical Neoplasms ... more >> Squamous Cell Carcinoma Adenocarcinoma Carcinoma, Adenosquamous Collapse << | Phase 2 | Recruiting | December 2019 | Canada, Alberta ... more >> Tom Baker Cancer Centre Not yet recruiting Calgary, Alberta, Canada, T2N 4N2 Contact: Corinne Doll, M.D. 403-521-3685 Cross Cancer Institute Not yet recruiting Edmonton, Alberta, Canada, T6G 1Z2 Contact: Ericka Wiebe, M.D. 480-432-8755 Canada, Ontario Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Kathy Han, M.D., M.Sc. 416-946-2919 Canada, Quebec Hôpital Maisonneuve-Rosemont Not yet recruiting Montréal, Quebec, Canada, H1T 2M4 Contact: Eve-Lyne Marchand, M.D., Ph.D. 514-252-3425 Centre Hospitalier De L'Université de Montréal Not yet recruiting Montréal, Quebec, Canada, H2L 4M1 Contact: Maroie Barkati, M.D. 514-890-8000 ext 28820 Collapse << |
| NCT00332488 | - | - | Completed | - | - |
| NCT03451006 | Aging Inflamm... more >>ation Frailty Collapse << | Phase 2 | Recruiting | April 30, 2020 | United States, Minnesota ... more >> Mayo Clinic in Rochester Recruiting Rochester, Minnesota, United States, 55905 Contact: Mandeep Singh, MD, MPH 507-255-5891 singh.mandeep@mayo.edu Contact: James Kirkland, MD, PhD 5072933346 kirkland.james@mayo.edu Collapse << |
| NCT02990728 | Endometrial Cancer | Phase 2 | Enrolling by invitation | March 2020 | - |
| NCT00660907 | - | - | Completed | - | - |
| NCT03467945 | Healthy | Phase 1 | Completed | - | Germany ... more >> Please Contact the Merck KGaA Communication Center Darmstadt, Germany, 64293 Collapse << |
| NCT01765023 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Pusan Paik Hospital Busan, Korea, Republic of, 614-735 Collapse << |
| NCT03486626 | Polycystic Ovary Syndrome ... more >> Anovulatory Collapse << | Phase 4 | Active, not recruiting | October 2018 | Egypt ... more >> Ain Shams University Maternity Hospital Cairo, Egypt, 1156 Collapse << |
| NCT01541956 | Type 2 Diabetes | Phase 4 | Completed | - | China ... more >> Novartis Investigative Site Beijing, China, 100028 Collapse << |
| NCT01954732 | Stage IA Pancreatic Cancer ... more >> Stage IB Pancreatic Cancer Stage IIA Pancreatic Cancer Stage IIB Pancreatic Cancer Collapse << | Phase 1 | Withdrawn(Slow accrual) | - | - |
| NCT00835861 | - | - | Completed | - | - |
| NCT01796028 | Prostatic Neoplasms | Phase 2 | Completed | - | France ... more >> Centre ANTOINE LACASSAGNE Nice, France, 06189 Collapse << |
| NCT03004612 | Prediabetic State ... more >> Insulin Resistance Collapse << | Phase 4 | Active, not recruiting | August 2018 | Mexico ... more >> Universidad de Guanajuato León, Guanajuato, Mexico, 37670 Collapse << |
| NCT03757923 | Menstrual Irregularities ... more >> Hyperandrogenism Collapse << | Phase 1 Phase 2 | Recruiting | January 2019 | Pakistan ... more >> Dow University Hospital OJHA Recruiting Karachi, Sindh, Pakistan, 75270 Collapse << |
| NCT03192293 | Breast Cancer | Phase 2 | Recruiting | January 20, 2019 | Singapore ... more >> National University Hospital Singapore Recruiting Singapore, Singapore, 119228 Collapse << |
| NCT02794792 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Russian Federation ... more >> Site RU70011 Moscow, Russian Federation, 117036 Site RU70005 Moscow, Russian Federation, 119034 Site RU70003 Moscow, Russian Federation, 121374 Site RU70009 Moscow, Russian Federation, 125315 Site RU70010 Nizhniy Novgorod, Russian Federation, 603018 Site RU70006 Samara, Russian Federation, 443067 Site RU70004 Saratov, Russian Federation, 410012 Site RU70008 St. Petersburg, Russian Federation, 191119 Site RU70014 St. Petersburg, Russian Federation, 194354 Site RU70007 St. Petersburg, Russian Federation, 197022 Site RU70002 St. Petersburg, Russian Federation, 197706 Site RU70015 Volgograd, Russian Federation, 400001 Site RU70001 Yaroslavl, Russian Federation, 150003 Site RU70013 Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT01528046 | Solid Tumors ... more >>Primary Brain Tumors Collapse << | Phase 1 | Recruiting | December 2020 | United States, Connecticut ... more >> Connecticut Children's Medical Center Recruiting Hartford, Connecticut, United States, 06106 Contact: Robin Arens 860-545-9637 Rarens@ccmckids.org Principal Investigator: Michael Isakoff, M.D. Sub-Investigator: Donna Boruchov, M.D. Sub-Investigator: Kerry Moss, M.D. Sub-Investigator: Andrea Orsey, M.D. Sub-Investigator: Nehal Parikh, M.D. Sub-Investigator: Arnold Altman, M.D. United States, Delaware Nemours/Alfred I. duPont Hospital for Children, Delaware Recruiting Wilmington, Delaware, United States, 19803 Contact: Debra J. Bertz 302-651-5757 debra.bertz@nemours.org Principal Investigator: Edward A. Kolb, M.D. Sub-Investigator: Andrew Walter, M.D. Sub-Investigator: Jonathan Powell, M.D. Sub-Investigator: Emi Caywood, M.D. Sub-Investigator: Robin Miller, M.D. Sub-Investigator: Gregory Griffin, M.D. United States, Florida University of Florida Terminated Gainesville, Florida, United States, 32611 Nemours Children's Clinic Recruiting Jacksonville, Florida, United States, 32207 Contact: Ingrid Ingram 904-697-3985 iingram@nemours.org Principal Investigator: Scott Bradfield, M.D. Sub-Investigator: Eric Sandler, M.D. Sub-Investigator: Michael Joyce, M.D. Sub-Investigator: Cynthia Gauger, M.D. Sub-Investigator: Manisha Bansal, M.D. Sub-Investigator: Paul Pitel, M.D. Holtz Children's Hospital at the University of Miami Recruiting Miami, Florida, United States, 33124 Contact: Myriam Zayas 305-243-7846 MZayas2@med.miami.edu Principal Investigator: John Goldberg, M.D. Sub-Investigator: Cristina Fernandes, M.D. Sub-Investigator: Joanna Davis, M.D. Sub-Investigator: Antonello Podda, M.D. Sub-Investigator: Martin Andreansky, M.D. Sub-Investigator: Julio Barredo, M.D. Sub-Investigator: Ofelia Alvarez, M.D. Johns Hopkins All Children's Hospital Recruiting Saint Petersburg, Florida, United States, 33701 Contact: Ashley Repp, RN 727-767-4784 Ashley.Repp@allkids.org Contact: Frances Hamblin 727-767-2423 frances.hamblin@allkids.org Principal Investigator: Damon Reed, M.D. Sub-Investigator: Irmel Ayala, M.D. Sub-Investigator: Gregory Hale, M.D. Sub-Investigator: Nanette Grana, M.D. Sub-Investigator: Stacie Stapleton, M.D. Sub-Investigator: Kelly Sawczyn, M.D. Sub-Investigator: Jennifer Mayer, M.D. Sub-Investigator: Jody Kerr, M.D. Tampa General Hospital Recruiting Tampa, Florida, United States, 33606 Contact: Denise Fife 813-844-7829 dafife@tgh.org Principal Investigator: Cameron Tebbi, M.D. H. Lee Moffitt Cancer Center and Research Institute Recruiting Tampa, Florida, United States, 33612 Contact: Tiffany Smith 813-745-6250 tiffany.smith@moffitt.org Principal Investigator: Damon Reed, M.D. United States, Kentucky University of Kentucky Recruiting Lexington, Kentucky, United States, 40536 Contact: Tammy Taylor 859-323-6975 tammy.taylor@uky.edu Principal Investigator: Lars Wagner, M.D. United States, Maryland Johns Hopkins Sidney Kimmel Comprehensive Cancer Center Recruiting Baltimore, Maryland, United States, 21231 Contact: David Loeb, M.D. 410-502-7247 loebda@jhmi.edu Principal Investigator: David Loeb, M.D. United States, New York The Children's Hospital at Montefiore Recruiting Bronx, New York, United States, 10467 Contact: Noam Zeffren 718-741-2356 nzeffren@montefiore.org Principal Investigator: Jonathan Gill, M.D. United States, North Carolina University of North Carolina at Chapel Hill Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Sharon Guerry 919-966-1178 Contact: Patrick Thompson, M.D 919-966-1178 patom@email.unc.edu Principal Investigator: Patrick Thompson, M.D. United States, Ohio Nationwide Children's Hospital Recruiting Columbus, Ohio, United States, 43205 Contact: Amy Yekisa 614-722-6570 Amy.Yekisa@nationwidechildrens.org Principal Investigator: Bhuvana Setty, M.D. United States, Utah Primary Children's Medical Center/Utah Recruiting Salt Lake City, Utah, United States, 84113 Contact: Melissa Bolton 801-213-3909 melissa.bolton@hsc.utah.edu Principal Investigator: Holly Spraker-Perlman, M.D. Sub-Investigator: Richard Lemons, M.D., Ph.D. Sub-Investigator: Jennifer Wright, M.D. Collapse << |
| NCT01192139 | Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT02417090 | - | - | Enrolling by invitation | December 2019 | - |
| NCT01215032 | Prostate Cancer | Phase 2 | Terminated(slow accrual, compe... more >>ting clinical trials) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00835861 | Non Insulin Dependent Diabetes... more >> Pregnancy Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> University of North Carolina Hospitals Obstetric Clinics Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01215032 | - | - | Terminated(slow accrual, compe... more >>ting clinical trials) Collapse << | - | - |
| NCT02438540 | Insulin Sensitivity ... more >> Obesity Collapse << | Not Applicable | Completed | - | - |
| NCT01529593 | Advanced Cancers | Phase 1 | Recruiting | March 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Collapse << |
| NCT01228240 | Diabetes Mellitus, Type 2 AND ... more >>Metformin Collapse << | Not Applicable | Completed | - | - |
| NCT03713801 | Aging Vaccine... more >> Response Impaired Collapse << | Phase 1 | Recruiting | December 31, 2019 | United States, Texas ... more >> UT Health San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Grace Lee, PhD 210-450-8097 leeg3@uthscsa.edu Collapse << |
| NCT01719614 | Healthy Participants | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT01192139 | - | - | Completed | - | - |
| NCT01122316 | Heart Failure ... more >> Diabetes Collapse << | Not Applicable | Completed | - | United States, California ... more >> Ahmanson-UCLA Cardiomyopathy Center Los Angeles, California, United States, 90095-7368 Collapse << |
| NCT02438540 | - | - | Completed | - | - |
| NCT01604213 | Type 2 Diabetes Mellitus ... more >> Ischemic Heart Disease Collapse << | Phase 4 | Completed | - | Israel ... more >> Sheba Medical Center, Cardiac Rehabilitation Institute Tel Hashomer, Israel, 52621 Collapse << |
| NCT01941953 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | Brazil ... more >> Instituto Do Cancer Do Estado de São Paulo Sao Paulo, Brazil, 01246-000 Collapse << |
| NCT01929863 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT01929863 | - | - | Completed | - | - |
| NCT01573377 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Not Applicable | Completed | - | China, Chongqing ... more >> The First Affiliated Hospital of Chongqing Medical University Chongqing, Chongqing, China, 400016 Collapse << |
| NCT03051451 | Overweight and Obesity | Phase 2 | Suspended(Business decision) | November 2018 | Mexico ... more >> Laboratorios SIlanes S.A. de C.V, Mexico Distrito Federal, Mexico City, Mexico, 03100 Collapse << |
| NCT02981329 | Sickle Cell Anemia ... more >> Sickle Cell Disease Hemoglobin Disorder Hemoglobin Disease; Sickle-Cell, Thalassemia Collapse << | Early Phase 1 | Recruiting | November 2022 | United States, Texas ... more >> Texas Children's Hospital Recruiting Houston, Texas, United States, 77030 Contact: Vivien Sheehan, MD 832-824-4459 vxsheeha@texaschildrens.org Contact: Bodgan Dinu brdinu@texaschildrens.org The University of Texas Health Science Center at Houston Recruiting Houston, Texas, United States, 77030 Contact: Harinder Juneja, MD Collapse << |
| NCT01677897 | Metastatic Prostate Cancer | Phase 2 | Unknown | April 2015 | Switzerland ... more >> Kantonsspital Graubünden Recruiting Chur, Graubünden, Switzerland, 7000 Principal Investigator: michael mark, md Collapse << |
| NCT01068743 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT02751307 | Metabolic Syndrome | Phase 4 | Completed | - | - |
| NCT01589445 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Bangladesh ... more >> Bangladesh Institute of Research and Rehabilitation in Diabetes, Endocrine and Metabolic Disorders (BIRDEM) Dhaka, Bangladesh, 1000 Collapse << |
| NCT01068743 | - | - | Completed | - | - |
| NCT01441869 | Bioequivalence, Log-transforme... more >>d AUCss and Cmax,ss Values for Saxagliptin and Metformin Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT02230995 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach an der Riss, Germany Collapse << |
| NCT02230995 | - | - | Completed | - | - |
| NCT01808690 | Type 1 Diabetes | Phase 3 | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado and University of Colorado Denver Health Sciences Center Aurora, Colorado, United States, 80045 Collapse << |
| NCT01905046 | Atypical Ductal Breast Hyperpl... more >>asia BRCA1 Mutation Carrier BRCA2 Mutation Carrier Ductal Breast Carcinoma in Situ Lobular Breast Carcinoma in Situ Collapse << | Phase 3 | Recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Suspended Duarte, California, United States, 91010 Los Angeles County-USC Medical Center Suspended Los Angeles, California, United States, 90033 USC / Norris Comprehensive Cancer Center Suspended Los Angeles, California, United States, 90033 City of Hope South Pasadena Suspended South Pasadena, California, United States, 91030 United States, Kansas University of Kansas Cancer Center Recruiting Kansas City, Kansas, United States, 66160 Contact: Site Public Contact 913-945-7552 ctnursenav@kumc.edu Principal Investigator: Carol J. Fabian United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Site Public Contact 855-776-0015 Principal Investigator: Sandhya Pruthi United States, New York Roswell Park Cancer Institute Suspended Buffalo, New York, United States, 14263 Mount Sinai Hospital Recruiting New York, New York, United States, 10029 Contact: Site Public Contact 212-824-7309 CCTO@mssm.edu Principal Investigator: Sarah P. Cate Columbia University/Herbert Irving Cancer Center Suspended New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center Suspended Durham, North Carolina, United States, 27710 United States, Ohio The Christ Hospital Recruiting Cincinnati, Ohio, United States, 45219 Contact: Site Public Contact 513-585-0844 Principal Investigator: Jennifer B. Manders Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Lisa D. Yee United States, Oklahoma University of Oklahoma Health Sciences Center Recruiting Oklahoma City, Oklahoma, United States, 73104 Contact: Site Public Contact 405-271-8777 ou-clinical-trials@ouhsc.edu Principal Investigator: William C. Dooley United States, South Carolina Greenville Health System Cancer Institute-Easley Recruiting Easley, South Carolina, United States, 29640 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Andrews Suspended Greenville, South Carolina, United States, 29601 Greenville Health System Cancer Institute-Butternut Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Faris Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Memorial Hospital Recruiting Greenville, South Carolina, United States, 29605 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Eastside Recruiting Greenville, South Carolina, United States, 29615 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Greer Recruiting Greer, South Carolina, United States, 29650 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Seneca Recruiting Seneca, South Carolina, United States, 29672 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere Greenville Health System Cancer Institute-Spartanburg Recruiting Spartanburg, South Carolina, United States, 29307 Contact: Site Public Contact 864-241-6251 kwilliams8@ghs.org Principal Investigator: Jeffrey K. Giguere United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Therese B. Bevers Doctor's Hospital of Laredo Active, not recruiting Laredo, Texas, United States, 78041 United States, Wisconsin University of Wisconsin Hospital and Clinics Recruiting Madison, Wisconsin, United States, 53792 Contact: Site Public Contact 800-622-8922 Principal Investigator: Lee G. Wilke Collapse << |
| NCT01929811 | pCR Rate BCT ... more >>Rate Safety Collapse << | Phase 2 | Recruiting | September 2020 | China, Shandong ... more >> Linyi People's Hospital Not yet recruiting Linyi, Shandong, China, 276003 Contact: Hong Liang Principal Investigator: Hong Liang China, Shanghai Ruijin Hospital, Shanghai Jiaotong University School of Medicine Recruiting Shanghai, Shanghai, China, 200025 Contact: Xiaosong Chen Principal Investigator: Kunwei Shen, Dr Collapse << |
| NCT01589445 | - | - | Completed | - | - |
| NCT01725672 | Diabetes Mellitus, Type 2 | Phase 1 | Terminated(Due to changes in E... more >>MA guidelines on modified release dosage forms (Feb 2013; EMA/CHMP/EWP/280/96). The Part B formulation would not meet the new guidelines.) Collapse << | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01775579 | Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Samgsung Seoul Hospital Seoul, Korea, Republic of Collapse << |
| NCT03271866 | Schizophrenia | Not Applicable | Recruiting | March 1, 2019 | China, Hunan ... more >> The Second Xiangya Hospital of Central South University Recruiting Changsha, Hunan, China, 410011 Contact: Tiannan Shao, M.D., Ph.D. +86 13341318057 shaostndoc@163.com Contact: Renrong Wu, M.D., Ph.D. wurenrong2013@163.com Principal Investigator: Dongyu Kang, M.D. Collapse << |
| NCT01992211 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02280057 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Gynecology Dept. Herning Hospital Herning, Denmark, 7400 Collapse << |
| NCT01310231 | Metastatic Breast Cancer | Phase 2 | Completed | - | Canada, Ontario ... more >> London Regional Cancer Program London, Ontario, Canada St. Michael's Hospital Toronto, Ontario, Canada, M5B 1N9 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Windsor Regional Cancer Centre Windsor, Ontario, Canada, N8W 2X3 Collapse << |
| NCT02145559 | Breast Neoplasms ... more >> Lung Neoplasms Cancer of Liver Lymphoma Cancer of Kidney Collapse << | Phase 1 | Active, not recruiting | July 2019 | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT01718093 | Type 1 Diabetes | Phase 4 | Completed | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT01037660 | Cardiovascular Disease | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT02084082 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.9.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT00892073 | Hypothalamic Obesity | Phase 2 | Completed | - | Canada, Ontario ... more >> The Hospital for Sick Children Toronto, Ontario, Canada Collapse << |
| NCT02121509 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.10.1 Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT01756417 | Healthy | Phase 1 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States Collapse << |
| NCT03764605 | ADPKD | Phase 3 | Not yet recruiting | January 30, 2022 | Italy ... more >> AOUC "Policlinico" Bari, Italy, 70124 AOUConsorziale Policlinico Di Bari Not yet recruiting Bari, Italy, 70124 Contact: Loreto Gesualdo +390805594040 trialnefrobari@gmail.com Contact: Giovanni Piscopo +393299664404 giopiscopo@gmail.com Collapse << |
| NCT02121509 | - | - | Completed | - | - |
| NCT00994812 | Polycystic Ovary Syndrome ... more >> Miscarriage Infertility Toxemia Gestational Diabetes Collapse << | Phase 3 | Completed | - | Finland ... more >> University Hospital Of Helsinki Helsinki, Finland University Hospital of Kuopio Kuopio, Finland University Hopsital of Oulu Oulu, Finland, 90029 University Hospital of Tampere Tampere, Finland University Hospital of Turku Turku, Finland Collapse << |
| NCT02212756 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju, Jeollabuk-do, Korea, Republic of, 561-712 Collapse << |
| NCT01523639 | Colorectal Cancer ... more >> Steatohepatitis Collapse << | Phase 2 | Terminated(Prematurely due to ... more >>slow recruitment (07/08/2013). Newly defined study end=LPLV=05/11/2013. ABCSG guaranteed completed treatment period for ethical reasons.) Collapse << | May 2014 | Austria ... more >> Medical University Graz, Oncology Graz, Styria, Austria, 8036 Medical University Innsbruck, Internal Medicine Innsbruck, Tyrol, Austria, 6020 Hospital St. Vinzenz Zams, Tyrol, Austria, 6511 Hospital BHS Ried Ried, Upper Austria, Austria, 4910 KH BHB Vienna Vienna, Austria, 1021 Med. Univ. Vienna, General Hospital Vienna Vienna, Austria, 1090 KH St. Josef KH Vienna, Austria, 1130 Collapse << |
| NCT02581137 | Erythroplakia ... more >> Hyperplasia Mild Dysplasia Moderate Dysplasia Oral Cavity Carcinoma Oral Leukoplakia Severe Dysplasia Collapse << | Phase 2 | Active, not recruiting | - | United States, California ... more >> University of California San Diego San Diego, California, United States, 92103 United States, Minnesota University of Minnesota/Masonic Cancer Center Minneapolis, Minnesota, United States, 55455 Canada, British Columbia BC Cancer Research Centre Vancouver, British Columbia, Canada, V5Z 1L3 University of British Columbia Hospital Vancouver, British Columbia, Canada, V6T 2B5 Collapse << |
| NCT03309007 | PreDiabetes A... more >>ging Collapse << | Phase 3 | Recruiting | July 31, 2018 | United States, New Mexico ... more >> University of New Mexico Clincal & Translational Science Center Recruiting Albuquerque, New Mexico, United States, 87131 Contact: Mark R Burge, MD 505-272-4658 mburge@salud.unm.edu Collapse << |
| NCT01864174 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02084082 | - | - | Completed | - | - |
| NCT02712619 | Healthy | Phase 1 | Completed | - | - |
| NCT01864174 | - | - | Completed | - | - |
| NCT01383356 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Canada, Ontario ... more >> 1288.19.1 Boehringer Ingelheim Investigational Site Toronto, Ontario, Canada Collapse << |
| NCT02266472 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT02853630 | TYpe 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | December 2018 | India ... more >> Jnana Sanjeevini Medical Centre Bangalore, Karnataka, India, 560 078 Vikas Pai Research Foundation Pune, Maharashtra, India, 411 005 Singhvi Health Centre Chennai, Tamil Nadu, India, 600 019 Dr V.Seshiah Diabetes research Institute, Dr V.Balaji Diabetes care centre Chennai, Tamil Nadu, India, 600029 Arthur Asirvatham Hospital Madurai, Tamil Nadu, India, 625 020 Ramana Maharishi Rangammal Hospital Tiruvannamalai, Tamil Nadu, India, 606 603 Collapse << |
| NCT00845936 | Drug Induced Weight Gain | Phase 4 | Unknown | May 2010 | Israel ... more >> Beer-Yaacov MHC Not yet recruiting Beer Yaacov, Israel, 70350 Contact: Pazit Leibovich, MD 972-9284065 pazit.leib@gmail.com Sub-Investigator: Pazit Leibovich, MD Collapse << |
| NCT00159536 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Norway ... more >> University hospital of Bergen Bergen, Norway Central Hospital of Northern Norway Bodø, Norway Buskerud Hospital Drammen, Norway Haugesund Hospital Haugesund, Norway Ringerike Hospital Hønefoss, Norway Elvebredden Gynekologpraksis Kristiansand, Norway Lillehammer Hospital Lillehammer, Norway Stavanger University Hospital Stavanger, Norway Departments of Obstetrics and Gynecology and Endocrinology, St. Olav's Hospital Trondheim, Norway Ålesund Hospital Ålesund, Norway Collapse << |
| NCT02372656 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT01383356 | - | - | Completed | - | - |
| NCT03651531 | Gestational Diabetes | Phase 3 | Not yet recruiting | July 2020 | - |
| NCT02266472 | - | - | Completed | - | - |
| NCT01551615 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01930864 | Colorectal Neoplasms ... more >> Adenocarcinoma Collapse << | Phase 2 | Recruiting | December 1, 2020 | Brazil ... more >> Barretos Cancer Hospital Recruiting Barretos, SP, Brazil, 14784-400 Contact: Joao Paulo SN Lima, MD 551733216600 ext 6829 joao.lima@accamargo.org.br Contact: Arinilda C Bragagnoli, MD 551733216600 ext 6829 aryscampos@yahoo.com.br Principal Investigator: Arinilda C Bragagnoli, MD State University of Campinas Not yet recruiting Campinas, São Paulo, Brazil, 1300000 Contact: Lígia T Macedo, MD ligaimed@gmail.com Principal Investigator: Ligia T Macedo, MD Collapse << |
| NCT00667498 | Pediatric Obesity ... more >> Insulin Resistance Hyperinsulinemia Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale Center for Clinical Investigation (YCCI) New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02440893 | - | - | Completed | - | - |
| NCT01463228 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT03627039 | - | - | Not yet recruiting | August 1, 2020 | - |
| NCT01901224 | Peripheral Artery Disease | Phase 4 | Terminated(study was not funde... more >>d) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01518712 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01901224 | - | - | Terminated(study was not funde... more >>d) Collapse << | - | - |
| NCT01426399 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT01778244 | Dyslipidemia | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT02312661 | Epithelial Ovarian Cancer | Phase 1 | Completed | - | Netherlands ... more >> University Mecdical Center Groningen Groningen, Netherlands, 9713 GZ Collapse << |
| NCT01280409 | - | - | Completed | - | - |
| NCT01280409 | Diabetes, Gestational | Phase 4 | Completed | - | United States, Texas ... more >> Memorial Hermann Hospital, Texas Medical Center Houston, Texas, United States, 77030 University of Texas Health Science Center at Houston, Professional Building Houston, Texas, United States, 77030 Collapse << |
| NCT01177709 | Schizophrenia ... more >> Obesity Collapse << | Not Applicable | Terminated(PI moved to differe... more >>nt institution. Decided to concentrate on different studies.) Collapse << | - | United States, New York ... more >> Manhatan Psychiatric Center New York, New York, United States, 10035 Collapse << |
| NCT01177709 | - | - | Terminated(PI moved to differe... more >>nt institution. Decided to concentrate on different studies.) Collapse << | - | - |
| NCT01882907 | - | - | Completed | - | - |
| NCT01963663 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Diabetes Clinic, Vali-Asr Hospital, Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT02986659 | Coronary Artery Disease ... more >> Mild Cognitive Impairment Obesity, Abdominal Hypertension Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> Wake Forest Health Sciences Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT01093794 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01882907 | Type 2 Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Busan Saint Mary's Medical Center Busan, Korea, Republic of Dong-AUniversity Medical Center Busan, Korea, Republic of Inje University Baik Hospital Busan, Korea, Republic of Kosin University Hospital Busan, Korea, Republic of Pusan National University Hospital Busan, Korea, Republic of Changwon Fatima Hospital Changwon, Korea, Republic of Daegu Catholic University Medical Center Daegu, Korea, Republic of Keimyung University Dongsan Medical Center Daegu, Korea, Republic of Kyungbuk National Universtiy Hospital Daegu, Korea, Republic of Chonnam National University Hospital Gwangju, Korea, Republic of Chosun University Hospital Gwangju, Korea, Republic of Chonbuk National University Hospital Jeonju, Korea, Republic of Gyeongsang National University Hospital Jinju, Korea, Republic of Masan Samsung Medical Center Masan, Korea, Republic of Ulsan University Hospital Ulsan, Korea, Republic of Collapse << |
| NCT01093794 | - | - | Completed | - | - |
| NCT02026310 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | China, Chongqing ... more >> The First Affiliated Hospital of Chongqing Medical University Chongqing,, Chongqing, China, 400016 Collapse << |
| NCT02473094 | Rectal Neoplasms ... more >> Adenocarcinoma Carcinoma Collapse << | Phase 2 | Terminated(Slow Accrual) | - | Brazil ... more >> Clinical Oncology Department - General Hospital - State University of Campinas (UNICAMP) Campinas, São Paulo, Brazil, 13083888 Collapse << |
| NCT02793505 | - | - | Completed | - | - |
| NCT02884089 | - | - | Completed | - | - |
| NCT01935804 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 2 | Completed | - | Saudi Arabia ... more >> Center of Excellence for Osteoporosis Research, King Abdulaziz University Jeddah, Makkah, Saudi Arabia, 21589 Collapse << |
| NCT01342614 | Diabetes Mellitus ... more >> Essential Hypertension Collapse << | Not Applicable | Completed | - | - |
| NCT02064374 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT01844531 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.6.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02802215 | Polycystic Ovary Syndrome ... more >> Diabetes, Gestational Collapse << | Phase 2 Phase 3 | Unknown | September 2016 | Egypt ... more >> Ain Shams maternity hospital Recruiting Cairo, Egypt Principal Investigator: Ahmed E Elbohoty, Ph.D. Collapse << |
| NCT00546741 | Healthy Male and Female Subjec... more >>ts Collapse << | Phase 1 | Completed | - | United States, New Jersey ... more >> Bristol-Myers Squibb Clinical Pharmacology Unit Hamilton, New Jersey, United States, 08690 Collapse << |
| NCT02336087 | Pancreatic Adenocarcinoma ... more >> Unresectable Pancreatic Carcinoma Collapse << | Phase 1 | Recruiting | August 2019 | United States, California ... more >> City of Hope Medical Center Recruiting Duarte, California, United States, 91010 Contact: Vincent Chung 626-471-9200 vchung@coh.org Principal Investigator: Vincent Chung City of Hope Rancho Cucamonga Recruiting Rancho Cucamonga, California, United States, 91730 Contact: Valerie Estala 626-256-4673 ext 81699 Sub-Investigator: Behnam Ebrahimi, MD City of Hope South Pasadena Recruiting South Pasadena, California, United States, 91030 Contact: Odessa Rodriguez 626-256-4673 ext 81409 orodriguez@coh.org Sub-Investigator: Christina Yeon, MD City of Hope West Covina Recruiting West Covina, California, United States, 91790 Contact: Meizi Zheng 626-256-4673 ext 81336 Sub-Investigator: Gargi Upadhyaya, MD Collapse << |
| NCT01068730 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT01844531 | - | - | Completed | - | - |
| NCT01068730 | - | - | Completed | - | - |
| NCT03029390 | Prediabetes I... more >>mpaired Fasting Glucose Impaired Glucose Tolerance Collapse << | Phase 4 | Active, not recruiting | May 2018 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02360059 | - | - | Terminated(Low Accrual) | - | - |
| NCT02360059 | Breast Cancer | Phase 2 | Terminated(Low Accrual) | - | United States, Texas ... more >> Lyndon B. Johnson General Hospital Houston, Texas, United States, 77026 Collapse << |
| NCT03655301 | Healthy Volunteers | Phase 1 | Active, not recruiting | January 18, 2019 | United States, Texas ... more >> Pharmaceutical Product Development, LLC Austin, Texas, United States, 78744 Collapse << |
| NCT01099618 | Ketosis Prone Diabetes ... more >> Diabetes Ketoacidosis Hyperglycemia Collapse << | Phase 4 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Collapse << |
| NCT00005669 | - | - | Completed | - | - |
| NCT02884089 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> Dallas Clinical Research Unit Dallas, Texas, United States, 75247 Collapse << |
| NCT00859898 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01008046 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals,OB/GYN department Mansoura, Dakahlia Governorate, Egypt Collapse << |
| NCT00859898 | - | - | Completed | - | - |
| NCT02394158 | Gestational Diabetes Mellitus | Phase 4 | Unknown | July 2017 | United Kingdom ... more >> Imperial College NHS Trust Recruiting London, United Kingdom, W2 1PG Contact: Stephen Robinson, FRCP, MD 0203 312 1253 Contact: Rochan Agha-Jaffar, BMBS, MRCP 0207 594 6140 r.agha-jaffar@imperial.ac.uk London North West Healthcare Trust Recruiting London, United Kingdom Contact: Mushtaqur Rahman, FRCP, PhD Contact: Rochan Agha-Jaffar, BMBS, MRCP 0207 594 6140 r.agha-jaffar@imperial.ac.uk Collapse << |
| NCT02696941 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> University of Oxford Oxford, United Kingdom Collapse << |
| NCT01799057 | Peripheral Arterial Disease ... more >> Intermittent Claudication Collapse << | Phase 4 | Terminated(The trial has been ... more >>terminated due to difficulties with recruitment.) Collapse << | - | Australia, Victoria ... more >> Baker IDI Heart and Diabetes Institute Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01273571 | Healthy | Phase 1 | Completed | - | - |
| NCT01196546 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Thailand ... more >> BMA Medical College and Vajira Hospital Bangkok, Thailand King Chulalongkorn Memorial Hospital Bangkok, Thailand Phamongkutklao Hospital Bangkok, Thailand Police General Hospital Bangkok, Thailand Siriraj Hospital Bangkok, Thailand Khon Kaen Hospital Khon Kaen, Thailand Fort Suranaree Hospital Nakhon Ratchasima, Thailand Maharat Nakhon Ratchasima Hospital Nakhon Ratchasima, Thailand Collapse << |
| NCT01593137 | Type 2 Diabetes | Phase 4 | Withdrawn(Lack of patients wit... more >>h the criteria established in the protocol.) Collapse << | December 2014 | Spain ... more >> Hospital Clínico San Carlos Madrid, Spain, 28040 Collapse << |
| NCT01099618 | - | - | Completed | - | - |
| NCT00918138 | Type 2 Diabetes | Phase 3 | Completed | - | United States, Arizona ... more >> Dedicated Phase I, Inc. Phoenix, Arizona, United States, 85013 United States, California Pacific Sleep Medicine Services (Avastra Clinical Trials) Redlands, California, United States, 92373 Orange County Research Center Tustin, California, United States, 92780 United States, Florida Clinical Research Of South Florida Coral Gables, Florida, United States, 33134 Palm Springs Research Institute Hialeah, Florida, United States, 33012 Healthcare Clinical Data, Inc. North Miami, Florida, United States, 33161 United States, Georgia River Birch Research Alliance, Llc Blue Ridge, Georgia, United States, 30513 United States, Michigan Jasper Clinic, Inc. Kalamazoo, Michigan, United States, 49007 United States, New York Clinilabs, Inc. New York, New York, United States, 10019 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 United States, Texas Clinical Trials Of Texas Inc. San Antonio, Texas, United States, 78229 United States, Utah Advanced Clinical Research West Jordan, Utah, United States, 84088 Argentina Local Institution Capital Federal, Buenos Aires, Argentina, 1429 Local Institution Buenos Aires, Argentina, 1425 Israel Local Institution Beer-Sheva, Israel, 84101 Local Institution Holon, Israel, 58100 Local Institution Kfar-Saba, Israel, 44281 Local Institution Zefat, Israel, 13100 Mexico Local Institution Monterrey, Nuevo Leon, Mexico, 64460 Local Institution Durango, Mexico, 34000 Local Institution Durango, Mexico, 34075 Collapse << |
| NCT01068717 | - | - | Completed | - | - |
| NCT02951260 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT01068717 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT01384279 | Obesity | Not Applicable | Completed | - | Taiwan ... more >> Beitou Armed Forces Hospital Taipei, Taiwan, 11243 Collapse << |
| NCT00918138 | - | - | Completed | - | - |
| NCT01968317 | Endometrial Atypical Hyperplas... more >>ia Endometrial Adenocarcinoma Collapse << | Phase 2 | Completed | - | China, Shanghai ... more >> Obstetrics and Gynecology Hospital, Fudan University Shanghai, Shanghai, China, 200011 Fudan University Shanghai Cancer Center Shanghai, Shanghai, China, 200032 Shanghai Changning Maternity & Infant Health Hospital Shanghai, Shanghai, China Shanghai Sixth People's Hospital Shanghai, Shanghai, China China, Zhejiang Zhejiang Cancer Hospital Hangzhou, Zhejiang, China Collapse << |
| NCT01544751 | Nonalcoholic Fatty Liver Disea... more >>se (NAFLD) Collapse << | Not Applicable | Unknown | June 2015 | Italy ... more >> Endocrinology Unit Recruiting Catanzaro, Italy, 88100 Contact: Angela Mazza, Post doc +03909613647110 angela_mazza@libero.it Contact: Barbara Fruci, Doctor +03909613647110 barbara.fruci@yahoo.it Principal Investigator: Antonino Belfiore, Professor Collapse << |
| NCT00005669 | Hyperinsulinemia ... more >> Obesity Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02250794 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | December 2015 | - |
| NCT01755494 | Type 2 Diabetes Mellitus(T2DM) | Phase 1 | Completed | - | China ... more >> Research Site Beijing, China Collapse << |
| NCT02278965 | Stage 0 Breast Carcinoma ... more >> Breast Neoplasms Stage I Breast Carcinoma Stage II Breast Carcinoma Stage III Breast Carcinoma Collapse << | Phase 1 | Active, not recruiting | October 2018 | United States, New York ... more >> Columbia University New York, New York, United States, 10032 Collapse << |
| NCT01077479 | Metabolic Syndrome ... more >> Hypercholesterolemia Collapse << | Phase 2 | Completed | - | Australia, New South Wales ... more >> Westmead Hospital Sydney, New South Wales, Australia, 2145 Collapse << |
| NCT03717701 | Pre-Eclampsia | Not Applicable | Not yet recruiting | January 1, 2021 | - |
| NCT00105066 | - | - | Completed | - | - |
| NCT02027103 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Diabetes Clinic, Vali-Asr Hospital, Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT00105066 | Obesity Hyper... more >>tension Hypercholesterolemia Hyperglycemia Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute on Aging (NIA), Harbor Hospital Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02605772 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | March 2017 | - |
| NCT01316042 | Premature Pubarche | Not Applicable | Terminated(poor recruitment at... more >> collaborating sites) Collapse << | - | United States, Indiana ... more >> Riley Hospital for Children, Indiana University School of Medicine Indianapolis, Indiana, United States, 46202 United States, Pennsylvania Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Children's Hospital of Pittsburgh at the University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT00842400 | Obesity | Phase 1 | Completed | - | United States, Arizona ... more >> MDS Pharma Services Phoenix, Arizona, United States, 85283 Collapse << |
| NCT01316042 | - | - | Terminated(poor recruitment at... more >> collaborating sites) Collapse << | - | - |
| NCT01302743 | Diabetes | Not Applicable | Terminated(lack of recruitment... more >>) Collapse << | - | United States, Nevada ... more >> Mike O'Callaghan Federal Hospital Nellis Air Force Base, Nevada, United States, 89191 Collapse << |
| NCT01302743 | - | - | Terminated(lack of recruitment... more >>) Collapse << | - | - |
| NCT03629340 | Pulmonary Hypertension ... more >> Heart Failure Collapse << | Phase 2 | Not yet recruiting | December 2021 | - |
| NCT01890122 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03200015 | Diffuse Large B Cell Lymphoma | Phase 2 | Active, not recruiting | January 27, 2019 | Mexico ... more >> Hospital Universitario Dr. José Eleuterio González Monterrey, Nuevo Leon, Mexico, 64610 Collapse << |
| NCT00969566 | Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, 463-707 Collapse << |
| NCT03490019 | Bardet-Biedl Syndrome ... more >> Visual Impairment Collapse << | Phase 2 | Not yet recruiting | August 2020 | - |
| NCT02099838 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Hubei ... more >> Tongji Hospital Wuhan, Hubei, China, 430030 Collapse << |
| NCT01890122 | - | - | Completed | - | - |
| NCT02099838 | - | - | Completed | - | - |
| NCT01070160 | - | - | Completed | - | United States, Connecticut ... more >> Yale-New Haven Hospital Women's Center New Haven, Connecticut, United States, 06520-8063 Collapse << |
| NCT01216397 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1288.6.1 Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT01216397 | - | - | Completed | - | - |
| NCT02088918 | Healthy Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Chungnam University Hospital Daejeon, Korea, Republic of Collapse << |
| NCT01300637 | Obesity Metab... more >>olic Syndrome Schizophrenia Collapse << | Not Applicable | Unknown | July 2011 | Taiwan ... more >> Taipei Medical University-WanFang Hospital Recruiting Taipei, Taiwan, 116 Contact: Chun-Hsin Chen, MD 886-2-29307930 ext 53961 chunhsin57@yahoo.com.tw Contact: Mong-Liang Lu, MD 886-2-29307930 ext 53961 mongliang@hotmail.com Collapse << |
| NCT02275845 | Gestational Diabetes Mellitus | Phase 3 | Unknown | September 2017 | Netherlands ... more >> Maasstad Ziekenhuis Recruiting Rotterdam, Netherlands Contact: Huguette Brink, Drs +31 10 291 2889 brinkH@maasstadziekenhuis.nl Contact: Wetenschapsbureau +31 10 291 3357 wetenschapsbureau@maasstadziekenhuis.nl Collapse << |
| NCT00944346 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St, Charles, Missouri, United States Collapse << |
| NCT03270553 | Healthy Volunteers | Phase 1 | Completed | - | United States, Nebraska ... more >> Clinical Site Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT00159575 | Polycystic Ovary Syndrome | Phase 4 | Terminated(Slow patient recrui... more >>tment and expiry of study medication(aug2009)) Collapse << | - | Norway ... more >> Sigrun Kjøtrød Trondheim, Norway, 7006 Fertility UNit- -St Olavs Hospital Trondheim, Norway, 7046 Collapse << |
| NCT00965991 | Gestational Diabetes | Not Applicable | Completed | - | United States, New Mexico ... more >> University of New Mexico Diabetes in Pregnancy Clinic Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT01565096 | Diabetes Mellitus Type II | Phase 4 | Unknown | - | Germany ... more >> ikfe GmbH Recruiting Mainz, Germany, 55116 Contact: Thomas Forst, Prof., MD +49 6131 576 36 16 thomasf@ikfe.de Contact: Swantje Anders +49 6131 327 90 22 s.anders@ikfe-cro.de Collapse << |
| NCT00679679 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Argentina ... more >> Hospital Privado de Córdoba Cordoba, Argentina, X5016KEH Collapse << |
| NCT00944177 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St, Charles, Missouri, United States Collapse << |
| NCT00727857 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02151461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 United States, Florida Palm Beach Research Palm Beach, Florida, United States, 33409 Meridien Research Tampa, Florida, United States, 33606 United States, Georgia River Birch Research Alliance Blue Ridge, Georgia, United States, 30513 Meridian Research Savannah, Georgia, United States, 31406 United States, Ohio Streling Research Group Cincinnati, Ohio, United States, 45219 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Meharry Medical College Nashville, Tennessee, United States, 37208 Vanderbilt University Medical Center, Diabetes, Endocrinology, and Metabolism Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02226510 | Left Ventricular Hypertrophy ... more >> Insulin Resistance Coronary Artery Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital & Medical School Dundee, Angus, United Kingdom, DD1 9SY Collapse << |
| NCT02151461 | - | - | Completed | - | - |
| NCT02018731 | Becker's Muscular Dystrophy (B... more >>MD) Collapse << | Phase 2 | Completed | - | Switzerland ... more >> University Children's Hospital Basel, BS, Switzerland, 4031 Collapse << |
| NCT01677299 | - | - | Completed | - | - |
| NCT01349387 | Type 2 Diabetes | Not Applicable | Completed | - | France ... more >> Centre Hospitalier Sud Francilien Corbeil Essonnes, France, 91106 Collapse << |
| NCT01677299 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> Celerion, Inc. Tempe, Arizona, United States, 85283 United States, Nebraska Celerion, Inc. Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT00727857 | - | - | Completed | - | - |
| NCT01433913 | Adenocarcinoma of the Prostate... more >> Recurrent Prostate Cancer Stage I Prostate Cancer Stage IIA Prostate Cancer Stage IIB Prostate Cancer Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Arizona Cancer Center - Tucson Tucson, Arizona, United States, 85724-5024 University of Arizona Health Sciences Center Tucson, Arizona, United States, 85724 United States, California University of Southern California/Norris Cancer Center Los Angeles, California, United States, 90033 Collapse << |
| NCT03690778 | Diabetes Hype... more >>rlipidemias Collapse << | Phase 1 | Recruiting | March 2019 | Korea, Republic of ... more >> Inha University Hospital Recruiting Incheon, Korea, Republic of Contact: Sang-Heon Cho, MD., Ph D Collapse << |
| NCT01697566 | Gynecology | Phase 3 | Active, not recruiting | May 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02982798 | Diabetes Hype... more >>rlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inha University Hospital Incheon, Korea, Republic of Collapse << |
| NCT01433913 | - | - | Completed | - | - |
| NCT01911247 | Endometrial Cancer | Early Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT02647827 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Hyperandrogenism Collapse << | Phase 2 | Recruiting | December 2019 | China ... more >> Peking University Recruiting Beijing, China, 100871 Contact: Jie Qiao, Professor +8615611908108 jie.qiao@263.net Contact: Haolin Zhang, PhD +8613911533466 zoe@bjmu.edu.cn Sweden Karolinska University Hospital Recruiting Stockholm, Sweden, 17176 Contact: Berit Legerstam, Study nurse +46851773782 berit.legerstam@karolinska.se Contact: Liselotte Blomberg, Study nurse +46851773782 liselott.blomberg@karolinska.se Principal Investigator: Elisabet Stener-Victorin, PhD Sub-Investigator: Angelica Linden-Hirschberg, Professor Sub-Investigator: Sebastian Gidlöf, PhD Collapse << |
| NCT02102932 | Healthy | Phase 1 | Completed | - | China ... more >> 1276.23.86002 Boehringer Ingelheim Investigational Site Beijing, China Collapse << |
| NCT02179788 | Low Milk Supply ... more >> Pre-diabetes Insulin Resistance Suppressed Lactation Collapse << | Phase 1 Phase 2 | Completed | - | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 Collapse << |
| NCT03151005 | Polycystic Ovary Syndrome (PCO... more >>S) Overweight Obesity Collapse << | Phase 4 | Recruiting | December 31, 2018 | China, Chongqing ... more >> The Second Affiliated Hospital, Third Military Medical University Recruiting Chongqing, Chongqing, China, 400037 Principal Investigator: Min Long, MD Collapse << |
| NCT00941239 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Pharmacology and toxicology department, UANL Monterrey, Nuevo León, Mexico, 64460 Collapse << |
| NCT02167620 | Diabetes Mellitus, Type 2 ... more >> Schizophrenia Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Center for Addiction and Mental Health Toronto, Ontario, Canada Collapse << |
| NCT01728740 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00882882 | Type II Diabetes | Phase 1 | Completed | - | - |
| NCT03570632 | Diabetes Mellitus, Type 1 ... more >> Pregnancy in Diabetes Collapse << | Phase 4 | Not yet recruiting | December 2020 | United States, Pennsylvania ... more >> Magee Womens Hospital of UPMC Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02375139 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01762046 | Diabetes Mellitus, Type 2 | Phase 1 | Active, not recruiting | October 2017 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Brigham and Women's Hospital Boston, Massachusetts, United States, 02116 Joslin Diabetes Center Boston, Massachusetts, United States, 02116 Collapse << |
| NCT02102932 | - | - | Completed | - | - |
| NCT03258723 | Diabetes | Phase 4 | Not yet recruiting | June 2021 | United States, New York ... more >> Flatbush Family Health Center at NYU Langone Not yet recruiting Brooklyn, New York, United States, 11203 Contact: Paula Pollard-Thomas, MD 347-380-0377 Paula.Pollard-Thomas@nyumc.org Contact: La Sharra Bennett 718-630-2197 ext 4040 Internal Medicine Associates at Mt. Sinai Not yet recruiting New York, New York, United States, 10029 Contact: Victoria L Mayer, MD 212-659-9194 victoria.mayer@mountsinai.org Contact: Carol Horowitz, MD 212-659-8551 carol.horowitz@mountsinai.org Barbados University of the West Indies, Cavehill and Barbados Ministry of Health Polyclinics Not yet recruiting Bridgetown, Barbados Contact: Joseph Herbert, MD 2462347715 familymedicinebarbados@gmail.com Contact: Natasha Sobers, MD 2462400313 natasha.sobers@cavehill.uwi.edu Puerto Rico Internal Medicine Clinic at the University of Puerto Rico Hospital Not yet recruiting Carolina, Puerto Rico, 00984 Contact: Elsie Cruz, MD elsie.cruz1@upr.edu Contact: Enid J Garcia-Rivera, MD (787) 701-1121 enid.garcia3@upr.edu Trinidad and Tobago Southwest Regional Health Authority, La Romaine Health Center Not yet recruiting San Fernando, Trinidad and Tobago Contact: Albert Persaud, MD 1-868-225-1802 albertpersaud@swrha.co.tt Contact: Kavita Dharamraj, MD 1-868-730-3418 kavitadharamraj@swrha.co.tt Collapse << |
| NCT02437812 | Epithelial Ovarian Carcinoma | Phase 2 | Recruiting | April 2021 | United States, California ... more >> Gynecologic Oncology Associates Recruiting Newport Beach, California, United States, 92663 Contact: Katrina Lopez, CCRC 949-642-5165 ext 259 Katrinal@gynoncology.com Collapse << |
| NCT01192152 | Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT02050009 | Ovarian Papillary Serous Carci... more >>noma Ovarian Serous Cystadenocarcinoma Recurrent Fallopian Tube Cancer Recurrent Ovarian Epithelial Cancer Recurrent Ovarian Germ Cell Tumor Recurrent Primary Peritoneal Cavity Cancer Collapse << | Phase 1 | Withdrawn(study was not initia... more >>ted) Collapse << | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT01192152 | - | - | Completed | - | - |
| NCT01206647 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | February 2011 | Germany ... more >> Diabetologische Schwerpunktpraxis Dr. Lorra / Dr. Bonnermann Bochum, Germany, 44869 Zentrum für klinische Studien Dresden, GWT-TUD GmbH Dresden, Germany, 01307 Gemeinschaftspraxis Partner der Gesundheit Essen, Germany, 45355 IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Zentrum für Diabetes und Gefäßerkrankungen Münster, Germany, 48145 Diabetes Zentrum Neuwied Neuwied, Germany, 56564 ikfe Studiencenter Potsdam GmbH Potsdam, Germany, 14469 Collapse << |
| NCT01966978 | Diabetes Mellitus, Type 2 ... more >> Diabetes Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> UT Southwestern Medical Center Dallas, Texas, United States, 02720 Collapse << |
| NCT02519543 | Bipolar Disorder | Phase 3 | Recruiting | September 2019 | United States, Pennsylvania ... more >> Western Psychiatric Institute and Clinic, University of Pittsburgh Medical Center, University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213-2593 Contact: Roy Chengappa, MD FRCPC chengappakn@upmc.edu Contact: Patricia Schlicht, BN 412-246-5258 schlpx@UPMC.EDU Principal Investigator: Roy Chengappa, MD FRCPC Canada, Nova Scotia Nova Scotia Health Authority - Dept. of Psychiatry Recruiting Halifax, Nova Scotia, Canada, B3H 2E2 Contact: Julie S Garnham, BN (902) 473 7144 jgarnham@dal.ca Principal Investigator: Cynthia Calkin, MD FRCPC Collapse << |
| NCT03236740 | PCOS | Phase 4 | Completed | - | India ... more >> SCB Medical College Cuttack, Orissa, India, 753007 Collapse << |
| NCT03312582 | Peri-Implantitis | Phase 4 | Completed | - | - |
| NCT01427595 | Polycystic Ovary Syndrome ... more >> Hyperandrogenism Collapse << | Not Applicable | Active, not recruiting | December 2019 | United States, Virginia ... more >> Center for Research in Reproduction, University of Virginia Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00063232 | - | - | Completed | - | - |
| NCT02438397 | Type 2 Diabetes | Phase 4 | Unknown | October 2016 | China, Shanghai ... more >> the 6th affliliated hospital of Shanghai Jiaotong university Recruiting Shanghai, Shanghai, China, 200233 Collapse << |
| NCT03311308 | Advanced Melanoma | Phase 1 | Recruiting | December 2028 | United States, Pennsylvania ... more >> Univ of Pittsburgh Medical Center Hillman Cancer Center Recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: Amy Rose, RN 412-647-8587 Collapse << |
| NCT00772174 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00670800 | - | - | Completed | - | - |
| NCT02827903 | Type 2 Diabetes ... more >> Dyslipidemia Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Buchoen St. mary's Hospital Bucheon, Korea, Republic of SeJong Hostpital Bucheon, Korea, Republic of SoonChunHyang University Bucheon Hospital Bucheon, Korea, Republic of InJe University Busan paik Hospital Busan, Korea, Republic of Inje University Haeundae paik hospital Busan, Korea, Republic of Daegu Catholic University Medical Center Daegu, Korea, Republic of Youngnam University Hostpital Daegu, Korea, Republic of Daeheon Eulji Medical Center Daejeon, Korea, Republic of KonYang University Hostpital Daejeon, Korea, Republic of Chosun University Hospital Gwangju, Korea, Republic of BunDang Jesang Hospital(DMC) Gyeonggi-do, Korea, Republic of Hallym University Sacred Heart hospital Gyeonggi-do, Korea, Republic of HanYang University Guri Hospital Gyeonggi-do, Korea, Republic of Inje University Ilsan paik hospital Gyeonggi-do, Korea, Republic of Myoung Ji Hospital Gyeonggi-do, Korea, Republic of Gachon Gil Hospital Incheon, Korea, Republic of Inchoen St. mary's Hospital Incheon, Korea, Republic of Eulji Hospital Nowon Seoul, Korea, Republic of Hallym University Kangnam Sacred Heart hospital Seoul, Korea, Republic of KangBuk SamSung Hospital Seoul, Korea, Republic of Kangdong Sacred Heart Hospital Seoul, Korea, Republic of KonKuk University Medical Center Seoul, Korea, Republic of Korea University Anam Hospital Seoul, Korea, Republic of KyungHee University Hospital at Gangdong Seoul, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of Seoul National University Hospital(Famlily medicine) Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Severance hospital Seoul, Korea, Republic of SoonChunHayng University Hospital Seoul Seoul, Korea, Republic of The catholic University of Korea YEOUIDO St mary's Hospital Seoul, Korea, Republic of Korea University Guro hospital Soeul, Korea, Republic of Ajou University hospital Suwon, Korea, Republic of Wonju Severance Christian hospital Wŏnju, Korea, Republic of Collapse << |
| NCT03184493 | Liver Cancer | Phase 3 | Recruiting | June 2021 | China, Guangxi ... more >> Affiliated Tumor Hospital of Guangxi Medical University Recruiting Nanjing, Guangxi, China, 530021 Contact: Bang-de Xiang, PhD 7715330855 xiangbangde@163.com Collapse << |
| NCT00670800 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | United States, Michigan ... more >> University of Michigan, Michigan Clinical Research Unit Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01208740 | Infertility P... more >>COS Prognosis for Poor Response Collapse << | Phase 4 | Terminated(Concerns about safe... more >>ty at the first interim analysis) Collapse << | - | Italy ... more >> Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT00953355 | Polycystic Ovary Syndrome ... more >> Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy Collapse << |
| NCT02495103 | Renal Cell Carcinoma ... more >> Hereditary Leiomyomatosis and Renal Cell Cancer Papillary Renal Cell Carcinoma, Sporadic Collapse << | Phase 1 Phase 2 | Recruiting | May 1, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01367054 | Nutritional and Metabolic Dise... more >>ases Collapse << | Not Applicable | Unknown | December 2011 | Brazil ... more >> Biocinese Toledo, Pr, Brazil, 85903-590 Collapse << |
| NCT00063232 | Hepatitis | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02186483 | Hyperlipidemia ... more >> Diabetes Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center(SMC) Seoul, Gangnam-Gu, Korea, Republic of, 135-710 Collapse << |
| NCT00899470 | - | - | Completed | - | - |
| NCT01302145 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Hungary ... more >> Balatonfured, Hungary, 8230 Budapest, Hungary, 1083 Netherlands Groningen, Netherlands, 9470 AE Poland Warsaw, Poland, 02-097 Slovakia Bratislava, Slovakia, 83305 Collapse << |
| NCT00899470 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Mds Pharma Services Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT00286442 | - | - | Completed | - | - |
| NCT02941445 | Polycystic Ovary Syndrome ... more >> Body Weight Collapse << | Phase 4 | Completed | - | - |
| NCT02526615 | Insulin Sensitivity ... more >> Type 2 Diabetes Collapse << | Phase 4 | Completed | - | Finland ... more >> Turku PET Centre Turku, Finland, 20520 Turku university hospital, PET center Turku, Finland, 20520 Turku PET Centre (Turku University Hospital) Turku, Finland, 20521 Collapse << |
| NCT00915772 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00286442 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00915772 | - | - | Completed | - | - |
| NCT02028689 | Healthy | Phase 1 | Completed | - | United States, Michigan ... more >> Kalamazoo, Michigan, United States, 49007 Collapse << |
| NCT01341886 | Malignant Neoplasm of Thyroid ... more >>Stage I Malignant Neoplasm of Thyroid Stage II Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Mashad University of Medical Sciences Mashad, Khorasan Razavi, Iran, Islamic Republic of Collapse << |
| NCT03349684 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | September 11, 2019 | - |
| NCT01877564 | - | - | Completed | - | - |
| NCT01881828 | Type 1 Diabetes | Phase 3 | Unknown | December 2014 | United States, Florida ... more >> Jaeb Center for Health Research Tampa, Florida, United States, 33647 Collapse << |
| NCT00391261 | Schizophrenia ... more >> Schizoaffective Disorder Schizophreniform Disorder Bipolar Disorder Autism Spectrum Disorders Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> Cambridge Health Alliance Medford, Massachusetts, United States, 02155 Collapse << |
| NCT03710460 | Obesity | Phase 4 | Recruiting | June 30, 2019 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: Manuel Gonzalez, PhD +52-10-58-52-00 ext 34212 uiec@prodigy.net.mx Contact: Esperanza Martinez, PhD +52-10-58-52-00 ext 34212 esperanzamartnezabundi@yahoo.com Collapse << |
| NCT02851095 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT03566810 | Healthy | Phase 1 | Recruiting | November 8, 2018 | Germany ... more >> Please Contact the Merck KGaA Communication Center Recruiting Darmstadt, Germany, 64293 Contact +49 6151 72 5200 service@merckgroup.com Collapse << |
| NCT00466622 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Norway ... more >> Dept. of Obstetrics and Gynecology, National Center for fetal Medicine, University Hospital of Trondheim Trondheim, Norway, 7006 Collapse << |
| NCT03071705 | Non-Small Cell Adenocarcinoma ... more >> Tyrosine Kinase Mutation EGFR Gene Mutation Collapse << | Not Applicable | Recruiting | December 2017 | Mexico ... more >> Instituto National de Cancerologia Recruiting Mexico, Mexico, 14080 Contact: Oscar Gerardo MD Arrieta, Oncology 015556280400 ext 71101 ogar@servidor.unam.mx Collapse << |
| NCT01438814 | - | - | Completed | - | - |
| NCT01560481 | Healthy | Phase 1 | Completed | - | Spain ... more >> Centre d'Investigació de Medicaments Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT01438814 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02143050 | Melanoma | Phase 1 Phase 2 | Recruiting | June 2020 | United States, Kentucky ... more >> James Graham Brown Cancer Center-Universityof Louisville Recruiting Louisville, Kentucky, United States, 40202 Contact: Jason A Chesney, MD, PhD 502-562-3429 jason.chesney@louisville.edu Contact: Christina LaDuke 502-217-5205 christy.laduke@louisville.edu Sub-Investigator: Donald M Miller, MD, PhD Sub-Investigator: Kelly M McMasters, MD, PhD Sub-Investigator: Sucheta Telang, MD James Graham Brown Cancer Center Recruiting Louisville, Kentucky, United States, 40202 Contact: Karen Carter, BSN 502-562-3690 kmcart05@louisville.edu Contact: Christina LaDuke, BS 502-217-5205 christi.laduke@louisville.edu Collapse << |
| NCT02865668 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT03475524 | Melasma | Phase 4 | Not yet recruiting | August 2019 | Egypt ... more >> Assuit University Not yet recruiting Assiut, Egypt, 71111 Contact: Radwa Bakr 01119988115 Contact: sahar Ismail saharsotohy@yahoo.com Collapse << |
| NCT03048500 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage III Non-Small Cell Lung Cancer Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 2 | Recruiting | February 2021 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Young K. Chae 312-926-6895 Principal Investigator: Young K. Chae, MD, MPH, MBA Sub-Investigator: Sachin Pai, MD Sub-Investigator: Nisha Mohindra, MD Sub-Investigator: Victoria Villaflor, MD Sub-Investigator: Ricardo Costa, MD Sub-Investigator: Valerie Nelson, MD Sub-Investigator: Timothy Taxter, MD Northwestern University- Lake Forest Hospital Recruiting Lake Forest, Illinois, United States, 60045 Contact: Valerie Nelson, MD 847-582-2134 Principal Investigator: Valerie Nelson, MD Collapse << |
| NCT01877564 | Adenocarcinoma of the Endometr... more >>ium Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT00682448 | Metabolic Complications | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT00778427 | Healthy | Not Applicable | Completed | - | United States, Nevada ... more >> NovumPharmaceutical Research Services Las Vegas, Nevada, United States, 89121 Collapse << |
| NCT00473876 | Congestive Heart Failure ... more >> Insulin Resistance Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Medicine and Therapeutics, Ninewells Hospital Dundee, Scotland, United Kingdom, DD1 9SY Collapse << |
| NCT00648869 | Healthy | Not Applicable | Withdrawn(Drug was not sent) | November 2008 | Israel ... more >> Diabetes Unit, Hadassah Hebew University Hospital Jerusalem, Israel, 91120 Collapse << |
| NCT00473876 | - | - | Completed | - | - |
| NCT02640534 | Cancer of the Prostate ... more >> Prostate Cancer Collapse << | Phase 2 | Recruiting | June 2026 | Switzerland ... more >> Kantonsspital Aarau Recruiting Aarau, Switzerland, 5001 Contact: Tobias Wehrhahn, Dr. med. +41 62 838 60 50 tobias.wehrhahn@ksa.ch Principal Investigator: Tobias Wehrhahn, Dr. med. Universitaetsspital Basel Recruiting Basel, Switzerland, 4031 Contact: Sacha Rothschild, MD +41 61 265 25 25 sacha.rothschild@usb.ch Principal Investigator: Sacha Rothschild, MD Istituto Oncologico della Svizzera Italiana (IOSI) Recruiting Bellinzona, Switzerland, 6500 Contact: Ricardo Pereira Mestre, MD +41 91 811 84 46 ricardo.pereiramestre@eoc.ch Principal Investigator: Ricardo Pereira Mestre, MD Kantonsspital Graubuenden Recruiting Chur, Switzerland, 7000 Contact: Richard Cathomas, MD +41 81 256 66 95 richard.cathomas@ksgr.ch Principal Investigator: Richard Cathomas, MD Spital Thurgau AG Recruiting Frauenfeld, Switzerland, CH-8500 Contact: Regina Woelkly, MD +41 52 723 76 91 regina.woelkly@stgag.ch Principal Investigator: Regina Woelkly, MD Hôpitaux Universitaires de Genève Recruiting Genève 14, Switzerland, 1211 Contact: Anna Patrikidou, MD +41 79 553 60 99 anna.patrikidou@hcuge.ch Principal Investigator: Anna Patrikidou, MD CCAC Lausanne Recruiting Lausanne, Switzerland, 1004 Contact: Pierre Bohanes, MD +41 (21) 646 79 94 pbohanes@bluewin.ch Principal Investigator: Pierre Bohanes, MD Luzerner Kantonsspital Recruiting Luzern 16, Switzerland, 6000 Contact: Beat Müller, MD +41 41 205 58 60 beat.mueller@luks.ch Principal Investigator: Beat Müller, MD Hôpital du Valais Recruiting Martigny, Switzerland, 1920 Contact: Sandro Anchisi, MD +41 27 603 90 00 sandro.anchisi@hopitalvs.ch Principal Investigator: Sandro Anchisi, MD Kantonsspital Olten Recruiting Olten, Switzerland, CH-4600 Contact: Walter Mingrone, MD 41-62-311-4241 wmingrone_ol@spital.ktso.ch Principal Investigator: Walter Mingrone, MD Hôpital du Valais Recruiting Sion, Switzerland, 1951 Contact: Sandro Anchisi, MD +41 27 603 45 00 sandro.anchisi@hopitalvs.ch Principal Investigator: Sandro Anchisi, MD Bürgerspital Solothurn Recruiting Solothurn, Switzerland, CH-4500 Contact: Philippe von Burg, MD +41 32 627 47 00 philippe.vonburg@spital.so.ch Principal Investigator: Philippe von Burg, MD Kantonsspital St. Gallen Recruiting St. Gallen, Switzerland, 9007 Contact: Christian Rothermundt, MD +41 71 494 11 63 christian.rothermundt@kssg.ch Principal Investigator: Christian Rothermundt, MD Kantonsspital Winterthur Recruiting Winterthur, Switzerland, 8401 Contact: Natalie Fischer, MD +41 052 266 40 87 natalie.fischer@ksw.ch Principal Investigator: Natalie Fischer, MD Stadtspital Triemli Recruiting Zürich, Switzerland, 8063 Contact: Aleksandra Marek, Dr. med. +41 (44) 466 11 11 aleksandra.marek@triemli.zuerich.ch Principal Investigator: Aleksandra Marek, Dr. med. UniversitätsSpital Zürich Recruiting Zürich, Switzerland, 8091 Contact: Thomas Hermanns, MD +41 44 255 11 11 thomas.hermanns@usz.ch Principal Investigator: Thomas Hermanns, MD Collapse << |
| NCT00546728 | Impaired Glucose Tolerance | Phase 4 | Completed | - | United States, Minnesota ... more >> International Diabetes Center at Park Nicollet St. Louis Park, Minnesota, United States, 55416 St. Paul Heart Clinic St. Paul, Minnesota, United States, 55102 Collapse << |
| NCT00778466 | Healthy | Not Applicable | Completed | - | United States, Nevada ... more >> NovumPharmaceutical Research Services Las Vegas, Nevada, United States, 89121 Collapse << |
| NCT02580331 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT02125227 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of, 561712 Collapse << |
| NCT02819869 | Hepatocellular Carcinoma | Phase 2 | Terminated(insufficient for th... more >>e fund) Collapse << | - | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT01210911 | Locally Advanced Pancreatic Ca... more >>ncer Metastatic Pancreatic Cancer Collapse << | Phase 2 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, Netherlands, 1105AZ Collapse << |
| NCT01521624 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT00546728 | - | - | Completed | - | - |
| NCT02984475 | Beta Thalassemia Major Anemia | Phase 4 | Recruiting | April 2019 | Egypt ... more >> Abo El Reesh Hospital Recruiting Cairo., Egypt Contact: Mona Gaber, Bachelor. 0223648368 mona.sobhy@pharma.cu.edu.eg El Demerdash (Ain Shams Teaching Hospital). Completed Cairo., Egypt Collapse << |
| NCT02506790 | Breast Cancer | Phase 2 | Recruiting | August 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting Saint - Petersburg, Russian Federation, 191124 Contact: Tatiana Y Semiglazova, MD, PhD, DSc +79219468072 tsemiglazova@mail.ru Sub-Investigator: Petr V Krivorotko, MD, PhD, DSc Principal Investigator: Tatiana Y Semiglazova, MD, PhD, DSc Collapse << |
| NCT00830076 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT02945813 | Prostate Cancer | Phase 2 | Recruiting | December 2030 | France ... more >> Centre Hospitalier Régional Universitaire (CHRU) Jean Minjoz Recruiting Besançon, France, 25030 Contact: Bernardino De Bari, MD +33 381 23 24 28 bdebari@chu-besancon.fr Principal Investigator: Bernardino De Bari Clinique Pasteur - Centre finistérien de radiothérapie et d'oncologie Recruiting Brest, France, 29220 Contact: Ali Hasbini, MD +33 298 31 32 00 alihasbini@oncologie-brest.fr Principal Investigator: Ali Hasbini, MD Centre de lutte contre le cancer Léon Bérard Recruiting Lyon, France, 69008 Contact: Pascal Pommer, MD +33 478 78 51 66 pascal.pommier@lyon.unicancer.fr Principal Investigator: Pascal Pommer, MD Hôpital Saint-Louis Recruiting Paris, France, 75010 Contact: Christophe Hennequin, Prof +33 142 49 90 24 christophe.hennequin2@aphp.fr Principal Investigator: Christophe Hennequin, Prof CHU de Poitiers - La Miletrie Recruiting Poitiers Cedex, France, 86021 Contact: Stéphane Guerif, MD +33 549 44 29 61 stephane.guerif@chu-poitiers.fr Principal Investigator: Stéphane Guerif, MD Institut de Cancérologie de L'Ouest René Gauducheau Recruiting Saint Herblain cedex, France, 44800 Contact: Stéphane Supiot, MD +33 240 679 913 stephane.supiot@ico.unicancer.fr Principal Investigator: Stéphane Supiot, MD Institut de Cancérologie de la Loire Lucien Neuwirth Recruiting Saint Priest en Jarez, France, 42270 Contact: Nicolas Magne, Prof +33 477 91 71 04 nicolas.magne@icloire.fr Principal Investigator: Nicolas Magne, Prof Clinique Pasteur - Oncorad Recruiting Toulouse Cedex 3, France, 31076 Contact: Igor Latorzeff, MD +33 567 20 44 00 ilatorzeff@clinique-pasteur.com Principal Investigator: Igor Latorzeff, MD Germany Universitätsmedizin Berlin Recruiting Berlin, Germany, 13353 Contact: Pirus Ghadjar, MD +49 450 657055 pirus.ghadjar@charite.de Principal Investigator: Pirus Ghadjar, MD Klinikum der Universität München Not yet recruiting München, Germany, 81377 Contact: Claus Belka, Prof +49 89 4400 74520 claus.belka@med.uni-muenchen.de Principal Investigator: Claus Belka, Prof Universitätsklinikum Rostock Recruiting Rostock, Germany, 18059 Contact: Guido Hildebrandt, Prof +49 381 494 90 00 guido.hildebrandt@uni-rostock.de Principal Investigator: Guido Hildebrandt, Prof Universitätsklinikum Würzburg Recruiting Würzburg, Germany, 97080 Contact: Michael Flentje, Prof +49 931 201 288 91 flentje_m@ukw.de Principal Investigator: Michael Flentje, Prof Switzerland Universitätsspital Basel Recruiting Basel, Switzerland, 4031 Contact: Alexandros Papachristofilou, MD +41 61 265 49 46 alexandros.papachristofilou@usb.ch Principal Investigator: Alexandros Papachristofilou, MD Istituto Oncologico della Svizzera Italiana IOSI - Ospedale San Giovanni Recruiting Bellinzona, Switzerland, 6500 Contact: Che Ngwa Azinwi, MD +41 091 811 89 32 ngwache.azinwi@eoc.ch Principal Investigator: Che Ngwa Azinwi, MD Inselspital Bern Recruiting Bern, Switzerland, 3010 Contact: Daniel Aebersold, Prof +41 31 632 26 32 daniel.aebersold@insel.ch Principal Investigator: Daniel Aebersold, Prof Kantonsspital Graubuenden Recruiting Chur, Switzerland, 7000 Contact: Daniel R. Zwahlen, MD +41 81 256 64 95 daniel.zwahlen@ksgr.ch Principal Investigator: Daniel R. Zwahlen, MD HFR - Hôpital cantonal Recruiting Fribourg, Switzerland, 1708 Contact: David Lauffer, MD +41 26 306 00 00 David.Lauffer@h-fr.ch Principal Investigator: David Lauffer, MD Hôpitaux Universitaires Genève HUG Recruiting Geneva, Switzerland, 1211 Contact: Thomas Zilli, MD +41 22 372 70 90 thomas.zilli@hcuge.ch Principal Investigator: Thomas Zilli, MD Clinique de Genolier Recruiting Genolier, Switzerland, 1272 Contact: Cristina Picardi, MD +41 22 366 94 59 cpicardi@genolier.net Principal Investigator: Cristina Picardi, MD Spital Thurgau Recruiting Münsterlingen, Switzerland, CH-8596 Contact: Christiane Reuter, MD +41 71 686 23 33 christiane.reuter@stgag.ch Principal Investigator: Christiane Reuter, MD Hopital de Sion Recruiting Sion, Switzerland, 1951 Contact: Kaouthar Khanfir, MD +41 27 603 87 72 kaouthar.khanfir@hopitalvs.ch Principal Investigator: Kaouthar Khanfir, MD Kantonsspital St. Gallen Recruiting St. Gallen, Switzerland, 9007 Contact: Paul Martin Putora, MD +41 71 494 22 68 paulmartin.putora@kssg.ch Principal Investigator: Paul Martin Putora, MD Kantonsspital Winterthur KSW Recruiting Winterthur, Switzerland, 8401 Contact: Binaya Kumar Shrestha, MD +41 52 266 55 05 binayakumar.shrestha@ksw.ch Principal Investigator: Binaya Kumar Shrestha, MD Klinik Hirslanden Recruiting Zurich, Switzerland, 8032 Contact: Marcin Sumila, MD +41 44 387 25 51 marcin.sumila@hirslanden.ch Principal Investigator: Marcin Sumila, MD Stadtspital Triemli Recruiting Zurich, Switzerland, 8063 Contact: Helmut Kranzbühler, MD +41 44 466 13 67 helmut.kranzbuehler@triemli.zuerich.ch Principal Investigator: Helmut Kranzbühler, MD UniversitätsSpital Zürich Recruiting Zurich, Switzerland, 8091 Contact: Matthias Guckenberger, Prof +41 44 255 29 30 Matthias.Guckenberger@usz.ch Principal Investigator: Matthias Guckenberger, Prof Collapse << |
| NCT01999686 | Polycystic Ovary Syndrome (PCO... more >>S) Insulin Resistance Collapse << | Phase 3 | Recruiting | October 2018 | Indonesia ... more >> Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Lambung Mangkurat, Ulin Banjarmasin Hospital Recruiting Banjarmasin, Indonesia Contact: Iwan D Putra, dr., SpOG-K +62811503472 i_darmaputra@yahoo.com Principal Investigator: Iwan D Putra, dr., SpOG-K Sub-Investigator: Hardyan Sauqi, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Udayana, Sanglah Hospital Recruiting Denpasar, Indonesia Contact: Putu Doster Mahayasa, dr., SpOG-K +628123812259 dmahayasa@gmail.com Principal Investigator: Putu Doster Mahayasa, dr., SpOG-K Sub-Investigator: Anom Suardika, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Hasanuddin, Dr. Wahidin Sudirohusodo Hospital Recruiting Makasar, Indonesia Contact: Nusratuddin Abdullah, Dr., dr., SpOG-K, MARS +6281342752561 nusraya@yahoo.com Principal Investigator: Nusratuddin Abdullah, Dr., dr., SpOG-K, MARS Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Brawijaya, Dr. Saiful Anwar Hospital Recruiting Malang, Indonesia Contact: Arsana Wiyasa IW, Dr., dr., SpOG-K +62811343876 abiyasa9@yahoo.com Principal Investigator: Arsana Wiyasa IW, Dr., dr., SpOG-K Sub-Investigator: Sutrisno Sutrisno, dr., SpOG-K Sub-Investigator: Widjajanto Ngartjono, dr., SpOG-K Sub-Investigator: Pande M Dwijayasa, dr., SpOG-K Sub-Investigator: Hermawan Wibisono, dr., SpOG Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Sam Ratulangi, Prof. Dr. Kandou Hospital Recruiting Manado, Indonesia Contact: Eddy Suparman, Prof., Dr., dr., SpOG-K +62811431600 obsgyn_manado@yahoo.com.sg Principal Investigator: Eddy Suparman, Prof., Dr., dr., SpOG-K Sub-Investigator: Freddy W Wagey, Dr., dr., SpOG-K Sub-Investigator: Maria F Loho, dr., SpOG-K Sub-Investigator: Juneke J Kaeng, dr., SpOG-K Sub-Investigator: Maya E Mewengkang, dr., SpOG Sub-Investigator: Linda M Mamengko, dr., SpOG-K Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, Diponegoro University, Dr. Kariadi Hospital Recruiting Semarang, Indonesia Contact: Syarief Taufik, dr., SpOG-K +628112715580 tofik_obg@yahoo.com Principal Investigator: Syarief Taufik, dr., SpOG-K Sub-Investigator: Inu Mulyantoro, dr., SpOG-K Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Airlangga, Dr. Soetomo Hospital. Recruiting Surabaya, Indonesia Contact: Soehartono Ds, Prof., dr., SpOG-K +62811347720 batabsby@yahoo.com Principal Investigator: Soehartono Ds, Prof., dr., SpOG-K Sub-Investigator: Budi Santoso, Prof., Dr., dr., SpOG-K Sub-Investigator: Relly Y Primariawan, SpOG-K Collapse << |
| NCT00830076 | - | - | Completed | - | - |
| NCT00780013 | Healthy | Not Applicable | Completed | - | Canada, Quebec ... more >> MDS Pharma Services Saint-Laurent, Quebec, Canada Collapse << |
| NCT00809536 | Generalized Anxiety Disorder | Phase 1 | Terminated(Please Detailed Des... more >>cription for termination reason.) Collapse << | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT02104804 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Changchun, China Research Site Fuzhou, China Research Site Guangzhou, China Research Site Ha'er bing, China Research Site Hefei, China Research Site Nanchang, China Research Site Nanjing, China Research Site Shanghai, China Research Site ShiJiazhuang, China Research Site Shiyan, China Collapse << |
| NCT02984722 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Italy ... more >> Catholic university of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT02104804 | - | - | Completed | - | - |
| NCT01329016 | Gestational Diabetes Mellitus | Phase 1 Phase 2 | Unknown | June 2015 | United States, Indiana ... more >> Indiana University School of Medicine Indianapolis, Indiana, United States, 46202 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01046032 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | United Kingdom ... more >> IVF Hammersmith London, United Kingdom Nuture IVF Unit Nottingham, United Kingdom Oxford Fertility Unit Oxford, United Kingdom, OX39DU Collapse << |
| NCT02383563 | HIV Coronary ... more >>Artery Disease Collapse << | Phase 2 | Unknown | February 2016 | United States, Hawaii ... more >> Hawaii Center for AIDS Recruiting Honolulu, Hawaii, United States, 96813 Contact: Cecilia Shikuma, MD 808-692-1328 shikuma@hawaii.edu Contact: Debra Ogata-Arakaki, RN 808 692-1332 ogataara@hawaii.edu Collapse << |
| NCT01447927 | Barrett Esophagus ... more >> Esophageal Cancer Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> Hines Veterans Administration Hospital Hines, Illinois, United States, 60141 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Ohio Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106-5065 Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 United States, Pennsylvania University of Pittsburgh Medical Center - Shadyside Hospital Pittsburgh, Pennsylvania, United States, 15232 Canada, Ontario University of Toronto Toronto, Ontario, Canada, M5S 1A1 Puerto Rico University of Puerto Rico San Juan, Puerto Rico, 00936 Collapse << |
| NCT01710527 | Diabetic Foot | Phase 2 | Completed | - | - |
| NCT01710527 | - | - | Completed | - | - |
| NCT03329651 | Impaired Glucose Tolerance ... more >> Diabetes Mellitus, Type 2 Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT01525823 | Healthy Volunteers | Phase 1 | Completed | - | Australia, Victoria ... more >> Local Institution Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01733836 | Prostate Cancer | Phase 2 | Withdrawn(The reason for this ... more >>decision is that this study is being merged with a larger multi-centre study using the same agent in the same population.) Collapse << | October 2014 | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT02040246 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Shanghai ... more >> Renji Hospital Shanghai, Shanghai, China, 200127 China Renji Hospital Shanghai, China, 200127 Collapse << |
| NCT00961480 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT01447927 | - | - | Completed | - | - |
| NCT02039245 | Healthy Volunteers | Phase 1 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States Collapse << |
| NCT01593371 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of, 13145-784 Collapse << |
| NCT02797340 | Heart Failure, Systolic | Phase 4 | Active, not recruiting | April 30, 2018 | Denmark ... more >> Aarhus University Hospital, Department of Cardiology, Palle Juul-Jensens Boulevard 99 Aarhus N, Denmark, 8200 Collapse << |
| NCT01857609 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul,, Korea, Republic of Collapse << |
| NCT00720161 | Spina Bifida ... more >>Neuromuscular Diseases Collapse << | Not Applicable | Completed | - | Belgium ... more >> UZLeuven Leuven, Brabant, Belgium, 3000 Collapse << |
| NCT01292993 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Lexicon Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT00988078 | Polycystic Ovary Syndrome | Not Applicable | Unknown | December 2013 | Brazil ... more >> Federal University of Sao Paulo Recruiting Sao Paulo, Brazil Contact: Margareth Iwata megiwata@yahoo.com Collapse << |
| NCT01915550 | Diabetes Mellitus | Phase 3 | Completed | - | Egypt ... more >> Ain Shams University Maternity Hospital Cairo, Egypt, 19931 Collapse << |
| NCT00960076 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02188654 | Psoriatic Arthritis | Not Applicable | Completed | - | Egypt ... more >> University of Alexandria Alexandria, Egypt, 00203 Collapse << |
| NCT01002807 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT03558867 | Pre Diabetes ... more >>Type 2 Diabetes Mellitus Collapse << | Not Applicable | Recruiting | December 23, 2020 | Australia, New South Wales ... more >> Garvan Institute of Medical Research Recruiting Sydney, New South Wales, Australia, 2010 Contact: Jerry R Greenfield, MD, PhD +61292958217 j.greenfield@garvan.org.au Contact: Dorit Samocha-Bonet, PhD +61292958309 d.samochabonet@garvan.org.au Principal Investigator: Dorit Samocha-Bonet, PhD Principal Investigator: Jerry R Greenfield, MD, PhD Collapse << |
| NCT00661362 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00960076 | - | - | Completed | - | - |
| NCT00661362 | - | - | Completed | - | - |
| NCT02183571 | Healthy | Phase 1 | Completed | - | - |
| NCT02874430 | Breast Carcinoma ... more >> Endometrial Clear Cell Adenocarcinoma Endometrial Serous Adenocarcinoma Uterine Corpus Cancer Uterine Corpus Carcinosarcoma Collapse << | Phase 2 | Recruiting | January 2022 | United States, Pennsylvania ... more >> Thomas Jefferson University Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Jennifer Johnson, MD, PhD 215-955-8874 Sub-Investigator: Adam Berger, MD Sub-Investigator: Theodore Tsangaris, MD Sub-Investigator: Melissa Lazar, MD Sub-Investigator: Christine Kim, MD Sub-Investigator: Norman Rosenblum, MD, PhD Sub-Investigator: Benjamin Leiby, PhD Sub-Investigator: Madalina Tuluc, MD Sub-Investigator: Joanna Chan, MD Sub-Investigator: Ubaldo Martinez Outschoorn, MD Sub-Investigator: Nicole Simone, MD Collapse << |
| NCT01443221 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01357876 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, Cambridgeshire, United Kingdom, CB2 2GG Collapse << |
| NCT01454622 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT02077803 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT02022007 | Polycystic Ovary Syndrome ... more >> Disorder of Glucose Regulation Collapse << | Phase 3 | Completed | - | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70817 Collapse << |
| NCT01961388 | - | - | Withdrawn | June 2017 | - |
| NCT00897390 | - | - | Completed | - | - |
| NCT02007278 | Type 2 Diabetes ... more >> Hypoglycemia Collapse << | Phase 4 | Completed | - | Colombia ... more >> Novartis Investigative Site Medellín, Antioquia, Colombia Novartis Investigative Site Manizales, Caldas, Colombia, 1700 Novartis Investigative Site Bogotá, Cundinamarca, Colombia Novartis Investigative Site Chía, Cundinamarca, Colombia, 11001000 Novartis Investigative Site Cali, Valle del Cauca, Colombia, 760001 Novartis Investigative Site Bogotá, Colombia, 00000 Novartis Investigative Site Cali, Colombia Novartis Investigative Site Monteria, Colombia Collapse << |
| NCT02007278 | - | - | Completed | - | - |
| NCT00897390 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Mds Pharma Services Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT01831674 | Fasting | Phase 1 | Completed | - | India ... more >> Veeda Clinical Research Pvt. Ltd. Ahmedabad, Gujarat, India, 380 015 Collapse << |
| NCT02483299 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02022007 | - | - | Completed | - | - |
| NCT03109847 | Thyroid | Phase 2 | Recruiting | January 2022 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Joseph Curry, MD 215-955-6760 Collapse << |
| NCT02574845 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02574845 | - | - | Completed | - | - |
| NCT02565862 | Hepatitis C D... more >>iabetes Mellitus Insulin Resistance Collapse << | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud University Medical Center Nijmegen, Netherlands Collapse << |
| NCT03246451 | Glucose Metabolism Disorders | Not Applicable | Completed | - | Denmark ... more >> Center for Diabetes Research Gentofte, Copenhagen, Denmark, 2900 Collapse << |
| NCT03618654 | Head and Neck Squamous Cell Ca... more >>rcinoma Oral Cavity Squamous Cell Carcinoma Oropharyngeal Squamous Cell Carcinoma Collapse << | Phase 1 | Active, not recruiting | January 2021 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00118937 | Diabetes Mellitus, Type 1 | Phase 4 | Completed | - | - |
| NCT02223026 | Healthy | Phase 1 | Completed | - | - |
| NCT01206153 | Amenorrhea | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT01871558 | - | - | Completed | - | - |
| NCT02338193 | Diabetes Prevention in Women A... more >>fter GDM Who Are at High-risk Collapse << | Phase 3 | Active, not recruiting | March 2019 | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70815 Collapse << |
| NCT01941199 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02484209 | Diabetes Mellitus | Phase 3 | Withdrawn(Inclusion criteria t... more >>oo strict making it difficult to recruit in primary care) Collapse << | November 2016 | - |
| NCT01622257 | - | - | Completed | - | - |
| NCT02722239 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Russian Federation ... more >> Research Site Moscow, Russian Federation, 123423 Collapse << |
| NCT01995032 | Duchenne's Muscular Dystrophy ... more >>(DMD) Collapse << | Phase 3 | Completed | - | Switzerland ... more >> University Children's Hospital Basel, BS, Switzerland, 4031 Collapse << |
| NCT02722239 | - | - | Completed | - | - |
| NCT01622257 | PCOS | Phase 2 | Completed | - | Egypt ... more >> Ain shams university Cairo, Egypt Collapse << |
| NCT01871558 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 3 | Completed | - | France ... more >> Novartis Investigative Site Antibes, France, 06600 Novartis Investigative Site Auxerre, France, 89000 Novartis Investigative Site Bar le Duc, France, 55012 Novartis Investigative Site Bondy, France, 93143 Novartis Investigative Site Brest, France, 29200 Novartis Investigative Site Caen, France, 14000 Novartis Investigative Site Corbeil Essonnes, France, 91100 Novartis Investigative Site Fleury sur Orne, France, 14123 Novartis Investigative Site Maisons Laffitte, France, 78600 Novartis Investigative Site Menton, France, 06500 Novartis Investigative Site Montpellier, France, 34090 Novartis Investigative Site Paris, France, 75010 Novartis Investigative Site Rennes, France, 35203 Novartis Investigative Site Saint Nazaire, France, 44600 Novartis Investigative Site Sanary Sur Mer, France, 83110 Novartis Investigative Site Strsbourg, France, 67000 Novartis Investigative Site Toulouse, France, 31300 Novartis Investigative Site Valence, France, 26000 Novartis Investigative Site Valenciennes, France, 59300 Collapse << |
| NCT01842620 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02851212 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT00909506 | Breast Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT03355469 | Metabolic Syndrome | Phase 4 | Recruiting | January 31, 2022 | United States, Virginia ... more >> University of Virginia Recruiting Charlottesville, Virginia, United States, 22904 Contact: Stephanie Miller, M.S.Ed. 434-243-8677 slm4ps@virginia.edu Sub-Investigator: Emily Heiston, M.S.Ed Sub-Investigator: Monique Francois, PhD Principal Investigator: Steven K Malin, PhD Collapse << |
| NCT00961857 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT03138356 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Harrow, United Kingdom, HA1 3UJ Collapse << |
| NCT01478308 | Prostate Cancer | Phase 2 | Withdrawn(No accrual in initia... more >>l period, PI decided to close study.) Collapse << | - | - |
| NCT00834587 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> PRACS Institute Ltd. East Grand Forks, Minnesota, United States, 56721 United States, North Dakota PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01842620 | - | - | Completed | - | - |
| NCT02846506 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT00743002 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> Artesia, California, United States Los Angeles, California, United States Santa Ana, California, United States Torrence, California, United States Walnut Creek, California, United States United States, Florida Deland, Florida, United States United States, Illinois Bloomingdale, Illinois, United States United States, Michigan Grand Rapids, Michigan, United States United States, Missouri St. Louis, Missouri, United States United States, Nevada Las Vegas, Nevada, United States United States, New Jersey Clifton, New Jersey, United States United States, North Carolina Asheboro, North Carolina, United States Wilmington, North Carolina, United States United States, Pennsylvania Bensalem, Pennsylvania, United States United States, Tennessee Athens, Tennessee, United States United States, Texas Corpus Christi, Texas, United States Dallas, Texas, United States Houston, Texas, United States Richmond, Texas, United States San Antonio, Texas, United States Collapse << |
| NCT01508182 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT01508195 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT00834587 | - | - | Completed | - | - |
| NCT02744131 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | December 2019 | India ... more >> KAR clinic and hospital pvt ltd Recruiting Bhubaneswar, Odisha, India, 751001 Contact: Sujata Kar, M.D +919437824163 suju2463@gmail.com Contact: Suneeta Panigrahi, B.A womenwing@karhospitals.com Collapse << |
| NCT01365091 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Brazil ... more >> Local Institution Campinas, Sao Paulo, Brazil, 13073 Collapse << |
| NCT00835497 | - | - | Completed | - | - |
| NCT01970046 | Type 2 Diabetes | Phase 3 | Unknown | January 2015 | China ... more >> Chinese PLA General Hospital Recruiting Beijing, China Contact: Changyu Pan, M.D. panchy301@aliyun.com Principal Investigator: Changyu Pan, M.D. Collapse << |
| NCT02186847 | Adenosquamous Lung Carcinoma ... more >> Bronchioloalveolar Carcinoma Large Cell Lung Carcinoma Lung Adenocarcinoma Non-Small Cell Lung Carcinoma Recurrent Non-Small Cell Lung Carcinoma Squamous Cell Lung Carcinoma Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Collapse << | Phase 2 | Active, not recruiting | March 2023 | - |
| NCT00835497 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> PRACS Institute Ltd. East Grand Forks, Minnesota, United States, 56721 United States, North Dakota PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT03756623 | Healthy | Not Applicable | Recruiting | June 30, 2019 | China, Jiangsu ... more >> The Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School Recruiting Nanjing, Jiangsu, China, 210008 Contact: Mingming Zhang, MD,PhD 18551831115 doczmm@126.com Collapse << |
| NCT01233206 | Infertility P... more >>olycystic Ovary Syndrome (PCOS) Collapse << | Phase 4 | Completed | - | Italy ... more >> Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT02045290 | Type 1 Diabetes | Phase 3 | Completed | - | United States, Florida ... more >> Jaeb Center for Health Research Tampa, Florida, United States, 33647 Collapse << |
| NCT03316690 | Impaired Glucose Tolerance ... more >> Diabetes Mellitus, Type 2 Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for aktiv sundhed Copenhagen, Copenhagen N, Denmark, 2200 Collapse << |
| NCT01087866 | Gestational Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | Finland ... more >> Oulu University Hospital Oulu, Finland, 90029 Collapse << |
| NCT00337610 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 3 | Completed | - | - |
| NCT01365091 | - | - | Completed | - | - |
| NCT01791647 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Italy ... more >> Catholic University of Sacred Heart Recruiting Rome, Italy, 00168 Contact: Maurizio Guido, Medical Doctor 063057794 Principal Investigator: Maurizio Guido, Medical Doctor Collapse << |
| NCT01353391 | Type 2 Diabetes | Phase 3 | Active, not recruiting | June 2019 | Canada, Ontario ... more >> The Centre for Mother, Infant, and Child Research, Sunnybrook Research Institute Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT00770653 | Diabetes Mellitus ... more >> Dyslipidemias Collapse << | Phase 3 | Completed | - | - |
| NCT01766778 | Type-2 Diabetes Mellitus | Phase 4 | Completed | - | Hong Kong ... more >> Novartis Investigative Site Hong Kong SAR, Hong Kong Novartis Investigative Site HongKong, Hong Kong Novartis Investigative Site Tuen Mun, Hong Kong Collapse << |
| NCT00875394 | - | - | Completed | - | - |
| NCT01489644 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Novo Nordisk Investigational Site Miami, Florida, United States, 33169 United States, Washington Novo Nordisk Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT00875394 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00770653 | - | - | Completed | - | - |
| NCT02551874 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03109873 | Larynx Lip ... more >> Oral Cavity Pharynx Collapse << | Early Phase 1 | Active, not recruiting | June 2020 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT01766778 | - | - | Completed | - | - |
| NCT03078725 | Gestational Diabetes Mellitus,... more >> Class A2 Collapse << | Phase 4 | Withdrawn(Research was competi... more >>ng with other projects in the department, and no patients were recruited.) Collapse << | May 10, 2018 | - |
| NCT00337610 | - | - | Completed | - | - |
| NCT01509001 | Type 2 Diabetes | Phase 4 | Completed | - | Brazil ... more >> Clinical Hospital of São Paulo Medical School são Paulo, SP, Brazil Collapse << |
| NCT00836472 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT00835991 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT00835991 | - | - | Completed | - | - |
| NCT02551874 | - | - | Completed | - | - |
| NCT01910441 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(The study was termi... more >>nated due to the unavailability of Continuous Glucose Monitoring sensors (CGMS) which were required to assess the primary end-point.) Collapse << | - | - |
| NCT01910441 | - | - | Terminated(The study was termi... more >>nated due to the unavailability of Continuous Glucose Monitoring sensors (CGMS) which were required to assess the primary end-point.) Collapse << | - | - |
| NCT02294006 | Well Differentiated Pancreatic... more >> Endocrine Tumor Collapse << | Phase 2 | Unknown | June 2017 | Italy ... more >> Fondazione IRCCS Istituto Tumori Milano Recruiting Milano, Italy, 20133 Contact: Sara Pusceddu, MD sara.pusceddu@istitutotumori.mi.it Collapse << |
| NCT01021579 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Zainabieh Hospital Shiraz, Fars, Iran, Islamic Republic of, 7173646199 Collapse << |
| NCT00468039 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, Texas ... more >> Diabetes and Glandular Disease Research Associates San Antonio, Texas, United States, 78229 Collapse << |
| NCT02244567 | Polycystic Ovarian Syndrome | Phase 1 | Recruiting | June 2019 | Egypt ... more >> Nesreen Abdel Fattah Abdullah Shehata Recruiting Cairo, Egypt Contact: Nesreen A Shehata, MD 00201227866337 nesoomar@yahoo.com Collapse << |
| NCT02980276 | Gestational Diabetes | Phase 3 | Recruiting | August 2020 | Ireland ... more >> Cork University Hospital Not yet recruiting Cork, Ireland Principal Investigator: Prof. Louise Kenny University Hospital Galway Recruiting Galway, Ireland Principal Investigator: Prof. Fidelma Dunne Collapse << |
| NCT01845077 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> 1288.8.1 Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT00836472 | - | - | Completed | - | - |
| NCT01845077 | - | - | Completed | - | - |
| NCT01578551 | Carcinoma, Non-Small-Cell Lung... more >> Lung Adenocarcinoma Collapse << | Phase 2 | Active, not recruiting | November 2019 | United States, Maryland ... more >> Johns Hopkins Bayview Medical Center Baltimore, Maryland, United States, 21224 Johns Hopkins University SKCCC Baltimore, Maryland, United States, 21231 Collapse << |
| NCT01811953 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.8.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02048761 | Intrabony Defects in Chronic P... more >>eriodontitis Collapse << | Phase 2 | Unknown | February 2014 | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT03513874 | Type 1 Diabetes Mellitus ... more >> Autoimmune Diabetes Collapse << | Phase 4 | Recruiting | March 1, 2019 | China, Hunan ... more >> Institute of Metabolism and Endocrinology, Second Xiangya Hospital, Central South University Recruiting Changsha, Hunan, China, 410011 Contact: Zhiguang Zhou, MD/PhD 86-731-85292154 zhouzg@hotmail.com Collapse << |
| NCT01811953 | - | - | Completed | - | - |
| NCT01211197 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Germany ... more >> 1276.5.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02173639 | Healthy | Phase 1 | Completed | - | - |
| NCT01917656 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT02083692 | Head and Neck Squamous Cell Ca... more >>ncer Collapse << | Early Phase 1 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT01917656 | - | - | Completed | - | - |
| NCT01587378 | Pregnancy Pol... more >>ycystic Ovary Syndrome Collapse << | Not Applicable | Completed | - | Iceland ... more >> Landspital University Hospital Reykjavik, Iceland, 101 Norway Haukeland University Hospital Bergen, Norway, 5000 Nordlandssykehuset HF Bodø, Norway, 1480 Sykehuset Innlandet Brumunddal, Norway, 2380 Vestre Viken Hospital Trust Drammen, Norway Vestre Viken Ringerike sykehus Honefoss, Norway, 3511 Sykehuset Telemark Skien, Norway, 3710 St Olavs Hospital Trondheim, Norway, 7006 Sentralsykehuset i Vestfold Tønsberg, Norway, 3116 Sykehuset Ålesund Ålesund, Norway, 6026 Sweden Karolinska Universitetssjukhus Stockholm, Sweden, 17176 Norrlands universitetssjukhus Umeå, Sweden, 901 85 Uppsala University Hospital Uppsala, Sweden, 75185 Collapse << |
| NCT01725490 | Familial Adenomatous Polyposis | Phase 2 | Unknown | September 2015 | Korea, Republic of ... more >> Department of Internal Medicine, Yonsei University College of Medicine Not yet recruiting Seoul, Korea, Republic of, 120-752 Contact: TAE IL KIM, MD 82-2-2228-1965 taeilkim@yuhs.ac Department of Internal Medicine, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 120-752 Contact: TAE IL KIM, MD 82-2-2228-1965 taeilkim@yuhs.ac Collapse << |
| NCT03380091 | PCOS Depressi... more >>on Vitamin D Deficiency Insulin Resistance Collapse << | Phase 4 | Recruiting | April 2019 | United States, California ... more >> UCSF Center for Reproductive Health Recruiting San Francisco, California, United States, 94158 Contact: Nik Lenhart 415-885-3598 Nik.Lenhart@ucsf.edu Collapse << |
| NCT01525225 | Type 2 Diabetes Mellitus | Phase 1 | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | United States, Arkansas ... more >> Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 United States, Kentucky Kosair Charities Pediatric Clinical Research Unit Louisville, Kentucky, United States, 40202 United States, Missouri The Children'S Mercy Hospital And Clinics Kansas City, Missouri, United States, 64108 United States, Ohio Promedica Toledo Children'S Hospital Toledo, Ohio, United States, 43606 Promedica Toledo Childrens Hospital Toledo, Ohio, United States, 43606 United States, Pennsylvania Childrens Hospital Of Pittsburgh Of Upmc Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT00778349 | Healthy | Not Applicable | Completed | - | - |
| NCT01975220 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> 1276.13.1 Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT03189732 | Diabetes Mellitus, Type 2 | Not Applicable | Active, not recruiting | December 2017 | Czechia ... more >> Third Faculty of Medicine, Charles University Prague, Czechia, 100 00 Collapse << |
| NCT01211197 | - | - | Completed | - | - |
| NCT01975220 | - | - | Completed | - | - |
| NCT02106923 | Healthy | Phase 1 | Completed | - | United States, Texas ... more >> Boehringer Ingelheim Investigational Site Austin, Texas, United States Collapse << |
| NCT02106923 | - | - | Completed | - | - |
| NCT00881725 | Prostate Cancer | Phase 2 | Terminated(Slow Accrual) | - | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G2M9 Collapse << |
| NCT00690456 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Indonesia Sanofi-Aventis Administrative Office Jakarta, Indonesia Lithuania Sanofi-Aventis Administrative Office Vilnius, Lithuania Malaysia Sanofi-Aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Slovakia Sanofi-Aventis Administrative Office Brastislava, Slovakia Taiwan Sanofi-Aventis Administrative Office Taipei, Taiwan Thailand Sanofi-Aventis Administrative Office Bangkok, Thailand Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT01187849 | Patients With Glucocorticoid T... more >>reatment Collapse << | Phase 4 | Completed | - | Switzerland ... more >> University Hospital Basel Basel, Basel-Stadt, Switzerland, 4031 Collapse << |
| NCT01525225 | - | - | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | - |
| NCT03088631 | Intracytoplasmatic Sperm Injec... more >>tion Collapse << | Phase 2 | Recruiting | May 2019 | Egypt ... more >> Women Health Hospital - Assiut university Recruiting Assiut, Egypt, 71111 Contact: Mohammed K ALi, MD +201005537951 m_khairy2001@yahoo.com Collapse << |
| NCT02068443 | - | - | Completed | - | - |
| NCT03259490 | Healthy | Phase 1 | Completed | - | Germany ... more >> Humanpharmakologisches Zentrum Biberach Biberach, Germany, 88397 Collapse << |
| NCT02068443 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Nagoya-shi, Aichi, Japan Chiba-shi, Chiba, Japan Fukuoka-shi, Fukuoka, Japan Kitakyushu-shi, Fukuoka, Japan Kurume-shi, Fukuoka, Japan Aki-gun, Hiroshima, Japan Hiroshima-shi, Hiroshima, Japan Koga-shi, Ibaraki, Japan Mito-shi, Ibaraki, Japan Tushiura-shi, Ibaraki, Japan Ushiku-shi, Ibaraki, Japan Kanazawa-shi, Ishikawa, Japan Takamatsu-chi, Kagawa, Japan Sendai-shi, Miyagi, Japan Ohita-shi, Ohita, Japan Okinawa-shi, Okinawa, Japan Shimajiri-gun, Okinawa, Japan Kashiwara-shi, Osaka, Japan Osaka-shi, Osaka, Japan Hiki-gun, Saitama, Japan Oyama-shi, Tochigi, Japan Shimotsuke-shi, Tochigi, Japan Koutoh-ku, Tokyo, Japan Meguro-ku, Tokyo, Japan Sagae-shi, Yamagata, Japan Yamagata-shi, Yamagata, Japan Yamaguchi-shi, Yamaguchi, Japan Collapse << |
| NCT02183506 | Healthy | Phase 1 | Completed | - | - |
| NCT01590797 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01333852 | Head and Neck Neoplasms ... more >> Squamous Cell Carcinoma Collapse << | Phase 2 | Terminated(Low accrual) | - | Brazil ... more >> Barretos Cancer Hospital Barretos, SP, Brazil, 14784400 Collapse << |
| NCT01590797 | - | - | Completed | - | - |
| NCT01885013 | Human Epidermal Growth Factor ... more >>2 Negative Carcinoma of Breast Collapse << | Phase 2 | Completed | - | Italy ... more >> P.O. M. Bufalini Cesena, FC, Italy UO Oncologia Medica IRCCS IRST Meldola (FC), FC, Italy, 47014 Ospedale Civile degli Infermi Faenza, RA, Italy Ospedale Umberto I Lugo, RA, Italy Ospedale Civile Santa Maria delle Croci Ravenna, RA, Italy ULSS n.8 Asolo Ospedale di Castelfranco Asolo, Italy Centro di Riferimento Oncologico CRO Aviano, Italy Ospedale S.Martino Belluno, Italy Azienda Ospedaliera "Antonio Cardarelli" Campobasso, Italy P.O. "SS. Annunziata" Chieti, Italy Presidio Ospedaliero E. Profili Fabriano, Italy E.O. Galliera Genova, Italy Ospedale di Guastalla Guastalla, Italy Azienda per i Servizi Sanitari n.5 "Bassa Friulana" Latisana, Italy Presidio Ospedaliero "Vito Fazzi" Lecce, Italy ULSS n.13 di Mirano Mirano, Italy Arcispedale S. Maria Nuova Modena, Italy Azienda Ospedaliera S. Salvatore di Pesaro Pesaro, Italy Ospedale S. Spirito Pescara, Italy Ospedale Civile di Piacenza Piacenza, Italy Azienda Ospedaliera Santa Maria degli Angeli Pordenone, Italy Arcispedale S. Maria Nuova Reggio Emilia, Italy Ospedale Civile degli Infermi Rimini, Italy IRCCS Centro di riferimento Oncologico di Basilicata di Rionero in Vulture Rionero in Vulture, Italy Ospedale Nuovo Regina Margherita Roma, Italy Collapse << |
| NCT00959101 | Diabetes Heal... more >>thy Collapse << | Phase 1 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100032 Collapse << |
| NCT01550900 | Colorectum | Not Applicable | Withdrawn | - | - |
| NCT01690091 | Insulin Resistance ... more >> Chronic Heart Failure Collapse << | Phase 2 Phase 3 | Completed | - | Czechia ... more >> Diabetes Center, Institute of Clinical and Experimental Medicine Prague, Czechia Collapse << |
| NCT00855166 | - | - | Completed | - | - |
| NCT03047837 | Tertiary Prevention in Colon C... more >>ancer Collapse << | Phase 2 | Recruiting | March 15, 2020 | Italy ... more >> Medical Oncology Ente Ospedaliero Ospedali Galliera Recruiting Genova, Italy, 16128 Contact: Andrea De Censi, MD +39010 5634501 andrea.decensi@galliera.it Contact: Marilena Petrera, PhD 00390105634580 marilena.petrera@galliera.it Collapse << |
| NCT00855166 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Bulgaria ... more >> Research Site Blagoevgrad, Bulgaria Research Site Sofia, Bulgaria Czech Republic Research Site Beroun, Czech Republic Research Site Brno, Czech Republic Research Site Praha, Czech Republic Research Site Semily, Czech Republic Research Site Slany, Czech Republic Hungary Research Site Balatonfured, Hungary Research Site Budapest, Hungary Research Site Csongrad, Hungary Research Site Kecskemet, Hungary Research Site TAT, Hungary Poland Research Site Elblag, Poland Research Site Krakow, Poland Research Site Torun, Poland Sweden Research Site Goteborg, Sweden Research Site Jarfalla, Sweden Research Site Lund, Sweden Research Site Malmo, Sweden Research Site Stockholm, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT03076281 | Larynx LIP ... more >> Oral Cavity Pharynx Collapse << | Phase 2 | Recruiting | August 2020 | United States, Pennsylvania ... more >> Sidney Kimmel Cancer Center at Thomas Jefferson University Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Jennifer Johnson, MD, PhD 215-955-8874 jennifer.johnson@jefferson.edu Collapse << |
| NCT03243851 | Glioblastoma | Phase 2 | Recruiting | December 31, 2018 | Korea, Republic of ... more >> National Cancer Center Korea Recruiting Ilsan, Gyeonggi-Do, Korea, Republic of, 10408 Contact: Heon Yoo, MD., PhD. +82-31-920-1249 heonyoo@ncc.re.kr Seoul National University Bundang Hospital Recruiting Seongnam-si, Gyeonggi-Do, Korea, Republic of, 13620 Contact: Yu Jung Kim, MD., PhD. +82-31-787-8210 cong1005@gmail.com Ajou University Hospital Recruiting Suwon-si, Gyeonggi-Do, Korea, Republic of, 16499 Contact: Se Hyuk Kim, MD., PhD. +82-31-219-3559 nsksh@ajou.ac.kr Chonnam National University Hwasun Hospital Recruiting Hwasun, Jeollanam-Do, Korea, Republic of, 58128 Contact: Shin Jung, MD., PhD. +82-2-379-8954 sjung@chonnam.ac.kr Incheon St. Mary's Hospital Recruiting Incheon, Korea, Republic of, 21431 Contact: Dong Sup Chung, MD., PhD. +82-32-280-5876 dschung@catholic.ac.kr Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Contact: Tae Min Kim, MD., PhD. +82-2-2072-1672 gabriel9@snu.ac.kr Severance Hospital Recruiting Seoul, Korea, Republic of, 03722 Contact: Jong Hee Chang, MD., PhD. +82-2-2228-2162 changjh@yuhs.ac Konkuk University Hospital Recruiting Seoul, Korea, Republic of, 05030 Contact: Young Cho Koh, MD., PhD. +82-2-2030-7622 kohyc@kuh.ac.kr Asan Medical Center Recruiting Seoul, Korea, Republic of, 05505 Contact: Jeong Hoon Kim, MD., PhD. +82-2-3010-3559 jhkim1@amc.seoul.kr Samsung Medical Center Recruiting Seoul, Korea, Republic of, 06351 Contact: Do Hyun Nam, MD., PhD. +82-2-3410-3497 nsnam@skku.edu Seoul St Mary's Hospital Recruiting Seoul, Korea, Republic of, 06591 Contact: Yong-Kil Hong, MD., PhD. +82-2-2258-2101 hongyk@catholic.ac.kr Saint Vincent's Hospital, Korea Recruiting Suwon-si, Korea, Republic of, 16247 Contact: Seung Ho Yang, MD., PhD. +82-31-249-8304 72ysh@catholic.ac.kr Collapse << |
| NCT02514629 | Hypogonadism ... more >>Obesity Collapse << | Phase 3 | Completed | - | Spain ... more >> Hospital Uiversitario Virgen de la Victoria Malaga, Spain, 29010 Collapse << |
| NCT01429818 | Endothelial Dysfunction ... more >> Diabetes Mellitus Collapse << | Phase 4 | Completed | - | Mexico ... more >> Unidad PET Ciclotrón. Facultad de Medicina. Universidad Nacional Autónoma de México Mexico city, Distrito Federal, Mexico, 04510 Collapse << |
| NCT03108963 | PCOS | Phase 2 | Completed | - | Egypt ... more >> Zagazig University Zagazig, Egypt Collapse << |
| NCT02910817 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Assiut University Assiut, Egypt, 71515 Collapse << |
| NCT01627067 | Breast Cancer | Phase 2 | Active, not recruiting | September 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02791490 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03238495 | HER2-positive Breast Cancer | Phase 2 | Recruiting | September 1, 2019 | United States, Kansas ... more >> The University of Kansas Cancer Center, West Clinic Recruiting Kansas City, Kansas, United States, 66112 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Westwood Campus Recruiting Kansas City, Kansas, United States, 66205 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Overland Park Clinic Recruiting Overland Park, Kansas, United States, 66210 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu United States, Missouri The University of Kansas Cancer Center, South Clinic Recruiting Kansas City, Missouri, United States, 64131 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, North Clinic Recruiting Kansas City, Missouri, United States, 64154 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu The University of Kansas Cancer Center, Lee's Summit Clinic Recruiting Lee's Summit, Missouri, United States, 64064 Contact: Clinical Trials Nurse Navigator 913-945-7552 ctnursenav@kumc.edu Collapse << |
| NCT00940472 | Diabetes Type 2 | Phase 2 Phase 3 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud, Universidad de Guadalajara Guadalajara, Jal, Mexico, 44340 Collapse << |
| NCT03600363 | Lymphoma, Large B-Cell, Diffus... more >>e Stage III Follicular Lymphoma Collapse << | Phase 2 | Not yet recruiting | December 2020 | - |
| NCT03603912 | Atrial Fibrillation | Phase 4 | Recruiting | June 30, 2022 | United States, Ohio ... more >> Cleveland Clinic Recruiting Cleveland, Ohio, United States, 44195 Contact: Mina K Chung, MD 216-444-2290 chungm@ccf.org Contact: Niraj Varma, MD 216-444-2142 varman@ccf.org Collapse << |
| NCT01797523 | Endometrial Cancer | Phase 2 | Active, not recruiting | October 2020 | United States, Florida ... more >> Sacred Heart Health Systems Pensacola, Florida, United States, 32504 United States, New Jersey MD Anderson Cooper Cancer Center Voorhees, New Jersey, United States, 08043 United States, Texas Memorial City Medical Center Houston, Texas, United States, 77024 Lyndon B Johnson General Hospital Houston, Texas, United States, 77026 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 The Woman's Hospital of Texas Houston, Texas, United States, 77054 Collapse << |
| NCT02049814 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Anhui ... more >> Hefei, Anhui, China Maanshan, Anhui, China China, Beijing Beijing, Beijing, China China, Guangdong Guangzhou, Guangdong, China Shenzhen, Guangdong, China Taishan, Guangdong, China China, Hunan Zhuzhou, Hunan, China China, Jiangsu Nanjing, Jiangsu, China Xuzhou, Jiangsu, China China, Jilin Changchun, Jilin, China Jilin, Jilin, China China, Liaoning Shenyang, Liaoning, China China, Shandong Qingdao, Shandong, China China, Shanghai Shanghai, Shanghai, China China, Shanxi Yan An, Shanxi, China Yanan, Shanxi, China China, Tianjin Tianjin, Tianjin, China China, Zhejiang Wenzhou, Zhejiang, China Collapse << |
| NCT00649350 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01434186 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States United States, Florida Research Site Tallahassee, Florida, United States United States, Michigan Research Site Dearborn, Michigan, United States United States, Minnesota Research Site St. Paul, Minnesota, United States United States, New York Research Site Mineola, New York, United States United States, Ohio Research Site Cleveland, Ohio, United States United States, Tennessee Research Site Memphis, Tennessee, United States Belgium Research Site Namur, Belgium Canada, Alberta Research Site Calgary, Alberta, Canada India Research Site Bangalore, India Mexico Research Site Aguascalientes, Mexico Research Site Meridas, Mexico Research Site Monterrey, Mexico Research Site Veracruz, Mexico Taiwan Research Site Taichung, Taiwan United Kingdom Research Site Leicester, United Kingdom Collapse << |
| NCT01434186 | - | - | Completed | - | - |
| NCT01107808 | Insulin Resistance ... more >> Insulin Sensitivity Obesity Vitamin D Deficiency Collapse << | Not Applicable | Withdrawn(Poor recruitment to ... more >>the study) Collapse << | March 2011 | - |
| NCT00501787 | Polycystic Ovary Syndrome | Phase 4 | Withdrawn | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT02644577 | Bipolar Disorder | Phase 4 | Completed | - | China, Liaoning ... more >> Department of Psychiatry, the First Hospital of China Medical University Shenyang, Liaoning, China, 110001 Collapse << |
| NCT02185066 | Healthy Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Busan Paik Hospital Busan, Korea, Republic of Collapse << |
| NCT01555164 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01555164 | - | - | Completed | - | - |
| NCT01864681 | NSCLC EGFR Ge... more >>ne Amplification Advanced Cancer Stage IIIB NSCLC Stage IV NSCLC Collapse << | Phase 2 | Completed | - | China, Chongqing ... more >> Department of Respiratory Diseases, Daping Hospital, Third Military Medical University Chongqing, Chongqing, China, 400042 Collapse << |
| NCT01528254 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | April 18, 2019 | - |
| NCT00528879 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00924573 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Sanofi-Aventis Administrative Office Tokyo, Japan Collapse << |
| NCT00501904 | Polycystic Ovary Syndrome | Phase 4 | Unknown | December 2016 | Italy ... more >> Pugliese Hospital Recruiting Catanzaro, Catanzaro, CZ, Italy, Italy, 88100 Contact: Ingrid Tomaino, MD +39-0961965097 angela.falbo@libero.it Principal Investigator: Stefano Palomba, MD Collapse << |
| NCT03010683 | Type 2 Diabetes Mellitus ... more >> Coronary Artery Disease Collapse << | Not Applicable | Recruiting | December 2017 | Greece ... more >> ''Attikon'' University General Hospital Recruiting Haidari, Athens, Attiki, Greece, 12462 Principal Investigator: Ignatios Ikonomidis, MD Collapse << |
| NCT00525330 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> San Diego, California, United States Collapse << |
| NCT01423487 | Weight Gain A... more >>menorrhea Collapse << | Not Applicable | Withdrawn(Difficult to obtain ... more >>informed consent) Collapse << | August 2013 | China, Hunan ... more >> Mental Health Institute of The Second Xiangya Hospital Changsha, Hunan, China, 410011 Collapse << |
| NCT00145379 | Type 1 Diabetes Mellitus | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Center Odense, Funen, Denmark, 5000 Collapse << |
| NCT02570672 | Frailty | Phase 2 | Recruiting | October 2018 | United States, Texas ... more >> University of Texas Health Science Center at San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Alicia Conde 210-617-5197 Collapse << |
| NCT00528879 | - | - | Completed | - | - |
| NCT01240759 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Texas ... more >> Juno Research, LLC Houston, Texas, United States, 77074 Collapse << |
| NCT01438190 | Infertility P... more >>COS Collapse << | Phase 4 | Unknown | December 2014 | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT00134303 | Obesity | Phase 4 | Unknown | July 2015 | Belgium ... more >> University Hospital Ghent Recruiting Ghent, Belgium, 9000 Contact: Hans Van Vlierberghe, MD, PhD + 32 9 332.23.70 hans.vanvlierberghe@UGent.be Collapse << |
| NCT02565368 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT00754689 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(Company decision tak... more >>en in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT02689843 | Polycystic Ovary Syndrome | Early Phase 1 | Completed | - | Iran, Islamic Republic of ... more >> Shahid Motahhari Clinic, Shiraz University of Medical Sciences Shiraz, Fars, Iran, Islamic Republic of Collapse << |
| NCT01089192 | Type 2 Diabetes | Phase 1 | Completed | - | India ... more >> Torrent Research Centre Bhat, Gujarat, India, 382 428 Collapse << |
| NCT00247117 | Liver Diseases | Not Applicable | Unknown | - | Israel ... more >> Institute of Endocrinology, Kaplan Medical Center Recruiting Rehovot, Israel, 76100 Collapse << |
| NCT01089205 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02024971 | - | - | Completed | - | - |
| NCT02024971 | - | - | Completed | - | - |
| NCT00857870 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> GWT -TUD, Center for Clinical Studies Dresden, Germany, 01307 Collapse << |
| NCT02614339 | Non-DM Stage III Colorectal Ca... more >>ncer Collapse << | Phase 3 | Not yet recruiting | November 2021 | - |
| NCT01477060 | Metastatic Breast Cancer | Phase 2 | Terminated(The study was stopp... more >>ed due to insufficient accrual) Collapse << | - | Italy ... more >> Cliniche Gavazzeni S.p.A. - Humanitas Gavazzeni Bergamo, BG, Italy, 24125 Fondazione Poliambulanza Brescia, BS, Italy, 25124 Azienda Ospedaliera San Gerardo Monza, MB, Italy, 20052 Azienda Ospedaliera "G. Salvini" - P.O. Garbagnate Milanese Garbagnate Milanese, MI, Italy, 20020 Ospedale Civile Di Legnano Legnano, MI, Italy, 20025 IRCCS Fondazione San Raffaele Monte Tabor Milano, MI, Italy, 20132 IRCCS Istituto Nazionale dei Tumori Milano, MI, Italy, 20133 Azienda Ospedaliera Ospedale Ca' Granda Milano, MI, Italy, 20162 Fondazione Salvatore Maugeri Clinica del Lavoro e della Riabilitazione - Reparto Riabilitazione Oncologica Pavia, PV, Italy, 27100 Fondazione Salvatore Maugeri Clinica del Lavoro e della Riabilitazione - U.O. Oncologia Pavia, PV, Italy, 27100 Azienda Ospedaliera della Valtellina e della Valchiavenna - P.O. Sondrio Sondrio, SO, Italy, 23100 Collapse << |
| NCT01037842 | Type 2 Diabetes | Phase 3 | Completed | - | Korea, Republic of ... more >> Pusan National University College of Medicine Busan, Korea, Republic of Kyungpook National University School of Medicine Daegu, Korea, Republic of Chungnam National University College of Medicine Daejeon, Korea, Republic of Chonnam National University College of Medicine Gwangju, Korea, Republic of Chonbuk National University College of Medicine Jeonju, Korea, Republic of Eulji University School of Medicine Seoul, Korea, Republic of Hallym University College of Medicine Seoul, Korea, Republic of Korea University College of Medicine Seoul, Korea, Republic of Seoul St. Mary's Hospital Seoul, Korea, Republic of SungKyunKwan University School of Medicine Seoul, Korea, Republic of The Catholic University of Korea Seoul, Korea, Republic of Yonsei University College of Medicine Seoul, Korea, Republic of Uijeongbu St. Mary Hospital Uijeongbu, Korea, Republic of Collapse << |
| NCT01195259 | - | - | Completed | - | - |
| NCT00359112 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 3 | Completed | - | - |
| NCT01135433 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Chubu, Japan Chugoku, Japan Hokkaido, Japan Kansai, Japan Kantou, Japan Kyushu, Japan Touhoku, Japan Collapse << |
| NCT02744976 | Coronary Artery Disease ... more >> Atherosclerosis Prediabetic State Collapse << | Phase 4 | Recruiting | December 2020 | Latvia ... more >> Pauls Stradins Clinical University hospital Recruiting Riga, Latvia Collapse << |
| NCT02911792 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | April 2019 | United States, Florida ... more >> Sanford Burnham Prebys Medical Discovery Institute Not yet recruiting Orlando, Florida, United States, 32827 Contact: Richard Pratley, MD 407-745-2145 rpratley@SBPdiscovery.org United States, Illinois Northwestern Medical School Not yet recruiting Chicago, Illinois, United States, 60611 Contact: Mark Molitch, MD 312-503-4130 molitch@northwestern.edu The University of Chicago Not yet recruiting Chicago, Illinois, United States, 60637 Contact: George Bakris, MD 773-702-7936 gbakris@medicine.bsd.uchicago.edu United States, Texas The University of Texas Health Science Center at San Antonio Not yet recruiting San Antonio, Texas, United States, 78229 Contact: Ralph A DeFronzo, MD 210-567-6691 defronzo@uthscsa.edu Contact: Monica Palomo, BS 210-567-6710 palomom@uthscsa.edu Sub-Investigator: Ralph A DeFronzo, MD Principal Investigator: Eugenio Cersosimo, MD University Health Systems Texas Diabetic Institute Recruiting San Antonio, Texas, United States Contact: Eugenio Cersosimo, MD 210-358-7200 cersosimo@uthscsa.edu United States, Washington Sacred Heart Medical Center Not yet recruiting Spokane, Washington, United States, 99204 Contact: Katherine Tuttle, MD 509-340-0930 Australia, Victoria Baker Medical Research Institute and Alfred Hospital Not yet recruiting Melbourne, Victoria, Australia Contact: Mark Cooper, MD 61385321362 mark.cooper@bakeridi.edu.au Collapse << |
| NCT02457286 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Collapse << | Phase 1 | Withdrawn(patients do not want... more >> to participate) Collapse << | - | United States, New York ... more >> North Shore Hospital Manhasset, New York, United States, 11030 Collapse << |
| NCT00666458 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00666458 | - | - | Completed | - | - |
| NCT01459809 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00396357 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative Site Investigative Centers, Germany Collapse << |
| NCT01089179 | Type 2 Diabetes | Phase 1 | Completed | - | India ... more >> Torrent Research Centre Bhat, Gujarat, India, 382 428 Collapse << |
| NCT02813863 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02866786 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | India ... more >> Department of Obstetrics & Gynecology Cuttack, Odisha, India, 753007 Collapse << |
| NCT03499704 | Diabetes Mellitus, Type 2 | Phase 4 | Not yet recruiting | December 31, 2020 | - |
| NCT01984489 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT02039258 | Healthy Volunteers | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT00376038 | Type 2 Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | Mexico ... more >> GSK Investigational Site Mexico, D.F., Mexico, 14050 Collapse << |
| NCT03670043 | Type2 Diabetes Mellitus | Phase 4 | Not yet recruiting | November 2020 | - |
| NCT02172248 | Healthy | Phase 1 | Completed | - | - |
| NCT01890629 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Severance hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02611518 | Healthy | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany Collapse << |
| NCT01809327 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02140788 | - | - | Terminated(PI left institution... more >>) Collapse << | - | - |
| NCT00819741 | - | - | Completed | - | - |
| NCT02140788 | Schizophrenia ... more >> Schizo-affective Disorder Collapse << | Phase 4 | Terminated(PI left institution... more >>) Collapse << | - | - |
| NCT02637037 | Bioequivalence ... more >> Fixed Dose Combination Tablets Healthy Male and Female Subjects Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00819741 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Fujian ... more >> Novo Nordisk Investigational Site Fuzhou, Fujian, China, 350001 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210009 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210012 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210029 Novo Nordisk Investigational Site Suzhou, Jiangsu, China, 215004 Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200001 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200025 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200092 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200120 China, Zhejiang Novo Nordisk Investigational Site Hangzhou, Zhejiang, China, 310003 Novo Nordisk Investigational Site Wenzhou, Zhejiang, China, 325000 China Novo Nordisk Investigational Site Hangzhou, China, 310016 Novo Nordisk Investigational Site Nantong, China, 226001 Novo Nordisk Investigational Site Shanghai, China, 200040 Collapse << |
| NCT02508168 | Healthy Volunteers | Phase 1 | Completed | - | - |
| NCT02637037 | - | - | Completed | - | - |
| NCT01318109 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | - |
| NCT01809327 | - | - | Completed | - | - |
| NCT02190838 | Melanoma | Phase 2 | Unknown | December 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Ambulatory Chemotherapy Department Recruiting St. Petersburg, Russian Federation, 197758 Contact: Elena V. Tkachenko, MD +78124399575 elenatkachen@yandex.ru Principal Investigator: Elena V. Tkachenko, MD N.N. Petrov Research Institute of Oncology Chemotherapy and Innovative Technologies Department Recruiting St. Petersburg, Russian Federation, 197758 Contact: Aleksei V. Novik, MD, PhD +78124399505 anovik@list.ru Principal Investigator: Svetlana A. Protsenko, MD, PhD, DSc N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting St.Petersburg, Russian Federation, 191124 Contact: Tatiana Y. Semiglazova, MD, PhD, DSc tsemiglazova@mail.ru Principal Investigator: Tatiana V Semiglazova, MD, PhD, DSc Collapse << |
| NCT02909933 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02431676 | Breast Cancer ... more >> Prostate Cancer Lung Cancer Colon Cancer Melanoma of Skin Endometrial Cancer Liver Cancer Pancreatic Cancer Rectal Cancer Kidney Cancer Other Solid Malignant Tumors Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, Maryland ... more >> Johns Hopkins ProHealth Baltimore, Maryland, United States, 21207 Collapse << |
| NCT02471404 | Type 2 Diabetes Mellitus ... more >> Inadequate Glycaemic Control Collapse << | Phase 4 | Completed | - | - |
| NCT01318109 | - | - | Completed | - | - |
| NCT01352026 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 2 | Withdrawn(Several therapeutic ... more >>have been developed making more difficult the eligibility of patients - Recruitment of new centers wasn't possible due to competing trials) Collapse << | - | France ... more >> CHU de Nantes Nantes, France, 44093 CHU de Bordeaux Pessac, France, 33604 Collapse << |
| NCT02506777 | Breast Cancer | Phase 2 | Recruiting | August 2017 | Russian Federation ... more >> N.N. Petrov Research Institute of Oncology Clinical Diagnostic Department Recruiting Saint - Petersburg, Russian Federation, 191124 Contact: Tatiana Y Semiglazova, MD, PhD, DSc +79219468072 tsemiglazova@mail.ru Contact: Michael A Osipov, MD +79052075653 Ocipovmixail@mail.ru Principal Investigator: Tatiana Y Semiglazova, MD, PhD, DSc Sub-Investigator: Petr V Krivorotko, MD, PhD, DSc Collapse << |
| NCT02167061 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center, Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT02109679 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1307.7.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01679574 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Assiut University Assiut, Egypt, 11111 Collapse << |
| NCT00948402 | Polycystic Ovarian Syndrome ... more >> Insulin Sensitivity Collapse << | Phase 3 | Completed | - | - |
| NCT02960711 | Metabolic Syndrome | Phase 3 | Unknown | July 2018 | Italy ... more >> Fondazione IRCCS Istituto Nazionale Tumori Recruiting Milan, Italy, 20133 Contact: Franco Berrino, MD +390223903515 Franco.berrino@istitutotumori.mi.it Contact: Patrizia Pasanisi, MD +390223903513 trialcenter@istitutotumori.mi.it Collapse << |
| NCT03709147 | Advanced LKB1-inactive Lung Ad... more >>enocarcinoma Collapse << | Phase 2 | Not yet recruiting | September 10, 2023 | Italy ... more >> Marina Chiara Garassino Not yet recruiting Milan, Italy, 20133 Contact: MARINA CHIARA GARASSINO, MD 0223903813 ext +39 marina.garassino@istitutotumori.mi.it Collapse << |
| NCT00617240 | - | - | Completed | - | - |
| NCT01693289 | Focus of Study | Phase 1 | Completed | - | Egypt ... more >> Zagazig University Zagazig, Egypt Collapse << |
| NCT00762957 | Diabetes Mellitus | Phase 3 | Terminated(Hepatic Safety Sign... more >>al Identified.) Collapse << | - | - |
| NCT02429258 | Type II Diabetes | Phase 4 | Completed | - | - |
| NCT01437813 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Investigación Farmacológica Y Biofarmaceutica, S.A. de C.V. México, Mexico, 14610 Collapse << |
| NCT00617240 | Weight Gain | Phase 2 | Completed | - | United States, North Carolina ... more >> University of North Carolina, Department of Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00449605 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Company decision ta... more >>ken in light of demands by certain national health authorities) Collapse << | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Chile Sanofi-Aventis Administrative Office Santiago, Chile Sanofi-aventis adminsitrative office Santiago, Chile Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark Finland Sanofi-Aventis Administrative Office Helsinki, Finland Hungary Sanofi-Aventis Administrative Office Budapest, Hungary India Sanofi-Aventis Administrative Office Mumbai, India Italy Sanofi-Aventis Administrative Office Milan, Italy Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Puerto Rico Sanofi-Aventis Administrative Office Puerto Rico, Puerto Rico Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Collapse << |
| NCT02429258 | - | - | Completed | - | - |
| NCT00934817 | Healthy | Phase 1 | Completed | - | - |
| NCT03253562 | Reducing the Elevated Blood Pr... more >>essure for Diabetic Hypertensive Patients Collapse << | Phase 4 | Completed | - | Egypt ... more >> National institute of diabetes and endocrinology Cairo, Egypt, 11311 Collapse << |
| NCT01702298 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02488564 | HER-2 Positive Breast Cancer | Phase 2 | Active, not recruiting | October 2019 | Italy ... more >> IRST Oncologia medica Meldola, FC, Italy, 47014 Oncologia Medica AOU Ferrara Ferrara, FE, Italy, 44124 Oncologia Medica, Policlinico di Modena Modena, MO, Italy, 41100 Oncologia Medica Ospedale Guglielmo da Saliceto Parma, PR, Italy, 29121 Oncologia , IRCCS azienda ospedaliera S.Maria Nuova Reggio Emilia, RE, Italy, 42123 U.O Oncologia AUSLdella Romagna Rimini, RN, Italy, 47923 Day Hospital Oncologico, Ospedale Guastalla Guastalla, Italy, 42016 Oncologia Medica AUSL Imola Imola, Italy, 40133 Oncologia Medica AOU Parma Parma, Italy, 43100 Collapse << |
| NCT02552355 | Physical Activity | Phase 2 | Completed | - | United States, Colorado ... more >> Colorado State University, Dept. of Health and Exercise Science Fort Collins, Colorado, United States, 80523-1582 Collapse << |
| NCT01666730 | Acinar Cell Adenocarcinoma of ... more >>the Pancreas Duct Cell Adenocarcinoma of the Pancreas Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Active, not recruiting | November 2018 | United States, Ohio ... more >> University Hospitals Cleveland Medical Center, Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT00883259 | Obesity Polyc... more >>ystic Ovary Syndrome Gestational Diabetes Collapse << | Phase 4 | Unknown | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 "Pugliese" Hospital Not yet recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39-0961-883234 angela.falbo@libero.it University of Catanzaro, Italy Catanzaro, Italy, 88100 Collapse << |
| NCT01675310 | Pregestational Obesity (BMI > ... more >>27kg/m2) History of Polycystic Ovary Syndrome Collapse << | Phase 4 | Completed | - | Mexico ... more >> National Institute of Perinatology Mexico, Mexico, 11000 Collapse << |
| NCT01856907 | - | - | Completed | - | - |
| NCT01702298 | - | - | Completed | - | - |
| NCT00911313 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Egypt ... more >> Mansoura University Hospitals,OB/GYN department Mansoura, Dakahlia, Egypt, 35511 Collapse << |
| NCT00390273 | Hyperlactatemia | Phase 1 Phase 2 | Completed | - | France ... more >> Centre Hospitalier Universitaire de Grenoble Grenoble cedex 9, France, 38043 Collapse << |
| NCT03629054 | Healthy | Phase 1 | Completed | - | Germany ... more >> Humanpharmakologisches Zentrum Biberach Biberach, Germany, 88397 Collapse << |
| NCT01856907 | Disorder of Glucose Regulation | Phase 4 | Completed | - | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70817 Collapse << |
| NCT00560690 | Hepatitis C | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14114 Collapse << |
| NCT02234440 | NASH Related Cirrhosis | Not Applicable | Completed | - | India ... more >> Institute of Liver & Biliary Sciences New Delhi, Delhi, India, 110070 Collapse << |
| NCT01545388 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02798172 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT03336840 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | June 2019 | China, Jiangsu ... more >> Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Recruiting Nanjing, Jiangsu, China, 210008 Contact: Dalong Zhu, MD,PhD 86-25-83-105302 zhudldr@gmail.com Collapse << |
| NCT01545388 | - | - | Completed | - | - |
| NCT03151772 | Glioblastoma | Early Phase 1 | Recruiting | June 2019 | Sweden ... more >> Sahlgrenska University Hospital Recruiting Göteborg, Sweden Contact: Louise Carstam, MD Collapse << |
| NCT01775813 | Obesity Insul... more >>in Resistance Gonadal Dysfunction Type 2 Diabetes Collapse << | Phase 4 | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado Aurora, Colorado, United States, 80045 Collapse << |
| NCT01969318 | Type 2 Diabetes | Phase 2 | Completed | - | China ... more >> Chinese PLA General Hospital Beijing, China Collapse << |
| NCT00367055 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT03651895 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Early Phase 1 | Not yet recruiting | August 31, 2020 | - |
| NCT02948283 | Anemia Fatigu... more >>e Fever Lymphadenopathy Lymphocytosis Night Sweats Recurrent Chronic Lymphocytic Leukemia Recurrent Plasma Cell Myeloma Refractory Chronic Lymphocytic Leukemia Refractory Plasma Cell Myeloma Splenomegaly Thrombocytopenia Weight Loss Collapse << | Phase 1 | Active, not recruiting | October 2019 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << |
| NCT00367055 | - | - | Completed | - | - |
| NCT02609815 | Diabetes | Not Applicable | Recruiting | December 31, 2019 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Tae Jung Oh, MD, PHD 82-31-787-7078 ohtjmd@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT00942123 | - | - | Completed | - | Italy ... more >> Ospedale Maggiore Policlinico Milano, MI, Italy, 20122 Collapse << |
| NCT00900146 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Terminated(Numerically modest ... more >>lowering of HbA1c with canakinumab in combination with metformin was inadequate to continue patients with T2DM into Period IV of this study.) Collapse << | - | - |
| NCT02497638 | Prostate Cancer | Phase 2 | Not yet recruiting | December 2022 | Canada, Ontario ... more >> Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Principal Investigator: Robert Hamilton, MD, MPH, FRCSC Principal Investigator: Neil Fleshner, MD, MPH, FRCSC Collapse << |
| NCT01535677 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT01612858 | Lipodystrophy ... more >> HIV Infection Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT00900146 | - | - | Terminated(Numerically modest ... more >>lowering of HbA1c with canakinumab in combination with metformin was inadequate to continue patients with T2DM into Period IV of this study.) Collapse << | - | - |
| NCT00543361 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT01612858 | - | - | Completed | - | - |
| NCT01410604 | Obesity | Phase 4 | Completed | - | Mexico ... more >> Hospital Infantil de México Federico Gómez México, Distrito Federal, Mexico, 06720 Hospital Regional de Alta Especialidad del Bajío León, Guanajuato, Mexico, 37670 Collapse << |
| NCT01410604 | - | - | Completed | - | - |
| NCT02063802 | Obesity | Phase 2 | Unknown | May 2014 | Mexico ... more >> General Hospital of Mexico Recruiting Mexico, Distrito Federal, Mexico, 06726 Contact: Nayely G Garibay, Pediatrician +52 (55) 59-48-26-37 gngaribay@hotmail.com Contact: Mayra A Bustos, Doctor +52 7773406509 mayibe2609@gmail.com Collapse << |
| NCT02231931 | Healthy | Phase 1 | Completed | - | Germany ... more >> 352.2082.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01658514 | - | - | Completed | - | - |
| NCT00414245 | Gestational Diabetes ... more >> Diabetes Mellitus Type 2 Collapse << | Phase 3 | Unknown | - | Israel ... more >> Division of Obstetrics and Gynecology, Soroka University Medical Center Not yet recruiting Beer-Sheva, Israel Contact: Boaz Sheizaf, MD 972-54-2401191 bsheizaf@bgu.ac.il Principal Investigator: Boaz Sheizaf, MD Collapse << |
| NCT02283554 | Periodontitis | Phase 2 | Completed | - | - |
| NCT01204580 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Indonesia ... more >> Sanofi-Aventis Administrative Office Jakarta, Indonesia Collapse << |
| NCT01474018 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, Texas ... more >> UT Southwestern Medical Center Dallas, Texas, United States, 75390 UT Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT03088670 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Russian Federation ... more >> Non-state healthcare Organization "Road Clinical Hospital at the station Chelyabinsk Open Joint Stock Company" Russian Railways Chelyabinsk, Russian Federation, 454048 State Budget Healthcare Institution of Moscow "Endocrinology Dispensary" Moscow, Russian Federation, 119034 SEI HPE "First Moscow State Medical University n.a. I.M. Sechenov of Ministry of Health and Social Development of the Russian Federation", Chair of faculty therapy #2 based at SHI of Moscow "City clinical hospital #61" Moscow, Russian Federation, 119048 State Budget Educational Institution higher professional education "1st Moscow State Medical University n.a. I.M. Sechenov of the Ministry of Healthcare of Russian Federation" Moscow, Russian Federation, 119435 Federal State Institution "Clinical Hospital № 1" of the Office of the President of the Russian Federation Moscow, Russian Federation, 121352 State Budget Healthcare Institution of Moscow city "City Clinical Hospital # 71 by the Department of Healthcare in Moscow" Moscow, Russian Federation, 121374 State Budget Educational Institution of higher professional education "1st Moscow State Medical University n.a. I.M. Sechenov of the Ministry of Healthcare of Russian Federation" based on Endocrinology department of Hospital № 67 n.a L A Vorohobova Moscow, Russian Federation, 123423 SEI APE "Russian Medical Academy of Postgraduate Education of Roszdrav", Chair of endocrinology and diabetology with course of endocrine surgery based at FSI "Central Clinical hospital of civil aviation" Moscow, Russian Federation, 125367 State budgetary institution "Nizhny Novgorod Regional Clinical Hospital n.a Semashko Nizhny Novgorod, Russian Federation, 603126 State Health Care Institution "Perm Regional Hospital for War Veterans" Perm, Russian Federation, 614097 State Educational Institution of Higher Professional Education "Perm State Medical Academy n.a. academician EA Wagner's "Ministry of Health of the Russian Federation on the basis of Department of Endocrinology Regional Clinical Hospital Perm, Russian Federation, 614990 SHI "Republic Hospital n.a. V.A. Baranov" Petrozavodsk, Russian Federation, 185019 State budgetary institution of Ryazan region "City Clinical Hospital № 11" Ryazan, Russian Federation, 390037 St. Petersburg State healthcare institution "City Outpatient Clinic #37" Saint Petersburg, Russian Federation, 191119 FSHI "Clinical Hospital #122 n.a. L.G. Sokolov of FMBA of Russia", Internal medicine department Saint Petersburg, Russian Federation, 194291 State Health Care Institution Leningrad Regional Hospital Saint Petersburg, Russian Federation, 194291 Autonomous Noncommercial Organization "Medical Centre "XXI сentury" Saint Petersburg, Russian Federation, 194354 St. Petersburg State healthcare institution "City Hospital of Saint Martyr Elizaveta" Saint Petersburg, Russian Federation, 195257 FSMEI HPE "Military Medical Academy n.a. S.M. Kirov" of the Ministry of Defence of the Russian Federation, 1st Internal medicine Chair of advanced training Saint Petersburg, Russian Federation, 198013 "Diabetes Center "LLC Samara, Russian Federation, 443041 State Budget Educational Institution higher professional education "Saratov State Medical University n.a. V.I. Razumovsky" of the Ministry of Healthcare of Russian Federation based on Clinical Hospital n.a S.R. Mirotvortsev Saratov, Russian Federation, , 410054 Municipal health care institution "City polyclinic №20 Saratov, Russian Federation, 410018 SBEI HPE "Smolensk State Medical Academy" of the Ministry of Health and Social Development of the Russian Federation, Center of clinical research of diagnostic and medicinal products, Chair of clinical pharmacology Smolensk, Russian Federation, 214019 State budget educational institution of higher education "Smolensk State Medical Academy," of Ministry of Health and Social Development of the Russian Federation on the basis of sanatorium SSMA Smolensk, Russian Federation, 214019 State autonomous healthcare institution of Yaroslavl Region "Сlinical hospital for emergency medical care n. a. N.V. Solovyov" Yaroslavl, Russian Federation, 150003 Municipal clinical health institution Health Service Novo-Yaroslavl Oil Refinery Yaroslavl, Russian Federation, 150023 SHI of Yaroslavl region "Regional Clinical Hospital", Endocrinology department Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT00975065 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals Seoul, Korea, Republic of Collapse << |
| NCT01286233 | - | - | Unknown | September 2016 | - |
| NCT00968812 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00968812 | - | - | Completed | - | - |
| NCT01167738 | Pancreatic Cancer | Phase 2 | Terminated(concern of detrimen... more >>tal effect) Collapse << | - | Italy ... more >> Istituto Scientifico H. San Raffaele Milan, Italy, 20132 Collapse << |
| NCT03352869 | Polycystic Ovary Syndrome ... more >> Overweight and Obesity Disorder of Glucose Regulation Collapse << | Phase 4 | Recruiting | July 31, 2018 | China ... more >> Renji Hospital Department of Endocrinology and Metabolism Recruiting Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Contact: Jing Ma, MD +86-15800983436 cherry1996@live.cn Collapse << |
| NCT01606007 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01658514 | Renal Insufficiency | Phase 2 | Completed | - | - |
| NCT02695810 | Prediabetic State ... more >> Obesity Collapse << | Phase 2 | Recruiting | February 2019 | Denmark ... more >> Steno Diabetes Center A/S Recruiting Gentofte, Denmark, 2820 Contact: Kristine Færch, PhD +4530791461 krif@steno.dk Contact: Marit E Jørgensen, PhD +4530756008 maej@steno.dk Sub-Investigator: Hanan Amadid, MD Sub-Investigator: Lea B Nielsen, MSc Collapse << |
| NCT01717911 | Type 2 Diabetes | Phase 4 | Unknown | December 2018 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD 886-2-28757515 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT01267448 | Diabetes Mellitus Type 2 ... more >> Severe Hyperglycemia - Blood Glucose Level >300mg/dl. Collapse << | Phase 4 | Unknown | December 2012 | United States, Illinois ... more >> John Stroger Hospital of Cook County Not yet recruiting Chicago, Illinois, United States, 60612 Contact: Ambika Babu, M.D. 312-864-0543 Ambika_Babu@rush.edu Contact: Leon Fogelfeld, M.D. 312-864-0539 lfogelfe@cchil.org Principal Investigator: Ambika Babu, M.D. John Stroger Hospital of Cook County Recruiting Chicago, Illinois, United States, 60612 Contact: Ambika Babu, M.D. 312-864-0543 aamblee@hotmail.com Contact: Leon Fogelfeld, M.D. 312-864-0539 lfogelfe@cchil.org Principal Investigator: Ambika Babu, M.D. Collapse << |
| NCT00292799 | Anovulation O... more >>besity Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Sheffield Teaching Hospitals NHS Trust, Jessop Wing Sheffield, United Kingdom, S10 2SF Collapse << |
| NCT01606007 | - | - | Completed | - | - |
| NCT01274130 | Healthy Adult | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00499707 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02202161 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21225 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT00451568 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Odense University Hospital Odense, Denmark Collapse << |
| NCT01013051 | - | - | Completed | - | Canada, Alberta ... more >> University of Alberta Hospital Clinical Investigation Unity Edmonton, Alberta, Canada, T6G2B7 Collapse << |
| NCT02202161 | - | - | Completed | - | - |
| NCT01998113 | - | - | Completed | - | Spain ... more >> Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT01101438 | Breast Cancer | Phase 3 | Active, not recruiting | February 2022 | - |
| NCT01825798 | Overweight Au... more >>tism Spectrum Disorder Collapse << | Phase 3 | Completed | - | United States, Ohio ... more >> Ohio State University/Nationwide Children's Hospital Columbus, Ohio, United States, 43210 United States, Pennsylvania University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15203 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37212 Canada, Ontario Holland Bloorview Kids Rehabilitation Hospital Toronto, Ontario, Canada, M4G 1R8 Collapse << |
| NCT02562664 | Reproductive Endocrinology ... more >> Polycystic Ovarian Syndrome Collapse << | Phase 2 | Completed | - | - |
| NCT00770445 | Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Lichtenfels, Bavaria, Germany Frankfurt, Hessen, Germany Kassel, Hessen, Germany Wiesbaden, Hessen, Germany Bad Oeynhausen, North Rhine-Westphalia, Germany Dinslaken, North Rhine-Westphalia, Germany Duisburg, North Rhine-Westphalia, Germany Essen, North Rhine-Westphalia, Germany Münster, North Rhine-Westphalia, Germany Wuppertal, North Rhine-Westphalia, Germany Mainz, Rheinland-Pfalz, Germany Diez, Rhineland-Palatinate, Germany Landau, Rhineland-Palatinate, Germany Dresden, Saxony, Germany Leipzig, Saxony, Germany Jena, Thüringen, Germany Berlin, Germany Hamburg, Germany Collapse << |
| NCT02434744 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Alabama ... more >> Achieve Clinical Research, LLC Birmingham, Alabama, United States, 35216 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 National Research Institute Los Angeles, California, United States, 90057 Infosphere Clinical Research, Inc West Hills, California, United States, 91307 United States, Florida Clinical Research of South Florida Coral Gables, Florida, United States, 33134 Med Research of Florida, LLC Miami, Florida, United States, 33186 United States, North Carolina High Point Clinical Trials Center High Point, North Carolina, United States, 27265 United States, South Carolina Coastal Carolina Research Center Mt. Pleasant, South Carolina, United States, 29464 United States, Texas Clinical Trial Network Houston, Texas, United States, 77074 United States, Virginia Clinical Research Associates of Tidewater Norfolk, Virginia, United States, 23507 Collapse << |
| NCT02531308 | Diffuse Large B-Cell Lymphoma | Phase 2 | Terminated(poor accrual) | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT01825798 | - | - | Completed | - | - |
| NCT02133118 | - | - | Completed | - | Netherlands ... more >> Isala Zwolle, Overijssel, Netherlands, 8000 AB Collapse << |
| NCT01704261 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01704261 | - | - | Completed | - | - |
| NCT03204071 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT00382096 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers, Germany Collapse << |
| NCT02531308 | - | - | Terminated(poor accrual) | - | - |
| NCT00712673 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Australia sanofi-aventis Australia & New Zealand administrative office Macquarie Park, Australia Canada Sanofi-Aventis Administrative Office Laval, Canada Chile Sanofi-Aventis Administrative Office Santiago, Chile Croatia Sanofi-Aventis Administrative Office Zagreb, Croatia Czech Republic Sanofi-Aventis Administrative Office Praha, Czech Republic Germany Sanofi-Aventis Administrative Office Berlin, Germany Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Morocco Sanofi-Aventis Administrative Office Casablanca, Morocco Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation South Africa Sanofi-Aventis Administrative Office Midrand, South Africa Spain Sanofi-Aventis Administrative Office Barcelona, Spain Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Venezuela Sanofi-Aventis Administrative Office Caracas, Venezuela Collapse << |
| NCT03267576 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Mexico ... more >> Consultorio Privado Guadalajara, Mexico, 44150 Investigación Clínica Especializada Guadalajara, Mexico, 44600 Consultorio Privado en Unidad de Patología Clínica Guadalajara, Mexico, 44650 Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán Mexico City, Mexico, 14080 Hospital Universitario 'Dr. Jose Eleuterio Gonzalez' Monterrey, Mexico, 64460 Collapse << |
| NCT00131664 | - | - | Completed | - | - |
| NCT00712673 | - | - | Completed | - | - |
| NCT00131664 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Canada, Ontario ... more >> Canadian Heart Research Centre Toronto, Ontario, Canada, m5b 2p9 Collapse << |
| NCT01699932 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Lebanon ... more >> Investigational Site Number 422-002 Beirut, Lebanon Investigational Site Number 422-001 Hazmieh, Lebanon Russian Federation Investigational Site Number 643001 Samara, Russian Federation Investigational Site Number 643002 St-Petersburg, Russian Federation Investigational Site Number 643-03 St.-Petersburg, Russian Federation Ukraine Investigational Site Number 804003 Chernivtsi, Ukraine, 58022 Investigational Site Number 804008 Donetsk, Ukraine, 83003 Investigational Site Number 804004 Donetsk, Ukraine, 83059 Investigational Site Number 804001 Donetsk, Ukraine, 83099 Investigational Site Number 804010 Odessa, Ukraine Investigational Site Number 804006 Poltava, Ukraine, 36011 Investigational Site Number 804007 Vinnytsya, Ukraine, 21010 Investigational Site Number 804002 Vinnytsya, Ukraine, 21029 Collapse << |
| NCT02065687 | Endometrial Adenocarcinoma ... more >> Endometrial Clear Cell Adenocarcinoma Endometrial Serous Adenocarcinoma Endometrial Undifferentiated Carcinoma Recurrent Uterine Corpus Carcinoma Stage III Uterine Corpus Cancer AJCC v7 Stage IIIA Uterine Corpus Cancer AJCC v7 Stage IIIB Uterine Corpus Cancer AJCC v7 Stage IIIC Uterine Corpus Cancer AJCC v7 Stage IV Uterine Corpus Cancer AJCC v7 Stage IVA Uterine Corpus Cancer AJCC v7 Stage IVB Uterine Corpus Cancer AJCC v7 Collapse << | Phase 2 Phase 3 | Active, not recruiting | - | - |
| NCT01590771 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02395237 | Healthy | Phase 1 | Unknown | May 2015 | Jordan ... more >> Arab Pharmaceutical industry Consulting/ Pharmaceutical Research Unit Amman, Jordan, 00962 Collapse << |
| NCT01023581 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02477969 | Type 2 Diabetes Mellitus | Phase 3 | Unknown | June 2017 | China, Shanghai ... more >> Shanghai sixth People's Hospital Shanghai, Shanghai, China, 200233 Collapse << |
| NCT00241605 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02249910 | Diabetes Heal... more >>thy Collapse << | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany, 14050 Collapse << |
| NCT01590771 | - | - | Completed | - | - |
| NCT00437333 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT02614859 | Cancer of Prostate | Phase 2 | Recruiting | April 2022 | United States, Maryland ... more >> National Cancer Institute Recruiting Bethesda, Maryland, United States, 20892-9760 Contact: Marijo Bilusic, MD marijo.bilusic@nih.gov United States, Pennsylvania Fox Chase Cancer Center - Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT01871415 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | China ... more >> Shanghai, China, 200003 ShenYang, China, 110004 Shiyan, China, 442000 Suzhou, China, 215004 Korea, Republic of Gyeonggi-do, Korea, Republic of, 463-712 Incheon, Korea, Republic of, 405-760 Seoul, Korea, Republic of, 150-950 Collapse << |
| NCT01136512 | - | - | Completed | - | United States, New York ... more >> Staten Island University Hospital Staten Island, New York, United States, 10305 Collapse << |
| NCT01023581 | - | - | Completed | - | - |
| NCT01561976 | - | - | Completed | - | - |
| NCT00701090 | Type 2 Diabetes Mellitus, Non ... more >>Insulin Dependent Diabetes Mellitus, Non-Insulin-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00303537 | Fatty Liver | Phase 2 Phase 3 | Unknown | June 2008 | Norway ... more >> Haukeland Universitetssykehus Bergen, Norway Aker University Hospital Oslo, Norway Akershus University Hospital Oslo, Norway Universitetssykehuset i Nord-Norge Tromsø, Norway Collapse << |
| NCT01947153 | Healthy | Phase 1 | Completed | - | Russian Federation ... more >> 1288.21.1 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation Collapse << |
| NCT00701090 | - | - | Completed | - | - |
| NCT01947153 | - | - | Completed | - | - |
| NCT03189407 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | - |
| NCT01336673 | - | - | Terminated(The study was sched... more >>uled for completion February 2013. Because of difficulties encountered in completing the trial the study was terminated by sponsor June 2013.) Collapse << | - | Thailand ... more >> Faculty of Medicine Vajira Hospital Bangkok, Thailand Collapse << |
| NCT01524705 | Type 2 Diabetes | Phase 4 | Completed | - | United States, California ... more >> So Calif. Permanente Medical Group San Diego, California, United States, 92109 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Georgia Atlanta Diabetes Associates Atlanta, Georgia, United States, 30309 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Minnesota International Diabetes Center Minneapolis, Minnesota, United States, 55416 United States, Missouri Washington University St. Louis, Missouri, United States, 63110 United States, New York Kaledia Health of Western New York Buffalo, New York, United States, 14209 United States, North Carolina Diabetes Care Center Durham, North Carolina, United States, 27713 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239 United States, Vermont University of Vermont Colchester, Vermont, United States, 05446 United States, Washington University of Washington Seattle, Washington, United States, 98105 Washington State University Spokane, College of Pharmacy Spokane WA 99202 USA Spokane, Washington, United States, 99202 Collapse << |
| NCT02719756 | Diabetes Mellitus ... more >> Hypoglycemic Episodes Collapse << | Phase 4 | Unknown | December 2016 | Russian Federation ... more >> Research Clinical Centre of the Russian Railways, JSC Not yet recruiting Moscow, Russian Federation, 125993 Contact: Alexander Ametov, prof., MD (499)1510951 ext 8107 alexander.ametov@gmail.ru Contact: Viktoria Blagova, PhD cand. (499)6130363 ext 8107 vika-blagova@mail.ru Collapse << |
| NCT01505426 | Type II Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Busan, Korea, Republic of Daegu, Korea, Republic of Daejeon, Korea, Republic of Gwangju, Korea, Republic of Incheon, Korea, Republic of Jeonju, Korea, Republic of Seoul, Korea, Republic of Wonju, Korea, Republic of Taiwan Kaohsiung, Taiwan Taichung, Taiwan Tainan, Taiwan Taipei, Taiwan Collapse << |
| NCT01965756 | Alzheimer's Disease ... more >> Vascular Dementia Dementia Memory Impairment Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania, Penn Memory Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01965756 | - | - | Completed | - | - |
| NCT01034111 | - | - | Completed | - | - |
| NCT01034111 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00327015 | Diabetes | Phase 3 | Completed | - | - |
| NCT01561976 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT00327015 | - | - | Completed | - | - |
| NCT02111967 | - | - | Completed | - | Switzerland ... more >> Pharmaceutical Care Research Group Basel, Switzerland, 4056 Collapse << |
| NCT02087826 | Genetics Meta... more >>bolism Type 2 Diabetes Collapse << | Not Applicable | Recruiting | January 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Varinderpal Kaur 617-643-5423 sigma2mgh@partners.org Principal Investigator: Jose C Florez, MD, PhD Sub-Investigator: Jennifer Todd, MD Sub-Investigator: Jamie L Garry, MS, RD Collapse << |
| NCT00575588 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02342925 | Healthy Volunteer | Phase 1 | Completed | - | United Kingdom ... more >> Leeds, United Kingdom, LS2 9LH Collapse << |
| NCT01691846 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00383578 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Investigative Centers Nurnberg, Germany Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT00650234 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT01512979 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT00575588 | - | - | Completed | - | - |
| NCT01512979 | - | - | Completed | - | - |
| NCT00659568 | Breast Cancer ... more >> Endometrial Cancer Kidney Cancer Lung Cancer Lymphoma Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> London Regional Cancer Program at London Health Sciences Centre London, Ontario, Canada, N6A 4L6 Collapse << |
| NCT00519480 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33169 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 Argentina GSK Investigational Site Buenos Aires, Argentina, 1425 Germany GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT00648518 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00650312 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00922194 | Obesity Type ... more >>2 Diabetes Mellitus Collapse << | Not Applicable | Completed | - | India ... more >> Sircar Diabetes Clinic Lucknow, UP, India, 226006 Collapse << |
| NCT01677923 | Obesity | Phase 3 | Completed | - | Lithuania ... more >> Rasa Verkauskiene Kaunas, Eiveniu str. 2, Lithuania, LT50009 Collapse << |
| NCT00885378 | - | - | Completed | - | - |
| NCT00922194 | - | - | Completed | - | - |
| NCT00151411 | Polycystic Ovary Syndrome | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Penn State Milton S. Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT01582243 | - | - | Completed | - | - |
| NCT00885378 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00151411 | - | - | Completed | - | - |
| NCT01117103 | - | - | Completed | - | Singapore ... more >> Kevin Tan Clinic For Diabetes, Thyroid & Hormones Pte Ltd Mount Elizabeth, Singapore Collapse << |
| NCT02881528 | Prediabetic State | Phase 4 | Withdrawn(Poor Recruitment pri... more >>or to end of funding timelines.) Collapse << | - | United Kingdom ... more >> NHS Ayrshire & Arran Kilmarnock, Ayrshire, United Kingdom, KA2 0BE NHS Grampian Aberdeen, Grampian, United Kingdom, AB25 2ZG NHS Lanarkshire Wishaw, Lanarkshire, United Kingdom, ML2 0DP NHS Tayside Dundee, Tayside, United Kingdom, DD1 9SY NHS Greater Glasgow & Clyde Glasgow, United Kingdom, G3 8SJ Collapse << |
| NCT01582243 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Taiwan ... more >> Novartis Investigative Site Changhua, Taiwan, 500 Collapse << |
| NCT03129113 | Hepatic Steatosis ... more >> HIV-1-infection Collapse << | Phase 2 Phase 3 | Recruiting | November 2020 | United Kingdom ... more >> Royal Free Hospital Recruiting London, United Kingdom, NW3 2QG Contact: PI St Thomas' Hospital Recruiting London, United Kingdom, SE1 9RT Contact: PI King's College Hospital Recruiting London, United Kingdom, SE5 9RJ Contact: PI UCL Mortimer Market Centre Recruiting London, United Kingdom, WC1E 6JB Contact: PI Collapse << |
| NCT00618072 | - | - | Completed | - | - |
| NCT00993187 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT00993187 | - | - | Completed | - | - |
| NCT00618072 | Hyperinsulinemia ... more >> Insulin Resistance Obesity Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Albert Einstein College of Medicine Bronx, New York, United States, 10461 Collapse << |
| NCT01490658 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Novo Nordisk Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00889876 | Cardiovascular Diseases | Not Applicable | Unknown | December 2012 | Sweden ... more >> Karolinska Institutet, Karolinska University Hospital Huddinge Recruiting Stockholm, Sweden, 141 86 Contact: Maria Westerståhl, PhD +468 585 873 96 maria.westerstahl@ki.se Contact: Pernilla Hedvall, PhDstudent +468 585 873 96 pernilla.hedvall@ki.se Principal Investigator: Claude Marcus, Professor Collapse << |
| NCT03608358 | Type 2 Diabetes Mellitus | Phase 3 | Not yet recruiting | March 1, 2021 | China ... more >> Research Site Not yet recruiting Baotou, China, 014016 Research Site Not yet recruiting Beijing, China, 100039 Research Site Not yet recruiting Beijing, China, 100044 Research Site Not yet recruiting Beijing, China, 100101 Research Site Not yet recruiting Beijing, China, 100144 Research Site Not yet recruiting Beijing, China, 101199 Research Site Not yet recruiting Beijing, China, 101200 Research Site Not yet recruiting Changchun, China, 130021 Research Site Not yet recruiting Chenzhou, China Research Site Withdrawn Chongqing, China, 400014 Research Site Not yet recruiting Fuzhou, China, 350025 Research Site Withdrawn Guangzhou, China, 510000 Research Site Withdrawn Guangzhou, China, 510080 Research Site Not yet recruiting Guangzhou, China, 510515 Research Site Not yet recruiting Haerbin, China, 150001 Research Site Not yet recruiting Hefei, China, 230001 Research Site Not yet recruiting Hefei, China Research Site Not yet recruiting Hengyang, China, CN-421001 Research Site Not yet recruiting Hohhot, China, 010017 Research Site Not yet recruiting Huizhou, China, 516001 Research Site Not yet recruiting Jinan, China, 250001 Research Site Withdrawn Jinzhou, China, 121004 Research Site Withdrawn Nanchang, China, 330006 Research Site Not yet recruiting Nanjing, China, 210012 Research Site Not yet recruiting Nanjing, China, 210029 Research Site Not yet recruiting Nantong, China, 226001 Research Site Not yet recruiting Qingdao, China, 266003 Research Site Not yet recruiting Shanghai, China, 200240 Research Site Not yet recruiting Shantou, China, 515065 Research Site Not yet recruiting Shengyang, China, 110004 Research Site Not yet recruiting Tangshan, China Research Site Not yet recruiting Tianjin, China, 300211 Research Site Not yet recruiting Wuhan, China, 430033 Research Site Not yet recruiting Wuhan, China, CN-430022 Research Site Not yet recruiting Wulumuqi, China, 830054 Research Site Not yet recruiting Xian, China, 710061 Research Site Withdrawn Xining, China, 810001 Research Site Not yet recruiting Yancheng, China, 224005 Research Site Withdrawn Yinchuan, China, 750004 Research Site Not yet recruiting Yueyang, China, 414000 Research Site Not yet recruiting Zhuzhou, China, 412007 Thailand Research Site Not yet recruiting Bangkok, Thailand, 10400 Vietnam Research Site Not yet recruiting Ho Chi Minh, Vietnam, 10000 Research Site Not yet recruiting Ho Chi Minh, Vietnam Collapse << |
| NCT01076088 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00684528 | Diabetes Type 2 | Phase 3 | Unknown | December 2012 | Israel ... more >> Clalit Health services center Recruiting Tel Aviv, Israel, 62098 Contact: Zinger 972-5062-63652 Principal Investigator: Zinger Joel, MD Collapse << |
| NCT01171456 | Gestational Diabetes Mellitus | Not Applicable | Withdrawn(Insufficient funding... more >> for enrollment of patients. The invesitgators realized the study could not be performed.) Collapse << | - | United States, Utah ... more >> Intermountain Medical Center Murray, Utah, United States, 84107 Collapse << |
| NCT01076088 | - | - | Completed | - | - |
| NCT03068065 | Non-Alcoholic Fatty Liver Dise... more >>ase Type2 Diabetes Collapse << | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT00648492 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00649948 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT02535975 | Mild Graves' Ophthalmopathy | Not Applicable | Unknown | June 2017 | Korea, Republic of ... more >> Endocrinology, Internal Medicine, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 03722 Contact: Eun Jig Lee, MD +82-2-2228-1983 ejlee432@yuhs.ac Collapse << |
| NCT00502229 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Phase 4 | Withdrawn | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT00683657 | - | - | Completed | - | - |
| NCT00683657 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03202563 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | October 30, 2018 | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 06351 Collapse << |
| NCT02091336 | Gestational Diabetes | Phase 4 | Unknown | December 2016 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Recruiting Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT00169624 | Coronary and Peripheral Endoth... more >>elial Dysfunction Collapse << | Phase 3 | Terminated | - | France ... more >> Laurent SEBBAG Lyon, France, 69677 Collapse << |
| NCT00941161 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico Collapse << |
| NCT01834274 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT00399204 | Type 2 Diabetes | Phase 4 | Unknown | - | India ... more >> PGIMER Recruiting Chandigarh, India, 160012 Contact: Samir Malhotra, MD, DM samirmalhotra345@yahoo.com Sub-Investigator: Samir Malhotra, MD,DM Collapse << |
| NCT00669864 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT00396227 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT01834274 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT00669864 | - | - | Completed | - | - |
| NCT02682121 | Prediabetes | Phase 1 | Completed | - | United States, Utah ... more >> University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00767351 | - | - | Completed | - | - |
| NCT00279045 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01001962 | Hypertension ... more >>Type II Diabetes Collapse << | Phase 4 | Not yet recruiting | January 2020 | Greece ... more >> Hypertension 24h ABPM center Papageorgiou Hospital Not yet recruiting Thessaloniki, Greece Contact: VASILEIOS KOTSIS, PROF Principal Investigator: VASILEIOS KOTSIS, PROF Collapse << |
| NCT00971243 | - | - | Completed | - | - |
| NCT02956044 | Type2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Indiana ... more >> Clinical Research Site Evansville, Indiana, United States, 47710 Collapse << |
| NCT00971243 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03317028 | T2DM With Inadequate Glycemic ... more >>Control Collapse << | Phase 2 | Recruiting | March 31, 2021 | United States, Florida ... more >> Clinical Research of South Florida Recruiting Coral Gables, Florida, United States, 33134 Contact: Rosen Jeffrey, MD 305-445-5637 jrosen@crsouthflorida.com Principal Investigator: Rosen Jeffrey, MD The Community Research of South Florida Recruiting Hialeah, Florida, United States, 33016 Contact: Carpio Jose, MD 305-441-6611 jcarpio@thecommunityresearch.com Principal Investigator: Carpio Jose, MD Taiwan Kaohsiung Veterans General Hospital Recruiting Kaohsiung, Taiwan, 81362 Contact: Chih-Hsun Chu, MD +886-7-3422-121 ext 8089 chchu@vghks.gov.tw Principal Investigator: Chih-Hsun Chu, MD Kaohsiung Chang Gung Memorial Hospital Recruiting Kaohsiung, Taiwan, 833 Contact: Jung-Fu Chen, MD +886-7-733-7166 0722cjf@cgmh.org.tw Principal Investigator: Jung-Fu Chen, MD Chung Shan Medical University Hospital Recruiting Taichung, Taiwan, 402 Contact: Chien-Ning Huang, MD +886-4-2473-9595 ext 11600 cshy049@csh.org.tw Principal Investigator: Chien-Ning Huang, MD China Medical University Hospital Recruiting Taichung, Taiwan, 404 Contact: Ching-Chu Chen, MD +886-4-2206-2121 ext 3489 d1918@mail.cmuh.org.tw Principal Investigator: Ching-Chu Chen, MD Taichung Veterans General Hospital Recruiting Taichung, Taiwan, 40705 Contact: I-Te Lee, MD, PhD +886-933-553-211 itlee@vghtc.gov.tw Principal Investigator: I-Te Lee, MD, PhD Chi-Mei Medical Center Recruiting Tainan, Taiwan, 710 Contact: Kai-Jen Tien, MD +886-6-2816-31 tkzzkimo@gmail.com Principal Investigator: Kai-Jen Tien, MD National Taiwan University Hospital Recruiting Taipei, Taiwan, 10048 Contact: Chih-Yuan Wang, MD, PhD +886-938-036-246 cyw1965@gmail.com Principal Investigator: Chih-Yuan Wang, MD, PhD Taipei Veterans general Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD +886-2-2875-7516 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Tri-Service General Hospital Recruiting Taipei, Taiwan, 114 Contact: Chang-Hsun Hsieh, MD +886-2-8792-7182 10324@yahoo.com.tw Principal Investigator: Chang-Hsun Hsieh, MD Chang Gung Memorial Hospital_Linkou Not yet recruiting Taoyuan, Taiwan, 333 Contact: Yu-Yao Huang, MD, PhD +886-975-368-137 yyh@cgmh.org.tw Principal Investigator: Yu-Yao Huang, MD, PhD Collapse << |
| NCT00775164 | Obesity Insul... more >>in Resistance Collapse << | Phase 4 | Withdrawn(Inadequate enrollmen... more >>t) Collapse << | February 2011 | - |
| NCT01117584 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03115112 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00451399 | Weight Gain | Phase 4 | Completed | - | China, Hunan ... more >> Institute of Mental Health of The Second Xiangya Hospital, Central South University Changsha, Hunan, China, 410011 Collapse << |
| NCT01459094 | Healthy | Phase 1 | Completed | - | - |
| NCT02080377 | Pregnancy Ges... more >>tational Diabetes Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Princess Royal Infirmary Glasgow, Lanarkshire, United Kingdom, G31 2ER Simpson Centre for Reproductive Health, Royal Infirmary Hospital, Edinburgh Edinburgh, Lothian, United Kingdom, EH16 4SA Western General Hospital Edinburgh, Lothian, United Kingdom, EH4 2XU St Johns Hospital Livingston, West Lothian, United Kingdom Queen Elizabeth Hospital Glasgow, United Kingdom Collapse << |
| NCT02050074 | Type 2 Diabetes | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Division, Department of Medicine, Gentofte Hospital Hellerup, Copenhagen, Denmark, 2900 Collapse << |
| NCT01518101 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Berlin, Germany, 10115 Novartis Investigative Site Berlin, Germany, 13055 Novartis Investigative Site Dortmund, Germany, 44137 Novartis Investigative Site Falkensee, Germany, 14612 Novartis Investigative Site Meissen, Germany, 01662 Novartis Investigative Site Neunkirchen, Germany, 57290 Novartis Investigative Site Saarlouis, Germany, 66740 Novartis Investigative Site Völlkingen, Germany, 66333 Collapse << |
| NCT01729403 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | Germany ... more >> Neuss, Germany, 41460 Collapse << |
| NCT01708902 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01318135 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | - |
| NCT03018665 | Diabetes Mellitus, Type 2 | Phase 4 | Not yet recruiting | - | China, Jilin ... more >> First Hospital of Jilin University Not yet recruiting Changchun, Jilin, China, 130021 Contact: Guixia Wang, PhD gwang168@jlu.edu.cn Collapse << |
| NCT01708902 | - | - | Completed | - | - |
| NCT00871936 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Burbank, California, United States, 91505 Los Angeles, California, United States, 90036 Los Angeles, California, United States, 90057 United States, Florida West Palm Beach, Florida, United States, 33401 United States, Illinois Addison, Illinois, United States, 60101 United States, Maine Auburn, Maine, United States, 04210 United States, North Carolina Charlotte, North Carolina, United States, 28211 Hickory, North Carolina, United States, 28602 Winston-Salem, North Carolina, United States, 27103 United States, South Carolina Greenville, South Carolina, United States, 29615 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT02960659 | Type 2 Diabetes | Phase 1 | Recruiting | August 31, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03179254 | Diabetes Type 2 | Phase 4 | Completed | - | - |
| NCT01318135 | - | - | Completed | - | - |
| NCT02821910 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT00283816 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, New York ... more >> University of Rochester Medical Center Rochester, New York, United States, 14642 Collapse << |
| NCT00362323 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT00283816 | - | - | Completed | - | - |
| NCT01973231 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT00501020 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01973231 | - | - | Completed | - | - |
| NCT01899430 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT02343926 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Russian Federation ... more >> Investigational Site 03 Vladimir, Vladimirskaya Oblast, Russian Federation, 600023 Collapse << |
| NCT03349775 | Obesity | Early Phase 1 | Recruiting | September 1, 2024 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Elizabeth Liu, BS 617-724-3064 ELIU9@MGH.HARVARD.EDU Collapse << |
| NCT01892254 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Denmark ... more >> Copenhagen University Hospital Gentofte Hellerup, Select A State, Denmark, 2900 Collapse << |
| NCT02366377 | Type 2 Diabetes Mellitus | Phase 1 | Unknown | - | - |
| NCT01065766 | - | - | Completed | - | - |
| NCT00934570 | Obesity Type ... more >>2 Diabetes Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Children's Hospital, London Health Sciences Centre London, Ontario, Canada, N6A 5W9 Collapse << |
| NCT00490178 | Diabetes Mellitus, Type 2 ... more >> Dyslipidemia Collapse << | Phase 3 | Completed | - | France ... more >> Site 3 Marseille, France Site 1 Nantes, France Site 2 Tours, France Collapse << |
| NCT01065766 | - | - | Completed | - | - |
| NCT01463774 | Healthy | Phase 1 | Completed | - | United States, Nebraska ... more >> Lincoln, Nebraska, United States Collapse << |
| NCT03141073 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 2019 | China ... more >> Hua Medicine Limited Recruiting Shanghai, China Contact: YI ZHANG, MD,PhD +86-021-58869997 yzhang@huamedicine.com Collapse << |
| NCT01926769 | Previously Treated Advanced Co... more >>lorectal Cancer Collapse << | Phase 2 | Terminated(Slow accrual rate) | - | Korea, Republic of ... more >> Gachon University Gil Medical Center Incheon, Korea, Republic of Collapse << |
| NCT02355145 | - | - | Completed | - | Bulgaria ... more >> Research Site Pleven, Bulgaria Research Site Sofia, Bulgaria Research Site Stara Zagora, Bulgaria Collapse << |
| NCT03122041 | PCOS | Phase 4 | Completed | - | - |
| NCT01787396 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> LG Life Sciences Seoul, Korea, Republic of, 110-062 Collapse << |
| NCT02022254 | Diabetes Diab... more >>etes Mellitus, Type 2 Healthy Collapse << | Phase 1 | Completed | - | Germany ... more >> Novo Nordisk Investigational Site Berlin, Germany, 10117 Collapse << |
| NCT03194009 | Diabetes Mellitus, Type 2 ... more >> Prediabetic State Collapse << | Not Applicable | Recruiting | December 31, 2022 | Mexico ... more >> Centros de Salud. Secretaría de Salud Ciudad de México Recruiting Mexico City, Mexico, 06820 Contact: Alberto Gallardo, MD +52 (55) 50381700 ext 1597 albgallardo@yahoo.com.mx Contact: Carmen Vanegas, MD +52(55) 50381700 ext 1067 carmenvanegas_5@hotmail.com Collapse << |
| NCT02926950 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | March 2019 | - |
| NCT02188186 | Type 2 Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, 463-707 Collapse << |
| NCT00643851 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02008682 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100071 Novo Nordisk Investigational Site Beijing, Beijing, China, 100700 Novo Nordisk Investigational Site Beijing, Beijing, China, 100853 China, Chongqing Novo Nordisk Investigational Site ChongQing, Chongqing, China, 404000 China, Hebei Novo Nordisk Investigational Site Cangzhou, Hebei, China, 061000 Novo Nordisk Investigational Site Hengshui, Hebei, China, 053000 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050000 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050051 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210011 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210012 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210029 Novo Nordisk Investigational Site Suzhou, Jiangsu, China, 215004 Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 Novo Nordisk Investigational Site Zhenjiang, Jiangsu, China, 212001 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Jilin Novo Nordisk Investigational Site Changchun, Jilin, China, 130041 China, Liaoning Novo Nordisk Investigational Site Dalian, Liaoning, China, 116033 Novo Nordisk Investigational Site Shenyang, Liaoning, China, 110021 China, Shandong Novo Nordisk Investigational Site Qingdao, Shandong, China, 266003 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200080 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200092 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200120 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200240 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 201199 China Novo Nordisk Investigational Site Beijing, China, 101200 Collapse << |
| NCT00657943 | Type 2 Diabetes ... more >> Atherosclerosis Arteriosclerosis Collapse << | Phase 4 | Completed | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark, 2100 Bispebjerg Hospital Copenhagen, Denmark, 2400 Frederiksberg Hospital Frederiksberg, Denmark, 2000 Gentofte Sygehus Gentofte, Denmark, 2820 Steno Diabetes Center Gentofte, Denmark, 2820 Herlev Hospital Herlev, Denmark, 2830 Hillerod Sygehus Hillerod, Denmark, 3400 Hvidovre Hospital Hvidovre, Denmark, 2650 Køge Sygehus Koge, Denmark, 4600 Collapse << |
| NCT00643851 | - | - | Completed | - | - |
| NCT02008682 | - | - | Completed | - | - |
| NCT02629627 | Metabolic Syndrome | Phase 2 | Completed | - | - |
| NCT00834743 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Biovail Contract Research Toronto, Ontario, Canada, M1L4S4 Collapse << |
| NCT02187250 | PCOS Obesity | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT01358994 | - | - | Completed | - | Sweden ... more >> Dept of Nephrology Malmö, Sweden, SE 20502 Collapse << |
| NCT00834743 | - | - | Completed | - | - |
| NCT00134290 | Body Weight I... more >>nsulin Resistance Collapse << | Phase 4 | Completed | - | Belgium ... more >> University Hospital Ghent Ghent, Belgium, 9000 Collapse << |
| NCT01296412 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00834613 | - | - | Completed | - | - |
| NCT01472367 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | October 1, 2019 | - |
| NCT00834613 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Biovail Contract Research Toronto, Ontario, Canada, M1L4S4 Collapse << |
| NCT01296412 | - | - | Completed | - | - |
| NCT03297879 | Type2 Diabetes | Phase 4 | Completed | - | - |
| NCT03076112 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 30, 2019 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Kyoung Min Kim, MD, PHD 82-31-787-7047 kyoungmin02@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT01563120 | Gestational Diabetes Mellitus | Phase 4 | Completed | - | Israel ... more >> Dep. OB/GYN, Emek Medical Center Afula, Israel Collapse << |
| NCT01672788 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1276.7.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01594697 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Italy ... more >> Catholic University of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT01638988 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 3 | Withdrawn(Previous studies hav... more >>e been done regarding same condition) Collapse << | December 2015 | Canada, Quebec ... more >> Clinique Ovo Montréal, Quebec, Canada, H4P 2S4 Collapse << |
| NCT01006590 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01832181 | - | - | Recruiting | July 2021 | Canada, Ontario ... more >> Dr. Denice Feig Recruiting Toronto, Ontario, Canada Collapse << |
| NCT01006590 | - | - | Completed | - | - |
| NCT00099866 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Novartis investigative Site Investigative Sites, Germany Collapse << |
| NCT00778622 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Beijing ... more >> Local Institution Beijing, Beijing, China, 100028 Local Institution Beijing, Beijing, China, 100034 Local Institution Beijing, Beijing, China, 100044 Local Institution Beijing, Beijing, China, 100088 Local Institution Beijing, Beijing, China, 100730 Local Institution Beijing, Beijing, China, 101100 Local Institution Beijing, Beijing, China, 200016 China, Guangdong Local Institution Guangdong Province, Guangdong, China, 510180 Local Institution Guangdong Province, Guangdong, China, 528000 Local Institution Guangdong, Guangdong, China, 510180 China, Shanghai Local Institution Shanghai, Shanghai, China, 200003 Local Institution Shanghai, Shanghai, China, 200092 Local Institution Shanghai, Shanghai, China, 201100 Local Institution Shanghai, Shanghai, China, 201200 Collapse << |
| NCT00778622 | - | - | Completed | - | - |
| NCT01581814 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Italy ... more >> Policlinico A. Gemelli Rome, Italy, 00168 Collapse << |
| NCT02516085 | Duchenne Muscular Dystrophy | Phase 1 | Completed | - | - |
| NCT01760447 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | September 13, 2019 | - |
| NCT01107717 | Diabetes | Not Applicable | Recruiting | December 2024 | United States, Texas ... more >> Texas Diabetes Institute Recruiting San Antonio, Texas, United States, 78229-3900 Contact: Muhammad Abdul-Ghani, MD, PhD 210-567-2391 ABDULGHANI@UTHSCSA.EDU Contact: Curtiss Puckett, PA (210) 358 7200 Curtiss.Puckett@uhs-sa.com Principal Investigator: Ralph DeFronzo, MD Collapse << |
| NCT00682890 | Polycystic Ovary Syndrome ... more >> PCOS Insulin Sensitivity Collapse << | Phase 4 | Terminated(Lack of recruitment... more >>) Collapse << | - | United States, Virginia ... more >> General Clinical Research Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01672788 | - | - | Completed | - | - |
| NCT01650181 | Liver Disease | Phase 4 | Completed | - | Mexico ... more >> Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán Mexico City, Mexico, 14000 Collapse << |
| NCT00682890 | - | - | Terminated(Lack of recruitment... more >>) Collapse << | - | - |
| NCT00325910 | Diabetes Mellitus, Type 2 ... more >> Heart Failure, Congestive Collapse << | Phase 3 | Terminated(Insufficient study ... more >>participants) Collapse << | - | Canada, Alberta ... more >> Misericordia Hospital Edmonton, Alberta, Canada, T5R 4H5 University of Alberta Hospital Edmonton, Alberta, Canada, T6G 2B7 Collapse << |
| NCT00707031 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00707031 | - | - | Completed | - | - |
| NCT00681460 | Diabetes, Gestational ... more >> Insulin Resistance Collapse << | Not Applicable | Completed | - | Poland ... more >> Div of Obstetrics and Women's Diseases, Dept of Obstetrics and Gynecology, K Marcinkowski Univ of Med Sciences Poznan, Poland, 60-535 Collapse << |
| NCT02252081 | Chronic Kidney Disease ... more >> Cardiovascular Disease Metabolic Syndrome Collapse << | Phase 2 | Recruiting | December 31, 2019 | United States, Tennessee ... more >> Tennessee Valley Healthcare System Nashville Campus, Nashville, TN Recruiting Nashville, Tennessee, United States, 37212-2637 Contact: Adriana M Hung, MD MPH 615-873-6731 Adriana.Hung@va.gov Contact: Cindy A Booker (615) 343-5828 cindy.a.booker@vanderbilt.edu Principal Investigator: Adriana M Hung, MD MPH Collapse << |
| NCT00745433 | - | - | Completed | - | Belgium ... more >> Brussels, Belgium, 1070 Collapse << |
| NCT01514149 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 2 | Terminated(Business decision) | - | - |
| NCT01457911 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational Site Number 156001 Beijing, China, 100044 Investigational Site Number 156016 Beijing, China, 100088 Investigational Site Number 156003 Beijing, China, 100730 Investigational Site Number 156018 Beijing, China, 100730 Investigational Site Number 156002 Beijing, China, 100853 Investigational Site Number 156019 Changchun, China, 130041 Investigational Site Number 156017 Chengdu, China, 610083 Investigational Site Number 156014 Guangzhou, China, 510080 Investigational Site Number 156022 Guangzhou, China, 510150 Investigational Site Number 156011 Hangzhou, China, 310003 Investigational Site Number 156012 Hefei, China, 230001 Investigational Site Number 156020 Jinan, China, 250012 Investigational Site Number 156023 Jinan, China, 250021 Investigational Site Number 156010 Shanghai, China, 200040 Investigational Site Number 156006 Shanghai, China, 200072 Investigational Site Number 156009 Shanghai, China, 200080 Investigational Site Number 156021 Shenyang, China, 110022 Investigational Site Number 156005 Shijiazhuang, China, 050051 Investigational Site Number 156004 Taiyuan, China, 030001 Investigational Site Number 156024 Tianjin, China, 300052 Investigational Site Number 156008 Wuhan, China, 430022 Investigational Site Number 156015 Xiamen, China, 361003 Collapse << |
| NCT02220647 | Healthy | Phase 1 | Completed | - | - |
| NCT00400231 | Metabolic Syndrome x | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01514149 | - | - | Terminated(Business decision) | - | - |
| NCT02221401 | Healthy | Phase 1 | Completed | - | - |
| NCT01106625 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01115140 | Pregnancy Pol... more >>ycystic Ovary Syndrome Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Italy, 88100 Collapse << |
| NCT01341717 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Jothydev's Diabetes and Research Center Thiruvananthapuram, Kerala, India, 695032 Collapse << |
| NCT01106625 | - | - | Completed | - | - |
| NCT02221414 | Healthy | Phase 1 | Completed | - | - |
| NCT01841697 | - | - | Completed | - | - |
| NCT03216278 | Healthy Subjects in Fasted and... more >> Fed State Collapse << | Phase 1 | Completed | - | Brazil ... more >> Research Site Aparecida de Goiania, Brazil, 74935-530 Collapse << |
| NCT00757588 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00757588 | - | - | Completed | - | - |
| NCT01841697 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03204058 | Chronic Periodontitis | Phase 2 Phase 3 | Completed | - | - |
| NCT02243176 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China ... more >> Research Site Shanghai, China Collapse << |
| NCT01755156 | - | - | Completed | - | - |
| NCT02243176 | - | - | Completed | - | - |
| NCT01755156 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00413179 | Polycystic Ovary Syndrome ... more >> Anovulation Oligoovulation Infertility Hyperandrogenism Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Wiford Hall Medical Center Lackland AFB, Texas, United States, 78236 Collapse << |
| NCT00970593 | Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site Miami Gardens, Florida, United States, 33169 Collapse << |
| NCT02100475 | - | - | Completed | - | - |
| NCT02100475 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02946632 | Diabetes Mellitus, Type II | Phase 3 | Not yet recruiting | December 2019 | Korea, Republic of ... more >> Korea University Anam Hospital Seoul, Korea, Republic of Collapse << |
| NCT03169959 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT01666171 | - | - | Unknown | - | - |
| NCT03363815 | Heathy Volunteer | Phase 1 | Not yet recruiting | February 23, 2018 | United States, Florida ... more >> Covance Clinical Research Unit Not yet recruiting Daytona Beach, Florida, United States, 32117 Collapse << |
| NCT03092635 | Breast Cancer | Phase 1 | Recruiting | December 1, 2019 | United States, South Carolina ... more >> Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Vistea Crawford 843-792-9321 hcc-clinical-trials@musc.edu Collapse << |
| NCT01232036 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02205528 | Diabetes Diab... more >>etes Mellitus Type 2 Collapse << | Phase 2 | Completed | - | United States, California ... more >> Novo Nordisk Investigational Site Chino, California, United States, 91710 Novo Nordisk Investigational Site Hawaiian Gardens, California, United States, 90716 Novo Nordisk Investigational Site Los Angeles, California, United States, 90057 Novo Nordisk Investigational Site Spring Valley, California, United States, 91978 United States, Florida Novo Nordisk Investigational Site Hialeah, Florida, United States, 33012 Novo Nordisk Investigational Site Port Orange, Florida, United States, 32127 Novo Nordisk Investigational Site Sanford, Florida, United States, 32771 United States, Illinois Novo Nordisk Investigational Site Chicago, Illinois, United States, 60607 Novo Nordisk Investigational Site Evanston, Illinois, United States, 60201 United States, Maryland Novo Nordisk Investigational Site Oxon Hill, Maryland, United States, 20745 United States, New Jersey Novo Nordisk Investigational Site Berlin, New Jersey, United States, 08009 United States, New Mexico Novo Nordisk Investigational Site Albuquerque, New Mexico, United States, 87102 United States, North Carolina Novo Nordisk Investigational Site Greensboro, North Carolina, United States, 27408 United States, Ohio Novo Nordisk Investigational Site Cincinnati, Ohio, United States, 45227 United States, Oklahoma Novo Nordisk Investigational Site Tulsa, Oklahoma, United States, 74136 United States, Oregon Novo Nordisk Investigational Site Eugene, Oregon, United States, 97404 United States, Tennessee Novo Nordisk Investigational Site Knoxville, Tennessee, United States, 37920 United States, Texas Novo Nordisk Investigational Site Corpus Christi, Texas, United States, 78404 Novo Nordisk Investigational Site Houston, Texas, United States, 77074 United States, Virginia Novo Nordisk Investigational Site Manassas, Virginia, United States, 20110 Collapse << |
| NCT00708578 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Collapse << |
| NCT00607867 | Diabetes Mellitus ... more >> Diet Collapse << | Not Applicable | Terminated(The funding ended b... more >>efore the study was completed.) Collapse << | - | United States, Minnesota ... more >> VA Medical Center, Minneapolis Minneapolis, Minnesota, United States, 55417 Collapse << |
| NCT01043965 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | - |
| NCT01763346 | Prediabetes T... more >>ype 2 Diabetes Obesity Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of Southern California Los Angeles, California, United States, 91011 Collapse << |
| NCT02532855 | - | - | Completed | - | - |
| NCT00638716 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Quebec ... more >> ConjuChem Biotechnologies Inc. Montreal, Quebec, Canada, H2X 3Y8 Collapse << |
| NCT02192424 | Type 2 Diabetes | Phase 3 | Recruiting | September 2020 | Canada, Ontario ... more >> Mount Sinai Hospital Recruiting Toronto, Ontario, Canada, M5G1X5 Principal Investigator: Ravi Retnakaran, MD Collapse << |
| NCT01889706 | - | - | Unknown | February 2014 | Poland ... more >> Department of Internal Medicine and Diabetology, Raszeja Hospital Recruiting Poznan, Great Poland, Poland, 60-834 Contact: Agnieszka Zawada, MD aga.zawada@gmail.com Collapse << |
| NCT00638716 | - | - | Completed | - | - |
| NCT00941148 | Type 2 Diabetic Patients ... more >> Insufficient Metabolic Control OAD Treatment Collapse << | Phase 4 | Completed | - | Germany ... more >> ikfe GmbH, Clinic Department Mainz, RLP, Germany, 55116 Collapse << |
| NCT00428311 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Spain ... more >> Department of Endocrinology, Hospital Ramón y Cajal Madrid, Spain, E-28034 Collapse << |
| NCT00607867 | - | - | Terminated(The funding ended b... more >>efore the study was completed.) Collapse << | - | - |
| NCT01159600 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00362765 | Type 2 Diabetes ... more >> Dyslipidemia Collapse << | Phase 2 Phase 3 | Terminated(The study was prema... more >>turely terminated, due to difficulties in the recruitment of T2DM patients who are not under statin therapy at inclusion.) Collapse << | - | Finland ... more >> Site 2 Turku, Finland Ireland Site 3 Dublin, Ireland Italy Site 1 Pisa, Italy Collapse << |
| NCT02532855 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02419612 | Diabetes | Phase 3 | Active, not recruiting | August 15, 2019 | - |
| NCT00699036 | Nonalcoholic Steatohepatitis ... more >> Nonalcoholic Fatty Liver Disease Collapse << | Phase 2 | Unknown | August 2009 | United States, Texas ... more >> Brooke Army Medical Center Recruiting San Antonio, Texas, United States, 78234 Contact: Dawn M Torres, MD 210-916-5649 Principal Investigator: Dawn M Torres, MD Sub-Investigator: Stephen A Harrison, MD Collapse << |
| NCT02419612 | - | - | Active, not recruiting | - | - |
| NCT00763451 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Chile Sanofi-Aventis Administrative Office Santiago, Chile Colombia Sanofi-Aventis Administrative Office Santafe de Bogota, Colombia Estonia Sanofi-Aventis Administrative Office Tallinn, Estonia Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Lithuania Sanofi-Aventis Administrative Office Vilnius, Lithuania Malaysia Sanofi-Aventis Administrative Office Kuala Lumpur, Malaysia Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Philippines Sanofi-Aventis Administrative Office Makati City, Philippines Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Slovakia Sanofi-Aventis Administrative Office Brastislava, Slovakia Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT00763451 | - | - | Completed | - | - |
| NCT00491725 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT01779375 | Prediabetes T... more >>ype 2 Diabetes Collapse << | Phase 3 | Completed | - | United States, Colorado ... more >> Childrens Hospital Colorado Denver, Colorado, United States, 80045 United States, Connecticut Yale School of Medicine Pediatric Obesity and Type 2 Diabetes Clinic New Haven, Connecticut, United States, 06511 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT01779375 | - | - | Completed | - | - |
| NCT00162240 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00885352 | - | - | Completed | - | - |
| NCT00885352 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01046500 | Metabolic Syndrome | Phase 2 | Unknown | December 2014 | Italy ... more >> University Hospital Recruiting Messina, Italy Contact: Rosario D'Anna, associate professor +39 090 2217324 rosariodanna@tin.it Principal Investigator: Rosario D'Anna, associate professor Collapse << |
| NCT01159600 | - | - | Completed | - | - |
| NCT02719132 | Diabetes Mellitus Type 2 | Phase 4 | Withdrawn | November 2017 | - |
| NCT00399711 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00995345 | - | - | Completed | - | - |
| NCT00282945 | Type 2 Diabetes Mellitus | Not Applicable | Terminated(See statement in De... more >>tailed Description.) Collapse << | - | Israel ... more >> Pfizer Investigational Site Holon, Israel, 58100 Pfizer Investigational Site Jerusalem, Israel, 91120 Collapse << |
| NCT00672464 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 4 | Withdrawn(lack of funding) | February 2010 | United States, North Carolina ... more >> John Umstead Hospital Butner, North Carolina, United States, 27509 Duke University Medical Center Durham, North Carolina, United States, 27705 Collapse << |
| NCT02393573 | Pre-Diabetes ... more >>Major Lung or Abdominal Surgery Insulin Resistance Collapse << | Not Applicable | Terminated(Poor recruitment) | - | Canada, Quebec ... more >> Montreal General Hospital Montreal, Quebec, Canada, H3G 1A4 Collapse << |
| NCT01907854 | - | - | Completed | - | - |
| NCT01907854 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01519674 | - | - | Completed | - | - |
| NCT03377335 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | January 2019 | Italy ... more >> University Hospital of Palermo Palermo, Italy, 90127 Collapse << |
| NCT01519674 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01758380 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT01596504 | - | - | Completed | - | - |
| NCT01437800 | Healthy | Phase 1 | Completed | - | Mexico ... more >> Investigacion Farmacologica Y Biofarmaceutica, S.A. de C.V. México, Mexico Collapse << |
| NCT01596504 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Investigational Site Number 276008 Berlin, Germany, 10117 Investigational Site Number 276006 Berlin, Germany, 14050 Investigational Site Number 276004 Kiel, Germany, 24105 Investigational Site Number 276002 Mainz, Germany, 55116 Investigational Site Number 276005 Mönchengladbach, Germany, 41061 Investigational Site Number 276007 München, Germany, 80636 Investigational Site Number 276003 Neu-Ulm, Germany, 89231 Investigational Site Number 276001 Neuss, Germany, 41460 Collapse << |
| NCT00995345 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT01582230 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China, Beijing ... more >> Novartis Investigative Site Beijing, Beijing, China, 100730 China, Hunan Novartis Investigative Site Changsha City, Hunan, China, 410011 Novartis Investigative Site Changsha, Hunan, China, 410003 China, Jiangsu Novartis Investigative Site Nanjing, Jiangsu, China, 210006 Novartis Investigative Site Wu Xi, Jiangsu, China, 214023 China, Jiangxi Novartis Investigative Site Nanchang, Jiangxi, China, 330006 China, Liaoning Novartis Investigative Site Shenyang, Liaoning, China, 110003 China, Shandong Novartis Investigative Site Jinan, Shandong, China, 250031 China, Shanxi Novartis Investigative Site Xi'an, Shanxi, China, 710032 China, Sichuan Novartis Investigative Site Chengdu, Sichuan, China, 610072 China Novartis Investigative Site Beijing, China Novartis Investigative Site Shanghai, China, 200025 Novartis Investigative Site Tianjin, China, 300052 Philippines Novartis Investigative Site Metro Manila, Philippines, 1500 Novartis Investigative Site Pasay City, Philippines, 1300 Novartis Investigative Site Quezon City, Philippines, 1102 Singapore Novartis Investigative Site Singapore, Singapore, 169608 Novartis Investigative Site Singapore, Singapore, 768825 Thailand Novartis Investigative Site Bangkok, Thailand, 10330 Novartis Investigative Site Bangkok, Thailand, 10400 Novartis Investigative Site Khon Kaen, Thailand, 40002 Collapse << |
| NCT03196362 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | December 31, 2018 | China, Guangdong ... more >> endocrinology department of the first affiliated hospital of Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510080 Contact: Yanbing Li, MD,PhD 8602087334331 easd04lyb@126.com Principal Investigator: Yanbing Li, MD,PhD Collapse << |
| NCT01800227 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd., Village Bhat, Gandhinagar, Gujarat, India Collapse << |
| NCT00344851 | Polycystic Ovary Syndrome | Phase 2 | Completed | - | United States, Louisiana ... more >> Facility: Metabolic Center of Louisiana Research Foundation Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT00975052 | Type 2 Diabetes | Phase 1 | Completed | - | - |
| NCT02341703 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Early Phase 1 | Completed | - | Egypt ... more >> faculty of medicine, Cairo University Cairo, Egypt, 002 Collapse << |
| NCT01381900 | - | - | Completed | - | - |
| NCT01324180 | Acute Lymphoblastic Leukemia | Phase 1 | Completed | - | United States, Florida ... more >> Holtz Children's Hospital University of Miami Miller School of Medicine Miami, Florida, United States, 33136 Arnold Palmer Hospital for Children Orlando, Florida, United States, 32806 All Children's Hospital Saint Petersburg, Florida, United States, 33701 United States, New York Montefiore Medical Center, The Children's Hospital at Montefiore The Bronx, New York, United States, 10467 United States, Ohio Nationwide Children's Hospital Columbus, Ohio, United States, 43205 Collapse << |
| NCT02184832 | Metformin Response ... more >> Fasting Glucose Tryptophan Concentration Metformin Concentration Diet Tolerability Collapse << | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01095666 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01095666 | - | - | Completed | - | - |
| NCT00121667 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01800240 | Healthy | Phase 1 | Completed | - | India ... more >> Bio Evaluation Centre, Torrent Pharmaceuticals Ltd., Village Bhat, Gandhinagar, Gujarat, India Collapse << |
| NCT01217892 | - | - | Completed | - | - |
| NCT01340664 | - | - | Completed | - | - |
| NCT01340664 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01381900 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | China ... more >> Baotou, China Beijing, China Changchun, China Changsha, China Chengdu, China Chongqing, China Guangzhou, China Hangzhou, China Harbin, China Nanchang, China Nanjing, China Nanning, China Shanghai, China Shenyang, China Shiyan, China Siping, China Su Zhou, China Tianjin, China Wuxi, China Xi'An, China Malaysia Kota Bharu, Malaysia Pulau Pinang, Malaysia Selangor, Malaysia Vietnam Hanoi, Vietnam Ho Chi Minh, Vietnam Collapse << |
| NCT00353691 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00121667 | - | - | Completed | - | - |
| NCT02947620 | Type II Diabetes ... more >> Dyslipidemia Collapse << | Phase 3 | Unknown | - | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Daewoong Clinical Collapse << |
| NCT02630706 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01497522 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Kitakyushu, Fukuoka, Japan, 800-0296 Novartis Investigative Site Kurume, Fukuoka, Japan, 830-8543 Novartis Investigative Site Ohkawa-city, Fukuoka, Japan, 831-0016 Novartis Investigative Site Kawasaki-city, Kanagawa, Japan, 210-0014 Novartis Investigative Site Yokohama, Kanagawa, Japan, 221-0802 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Takatsuki, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-0031 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Kiyose, Tokyo, Japan, 204-0021 Novartis Investigative Site Minato-ku, Tokyo, Japan, 105-7390 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Novartis Investigative Site Fukuoka, Japan, 810-0001 Novartis Investigative Site Fukuoka, Japan, 819-0168 Novartis Investigative Site Kyoto, Japan, 607-8062 Collapse << |
| NCT01217892 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02630706 | - | - | Completed | - | - |
| NCT01720290 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100191 China Novo Nordisk Investigational Site Beijing, China, 100029 Novo Nordisk Investigational Site Shanghai, China Collapse << |
| NCT02831361 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00792935 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02280486 | Type 2 Diabetes | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT01422876 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00792935 | - | - | Completed | - | - |
| NCT01719003 | Diabetes Mellitus, Type 2 ... more >> Hyperglycemia Collapse << | Phase 3 | Completed | - | - |
| NCT02409238 | Mild Cognitive Impairment ... more >> Insulin Resistance Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Unknown | - | Singapore ... more >> SingHealth Polyclinics - Marine Parade Polyclinic Recruiting Singapore, Singapore, 440080 Contact: WEE KIEN HAN ANDREW, MCI (65) 63450049 andrew.wee@singhealth.com.sg Contact: NG CHIAT ENG (65)90718977 ng.chiat.eng@singhealth.com.sg Collapse << |
| NCT01399645 | Nonalcoholic Fatty Liver Disea... more >>se Nonalcoholic Steatohepatitis Type 2 Diabetes Collapse << | Phase 2 | Completed | - | Canada, Quebec ... more >> Centre hospitalier de l'Université de Montréal Montréal, Quebec, Canada, H2W 1T7 Collapse << |
| NCT00349635 | Obesity | Phase 2 | Completed | - | Finland ... more >> Site 1 Helsinki, Finland Site 3 Kuopio, Finland Site 2 Oulu, Finland Collapse << |
| NCT00015691 | HIV Infections ... more >> Lipodystrophy Hyperinsulinemia Collapse << | Not Applicable | Completed | - | - |
| NCT02681094 | - | - | Completed | - | - |
| NCT00814294 | Diabetes | Phase 2 Phase 3 | Unknown | October 2009 | - |
| NCT00348725 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 3 | Completed | - | France ... more >> Site 24 Anzin, France Site 23 Bachant, France Site 21 Baune, France Site 32 Bersee, France Site 34 Briollay, France Site 27 Denain, France Site 28 Denain, France Site 5 Le Temple de Bretagne, France Site 29 Nantes, France Site 2 Nantes, France Site 8 Nantes, France Site 9 Nantes, France Site 1 Nort sur Erdre, France Site 13 Parcay les Pins, France Site 26 Quarouble, France Site 7 Saint Aignan le Grand, France Site 4 Saint Herblain, France Site 30 Saint-Amand, France Site 11 Sautron, France Site 17 Segre, France Site 10 St Etienne de Montluc, France Site 33 Thiant, France Site 12 Thouars, France Site 25 Vieux Conde, France Site 20 Vihiers, France Collapse << |
| NCT00349128 | Dyslipidemia/Glucose Metabolis... more >>m Disorder Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT01422876 | - | - | Completed | - | - |
| NCT02681094 | Type 2 Diabetes Mellitus ... more >> Inadequate Glycaemic Control Collapse << | Phase 3 | Completed | - | - |
| NCT01850615 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01465152 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Spain ... more >> Novo Nordisk Investigational Site Barcelona, Spain, 08020 Novo Nordisk Investigational Site Benamargosa, Spain, 29718 Novo Nordisk Investigational Site Burlada, Spain, 31600 Novo Nordisk Investigational Site Camas, Spain, 41900 Novo Nordisk Investigational Site Fuenlabrada, Spain, 28942 Novo Nordisk Investigational Site Gijón, Spain, 33212 Novo Nordisk Investigational Site Granada, Spain, 18012 Novo Nordisk Investigational Site Lugo, Spain, 27004 Novo Nordisk Investigational Site Madrid, Spain, 28021 Novo Nordisk Investigational Site Madrid, Spain, 28030 Novo Nordisk Investigational Site Madrid, Spain, 28035 Novo Nordisk Investigational Site Portugalete, Spain, 48920 Novo Nordisk Investigational Site San Adria del Besos, Spain, 08930 Novo Nordisk Investigational Site Santander, Spain, 39009 Novo Nordisk Investigational Site Santander, Spain, 39011 Novo Nordisk Investigational Site Tegueste, Spain, 38280 Novo Nordisk Investigational Site Valencia, Spain, 46021 Novo Nordisk Investigational Site Zaragoza, Spain, 50007 Collapse << |
| NCT01389778 | Polycystic Ovary Syndrome | Not Applicable | Active, not recruiting | October 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01850615 | - | - | Completed | - | - |
| NCT01719003 | - | - | Completed | - | - |
| NCT03610412 | Diabetes Mellitus, Type 2 | Phase 2 | Not yet recruiting | December 2018 | - |
| NCT00432276 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00118950 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Denmark ... more >> Steno Diabetes Center Gentofte, Denmark, 2820 Collapse << |
| NCT00432276 | - | - | Completed | - | - |
| NCT00813995 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00266253 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01822548 | Type 2 Diabetes | Phase 3 | Completed | - | Italy ... more >> Azienda Opedaliera-Universitaria Parma, Italy, 43126 Azienda Ospedaliera-Universitaria Parma, Italy, 43126 Collapse << |
| NCT01430351 | Brain Cancer | Phase 1 | Active, not recruiting | September 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01822548 | - | - | Completed | - | - |
| NCT00396513 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | Poland ... more >> Division of Infertility and Reproductive Endocrinology, Department of Gynecology and Obstetrics Recruiting Poznan, Poland, 60-184 Collapse << |
| NCT03566225 | Pioglitazone | Early Phase 1 | Not yet recruiting | December 25, 2018 | - |
| NCT00813995 | - | - | Completed | - | - |
| NCT00815399 | Type 2 Diabetes | Phase 4 | Completed | - | Italy ... more >> Department of Geriatrics and Metabolic Diseases Naples, Italy, 80138 Collapse << |
| NCT01757587 | Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> CH Sud Francilien Evry, France, 91000 Collapse << |
| NCT02526524 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01490918 | Type-II Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> MedicalExcellence Seoul, Korea, Republic of, 137-701 Collapse << |
| NCT01709305 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02875821 | Type 2 Diabetes With Non-alcoh... more >>olic Fatty Liver (NAFLD) Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes Center Seoul, Korea, Republic of, 03722 Collapse << |
| NCT00856986 | - | - | Completed | - | - |
| NCT00856986 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01099137 | Diabetes | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Korea, Republic of Collapse << |
| NCT02526524 | - | - | Completed | - | - |
| NCT00798161 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00798161 | - | - | Completed | - | - |
| NCT00251953 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT01709305 | - | - | Completed | - | - |
| NCT03489668 | PCOS | Not Applicable | Recruiting | March 31, 2020 | Poland ... more >> Division of Infertility and Reproductive Endocrinology, Department of Gynecology and Obsterics Recruiting Poznan, Poland Contact: Beata Banaszewska, MD PhD bbeata48@gmail.com Contact: Mateusz Trzciński, MD mtrzcinski13@gmail.com Sub-Investigator: Antoni J Duleba, Prof Sub-Investigator: Leszek A Pawelczyk, Prof Sub-Investigator: Robert Z Spaczyński, Prof Sub-Investigator: Piotr Piekarski, MD Sub-Investigator: Jeff Chang, Prof Collapse << |
| NCT01106690 | - | - | Completed | - | - |
| NCT01106690 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00698789 | Type 2 Diabetes | Phase 2 | Terminated(Study ended after d... more >>iabetes development plan review. .) Collapse << | - | United States, Arizona ... more >> Phoenix, Arizona, United States, 85029 Tucson, Arizona, United States, 85705 United States, California Chula Vista, California, United States, 91911 Los Angeles, California, United States Mission Viejo, California, United States, 92691 Orange, California, United States, 92869 Paramount, California, United States, 90723 San Diego, California, United States, 92117 Santa Ana, California, United States, 92701 Tustin, California, United States, 92780 United States, Colorado Colorado Springs, Colorado, United States, 80909 United States, Florida St. Petersburg, Florida, United States, 33709 United States, Kansas Shawnee Mission, Kansas, United States, 66216 United States, Louisiana Shreveport, Louisiana, United States, 71101 United States, Michigan Dearborn, Michigan, United States, 48126 United States, Missouri St. Louis, Missouri, United States, 63110 United States, Nebraska Omaha, Nebraska, United States, 68154 United States, New Jersey Elizabeth, New Jersey, United States, 07202 United States, North Carolina Charlotte, North Carolina, United States, 28211 United States, Oklahoma Oklahoma City, Oklahoma, United States, 73103 United States, Rhode Island East Providence, Rhode Island, United States, 02914 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT00035568 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 4 | Completed | - | United States, Arkansas ... more >> Local Institution Little Rock, Arkansas, United States United States, California Local Institution Los Angeles, California, United States United States, Illinois Local Institution Chicago, Illinois, United States United States, Indiana Local Institution Indianapolis, Indiana, United States United States, New York Local Institution Brooklyn, New York, United States Local Institution Rochester, New York, United States United States, Pennsylvania Local Institution Pittsburgh, Pennsylvania, United States United States, Texas Local Institution Dallas, Texas, United States Local Institution San Antonio, Texas, United States Collapse << |
| NCT03689374 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | November 13, 2020 | - |
| NCT00976937 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00519142 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00370617 | Chronic Hepatitis C ... more >> Insulin Resistance Collapse << | Phase 4 | Unknown | - | Italy ... more >> Division of Gastroenterology, University of Torino, Ospedale San Giovanni Battista Recruiting Torino, Italy, 10126 Contact: Mario Rizzetto, MD +39-011-6336397 mrizzetto@molinette.piemonte.it Contact: Elisabetta Bugianesi, MD +39-011-6336397 ebugianesi@yahoo.it Sub-Investigator: Elisabetta Bugianesi, MD Collapse << |
| NCT02069379 | Depression In... more >>sulin Resistance Metabolic Syndrome Collapse << | Phase 4 | Completed | - | United States, Michigan ... more >> University of Michigan Medical School Ann Arbor, Michigan, United States, 48103 Collapse << |
| NCT01529541 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Hospital Seoul, Korea, Republic of Collapse << |
| NCT02150824 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Germany ... more >> 1307.4.49003 Boehringer Ingelheim Investigational Site Berlin, Germany 1307.4.49002 Boehringer Ingelheim Investigational Site Mainz, Germany 1307.4.49001 Boehringer Ingelheim Investigational Site Neuss, Germany Collapse << |
| NCT00976937 | - | - | Completed | - | - |
| NCT02069379 | - | - | Completed | - | - |
| NCT03136484 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00614120 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01471275 | Type 2 Diabetes Mellitus ;Obes... more >>ity,High Triglycerides;TCM Collapse << | Not Applicable | Unknown | December 2013 | China, Beijing ... more >> Guang'anmen Hospital of China Academy ofChinese Medical Sciences Recruiting Beijing, Beijing, China Contact: Zhou Qiang, doctor 15101016416 15101016416@126.com Principal Investigator: Tong Xianlin, professor Sub-Investigator: Lian Fengmei, professor Sub-Investigator: Zhou Qiang, doctor China, Hebei Baoding Hospital of TCM Recruiting Baoding, Hebei, China Contact: Li Liping lilipingm@163.com Principal Investigator: Li Liping, professor China, Jilin Affiliated Hospital of Changchun University of TCM Recruiting Changchun, Jilin, China Contact: Piao Chunli, professor piaochunli981027@yahoo.com.cn Principal Investigator: Piao Chunli, professor Sub-Investigator: Wang Yanyan China, Qinghai Qinghai Provence Hospital of TCM Recruiting Xining, Qinghai, China Contact: Pu Weirong puweirong123@163.com Principal Investigator: Ba Zhuoma, professor Sub-Investigator: Pu Weirong China, Tianjin Tianjin Dagang Hospital Recruiting Dagang, Tianjin, China Contact: Jin Chuan 58023442@qq.com Principal Investigator: Guo Hailong Sub-Investigator: Jin Chuan Tianjin University of Traditional Chinese Medicine, First Affiliated Hospital Recruiting Tianjin, Tianjin, China Contact: Wu Shentao, professor 13752262891 wushentao@yahoo.com.cn Principal Investigator: Wu Shentao, professor Sub-Investigator: Wang Bin, doctor China, Yichang Yichang City Yiling Hospital Recruiting Yichang, Yichang, China Contact: Yang Xingzhong clcy0401@163.com Principal Investigator: Yang Xingzhong China, Zhejiang Hangzhou Hospital of TCM Recruiting Hangzhou, Zhejiang, China Contact: Hong Yuzhi, professor hyzhcy@yahoo.cn Principal Investigator: Hong Yuzhi, professor Sub-Investigator: Wei Yan Collapse << |
| NCT00095030 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00754988 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00614120 | - | - | Completed | - | - |
| NCT02129985 | Type 2 Diabetes | Phase 4 | Unknown | September 2015 | China, Shanghai ... more >> Xiaolong Zhao Recruiting Jingan, Shanghai, China, 20041 Contact: xiaolong zhao, MD. 86-18918067241 xiaolongzhao@163.com Principal Investigator: xiaolong zhao, MD. Collapse << |
| NCT00319293 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | - |
| NCT01128621 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT00386100 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01257334 | Type 2 Diabetes | Phase 3 | Unknown | July 2012 | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT01895569 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | June 2016 | Italy ... more >> IRCCS Policlinico S. Matteo Foundation Recruiting Pavia, Italy, 27100 Contact: Giuseppe Derosa, MD, PhD giuseppe.derosa@unipv.it Contact: Pamela Maffioli, MD pamelamaffioli@hotmail.it Principal Investigator: Giuseppe Derosa, MD, PhD Sub-Investigator: Pamela Maffioli, MD Collapse << |
| NCT00700817 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00386100 | - | - | Completed | - | - |
| NCT00978263 | Diabetes | Not Applicable | Completed | - | China, Chongqing ... more >> the First Affiliated Hospital of Chongqing Medical University Chongqing, Chongqing, China, 400016 Collapse << |
| NCT00102466 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Investigative Centers Nurnberg, Germany Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT00703755 | Patients With Metabolic Syndro... more >>me Collapse << | Phase 2 | Completed | - | - |
| NCT00700817 | - | - | Completed | - | - |
| NCT02036515 | - | - | Completed | - | - |
| NCT02854527 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Pharma GmbH & Co KG Biberach, Germany, 88397 Collapse << |
| NCT01686126 | Complex Endometrial Hyperplasi... more >>a With Atypia Grade 1 Endometrial Endometrioid Adenocarcinoma Collapse << | Phase 2 | Recruiting | December 2020 | Australia, New South Wales ... more >> Chris O'Brien Lifehouse Recruiting Camperdown, New South Wales, Australia, 2050 Principal Investigator: Jonathan Carter Sub-Investigator: Selvan Pather John Hunter Hospital Not yet recruiting New Lambton, New South Wales, Australia, 2305 Principal Investigator: Geoffrey Otton Australia, Queensland The Wesley Hospital Recruiting Auchenflower, Queensland, Australia, 4066 Principal Investigator: Jim Nicklin Sub-Investigator: Andrea Garrett Brisbane Private Hospital Withdrawn Brisbane, Queensland, Australia, 4000 Royal Brisbane and Women's Hospital Recruiting Brisbane, Queensland, Australia, 4029 Principal Investigator: Andreas Obermair Sub-Investigator: James Nicklin Sub-Investigator: Russell Land Sub-Investigator: Andrea Garrett Greenslopes Private Hospital Recruiting Greenslopes, Queensland, Australia, 4120 Principal Investigator: Andreas Obermair Sub-Investigator: Russell Land Sub-Investigator: Naven Chetty Mater Health Services, Brisbane Recruiting South Brisbane, Queensland, Australia, 4101 Principal Investigator: Lewis Perrin Sub-Investigator: Naven Chetty Sub-Investigator: Alex Crandon Mater Private Hospital Recruiting South Brisbane, Queensland, Australia, 4101 Principal Investigator: Lewis Perrin Sub-Investigator: Naven Chetty Gold Coast Hospital Recruiting Southport, Queensland, Australia, 4215 Principal Investigator: Marcelo Nascimento Townsville Hospital Recruiting Townsville, Queensland, Australia, 4810 Principal Investigator: Andreas Obermair Sub-Investigator: James Nicklin Sub-Investigator: Lewis Perrin Sub-Investigator: Russell Land Sub-Investigator: Andrea Garrett Sub-Investigator: Naven Chetty John Flynn Hospital Not yet recruiting Tugun, Queensland, Australia, 4224 Principal Investigator: Marcelo Nascimento Sub-Investigator: Alex Crandon Australia, South Australia Royal Adelaide Hospital Active, not recruiting Adelaide, South Australia, Australia, 5000 Queen Elizabeth Hospital Not yet recruiting Woodville, South Australia, Australia, 5011 Principal Investigator: John Miller Australia, Victoria Royal Women's Hospital Recruiting Carlton, Victoria, Australia, 3053 Principal Investigator: Orla McNally Sub-Investigator: Michael Quinn Sub-Investigator: Deborah Neesham Sub-Investigator: David Wrede Monash Medical Centre Active, not recruiting Clayton, Victoria, Australia, 3168 Australia, Western Australia King Edward Memorial Hospital for Women Recruiting Perth, Western Australia, Australia, 6008 Principal Investigator: Yee Leung Sub-Investigator: Stuart Salfinger Sub-Investigator: Jason Tan St John of God Hospital Recruiting Subiaco, Western Australia, Australia, 6904 Principal Investigator: Stuart Salfinger Sub-Investigator: Jason Tan New Zealand Christchurch Women's Hospital Recruiting Christchurch, New Zealand Contact: Dianne Harker dianne.harker@otago.ac.nz Principal Investigator: Peter Sykes Sub-Investigator: Bryony Simcock Wellington Hospital Recruiting Wellington, New Zealand Contact: Cecile Bergzoll cecile.bergzoll@ccdhb.org.nz Principal Investigator: Cecile Bergzoll Collapse << |
| NCT00617058 | - | - | Terminated(Terminated in lieu ... more >>of similar,competing large, multi-site study.) Collapse << | - | - |
| NCT02036515 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02854527 | - | - | Completed | - | - |
| NCT01128621 | - | - | Completed | - | - |
| NCT00617058 | Weight Gain | Phase 2 | Terminated(Terminated in lieu ... more >>of similar,competing large, multi-site study.) Collapse << | - | United States, North Carolina ... more >> University of North Carolina, Department of Psychiatry Chapel HIll, North Carolina, United States, 27599 Collapse << |
| NCT00138515 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT00740051 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01208012 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Germany ... more >> IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Collapse << |
| NCT02685774 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT01547104 | Diabetes Mellitus Type 2 | Phase 4 | Unknown | October 2012 | Germany ... more >> ikfe GmbH Recruiting Mainz, Rhineland-Palatinate, Germany, 55116 Contact: Thomas Forst, MD, PhD + 49 6131 57636 ext 16 ThomasF@ikfe.de Contact: Daniela Sachsenheimer, MD + 49 6131 57636 ext 46 DanielaS@ikfe.de Sub-Investigator: Daniela Sachsenheimer, MD Sub-Investigator: Stephanie Helleberg, MD Sub-Investigator: Stefan Diessel Sub-Investigator: Michael Mitry, MD Collapse << |
| NCT00740051 | - | - | Completed | - | - |
| NCT01319994 | Glucocorticoid Treatment | Phase 2 Phase 3 | Unknown | January 2015 | United Kingdom ... more >> Barts and the London Recruiting London, United Kingdom Collapse << |
| NCT00676338 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02521753 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Not Applicable | Recruiting | December 2018 | Mexico ... more >> Hospital de Gineco Obstetricia Centro Médico "La Raza" Recruiting Mexico City, D.f., Mexico, 2990 Contact: Victor S Vital-Reyes, MD, PhD 52 55 57245900 ext 23719 vitalito23@hotmail.com Collapse << |
| NCT00318656 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 4 | Completed | - | - |
| NCT00676338 | - | - | Completed | - | - |
| NCT00538486 | Hypertension ... more >>Obesity Collapse << | Phase 4 | Completed | - | China ... more >> The third hospital affiliated to the Third Military Medical University Chongqing, China, 400042 Collapse << |
| NCT02276274 | - | - | Completed | - | - |
| NCT02276274 | Clinical Pharmacology | Phase 3 | Completed | - | Japan ... more >> Fukuoka-shi, Fukuoka, Japan Collapse << |
| NCT03135301 | Infertility, Female | Phase 3 | Recruiting | March 2019 | Egypt ... more >> Assiut Faculty of Medicine Recruiting Assiut, Egypt Contact: Ahmed Abbas, MD 0100 bmr90@hotmail.com Collapse << |
| NCT01971034 | Pancreatic Adenocarcinoma Adva... more >>nced or Metastatic Collapse << | Phase 2 | Completed | - | Brazil ... more >> ICESP Sao Paulo, SP, Brazil, 01246000 Collapse << |
| NCT00947557 | Diabetes Mellitus, Type II | Phase 3 | Terminated | - | - |
| NCT00318656 | - | - | Completed | - | - |
| NCT01241669 | Healthy Subjects | Phase 1 | Completed | - | United States, Kansas ... more >> Quintiles Phase I Services Overland Park, Kansas, United States, 661200 Collapse << |
| NCT00110851 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Connecticut ... more >> Phoenix Internal Medicine Associates Waterbury, Connecticut, United States, 06708 United States, Florida University Clinical Research-DeLand DeLand, Florida, United States, 32720 Center for Diabetes and Endocrine Care Hollywood, Florida, United States, 33021 Genesis Research International Longwood, Florida, United States, 32779 Baptist Diabetes Associates Miami, Florida, United States, 33156 Andres Patron, DO PA Pembroke Pines, Florida, United States, 33028 CLIRECO, Inc. Tamarac, Florida, United States, 33321 Endocrine Clinical Research Winter Park, Florida, United States, 32789, United States, Kansas PRN of Kansas Wichita, Kansas, United States, 67203 United States, North Carolina Medical Research Associates of Charlotte Charlotte, North Carolina, United States, 28211 Neem Research Group of Charlotte Charlotte, North Carolina, United States, 28262 Unifour Medical Research Associates Hickory, North Carolina, United States, 28601 Neem Research Group of Raleigh Raleigh, North Carolina, United States, 27609 Piedmont Medical Research Associates Winston-Salem, North Carolina, United States, 27103 United States, Oklahoma COR Clinical Research, LLC Oklahoma City, Oklahoma, United States, 73103 United States, Oregon Clinical Research Institute of Southern Oregon, PC Medford, Oregon, United States, 97504 United States, Pennsylvania Camp Hill Clinical Research Center Camp Hill, Pennsylvania, United States, 17011 United States, South Carolina Neem Research Group Columbia, South Carolina, United States, 29201 Palmetto Medical Research Associates Mt. Pleasant, South Carolina, United States, 29464 United States, Texas Oaks Medical Center Spring, Texas, United States, 77386 Collapse << |
| NCT00814372 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(MBX-102 did not mee... more >>t the target efficacy profile of HbA1c change.) Collapse << | - | United States, California ... more >> Providence Clinical Research Burbank, California, United States, 91505 American Institute of Research Los Angeles, California, United States, 90017 United States, Nevada Nevada Alliance Against Diabetes Las Vegas, Nevada, United States, 89101 United States, North Carolina Piedmont Medical Research Associates Winston-Salem, North Carolina, United States, 27103 United States, Texas DGD Research, Inc. San Antonio, Texas, United States, 78229 India Kamineni Hospitals Pvt, Ltd. Hyderabad, Andhra Pradesh, India, 50008 Bangalore Diabetes Centre Bangalore, India Diacon Hospital Diabetes & Research Centre Bangalore, India M. S. Ramaiah Memorial Hospital Bangalore, India St. John's Medical College & Hospital Bangalore, India Dr. V. Seshiah Diabetes Care & Research Institute Chennai, India Mediciti Hospital Hyderabad, India Medwin Hospital Hyderabad, India Diabetes Thyroid Hormone Research Institute Pvt. Ltd. Indore, India Fortis Hospital Jaipur, India Bharti Research Institute of Diabetes & Endocrinology Karnal, India Amrita Institute of Medical Sciences Kochi, India Diabetes Action Centre Mumbai, India, 400067 BYL Nair Hospital Mumbai, India PD Hindhuja National Hospital Mumbai, India Gandhi Endocrinology & Diabetes Centre Nagpur, India All India Institute of Medical Sciences New Delhi, India Deenanath Mangeshkar Hospital & Research Centre Pune, India Diabetes Care & Research Centre Pune, India Health & Research Centre Trivendrum, India Christian Medical College Vellore, India Collapse << |
| NCT01068652 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Algeria ... more >> Novo Nordisk Investigational Site Ain Témouchent, Algeria, 46000 Novo Nordisk Investigational Site Alger, Algeria, 16000 Novo Nordisk Investigational Site Algiers, Algeria, 16000 Novo Nordisk Investigational Site Constantine, Algeria, 25000 Novo Nordisk Investigational Site Oran, Algeria, 31000 Novo Nordisk Investigational Site Setif, Algeria, 19000 Novo Nordisk Investigational Site Sidi Bel Abbes, Algeria, 22000 Egypt Novo Nordisk Investigational Site Alexandria, Egypt, 21131 Morocco Novo Nordisk Investigational Site Casablanca, Morocco, 20000 South Africa Novo Nordisk Investigational Site Port Elizabeth, Eastern Cape, South Africa, 6014 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1812 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0002 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0083 Novo Nordisk Investigational Site Vaderbijlpark, Gauteng, South Africa, 1900 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4170 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4450 Novo Nordisk Investigational Site Kimberly, Northern Cape, South Africa, 8301 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7945 Novo Nordisk Investigational Site Sandton, South Africa, 2146 Tunisia Novo Nordisk Investigational Site Tunisia, Tunisia, 1053 Collapse << |
| NCT01444248 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals Seoul, Korea, Republic of Collapse << |
| NCT02588859 | - | - | Completed | - | India ... more >> Research Site Nellure, Andhra Pradesh, India Research Site Vishakhapatnam, Andhra Pradesh, India Research Site Guwahati, Assam, India Research Site New Delhi, Delhi, India Research Site Ahmedabad, Gujarat, India Research Site Rajkot, Gujarat, India Research Site Vadodara, Gujarat, India Research Site Karnal, Haryana, India Research Site Yamunanagar, Haryana, India Research Site Bangalore, Karnataka, India Research Site Trivandrum, Kerala, India Research Site Cochin, Kochi, India Research Site Bhopal, Madhya Pradesh, India Research Site Indore, Madhya Pradesh, India Research Site Mumbai, Maharashtra, India Research Site Pune, Maharashtra, India Research Site Amritsar, Punjab, India Research Site Chandigarh, Punjab, India Research Site Ludhiana, Punjab, India Research Site Jaipur, Rajasthan, India Research Site Udaipur, Rajasthan, India Research Site Madurai, Tamiladu, India Research Site Chennai, Tamilnadu, India Research Site Hyderabad, Telangana, India Research Site Hyderebad, Telangana, India Research Site Allahabad, Uttar Pradesh, India Research Site Kanpur, Uttar Pradesh, India Research Site Lucknow, Uttar Pradesh, India Research Site Noida, Uttar Pradesh, India Research Site Kolkata, West Bengal, India Research Site new Delhi, India Collapse << |
| NCT02497313 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for Diabetes Research, Herlev-Gentofte Hospital Hellerup, Denmark, 2900 Collapse << |
| NCT01768559 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01068652 | - | - | Completed | - | - |
| NCT00795808 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | New Zealand ... more >> University of Auckland Auckland, New Zealand Collapse << |
| NCT01678820 | - | - | Terminated(Merck terminated th... more >>e study for business reasons in November 2013.) Collapse << | - | - |
| NCT01020123 | - | - | Completed | - | - |
| NCT00558077 | Polycystic Ovary Syndrome ... more >> Infertility Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese" Hospital Catanzaro, Catanzaro/Italy, Italy, 88100 Collapse << |
| NCT03066830 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | May 2019 | - |
| NCT01020123 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03125694 | Uncontrolled Type 2 Diabetes M... more >>ellitus Collapse << | Phase 3 | Completed | - | Iran, Islamic Republic of ... more >> Tehran University of Medical Sciences Tehran, Iran, Islamic Republic of Collapse << |
| NCT01678820 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(Merck terminated th... more >>e study for business reasons in November 2013.) Collapse << | - | - |
| NCT01768559 | - | - | Completed | - | - |
| NCT00437970 | Diabetes Mellitus, Type 2 | Phase 4 | Withdrawn(Unable to secure sup... more >>ply of the study medication) Collapse << | February 2009 | Australia, Northern Territory ... more >> Menzies School of Health Research Darwin, Northern Territory, Australia, 0810 Collapse << |
| NCT01481116 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01106131 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Hospital Seoul, Korea, Republic of Collapse << |
| NCT02147925 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 4 | Completed | - | China, Guangdong ... more >> The Third Affiliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT02931591 | Small for Gestational Age | Not Applicable | Not yet recruiting | June 2020 | - |
| NCT01481116 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT03761134 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | September 2020 | China ... more >> Investigational Site Number 1560013 Recruiting Shanghai, China Collapse << |
| NCT02131272 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated | - | - |
| NCT02305381 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02131272 | - | - | Terminated | - | - |
| NCT02305381 | - | - | Completed | - | - |
| NCT01012037 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01392677 | Type 2 Diabetes Mellitus ... more >> High HbA1c Level Inadequate Glycaemic Control Collapse << | Phase 3 | Completed | - | - |
| NCT00240370 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03770728 | Type 2 Diabetes Mellitus | Phase 3 | Not yet recruiting | April 2021 | - |
| NCT01392677 | - | - | Completed | - | - |
| NCT03118713 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | January 2019 | Korea, Republic of ... more >> Site 13 Busan, Korea, Republic of Site 02 Deagu, Korea, Republic of Site 11 Guri-si, Korea, Republic of Site 01 Seoul, Korea, Republic of Site 05 Seoul, Korea, Republic of Site 07 Seoul, Korea, Republic of Site 09 Seoul, Korea, Republic of Site 12 Suwon-si, Korea, Republic of Collapse << |
| NCT02769481 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | December 2019 | - |
| NCT01426802 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Egypt ... more >> Investigative site Cairo, Egypt Collapse << |
| NCT01012037 | - | - | Completed | - | - |
| NCT01169779 | - | - | Completed | - | - |
| NCT01123980 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT03155087 | - | - | Completed | - | - |
| NCT01123980 | - | - | Completed | - | - |
| NCT01169779 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational Site Number 156011 Beijing, China, 100034 Investigational Site Number 156012 Beijing, China, 100101 Investigational Site Number 156019 Beijing, China, 100191 Investigational Site Number 156002 Beijing, China, 100700 Investigational Site Number 156003 Beijing, China, 100730 Investigational Site Number 156009 Beijing, China, 100730 Investigational Site Number 156001 Beijing, China, 100853 Investigational Site Number 156036 Changchun, China, 130041 Investigational Site Number 156016 Changsha, China, 410008 Investigational Site Number 156015 Changsha, China, 410011 Investigational Site Number 156006 Chengdu, China, 610041 Investigational Site Number 156032 Chengdu, China, 610072 Investigational Site Number 156010 Dalian, China, 116027 Investigational Site Number 156004 Guangzhou, China, 510080 Investigational Site Number 156008 Guangzhou, China, 510080 Investigational Site Number 156025 Guangzhou, China, 510630 Investigational Site Number 156031 Haikou, China, 57028 Investigational Site Number 156014 Harbin, China, 150001 Investigational Site Number 156029 Hefei, China, 230022 Investigational Site Number 156013 Qingdao, China, 266003 Investigational Site Number 156007 Shanghai, China, 200003 Investigational Site Number 156030 Shanghai, China, 200065 Investigational Site Number 156020 Shenyang, China, 110004 Investigational Site Number 156035 Suzhou, China, 215004 Investigational Site Number 156033 Taiyuan, China, 030001 Investigational Site Number 156037 Tianjin, China, 300052 Investigational Site Number 156022 Xi'An, China, 710032 Investigational Site Number 156023 Xi'An, China, 710061 Hong Kong Investigational Site Number 344001 Hong Kong, Hong Kong Investigational Site Number 344003 Hong Kong, Hong Kong Investigational Site Number 344002 Shatin, Nt, Hong Kong Malaysia Investigational Site Number 458001 Kelantan, Malaysia, 16150 Investigational Site Number 458003 Kuala Lumpur, Malaysia, 59100 Investigational Site Number 458002 Putrajaya, Malaysia, 62250 Thailand Investigational Site Number 764002 Bangkok, Thailand, 10400 Collapse << |
| NCT00486187 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Canada, Ontario ... more >> Partners Research Brampton, Ontario, Canada, L6V 1B4 Collapse << |
| NCT01233622 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03684642 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 2020 | United States, Alabama ... more >> Investigational Site Number 8400038 Recruiting Birmingham, Alabama, United States, 35211 United States, Arizona Investigational Site Number 8400035 Recruiting Chandler, Arizona, United States, 85224 Investigational Site Number 8400005 Recruiting Glendale, Arizona, United States, 85306 United States, California Investigational Site Number 8400040 Recruiting Tustin, California, United States, 92780 United States, Florida Investigational Site Number 8400041 Recruiting Pembroke Pines, Florida, United States, 33026 United States, Georgia Investigational Site Number 8400025 Recruiting Snellville, Georgia, United States, 30078 United States, Kentucky Investigational Site Number 8400044 Recruiting Lexington, Kentucky, United States, 40503 United States, New Jersey Investigational Site Number 8400001 Recruiting Bridgeton, New Jersey, United States, 08302 United States, North Carolina Investigational Site Number 8400036 Recruiting Morehead City, North Carolina, United States, 28557 United States, Utah Investigational Site Number 8400037 Recruiting Layton, Utah, United States, 84041 Collapse << |
| NCT00379769 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01294553 | - | - | Completed | - | - |
| NCT00118963 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Denmark ... more >> Steno Diabetes Center Gentofte, Denmark, 2820 Collapse << |
| NCT00995787 | Type II Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site San Diego, California, United States Collapse << |
| NCT01819129 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00568984 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100034 Novo Nordisk Investigational Site Beijing, Beijing, China, 100044 Novo Nordisk Investigational Site Beijing, Beijing, China, 100730 China, Tianjin Novo Nordisk Investigational Site Tianjin, Tianjin, China, 300052 Malaysia Novo Nordisk Investigational Site Cheras, Malaysia, 56000 Novo Nordisk Investigational Site Ipoh, Malaysia, 30450 Novo Nordisk Investigational Site Kota Bharu, Kelantan, Malaysia, 16150 Philippines Novo Nordisk Investigational Site Cebu City, Philippines, 6000 Novo Nordisk Investigational Site Manila, Philippines, 1003 Novo Nordisk Investigational Site Marikina City, Philippines, 1800 Novo Nordisk Investigational Site Pampanga, Philippines, 2000 Novo Nordisk Investigational Site Quezon City, Philippines, 1100 Novo Nordisk Investigational Site Quezon, Philippines, 4301 Thailand Novo Nordisk Investigational Site Bangkok, Thailand, 10300 Novo Nordisk Investigational Site Bangkok, Thailand, 10330 Collapse << |
| NCT00463502 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03606694 | Type 2 Diabetes Mellitus | Phase 2 | Not yet recruiting | September 30, 2020 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00375388 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Netherlands ... more >> Bethesda General Hospital and Bethesda Diabetes Center Hoogeveen, Drenthe, Netherlands, 7909 AA Collapse << |
| NCT01310452 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | - | China ... more >> Zhong Shan Hospital, Fudan University Shang hai, China Collapse << |
| NCT01294553 | - | - | Completed | - | - |
| NCT00379769 | - | - | Completed | - | - |
| NCT00361868 | Dyslipidemia ... more >>Glucose Metabolism Disorder Collapse << | Phase 3 | Terminated(The study was disco... more >>ntinued prematurely at the end of March 2007 due to slow enrolment.) Collapse << | - | - |
| NCT01867502 | Diabetes Mellitus, Type 2 ... more >> Hypoglycemic Agents Diabetic Blood Glucose Monitoring Exercise Collapse << | Phase 4 | Unknown | August 2015 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre Recruiting Porto Alegre, Rio Grande do Sul, Brazil, 90035-903 Contact: Aline Fofonka, MSc 55-5192339191 alinefofonka@hotmail.com Principal Investigator: Beatriz D Schaan, PhD Collapse << |
| NCT01874080 | Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT00261352 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT00744367 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00592969 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Argentina ... more >> Novo Nordisk Investigational Site Mariano Acosta s/n, Argentina, 5016 Brazil Novo Nordisk Investigational Site Santa Efigenia, Brazil, 30150-320 Collapse << |
| NCT00018616 | Diabetes | Phase 2 | Completed | - | United States, Connecticut ... more >> VA Connecticut Health Care System (West Haven) West Haven, Connecticut, United States, 06516 Collapse << |
| NCT00317928 | Polycystic Ovary Syndrome | Not Applicable | Unknown | - | - |
| NCT00960661 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00698230 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00934323 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT02949193 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Kangbuk Samsung Medical Center Seoul, Korea, Republic of, 110-746 Collapse << |
| NCT00755287 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00960661 | - | - | Completed | - | - |
| NCT01819129 | - | - | Completed | - | - |
| NCT00324363 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Research Site Beijing, China Research Site Guangzhou, China Research Site Nanjing, China Research Site Shanghai, China Research Site Sichuan, China India Research Site Chennai, India Research Site Mumbai, India Research Site Pune, India Korea, Republic of Research Site Seoul, Korea, Republic of Research Site Sungnam City, Korea, Republic of Taiwan Research Site Chiayi, Taiwan Research Site Taichung, Taiwan Research Site Tainan, Taiwan Research Site Taipei, Taiwan Collapse << |
| NCT00817778 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00817778 | - | - | Completed | - | - |
| NCT02512523 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | - | Korea, Republic of ... more >> HANDOK Inc. Recruiting Seoul, Korea, Republic of Contact: Sejin Kim, CRM SeJin.Kim@handok.com Contact: Sojung Park, CPL SoJung.Park@handok.com Collapse << |
| NCT00774553 | Type 2 Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States India Research site Ahmedabad, India Collapse << |
| NCT00603239 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Alabama ... more >> Research Site Birmingham, Alabama, United States United States, California Research Site Concord, California, United States Research Site Fresno, California, United States United States, Colorado Research Site Denver, Colorado, United States United States, Nevada Research Site Las Vegas, Nevada, United States United States, Ohio Research Site Mogadore, Ohio, United States United States, Oregon Research Site Corvallis, Oregon, United States Canada, British Columbia Research Site New Westminster, British Columbia, Canada Canada, Manitoba Research Site Winnipeg, Manitoba, Canada Canada, Ontario Research Site Ajax, Ontario, Canada Research Site Cambridge, Ontario, Canada Research Site Windsor, Ontario, Canada Mexico Research Site Mexico City, Distrito Federal, Mexico Research Site Celaya, Guanajuato, Mexico Research Site Monterrey, Nuevo Leon, Mexico Research Site Chihuahua, Mexico Research Site Mexico City, Mexico Romania Research Site Baia Mare, Romania Research Site Brasov, Romania Research Site Bucuresti, Romania Research Site Dolj, Romania Research Site Iasi, Romania Research Site Suceava, Romania South Africa Research Site Johannesburg, South Africa Research Site Pretoria, South Africa Collapse << |
| NCT00139477 | - | - | Completed | - | - |
| NCT00139477 | Obesity | Not Applicable | Completed | - | United States, Florida ... more >> Nemours Children's Clinic Jacksonville, Florida, United States, 32207 Collapse << |
| NCT03604419 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 2 | Recruiting | June 1, 2019 | United States, Florida ... more >> Clinical Pharmacology of Miami Recruiting Miami, Florida, United States, 33014 Contact: KENNETH Lasseter, MD Collapse << |
| NCT02288273 | - | - | Completed | - | - |
| NCT02288273 | Type 2 Diabetes | Phase 4 | Completed | - | - |
| NCT01126580 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01336751 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States Collapse << |
| NCT00351884 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative Site Investigative Centers, Germany Collapse << |
| NCT01376557 | - | - | Completed | - | - |
| NCT01126580 | - | - | Completed | - | - |
| NCT02025907 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT00603239 | - | - | Completed | - | - |
| NCT01376557 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02025907 | - | - | Completed | - | - |
| NCT00541437 | - | - | Completed | - | Taiwan ... more >> National Taiwan university hospital Taipei, Taiwan Collapse << |
| NCT01496430 | Hypertension ... more >>Diabetes Collapse << | Phase 3 | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT00469092 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01496430 | - | - | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT00469092 | - | - | Completed | - | - |
| NCT01486966 | - | - | Terminated(Trial terminated pr... more >>ematurely due to slow recruitment.) Collapse << | - | - |
| NCT00613951 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Finland ... more >> Novo Nordisk Investigational Site Helsinki, Finland, 00260 Novo Nordisk Investigational Site Kuopio, Finland, 70210 Novo Nordisk Investigational Site Lahti, Finland, 15110 Novo Nordisk Investigational Site Pori, Finland, FI-28100 France Novo Nordisk Investigational Site Bar-Le-Duc, France, 55000 Novo Nordisk Investigational Site GRENOBLE cedex, France, 38043 Novo Nordisk Investigational Site Hayange, France, 57700 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Nanterre, France, 92014 Novo Nordisk Investigational Site NEVERS cedex, France, 58033 Novo Nordisk Investigational Site Pointe à Pitre, France, 97159 Germany Novo Nordisk Investigational Site Berlin, Germany, 12163 Novo Nordisk Investigational Site Pirna, Germany, 01796 Novo Nordisk Investigational Site Riesa, Germany, 01587 Novo Nordisk Investigational Site Saarbrücken, Germany, 66121 Novo Nordisk Investigational Site St. Ingbert, Germany, 66386 Novo Nordisk Investigational Site Völklingen, Germany, 66333 Novo Nordisk Investigational Site Wangen, Germany, 88239 Poland Novo Nordisk Investigational Site Bydgoszcz, Poland, 85-822 Novo Nordisk Investigational Site Gniewkowo, Poland, 88-140 Novo Nordisk Investigational Site Nysa, Poland, 48-300 Novo Nordisk Investigational Site Plock, Poland, 09-400 Novo Nordisk Investigational Site Szczecin, Poland, 70-483 Novo Nordisk Investigational Site Tychy, Poland, 43-100 Novo Nordisk Investigational Site Warszawa, Poland, 02-507 Novo Nordisk Investigational Site Wroclaw, Poland, 50-127 Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Barcelona, Spain, 08035 Novo Nordisk Investigational Site Granada, Spain, 18012 Novo Nordisk Investigational Site Madrid, Spain, 28034 Novo Nordisk Investigational Site San Juan, Spain, 03550 Collapse << |
| NCT00231647 | Obesity Diabe... more >>tes Mellitus, Type 2 Diabetes Mellitus, Adult-Onset Collapse << | Phase 2 | Completed | - | - |
| NCT03714594 | Glucose, Low Blood | Phase 2 | Recruiting | September 2021 | Italy ... more >> Department of Endocrinology and Metabolism, University of Pisa Recruiting Pisa, Italy, 56124 Contact: Stefano Del Prato, MD +39 050 995103 stefano.delprato@med.unipi.it Sub-Investigator: Giuseppe Daniele, MD Collapse << |
| NCT02647320 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01486966 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Terminated(Trial terminated pr... more >>ematurely due to slow recruitment.) Collapse << | - | China, Beijing ... more >> Novo Nordisk Investigational Site Beijing, Beijing, China, 100034 China, Chongqing Novo Nordisk Investigational Site Chongqing, Chongqing, China, 400016 China, Jiangsu Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200003 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 Collapse << |
| NCT01051011 | Diabetes Mellitus Type 2 | Phase 3 | Terminated(high discontinuatio... more >>n rates mainly due to GI tolerability and implementation of risk mitigation plan to address hypersensitivity reactions) Collapse << | - | - |
| NCT00653302 | Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> Sanofi-aventis administrative office Paris, France Collapse << |
| NCT00631488 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00614055 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | France ... more >> Novo Nordisk Investigational Site Alès, France, 30100 Novo Nordisk Investigational Site Brest, France, 29609 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Le Creusot, France, 71200 Novo Nordisk Investigational Site Mont de Marsan, France, 40024 Novo Nordisk Investigational Site Venissieux, France, 69200 Germany Novo Nordisk Investigational Site Bad Kreuznach, Germany, 55545 Novo Nordisk Investigational Site Bad Mergentheim, Germany, 97980 Novo Nordisk Investigational Site Dormagen, Germany, 41539 Novo Nordisk Investigational Site Hohenmölsen, Germany, 06679 Novo Nordisk Investigational Site Neuss, Germany, 41460 Novo Nordisk Investigational Site Neuwied, Germany, 56564 Norway Novo Nordisk Investigational Site Elverum, Norway, 2408 Novo Nordisk Investigational Site Hamar, Norway, 2317 Novo Nordisk Investigational Site Kongsvinger, Norway, 2212 Novo Nordisk Investigational Site Oslo, Norway, 0586 Novo Nordisk Investigational Site Stavanger, Norway, 4011 Novo Nordisk Investigational Site Tromsø, Norway, 9038 Romania Novo Nordisk Investigational Site Bucharest, Romania, 011234 Novo Nordisk Investigational Site Bucharest, Romania, 020042 Novo Nordisk Investigational Site Bucharest, Romania, 020475 Novo Nordisk Investigational Site Bucharest, Romania, 020992 Novo Nordisk Investigational Site Bucharest, Romania Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Partida de Bacarot, Spain, 03114 Novo Nordisk Investigational Site Sevilla, Spain, 41010 Novo Nordisk Investigational Site Valencia, Spain, 46014 Novo Nordisk Investigational Site Valladolid, Spain, 47011 Collapse << |
| NCT00614055 | - | - | Completed | - | - |
| NCT00823992 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT02053272 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Romania ... more >> Diabol SRL Brasov, Romania, 500365 Nicodiab SRL Bucuresti, Romania, 010507 Spitalul Universitar de Urgenta Militar Central "Dr. Carol Davila" Bucuresti, Romania, 010825 Institutul National de Diabet Bucuresti, Romania, 020475 Centrul Medical 'Sanatatea Ta' SRL Bucuresti, Romania, 020614 ArtMedical Clinic Bucuresti, Romania, 021107 Societatea Civila Medicala dr. Paveliu Bucuresti, Romania, 050538 Consultmed SRL Iasi, Romania, 700547 Cabinet Medical Individual Diabet Nutritie si Boli Metabolice Maramures, Romania, 430123 Grandmed SRL Oradea, Romania, 410159 Spitalul Judetean de Urgente Satu Mare Satu Mare, Romania, 440055 Spitalul Clinic Judetean de Urgenta Sibiu Sibiu, Romania, 550245 Gagiu D. Remus Cabinet Medical Individual Targoviste, Romania, 130083 Mediab SRL Diabet Zaharat Targu Mures, Romania, 540142 United Kingdom Avondale Surgery Chesterfield, Derbyshire, United Kingdom, S40 4TF Hull and East Yorkshire Hospitals NHS Trust Hull, East Yorkshire, United Kingdom, HU3 2JZ Salford Royal NHS Foundation Trust Salford, Greater Manchester, United Kingdom, M6 8HD University Hospitals of Leicester NHS Trust Leicester, Leicestershire, United Kingdom, LE5 4QF Aintree University Hospitals NHS Foundation Trust Liverpool, Merseyside, United Kingdom, L9 7AL Strensall Medical Practice York, North Yorkshire, United Kingdom, YO32 5UA Churchill Hospital Oxford, Oxfordshire, United Kingdom, OX3 9DU Hathaway Surgery Chippenham, Wiltshire, United Kingdom, SN14 6GT Avenue Surgery Warminster, Wiltshire, United Kingdom, BA12 9AA St Chad's Surgery Bath, United Kingdom, BA3 2UH Barts and The London NHS Trust London, United Kingdom, E1 1BB Collapse << |
| NCT02053103 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(On 25April2014, stu... more >>dy was terminated before any dosing due to an AE of safety concern that occurred in protocol B1731003 with the same drug.) Collapse << | - | United States, Florida ... more >> Pfizer Investigational Site Orlando, Florida, United States, 32809 Collapse << |
| NCT00613951 | - | - | Completed | - | - |
| NCT00770835 | Diabetes Mellitus | Phase 4 | Completed | - | Italy ... more >> Padova, Italy Pisa, Italy Collapse << |
| NCT01951183 | Diabetes Mellitus Type 2 | Phase 2 | Withdrawn(Cancelled due to Spo... more >>nsor's decision) Collapse << | December 2013 | - |
| NCT00753896 | Type 2 Diabetes | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Mesa, Arizona, United States Research Site Tempe, Arizona, United States United States, California Research Site Concord, California, United States Research Site Fresno, California, United States Research Site La Mesa, California, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Idaho Research Site Idaho Falls, Idaho, United States United States, Kentucky Research Site Bowling Green, Kentucky, United States United States, Oregon Research Site Corvallis, Oregon, United States United States, Tennessee Research Site Chattanooga, Tennessee, United States Research Site Memphis, Tennessee, United States Canada, British Columbia Research Site New Westminister, British Columbia, Canada Canada, Ontario Research Site Ajax, Ontario, Canada Research Site Cambridge, Ontario, Canada Research Site Windsor, Ontario, Canada Mexico Research Site Chihuahua, Chiuahua, Mexico Research Site Monterrey, Nuevo Leon, Mexico Research Site Distrito Federal, Mexico Romania Research Site Baia Mare, Romania Research Site Brasov, Romania Research Site Bucharesti, Romania Research Site Craiova, Romania Research Site Iasi, Romania Research Site Suceava, Romania South Africa Research Site Johannesburg, South Africa Research Site Pretoria, South Africa Collapse << |
| NCT02872818 | Apoptotic Signal Pathways in E... more >>ndometrial Hyperplasia Collapse << | Phase 4 | Completed | - | Turkey ... more >> Kayseri Training and Research Hospital Kayseri, Turkey, 38000 Collapse << |
| NCT02647320 | - | - | Completed | - | - |
| NCT00631488 | - | - | Completed | - | - |
| NCT02033889 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00106340 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers Nurnberg, Germany Collapse << |
| NCT00753896 | - | - | Completed | - | - |
| NCT00209482 | Obesity | Phase 2 Phase 3 | Unknown | - | United States, California ... more >> University of California, Los Angeles Los Angeles, California, United States, 90095 University of California, San Francisco San Francisco, California, United States, 94143 Stanford University Stanford, California, United States, 94305 United States, Massachusetts Children's Hospital, Boston Boston, Massachusetts, United States, 02115 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00484419 | Type 2 Diabetes ... more >> Hyperlipidemia Collapse << | Phase 3 | Completed | - | United States, California ... more >> La Mesa, California, United States Los Angeles, California, United States Los Gatos, California, United States San Antonio, California, United States United States, Florida Chiefland, Florida, United States United States, Indiana Gary, Indiana, United States United States, Maryland Baltimore, Maryland, United States United States, Michigan Dearborn, Michigan, United States W. Bloomfield, Michigan, United States United States, Nevada Las Vegas, Nevada, United States United States, New York Yonkers, New York, United States United States, North Carolina Lexington, North Carolina, United States Winston-Salem, North Carolina, United States United States, Ohio Munroe Falls, Ohio, United States Zaneville, Ohio, United States United States, Oregon Portland, Oregon, United States United States, Pennsylvania Jersey Shore, Pennsylvania, United States United States, South Carolina Clemson, South Carolina, United States United States, Tennessee Harriman, Tennessee, United States United States, Texas Dallas, Texas, United States Collapse << |
| NCT03452267 | Insulin Sensitivity | Phase 2 Phase 3 | Not yet recruiting | June 2020 | - |
| NCT00484419 | - | - | Completed | - | - |
| NCT00214591 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT01892020 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT02033889 | - | - | Completed | - | - |
| NCT00279240 | Diabetes | Phase 1 | Completed | - | India ... more >> Diabetes Research Centre & MV Hospital for Diabetes Chennai, Tamilnadu, India, 600013 Collapse << |
| NCT00975286 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00038727 | Diabetes Mellitus ... more >> Cancer CVD Collapse << | Phase 3 | Active, not recruiting | January 2025 | United States, Maryland ... more >> George Washington University Rockville, Maryland, United States, 20852 Collapse << |
| NCT01892020 | - | - | Completed | - | - |
| NCT00975286 | - | - | Completed | - | - |
| NCT00318461 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00038727 | - | - | Active, not recruiting | - | - |
| NCT01868542 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 301-721 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 301-723 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 302-120 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 302-718 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 330-721 Novo Nordisk Investigational Site Daejeon, Korea, Republic of, 361-711 Collapse << |
| NCT00700856 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 4 | Unknown | December 2018 | - |
| NCT00822211 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> China-Japan Friendship Hospital Beijing, China General Hospital of Beijing Military Region of PLA Beijing, China Peking Union Medical College Hospital Beijing, China Peking University First Hospital Beijing, China The General Hospital of the PLA Beijing, China The General Hospital of the Second Artilleryman of PLA Beijing, China The Affiliated Union Hospital of Fujian Medical University Fuzhou, China The Fuzhou General Hospital of the PLA Nanjing Military Area Fuzhou, China Guangdong Provincial People's Hospital Guangzhou, China Second Affiliated Hospital of Nanchang University Nanchang, China The Jiangxi Provincial People's Hospital Nanchang, China Te Affiliated Drum Tower of Nanjing University Medical School Nanjing, China Tongji Hospital of Tongji University Nanjing, China Shanghai Changzheng Hospital Shanghai, China The Second Affiliated Hospital of China Medical University Shenyang, China The Second Affiliated Hospital of Tianjin Medical University Tianjin, China First Affiliated Hospital of 4th Military Medical University Xi'an, China Collapse << |
| NCT01868542 | - | - | Completed | - | - |
| NCT00265746 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | - | Poland ... more >> Department of Endocrinology and Isotope Therapy, Polish Mother’s Memorial Hospital, Research Institute, Lodz Recruiting Lodz, Poland Contact: Katarzyna Cypryk, M D, PhD , Habilitation 0048422711159 kcypryk@mp.pl Principal Investigator: Katarzyna Cypryk, PhD, MD , Sub-Investigator: Marcin Kosiński, MD Collapse << |
| NCT02135549 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, New York ... more >> Accumed Garden City, New York, United States, 11530 Collapse << |
| NCT00318461 | - | - | Completed | - | - |
| NCT00544518 | Type 2 Diabetes | Phase 2 | Unknown | October 2008 | China, Shanxi ... more >> XiJing hospital of Fourth Military Medical University Xi'an, Shanxi, China, 710032 Collapse << |
| NCT03259789 | Type2 Diabetes Mellitus | Phase 3 | Active, not recruiting | January 2019 | - |
| NCT00231660 | Obesity Diabe... more >>tes Mellitus, Type 2 Diabetes Mellitus, Adult-Onset Collapse << | Phase 3 | Completed | - | - |
| NCT03279263 | Type 2 Diabetes Mellitus | Phase 2 | Recruiting | September 2019 | United States, California ... more >> National Research Institute Recruiting Los Angeles, California, United States, 90057 Contact: Juan Frias, MD Collapse << |
| NCT00399360 | - | - | Completed | - | - |
| NCT01511198 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00399360 | HIV Infections ... more >> Metabolic Syndrome X Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03165877 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT02932475 | Diabetes Preg... more >>nancy Collapse << | Phase 3 | Recruiting | June 30, 2021 | United States, Alabama ... more >> University of Alabama Not yet recruiting Birmingham, Alabama, United States, 35294 Contact: Lori Harper, MD 205-934-5611 lmharper@uabmc.edu United States, California University of California - San Diego Not yet recruiting San Diego, California, United States, 92093 Contact: Gladys Ramos, MD 619-543-6906 gramos@ucsd.edu United States, Mississippi University of Mississippi Not yet recruiting Jackson, Mississippi, United States, 38677 Contact: Kedra Wallace, MD 601-984-5396 kwallace2@umc.edu United States, New York Columbia University Not yet recruiting New York, New York, United States, 10027 Contact: Ronald Wapner, MD 212-305-1521 rw2191@columbia.edu United States, North Carolina University of North Carolina Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Kim Boggess, MD 919-966-1601 kboggess@med.unc.edu Contact: Karen Dorman 984-974-9012 kdorman@med.unc.edu United States, Ohio Ohio State University Not yet recruiting Columbus, Ohio, United States, 43210 Contact: Mark Landon, MD 614-293-8513 mark.landon@osumc.edu United States, Pennsylvania University of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Celeste Durswald, MD 215-615-3817 celeste.durnwald@uphs.upenn.edu United States, Texas University of Texas Medical Branch Not yet recruiting Galveston, Texas, United States, 77555 Contact: Gayle Olson, MD 409-747-4905 golson@utmb.edu University of Texas - Houston Not yet recruiting Houston, Texas, United States, 77030 Contact: Jeffie Refuerzo 713-500-6416 jerrie.s.refuerzo@uth.tmc.edu United States, Utah University of Utah Not yet recruiting Salt Lake City, Utah, United States, 84112 Contact: Michael Varner 801-581-4173 michael.varner@hsc.utah.edu Collapse << |
| NCT01999218 | - | - | Completed | - | - |
| NCT03444584 | Type 2 Diabetes Mellitus | Phase 2 | Active, not recruiting | December 10, 2018 | Germany ... more >> Research Site Magdeburg, Germany, 39120 Research Site Mannheim, Germany, 68167 Research Site München, Germany, 81241 Hungary Research Site Balatonfüred, Hungary, 8230 Research Site Miskolc, Hungary, 3529 Research Site Szeged, Hungary, 6720 United Kingdom Research Site Liverpool, United Kingdom, L7 8XP Research Site Manchester, United Kingdom, M13 9NQ Research Site Rotherham, United Kingdom, S65 1DA Research Site Swansea, United Kingdom, SA2 8PP Collapse << |
| NCT01557504 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT01427920 | - | - | Completed | - | - |
| NCT01427920 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT02989649 | - | - | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns) Collapse << | - | China, Beijing ... more >> Beijing, Beijing, China China, Hubei Wuhan, Hubei, China China, Liaoning Dalian, Liaoning, China Shenyang, Liaoning, China China, Shandong Jinan, Shandong, China Qingdao, Shandong, China Tai'an, Shandong, China Taian, Shandong, China China, Tianjin Tianjin, Tianjin, China Hong Kong Sai Ying Pun, Hong Kong Sha Tin, Hong Kong Collapse << |
| NCT01557504 | - | - | Completed | - | - |
| NCT02491333 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 3 | Unknown | December 2017 | China, Guangdong ... more >> The First Affiliated Hospital of Guangzhou Medical University Recruiting Guangzhou, Guangdong, China, 510120 Contact: Wanting Wu, Master 15818153730 Contact: Meifang Li, Master 13544426679 Principal Investigator: Hongxia Ma, PhD Collapse << |
| NCT01303055 | Diabetes Mellitus, Type 2 | Not Applicable | Unknown | December 2014 | Japan ... more >> Dept. of Intern. Med., School of Dentistry, Aichi Gakuin University Recruiting Nagoya, Aichi, Japan, 464-8650 Contact: Takahiro Tosaki, MD, PhD +81-52-759-2111 nrd49075@nifty.com Principal Investigator: Takahiro Tosaki, MD, PhD Sub-Investigator: Keiko Naruse, MD, PhD Tosaki Clinic for Diabetes and Endocrinology Not yet recruiting Nagoya, Aichi, Japan, 468-0009 Contact: Takahiro Tosaki, MD, PhD nrd49075@nifty.com Principal Investigator: Takahiro Tosaki, MD, PhD Diabetes Clinic, Okazaki East Hospital Recruiting Okazaki, Aichi, Japan, 444-0008 Contact: Takahiro Tosaki, MD, PhD +81-564-22-6616 nrd49075@nifty.com Sub-Investigator: Chizuko Suzuki, MD, PhD Sub-Investigator: Maiko Mizutani, MD, PhD Principal Investigator: Takahiro Tosaki, MD, PhD Diabetes Center, Yokkaichi Social Insurance Hospital Recruiting Yokkaichi, Mie, Japan, 510-0016 Contact: Takahiro Tosaki, MD, PhD +81-59-331-2000 nrd49075@nifty.com Sub-Investigator: Miho Miyoshi, MD Sub-Investigator: Tomoki Majima, MD Principal Investigator: Takahiro Tosaki, MD, PhD Collapse << |
| NCT00469586 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT01632163 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01999218 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02730741 | Antidiarrhoea | Phase 2 | Recruiting | - | France ... more >> BIOSE Recruiting Aurillac, France Contact: NIVOLIEZ Mr Adrien +330471465101 a.nivoliez@biose.com Principal Investigator: Pr. Jacques MOREAU Collapse << |
| NCT01084486 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Phase 1 Phase 2 | Completed | - | Israel ... more >> Wolfson Medical Center Holon, Israel, 58100 Collapse << |
| NCT02149459 | Brain Neoplasms | Phase 1 | Recruiting | July 2018 | Israel ... more >> Sheba Medical Center Recruiting Ramat Gan, Israel, 52621 Collapse << |
| NCT01733459 | Polycystic Ovary Syndrome (PCO... more >>S) Collapse << | Phase 3 | Completed | - | Indonesia ... more >> Department of Obstetrics and Gynecology, Faculty of Medicine, University of Padjadjaran, Hasan Sadikin Hospital Bandung, West Java, Indonesia, 40161 Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Indonesia, dr. Cipto Mangunkusumo Hospital Jakarta, Indonesia, 10430 Collapse << |
| NCT02076347 | Type 2 Diabetes ... more >> Vitamin B12 Deficiency Neuropathy Anemia Collapse << | Not Applicable | Completed | - | United States, Ohio ... more >> The Ohio State University General Internal Medicine Columbus, Ohio, United States, 43221 Collapse << |
| NCT01210001 | - | - | Completed | - | - |
| NCT00734474 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT02501616 | Endothelial Function ... more >> Type 2 Diabetes Collapse << | Phase 4 | Unknown | - | Korea, Republic of ... more >> Boramae medical center Recruiting Seoul, Korea, Republic of, 156-707 Contact: Bokyung Koo bokyungkoomd@gmail.com Principal Investigator: Min Kyong Moon Collapse << |
| NCT00536211 | Metabolic Syndrome X ... more >> Insulin Resistance Hypertension Hypercholesterolemia Obesity Collapse << | Not Applicable | Withdrawn(protocol not approve... more >>d by VA R&D) Collapse << | December 2011 | United States, Missouri ... more >> Harry S. Truman Memorial Veterans' Hospital Columbia, Missouri, United States, 65201 Collapse << |
| NCT00184626 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novo Nordisk Investigational Site Istanbul, Turkey, 111 Collapse << |
| NCT02752412 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01549964 | Glycemic Control | Phase 3 | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT03538704 | Endometrial Cancer ... more >> Fertility Collapse << | Not Applicable | Recruiting | December 31, 2020 | China, Beijing ... more >> Peking University People's Hospital Recruiting Beijing, Beijing, China, 100044 Contact: Jianliu Wang, Professor 010-88324381 wangjianliu1203@163.com Collapse << |
| NCT01549964 | - | - | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01476475 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01511172 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT01210001 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01462266 | - | - | Completed | - | - |
| NCT01137812 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00191464 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Hampshire ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lebanon, New Hampshire, United States, 03756 Australia, New South Wales For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. St Leonards, New South Wales, Australia, 2065 Greece For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Thessaloniki, Greece, 56429 India For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mumbai, Maharstra, India, 400 007 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Den Helder, Netherlands, 1783 GZ Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Olesnica, Poland, 56-400 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon-Fri from 9AM to 5PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ponce, Puerto Rico, 00731 Collapse << |
| NCT01476475 | - | - | Completed | - | - |
| NCT03061981 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Maryland ... more >> Early Phase Clinical Unit Baltimore, Maryland, United States, 21225 Collapse << |
| NCT03332771 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | August 6, 2019 | - |
| NCT03194945 | Type 2 Diabetes Mellitus | Phase 4 | Not yet recruiting | December 31, 2020 | China, Guangdong ... more >> endocrinology department of the first affiliated hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT02302326 | Polycystic Ovary Syndrome | Not Applicable | Recruiting | December 2017 | Spain ... more >> Antonio Hernández Recruiting Valencia, Spain, 46017 Contact: Antonio Hernández, Phd, MD 34961622492 hernandez_antmij@gva.es Contact: Celia Bañuls, PhD 34961622757 celia.banuls@uv.es Collapse << |
| NCT01462266 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00683735 | Diabetes | Phase 2 Phase 3 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Collapse << |
| NCT01137812 | - | - | Completed | - | - |
| NCT03514108 | Heart Failure ... more >> Diabetes Collapse << | Phase 4 | Recruiting | September 1, 2023 | Denmark ... more >> Sygehus Sønderjylland, Aabenraa Not yet recruiting Aabenraa, Denmark Contact: Bartlomiej Jonczy Aalborg University Hospital Not yet recruiting Aalborg, Denmark Contact: Søren Vraa Aarhus University Hospital Recruiting Aarhus, Denmark Contact: Henrik Wiggers +45 40136627 henrikwiggers@dadlnet.dk Sub-Investigator: Anders Jorsal Amager Hospital Not yet recruiting Copenhagen, Denmark Contact: Margrethe Ege Olsen Bispebjerg Hospital Not yet recruiting Copenhagen, Denmark Contact: Olav Wendelbo Nielsen Gentofte Hospital Recruiting Copenhagen, Denmark Contact: Morten Schou Glostrup Hospital Not yet recruiting Copenhagen, Denmark Contact: Morten Petersen Herlev Hospital Recruiting Copenhagen, Denmark Contact: Morten Schou Hvidovre Hospital Not yet recruiting Copenhagen, Denmark Contact: Jens Hove Rigshospitalet Recruiting Copenhagen, Denmark Contact: Lars Køber Principal Investigator: Finn Gustafsson Sydvestjysk Sygehus, Esbjerg Not yet recruiting Esbjerg, Denmark Contact: Kirsten Vilain Mikkelsen Herning Hospital Not yet recruiting Herning, Denmark Contact: Morten Bøttcher Regionshospital Nordjylland, Hjørring Not yet recruiting Hjørring, Denmark Contact: Gitte Nielsen Holbæk Hospital Not yet recruiting Holbæk, Denmark Contact: Ilan Raymond Horsens Hospital Not yet recruiting Horsens, Denmark Contact: Karen Kaae Dodt Kolding Hospital Not yet recruiting Kolding, Denmark Contact: Monica Petronela Poenaru Nykøbing Falster Hospital Not yet recruiting Nykøbing Falster, Denmark Contact: Lisbeth Tingsted Odense University Hospital Not yet recruiting Odense, Denmark Contact: Mikael Kjær Poulsen Sjællands Universitetshospital, Roskilde Not yet recruiting Roskilde, Denmark Contact: Nina Pernille Gardshodn Buch Silkeborg Hospital Not yet recruiting Silkeborg, Denmark Contact: Anne Sejr Knudsen Slagelse Sygehus Not yet recruiting Slagelse, Denmark Contact: Henrik Ryde Vejle Hospital Not yet recruiting Vejle, Denmark Contact: Vibeke Brogaard Viborg Hospital Not yet recruiting Viborg, Denmark Contact: Anne Pauline Schrøder Collapse << |
| NCT02924064 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> Investigational center Beijing, China Collapse << |
| NCT00734474 | - | - | Completed | - | - |
| NCT00350779 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03639545 | Vascular Stiffness ... more >> Hypoglycemic Agents Diabetes Complications Diabetes Mellitus, Type 1 Collapse << | Phase 4 | Recruiting | January 30, 2019 | Slovenia ... more >> University Medical Centre Ljubljana Recruiting Ljubljana, Slovenia, SI-1000 Contact: Mojca Lunder, MD, PhD +386 1 5223140 mojca.lunder@kclj.si Contact: Andrej Janez, Prof +386 1 5223564 andrej.janez@kclj.si Principal Investigator: Miodrag Janic, MD, PhD Principal Investigator: Miso Sabovic, Prof Collapse << |
| NCT02058147 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00350779 | - | - | Completed | - | - |
| NCT00860288 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT02730377 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Active, not recruiting | August 12, 2019 | - |
| NCT02058147 | - | - | Completed | - | - |
| NCT01682759 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01682759 | - | - | Completed | - | - |
| NCT01972724 | Type II Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Chagwon, Korea, Republic of Daegu, Korea, Republic of Daejeon, Korea, Republic of Gwangju, Korea, Republic of Gyeonggi-do, Korea, Republic of Jeonju, Korea, Republic of Seoul, Korea, Republic of Ulsan, Korea, Republic of Wonju, Korea, Republic of Collapse << |
| NCT01972724 | - | - | Completed | - | - |
| NCT01909141 | Polycystic Ovary Syndrome ... more >> Infertility Collapse << | Early Phase 1 | Completed | - | Egypt ... more >> Cairo University Cairo, Egypt Collapse << |
| NCT00343395 | Coronary Artery Disease | Phase 4 | Terminated(FDA concerns regard... more >>ing Avandamet) Collapse << | - | United States, Utah ... more >> Intermountain Medical Center Murray, Utah, United States, 84157 Collapse << |
| NCT01617434 | - | - | Completed | - | - |
| NCT01617434 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01909141 | - | - | Completed | - | - |
| NCT01602003 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> LG Life Sciences Seoul, Korea, Republic of, 110-062 Collapse << |
| NCT00807092 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100730 Collapse << |
| NCT00627445 | - | - | Completed | - | - |
| NCT00627445 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100034 Collapse << |
| NCT01664247 | - | - | Completed | - | - |
| NCT01664247 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00420511 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Ontario ... more >> Leadership Sinai Centre for Diabetes Toronto, Ontario, Canada, M5T 3L9 Collapse << |
| NCT00420511 | - | - | Completed | - | - |
| NCT02693392 | Type-2 Diabetes Mellitus | Not Applicable | Completed | - | India ... more >> Department of Medicine and Department of Pharmacology, AIIMS Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT00402909 | Type 2 Diabetes | Phase 4 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07974 Collapse << |
| NCT00046462 | Diabetes Mellitus | Phase 3 | Completed | - | United States, New Jersey ... more >> Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00044447 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Dallas Diabetes & Endocrine Center Dallas, Texas, United States, 75230 Collapse << |
| NCT02338921 | Type 2 Diabetes | Phase 4 | Recruiting | December 2018 | Korea, Republic of ... more >> Soo Lim Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Dong Hwa Lee, MD Collapse << |
| NCT00035542 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00237237 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Germany Investigative Centers Nurnberg, Germany Collapse << |
| NCT01779362 | Prediabetes T... more >>ype 2 Diabetes Collapse << | Phase 3 | Active, not recruiting | August 2019 | United States, Illinois ... more >> Jesse Brown VA Medical Center Chicago, Illinois, United States, 60612 University of Chicago Chicago, Illinois, United States, 60637 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Washington VA Puget Sound Health Care System Seattle, Washington, United States, 98108 Collapse << |
| NCT00097500 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Finland ... more >> Research Site Helsinki, Finland Netherlands Research Site Amsterdam, Netherlands Sweden Research Site Goteborg, Sweden Collapse << |
| NCT00097500 | - | - | Completed | - | - |
| NCT00039013 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT03766256 | Gestational Diabetes | Not Applicable | Not yet recruiting | December 31, 2019 | United States, California ... more >> Toluca Lake Health Center Burbank, California, United States, 91505 100 Medical Plaza Primary Care Suites 455 & 490 Los Angeles, California, United States, 90024 Internal Medicine Women's Health Clinic Suites 250 & 290 Los Angeles, California, United States, 90024 UCLA Internal Medicine Family Westwood Suite 465 Los Angeles, California, United States, 90024 UCLA Internal Medicine Geriatrics Suite 365 & 420 Los Angeles, California, United States, 90024 CPN Brentwood Los Angeles, California, United States, 90049 CPN West Washington Internal Medicine Los Angeles, California, United States, 90066 Pacific Palisades Pacific Palisades, California, United States, 90272 12th Street Clinic Santa Monica, California, United States, 90401 Wilshire Office Santa Monica, California, United States, 90401 CPN Santa Monica 15th Street Family Medicine Practice Santa Monica, California, United States, 90404 CPN/Santa Monica Parkside Santa Monica, California, United States, 90404 Santa Moica Bay Physicians/20th Street 10th Floor Santa Monica, California, United States, 90404 Santa Monica Bay Physicians Plaza Santa Monica, California, United States, 90404 Santa Monica Bay Physicians/20th Street 3rd Floor Santa Monica, California, United States, 90404 Santa Monica Internal Medicine Santa Monica, California, United States, 90404 UCLA Family Health Clinic Santa Monica, California, United States, 90404 Santa Moica Bay Physicians Ocean Park Santa Monica, California, United States, 90405 Jack H. Skirball Health Center Woodland Hills, California, United States, 91364 CPN Woodland Hills Practice Woodland Hills, California, United States, 91367 Collapse << |
| NCT00384215 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT03351478 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | May 2019 | - |
| NCT00283049 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(Due to technical is... more >>sues relating to the Electronic diary data.) Collapse << | - | United States, New Jersey ... more >> sanofi-aventis, US Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00283049 | - | - | Terminated(Due to technical is... more >>sues relating to the Electronic diary data.) Collapse << | - | - |
| NCT00044668 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Hungary ... more >> Diagnostic Units Hungary Kft. Budapest, Hungary, H 1036 Peterfy Teaching Hospital Budapest, Hungary, H 1076 Uzsoki Street Municipal Hospital Budapest, Hungary, H 1145 Collapse << |
| NCT01101204 | Diabetes Dysl... more >>ipidemia Inflammation Cytokines Collapse << | Not Applicable | Unknown | December 2012 | Poland ... more >> Department of Clinical Pharmacology Not yet recruiting Katowice, Poland, 40752 Contact: Krzysztof Labuzek, MD, PhD +48 32 252 39 02 lbuldak@gmail.com Sub-Investigator: Łukasz Bułdak, MD Principal Investigator: Bogusław Okopień, MD, PhD Sub-Investigator: Krzysztof Labuzek, MD, PhD Collapse << |
| NCT01140438 | Diabetes | Not Applicable | Active, not recruiting | December 2018 | Norway ... more >> Valdemar Grill Trondheim, Norway, 7006 Collapse << |
| NCT00373178 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Greece ... more >> AHEPA University Hospital of Thessaloniki Thessaloniki, Greece Aristotle University of Thessaloniki Thessaloniki, Greece Collapse << |
| NCT00996658 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01778049 | - | - | Completed | - | - |
| NCT00622284 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00996658 | - | - | Completed | - | - |
| NCT01778049 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00717457 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT02142309 | Type 2 Diabetes | Phase 4 | Unknown | January 2017 | Italy ... more >> Department of Geriatrics and Metabolic Diseases Active, not recruiting Naples, Italy, 80138 Katherine Esposito Recruiting Naples, Italy, 80138 Contact: Maria Ida Maiorino, MD mariaida.maiorino@unina2.it Sub-Investigator: Giuseppe Bellastella, MD Collapse << |
| NCT00870194 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Argentina ... more >> Research Site Buenos Aires, Argentina Research Site Morón, Argentina Australia Research Site Adelaide, Australia Research Site Geelong, Australia Research Site Melbourne, Australia Germany Research Site Aschaffenburg, Germany Research Site Asslar, Germany Research Site Beckum-Neubeckum, Germany Research Site Berlin, Germany Research Site Bosenheim, Germany Research Site Essen, Germany Research Site Falkensee, Germany Research Site Furth im Wald, Germany Research Site Grevenbroich, Germany Research Site Hamburg-Othmarschen, Germany Research Site Hohenmolsen, Germany Research Site Leipzig, Germany Research Site Neuwied, Germany Research Site Pohlheim, Germany Research Site Speyer, Germany Greece Research Site Athens, Greece Research Site Thessaloniki, Greece India Research Site Ahmedabad, India Research Site Bangalore, India Research Site Coimbatore, India Research Site Indore, India Research Site Jaipur, India Korea, Republic of Research Site Daegu, Korea, Republic of Research Site Gwangju, Korea, Republic of Research Site Seoul, Korea, Republic of Research Site Ulsan, Korea, Republic of Mexico Research Site Coatzacoalcos, Mexico Research Site Guadalajara, Mexico Research Site Merida, Mexico Research Site Monterrey, Mexico Research Site Tampico, Mexico Collapse << |
| NCT02002221 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 4 | Completed | - | Japan ... more >> Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 810-8798 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 815-0071 Novartis Investigative Site Fukutsu-city, Fukuoka, Japan, 811-3217 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 800-0295 Novartis Investigative Site Koriyama-city, Fukushima, Japan, 963-8851 Novartis Investigative Site Mito-city, Ibaraki, Japan, 311-4153 Novartis Investigative Site Ibusuki-city, Kagoshima, Japan, 891-0401 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0802 Novartis Investigative Site Kumamoto-city, Kumamoto, Japan, 861-8039 Novartis Investigative Site Kumamoto-city, Kumamoto, Japan, 861-8520 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8533 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Sendai-city, Miyagi, Japan, 980-0021 Novartis Investigative Site Hirakata-City, Osaka, Japan, 573-0153 Novartis Investigative Site Izumisano-city, Osaka, Japan, 598-8577 Novartis Investigative Site Osaka-city, Osaka, Japan, 530-0001 Novartis Investigative Site Osaka-city, Osaka, Japan, 534-0021 Novartis Investigative Site Takatsuki-city, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Saitama-city, Saitama, Japan, 336-0963 Novartis Investigative Site Sayama-city, Saitama, Japan, 350-1305 Novartis Investigative Site Tokorozawa-city, Saitama, Japan, 359-1161 Novartis Investigative Site Adachi-ku, Tokyo, Japan, 123-0845 Novartis Investigative Site Chiyoda-ku, Tokyo, Japan, 101-0024 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 103-0027 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 103-0028 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 104-0061 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0041 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Ota-ku, Tokyo, Japan, 144-0051 Novartis Investigative Site Suginami-ku, Tokyo, Japan, 166-0004 Collapse << |
| NCT00870194 | - | - | Completed | - | - |
| NCT02002221 | - | - | Completed | - | - |
| NCT02284893 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02284893 | - | - | Completed | - | - |
| NCT00086515 | - | - | Completed | - | - |
| NCT01755468 | Type 2 Diabetes | Phase 3 | Completed | - | Canada, Ontario ... more >> Mount Sinai Hospital Toronto, Ontario, Canada, M5G1X5 Collapse << |
| NCT00332085 | Type 2 Diabetes | Phase 1 | Terminated(Issues w/recruitmen... more >>t) Collapse << | - | United States, Washington ... more >> Bastyr University Campus SPR Kenmore, Washington, United States, 98036 Bastyr Center for Natural Health Seattle, Washington, United States Collapse << |
| NCT00622284 | - | - | Completed | - | - |
| NCT00086515 | Diabetes Mellitus, Type II | Phase 3 | Completed | - | - |
| NCT01006603 | - | - | Completed | - | - |
| NCT02426294 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | May 2016 | Korea, Republic of ... more >> Pusan National University Hospital Recruiting Busan, Korea, Republic of, 602-739 Contact: Sang Soo Kim, MD., PhD. +82-51-240-7228 drsskim7@gmail.com Contact: Jong Ho Kim, MD. +82-51-240-7983 bedaya@hanmail.net Collapse << |
| NCT01006603 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT00138567 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Investigative Site East Hanover, New Jersey, United States, 07936 Germany Novartis Investigative site Investigative Centers, Germany Collapse << |
| NCT01340768 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01340768 | - | - | Completed | - | - |
| NCT01814748 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01814748 | - | - | Completed | - | - |
| NCT00309608 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01167881 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01167881 | - | - | Completed | - | - |
| NCT00309608 | - | - | Completed | - | - |
| NCT00545584 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00709917 | - | - | Completed | - | Belgium ... more >> Brussels, Belgium, 1070 Luxembourg Luxembourg, Luxembourg Collapse << |
| NCT00150410 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00099892 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis Pharmaceuticals East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT00545584 | - | - | Completed | - | - |
| NCT01035528 | Coronary Artery Disease ... more >> Type 2 Diabetes Collapse << | Phase 4 | Unknown | August 2010 | Germany ... more >> Staedt. Klinikum Muenchen Bogenhausen Munich, Bavaria, Germany, 81925 Collapse << |
| NCT02291510 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT02291510 | - | - | Completed | - | - |
| NCT00361738 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Argentina ... more >> Sanofi-Aventis Administrative Office San Isidro, Argentina Australia, New South Wales Sanofi-Aventis Administrative Office Cove, New South Wales, Australia Belgium Sanofi-Aventis Administrative Office Diegem, Belgium Chile Sanofi-Aventis Administrative Office Santiago, Chile Denmark Sanofi-Aventis Administrative Office Horsholm, Denmark France Sanofi-Aventis Administrative Office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Poland Sanofi-Aventis Administrative Office Warszawa, Poland South Africa Sanofi-Aventis Administrative Office Midrand, South Africa Collapse << |
| NCT00471523 | Infertility P... more >>olycystic Ovary Syndrome Anovulation Collapse << | Phase 4 | Completed | - | Italy ... more >> "Pugliese Hospital" Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT00601250 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01195090 | - | - | Completed | - | - |
| NCT01195090 | Type 2 Diabetes | Phase 4 | Completed | - | Taiwan ... more >> Division of Endocrinology and Metabolism, Department of Internal Medicine, Mackay Memorial Hospital Taipei, Taiwan, 10449 Collapse << |
| NCT03060980 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Decision by Sponsor... more >>) Collapse << | - | - |
| NCT00602472 | - | - | Completed | - | - |
| NCT00601250 | - | - | Completed | - | - |
| NCT00299871 | Type 2 Diabetes | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Canada Sanofi-Aventis Administrative Office Laval, Canada Poland Sanofi-Aventis Administrative Office Warszawa, Poland Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Ukraine Sanofi-Aventis Administrative Office Kiev, Ukraine Collapse << |
| NCT00417729 | Diabetes Mellitus | Phase 4 | Completed | - | Taiwan ... more >> Taichung Veterans General Hospital Taichung, Taiwan Collapse << |
| NCT03061214 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | April 17, 2019 | - |
| NCT00602472 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02467920 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Hubei ... more >> Tongji Hospital, Tongji Medical College, Huazhong University of Science and technology Wuhan, Hubei, China, 430000 Collapse << |
| NCT00116922 | Impaired Glucose Tolerance ... more >> Diabetes Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> UWO Research Park London, Ontario, Canada, N6G 4X8 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 Collapse << |
| NCT01859793 | Diabetes Athe... more >>rosclerosis Collapse << | Phase 4 | Completed | - | United States, Wisconsin ... more >> Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT01859793 | - | - | Completed | - | - |
| NCT00103857 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00441363 | Type 2 Diabetes | Phase 3 | Terminated(Failure to Recruit ... more >>in a Timely manner) Collapse << | - | - |
| NCT01798875 | PCOS | Phase 4 | Unknown | December 2015 | Poland ... more >> Department of Clinical and Experimental Endocrinology, Medical University of Gdansk Recruiting Gdansk, Poland Contact: Dominik Rachon, MD, PhD +48583491947 drachon@gumed.edu.pl Principal Investigator: Monika Kuligowska-Jakubowska, MD Principal Investigator: Jolanta Dardzinska, MD, PhD Sub-Investigator: Agnieszka Kowalewska-Wlas, MD, PhD Principal Investigator: Aleksandra Rutkowska, MSc, PhD Principal Investigator: Justyna Breska-Kruszewska, MSc Collapse << |
| NCT00842140 | Polycystic Ovary Syndrome | Not Applicable | Completed | - | Brazil ... more >> University of Sao Paulo Ribeirao Preto, Sao Paulo, Brazil, 14049-900 Collapse << |
| NCT00103857 | - | - | Completed | - | - |
| NCT01169090 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT01206712 | - | - | Completed | - | Germany ... more >> IKFE Institute for Clinical Research and Development Mainz, Germany, 55116 Collapse << |
| NCT02627027 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Jeonju-si, Jeollabuk-do, Korea, Republic of Collapse << |
| NCT00789035 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00441363 | - | - | Terminated(Failure to Recruit ... more >>in a Timely manner) Collapse << | - | - |
| NCT00147719 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00789035 | - | - | Completed | - | - |
| NCT01215097 | - | - | Completed | - | - |
| NCT01863667 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | - | - |
| NCT01215097 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | China ... more >> 1218.65.86007 Boehringer Ingelheim Investigational Site Beijing, China 1218.65.86011 Boehringer Ingelheim Investigational Site Chongqing, China 1218.65.86008 Boehringer Ingelheim Investigational Site Dalian, China 1218.65.86010 Boehringer Ingelheim Investigational Site Fuzhou, China 1218.65.86014 Boehringer Ingelheim Investigational Site Hangzhou, China 1218.65.86005 Boehringer Ingelheim Investigational Site Hefei, China 1218.65.86006 Boehringer Ingelheim Investigational Site Hefei, China 1218.65.86012 Boehringer Ingelheim Investigational Site Nanjing, China 1218.65.86001 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86002 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86003 Boehringer Ingelheim Investigational Site Shanghai, China 1218.65.86004 Boehringer Ingelheim Investigational Site Suzhou, China 1218.65.86015 Boehringer Ingelheim Investigational Site Wenzhou, China 1218.65.86009 Boehringer Ingelheim Investigational Site Wuhan, China 1218.65.86013 Boehringer Ingelheim Investigational Site Yangzhou, China Malaysia 1218.65.60002 Boehringer Ingelheim Investigational Site Johor Bahru,, Malaysia 1218.65.60001 Boehringer Ingelheim Investigational Site Kelantan, Malaysia Philippines 1218.65.63001 Boehringer Ingelheim Investigational Site Marikina, Philippines 1218.65.63002 Boehringer Ingelheim Investigational Site San Juan, Philippines Collapse << |
| NCT00401973 | Schizophrenia ... more >> Schizoaffective Disorders Collapse << | Phase 3 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. National City, California, United States, 91950 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Canton, Ohio, United States, 44708 United States, Tennessee For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Johnson City, Tennessee, United States, 37614 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Memphis, Tennessee, United States, 38105 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Desoto, Texas, United States, 75115 Brazil For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rio De Janeiro, Brazil, 21020-130 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Salvador, Brazil, 40301500 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beer Yaacov, Israel, 70350 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 143-711 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mexico City, Mexico, 14050 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bayamon, Puerto Rico, 00961 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ponce, Puerto Rico, 00717 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico, 00907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 117603 Collapse << |
| NCT00729326 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00401973 | - | - | Completed | - | - |
| NCT03307252 | Healthy | Phase 1 | Completed | - | Germany ... more >> CTC North GmbH & Co. KG, Hamburg Hamburg, Germany, 20251 Collapse << |
| NCT00729326 | - | - | Completed | - | - |
| NCT00577590 | Diabetes Mellitus, Type II | Not Applicable | Completed | - | United States, Missouri ... more >> Washington University in St. Louis Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00577590 | - | - | Completed | - | - |
| NCT02648204 | - | - | Completed | - | - |
| NCT01863667 | - | - | Terminated | - | - |
| NCT00674466 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Quebec ... more >> Maggie Wang Montreal, Quebec, Canada, H2X 3Y8 Collapse << |
| NCT00674466 | - | - | Completed | - | - |
| NCT01915264 | - | - | Withdrawn | March 2017 | - |
| NCT02229383 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02229383 | - | - | Completed | - | - |
| NCT02177643 | Diabetes-Related Complications... more >> Diabetes Mellitus, Type 2 Insulin Resistance Collapse << | Phase 2 | Unknown | - | Brazil ... more >> Universidade Estadual de Feira de Santana Recruiting Feira de Santana, Bahia, Brazil Contact: Dra. Ana Mayra A. Oliveira, PhD +55 (075) 3625-4027 anamayra@uol.com.br Principal Investigator: Dra. Ana Mayra A. Oliveira, PhD Centro de Estudos em Diabetes e Hipertensão Recruiting Fortaleza, Ceará, Brazil Contact: Dra. Adriana C. Forti, PhD +55 (085) 3105-8300 cedh@cedh.med.br Principal Investigator: Dra. Adriana C. Forti, PhD Universidade Estadual de Campinas Recruiting Campinas, São Paulo, Brazil Contact: Dra. Maria Cândida R. Parisi, PhD +55 (019) 98196-2242 emaildacandida@uol.com.br Contact: Dr. Daniel Minuti, MD Msc +55 (019) 98128-2535 danielminutti@yahoo.com.br Sub-Investigator: Dr. Daniel Minuti, MD Msc Collapse << |
| NCT00396851 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | - | India ... more >> PGIMER Not yet recruiting Chandigarh, India, 160012 Contact: Samir Malhotra samirmalhotra345@yahoo.com Sub-Investigator: Promila Pandhi, MD,DM Collapse << |
| NCT02648204 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01517412 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01691989 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01517373 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01517412 | - | - | Completed | - | - |
| NCT00807092 | - | - | Completed | - | - |
| NCT01930188 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01612741 | - | - | Completed | - | India ... more >> Many Locations, India Collapse << |
| NCT00749190 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00749190 | - | - | Completed | - | - |
| NCT00460941 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01106677 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01517373 | - | - | Completed | - | - |
| NCT00611884 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00423501 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT00611884 | - | - | Completed | - | - |
| NCT00473525 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01106677 | - | - | Completed | - | - |
| NCT01649297 | - | - | Completed | - | - |
| NCT01649297 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00833027 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02452632 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Site KR00002 Busan, Korea, Republic of Site KR00003 Busan, Korea, Republic of Site KR00007 Daegu, Korea, Republic of Site KR00011 Daegu, Korea, Republic of Site KR00019 Dong Gu Gwangju, Korea, Republic of Site KR00008 Gangwon-do, Korea, Republic of Site KR00018 Gyeonggi-do, Korea, Republic of Site KR00022 Gyeonggi-do, Korea, Republic of Site KR00014 Incheon, Korea, Republic of Site KR00020 Jeollabuk-do, Korea, Republic of Site KR00021 Kyungsangnam-do, Korea, Republic of Site KR00001 Seoul, Korea, Republic of Site KR00004 Seoul, Korea, Republic of Site KR00005 Seoul, Korea, Republic of Site KR00006 Seoul, Korea, Republic of Site KR00009 Seoul, Korea, Republic of Site KR00010 Seoul, Korea, Republic of Site KR00012 Seoul, Korea, Republic of Site KR00013 Seoul, Korea, Republic of Site KR00016 Seoul, Korea, Republic of Site KR00017 Seoul, Korea, Republic of Site KR00015 Suwon, Korea, Republic of Collapse << |
| NCT02973100 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00833027 | - | - | Completed | - | - |
| NCT03208309 | Complications of Diabetes Mell... more >>itus Diabetes Mellitus Diabetes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Brazil ... more >> Universidade Estadual de Feira de Santana Feira de Santana, Bahia, Brazil Centro de Estudos em Diabetes e Hipertensão Fortaleza, Ceará, Brazil Universidade Estadual de Campinas Campinas, São Paulo, Brazil Collapse << |
| NCT01541215 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02973100 | - | - | Completed | - | - |
| NCT01289119 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00624364 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | South Africa ... more >> Novo Nordisk Investigational Site Bloemfontein, Free State, South Africa, 9301 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1827 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 1829 Novo Nordisk Investigational Site Johannesburg, Gauteng, South Africa, 2001 Novo Nordisk Investigational Site Pretoria, Gauteng, South Africa, 0116 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4001 Novo Nordisk Investigational Site Durban, KwaZulu-Natal, South Africa, 4091 Novo Nordisk Investigational Site Pietermaritzburg, KwaZulu-Natal, South Africa, 3235 Novo Nordisk Investigational Site Brits, North West, South Africa, 0250 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7708 Novo Nordisk Investigational Site Cape Town, Western Cape, South Africa, 7800 Novo Nordisk Investigational Site Benoni, South Africa, 1500 Novo Nordisk Investigational Site Bloemfontein, South Africa, 27 51 Novo Nordisk Investigational Site Kimberley, South Africa, 8301 Novo Nordisk Investigational Site Richards Bay, South Africa, 3900 Collapse << |
| NCT01541215 | - | - | Completed | - | - |
| NCT01289119 | - | - | Completed | - | - |
| NCT00108615 | Glucose Metabolism Disorders ... more >> Diabetes Collapse << | Phase 4 | Completed | - | United States, Arkansas ... more >> Central Arkansas Veterans HCS Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT02033876 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00479466 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Sufficient data reg... more >>arding the dose-response to MK-0893 had been obtained from the first cohort of the study to assess the safety and efficacy) Collapse << | - | - |
| NCT00839527 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00479466 | - | - | Terminated(Sufficient data reg... more >>arding the dose-response to MK-0893 had been obtained from the first cohort of the study to assess the safety and efficacy) Collapse << | - | - |
| NCT00622089 | Type 2 Diabetes | Phase 2 Phase 3 | Terminated(Program terminated) | December 2008 | - |
| NCT00998686 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | November 2011 | - |
| NCT00348712 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT00839527 | - | - | Completed | - | - |
| NCT01376323 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Anniston, Alabama, United States, 36207 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 GSK Investigational Site Miramar, Florida, United States, 33025 France GSK Investigational Site Nantes cedex 01, France, 44093 GSK Investigational Site Pierre-Bénite Cedex, France, 69495 GSK Investigational Site Rennes Cedex, France, 35046 GSK Investigational Site Rueil-Malmaison, France, 92502 Spain GSK Investigational Site Alicante, Spain, 03114 GSK Investigational Site Badalona, Spain, 08916 GSK Investigational Site Granada, Spain, 18004 GSK Investigational Site Madrid, Spain, 28046 United Kingdom GSK Investigational Site Edinburgh, Midlothian, United Kingdom, EH4 2XU GSK Investigational Site Cambridge, United Kingdom, CB2 0GG GSK Investigational Site Coventry, United Kingdom, CV2 2DX GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT00701935 | Diabetes Mellitus | Phase 2 | Terminated(Enrollment was much... more >> slower than anticipated, leading to a decision to terminate the study early for enrollment futility.) Collapse << | - | United States, Arizona ... more >> Research Site Temple, Arizona, United States United States, Colorado Research Site Colorado Springs, Colorado, United States United States, Hawaii Research Site Honolulu, Hawaii, United States United States, Nebraska Research Site Bellevue, Nebraska, United States United States, Nevada Research Site Las Vegas, Nevada, United States Canada, Alberta Research Site Calgary, Alberta, Canada Canada, British Columbia Research Site Vancouver, British Columbia, Canada Research Site Victoria, British Columbia, Canada Canada, Manitoba Research Site Winnipeg, Manitoba, Canada Canada, New Brunswick Research Site Saint John, New Brunswick, Canada Canada, Ontario Research Site Brampton, Ontario, Canada Research Site London, Ontario, Canada Research Site Ottawa, Ontario, Canada Research Site Toronto, Ontario, Canada Canada, Quebec Research Site Chicoutimi, Quebec, Canada Research Site Pointe-Claire, Quebec, Canada Canada Research Site Quebec, Canada Collapse << |
| NCT02635386 | Polycystic Ovary Syndrome ... more >> Obesity Collapse << | Phase 3 | Recruiting | August 2019 | United States, Louisiana ... more >> Woman's Hospital Recruiting Baton Rouge, Louisiana, United States, 70815 Contact: Karen Elkind-Hirsch, PhD. 225-231-5278 Contact: Ericka Seidemann, MS 225-231-5275 ericka.seidemann@womans.org Principal Investigator: Karen Elkind-Hirsch, Ph.D. Sub-Investigator: Renee Harris, M.D. Collapse << |
| NCT00837590 | Pre-diabetes ... more >>Obesity Collapse << | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana Clinical Research Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00701935 | - | - | Terminated(Enrollment was much... more >> slower than anticipated, leading to a decision to terminate the study early for enrollment futility.) Collapse << | - | - |
| NCT02373865 | Diabetes Mellitus Type 2 | Phase 4 | Terminated(Premature terminati... more >>on due to insufficient patient recruitement..) Collapse << | - | Germany ... more >> GWT-TUD GmbH / Studienzentrum Hanefeld Dresden, Germany, 01307 Collapse << |
| NCT01175473 | - | - | Completed | - | - |
| NCT01482286 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT02549924 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(no financial suppor... more >>t) Collapse << | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02004886 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02786823 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | India ... more >> All India Institute of Medical Sciences, Bhubaneswar Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT01175473 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Sanofi-Aventis Investigational Site Number 276006 Berlin, Germany, 14050 Sanofi-Aventis Investigational Site Number 276004 Kiel, Germany, 24105 Sanofi-Aventis Investigational Site Number 276002 Mainz, Germany, 55116 Sanofi-Aventis Investigational Site Number 276003 Mannheim, Germany, 68167 Sanofi-Aventis Investigational Site Number 276005 Mönchengladbach, Germany, 41061 Sanofi-Aventis Investigational Site Number 276007 München, Germany, 80636 Sanofi-Aventis Investigational Site Number 276001 Neuss, Germany, 41460 Collapse << |
| NCT02004886 | - | - | Completed | - | - |
| NCT02564926 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Research Site Ansan-si, Korea, Republic of, 15355 Research Site Changwon-si, Korea, Republic of, 51353 Research Site Daegu, Korea, Republic of, 41931 Research Site Daejeon-si, Korea, Republic of, 35233 Research Site Goyang-si, Korea, Republic of, 10326 Research Site Incheon, Korea, Republic of, 21565 Research Site Seongnam-si, Korea, Republic of, 13620 Research Site Seoul, Korea, Republic of, 01830 Research Site Seoul, Korea, Republic of, 04401 Research Site Seoul, Korea, Republic of, 05278 Research Site Seoul, Korea, Republic of, 06273 Research Site Suwon-si, Korea, Republic of, 16499 Research Site Uijeongbu-si, Korea, Republic of, 11765 Research Site Wonju-si, Korea, Republic of, 220-701 Collapse << |
| NCT01376323 | - | - | Completed | - | - |
| NCT03353948 | Polycystic Ovary Syndrome ... more >> Obesity Infertility, Female Collapse << | Phase 4 | Completed | - | - |
| NCT00961909 | Diabetes Mellitus Type 2 | Phase 1 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Chula Vista, California, United States, 91911 United States, Florida Ft. Myers, Florida, United States, 33901 Miramar, Florida, United States, 33025 United States, Texas San Antonio, Texas, United States, 78209 San Antonio, Texas, United States, 78229-4801 Collapse << |
| NCT00333151 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01471808 | Diabetes Mellitus, Type 2 | Phase 4 | Enrolling by invitation | December 2021 | - |
| NCT02235298 | Type 2 Diabetes | Phase 4 | Recruiting | December 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Gianluca Iacobellis, MD PhD giacobellis@med.miami.edu Principal Investigator: Gianluca Iacobellis, MD PhD University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Gianluca Iacobellis, MD PhD giacobellis@med.miami.edu Collapse << |
| NCT03268005 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | January 29, 2019 | - |
| NCT01059825 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT03757910 | Alzheimer Disease | Early Phase 1 | Not yet recruiting | August 2019 | United States, New York ... more >> Columbia University Medical Center Not yet recruiting New York, New York, United States, 10032 Contact: Jose A Luchsinger, MD 212-305-4730 jal94@cumc.columbia.edu Principal Investigator: Jose A Luchsinger, MD Sub-Investigator: F X Pi-Sunyer, MD Albert Einstein College of Medicine Not yet recruiting New York, New York, United States, 10461 Principal Investigator: Jill Crandall, MD Collapse << |
| NCT00449930 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00850239 | Type 2 Diabetes Mellitus | Phase 3 | Terminated | December 2010 | - |
| NCT00449930 | - | - | Completed | - | - |
| NCT02981121 | PreDiabetes | Phase 4 | Recruiting | November 2020 | Korea, Republic of ... more >> Kyunghee University Medical Center Recruiting Seoul, Korea, Republic of, 02447 Contact: Jeong-taek Woo, MD, PhD +82-2-958-8128 jtwoomd@khmc.or.kr Contact: Sang Youl Rhee, MD, PhD +82-2-958-8220 bard95@hanmail.net Principal Investigator: Jeong-taek Woo, MD, PhD Sub-Investigator: Suk Chon, MD, PhD Sub-Investigator: Sang Youl Rhee, MD, PhD Collapse << |
| NCT01066715 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03603080 | Obesity | Phase 4 | Not yet recruiting | November 1, 2019 | United States, Connecticut ... more >> Hartford Hospital Not yet recruiting Hartford, Connecticut, United States, 06102 Contact: Pavlos K Papasavas, MD 860-246-2071 pavlos.papasavas@hhchealth.org Contact: Darren Tishler, MD 8605244566 darren.tishler@hhchealth.org Sub-Investigator: Pavlos K Papasavas, MD Sub-Investigator: Darren Tishler, MD Principal Investigator: Devika Umashanker, MD Collapse << |
| NCT02443922 | Type 2 Diabetes | Not Applicable | Unknown | December 2015 | - |
| NCT03770052 | Type 2 Diabetes Mellitus ... more >> Inadequate Glucose Control Collapse << | Phase 4 | Not yet recruiting | December 31, 2020 | Korea, Republic of ... more >> Pusan National University Hospital Busan, Korea, Republic of Collapse << |
| NCT01933256 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 Collapse << |
| NCT01075282 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02621489 | Atherosclerosis ... more >> Diabetes Restenosis Collapse << | Phase 4 | Recruiting | December 2019 | Sweden ... more >> Dept of clinical science and education Karolinska Institutet Södersjukhuset Recruiting Stockholm, Other, Sweden, 11883 Contact: Thomas Nyström, MD, PhD +4686163211 thomas.nystrom@sodersjukhuset.se Contact: Nils Witt, Md, PhD +4686161000 Sub-Investigator: Nils Witt, MD, PhD Sub-Investigator: Irene Santos, MD Collapse << |
| NCT01269996 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Hong Kong ... more >> Department of Medicine & Therapeutics, Prince of Wales Hospital, The Chinese University of Hong Kong Hong Kong, Hong Kong Collapse << |
| NCT00783744 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Austria ... more >> Sanofi aventis administrative office Vienna, Austria Finland Sanofi-aventis administrative office Helsinki, Finland France Sanofi-aventis administrative office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Spain Sanofi-Aventis Administrative Office Barcelona, Spain Sweden Sanofi-Aventis Administrative Office Bromma, Sweden Switzerland Sanofi-Aventis Administrative Office Geneva, Switzerland United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00474838 | Type 2 Diabetes Mellitus ... more >> Pancreatic Beta Cell Function Glucotoxicity Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Hanyang University Medical Center Kuri, Kyunggi-do, Korea, Republic of, 471-020 The Catholic University of Korea Bucheon St.Mary's Hospital Bucheon, Korea, Republic of Inha University Hospital In Cheon, Korea, Republic of, 400-711 Jeju National University Hospital Jeju-do, Korea, Republic of Kyunghee University Medical Center Seoul, Korea, Republic of, 130-702 Korea University Guro Hospital Seoul, Korea, Republic of, 152-730 Kyung Hee University East Weast Neo Medicalcenter Seoul, Korea, Republic of Ajou University Medical Center Suwon, Korea, Republic of, 443-721 Collapse << |
| NCT00991055 | Type 2 Diabetes | Phase 4 | Unknown | October 2010 | Taiwan ... more >> National Cheng Kung University Hospital Recruiting Tainan, Taiwan Contact: Ta-Jen Wu, Dr 886-6-2353535 ext 5387 djwu@mail.ncku.edu.tw Principal Investigator: Ta-Jen Wu, Dr. Collapse << |
| NCT01059825 | - | - | Completed | - | - |
| NCT03746106 | Vitamin B1 Deficiency ... more >> Thiamine Deficiency Collapse << | Phase 4 | Not yet recruiting | October 2019 | - |
| NCT02554877 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01805830 | Type 2 Diabetes | Phase 3 | Completed | - | Korea, Republic of ... more >> Handok Pharmaceuticals CO. LTD Seoul, Korea, Republic of Collapse << |
| NCT00358124 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT02554877 | - | - | Completed | - | - |
| NCT02480465 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | March 2017 | Korea, Republic of ... more >> Korea University Anam Hospital Recruiting Seoul, Korea, Republic of Contact: Dong-sub Choi Collapse << |
| NCT01003483 | Obesity | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Research and Clinical Center for Infertility Yazd, Iran, Islamic Republic of, 8916877391 Collapse << |
| NCT01903356 | - | - | Completed | - | Korea, Republic of ... more >> One or multiple Investigational sites, Korea, Republic of Collapse << |
| NCT01144728 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Kazakhstan ... more >> Sanofi-Aventis Administrative Office Almaty, Kazakhstan Collapse << |
| NCT01075282 | - | - | Completed | - | - |
| NCT02304081 | Type2 Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Profil Mainz GmbH & Co. KG Mainz, Germany, 55116 Collapse << |
| NCT01341795 | - | - | Unknown | - | Korea, Republic of ... more >> Ajou University School of Medicine Recruiting Suwon, Gyeonggi, Korea, Republic of, 433-721 Contact: Doo-Yeoun Cho, MD +82-31-219-4271 dooycho@ajou.ac.kr Sub-Investigator: Young-Sang Kim, MD Collapse << |
| NCT00546442 | Chronic Hepatitis C | Phase 4 | Completed | - | Spain ... more >> Hospital Germans Trias i Pujol Badalona, Barcelona, Spain, 08916 Hospital Parc Taulí Sabadell, Barcelona, Spain, 08208 Hospital de Donostia San Sebastian, Guipuzcoa, Spain, 20014 Fundación Hospital de Alcorcón Alcorcón, Madrid, Spain, 28922 Hospital del Mar Barcelona, Spain, 08003 Hospital General de Ciudad Real Ciudad Real, Spain, 13005 Hospital Reina Sofía Córdoba, Spain, 14004 Hospital Clínico Universitario San Cecilio Granada, Spain, 18003 Hospital Virgen de las Nieves Granada, Spain, 18004 Hospital de Leon Leon, Spain, 24071 Hospital de la Princesa Madrid, Spain, 28006 Hospital Gregorio Marañón Madrid, Spain, 28007 Hospital Carlos III Madrid, Spain, 28029 Hospital Puerta de Hierro Madrid, Spain, 28035 Hospital La Paz Madrid, Spain, 28046 Hospital Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Virgen Macarena Sevilla, Spain, 41009 Hospital Universitario de Valme Sevilla, Spain, 41014 Hospital General Universitario Valencia, Spain, 46014 Collapse << |
| NCT01648582 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100730 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chengdu, China, 610041 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chongqing, China, 400030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dalian, China, 116011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guiyang, China, 550004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hefei, China, 230022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanchang, China, 330006 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210006 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanning, China, 530021 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shenyang, China, 110004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shijiazhuang, China, 050051 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wu Han, China, 430022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yangzhou, China, 225001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Zhenjiang, China, 212000 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bucheon,, Korea, Republic of, 420-717 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyunggi-Do, Korea, Republic of, 425-020 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 137-701 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wonju-Si, Korea, Republic of, 220-701 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chihuahua, Mexico, 31238 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Monterrey, Mexico, 64460 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chelyabinsk, Russian Federation, 454047 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 127018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 192012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sankt-Petersburg, Russian Federation, 190000 Collapse << |
| NCT01364675 | Pre-diabetes ... more >>Pre-hypertension Collapse << | Not Applicable | Unknown | December 2016 | Thailand ... more >> Ubon ratchathani Public Health Office Not yet recruiting Muang District, Ubon ratchathani, Thailand, 34000 Principal Investigator: Phisitt Vejakama, M.D. Collapse << |
| NCT00263965 | Type 2 Diabetes | Phase 2 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | Finland ... more >> Research Site Helsinki, Finland Italy Research Site Pisa, Italy United Kingdom Research Site Oxford, United Kingdom Collapse << |
| NCT02027753 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Collapse << |
| NCT00282451 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Egypt ... more >> Novo Nordisk Investigational Site Aghakhan, Egypt Novo Nordisk Investigational Site Giza, Egypt Saudi Arabia Novo Nordisk Investigational Site Jeddah, Saudi Arabia, 80215 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 245 Collapse << |
| NCT01968265 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Canada, Alberta ... more >> Isis Investigative Site Red Deer, Alberta, Canada Canada, British Columbia Isis Investigative Site Penticton, British Columbia, Canada Canada, Ontario Isis Investigative Site Cornwall, Ontario, Canada Isis Investigative Site Courtice, Ontario, Canada Isis Investigative Site Sudbury, Ontario, Canada Canada, Quebec Isis Investigative Site Montreal, Quebec, Canada, H2R1V6 Romania Isis Investigative Site Cluj Napoca, Cluj, Romania, 400349 Isis Investigative Site Bucharest, Romania, 010507 Isis Investigative Site Bucharest, Romania, 011794 Isis Investigative site Bucharest, Romania, 022441 South Africa Isis Investigative Site Bloemfontein, Free State, South Africa, 9300 Isis Investigative Site Benoni, Gauteng, South Africa, 1501 Isis Investigative Site Soweto, Gauteng, South Africa, 1818 Isis Investigative Site Somerset West, Western Cape, South Africa, 7130 Collapse << |
| NCT00619697 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT00933309 | Breast Cancer | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00881530 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT03503942 | Pre-diabetes | Not Applicable | Recruiting | December 31, 2022 | Singapore ... more >> Singapore General Hospital Recruiting Singapore, Singapore, 169608 Contact: Yong Mong Bee 6321 3753 ext +65 bee.yong.mong@singhealth.com.sg Collapse << |
| NCT00313001 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Novo Nordisk Investigational Site Plainsboro, New Jersey, United States, 08536 Collapse << |
| NCT00881530 | - | - | Completed | - | - |
| NCT01725126 | Obesity | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Kansas GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT02613897 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Texas ... more >> The University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT01918865 | Type 2 Diabetes Mellitus ... more >> Obese Collapse << | Phase 2 | Completed | - | Argentina ... more >> Isis Investigative Site Mar del Plata, Buenos Aires, Argentina, B7600FZN Isis Investigative Site Rosario, Santa Fe, Argentina, S2000CVD Canada, Alberta Isis Investigational Site Edmonton, Alberta, Canada, T6G 2E1 Canada, British Columbia Isis Investigational Site Kelowna, British Columbia, Canada, V1Y 3G8 Isis Investigational Site Vancouver, British Columbia, Canada, V6J 1S3 Canada, Ontario Isis Investigational Site Etobicoke, Ontario, Canada, M9R 4E1 Isis Investigational Site Toronto, Ontario, Canada, M4G 3E8 Isis Investigative Site Toronto, Ontario, Canada, M9V 4B4 Canada, Saskatchewan Isis Investigational Site Saskatoon, Saskatchewan, Canada, S7K 3H3 South Africa Isis Investigational Site Centurion, Gauteng, South Africa, 0154 Isis Investigational Site Mamelodi, Gauteng, South Africa, 0122 Isis Investigational Site Pretoria, Gauteng, South Africa, 0001 Isis Investigational Site Port Elizabeth, Korsten, South Africa Isis Investigational Site Durban, KwaZulu-Natal, South Africa Isis Investigational Site Middelburg, Mpumalanga, South Africa Isis Investigational Site Limpopo, Thabazimbi, South Africa, 0380 Isis Investigational Site Cape Town, Western Cape, South Africa, 7570 Isis Investigational Site Cape Town, Western Cape, South Africa Isis Investigational Site Benoni, South Africa, 1500 Isis Investigational Site Bloemfontein, South Africa, 9301 Isis Investigational Site Pretoria, South Africa, 0184 Collapse << |
| NCT01464437 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02683226 | Polycystic Ovary Syndrome ... more >> Insulin Resistance Collapse << | Phase 4 | Completed | - | Slovenia ... more >> University Medical Center Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT01648582 | - | - | Completed | - | - |
| NCT00111631 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT00396071 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Diabetes Zentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Switzerland Novartis Pharmaceuticals Basel, Switzerland Collapse << |
| NCT01804842 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | - |
| NCT00909480 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01804842 | - | - | Completed | - | - |
| NCT02648854 | Diabetes Mellitus, Type II | Phase 1 | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Deokjin-gu, Jeonju-si, Jeollabuk-do, Korea, Republic of, 561-712 Collapse << |
| NCT00909480 | - | - | Completed | - | - |
| NCT03527537 | Gestational Diabetes | Phase 4 | Not yet recruiting | June 1, 2028 | United States, Wisconsin ... more >> Medical College of Wisconsin Not yet recruiting Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00328172 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01819272 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00328172 | - | - | Completed | - | - |
| NCT01131182 | - | - | Completed | - | - |
| NCT01819272 | - | - | Completed | - | - |
| NCT01388361 | - | - | Completed | - | - |
| NCT01131182 | Type 2 Diabetes Mellitus (T2DM... more >>) Collapse << | Phase 4 | Completed | - | - |
| NCT00378729 | Polycystic Ovary Syndrome | Phase 4 | Unknown | October 2011 | France ... more >> Hopital Antoine Beclere Recruiting Clamart, France, 92170 Contact: Hervé FERNANDES, MD,PhD +33(0)- 1 45 37 44 69 herve.fernandez@abc.aphp.fr Principal Investigator: Hervé FERNANDEZ, MD Collapse << |
| NCT01725126 | - | - | Completed | - | - |
| NCT00972283 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00972283 | - | - | Completed | - | - |
| NCT02737657 | - | - | Completed | - | Kuwait ... more >> Kuwait City, Kuwait Lebanon Amioun, Lebanon Beirut, Lebanon Nabatieh, Lebanon Saida, Lebanon Taalabya, Lebanon Tripoli, Lebanon United Arab Emirates Al Ain, United Arab Emirates Dubai, United Arab Emirates Collapse << |
| NCT01947790 | LVM Type 2 Di... more >>abetic Patients With IHD Endothelial Function Collapse << | Phase 4 | Completed | - | China, Hubei ... more >> Wuhan City, Hubei, China, 43000 Xiang Guangda Wuhan, Hubei, China, 430070 Collapse << |
| NCT01388361 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00434954 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Germany ... more >> Research Site Bad Mergentheim, Germany Research Site Berlin, Germany Research Site Bosenheim, Germany Research Site Burghausen, Germany Research Site Datteln, Germany Research Site Dresden, Germany Research Site Essen, Germany Research Site Friedrichsthal, Germany Research Site Hildesheim, Germany Research Site Hirschhorn, Germany Research Site Hohenmolsen, Germany Research Site Jena, Germany Research Site Lehrte, Germany Research Site Leipzig, Germany Research Site Ludwigsburg, Germany Research Site Mannheim, Germany Research Site Marburg, Germany Research Site Marktheidenfeld, Germany Research Site Meissen, Germany Research Site Munchen, Germany Research Site Offenbach, Germany Research Site Oschatz, Germany Research Site Pohlheim, Germany Research Site Regensburg, Germany Research Site Riesa, Germany Research Site Rodgau, Germany Research Site Roding, Germany Research Site Rosenheim, Germany Research Site Schluchtern, Germany Research Site Schwedt/Oder, Germany Research Site Sinsheim, Germany Research Site Speyer, Germany Research Site Unterhaching, Germany Research Site Volklingen, Germany Research Site Wallerfing, Germany Research Site Wangen, Germany Research Site Warburg, Germany Research Site Wiesbaden, Germany Research Site Wolfsburg, Germany Collapse << |
| NCT00434954 | - | - | Completed | - | - |
| NCT03222765 | PreDiabetes | Not Applicable | Recruiting | December 31, 2019 | Spain ... more >> Centro de Salud Jose Marva Recruiting Madrid, Spain, 28030 Contact: Esther Sanchez, MD +34629 Contact: Ana M Santos Collapse << |
| NCT00851903 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Vienna, Austria Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Colombia Sanofi-Aventis Administrative Office Bogota, Colombia Egypt Sanofi-Aventis Administrative Office Cairo, Egypt Greece Sanofi-Aventis Administrative Office Kallithea, Greece Hong Kong Sanofi-Aventis Administrative Office Hong Kong, Hong Kong India Sanofi-Aventis Administrative Office Mumbai, India Israel Sanofi-Aventis Administrative Office Natanya, Israel Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Lebanon Sanofi-Aventis Administrative Office Beirut, Lebanon Mexico Sanofi-Aventis Administrative Office Col. Coyoacan, Mexico Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Portugal Sanofi-Aventis Administrative Office Porto Salvo, Portugal Spain Sanofi-Aventis Administrative Office Barcelona, Spain United Kingdom Sanofi-Aventis Administrative Office Guildford Surrey, United Kingdom Collapse << |
| NCT00851903 | - | - | Completed | - | - |
| NCT00637273 | - | - | Completed | - | - |
| NCT00532935 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03175120 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | April 8, 2019 | - |
| NCT00532935 | - | - | Completed | - | - |
| NCT01215331 | Gestational Diabetes Mellitus | Phase 3 | Completed | - | Canada, Quebec ... more >> Centre de recherche clinique du CHUS Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT02503943 | Type 2 Diabetes ... more >> Overweight Obesity Masked Hypertension Collapse << | Phase 4 | Unknown | September 2016 | - |
| NCT00637273 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00494884 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Germany ... more >> Novartis Investigative Site Bochum, Germany Collapse << |
| NCT03030300 | Type2 Diabetes | Phase 4 | Completed | - | China, Shandong ... more >> Shandong Provincial Hospital Jinan, Shandong, China, 250001 Qianfoshan Hospital Jinan, Shandong, China, 250012 Qilu Hospital Jinan, Shandong, China, 250014 Collapse << |
| NCT03700801 | Type 2 Diabetes Mellitus | Phase 4 | Not yet recruiting | March 2019 | - |
| NCT02911948 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Japan ... more >> Novo Nordisk Investigational Site Asahikawa-shi, Hokkaido, Japan, 078-8211 Novo Nordisk Investigational Site Bunkyo-ku, Tokyo, Japan, 113 8431 Novo Nordisk Investigational Site Chigasaki-shi, Kanagawa, Japan, 253-0044 Novo Nordisk Investigational Site Chitose, Hokkaido, Japan, 066-0032 Novo Nordisk Investigational Site Chiyoda-ku, Tokyo, Japan, 101-0024 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0027 Novo Nordisk Investigational Site Fujisawa-shi, Kanagawa, Japan, 251-0041 Novo Nordisk Investigational Site Fukui-shi, Fukui, Japan, 918-8503 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 810-0001 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 819-0006 Novo Nordisk Investigational Site Fukuoka, Japan, 830 8522 Novo Nordisk Investigational Site Kashiwara-shi, Osaka, Japan, 582 0005 Novo Nordisk Investigational Site Kawagoe-shi, Saitama, Japan, 350 0851 Novo Nordisk Investigational Site Kawaguchi-shi, Saitama, Japan, 332-0012 Novo Nordisk Investigational Site Koriyama-shi, Fukushima, Japan, 963 8851 Novo Nordisk Investigational Site Kumamoto-shi, Kumamoto, Japan, 862-0965 Novo Nordisk Investigational Site Kumamoto-shi,Kumamoto, Japan, 862 0976 Novo Nordisk Investigational Site Matsumoto-shi, Nagano, Japan, 390-8621 Novo Nordisk Investigational Site Miyazaki-shi, Japan, 880 0034 Novo Nordisk Investigational Site Naka-shi, Ibaraki, Japan, 311 0113 Novo Nordisk Investigational Site Nakagami, Okinawa, Japan, 901-2393 Novo Nordisk Investigational Site Neyagawa-shi, Osaka, Japan Novo Nordisk Investigational Site Niigata-shi, Niigata, Japan, 950 1104 Novo Nordisk Investigational Site Okawa-shi, Fukuoka, Japan, 831 0016 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 536-0001 Novo Nordisk Investigational Site Ota-ku, Tokyo, Japan, 1430015 Novo Nordisk Investigational Site Oyama-shi, Tochigi, Japan, 323 0022 Novo Nordisk Investigational Site Saitama-shi, Saitama, Japan, 336-0963 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060 0062 Novo Nordisk Investigational Site Sendai-shi, Japan, 980 0021 Novo Nordisk Investigational Site Shimotsuke-shi, Tochigi, Japan, 329 0433 Novo Nordisk Investigational Site Takatsuki-shi, Osaka, Japan, 569 1096 Novo Nordisk Investigational Site Tokyo, Japan, 103-0028 Novo Nordisk Investigational Site Tokyo, Japan, 181-0013 Novo Nordisk Investigational Site Tomigusuku-shi, Okinawa, Japan, 901-0243 Novo Nordisk Investigational Site Tomigusuku-shi, Okinawa, Japan, 901-0244 Novo Nordisk Investigational Site Yamaguchi-shi, Yamaguchi, Japan, 754-0002 Novo Nordisk Investigational Site Yokohama-shi, Japan, 235 0045 Collapse << |
| NCT00494663 | Type 2 Diabetes | Phase 2 | Terminated(Program Terminated) | December 2008 | - |
| NCT00605475 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Arkansas ... more >> Arkansas Research Medical Testing Little Rock, Arkansas, United States, 72202 United States, Florida Allied Research International - Cetero Research Miami Miami, Florida, United States, 33169 Elite Research Institute Miami Miami, Florida, United States, 33169 United States, Maryland International Research Center Towson MD, Maryland, United States, 21286 United States, Oregon Covance Clinical Research Unit Inc Portland, Oregon, United States, 97239 United States, Washington Charles River Clinical Services NW Tacoma, Washington, United States, 98418 Germany Novartis Investigator Site Berlin, Germany Novartis Investigator Site Kiel, Germany Novartis Investigator Site Moenchengladbach, Germany Novartis Investigator Site Munich, Germany Novartis Investigative Site Neuss, Germany Russian Federation Novartis Investigative Site Moscow, Russian Federation Novartis Investigative Site St. Petersberg, Russian Federation Collapse << |
| NCT01618162 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01425580 | Congestive Heart Failure ... more >> Type 2 Diabetes Mellitus Collapse << | Phase 2 | Completed | - | Sweden ... more >> Karolinska Institutet, Department of Clinical Science and Education, Södersjukhuset Stockholm, Sweden, 118 83 Karolinska Institutet, Division of Internal Medicine Södersjukhuset AB Stockholm, Sweden, 118 83 Collapse << |
| NCT01618162 | - | - | Completed | - | - |
| NCT02014740 | Type 2 Diabetes ... more >> Overweight Obesity Collapse << | Phase 4 | Completed | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT01589653 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Egypt ... more >> Novo Nordisk Investigational Site Alexandria, Egypt, 21131 Novo Nordisk Investigational Site Beni-Suef, Egypt, 62511 Novo Nordisk Investigational Site Cairo, Egypt, 11562 Indonesia Novo Nordisk Investigational Site Bandung, Indonesia, 40161 Novo Nordisk Investigational Site Malang, Indonesia, 65111 Morocco Novo Nordisk Investigational Site Casablanca, Morocco, 20100 Novo Nordisk Investigational Site Marrakech, Morocco, 40000 Saudi Arabia Novo Nordisk Investigational Site Jeddah, Saudi Arabia, 80215 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 11211 Novo Nordisk Investigational Site Riyadh, Saudi Arabia, 11426 Novo Nordisk Investigational Site Taif, Saudi Arabia, 21944 Tunisia Novo Nordisk Investigational Site Tunis, Tunisia, 1007 Novo Nordisk Investigational Site Tunis, Tunisia, 1008 Vietnam Novo Nordisk Investigational Site Ha Noi City, Vietnam Novo Nordisk Investigational Site Ho Chi Minh City, Vietnam Collapse << |
| NCT02231021 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | November 2018 | Korea, Republic of ... more >> Seoul St Mary's Hospital, The Catholic University of Korea Recruiting Seoul, Korea, Republic of, 137-701 Principal Investigator: Kun-Ho Yoon, MD, PhD Sub-Investigator: Yoon-Hee Choi, MD, PhD Sub-Investigator: Hye-Kyung Yang, MD Collapse << |
| NCT00605475 | - | - | Completed | - | - |
| NCT02315287 | Type 2 Diabetes | Phase 4 | Recruiting | December 2018 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Gyeonggi, Korea, Republic of, 463-707 Contact: Soo Lim, MD, PHD 82-31-787-7035 limsoo@snu.ac.kr Contact: Eu Jeong Ku, MD 82-31-787-6232 eujeong.ku@gmail.com Principal Investigator: Soo Lim, MD, PHD Collapse << |
| NCT02014740 | - | - | Completed | - | - |
| NCT01589653 | - | - | Completed | - | - |
| NCT00694759 | Polycystic Ovary Syndrome | Not Applicable | Unknown | July 2011 | United States, Oregon ... more >> Oregon Health & Science University Recruiting Portland, Oregon, United States, 97239 Principal Investigator: Bethany J. Klopfenstein, MD Collapse << |
| NCT00362440 | HIV Lipodystrophy | Phase 2 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01370707 | Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Bucheon St. Mary's Hospital Bucheon, Korea, Republic of Seoul National University Bundang Hospital Bundang, Korea, Republic of Chonbuk National University Hospital Chonju, Korea, Republic of Kyungpook University Hospital Daegu, Korea, Republic of Yeungnam University Medical Center Daegu, Korea, Republic of National Health Insurance Coporation Ilsan Hospital Ilsan, Korea, Republic of Inha University Hospital Incheon, Korea, Republic of Inje University Pusan Baik Hospital Pusan, Korea, Republic of Pusan National University Hospital Pusan, Korea, Republic of Asan Medical Center Seoul, Korea, Republic of Eulji Medical Center Seoul, Korea, Republic of Hallym University Medical Center(Gangdong) Seoul, Korea, Republic of Hallym University Medical Center(Gangnam) Seoul, Korea, Republic of Kangbuk Samsung Hospital Seoul, Korea, Republic of Korea University Guro Hospital Seoul, Korea, Republic of Kyung Hee University Hospital Seoul, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of Seoul St. Mary's hospital Seoul, Korea, Republic of Severance Hospital Seoul, Korea, Republic of Ajou University Hospital Suwon, Korea, Republic of Uijeongbu St. Mary's Hospital Uijeongbu, Korea, Republic of Collapse << |
| NCT02501850 | Diabetes Mellitus Type 2 | Phase 4 | Unknown | October 2015 | Spain ... more >> Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Contact: Andreea Ciudin, PhD +34932744736 aciudin@vhebron.net Contact: Cristina Hernandez, MD, PhD +34 932746591 cristina.hernandez@vhir.org Principal Investigator: Cristina Hernandez, MD, PhD Sub-Investigator: Andreea Ciudin, MD Sub-Investigator: Olga Simó-Servat, MD Collapse << |
| NCT00437554 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Korea, Republic of ... more >> Handok Seoul, Korea, Republic of Collapse << |
| NCT02502344 | Insulin Resistance | Phase 4 | Unknown | - | China, Beijing ... more >> BEI JING Tsinghua Changguang Hospital Recruiting Beijing, Beijing, China, 102218 Contact: jian Z xiao, doctor 0086-010-56118899 ext 13683685711 xiaojzh858@sina.com Collapse << |
| NCT00362440 | - | - | Completed | - | - |
| NCT00184574 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01232491 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | - |
| NCT01771185 | Type 2 Diabetes | Not Applicable | Unknown | December 2013 | - |
| NCT00081328 | Diabetes Mellitus, Type II | Phase 3 | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Colorado University of Colorado Health Sciences Center, The Children's Hospital Denver, Colorado, United States, 80262 United States, Connecticut Yale University New Haven, Connecticut, United States, 06520 United States, Maryland George Washington University Biostatistics Center Rockville, Maryland, United States, 20852 United States, Massachusetts Massachusetts General Hospital Diabetes Center Boston, Massachusetts, United States, 02114 Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Saint Louis University Health Sciences Center St Louis, Missouri, United States, 63104 Washington University Department of Pediatrics St. Louis, Missouri, United States, 63110 United States, New York Columbia University Medical Center New York City, New York, United States, 10032 State University of New York Upstate Medical University Syracuse, New York, United States, 13210 United States, Ohio Case Western Reserve Cleveland, Ohio, United States, 44106 United States, Oklahoma University of Oklahoma Oklahoma City, Oklahoma, United States, 93104 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00081328 | - | - | Completed | - | - |
| NCT00068861 | Polycystic Ovary Syndrome ... more >> Infertility Pregnancy Collapse << | Phase 3 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35249 United States, California University of California at San Diego La Jolla, California, United States, 92037 Stanford University Stanford, California, United States, 94305 United States, Colorado University of Colorado Aurora, Colorado, United States, 80045 United States, Michigan Wayne State Detroit, Michigan, United States, 48201 United States, New Jersey University of Medicine and Dentistry New Jersey Newark, New Jersey, United States, 07103 United States, Pennsylvania Penn State Hershey, Pennsylvania, United States, 17033 University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Baylor College of Medicine Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT01624259 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01232491 | - | - | Completed | - | - |
| NCT02623998 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | December 2018 | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2T 5C7 Canada, Manitoba Health Science Centre Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario McMaster University Hamilton, Ontario, Canada St. Joseph's Hospital London, Ontario, Canada, N6A 4V2 LMC Markham, Ontario, Canada Canada, Quebec Univeristy of Montreal Montreal, Quebec, Canada Collapse << |
| NCT03000218 | Overweight Ab... more >>normal Liver Function Tests Collapse << | Phase 1 | Unknown | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seoul, Korea, Republic of Contact: Jae-Yong Chung, MD, PhD +82-031-787-3955 Principal Investigator: Jae-Yong Chung, MD, PhD Collapse << |
| NCT00951899 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01215968 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United Kingdom ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Glasgow, United Kingdom Collapse << |
| NCT01624259 | - | - | Completed | - | - |
| NCT01215968 | - | - | Completed | - | - |
| NCT01404676 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Division of Endocrinology and Metabolism, Department of Internal Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01307306 | Burns | Not Applicable | Recruiting | September 2019 | Canada, Ontario ... more >> Ross Tilley Burn Centre, Sunnybrook Health Sciences Centre Recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Marc G Jeschke, MD PhD 416-480-6703 marc.jeschke@sunnybrook.ca Contact: Marjorie Burnett, HonsBSc 416-480-6100 ext 88021 marjorie.burnett@sunnybrook.ca Sub-Investigator: Robert Cartotto, MD Sub-Investigator: Shahriar Shahrokhi, MD Sub-Investigator: Alison Cheung, PhD Collapse << |
| NCT00951899 | - | - | Completed | - | - |
| NCT01786707 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Diabetes Research Institute, University of Miami Miller School of Medicine Miami, Florida, United States, 33136 Collapse << |
| NCT01786707 | - | - | Completed | - | - |
| NCT00443755 | Type 2 Diabetes ... more >> Insulin Resistance Metabolic Syndrome Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00506298 | Type 2 Diabetes | Phase 2 | Completed | - | Canada, British Columbia ... more >> Chilliwack, British Columbia, Canada Vancouver, British Columbia, Canada Canada, Manitoba Winnipeg, Manitoba, Canada Canada, Newfoundland and Labrador Bay Roberts, Newfoundland and Labrador, Canada Holyroad, Newfoundland and Labrador, Canada St. Johns, Newfoundland and Labrador, Canada Canada, Ontario Aylmer, Ontario, Canada Burlington, Ontario, Canada Collingwood, Ontario, Canada Corunna, Ontario, Canada Robarts Research Institute London, Ontario, Canada London, Ontario, Canada Newmarket, Ontario, Canada Sarnia, Ontario, Canada Sudbury, Ontario, Canada Toronto, Ontario, Canada Canada, Quebec Montreal, Quebec, Canada Canada, Saskatchewan Saskatoon, Saskatchewan, Canada Collapse << |
| NCT01958671 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03738449 | Type 2 Diabetes Mellitus | Phase 1 | Not yet recruiting | December 26, 2018 | Korea, Republic of ... more >> Severance Hospital Not yet recruiting Seoul, Korea, Republic of, 03722 Contact: Min Soo Park, Ph.D. M.D +82-2-2228-0270 minspark@yuhs.ac Principal Investigator: Minsoo Park, Ph.D. M.D Collapse << |
| NCT01332370 | - | - | Completed | - | - |
| NCT02088034 | Diabetes | Phase 4 | Unknown | February 2016 | United States, Pennsylvania ... more >> Temple University - Center for Obesity Research and Education Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT00518115 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00443755 | - | - | Completed | - | - |
| NCT01958671 | - | - | Completed | - | - |
| NCT03751657 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | November 7, 2019 | - |
| NCT01076075 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00510705 | Severe Coronary Artery Disease... more >> Obesity Impaired Glucose Tolerance Collapse << | Not Applicable | Terminated | - | Germany ... more >> University of Leipzig - Heart Center Leipzig, Germany, 04289 Collapse << |
| NCT01624116 | Diabetes, Type 2 | Not Applicable | Completed | - | Pakistan ... more >> Endocrinology Unit & Diabetes Management Centre, Services Hospital. Lahore, Pakistan Collapse << |
| NCT01090752 | - | - | Completed | - | - |
| NCT01090752 | Diabetes Hype... more >>rtension Collapse << | Phase 4 | Completed | - | Switzerland ... more >> Pechère-Bertschi Antoinette Geneva, Switzerland, 1211 Collapse << |
| NCT00751114 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01111955 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> Farid Marquez, Md Hialeah, Florida, United States, 33012 United States, Washington Capital Clinical Reserch Center Olympia, Washington, United States, 98502 United States, Wisconsin Aurora Advanced Healthcare Milwaukee, Wisconsin, United States, 53209 Australia, New South Wales Local Institution Blacktown, New South Wales, Australia, 2148 Australia, Victoria Local Institution Box Hill, Victoria, Australia, 3128 Australia, Western Australia Local Institution Freemantle, Western Australia, Australia, 6959 Canada, British Columbia Local Institution Surrey, British Columbia, Canada, V4A 2H9 Canada, Manitoba Local Institution Winnipeg, Manitoba, Canada, R3E 3P4 Canada, New Brunswick Local Institution Bathurst, New Brunswick, Canada, E2A 4X7 Canada, Newfoundland and Labrador Local Institution St. John'S, Newfoundland and Labrador, Canada, A1A 3R5 Canada, Ontario Local Institution Thornhill, Ontario, Canada, L4J 8L7 Canada, Quebec Local Institution Mirabel, Quebec, Canada, J7J 2K8 Canada, Saskatchewan Local Institution Saskatoon, Saskatchewan, Canada, S7K 7H9 Canada Local Institution Quebec, Canada, G1N 4V3 Collapse << |
| NCT00984867 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00518115 | - | - | Completed | - | - |
| NCT00715624 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Brazil Sanofi-Aventis Administrative Office Sao Paulo, Brazil Canada Sanofi-Aventis Administrative Office Laval, Canada Chile Sanofi-Aventis Administrative Office Santiago, Chile Egypt Sanofi-Aventis Administrative Office Cairo, Egypt France Sanofi-Aventis Administrative Office Paris, France Germany Sanofi-Aventis Administrative Office Berlin, Germany India Sanofi-Aventis Administrative Office Mumbai, India Italy Sanofi-Aventis Administrative Office Milano, Italy Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Mexico Sanofi-Aventis Administrative Office Mexico, Mexico Puerto Rico Sanofi-Aventis Administrative Office Puerto Rico, Puerto Rico Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey United Kingdom Sanofi-Aventis Administrative Office Guildford Surrey, United Kingdom Collapse << |
| NCT00751114 | - | - | Completed | - | - |
| NCT00715624 | - | - | Completed | - | - |
| NCT01076075 | - | - | Completed | - | - |
| NCT01751321 | Type 2 Diabetes | Not Applicable | Unknown | October 2013 | - |
| NCT02384109 | Prediabetic State | Not Applicable | Active, not recruiting | June 2019 | United States, California ... more >> Toluca Lake Health Center Burbank, California, United States, 91505 100 Medical Plaza Primary Care Suites 455 & 490 Los Angeles, California, United States, 90024 Internal Medicine Women's Health Clinic Suites 250 & 290 Los Angeles, California, United States, 90024 UCLA Internal Medicine Family Westwood Suite 465 Los Angeles, California, United States, 90024 UCLA Internal Medicine Geriatrics Suite 365 & 420 Los Angeles, California, United States, 90024 CPN Brentwood Los Angeles, California, United States, 90049 CPN West Washington Internal Medicine Los Angeles, California, United States, 90066 Pacific Palisades Pacific Palisades, California, United States, 90272 12th Street Clinic Santa Monica, California, United States, 90401 Wilshire Office Santa Monica, California, United States, 90401 CPN Santa Monica 15th Street Family Medicine Practice Santa Monica, California, United States, 90404 CPN/Santa Monica Parkside Santa Monica, California, United States, 90404 Santa Moica Bay Physicians/20th Street 10th Floor Santa Monica, California, United States, 90404 Santa Monica Bay Physicians Plaza Santa Monica, California, United States, 90404 Santa Monica Bay Physicians/20th Street 3rd Floor Santa Monica, California, United States, 90404 Santa Monica Internal Medicine Santa Monica, California, United States, 90404 UCLA Family Health Clinic Santa Monica, California, United States, 90404 Santa Moica Bay Physicians Ocean Park Santa Monica, California, United States, 90405 Jack H. Skirball Health Center Woodland Hills, California, United States, 91364 CPN Woodland Hills Practice Woodland Hills, California, United States, 91367 Collapse << |
| NCT00714233 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | - |
| NCT00510588 | Stable Coronary Artery Disease... more >> Obesity Impaired Glucose Tolerance Collapse << | Not Applicable | Completed | - | Germany ... more >> University of Leipzig - heart center Leipzig, Germany, 04289 Collapse << |
| NCT00082407 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00984867 | - | - | Completed | - | - |
| NCT01452451 | Diabetes Mellitus | Phase 2 | Completed | - | United States, Ohio ... more >> Ohio, Ohio, United States Collapse << |
| NCT00219440 | Diabetes Mellitus | Phase 4 | Completed | - | United States, Louisiana ... more >> Pennington Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT00082407 | - | - | Completed | - | - |
| NCT02964247 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02803918 | Type 2 Diabetes Mellitus | Phase 1 | Recruiting | January 27, 2020 | United States, Louisiana ... more >> Investigational Site Number 840002 Recruiting Baton Rouge, Louisiana, United States, 70808 Investigational Site Number 840006 Recruiting New Orleans, Louisiana, United States, 70115-6969 United States, Pennsylvania Investigational Site Number 840009 Recruiting Philadelphia, Pennsylvania, United States, 19104 United States, Texas Investigational Site Number 840007 Recruiting Lufkin, Texas, United States, 75904 Mauritius Investigational Site Number 480001 Recruiting Phoenix, Mauritius Mexico Investigational Site Number 484001 Recruiting Monterrey, Mexico, 64460 Investigational Site Number 484002 Recruiting Puebla, Mexico, 72190 South Africa Investigational Site Number 710001 Recruiting Cape Town, South Africa, 7530 Spain Investigational Site Number 724002 Recruiting Barcelona, Spain, 08009 Investigational Site Number 724001 Recruiting Vitoria, Spain, 01009 Collapse << |
| NCT00714233 | - | - | Completed | - | - |
| NCT00331851 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01128153 | - | - | Completed | - | - |
| NCT01128153 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT02446834 | Polycystic Ovary Syndrome | Not Applicable | Unknown | August 2017 | China, Shanghai ... more >> Renji Hospital Department of Endocrinology and Metabolism Recruiting Shanghai, Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Contact: Jing Ma, MD +86-15800983436 cherry1996@live.cn Collapse << |
| NCT00371306 | Gestational Diabetes ... more >> Type 2 Diabetes Pregnancy Collapse << | Not Applicable | Unknown | September 2008 | United States, Tennessee ... more >> Regional Obstetrical Consultants Recruiting Chattanooga, Tennessee, United States, 37403 Collapse << |
| NCT00598793 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03196154 | - | - | Completed | - | Malaysia ... more >> Clinical Research Centre, Sarawak General Hospital Kuching, Sarawak, Malaysia, 93586 Collapse << |
| NCT02763007 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | September 2020 | Korea, Republic of ... more >> Seoul St Mary's Hospital, The Catholic University of Korea Recruiting Seoul, Korea, Republic of, 137-701 Contact: YOON HEE CHOI, MD Principal Investigator: Kun-Ho Yoon, MD, PhD Sub-Investigator: Yoon-Hee Choi, MD, PhD Sub-Investigator: Eun-Young Lee, MD, PhD Collapse << |
| NCT01765894 | - | - | Completed | - | Denmark ... more >> XLab, Center for Healthy Aging, Department of Biomedical Sciences, University of Copenhagen Copenhagen, Denmark, 2200 Collapse << |
| NCT00082381 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00082381 | - | - | Completed | - | - |
| NCT00838903 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00849056 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00647023 | Polycystic Ovary Syndrome | Not Applicable | Terminated | - | - |
| NCT01775176 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | United States, Louisiana ... more >> Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Collapse << |
| NCT02919345 | Diabetes Mellitus, Type 2 ... more >> Coronary Artery Disease Carotid Artery Diseases Collapse << | Phase 4 | Unknown | October 2017 | Brazil ... more >> State University of Campinas Campinas, Sao Paulo, Brazil, 13083-887 Collapse << |
| NCT00838903 | - | - | Completed | - | - |
| NCT02365233 | Type II Diabetes ... more >> Nonalcoholic Fatty Liver Collapse << | Phase 4 | Terminated(IRB withheld the da... more >>ta due to inadequate supporting documentation) Collapse << | - | United States, Texas ... more >> University of Texas Medical Branch -Galveston Galveston, Texas, United States, 77555 Collapse << |
| NCT00427154 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT00849056 | - | - | Completed | - | - |
| NCT03364335 | Obesity | Phase 2 | Completed | - | United States, Alabama ... more >> AMR Mobile, Alabama, United States, 36608 United States, California Catalina Research Institute Montclair, California, United States, 91763 Northern California Research Sacramento, California, United States, 95821 United States, Idaho ACR Meridian, Idaho, United States, 83642 United States, Illinois Synexus Chicago, Illinois, United States, 60602 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 Clinical Trials Management Metairie, Louisiana, United States, 70006 United States, Missouri Synexus Saint Louis, Missouri, United States, 63141 United States, Nevada AMR Las Vegas, Nevada, United States, 89119 United States, Ohio Rapid Medical Research Cleveland, Ohio, United States, 44122 United States, South Carolina Medical Research South Charleston, South Carolina, United States, 29407 United States, Tennessee Premier Clarksville, Tennessee, United States, 37040 United States, Texas Synexus Dallas, Texas, United States, 75234 Synexus San Antonio, Texas, United States, 78229 United States, Utah ACR Jordan, Utah, United States, 84088 Collapse << |
| NCT00806234 | Psychotic Disorders | Phase 4 | Completed | - | United States, Maryland ... more >> University of Maryland Baltimore, Maryland, United States, 21201 Johns Hopkins Hospital Baltimore, Maryland, United States, 21205 United States, New York The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 United States, North Carolina University of North Carolina, Division of Child and Adolescent Psychiatry Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01089790 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT01258075 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | June 2019 | - |
| NCT00806234 | - | - | Completed | - | - |
| NCT01717313 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03364335 | - | - | Completed | - | - |
| NCT01302379 | Breast Neoplasms | Not Applicable | Unknown | December 2016 | United States, California ... more >> Moores UCSD Cancer Center La Jolla, California, United States, 92093 Collapse << |
| NCT02099110 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02365233 | - | - | Terminated(IRB withheld the da... more >>ta due to inadequate supporting documentation) Collapse << | - | - |
| NCT00970528 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of ... more >> Wonju-si, Gang''weondo, Korea, Republic of, 220-701 Busan, Korea, Korea, Republic of, 602-714 Gyeonggi-do, Korea, Korea, Republic of, 463-500 Gyeonggi-do, Korea, Korea, Republic of, 471-701 Pusan, Korea, Korea, Republic of, 602-739 Seoul, Korea, Korea, Republic of, 150-713 Daegu, Korea, Republic of, 705-717 Jeonju-si, Korea, Republic of, 456-712 Seongbuk-gu Seoul, Korea, Republic of, 136-705 Seoul, Korea, Republic of, 134-701 Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01717313 | - | - | Completed | - | - |
| NCT01794143 | Type 2 Diabetes ... more >> Comparative Effectiveness of Glycemia-lowering Medications Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01792830 | - | - | Completed | - | - |
| NCT01792830 | Poor Glycemic Control | Phase 3 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory Midtown Hospital Atlanta, Georgia, United States, 30308 Emory University Hospital Atlanta, Georgia, United States, 30326 Collapse << |
| NCT02099110 | - | - | Completed | - | - |
| NCT01526317 | Pharmacokinetics and Drug Inte... more >>raction of Crestor and Glucodown SR Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Severance Hospital, Yonsei University Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT03421119 | Diabetes Mellitus, Type 2 | Phase 3 | Not yet recruiting | December 1, 2019 | - |
| NCT02920801 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Shanghai ... more >> Fang Li Shanghai, Shanghai, China, 200032 Collapse << |
| NCT02787551 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | November 20, 2018 | - |
| NCT00366301 | Type 2 Diabetes | Phase 4 | Terminated(Interim analyses de... more >>monstrated futility. Thus, recruitment curtailed 10/08.) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Collapse << |
| NCT02058160 | - | - | Completed | - | - |
| NCT02128932 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00099320 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02128932 | - | - | Completed | - | - |
| NCT00366301 | - | - | Terminated(Interim analyses de... more >>monstrated futility. Thus, recruitment curtailed 10/08.) Collapse << | - | - |
| NCT03207698 | Furcation Defects | Phase 2 Phase 3 | Completed | - | India ... more >> Government Dental College and Research Institute Bangalore, Karnataka, India, 560002 Collapse << |
| NCT02058160 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01485614 | Diabetes Mellitus ... more >> Type 2 Diabetes Collapse << | Phase 3 | Active, not recruiting | October 17, 2019 | - |
| NCT00948324 | Type 2 Diabetes Mellitus | Phase 4 | Suspended | December 2021 | - |
| NCT00834652 | Depression | Phase 4 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00834652 | - | - | Completed | - | - |
| NCT03230786 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Czechia ... more >> Vdoviak Miroslav MUDr. - Interní Ambulance Karlovy Vary, Czechia, 360 17 General University Hospital Prague 2, Czechia, 128 00 Institute for Clinical and experimental Medicine Praha 4, Czechia, 140 21 Interní a Diabetologická ambulance, Clintrial s.r.o Praha, Czechia, 100 00 Endokrinologicky Ústav Praha, Czechia, 113 94 Diabet2 s.r.o Praha, Czechia Interní a Diabetologická ambulance Uherské Hradiště, Czechia, 68 601 Diabetologická a Obezitologická ambulance České Budějovice, Czechia Denmark Bispebjerg Hospital, Endokrinologisk Afdeling Bispebjerg, Copenhagen NV, Denmark, 2400 Bioclinica Ballerup, Copenhagen, Denmark, 2750 Bioclinica Aalborg, Jutland, Denmark, 9000 Aarhus University Hospital, Endocrinlogy Department Aarhus, Jutland, Denmark, 8000 Sydvestjysk Sygehus Esbjerg, Jutland, Denmark, 6700 Bioclinica Vejle, Jutland, Denmark, 7100 Holbæk Sygehus Holbæk, Denmark, 4300 Moldova, Republic of Rtl Sm Srl/Scr Chisinau, Moldova, Republic of, MD2025 Rtl Sm Srl Chisinau, Moldova, Republic of, MD2025 Poland Centrum Badań Klinicznych PI-House sp. z o.o. Gdańsk, Poland, 80-546 Specjalistyczna Praktyka Lekarska Piotr Kubalski Grudziądz, Poland, 86-302 Medyczne Centrum Diabetologiczno Endokrynologiczno Metaboliczno Kraków, Poland, 31-261 Prywatny Gabinet Lekarski M. Horodecki (budynek Medicus) Opole, Poland, 45-367 Centrum Medyczne Hygea Tychy, Poland, 43-100 Centrum Medyczne AMED Warsaw, Poland, 01-518 Prywatny Gabinet Lekarski i Wizyty Lekarskie Jan Ruxer Łódź, Poland, 90-074 Romania S.C. Policlinica CCBR S.R.L. Bucharest, Romania, 030463 Institutului Național de Diabet, Nutriție și Boli Metabolice "Prof.Dr. N.C. Paulescu" Bucharest, Romania S.C Nicodiab S.R.L. Bucharest, Romania S.C. Sfinx Medica S.R.L. Constanţa, Romania, 900412 S.C. Mediab S.R.L. Targu Mures, Romania S.C. Mediab S.R.L. Târgu-Mureş, Romania United Kingdom St. Pancras London, United Kingdom, WC1X 8QD Collapse << |
| NCT01357135 | - | - | Completed | - | - |
| NCT00513630 | Type 2 Diabetes ... more >> Coronary Artery Disease Collapse << | Phase 4 | Completed | - | China ... more >> Shanghai Jiao Tong University School of Medicine Shanghai, China, 200025 Collapse << |
| NCT01332071 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Brazil ... more >> GSK Investigational Site Goiania, Goiás, Brazil Collapse << |
| NCT02160743 | Food Effect Study of CJ-30056 ... more >>20mg/500mg Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inje University Busan Paik Hospital Busan, Korea, Republic of, 614-735 Collapse << |
| NCT00631033 | Obesity | Phase 2 | Completed | - | Netherlands ... more >> Rijnstate Hospital Arnhem, Netherlands Collapse << |
| NCT01357135 | - | - | Completed | - | - |
| NCT01646320 | - | - | Completed | - | - |
| NCT01332071 | - | - | Completed | - | - |
| NCT00482729 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00482729 | - | - | Completed | - | - |
| NCT00641056 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00641056 | - | - | Completed | - | - |
| NCT01646320 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03593135 | Randomized Controlled Trial ... more >> Diabetes Mellitus Collapse << | Not Applicable | Completed | - | Pakistan ... more >> Shaikh Zayed Medical Complex Lahore, Punjab, Pakistan, 54000 Collapse << |
| NCT00232583 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, Texas ... more >> University of Texas Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT03283657 | PreDiabetes D... more >>iabetes Mellitus Diabetes Mellitus, Type 2 Body Weight Body Weight Changes Collapse << | Not Applicable | Enrolling by invitation | March 31, 2019 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00483392 | Insulin Resistance | Not Applicable | Completed | - | Slovenia ... more >> Department of Endocrinology, Diabetes and Metabolic diseases Ljubljana, Slovenia, 1525 Collapse << |
| NCT01064687 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00232583 | - | - | Completed | - | - |
| NCT00562172 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> Sanofi-aventis Seoul, Korea, Republic of Collapse << |
| NCT01238978 | Type 2 Diabetes Mellitus ... more >> Elderly Collapse << | Phase 4 | Completed | - | France ... more >> Novartis Investigative Site Armentieres, France, 59208 Novartis Investigative Site Brest, France, 29200 Novartis Investigative Site Caen, France, 14000 Novartis Investigative Site Creil, France, 60100 Novartis Investigative Site #2 Paris, France, 75012 Novartis Investigative Site Paris, France, 75020 Novartis Investigative Site Pau, France, 64000 Novartis Investigative Site St Jean de la Ruelle, France, 45140 Novartis Investigative Site Strasbourg, France, 67000 Novartis Investigative Site Strasbourg, France Novartis Investigative Site Toulouse, France, 31200 Novartis Investigative Site Tours, France, 37000 Novartis Investigative Site Venissieux, France, 69200 Novartis Investigative Site #2 Venissieux, France Novartis Investigative Site Versailles, France, 78000 Collapse << |
| NCT01619059 | - | - | Completed | - | - |
| NCT01064687 | - | - | Completed | - | - |
| NCT01619059 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00067951 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00308373 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00755846 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03430856 | Type2 Diabetes Mellitus | Phase 2 Phase 3 | Recruiting | September 2020 | India ... more >> Diacon Hospital Recruiting Bangalore, Karnataka, India, 560010 Contact: Aravind Aravind, MBBS, DNB 8028085305 draravind@hotmail.com Contact: Geethanjali Rangaswamy 9008465877 geethanjali.kst@gmail.com Collapse << |
| NCT01984606 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn | February 2017 | - |
| NCT02229396 | Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00541450 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00755846 | - | - | Completed | - | - |
| NCT00395343 | - | - | Completed | - | - |
| NCT00099619 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Australia, New South Wales ... more >> Research Site Westmead, New South Wales, Australia Australia, South Australia Research Site Daw Park, South Australia, Australia Research Site Fullarton, South Australia, Australia Australia, Victoria Research Site Box Hill, Victoria, Australia Research Site East Ringwood, Victoria, Australia Greece Research Site Athens, Greece Research Site Piraeus, Greece Research Site Thessaloniki, Greece Hungary Research Site Budapest, Hungary Research Site Gyula, Hungary Research Site Pecs, Hungary Research Site Veszprem, Hungary Research Site Zalaegerszeg, Hungary Italy Research Site Bari, Italy Research Site Bergamo, Italy Research Site Catania, Italy Research Site Milan, Italy Research Site Perugia, Italy Research Site Rome, Italy Mexico Research Site Guadalajara, Jalisco, Mexico Research Site Zapopan, Jalisco, Mexico Research Site Monterrey, N.l., Mexico Research Site Mexico City, Mexico Poland Research Site Bydgoszcz, Poland Research Site Gdansk, Poland Research Site Lublin, Poland Collapse << |
| NCT02229396 | - | - | Completed | - | - |
| NCT01278160 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Beijing, Beijing, China, 100029 Collapse << |
| NCT00541450 | - | - | Completed | - | - |
| NCT00395343 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01144975 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Mexico ... more >> Mexico City, Mexico Collapse << |
| NCT02584075 | Coronary Endothelial Function | Phase 4 | Unknown | January 2018 | - |
| NCT01071850 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT02969798 | Diabetes Mellitus, Type 2 ... more >> Impaired Glucose Tolerance (IGT) Impaired Fasting Glucose (IFG) Collapse << | Not Applicable | Recruiting | July 2020 | United States, Texas ... more >> The University of Texas Health Science Center at San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Ralph A DeFronzo, MD 210-567-6691 defronzo@uthscsa.edu Contact: Monica Palomo, BS 210-567-6710 palomom@uthscsa.edu Principal Investigator: Ralph A DeFronzo, MD Sub-Investigator: Eugenio Cersosimo, MD Collapse << |
| NCT01278160 | - | - | Completed | - | - |
| NCT02020616 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Terminated(Lack of Efficacy) | - | United States, California ... more >> Orange County Research Center Orange, California, United States, 92868 United States, Florida Miami Research Associates Miami, Florida, United States, 33143 Compass Research Orlando, Florida, United States, 32806 United States, New York Clinilabs, Inc (New York) New York, New York, United States, 10019 United States, Texas Dallas Diabetes Endocrine Center Dallas, Texas, United States, 75230 Collapse << |
| NCT03642717 | - | - | Recruiting | March 31, 2020 | Korea, Republic of ... more >> Hyewon Medical Foundation Sejong Hospital Recruiting Bucheon, Korea, Republic of, 14754 Inje University Busan Paik Hospital Recruiting Busan, Korea, Republic of, 47392 Keimyung University Dongsan Hospital Recruiting Daegu, Korea, Republic of, 41931 Inha University Hospital Recruiting Incheon, Korea, Republic of, 22332 Seoul Medical Center Recruiting Seoul, Korea, Republic of, 02053 Hongik Hospital Recruiting Seoul, Korea, Republic of, 07937 Ajou University Hospital Recruiting Suwon, Korea, Republic of, 16499 Yonsei University Wonju Severance Christian Hospital Recruiting Wonju, Korea, Republic of, 26426 Collapse << |
| NCT02686177 | Type 2 Diabetes | Phase 4 | Recruiting | June 18, 2019 | France ... more >> Nutrition department, pole ENDO - Assistance Publique Hôpitaux Marseille Recruiting Marseille, France, 13005 Contact: Anne DUTOUR, MD, PhD 33 4 91 38 29 63 anne.dutour@ap-hm.fr Collapse << |
| NCT01193296 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> Investigative Site Bondy, France Investigative Site Corbeil, France Investigative Site Marseille, France Investigative Site Nancy, France Investigative Site Paris, France Investigative Site Toulouse, France Investigative Site Venissieux, France Collapse << |
| NCT02020616 | - | - | Terminated(Lack of Efficacy) | - | - |
| NCT01789957 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00699322 | Type 2 Diabetes | Phase 4 | Unknown | June 2010 | Korea, Republic of ... more >> Kangnam St. Mary's hospital Recruiting Seoul, Korea, Republic of, 137-701 Sub-Investigator: Seung-Hwan Lee, M.D. Sub-Investigator: Eun-Sook Kim, M.D. Principal Investigator: Kun-Ho Yoon, M.D., Ph.D. Collapse << |
| NCT02712632 | - | - | Recruiting | January 2019 | United Kingdom ... more >> University of Dundee Recruiting Dundee, Tayside, United Kingdom, DD1 9SY Contact: Louise Greig, RGN, MBA 01382 383208 louise.greig@nhs.net Collapse << |
| NCT01734785 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01356381 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United Kingdom ... more >> Novartis Investigative Site Newcastle Upon Tyne, United Kingdom, NE4 5PL Collapse << |
| NCT00135330 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Phoenix, Arizona, United States United States, California Research Site San Francisco, California, United States United States, Florida Research Site Jacksonville, Florida, United States Research Site Orlando, Florida, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Hawaii Research Site Honolulu, Hawaii, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Massachusetts Research Site Boston, Massachusetts, United States Research Site Pittsfield, Massachusetts, United States United States, Minnesota Research Site Minneapolis, Minnesota, United States Research Site Rochester, Minnesota, United States United States, New York Research Site Bronx, New York, United States Research Site Syracuse, New York, United States United States, Ohio Research Site Cincinatti, Ohio, United States United States, Texas Research Site League City, Texas, United States Research Site New Braunfels, Texas, United States Research Site San Antonio, Texas, United States United States, Washington Research Site Renton, Washington, United States Research Site Spokane, Washington, United States Collapse << |
| NCT00135330 | - | - | Completed | - | - |
| NCT01734785 | - | - | Completed | - | - |
| NCT00316758 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01278485 | - | - | Completed | - | - |
| NCT01278485 | - | - | Completed | - | - |
| NCT02682563 | Diabetes Mellitus, Type 2 ... more >> Diabetic Nephropathies Collapse << | Phase 4 | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Noord-Holland, Netherlands, 1081 HV Collapse << |
| NCT01339143 | Diabetes Mellitus ... more >> Oxidative Stress Collapse << | Phase 4 | Unknown | May 2013 | Korea, Republic of ... more >> Korea University Anam Hospital Recruiting Seoul, Korea, Republic of Collapse << |
| NCT00817271 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT00069836 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02401880 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Profil Mainz GmbH & Co. KG Mainz, Germany, 55116 Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT01576328 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | United States, California ... more >> SC Clinical Research Garden Grove, California, United States, 92844 United States, Florida Diabetes Research Institute Miami, Florida, United States, 33136 Compass Research Orlando, Florida, United States, 32806 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States Tulane University Medical Center New Orleans, Louisiana, United States United States, Montana Big Sky Clinical Research Butte, Montana, United States, 59701 United States, Nevada Desert Endocrinology Clinical Research Center-Henderson Henderson, Nevada, United States, 89052 Alliance Against Diabetes/AAD Clinical Research Las Vegas, Nevada, United States, 89101 United States, New Hampshire Active Practices and Research Newington, New Hampshire, United States, 03801 United States, Ohio The Carl and Edyth Lindner Center for Research and Education at The Christ Hospital Cincinnati, Ohio, United States, 45219 Providence Health Partners - Center for Clinical Reseach Dayton, Ohio, United States, 45439 United States, Texas Dallas Diabetes and Endocrine Center Dallas, Texas, United States, 75230 West Houston Clinical Research Services Houston, Texas, United States Paragon Research Center San Antonio, Texas, United States, 78205 United States, Utah Wasatch Clinical Research Salt Lake City, Utah, United States United States, Virginia National Clinical Research - Norfolk, Inc Norfolk, Virginia, United States, 23502 National Clinical Research - Richmond, Inc. Richmond, Virginia, United States, 23294 United States, Washington Capital Clinical Research Center Olympia, Washington, United States, 98502 Collapse << |
| NCT01674348 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(Due to non-recruitm... more >>ent, the study is being halted. There are no major safety or tolerability concerns in the study conducted so far.) Collapse << | - | Canada, Ontario ... more >> LMC Endocrinolgy Centres Ltd Toronto, Ontario, Canada, M4G 3E8 Collapse << |
| NCT00819884 | Type II Diabetes | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00363948 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> Chubu Region Chubu, Japan Chugoku Region Chugoku, Japan Hokkaido Region Hokkaido, Japan Hokuriku Region Hokuriku, Japan Kanto Region Kanto, Japan Kinki Region Kinki, Japan Kyushu Region Kyushu, Japan Tohoku Region Tohoku, Japan Collapse << |
| NCT00679939 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT02653599 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01017302 | Diabetes Mellitus Type 2 | Phase 1 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States, 78229 Collapse << |
| NCT01455922 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT00174642 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00618007 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01455870 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00679939 | - | - | Completed | - | - |
| NCT01455909 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT01455883 | Type 2 Diabetes | Phase 3 | Withdrawn | July 2014 | United States, California ... more >> Intarcia Therapeutics, Inc Hayward, California, United States, 94545 Collapse << |
| NCT01045707 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03029702 | Gestational Diabetes ... more >> Diabetes, Gestational Pregnancy in Diabetes Collapse << | Phase 4 | Recruiting | March 30, 2021 | United States, Pennsylvania ... more >> Magee Womens Hospital of UPMC Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Maisa Feghali, MD 412-641-4874 feghalim@upmc.edu Principal Investigator: Maisa Feghali, MD Collapse << |
| NCT01045707 | - | - | Completed | - | - |
| NCT02111096 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(The overall benefit... more >>-risk profile did not support continued development of LY2409021 for type 2 diabetes.) Collapse << | - | - |
| NCT02462369 | Microalbuminuria ... more >> Microalbuminuria /Creatinine Ratios ACR Collapse << | Phase 4 | Unknown | October 2017 | - |
| NCT02887625 | Type 2 Diabetes | Not Applicable | Unknown | May 2017 | Qatar ... more >> Hamad General Hospital Recruiting Doha, Qatar Contact: Osama Mujahed, MD 66703172 pvedromigahid@gmail.com Contact: Ayman Mujahed, MD 44391208 ymndodo20@gmail.com Collapse << |
| NCT02111096 | - | - | Terminated(The overall benefit... more >>-risk profile did not support continued development of LY2409021 for type 2 diabetes.) Collapse << | - | - |
| NCT02824003 | Type 2 Diabetes | Phase 2 | Completed | - | Austria ... more >> Ionis Investigator Site Vienna, Austria, A-1130 Collapse << |
| NCT01933672 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Florida ... more >> Avail Clinical Research, LLC DeLand, Florida, United States, 32720 Miami Research Associates, Inc. South Miami, Florida, United States, 33143 MRA Clinical Research, LLC South Miami, Florida, United States, 33143 United States, North Carolina High Point Clinical Trials Center, LLC High Point, North Carolina, United States, 27265 United States, Ohio Community Research Cincinnati, Ohio, United States, 45255 United States, Texas Clinical Trials of Texas, Inc San Antonio, Texas, United States, 78229 Diabetes and Glandular Disease Clinic San Antonio, Texas, United States, 78229 United States, Wisconsin Spaulding Clinical Research, LLC West Bend, Wisconsin, United States, 53095 Collapse << |
| NCT02567994 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT00097279 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01933672 | - | - | Completed | - | - |
| NCT02583919 | Type 2 Diabetes | Phase 2 | Completed | - | United States, California ... more >> Ionis Investigational Site Greenbrae, California, United States, 94904 Ionis Investigational Site Huntington Park, California, United States, 90255 Ionis Investigational Site La Mesa, California, United States, 91942 Ionis Investigational Site Los Angeles, California, United States, 90057 Isis Investigational Site Spring Valley, California, United States, 91978 United States, Florida Ionis Investigational Site Port Orange, Florida, United States, 32127 United States, Georgia Ionis Investigational Site Marietta, Georgia, United States, 30060 United States, Maryland Ionis Investigational Site Oxon Hill, Maryland, United States, 20745 United States, New York Ionis Investigational Site New York, New York, United States, 10016 United States, North Carolina Ionis Investigational Site Greensboro, North Carolina, United States, 27410 Ionis Investigational Site Morehead City, North Carolina, United States, 28557 United States, Ohio Ionis Investigational Site Cincinnati, Ohio, United States, 45219 Ionis Investigational Site Cincinnati, Ohio, United States, 45245 United States, Oregon Ionis Investigational Site Eugene, Oregon, United States, 97404 United States, South Carolina Ionis Investigational Site Spartanburg, South Carolina, United States, 29303 United States, Texas Ionis Investigational Site Houston, Texas, United States, 77036 Ionis Investigational Site Houston, Texas, United States, 77074 Ionis Investigational Site Katy, Texas, United States, 77450 United States, Washington Ionis Investigational Site Olympia, Washington, United States, 98502 Collapse << |
| NCT00032487 | - | - | Completed | - | - |
| NCT02475070 | Type 2 Diabetes | Phase 4 | Completed | - | Sweden ... more >> Lund University Lund, Sweden, 22184 Collapse << |
| NCT01885260 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Alabama ... more >> Isis Investigational Site Birmingham, Alabama, United States, 35216 Isis Investigational Site Muscle Shoals, Alabama, United States, 35662 United States, California Isis Investigational Site Anaheim, California, United States, 92801 Isis Investigational Site Chino, California, United States, 91710 Isis Investigational Site Los Angeles, California, United States, 90057 United States, Florida Isis Investigational Site Deland, Florida, United States, 32720 Isis Investigational Site Ft. Lauderdale, Florida, United States, 33306 Isis Investigational Site Miami, Florida, United States, 33143 United States, Georgia Isis Investigational Site Marietta, Georgia, United States, 30066 United States, Nevada Isis Investigational Site Las Vegas, Nevada, United States, 89101 United States, Ohio Isis Investigational Site Cincinnati, Ohio, United States, 45245 United States, Oklahoma Isis Investigational Site Oklahoma City, Oklahoma, United States, 73103 United States, Oregon Isis Investigational Site Eugene, Oregon, United States, 97404 United States, Texas Isis Investigational Site Corpus Christi, Texas, United States, 78404 Isis Investigational Site Katy, Texas, United States, 77450 United States, Utah Isis Investigational Site Salt Lake City, Utah, United States, 84107 United States, Washington Isis Investigational Site Renton, Washington, United States, 98057 Isis Investigational Site Spokane, Washington, United States, 99202 South Africa Isis Investigational Site Lenasia South, Gauteng, South Africa, 1829 Isis Investigational Site Soweto, Gauteng, South Africa, 1818 Isis Investigational Site Durban, KwaZulu-Natal, South Africa, 4091 Isis Investigational Site Cape Town, Western Cape, South Africa, 7700 Isis Investigational Site Parow, Cape Town, Western Cape, South Africa, 7500 Isis Investigational Site Western Cape, South Africa, 7130 Collapse << |
| NCT02303405 | Diabetes Mellitus Type 2 With ... more >>Hyperglycemia Collapse << | Phase 2 | Terminated(Investigator decisi... more >>on) Collapse << | - | United States, California ... more >> Charles Drew University of Medicine and Science Los Angeles, California, United States, 90059 Collapse << |
| NCT00032487 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02256189 | Type 2 Diabetes | Phase 4 | Completed | - | Sweden ... more >> Skane University Hospital Malmö Malmö, Sweden, 20502 Collapse << |
| NCT02325960 | Type 2 Diabetes | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT00942318 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> University Hospital Toulouse Toulouse, France, 31059 Collapse << |
| NCT00823680 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United States, California ... more >> Chula Vista, California, United States, 91911 United States, Florida Miami, Florida, United States, 330014 Austria Graz, Austria, 8036 Germany München, Germany, 80636 Neuss, Germany, 41460 Collapse << |
| NCT03495102 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | October 7, 2019 | - |
| NCT00461643 | Anovulatory Infertility Relate... more >>d to Polycystic Ovary Syndrome Collapse << | Phase 4 | Unknown | - | Italy ... more >> "Pugliese" Hospital Recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39 0961 883234 angela.falbo@libero.it Principal Investigator: Stefano Palomba, MD "Pugliese" Hospital Recruiting Catanzaro, Italy, 88100 Contact: Ingrid Tomaino, MD +39 0961 883234 zullo@unicz.it Principal Investigator: Stefano Palomba, MD Principal Investigator: Francesco Orio, MD Principal Investigator: Achille Tolino, MD Principal Investigator: Pasquale Mastrantonio, MD Principal Investigator: Brian Dale, MD Principal Investigator: Ettore Cittadini, MD Principal Investigator: Andrea Riccardo Genazzani, MD Pugliese-Ciaccio Hospital Recruiting Catanzaro, Italy, 88100 Contact: , MD Collapse << |
| NCT02546609 | NAFLD | Phase 2 | Completed | - | United States, California ... more >> Catalina Research Institute Chino, California, United States, 91710 University of California San Diego San Diego, California, United States, 92103 United States, Colorado Rocky Mountain Research Wheat Ridge, Colorado, United States, 80033 United States, Georgia Atlanta Gastroenterology Associates Atlanta, Georgia, United States, 30312 GI Specialists of Georgia Marietta, Georgia, United States, 30060 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, North Carolina University of North Carolina Chapel Hill Chapel Hill, North Carolina, United States, 27599 United States, Ohio Sterling Research Cincinnati, Ohio, United States, 45246 United States, Tennessee Premier Clinical Research Clarksville, Tennessee, United States, 37043 Gastro One Germantown, Tennessee, United States, 38138 Quality Medical Research Nashville, Tennessee, United States, 37211 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT00506194 | Type 2 Diabetes | Not Applicable | Completed | - | Taiwan ... more >> Division of Endocrinology and Metabolism, Department of Medicine Taipei, Taiwan, 112 Collapse << |
| NCT02546609 | - | - | Completed | - | - |
| NCT00633425 | Diabetes Mellitus | Phase 4 | Completed | - | France ... more >> Sanofi-Aventis Paris, France Collapse << |
| NCT00494312 | Diabetes Mellitus ... more >> Liver Collapse << | Phase 4 | Completed | - | - |
| NCT01595789 | Coronary Artery Disease ... more >> Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Denmark ... more >> Copenhagen University Hospital, Bispebjerg Copenhagen, Bispebjerg, Denmark, 2400 Collapse << |
| NCT00543751 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated | - | - |
| NCT00818077 | - | - | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT01219582 | - | - | Completed | - | India ... more >> Many Locations, India Collapse << |
| NCT02492763 | Type II Diabetes Mellitus | Phase 2 | Terminated | - | - |
| NCT00964184 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Fortis Flt Lt Rajan Dhall Hospital New Delhi, Delhi, India, 110070 Collapse << |
| NCT03642600 | PCOS Obesity ... more >> Metabolic Syndrome Collapse << | Phase 4 | Not yet recruiting | December 30, 2019 | - |
| NCT02492763 | - | - | Terminated | - | - |
| NCT00780715 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United Kingdom ... more >> Ninewells Hospital & Medical School Dundee, United Kingdom, DD1 9SY Collapse << |
| NCT00780715 | - | - | Completed | - | - |
| NCT01990469 | T2DM | Phase 3 | Completed | - | - |
| NCT00123110 | Insulin Resistance ... more >> Postmenopause Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Clinical and Translational Research Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT02496000 | Diabetes Type 2 | Phase 2 | Completed | - | - |
| NCT00543738 | Diabetes Mellitus | Phase 3 | Terminated | - | - |
| NCT02496000 | - | - | Completed | - | - |
| NCT00347100 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China ... more >> Sanofi-Aventis Beijing, China Collapse << |
| NCT02726490 | Gestational Diabetes | Not Applicable | Terminated(Lack of Recruitment... more >>) Collapse << | - | United States, Texas ... more >> TTUHSC El Paso El Paso, Texas, United States, 79905 Collapse << |
| NCT03239119 | Type2 Diabetes Mellitus | Phase 3 | Not yet recruiting | January 15, 2019 | - |
| NCT01025765 | Chronic Hepatitis C | Phase 4 | Unknown | December 2012 | Taiwan ... more >> National Taiwan University Hospital Department of Internal Medicine, Taipei, Taiwan, 10002 Collapse << |
| NCT00541775 | - | - | Completed | - | - |
| NCT02274740 | Type II Diabetes Mellitus | Phase 2 | Terminated(Sponsor decision no... more >>t related to safety reasons) Collapse << | - | - |
| NCT03130426 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | December 2019 | Canada, Alberta ... more >> University of Calgary Recruiting Calgary, Alberta, Canada, T2T 5C7 Contact: Ron Sigal, MD 403-955-8107 rsigal@ucalgary.ca Canada, Ontario LMC Recruiting Burlington, Ontario, Canada, M4G 3E8 Contact: Mohammed Azharuddin, MD Joanne Liutkus Recruiting Cambridge, Ontario, Canada, N1R 7L6 Contact: Danielle Gelinas 519-624-8977 McMaster University Recruiting Hamilton, Ontario, Canada, L8N 3Z5 Contact: Natalia McInnes, MD 905-521-2100 ext 73794 natalia.mcinnes@mcmaster.ca Contact: Ada Smith 905-521-2100 ext 22205 smithad@mcmaster.ca St. Joseph's Hospital Recruiting London, Ontario, Canada, N6A 4V2 Contact: Irene Hramiak, MD 519-646-6353 Irene.Hramiak@sjhc.london.on.ca Western University Recruiting London, Ontario, Canada, N6G 2M1 Contact: Stewart Harris, MD 519-661-2111 Stewart.Harris@schulich.uwo.ca The Ottawa Hospital Recruiting Ottawa, Ontario, Canada, K1Y 4E9 Contact: Heather Lochnan, MD hlochnan@toh.ca Canada, Quebec McGill University Recruiting Montreal, Quebec, Canada, H4A 3J1 Contact: Angie Lombardo 514-934-1934 ext 36492 angie.lombardo@muhc.mcgill.ca Collapse << |
| NCT00541775 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02561130 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | October 2018 | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2T 5C7 Canada, Manitoba Health Science Centre Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario McMaster University Hamilton, Ontario, Canada, L8N 3Z5 St. Joseph's Hospital London, Ontario, Canada, N6A 4V2 Western University London, Ontario, Canada, N6G 2M1 LMC Toronto, Ontario, Canada, M4G 3E8 St. Michaels's Hospital Toronto, Ontario, Canada, M5C 2T2 Collapse << |
| NCT01937598 | Type 2 Diabetes | Phase 3 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Germany, 37431 Collapse << |
| NCT00971659 | Type 2 Diabetes | Phase 1 | Completed | - | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT02042664 | Obesity Diabe... more >>tes Collapse << | Phase 3 | Completed | - | France ... more >> Assistance Publique Hopitaux de Marseille Marseille, France, 13354 Collapse << |
| NCT01937598 | - | - | Completed | - | - |
| NCT03378297 | Ovarian Cancer | Early Phase 1 | Recruiting | June 1, 2020 | Norway ... more >> Helse Bergen HF, Haukeland University Hospital Recruiting Bergen, Norway, 5021 Contact: Liv Cecilie V Thomsen, MD, PhD +47 55975000 Liv.Vestrheim@uib.no Contact: Line Bjørge, MD, PhD +47 55975000 line.bjorge@uib.no Principal Investigator: Line Bjørge, MD, PhD Sub-Investigator: Liv Cecilie V Thomsen, MD, PhD Sub-Investigator: Kathrine Woie, MD, PhD Sub-Investigator: Bjørn Tore Gjertsen, MD, PhD Oslo University Hospital Recruiting Oslo, Norway, 0424 Contact: Erik Rokkones, MD, PhD +47 915 02770 erk@ous-hf.no Contact: Olysea Solheim, MD, PhD +47 915 02770 olsolh@ous-hf.no Principal Investigator: Erik Rokkones, MD, PhD Sub-Investigator: Olysea Solheim, MD, PhD Helse Stavanger HF, Stavanger University Hospital Recruiting Stavanger, Norway, 4068 Contact: Bent Fiane, MD +47 51518000 bent.eltvedt.fiane@sus.no Contact: Cecilie F Torkildsen, MD +47 51518000 cecilie.torkildsen@gmail.com Principal Investigator: Bent Fiane, MD Sub-Investigator: Cecilie F Torkildsen, MD Sub-Investigator: Ragnar K Sande, MD, PhD Collapse << |
| NCT02587741 | Diabetic Retinopathy | Early Phase 1 | Recruiting | July 2025 | China, Guangdong ... more >> the third affiliated hospital of Sun yet-san university Recruiting Guangzhou, Guangdong, China, 510000 Contact: Zhu Bilian, master +8613580364394 bilianzhu@qq.com Contact: Tang Xixiang, master +8613570434387 txx_8711@126.com Collapse << |
| NCT00374907 | Type 2 Diabetes | Phase 3 | Completed | - | United States, California ... more >> Va San Diego Healthcare System San Diego, California, United States, 92161 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 United States, Texas Diabetes & Glandular Disease Research Assoc,, Inc. San Antonio, Texas, United States, 78229 Collapse << |
| NCT00673465 | - | - | Completed | - | - |
| NCT00374907 | - | - | Completed | - | - |
| NCT03072485 | Aging | Phase 1 | Recruiting | December 31, 2017 | United States, California ... more >> Stanford Dermatology Recruiting Redwood City, California, United States, 94603 Contact: Anne Chang, MD 650-721-7151 alschang@stanford.edu Contact: Michaella Montana, BS 650 721 7159 mmontana@stanford.edu Collapse << |
| NCT03617458 | Pulmonary Artery Hypertension | Phase 2 | Recruiting | December 2022 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01965509 | Type 2 Diabetes | Phase 2 | Completed | - | China ... more >> China-Japan Friendship Hospital Beijing, China Collapse << |
| NCT01224366 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01477853 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(The study was termi... more >>nated early by the Sponsor for business reasons.) Collapse << | - | - |
| NCT00673465 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT01477853 | - | - | Terminated(The study was termi... more >>nated early by the Sponsor for business reasons.) Collapse << | - | - |
| NCT01046422 | Diabetes Type 2 | Phase 2 | Completed | - | United States, California ... more >> Marina Raikhel, Md Lomita, California, United States, 90717 United States, Nevada Nevada Alliance Against Diabetes Las Vegas, Nevada, United States, 89101 Australia, Queensland Local Institution Caboolture, Queensland, Australia, 4510 Local Institution Meadowbrook, Queensland, Australia, 4131 Australia, South Australia Local Institution Daw Park, South Australia, Australia, 5041 Australia, Victoria Local Institution Geelong, Victoria, Australia, 3220 Australia, Western Australia Local Institution Nedlands, Western Australia, Australia, 6009 Canada, Manitoba Local Institution Winnipeg, Manitoba, Canada, R3P 1R9 Canada, Newfoundland and Labrador Local Institution Mount Pearl, Newfoundland and Labrador, Canada, A1N 1W7 Canada, Ontario Local Institution Brampton, Ontario, Canada, L6T 3J1 Local Institution Toronto, Ontario, Canada, M4G 3E8 Canada, Prince Edward Island Local Institution Charlottetown, Prince Edward Island, Canada, C1A 5Y9 Canada, Quebec Local Institution Drummondville, Quebec, Canada, J2B 7T1 Local Institution Lachine, Quebec, Canada, H8S 2E4 Local Institution Laval, Quebec, Canada, H7T 2P5 Local Institution Montreal, Quebec, Canada, H3J 2V5 Korea, Republic of Local Institution Incheon, Korea, Republic of, 405-760 Local Institution Suwon, Korea, Republic of, 443-721 Collapse << |
| NCT02290184 | Insulin Resistance, ... more >> Metabolic Syndrome Type 2 Diabetes Collapse << | Not Applicable | Unknown | October 2016 | United States, California ... more >> San Francisco / Daly City Communities San Francisco, California, United States, 94115 Collapse << |
| NCT02846233 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | December 2018 | United States, California ... more >> UCSF Fresno Recruiting Fresno, California, United States, 93701 Contact: SOE NAING snaing@fresno.ucsf.edu Collapse << |
| NCT03053518 | Type2 Diabetes | Not Applicable | Recruiting | December 2019 | United States, New York ... more >> New York University School of Medicine Recruiting New York, New York, United States, 10016 Contact: Ram Jagannathan Ram.Jagannathan@nyumc.org Principal Investigator: Mary Sevick, MD Collapse << |
| NCT00686634 | - | - | Completed | - | - |
| NCT00694057 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 Anaheim, California, United States, 92805 Beverly Hills, California, United States, 90211 Buena Park, California, United States, 90620 Burbank, California, United States, 91505 Carmichael, California, United States, 95608 Escondido, California, United States, 92025 Fresno, California, United States, 93726 Huntington Park, California, United States, 90255 Long Beach,, California, United States, 90806 Los Angeles, California, United States, 90057 National City, California, United States, 91950 Roseville, California, United States, 95661 United States, Illinois Chicago, Illinois, United States, 60616 United States, Louisiana Baton Rouge, Louisiana, United States, 70808 United States, Missouri St. Louis, Missouri, United States, 63141 United States, Nevada Las Vegas, Nevada, United States, 89106 Las Vegas, Nevada, United States, 89148 United States, Texas El Paso, Texas, United States, 79905 San Antonio, Texas, United States, 78229 United States, Utah Salt Lake City, Utah, United States, 84107 United States, Wisconsin Kenosha, Wisconsin, United States, 53142 Collapse << |
| NCT00686634 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, California ... more >> Charles Drew University of Medicine and Science Los Angeles, California, United States, 90059 Collapse << |
| NCT02941367 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | India ... more >> Investigational Site Number 356005 Ahmedabad, India, 380007 Investigational Site Number 356002 Bangalore, India, 560043 Investigational Site Number 356015 Bangalore, India, 560055 Investigational Site Number 356008 Bangalore, India, 560060 Investigational Site Number 356009 Hyderabad, India, 500001 Investigational Site Number 356018 Hyderabad, India, 500004 Investigational Site Number 356010 Hyderabad, India, 500072 Investigational Site Number 356003 Hyderabad, India, 500095 Investigational Site Number 356007 Jaipur, India Investigational Site Number 356019 Madurai, India, 625020 Investigational Site Number 356014 Mumbai, India, 400007 Investigational Site Number 356022 Nagpur, India, 440010 Israel Investigational Site Number 376001 Haifa, Israel, 31096 Investigational Site Number 376002 Safed, Israel, 13100 Kuwait Investigational Site Number 414001 Kuwait, Kuwait Turkey Investigational Site Number 792002 Zonguldak, Turkey Collapse << |
| NCT02856113 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | June 11, 2021 | - |
| NCT01162772 | - | - | Unknown | - | Austria ... more >> Medical University of Viemma Recruiting Vienna, Austria, 1090 Contact: Jeanette Strametz-Juranek, MD 0043140400 ext 4618 jeanette.strametz-juranek@meduniwien.ac.at Contact: Alexandra Kautzky-Willer, MD 0140400 ext 4316 alexandra.kautzky-willer@meduniwien.ac.at Principal Investigator: Jeanette Strametz-Juranek, MD Principal Investigator: Alexandra Kautzky-Willer, MD Sub-Investigator: Alia Sabri, MD Sub-Investigator: Lana Kosi, MD Collapse << |
| NCT02882477 | Diabetes Mellitus ... more >> Iron Metabolism Disorders Gastroduodenal Ulcer Optic Atrophy Sensorineural Hearing Loss Platelet Dysfunction Collapse << | Phase 2 Phase 3 | Unknown | December 2018 | Israel ... more >> Hadassah medical center Recruiting Jerusalem, Israel Contact: David Zangen, Professor 0097507874488 zanegnd@hadassah.org.il Contact: Ulla Najwa Abdulhag, MD 0097505172866 najwa.ped@gmail.com Collapse << |
| NCT02638805 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00360698 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, New Jersey ... more >> Sanofi-aventis Bridgewater, New Jersey, United States, 08807 Russian Federation Sanofi-Aventis Moscow, Russian Federation United Kingdom Sanofi-aventis Guildford, United Kingdom Collapse << |
| NCT02201381 | Cancer Overal... more >>l Survival Collapse << | Phase 3 | Recruiting | October 22, 2023 | United Kingdom ... more >> Care Oncology Clinic Recruiting London, United Kingdom, W1G 9PP Contact: Gregory Stoloff +44 207 580 3266 info@careoncologyclinic.com Contact: Robin Bannister, PhD +44 207 153 6577 robin.bannister@seekacure.com Collapse << |
| NCT03646799 | Type II Diabetes | Phase 1 | Recruiting | September 14, 2018 | Korea, Republic of ... more >> Yonsei University Severance Hospital Recruiting Soeul, Korea, Republic of Contact: Min Soo Park, Ph.D. M.D +82 2 2228 0400 minspark@yuhs.ac Principal Investigator: Min Soo Park, Ph.D. M.D Collapse << |
| NCT02152371 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01028391 | - | - | Completed | - | - |
| NCT01952145 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00360698 | - | - | Completed | - | - |
| NCT01028391 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02152371 | - | - | Completed | - | - |
| NCT02743260 | Healthy | Phase 4 | Unknown | December 2017 | Germany ... more >> Department of Pharmacology I, University Hospital Cologne Cologne, NRW, Germany, 50931 Collapse << |
| NCT01952145 | - | - | Completed | - | - |
| NCT01901757 | Diabetes Mellitus | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01217073 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02091362 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01217073 | - | - | Completed | - | - |
| NCT01181674 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Canada, Ontario ... more >> McMaster University Medical Centre, Diabetes Care and Research Program Hamilton, Ontario, Canada, L8N 3Z5 Collapse << |
| NCT02091362 | - | - | Completed | - | - |
| NCT02229227 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02229240 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(Study was cancelled ... more >>prior to enrolling any patients) Collapse << | December 2015 | - |
| NCT01089569 | Type 2 Diabetes | Not Applicable | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT02304536 | Infertility | Phase 3 | Recruiting | - | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Cairo University Hospitals Recruiting Cairo, Egypt Contact: AbdelGany MA Hassan, MRCOG, MD 00217801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT01089569 | - | - | Completed | - | - |
| NCT02635646 | Prediabetes | Not Applicable | Completed | - | Mexico ... more >> Universidad de Guanajuato León, Guanajuato, Mexico, 37670 Collapse << |
| NCT02897349 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | January 30, 2019 | China ... more >> China-Japan Friendship Hospital Beijing, China, 100029 Beijing Tongren Hospital Beijing, China, 100730 The General Hospital of Chinese People's Armed Police Forces Beijing, China, 100854 First Hospital of Jilin University Changchun, China, 130021 Fuzhou General Hospital of Nanjing Militray Command Fuzhou, China, 350025 Third Affiliated Hospital of Guangzhou Medical University Guangzhou, China, 510150 The Affiliated Hospital of Guizhou Medical University Guiyang, China, 550004 Hangzhou Second People's Hospital Hangzhou, China, 310015 General Hospital of Jinan Military Area Jinan, China, 250000 Nanjing First Hospital Nanjing, China, 210006 The affiliated hospital of medicalcollege qingdao university Qingdao, China, 266005 Centre Hospital of Putuo District, Shanghai Shanghai, China, 200062 Shanghai Tenth People's Hospital Shanghai, China, 200072 Yangpu Hospital, Tongji University Shanghai, China, 200090 Second Hospital Affiliated to Shantou Medical University Shantou, China, 515041 Shengjing Hospital of China Medical University Shengyang, China, 110072 Siping Central People's Hospital Siping, China, 136000 The First Affiliated Hospital of Soochow University Suzhou, China, 215006 Nankai University Affiliated Hospital Tianjin, China, 300000 The 2nd Hospital of Tianjin Medical University Tianjin, China, 300000 Tianjin Medical University General Hospital Tianjin, China, 30052 Renmin Hospital of Wuhan University Wuhan, China, 430060 First Affiliated Hospital of Xiamen University Xiamen, China, 361003 the first people hospital of Yue Yang Yueyang, China, 414000 Affiliated Hospital of Jiangsu University Zhenjiang, China, 212013 Collapse << |
| NCT02836704 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China ... more >> China, China Collapse << |
| NCT02635646 | - | - | Completed | - | - |
| NCT01398592 | Diabetes Mellitus Type 2 | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Oeynhausen, Germany, 32549 Novartis Investigative Site Berlin, Germany, 12347 Novartis Investigative Site Berlin, Germany, 13597 Novartis Investigative Site Berlin, Germany, 14089 Novartis Investigative Site Celle, Germany, 29221 Novartis Investigative Site Hannover, Germany, 30165 Novartis Investigative Site Hannover, Germany, 30167 Novartis Investigative Site Kassel, Germany, 34117 Novartis Investigative Site Kirchhain, Germany, 35274 Novartis Investigative Site Lehrte, Germany, 31275 Novartis Investigative Site Mülheim, Germany, 45468 Novartis Investigative Site Oberhausen, Germany, 46049 Novartis Investigative Site Osnabrück, Germany, 49080 Novartis Investigative Site Potsdam, Germany, 14469 Novartis Investigative Site Stuttgart, Germany, 70378 Collapse << |
| NCT03226210 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | China, Jiangsu ... more >> Nanjing First Hostital Nanjing, Jiangsu, China, 210000 Collapse << |
| NCT01191268 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02229227 | - | - | Completed | - | - |
| NCT00789191 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00789191 | - | - | Completed | - | - |
| NCT03323294 | - | - | Recruiting | October 2019 | United States, Arkansas ... more >> Arkansas Children's Hospital Recruiting Little Rock, Arkansas, United States, 72202 Contact: Leanna Delhey, BS, MPH 501-364-4519 delheylm@archildrens.org Principal Investigator: Eugenia Carvalho, PhD Sub-Investigator: Shannon Rose, PhD Sub-Investigator: Elisabet Borsheim, PhD Sub-Investigator: Jon Oden, MD Sub-Investigator: Shipra Bansal, MD Sub-Investigator: Emir Tas, MD Sub-Investigator: Stepan Melnyk, MD, PhD Sub-Investigator: Judith Weber, PhD, RD Collapse << |
| NCT01191268 | - | - | Completed | - | - |
| NCT00804986 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00776607 | Latent Autoimmune Diabetes in ... more >>Adults LADA Collapse << | Not Applicable | Unknown | April 2011 | United Kingdom ... more >> Diabetes Unit Not yet recruiting Cardiff, Wales, United Kingdom, CF64 2XX Contact: Julia Platts 02920 711 711 ext 5149 Julia.Platts@CardiffandVale.wales.nhs.uk Principal Investigator: Julia Platts Clinical Research Unit Recruiting Swansea, Wales, United Kingdom, SA6 6NL Contact: Lucy Barlow +44 (0) 1792 703722 ext 3722 Lucy.Barlow@swansea-tr.wales.nhs.uk Principal Investigator: Jeffrey Stephens Collapse << |
| NCT00760344 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02463357 | Mountain Sickness | Phase 4 | Active, not recruiting | November 30, 2018 | United States, Michigan ... more >> Alma College Alma, Michigan, United States, 48801 Collapse << |
| NCT00763815 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01960205 | Prediabetes | Phase 4 | Unknown | - | China, Shandong ... more >> Shandong Provincial Hospital Recruiting Jinan, Shandong, China, 250021 Contact: Xinli Zhou, MD, PhD 15168889976 zhouxinli0301@163.com Principal Investigator: Xinli Zhou, MD,PhD Collapse << |
| NCT01342042 | Type 2 Diabetes | Phase 2 | Unknown | December 2011 | China, Beijing ... more >> Peking University First Hospital Recruiting Peking, Beijing, China, 010 Contact: Cui Yi Min, MD 86-10-88325988 Principal Investigator: Cui Yi Min, MD Collapse << |
| NCT02449187 | Diabetes Hype... more >>rlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Inha University Hospital Incheon, Korea, Republic of Collapse << |
| NCT00763815 | - | - | Completed | - | - |
| NCT00804986 | - | - | Completed | - | - |
| NCT03748810 | - | - | Recruiting | December 31, 2018 | Korea, Republic of ... more >> Chungbuk National University Hospital Recruiting Cheonju, Chungcheongbuk-do, Korea, Republic of, 28644 Contact: Eu Jeong Ku, MD 82-10-3462-8863 eujeong.ku@gmail.com Collapse << |
| NCT03383068 | PCOS Impaired... more >> Glucose Tolerance Collapse << | Phase 4 | Not yet recruiting | June 30, 2020 | China, Shanghai ... more >> Renji Hospital Department of Endocrinology and Metabolism Not yet recruiting Shanghai, Shanghai, China, 200127 Contact: Wei Liu, MD +86-18918358342 sue_liuwei@163.com Collapse << |
| NCT00268476 | Prostate Cancer | Phase 2 Phase 3 | Recruiting | September 2024 | - |
| NCT02411825 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Germany ... more >> Investigational Site Number 276001 Berlin, Germany, 10117 Collapse << |
| NCT03006172 | Breast Cancer ... more >> Solid Tumor Collapse << | Phase 1 | Recruiting | February 29, 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital. Recruiting Boston, Massachusetts, United States, 02114 Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 United States, New York Columbia University Medical Center Recruiting New York, New York, United States, 10032 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 United States, Tennessee Sarah Cannon Research Institute Recruiting Nashville, Tennessee, United States, 37203 Canada, Ontario Philippe Bedard Clinic Toronto Recruiting Toronto, Ontario, Canada, M5G 1L7 France Institut Bergonie Recruiting Bordeaux, France, 33076 Institut Gustave Roussy Recruiting Villejuif, France, 94805 Spain Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Hospital Clinico Universitario de Valencia Recruiting Valencia, Spain, 46010 United Kingdom Barts and the London NHS Trust. Recruiting London, United Kingdom, EC1A 7BE Royal Marsden Hospital - London Recruiting London, United Kingdom, SW3 6JJ Royal Marsden Hospital - Surrey Recruiting Surrey, United Kingdom, SM2 5PT Collapse << |
| NCT03359590 | Pharmacological Action | Phase 2 | Enrolling by invitation | July 30, 2019 | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany Collapse << |
| NCT01033773 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, District of Col... more >>umbia Washington Hospital Center Washington, District of Columbia, United States, 20010 Collapse << |
| NCT01392573 | - | - | Completed | - | - |
| NCT01392573 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00667732 | Type 2 Diabetes | Phase 4 | Completed | - | United States, District of Col... more >>umbia Medstar Research Institute Washington DC, District of Columbia, United States, 20003 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT02694263 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | September 2018 | United Kingdom ... more >> University Hospitals of Leicester NHS Trust Recruiting Leicester, Leicestershire, United Kingdom, LE5 4PW Contact: Emer Brady, PhD 0116248 ext 8959 emer.brady@uhl-tr.nhs.uk Contact: Natasha Wileman, MSc 0116258 ext 8929 natasha.wileman@uhl-tr.nhs.uk University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, West Midlands, United Kingdom, B15 2TH Contact: Wasim Hanif 0121371 ext 4404 wasim.hanif@uhb.nhs.uk Contact: Deborah Smith 0121371 ext 4707 deborah.smith@uhb.nhs.uk Collapse << |
| NCT01963130 | - | - | Completed | - | Turkey ... more >> Bezmialem VU Istanbul, Turkey, 34093 Collapse << |
| NCT03717194 | Type2 Diabetes ... more >> Heart Failure Collapse << | Phase 3 | Not yet recruiting | October 30, 2020 | Korea, Republic of ... more >> Seoul National University Bundang Hospita; Not yet recruiting Seongnam-si, Gyeonggi-do, Korea, Republic of, 13620 Collapse << |
| NCT00622323 | - | - | Completed | - | Philippines ... more >> Research Site Paranaque City, Philippines Research Site Pasig City, Philippines Research Site Taytay, Philippines Collapse << |
| NCT02292290 | Type 2 Diabetes | Phase 4 | Completed | - | United Kingdom ... more >> University Hospitals of Leicester Leicester, Leicestershire, United Kingdom University Hospital Birmingham Birmingham, West Midlands, United Kingdom Collapse << |
| NCT01503008 | Type 2 Diabetes Mellitus | Not Applicable | Withdrawn | August 2012 | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 United States, Massachusetts Center for Connected Health, Partners Healthcare Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00121966 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Denmark ... more >> Diabetes Research Center Odense, Denmark, 5000 Collapse << |
| NCT02586129 | Dyslipidemia ... more >>Type II Diabetes Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul National Universitiy Bundang Hospital Seongnam-Si, Gyeonggi-Do, Korea, Republic of, 463-707 Collapse << |
| NCT00667732 | - | - | Completed | - | - |
| NCT02597101 | Type 2 Diabetes Mellitus ... more >> Insulin Resistance Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, Pennsylvania ... more >> Allegheny Health Network Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT01658501 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00868790 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(Upon interim analys... more >>is, sufficient data was accrued to assess study hypotheses.) Collapse << | - | - |
| NCT00004992 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Glucose Intolerance Collapse << | Phase 3 | Completed | - | United States, Maryland ... more >> George Washington University Biostatistics Center, Suite 750, 6110 Executive Blvd Rockville, Maryland, United States, 20852 Collapse << |
| NCT02749032 | Type 2 Diabetes | Phase 1 | Completed | - | Germany ... more >> Diabeteszentrum Bad Lauterberg Bad Lauterberg, Niedersachsen, Germany, 37431 Collapse << |
| NCT01009580 | - | - | Completed | - | - |
| NCT00868790 | - | - | Terminated(Upon interim analys... more >>is, sufficient data was accrued to assess study hypotheses.) Collapse << | - | - |
| NCT00672386 | Diabetes Mellitus, Type 2 ... more >> Diabetes Mellitus Endocrine System Diseases Nutritional and Metabolic Diseases Collapse << | Phase 2 | Completed | - | - |
| NCT01488552 | Stage IV Pancreatic Cancer | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> TGen Clinical Research Services (TCRS) Scottsdale, Arizona, United States, 85258 United States, California Disney Family Cancer Center Burbank, California, United States, 91505 United States, Minnesota Virginia Piper Cancer Institute (VPCI) Minneapolis, Minnesota, United States, 55407 United States, Washington Virginia Mason Medical Center Seattle, Washington, United States, 98101 Evergreen Hematology and Oncology Spokane, Washington, United States, 99218 Collapse << |
| NCT01009580 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01513590 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01513590 | - | - | Completed | - | - |
| NCT00005654 | Hyperinsulinism ... more >> Polycystic Ovary Syndrome Collapse << | Not Applicable | Completed | - | - |
| NCT03199053 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 20, 2021 | - |
| NCT01351753 | Obesity Metab... more >>olic Syndrome X Collapse << | Phase 2 | Suspended(Change of staff) | October 2019 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52241 Collapse << |
| NCT01125150 | - | - | Completed | - | United States, Michigan ... more >> Department of Dermatology, NEW CENTER ONE Detroit, Michigan, United States, 48202 Collapse << |
| NCT03278158 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | December 2018 | Denmark ... more >> Odense University Hospital Recruiting Odense, Denmark, 5000 Contact: Henning Beck-Nielsen, Dr henning.beck-nielsen@rsyd.dk Collapse << |
| NCT00159211 | Type 2 Diabetes | Not Applicable | Terminated(inclusion was finis... more >>hed) Collapse << | - | France ... more >> Sce de Diabétologie, hôpital de la Pitié-salpêtrière, 83bld de l'hôpital Paris, France, 75013 Collapse << |
| NCT00449553 | - | - | Completed | - | Denmark ... more >> Multiple, Denmark Iceland Multiple, Iceland Norway Multiple, Norway Sweden Multiple, Sweden Collapse << |
| NCT00641407 | Type 2 Diabetes Mellitus ... more >> Hypoglycemia Collapse << | Phase 4 | Completed | - | Italy ... more >> Department of Geriatrics and Metabolic Disease Naples, Italy, 80138 Collapse << |
| NCT01241448 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01827280 | 1- Microvascular Function ... more >> 2-oxidative Stress 3-inflammation Collapse << | Phase 4 | Completed | - | Brazil ... more >> Laboratory for Clinical and Experimental Research on Vascular Biology Rio de Janeiro, Brazil, 20550-900 Collapse << |
| NCT01241448 | - | - | Completed | - | - |
| NCT01318564 | - | - | Completed | - | United States, Texas ... more >> Kelsey Seybold Clinics Houston, Texas, United States, 77025 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01884051 | - | - | Recruiting | July 2022 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00035984 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT00182494 | Diabetes Mellitus ... more >> Schizophrenia Collapse << | Phase 4 | Unknown | - | Canada, Ontario ... more >> McMaster University Recruiting Hamilton, Ontario, Canada Contact: Lakshmi P Voruganti, MD 905-522-1155 ext 6355 vorugl@mcmaster.ca Contact: Susan Strong, MSc 905 522 1155 strongs@mcmaster.ca Principal Investigator: Lakshmi P Voruganti, MD Collapse << |
| NCT01368081 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01368081 | - | - | Completed | - | - |
| NCT01656057 | To Assess the Impact of Bile A... more >>cids on Human Glukagon-like-peptide-1 Secretion Collapse << | Not Applicable | Completed | - | Denmark ... more >> University Hospital of Copenhagen, Gentofte Hospital, Diabetic Research Division Copenhagen, Hellerup, Denmark, 2900 Collapse << |
| NCT00713830 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Bulgaria Sanofi-Aventis Administrative Office Sofia, Bulgaria Czech Republic Sanofi-Aventis Administrative Office Praha, Czech Republic Egypt Sanofi-Aventis Administrative Office Cairo, Egypt Germany Sanofi-Aventis Administrative Office Berlin, Germany India Sanofi-Aventis Administrative Office Mumbai, India Israel Sanofi-Aventis Administrative Office Natanya, Israel Japan Sanofi-Aventis Administrative Office Tokyo, Japan Korea, Republic of Sanofi-Aventis Administrative Office Seoul, Korea, Republic of Netherlands Sanofi-Aventis Administrative Office Gouda, Netherlands Romania Sanofi-Aventis Administrative Office Bucuresti, Romania Russian Federation Sanofi-Aventis Administrative Office Moscow, Russian Federation Taiwan Sanofi-Aventis Administrative Office Taipei, Taiwan Thailand Sanofi-Aventis Administrative Office Bangkok, Thailand Tunisia Sanofi-Aventis Administrative Office Megrine, Tunisia Turkey Sanofi-Aventis Administrative Office Istanbul, Turkey Collapse << |
| NCT00518882 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00713830 | - | - | Completed | - | - |
| NCT00909597 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT00518882 | - | - | Completed | - | - |
| NCT00357370 | - | - | Completed | - | - |
| NCT00097877 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01420692 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | Turkey ... more >> Ankara University Medical Faculty Dept. of Endocrinology and Metabolism Ankara, Turkey, 06100 Collapse << |
| NCT03375580 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Not Applicable | Recruiting | December 31, 2019 | China, Guangdong ... more >> First Affiliated Hospital of Guangdong Pharmaceutical University Recruiting Guangzhou, Guangdong, China, 510080 Contact: Guobiao Huang 86-20-39352064 153706227@qq.com Collapse << |
| NCT03529123 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | August 30, 2019 | India ... more >> Investigational Site Number 01 Recruiting Bangalore, India, 560092 Investigational Site Number 012 Recruiting Belgaum, India, 590010 Investigational Site Number 017 Recruiting Coimbatore, India, 641009 Investigational Site Number 06 Recruiting Hyderabad, India, 500063 Investigational Site Number 07 Recruiting Hyderabad, India, 500072 Investigational Site Number 08 Recruiting Jaipur, India Investigational Site Number 04 Recruiting Kolkata, India, 700107 Investigational Site Number 018 Recruiting Lucknow, India Investigational Site Number 014 Recruiting Madurai, India, 625020 Investigational Site Number 05 Recruiting Nasik, India, 422002 Investigational Site Number 010 Recruiting New Delhi, India, 110029 Investigational Site Number 016 Recruiting Pune, India, 411040 Collapse << |
| NCT00005104 | Polycystic Ovary Syndrome ... more >> Hyperinsulinism Collapse << | Not Applicable | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294-3300 United States, Louisiana Louisiana State University School of Medicine Shreveport, Louisiana, United States, 71130-3932 United States, Missouri Washington University - St. Louis St. Louis, Missouri, United States, 63110 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908 Medical College of Virginia School of Medicine Richmond, Virginia, United States, 23298-0230 Italy University Alma Mater Bologna, Italy, 40100 Collapse << |
| NCT00357370 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | - |
| NCT01680978 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United Kingdom ... more >> Oxford, United Kingdom, OX3 9DU Collapse << |
| NCT00541567 | Obesity and Type 2 Diabetes | Phase 2 Phase 3 | Withdrawn | June 2010 | - |
| NCT03204799 | - | - | Recruiting | December 16, 2018 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT01069965 | Diabetes Mellitus | Phase 2 | Terminated(Funding support wit... more >>hdrawn.) Collapse << | - | - |
| NCT01956305 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Worldwide Clinical Trials San Antonio, Texas, United States, 78217 Collapse << |
| NCT01306214 | - | - | Completed | - | - |
| NCT01306214 | Diabetes Mellitus, Type 2 ... more >> Obesity Collapse << | Phase 3 | Completed | - | - |
| NCT01516476 | Diabetes Mellitus Type 2 | Phase 2 | Terminated | - | United States, Ohio ... more >> Cincinnati, Ohio, United States, 45227 Collapse << |
| NCT03655535 | Type2 Diabetes Mellitus | Phase 2 | Recruiting | August 30, 2019 | United States, California ... more >> Coastal Metabolic Research Center, Inc. Recruiting Ventura, California, United States, 93003 Contact: Ronald Chochinov, MD 805-658-8460 rchochinov@yahoo.com Contact: Sagrario Olmos-Garcia, MD sagy68@gmail.com United States, New Mexico Albuquerque Clinical Trials Recruiting Albuquerque, New Mexico, United States, 87102 Contact: Ellen Kim, MD 505-224-7407 ekim@abqct.com Contact: Jose Atencio JAtencio@abqct.com United States, South Carolina Coastal Carolina Research Center Recruiting Mount Pleasant, South Carolina, United States, 29464 Contact: Nathan Morton 843-352-6977 nmorton@coastalcarolinaresearch.com Contact: Yvonne Davis, MD ydavis@coastalcarolinaresearch.com United States, Utah Advanced Research Institute Recruiting Ogden, Utah, United States, 84405 Contact: Jack Wahlen, MD 801-409-2040 jwahlen@advresearch.org Contact: Jonathan Winkfield, PA-C jon@advresearch.org Collapse << |
| NCT02006836 | Diabetes | Not Applicable | Completed | - | China, Hubei ... more >> Tongji Hospital Wuhan, Hubei, China, 430030 Collapse << |
| NCT00121641 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01237314 | - | - | Completed | - | United States, Minnesota ... more >> International Diabetes Center Minneapolis, Minnesota, United States, 55416 Collapse << |
| NCT00121641 | - | - | Completed | - | - |
| NCT00997152 | Type II Diabetes Mellitus | Phase 2 | Terminated | - | - |
| NCT02246257 | Rheumatoid Arthritis ... more >> Cardiovascular Diseases Collapse << | Not Applicable | Recruiting | September 2020 | Denmark ... more >> Department of Rheumathology, Frederiksberg and Bispebjerg univeristy Hospital Recruiting Frederiksberg, Region Of Copenhagen, Denmark, 2000 Contact: Annemarie Lyng Svesson, MD, PhD 004528555126 lyng.annemarie@gmail.com Contact: Torkell J Ellingsen, MD, PhD, Professor 004565413523 torkell.ellingsen@rsyd.dk Collapse << |
| NCT00653185 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00767000 | - | - | Terminated | - | - |
| NCT00767000 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated | - | - |
| NCT03235050 | Diabetes Mellitus, Type 2 | Phase 2 | Active, not recruiting | March 9, 2020 | - |
| NCT00901979 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01364350 | - | - | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Colorado University of Colorado Denver Denver, Colorado, United States, 80262 United States, Connecticut Yale University New Haven, Connecticut, United States, 06519 United States, Maryland George Washington University Biostatistics Center Rockville, Maryland, United States, 20852 United States, Massachusetts Massachusetts General Hospital Diabetes Center Boston, Massachusetts, United States, 02114 Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Saint Louis University St Louis, Missouri, United States, 63104 Washington University St Louis, Missouri, United States, 63110 United States, New York Columbia University Medical Center New York City, New York, United States, 10032 State University of New York Upstate Medical University Syracuse, New York, United States, 13214 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Oklahoma University of Oklahoma Health Sciences Center Oklahoma City, Oklahoma, United States, 73117 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT02006836 | - | - | Completed | - | - |
| NCT02526810 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | September 2016 | China, Guangdong ... more >> Endocrinology department of the Third Affiliated Hospital of Sun Yet-san University Recruiting Guangzhou City, Guangdong, China, 510630 Contact: Zeng Longyi, Professor 0086-020-85252160 zssynfmk@163.com Contact: Lin Shuo, Doctor 0086-020-85253408 littltpig@yeah.net Principal Investigator: Zeng Longyi, Professor Sub-Investigator: lin Shuo, Doctor Collapse << |
| NCT00313313 | Diabetes | Phase 3 | Completed | - | - |
| NCT00425009 | Type 2 Diabetes | Phase 1 Phase 2 | Completed | - | China ... more >> Department of Endocrinology, Xinhua Hospital Affiliated to Shanghai Jiaotong University School of Medicine Shanghai, China, 200092 Collapse << |
| NCT00313313 | - | - | Completed | - | - |
| NCT00636766 | Carotid Atherosclerosis ... more >> Stroke Type 2 Diabetes Metabolic Syndrome Collapse << | Not Applicable | Completed | - | Greece ... more >> General Hospital of Thessaloniki "Hippokratio" Thessaloniki, Greece Collapse << |
| NCT00086502 | - | - | Completed | - | - |
| NCT02183935 | Obesity Diabe... more >>tes Metabolic Syndrome Collapse << | Phase 3 | Unknown | December 2016 | Slovenia ... more >> UMC Ljubljana Recruiting Ljubljana, Slovenia Contact: Tadej Battelino, PhD 00386 01 522 9235 tadej.battelino@mf.uni-lj.si Principal Investigator: Tadej Battelino, MD, PhD Sub-Investigator: Rok Orel, MD, PhD Sub-Investigator: Matjaž Homan, MD, PhD Sub-Investigator: Primož Kotnik, MD, PhD Collapse << |
| NCT03489317 | - | - | Recruiting | December 2021 | Hong Kong ... more >> Department of Microbiology, Chinese University of Hong Kong Recruiting Sha Tin, Hong Kong Contact: Yun Kit Yeoh 85235053333 yeohyunkit@cuhk.edu.hk Lek Yuen Health Centre Recruiting Sha Tin, Hong Kong Contact: Martin Wong 85222528782 wong_martin@cuhk.edu.hk Collapse << |
| NCT00290940 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00680745 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01283308 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | India ... more >> Madras Diabetes Research Foundation Chennai, Tamil Nadu, India, 600 086 Collapse << |
| NCT00680745 | - | - | Completed | - | - |
| NCT01845831 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory University Hospital Atlanta, Georgia, United States, 30324 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Pennsylvania Temple University Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT00806520 | - | - | Completed | - | United States, Massachusetts ... more >> Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Minnesota International Diabetes Center Minneapolis, Minnesota, United States, 55416 United States, Nevada Palm Medical Group Las Vegas, Nevada, United States, 89148 United States, North Carolina University of North Carolina Diabetes Care Center Durham, North Carolina, United States, 27713 United States, Washington Rockwood Clinic Spokane, Washington, United States, 99202 Collapse << |
| NCT00086502 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02973321 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01011868 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01845831 | - | - | Completed | - | - |
| NCT00696657 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00106704 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02946996 | Prostate Cancer | Phase 2 | Recruiting | March 1, 2020 | United States, South Carolina ... more >> Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Mike Wheeler 843-792-9321 hcc-clinical-trials@musc.edu Collapse << |
| NCT02879409 | Diabetes Mellitus Type 2 | Not Applicable | Recruiting | August 2019 | Qatar ... more >> Hamad Medical Corporation Recruiting Doha, Qatar, 3050 Contact: Ibrahim Janahi, MD 4439-2434 ext 00974 Imohd@hamad.qa Principal Investigator: Ibrahim Janahi, MD Sub-Investigator: Mashhood Siddique, MD Sub-Investigator: Hamda Ali, MD Collapse << |
| NCT01084005 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00094770 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00106704 | - | - | Completed | - | - |
| NCT01084005 | - | - | Completed | - | - |
| NCT00156897 | Non-Insulin-Dependent Diabetes... more >> Mellitus Obesity Collapse << | Phase 2 | Completed | - | Denmark ... more >> Aalborg Sygehus Nord Aalborg, Denmark Aarhus University Hospital Aarhus, Denmark, DK-8000 Bispebjerg Hospital Copenhagen, Denmark, DK-2400 Gentofte Hospital Hellerup, Denmark, DK-2900 Hvidovre Hospital Hvidovre, Denmark, DK-2650 Odense University Hospital Odense, Denmark Finland Lääkärikeskus Minerva Eura, Finland, 27510 Obesity Research Unit Helsinki, Finland, 00100 Suomen Terveystalo Jyvaskyla, Finland, 40100 Oy Foodfiles Limited Kuopio, Finland Oulun Diakonissalaitos Oulu, Finland, 90100 University of Oulu Oulu, Finland Turku University Hospital Turku, Finland, 20520 Netherlands Allevon Den Bosch Ziekenhuis, Netherlands Allevon DN de Bilt, Netherlands Zeikenhuisgroep Twente Hengelo, Netherlands Nederlandse Obesitas Klinik Hilversum, Netherlands Sint Franciscus Gasthuis Rotterdam, Netherlands Sweden Sahlgrenska University Hospital Goteborg, Sweden Linkoping University Hospital Linkoping, Sweden Karolinsaka University Hospital Stockholm, Sweden Norrlands Universitetssjukhus Umea, Sweden Samariterhemmets Sjukhus Uppsala, Sweden United Kingdom Aberdeen Royal Infirmary Aberdeen, United Kingdom Royal United Hospital Bath, United Kingdom Clinical Research Centre Edgbaston Birmingham, United Kingdom Walsgrave Hospital Coventry, United Kingdom Glasgow Royal Infirmary Glasgow, United Kingdom Clinical Research Centre, Crosby Liverpool, United Kingdom Liverpool University Hospital Liverpool, United Kingdom Queen Mary's School of Medicine & Dentistry London, United Kingdom, E1 2AA Hammersmith Hospital London, United Kingdom Luton & Dunstable Hospital Luton, United Kingdom Clinical Research Centre Manchester, United Kingdom James Cook University Hospital Middlesborough, United Kingdom Royal Shrewsbury Hospital Shrewsbury, United Kingdom Clinical Research Centre Wigan, United Kingdom Collapse << |
| NCT01096940 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Research Site San Antonio, Texas, United States Collapse << |
| NCT00087516 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00094770 | - | - | Completed | - | - |
| NCT01011868 | - | - | Completed | - | - |
| NCT01175824 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Argentina ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mar Del Plata, Argentina, B7600FZN For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ramos Mejia, Argentina, B1704ETD Brazil For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Porto Alegre, Brazil, 90035-170 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. São Paulo, Brazil, 01244-030 China For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changzhou, China, 213003 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150086 Egypt For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alexandria, Egypt For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cairo, Egypt, 11562 India For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jaipur, India, 302018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vishakhapatnam, India, 530002 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daegu, Korea, Republic of, 700-712 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Goyang-Si, Korea, Republic of, 410-719 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyunggi-Do, Korea, Republic of, 425-020 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 134-090 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Málaga, Spain, 29010 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Boi De Llobregat, Spain, 08830 Turkey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Antalya, Turkey, 07070 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Istanbul, Turkey, 34865 Collapse << |
| NCT01175824 | - | - | Completed | - | - |
| NCT00802100 | Schizophrenia | Phase 4 | Completed | - | United States, California ... more >> SHANTI Clinical Trials Colton, California, United States, 92324 Stanford University Palo Alto, California, United States, 94305 United States, Connecticut Yale University New Haven, Connecticut, United States, 06519 United States, Florida University of Miami School of Medicine Miami, Florida, United States, 33316 United States, Georgia Medical College of Georgia Augusta, Georgia, United States, 30912 United States, Maryland Clinical Insights Glen Burnie, Maryland, United States, 21061 United States, Massachusetts University of Massachusetts Worcester, Massachusetts, United States, 01605 United States, Michigan Wayne State University Detroit, Michigan, United States, 48201 United States, Minnesota University of Minnesota School of Medicine Minneapolis, Minnesota, United States, 55454 United States, New York Research Foundation for Mental Hygiene New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center-John Umstead Hospital Butner, North Carolina, United States, 27509 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75235 Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00087516 | - | - | Completed | - | - |
| NCT00696657 | - | - | Completed | - | - |
| NCT00802100 | - | - | Completed | - | - |
| NCT02106104 | Type 2 Diabetes | Phase 4 | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT03225209 | Type 2 Diabetes Mellitus | Phase 2 | Active, not recruiting | August 15, 2018 | United States, North Carolina ... more >> Department of Veteran Affairs, Greenville Health Care Center Greenville, North Carolina, United States, 27834 Collapse << |
| NCT02043054 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> University Hospitals of Leicester NHS Trust, Diabetes Research Centre Leicester, Leicestershire, United Kingdom, LE5 4PW Collapse << |
| NCT01338870 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01664624 | - | - | Completed | - | - |
| NCT01103609 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT01338870 | - | - | Completed | - | - |
| NCT01664624 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Chula Vista, California, United States United States, Florida Orlando, Florida, United States Collapse << |
| NCT01119846 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Phoenix, Arizona, United States, 85013 United States, California GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio GSK Investigational Site Columbus, Ohio, United States, 43212 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 United States, Washington GSK Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT01083212 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Sweden ... more >> Reserach Site Linkoping, Sweden Research Site Lulea, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT01119846 | - | - | Completed | - | - |
| NCT00929539 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03640221 | Hypertension ... more >>Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Not yet recruiting | August 31, 2020 | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States, 90048 Collapse << |
| NCT01336738 | - | - | Completed | - | - |
| NCT01103622 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT01475461 | - | - | Completed | - | - |
| NCT01298518 | Type 2 Diabetes Patients | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site DeLand, Florida, United States, 32720 Pfizer Investigational Site Miami Gardens, Florida, United States, 33169 Collapse << |
| NCT01475461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01336738 | Diabetes, Type 2 | Phase 2 | Completed | - | - |
| NCT00924534 | Type 2 Diabetes | Phase 2 | Completed | - | Bulgaria ... more >> Site Reference ID/Investigator# 54183 Dimitrovgrad, Bulgaria, 6400 Site Reference ID/Investigator# 54182 Pleven, Bulgaria, 5800 Site Reference ID/Investigator# 44722 Plovdiv, Bulgaria, 4000 Site Reference ID/Investigator# 44723 Sofia, Bulgaria, 1407 Poland Site Reference ID/Investigator# 44725 Lubin, Poland, 59-301 Site Reference ID/Investigator# 44724 Pulawy, Poland, 24-100 Site Reference ID/Investigator# 54185 Radzymin, Poland, 05-250 Site Reference ID/Investigator# 44727 Ruda Slaska, Poland Site Reference ID/Investigator# 54184 Wroclaw, Poland, 50-349 South Africa Site Reference ID/Investigator# 44728 Cape Town, South Africa, 7130 Site Reference ID/Investigator# 44730 Cape Town, South Africa, 7937 Site Reference ID/Investigator# 44729 Johannesburg, South Africa, 2198 Collapse << |
| NCT01298518 | - | - | Completed | - | - |
| NCT01232946 | Type 2 Diabetes | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana Clinical Research Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT02000700 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Arkansas ... more >> Little Rock, Arkansas, United States United States, California Los Angeles, California, United States United States, Colorado Aurora, Colorado, United States United States, Connecticut New Haven, Connecticut, United States United States, Florida Jacksonville, Florida, United States United States, Indiana Indianapolis, Indiana, United States United States, Kansas Lenexa, Kansas, United States United States, Maryland Baltimore, Maryland, United States United States, Missouri Kansas City, Missouri, United States United States, Ohio Toledo, Ohio, United States United States, Pennsylvania Pittsburgh, Pennsylvania, United States United States, Texas Houston, Texas, United States United States, Utah Salt Lake City, Utah, United States Brazil Campinas, Brazil Curitiba, Brazil Ribeirão Preto, Brazil Sao Paulo, Brazil Collapse << |
| NCT01105429 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute For Clinical Research, Inc. (Picr) Chula Vista, California, United States, 91911 United States, Florida Clinical Pharmacology Of Miami Inc. Miami, Florida, United States, 33014 Elite Research Institute Miami, Florida, United States, 33169 Collapse << |
| NCT00241423 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, District of Col... more >>umbia Research Site Washington, District of Columbia, United States United States, Texas Research Site San Antonio, Texas, United States Collapse << |
| NCT00942188 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Alabama ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mobile, Alabama, United States, 36689 United States, California For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Anaheim, California, United States, 92801 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chula Vista, California, United States, 91911 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. La Mesa, California, United States, 91942 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Walnut Creek, California, United States, 94598 United States, District of Columbia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Washington, District of Columbia, United States, 20003 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miami Gardens, Florida, United States, 33169 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miami, Florida, United States, 33169 United States, Idaho For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Boise, Idaho, United States, 83702 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21287 United States, Michigan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dearborn, Michigan, United States, 48126 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Philadelphia, Pennsylvania, United States, 19107 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Arlington, Texas, United States, 76014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dallas, Texas, United States, 75230 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tomball, Texas, United States, 77375 United States, Utah For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Salt Lake City, Utah, United States, 84107 United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Richmond, Virginia, United States, 23294 Collapse << |
| NCT02286128 | - | - | Completed | - | Thailand ... more >> Mattabhorn Phornputkul Chiang Mai, Thailand, 50200 Collapse << |
| NCT02405260 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Arizona ... more >> Mesa, Arizona, United States, 85213 Tuscon, Arizona, United States, 85741 United States, California Canoga Park, California, United States, 91303 Huntington Park, California, United States, 90255 Los Angeles, California, United States, 90056 West Hills, California, United States, 91307 United States, Colorado Centennial, Colorado, United States, 80112 Littleton, Colorado, United States, 80127 United States, Connecticut Waterbury, Connecticut, United States, 06708 United States, Florida West Palm Beach, Florida, United States, 33409 United States, Illinois Gurnee, Illinois, United States, 60031 United States, Indiana Evansville, Indiana, United States, 47725 United States, Minnesota Rochester, Minnesota, United States, 55905 United States, North Carolina Greensboro, North Carolina, United States, 27408 United States, South Carolina Moncks Corner, South Carolina, United States, 29461 United States, Texas San Antonio, Texas, United States, 78209 San Antonio, Texas, United States, 78229 Schertz, Texas, United States, 78154 United States, Virginia Norfolk, Virginia, United States, 23507 United States, Washington Richland, Washington, United States, 99352 Collapse << |
| NCT02081118 | Type 2 Diabetes | Phase 2 | Completed | - | United States, Florida ... more >> Hanmi pharmaceutical Miami, Florida, United States Collapse << |
| NCT01889667 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | United States, California ... more >> Orange County Research Center Tustin, California, United States, 92780 Collapse << |
| NCT01665352 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, California ... more >> Site 18 Chula Vista, California, United States, 91911 Site 1 Los Angeles, California, United States, 90057 United States, Colorado Site 13 Denver, Colorado, United States, 80220 United States, Florida Site 4 Coral Gables, Florida, United States, 33134 Site 5 Port Orange, Florida, United States, 32127 United States, Hawaii Site 3 Honolulu, Hawaii, United States, 96814 United States, Indiana Site 17 Indianapolis, Indiana, United States, 46254 United States, Kentucky Site 7 Louisville, Kentucky, United States, 40213 United States, Maine Site 8 Auburn, Maine, United States, 04210 United States, Nevada Site 9 Las Vegas, Nevada, United States, 89101 United States, New Jersey Site 26 Trenton, New Jersey, United States, 08611 United States, New York Site 12 Rochester, New York, United States, 14609 United States, Ohio Site 14 Cincinnati, Ohio, United States, 45219 United States, South Carolina Site 6 Mount Pleasant, South Carolina, United States, 29464 United States, Texas Site 16 Dallas, Texas, United States, 75230 Site 10 Houston, Texas, United States, 77072 Site 2 Houston, Texas, United States, 77074 Site 11 Katy, Texas, United States, 77450 United States, Virginia Site 15 Richmond, Virginia, United States, 23294 Collapse << |
| NCT02206607 | - | - | Completed | - | - |
| NCT01249677 | Type 2 Diabetes | Phase 4 | Completed | - | Germany ... more >> Department of Medicine I; University Hospital St. Josef Hospital Bochum, Germany Collapse << |
| NCT01290575 | Type 2 Diabetes | Phase 1 | Completed | - | United States, Arizona ... more >> Dedicated Phase I, Inc. Phoenix, Arizona, United States, 85013 United States, Arkansas Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, Florida Clinical Pharmacology Of Miami Inc. Miami, Florida, United States, 33014 Mra Clinical Research Miami, Florida, United States, 33143 United States, Texas Cetero Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT00359762 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00942188 | - | - | Completed | - | - |
| NCT02206607 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, North Carolina ... more >> High Point Clinical Trials Center High Point, North Carolina, United States, 27265 United States, Texas Clinical Trials of Texas, Inc. San Antonio, Texas, United States, 78229 Collapse << |
| NCT01889667 | - | - | Completed | - | - |
| NCT01024244 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | United States, Kansas ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Topeka, Kansas, United States, 66606 United States, New Jersey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Toms River, New Jersey, United States, 08753 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Syracuse, New York, United States, 13210 Australia, New South Wales For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. St. Leonards, New South Wales, Australia, 2065 Australia, South Australia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Daw Park, South Australia, Australia, 5041 Australia, Victoria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Box Hill, Victoria, Australia, 3128 Puerto Rico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Manati, Puerto Rico, 00674 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Juan, Puerto Rico, 00907 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Arkhangelsk, Russian Federation, 163045 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 119435 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rostov-On-Don, Russian Federation, 344022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 194354 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alcira, Spain, 46600 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alicante, Spain, 03114 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dos Hermanas, Spain, 41014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Santa Cruz De Tenerife, Spain, 38320 Collapse << |
| NCT00359762 | - | - | Completed | - | - |
| NCT01082120 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT01029795 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | Australia, South Australia ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Elizabeth Vale, South Australia, Australia, 5112 Australia, Victoria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. East Ringwood, Victoria, Australia, 3135 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Heidelberg, Victoria, Australia, 3081 Austria For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vienna, Austria, 1090 Czech Republic For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Holesov, Czech Republic, 769 01 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Prague, Czech Republic, 140 59 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55116 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Neuss, Germany, D-41460 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jerusalem, Israel, 91120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nahariya, Israel, 22100 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rostov-On-Don, Russian Federation, 344022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saint Petersburg, Russian Federation, 191126 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cordoba, Spain, 14004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dos Hermanas, Spain, 41014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Teruel, Spain, 44002 Collapse << |
| NCT01095991 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Germany ... more >> Research Site Neuss, Germany Collapse << |
| NCT01003184 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Ireland ... more >> Research Site Dublin, Ireland United Kingdom Research Site Addlestone, England, United Kingdom Research Site Bath, England, United Kingdom Research Site Birmingham, England, United Kingdom Research Site Blackburn, England, United Kingdom Research Site Bournemouth, England, United Kingdom Research Site Chippenham, England, United Kingdom Research Site Derby, England, United Kingdom Research Site Exeter, England, United Kingdom Research Site High Wycombe, England, United Kingdom Research Site Hull, England, United Kingdom Research Site Leicester, England, United Kingdom Research Site Liverpool, England, United Kingdom Research Site London, England, United Kingdom Research Site Manchester, England, United Kingdom Research Site Merseyside, England, United Kingdom Research Site Middlesborough, England, United Kingdom Research Site Newcastle, England, United Kingdom Research Site Northampton, England, United Kingdom Research Site Oldham, England, United Kingdom Research Site Plymouth, England, United Kingdom Research Site Portsmouth, England, United Kingdom Research Site Sheffield, England, United Kingdom Research Site Stevenage, England, United Kingdom Research Site Suffolk, England, United Kingdom Research Site Wakefield, England, United Kingdom Research Site Wiltshire, England, United Kingdom Research Site Aberdeen, Scotland, United Kingdom Research Site Dundee, Scotland, United Kingdom Research Site Carmathen, Wales, United Kingdom Research Site Swansea, Wales, United Kingdom Research Site Wrexham, Wales, United Kingdom Research Site Leytonstone, United Kingdom Research Site Livingston, United Kingdom Collapse << |
| NCT02653209 | Type 2 Diabetes | Phase 4 | Recruiting | December 2019 | United Kingdom ... more >> Exeter Clinical Research Facility Recruiting Exeter, United Kingdom, EX2 5DW Contact: Catherine Angwin 01392408185 C.Angwin@exeter.ac.uk Contact: Andrew Hattersley +441392 408231 A.T.Hattersley@exeter.ac.uk Principal Investigator: Andrew Hattersley Collapse << |
| NCT01003184 | - | - | Completed | - | - |
| NCT01024244 | - | - | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | - |
| NCT00203632 | Diabetes Mellitus | Phase 3 | Terminated(Terminated at 50% e... more >>nrollment due to recent concerns about rosiglitazone) Collapse << | - | Canada, Alberta ... more >> Foothills Medical Centre Calgary, Alberta, Canada, T2N 2T9 Collapse << |
| NCT01408095 | Diabetes Mellitus, Type 2 | Phase 2 | Withdrawn(Trial terminated no ... more >>patients were screened or enrolled) Collapse << | September 2012 | - |
| NCT01784380 | - | - | Completed | - | China, Guangdong ... more >> Elecsys2010,French KONTRON EUB-5500 color Doppler ultrasonic diagnostic apparatus ZHONGSHAN City, Guangdong, China, 528400 Reproductive Center of Bo Ai Hospital, Zhongshan Zhongshan, Guangdong, China, 528403 Collapse << |
| NCT02097342 | Type 2 Diabetes Mellitus ... more >> Insulin Sensitivity/Resistance Collapse << | Phase 4 | Unknown | January 2015 | India ... more >> Postgraduate Institute of Medical Education & Research Recruiting Chandigarh, India, 160012 Contact: Anil Bhansali, MD DM anilbhansaliendocrine@gmail.com Collapse << |
| NCT03167918 | Type 2 Diabetes Mellitus ... more >> Pulmonary Function Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT01029795 | - | - | Terminated(Terminated due to n... more >>onclinical safety findings) Collapse << | - | - |
| NCT01811485 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Chikushino-city, Fukuoka, Japan, 818-0083 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 810-0014 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 819-0006 Novartis Investigative Site Iizuka-city, Fukuoka, Japan, 820-8505 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 800-0296 Novartis Investigative Site Kitakyushu-city, Fukuoka, Japan, 807-0857 Novartis Investigative Site Koga-city, Ibaraki, Japan, 306-0232 Novartis Investigative Site Kawasaki-city, Kanagawa, Japan, 210-0852 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0802 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 231-0023 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8533 Novartis Investigative Site Yatsushiro-city, Kumamoto, Japan, 866-8660 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 607-8062 Novartis Investigative Site Kyoto-city, Kyoto, Japan, 615-0035 Novartis Investigative Site Sakai-city, Osaka, Japan, 590-0064 Novartis Investigative Site Takatsuki-city, Osaka, Japan, 569-1096 Novartis Investigative Site Ageo-city, Saitama, Japan, 362-8588 Novartis Investigative Site Tokorozawa-city, Saitama, Japan, 359-1161 Novartis Investigative Site Bunkyo-ku, Tokyo, Japan, 113-0031 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji-city, Tokyo, Japan, 192-0918 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Katsushika-ku, Tokyo, Japan, 124-0024 Novartis Investigative Site Kiyose-city, Tokyo, Japan, 204-0021 Novartis Investigative Site Minato-ku, Tokyo, Japan, 105-7390 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Nerima-ku, Tokyo, Japan, 177-0051 Novartis Investigative Site Shibuya-ku, Tokyo, Japan, 150-0002 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Collapse << |
| NCT02145611 | Type 2 Diabetes Mellitus ... more >> Hypertension Endothelial Dysfunction Collapse << | Phase 4 | Completed | - | Brazil ... more >> Fundação Faculdade Regional de Medicina de São José do Rio Preto São José do Rio Preto, São Paulo, Brazil, 15090-000 Collapse << |
| NCT01811485 | - | - | Completed | - | - |
| NCT01087567 | Type 2 Diabetes | Phase 4 | Completed | - | United States, Ohio ... more >> Ohio University Athens, Ohio, United States, 45701 Collapse << |
| NCT03038789 | - | - | Recruiting | January 31, 2020 | Korea, Republic of ... more >> Research Site Recruiting Busan, Korea, Republic of Research Site Recruiting ChungCheong, Korea, Republic of Research Site Recruiting DaeGu, Korea, Republic of Research Site Recruiting DaeJeon, Korea, Republic of Research Site Recruiting GyeongGi, Korea, Republic of Research Site Active, not recruiting GyeongGi, Korea, Republic of Research Site Recruiting Seoul, Korea, Republic of Research Site Withdrawn Seoul, Korea, Republic of Research Site Recruiting WonJu, Korea, Republic of Collapse << |
| NCT00162305 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> Local Institution Fort Lauderdale, Florida, United States, 33301 Local Institution Miami, Florida, United States, 33169 Local Institution Orlando, Florida, United States, 32809 United States, Louisiana Local Institution New Orleans, Louisiana, United States, 70119 United States, Texas Local Institution San Antonio, Texas, United States, 78229-3894 Collapse << |
| NCT03737240 | Diabetes Mellitus | Phase 3 | Not yet recruiting | December 2021 | United States, Georgia ... more >> Grady Health System Not yet recruiting Atlanta, Georgia, United States, 30303 Contact: Rodolfo Galindo, MD 404-251-8961 rodolfo.galindo@emory.edu Emory Clinic Not yet recruiting Atlanta, Georgia, United States, 30322 Contact: Rodolfo Galindo, MD 404-251-8961 rodolfo.galindo@emory.edu Collapse << |
| NCT00464594 | Diabetes Mellitus | Not Applicable | Unknown | - | Japan ... more >> Tokyo Medical and Dental University Recruiting Tokyo, Japan, 113-8519 Contact 03-3813-6111 Collapse << |
| NCT02470039 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Germany ... more >> Novo Nordisk Investigational Site Mainz, Germany, 55116 Novo Nordisk Investigational Site Neuss, Germany, 41460 Collapse << |
| NCT01465113 | Short Segment Barrett's Esopha... more >>gus Long Segment Barrett's Esophagus Collapse << | Early Phase 1 | Completed | - | United States, Ohio ... more >> University Hospitals Ahuja Medical Center Beachwood, Ohio, United States, 44122 Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02420262 | - | - | Completed | - | - |
| NCT01754142 | - | - | Completed | - | Korea, Republic of ... more >> Research Site Seoul, Korea, Republic of Collapse << |
| NCT02420262 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03674385 | Clomiphene Citrate Resistant P... more >>olycystic Ovary Syndrome Collapse << | Phase 2 | Recruiting | January 20, 2019 | Egypt ... more >> Medical Center of Infertility Recruiting Beni-suef, Egypt, 62521 Contact: eman megahed, M.D-PHD 01095115944 dr_emanmojahed@hotmail.com Collapse << |
| NCT00883766 | Polycystic Ovary Syndrome ... more >> In Vitro Fertilization Collapse << | Not Applicable | Terminated | - | Spain ... more >> Institut Universitari Dexeus Barcelona, Spain, 08028 Collapse << |
| NCT00063635 | Fatty Liver | Phase 3 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94143 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 United States, Missouri St. Louis University St. Louis, Missouri, United States, 63110 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44109 United States, Texas Texas Children's Hospital Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT02061969 | Diabetes | Phase 4 | Completed | - | United States, Georgia ... more >> Crestvew Nursing Home Atlanta, Georgia, United States, 30315 Budd Terrace Nursing Home Atlanta, Georgia, United States, 30322 Wesley Woods Nursing Home Atlanta, Georgia, United States, 30322 VA Nursing Home Decatur, Georgia, United States, 30033 Collapse << |
| NCT01117350 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT00135096 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | United States, New Jersey ... more >> Sanofi-aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT00269061 | Non-Insulin-Dependent Diabetes... more >> Mellitus Collapse << | Phase 1 | Completed | - | United States, Massachusetts ... more >> GSK Investigational Site Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT00063635 | - | - | Completed | - | - |
| NCT01117350 | - | - | Completed | - | - |
| NCT00481663 | Type II Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02053116 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(On 25April2014, stu... more >>dy was terminated before any dosing due to an AE of safety concern that occurred in protocol B1731003 with the same drug.) Collapse << | - | United States, Florida ... more >> Pfizer Investigational Site DeLand, Florida, United States, 32720 Collapse << |
| NCT02061969 | - | - | Completed | - | - |
| NCT02597049 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02607410 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Brazil ... more >> University of Sao Paulo General Hospital Sao Paulo, Brazil, 05403-000 Collapse << |
| NCT02597049 | - | - | Completed | - | - |
| NCT01426737 | Type 2 Diabetes | Phase 4 | Unknown | January 2013 | Switzerland ... more >> Universitiy Hospital Recruiting Zurich, Switzerland Collapse << |
| NCT01095653 | - | - | Completed | - | - |
| NCT01095653 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03772262 | Interaction Drug Food | Early Phase 1 | Recruiting | March 31, 2019 | United States, Washington ... more >> Washington State University Recruiting Spokane, Washington, United States, 99202 Contact: Malathi Jandhyala 509-335-3668 irb@wsu.edu Collapse << |
| NCT00853151 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | - |
| NCT03763240 | Blood Glucose, High | Not Applicable | Recruiting | December 1, 2020 | Sweden ... more >> Gothia Forum Recruiting Gothenburg, Sweden, 41345 Contact: Helen Svanström, Nurse 031-3439919 rekrytering.ctc.su@vgregion.se Collapse << |
| NCT00853151 | - | - | Completed | - | - |
| NCT00280046 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Russian Federation ... more >> Novo Nordisk Investigational Site Arkhangelsk, Russian Federation, 163045 Novo Nordisk Investigational Site Belgorod, Russian Federation, 308007 Novo Nordisk Investigational Site Ekaterinburg, Russian Federation, 620102 Novo Nordisk Investigational Site Kazan, Russian Federation, 420064 Novo Nordisk Investigational Site Khabarovsk, Russian Federation, 680067 Novo Nordisk Investigational Site Nizhniy Novgorod, Russian Federation, 603126 Novo Nordisk Investigational Site Novosibirsk, Russian Federation, 630090 Novo Nordisk Investigational Site Omsk, Russian Federation, 644070 Novo Nordisk Investigational Site Saint-Petersburg, Russian Federation, 194291 Novo Nordisk Investigational Site Vladivostok, Russian Federation, 690105 Novo Nordisk Investigational Site Voronezh, Russian Federation, 394053 Collapse << |
| NCT00934219 | Hypertriglyceridemia | Phase 4 | Unknown | December 2012 | United States, Ohio ... more >> Jewish Hospital Cholesterol Center Recruiting Cincinnati, Ohio, United States, 45229 Collapse << |
| NCT00032474 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> University of Texas Health Sciences Center San Antonio, Texas, United States, 78229 Collapse << |
| NCT01068665 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01068665 | - | - | Completed | - | - |
| NCT00511667 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | - |
| NCT01608724 | - | - | Completed | - | - |
| NCT02695121 | - | - | Recruiting | April 30, 2024 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States Netherlands Research Site Recruiting Utrecht, Netherlands United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT01076647 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01608724 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 4 | Completed | - | China ... more >> Research Site Baotou, China Research Site Beijing, China Research Site Benxi, China Research Site Changchun, China Research Site Chengdu, China Research Site Chongqing, China Research Site Dalian, China Research Site Dongguan, China Research Site Foshan, China Research Site Fuzhou, China Research Site Guangzhou, China Research Site Guiyang, China Research Site Haikou, China Research Site Hangzhou, China Research Site Hhht, China Research Site Hubei, China Research Site Jimo, China Research Site Jinan, China Research Site Kunming, China Research Site Linyi, China Research Site Nanjing, China Research Site Nantong, China Research Site Qingdao, China Research Site Shanghai, China Research Site Shantou, China Research Site Shenyang, China Research Site Shiyan, China Research Site Suzhou, China Research Site Taiyuan, China Research Site Tianjin, China Research Site Urumqi, China Research Site Wenzhou, China Research Site Wuhan, China Research Site Wuxi, China Research Site Xian, China Research Site Zhanjiang, China Research Site Zhengzhou, China Collapse << |
| NCT00500955 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00511667 | - | - | Completed | - | - |
| NCT01076647 | - | - | Completed | - | - |
| NCT01068678 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01068678 | - | - | Completed | - | - |
| NCT01046110 | - | - | Completed | - | - |
| NCT01046110 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01326026 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00871572 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Buena Park, California, United States, 90620 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Huntington Park, California, United States, 90255 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Westlake Village, California, United States, 91361 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fort Lauderdale, Florida, United States, 33306 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jupiter, Florida, United States, 33458 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New Port Richey, Florida, United States, 34652 United States, Idaho For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Idaho Falls, Idaho, United States, 83404 United States, Kansas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Topeka, Kansas, United States, 66606 United States, Kentucky For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Madisonville, Kentucky, United States, 42431 United States, Nevada For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Las Vegas, Nevada, United States, 89101 United States, South Dakota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rapid City, South Dakota, United States, 57702 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Norfolk, Virginia, United States, 23502 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aschaffenburg, Germany, 63739 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55116 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Munchen, Germany, 80336 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saarlouis, Germany, 66740 Lithuania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kaunas, Lithuania, LT-51270 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vilnius, Lithuania, LT-08661 Collapse << |
| NCT00274274 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Germany ... more >> Düsseldorf, Germany Collapse << |
| NCT01326026 | - | - | Completed | - | - |
| NCT01365507 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01365507 | - | - | Completed | - | - |
| NCT00789750 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00789750 | - | - | Completed | - | - |
| NCT00871572 | - | - | Completed | - | - |
| NCT01159249 | Type 2 Diabetes | Phase 3 | Completed | - | Japan ... more >> Novartis Investigative Site Hunabashi, Chiba, Japan, 274-0805 Novartis Investigative Site Chikushino, Fukuoka, Japan, 818-0036 Novartis Investigative Site Itoshima-shi, Fukuoka, Japan, 819-1102 Novartis Investigative Site Koriyama, Fukushima, Japan, 963-8851 Novartis Investigative Site Kobe, Hyogo, Japan, 658-0064 Novartis Investigative Site Kawasaki, Kanagawa, Japan, 210-0852 Novartis Investigative Site Kawasaki, Kanagawa, Japan, 212-0024 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 221-0077 Novartis Investigative Site Yokohama, Kanagawa, Japan, 221-0065 Novartis Investigative Site Izumisano, Osaka, Japan, 598-0048 Novartis Investigative Site Hannou, Saitama, Japan, 357-0024 Novartis Investigative Site Hiki-Gun, Saitama, Japan, 355-0328 Novartis Investigative Site Kawaguchi, Saitama, Japan, 332-0012 Novartis Investigative Site Koshigaya city, Saitama, Japan, 343-0826 Novartis Investigative Site Tokorozawa, Saitama, Japan, 359-1161 Novartis Investigative Site Edogawa-ku, Tokyo, Japan, 134-0084 Novartis Investigative Site Hachioji, Tokyo, Japan, 192-0046 Novartis Investigative Site Katsushika-ku, Tokyo, Japan, 125-0041 Novartis Investigative Site Minato-ku, Tokyo, Japan, 108-0075 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141-0032 Novartis Investigative Site Toshima-ku, Tokyo, Japan, 171-0021 Novartis Investigative Site Fukuoka, Japan, 807-0857 Novartis Investigative Site Fukuoka, Japan, 816-0094 Novartis Investigative Site Fukuoka, Japan, 819-0168 Collapse << |
| NCT00620282 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | United States, Minnesota ... more >> Novo Nordisk Investigational Site Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00620282 | - | - | Completed | - | - |
| NCT01068860 | Type 2 Diabetes Mellitus ... more >> Impaired Glucose Tolerance Collapse << | Phase 2 | Completed | - | - |
| NCT01068860 | - | - | Completed | - | - |
| NCT00482950 | Type 2 Diabetes Mellitus | Phase 2 | Unknown | February 2008 | - |
| NCT00766441 | Diabetes Hypo... more >>glycemia Collapse << | Phase 4 | Terminated(inadequate recruitm... more >>ent) Collapse << | - | United Kingdom ... more >> Wellcome Trust Clinical Research Facility Manchester, United Kingdom, M13 9NT Collapse << |
| NCT00952198 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research Chula Vista, California, United States, 91911 United States, Texas Cetero San Antonio, Texas, United States, 78229 Collapse << |
| NCT01059812 | - | - | Completed | - | - |
| NCT01970033 | Type 2 Diabetes | Phase 3 | Unknown | January 2015 | China ... more >> Chinese PLA General Hospital Recruiting Beijing, China Contact: Changyu Pan, M.D. panchy301@aliyun.com Principal Investigator: Changyu Pan, M.D. Collapse << |
| NCT00747383 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United Kingdom ... more >> Royal Bournemouth Hospital Bournemouth, Dorset, United Kingdom, BH7 7DW Collapse << |
| NCT00862875 | Type 2 Diabetes | Phase 4 | Completed | - | Canada, Quebec ... more >> Institut de Recherches Cliniques de Montréal Montréal, Quebec, Canada, H2W 1R7 Collapse << |
| NCT01059812 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00658021 | Type 2 Diabetes | Phase 3 | Recruiting | March 6, 2023 | - |
| NCT03164187 | - | - | Completed | - | Azerbaijan ... more >> Azerbaijan Republic Ministry of Health Republic Endocrinology Center Baku, Azerbaijan, AZ 1/122 Azer-Turk Med clinic Baku, Azerbaijan, AZ 1025 Hospital of oil workers, polyclinics and private clinics Baku, Azerbaijan, AZ 1025 Medical Clinic of Azerbaijan Medical University Baku, Azerbaijan, AZ-1078 Collapse << |
| NCT01495013 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Bucheon, Korea, Republic of, 134 727 GSK Investigational Site Busan, Korea, Republic of, 602-739 GSK Investigational Site Kangwondo, Korea, Republic of, 220-701 GSK Investigational Site Seocho-ku, Seoul, Korea, Republic of, 137-701 GSK Investigational Site Seongnam-si Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 110-746 GSK Investigational Site Seoul, Korea, Republic of, 135-710 GSK Investigational Site Seoul, Korea, Republic of, 138-736 GSK Investigational Site Seoul, Korea, Republic of, 139-872 GSK Investigational Site Seoul, Korea, Republic of, 152-703 GSK Investigational Site Suwon, Korea, Republic of, 463442 Malaysia GSK Investigational Site Ipoh, Malaysia, 30450 GSK Investigational Site Johor Bahru, Malaysia, 80100 GSK Investigational Site Kuala Lumpur, Malaysia, 59100 GSK Investigational Site Kubang Kerian, Malaysia, 16150 Mexico GSK Investigational Site Cuernavaca, Mexico, 62250 GSK Investigational Site Del. Cuauhtémoc, Mexico, 06700 GSK Investigational Site Gadalajara, Jalisco, Mexico, C.P. 44130 GSK Investigational Site Guadalajara Jalisco, Mexico, C.P. 44210 GSK Investigational Site Mexico City, Mexico, 06700 GSK Investigational Site Mexico, D.F., Mexico, 11650 Philippines GSK Investigational Site Cebu City, Philippines, 6000 GSK Investigational Site Davao City, Philippines, 8000 GSK Investigational Site Manila, Philippines, 1008 GSK Investigational Site Pasig City, Philippines, 1600 GSK Investigational Site Sta. Cruz, Manila, Philippines, 1012 Russian Federation GSK Investigational Site Moscow, Russian Federation, 109240 GSK Investigational Site Saratov, Russian Federation, 410054 GSK Investigational Site St. Petersburg, Russian Federation, 191119 Thailand GSK Investigational Site Chiangrai, Thailand, 57000 GSK Investigational Site Nakhon Ratchasima, Thailand, 30000 GSK Investigational Site Rajathevee, Thailand, 10400 Collapse << |
| NCT00479986 | Type 2 Diabetes Mellitus ... more >> Cardiovascular Disease Collapse << | Phase 4 | Completed | - | Germany ... more >> Dr. Michael Morcos Heidelberg, Germany IKFE Mainz, Germany, 55116 Collapse << |
| NCT00679237 | Coronary Heart Disease | Not Applicable | Active, not recruiting | December 2024 | Norway ... more >> Sorlandet Hospital Arendal, Norway Collapse << |
| NCT00062764 | - | - | Completed | - | - |
| NCT00062764 | Hepatitis | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02695173 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT01755442 | Diabetes Mellitus | Phase 1 | Terminated(Amgen determined no... more >> further need for this study.) Collapse << | - | United States, California ... more >> Research Site San Diego, California, United States, 92161 Collapse << |
| NCT01285076 | - | - | Completed | - | Taiwan ... more >> Merck Sharp & Dohme (I.A.) Corp. Taipei, Taiwan, 106 Collapse << |
| NCT00675857 | Diabetes Mellitus, Type 2 ... more >> Metabolic Syndrome X Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Calgary, Alberta, Canada, T3B 0M3 Canada, Manitoba Winnipeg, Manitoba, Canada, R3E 3P4 Canada, Ontario Etobicoke, Ontario, Canada, M9R 4E1 London, Ontario, Canada, N6A 4V2 Oakville, Ontario, Canada, L6H 3P1 Ottawa, Ontario, Canada, K1H 1A2 Ottawa, Ontario, Canada, K1K 4L2 Thornhill, Ontario, Canada, L4J 8L7 Toronto, Ontario, Canada, M4R 2G4 Toronto, Ontario, Canada, M5C 2T2 Canada, Quebec Chicoutimi, Quebec, Canada, G7H 5H6 Greenfield Park, Quebec, Canada, J4V 2H1 Laval, Quebec, Canada, H7T 2P5 Montreal, Quebec, Canada, H1T 2M4 Montreal, Quebec, Canada, H2W 1T7 Montréal, Quebec, Canada, H3A 1A1 Montréal, Quebec, Canada, H3H 2L9 Montréal, Quebec, Canada, H3N 1S4 Québec, Quebec, Canada, G1V 4G5 Sherbrooke, Quebec, Canada, J1G 1B8 Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT00603291 | Obesity | Phase 3 | Completed | - | United States, California ... more >> Arena Pharmaceuticals, Inc. San Diego, California, United States, 92121 Collapse << |
| NCT00603291 | - | - | Completed | - | - |
| NCT01494987 | - | - | Completed | - | - |
| NCT00445627 | - | - | Recruiting | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT02695082 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT01285076 | - | - | Completed | - | - |
| NCT01451398 | - | - | Completed | - | - |
| NCT01494987 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT02695095 | - | - | Recruiting | November 30, 2019 | United States, Delaware ... more >> Research Site Recruiting Wilmington, Delaware, United States United States, District of Columbia Research Site Recruiting Washington, District of Columbia, United States United Kingdom Research Site Recruiting London, United Kingdom Collapse << |
| NCT01451398 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00778765 | Healthy | Not Applicable | Completed | - | Canada, Quebec ... more >> MDS Pharma Services Saint-Laurent,, Quebec, Canada Collapse << |
| NCT02768948 | Diabetic Nephropathy | Not Applicable | Recruiting | July 2019 | France ... more >> CHU de Nice Recruiting Nice, France, 06000 Contact: Guillaume FAVRE, PH favre.g@chu-nice.fr Collapse << |
| NCT02871882 | Type 2 Diabetes Mellitus ... more >> Obese Overweight Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00795600 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Spain ... more >> Málaga, Spain, 29009 Collapse << |
| NCT00763347 | Diabetes Mellitus | Phase 2 | Terminated(Voluntarily termina... more >>ted based on preliminary non-clinical findings.) Collapse << | - | - |
| NCT00272064 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Italy ... more >> Sanofi-Aventis Administrative Office Milan, Italy Collapse << |
| NCT00058981 | Diabetes, Autoimmune | Phase 2 | Terminated | - | United States, Alabama ... more >> University of Alabama at Birmingham Endocrinology Department Birmingham, Alabama, United States, 35294 United States, Colorado University of Colorado Hospital Endocrinology Practice Aurora, Colorado, United States, 80010 United States, Kentucky University of Kentucky Department of Internal Medicine Lexington, Kentucky, United States, 40536 United States, Missouri Washington University School of Medicine Endocrinology/Metabolic Dept Saint Louis, Missouri, United States, 63110 United States, New York North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States, 11042 United States, Texas Diabetes & Glandular Disease Research Associates San Antonio, Texas, United States, 78229-4801 United States, Washington DVA Puget Sound Health Care System Endocrinology (III) Department Seattle, Washington, United States, 98108 Collapse << |
| NCT01699737 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT03406377 | Type 2 Diabetes Mellitus | Phase 2 | Recruiting | June 2019 | United States, California ... more >> National Research Institute - Huntington Park Recruiting Huntington Park, California, United States, 90255 Contact: Stanley Hsin-Wei Hsia National Research Institute - Wilshire Recruiting Los Angeles, California, United States, 90057 Contact: Juan Pablo Frias United States, Florida Clinical Pharmacology of Miami, LLC Recruiting Hialeah, Florida, United States, 33014 Contact: Kenneth Lasseter, MD Collapse << |
| NCT00673309 | Burns | Phase 2 Phase 3 | Completed | - | United States, Texas ... more >> University of Texas Medical Branch Galveston, Texas, United States, 77550 Collapse << |
| NCT01700075 | Myocardial Infarction ... more >> Coronary Artery Diseases Diabetes Mellitus, Type 2 Atherosclerosis of Femoral Artery Collapse << | Phase 4 | Completed | - | Kazakhstan ... more >> Scientific Research Institute of Cardiology and Internal Diseases Almaty, Kazakhstan, 050000 Collapse << |
| NCT01923350 | Weight Reduction | Not Applicable | Completed | - | United States, Maryland ... more >> Social & Scientific Systems Inc. Silver Spring, Maryland, United States, 20910 Collapse << |
| NCT00528372 | - | - | Completed | - | - |
| NCT00795600 | - | - | Completed | - | - |
| NCT00873821 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | - |
| NCT02449603 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Shaanxi ... more >> Xijing Hospital, Fourth Military Medical university Xi'an, Shaanxi, China, 710032 Collapse << |
| NCT00873821 | - | - | Completed | - | - |
| NCT00528372 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01301456 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Metabolic Diseases Endocrine System Diseases Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 Comprehensive Phase One (A Division of Comprehensive NeuroScience, Inc.) Miramar, Florida, United States, 33025 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Nebraska ICON Clinical Pharmacology, LLC Omaha, Nebraska, United States, 68154 United States, Pennsylvania CRI Worldwide, LLC Philadelphia, Pennsylvania, United States, 19139 United States, Texas Healthcare Discoveries LLC d/b/a ICON Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT00300911 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Turkey ... more >> Baskent University Ankara Hospital Ankara, Turkey, 06490 Collapse << |
| NCT01647308 | Type 2 Diabetes | Phase 2 | Terminated(The study was termi... more >>nated for business planning purposes. Assessments of APOCIIIRx in this population will be explored in a larger efficacy study) Collapse << | - | United States, California ... more >> Profil Institute for Clinical Research Chula Vista, California, United States, 91911 Collapse << |
| NCT01301456 | - | - | Completed | - | - |
| NCT00454233 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | Russian Federation ... more >> Ekaterinburg, Russian Federation 4 Sites Moscow, Russian Federation N. Novgorod, Russian Federation Petrozavodsk, Russian Federation Samara, Russian Federation 10 Sites St. Petersburg, Russian Federation Collapse << |
| NCT03526289 | Type 2 Diabetes Mellitus ... more >> Overweight and Obesity Collapse << | Not Applicable | Completed | - | Denmark ... more >> Center for diabetes research Hellerup, Denmark, DK-2900 Collapse << |
| NCT03433248 | Type2 Diabetes | Phase 4 | Recruiting | December 1, 2020 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1081HV Contact: Michaël JB van Baar, MD +31 20 444 4709 m.vanbaar@vumc.nl Contact: Daniël H van Raalte, MD PhD +31 20 444 0534 d.vanraalte@vumc.nl Principal Investigator: Mark HH Kramer, MD PhD Principal Investigator: Daniël H van Raalte, MD PhD Collapse << |
| NCT02367066 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Research Site Chula Vista, California, United States Collapse << |
| NCT02049034 | Type 2 Diabetes | Phase 4 | Completed | - | United Kingdom ... more >> University of Surrey FHMS Diabetes and Metabolic Medicine Guildford, Surrey, United Kingdom, GU2 7WG Collapse << |
| NCT02367066 | - | - | Completed | - | - |
| NCT03538743 | Type 2 Diabetes Mellitus | Phase 1 | Recruiting | April 20, 2019 | United States, California ... more >> Anaheim Clinical Trials, LLC Recruiting Anaheim, California, United States, 92801 United States, Florida Qps-Mra, Llc Recruiting South Miami, Florida, United States, 33143 Qps-Mra,Llc Recruiting South Miami, Florida, United States, 33143 United States, Kansas Vince & Associates Clinical Research, Inc. Recruiting Overland Park, Kansas, United States, 66212 Vince and Associates Clinical Research, Inc. Recruiting Overland Park, Kansas, United States, 66212 Collapse << |
| NCT00377676 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03098654 | Human Immunodeficiency Virus ... more >> Diabetes Mellitus, Type 2 Hypertension Collapse << | Not Applicable | Recruiting | June 30, 2021 | Tanzania ... more >> Muhimbili University of Health and Allied Sciences Recruiting Dar Es Salaam, Tanzania Contact: Jessie Mbwambo, MD jmbwambo@gmail.com Collapse << |
| NCT03061409 | Nutrient Deficiency | Not Applicable | Recruiting | July 2018 | United States, Massachusetts ... more >> Jean Mayer USDA Human Nutrition Research Center on Aging, Tufts University Recruiting Boston, Massachusetts, United States, 02111 Contact: Kimberly Dupiton, BA 617-556-3012 Kimberly.Dupiton@tufts.edu Collapse << |
| NCT02072096 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(The trial was termi... more >>nated per protocol because of lack of feasibility.) Collapse << | - | - |
| NCT00377676 | - | - | Completed | - | - |
| NCT00722371 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00722371 | - | - | Completed | - | - |
| NCT00155545 | Leg Length Discrepancy ... more >> Scoliosis Collapse << | Phase 1 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT00913367 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Korea, Republic of ... more >> HeeYoung Lee Seoul, Korea, Republic of Collapse << |
| NCT00887094 | Diabetes Mellitus, Type 2 ... more >> Treatment Exercise Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital de Clinicas de Porto Alegre Porto Alegre, RS, Brazil, 90035-003 Collapse << |
| NCT02072096 | - | - | Terminated(The trial was termi... more >>nated per protocol because of lack of feasibility.) Collapse << | - | - |
| NCT03168880 | Triple Negative Breast Cancer | Phase 3 | Recruiting | November 30, 2024 | India ... more >> Tata memorial Centre Recruiting Mumbai, Maharashtra, India, 400012 Contact: Rohini Hawaldar 24168601 rwhawaldar@gmail.com Collapse << |
| NCT01922492 | Partial Pancreatectomy Due to ... more >>Benign Pancreatic Neoplasm Collapse << | Not Applicable | Unknown | December 2017 | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT03624803 | - | - | Not yet recruiting | May 31, 2019 | - |
| NCT00502710 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT03462069 | Diabetes Mellitus | Phase 2 | Recruiting | August 7, 2019 | Germany ... more >> Investigational Site Number 2760001 Recruiting Berlin, Germany, 13353 Collapse << |
| NCT00794963 | Metabolic Syndrome | Not Applicable | Terminated(Difficulty recruiti... more >>ng) Collapse << | - | United States, New York ... more >> Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 Collapse << |
| NCT00147836 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | China, Guangdong ... more >> the first Affiliated Hospital of Sun Yat-Sen university Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT01697592 | - | - | Completed | - | - |
| NCT01697592 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00794963 | - | - | Terminated(Difficulty recruiti... more >>ng) Collapse << | - | - |
| NCT01204294 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan ... more >> 1218.78.008 Boehringer Ingelheim Investigational Site Akishima, Tokyo, Japan 1218.78.030 Boehringer Ingelheim Investigational Site Amagasaki, Hyogo, Japan 1218.78.017 Boehringer Ingelheim Investigational Site Annaka, Gunma, Japan 1218.78.006 Boehringer Ingelheim Investigational Site Aomori, Aomori, Japan 1218.78.007 Boehringer Ingelheim Investigational Site Aomori, Aomori, Japan 1218.78.009 Boehringer Ingelheim Investigational Site Chuo-ku, Tokyo, Japan 1218.78.013 Boehringer Ingelheim Investigational Site Chuo-ku, Tokyo, Japan 1218.78.032 Boehringer Ingelheim Investigational Site Fukuoka, Fukuoka, Japan 1218.78.040 Boehringer Ingelheim Investigational Site Fukuoka, Fukuoka, Japan 1218.78.037 Boehringer Ingelheim Investigational Site Higashi Osaka, Osaka, Japan 1218.78.043 Boehringer Ingelheim Investigational Site Hitachinaka, Ibaraki, Japan 1218.78.019 Boehringer Ingelheim Investigational Site Isesaki, Gunma, Japan 1218.78.036 Boehringer Ingelheim Investigational Site Kashiwara, Osaka, Japan 1218.78.026 Boehringer Ingelheim Investigational Site Kasugai, Aichi, Japan 1218.78.038 Boehringer Ingelheim Investigational Site Kawachinagano, Osaka, Japan 1218.78.021 Boehringer Ingelheim Investigational Site Kitaazumi-gun, Nagano, Japan 1218.78.022 Boehringer Ingelheim Investigational Site Matsumoto, Nagano, Japan 1218.78.033 Boehringer Ingelheim Investigational Site Matsumoto, Nagano, Japan 1218.78.012 Boehringer Ingelheim Investigational Site Meguro-ku, Tokyo, Japan 1218.78.041 Boehringer Ingelheim Investigational Site Meguro-ku, Tokyo, Japan 1218.78.034 Boehringer Ingelheim Investigational Site Morioka, Iwate, Japan 1218.78.035 Boehringer Ingelheim Investigational Site Morioka, Iwate, Japan 1218.78.018 Boehringer Ingelheim Investigational Site Moriya, Ibaraki, Japan 1218.78.024 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.025 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.027 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.028 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.031 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1218.78.039 Boehringer Ingelheim Investigational Site Oita, Oita, Japan 1218.78.004 Boehringer Ingelheim Investigational Site Okinawa, Okinawa, Japan 1218.78.016 Boehringer Ingelheim Investigational Site Sagae, Yamagata, Japan 1218.78.001 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.020 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.042 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1218.78.005 Boehringer Ingelheim Investigational Site Shimajiri-gun, Okinawa, Japan 1218.78.014 Boehringer Ingelheim Investigational Site Shinjuku-ku, Tokyo, Japan 1218.78.002 Boehringer Ingelheim Investigational Site Shinjyuku-ku. Tokyo, Japan 1218.78.011 Boehringer Ingelheim Investigational Site Shizuoka, Shizuoka, Japan 1218.78.003 Boehringer Ingelheim Investigational Site Suita, Osaka, Japan 1218.78.023 Boehringer Ingelheim Investigational Site Tokorozawa, Saitama, Japan 1218.78.029 Boehringer Ingelheim Investigational Site Tokorozawa, Saitama, Japan 1218.78.010 Boehringer Ingelheim Investigational Site Tsuchiura, Ibaraki, Japan 1218.78.015 Boehringer Ingelheim Investigational Site Yamagata, Yamagata, Japan Collapse << |
| NCT00295633 | - | - | Completed | - | - |
| NCT00316082 | Diabetes | Phase 3 | Completed | - | - |
| NCT01204294 | - | - | Completed | - | - |
| NCT00316082 | - | - | Completed | - | - |
| NCT00631007 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00295633 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02738879 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00631007 | - | - | Completed | - | - |
| NCT01429506 | Diabetes Mellitus ... more >> Obesity Collapse << | Not Applicable | Completed | - | India ... more >> PGIMER Chandigarh, India, 160012 Collapse << |
| NCT01204775 | Type 2 Diabetes | Phase 3 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States Research Site San Diego, California, United States United States, Florida Research Site Hollywood, Florida, United States Research Site Tallahassee, Florida, United States Research Site Tampa, Florida, United States United States, Michigan Research Site Dearborn, Michigan, United States United States, New York Research Site Buffalo, New York, United States United States, Tennessee Research Site Memphis, Tennessee, United States United States, Virginia Research Site Richmond, Virginia, United States Russian Federation Research Site Moscow, Russian Federation Research Site Novosibirsk, Russian Federation Research Site Saint Petersburg, Russian Federation South Africa Research Site Cape Town, South Africa Research Site Pretoria, South Africa Taiwan Research Site Taichung, Taiwan Research Site Taipei, Taiwan Collapse << |
| NCT01204775 | - | - | Completed | - | - |
| NCT02052011 | Microvascular Angina | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale New Haven Hospital New Haven, Connecticut, United States, 06510 Collapse << |
| NCT02009488 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> San Diego, La Jolla, California, United States United States, Florida Gainesville, Florida, United States Collapse << |
| NCT00765817 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03711682 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | El Salvador ... more >> Unidad Comunitaria de Salud Familiar Intermedia de Comasagua (UCSFI-Comasagua) Comasagua, La Libertad, El Salvador, 0504 Collapse << |
| NCT02052011 | - | - | Completed | - | - |
| NCT01244646 | - | - | Terminated | - | Netherlands ... more >> Research Site Alkmaar, Netherlands Research Site Den Haag, Netherlands Research Site Edam, Netherlands Research Site Gorinchem, Netherlands Research Site Heerhugowaard, Netherlands Research Site Heiloo, Netherlands Research Site Hoogvliet, Netherlands Research Site Lichtenvoorde, Netherlands Research Site Limmen, Netherlands Research Site Maasbracht, Netherlands Research Site Noord-Scharwoude, Netherlands Research Site Poortvliet, Netherlands Research Site Rotterdam, Netherlands Research Site Spijkenisse, Netherlands Research Site Tuitjenhorn, Netherlands Research Site Vaassen, Netherlands Research Site Valkenswaard, Netherlands Research Site Voerendaal, Netherlands Research Site Volendam, Netherlands Research Site Wamel, Netherlands Research Site Zuid-Scharwoude, Netherlands Research Site Zutphen, Netherlands Collapse << |
| NCT02823808 | Diabetes | Phase 4 | Recruiting | December 2021 | Korea, Republic of ... more >> Kyunghee University Medical Center Recruiting Seoul, Korea, Republic of Contact: Sang Youl Rhee Collapse << |
| NCT01483781 | Diabetes Mellitus, Type 2 ... more >> Plasma Volume Collapse << | Phase 1 | Completed | - | Germany ... more >> Neuss, Germany Collapse << |
| NCT00765817 | - | - | Completed | - | - |
| NCT02708758 | Gestational Diabetes | Not Applicable | Recruiting | December 2019 | Mexico ... more >> Instituto Nacional de Perinatología Isidro Espinosa de los Reyes Recruiting Mexico City, Mexico, 11000 Contact: Nayeli Martínez-Cruz, MD +525555209900 ext 299 nayemc_21@hotmail.com Contact: Enrique Reyes-Muñoz, MD, PhD. +525555209900 ext 299 dr.enriquereyes@gmail.com Collapse << |
| NCT00393705 | - | - | Completed | - | - |
| NCT00393705 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Croatia ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Zagreb, Croatia, HR-10000 Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bialystok, Poland, 15-276 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rzeszow, Poland, 35-0723 Romania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bucharest, Romania, 020475 South Africa For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kempton Park, South Africa, 1619 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kenilworth, South Africa, 7708 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kuilsriver, South Africa, 7580 Turkey For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Istanbul, Turkey, 34662 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Konya, Turkey, 42075 Collapse << |
| NCT00341406 | - | - | Completed | - | United States, Maryland ... more >> National Institute of Diabetes and Digestive and Kidney Disorders (NIDDK), 9000 Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00465335 | - | - | Completed | - | - |
| NCT01638260 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(Terminated because ... more >>of insufficient number of subjects included.) Collapse << | - | Netherlands ... more >> Maastricht University Medical Center Maastricht, Netherlands, 6229HX Collapse << |
| NCT01944618 | - | - | Terminated() | - | Australia, Victoria ... more >> Local Institution Clayton, Victoria, Australia, VIC 3168 Collapse << |
| NCT03041129 | - | - | Recruiting | June 1, 2022 | United States, Colorado ... more >> University of Colorado Anshutz Medical Campus/Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Yesenia Garcia 720-777-6984 yesenia.garciareyes@childrenscolorado.org Collapse << |
| NCT00664846 | Stroke | Not Applicable | Completed | - | - |
| NCT01156597 | - | - | Completed | - | - |
| NCT00732121 | Type 2 Diabetes | Phase 4 | Unknown | August 2010 | United States, Vermont ... more >> University of Vermont South Burlington, Vermont, United States, 05403 Collapse << |
| NCT01203111 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Algeria ... more >> Administrative office Algiers, Algeria Brazil Administrative office Sao Paulo, Brazil Israel Administrative office Natanya, Israel Lebanon Administrative office Beirut, Lebanon Mexico Administrative office Col. Coyoacan, Mexico Morocco Administrative office Casablanca, Morocco Peru Administrative office Lima, Peru Saudi Arabia Administrative office Jeddah, Saudi Arabia United Arab Emirates Administrative office Dubaï, United Arab Emirates Venezuela Administrative office Caracas, Venezuela Collapse << |
| NCT00353834 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, Massachusetts ... more >> Joslin Diabetes Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT03419624 | Obesity Diabe... more >>tes Mellitus, Type 2 Collapse << | Phase 3 | Recruiting | June 15, 2019 | Germany ... more >> Diabeteszentrum Oldenburg Recruiting Oldenburg, Lower Saxony, Germany, 23758 Contact: Thomas Schaum, MD +494361513622 info@diabeteszentrum-oldenburg.de Principal Investigator: Thomas Schaum, MD University Medical Center Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Contact: Anne Lautenbach, MD +4915222816188 a.lautenbach@uke.de Contact: Hilko TD Wittmann h.wittmann@uke.de Diabetologische Schwerpunktpraxis Harburg Recruiting Hamburg, Germany, 21073 Contact: Bjoern Paschen, MD +4940557753300 info@diabetologie-harburg.de Principal Investigator: Bjoern Paschen, MD Gemeinschaftspraxis für Innere Medizin und Diabetologie Recruiting Hamburg, Germany, 22607 Contact: Dominik Dahl, MD +4940896295 Principal Investigator: Dominik Dahl, MD Collapse << |
| NCT00656864 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, Vermont ... more >> University of Vermont South Burlington, Vermont, United States, 05403 Collapse << |
| NCT01156597 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Florida ... more >> Diabetes Research Institute Miami, Florida, United States, 33136 Collapse << |
| NCT01175486 | Diabetes | Phase 4 | Unknown | December 2015 | Taiwan ... more >> Taipei Veterans General Hospital, Taiwan Recruiting Taipei, Taiwan Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT03703115 | Infertility P... more >>olycystic Ovary Syndrome Collapse << | Not Applicable | Recruiting | September 1, 2019 | Egypt ... more >> KasrELAiniH Recruiting Cairo, Egypt Contact: Abdelmaguid Ramzy, MD 00223682030 amramzy@gmail.com Contact: Amira Dieb 00223682030 amirasaied2026@cu.edu.gov Principal Investigator: Ramzy, MD Collapse << |
| NCT02686476 | Non Alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Completed | - | India ... more >> Division Of Endocrinology , Medanta The Medicity Sec 38 Gurgaon, Haryana, India, 122001 Collapse << |
| NCT00353834 | - | - | Completed | - | - |
| NCT01623024 | Non.Alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 3 | Unknown | - | Italy ... more >> Sezione di Gastroenterologia, Di.Bi.M.I.S. AOUP Paolo Giaccone, University of Palermo, Italy Not yet recruiting Palermo, Italy, 90127 Contact: Antonio Craxì, Professor 00390916552280 antonio.craxi@unipa.it Principal Investigator: Antonio Craxì, Professor Collapse << |
| NCT03503045 | - | - | Completed | - | Turkey ... more >> Gulhane School of Medicine Ankara, Turkey, 06018 Collapse << |
| NCT01072578 | Healthy Volunteers | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT03249896 | Gestational Diabetes ... more >> Pregnancy Complications Weight Gain Collapse << | Not Applicable | Recruiting | April 2019 | Singapore ... more >> National University Hospital Recruiting Singapore, Singapore, 119228 Contact: E Shyong Tai, PhD +65 67724371 e_shyong_tai@nuhs.edu.sg Contact: Tong Wei Yew, MRCP +65 67723943 tong_wei_yew@nuhs.edu.sg Collapse << |
| NCT02830191 | - | - | Completed | - | - |
| NCT02420392 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00696982 | Diabetes Mellitus ... more >> Arterial Stiffness Collapse << | Not Applicable | Unknown | June 2009 | Israel ... more >> Department of Internal Medicine A , Research & Development unit Assaf Harofeh Medical Center, Zerifin, affiliated to Sackler School of Medicine, Tel Aviv, Israel Recruiting Zrifin, Israel, 70300 Contact: Shlomit Koren, MD 972-546-888=385 shlomitks@gmail.com Contact: Shai Efrati, MD 972-577-346-364 efratishai@013.net Principal Investigator: Shlomit Koren, MD Collapse << |
| NCT02051842 | NAFLD Pre-dia... more >>betes Collapse << | Phase 4 | Unknown | January 2017 | Mexico ... more >> Instituto Nacional de Ciencias Médicas y Nutrición "Salvador Zubirán" Recruiting Mexico City, Mexico, 14000 Contact: Aldo Torre, M.D, M.Sc. 54870900 ext 2711 detoal@yahoo.com Principal Investigator: Aldo Torre, MD MSci Collapse << |
| NCT03023137 | Recurrent Miscarriage | Not Applicable | Completed | - | Brazil ... more >> Hospital Federal dos Servidores do Estado, Ministry of Health Rio de Janeiro, RJ, Brazil, 20221-903 Collapse << |
| NCT02925559 | Diabetes Mellitus, Type 2 | Phase 4 | Unknown | December 2017 | Brazil ... more >> Centro de Diabetes Curitiba Curitiba, Parana, Brazil, 80810040 Collapse << |
| NCT03023137 | - | - | Completed | - | - |
| NCT00832390 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT03732690 | Type 2 Diabetes | Not Applicable | Not yet recruiting | December 30, 2020 | France ... more >> Hôpital PITIE SALPETRIERE - APHP Not yet recruiting Paris, France, 75013 Contact: Karine CLEMENT, MD, PhD Collapse << |
| NCT00832390 | - | - | Completed | - | - |
| NCT02444156 | Diabetes Mellitus, Type 2 ... more >> Glucose Intolerance Hyperglycemia Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Leicester Diabetes Centre Leicester, United Kingdom, LE5 4PW Collapse << |
| NCT03620773 | Type 2 Diabetes Mellitus ... more >> Obesity Bariatric Surgery Candidate Nephropathy Diabetic Kidney Disease Diabetes Mellitus, Type 2 Diabetes Mellitus Diabetes Complications Weight Loss Diabetic Nephropathies Adolescent Obesity Pediatric Obesity Collapse << | Phase 1 Phase 2 | Recruiting | August 2023 | United States, Colorado ... more >> Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Petter Bjornstad, MD 720-777-4659 petter.bjornstad@childrenscolorado.org Principal Investigator: Petter Bjornstad, M.D. Sub-Investigator: Kristin Nadeau, M.D. Collapse << |
| NCT02627898 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02176681 | Haemodialyzed, Type 2 Diabetes | Phase 4 | Completed | - | France ... more >> CH d'Amiens Amiens, France, 80054 CH de Besançon Besancon, France, 25030 AURAL Colmar Colmar, France, 68000 Hospices civils de Colmar Colmar, France, 68024 CH de Dijon Dijon, France, 21079 AURAL Mulhouse Mulhouse, France, 68070 CH de Mulhouse Mulhouse, France, 68070 CH de Nancy Nancy, France, 54000 AURAL Clinique Sainte Anne Strasbourg, France, 67000 Clinique Sainte Anne Strasbourg, France, 67000 Hôpitaux Universitaires de Strasbourg Strasbourg, France, 67091 AURAL Strasbourg Strasbourg, France, 67200 CH de Valenciennes Valenciennes, France, 59300 Collapse << |
| NCT01472432 | Chronic Foot Ulcers | Phase 4 | Completed | - | Italy ... more >> Second university of Naples Naples, Italy, I-80100 Collapse << |
| NCT02941770 | Overweight | Not Applicable | Recruiting | February 2019 | Portugal ... more >> Hospital de Santa Maria Recruiting Lisbon, Portugal, 1649-035 Contact: António Videira-Silva, MSc 969172368 antonioascenso@campus.ul.pt Collapse << |
| NCT01919489 | Type 2 Diabetes | Phase 4 | Recruiting | June 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States Contact: Gianluca Iacobellis, MD GIacobellis@med.miami.edu Principal Investigator: Gianluca Iacobellis, MD United States, Georgia Grady Memorial Hospital Recruiting Atlanta, Georgia, United States, 30303 Contact: Guillermo E Umpierrez, MD 404-778-1665 geumpie@emory.edu Contact: Francisco J Pasquel, MD 404 778 1697 fpasque@emory.edu Principal Investigator: Guillermo Umpierrez, MD Sub-Investigator: Francisco J Pasquel, MD Sub-Investigator: Priya Vellanki, MD Emory University Hospital Recruiting Atlanta, Georgia, United States, 30324 Contact: Francisco Pasquel, MD fpasque@emory.edu Sub-Investigator: Francisco Pasquel, MD Emory Universtiy Hospital at MIdtown Recruiting Atlanta, Georgia, United States Contact: Francisco Pasquel, MD Sub-Investigator: Rodolfo J Galindo, MD United States, New York State University of NY at Buffalo Recruiting New York, New York, United States Contact: Ajay Chaudhuri, MD AChaudhuri@KaleidaHealth.Org Principal Investigator: Ajay Chaudhuri, MD Collapse << |
| NCT02076880 | Severe Obesity ... more >> Type 2 Diabetes Collapse << | Not Applicable | Unknown | November 2017 | France ... more >> CHU Montpellier Recruiting Montpellier, France, 34295 Contact: GALTIER Florence f-galtier@chu-montpellier.fr Principal Investigator: GALTIER Florence, MD Collapse << |
| NCT01849289 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00451620 | Type 2 Diabetes | Phase 2 | Completed | - | Canada, British Columbia ... more >> University of British Columbia Vancouver, British Columbia, Canada Collapse << |
| NCT00965549 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01122641 | Insulin Resistance ... more >> Microvascular Disease Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Diabetes and Vascular Research Department Exeter, Devon, United Kingdom, EX2 5AX Collapse << |
| NCT03429543 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | January 31, 2022 | - |
| NCT00658164 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Phase 3 | Unknown | - | Italy ... more >> U.O. Medicina Interna 1/B Recruiting Milan, Italy, 20122 Contact: Silvia Fargion, Prof. 39-02-5503-3301 silvia.fargion@unimi.it Principal Investigator: Silvia Fargion, Prof. Sub-Investigator: Luca Valenti, Md Collapse << |
| NCT01039532 | - | - | Completed | - | India ... more >> AnilBhansali Chandigarh, India, 160012 Collapse << |
| NCT00353587 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | - |
| NCT01849289 | - | - | Completed | - | - |
| NCT01018017 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Neuss, Nordrhein-Westfalen, Germany, 41460 Collapse << |
| NCT00736879 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT03739268 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | September 1, 2020 | Denmark ... more >> Steno Diabetes Center Copenhagen, Gentofte Hospital Recruiting Hellerup, Denmark, 2900 Contact: Henriette H Nerild, M.D. +45 3867 4264 henriette.holst.nerild@regionh.dk Collapse << |
| NCT01022658 | Type 2 Diabetes | Not Applicable | Completed | - | France ... more >> Jean Verdier hospital, Department of Endocrinology-Diabetology-Nutrition Bondy, France, 93140 Collapse << |
| NCT02830048 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United Kingdom ... more >> Clinical Research Unit, OCDEM, Churchill Hospital Oxford, Oxfordshire, United Kingdom, OX3 7LE Collapse << |
| NCT01472432 | - | - | Completed | - | - |
| NCT02922179 | - | - | Unknown | January 2017 | - |
| NCT00982644 | - | - | Completed | - | - |
| NCT00982644 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03444142 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 30, 2018 | Mexico ... more >> Instituto de terapeutica Experimental y Clinica. Centro universitario de Ciencias de la Salud. Universidad de Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT01379690 | - | - | Completed | - | Singapore ... more >> Changi General Hospital Singapore, Singapore, 529889 Collapse << |
| NCT03705195 | Type 2 Diabetes Mellitus | Not Applicable | Recruiting | June 20, 2019 | United Kingdom ... more >> Ninewells Hospital and Medical School Recruiting Dundee, United Kingdom, DD1 9SY Contact: Ruth LM Cordiner, MBChB, MRCP 0044 1382 386 715 ext 00441382386715 r.cordiner@dundee.ac.uk Principal Investigator: Ruth LM Cordiner Collapse << |
| NCT02024477 | Type 2 Diabetes | Phase 4 | Active, not recruiting | January 2018 | United States, District of Col... more >>umbia Medical Faculty Associates Inc Washington, District of Columbia, United States, 20037 Collapse << |
| NCT00736879 | - | - | Completed | - | - |
| NCT03766308 | Type 2 Diabetes Mellitus | Not Applicable | Active, not recruiting | May 31, 2019 | China, Hubei ... more >> Tongji Hospital, Tongji Medical College, Huazhong University of Science and technology Wuhan, Hubei, China Collapse << |
| NCT01514942 | PCOS | Phase 4 | Completed | - | Italy ... more >> Istituto di Patologia Ostetrica e Ginecologica Catania, Italy Collapse << |
| NCT03652987 | - | - | Not yet recruiting | September 2023 | - |
| NCT03376490 | - | - | Completed | - | Singapore ... more >> SingHealth Polyclinics - Marine Parade Polyclinic Singapore, Singapore, 440080 Collapse << |
| NCT03074630 | Diabetes Mellitus, Type 2 ... more >> Hypercholesterolemia Collapse << | Phase 4 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, North Holland, Netherlands, 1105AZ Collapse << |
| NCT01055223 | - | - | Completed | - | - |
| NCT00036504 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | United States, California ... more >> LaJolla, California, United States Walnut Creek, California, United States United States, Connecticut New Britain, Connecticut, United States United States, Florida Longwood, Florida, United States United States, Georgia Atlanta, Georgia, United States Fayetteville, Georgia, United States United States, Idaho Boise, Idaho, United States United States, Illinois Springfield, Illinois, United States United States, New Jersey Mt. Laurel, New Jersey, United States New Brunswick, New Jersey, United States United States, New York Syracuse, New York, United States United States, Tennessee Chattanooga, Tennessee, United States United States, Texas Dallas, Texas, United States Collapse << |
| NCT02826525 | T2DM With NAFLD | Phase 1 Phase 2 | Active, not recruiting | December 28, 2020 | - |
| NCT01986855 | - | - | Completed | - | - |
| NCT02608905 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | May 2019 | United States, Texas ... more >> Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Mandeep Bajaj, MD 713-798-1712 bajaj@bcm.edu Collapse << |
| NCT03536364 | Obesity Pre-D... more >>iabetes Collapse << | Not Applicable | Completed | - | United States, New York ... more >> Comprehensive Weight Control Center New York, New York, United States, 10065 Collapse << |
| NCT01055223 | - | - | Completed | - | - |
| NCT01272232 | Metabolism and Nutrition Disor... more >>der Obesity Collapse << | Phase 3 | Completed | - | - |
| NCT00642278 | Diabetes Mellitus, Type II ... more >> Diabetes Mellitus, Non Insulin Dependent Collapse << | Phase 2 | Completed | - | - |
| NCT01272232 | - | - | Completed | - | - |
| NCT01606397 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | Singapore ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Singapore, Singapore United Kingdom For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Plymouth, United Kingdom Collapse << |
| NCT00659711 | Type 2 Diabetes | Not Applicable | Unknown | December 2013 | United States, New York ... more >> 115 Flint Road Buffalo, New York, United States, 14221 Collapse << |
| NCT01986855 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01869959 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cypress, California, United States, 90630 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tustin, California, United States, 92780 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Deland, Florida, United States, 32720 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Miramar, Florida, United States, 33025 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45212 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97239 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 Collapse << |
| NCT03735108 | - | - | Recruiting | June 30, 2019 | China ... more >> Shanghai First Maternity and Infant Hospital Recruiting Shanghai, China Contact: Shihua Bao 86-021-20261599 baoshihua@tongji.edu.cn Collapse << |
| NCT01869959 | - | - | Completed | - | - |
| NCT02798757 | Diabetes | Phase 4 | Recruiting | May 2018 | Greece ... more >> University of Ioannina Recruiting Ioánnina, Greece, 45500 Contact: Vasilis Tsimihodimos, Assistant Professor +306971705609 vtsimi@uoi.gr Collapse << |
| NCT00642278 | - | - | Completed | - | - |
| NCT02235194 | Type 2 Diabetes, ... more >> Prediabetes Collapse << | Not Applicable | Completed | - | Germany ... more >> Stoffwechselzentrum Rhein Pfalz Mannheim, Germany, 68163 Collapse << |
| NCT00546325 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Australia ... more >> Sanofi-Aventis Administrative Office North Ryde, Australia Collapse << |
| NCT02197520 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research Inc Chula Vista, California, United States, 91911 Collapse << |
| NCT02197520 | - | - | Completed | - | - |
| NCT01357889 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01357889 | - | - | Completed | - | - |
| NCT00502281 | Polycystic Ovary Syndrome | Phase 4 | Suspended | - | Italy ... more >> Pugliese Hospital Catanzaro, Catanzaro, CZ, Italy, 88100 Collapse << |
| NCT03021187 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01936025 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Germany ... more >> Profil Institut für Stoffwechselforschung GmbH Neuss, Germany, 41460 Collapse << |
| NCT00800176 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT00044694 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 2 | Completed | - | - |
| NCT03409523 | - | - | Enrolling by invitation | December 2018 | United States, New York ... more >> New York University School of Medicine New York, New York, United States, 10016 Collapse << |
| NCT00482742 | Catheterization, Peripheral | Not Applicable | Terminated(product development... more >> was cancelled) Collapse << | - | Croatia ... more >> University Hospital Split Split, Croatia, 21000 Clinical Hospital Centre Zagreb Zagreb, Croatia, 10000 Israel Rabin Campus, Belinson Hospital Petah-Tikva, Israel, 49100 Collapse << |
| NCT01986881 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | September 11, 2019 | - |
| NCT01512108 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Japan ... more >> Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0002 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0027 Novo Nordisk Investigational Site Ehime, Japan, Japan, 790 0067 Novo Nordisk Investigational Site Fukuoka, Japan, Japan, 810 8798 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan, 815 8555 Novo Nordisk Investigational Site Fukuoka, Japan, 812 0025 Novo Nordisk Investigational Site Fukuoka, Japan, 818 8502 Novo Nordisk Investigational Site Fukuoka, Japan, 820 8505 Novo Nordisk Investigational Site Kamagaya-shi, Chiba, Japan, 273 0121 Novo Nordisk Investigational Site Kashiwara-shi, Osaka, Japan, 582 0005 Novo Nordisk Investigational Site Kawagoe-shi, Saitama, Japan, 350 0851 Novo Nordisk Investigational Site Kitakyushu-shi, Fukuoka, Japan, 800 0252 Novo Nordisk Investigational Site Koriyama-shi, Fukushima, Japan, 963 8851 Novo Nordisk Investigational Site Kumamoto-shi,Kumamoto, Japan, 862 0976 Novo Nordisk Investigational Site Kurume-shi, Fukuoka, Japan, 839 0863 Novo Nordisk Investigational Site Miyazaki-shi, Japan, 880 0034 Novo Nordisk Investigational Site Naka-shi, Ibaraki, Japan, 311 0113 Novo Nordisk Investigational Site Niigata-shi, Niigata, Japan, 950 1104 Novo Nordisk Investigational Site Oita, Japan, 870 8511 Novo Nordisk Investigational Site Okawa-shi, Fukuoka, Japan, 831 0016 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 532 0003 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 545 8586 Novo Nordisk Investigational Site Ota-ku, Tokyo, Japan, 144 0035 Novo Nordisk Investigational Site Oyama-shi, Tochigi, Japan, 323 0022 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060-0001 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 062 0007 Novo Nordisk Investigational Site Shimotsuke-shi, Tochigi, Japan, 329 0433 Novo Nordisk Investigational Site Shizuoka-shi, Japan, 424 0853 Novo Nordisk Investigational Site Tagajo-shi, Japan, 985 0852 Novo Nordisk Investigational Site Takatsuki-shi, Osaka, Japan, 569 1096 Novo Nordisk Investigational Site Tochigi, Japan, 329 0498 Novo Nordisk Investigational Site Tokyo, Japan, 104 0044 Novo Nordisk Investigational Site Tokyo, Japan, 113 0031 Novo Nordisk Investigational Site Tokyo, Japan, 125 0054 Novo Nordisk Investigational Site Yamaguchi, Japan, 755 0067 Novo Nordisk Investigational Site Yokohama-shi, Japan, 235 0045 Collapse << |
| NCT02119819 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT03244579 | Gestational Diabetes Mellitus ... more >>With Baby Delivered Collapse << | Not Applicable | Recruiting | October 20, 2019 | Jordan ... more >> Sabika Allehdan Recruiting Amman, Jordan, 009626 Contact: Sabika Allehdan, MSc 799232789 sabika_1986@Yahoo.com Collapse << |
| NCT01618214 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Anhui ... more >> Novo Nordisk Investigational Site Hefei, Anhui, China, 230001 Novo Nordisk Investigational Site Hefei, Anhui, China, 230022 China, Beijing Novo Nordisk Investigational Site Beijing, Beijing, China, 100029 Novo Nordisk Investigational Site Beijing, Beijing, China, 100088 Novo Nordisk Investigational Site Beijing, Beijing, China, 100700 Novo Nordisk Investigational Site Beijing, Beijing, China, 100730 China, Chongqing Novo Nordisk Investigational Site Chongqing, Chongqing, China, 400010 Novo Nordisk Investigational Site ChongQing, Chongqing, China, 404000 China, Guangdong Novo Nordisk Investigational Site Guangzhou, Guangdong, China, 510080 China, Hebei Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050051 Novo Nordisk Investigational Site Shijiazhuang, Hebei, China, 050082 China, Hubei Novo Nordisk Investigational Site Wuhan, Hubei, China, 430034 China, Inner Mongolia Novo Nordisk Investigational Site Tongliao, Inner Mongolia, China, 028007 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210011 Novo Nordisk Investigational Site Yangzhou, Jiangsu, China, 225001 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Jilin Novo Nordisk Investigational Site Changchun, Jilin, China, 130041 Novo Nordisk Investigational Site Siping, Jilin, China, 136000 China, Shaanxi Novo Nordisk Investigational Site Xi'an, Shaanxi, China, 710032 China, Shandong Novo Nordisk Investigational Site Jinan, Shandong, China, 250013 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 China, Zhejiang Novo Nordisk Investigational Site Hangzhou, Zhejiang, China, 310003 Collapse << |
| NCT01512108 | - | - | Completed | - | - |
| NCT03086330 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT03278470 | Rheumatoid Arthritis | Phase 1 | Completed | - | Korea, Republic of ... more >> The catholic university of korea seoul ST. mary's hospital Soeul, Korea, Republic of Collapse << |
| NCT00375492 | - | - | Completed | - | - |
| NCT00375492 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | United States, Arizona ... more >> Research Site Peoria, Arizona, United States United States, Florida Research Site Jacksonville, Florida, United States Research Site Orlando, Florida, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Minnesota Research Site Minneapolis, Minnesota, United States United States, Missouri Research Site St. Louis, Missouri, United States United States, South Carolina Research Site Spartanburg, South Carolina, United States United States, Texas Research Site San Antonio, Texas, United States United States, Washington Research Site Renton, Washington, United States Collapse << |
| NCT01618214 | - | - | Completed | - | - |
| NCT00251940 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT01336023 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01336023 | - | - | Completed | - | - |
| NCT02371187 | Physical Activity | Phase 2 | Completed | - | United States, Colorado ... more >> Colorado State University, Dept. of Health and Exercise Science Fort Collins, Colorado, United States, 80523-1582 Collapse << |
| NCT00736099 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02964572 | Type2 Diabetes Mellitus | Not Applicable | Recruiting | October 2018 | Korea, Republic of ... more >> Yonsei University College of Medicine, Department of Internal Medicine, Division of Endocrinology, Severance Hospital, Diabetes center Recruiting Seoul, Korea, Republic of, 03722 Contact: Yong-ho Lee, M.D., Ph.D. 82-2-2228-1943 yholee@yuhs.ac Collapse << |
| NCT01628445 | Type 2 Diabetes | Phase 3 | Terminated(Error made by local... more >> pharmacy caused mixed randomization of 20 participants) Collapse << | - | Canada, Manitoba ... more >> Winnipeg Regional Health Authority Health Sciences Centre Winnipeg Diabetes Research Group Winnipeg, Manitoba, Canada, R3E3P4 Collapse << |
| NCT02240680 | - | - | Completed | - | - |
| NCT01570751 | - | - | Completed | - | - |
| NCT01098539 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00736099 | - | - | Completed | - | - |
| NCT01098539 | - | - | Completed | - | - |
| NCT01570751 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02240680 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT03747107 | - | - | Recruiting | March 30, 2020 | United Kingdom ... more >> NHS Tayside Recruiting Dundee, Scotland, United Kingdom, DD2 4BF Contact: Tobias Dreischulte, PhD ++44(0)7540736211 t.dreischulte@dundee.ac.uk Collapse << |
| NCT03015519 | Diabetes Mellitus, Type 2 | Phase 3 | Withdrawn(The study was stoppe... more >>d as the asset will be withdrawn from the market prior to study end.) Collapse << | April 20, 2020 | United States, Illinois ... more >> GSK Investigational Site Chicago, Illinois, United States, 60634 United States, North Carolina GSK Investigational Site Morehead City, North Carolina, United States, 28557 United States, Texas GSK Investigational Site El Paso, Texas, United States, 79935 Collapse << |
| NCT02588729 | Gestational Diabetes Mellitus | Not Applicable | Completed | - | Norway ... more >> Oslo university hospital Oslo, Norway, 4950 Collapse << |
| NCT03626909 | - | - | Recruiting | January 13, 2021 | Guatemala ... more >> Hospital Obras Sociales Recruiting San Lucas Tolimán, Solala, Guatemala Contact: Sean Duffy, MD +502 4607-0561 Sean.Duffy@fammed.wisc.edu Contact: , MD Collapse << |
| NCT02456428 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02476760 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT01916694 | Gestational Diabetes Mellitus ... more >> Pregnancy Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> John Radcliffe Hospital Oxford, Oxfordshire, United Kingdom, OX3 9DU Collapse << |
| NCT00690755 | - | - | Completed | - | United States, Florida ... more >> VA Medical Center Tampa, Florida, United States, 33612 Collapse << |
| NCT00580710 | - | - | Recruiting | August 2020 | United States, Connecticut ... more >> Yale University School of Medicine Recruiting New Haven, Connecticut, United States, 06520 Principal Investigator: Robert Sherwin, M.D. Collapse << |
| NCT02033499 | - | - | Completed | - | - |
| NCT02475499 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02259972 | Healthy | Phase 1 | Completed | - | - |
| NCT00000620 | - | - | Completed | - | - |
| NCT02033499 | DIABETES MELLITUS, NONINSULIN-... more >>DEPENDENT, 2 (Disorder) Collapse << | Not Applicable | Completed | - | United States, North Carolina ... more >> The University of North Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00000620 | Atherosclerosis ... more >> Cardiovascular Diseases Hypercholesterolemia Hypertension Diabetes Mellitus, Type 2 Diabetes Mellitus Coronary Disease Collapse << | Phase 3 | Completed | - | United States, Minnesota ... more >> Minneapolis Medical Research Foundation Minneapolis, Minnesota, United States, 55404 United States, New York Columbia University New York, New York, United States, 10027 United States, North Carolina Wake Forest University Winston-Salem, North Carolina, United States, 27106 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Tennessee Veterans Affairs Memphis, Tennessee, United States, 38104 United States, Washington University of Washington Seattle, Washington, United States, 98195 Canada, Ontario McMaster University Hamilton, Ontario, Canada Collapse << |
| NCT01868646 | Type II Diabetes Mellitus | Phase 4 | Completed | - | Russian Federation ... more >> Municipal budgetary authority "Khimki Central Clinical Hospital" Moscow region, Russian Federation, 141400 State Healthcare Institution of Moscow "Central research institute of gastroenterology" of Department of health care of Moscow Moscow, Russian Federation, 111123 State Educational Institution of Higher Professional Education "Moscow State Medical Academy named after I.M. Sechenov" Moscow, Russian Federation, 119991 Nonstate Health Care Institution "Central Clinical Hospital №2 named after N.A. Semashko of Public Limited Company "Russian Railways" Moscow, Russian Federation, 129128 Nizhny Novgorod regional State Budgetary Health Institution " Nizhny Novgorod regional Clinical Hospital named after N.A. Semashko " Nizhny Novgorod Region, Russian Federation, 603126 State Budgetary Educational Institution of High Professional Training "Rostov State Medical University" of Ministry of Health of Russian Federation, Department of Endocrinology Rostov-on-Don, Russian Federation, 344022 Co.Ltd " Diabet Center" Samara, Russian Federation, 443067 The State Budgetary Educational Institution of Higher Professional Education "Smolensk National Research Medical University" Ministry of Health of the Russian Federation Smolensk, Russian Federation, 214019 St. Petersburg State Budgetary Health Care Institution "City Polyclinic №6" St. Petersburg, Russian Federation, 191482 St. Petersburg State Budgetary Health Care Institution "Saint Venerable Martyr Elizaveta Municipal Hospital" St. Petersburg, Russian Federation, 195257 State Budgetary Educational Institution of High Professional Training "St. Petersburg State Medical University named after academician I.P. Pavlov" of Ministry of Health of Russian Federation, Therapy Faculty Board St. Petersburg, Russian Federation, 197022 St. Petersburg Sate Budgetary Institution "Consultative-Diagnostic Polyclinic №1 of Coastal Area" St. Petersburg, Russian Federation, 197183 St. Petersburg State Budgetary Health Care Institution "Consultative-Diagnostic Center № 85", Diabetes Center №2 St. Petersburg, Russian Federation, 198260 St. Petersburg State Budgetary Health Care Institution "City Polyclinic №77 of Nevsky District", The City Diabetes Center №4 St.Petersburg, Russian Federation, 192177 Vladimir region State budgetary institution of health care "Regional clinical hospital" Vladimir, Russian Federation, 600023 Independent Health Care Institution of Voronezh Region "Voronezh Regional Clinical Consultative-Diagnostic Center" Voronezh, Russian Federation, 394018 State Budgetary Health Care Institution of Yaroslavl Region "Regional Сlinical Hospital" Yaroslavl, Russian Federation, 150062 Collapse << |
| NCT01974544 | Type 2 Diabetes | Not Applicable | Unknown | December 2016 | Chile ... more >> Pontificia Universidad Católica de Chile Recruiting Santiago, Región Metropolitana, Chile, 8330033 Contact: Castillo Alejandra, Nurse 56223543527 macastia@uc.cl Contact: Vega Andrea, Nurse 56223543879 abvega@uc.cl Principal Investigator: Boza Camilo, MD surgeon Sub-Investigator: Valderas Juan Patricio, MD Sub-Investigator: Arrese Marco, MD Internist Sub-Investigator: Muñoz Rodrigo, MD Collapse << |
| NCT03426228 | - | - | Recruiting | December 31, 2019 | United Arab Emirates ... more >> Fakih IVF Recruiting Dubai, United Arab Emirates, 72960 Contact: Ayla Coussa, Dietitian 00971 4 349 7600 ext 162 a.coussa@warwick.ac.uk Contact: Michael Fakih, MD 00971 4 349 7600 ext 117 mfakih@fakihivf.com Collapse << |
| NCT03732209 | Type 2 Diabetes | Phase 1 Phase 2 | Not yet recruiting | July 31, 2020 | United States, Virginia ... more >> Virginia Tech Carilion Research Institute Roanoke, Virginia, United States, 24016 Collapse << |
| NCT02138097 | - | - | Completed | - | United States, Massachusetts ... more >> 1218.161.1 Boehringer Ingelheim Investigational Site Boston, Massachusetts, United States Collapse << |
| NCT00690755 | - | - | Completed | - | - |
| NCT02430870 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California ... more >> Anaheim, California, United States Chula Vista, California, United States Collapse << |
| NCT02138097 | - | - | Completed | - | - |
| NCT00392678 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | United States, Connecticut ... more >> Chapel Medical Group New Haven, Connecticut, United States, 06511 United States, District of Columbia MedStar Research Institute Washington, District of Columbia, United States, 20003-4393 United States, Florida Endocrine Clinical Research Winter Park, Florida, United States, 32746 United States, Georgia Kaiser Permanente Atlanta, Georgia, United States, 30084 Emory School of Medicine Atlanta, Georgia, United States, 30303 United States, Illinois University of Illinois at Chicago Chicago, Illinois, United States, 60612 United States, Louisiana Tulane University New Orleans, Louisiana, United States, 70112 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68105 United States, New York Kaleida Health Center Buffalo, New York, United States, 14226 North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States, 11042 Columbia University New York, New York, United States, 10032 University of Rochester Medical Center Rochester, New York, United States, 14642 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Texas University of Texas Southwestern Dallas, Texas, United States, 75390 Collapse << |
| NCT00392678 | - | - | Completed | - | - |
| NCT00328627 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00661921 | Diabetes Mellitus | Phase 2 | Withdrawn(Program Development ... more >>discontinued) Collapse << | October 2009 | - |
| NCT01703208 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(The study was termi... more >>nated for business reasons and not due to any safety or efficacy concerns related to omarigliptin) Collapse << | - | - |
| NCT00799643 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham, Alabama, United States United States, California University of California, San Diego San Diego, California, United States United States, Connecticut Chapel Medical Group New Haven, Connecticut, United States United States, Georgia Emory University School of Medicine Atlanta, Georgia, United States Kaiser Permanente Tucker, Georgia, United States United States, Indiana Indiana University Indianapolis, Indiana, United States United States, Louisiana Tulane University Health Sciences Center New Orleans, Louisiana, United States United States, Maryland Medstar Research Institute Hyattsville, Maryland, United States Dr. Rudo, Westminster, MD Westminster, Maryland, United States, 21157 United States, Massachusetts Joslin Diabetes Center Boston, Massachusetts, United States, 02215 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48106 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States United States, New York North Shore Diabetes and Endocrine Associates New Hyde Park, New York, United States Columbia University New York, New York, United States Lang Medical Center Queens, New York, United States Albert Einstein College of Medicine The Bronx, New York, United States United States, North Carolina Carolina's Health Care Charlotte, North Carolina, United States University of North Carolina Durham, North Carolina, United States United States, Texas University of Texas Southwestern Dallas, Texas, United States Scott and White Temple, Texas, United States Collapse << |
| NCT00263276 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT00799643 | - | - | Completed | - | - |
| NCT01106651 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02282423 | Diabetes Obes... more >>ity Collapse << | Not Applicable | Terminated(PI job change) | - | United States, Arizona ... more >> Mayo Clinic Scottsdale, Arizona, United States, 85259 Collapse << |
| NCT01923181 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00571519 | Type 2 Diabetes Mellitus | Phase 3 | Terminated(DSPD focussing on S... more >>tudy 301 to confirm clinical profile before proceeding.) Collapse << | - | United States, Alabama ... more >> Huntsville, Alabama, United States, 35801 Huntsville, Alabama, United States, 35802 United States, California San Diego, California, United States, 92128 San Francisco, California, United States, 94115 United States, Connecticut Ridgefield, Connecticut, United States, 06877 United States, Florida Fort Lauderdale, Florida, United States, 33308 Pembroke Pines, Florida, United States, 33026 United States, Mississippi Port Gibson, Mississippi, United States, 39150 United States, Nevada Las Vegas, Nevada, United States, 89119 United States, New Mexico Albuquerque, New Mexico, United States, 87109 United States, Ohio Kettering, Ohio, United States, 45429 United States, Pennsylvania Fleetwood, Pennsylvania, United States, 19522 Harrisburg, Pennsylvania, United States, 17112 United States, South Carolina Simpsonville, South Carolina, United States, 29681 United States, Texas Daingerfield, Texas, United States, 75638 Dallas, Texas, United States, 75216 Plano, Texas, United States, 75093 San Antonio, Texas, United States, 78205 Collapse << |
| NCT01703208 | - | - | Terminated(The study was termi... more >>nated for business reasons and not due to any safety or efficacy concerns related to omarigliptin) Collapse << | - | - |
| NCT01106651 | - | - | Completed | - | - |
| NCT00328627 | - | - | Completed | - | - |
| NCT02430870 | - | - | Completed | - | - |
| NCT01221519 | Type 2 Diabetes Mellitus ... more >> High Blood Sugar Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Research Site St. Paul, Minnesota, United States Collapse << |
| NCT03359837 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | November 7, 2019 | China ... more >> Recruiting Not Listed, China Collapse << |
| NCT02954692 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Turkey ... more >> Turkey, Turkey Collapse << |
| NCT00935805 | - | - | Unknown | July 2012 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT01798706 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT03568630 | - | - | Recruiting | July 2023 | United States, Nebraska ... more >> Nebraska Medical Center Recruiting Omaha, Nebraska, United States, 68198 Contact: Christina Hoy, DNP 402-559-1577 christina.hoy@unmc.edu Collapse << |
| NCT03685357 | - | - | Recruiting | January 2020 | Egypt ... more >> AinShams University Recruiting Cairo, Egypt Principal Investigator: Afnan Elgnainy Collapse << |
| NCT01798706 | - | - | Completed | - | - |
| NCT03049228 | - | - | Enrolling by invitation | April 2, 2019 | Netherlands ... more >> Human Biology Maastricht, Limburg, Netherlands, 6200MD Collapse << |
| NCT01922791 | Pregnancy Obe... more >>sity Gestational Diabetes Mellitus Collapse << | Not Applicable | Active, not recruiting | January 2020 | Finland ... more >> University of Turku Turku, Finland, 20014 Turku University Hospital Turku, Finland, 20521 Collapse << |
| NCT00940797 | Diabetes Type 2 | Phase 2 | Completed | - | Mexico ... more >> Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara, México Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02157974 | Hepatic Steatosis ... more >> Polycystic Ovarian Syndrome Obesity Collapse << | Phase 2 Phase 3 | Recruiting | December 2018 | United States, Colorado ... more >> University of Colorado Anshutz Medical Campus/Children's Hospital Colorado Recruiting Aurora, Colorado, United States, 80045 Contact: Yesenia Garcia 720-777-6984 yesenia.garciareyes@childrenscolorado.org Collapse << |
| NCT03491241 | Pre-diabetes ... more >>Obesity Collapse << | Not Applicable | Completed | - | Italy ... more >> Raffaele Marfella Naples, Italy, 80138 Collapse << |
| NCT02272556 | Hyperglycemia | Early Phase 1 | Recruiting | November 2019 | United States, Connecticut ... more >> Yale University Recruiting New Haven, Connecticut, United States, 06519 Contact: Janice Hwang, MD 203-785-6222 janice.hwang@yale.edu Collapse << |
| NCT01077323 | - | - | Completed | - | United States, Massachusetts ... more >> Research Site Waltham, Massachusetts, United States Collapse << |
| NCT01767389 | - | - | Completed | - | - |
| NCT01077323 | - | - | Completed | - | - |
| NCT03585777 | - | - | Not yet recruiting | March 2019 | Egypt ... more >> Fayoum university Hospital Recruiting Fayoum, Egypt Contact: Ahmed Hammad, MD Collapse << |
| NCT02269098 | Diabetes Mellitus, Type 2 ... more >> Medication Adherence HYPERGLYCEMIA Collapse << | Not Applicable | Completed | - | United States, District of Col... more >>umbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 Collapse << |
| NCT02219750 | Diabetes | Phase 4 | Completed | - | Taiwan ... more >> Mackay Memerial Hospital Taipei, Taiwan Collapse << |
| NCT01213563 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United Kingdom ... more >> Imperial College London, Charing Cross Hospital London, United Kingdom, W68NA Collapse << |
| NCT02368704 | - | - | Unknown | November 2016 | France ... more >> Saint-Louis Hospital Paris, France, 75010 Collapse << |
| NCT00015626 | Insulin Resistance ... more >> Diabetes Mellitus Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Cornell University New York, New York, United States, 10021 Collapse << |
| NCT01289990 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00894322 | Type 2 Diabetes Mellitus | Phase 1 Phase 2 | Completed | - | United States, Nebraska ... more >> Research Site Lincoln, Nebraska, United States Collapse << |
| NCT02269098 | - | - | Completed | - | - |
| NCT03552991 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01421459 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01417897 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | May 2012 | Germany ... more >> ife GmbH, Clinic Mainz, Rhineland-Palatinate, Germany, 55116 Collapse << |
| NCT01042379 | Breast Neoplasms ... more >> Breast Cancer Breast Tumors Collapse << | Phase 2 | Recruiting | - | - |
| NCT01121315 | - | - | Completed | - | Sweden ... more >> Research Site Uppsala, Sweden Collapse << |
| NCT01289990 | - | - | Completed | - | - |
| NCT02029846 | Type 2 Diabetes Mellitus | Phase 4 | Terminated(No sufficient numbe... more >>rs of eligible participants) Collapse << | - | United States, Maryland ... more >> Bay West Endocrinology Associates Baltimore, Maryland, United States, 21204 Collapse << |
| NCT02029846 | - | - | Terminated(No sufficient numbe... more >>rs of eligible participants) Collapse << | - | - |
| NCT00894322 | - | - | Completed | - | - |
| NCT01421459 | - | - | Completed | - | - |
| NCT02453685 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Australia, New South Wales ... more >> Novo Nordisk Investigational Site Coffs Harbour, New South Wales, Australia, 2450 Bulgaria Novo Nordisk Investigational Site Petrich, Bulgaria, 2850 Hungary Novo Nordisk Investigational Site Budapest, Hungary, 1042 India Novo Nordisk Investigational Site New Delhi, India, 110001 Korea, Republic of Novo Nordisk Investigational Site Seoul, Korea, Republic of, 138-736 Serbia Novo Nordisk Investigational Site Belgrade, Serbia, 11080 Thailand Novo Nordisk Investigational Site Bangkok, Thailand, 10330 Turkey Novo Nordisk Investigational Site Istanbul, Turkey, 34752 United Arab Emirates Novo Nordisk Investigational Site Ras Al Khaimah, United Arab Emirates, 4727 Collapse << |
| NCT01935466 | - | - | Recruiting | July 2021 | India ... more >> Deptt of Endocrinology Recruiting Chandigarh, India, 160012 Contact: Ashu Rastogi, MD, DM 919781001046 ashuendo@gmail.com Principal Investigator: Ashu Rastogi, MD, DM Collapse << |
| NCT01940302 | Type 2 Diabetes | Early Phase 1 | Completed | - | China, Guangdong ... more >> Shaoguan University Shaoguan, Guangdong, China, 512005 Collapse << |
| NCT02805283 | - | - | Completed | - | United States, Minnesota ... more >> Reasearch Site Eden Prairie, Minnesota, United States Collapse << |
| NCT01270191 | Type 2 Diabetes | Phase 4 | Unknown | December 2015 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 11217 Contact: Harn-Shen Chen, MD, PhD 886-2-28757515 chenhs@vghtpe.gov.tw Principal Investigator: Harn-Shen Chen, MD, PhD Collapse << |
| NCT00481429 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United Kingdom ... more >> Addenbrooke's Hospital Cambridge, United Kingdom, CB2 2GG Ealing Hospital London, United Kingdom, UB1 3HW Imperial College London - Hammersmith Campus London, United Kingdom, W12 ONN Charing Cross Hospital London, United Kingdom, W6 8RF Collapse << |
| NCT00790348 | - | - | Unknown | - | United States, Michigan ... more >> Wayne State University Recruiting Detroit, Michigan, United States, 48201 Contact: Berhane Seyoum, MD 313-745-4008 bseyoum@med.wayne.edu Principal Investigator: Berhane Seyoum, MD, MPH Collapse << |
| NCT02417012 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Denmark ... more >> Center for Physical Activity Research, Copenhagen University Hospital Copenhagen, Denmark, 2100 Collapse << |
| NCT01759823 | Type 2 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | India ... more >> Postgraduate Institute of Medical Education and Research Chandigarh, UT, India, 160012 Collapse << |
| NCT02060383 | Cushing's Disease ... more >> Acromegaly Collapse << | Phase 4 | Completed | - | - |
| NCT03172494 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Recruiting | July 29, 2019 | - |
| NCT01584232 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 448-0852 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 286-0048 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ehime, Japan, 790-0024 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 819-0168 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 0530018 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kagoshima, Japan, 8910401 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 247-0055 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kumamoto, Japan, 860-0811 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 606-8397 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 385-0022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagasaki, Japan, 857-1195 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ooita, Japan, 8700039 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 598-0048 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 103-0028 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Yamaguchi, Japan, 754-0002 Collapse << |
| NCT01095822 | Diabetes Mellitus | Phase 4 | Unknown | March 2011 | United States, Maryland ... more >> Dr. Pokov's Polyclinic. 6821 Reisterstown Road Suite 206 Baltimore, Maryland, United States, 21215 Collapse << |
| NCT02244164 | Type 2 Diabetes ... more >> Incretinomimetics Pancreas Collapse << | Not Applicable | Recruiting | - | Belgium ... more >> Gastroenterology Department Erasme Hospital Recruiting Brussels, Belgium, 1070 Contact: Vincent Huberty, MD +3225553715 vincent.huberty@erasme.ulb.ac.be Collapse << |
| NCT01584232 | - | - | Completed | - | - |
| NCT01785043 | DIABETES Mellitus Type 2 Not W... more >>ell Controlled Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Clínic de Barcelona Barcelona, Spain, 08036 Collapse << |
| NCT01586897 | Diabetes Mellitus, Type 2 ... more >> Hypertension Hyperlipidemia Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT02917629 | Oral Cavity Neoplasm ... more >> Oropharyngeal Neoplasm Stage 0 Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage 0 Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage I Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage I Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage II Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage II Oropharyngeal Squamous Cell Carcinoma AJCC v6 and v7 Stage III Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage III Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVA Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVA Oropharyngeal Squamous Cell Carcinoma AJCC v7 Stage IVB Oral Cavity Squamous Cell Carcinoma AJCC v6 and v7 Stage IVB Oropharyngeal Squamous Cell Carcinoma AJCC v7 Collapse << | Phase 2 | Recruiting | March 31, 2019 | United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Recruiting Baltimore, Maryland, United States, 21287 Contact: Wayne M. Koch 410-995-4906 wkoch@jhmi.edu Principal Investigator: Wayne M. Koch United States, Minnesota University of Minnesota/Masonic Cancer Center Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Frank G. Ondrey 612-625-3200 ondre002@umn.edu Principal Investigator: Frank G. Ondrey United States, New York University of Rochester Recruiting Rochester, New York, United States, 14642 Contact: Shawn D. Newlands 585-273-1943 Shawn_Newlands@URMC.Rochester.edu Principal Investigator: Shawn D. Newlands United States, Wisconsin University of Wisconsin Hospital and Clinics Recruiting Madison, Wisconsin, United States, 53792 Contact: Aaron M. Wieland 608-265-8207 wieland@surgery.wisc.edu Principal Investigator: Aaron M. Wieland Collapse << |
| NCT00819325 | Coronary Artery Disease ... more >> Angina Pectoris Type 2 Diabetes Mellitus Percutaneous Coronary Intervention Collapse << | Phase 4 | Completed | - | Canada, Nova Scotia ... more >> Queen Elizabeth II Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 3A7 Collapse << |
| NCT01586897 | - | - | Completed | - | - |
| NCT02110979 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | United States, Illinois ... more >> EPI-Q Inc Oak Brook, Illinois, United States, 60523 EPI-Q Oak Brook, Illinois, United States, 60523 Collapse << |
| NCT00990444 | Diabetes Mellitus | Phase 1 Phase 2 | Suspended | December 2010 | Sweden ... more >> Karolinska Trial Alliance, Phase I Unit Stockholm, Solna, Stockholm, Sweden, 17176 Collapse << |
| NCT03173391 | Diabetes Mellitus, Type 2 | Phase 3 | Recruiting | December 31, 2019 | China, Shanghai ... more >> Hua Medicine Limited Recruiting Shanghai, Shanghai, China, 201203 Contact: YI ZHANG, PhD +86-021-58863005 yzhang@huamedicine.com Principal Investigator: Dalong Zhu, PhD Collapse << |
| NCT03659461 | Type 2 Diabetes Mellitus ... more >> PreDiabetes Collapse << | Not Applicable | Recruiting | January 2020 | Malaysia ... more >> Hospital Sultan Ismail Recruiting Johor Bahru, Johor, Malaysia, 81100 Contact: Chong Shiau Chin, MPHARM +6073565000 shiauchin83@hotmail.com Contact +60361455555 Collapse << |
| NCT01781754 | - | - | Unknown | January 2014 | Israel ... more >> Tel Aviv Sourasky Medical Center Recruiting Tel-Aviv, Israel, 64239 Contact: Nachum Vaisman, MD 972-3-6974807 vaisman@tasmc.health.gov.il Principal Investigator: Nachum Vaisman, MD Collapse << |
| NCT02298192 | - | - | Completed | - | - |
| NCT02298192 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00133718 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Norway ... more >> Asker and Baerum Hospital, Medical Department RUD, Norway, 1309 Collapse << |
| NCT02616666 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | December 31, 2021 | - |
| NCT03511118 | - | - | Recruiting | September 22, 2019 | United States, North Carolina ... more >> Duke University Maternal and Fetal Medicine Recruiting Durham, North Carolina, United States, 27705 Contact: Kristin Weaver 919-613-9630 obresearch@duke.edu Principal Investigator: Brenna Hughes, MD United States, Washington University of Washington Recruiting Seattle, Washington, United States, 98195-7630 Contact: Maggie Leahy 540-525-0121 maleahy@u.washington.edu Principal Investigator: Mary Hébert, Pharm.D Collapse << |
| NCT02582242 | Diabetes Type... more >> 2 Diabetes Mellitus Collapse << | Phase 4 | Completed | - | - |
| NCT03596450 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | November 19, 2021 | United States, California ... more >> Novo Nordisk Investigational Site Not yet recruiting Monterey, California, United States, 93940 Novo Nordisk Investigational Site Not yet recruiting San Jose, California, United States, 95148 Novo Nordisk Investigational Site Not yet recruiting Tulare, California, United States, 93274 United States, Connecticut Novo Nordisk Investigational Site Not yet recruiting Milford, Connecticut, United States, 06460 United States, Georgia Novo Nordisk Investigational Site Not yet recruiting Lilburn, Georgia, United States, 30047 Novo Nordisk Investigational Site Not yet recruiting Sandersville, Georgia, United States, 31082 Novo Nordisk Investigational Site Not yet recruiting Savannah, Georgia, United States, 31406 Novo Nordisk Investigational Site Recruiting Statesboro, Georgia, United States, 30461 Novo Nordisk Investigational Site Not yet recruiting Stockbridge, Georgia, United States, 30281 Novo Nordisk Investigational Site Not yet recruiting Swainsboro, Georgia, United States, 30401 United States, Kentucky Novo Nordisk Investigational Site Not yet recruiting Covington, Kentucky, United States, 41011 Novo Nordisk Investigational Site Not yet recruiting Owensboro, Kentucky, United States, 42303 United States, Virginia Novo Nordisk Investigational Site Not yet recruiting Chatham, Virginia, United States, 24531 Novo Nordisk Investigational Site Not yet recruiting Colonial Heights, Virginia, United States, 23834 Novo Nordisk Investigational Site Not yet recruiting Falls Church, Virginia, United States, 22044 Novo Nordisk Investigational Site Not yet recruiting Lynchburg, Virginia, United States, 24501 Novo Nordisk Investigational Site Not yet recruiting Warrenton, Virginia, United States, 20186 Collapse << |
| NCT02582242 | - | - | Completed | - | - |
| NCT00802971 | Hypoglycemia | Not Applicable | Completed | - | - |
| NCT00691314 | Stroke Transi... more >>ent Ischemic Attack Atherosclerosis Stenosis Stents Collapse << | Not Applicable | Completed | - | China, Beijing ... more >> Beijing Tiantan Hospital Beijing, Beijing, China, 100050 Xuanwu Hospital Capital Medical University Beijing, Beijing, China, 100053 Peking Union Medical College Hospital Beijing, Beijing, China, 100730 China, Chongqing Southwest Hospital Chongqing, Chongqing, China, 400038 Daping Hospital Chongqing, Chongqing, China, 400042 China, Jiangsu Nanjing General Hospital of Nanjing Military Command Nanjing, Jiangsu, China, 210002 China, Jilin Jilin University Changchun, Jilin, China, 130021 China, Shanghai Huashan Hospital Fudan University Shanghai, Shanghai, China, 200040 China, Sichuan West China Center of Medical Sciences Chengdu, Sichuan, China, 610041 China, Tianjin The General Hospital Under Tianjin Medical Sciences University Tianjin, Tianjin, China, 300052 Collapse << |
| NCT01065298 | - | - | Completed | - | India ... more >> Post Graduate Institute of Medical Education and Research Chandigarh, Chandigarh UT, India Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
