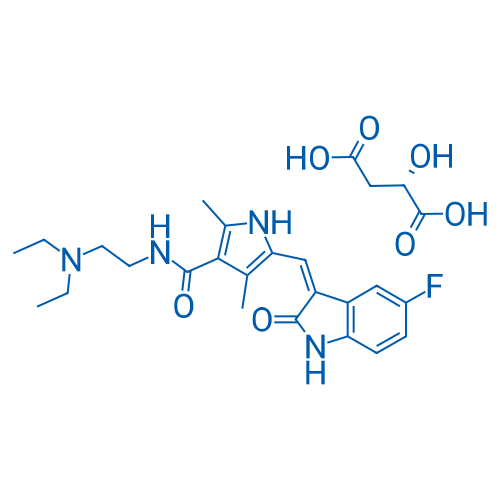
CAS No.: 341031-54-7
苹果酸舒尼替尼 Catalog No. CSN12411
Synonyms: SU 11248 Malate;Sunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib Malate;Sunitinib (malate)
Sunitinib Malate is a multi-targeted RTK inhibitor targeting VEGFR2 (Flk-1) and PDGFRβ with IC50 of 80 nM and 2 nM, and also inhibits c-Kit.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 PDGFRβ
IC50:2nMVEGFR2
IC50:80nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Rat: 0.5 mg/kg[5] (p.o.), 1 mg/kg - 10 mg/kg[6] (p.o.) Mice: 40 mg/kg[7] (p.o.)
给药途径 p.o.
动物药代数据 Animal Rats[8] Dose 2 mg/kg (i.v. or p.o.) Administration i.v.
p.o.F 112 ± 34% (p.o.) T1/2 2.52 ± 0.04 h (i.v.) AUCtlast 896 ± 461 ng·h/ml (p.o.) Tmax 2.0 ± 2.0 h (p.o.) Cmax 177 ± 86 ng/ml (p.o.) AUC0→∞ 996 ± 439 ng·h/ml (i.v.)
1178 ± 691 ng·h/ml (p.o.)CLs 40 ± 23 ml/min/kg (i.v.) - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00663559 Carcinoma Renal Cells Phase 2 Completed - Spain ... more >> Hospital Central de Asturias Oviedo, Asturias, Spain, 33006 Hospital Parc Taulí Sabadell, Barcelona, Spain, 08208 Hospital CLINIC Barcelona, Spain, 08036 Hospital Reina Sofía Córdoba, Spain, 14004 Hospital de Jaén Jaén, Spain, 23007 Hospital Clínico San Carlos Madrid, Spain, 28040 Hospital lozano Blesa Zaragoza, Spain, 50009 Collapse << NCT03097601 - - Active, not recruiting December 30, 2019 France ... more >> Centre Antoine LACASSAGNE Nice, France, 06189 Collapse << NCT01243359 Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Cancer Stage I Renal Cell Cancer Stage II Renal Cell Cancer Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << Phase 1 Completed - United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << - 更多
- 参考文献
- [1] Shukla S, Robey RW, et al. Sunitinib (Sutent, SU11248), a small-molecule receptor tyrosine kinase inhibitor, blocks function of the ATP-binding cassette (ABC) transporters P-glycoprotein (ABCB1) and ABCG2. Drug Metab Dispos. 2009 Feb;37(2):359-65.
- [2] Sun L, Liang C, et al. Discovery of 5-[5-fluoro-2-oxo-1,2- dihydroindol-(3Z)-ylidenemethyl] -2,4- dimethyl-1H-pyrrole-3-carboxylic acid (2-diethylaminoethyl)amide, a novel tyrosine kinase inhibitor targeting vascular endothelial and platelet-derived growth factor receptor tyrosine kinase. J Med Chem. 2003 Mar 27;46(7):1116-9.
- [3] Mendel DB, Laird AD, et al. In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res. 2003 Jan;9(1):327-37.
- [4] Zhou Q, Gallo JM. Differential effect of sunitinib on the distribution of temozolomide in an orthotopic glioma model. Neuro Oncol. 2009;11(3):301-10.
- [5] sunitinib malate
- [6] Blasi E, Heyen J, et al. Sunitinib, a receptor tyrosine kinase inhibitor, increases blood pressure in rats without associated changes in cardiac structure and function. Cardiovasc Ther. 2012 Oct;30(5):287-94.
- [7] Gotink KJ, Broxterman HJ, et al. Acquired tumor cell resistance to sunitinib causes resistance in a HT-29 human colon cancer xenograft mouse model without affecting sunitinib biodistribution or the tumor microvasculature. Oncoscience. 2014 Dec 15;1(12):844-53. eCollection 2014.
- [8] Sutent
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 341031-54-7 | 储存条件 |
|
|||||||||
| 分子式 | C26H33FN4O7 | 运输 | 蓝冰 | |||||||||
| 分子量 | 532.56 | 别名 | SU 11248 Malate;Sunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib MalateSunitinib Malate;Sunitinib (malate);SU-11248;SU011248;Sutent | |||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 3T3 | - | Kinase Assay | - | Inhibition of PDGF-induced BrdU incorporation with IC50 of 0.007 μM | 12646019 |
| 3T3 | - | Growth Inhibition Assay | - | Inhibition of Platelet-derived growth factor induced 3T3 cell proliferation with IC50 of 0.01 μM | 12646019 |
| 3T3 | - | Function Assay | - | Inhibition of Vascular endothelial growth factor receptor with IC50 of 0.05 μM | 12646019 |
| 3T3 | - | Kinase Assay | 20 min | Cellular inhibition of VEGF induced human KDR phosphorylation with IC50 of 0.022 μM | 16162008 |
| 4T1 | - | Cytotoxic Assay | 72 h | IC50=0.016 μM | 24890652 |
| 697 | - | Growth Inhibition Assay | - | IC50=614.25 nM | SANGER |
| 8-MG-BA | - | Growth Inhibition Assay | - | IC50=6.51507 μM | SANGER |
| A101D | - | Growth Inhibition Assay | - | IC50=2.78304 μM | SANGER |
| A375 | - | Cytotoxic Assay | 72 h | IC50=5.4 μM | 19654408 |
| A388 | - | Growth Inhibition Assay | - | IC50=28.0074 μM | SANGER |
| A3-KAW | - | Growth Inhibition Assay | - | IC50=1.62546 μM | SANGER |
| A431 | - | Kinase Assay | - | Inhibition of PDGFRbeta expressed with IC50 of 12.2 μM | 20558072 |
| A431 | - | Kinase Assay | - | Inhibition of VEGFR2 expressed with IC50 of 18.9 μM | 20558072 |
| A431 | 10 μM | Kinase Assay | 1 h | Inhibition of EGFR with IC50 of 0.1721 μM | 24890652 |
| A498 | 100 μM | Cytotoxic Assay | 72 h | IC50=4.3 μM | 23489626 |
| A4-Fuk | - | Growth Inhibition Assay | - | IC50=1.34141 μM | SANGER |
| A549 | - | Function Assay | - | Inhibition of c-Met dependent HGF-induced human A549 cell migration with IC50 of 2 μM | 18434145 |
| A549 | - | Growth Inhibition Assay | 16 h | Antitumor activity against human A549 cells | 21450463 |
| A549 | - | Cytotoxic Assay | 72 h | IC50=2.44 μM | 23602441 |
| A704 | - | Growth Inhibition Assay | - | IC50=17.9904 μM | SANGER |
| ACHN | - | Cytotoxic Assay | 6 d | IC50=2.5 μM | 23360104 |
| ALL-PO | - | Growth Inhibition Assay | - | IC50=79.89 nM | SANGER |
| ARH-77 | - | Growth Inhibition Assay | - | IC50=7.53597 μM | SANGER |
| ATN-1 | - | Growth Inhibition Assay | - | IC50=3.67114 μM | SANGER |
| Ba/F3 | - | Growth Inhibition Assay | 72 h | IC50=1.2 μM | 20117004 |
| BaPTC2 | - | Growth Inhibition Assay | 72 h | IC50=0.22 μM | 20117004 |
| BB30-HNC | - | Growth Inhibition Assay | - | IC50=2.22375 μM | SANGER |
| BB49-HNC | - | Growth Inhibition Assay | - | IC50=3.70508 μM | SANGER |
| BB65-RCC | - | Growth Inhibition Assay | - | IC50=18.4162 μM | SANGER |
| BC-1 | - | Growth Inhibition Assay | - | IC50=7.24812 μM | SANGER |
| BC-3 | - | Growth Inhibition Assay | - | IC50=12.4651 μM | SANGER |
| BE-13 | - | Growth Inhibition Assay | - | IC50=3.23111 μM | SANGER |
| Bel7402 | - | Growth Inhibition Assay | 72 h | IC50=2.67 μM | 24904961 |
| BGC | - | Growth Inhibition Assay | 72 h | IC50=4.78 μM | 23999040 |
| BL-41 | - | Growth Inhibition Assay | - | IC50=2.91222 μM | SANGER |
| BL-70 | - | Growth Inhibition Assay | - | IC50=1.11846 μM | SANGER |
| BV-173 | - | Growth Inhibition Assay | - | IC50=2.3366 μM | SANGER |
| BxPC3 | - | Growth Inhibition Assay | 72 h | IC50=3.63 μM | 23999040 |
| CA46 | - | Growth Inhibition Assay | - | IC50=7.58012 μM | SANGER |
| CAKI-1 | 100 μM | Growth Inhibition Assay | 48 h | GI50=0.63 μM | 22560627 |
| CCRF-CEM | - | Growth Inhibition Assay | - | IC50=14.4169 μM | SANGER |
| CESS | - | Growth Inhibition Assay | - | IC50=7.10438 μM | SANGER |
| CGTH-W-1 | - | Growth Inhibition Assay | - | IC50=30.94 nM | SANGER |
| CI-1 | - | Growth Inhibition Assay | - | IC50=8.20411 μM | SANGER |
| CMK | - | Growth Inhibition Assay | - | IC50=1.95517 μM | SANGER |
| COLO-320-HSR | - | Growth Inhibition Assay | - | IC50=12.1565 μM | SANGER |
| COLO-668 | - | Growth Inhibition Assay | - | IC50=30.792 μM | SANGER |
| COR-L279 | - | Growth Inhibition Assay | - | IC50=9.3909 μM | SANGER |
| COR-L88 | - | Growth Inhibition Assay | - | IC50=16.0012 μM | SANGER |
| CPC-N | - | Growth Inhibition Assay | - | IC50=8.29199 μM | SANGER |
| CTB-1 | - | Growth Inhibition Assay | - | IC50=13.7294 μM | SANGER |
| CTV-1 | - | Growth Inhibition Assay | - | IC50=1.72751 μM | SANGER |
| D-283MED | - | Growth Inhibition Assay | - | IC50=16.8489 μM | SANGER |
| Daudi | - | Growth Inhibition Assay | - | IC50=8.23546 μM | SANGER |
| DEL | - | Growth Inhibition Assay | - | IC50=2.08482 μM | SANGER |
| DG-75 | - | Growth Inhibition Assay | - | IC50=6.43069 μM | SANGER |
| DOHH-2 | - | Growth Inhibition Assay | - | IC50=2.33431 μM | SANGER |
| DU145 | - | Function Assay | - | Inhibition of c-Met dependent HGF-induced human DU145 cell scattering with IC50 of 10 μM | 18434145 |
| DU-4475 | - | Growth Inhibition Assay | - | IC50=4.36729 μM | SANGER |
| EB-3 | - | Growth Inhibition Assay | - | IC50=29.5309 μM | SANGER |
| EHEB | - | Growth Inhibition Assay | - | IC50=34.1193 μM | SANGER |
| EKVX | 100 μM | Growth Inhibition Assay | 48 h | GI50=7.9 μM | 22560627 |
| EM-2 | - | Growth Inhibition Assay | - | IC50=18.5672 μM | SANGER |
| EMG-01 | - | Growth Inhibition Assay | - | IC50=1.83563 μM | SANGER |
| EoL-1-cell | - | Growth Inhibition Assay | - | IC50=1.64 pM | SANGER |
| ES1 | - | Growth Inhibition Assay | - | IC50=10.2984 μM | SANGER |
| ES3 | - | Growth Inhibition Assay | - | IC50=37.5004 μM | SANGER |
| ES4 | - | Growth Inhibition Assay | - | IC50=2.80628 μM | SANGER |
| ES5 | - | Growth Inhibition Assay | - | IC50=24.7349 μM | SANGER |
| ES6 | - | Growth Inhibition Assay | - | IC50=981.06 nM | SANGER |
| ETK-1 | - | Growth Inhibition Assay | - | IC50=1.2858 μM | SANGER |
| EVSA-T | - | Growth Inhibition Assay | - | IC50=17.0485 μM | SANGER |
| EW-1 | - | Growth Inhibition Assay | - | IC50=4.67911 μM | SANGER |
| EW-11 | - | Growth Inhibition Assay | - | IC50=34.4725 μM | SANGER |
| EW-16 | - | Growth Inhibition Assay | - | IC50=2.13508 μM | SANGER |
| EW-3 | - | Growth Inhibition Assay | - | IC50=6.43207 μM | SANGER |
| GB-1 | - | Growth Inhibition Assay | - | IC50=710.23 nM | SANGER |
| GDM-1 | - | Growth Inhibition Assay | - | IC50=21.9414 μM | SANGER |
| GOTO | - | Growth Inhibition Assay | - | IC50=31.6085 μM | SANGER |
| GR-ST | - | Growth Inhibition Assay | - | IC50=10.2207 μM | SANGER |
| GT3TKB | - | Growth Inhibition Assay | - | IC50=6.60763 μM | SANGER |
| H4 | 10 μM | Cytotoxic Assay | - | Toxicity in human H4 cells | 20350806 |
| H460 | - | Cytotoxic Assay | 72 h | IC50=2.7 μM | 21621880 |
| HAEC | 100 μM | Growth Inhibition Assay | 72 h | Antiproliferative activity against human HAEC cells expressing VEGFR with IC50 of 0.1 μM | 22444679 |
| HAEC | 100 μM | Cytotoxic Assay | 72 h | Cytotoxicity against VEGFR expressing HAEC with IC50 of 0.1 μM | 23131541 |
| HAL-01 | - | Growth Inhibition Assay | - | IC50=2.05946 μM | SANGER |
| HC-1 | - | Growth Inhibition Assay | - | IC50=5.43291 μM | SANGER |
| HCC1187 | - | Growth Inhibition Assay | - | IC50=18.0187 μM | SANGER |
| HCC1599 | - | Growth Inhibition Assay | - | IC50=8.19987 μM | SANGER |
| HCE-4 | - | Growth Inhibition Assay | - | IC50=3.78642 μM | SANGER |
| HCT116 | - | Function Assay | 24 h | Antimigratory activity assessed as inhibition of cell migration | 23153200 |
| HCT116 | - | Cytotoxic Assay | 72 h | IC50=4.71 μM | 23602441 |
| HDLM-2 | - | Growth Inhibition Assay | - | IC50=6.32109 μM | SANGER |
| HD-MY-Z | - | Growth Inhibition Assay | - | IC50=5.17301 μM | SANGER |
| HEK293 | 100 nM | Kinase Assay | 1 h | does not inhibit VEGF-induced autophosphorylation of tyrosine 1175 residue on VEGFR2 expressed in HEK293 cells | 21885287 |
| HEL | - | Growth Inhibition Assay | - | IC50=3.48005 μM | SANGER |
| HepG2 | - | Growth Inhibition Assay | 72 h | IC50=3.81 μM | 20570526 |
| HepG2 | - | Cytotoxic Assay | 48 h | IC50=13.24 μM | 22483608 |
| HH | - | Growth Inhibition Assay | - | IC50=4.20914 μM | SANGER |
| HK2 | - | Growth Inhibition Assay | 72 h | IC50=5.85 μM | 24904961 |
| HL60 | - | Growth Inhibition Assay | 16 h | Antitumor activity against human HL60 cells | 21450463 |
| HL60 | 50 μM | Cytotoxic Assay | 48 h | IC50=15.5 μM | 22019188 |
| HL-60 | - | Growth Inhibition Assay | - | IC50=4.21065 μM | SANGER |
| HOP-62 | - | Growth Inhibition Assay | - | IC50=4.3352 μM | SANGER |
| HT-144 | - | Growth Inhibition Assay | - | IC50=28.69 μM | SANGER |
| HT-29 | 100 μM | Growth Inhibition Assay | 72 h | Antiproliferative activity against human HT-29 cells expressing VEGFR with IC50 of 0.33 μM | 22444679 |
| HT-29 | 100 μM | Cytotoxic Assay | 72 h | Cytotoxicity against VEGFR expressing human HT-29 cells with IC50 of 0.33 μM | 23131541 |
| HT-29 | - | Growth Inhibition Assay | 72 h | IC50=1.47 μM | 23999040 |
| HUTU-80 | - | Growth Inhibition Assay | - | IC50=13.7573 μM | SANGER |
| HUVEC | - | Growth Inhibition Assay | 16 h | Inhibition of VEGF-induced cell proliferation with IC50 of 2.75 μM | 21450463 |
| HUVEC | - | Growth Inhibition Assay | 16 h | Inhibition of bFGF-induced cell proliferation with IC50 of 4.04 μM | 21450463 |
| HUVEC | 1 μM | Function Assay | 24 h | Antiangiogenic activity assessed as decrease in VEGF-induced cell migration | 21963305 |
| HUVEC | 1 μM | Function Assay | 1 h | Inhibition of ERK phosphorylation at Thr202/Tyr204 in VEGF-stimulated HUVEC | 21963305 |
| HUVEC | 1 μM | Function Assay | 1 h | Inhibition of eNOS phosphorylation at Ser-1172 in VEGF-stimulated HUVEC | 21963305 |
| HUVEC | 1 μM | Kinase Assay | 1 h | Inhibition of VEGFR2 phosphorylation at tyr-1175 in VEGF-stimulated HUVEC | 21963305 |
| HUVEC | 1 μM | Function Assay | 1 h | does not inhibit AKT phosphorylation at Ser-473 in VEGF-stimulated HUVEC | 21963305 |
| HUVEC | 2 μM | Function Assay | 18 h | Antiangiogenic activity in HUVECs under hypoxic condition assessed as inhibition of tube formation | 23153200 |
| HUVEC | 3 μM | Growth Inhibition Assay | 24 h | Antiangiogenic activity assessed as inhibition of VEGF-induced tube formation with EC50 of 0.645 μM | 23583911 |
| HUVEC | 200 μM | Kinase Assay | - | Inhibition of VEGFR2 phosphorylation in growth factor-stimulated HUVEC | 23583911 |
| HUVEC | 200 μM | Kinase Assay | - | Inhibition of VEGFR1 phosphorylation in growth factor-stimulated HUVEC | 23583911 |
| IM9 | - | Growth Inhibition Assay | 16 h | Antitumor activity against human IM9 cells | 21450463 |
| IMR-5 | - | Growth Inhibition Assay | - | IC50=7054876 μM | SANGER |
| IST-MEL1 | - | Growth Inhibition Assay | - | IC50=12.8694 μM | SANGER |
| IST-MES1 | - | Growth Inhibition Assay | - | IC50=13.6729 μM | SANGER |
| IST-SL1 | - | Growth Inhibition Assay | - | IC50=8.02417 μM | SANGER |
| IST-SL2 | - | Growth Inhibition Assay | - | IC50=6.12519 μM | SANGER |
| JAR | - | Growth Inhibition Assay | - | IC50=3.98832 μM | SANGER |
| JiyoyeP-2003 | - | Growth Inhibition Assay | - | IC50=26.2742 μM | SANGER |
| J-RT3-T3-5 | - | Growth Inhibition Assay | - | IC50=6.07909 μM | SANGER |
| JVM-2 | - | Growth Inhibition Assay | - | IC50=28.2898 μM | SANGER |
| K052 | - | Growth Inhibition Assay | - | IC50=2.40202 μM | SANGER |
| K5 | - | Growth Inhibition Assay | - | IC50=12.2985 μM | SANGER |
| K562 | - | Growth Inhibition Assay | 16 h | Antitumor activity against human K562 cells | 21450463 |
| K562 | 50 μM | Cytotoxic Assay | 48 h | IC50=21.9 μM | 22019188 |
| K562 | - | Growth Inhibition Assay | - | IC50=11.8702 μM | SANGER |
| KALS-1 | - | Growth Inhibition Assay | - | IC50=16.5931 μM | SANGER |
| KARPAS-299 | - | Growth Inhibition Assay | - | IC50=2.11313 μM | SANGER |
| KARPAS-422 | - | Growth Inhibition Assay | - | IC50=7.57206 μM | SANGER |
| KARPAS-45 | - | Growth Inhibition Assay | - | IC50=2.49446 μM | SANGER |
| Kasumi-1 | - | Growth Inhibition Assay | 72 h | IC50=0.016 μM | 20570526 |
| Kasumi-1 | - | Function Assay | - | Inhibition of c-Kit autophosphorylation with IC50 of 0.015 μM | 20833039 |
| KB3-1 | - | Cytotoxic Assay | 72 h | Cytotoxicity against human P-gp-negative KB-3-1 cells with IC50 of 2.3 μM | 19397322 |
| KBV1 | - | Cytotoxic Assay | 72 h | Cytotoxicity against human P-glycoprotein-expressing KBV1 cells with IC50 of 4.1 μM | 19397322 |
| KE-37 | - | Growth Inhibition Assay | - | IC50=4.06158 μM | SANGER |
| KG-1 | - | Growth Inhibition Assay | - | IC50=6.02878 μM | SANGER |
| KGN | - | Growth Inhibition Assay | - | IC50=2.60339 μM | SANGER |
| KLE | - | Growth Inhibition Assay | - | IC50=29.619 μM | SANGER |
| KM12 | - | Growth Inhibition Assay | - | IC50=350.14 nM | SANGER |
| KM-H2 | - | Growth Inhibition Assay | - | IC50=44.9577 μM | SANGER |
| KMOE-2 | - | Growth Inhibition Assay | - | IC50=8.1942 μM | SANGER |
| KMS-12-PE | - | Growth Inhibition Assay | - | IC50=22.274 μM | SANGER |
| KP-N-YS | - | Growth Inhibition Assay | - | IC50=14.2894 μM | SANGER |
| KS-1 | - | Growth Inhibition Assay | - | IC50=1.69247 μM | SANGER |
| KU812 | - | Growth Inhibition Assay | - | IC50=6.6942 μM | SANGER |
| KY821 | - | Growth Inhibition Assay | - | IC50=2.79758 μM | SANGER |
| L-363 | - | Growth Inhibition Assay | - | IC50=4.80621 μM | SANGER |
| L-428 | - | Growth Inhibition Assay | - | IC50=18.0151 μM | SANGER |
| LAMA-84 | - | Growth Inhibition Assay | - | IC50=2.69545 μM | SANGER |
| LAN-6 | - | Growth Inhibition Assay | - | IC50=14.2457 μM | SANGER |
| LB1047-RCC | - | Growth Inhibition Assay | - | IC50=1.81624 μM | SANGER |
| LB831-BLC | - | Growth Inhibition Assay | - | IC50=32.3843 μM | SANGER |
| LB996-RCC | - | Growth Inhibition Assay | - | IC50=4.12168 μM | SANGER |
| LC-1F | - | Growth Inhibition Assay | - | IC50=43.3682 μM | SANGER |
| LC-2-ad | - | Growth Inhibition Assay | - | IC50=1.11407 μM | SANGER |
| LC4-1 | - | Growth Inhibition Assay | - | IC50=4.3807 μM | SANGER |
| LO2 | - | Growth Inhibition Assay | 72 h | IC50=19.93 μM | 24904961 |
| LOUCY | - | Growth Inhibition Assay | - | IC50=11.9875 μM | SANGER |
| LOXIMVI | - | Growth Inhibition Assay | - | IC50=16.256 μM | SANGER |
| LP-1 | - | Growth Inhibition Assay | - | IC50=9.78083 μM | SANGER |
| LS-123 | - | Growth Inhibition Assay | - | IC50=9.68161 μM | SANGER |
| LXF-289 | - | Growth Inhibition Assay | - | IC50=2.72989 μM | SANGER |
| MC116 | - | Growth Inhibition Assay | - | IC50=4.43081 μM | SANGER |
| MC-CAR | - | Growth Inhibition Assay | - | IC50=8.3358 μM | SANGER |
| MCF7 | 100 μM | Growth Inhibition Assay | 48 h | GI51=2 μM | 22560627 |
| MCF7 | - | Cytotoxic Assay | 72 h | IC50=6.29 μM | 23602441 |
| MCF7 | - | Cytotoxic Assay | 72 h | IC50=0.0271 μM | 24890652 |
| MCF7 | - | Cytotoxic Assay | 72 h | IC50=0.0293 μM | 24890652 |
| MDA-MB-231 | - | Growth Inhibition Assay | 16 h | Antitumor activity against human MDA-MB-231 cells | 21450463 |
| MDA-MB-231 | - | Cytotoxic Assay | 72 h | IC50=0.0223 μM | 24890652 |
| MDA-MB-435 | 100 μM | Growth Inhibition Assay | 48 h | GI52=2 μM | 22560627 |
| MDA-MB-435 | - | Cytotoxic Assay | 72 h | IC50=0.0097 μM | 24890652 |
| MDA-MB-468 | - | Cytotoxic Assay | 72 h | IC50=0.0061 μM | 24890652 |
| MHH-CALL-2 | - | Growth Inhibition Assay | - | IC50=5.76719 μM | SANGER |
| MHH-NB-11 | - | Growth Inhibition Assay | - | IC50=20.5521 μM | SANGER |
| MHH-PREB-1 | - | Growth Inhibition Assay | - | IC50=5.32312 μM | SANGER |
| ML-2 | - | Growth Inhibition Assay | - | IC50=2.63512 μM | SANGER |
| MLMA | - | Growth Inhibition Assay | - | IC50=16.9907 μM | SANGER |
| MMAC-SF | - | Growth Inhibition Assay | - | IC50=6.10949 μM | SANGER |
| MN-60 | - | Growth Inhibition Assay | - | IC50=7.54088 μM | SANGER |
| MOLT-16 | - | Growth Inhibition Assay | - | IC50=631.32 nM | SANGER |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=3.39838 μM | SANGER |
| MONO-MAC-6 | - | Growth Inhibition Assay | - | IC50=33.8 nM | SANGER |
| MPP-89 | - | Growth Inhibition Assay | - | IC50=42.0586 μM | SANGER |
| MS-1 | - | Growth Inhibition Assay | - | IC50=3.83778 μM | SANGER |
| MSTO-211H | - | Growth Inhibition Assay | - | IC50=31.8678 μM | SANGER |
| MV4-11 | - | Cytotoxic Assay | 72 h | IC50=0.003 μM | 22452518 |
| MV4-11 | - | Growth Inhibition Assay | 48 h | IC50=0.003 μM | 24904961 |
| MV-4-11 | - | Growth Inhibition Assay | - | IC50=272 pM | SANGER |
| MZ1-PC | - | Growth Inhibition Assay | - | IC50=7.30389 μM | SANGER |
| MZ2-MEL | - | Growth Inhibition Assay | - | IC50=15.2251 μM | SANGER |
| NALM-6 | - | Growth Inhibition Assay | - | IC50=2.90473 μM | SANGER |
| NB1 | - | Growth Inhibition Assay | - | IC50=1.96117 μM | SANGER |
| NB13 | - | Growth Inhibition Assay | - | IC50=9.79994 μM | SANGER |
| NB14 | - | Growth Inhibition Assay | - | IC50=10.9277 μM | SANGER |
| NB7 | - | Growth Inhibition Assay | - | IC50=27.9229 μM | SANGER |
| NCCIT | - | Growth Inhibition Assay | - | IC50=8.71745 μM | SANGER |
| NCI-H128 | - | Growth Inhibition Assay | - | IC50=13.0723 μM | SANGER |
| NCI-H1304 | - | Growth Inhibition Assay | - | IC50=8.20661 μM | SANGER |
| NCI-H1355 | - | Growth Inhibition Assay | - | IC50=7.45804 μM | SANGER |
| NCI-H1581 | - | Growth Inhibition Assay | - | IC50=18.0866 μM | SANGER |
| NCI-H1648 | - | Growth Inhibition Assay | - | IC50=9.13834 μM | SANGER |
| NCI-H1650 | - | Growth Inhibition Assay | - | IC50=29.0176 μM | SANGER |
| NCI-H1694 | - | Growth Inhibition Assay | - | IC50=13.1144 μM | SANGER |
| NCI-H1770 | - | Growth Inhibition Assay | - | IC50=14.5369 μM | SANGER |
| NCI-H1882 | - | Growth Inhibition Assay | - | IC50=17.9834 μM | SANGER |
| NCI-H209 | - | Growth Inhibition Assay | - | IC50=8.11652 μM | SANGER |
| NCI-H2107 | - | Growth Inhibition Assay | - | IC50=11.5984 μM | SANGER |
| NCI-H2141 | - | Growth Inhibition Assay | - | IC50=38.0843 μM | SANGER |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=31.1063 μM | SANGER |
| NCI-H3122 | - | Growth Inhibition Assay | 72 h | IC50=0.83 μM | 24904961 |
| NCI-H446 | - | Growth Inhibition Assay | - | IC50=27.4946 μM | SANGER |
| NCI-H460 | - | Growth Inhibition Assay | 72 h | IC50=4.31 μM | 24904961 |
| NCI-H510A | - | Growth Inhibition Assay | - | IC50=24.1278 μM | SANGER |
| NCI-H526 | - | Growth Inhibition Assay | 72 h | IC50=1.01 μM | 24904961 |
| NCI-H719 | - | Growth Inhibition Assay | - | IC50=16.9161 μM | SANGER |
| NCI-H747 | - | Growth Inhibition Assay | - | IC50=28.9195 μM | SANGER |
| NCI-H82 | - | Growth Inhibition Assay | - | IC50=7.3785 μM | SANGER |
| NCI-SNU-1 | - | Growth Inhibition Assay | - | IC50=13.345 μM | SANGER |
| NCI-SNU-16 | - | Growth Inhibition Assay | - | IC50=1.46486 μM | SANGER |
| NEC8 | - | Growth Inhibition Assay | - | IC50=12.6887 μM | SANGER |
| NH-12 | - | Growth Inhibition Assay | - | IC50=43.9359 μM | SANGER |
| NIH3T3 | - | Kinase Assay | 20 min | inhibit human KDR kinase expressed with IC50 of 0.018 μM | 16162008 |
| NKM-1 | - | Growth Inhibition Assay | - | IC50=98.52 nM | SANGER |
| NMC-G1 | - | Growth Inhibition Assay | - | IC50=27.1822 μM | SANGER |
| no-10 | - | Growth Inhibition Assay | - | IC50=21.0264 μM | SANGER |
| no-11 | - | Growth Inhibition Assay | - | IC50=7.77557 μM | SANGER |
| NOMO-1 | - | Growth Inhibition Assay | - | IC50=4.33699 μM | SANGER |
| NOS-1 | - | Growth Inhibition Assay | - | IC50=15.3 nM | SANGER |
| NTERA-S-cl-D1 | - | Growth Inhibition Assay | - | IC50=17.8572 μM | SANGER |
| OCI-AML2 | - | Growth Inhibition Assay | - | IC50=1.36851 μM | SANGER |
| OCUB-M | - | Growth Inhibition Assay | - | IC50=8.56508 μM | SANGER |
| ONS-76 | - | Growth Inhibition Assay | - | IC50=9.81016 μM | SANGER |
| OPM-2 | - | Growth Inhibition Assay | - | IC50=3.05954 μM | SANGER |
| OS-RC-2 | - | Growth Inhibition Assay | - | IC50=4.8731 μM | SANGER |
| OVCAR3 | 100 μM | Growth Inhibition Assay | 48 h | GI53=3.2 μM | 22560627 |
| OVCAR-4 | - | Growth Inhibition Assay | - | IC50=8.50351 μM | SANGER |
| PC3 | 50 μM | Cytotoxic Assay | 48 h | IC50=25.1 μM | 22019188 |
| PC9 | - | Cytotoxic Assay | 48 h | IC50=10.97 μM | 22483608 |
| PF-382 | - | Growth Inhibition Assay | - | IC50=3.4457 μM | SANGER |
| PSN1 | - | Growth Inhibition Assay | - | IC50=1.50676 μM | SANGER |
| QIMR-WIL | - | Growth Inhibition Assay | - | IC50=3.63391 μM | SANGER |
| Raji | - | Growth Inhibition Assay | - | IC50=19.9565 μM | SANGER |
| Ramos-2G6-4C10 | - | Growth Inhibition Assay | - | IC50=11.265 μM | SANGER |
| REH | - | Growth Inhibition Assay | - | IC50=12.6498 μM | SANGER |
| RKO | - | Growth Inhibition Assay | - | IC50=44.1252 μM | SANGER |
| RL95-2 | - | Growth Inhibition Assay | - | IC50=2.11137 μM | SANGER |
| RPMI-6666 | - | Growth Inhibition Assay | - | IC50=3.54171 μM | SANGER |
| RPMI-8226 | - | Growth Inhibition Assay | - | IC50=7.50739 μM | SANGER |
| RPMI-8402 | - | Growth Inhibition Assay | - | IC50=2.33618 μM | SANGER |
| RS4-11 | - | Function Assay | 2 h | Inhibition of FLT3 autophosphorylation with IC50 of 0.0099 μM | 19654408 |
| RS4-11 | - | Function Assay | 2 h | Inhibition of FLT3 ITD mutant autophosphorylation with IC50 of 0.034 μM | 19654408 |
| RS4-11 | - | Growth Inhibition Assay | 72 h | IC50=1 μM | 20570526 |
| RS4-11 | - | Growth Inhibition Assay | - | IC50=2.17924 μM | SANGER |
| RXF393 | - | Growth Inhibition Assay | - | IC50=11.4834 μM | SANGER |
| SCC-3 | - | Growth Inhibition Assay | - | IC50=2.82891 μM | SANGER |
| SCH | - | Growth Inhibition Assay | - | IC50=32.8485 μM | SANGER |
| SF126 | - | Growth Inhibition Assay | - | IC50=3.06198 μM | SANGER |
| SF268 | - | Growth Inhibition Assay | - | IC50=3.32869 μM | SANGER |
| SF-539 | 333 μM | Kinase Assay | 60 min | Inhibition of PDGFRbeta phosphorylation with IC50 of 12.2 μM | 20403700 |
| SF-539 | 333 μM | Kinase Assay | 60 min | Inhibition of PDGFRbeta tyrosine kinase activity in PDGF-BB-stimulated human SF-539 cells with IC50 of 12.2 μM | 22204741 |
| Sf9 | - | Kinase Assay | 30 min | Inhibition of GST-tagged VEGFR expressed with IC50 of 0.185 μM | 19854051 |
| Sf9 | - | Function Assay | 1 h | Inhibition of human recombinant His-tagged RET expressed with IC50 of 1.3 μM | 20117004 |
| SH-4 | - | Growth Inhibition Assay | - | IC50=5.4837 μM | SANGER |
| SH-SY5Y | 10 μM | Kinase Assay | 1 h | Inhibition of PDGFRbeta with IC50 of 0.0831 μM | 24890652 |
| SIG-M5 | - | Growth Inhibition Assay | - | IC50=1.37009 μM | SANGER |
| SJSA-1 | - | Growth Inhibition Assay | - | IC50=7.66061 μM | SANGER |
| SK-LMS-1 | - | Growth Inhibition Assay | - | IC50=3.83338 μM | SANGER |
| SK-MEL-2 | - | Growth Inhibition Assay | - | IC50=42.6405 μM | SANGER |
| SK-MM-2 | - | Growth Inhibition Assay | - | IC50=5.4782 μM | SANGER |
| SK-N-DZ | - | Growth Inhibition Assay | - | IC50=4.67099 μM | SANGER |
| SK-NEP-1 | - | Growth Inhibition Assay | - | IC50=2.60237 μM | SANGER |
| SK-N-FI | - | Growth Inhibition Assay | - | IC50=17.6913 μM | SANGER |
| SK-PN-DW | - | Growth Inhibition Assay | - | IC50=8.60985 μM | SANGER |
| SK-UT-1 | - | Growth Inhibition Assay | - | IC50=49.8825 μM | SANGER |
| SMMC7721 | - | Cytotoxic Assay | 72 h | IC50=6.47 μM | 21621880 |
| SNB19 | 100 μM | Growth Inhibition Assay | 48 h | GI54=10 μM | 22560627 |
| SR | - | Growth Inhibition Assay | - | IC50=1.54572 μM | SANGER |
| ST486 | - | Growth Inhibition Assay | - | IC50=6.3471 μM | SANGER |
| SU-DHL-1 | - | Growth Inhibition Assay | - | IC50=4.80174 μM | SANGER |
| SW620 | 100 μM | Growth Inhibition Assay | 48 h | GI55=1.3 μM | 22560627 |
| SW872 | - | Growth Inhibition Assay | - | IC50=8.34758 μM | SANGER |
| SW954 | - | Growth Inhibition Assay | - | IC50=6.27517 μM | SANGER |
| SW962 | - | Growth Inhibition Assay | - | IC50=20.4293 μM | SANGER |
| SW982 | - | Growth Inhibition Assay | - | IC50=4.55307 μM | SANGER |
| T24 | - | Growth Inhibition Assay | 72 h | IC50=2.44 μM | 23999040 |
| TALL-1 | - | Growth Inhibition Assay | - | IC50=8.18384 μM | SANGER |
| TE-1 | - | Growth Inhibition Assay | - | IC50=20.4104 μM | SANGER |
| TE-10 | - | Growth Inhibition Assay | - | IC50=2.34384 μM | SANGER |
| TE-11 | - | Growth Inhibition Assay | - | IC50=1.83985 μM | SANGER |
| TE-12 | - | Growth Inhibition Assay | - | IC50=804.55 nM | SANGER |
| TE-15 | - | Growth Inhibition Assay | - | IC50=507.61 nM | SANGER |
| TE-8 | - | Growth Inhibition Assay | - | IC50=2.37038 μM | SANGER |
| TE-9 | - | Growth Inhibition Assay | - | IC50=37.0401 μM | SANGER |
| TGBC1TKB | - | Growth Inhibition Assay | - | IC50=12.002 μM | SANGER |
| TGW | - | Growth Inhibition Assay | - | IC50=13.3276 μM | SANGER |
| THP1 | - | Growth Inhibition Assay | 72 h | IC50=0.5 μM | 20570526 |
| TK10 | 100 μM | Growth Inhibition Assay | 48 h | GI56=6.3 μM | 22560627 |
| TK10 | - | Growth Inhibition Assay | - | IC50=30.126 μM | SANGER |
| TT | - | Growth Inhibition Assay | 72 h | IC50=0.04 μM | 24904961 |
| U251 | 333 μM | Kinase Assay | 60 min | Inhibition of VEGFR2 phosphorylation with IC50 of 18.9 μM | 20403700 |
| U251 | 10 μM | Kinase Assay | 1 h | Inhibition of VEGFR2 with IC50 of 0.0189 μM | 24890652 |
| U-266 | - | Growth Inhibition Assay | - | IC50=34.2781 μM | SANGER |
| UACC257 | 100 μM | Growth Inhibition Assay | 48 h | GI57=4 μM | 22560627 |
| VA-ES-BJ | - | Growth Inhibition Assay | - | IC50=9.99933 μM | SANGER |
| WI38 | - | Cytotoxic Assay | 72 h | IC50=8.56 μM | 21621880 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00663559 | Carcinoma Renal Cells | Phase 2 | Completed | - | Spain ... more >> Hospital Central de Asturias Oviedo, Asturias, Spain, 33006 Hospital Parc Taulí Sabadell, Barcelona, Spain, 08208 Hospital CLINIC Barcelona, Spain, 08036 Hospital Reina Sofía Córdoba, Spain, 14004 Hospital de Jaén Jaén, Spain, 23007 Hospital Clínico San Carlos Madrid, Spain, 28040 Hospital lozano Blesa Zaragoza, Spain, 50009 Collapse << |
| NCT03097601 | - | - | Active, not recruiting | December 30, 2019 | France ... more >> Centre Antoine LACASSAGNE Nice, France, 06189 Collapse << |
| NCT01243359 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Cancer Stage I Renal Cell Cancer Stage II Renal Cell Cancer Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00372775 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site Norwalk, Connecticut, United States, 06856 United States, Florida Pfizer Investigational Site Cocoa Beach, Florida, United States, 32931 Pfizer Investigational Site Merritt Island, Florida, United States, 32952 Pfizer Investigational Site Titusville, Florida, United States, 32796 United States, Missouri Pfizer Investigational Site Creve Coeur, Missouri, United States, 63141 Pfizer Investigational Site St. Louis, Missouri, United States, 63110-1094 Pfizer Investigational Site St. Louis, Missouri, United States, 63110 Pfizer Investigational Site St. Peters, Missouri, United States, 63376 United States, New Jersey Pfizer Investigational Site Basking Ridge, New Jersey, United States, 07920 United States, New York Pfizer Investigational Site Commack, New York, United States, 11725 Pfizer Investigational Site New York, New York, United States, 10022 United States, Pennsylvania Pfizer Investigational Site Sayre, Pennsylvania, United States, 18840 United States, Texas Pfizer Investigational Site Austin, Texas, United States, 78705 Pfizer Investigational Site Austin, Texas, United States, 78745 Pfizer Investigational Site Austin, Texas, United States, 78758 Pfizer Investigational Site Austin, Texas, United States, 78759 Pfizer Investigational Site Round Rock, Texas, United States, 78664 France Pfizer Investigational Site Pessac, Be1 04495, France, 33604 Pfizer Investigational Site Marseille Cedex 09, France, 13009 Pfizer Investigational Site Saint-Priest en Jarez Cedex, France, 42271 Pfizer Investigational Site Toulouse cedex 9, France, 31059 Italy Pfizer Investigational Site Bologna, Italy, 40139 Pfizer Investigational Site Genova, Italy, 16132 Pfizer Investigational Site Orbassano (TO), Italy, 10043 Pfizer Investigational Site Roma, Italy, 00151 Spain Pfizer Investigational Site Madrid, Spain, 28046 Pfizer Investigational Site Valencia, Spain, 46014 Collapse << |
| NCT00399152 | Renal Cancer ... more >>GIST Collapse << | Phase 1 | Completed | - | - |
| NCT00864864 | Glioblastoma ... more >>Brain Tumor Collapse << | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01005472 | Metastatic Melanoma | Phase 1 | Completed | - | United States, California ... more >> University of California Los Angeles (UCLA) Los Angeles, California, United States, 90095 Collapse << |
| NCT02402062 | Neuroendocrine Tumors ... more >> Pancreatic Neoplasms Collapse << | Phase 2 | Active, not recruiting | June 2019 | Spain ... more >> Institut Catalá d'Oncologia L'Hospitalet L'Hospitalet de Llobregat, Barcelona, Spain, 08907 Hospital Universitario Marqués de Valdecilla Santander, Cantabria, Spain, 39008 Hospital Provincial de Castellón Castelló, Valencia, Spain Hospital Universitari Vall d'Hebron Barcelona, Spain, 08035 Hospital Universitario Virgen de las Nieves Granada, Spain, 18014 Hospital Universitario Ramón y Cajal Madrid, Spain, 28034 Hospital Universitario 12 de Octubre Madrid, Spain, 28041 Hospital Universitario La Paz Madrid, Spain, 28046 Hospital Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Universitario Virgen del Rocío Sevilla, Spain, 41013 Collapse << |
| NCT00699374 | Carcinoma, Hepatocellular | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00699374 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00372775 | - | - | Completed | - | - |
| NCT00577382 | - | - | Completed | - | - |
| NCT00577382 | Mucosal Lentiginous Melanoma ... more >> Acral Lentiginous Malignant Melanoma Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Missouri Washington University in St. Louis St. Louis, Missouri, United States, 63110 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37212 Collapse << |
| NCT00422344 | Renal Cell Carcinoma ... more >> Kidney Cancer Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center 1275 York Avenue New York, New York, United States, 10021 Collapse << |
| NCT00961571 | Metastatic Colorectal Cancer | Phase 2 | Terminated(Unanticipated side ... more >>effects and futility) Collapse << | - | United States, District of Col... more >>umbia Lombardi Cancer Center at Georgetown University Washington, District of Columbia, United States, 20007 Collapse << |
| NCT01308034 | Non GIST Sarcomas | Phase 1 | Completed | - | France ... more >> Institut Bergonié Bordeaux, France Centre Oscar Lambret Lille, France Centre Léon Bérard Lyon, France, 69373 CHU La Timone Marseille, France Institut de Cancérologie de l'ouest Saint Herblain, France Institut Gustave Roussy Villejuif, France Collapse << |
| NCT00499473 | - | - | Completed | - | - |
| NCT01462695 | Childhood Cerebellar Anaplasti... more >>c Astrocytoma Childhood Cerebral Anaplastic Astrocytoma Childhood Cerebral Astrocytoma Childhood Infratentorial Ependymoma Childhood Mixed Glioma Childhood Oligodendroglioma Childhood Supratentorial Ependymoma Recurrent Childhood Cerebellar Astrocytoma Recurrent Childhood Cerebral Astrocytoma Recurrent Childhood Ependymoma Recurrent Childhood Subependymal Giant Cell Astrocytoma Collapse << | Phase 2 | Completed | - | - |
| NCT00499473 | Adult Anaplastic Astrocytoma ... more >> Adult Diffuse Astrocytoma Adult Giant Cell Glioblastoma Adult Glioblastoma Adult Gliosarcoma Adult Mixed Glioma Adult Oligodendroglioma Adult Pineal Gland Astrocytoma Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT01462695 | - | - | Completed | - | - |
| NCT01064310 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT00392444 | Advanced Malignant Mesotheliom... more >>a Recurrent Malignant Mesothelioma Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> National Cancer Institute of Canada Clinical Trials Group Kingston, Ontario, Canada, K7L 3N6 Collapse << |
| NCT01064310 | - | - | Completed | - | - |
| NCT01402817 | Neurofibromatosis ... more >> NF1 Plexiform Neurofibromas Collapse << | Phase 2 | Terminated(1 patient died of u... more >>ncertain cause but possibly related to drug.) Collapse << | - | United States, Indiana ... more >> Riley Hospital for Children at IU Health Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT03025893 | Glioblastoma Multiforme ... more >> Glioblastoma, Adult Glioblastoma Recurrent Brain Tumor GBM Collapse << | Phase 2 Phase 3 | Recruiting | January 2020 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Netherlands Contact: Cyrillo G Brahm, MD +31 20 444 4321 Principal Investigator: Henk WM Verheul, MD PhD Sub-Investigator: Cyrillo G Brahm, MD University Medical Center Groningen Not yet recruiting Groningen, Netherlands Contact: Cyrillo G Brahm, MD PhD Principal Investigator: Annemiek ME Walenkamp, MD PhD Sub-Investigator: Cyrillo G Brahm, MD Collapse << |
| NCT01122615 | Renal Cell Cancer ... more >> Kidney Cancer Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02928575 | Glioblastoma Multiforme | Phase 2 | Unknown | June 2017 | Canada, Alberta ... more >> Tom Baker Cancer Center and University of Calgary Recruiting Calgary, Alberta, Canada, T2N 4N2 Contact: Jacob Easaw, MD PhD FRCPC 403-521-3446 Jay.Easaw@albertahealthservices.ca Canada, Quebec McGill University Health Centre Recruiting Montreal, Quebec, Canada, H4A 3J1 Contact: Bassam Abdulkarim, MD PhD FRCPC 514-934-1934 ext 43915 bassam.abdulkarim@mcgill.ca Collapse << |
| NCT01158222 | Clear Cell Renal Cell Carcinom... more >>a Stage IV Renal Cell Cancer Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01158222 | - | - | Completed | - | - |
| NCT00392444 | - | - | Completed | - | - |
| NCT00656669 | Breast Cancer | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00618124 | Solid Tumors ... more >>Breast Cancer Neoplasms Collapse << | Phase 1 | Completed | - | United States, Indiana ... more >> Pfizer Investigational Site Indianapolis, Indiana, United States, 46202 United States, New Mexico Pfizer Investigational Site Albuqurque, New Mexico, United States, 87131 United States, Tennessee Pfizer Investigational Site Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00961571 | - | - | Terminated(Unanticipated side ... more >>effects and futility) Collapse << | - | - |
| NCT00843037 | Paraganglioma ... more >> Pheochromocytoma Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Canada, Ontario University Health Network, Princess Margaret Cancer Centre Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec Hôpital Notre-Dame du CHUM Montreal, Quebec, Canada, H2L 4M1 Netherlands University Medical Centre Groningen Groningen, Netherlands Collapse << |
| NCT00656669 | - | - | Completed | - | - |
| NCT02164240 | Gastrointestinal Stromal Tumor | Phase 1 | Active, not recruiting | May 2020 | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00631618 | Metastatic Melanoma | Phase 2 | Completed | - | United States, California ... more >> California Pacific Medical Center San Francisco, California, United States, 94115 Collapse << |
| NCT00967603 | Pancreatic Cancer | Phase 2 | Completed | - | Italy ... more >> San Raffaele Scientific Institute Milan, Italy, 20132 Collapse << |
| NCT00299741 | Prostate Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel-Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00615446 | Solid Tumors | Phase 1 | Completed | - | United States, Massachusetts ... more >> Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00299741 | - | - | Completed | - | - |
| NCT02386111 | Carcinoma, Renal Cell ... more >> Kidney Diseases Kidney Neoplasms Urogenital Neoplasms Urologic Diseases Urologic Neoplasms Neoplasms Neoplasms by Histologic Type Clear-cell Metastatic Renal Cell Carcinoma Collapse << | Phase 1 | Terminated(Portfolio re-priori... more >>tization) Collapse << | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, California UC Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 UCSF Helen Diller Comprehensive Cancer Center San Francisco, California, United States, 94158 United States, District of Columbia George Washington University-Medical Faculty Associates Washington, District of Columbia, United States, 20037 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Nebraska Nebraska Cancer Specialists Omaha, Nebraska, United States, 68130 United States, New York Mount Sinai Medical Center New York, New York, United States, 10029 United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02058901 | Solid Tumors | Phase 1 | Recruiting | December 2017 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Netherlands, 1081HV Contact: Henk MW Verheul, MD, PhD +31-20-4444321 h.verheul@vumc.nl Principal Investigator: Henk MW Verheul, MD, PhD Collapse << |
| NCT01740154 | - | - | Terminated(Slow Accrual) | - | - |
| NCT01402817 | - | - | Terminated(1 patient died of u... more >>ncertain cause but possibly related to drug.) Collapse << | - | - |
| NCT00526656 | Bladder Cancer | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01740154 | Fatigue Recur... more >>rent Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Not Applicable | Terminated(Slow Accrual) | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT00787787 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Collapse << | Phase 2 | Terminated | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT00270413 | Metastatic Breast Cancer | Phase 2 | Completed | - | Belgium ... more >> Imelda Bonheiden Bonheiden, Belgium AZ st-jan brugge Brugge, Belgium AZ VUB Brussels, Belgium UCL Brussels, Belgium Charleroi Charleroi, Belgium UZ Gent Gent, Belgium, 9000 ZOL Hasselt, Belgium UZ Leuven Leuven, Belgium, 3000 Liege sart-tilman Liege, Belgium Namur st-elisabeth Namur, Belgium St-Elisabeth Turnhout Turnhout, Belgium AZ st-augustinus Wilrijk, Belgium Mont-Godinne Yvoir, Belgium Collapse << |
| NCT00730353 | Esophageal Cancer | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 Rush-Presbyterian St. Luke's Medical Center Chicago, Illinois, United States, 60612 Medical & Surgical Specialists, LLC Galesburg, Illinois, United States, 61401 United States, Indiana Cancer Care Center of Southern Indiana Bloomington, Indiana, United States, 47403 Oncology Hematology Associates of SW Indiana Evansville, Indiana, United States, 47714 Fort Wayne Oncology & Hematology, Inc Fort Wayne, Indiana, United States, 46815 IN Onc/Hem Associates Indianapolis, Indiana, United States, 46202 Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 Arnett Cancer Care Lafayette, Indiana, United States, 47904 Horizon Oncology Center Lafayette, Indiana, United States, 47905 Medical Consultants, P.C. Muncie, Indiana, United States, 47303 Monroe Medical Associates Munster, Indiana, United States, 46321 Northern Indiana Cancer Research Consortium South Bend, Indiana, United States, 46601 Providence Medical Group Terre Haute, Indiana, United States, 47802 United States, Ohio Ireland Cancer Center - University Hospitals of Cleveland Cleveland, Ohio, United States, 44106 Collapse << |
| NCT00388037 | - | - | Completed | - | - |
| NCT00388037 | Recurrent Fallopian Tube Cance... more >>r Recurrent Ovarian Epithelial Cancer Recurrent Primary Peritoneal Cavity Cancer Stage IIIA Fallopian Tube Cancer Stage IIIA Ovarian Epithelial Cancer Stage IIIA Primary Peritoneal Cavity Cancer Stage IIIB Fallopian Tube Cancer Stage IIIB Ovarian Epithelial Cancer Stage IIIB Primary Peritoneal Cavity Cancer Stage IIIC Fallopian Tube Cancer Stage IIIC Ovarian Epithelial Cancer Stage IIIC Primary Peritoneal Cavity Cancer Stage IV Fallopian Tube Cancer Stage IV Ovarian Epithelial Cancer Stage IV Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> National Cancer Institute of Canada Clinical Trials Group Kingston, Ontario, Canada, K7L 3N6 Collapse << |
| NCT01743300 | Healthy | Phase 1 | Completed | - | - |
| NCT00753727 | Soft Tissue Sarcoma | Phase 1 Phase 2 | Unknown | March 2016 | Australia, Victoria ... more >> Peter MacCallum Cancer Centre Recruiting Melbourne, Victoria, Australia, 3002 Contact: Ania Matera +61396563661 Ania.Matera@petermac.org Contact: Sally Whyte, PhD +61396563605 sally.whyte@petermac.org Principal Investigator: David Thomas, MB BS PhD Collapse << |
| NCT00788060 | Renal Cell Carcinoma | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27705 Collapse << |
| NCT00768144 | - | - | Completed | - | - |
| NCT01100177 | Glioblastoma | Phase 2 | Completed | - | Spain ... more >> Grupo Español de Investigacion en Neurooncologia Madrid, Spain, 28001 Collapse << |
| NCT00787787 | - | - | Terminated | - | - |
| NCT00329043 | Prostate Cancer | Phase 2 | Active, not recruiting | May 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00730353 | - | - | Completed | - | - |
| NCT00616109 | - | - | Completed | - | - |
| NCT00378911 | Recurrent Uterine Sarcoma ... more >> Uterine Leiomyosarcoma Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Gynecologic Oncology Group Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT00768144 | Ovarian Cancer ... more >> Fallopian Tube Cancer Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02214 Beth-Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01215578 | Neuroendocrine Tumors ... more >> Pancreatic Neoplasms Advanced Disease Sunitinib Collapse << | Phase 2 | Terminated(lack of recruitemen... more >>t) Collapse << | - | France ... more >> Hôpital Beaujon Clichy, Hauts de Seine, France, 92110 Collapse << |
| NCT00702884 | Esophageal Cancer | Phase 2 | Completed | - | United States, Ohio ... more >> Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00813423 | Adult Solid Neoplasm | Phase 1 | Active, not recruiting | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, New Jersey Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT02919371 | Renal Cell Carcinoma | Phase 1 Phase 2 | Recruiting | December 2021 | Saudi Arabia ... more >> Oncology Centre, King Faisal Specialist Hospital and Research Centre Recruiting Riyadh, Saudi Arabia, 11211 Contact: Shouki Bazarbashi, MD 00966 11 442 3935 bazarbashi@kfshrc.edu.sa Contact: Fazal Hussain, MD 966-11-4423949 fhussain@kfshrc.edu.sa Collapse << |
| NCT01824615 | Ovarian Cancer ... more >> Adverse Effects Collapse << | Phase 2 | Completed | - | Taiwan ... more >> Cathay General Hospital Taipei, Taiwan, 106 Collapse << |
| NCT00616109 | Extensive-Stage Small Cell Lun... more >>g Cancer Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00702884 | - | - | Completed | - | - |
| NCT00793871 | Gastrointestinal Neoplasms, Ga... more >>strointestinal Stromal Tumors Collapse << | Phase 4 | Completed | - | China, Jiangsu ... more >> Nanjing Bayi Hospital Nanjing, Jiangsu, China, 210002 China Beijing Cancer Hospital Beijing, China, 100035 307 Hospital of PLA Beijing, China, 100071 Cancer Institute & Hospital Chinese Academy of Medical Sciences and PUMC Bejing, China, 100021 Collapse << |
| NCT00793871 | - | - | Completed | - | - |
| NCT00459979 | Kidney Cancer | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT00459979 | - | - | Completed | - | - |
| NCT00910039 | Cognitive/Functional Effects ... more >> Metastatic Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 2 | Terminated(Slow Accrual) | - | United States, Michigan ... more >> Henry Ford Health System Detroit, Michigan, United States, 48202 United States, Ohio Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01827254 | - | - | Completed | - | - |
| NCT03463460 | Thymic Carcinoma | Phase 2 | Suspended(pending amendment) | July 30, 2020 | United States, Ohio ... more >> Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00890747 | Accelerated Phase Chronic Myel... more >>ogenous Leukemia Acute Myeloid Leukemia With Multilineage Dysplasia Following Myelodysplastic Syndrome Acute Undifferentiated Leukemia Adult Acute Lymphoblastic Leukemia in Remission Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(15;17)(q22;q12) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Grade III Lymphomatoid Granulomatosis Adult Langerhans Cell Histiocytosis Adult Nasal Type Extranodal NK/T-cell Lymphoma Aggressive NK-cell Leukemia AIDS-related Diffuse Large Cell Lymphoma AIDS-related Diffuse Mixed Cell Lymphoma AIDS-related Diffuse Small Cleaved Cell Lymphoma AIDS-related Immunoblastic Large Cell Lymphoma AIDS-related Lymphoblastic Lymphoma AIDS-related Malignancies AIDS-related Small Noncleaved Cell Lymphoma Anaplastic Large Cell Lymphoma Angioimmunoblastic T-cell Lymphoma Atypical Chronic Myeloid Leukemia, BCR-ABL1 Negative Chronic Eosinophilic Leukemia Chronic Myelomonocytic Leukemia Chronic Neutrophilic Leukemia Chronic Phase Chronic Myelogenous Leukemia Clear Cell Renal Cell Carcinoma Cutaneous B-cell Non-Hodgkin Lymphoma de Novo Myelodysplastic Syndromes Essential Thrombocythemia Extramedullary Plasmacytoma Extranodal Marginal Zone B-cell Lymphoma of Mucosa-associated Lymphoid Tissue Hepatosplenic T-cell Lymphoma HIV Infection HIV-associated Hodgkin Lymphoma Intraocular Lymphoma Isolated Plasmacytoma of Bone Light Chain Deposition Disease Mast Cell Leukemia Myelodysplastic Syndrome With Isolated Del(5q) Myelodysplastic/Myeloproliferative Neoplasm, Unclassifiable Myeloid/NK-cell Acute Leukemia Nodal Marginal Zone B-cell Lymphoma Noncutaneous Extranodal Lymphoma Osteolytic Lesions of Multiple Myeloma Peripheral T-cell Lymphoma Plasma Cell Neoplasm Polycythemia Vera Post-transplant Lymphoproliferative Disorder Previously Treated Myelodysplastic Syndromes Primary Myelofibrosis Primary Systemic Amyloidosis Progressive Hairy Cell Leukemia, Initial Treatment Prolymphocytic Leukemia Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Adult Burkitt Lymphoma Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Adult Diffuse Mixed Cell Lymphoma Recurrent Adult Diffuse Small Cleaved Cell Lymphoma Recurrent Adult Grade III Lymphomatoid Granulomatosis Recurrent Adult Hodgkin Lymphoma Recurrent Adult Immunoblastic Large Cell Lymphoma Recurrent Adult Lymphoblastic Lymphoma Recurrent Adult T-cell Leukemia/Lymphoma Recurrent Cutaneous T-cell Non-Hodgkin Lymphoma Recurrent Grade 1 Follicular Lymphoma Recurrent Grade 2 Follicular Lymphoma Recurrent Grade 3 Follicular Lymphoma Recurrent Mantle Cell Lymphoma Recurrent Marginal Zone Lymphoma Recurrent Mycosis Fungoides/Sezary Syndrome Recurrent Renal Cell Cancer Recurrent Small Lymphocytic Lymphoma Refractory Chronic Lymphocytic Leukemia Refractory Hairy Cell Leukemia Refractory Multiple Myeloma Relapsing Chronic Myelogenous Leukemia Stage IV Renal Cell Cancer T-cell Large Granular Lymphocyte Leukemia Testicular Lymphoma Unspecified Adult Solid Tumor, Protocol Specific Waldenström Macroglobulinemia Collapse << | Phase 1 | Completed | - | United States, California ... more >> Jonsson Comprehensive Cancer Center Los Angeles, California, United States, 90095 United States, District of Columbia Lombardi Comprehensive Cancer Center at Georgetown University Washington, District of Columbia, United States, 20057 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Maryland AIDS - Associated Malignancies Clinical Trials Consortium Rockville, Maryland, United States, 20850 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Missouri Washington University - Jewish Saint Loius, Missouri, United States, 63110 United States, New York Albert Einstein College of Medicine Bronx, New York, United States, 10461 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Pennsylvania Abramson Cancer Center of The University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Washington Virginia Mason Medical Center Seattle, Washington, United States, 98101 Collapse << |
| NCT00397787 | Acinar Cell Adenocarcinoma of ... more >>the Pancreas Duct Cell Adenocarcinoma of the Pancreas Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> Cancer and Leukemia Group B Chicago, Illinois, United States, 60606 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00389974 | Cervical Adenocarcinoma ... more >> Cervical Adenosquamous Cell Carcinoma Cervical Squamous Cell Carcinoma Recurrent Cervical Cancer Stage IVB Cervical Cancer Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> National Cancer Institute of Canada Clinical Trials Group Kingston, Ontario, Canada, K7L 3N6 Collapse << |
| NCT00910039 | - | - | Terminated(Slow Accrual) | - | - |
| NCT00389974 | - | - | Completed | - | - |
| NCT00599313 | Metastatic Prostate Cancer | Phase 2 | Completed | - | - |
| NCT01827254 | - | - | Completed | - | France ... more >> CHU de la Timone Marseille, Cedex 5, France, 13335 Hopital albert Michalon Grenoble, Cedex 9, France, 38043 Centre Paul Papin Angers, France, 49100 Hôpital Saint-André Bordeaux Cedex, France, 33075 Centre Catalan Urologie Andrologie Cabestany, France, 66330 Centre Oscar Lambret - Cancérologie Urologie Digestive Lille Cedex, France, 59020 clinique de la Louvière Lille Cedex, France, 59042 Hopital Dupuytren - Oncologie Medicale Limoges Cedex, France, 87042 Centre Leon Berard, Service d'Oncologie Medicale Lyon Cedex 08, France, 69373 Institut Paoli-Calmettes / Hôpital de jour Marseille Cedex 9, France, 13273 Cedex 9 Hopital Timone Adultes Marseille Cedex, France, 13385 Hopital Europeen Georges Pompidou Paris Cedex 15, France, 75908 Centre Eugene Marquis Rennes, France, 35042 CHU Charles Nicolle ROUEN Cedex, France, 76031 Departement d'Oncologie Medicale-Institut de Cancerologie de la Loire Saint Priest en Jarez Cedex, France, 42271 Centre Rene Gauducheau - Service Oncologie Medicale St Herblain Cedex, France, 44805 Institut Claudius Regaud Toulouse, France, 31000 Centre Alexis Vautrin Vandoeuvre les Nancy, France, 54511 Collapse << |
| NCT00361309 | Hepatocellular Carcinoma ... more >> Liver Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00599313 | - | - | Completed | - | - |
| NCT00519896 | Recurrent Thyroid Cancer ... more >> Stage IVA Follicular Thyroid Cancer Stage IVA Papillary Thyroid Cancer Stage IVB Follicular Thyroid Cancer Stage IVB Papillary Thyroid Cancer Stage IVC Follicular Thyroid Cancer Stage IVC Papillary Thyroid Cancer Thyroid Gland Medullary Carcinoma Collapse << | Phase 2 | Completed | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT00444314 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | United States, Indiana ... more >> Indiana University Cancer Center Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University School of Medicine Baltimore, Maryland, United States, 21205 United States, Wisconsin University of Wisconsin Paul P. Carbone Comprehensive Cancer Center Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00434109 | Neuroendocrine Tumor ... more >> Islet Cell Tumor Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00457743 | - | - | Completed | - | - |
| NCT00434109 | - | - | Completed | - | - |
| NCT00457743 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 1 Phase 2 | Completed | - | Japan ... more >> Pfizer Investigational Site Kashiwa, Chiba, Japan Pfizer Investigational Site Sapporo, Hokkaido, Japan Pfizer Investigational Site Suita, Osaka, Japan Pfizer Investigational Site Chuo-ku, Tokyo, Japan Collapse << |
| NCT00715442 | Renal Cell Carcinoma | Phase 2 | Active, not recruiting | June 2020 | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00519896 | - | - | Completed | - | - |
| NCT01718327 | Unresectable and Advanced Chol... more >>angiocarcinoma Collapse << | Phase 2 | Completed | - | France ... more >> Hôpital Beaujon Clichy, France, 92118 Hôpital privé Jean Mermoz Lyon, France, 69008 CHU La Timone Marseille, France, 13005 Hôpital Saint Antoine Paris, France, 75012 Institut Mutualiste Montsouris Paris, France, 75014 Institut Gustave Roussy Villejuif, France, 94805 Collapse << |
| NCT01498835 | Soft Tissue Sarcoma | Phase 1 | Completed | - | Germany ... more >> Helios Klinikum Bad Saarow Bad Saarow, Germany, 15526 University Medical Center Mannheim Mannheim, Germany, 68167 Collapse << |
| NCT01286896 | Malignant Solid Tumour | Phase 1 | Unknown | December 2011 | Netherlands ... more >> The Netherlands Cancer Institute - Antoni van Leeuwenhoek Hospital Recruiting Amsterdam, Netherlands, 1066CX Contact: Neeltje Steeghs Principal Investigator: Neeltje Steeghs, MD, PhD Collapse << |
| NCT00979992 | - | - | Active, not recruiting | - | - |
| NCT00886132 | Adenoid Cystic Carcinoma ... more >> Salivary Gland Cancer Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00478426 | Endometrial Adenocarcinoma ... more >> Endometrial Endometrioid Adenocarcinoma Endometrial Serous Adenocarcinoma Recurrent Uterine Corpus Carcinoma Stage IVA Uterine Corpus Cancer AJCC v7 Stage IVB Uterine Corpus Cancer AJCC v7 Uterine Carcinosarcoma Uterine Corpus Carcinosarcoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00338884 | Carcinoma, Renal Cell | Phase 2 | Completed | - | Argentina ... more >> Pfizer Investigational Site Rosario, Santa Fé, Argentina, (2000) Pfizer Investigational Site Buenos Aires, Argentina, 1431 Pfizer Investigational Site Cordoba, Argentina, X5000AAI Australia, South Australia Pfizer Investigational Site Adelaide, South Australia, Australia, 5000 Australia, Victoria Pfizer Investigational Site Clayton, Victoria, Australia, 3168 Pfizer Investigational Site East Bentleigh, Victoria, Australia, 3165 Brazil Pfizer Investigational Site Porto Alegre, RS, Brazil, 90610-000 Pfizer Investigational Site São Paulo, SP, Brazil, 01308-050 Korea, Republic of Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Pfizer Investigational Site Seoul, Korea, Republic of, 120-752 Mexico Pfizer Investigational Site Guadalajara, Jalisco, Mexico, 44280 Pfizer Investigational Site Monterrey, Nuevo Leon, Mexico, 64460 Taiwan Pfizer Investigational Site Taichung, Taiwan, 407 Pfizer Investigational Site Tainan, Taiwan, 710 Pfizer Investigational Site Taipei, Taiwan, 112 Collapse << |
| NCT00706706 | Carcinoma, Renal Cell | Phase 4 | Completed | - | China, Guangdong ... more >> Pfizer Investigational Site Guangzhou, Guangdong, China, 510060 China, Hubei Pfizer Investigational Site Wuhan, Hubei, China, 430030 China, Jiangsu Pfizer Investigational Site Nanjing, Jiangsu, China, 210002 China Pfizer Investigational Site Beijing, China, 100021 Pfizer Investigational Site Beijing, China, 100034 Pfizer Investigational Site Beijing, China, 100036 Pfizer Investigational Site Chong qing, China, 400038 Pfizer Investigational Site Shanghai, China, 200032 Pfizer Investigational Site Shanghai, China, 200127 Pfizer Investigational Site Tianjin, China, 300060 Pfizer Investigational Site Xi'an, China, 710032 Collapse << |
| NCT03140176 | - | - | Recruiting | December 30, 2020 | Algeria ... more >> Pierre Et Marie Curie Center Recruiting Algers, Algeria, 16005 CAC Annaba Recruiting Annaba, Algeria Hanene Djedi Recruiting Annaba, Algeria Egypt National Cancer Institute Recruiting Cairo, Egypt, 11796 Demerdash Hospital Recruiting Cairo, Egypt Collapse << |
| NCT00979992 | Ovarian Clear Cell Adenocarcin... more >>oma Recurrent Ovarian Carcinoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT00338884 | - | - | Completed | - | - |
| NCT03109015 | Carcinoma, Renal Cell | Phase 2 | Recruiting | May 1, 2019 | United States, North Carolina ... more >> Duke Cancer Institute Recruiting Durham, North Carolina, United States, 27705 Contact: Julia Rasmussen 919-681-9822 julia.rasmussen@duke.edu Contact: Diane Pinder 919-668-8375 diane.pinder@duke.edu Principal Investigator: Michael Harrison, MD Collapse << |
| NCT02398552 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Unknown | March 2017 | China, Beijing ... more >> Chinese acadamy of medical science cancer institute & hospital Not yet recruiting Beijing, Beijing, China, 100021 Contact: Changling Li, MD 0086-10-67781331 changllss@yahoo.com.cn Principal Investigator: Changling Li, MD Peking University First Hospital Not yet recruiting Beijing, Beijing, China, 100034 Contact: Zhisong He, MD 0086-10-83572211 wyj7074@sohu.com Principal Investigator: Zhisong He, MD Beijing Cancer Hospital Recruiting Beijing, Beijing, China, 100142 Contact: Chuanliang Cui, MD 0086-10-88196951 1008ccl@163.com Contact: Jun Guo, MD,PHD 0086-10-88196317 guoj307@126.com Principal Investigator: Jun Guo, MD,PHD China, Guangdong Sun Yat-sen university cancer center Not yet recruiting Guangzhou, Guangdong, China, 510060 Contact: Fangjian Zhou, MD 0086-20-87343088 zhoufj@sysucc.org.cn Principal Investigator: Fangjian Zhou, MD China, Shanghai Cancer Hospital, Fudan University Not yet recruiting Shanghai, Shanghai, China, 200032 Contact: Dingwei Ye, MD 0086-21-64175590 dwye@163.com Principal Investigator: Dingwei Ye, MD China, Tianjin Tianjin medical university cancer institute & hospital Not yet recruiting Tianjin, Tianjin, China, 300060 Contact: Xin Yao, MD 0086-22-23340123 yaoxin1969@yahoo.com.cn Principal Investigator: Xin Yao, MD Collapse << |
| NCT00330564 | - | - | Terminated(Slow accrual.) | - | - |
| NCT00495625 | Liver Cancer | Phase 2 | Terminated | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01061411 | Clear Cell Sarcoma of the Kidn... more >>ey Recurrent Renal Cell Carcinoma Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Netherlands Academ Zienkenhuis Bij De University Amsterdam, Netherlands, 1007 MB VU University Medical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT00330564 | Von Hippel-Lindau Syndrome ... more >> Renal Cell Carcinoma Hemangioblastoma Collapse << | Phase 2 | Terminated(Slow accrual.) | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00495625 | - | - | Terminated | - | - |
| NCT00706706 | - | - | Completed | - | - |
| NCT00716820 | - | - | Completed | - | - |
| NCT00873210 | - | - | Completed | - | Slovakia ... more >> Pfizer Investigational Site Banska Bystrica, Slovakia, 975 17 Pfizer Investigational Site Bojnice, Slovakia, 972 01 Pfizer Investigational Site Bratislava, Slovakia, 812 50 Pfizer Investigational Site Bratislava, Slovakia, 833 10 Pfizer Investigational Site Kosice, Slovakia, 043 91 Pfizer Investigational Site Michalovce, Slovakia, 071 01 Pfizer Investigational Site Nitra, Slovakia, 949 01 Pfizer Investigational Site Poprad, Slovakia, 058 01 Pfizer Investigational Site Presov, Slovakia, 081 81 Pfizer Investigational Site Ruzomberok, Slovakia, 034 26 Pfizer Investigational Site Trebisov, Slovakia, 075 01 Pfizer Investigational Site Trencin, Slovakia, 911 71 Pfizer Investigational Site Trnava, Slovakia, 917 75 Pfizer Investigational Site Zilina, Slovakia, 012 07 Collapse << |
| NCT00716820 | - | - | Completed | - | - |
| NCT00668811 | Papillary Thyroid Cancer ... more >> Follicular Thyroid Cancer Differentiated Thyroid Cancer Collapse << | Phase 2 | Completed | - | United States, District of Col... more >>umbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 Collapse << |
| NCT00668811 | - | - | Completed | - | - |
| NCT03592199 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | December 11, 2022 | Brazil ... more >> Instituto do Cancer do Estado de São Paulo Recruiting São Paulo, Brazil, 01246000 Contact: David QB Muniz, MD +551138932691 david.muniz@hc.fm.usp.br Contact: Diogo A Bastos, MD +551138932691 diogo.bastos@hc.fm.usp.br Principal Investigator: David QB Muniz, MD Sub-Investigator: Carlos Dzik, MD, PHD Sub-Investigator: Diogo A Bastos, MD Sub-Investigator: Jamile A Silva, MD Sub-Investigator: Pedro HI Velho, MD Collapse << |
| NCT00716625 | - | - | Completed | - | - |
| NCT00606008 | Anaplastic Astrocytoma ... more >> Glioblastoma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00606008 | - | - | Completed | - | - |
| NCT00480935 | Renal Cell Carcinoma | Phase 2 | Terminated(poor recruitment) | - | Canada, Ontario ... more >> University Health Network, Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00465179 | Renal Cell Cancer ... more >> Kidney Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00465179 | - | - | Completed | - | - |
| NCT00673816 | - | - | Terminated(Inability to recrui... more >>t and adequate number of participants) Collapse << | - | - |
| NCT00400569 | - | - | Completed | - | - |
| NCT00864721 | Non Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Florida ... more >> Ocala Oncology Center Ocala, Florida, United States, 34471 Cancer Centers of Florida, P.A. Ocoee, Florida, United States, 34761 United States, Minnesota Minnesota Oncology Hematology, P.A. Minneapolis, Minnesota, United States, 55404 United States, North Carolina Cancer Centers of North Carolina Raleigh, North Carolina, United States, 27607 United States, Oregon Willamette Valley Cancer Institute and Research Center Eugene, Oregon, United States, 97401-8122 United States, South Carolina Cancer Centers of the Carolinas Greenville, South Carolina, United States, 29605 United States, Texas Texas Oncology - Arlington South Arlington, Texas, United States, 76014 Texas Oncology, P.A. - Bedford Bedford, Texas, United States, 76022 Methodist Charlton Cancer Ctr. Dallas, Texas, United States, 75237 Texas Cancer Center of Mesquite Mesquite, Texas, United States, 75150 Texas Oncology Cancer Care and Research Center Waco, Texas, United States, 76712 United States, Virginia Virginia Oncology Associates Norfolk, Virginia, United States, 23502 United States, Washington Yakima Valley Mem Hosp/North Star Lodge Yakima, Washington, United States, 98902 Collapse << |
| NCT00864721 | - | - | Completed | - | - |
| NCT00716625 | - | - | Completed | - | - |
| NCT03066427 | Clear Cell Renal Carcinoma | Phase 2 | Recruiting | May 31, 2020 | Spain ... more >> ICO Duran i Reynals Recruiting L'Hospitalet de Llobregat, Barcelona, Spain, 08908 Contact: Xavier García del Muro, MD garciadelmuro@iconcologia.net Principal Investigator: Xavier García del Muro, MD Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Contact: Cristina Suárez, MD +34 932746000 ext 4919 crsuarez@vhebron.net Principal Investigator: Cristina Suárez, MD Hospital Clínic i Provincial de Barcelona Recruiting Barcelona, Spain, 08036 Contact: Oscar Reig, MD 08036 ext 2811 OREIG@clinic.cat Principal Investigator: Oscar Reig, MD Complejo Hospitalario Regional Reina Sofía Recruiting Córdoba, Spain, 14004 Contact: María J Méndez, MD +34 957012408 mjosemv@yahoo.es Principal Investigator: María J Méndez, MD Hospital Ramón Y Cajal Recruiting Madrid, Spain, 28034 Contact: Teresa Alonso, MD +34 913368263 talonso@oncologiahrc.com Principal Investigator: Teresa Alonso, MD Hospital Universitario 12 de Octubre Recruiting Madrid, Spain, 28041 Contact: Daniel Castellano, MD +34 913908339 cdanicas@hotmail.com Principal Investigator: Daniel Castellano, MD Collapse << |
| NCT00400569 | Liposarcoma L... more >>eiomyosarcoma Fibrosarcoma Malignant Fibrous Histiocytoma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01499121 | Clear Cell, Metastatic Renal C... more >>ell Carcinoma Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia BC Cancer Agency - Vancouver Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Manitoba CancerCare Manitoba Winnipeg, Manitoba, Canada, R3E 0V9 Canada, Nova Scotia QEII Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 3A7 Canada, Ontario Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Kingston General Hospital Research Institute Kingston, Ontario, Canada, K7L 2V7 London Health Sciences Centre London, Ontario, Canada, N6A 4L6 Durham Regional Cancer Centre Oshawa, Ontario, Canada, L1G 2B9 Ottawa Hospital Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec Notre-Dame Hospital Montreal, Quebec, Canada, H2W 1T8 Collapse << |
| NCT00673816 | Von Hippel-Lindau Syndrome | Phase 1 Phase 2 | Terminated(Inability to recrui... more >>t and adequate number of participants) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02713763 | Pancreatic Neuroendocrine Tumo... more >>ur Metastatic Collapse << | Phase 2 | Recruiting | July 2019 | Spain ... more >> Hospital Universitario Central de Asturias Recruiting Oviedo, Asturias, Spain, 33011 Contact: Paula Jiménez +34 646662756 palucaji@hotmail.com Principal Investigator: Paula Jiménez Hospital Universitario Valle de Hebrón Active, not recruiting Barcelona, Spain, 08035 Hospital Reina Sofía Recruiting Córdoba, Spain, 14004 Contact: Raquel Serrano +34 957011464 rsblanch@hotmail.com Principal Investigator: Raquel Serrano Hospital Universitario Donostia Active, not recruiting Donostia/San Sebastián, Spain, 20014 Hospital Ramón y Cajal Active, not recruiting Madrid, Spain, 28034 Hospital Universitario 12 de Octubre Recruiting Madrid, Spain, 28041 Contact: Rocío García Carbonero 34 91 390 83 39 rgcarbonero@gmail.com Principal Investigator: Rocio Garcia Carbonero Hospital General Universitario J.M. Morales Meseguer Active, not recruiting Murcia, Spain, 30008 Complejo Hospitalario Regional Virgen Del Rocío Recruiting Sevilla, Spain, 41013 Contact: Marta Benavent martabenavent.v@gmail.com Principal Investigator: Marta Benavent Instituto Valenciano de Oncología Active, not recruiting Valencia, Spain, 46009 Collapse << |
| NCT00794950 | Urinary Tract Urothelial Carci... more >>noma Collapse << | Phase 2 | Active, not recruiting | December 2018 | United States, Maryland ... more >> Mark P. Schoenberg, MD Baltimore, Maryland, United States, 21287-2101 United States, Michigan Alon Weizer, MD Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT03449173 | Type B3 Thymoma ... more >> Thymic Carcinoma Collapse << | Phase 2 | Recruiting | March 2, 2021 | Italy ... more >> National Cancer Institute Recruiting Milan, Italy, 20133 Collapse << |
| NCT01073644 | - | - | Terminated(The study was prema... more >>turely discontinued due to poor recruitment on 18th April 2013. There were no safety concerns that led to the decision to terminate.) Collapse << | - | Philippines ... more >> Chong Hua Hospital Cebu City, Philippines Private Clinic Manila, Philippines Collapse << |
| NCT00672594 | Prostate Cancer ... more >> Prostatectomy Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Texas MD Anderson, University of Texas Houston, Texas, United States, 77030 Collapse << |
| NCT00672594 | - | - | Completed | - | - |
| NCT02626754 | Renal Cell Carcinoma | Phase 2 | Terminated(In most sites had d... more >>ifficult to recruitting subjects.) Collapse << | - | Taiwan ... more >> Kaohsiung Medical University Chung-HO Memorial Hospital Kaohsiung, Taiwan, 80756 Kaohsiung Veterans General Hospital Kaohsiung, Taiwan, 81362 Taichung Veterans General Hospital Taichung, Taiwan, 40705 National Taiwan University Hospital Taipei, Taiwan, 10002 Taipei Vterans General Hospital Taipei, Taiwan, 11217 Tri-Service General Hospital Taipei, Taiwan, 11490 Chang Gung Memorial Hospital, Linkou Taoyuan City, Taiwan, 333 Collapse << |
| NCT00912912 | Genitourinary Disease | Phase 2 | Terminated(Slow Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00912912 | - | - | Terminated(Slow Accrual) | - | - |
| NCT02072031 | Anlotinib RCC | Phase 2 | Unknown | December 2016 | China, Beijing ... more >> Peking Union Medical College Hospital Beijing, Beijing, China, 100005 Cancer Institute and Hospital, Chinese Academy of Medical Sciences Beijing, Beijing, China, 100021 China, Chongqing Chongqing Cancer Hospital Chongqing, Chongqing, China, 404100 China, Gansu Gansu Province Tumor Hospital Lanzhou, Gansu, China, 730050 China, Guangdong Sun Yat-Sen University Cancer Center Guangzhou, Guangdong, China, 510000 China, Guangxi Guangxi medical university affiliated tumor hospital Nanning, Guangxi, China, 530021 China, Heilongjiang Harbin medical university affiliated tumor hospital Haerbin, Heilongjiang, China, 150000 China, Jiangsu The 81st Hospital of Chinese PLA Nanjing, Jiangsu, China, 210000 China, Jilin Jilin Cancer Hospital Changchun, Jilin, China, 132000 China, Liaoning China General Hospital of Shenyang Military Region Shenyang, Liaoning, China, 110000 Liaoning Province Tumor Hospital Shenyang, Liaoning, China, 110042 China, Shandong Qilu Hospital,Shandong University Jinan, Shandong, China, 250012 China, Shanghai Cancer Hospital of Fudan University Shanghai, Shanghai, China, 200000 China, Sichuan West China Hospital,Sichuan University Chengdu, Sichuan, China, 610041 China, Tianjin Tianjin Medical University Cancer Hospital Tianjin, Tianjin, China, 300000 Collapse << |
| NCT02790580 | Breast Cancer | Phase 2 | Recruiting | November 2020 | Singapore ... more >> National University Hospital, Singapore Recruiting Singapore, Singapore, 119228 Contact: Soo Chin Lee (65) 6779 5555 soo_chin_lee@nuhs.edu.sg Collapse << |
| NCT01391130 | Metastatic Clear Cell Renal Ce... more >>ll Carcinoma Collapse << | Phase 2 | Terminated(Study Terminated du... more >>e to Insufficient Efficacy) Collapse << | - | - |
| NCT00943839 | Kidney Cancer | Not Applicable | Completed | - | France ... more >> Centre Antoine Lacassagne Nice, France, 06189 Collapse << |
| NCT00847015 | Bladder Cancer ... more >> Urinary Bladder Collapse << | Phase 2 | Completed | - | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan-Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memoral Sloan Kettering Cancer Center@Phelps Sleepy Hollow, New York, United States Collapse << |
| NCT03456401 | Renal Cancer Cell | Phase 2 | Terminated(availability of new... more >> and more promising therapeutic agents than expected in the experimentation in question) Collapse << | - | Italy ... more >> Istituto Tumori Milan, Mi, Italy, 20156 Collapse << |
| NCT00847015 | - | - | Completed | - | - |
| NCT00583063 | Solid Tumors | Early Phase 1 | Completed | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00859456 | Sarcoma, Soft Tissue | Phase 2 | Unknown | December 2013 | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00361244 | Colorectal Carcinoma | Phase 1 Phase 2 | Terminated(Slow Accrual) | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01019798 | Non Small Cell Lung Cancer | Phase 2 | Unknown | December 2011 | Taiwan ... more >> Section of Hematology, Department of Medicine,Taipei Medical University Hospital Recruiting Taipei, Taiwan, 110 Contact: Cheng-Jeng Tai, M.D. 886-2-27372181 ext 3903 cjtai@tmu.edu.tw Principal Investigator: Cheng-Jeng Tai, M.D. Collapse << |
| NCT01616186 | Locally Metastatic Malignant N... more >>eoplasm Collapse << | Phase 2 | Withdrawn | April 2016 | - |
| NCT01026337 | Renal Cell Carcinoma ... more >> Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Not Applicable | Active, not recruiting | - | United States, Pennsylvania ... more >> Abramson Cancer Center of The University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00482911 | Intraocular Melanoma ... more >> Malignant Conjunctival Neoplasm Collapse << | Phase 2 | Terminated(Investigator left t... more >>he institute.) Collapse << | - | United States, Maryland ... more >> Warren Grant Magnuson Clinical Center - NCI Clinical Trials Referral Office Bethesda, Maryland, United States, 20892-1182 Collapse << |
| NCT00720941 | - | - | Active, not recruiting | - | - |
| NCT00720941 | Carcinoma, Renal Cell | Phase 3 | Active, not recruiting | June 30, 2019 | - |
| NCT00482911 | - | - | Terminated(Investigator left t... more >>he institute.) Collapse << | - | - |
| NCT00089648 | - | - | Completed | - | - |
| NCT00678119 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, California ... more >> City of Hope Duarte, California, United States, 91010 UCLA Los Angeles, California, United States, 90095 United States, Colorado The Urology Center of Colorado Denver, Colorado, United States, 80211 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Indiana The Indiana University Cancer Center Indianapolis, Indiana, United States, 46202 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 United States, Kansas University of Kansas Hospital Kansas City, Kansas, United States, 66160 United States, Minnesota University of Minnesota Cancer Center Minneapolis, Minnesota, United States, 55455 United States, North Carolina Carolina's Medical Center / Blumenthal Cancer Center Charlotte, North Carolina, United States, 28204 United States, Ohio Barrett Cancer Cincinnati, Ohio, United States, 45267 United States, Texas CORTPA Dallas, Texas, United States, 75230 United States, Virginia Urology of Virginia-Sentara Medical Group Norfolk, Virginia, United States, 23502 Canada, Ontario Princess Margaret Hospital Toronto, Ontario, Canada, M5G2M9 Canada, Quebec Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00662025 | Advanced/Metastatic Breast Can... more >>cer Collapse << | Phase 2 | Completed | - | Japan ... more >> Pfizer Investigational Site Anjo-city, Aichi, Japan Pfizer Investigational Site Nagoya, Aichi, Japan Pfizer Investigational Site Okazaki-City, Aichi, Japan Pfizer Investigational Site Toyoake, Aichi, Japan Pfizer Investigational Site Matsuyama-shi, Ehime, Japan Pfizer Investigational Site Kure, Hiroshima, Japan Pfizer Investigational Site Morioka, Iwate, Japan Pfizer Investigational Site Yokohama, Kanagawa, Japan Pfizer Investigational Site Sakai, Osaka, Japan Pfizer Investigational Site Bunkyo-ku, Tokyo, Japan Pfizer Investigational Site Koto-ku, Tokyo, Japan Pfizer Investigational Site Chiba, Japan Pfizer Investigational Site Fukuoka, Japan Pfizer Investigational Site Kagoshima, Japan Pfizer Investigational Site Kyoto, Japan Pfizer Investigational Site Niigata, Japan Pfizer Investigational Site Osaka, Japan Collapse << |
| NCT00890110 | Renal Cell Cancer | Phase 1 Phase 2 | Unknown | December 2011 | - |
| NCT00089648 | Carcinoma, Renal Cell | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Duarte, California, United States, 91010 Pfizer Investigational Site Pasadena, California, United States, 91105 Pfizer Investigational Site San Francisco, California, United States, 94115 United States, Illinois Pfizer Investigational Site Chicago, Illinois, United States, 60637-1460 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 United States, North Carolina Pfizer Investigational Site Durham, North Carolina, United States, 27710 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 United States, Tennessee Pfizer Investigational Site Nashville, Tennessee, United States, 37232-5536 United States, Texas Pfizer Investigational Site Dallas, Texas, United States, 75246 Collapse << |
| NCT03249740 | Neovascular Age-Related Macula... more >>r Degeneration Collapse << | Phase 1 Phase 2 | Recruiting | March 2020 | United States, Arizona ... more >> Retinal Consultants of Arizona Recruiting Gilbert, Arizona, United States, 85296 Contact: Elisa Martinez 602-222-2221 ext 4115 emartinez@retinalconsultantsaz.com United States, California Retina Institute of California Recruiting Arcadia, California, United States, 91007 Contact: Valentina Bozikovic 626-574-0188 vbozikovic@retina2020.com Retina-Vitreous Associates Medical Group Recruiting Beverly Hills, California, United States, 90211 Contact: Janet Kurokouch 310-289-2478 ext 1243 jkurokouchi@laretina.com United States, Florida Retina Macula Specialists of Miami, LLC Withdrawn Miami, Florida, United States, 33126 United States, Indiana Midwest Eye Institute Recruiting Indianapolis, Indiana, United States, 46290 Contact: Cari Novak 317-805-4596 cari.novak@gmail.com United States, New York Ophthalmic Consultants of Long Island Recruiting Lynbrook, New York, United States, 11563 Contact: Isabel Lezcano 516-593-4026 ilezcano@ocli.net United States, Texas Retina Research Institute of Texas Recruiting Abilene, Texas, United States, 79606 Contact: Courtney Wheeler 325-690-4414 Courtney.Wheeler@txicr.clinic Texas Retina Associates Recruiting Arlington, Texas, United States, 76012 Contact: Michelle Curry 682-433-5457 mcurry@texasretina.com Retina Research Center, PLLC Recruiting Austin, Texas, United States, 78705 Contact: Ivana Gunderson 512-279-1251 igunderson@e-retina.net Medical Center Ophthalmology Associates Recruiting San Antonio, Texas, United States, 78240 Contact: Beatrice Guajardo 210-697-2036 ext 2245 bguajardo@mcoaeyecare.com Collapse << |
| NCT00662025 | - | - | Completed | - | - |
| NCT02231749 | Advanced Renal Cell Carcinoma ... more >> Metastatic Renal Cell Carcinoma Collapse << | Phase 3 | Active, not recruiting | September 30, 2019 | - |
| NCT01711268 | - | - | Unknown | December 2014 | Australia, New South Wales ... more >> Crown Princess Mary Cancer Centre, Westmead Hospital Recruiting Westmead, New South Wales, Australia, 2145 Contact: Howard Gurney, MBBS (Hon), FRACP +61298455200 howard.gurney@sydney.edu.au Principal Investigator: Howard Gurney, MBBS (Hon), FRACP Collapse << |
| NCT00443534 | Neoplasms | Not Applicable | Completed | - | - |
| NCT00573404 | Gastrointestinal Stromal Tumor | Phase 1 | Terminated(slow accrual) | - | United States, Tennessee ... more >> Vanderbilt-Ingram Cancer Center - Cool Springs Nashville, Tennessee, United States, 37064 Vanderbilt-Ingram Cancer Center at Franklin Nashville, Tennessee, United States, 37064 Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << |
| NCT00443534 | - | - | Completed | - | - |
| NCT01225848 | Urothelial Carcinoma | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01784978 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(Lack of recruitment... more >>) Collapse << | - | France ... more >> Hopital Bordeaux University Bordeaux, France Greece ALEXANDRA General Hospital of Athens Athens, Greece, 11528 Spain Hospital Universitario Central de Asturias Oviedo, Asturias, Spain, 33006 Hospital Marqués de Valdecilla Santander, Cantabria, Spain, 39008 Hospital del Mar Barcelona, Spain, 08003 Hospital Universitario 12 de Octubre Madrid, Spain, 28026 Hospital Clínico San Carlos Madrid, Spain, 28040 Clara Campal. Hospital Sanchinarro Madrid, Spain, 28050 Hospital Universitario Virgen de la Victoria Málaga, Spain Hospital General Universitario de Valencia Valencia, Spain Collapse << |
| NCT00620347 | Lung Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> National Cancer Center, Korea Goyang-si, Gyeonggi-do, Korea, Republic of, 411-764 Collapse << |
| NCT00265317 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | - |
| NCT02231749 | - | - | Active, not recruiting | - | - |
| NCT01265901 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT00609622 | Colorectal Neoplasms | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00555256 | Non-Small Cell Lung Cancer | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01829217 | Lung Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02214 Beth Isreal Deaconess Medical Center Boston, Massachusetts, United States, 02215 Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00609622 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01829217 | - | - | Completed | - | - |
| NCT00265317 | - | - | Completed | - | - |
| NCT01033981 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | Panama ... more >> Pfizer Investigational Site Panama City, Panama Collapse << |
| NCT00400114 | Esophageal Cancer | Phase 2 | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital Regional Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 University Health Network (Princess Margaret & Toronto General Hospitals) Toronto, Ontario, Canada, M5G 2C4 Collapse << |
| NCT01118039 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Urethral Cancer Collapse << | Phase 2 | Unknown | - | Spain ... more >> Hospital Del Mar Recruiting Barcelona, Spain, 08003 Contact: Contact Person 34-39-3248-3861 XVillanueva@imas.imim.es Hospital de la Santa Cruz i Sant Pau Recruiting Barcelona, Spain, 08025 Contact: Contact Person 34-93-553-7118 jmaroto@santpau.cat Hospital Universitario San Carlos Recruiting Madrid, Spain, 28040 Contact: Contact Person 34-91-330-3000, Ext. 7935 jgonzalezl.hcsc@salud.madrid.org Hospital Universitario 12 de Octubre Recruiting Madrid, Spain, 28041 Contact: Contact Person 34-91-469-2313 cdanicas@hotmail.com Collapse << |
| NCT00474994 | Adult Malignant Fibrous Histio... more >>cytoma of Bone Desmoid Tumor Endometrial Cancer Ovarian Cancer Sarcoma Small Intestine Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber/Harvard Cancer Center at Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT02853331 | Renal Cell Carcinoma | Phase 3 | Active, not recruiting | January 27, 2020 | - |
| NCT00474994 | - | - | Completed | - | - |
| NCT00887575 | Breast Cancer | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Holy Cross Hospital Ft. Lauderdale, Florida, United States, 33308 Florida Cancer Specialists North Ft. Myers, Florida, United States, 33916 Florida Cancer Specialists South Ft. Myers, Florida, United States, 33916 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Indiana Providence Medical Group Terre Haute, Indiana, United States, 47802 United States, Kentucky Baptist Hospital East Louisville, Kentucky, United States, 40207 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 National Capital Clinical Research Consortium Bethesda, Maryland, United States, 20817 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63017 United States, Nebraska Nebraska Methodist Cancer Center Omaha, Nebraska, United States, 68114 United States, New Jersey Hematology Oncology Associates of Northern NJ Morristown, New Jersey, United States, 07960 United States, Oklahoma Cancer Centers of Southwest Oklahoma Lawton, Oklahoma, United States, 73505 United States, Tennessee Family Cancer Center Collierville, Tennessee, United States, 38017 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00887575 | - | - | Completed | - | - |
| NCT00524316 | Liver Cancer | Phase 2 | Terminated(low accrual) | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Collapse << |
| NCT00077974 | Carcinoma, Renal Cell | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Duarte, California, United States, 91010-3000 Pfizer Investigational Site Pasadena, California, United States, 91105 Pfizer Investigational Site San Francisco, California, United States, 94115 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 United States, Michigan Pfizer Investigational Site Ann Arbor, Michigan, United States, 48109 United States, Minnesota Pfizer Investigational Site Rochester, Minnesota, United States, 55905 United States, New York Pfizer Investigational Site New York, New York, United States, 10021 Pfizer Investigational Site New York, New York, United States, 10022 United States, North Carolina Pfizer Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 United States, Oregon Pfizer Investigational Site Portland, Oregon, United States, 97213 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00387920 | Central Nervous System Metasta... more >>ses Childhood Central Nervous System Choriocarcinoma Childhood Central Nervous System Embryonal Tumor Childhood Central Nervous System Germ Cell Tumor Childhood Central Nervous System Germinoma Childhood Central Nervous System Mixed Germ Cell Tumor Childhood Central Nervous System Teratoma Childhood Central Nervous System Yolk Sac Tumor Recurrent Childhood Central Nervous System Embryonal Tumor Unspecified Childhood Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, California Childrens Hospital of Orange County Orange, California, United States, 92868-3874 UCSF-Mount Zion San Francisco, California, United States, 94115 University of California San Francisco Medical Center-Parnassus San Francisco, California, United States, 94143 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Michigan C S Mott Children's Hospital Ann Arbor, Michigan, United States, 48109 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, New York Columbia University Medical Center New York, New York, United States, 10032 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 United States, Texas University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Baylor College of Medicine Houston, Texas, United States, 77030 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Canada, Ontario Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Canada, Quebec Centre Hospitalier Universitaire Sainte-Justine Montreal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT00077974 | - | - | Completed | - | - |
| NCT00373113 | Breast Neoplasms | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT02420821 | - | - | Active, not recruiting | - | - |
| NCT00747305 | Kidney Cancer | Phase 1 | Terminated(Study terminated ea... more >>rly due to low accrual.) Collapse << | - | United States, South Carolina ... more >> Hollings Cancer Center at Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT02420821 | Renal Cell Carcinoma | Phase 3 | Active, not recruiting | December 1, 2021 | - |
| NCT01803503 | Solid Tumors ... more >>Breast Cancer Non-small Cell Lung Cancer Prostate Cancer Gastric Cancer Collapse << | Phase 2 | Unknown | April 2018 | Singapore ... more >> National University Hospital Not yet recruiting Singapore, Singapore, 119074 Contact: Soo Chin Lee, MBBS (65) 6779 5555 soo_chin_lee@nuhs.edu.sg Principal Investigator: Soo Chin Lee, MBBS National University Hospital Recruiting Singapore, Singapore, 119228 Contact: Soo Chin Lee, MBBS +65 6779 5555 soo_chin_lee@nuhs.edu.sg Principal Investigator: Soo Chin Lee, MBBS Collapse << |
| NCT00668863 | Unresectable or Metastatic Col... more >>orectal Cancer Collapse << | Phase 2 | Completed | - | Japan ... more >> Pfizer Investigational Site Nagoya, Aichi, Japan Pfizer Investigational Site Chiba-shi, Chiba-ken, Japan Pfizer Investigational Site Matsuyama-shi, Ehime, Japan Pfizer Investigational Site Sapporo-shi, Hokkaido, Japan Pfizer Investigational Site Sapporo, Hokkaido, Japan Pfizer Investigational Site Kochi-shi, Kochi, Japan Pfizer Investigational Site Saku, Nagano, Japan Pfizer Investigational Site Osakasayama-shi, Osaka, Japan Pfizer Investigational Site Takatsuki, Osaka, Japan Pfizer Investigational Site Shimotsuke-shi, Tochigi, Japan Pfizer Investigational Site Minoh/Osaka, Japan Collapse << |
| NCT00879619 | - | - | Terminated(Terminated due to s... more >>low accrual.) Collapse << | - | - |
| NCT00373113 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00524316 | - | - | Terminated(low accrual) | - | - |
| NCT00668863 | - | - | Completed | - | - |
| NCT00879619 | Prostate Cancer ... more >> Adenocarcinoma of the Prostate Recurrent Prostate Cancer Stage I Prostate Cancer Stage III Prostate Cancer Stage IV Prostate Cancer Collapse << | Phase 1 Phase 2 | Terminated(Terminated due to s... more >>low accrual.) Collapse << | - | United States, California ... more >> Chao Comprehensive Cancer Center Orange, California, United States, 92868 Collapse << |
| NCT00930033 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Active, not recruiting | February 2020 | France ... more >> Hopital Necker Paris, France, 75015 Collapse << |
| NCT01070186 | Renal Cell Carcinoma | Phase 2 | Withdrawn(No participants were... more >> enrolled.) Collapse << | - | - |
| NCT00372567 | Gastrointestinal Stromal Tumor | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | United States, Michigan ... more >> Pfizer Investigational Site Detroit, Michigan, United States, 48201 Pfizer Investigational Site Farmington Hills, Michigan, United States, 48334 United States, Missouri Pfizer Investigational Site Creve Coeur, Missouri, United States, 63141 Pfizer Investigational Site St. Louis, Missouri, United States, 63110 Pfizer Investigational Site St. Peters, Missouri, United States, 63376 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 Germany Pfizer Investigational Site Goettingen, Germany, 37075 Pfizer Investigational Site Hamburg, Germany, 22767 Hong Kong Pfizer Investigational Site Lai Chi Kok, Kowloon, Hong Kong, 0 Pfizer Investigational Site Tuen Mun, New Territories, Hong Kong, 0 Pfizer Investigational Site Hong Kong, Hong Kong, 0 Italy Pfizer Investigational Site Bologna, Italy, 40138 Pfizer Investigational Site Milano, Italy, 20133 Pfizer Investigational Site San Giovanni Rotondo, Italy, 71013 Korea, Republic of Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Pfizer Investigational Site Seoul, Korea, Republic of, 135-710 Pfizer Investigational Site Seoul, Korea, Republic of, 138-736 Spain Pfizer Investigational Site Barcelona, Spain, 08036 Pfizer Investigational Site Valencia, Spain, 46009 United Kingdom Pfizer Investigational Site Glasgow, United Kingdom, G12 0YH Pfizer Investigational Site Leeds, United Kingdom, LS9 7TF Pfizer Investigational Site London, United Kingdom, NW1 2PG Pfizer Investigational Site London, United Kingdom, SW3 6JJ Pfizer Investigational Site London, United Kingdom, W1 Pfizer Investigational Site Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00372567 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00496223 | Melanoma | Phase 1 Phase 2 | Terminated(toxicities required... more >> dose reduction compromising effectiveness and PI left Moffitt) Collapse << | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00905021 | Metastatic Breast Cancer | Phase 1 Phase 2 | Terminated(Sponsor withdrew su... more >>pport) Collapse << | - | United States, Texas ... more >> Baylor College of Medicine, Lester and Sue Smith Breast Center Houston, Texas, United States, 77030 Collapse << |
| NCT00798889 | Solid Tumors | Not Applicable | Completed | - | - |
| NCT00453895 | Adrenocortical Carcinoma | Phase 2 | Unknown | June 2011 | Germany ... more >> Charite Berlin Berlin, Germany Dept. of Medicine I, University of Wuerzburg Wuerzburg, Germany, 97080 Collapse << |
| NCT00499135 | Adult Solid Neoplasm ... more >> Clear Cell Renal Cell Carcinoma Recurrent Renal Cell Carcinoma Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 1 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00824538 | Breast Cancer | Phase 2 | Terminated(Closed prematurely ... more >>due to emerging data on toxicity and competing trials.) Collapse << | - | - |
| NCT00905021 | - | - | Terminated(Sponsor withdrew su... more >>pport) Collapse << | - | - |
| NCT00853125 | Kidney Cancer | Phase 2 | Terminated(Slow accrual) | - | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT00720148 | Solid Tumors | Phase 1 | Completed | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT00695292 | Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33901 Florida Hospital Cancer Insitute Orlando, Florida, United States, 32804 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Louisiana Baton Rouge General Medical Center Baton Rouge, Louisiana, United States, 70806 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63017 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, South Carolina Spartanburg Regional Medical Center Spartanburg, South Carolina, United States, 29303 United States, Tennessee Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00798889 | - | - | Completed | - | - |
| NCT00524186 | Esophageal Cancer ... more >> Gastric Cancer Collapse << | Phase 1 | Terminated(PI left institute) | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Collapse << |
| NCT00695292 | - | - | Completed | - | - |
| NCT01238055 | Advanced Gastric Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02555748 | Metastatic Renal Cell Cancer | Phase 4 | Recruiting | June 2021 | France ... more >> CHU DE BORDEAUX - Hôpital Saint-André Recruiting Bordeaux, France, 33035 Contact: Marine GROSS-GOUPIL, MD + 33 (0)5 56 79 58 08 marine.gross-goupil@chu-bordeaux.fr Principal Investigator: Marine GROSS-GOUPIL, MD Institut Bergonie Recruiting Bordeaux, France, 33076 Contact: Guilhem ROUBAUD, MD + 33 (0)5 56 33 33 41 g.roubaud@bordeaux.unicancer.fr Principal Investigator: Guilhem ROUBAUD, MD CHU DE LIMOGES - Hôpital Dupuytren Recruiting Limoges, France, 87042 Contact: Sabrina FALKOWSKI, MD + 33 (0)5 55 05 61 00 sabrina.falkowski@chu-limoges.fr Principal Investigator: Sabrina FALKOWSKI, MD Centre Leon Berard Recruiting Lyon, France, 69373 Contact: Helen BOYLE, MD +33 (0)4 78 78 28 28 helen.boyle@lyon.unicancer.fr Principal Investigator: Aude FLECHON, MD Institut Paoli Calmettes Recruiting Marseille, France, 13273 Contact: Gwénaëlle GRAVIS-MESCAM, MD + 33 (0)4 91 22 36 77 gravisg@ipc.unicancer.fr Principal Investigator: Gwénaëlle GRAVIS-MESCAM, MD Institut Regional Du Cancer Montpellier Recruiting Montpellier, France, 34298 Contact: Diego TOSI, MD + 33 (0)4 67 61 23 04 diego.tosi@icm.unicancer.fr Principal Investigator: Diego TOSI, MD CH RODEZ Recruiting Rodez, France, 12027 Contact: Guillermo REYES ORTEGA, MD 05 65 55 22 10 g.reyes-ortega@ch-rodez.fr Institut Claudius Regaud Recruiting Toulouse, France, 31059 Contact: Christine CHEVREAU, MD + 33 5 31 15 51 03 Chevreau.christine@iuct-oncopole.fr Principal Investigator: Christine CHEVREAU, MD Gustave Roussy Withdrawn Villejuif, France, 94805 Collapse << |
| NCT01098903 | - | - | Completed | - | Australia, New South Wales ... more >> Westmead Hospital Westmead, New South Wales, Australia, 2145 Netherlands Academic medical center Amsterdam Amsterdam, Netherlands, 1105AZ Erasmus Medical Center, Daniel Den Hoed Cancer Center Rotterdam, Netherlands, 3075EA Collapse << |
| NCT00824538 | - | - | Terminated(Closed prematurely ... more >>due to emerging data on toxicity and competing trials.) Collapse << | - | - |
| NCT00462982 | - | - | Completed | - | - |
| NCT00729833 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00462982 | Kidney Cancer ... more >> Melanoma (Skin) Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00729833 | Advanced Cancer ... more >> Advanced Solid Tumors Collapse << | Phase 1 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | United States, California ... more >> Pfizer Investigational Site Los Angeles, California, United States, 90095 Pfizer Investigational Site Santa Monica, California, United States, 90404 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Texas Pfizer Investigational Site San Antonio, Texas, United States, 78229 Collapse << |
| NCT00849186 | Kidney Cancer | Not Applicable | Completed | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Collapse << |
| NCT00891878 | - | - | Completed | - | - |
| NCT00891878 | Adenocarcinoma of the Gastroes... more >>ophageal Junction Esophageal Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00698815 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IIIB Non-Small Cell Lung Cancer AJCC v7 Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 2 | Completed | - | - |
| NCT00372424 | Breast Cancer | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, 1000 Pfizer Investigational Site Bruxelles, Belgium, 1200 Pfizer Investigational Site Charleroi, Belgium, 6000 Pfizer Investigational Site Sint-Niklaas, Belgium, 9100 Pfizer Investigational Site Wilrijk, Belgium, 2610 Italy Pfizer Investigational Site Meldola, FC, Italy, 47014 Pfizer Investigational Site Milano, Italy, 20132 Collapse << |
| NCT00408252 | Recurrent Disease ... more >> Squamous Cell Head and Neck Carcinoma Palliative Treatment Collapse << | Phase 2 | Terminated(Too frequent grade ... more >>3-4 toxicities) Collapse << | - | Belgium ... more >> Cliniques Universiatires St LUC UCL Bruxelles, Belgium, 1200 clinique Sainte Elisabeth Namur, Belgium, 5000 Clinique universiataire de Mont Godinnes UCL Yvoir, Belgium, 5004 France Centre Jean Perrin Clermont Ferrand, France, 263011 Centre G-f Leclerc DIJON Cedex, France, 8021079 Centre Alexis Vautrin Nancy, France, 4511 René Gauducheau Saint Herblain Cedex, France, 44805 CHU Bretonneau Tours cedex 1, France, 37044 Institut Gustave Roussy Villejuif Cedex, France, 94805 Collapse << |
| NCT00698815 | - | - | Completed | - | - |
| NCT01147822 | Carcinoma, Renal Cell | Phase 2 | Active, not recruiting | September 30, 2019 | China, Guangdong ... more >> Novartis Investigative Site Guangzhou, Guangdong, China, 510060 China, Jiangsu Novartis Investigative Site Nanjing, Jiangsu, China, 210002 China, Zhejiang Novartis Investigative Site Hangzhou, Zhejiang, China, 310003 China Novartis Investigative Site Beijing, China, 100021 Novartis Investigative Site Beijing, China, 100036 Novartis Investigative Site Beijing, China, 100853 Novartis Investigative Site Beijing, China Novartis Investigative Site Shanghai, China, 200032 Novartis Investigative Site Shanghai, China, 200127 Novartis Investigative Site Tianjin, China, 300060 Korea, Republic of Novartis Investigative Site Daejeon, Korea, Republic of, 301-721 Novartis Investigative Site Goyang-si, Gyeonggi-Do, Korea, Republic of, 410-769 Novartis Investigative Site Seoul, Korea, Republic of, 120-752 Novartis Investigative Site Seoul, Korea, Republic of, 135-710 Novartis Investigative Site Seoul, Korea, Republic of, 138-736 Taiwan Novartis Investigative Site Kaohsiung Hsien, Taiwan, 833 Novartis Investigative Site Taichung, Taiwan, 40402 Novartis Investigative Site Taichung, Taiwan, 40705 Novartis Investigative Site Taipei, Taiwan, 10002 Novartis Investigative Site Taipei, Taiwan, 11217 Novartis Investigative Site Taoyuan County, Taiwan, 333 Collapse << |
| NCT00814021 | Kidney Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | France ... more >> Institut Claudius Regaud Toulouse, France, 31052 Collapse << |
| NCT01147822 | - | - | Active, not recruiting | - | - |
| NCT01309633 | Nasopharyngeal Carcinoma | Phase 2 | Unknown | September 2018 | Singapore ... more >> National University Hospital Recruiting Kent Ridge, Singapore, 119074 Contact: Boon Cher Goh, MBBS (65) 6772 4140 Boon_Cher_Goh@nuhs.edu.sg Contact: Christine Vergara (65) 6772 4619 christine_vergara@nuhs.edu.sg Sub-Investigator: Boon Cher Goh, MBBS Collapse << |
| NCT02404584 | - | - | Recruiting | September 2018 | France ... more >> CHRU de Montpellier - Hôpital Saint-Eloi Recruiting Montpellier cedex 5, France, 34295 Principal Investigator: Délphine Topart, MD CHRU de Nîmes - Hôpital Universitaire Carémeau Recruiting Nîmes Cedex 09, France, 30029 Principal Investigator: Nadine Houédé, MD, PhD Sub-Investigator: Alexandre Evard, Pharmd, PhD Sub-Investigator: Jean-Christophe Boyer, MD, PhD Institut de Cancérologie du Gard (ICG) Recruiting Nîmes Cedex 9, France, 30029 Principal Investigator: Angélique Chapellie, MD Collapse << |
| NCT00393796 | Bladder Cancer | Phase 2 | Terminated(Low accrual) | - | United States, California ... more >> David Geffen School of Medicine at UCLA Los Angeles, California, United States, 90095 United States, Illinois The University of Chicago Chicago, Illinois, United States, 60637 United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 United States, Minnesota Mayo Clinic - Rochester Rochester, Minnesota, United States, 55905 United States, New York Weill Medical College of Cornell University New York, New York, United States, 10021 Columbia University Medical Center New York, New York, United States, 10032 United States, Ohio Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 United States, Pennsylvania Abramson Cancer Center of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Wisconsin Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00372424 | - | - | Completed | - | - |
| NCT00578526 | Urothelial Cancer ... more >> Bladder Cancer Adult Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia British Columbia Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 London Health Sciences Centre London, Ontario, Canada, T6A 4L6 Collapse << |
| NCT00393796 | - | - | Terminated(Low accrual) | - | - |
| NCT00254540 | Carcinoma, Renal Cell | Phase 2 | Completed | - | Japan ... more >> Pfizer Investigational Site Sapporo, Hokkaido, Japan Pfizer Investigational Site Tsukuba, Ibaragi, Japan Pfizer Investigational Site Osakasayama, Osaka, Japan Pfizer Investigational Site Hamamatsu, Shizuoka, Japan Pfizer Investigational Site Sunto-gun, Shizuoka, Japan Pfizer Investigational Site Chuo-ku, Tokyo, Japan Pfizer Investigational Site Akita, Japan Pfizer Investigational Site Fukuoka, Japan Pfizer Investigational Site Osaka, Japan Pfizer Investigational Site Tokushima, Japan Pfizer Investigational Site Yamagata, Japan Collapse << |
| NCT00694096 | Renal Cell Cancer | Phase 1 | Completed | - | United States, Utah ... more >> Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00291577 | Breast Neoplasms | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, 1000 Italy Pfizer Investigational Site Milano, Italy, 20133 Sweden Pfizer Investigational Site Stockholm, Sweden, 171 76 Collapse << |
| NCT00694096 | - | - | Completed | - | - |
| NCT00849186 | - | - | Completed | - | - |
| NCT01934452 | - | - | Recruiting | April 15, 2020 | France ... more >> CHU de la Timone Recruiting Marseille, Cedex 5, France, 13335 CHRU HOTEL DIEU - Service Urologie Recruiting Angers Cedex, France, 49933 C.H.U Morvan Recruiting Brest, France, 29000 Clinique Victor Hugo Recruiting Le Mans, France, 72015 Institut Paoli-Calmettes Recruiting Marseille Cedex 09, France, 13273 Institut Paoli-Calmettes / Hôpital de jour Recruiting Marseille Cedex 9, France, 13273 Cedex 9 Hopital Timone Adultes Recruiting Marseille Cedex, France, 13385 CRLC Val d'Aurelle Recruiting Montpellier cedex 05, France, 34295 Hopital Europeen Georges Pompidou Recruiting Paris Cedex 15, France, 75908 Centre Hospitalier Lyon Sud Recruiting Pierre Bénite Cedex, France, 69495 Clinique Chirurgicale de l'Orangerie, Chiliotherapie Recruiting Strasbourg, France, 67010 Centre Alexis Vautrin Recruiting Vandoeuvre les Nancy, France, 54511 Institut Gustave Roussy Active, not recruiting Villejuif Cedex, France, 94805 Collapse << |
| NCT00291577 | - | - | Completed | - | - |
| NCT02282059 | - | - | Recruiting | December 12, 2022 | China, Anhui ... more >> Oncology Department, The Second Affiliated Hospital of Anhui Medical University Active, not recruiting Hefei, Anhui, China, 230061 China, Beijing Cancer Institute and Hospital, Chinese Academy of Medical Science Not yet recruiting Beijing, Beijing, China, 100021 Department of Medical Oncology, Cancer Institute and Hospital, Chinese Academy of Medical Sciences Active, not recruiting Beijing, Beijing, China, 100021 China, Guangdong SUN Yat-Sen University Cancer Center Active, not recruiting Guangzhou, Guangdong, China, 510060 China, Heilongjiang The Affiliated Tumour Hospital of Harbin Medical University Recruiting Haerbin, Heilongjiang, China, 150081 China, Henan The First Affiliated Hospital of Zhengzhou University Recruiting Zhengzhou, Henan, China, 450052 China, Jiangsu Nanjing Bayi Hospital Active, not recruiting Nanjing, Jiangsu, China, 210002 Nanjing General Hospital of Nanjing Military Command/Hepatobiliary Surgery Department Active, not recruiting Nanjing, Jiangsu, China, 210002 Jiangsu Cancer Hospital Active, not recruiting Nanjing, Jiangsu, China, 210009 Jiangsu Province Hospital Active, not recruiting Nanjing, Jiangsu, China, 210029 China, Shannxi Digestive surgery Department, The First Affiliated Hospital, The Fourth Military Medical University Recruiting Xi'an, Shannxi, China, 710032 China, Sichuan West China Hospital of Sichuan University/Hepatobiliary Pancreatic Surgery Recruiting Chengdu, Sichuan, China, 610041 China, Tianjin Tianjin Cancer Hospital Active, not recruiting Tianjin, Tianjin, China, 300060 China, Zhejiang Zhejiang Cancer Hospital, Oncology department Active, not recruiting Hangzhou, Zhejiang, China, 310022 China The PLA of 307 Hospital Active, not recruiting Beijing, China, 100071 Fudan University Shanghai Cancer Center Active, not recruiting Shanghai, China, 200032 Tianjin Medical University General Hospital/General Surgery Active, not recruiting Tianjin, China, 300052 Collapse << |
| NCT00712504 | Advanced Solid Tumors ... more >> Non Small Cell Lung Cancer Collapse << | Phase 1 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmingham, Alabama, United States, 35233-2115 Pfizer Investigational Site Birmingham, Alabama, United States, 35233 Pfizer Investigational Site Birmingham, Alabama, United States, 35294 United States, Tennessee Pfizer Investigational Site Nashville, Tennessee, United States, 37232 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01441661 | - | - | Completed | - | China, Beijing ... more >> Cancer Institute and Hospital, Chinese Academy of Medical Sciences Beijing, Beijing, China, 100021 China, Hubei Tongji Hospital, Tongji Medical College of Hust Wuhan, Hubei, China, 430030 Collapse << |
| NCT01121562 | Pancreatic Neuroendocrine Tumo... more >>rs Collapse << | Phase 2 | Completed | - | Japan ... more >> Aichi Cancer Center Central Hospital Nagoya, Aichi, Japan National Cancer Center Hospital Chuo-ku, Tokyo, Japan Kyushu University Hospital Fukuoka, Japan Osaka Police Hospital Osaka, Japan Collapse << |
| NCT00254540 | - | - | Completed | - | - |
| NCT00398112 | - | - | Completed | - | - |
| NCT01164202 | Liver Cancer | Phase 2 Phase 3 | Completed | - | France ... more >> CHU Nord Amiens, France CHR Annecy, France Institut Sainte Catherine Avignon, France CHU J Minjoz Besancon, France Institut Bergonié Bordeaux, France CH Béziers, France CHU Côte de Nacre Caen, France CHU Clermont Ferrand, France Clinique des Cèdres Cornebarrieu, France CHU Bocage Dijon, France CH Guilherand Granges, France Hôpital Claude Huriez Lille, France, 59037 Hopital Dupuytren Limoges, France CHBS Lorient, France Hôpital Privé Jean Mermoz Lyon, France CH Ambroise Paré Marseille, France CH Conception Marseille, France CHU La Timone Marseille, France Hôpital Saint Joseph Marseille, France CHRU Saint Eloi Montpellier, France CHU -Hôpital de l'Archet II Nice, France CHR Orléans, France Hopital Tenon Paris, France, 75970 Groupe Hospitalier Paris St Joseph Paris, France Hôpital Européen Georges Pompidou Paris, France CHU Jean Bernard Poitiers, France CHU Robert Debré Reims, France CAC Rennes, France CHU Rouen, France CHU Tours, France Institut Gustave Roussy Villejuif, France Collapse << |
| NCT00906360 | Metastatic Squamous Neck Cance... more >>r With Occult Primary Squamous Cell Carcinoma Recurrent Metastatic Squamous Neck Cancer With Occult Primary Recurrent Salivary Gland Cancer Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Nasopharynx Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage III Salivary Gland Cancer Stage III Squamous Cell Carcinoma of the Hypopharynx Stage III Squamous Cell Carcinoma of the Larynx Stage III Squamous Cell Carcinoma of the Lip and Oral Cavity Stage III Squamous Cell Carcinoma of the Nasopharynx Stage III Squamous Cell Carcinoma of the Oropharynx Stage III Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage III Verrucous Carcinoma of the Larynx Stage III Verrucous Carcinoma of the Oral Cavity Stage IV Salivary Gland Cancer Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Larynx Stage IV Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IV Squamous Cell Carcinoma of the Nasopharynx Stage IV Squamous Cell Carcinoma of the Oropharynx Stage IV Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IV Verrucous Carcinoma of the Larynx Stage IV Verrucous Carcinoma of the Oral Cavity Tongue Cancer Untreated Metastatic Squamous Neck Cancer With Occult Primary Collapse << | Phase 1 | Terminated | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637-1470 Decatur Memorial Hospital Decatur, Illinois, United States, 62526 Evanston CCOP-NorthShore University HealthSystem Evanston, Illinois, United States, 60201 Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 Loyola University Medical Center Maywood, Illinois, United States, 60153 Illinois CancerCare-Peoria Peoria, Illinois, United States, 61615 Central Illinois Hematology Oncology Center Springfield, Illinois, United States, 60702 United States, Indiana Fort Wayne Medical Oncology and Hematology Inc - State Boulevard Fort Wayne, Indiana, United States, 46845 United States, Maryland University of Maryland Greenebaum Cancer Center Baltimore, Maryland, United States, 21201-1595 United States, Michigan University of Michigan University Hospital Ann Arbor, Michigan, United States, 48109 United States, Missouri Saint John's Mercy Medical Center Saint Louis, Missouri, United States, 63141 United States, Wisconsin Froedtert and the Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00398112 | B-cell Chronic Lymphocytic Leu... more >>kemia Recurrent Small Lymphocytic Lymphoma Refractory Chronic Lymphocytic Leukemia Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> North Central Cancer Treatment Group Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00511849 | Neoplasms | Phase 1 | Completed | - | United States, Michigan ... more >> Pfizer Investigational Site Detroit, Michigan, United States, 48201 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77030 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01121562 | - | - | Completed | - | - |
| NCT01215565 | Hepatocellular Carcinoma ... more >> Fibrolamellar Hepatocellular Carcinoma Collapse << | Phase 2 | Terminated(lack of inclusion) | - | France ... more >> Hôpital Beaujon Clichy, Hauts de Seine, France, 92110 Collapse << |
| NCT01402089 | Non Small-cell Lung Cancer ... more >> Renal-cell Cancer Gastrointestinal Stroma Tumor Collapse << | Phase 4 | Completed | - | Switzerland ... more >> Cantonal Hospital St.Gallen St.Gallen, Switzerland, 9007 Collapse << |
| NCT00903175 | - | - | Completed | - | - |
| NCT01210053 | Non-small Cell Lung Cancer | Phase 2 | Unknown | March 2012 | China, Chongqing ... more >> Daping Hospital and the Research Institute of Surgery of the Third Military Medical University Recruiting Chongqing, Chongqing, China, 400042 Contact: yang zhenzhou, doctor yangzhenzhou@sohu.com Collapse << |
| NCT00903175 | Renal Cell Carcinoma | Phase 2 | Completed | - | - |
| NCT00421512 | Metastatic Renal Cell Carcinom... more >>a Kidney Cancer Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center 1275 York Avenue New York, New York, United States, 10021 Collapse << |
| NCT01850147 | Cancer Lung C... more >>ancer Collapse << | Phase 2 | Unknown | December 2014 | China ... more >> Cancer Institute and Hospital, Chinese Academy of Medical Sciences Recruiting Beijing, China, 100021 Collapse << |
| NCT01673386 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | - |
| NCT01070706 | Breast Cancer | Phase 1 Phase 2 | Completed | - | Korea, Republic of ... more >> Center for breast cancer, National Cancer Center Goyang, Kyeonggido, Korea, Republic of, 410-769 Collapse << |
| NCT01185366 | Kidney Cancer | Phase 2 | Active, not recruiting | August 2019 | United States, Massachusetts ... more >> Dana-Farber Cancer Institute/Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Texas University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 United States, Utah Huntsman Cancer Institute - University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00806663 | Metastatic Colorectal Cancer ... more >> Liver Metastases Collapse << | Phase 2 | Completed | - | Germany ... more >> Innere Univ.-Klinik u. Poliklinik Tumorforschung Essen, Germany, D-45122 Klinik für Tumorbiologie Freiburg, Germany, D-79106 Medizinische Universitätsklinik der Albert-Ludwigs-Universität Freiburg Freiburg, Germany, D-79106 Marienhospital Herne, Germany, D-44625 Collapse << |
| NCT03641326 | Gliosarcoma C... more >>entral Nervous System Sarcoma Collapse << | Phase 2 | Not yet recruiting | September 30, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Not yet recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01164228 | Kidney Cancer | Phase 2 | Active, not recruiting | May 31, 2023 | - |
| NCT02761057 | Stage III Renal Cell Cancer AJ... more >>CC v7 Stage IV Renal Cell Cancer AJCC v7 Type 1 Papillary Renal Cell Carcinoma Type 2 Papillary Renal Cell Carcinoma Unresectable Renal Cell Carcinoma Collapse << | Phase 2 | Suspended(Scheduled Interim Mo... more >>nitoring) Collapse << | - | - |
| NCT00617253 | Cancer Renal ... more >>Cell Carcinoma Collapse << | Phase 2 | Completed | - | Germany ... more >> Novo Nordisk Investigational Site Frankfurt, Germany, 60488 Novo Nordisk Investigational Site Hamburg, Germany, 20246 Novo Nordisk Investigational Site Hannover, Germany, 30625 Netherlands Novo Nordisk Investigational Site Amsterdam, Netherlands, 1066 CX Novo Nordisk Investigational Site Nijmegen, Netherlands, 6525 GA Collapse << |
| NCT01829841 | Renal Cell Cancer ... more >> Metastatic Renal Cell Cancer Collapse << | Phase 2 | Completed | - | China ... more >> Cancer Institute and Hospital Chinese Academy of Medical Sciences Beijing, China, 100021 Collapse << |
| NCT01907607 | Advanced Gastrointestinal Stro... more >>mal Tumors Collapse << | Phase 2 | Active, not recruiting | April 2019 | France ... more >> Institut Bergonié Bordeaux, Gironde, France, 33076 Centre Georges-Francois Leclerc Dijon, France, 21079 Centre Oscar Lambret Lille, France, 59020 Centre Léon Bérard Lyon, France, 69373 Hôpital de la Timone - AP-HM Marseille, France, 13385 Centre René Gauducheau Nantes, France, 44805 Hôpital Saint-Antoine (AP-HP) Paris, France, 75571 CHU de REIMS - Hôpital Robert Debré Reims, France, 51092 Institut Gustave Roussy Villejuif, France, 94800 Collapse << |
| NCT00570635 | Gastrointestinal Stromal Tumor... more >>s Gastrointestinal Neoplasms Collapse << | Phase 2 | Completed | - | United States, California ... more >> Los Angeles, California, United States, 90095 United States, Illinois Park Ridge, Illinois, United States, 60068 United States, Massachusetts Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00489944 | Intraocular Melanoma | Phase 2 | Unknown | - | United States, California ... more >> San Diego Pacific Oncology and Hematology Associates, Incorporated - Encinitas Recruiting Encinitas, California, United States, 92024 Contact: Edward F. McClay, MD 760-452-3340 emcclay@pacificoncology.com Collapse << |
| NCT00979329 | - | - | Completed | - | Netherlands ... more >> University Medical Center Nijmegen st Radboud Nijmegen, Netherlands, 6525 GH Collapse << |
| NCT00930345 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 4 | Terminated | - | France ... more >> HEGP Paris, France, 75015 Collapse << |
| NCT00471328 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT00471328 | - | - | Completed | - | - |
| NCT00491738 | Renal Cell Carcinoma | Phase 2 | Terminated(Based on the data c... more >>ollected, the combination of bevacizumab and sunitinib appeared to be poorly tolerated.) Collapse << | - | - |
| NCT01216371 | Renal Cell Carcinoma ... more >> Pulmonary Metastases Collapse << | Phase 2 | Unknown | October 2015 | Germany ... more >> Universitätsmedizin Charité Berlin Not yet recruiting Berlin, Germany, 10117 Contact: Steffen Weikert, Priv. Doz. Dr. med. 030/84 45-40 84 Principal Investigator: Steffen Weikert, Priv. Doz. Dr. med. Sub-Investigator: Kempkensteffen Carsten, Dr. med. Sub-Investigator: Jonas Busch, Dr. med. Sub-Investigator: Jan Gregor, Dr. med. Sub-Investigator: Jens Neudecker, Dr. med. Sub-Investigator: Jens C. Rückert, Dr. med. Sub-Investigator: Gertrud Feldmann Franziskus Krankenhaus Not yet recruiting Berlin, Germany, 10787 Contact: Jörg Neymeyer, Dr. med. 030/26 38-38 01 Principal Investigator: Jörg Neymeyer, Dr. med. Sub-Investigator: Thomas Wülfing, Dr. med. Helios Klinikum Emil von Behring Not yet recruiting Berlin, Germany, 14165 Contact: Kollmeier Jens, Dr. med. 030/81 02-14 47 Sub-Investigator: Jens Kollmeier, Dr. med. Sub-Investigator: Bettina Schlolaut, Dr. med. university hospital of Düsseldorf Recruiting Düsseldorf, Germany, 40255 Contact: Peter Albers, Prof. Dr. med. 0211/811-81 10 Principal Investigator: Peter Albers, Prof. Dr. med. Sub-Investigator: Jasper Decoene Sub-Investigator: Zenginli Hakan Sub-Investigator: Claus F. Eisenberger, Prof. Dr. med. Sub-Investigator: Wolfram T. Knoefel, Prof. Dr. med. Sub-Investigator: Almut Diem university hospital of Essen Recruiting Essen, Germany, 45122 Contact: Herbert Rübben, Prof. Dr. med. 0201/723-32 10 Principal Investigator: Herbert Rübben, Prof. Dr. med. Sub-Investigator: Marcus Schenk, Dr. med. Sub-Investigator: Farnk vom Dorp, Dr. med. Sub-Investigator: Min Ju, Dr. med. Sub-Investigator: Stephan Tschirdewahn Sub-Investigator: Michaela Löbert Ruhrlandklinik Department of Thoracic Surgery Not yet recruiting Essen, Germany, 45239 Contact: Christiane Zimmermann 0201/433-43 24 Sub-Investigator: Georgios Stamatis, Prof. Dr. med. Sub-Investigator: Stefan Welter, Dr. med. Sub-Investigator: Silvia Fechner, Dipl. med. university hospital of Freiburg Recruiting Freiburg, Germany, 79106 Contact: Ulrich Wetterauer, Prof. Dr. med. 0761/270-2891 Principal Investigator: Ulrich Wetterauer, Prof. Dr. med. Sub-Investigator: Christian Leiber, Dr. med. Sub-Investigator: Christian Stremmel, Prof. Dr. med. Sub-Investigator: Jutta Günter, Dr. med. university Hospital of Heidelberg Recruiting Heidelberg, Germany, 69119 Contact: Markus Hohenfellner, Prof. Dr. med. 06221/56-63 21 Principal Investigator: Markus Hohenfellner, Prof. Dr. med. Sub-Investigator: Pahernick Sascha, Priv. Doz. Dr. med. Sub-Investigator: Gencay Hatiboglu, Dr. med. Sub-Investigator: Johannes Huber, Dr. Dr. med. Sub-Investigator: Joachim Pfannschmidt, Priv. Doz. Dr. med. Sub-Investigator: Hendrik Dienemann, Prof. Dr. med. urological hospital of Maria Hilf Krankenhaus Krefeld Recruiting Krefeld, Germany, 47805 Contact: Susanne Krege, Priv. Doz. Dr. med. 02151/334-52 74 Principal Investigator: Susanne Krege, Priv. Doz. Dr. med. Sub-Investigator: Florian Hartmann, Dr. med. Hospital of Großhadern Not yet recruiting München, Germany, 81377 Contact: Michael Staehler, Dr. med. 089/70 95-0 Principal Investigator: Michael Staehler, Dr. med. Sub-Investigator: Cordula Nordhaus, Dr. med. Sub-Investigator: Philipp Nuhn, Dr. med. Sub-Investigator: Hauke Winter, Priv. Doz. Dr. med. Sub-Investigator: Rudolf Hatz, Prof. Dr. med. Sub-Investigator: Sylvia Dondl Dr.-Horst-Schmidt-Kliniken GmbH Recruiting Wiesbaden, Germany, 65199 Contact: Norbert Frickhofen, Prof. Dr. med. 0611/43-30 09 Principal Investigator: Norbert Frickhofen, Prof. Dr. med. Sub-Investigator: Bernd Jung, Dr. med. Sub-Investigator: Heinz G. Fuhr, Dr. med. Sub-Investigator: Joachim Schirren, Prof. Dr. med. Sub-Investigator: Servet Bölükbas, Dr. med. Sub-Investigator: Sabine Labenz Sub-Investigator: Birgit Wilde Sub-Investigator: Klaudia Fischbach Collapse << |
| NCT00532064 | - | - | Terminated(Terminated per PI's... more >> request) Collapse << | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00075218 | Gastrointestinal Stromal Tumor | Phase 3 | Completed | - | - |
| NCT00457392 | Carcinoma, Non-Small Cell Lung | Phase 3 | Completed | - | - |
| NCT00075218 | - | - | Completed | - | - |
| NCT00434356 | Metastatic Breast Cancer | Phase 2 | Terminated(Based on data colle... more >>cted, the combination appeared to be poorly tolearated.) Collapse << | - | - |
| NCT00491738 | - | - | Terminated(Based on the data c... more >>ollected, the combination of bevacizumab and sunitinib appeared to be poorly tolerated.) Collapse << | - | - |
| NCT00626509 | Kidney Cancer | Phase 2 | Unknown | - | Italy ... more >> Ospedale Sacro Cuore Recruiting Negrar, Italy, 37024 Contact: Contact Person 39-045-601-3912 Collapse << |
| NCT00434356 | - | - | Terminated(Based on data colle... more >>cted, the combination appeared to be poorly tolearated.) Collapse << | - | - |
| NCT00457392 | - | - | Completed | - | - |
| NCT00709995 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Active, not recruiting | December 2018 | Austria ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vienna, Austria, A1090 France For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Villejuif, France, 94805 Italy For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rome, Italy, 00149 Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Warsaw, Poland, 00909 Collapse << |
| NCT03519542 | - | - | Active, not recruiting | June 2018 | Spain ... more >> Hospital Clínico Universitario de Santiago de Compostela Santiago de Compostela, A Coruña, Spain, 15706 Hospital Universitario central de Asturias Oviedo, Asturias, Spain, 33011 Hospital Marqués de Valdecilla Santander, Cantabria, Spain, 39008 Hospital Universitarios de Burgos Burgos, Spain, 09006 Hospital Universiario de León León, Spain, 24080 Hospital Gregorio Marañón Madrid, Spain, 28007 Hospital Universitario Ramón y Cajal Madrid, Spain, 28024 Hospital Clínico (Madrid) Madrid, Spain, 28040 Hospital 12 de Octubre Madrid, Spain, 28041 Hospital Universitario La Paz Madrid, Spain Hospital Universitario Carlos Haya Málaga, Spain, 29010 Hospital Clínico Lozano Blesa Zaragoza, Spain, 50009 Collapse << |
| NCT01246843 | - | - | Completed | - | Netherlands ... more >> University Medical Centre Nijmegen Nijmegen, Gelderland, Netherlands, 6500 HB Collapse << |
| NCT02428335 | - | - | Unknown | June 2017 | Canada, Alberta ... more >> Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Collapse << |
| NCT00537056 | Kidney Neoplasms ... more >> Carcinoma, Renal Cell Kidney (Renal Cell) Cancer Collapse << | Not Applicable | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00732914 | Renal Cell Carcinoma | Phase 3 | Completed | - | Germany ... more >> Urologische Klinik Mannheim, Baden-Wuerttemberg, Germany, 68167 Collapse << |
| NCT01014065 | - | - | Completed | - | Canada, Alberta ... more >> University of Alberta/ Cross Cancer Institute Edmonton, Alberta, Canada Collapse << |
| NCT00077987 | Colorectal Neoplasms | Phase 2 | Completed | - | - |
| NCT01122888 | Adult Giant Cell Glioblastoma ... more >> Adult Glioblastoma Adult Gliosarcoma Adult Solid Neoplasm Recurrent Adult Brain Neoplasm Collapse << | Phase 1 | Terminated | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637 Collapse << |
| NCT03013946 | Renal Cell Carcinoma, Metastat... more >>ic Renal Cell Cancer, Recurrent Collapse << | Phase 3 | Recruiting | May 2020 | Germany ... more >> Medizinische Hochschule Hannover Not yet recruiting Hannover, Germany, 30625 Contact: Viktor Grünwald, Dr. +49 511 532 ext 2301 gruenwald.viktor@mh-hannover.de Zentrum für Onkologie und Urologie Rostock Recruiting Rostock, Germany, 18107 Contact: Andreas Hübner, Dr. Collapse << |
| NCT00915993 | Kidney Cancer | Not Applicable | Unknown | - | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104-4283 Contact: Clinical Trials Office - Abramson Cancer Center of the Univers 800-474-9892 Collapse << |
| NCT00553696 | Stomach Neoplasms | Phase 1 | Completed | - | Japan ... more >> Aichi cancer center central hospital / Medical Oncology Nagoya, Aichi, Japan, 464-8681 Saku Central Hospital, GI Devision Saku, Nagano, Japan Shizuoka Cancer Center Suntougun, Shizuoka, Japan, 411-8777 Collapse << |
| NCT02184416 | - | - | Recruiting | July 20, 2019 | - |
| NCT00553696 | - | - | Completed | - | - |
| NCT00226811 | - | - | Completed | - | - |
| NCT03075423 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | December 31, 2022 | Germany ... more >> Medizinische Hochschule Hannover Recruiting Hannover, Niedersachsen, Germany, 30625 Charité Universitätsmedizin Berlin Recruiting Berlin, Germany University Hospital Erlangen Recruiting Erlangen, Germany, 91054 Contact: Stefan Krause, Prof. Universitätsklinikum Essen Recruiting Essen, Germany Goethe University Frankfurt Recruiting Frankfurt am Main, Germany, D-60590 Contact: Lothar Prof. Dr. med. Bergmann, MD +40 69 63015121 l.bergmann@em.uni-frankfurt.de Contact: Nicola Goekbuget, MD Uiversitätsklinikum Hamburg-Eppendorf Recruiting Hamburg, Germany Uniklinikum Recruiting Jena, Germany Universitätsklinkum Gießen und Marburg Recruiting Marburg, Germany Klinikum Rechts der Isar der TU München Recruiting München, Germany University of Muenster Recruiting Münster, Germany, 48149 Universitätsklinik Tübingen Recruiting Tübingen, Germany Medizinische Universitätsklinik Ulm Recruiting Ulm, Germany, 89070 Collapse << |
| NCT00078000 | Breast Neoplasms | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Greenbrae, California, United States, 94904 Pfizer Investigational Site Los Angeles, California, United States, 90095 Pfizer Investigational Site San Francisco, California, United States, 94115 United States, Colorado Pfizer Investigational Site Aurora, Colorado, United States, 80010 United States, Illinois Pfizer Investigational Site Chicago, Illinois, United States, 60612-3824 United States, Indiana Pfizer Investigational Site Indianapolis, Indiana, United States, 46202 United States, Maryland Pfizer Investigational Site Baltimore, Maryland, United States, 21231 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00434226 | Non-Small Cell Lung Cancer | Phase 2 | Terminated(Based on data colle... more >>cted, the combination appeared to be poorly tolearated.) Collapse << | - | - |
| NCT00434226 | - | - | Terminated(Based on data colle... more >>cted, the combination appeared to be poorly tolearated.) Collapse << | - | - |
| NCT00226811 | Stomach Neoplasms | Phase 2 | Completed | - | China, Jiangsu ... more >> Pfizer Investigational Site Nanjing, Jiangsu, China, 210002 China Pfizer Investigational Site Beijing, China, 100036 Pfizer Investigational Site Beijing, China Pfizer Investigational Site Guangzhou, China, 510515 Hong Kong Pfizer Investigational Site Hong Kong, Hong Kong Pfizer Investigational Site Shatin, Hong Kong Italy Pfizer Investigational Site Ancona, Italy, 60020 Pfizer Investigational Site Genova, Italy, 16132 Japan Pfizer Investigational Site Kashiwa, Chiba, Japan Pfizer Investigational Site Suntougun, Shizuoka, Japan Pfizer Investigational Site Chuo-ku, Tokyo, Japan Korea, Republic of Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Pfizer Investigational Site Seoul, Korea, Republic of, 120-752 Pfizer Investigational Site Seoul, Korea, Republic of, 135-710 Pfizer Investigational Site Seoul, Korea, Republic of, 138-736 Portugal Pfizer Investigational Site Porto, Portugal, 4200-072 Taiwan Pfizer Investigational Site Kwei-Shan, Taoyuan, Taiwan, 333 Pfizer Investigational Site Taipei, Taiwan, 100 Collapse << |
| NCT00836745 | - | - | Completed | - | India ... more >> Pfizer Investigational Site New Delhi, Delhi, India, 110 085 Pfizer Investigational Site Bangalore, Karnataka, India, 560 027 Pfizer Investigational Site Mumbai, Maharashtra, India, 400014 Pfizer Investigational Site Chandigard, Punjab, India, 141402 Pfizer Investigational Site Kolkata, West Bengal, India, 700 106 Pfizer Investigational Site Delhi, India, 110 060 Pfizer Investigational Site Jaipur, India, 302006 Collapse << |
| NCT00836745 | - | - | Completed | - | - |
| NCT00137449 | - | - | Completed | - | - |
| NCT00137449 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Pfizer Investigational Site Boston, Massachusetts, United States, 02115 France Pfizer Investigational Site Lyon Cedex 08, France, 69373 Pfizer Investigational Site Villejuif, France, 94805 Italy Pfizer Investigational Site Milano, Italy, 20133 Collapse << |
| NCT02315625 | Neuroendocrine Tumors ... more >> Neuroendocrine Carcinoma Neuroendocrine Neoplasms Carcinoma, Neuroendocrine Neuroendocrine Tumors of the Gastrointestinal Tract and Pancreas Collapse << | Phase 2 | Recruiting | December 31, 2025 | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: Xavier Keutgen, MD 312-563-1557 xavier_keutgen@rush.edu United States, Maryland National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00718562 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative Site Nagoya, Aichi, Japan, 464-8681 Novartis Investigative Site Kashiwa, Chiba, Japan, 277-8577 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 812-8582 Novartis Investigative Site Sapporo-city, Hokkaido, Japan, 060-8648 Novartis Investigative Site Niigata-city, Niigata, Japan, 951-8520 Novartis Investigative Site Suita-city, Osaka, Japan, 565-0871 Novartis Investigative Site Sunto-gun, Shizuoka, Japan, 411-8777 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 104-0045 Collapse << |
| NCT00537056 | - | - | Completed | - | - |
| NCT00435409 | Breast Neoplasms | Phase 3 | Completed | - | - |
| NCT01396408 | Advanced Rare Tumours | Phase 2 | Active, not recruiting | December 2018 | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia BCCA - Cancer Centre for the Southern Interior Kelowna, British Columbia, Canada, V1Y 5L3 BCCA - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Manitoba CancerCare Manitoba Winnipeg, Manitoba, Canada, R3E 0V9 Canada, Nova Scotia QEII Health Sciences Centre Halifax, Nova Scotia, Canada, B3H 1V7 Izaak Walton Killam (IWK) Health Centre Halifax, Nova Scotia, Canada, B3K 6R8 Canada, Ontario Juravinski Cancer Centre at Hamilton Health Sciences Hamilton, Ontario, Canada, L8V 5C2 London Regional Cancer Program London, Ontario, Canada, N6A 4L6 Ottawa Hospital Research Institute Ottawa, Ontario, Canada, K1H 8L6 Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Univ. Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec CHUM - Hopital Notre-Dame Montreal, Quebec, Canada, H2L 4M1 McGill University - Dept. Oncology Montreal, Quebec, Canada, H2W 1S6 CHU Sainte-Justine Montreal, Quebec, Canada, H3T 1C5 Canada, Saskatchewan Allan Blair Cancer Centre Regina, Saskatchewan, Canada, S4T 7T1 Saskatoon Cancer Centre Saskatoon, Saskatchewan, Canada, S7N 4H4 Collapse << |
| NCT03424876 | - | - | Recruiting | August 8, 2018 | China, Beijing ... more >> Beijing Cancer Hospital Recruiting Beijing, Beijing, China, 100142 Contact: Jian Li, MD Collapse << |
| NCT02811861 | Renal Cell Carcinoma | Phase 3 | Recruiting | February 2021 | - |
| NCT00923117 | - | - | Terminated(The study was termi... more >>nated based on results of an interim analysis for futility.) Collapse << | - | - |
| NCT00435409 | - | - | Completed | - | - |
| NCT00923117 | Glioblastoma Multiforme ... more >> Malignant Gliomas Anaplastic Gliomas Collapse << | Phase 2 | Terminated(The study was termi... more >>nated based on results of an interim analysis for futility.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01506336 | Gastro Intestinal Stromal Tumo... more >>r Collapse << | Phase 2 | Completed | - | France ... more >> Institute Gustave Roussy (IGR) Villejuif, France, 94815 Collapse << |
| NCT00417677 | Renal Cell Carcinoma | Phase 1 Phase 2 | Terminated(Decision was made n... more >>ot to attempt a lower dose) Collapse << | - | United States, Maryland ... more >> Baltimore, Maryland, United States, 21231 United States, Massachusetts Boston, Massachusetts, United States, 02215 United States, New York New York, New York, United States, 10021 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT00790595 | Prostate Cancer | Phase 1 | Completed | - | United States, California ... more >> University of California, Los Angeles, Jonsson Comprehensive Cancer Center Los Angeles, California, United States, 90095 Collapse << |
| NCT00979966 | Non-clear Cell Renal Cell Canc... more >>er Collapse << | Phase 2 | Completed | - | Germany ... more >> Charité - Campus Virchow Klinikum Berlin, Germany Charité - Mitte Berlin, Germany Vivantes Klinikum am Urban Berlin, Germany Evangelische Kliniken Bonn gGmbH - Johanniter-Krankenhaus Bonn, Germany Universitätsklinikum Düsseldorf Düsseldorf, Germany Universitätsklinikum Essen Essen, Germany Klinikum der J.W. Goethe Universität Frankfurt, Germany Martin-Luther-Universität Halle-Wittenberg Halle, Germany Universitätskrankenhaus Jena Jena, Germany UK-SH Campus Lübeck Lübeck, Germany Klinikum Oldenburg gGmbH Oldenburg, Germany Klinikum Stuttgart, Katharinenhospital Stuttgart, Germany Facharzt für Innere Medizin, Viersen, Germany Kliniken Nordoberpfalz AG - Klinikum Weiden Weiden, Germany Collapse << |
| NCT02570789 | Clear-cell Metastatic Renal Ce... more >>ll Carcinoma Collapse << | Phase 4 | Active, not recruiting | December 2018 | Belgium ... more >> Cliniques universitaires Saint-Luc Brussel, Belgium, 1200 Collapse << |
| NCT00936832 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Withdrawn(because the sunitini... more >>b showed futility in anotehr trial) Collapse << | - | France ... more >> Hopital Ambroise Pare Boulogne Billancourt, France, 92100 Collapse << |
| NCT03141177 | Renal Cell Carcinoma | Phase 3 | Recruiting | April 22, 2023 | - |
| NCT00570908 | Breast Cancer | Phase 2 | Terminated(Due to poor accrual... more >> this study is being closed to accrual) Collapse << | - | United States, Texas ... more >> Lester and Sue Smith Breast Center at Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00570908 | - | - | Terminated(Due to poor accrual... more >> this study is being closed to accrual) Collapse << | - | - |
| NCT03729245 | Renal Cell Carcinoma ... more >> Metastatic Renal Cell Carcinoma Collapse << | Phase 3 | Not yet recruiting | June 2024 | United States, California ... more >> Investigator Site - Whittier Not yet recruiting Whittier, California, United States, 90603 United States, Texas Investigator Site - Houston Not yet recruiting Houston, Texas, United States, 77030 United States, Virginia Investigator Site - Leesburg Not yet recruiting Leesburg, Virginia, United States, 20176 Australia, South Australia Investigator Site - Kurralta Park Not yet recruiting Kurralta Park, South Australia, Australia, 5037 Collapse << |
| NCT02401815 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 1 Phase 2 | Active, not recruiting | March 2020 | United States, Florida ... more >> Sylvester Comprehensive Cancer Center/ UMHC Miami, Florida, United States, 33136 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, Ohio OSU Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT02230176 | Pancreatic Neuroendocrine Carc... more >>inoma Collapse << | Phase 2 | Recruiting | October 2023 | France ... more >> Gustave Roussy Recruiting Villejuif, Val De Marne, France, 94805 Contact: Lisa LAMBERT 0142116643 ext +33 lisa.lambert@gustaveroussy.fr Principal Investigator: Eric BAUDIN, MD, PhD Collapse << |
| NCT00373256 | Breast Neoplasms | Phase 3 | Completed | - | - |
| NCT00054886 | Kidney Neoplasms | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Los Angeles, California, United States, 90095 Pfizer Investigational Site Mission Hills, California, United States, 91345 Pfizer Investigational Site Monterey Park, California, United States, 91754 Pfizer Investigational Site Northridge, California, United States, 91328 Pfizer Investigational Site San Francisco, California, United States, 94115 Pfizer Investigational Site San Francisco, California, United States, 94121 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 United States, Michigan Pfizer Investigational Site Ann Arbor, Michigan, United States, 48109 United States, Nevada Pfizer Investigational Site Henderson, Nevada, United States, 89052 Pfizer Investigational Site Las Vegas, Nevada, United States, 89109 Pfizer Investigational Site Las Vegas, Nevada, United States, 89128 United States, New York Pfizer Investigational Site New York, New York, United States, 10021 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01381822 | Advanced Renal Cell Carcinoma ... more >> Gastrointestinal Stromal Tumors Pancreatic Neuroendocrine Tumors Collapse << | Phase 1 | Unknown | June 2014 | United States, Indiana ... more >> IU Health Goshen Center for Cancer Care Goshen, Indiana, United States, 46526 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 Collapse << |
| NCT01371201 | Malignant Progressive Pheochro... more >>mocytoma and Paraganglioma (PPGL) Collapse << | Phase 2 | Recruiting | June 2021 | France ... more >> Institut de Cancérologie Gustave roussy Recruiting Villejuif, France, 94805 Contact: Eric Baudin, MD eric.baudin@igr.fr Principal Investigator: Eric Baudin, MD Sub-Investigator: Martin Schlumberger, MD Sub-Investigator: Sophie Leboulleux, MD Sub-Investigator: Cécile Chougnet, MD Germany Universitätsklinikum Würzburg Recruiting Würburg, Germany, 97080 Contact: Martin FASSNACHT, MD 0931-201-39021 Principal Investigator: Martin FASSNACHT, MD Italy University of Padova Recruiting Padova, Italy, 35128 Contact: Giusepe OPOCHER, MD 0498215503 ext +39 Principal Investigator: Giusepe OPOCHER, MD Netherlands Radboud University Nijmegen Medical Centre Recruiting Nijmegen, GA, Netherlands, 6525 Contact: Henry TIMMERS, MD 0243614599 ext +31 Principal Investigator: Henry TIMMERS, MD Collapse << |
| NCT00094029 | - | - | - | - | - |
| NCT00555672 | - | - | Completed | - | - |
| NCT00616122 | Breast Cancer | Phase 1 Phase 2 | Completed | - | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 Collapse << |
| NCT00373256 | - | - | Completed | - | - |
| NCT01246778 | - | - | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Centre Nijmegen, Netherlands, 6500HB Collapse << |
| NCT00748358 | Prostatic Neoplasms ... more >> Neoplasms, Hormone-Dependent Tumor Markers, Biological Survival Rate Disease-Free Survival Collapse << | Phase 2 | Completed | - | France ... more >> Service Oncologie Médicale, Hopital Europeen Georges Pompidou Paris, France, 75015 Collapse << |
| NCT00555672 | Stomach Neoplasms | Phase 1 | Completed | - | Spain ... more >> Pfizer Investigational Site L'hospitalet de Llobregat, Barcelona, Spain, 08907 Pfizer Investigational Site Barcelona, Spain, 08003 Pfizer Investigational Site Madrid, Spain, 28041 Collapse << |
| NCT00616122 | - | - | Completed | - | - |
| NCT01664182 | Stage III Renal Cell Cancer AJ... more >>CC v7 Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 2 | Active, not recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 USC / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 City of Hope South Pasadena South Pasadena, California, United States, 91030 United States, Michigan Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Metro Minnesota Community Oncology Research Consortium Saint Louis Park, Minnesota, United States, 55416 United States, New Jersey Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Pennsylvania Penn State Milton S Hershey Medical Center Hershey, Pennsylvania, United States, 17033-0850 University of Pittsburgh Cancer Institute (UPCI) Pittsburgh, Pennsylvania, United States, 15232 United States, Virginia VCU Massey Cancer Center at Hanover Medical Park Mechanicsville, Virginia, United States, 23116 Collapse << |
| NCT00372853 | Carcinoma, Renal Cell | Phase 1 | Completed | - | United States, Arizona ... more >> Research Site Scottsdale, Arizona, United States, 85258 United States, New Jersey Research Site New Brunswick, New Jersey, United States, 08903 United States, Ohio Research Site Cleveland, Ohio, United States, 44195 United States, South Carolina Research Site Greenville, South Carolina, United States, 29605 United States, Tennessee Research Site Memphis, Tennessee, United States, 38120 Collapse << |
| NCT00113516 | Non-small Cell Lung Cancer | Phase 2 | Completed | - | - |
| NCT00130897 | - | - | - | - | - |
| NCT00137423 | Carcinoma, Renal Cell Metastas... more >>is Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Stanford, California, United States, 94305 United States, Nevada Pfizer Investigational Site Las Vegas, Nevada, United States, 89135 France Pfizer Investigational Site Villejuif, France, 94805 Germany Pfizer Investigational Site Berlin, Germany, 10117 Pfizer Investigational Site Muenchen, Germany, 81664 Greece Pfizer Investigational Site Thessaloniki, Greece, 56429 Netherlands Pfizer Investigational Site Nijmegen, Gld, Netherlands, 6525 GA Sweden Pfizer Investigational Site Lund, Sweden, SE-221 85 Pfizer Investigational Site Stockholm, Sweden, 171 76 Switzerland Pfizer Investigational Site St. Gallen, Switzerland, CH-9007 Collapse << |
| NCT00430261 | Lung Cancer A... more >>denocarcinoma Collapse << | Phase 2 | Completed | - | United States, Washington ... more >> Swedish Cancer Institute Seattle, Washington, United States, 98104 Collapse << |
| NCT01694277 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Recruiting | December 2022 | France ... more >> Institut Bergonié Recruiting Bordeaux, France, 33000 Collapse << |
| NCT00083889 | - | - | Completed | - | - |
| NCT00137423 | - | - | Completed | - | - |
| NCT00243503 | - | - | Completed | - | - |
| NCT00113516 | - | - | Completed | - | - |
| NCT01459757 | - | - | Completed | - | - |
| NCT00631410 | Colorectal Neoplasms | Phase 1 | Completed | - | Japan ... more >> Pfizer Investigational Site Kashiwa, Chiba, Japan Pfizer Investigational Site Suntougun, Shizuoka, Japan Pfizer Investigational Site Chuo-ku, Tokyo, Japan Collapse << |
| NCT00083889 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT00631410 | - | - | Completed | - | - |
| NCT02187042 | - | - | Completed | - | France ... more >> CHU de la Timone Marseille, Cedex 5, France, 13335 CHU Strasbourg Strasbourg, Cedex, France, 67091 Centre Hospitalier d'Annecy Annecy Cedex, France, 74011 Centre Catalan Urologie Andrologie Cabestany, France, 66330 Centre hospitalier de Chambery Chambery, France, 73000 Hopital prive La Louviere Lille, France, 59042 Clinique Claude Bernard Chimiotherapie Ambulatoire Metz, France, 57000 CRLC Val d'Aurelle Montpellier cedex 05, France, 34295 Polyclinique Gentilly Nancy, France, 54000 Polyclinique de Gentilly Service Oncologie Médicale Nancy, France, 54100 CHI de Cornouailles - Oncologie hospitalisation Quimper Cedex, France, 29107 Hopital Jean BERNARD - Tours - 7eme etage Valenciennes, France, 59300 Centre Alexis Vautrin Vandoeuvre les Nancy, France, 54511 Collapse << |
| NCT00371553 | Relapsed or Cisplatin-Refracto... more >>ry Germ Cell Cancer Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T4N 4N2 Canada, British Columbia BC Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Manitoba Cancer Care Manitoba Winnipeg, Manitoba, Canada, R3E 0V9 Canada, Nova Scotia QEII Health Science Center Halifax, Nova Scotia, Canada, B3H 2Y9 Canada, Ontario London Regional Cancer Centre London, Ontario, Canada, N6A 4L6 Princess Margaret Hospital Toronto, Ontario, Canada, M5G2M9 Collapse << |
| NCT00904657 | Blood Pressure | Phase 1 | Completed | - | - |
| NCT00531544 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Not Applicable | Completed | - | Saudi Arabia ... more >> KFSH&RC Riyadh, Saudi Arabia, 1211 Collapse << |
| NCT00676650 | Prostatic Neoplasms | Phase 3 | Terminated(Study A6181120 was ... more >>prematurely discontinued due to futility on 27 September 2010. No new or unexpected safety issues were identified.) Collapse << | - | - |
| NCT02019576 | Clear Cell Metastatic Renal Ce... more >>ll Carcinoma Collapse << | Phase 2 | Recruiting | December 2019 | Canada, Alberta ... more >> Tom Baker Cancer Centre Recruiting Calgary, Alberta, Canada, T2N 4N2 Contact: Gerald Lim, MD Gerald.Lim@albertahealthservices.ca Principal Investigator: Gerald Lim, MD Sub-Investigator: Daniel Heng, MD Cross Cancer Institute Recruiting Edmonton, Alberta, Canada, T6G 1Z2 Contact: Samir Patel, MD Samir.Patel2@albertahealthservices.ca Principal Investigator: Samir Patel, MD Sub-Investigator: Scott North, MD Canada, British Columbia BC Cancer Agency Recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Christian Kollmannsberger, MD ckollmannsberger@bccancer.bc.ca Principal Investigator: Christian Kollmannsberger, MD Canada, Manitoba Manitoba CancerCare Institute Recruiting Winnipeg, Manitoba, Canada, R3E 0V9 Contact: Arbind Dubey, MD adubey@cancercare.mb.ca Sub-Investigator: Piotr Czaykowski, MD Principal Investigator: Arbind Dubey, MD Canada, Nova Scotia QEII Health Sciences Centre Recruiting Halifax, Nova Scotia, Canada, B3H 2Y9 Contact: Lori Wood, MD lori.wood@cdha.nshealth.ca Principal Investigator: Lori Wood, MD Sub-Investigator: Derek Wilke, MD Canada, Ontario Juravinski Cancer Centre Recruiting Hamilton, Ontario, Canada, L8V 5C2 Contact: Anand Swaminath, MD Anand.swaminath@jcc.hhsc.ca Principal Investigator: Anand Swaminath, MD Sub-Investigator: Sebastian Hotte, MD London Health Sciences Centre Recruiting London, Ontario, Canada, N6A 4L6 Contact: Belal Ahmad, MD Belal.Ahmad@lhsc.on.ca Principal Investigator: Belal Ahmad, MD Sub-Investigator: George Rodrigues, MD Ottawa Hospital Cancer Centre Recruiting Ottawa, Ontario, Canada, K1H 8L6 Contact: Scott Morgan, MD smorgan@Ottawahospital.on.ca Principal Investigator: Scott Morgan, MD Sub-Investigator: Christina Canil, MD Odette Cancer Centre, Sunnybrook Health Sciences Centre Recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Georg Bjarnason, MD, FRCPC 416-480-5847 georg.bjarnason@sunnybrook.ca Contact: Nesan Bandali, RN 416-480-6100 ext 82139 nesan.bandali@sunnybrook.ca Principal Investigator: Georg Bjarnason, MD, FRCPC Principal Investigator: Patrick Cheung, MD Princess Margaret Hospital Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Srikala Sridhar, MD srikala.sridhar@uhn.on.ca Principal Investigator: Srikala Sridhar, MD Sub-Investigator: Anthony Brade, MD Canada, Quebec McGill University Health Centre Recruiting Montreal, Quebec, Canada, H3G 1A4 Contact: Fabio Cury, MD fabio.cury@gmail.com Principal Investigator: Fabio Cury, MD Jewish General Hospital Recruiting Montreal, Quebec, Canada, H3T 1E2 Contact: Tamim Niazi, MD tniazi@jgh.mcgill.ca Principal Investigator: Tamim Niazi, MD Collapse << |
| NCT00982657 | Solid Tumor | Phase 2 | Terminated(Refer to statement ... more >>in Summary Section/Detailed Description) Collapse << | - | United States, Arizona ... more >> Premiere Oncology of Arizona Scottsdale, Arizona, United States, 85258 United States, California Premiere Oncology, A Medical Corporation Santa Monica, California, United States, 90404 United States, Mississippi Boston Baskin Cancer Foundation Southaven, Mississippi, United States, 38671 United States, Oregon Providence Portland Medical Center Portland, Oregon, United States, 97213 United States, Tennessee Boston Baskin Cancer Foundation Bartlett, Tennessee, United States, 38133 Boston Baskin Cancer Foundation Germantown, Tennessee, United States, 38138 Boston Baskin Cancer Foundation Memphis, Tennessee, United States, 38120 Collapse << |
| NCT00673504 | Pancreatic Cancer | Phase 2 | Completed | - | Germany ... more >> Städtisches Klinikum Braunschweig Braunschweig, Germany, 38114 Universitätsklinikum Essen Essen, Germany, 45122 Krankenhaus Frankfurt, Germany, 60488 Klinikum der J. W. Goethe Universität; Zentrum der Inneren Medizin Frankfurt, Germany, 60590 Internistisches Facharztzentrum Frankfurt, Germany, 60596 Martin-Luther-Universität Halle-Wittenberg; Medizinische Fakultät Halle (Saale), Germany, 06097 Facharztpraxis Heidelberg, Germany, 69115 Klinikum Lüdenscheid Luedenscheid, Germany, 58515 Klinikum Nürnberg Nord Nürnberg, Germany, 90419 Robert-Bosch-Krankenhaus Stuttgart, Germany, 70376 Switzerland Kantonsspital St. Gallen St. Gallen, Switzerland, 9007 Collapse << |
| NCT00982657 | - | - | Terminated(Refer to statement ... more >>in Summary Section/Detailed Description) Collapse << | - | - |
| NCT01024920 | Carcinoma, Renal Cell | Phase 2 | Active, not recruiting | February 15, 2019 | Hungary ... more >> University of Pecs Medical School, Dept. of Oncotherapy Pecs, Hungary, 7624 Poland Ziemia Lubelska Oncological Center, Lublin Lublin, Poland, 20-099 Onco.Cent. - Instit. of Maria Sklodowskiej-Curie Warszawa, Poland, 02-781 Romania Military Central Clinical Emergency Hospital Bucharest, Romania, 010825 Sf. Nectarie Oncology Center, Craiova Craiova, Romania, 200347 ONCOLAB SRL, Craiova Craiova, Romania, 200385 Ukraine Municipal Establishment Cherkasy Oncology Centre Cherkasy, Ukraine, 18009 Bukovynsk State Medical University Chernivtsi, Ukraine, 58013 City Clinical Hospital #4, Dnipropetrovsk State Medical Academy Dnipropetrovks, Ukraine, 49102 CI of LRC Lviv Onco.Reg.Treat.&Diag.Cent. Lviv, Ukraine, 79031 Uzhgorod National University, Oncology Centre Uzhgorod, Ukraine, 88000 United Kingdom Addenbrooke's Hospital Cambridge, United Kingdom, CB2 0QQ Beatson West of Scotland Cancer Centre Glasgow, United Kingdom, G12 0YN Surrey Cancer Research Institute Guildford, United Kingdom, GU2 7WG St James's University Hospital Leeds, United Kingdom, LS9 7TF Collapse << |
| NCT00513695 | Inflammatory Breast Cancer ... more >> Male Breast Cancer Stage II Breast Cancer Stage IIIA Breast Cancer Stage IIIB Breast Cancer Stage IIIC Breast Cancer Collapse << | Phase 2 | Active, not recruiting | - | United States, Alaska ... more >> Anchorage Oncology Centre Anchorage, Alaska, United States, 99508 Katmai Oncology Group Anchorage, Alaska, United States, 99508 United States, Arizona Arizona Cancer Center Tucson, Arizona, United States, 85724-5024 United States, Idaho Saint Luke's Mountain States Tumor Institute Boise, Idaho, United States, 83712 United States, Washington Skagit Valley Hospital Mount Vernon, Washington, United States, 98273 Olympic Medical Center Port Angeles, Washington, United States, 98362 Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT03091192 | Carcinoma Car... more >>cinoma, Renal Cell Kidney Neoplasms Urologic Neoplasms Kidney Diseases Neoplasms by Site Enzyme Inhibitors Protein Kinase Inhibitors Collapse << | Phase 3 | Active, not recruiting | June 24, 2020 | - |
| NCT00243503 | Breast Neoplasms | Phase 2 | Completed | - | - |
| NCT00375674 | Kidney Neoplasms | Phase 3 | Completed | - | - |
| NCT00513695 | - | - | Active, not recruiting | - | - |
| NCT02450123 | Refractory Solid Tumors | Not Applicable | Recruiting | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of, 135-710 Contact: Joon Oh Park, M.D., Ph.D 82 2 3410 3459 oncopark@skku.edu Contact: yoon Jeong Ahn 221487395 ext 82 Collapse << |
| NCT00522249 | Carcinoma, Renal Cell | Phase 1 Phase 2 | Terminated(PI decision) | - | United States, Texas ... more >> Baylor College of Medicine - Methodist Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT00541008 | Kidney Cancer | Phase 2 | Unknown | - | France ... more >> Centre Paul Papin Recruiting Angers, France, 49036 Contact: Sophie Abadie-Lacourtoisie, MD 33-2-4135-2734 Centre Hospitalier Regional de Besancon - Hopital Jean Minjoz Recruiting Besancon, France, 25030 Contact: Ulrich Stein, MD 33-3-8166-8240 Hopital Saint Andre Recruiting Bordeaux, France, 33075 Contact: Alain Ravaud, MD, PhD 33-5-5679-5808 alain.ravaud@chu-bordeaux.fr C.H.U. de Brest Recruiting Brest, France, 29609 Contact: Jean-Pierre Malhaire, MD 33-2-9822-3395 Centre Regional Francois Baclesse Recruiting Caen, France, 14076 Contact: Emmanuel Sevin, MD 33-2-3145-5017 e.sevin@baclesse.fr CHU de Grenoble - Hopital de la Tronche Recruiting Grenoble, France, 38043 Contact: Matthieu Laramas, MD 33-4-7676-5333 Centre Hospitalier Departemental Recruiting La Roche Sur Yon, France, 85025 Contact: Franck Priou, MD 33-2-5144-6173 frank.priou@chd-vendee.fr Centre Oscar Lambret Recruiting Lille, France, 59020 Contact: Armelle Caty, MD 33-3-2029-5942 acaty@o-lambret.fr Centre Leon Berard Recruiting Lyon, France, 69373 Contact: Sylvie Negrier, MD 33-4-7878-2751 negrier@lyon.fnclcc.fr Marseille Institute of Cancer - Institut J. Paoli and I. Calmettes Recruiting Marseille, France, 13273 Contact: Gwenaelle Gravis, MD 33-4-9122-3740 gravisg@marseille.fnclcc.fr CHU de la Timone Recruiting Marseille, France, 13385 Contact: Marjorie Baciuchka-Palmaro 33-91-385-708 Hopital Notre-Dame de Bon Secours Recruiting Metz, France, 57038 Contact: Sabine Walter, MD 33-387-553-554 s.walter@chr-metz-thionville.rss.fr Centre Regional de Lutte Contre le Cancer - Centre Val d'Aurelle Recruiting Montpellier, France, 34298 Contact: Stephane Culine, MD 33-4-6761-8555 stculine@valdorel.fnclcc.fr CRLCC Nantes - Atlantique Recruiting Nantes-Saint Herblain, France, 44805 Contact: Frederic Rolland, MD 33-2-40-67-99-76 F-rolland@nantes.fnclcc.fr Centre Antoine Lacassagne Recruiting Nice, France, 06189 Contact: Jean Marc Ferrero, MD 33-4-9203-1511 jean-marc.ferrerero@nice.fnclcc.fr Hopital Europeen Georges Pompidou Recruiting Paris, France, 75015 Contact: Stephane Oudard, MD, PhD 33-1-5609-3434 stephane.oudard@hop.egp.ap-hop-paris.fr Hopital Saint Michel Recruiting Paris, France, 75015 Contact: Gael Deplanque, MD, MSC, PhD 33-1-4412-3078 Institut Jean Godinot Recruiting Reims, France, 51056 Contact: Herve Cure, MD, PhD 33-3-2650-4382 Hopital Foch Recruiting Suresnes, France, 92151 Contact: Christine Theodore, MD 33-1-4625-2149 Institut Claudius Regaud Recruiting Toulouse, France, 31052 Contact: Christine Chevreau-Dalbianco, MD 33-5-6142-4118 chevreau.christine@claudiusregaud.fr Centre Hospitalier Universitaire Bretonneau de Tours Recruiting Tours, France, 37044 Contact: Claude Linassier, MD, PhD 33-2-4747-3827 linassier@med.univ-tours.fr Institut Gustave Roussy Recruiting Villejuif, France, F-94805 Contact: Bernard Escudier, MD 33-1-4211-5410 escudier@igr.fr Collapse << |
| NCT00676650 | - | - | Terminated(Study A6181120 was ... more >>prematurely discontinued due to futility on 27 September 2010. No new or unexpected safety issues were identified.) Collapse << | - | - |
| NCT01176799 | Breast Cancer | Phase 2 | Unknown | December 2017 | Singapore ... more >> National University Hospital Recruiting Singapore, Singapore, 119074 Contact: Soo Chin Lee, MBBS, MRCP 65 6772 4629 Soo_Chin_Lee@nuhs.edu.sg Principal Investigator: Soo Chin Lee, MBBS, MRCP National University Hospital Recruiting Singapore, Singapore, 119228 Collapse << |
| NCT00953459 | Lung Cancer | Phase 2 | Terminated(poorly recruiting) | - | Netherlands ... more >> Vrije Universiteit Medisch Centrum Amsterdam, Netherlands, 1007 MB Collapse << |
| NCT00555620 | Stomach Neoplasms | Phase 1 | Completed | - | Korea, Republic of ... more >> Pfizer Investigational Site Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-707 Pfizer Investigational Site Goyang, Gyeonggi, Korea, Republic of, 410-769 Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00393939 | Breast Neoplasms | Phase 3 | Completed | - | - |
| NCT00748163 | Lung Cancer | Phase 2 | Withdrawn(Study has been aband... more >>oned for lack of available funding.) Collapse << | August 2008 | - |
| NCT00393939 | - | - | Completed | - | - |
| NCT00599924 | Colorectal Neoplasms ... more >> Neoplasms Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> Pfizer Investigational Site Aurora, Colorado, United States, 80045 United States, Tennessee Pfizer Investigational Site Nashville, Tennessee, United States, 37232 United States, Texas Pfizer Investigational Site Dallas, Texas, United States, 75246 Collapse << |
| NCT00792025 | Bladder Cancer ... more >> Transitional Cell Cancer of the Renal Pelvis and Ureter Collapse << | Phase 2 | Completed | - | France ... more >> Centre Rene Huguenin Saint Cloud, France, 92211 Hopital Foch Suresnes, France, 92151 Collapse << |
| NCT01258348 | Metastatic Renal Cell Cancer | Phase 1 | Completed | - | United States, Indiana ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Indianapolis, Indiana, United States, 46219 United States, South Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Greenville, South Carolina, United States, 29605 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. The Woodlands, Texas, United States, 77380 United States, Virginia For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Norfolk, Virginia, United States, 23502 Canada, Quebec For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Montreal, Quebec, Canada, H3T 1E2 Collapse << |
| NCT00555620 | - | - | Completed | - | - |
| NCT00375674 | - | - | Completed | - | - |
| NCT01258348 | - | - | Completed | - | - |
| NCT01984242 | Renal Cell Carcinoma | Phase 2 | Active, not recruiting | August 31, 2019 | - |
| NCT00589784 | CNS Cancer Me... more >>ningioma Intracranial Hemangiopericytoma Hemangioblastoma Neurofibromatosis Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New Jersey Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan-Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 United States, Pennsylvania University of Pittsburgh Medical Center Pittsburg, Pennsylvania, United States United States, Virginia University of Virginia Health Science Center Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00411151 | Gastric Adenocarcinoma ... more >> Barrett Esophagus Collapse << | Phase 2 | Completed | - | Germany ... more >> Universität Heidelberg, Nationales Zentrum für Tumorerkrankungen Heidelberg, Baden-Würtemberg, Germany, 69120 Universitätsklinikum Tübingen Tübingen, Baden-Würtemberg, Germany, 72076 TU München, Klinikum rechts der Isar, II. Med. Klinik und Poliklinik München, Bayern, Germany, 81675 Klinikum der Universität Würzburg Würzburg, Bayern, Germany, 97070 Universitätsklinikum der GHS Essen, Innere Klinik und Poliklinik, Tumorforschung Essen, Nordrhein-Westfalen, Germany, 45122 Universitätsklinik zu Köln, Klinik I für Innere Medizin Köln, Nordrhein-Westfalen, Germany, 50937 Klinikum der Johannes Gutenberg-Universität Mainz Mainz, Rheinland-Pfalz, Germany, 55131 Universitätsklinikum des Saarlandes Homburg/Saar, Saarland, Germany, 66421 Otto-von-Guericke-Universität Magdeburg Magdeburg, Sachsen-Anhalt, Germany, 39120 Universitätsklinikum Carl Gustav Carus, Med. Klinik I Dresden, Sachsen, Germany, 01307 Charité, Campus Benjamin Franklin Berlin, Germany, 12200 KH Nordwest, Abteilung für Hämatologie und Onkologie Frankfurt, Germany, 60488 Collapse << |
| NCT01984242 | - | - | Active, not recruiting | - | - |
| NCT00428597 | Carcinoma, Islet Cell ... more >> Carcinoma, Pancreas Collapse << | Phase 3 | Terminated(Refer to Detailed D... more >>escription.) Collapse << | - | - |
| NCT00599924 | - | - | Completed | - | - |
| NCT01472081 | Renal Cell Carcinoma ... more >> Clear-cell Metastatic Renal Cell Carcinoma Collapse << | Phase 1 | Active, not recruiting | June 2018 | United States, California ... more >> City Of Hope Duarte, California, United States, 91010 United States, Maryland Sidney Kimmel Comprehensive Cancer Center At Johns Hopkins Baltimore, Maryland, United States, 21231 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, New Hampshire Dartmouth-Hitchcock Medical Center Lebanon, New Hampshire, United States, 03756 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Levine Cancer Institute Charlotte, North Carolina, United States, 28204 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, Tennessee Tennessee Oncology, Pllc Nashville, Tennessee, United States, 37203 United States, Texas The University Of Texas Md Anderson Cancer Center Houston, Texas, United States, 77030 Canada, Alberta Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Canada, British Columbia Bc Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Princess Margaret Hospital-Uhn Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00411151 | - | - | Completed | - | - |
| NCT00589784 | - | - | Completed | - | - |
| NCT00428597 | - | - | Terminated(Refer to Detailed D... more >>escription.) Collapse << | - | - |
| NCT01446224 | - | - | Completed | - | - |
| NCT01039519 | Gastrointestinal Stromal Tumor | Phase 2 | Completed | - | United States, California ... more >> UCLA Medical Center Los Angeles, California, United States, 90095 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Oregon Oregon Health and Science University-Knight Cancer Institute Portland, Oregon, United States, 97239 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT01468688 | 3rd Line GIST | Phase 1 | Completed | - | United States, Massachusetts ... more >> Dana Farber Cancer Institute SC (2) Boston, Massachusetts, United States, 02215 United States, Washington Seattle Cancer Care Alliance Onc Seattle, Washington, United States, 98105 Belgium Novartis Investigative Site Leuven, Belgium, 3000 Canada, British Columbia Novartis Investigative Site Vancouver, British Columbia, Canada, V5Z 4E6 France Novartis Investigative Site Lyon Cedex, France, 69373 Novartis Investigative Site Villejuif Cedex, France, 94805 Japan Novartis Investigative Site Kashiwa, Chiba, Japan, 277-8577 Netherlands Novartis Investigative Site Leiden, Netherlands, 2300 RC Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 United Kingdom Novartis Investigative Site London, United Kingdom, SW3 6JJ Novartis Investigative Site Manchester, United Kingdom, M20 9BX Collapse << |
| NCT01735968 | 3rd Line GIST | Phase 1 | Completed | - | United States, Oregon ... more >> Oregon Health and Science University Dept. of OHSU (3) Portland, Oregon, United States, 97239 Belgium Novartis Investigative Site Leuven, Belgium, 3000 France Novartis Investigative Site Bordeaux, France, 33076 Germany Novartis Investigative Site Berlin, Germany, 13125 Novartis Investigative Site Essen, Germany, 45122 Italy Novartis Investigative Site Bologna, BO, Italy, 40138 Novartis Investigative Site Candiolo, TO, Italy, 10060 Netherlands Novartis Investigative Site Groningen, Netherlands, 9713 GZ Novartis Investigative Site Leiden, Netherlands, 2300 RC Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 United Kingdom Novartis Investigative Site Leeds, United Kingdom, LS9 7TF Collapse << |
| NCT00618319 | GIST | Phase 2 | Completed | - | United States, California ... more >> Reseach Facility Encinitas, California, United States Research Site Santa Monica, California, United States, 90404 United States, Texas Reseach Facility San Antonio, Texas, United States Collapse << |
| NCT00511511 | - | - | Completed | - | Netherlands ... more >> Erasmus MC Rotterdam, Netherlands, 3015 CE Collapse << |
| NCT01689376 | - | - | - | - | - |
| NCT01646593 | - | - | - | - | - |
| NCT02268435 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 1 | Withdrawn(Dovitinib Production... more >> stopped) Collapse << | November 2016 | - |
| NCT02013089 | Gastrointestinal Cancers | Not Applicable | Unknown | December 2017 | China, Guangdong ... more >> Gastrointestinal Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510655 Contact: Yanghong Deng, PhD 008613925106525 13925106525@163.com Principal Investigator: Yanhong Deng, PhD Collapse << |
| NCT02560012 | Carcinoma, Renal Cell | Phase 2 | Terminated(Loss of laboratory ... more >>performing molecular analysis) Collapse << | - | United States, Texas ... more >> UTHealth Memorial Hermann Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01039519 | - | - | Completed | - | - |
| NCT01860118 | - | - | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Sparks Center Birmingham, Alabama, United States, 35294 Collapse << |
| NCT02560012 | - | - | Terminated(Loss of laboratory ... more >>performing molecular analysis) Collapse << | - | - |
| NCT00410124 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT01219023 | Hand Foot Syndrome ... more >> Palmar Plantar Erythrodysesthesia Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT02779283 | Acute Lymphoblastic Leukemia ... more >> Acute Myeloid Leukemia Collapse << | Phase 1 | Recruiting | June 1, 2019 | United States, Oregon ... more >> OHSU Knight Cancer Institute Recruiting Portland, Oregon, United States, 97239 Contact: Stephen E. Spurgeon 503-494-8950 spurgeos@ohsu.edu Principal Investigator: Stephen E. Spurgeon Collapse << |
| NCT01582672 | Advanced Renal Cell Carcinoma ... more >> Renal Cell Carcinoma Metastatic Renal Cell Carcinoma Collapse << | Phase 3 | Terminated(Lack of efficacy) | - | - |
| NCT01441388 | Carcinoma, Renal Cell ... more >> Glioblastoma Carcinoma, Hepatocellular Collapse << | Phase 1 | Withdrawn(Business/Operational... more >> issues) Collapse << | November 2013 | - |
| NCT01731158 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Unknown | May 2016 | Germany ... more >> Charité Berlin Berlin, Germany University Hospital Bonn Bonn, Germany University Hospital Dresden Dresden, Germany University Hospital Düsseldorf Düsseldorf, Germany Waldkrankenhaus St. Marien Erlangen, Germany University Hospital Essen Essen, Germany University Hospital Frankfurt Frankfurt, Germany University Hospital Freiburg Freiburg, Germany University Hospital Göttingen Göttingen, Germany Oncological Practice Hannover Hannover, Germany University Hospital Hannover Hannover, Germany University Hospital Magdeburg Magdeburg, Germany University Hospital Tübingen Tübingen, Germany Collapse << |
| NCT01306045 | Carcinoma, Non-Small-Cell Lung... more >> Carcinoma, Small Cell Lung Carcinoma, Thymic Collapse << | Phase 2 | Recruiting | December 30, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office (888) NCI-1937 Collapse << |
| NCT00891475 | Carcinoma, Renal Cell | Phase 1 Phase 2 | Completed | - | Russian Federation ... more >> Ilya Tsimafeyeu Moscow, Russian Federation, 109147 Collapse << |
| NCT02187302 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | - |
| NCT01271712 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Active, not recruiting | May 31, 2019 | - |
| NCT00410124 | - | - | Completed | - | - |
| NCT02068170 | - | - | Completed | - | Belgium ... more >> UZ Leuven Leuven, Belgium, 3000 Collapse << |
| NCT01271712 | - | - | Active, not recruiting | - | - |
| NCT02239952 | Cancer High-g... more >>rade Glioma Collapse << | Not Applicable | Recruiting | January 2019 | Netherlands ... more >> VU University Medical Center Recruiting Amsterdam, Noord-Holland, Netherlands, 1081 HV Contact: H.M.W Verheul, MD, PhD +31 (0)20 4444321 h.verheul@vumc.nl Collapse << |
| NCT02216578 | Metastatic Gastrointestinal St... more >>romal Tumor Collapse << | Phase 2 | Recruiting | August 2019 | Belgium ... more >> U.Z. Leuven - Campus Gasthuisberg (147) Recruiting Leuven, Belgium Contact: Patrick Schöffski Czechia University Hospital Motol (1905) Recruiting Prague, Czechia Contact: Katerina Kopeckova-Kubackova France Institut Bergonie (228) Recruiting Bordeaux, France Contact: Antoine Italiano Centre Leon Berard (227) Recruiting Lyon, France Contact: Jean-Yves Blay Gustave Roussy (225) Recruiting Villejuif, France Contact: Olivier Mir Germany UniversitaetsMedizin Mannheim (527) Recruiting Mannheim, Germany Contact: Bernd Kasper Hungary Military Hospital - State Health Centre (3769) Recruiting Budapest, Hungary Contact: Zsuzanna papai United Kingdom Clatterbridge Centre for Oncology NHS Trust - Clatterbridge Cancer Centre NHS Foundation Trust (659) Recruiting Bebington, United Kingdom Contact: Ali Nasim Royal Marsden Hospital - Chelsea, London (613) Recruiting Chelsea, United Kingdom Contact: Charlotte Benson University College London Hospitals NHS Foundation Trust - University College Hospital (622) Recruiting London, United Kingdom Contact: Palma Dileo The Christie NHS Foundation Trust (610) Not yet recruiting Manchester, United Kingdom Contact: Michael Leahy Collapse << |
| NCT00782834 | - | - | Terminated(Stopped early for f... more >>utility, unable to meet accrual goals) Collapse << | - | - |
| NCT02889328 | Gastrointestinal Stromal Tumor... more >>s (GISTs) Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center, University of Ulsan College of Medicine Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT00914043 | - | - | Withdrawn(Withdrawn as NCI res... more >>cinded approval for parent study NSABP-B-45) Collapse << | - | - |
| NCT01190241 | Advanced Solid Tumors ... more >> Inoperable Metastasis Collapse << | Not Applicable | Unknown | - | Netherlands ... more >> Medical Center Alkmaar Alkmaar, Netherlands, 1815 JD VU University Medical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT01323400 | GIST | Phase 2 | Completed | - | France ... more >> Institut Bergonié Bordeaux, France Centre Georges François Leclerc Dijon, France Centre Oscar Lambret Lille, France Centre Léon Bérard Lyon, France, F-69008 Hôpital de la Timone Marseille, France Institut Paoli Calmette Marseille, France Centre Alexis Vautrin Nancy, France Institut de Cancérologie de l'Ouest Nantes, France Hôpital St Antoine Paris, France Hôpital Tenon Paris, France CHU de Reims Reims, France Institut Cancerologie Neuwirth St Priest en Jarez, France Institut Gustave Roussy Villejuif, France Collapse << |
| NCT01502228 | Renal Cell Carcinoma | Not Applicable | Terminated(No further funding) | - | United States, Indiana ... more >> Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00782834 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Terminated(Stopped early for f... more >>utility, unable to meet accrual goals) Collapse << | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT01502228 | - | - | Terminated(No further funding) | - | - |
| NCT00731211 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, California ... more >> San Francisco Oncology Associates San Francisco, California, United States, 94115 United States, Florida Florida Cancer Specialists Fort Myers, Florida, United States, 33901 Watson Clinic Center for Cancer Care and Research Lakeland, Florida, United States, 33805 Florida Hospital Cancer Institute Orlando, Florida, United States, 32804 Gulfcoast Oncology Associates St. Petersburg, Florida, United States, 33705 United States, Georgia Medical Oncology Associates of Augusta Augusta, Georgia, United States, 30901 Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Kentucky Baptist Hospital East Louisville, Kentucky, United States, 40207 United States, Maine Central Maine Medical Center Lewiston, Maine, United States, 04240 United States, Michigan Grand Rapids Clinical Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63017 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, South Carolina South Carolina Oncology Associates, PA Columbia, South Carolina, United States, 29210 United States, Tennessee Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 United States, Virginia Virginia Cancer Institute Richmond, Virginia, United States, 23235 Collapse << |
| NCT02848768 | - | - | Completed | - | France ... more >> CHU de Rennes Rennes, France, 35033 Collapse << |
| NCT01068769 | Gastrointestinal Stromal Tumor | Phase 2 | Active, not recruiting | December 2018 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Oregon Oregon Health Sciences University Portland, Oregon, United States, 97239 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT00731211 | - | - | Completed | - | - |
| NCT02264665 | - | - | Recruiting | June 5, 2019 | - |
| NCT01440959 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center, University of Ulsan College of Medicine Seoul,, Korea, Republic of, 138-736 Collapse << |
| NCT01440959 | - | - | Completed | - | - |
| NCT01449825 | - | - | Completed | - | - |
| NCT03062410 | Renal Carcinoma Metastatic ... more >> Kidney Neoplasms Renal Cancer Collapse << | Not Applicable | Recruiting | February 4, 2020 | France ... more >> CHU de Besançon Active, not recruiting Besançon, France, 25030 Hôpital privé Sainte Marie Recruiting Chalon-sur-Saône, France, 71100 Contact: Erika Viel, MD 0033385488989 Centre de lutte contre le cancer Georges François Leclerc Recruiting Dijon, France, 21000 Contact: Sylvain Ladoire, MD 0033380737506 sladoire@cgfl.fr Hôpital Nord Franche-Comté Recruiting Montbéliard, France, 25200 Contact: Guillaume Mouillet, MD mouillet.guillaume@gmail.com Centre de lutte contre le cancer Jean Godinot Recruiting Reims, France, 51100 Contact: Jean-Christophe Eymard, MD Groupe hospitaliser St-Vincent Recruiting Strasbourg, France, 67000 Contact: Louis-Marie Dourthe, MD lm.dourthe@solcrr.org Hôpitaux Universitaires de Strasbourg Recruiting Strasbourg, France, 67000 Contact: Philippe Barthelemy, MD philippe.barthelemy@chru-strasbourg.fr Centre de lutte contre le cancer Alexis Vautrin - Institut de Cancérologie de Lorraine Recruiting Vandœuvre-lès-Nancy, France, 54519 Contact: Lionnel Geoffrois, MD l.geoffrois@nancy.unicancer.fr Collapse << |
| NCT01482949 | Renal Cell Carcinoma | Phase 2 | Terminated(Lack of efficacy) | - | United States, Minnesota ... more >> University of Minnesota Cancer Center Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT01566747 | Renal Cell Carcinoma | Phase 2 | Active, not recruiting | September 2018 | Ireland ... more >> Beaumont Hospital Dublin, Leinster, Ireland Mater Misericordiae University Hospital Dublin, Leinster, Ireland Mater Private Hospital Dublin, Leinster, Ireland Cork University Hospital Cork, Ireland The Adelaide & Meath Hospital Dublin incorporating the National Children's Hospital Dublin, Ireland Galway University Hospital Galway, Ireland Mid-Western Regional Hospital Limerick, Ireland Waterford Regional Hospital Waterford, Ireland Sweden Karolinska University Hospital Stockholm, Sweden Collapse << |
| NCT01068769 | - | - | Active, not recruiting | - | - |
| NCT02607332 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center, University of Ulsan College of Medicine Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT03609424 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 1 Phase 2 | Not yet recruiting | August 30, 2020 | - |
| NCT02380625 | Ebola Virus Disease | Phase 1 Phase 2 | Unknown | March 2016 | - |
| NCT01620216 | Acute Myeloid Leukemia Arising... more >> From Previous Myelodysplastic Syndrome Chronic Myelomonocytic Leukemia Myelodysplastic Syndrome Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Collapse << | Phase 2 | Recruiting | March 30, 2019 | United States, Maryland ... more >> National Heart Lung and Blood Institute Not yet recruiting Bethesda, Maryland, United States, 20824 Contact: Christopher S. Hourigan 301-803-8029 hourigan@nih.gov Principal Investigator: Christopher S. Hourigan United States, Oregon OHSU Knight Cancer Institute Not yet recruiting Portland, Oregon, United States, 97239 Contact: Marc Loriaux 503-494-9383 loriauxm@ohsu.edu Principal Investigator: Marc Loriaux United States, Texas UT Southwestern/Simmons Cancer Center-Dallas Recruiting Dallas, Texas, United States, 75390 Contact: Robert H. Collins 214-648-4155 robert.collins@utsouthwestern.edu Principal Investigator: Robert H. Collins United States, Utah Huntsman Cancer Institute/University of Utah Not yet recruiting Salt Lake City, Utah, United States, 84112 Contact: Tibor J. Kovacsovics 801-585-3229 tibor.kovacsovics@hci.utah.edu Principal Investigator: Tibor J. Kovacsovics Collapse << |
| NCT00976612 | - | - | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01058044 | Cancer | Not Applicable | Completed | - | France ... more >> Centre Jean Perrin Clermont-Ferrand, France, 63011 Collapse << |
| NCT00495872 | Solid Tumors | Phase 1 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01151852 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Songpa-gu, Korea, Republic of, 138-736 Collapse << |
| NCT02960906 | Clear Cell Metastatic Renal Ce... more >>ll Carcinoma Collapse << | Phase 2 | Recruiting | May 2020 | France
... more >> Hôpital Saint André, CHU de Bordeaux
Recruiting
Bordeaux, France, 33075
Contact: Marine GROSS-GOUPIL, MD marine.gross-goupil@chu-bordeaux.fr
Centre Francois Baclesse
Recruiting
Caen, France, 14000
Contact: Florence JOLY, MD,PHD f.joly@baclesse.unicancer.fr
CHU Henri-Mondor
Recruiting
Creteil, France, 94000
Contact: Christophe TOURNIGAND, MD, PHD christophe.tournigand@aphp.fr
CHD Vendée
Not yet recruiting
La Roche-sur-Yon, France, 85925
Contact: Franck PRIOU, MD frank.priou@chd-vendee.fr
Centre OSCAR LAMBRET LILLE
Recruiting
Lille, France, 59000
Contact: Yaovi Messan AMELA, MD e-amela@o-lambret.fr
Hôpital Européen Marseille
Not yet recruiting
Marseille, France, 13003
Contact: Philippe DALIVOUST philippe.dalivoust@gmail.com
Institut Paoli Calmettes (IPC)
Recruiting
Marseille, France, 13009
Contact: Gwenaëlle GRAVIS, MD gravisg@ipc.unicancer.fr
Centre Antoine Lacassagne
Recruiting
Nice, France, 06100
Contact: Delphine BORCHIELLINI, MD Delphine.BORCHIELLINI@nice.unicancer.fr
Institut de Cancérologie du Gard - CHU Caremeau
Recruiting
Nîmes, France, 30029
Contact: Nadine HOUEDE, MD,PHD HOUEDE nadine |
| NCT01389583 | Gastrointestinal Stromal Tumor | Phase 2 | Recruiting | October 2019 | Taiwan ... more >> National Health Research of Institutes, Taiwan Cooperative Oncology Group Recruiting Tainan, Taiwan Principal Investigator: Kun-Huei Yeh, PhD Principal Investigator: Chueh-Chuan Yen, PhD Principal Investigator: Ken-Hong Lim, MD Principal Investigator: Jen-Shi Chen Principal Investigator: Cheng-Chung Wu, MS Principal Investigator: Chang-Fang Chiu, PhD Principal Investigator: Kuan-Der Lee, PhD Principal Investigator: Kun-Ming Rau, MPH Sub-Investigator: Ann-Lii Cheng, PhD Sub-Investigator: Yu-Lin Lin, MD Sub-Investigator: Ta-Chung Chao, MD Sub-Investigator: Wen-Liang Fang, MD Sub-Investigator: Ruey-Kuen Hsieh, MD Sub-Investigator: Chun-Nan Yeh, MD Sub-Investigator: Youngsen Yang, MD Sub-Investigator: Tseng-Hsi Lin, PhD Sub-Investigator: Mei-Due Yang, PhD Sub-Investigator: Li-Yuan Bai, MD Sub-Investigator: Wu-Chou Su, MD Sub-Investigator: Yan-Shen Shan, PhD Sub-Investigator: Nai-Jung Chiang, MD Sub-Investigator: Yen-Yang Chen, MD Collapse << |
| NCT01977586 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Not Applicable | Withdrawn(Difficulty in recrui... more >>ting suitable patients at centre) Collapse << | - | United Kingdom ... more >> Imperial College Healthcare NHS Trust London, United Kingdom Collapse << |
| NCT00749320 | Renal Cell Carcinoma | Not Applicable | Active, not recruiting | February 2019 | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT03379012 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | July 30, 2019 | Russian Federation ... more >> A.I. Kryzhanovsky Krasnoyarsk Cancer Center Recruiting Krasnoyarsk, Russian Federation Contact: Ruslan Zukov kidneycancer@yandex.ru Kidney Cancer Research Bureau Recruiting Moscow, Russian Federation Contact: Ilya Tsimafeyeu kidneycancer@yandex.ru RUDN University Recruiting Moscow, Russian Federation Contact: Yuliya Tishova kidneycancer@yandex.ru City Clinical Oncology Center Recruiting Saint Petersburg, Russian Federation Contact: Pavel Borisov kidneycancer@yandex.ru Collapse << |
| NCT03571438 | Kidney Cancer | Not Applicable | Recruiting | September 30, 2024 | France ... more >> Grenoble Alps Hospital Recruiting Grenoble, France, 38043 Contact: Jean-Luc Descotes, PU-PH Collapse << |
| NCT03170180 | Stomach Neoplasms | Phase 2 | Recruiting | October 31, 2020 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Sung Ju Park 234106820 Principal Investigator: Jeeyun Lee, M.D., Ph.D. Collapse << |
| NCT02086695 | - | - | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Canada, Manitoba St. Boniface General Hospital Winnipeg, Manitoba, Canada, R2H 2A6 Collapse << |
| NCT01151852 | - | - | Completed | - | - |
| NCT01582009 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Cancer Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 1 Phase 2 | Terminated(pts off study, PI l... more >>eft institute) Collapse << | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 University of Rochester Medical Center Rochester, New York, United States, 14642 Collapse << |
| NCT00876382 | - | - | Completed | - | United States, Arkansas ... more >> Northeast Arkansas Clinic Jonesboro, Arkansas, United States, 72401 United States, Florida Gulfcoast Oncology Associates St. Petersburg, Florida, United States, 33705 United States, Georgia Medical Oncology Associates of Augusta Augusta, Georgia, United States, 30901 United States, Kentucky Consultants in Blood Disorders and Cancer Louisville, Kentucky, United States, 40207 United States, Mississippi Jackson Oncology Associates Jackson, Mississippi, United States, 39202 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63017 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT01582204 | Renal Cancer | Not Applicable | Active, not recruiting | April 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00619268 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | France ... more >> Centre Paul Papin Angers, France Centre Hospitalier Universitaire de Besançon Besançon, France Centre Hospitalier Universitaire de Bordeaux - Hôpital St André Bordeaux, France Institut Bergonié Bordeaux, France Centre François Baclesse Caen, France Centre Jean Perrin Clermont Ferrand, France Centre Georges François Leclerc Dijon, France Centre Hospitalier de Versailles Le Chesnay, France Centre Hospitalier Universitaire de Lille - Hôpital Claude Huriez Lille, France Centre Oscar Lambret Lille, France Centre Hospitalier Universitaire DUPUTRYEN Limoges, France Centre Léon Bérard Lyon, France, 69373 Centre Hospitalier Universiariare Lyon, Hôpital Lyon Sud Lyon, France Institut Paoli Calmette Marseille, France Centre Val d'Aurelle Montpellier, France Clinique Valdegour-Centre médical Oncogard Nîmes, France Fondation Hôpital Saint Joseph Paris, France Hopital du Val de Grâce Paris, France Hôpital Européen Georges Pompidou Paris, France Centre Hospilier Universitaire de Poitiers Poitiers, France Institut Jean Godinot Reims, France Centre Eugène Marquis Rennes, France Centre René Gauducheau Saint Herblain, France Institut de Cancérologie de la Loire Saint Priest en Jarez, France Centre Hospitalier Starsbourg Strasbourg, France Hôpital FOCH Suresnes, France Institut Claudius Regaud Toulouse, France Centre Alexis Vautrin Vandoeuvre les Nancy, France Institut Gustave Roussy Villejuif, France, 94805 Collapse << |
| NCT00691730 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Not Applicable | Active, not recruiting | December 2018 | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Canada, Ontario London Regional Cancer Program London, Ontario, Canada, N6A 4L6 Mount Sinai Hospital Toronto, Ontario, Canada, M5G 1X5 University Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT02208128 | Renal Cell Carcinoma | Not Applicable | Unknown | February 2018 | Germany ... more >> Urological Department of the University Hospital Heidelberg Recruiting Heidelberg, Germany, 69120 Contact: Sascha Pahernik, PD Dr. +49 6221 56 39689 sascha.pahernik@med.uni-heidelberg.de Principal Investigator: Sascha Pahernik, PD Dr. Collapse << |
| NCT02959554 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Active, not recruiting | December 2021 | Austria ... more >> Medizinische Universität Wien - Universitätsklinik für Innere Medizin I, Klinische Abteilung für Onkologie Wien, Austria, 1090 Germany Universitätsklinikum Essen (AöR) - Klinik und Poliklinik für Urologie / Kinderurologie und Uroonkologie Essen, Germany, 45147 Collapse << |
| NCT00866320 | - | - | Completed | - | - |
| NCT01381614 | - | - | Completed | - | - |
| NCT01582009 | - | - | Terminated(pts off study, PI l... more >>eft institute) Collapse << | - | - |
| NCT00474786 | Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT00953446 | Renal Cell Carcinoma | Not Applicable | Suspended | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00474786 | - | - | Completed | - | - |
| NCT00866320 | Kidney Cancer | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 United States, Texas Baylor Sammons Cancer Center Dallas, Texas, United States, 75246 Collapse << |
| NCT00979381 | - | - | Completed | - | Netherlands ... more >> University Medical Center Nijmegen st Radboud Nijmegen, Netherlands, 6525 GH Collapse << |
| NCT00678288 | - | - | Terminated | - | - |
| NCT01603004 | - | - | Recruiting | May 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Nitya Raj, MD 646-888-4849 Contact: Kinh Gian Do, MD, PhD 212-639-8591 Principal Investigator: Nitya Raj, MD Collapse << |
| NCT00543504 | Advanced Cancer | Phase 1 | Active, not recruiting | October 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00538772 | - | - | Withdrawn | - | Canada, British Columbia ... more >> BC Cancer Agency - Centre for Southern Interior Kelowna, British Columbia, Canada, V1Y 5L3 BC Cancer Agency - Vancouver Vancouver, British Columbia, Canada, V5Z 4E6 BC Cancer Agency - Vancouver Island Victoria, British Columbia, Canada, V8R 6V5 Collapse << |
| NCT00678288 | Carcinoma, Renal Cell | Phase 2 | Terminated | - | Austria ... more >> Salzburg, Austria, 5020 Wien, Austria, 1090 France Avignon, France, 84000 Bayonne, France, 64100 Bordeaux, France, 33000 Marseille, France, 13015 Marseille, France, 13385 Nantes, France, 44020 Paris, France, 75014 Paris, France, 75651 Reims, France, 51100 Strasbourg, France, 67000 Toulouse, France, 31052 Vandoeuvre Les Nancy, France, 54000 Ireland Cork, Ireland Dublin, Ireland, 24 Italy Aviano, Pordenone, Italy, 33081 Legnago, Verona, Italy, 37045 Napoli, Italy, 80131 Pavia, Italy, 27100 Perugia, Italy, 06156 Reggio Emilia, Italy, 42100 Poland Gdansk, Poland, 80-219 Lublin, Poland, 20-090 Warszawa, Poland, 04-141 Wroclaw, Poland, 50 - 556 Spain Barcelona, Spain, 08035 Madrid, Spain, 28041 Madrid, Spain, 28046 Oviedo, Spain, 33006 Pamplona, Spain, 31008 Valencia, Spain, 46009 United Kingdom Northwood, Middlesex, United Kingdom, HA6 2RN Newcastle Upon Tyne, Tyne and Wear, United Kingdom, NE4 6BE London, United Kingdom, SW3 6JJ Collapse << |
| NCT01254864 | Prostate Cancer | Phase 2 | Active, not recruiting | March 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00457691 | Metastatic Colorectal Cancer | Phase 3 | Completed | - | - |
| NCT02688491 | Renal Neoplasms ... more >> Targeted Molecular Therapy Collapse << | Not Applicable | Not yet recruiting | July 2026 | - |
| NCT00831857 | - | - | Completed | - | Netherlands ... more >> University Medical Center Groningen Groningen, Netherlands, 9700 RB Collapse << |
| NCT01524848 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Denmark ... more >> Aarhus University Hospital, dept. of Oncology Aarhus, Denmark, DK-8000 Aarhus C Herlev Hospital, dept. of Oncology Herlev, Denmark, 2730 Finland Helsinki University Hospital, dept. of oncology Helsingfors, Finland, FI-00029 Kuopio University Hospital Cancer Center Kuopio, Finland, FI-70029 Germany Klinik für Interdisziplinäre Onkologie, Sarkomzentrum Berlin-Brandenburg Berlin, Germany, 13125 Universitätsklinikum Essen, Innere klinik und Poliklinik Essen, Germany, DE-45122 Studienzentrale chirurgische klinik, Universitäts medizin Mannheim Mannheim, Germany, DE-68167 Norway Dept of Oncology, Haukeland University Hospital Bergen, Norway, N-5021 Norwegian Radium Hospital Oslo, Norway, N-0310 Dept of Oncology, St Olav Hospital Trondheim, Norway, N-7006 Sweden Dept of Oncology, Sahlgrenska University Hospital Gothenburg, Sweden, SE-413 45 Dept of Oncology, Linköping University Hospital Linköping, Sweden, SE-581 85 Dept of Oncology, Skane University Hospital Lund, Sweden, SE-221 85 Radiumhemmet, Karolinska University Hospital Stockholm, Sweden, SE-171 76 Dept of Oncology, Norrland University Hospital Umeå, Sweden, SE-901 85 Dept of Oncology, Academic Hospital Uppsala, Sweden, SE-751 85 Collapse << |
| NCT03275558 | Recurrent Glioblastoma ... more >> Gliosarcoma Anaplastic Gliomas Collapse << | Phase 1 | Withdrawn(Study halted prior t... more >>o enrollment of first participant. (It has been determined on 2018 May 15 that the NCT03275558 study will be stopped by sponsor decision)) Collapse << | - | Hong Kong ... more >> The Hong Kong Cancer Institute Hong Kong, Hong Kong, 518031 Singapore National University Cancer Institute, Singapore, Singapore, 119074 Collapse << |
| NCT00869011 | Renal Cell Carcinoma | Phase 3 | Unknown | - | Germany ... more >> Section Sports Medicine, Charité Universitätsmedizin Berlin, Hindenburgdamm 30 Recruiting Berlin, Germany, 12200 Contact: Fernando C. Dimeo, MD +493084452098 fernando.dimeo@charite.de Principal Investigator: Fernando C. Dimeo, MD Collapse << |
| NCT00734851 | - | - | Terminated(Dose Limiting Toxic... more >>ities) Collapse << | - | - |
| NCT00444795 | - | - | Completed | - | Korea, Republic of ... more >> Keimyung University Dongsan Medical Center Jung-gu, Daegu, Korea, Republic of, 100-712 Daegu Catholic University Medical Center Nam-gu, Daegu, Korea, Republic of, 705-718 GangNeung Asan Hospital Gangneung-si, Gangwon-do, Korea, Republic of, 210-711 Soonchunhyang University Bucheon Hospital Bucheon, Gyeonggi-do, Korea, Republic of, 420-767 The Catholic University of Korea, St. Vincent's Hospital Suwon, Gyeonggi-do, Korea, Republic of, 442-723 Hwasun Hospital, Chonnam National University Cheonnam, South Jeolla Province, Korea, Republic of, 519-809 Hallym University Sacred Heart Hospital Anyang, Korea, Republic of, 431-070 Pusan National University Hospital Busan, Korea, Republic of, 602 739 Kosin University Gospel Hospital Busan, Korea, Republic of, 602-702 Dong-A University Hospital Busan, Korea, Republic of, 602-715 Dong-A University Medical Center (Dong-A University Hospital) Busan, Korea, Republic of, 602-715 Dong-A University Medical Center, Department of Medicine, Division of Hemato-Oncology Busan, Korea, Republic of, 602-715 Pusan National University Hospital Busan, Korea, Republic of, 602-739 Hwasun Hospital, Chonnam National University Cheonnam, Korea, Republic of, 519-809 Keimyung University Dongsan Hospital Daegu, Korea, Republic of, 700 712 Kyungpook National University Medical Center Daegu, Korea, Republic of, 702-210 Daegu Catholic University Medical Center (DCUMC) Daegu, Korea, Republic of, 705-718 Chungnam National University Hospital Daejeon, Korea, Republic of, 301-721 Eulji University Hospital Daejeon, Korea, Republic of, 302-799 Inje University Ilsan Paik Hospital Goyang, Korea, Republic of, 411-706 Chosun University Hospital Gwang Joo, Korea, Republic of, 501-717 Pusan National University Hospital Gyeongsangnam-do, Korea, Republic of, 626-770 Wonkwang University School of Medicine and Hospital (WUH) Iksan -Si, Korea, Republic of Inha University Hospital Incheon, Korea, Republic of, 400-711 Gachon University Gil Hospital Incheon, Korea, Republic of, 405-760 Chonbuk National University Hospital Jeonju, Korea, Republic of, 561-712 Yeungnam University Medical Center Nam-gu, Korea, Republic of, 705-717 Seoul National University Bundang Hospital Seongnam-si, Korea, Republic of, 463-707 Seoul National University Hospital (SNUH) Seoul, Korea, Republic of, 110-744 Seoul National University Hospital / Department of Internal Medicine Seoul, Korea, Republic of, 110-744 Kangbuk Samsung Hospital Seoul, Korea, Republic of, 110-746 Severance Hospital, Yonsei University Health System Seoul, Korea, Republic of, 120-752 Kyung Hee University Medical Center Seoul, Korea, Republic of, 130-872 KyungHee University Medical Center Seoul, Korea, Republic of, 130-872 Samsung Medical Center, Dept. of Medicine, Div. of Hematology/Oncology Seoul, Korea, Republic of, 135-710 Samsung Medical Center/Division of Hematology-Oncology, Department of Medicine Seoul, Korea, Republic of, 135-710 Gangnam Severance Hospital, Yonsei University Health System Seoul, Korea, Republic of, 135-720 Korea University Anam Hospital Seoul, Korea, Republic of, 136-705 Asan Medical Center/Department of Oncology Seoul, Korea, Republic of, 138-736 Asan Medical Center Seoul, Korea, Republic of, 138-736 Korea Cancer Center Hospital Seoul, Korea, Republic of, 139-706 Korea University Guro Hospital Seoul, Korea, Republic of, 152-703 Ewha Womans University Mokdong Hospital Seoul, Korea, Republic of, 158-710 Seoul National University Hospital Seoul, Korea, Republic of, 3080 Seoul ST. Mary's Hospital Seoul, Korea, Republic of Ulsan University Hospital Ulsan, Korea, Republic of, 682-714 Yonsei University Wonju College of Medicine, Wonju Christian Hospital Wonju-si, Korea, Republic of, 220-701 Collapse << |
| NCT00688766 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Terminated(Based on Independen... more >>t Data Monitoring Committee (IDMC) recommendation.) Collapse << | - | - |
| NCT00457691 | - | - | Completed | - | - |
| NCT02324803 | Self Efficacy ... more >> Adverse Drug Event Carcinoma, Renal Cell Collapse << | Phase 2 | Unknown | December 2015 | China, Guangdong ... more >> The First Affiliated Hospital of Guangzhou Medical University Recruiting Guangzhou, Guangdong, China, 510120 Contact: Mian Xie Collapse << |
| NCT01381601 | - | - | Completed | - | - |
| NCT00734851 | Prostate Cancer | Phase 2 | Terminated(Dose Limiting Toxic... more >>ities) Collapse << | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21218 United States, New Jersey The Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08901 United States, North Carolina Duke University Durham, North Carolina, United States, 27705 Collapse << |
| NCT00444795 | - | - | Completed | - | - |
| NCT01289028 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Saarow, Germany, 155226 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Duesseldorf, Germany, 40479 Novartis Investigative Site Essen, Germany, 45122 Novartis Investigative Site Frankfurt, Germany, 60488 Novartis Investigative Site Freiburg, Germany, 79106 Novartis Investigative Site Halle/'Saale, Germany, 06120 Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site Mannheim, Germany, 68167 Novartis Investigative Site Muenchen, Germany, 81377 Novartis Investigative Site Muenchen, Germany, 81675 Novartis Investigative Site Ulm, Germany, 89081 Italy Novartis Investigative Site Bologna, BO, Italy, 40138 Novartis Investigative Site Genova, GE, Italy, 16132 Novartis Investigative Site Taormina, ME, Italy, 98039 Novartis Investigative Site Milano, MI, Italy, 20133 Novartis Investigative Site Padova, PD, Italy, 35100 Novartis Investigative Site Aviano, PN, Italy, 33081 Novartis Investigative Site Torino, TO, Italy, 10153 Collapse << |
| NCT00705874 | Cancer | Phase 1 | Completed | - | United States, Colorado ... more >> Rocky Mountain Cancer Centre Denver, Colorado, United States United States, Florida Cancer Centres of Florida Ocoee, Florida, United States, 34761 United States, Indiana Central Indiana Cancer Centres Indianapolis, Indiana, United States, 46219 United States, Nevada Comprehensive Cancer Centres of Nevada Las Vegas, Nevada, United States, 89169 United States, New York New York Oncology Hematology PC Albany, New York, United States, 12206 United States, Ohio Dayton Oncology and Hematology, PA Kettering, Ohio, United States, 45409 United States, South Carolina Cancer Centres of the Carolinas Greenville, South Carolina, United States, 29605 United States, Texas Texas Oncology, PA Dallas, Texas, United States, 75246 Tyler Cancer Centre Tyler, Texas, United States, 75702 United States, Virginia Virginia Oncology Associates Norfolk, Virginia, United States, 23502 United States, Washington Northwest Cancer Specialists - Vancouver Cancer Centre Vancouver, Washington, United States, 98684 North Star Lodge Cancer Centre Yakima, Washington, United States, 98902 Collapse << |
| NCT00705874 | - | - | Completed | - | - |
| NCT00700258 | - | - | Recruiting | December 1, 2023 | - |
| NCT00717587 | Kidney Cancer | Phase 2 | Unknown | - | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104-4283 Contact: Clinical Trials Office - Abramson Cancer Center of the Univers 800-474-9892 Collapse << |
| NCT01514448 | Metastatic Renal Cell Carcinom... more >>a (mRCC) Collapse << | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Chemnitz, Germany, 09130 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Hannover, Germany, 30559 Novartis Investigative Site Hof, Germany, 95028 Novartis Investigative Site Kassel, Germany, 34125 Novartis Investigative Site Langen, Germany, 63225 Novartis Investigative Site Leipzig, Germany, 04357 Novartis Investigative Site Magdeburg, Germany, 39120 Novartis Investigative Site Muenster, Germany, 48149 Novartis Investigative Site Ravensburg, Germany, 88214 Novartis Investigative Site Velbert, Germany, 42551 Novartis Investigative Site Wiesbaden, Germany, 65191 Novartis Investigative Site Wolfsburg, Germany, 38440 Collapse << |
| NCT00486538 | Advanced Renal Cell Carcinoma | Phase 2 | Completed | - | United States, California ... more >> Site Reference ID/Investigator# 7193 Sacramento, California, United States, 95817 United States, District of Columbia Site Reference ID/Investigator# 5243 Washington, District of Columbia, United States, 20010 United States, Illinois Site Reference ID/Investigator# 5384 Chicago, Illinois, United States, 60637 United States, Massachusetts Site Reference ID/Investigator# 11662 Boston, Massachusetts, United States, 02114 Site Reference ID/Investigator# 11663 Boston, Massachusetts, United States, 02114 Site Reference ID/Investigator# 5379 Boston, Massachusetts, United States, 02114 United States, New Hampshire Site Reference ID/Investigator# 5380 Lebanon, New Hampshire, United States, 03756-0001 United States, Pennsylvania Site Reference ID/Investigator# 5249 Philadelphia, Pennsylvania, United States, 19111 Site Reference ID/Investigator# 6278 Philadelphia, Pennsylvania, United States, 19111 Site Reference ID/Investigator# 6269 Pittsburgh, Pennsylvania, United States, 15232 United States, South Carolina Site Reference ID/Investigator# 7300 Charleston, South Carolina, United States, 29425 United States, Texas Site Reference ID/Investigator# 6796 Houston, Texas, United States, 77030-4009 Canada Site Reference ID/Investigator# 6566 Vancouver, Canada, V5Z 4E6 Collapse << |
| NCT01514448 | - | - | Completed | - | - |
| NCT02535351 | Clear-cell Metastatic Renal Ce... more >>ll Carcinoma Collapse << | Phase 3 | Terminated(low enrolment) | - | Italy ... more >> Fondazione IRCCS Istituto Nazionale Tumori Milan, Mi, Italy, 20133 Collapse << |
| NCT00265798 | - | - | Active, not recruiting | - | - |
| NCT00265798 | Gastrointestinal Stromal Tumor | Phase 2 | Active, not recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 United States, Illinois University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637 Decatur Memorial Hospital Decatur, Illinois, United States, 62526 Central Illinois Hematology Oncology Center Springfield, Illinois, United States, 62702 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00861419 | Advanced Solid Tumors | Phase 1 | Completed | - | - |
| NCT01289028 | - | - | Completed | - | - |
| NCT01227213 | - | - | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Centre Nijmegen, Netherlands, 6500HB Collapse << |
| NCT03260894 | Renal Cell Carcinoma (RCC) | Phase 3 | Active, not recruiting | June 2020 | - |
| NCT01091207 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135710 Collapse << |
| NCT00509704 | - | - | Completed | - | Netherlands ... more >> UMC St Radboud Nijmegen, Netherlands Collapse << |
| NCT00482755 | Breast Cancer | Phase 2 | Completed | - | Canada, British Columbia ... more >> British Columbia Cancer Agency - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Edmond Odette Cancer Centre at Sunnybrook Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT01370109 | - | - | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center, University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01099423 | Kidney Cancer | Phase 3 | Active, not recruiting | May 2017 | Belgium ... more >> Onze Lieve Vrouw Ziekenhuis Aalst, Belgium Cliniques Universitaires St. Luc Brussels, Belgium Hôpitaux Universitaires Bordet-Erasme - Institut Jules Bordet Brussels, Belgium Universitair Ziekenhuis Gent Gent, Belgium Virga Jesse Hospital Hasselt, Belgium AZ Groeninghe - Campus Loofstraat Kortrijk, Belgium AZ Damiaan - Campus Sint-Jozef Oostende, Belgium Canada, Nova Scotia Queen Elizabeth II Health Sciences Centre Halifax, Nova Scotia, Canada Canada, Quebec CHUM - Pavillon Saint-Luc Montreal,, Quebec, Canada Canada Montreal General Hospital Montreal, Canada The Ottawa Hospital, The Integrated Cancer Program- General Campus Ottawa, Canada University Health Network - Oci / Princess Margaret Hospital Toronto, Canada Diamond Health Care Centre Vancouver, Canada Italy San Camillo Forlanini Hospitals Roma, Italy Netherlands Jeroen Bosch Ziekenhuis 's-Hertogenbosch, Netherlands Academisch Medisch Centrum - Universiteit van Amsterdam Amsterdam, Netherlands The Netherlands Cancer Institute-Antoni Van Leeuwenhoekziekenhuis Amsterdam, Netherlands Vrije Universiteit Medisch Centrum Amsterdam, Netherlands University Medical Center Groningen Groningen, Netherlands Academisch Ziekenhuis Maastricht Maastricht, Netherlands Radboud University Nijmegen Medical Centre Nijmegen, Netherlands Universitair Medisch Centrum - Academisch Ziekenhuis Utrecht, Netherlands United Kingdom Royal United Hospital Bath, United Kingdom University Hospitals Bristol NHS Foundation Trust - Bristol Haematology And Oncology Centre Bristol, United Kingdom St. James'S University Hospital Leeds, United Kingdom Barts and The London NHS Trust - St. Bartholomew'S Hospital London, United Kingdom Imperial College Healthcare NHS Trust - Charing Cross Hospital London, United Kingdom Christie NHS Foundation Trust Manchester, United Kingdom Singelton Hospital Swansea, United Kingdom Collapse << |
| NCT01876212 | Metastatic Melanoma | Phase 2 | Active, not recruiting | June 6, 2019 | United States, Pennsylvania ... more >> Hillman Cancer Center Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT02645149 | Melanoma | Phase 4 | Not yet recruiting | August 2021 | Australia, New South Wales ... more >> Royal Prince Alfred Hospital Not yet recruiting Camperdown, New South Wales, Australia, 2050 Contact: Maria Gonzalez 612 9911 7200 maria.gonzalez@melanoma.org.au Principal Investigator: John Thompson Westmead Hospital Westmead, New South Wales, Australia, 2145 Melanoma Institute Australia Not yet recruiting Wollstonecraft, New South Wales, Australia, 2065 Contact: Maria Gonzalez +612 9911 7200 maria.gonzalez@melanoma.org.au Contact: Alex Menzies +612 9911 7200 info@melanoma.org.au Sub-Investigator: Georgina Long Principal Investigator: Alex Menzies Collapse << |
| NCT00610012 | - | - | Active, not recruiting | August 2020 | Germany ... more >> iOMEDICO Freiburg, Germany Collapse << |
| NCT02551718 | Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << |
| NCT01246817 | Renal Cell Cancer | Phase 2 | Unknown | - | Netherlands ... more >> University Medical Centre Nijmegen Recruiting Nijmegen, Gelderland, Netherlands, 6500 HB Principal Investigator: C.M.L. van Herpen, Md PhD Collapse << |
| NCT01758575 | - | - | Terminated | - | Netherlands ... more >> VU University Medical Center Amsterdam, Netherlands, 1081HV Collapse << |
| NCT00877045 | - | - | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33901 United States, Georgia Georgia Cancer Specialists Atlanta, Georgia, United States, 30341 United States, Tennessee Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 United States, Virginia Virginia Cancer Institute Richmond, Virginia, United States, 23235 Collapse << |
| NCT03419572 | - | - | Recruiting | January 5, 2021 | - |
| NCT02776644 | - | - | Completed | - | - |
| NCT00601120 | Renal Cell Carcinoma | Not Applicable | Withdrawn(This protocol was ne... more >>ver officially opened and no patients were recruited.) Collapse << | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01984931 | Nausea Vomiti... more >>ng Collapse << | Phase 3 | Unknown | December 2013 | Korea, Republic of ... more >> Chungmu General Hopsital Recruiting Seoul, Yeongdeungpo-gu, Korea, Republic of, 150-034 Contact: Sang Hoon Lhee, MD, Phd +82 (2) 2068/4525 cmirb@naver.com Principal Investigator: Paolo Alan B Tabar, MD Collapse << |
| NCT02208583 | Hormone Refractory Prostate Ca... more >>ncer Collapse << | Not Applicable | Unknown | December 2016 | China, Tianjin ... more >> Department of Interventional Oncology, Tianjin Medical University Cancer Institute and Hospital Recruiting Tianjin, Tianjin, China, 300060 Contact: Wang HaiTao, Ph.D +86-18630955984 peterrock2000@126.com Principal Investigator: Wang HaiTao, Ph.D Collapse << |
| NCT03387514 | Renal Cell Carcinoma | Phase 2 | Recruiting | January 15, 2022 | United States, Wisconsin ... more >> University of Wisconsin Carbone Cancer Center Recruiting Madison, Wisconsin, United States, 53705 Contact: Cancer Connect 800-622-8922 cancerconnect@uwcarbone.wisc.edu Collapse << |
| NCT01636908 | Cancer Advanc... more >>ed Solid Tumors Collapse << | Not Applicable | Active, not recruiting | - | Netherlands ... more >> VUMedical Center Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT02142036 | Metastatic Cancer | Phase 2 | Active, not recruiting | January 2022 | Norway ... more >> Akershus University Hospital Lillestrøm, Norway, 1478 The Norwegian Radium Hospital Oslo, Norway, 0379 Collapse << |
| NCT03413176 | - | - | Completed | - | France ... more >> AP-HP, Pitié-Salpêtrière Hospital, Department of Pharmacology, CIC-1421, Pharmacovigilance Unit, INSERM. Paris, France, 75013 Collapse << |
| NCT00698659 | - | - | Terminated | - | Singapore ... more >> National University Hospital Singapore, Singapore Collapse << |
| NCT03297606 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Advanced Solid Tumors Collapse << | Phase 2 | Recruiting | September 2021 | Canada, British Columbia ... more >> BCCA - Vancouver Cancer Centre Recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Daniel John Renouf 604 877-6000 ext 672357 Canada, Ontario London Regional Cancer Program Recruiting London, Ontario, Canada, N6A 5W9 Contact: Stephen Welch 519 685-8640 Ottawa Hospital Research Institute Recruiting Ottawa, Ontario, Canada, K1H 8L6 Contact: John Hilton 613 737-7700 ext 70179 University Health Network Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Lillian Siu 416 946-2911 Canada, Quebec The Jewish General Hospital Recruiting Montreal, Quebec, Canada, H3T 1E2 Contact: Cristiano Ferrario 514 398-8307 Collapse << |
| NCT01200134 | Anaplastic Glioma | Phase 2 | Completed | - | France ... more >> Caen University Hospital Caen, France, 14033 Collapse << |
| NCT02747173 | - | - | Terminated | - | Spain ... more >> Hospital Universitario central de Asturias Oviedo, Asturias, Spain, 33011 Hospital de Donostia San Sebastián, Guipuzcoa, Spain, 20080 Complejo Hospitalario Univ de Santiago de Compostela Santiago de Compostela, La Coruña, Spain, 15706 Hospital de Navarra Pamplona, Navarra, Spain, 31008 Complejo Hospitalario Univ de Vigo Vigo, Pontevedra, Spain, 36204 Consorcio Hospitalario Parc Tauli Barcelona, Spain, 08208 Complejo Hospitalario de León Leon, Spain, 24071 Hospital Univ Lucus Augusti Lugo, Spain, 27003 Complejo Hospitalario de Ourense Ourense, Spain, 32005 Instituto Valenciano de Oncologia Valencia, Spain, 46009 Hospital Universitario La Fe Valencia, Spain, 46026 Hospital Miguel Servet Zaragoza, Spain, 50009 Collapse << |
| NCT01858441 | Pharmacogenetic Study | Not Applicable | Active, not recruiting | December 2020 | France ... more >> Centre Antoine Lacassagne Nice, France, 06000 Collapse << |
| NCT02228954 | - | - | Active, not recruiting | December 2019 | Netherlands ... more >> VU University Medical Center Amsterdam, Netherlands University Medical Center Groningen Groningen, Netherlands Radboudumc Nijmegen, Netherlands, 6525GA Collapse << |
| NCT02086734 | - | - | Completed | - | Germany ... more >> Urologische Klinik und Poliklinik der Universität München München, Bavaria, Germany, 81377 Collapse << |
| NCT00498966 | Kidney Cancer | Phase 2 | Completed | - | United States, Kentucky ... more >> Investigative Site Louisville, Kentucky, United States, 40202 United States, New Jersey Investigative Site Morristown, New Jersey, United States, 07962 United States, New York Investigative Site Armonk, New York, United States, 10504 Collapse << |
| NCT01810744 | Metastatic Colorectal Cancer ... more >> Glioblastoma Bevacizumab Capillaroscopy Collapse << | Not Applicable | Active, not recruiting | June 2018 | France ... more >> Centre Georges François Leclerc Dijon, France, 21079 Collapse << |
| NCT01498458 | Metastatic Breast Cancer | Phase 1 | Completed | - | Germany ... more >> GBG Forschungs GmbH Neu-Isenburg, Germany, 63263 Collapse << |
| NCT01498458 | - | - | Completed | - | - |
| NCT03091465 | - | - | Completed | - | - |
| NCT02847377 | Carcinoma, Non-Small-Cell Lung | Early Phase 1 | Recruiting | March 27, 2019 | France ... more >> CGFL Recruiting Dijon, France, 21079 Collapse << |
| NCT01538238 | Locally Advanced or Metastatic... more >> Non-clear Cell Type Renal Cell Carcinoma Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung medical Center Seoul, Korea, Republic of Collapse << |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT02044354 | Metastatic Castration-resistan... more >>t Prostate Cancer Collapse << | Phase 3 | Active, not recruiting | April 2018 | France ... more >> Gustave Roussy Villejuif, Val de Marne, France, 94805 Collapse << |
| NCT01372813 | - | - | Terminated(Terminated for insu... more >>fficient accrual.) Collapse << | - | - |
| NCT02056587 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 4 | Completed | - | Russian Federation ... more >> Tatarstan Regional Cancer Center Kazan, Tatarstan, Russian Federation, 420029 Altai Regional Cancer Center Barnaul, Russian Federation N.N. Blokhin Russian Cancer Research Center Moscow, Russian Federation, 115478 Collapse << |
| NCT03024437 | Metastatic Cancer ... more >> Renal Cancer Collapse << | Phase 1 Phase 2 | Recruiting | January 31, 2020 | United States, Indiana ... more >> Indiana University Health Hospital Recruiting Indianapolis, Indiana, United States, 46202 Contact: Sheila Dropcho, RN 317-278-4191 marlmoor@iu.edu Principal Investigator: Roberto Pili, MD Indiana University Melvin and Bren Simon Cancer Center Recruiting Indianapolis, Indiana, United States, 46202 Contact: Sheila Dropcho, RN 317-278-4191 sdropcho@iupui.edu Principal Investigator: Roberto Pili, MD United States, Pennsylvania University of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Julia Wolinkski 215-360-0928 julia.wolinksi@uphs.upenn.edu Principal Investigator: Vivek Narayan, MD Collapse << |
| NCT01466504 | - | - | Terminated(Accrual did not mee... more >>t expectations. Over nearly 2 years just 4 subjects were treated on study. The goal of 30 subjects was not attainable.) Collapse << | - | United States, New Hampshire ... more >> Dartmouth-Hitchcock Medical Center, Norris Cotton Cancer Center Lebanon, New Hampshire, United States, 03756 United States, Vermont White River Junction VA Medical Center White River Junction, Vermont, United States, 05009 Collapse << |
| NCT03229512 | Hand Foot Skin Reaction | Early Phase 1 | Recruiting | December 2018 | United States, Illinois ... more >> Department of Dermatology, Northwestern University Feinberg School of Medicine Recruiting Chicago, Illinois, United States, 60611 Contact: Jennifer N Choi, MD 312-695-8106 jennifer.choi@northwestern.edu Contact: Linda Serrano, MD 312-503-5903 linda.serrano@northwestern.edu Principal Investigator: Jennifer N Choi, MD Sub-Investigator: Linda Serrano, MD Northwestern Memorial Hospital Recruiting Chicago, Illinois, United States, 60611 Contact: Jennifer N Choi, MD 312-695-8106 jennifer.choi@northwestern.edu Contact: Linda Serrano, MD 312-503-5903 linda.serrano@northwestern.edu Principal Investigator: Jennifer N Choi, MD Sub-Investigator: Linda Serrano, MD Collapse << |
| NCT02877368 | - | - | Completed | - | France ... more >> Chu de Reims Reims, France, 51092 Collapse << |
| NCT01372813 | Advanced Clear Cell Renal Carc... more >>inoma Collapse << | Phase 2 | Terminated(Terminated for insu... more >>fficient accrual.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02712112 | Gastrointestinal Stromal Tumor... more >>s (GISTs) Collapse << | Phase 2 | Recruiting | December 2018 | Korea, Republic of ... more >> Asan Medical Center Recruiting Seoul, Korea, Republic of, 138-736 Contact: Min-Hee Ryu, MD., Ph.D. +82-10-5285-9492 miniryu@amc.seoul.kr Principal Investigator: Min-Hee Ryu, M.D., Ph.D. Collapse << |
| NCT00715182 | Advanced/ Metastatic Renal Cel... more >>l Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> Novartis Investigative Site San Francisco, California, United States United States, North Carolina Novartis Investigative Site Durham, North Carolina, United States, 27710 United States, Washington Novartis Investigative Site Seattle, Washington, United States, 98109-1023 France Novartis Investigative Site Bordeaux Cedex, France, 33075 Novartis Investigative Site Villejuif Cedex, France, 94805 Germany Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site München, Germany, 81675 Netherlands Novartis Investigative Site Rotterdam, Netherlands, 3075 EA Spain Novartis Investigative Site Madrid, Spain, 28041 Taiwan Novartis Investigative Site Taipei, Taiwan, ROC, Taiwan, 112 Novartis Investigative Site Taichung, Taiwan, 40705 Novartis Investigative Site Taipei, Taiwan, 10002 Collapse << |
| NCT01339962 | - | - | Completed | - | Denmark ... more >> University of Copenhagen Herlev Hospital Herlev, Denmark, 2730 Collapse << |
| NCT02599779 | Stage IV Renal Cell Cancer AJC... more >>C V7 Collapse << | Phase 2 | Recruiting | March 2021 | Canada, Alberta ... more >> Tom Baker Cancer Centre Recruiting Calgary, Alberta, Canada, T2N 4N2 Contact: Richard Lee-Ying, MD richard.lee-ying@albertahealthservices.ca Principal Investigator: Richard Lee-Ying, MD Canada, Ontario Sunnybrook Health Sciences Centre Recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Georg A. Bjarnason, MD 416-480-5847 georg.bjarnason@sunnybrook.ca Contact: Nesan Bandali, RN 416-480-5000 ext 82139 nesan.bandali@sunnybrook.ca Principal Investigator: Georg A. Bjarnason, MD Princess Margaret Hospital Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Aaron Hansen, MD 416-946-4501 aaron.hansen@uhn.ca Principal Investigator: Aaron Hansen, MD Collapse << |
| NCT00782275 | - | - | Completed | - | - |
| NCT02558387 | Recurrent or Metastatic Saliva... more >>ry Gland Cancer of the Head and Neck Collapse << | Phase 2 | Unknown | January 2017 | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01798446 | Renal Cell Carcinoma ... more >> Nonclear Cell Temsirolimus Resistance Collapse << | Phase 2 | Unknown | December 2016 | Korea, Republic of ... more >> Seou National University Hospital Recruiting Seoul, Korea, Republic of, 110744 Contact: Se-Hoon Lee, MD, PhD 82220722199 shlee119@snu.ac.kr Principal Investigator: Se-Hoon Lee, MD, PhD Sub-Investigator: Dae Seog Heo, MD, PhD Sub-Investigator: Dong-Wan Kim, MD, PhD Sub-Investigator: Tae Min Kim, MD, PhD Sub-Investigator: Bhumsuk Keam, MD Collapse << |
| NCT02693535 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Advanced Solid Tumors Collapse << | Phase 2 | Recruiting | - | - |
| NCT00782275 | Renal Cell Carcinoma ... more >> Kidney Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Tennessee Vanderbilt Univeristy Medical Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02113826 | Metastatic Alveolar Soft Part ... more >>Sarcoma Collapse << | Phase 2 | Terminated(Low accrual rate) | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02816515 | - | - | Completed | - | Hungary ... more >> Department of Oncological Internal Medicine and Clinical Pharmacology "C", National Institute of Oncology Hungary Budapest, Hungary, 1026 National Koranyi Institute of TB and Pulmonology Budapest, Hungary, 1121 National Institute of Oncology, Department of Radiotherapy 7-9 Ráth György u. Budapest, Hungary Collapse << |
| NCT01342627 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(low enrollment rate... more >>) Collapse << | - | Italy ... more >> Centro di Riferimento Oncologico Aviano, Pordenone, Italy, 33081 Collapse << |
| NCT01491672 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center NY, New York, United States, 90033 Argentina Novartis Investigative Site Rio Negro, Viedma, Argentina, 8500 Novartis Investigative Site Tucuman, Argentina, T4000 Brazil Novartis Investigative Site Florianopolis, SC, Brazil, 88034-000 Novartis Investigative Site Barretos, SP, Brazil, 14784-400 Novartis Investigative Site São Paulo, SP, Brazil, 01246-000 Novartis Investigative Site São Paulo, SP, Brazil, 01509-900 Bulgaria Novartis Investigative Site Sofia, Bulgaria, 1784 China, Beijing Novartis Investigative Site Beijing, Beijing, China, 100730 China, Zhejiang Novartis Investigative Site Hangzhou, Zhejiang, China, 310003 China Novartis Investigative Site Beijing, China, 100021 Novartis Investigative Site Guangzhou, China, 510060 Novartis Investigative Site Shanghai, China, 200032 Russian Federation Novartis Investigative Site Leningrad Region, Russia, Russian Federation, 188663 Novartis Investigative Site Moscow, Russia, Russian Federation, 125284 Novartis Investigative Site Nizhny Novgorod, Russia, Russian Federation, 603001 Novartis Investigative Site Obninsk, Russia, Russian Federation, 249036 Collapse << |
| NCT02465060 | Advanced Malignant Solid Neopl... more >>asm Bladder Carcinoma Breast Carcinoma Cervical Carcinoma Colon Carcinoma Colorectal Carcinoma Endometrial Carcinoma Esophageal Carcinoma Gastric Carcinoma Glioma Head and Neck Carcinoma Kidney Carcinoma Liver and Intrahepatic Bile Duct Carcinoma Lung Carcinoma Lymphoma Malignant Uterine Neoplasm Melanoma Ovarian Carcinoma Pancreatic Carcinoma Plasma Cell Myeloma Prostate Carcinoma Rectal Carcinoma Recurrent Bladder Carcinoma Recurrent Breast Carcinoma Recurrent Cervical Carcinoma Recurrent Colon Carcinoma Recurrent Colorectal Carcinoma Recurrent Esophageal Carcinoma Recurrent Gastric Carcinoma Recurrent Glioma Recurrent Head and Neck Carcinoma Recurrent Liver Carcinoma Recurrent Lung Carcinoma Recurrent Lymphoma Recurrent Malignant Solid Neoplasm Recurrent Melanoma Recurrent Ovarian Carcinoma Recurrent Pancreatic Carcinoma Recurrent Plasma Cell Myeloma Recurrent Prostate Carcinoma Recurrent Rectal Carcinoma Recurrent Skin Carcinoma Recurrent Thyroid Gland Carcinoma Recurrent Uterine Corpus Carcinoma Refractory Lymphoma Refractory Malignant Solid Neoplasm Refractory Plasma Cell Myeloma Skin Carcinoma Thyroid Gland Carcinoma Uterine Corpus Cancer Collapse << | Phase 2 | Recruiting | - | - |
| NCT01491672 | - | - | Completed | - | - |
| NCT03556384 | Gastrointestinal Stromal Tumor... more >>s Sdh GIST Cancer Collapse << | Phase 2 | Recruiting | September 2024 | United States, California ... more >> UC San Diego Moores Cancer Center Recruiting La Jolla, California, United States, 92093 Contact: Elaine Eng e1eng@ucsd.edu Principal Investigator: Adam M Burgoyne, MD, PhD Sub-Investigator: Jason K Sicklick, MD Collapse << |
| NCT02751827 | - | - | Recruiting | February 2019 | France ... more >> Institut Bergonié Recruiting Bordeaux, France, 33076 Contact: Antoine ITALIANO, MD,PhD Principal Investigator: Antoine ITALIANO, MD,PhD Centre Léon Bérard Not yet recruiting Lyon Cedex 08, France, 69373 Contact: Jean-Yves BLAY, MD, PhD Principal Investigator: Jean-Yves BLAY, MD, PhD Collapse << |
| NCT02789137 | - | - | Recruiting | October 30, 2018 | Spain ... more >> Hospital Arquitecto Marcide de Ferrol Recruiting Ferrol, A Coruña, Spain, 15405 Hospital Asil de Granollers Recruiting Granollers, Barcelona, Spain, 08402 Hospital Parc Taulí Recruiting Sabadell, Barcelona, Spain, 08208 Hospital Galdakano Recruiting Usansolo, Bizkaia, Spain, 48960 Complejo Hospitalario la Mancha Centro Recruiting Alcazar de San Juan, Ciudad REAL, Spain, 13600 Hospital Universitari Son Espases Recruiting Palma, Mallorca, Spain, 07120 H. Universitario Infanta Cristina Recruiting Badajoz, Spain, 06006 Hospital Vall de Hebrón Recruiting Barcelona, Spain, 08035 Hospital Universitario Ciudad Real Recruiting Ciudad Real, Spain, 13005 H. Reina Sofía de Cordoba Recruiting Córdoba, Spain, 14004 Hospital de Cabueñes Not yet recruiting Gijón, Spain, 33394 ICO Girona (Hospital Dr.Josep Trueta) Recruiting Girona, Spain, 17007 Hospital Universitario de Guadalajara Not yet recruiting Guadalajara, Spain, 19002 Complejo Hospitalario de Jaén Recruiting Jaén, Spain, 23007 Complejo Hospitalario de León Recruiting León, Spain, 24071 Complejo Hospitalario Ourense. Hospital Santa María Nai Recruiting Ourense, Spain, 32005 Hospital General de Asturias Not yet recruiting Oviedo, Spain, 33011 Complejo Hospitalario de Navarra Recruiting Pamplona, Spain, 31008 Hospital Universitario Canarias Not yet recruiting Santa Cruz de Tenerife, Spain, 38320 Hospital de Sant Pau i Santa Tecla Recruiting Tarragona, Spain, 43003 Hospital Virgen de la Salud Recruiting Toledo, Spain, 45071 Hospital Clínico Universitario de Valencia Recruiting Valencia, Spain, 46011 H Clínico Zaragoza Recruiting Zaragoza, Spain, 50009 Collapse << |
| NCT01390519 | - | - | Completed | - | Norway ... more >> Novartis Investigative Site Kristiansand, Norway, 4615 Novartis Investigative Site Tromsø, Norway, 9038 Novartis Investigative Site Trondheim, Norway, 7006 Collapse << |
| NCT03353753 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 3 | Active, not recruiting | December 2020 | - |
| NCT01370980 | - | - | Unknown | September 2014 | Switzerland ... more >> Policlinique Médicale Universitaire and Centre pluridisciplinaire d'oncologie, CHUV Recruiting Lausanne, Vaud, Switzerland, 1010 Contact: Bugnon Olivier +41 21 314 48 48 Olivier.Bugnon@hospvd.ch Contact: Schneider Marie-Paule +41 21 314 48 46 Marie-Paule.Schneider@hospvd.ch Collapse << |
| NCT01377298 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 2 | Unknown | May 2014 | Taiwan ... more >> National Taiwan University Hospital Recruiting Taipei, Taiwan, 100 Contact: Ruey-Long Hong, MD +886 2 23123456 ext 67510 rlhong@ntu.edu.tw Sub-Investigator: Jo-Pai Chen, MD Principal Investigator: Ruey-Long Hong, MD Collapse << |
| NCT00633295 | Gastrointestinal Stromal Tumor... more >>s (GIST) Collapse << | Phase 2 | Completed | - | Israel ... more >> Novartis Investigative Site Jerusalem, Israel Novartis Investigative Site Tel Aviv, Israel Novartis Investigative Site Tel Hashomer, Israel Collapse << |
| NCT02338570 | Renal Cell Carcinoma | Phase 4 | Terminated(Stopped on November... more >> 16th 2016, because of recruitment failure.) Collapse << | - | Italy ... more >> Spedali Civili Brescia, Italy Ospedale A Perrino Brindisi, Italy Istituto Europeo di Oncologia Milano, Italy Istituto Nazionale Tumori - IRCCS Fondazione G. Pascale Napoli, Italy AOU Maggiore della Carità Novara, Italy Casa di Cura La Maddalena Palermo, Italy Unicampus Biomedico Roma, Italy AOU San Giovanni di Dio e Ruggi D'Aragona Salerno, Italy Collapse << |
| NCT02700568 | Carcinoma, Renal Cell ... more >> Clear Cell Renal Cell Carcinoma Collapse << | Phase 2 | Recruiting | August 2018 | Russian Federation ... more >> Demchenkova Marina Viktorovna Recruiting Irkutsk, Russian Federation Semenov Andrey Vladimirovich Recruiting Ivanovo, Russian Federation Mikhailova Nadezhda Vasilievna Recruiting Kazan, Russian Federation Eskerov Kurban Abdulmutalibovich Active, not recruiting Kirov, Russian Federation Zukov Ruslan Aleksandrovich Recruiting Krasnoyarsk, Russian Federation Ovchinnikova Elena Georgievna Recruiting Nizhny Novgorod, Russian Federation Guseva Irina Vasilievna Recruiting Penza, Russian Federation Vladimirova Lyubov Yur'evna Active, not recruiting Rostov-on-Don, Russian Federation Zolotoreva Tatiana Gennadievna Recruiting Samara, Russian Federation Katkov Alexey Aleksandrovich Active, not recruiting Saratov, Russian Federation Ivannikov Andrey Andreyevich Recruiting Tambov, Russian Federation Usynin Evgeny Anatolievich Recruiting Tomsk, Russian Federation Evstegneyeva Irina Vladimirovna Recruiting Tver, Russian Federation Khmelevsky Andrey Anatolievich Recruiting Ufa, Russian Federation Gurina Ludmila Ivanovna Active, not recruiting Vladivostok, Russian Federation Collapse << |
| NCT03185039 | Kidney Cancer | Not Applicable | Recruiting | November 29, 2020 | France ... more >> Institut Paoli Calmettes Recruiting Marseille, France, 13009 Contact: GENRE Dominique, MD 33 (0)4 91 22 37 78 drci.up@ipc.unicancer.fr Contact: Isabelle Boquet, PhD 33 (0)4 91 22 37 78 drci.up@ipc.unicancer.fr Principal Investigator: GRAVIS Gwénaëlle, MD Collapse << |
| NCT00923130 | - | - | Completed | - | - |
| NCT02921724 | Cancer Oral D... more >>rug Administration Collapse << | Not Applicable | Recruiting | May 2019 | Italy ... more >> Azienda Ospedaliera Papa Giovanni XXIII, Bergamo Recruiting Bergamo, Italy, 24127 Contact: Carlo Tondini, MD Principal Investigator: Carlo Tondini, MD Irst-Irccs Recruiting Meldola (FC), Italy, 47014 Contact: Alessandro Passardi, MD +39 0543 739238 alessandro.passardi@irst.emr.it Principal Investigator: Alessandro Passardi, MD Azienda Provinciale per i Servizi Sanitari della Provincia Autonoma di Trento Recruiting Trento, Italy, 38123 Contact: Mimma Rizzo, MD Principal Investigator: Mimma Rizzo, MD Collapse << |
| NCT00448721 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, California ... more >> Investigative Site Duarte, California, United States, 91010 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Pennsylvania Investigative Site Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Investigative Site Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02483247 | Cancer | Phase 1 Phase 2 | Active, not recruiting | - | United States, Indiana ... more >> Parkview Research Center Fort Wayne, Indiana, United States Indiana University Health Goshen Goshen, Indiana, United States, 46526 Indiana University -Ball Indianapolis, Indiana, United States Indiana University-SCC Indianapolis, Indiana, United States United States, Louisiana Louisiana State Univesity New Orleans, Louisiana, United States United States, Virginia US Oncology Research Fairfax, Virginia, United States Canada, Ontario Ottawa Hospital Research Institute Ottawa, Ontario, Canada Princess Margaret Cancer Centre Toronto, Ontario, Canada Collapse << |
| NCT01265810 | Oral Complaints ... more >> Renal Cell Carcinoma Hepatocellular Carcinoma Gastrointestinal Stromal Tumors Collapse << | Phase 3 | Completed | - | Netherlands ... more >> The Netherlands Cancer Institute-Antoni van Leeuwenhoek Hospital Amsterdam, Netherlands, 1066 CX Leiden University Medical Centre (LUMC) Leiden, Netherlands, 2300 RC Collapse << |
| NCT01793636 | Metastatic Clear Cell Renal Ca... more >>rcinoma Collapse << | Phase 2 | Terminated(Inferior efficacy o... more >>f study drug in renal indication.) Collapse << | November 2015 | United Kingdom ... more >> Royal Bournemouth Hospital Bournemouth, United Kingdom, BH7 7DW Royal Sussex County Hospital Brighton, United Kingdom, BN2 5BE University Hospitals Coventry & Warwickshire NHS Trust Coventry, United Kingdom, CV2 2DX Beatson West of Scotland Cancer Center Glasgow, United Kingdom, G12 0YN St. James' Hospital Leeds, United Kingdom, LS9 7TF Barts Health NHS Trust London, United Kingdom, EC1A 6BE Royal Free London Hospital London, United Kingdom, NW3 2QG Southampton General Hospital Southampton, United Kingdom, SO16 6YD Southend University Hospital NHS Foundation Trust Westcliff-On-Sea, United Kingdom, SS0 0RY Collapse << |
| NCT00923130 | Renal Cell Carcinoma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 United States, South Carolina University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT01700699 | - | - | Unknown | October 2013 | Italy ... more >> Department of Medicine and Surgery, Univeristy of Salerno Recruiting Baronissi, Salerno, Italy, 84081 Contact: Mario Vitale, MD +39 089672539 mavitale@unisa.it Principal Investigator: Mario Vitale, MD Collapse << |
| NCT01714765 | Metastatic Clear Cell Renal Ca... more >>ncer Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Barts Health NHS Trust London, United Kingdom, EC1M 7BE Collapse << |
| NCT00767819 | Progressive Sarcoma | Phase 2 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Saarow, Germany, 15526 Novartis Investigative Site Duesseldorf, Germany, 40479 Novartis Investigative Site Essen, Germany, 45147 Novartis Investigative Site Mannheim, Germany, 68167 Novartis Investigative Site Muenchen, Germany, 81377 Italy Novartis Investigative Site Milano, MI, Italy, 20133 Collapse << |
| NCT03598816 | Renal Cell Carcinoma | Phase 2 | Not yet recruiting | November 30, 2021 | United States, Missouri ... more >> Washington University School of Medicine Not yet recruiting Saint Louis, Missouri, United States, 63110 Contact: James J Hsieh, M.D., Ph.D. 314-273-1688 jhsieh@wustl.edu Principal Investigator: James J Hsieh, M.D., Ph.D. Sub-Investigator: Vivek Arora, M.D., Ph.D. Sub-Investigator: William E Gillanders, M.D. Sub-Investigator: Eric Knoche, M.D. Sub-Investigator: Jingqin (Rosy) Luo, Ph.D. Sub-Investigator: Peter Oppelt, M.D. Sub-Investigator: Russell Pachynski, M.D. Sub-Investigator: Joel Picus, M.D. Sub-Investigator: Bruce Roth, M.D. Sub-Investigator: Malachi Griffith, Ph.D. Sub-Investigator: Obi Griffith, Ph.D. Collapse << |
| NCT01649180 | - | - | Terminated(Closed due to prolo... more >>nged enrollment timelines) Collapse << | - | - |
| NCT02533258 | - | - | Completed | - | - |
| NCT02533258 | - | - | Completed | - | - |
| NCT01105364 | Kidney Cancer ... more >> Metastatic Cancer Collapse << | Not Applicable | Unknown | - | France ... more >> Centre Hospitalier Universitaire Bretonneau de Tours Recruiting Tours, France, 37044 Contact: Contact Person 33-2-4747-4776 Collapse << |
| NCT01649180 | Renal Cell Carcinoma | Phase 2 | Terminated(Closed due to prolo... more >>nged enrollment timelines) Collapse << | - | United States, Nebraska ... more >> Missouri Valley Cancer Consortium Omaha, Nebraska, United States, 68106 United States, Ohio Cleveland Clinic, Taussig Cancer Institute Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania, Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT02048371 | Liposarcoma O... more >>steogenic Sarcoma Ewing/Ewing-like Sarcoma Rhabdomyosarcoma Collapse << | Phase 2 | Recruiting | December 2020 | United States, California ... more >> City of Hope National Medical Center Recruiting Duarte, California, United States, 91010 Contact: Torie Fechner 626-256-4673 tfechner@coh.org Principal Investigator: Warren Chow, MD Children's Hospital Los Angeles Recruiting Los Angeles, California, United States, 90027 Contact: Magnolia Manzano 323-361-8156 mmanzano@chla.usc.edu Sarcoma Oncology Research Center Recruiting Santa Monica, California, United States, 90403 Contact: Victoria Chua 310-552-9999 vchua@sarcomaoncology.com Principal Investigator: Sant Chawla, MD Stanford University Recruiting Stanford, California, United States, 94305 Contact: Maria Ahern 650-725-6413 mahern@stanford.edu Principal Investigator: Kristen Ganjoo, MD United States, Florida Mayo Clinic - Florida Recruiting Jacksonville, Florida, United States, 32224 Contact: Steven Attia, DO 904-953-7292 Attia.Steven@mayo.edu Principal Investigator: Steven Attia, DO H. Lee Moffitt Terminated Tampa, Florida, United States, 33612 United States, Illinois Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Rasima Cehic 312-695-2082 rasima.cehic@northwestern.edu Principal Investigator: Mark Agulnik, MD United States, Indiana Indiana University Recruiting Indianapolis, Indiana, United States, 46202 Contact: Michele Niland 317-278-0778 mniland@iupui.edu Principal Investigator: Daniel Rushing, MD United States, Massachusetts Dana Farber/Partners Cancer Care Terminated Boston, Massachusetts, United States, 02215 United States, Minnesota Mayo Clinic - Minnesota Recruiting Rochester, Minnesota, United States, 55905 Contact: Elizabeth Smith 507-293-3817 Smith.Elizabeth6@mayo.edu Principal Investigator: Scott Okuno, MD United States, North Carolina Carolinas Healthcare System Recruiting Charlotte, North Carolina, United States, 28203 Contact: Michelle Treadwell 980-442-2332 Michelle.Treadwell@carolinashealthcare.org Principal Investigator: Michael Livingston, MD Duke University Recruiting Durham, North Carolina, United States, 27705 Contact: Colleen Piechocki 919-681-1883 colleen.piechocki@duke.edu Principal Investigator: Richard Riedel, MD United States, Ohio Ohio State University Recruiting Columbus, Ohio, United States, 43202 Contact: Comprehensive Cancer Center 800-293-5066 Jamesline@osumc.edu Principal Investigator: David Liebner, MD United States, Oregon Oregon Health and Science University Recruiting Portland, Oregon, United States, 97239 Contact: David Boody-Alter 503-418-9655 boodyalt@ohsu.edu Principal Investigator: Lara Davis, MD United States, Tennessee Vanderbilt University Recruiting Nashville, Tennessee, United States, 37232 Contact: Melissa Sindler 615-875-0060 melissa.m.sindler@vanderbilt.edu Principal Investigator: Vicky Keedy, MD United States, Utah Huntsman Cancer Institute Recruiting Salt Lake City, Utah, United States, 84112 Contact: Justin Parker 801-587-4702 justin.parker@hci.utah.edu Principal Investigator: Jennifer Wright, MD United States, Washington Seattle Cancer Care Alliance Recruiting Seattle, Washington, United States, 98109 Contact: Roxanne Moore 206-288-6425 romoore@seattlecca.org Principal Investigator: Elizabeth Loggers, MD, PhD Collapse << |
| NCT02616185 | Prostatic Neoplasms | Phase 1 | Recruiting | March 15, 2021 | United States, Connecticut ... more >> Smilow Cancer Hospital at Yale-New Haven Recruiting New Haven, Connecticut, United States, 06510 United States, Maryland National Institutes of Health Active, not recruiting Bethesda, Maryland, United States, 20892-1196 National Institutes of Health Clinical Center Active, not recruiting Bethesda, Maryland, United States, 20892 United States, Nebraska GU Research Network Recruiting Omaha, Nebraska, United States, 68130 United States, Nevada Comprehensive Cancer Centers of Nevada Recruiting Las Vegas, Nevada, United States, 89169 United States, New York Memorial Sloan-Kettering Cancer Center 53rd Street Recruiting New York, New York, United States, 10022 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Memorial Sloan-Kettering Cancer Center, Sidney Kimmel Center Recruiting New York, New York, United States, 10065 Memorial Sloan-Kettering Cancer Center Recruiting New York, New York, United States, 10065 United States, North Carolina Duke University Medical Center Recruiting Durham, North Carolina, United States, 27710 Investigational Chemotherapy Services Recruiting Durham, North Carolina, United States, 27710 United States, Pennsylvania UPMC Cancer Center Recruiting Pittsburgh, Pennsylvania, United States, 15232 UPMC CancerCenter- Hillman Cancer Center Recruiting Pittsburgh, Pennsylvania, United States, 15232 United States, Washington Seattle Cancer Care Alliance Recruiting Seattle, Washington, United States, 98109 University of Washington Medical Center Recruiting Seattle, Washington, United States, 98195 Collapse << |
| NCT00640692 | - | - | Terminated(Insufficient enroll... more >>ment) Collapse << | April 2011 | - |
| NCT02342600 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Withdrawn(Supporting company w... more >>ithdrew interest) Collapse << | - | - |
| NCT03171389 | GIST, Malignant ... more >> KIT Exon 13 Mutation KIT Gene Mutation Collapse << | Phase 2 | Recruiting | September 22, 2020 | Germany ... more >> West German Cancer Center Recruiting Essen, NRW, Germany, 45122 Principal Investigator: Sebastian Bauer, Prof. Dr. Collapse << |
| NCT02071719 | - | - | Terminated | October 2017 | Netherlands ... more >> VU Medical Center Amsterdam, Noord Holland, Netherlands, 1081 HV Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| PDGFRβ | IC50:2nM | |
| VEGFR2 | IC50:80nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
