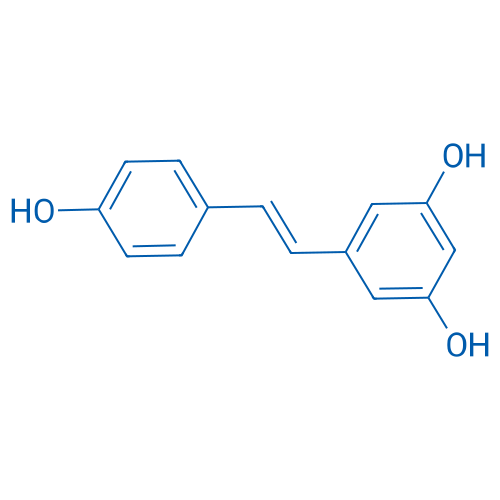
CAS No.: 501-36-0
白藜芦醇 Catalog No. CSN17016
Synonyms: 白藜芦醇;trans-Resveratrol;SRT501
Resveratrol is one of the numerous polyphenolic compounds found in several vegetal sources, has a wide spectrum of targets with IC50 of 0.8, 1, 3.3 and 5 μM for Adenylyl cyclase, IKKβ, DNA polymerase α and δ, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00920556 Multiple Myeloma Phase 2 Terminated(Study terminated.24... more >> subjects enrolled;provided adequate data for decision making.) Collapse << - Denmark ... more >> GSK Investigational Site Vejle, Denmark, 7100 United Kingdom GSK Investigational Site Sutton, Surrey, United Kingdom, SM2 5NG GSK Investigational Site Birmingham, United Kingdom, B15 2TH GSK Investigational Site Leeds, United Kingdom, LS9 7TF GSK Investigational Site London, United Kingdom, EC1A 7BE Collapse << NCT00654667 Insulin Resistance Phase 2 Withdrawn(no participant enrol... more >>lment) Collapse << - United States, California ... more >> University of California San Francisco, California, United States, 94143-0608 Collapse << NCT02256540 Endothelial Dysfunction Not Applicable Active, not recruiting January 2019 United States, Colorado ... more >> University of Colorado Anschutz Medical Campus Aurora, Colorado, United States, 80045 Collapse << - 更多
- 参考文献
- [1] Pirola L, Frojdo S. Resveratrol: one molecule, many targets. IUBMB Life. 2008 May;60(5):323-32.
- [2] Howitz KT, Bitterman KJ, et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 2003 Sep 11;425(6954):191-6.
- [3] Zunino SJ, Storms DH, et al. Resveratrol given intraperitoneally does not inhibit the growth of high-risk t(4;11) acute lymphoblastic leukemia cells in a NOD/SCID mouse model. Int J Oncol. 2012;40(4):1277-84.
- [4] Thirunavukkarasu M, Penumathsa SV, et al. Resveratrol alleviates cardiac dysfunction in streptozotocin-induced diabetes: Role of nitric oxide, thioredoxin, and heme oxygenase. Free Radic Biol Med. 2007;43(5):720-9.
- [5] Harper CE, Cook LM, et al. Genistein and resveratrol, alone and in combination, suppress prostate cancer in SV-40 tag rats. Prostate. 2009;69(15):1668-82.
- [6] Singh M, Parent S, et al. Resveratrol modulates the expression of PTGS2 and cellular proliferation in the normal rat endometrium in an AKT-dependent manner. Biol Reprod. 2011;84(5):1045-52.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 501-36-0 | 储存条件 |
|
|||||||||||
| 分子式 | C14H12O3 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 228.24 | 别名 | 白藜芦醇;trans-Resveratrol;SRT501 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| HEK293 cells | - | Function assay | 48 h | Inhibition of TNF-alpha-induced NF-kappaB activity expressed in HEK293 cells after 48 hrs by luciferase reporter gene assay, IC50=0.173 μM | 20527891 |
| HEK293T cells | - | Function assay | - | Inhibition of TNF-alpha-induced NF-kappaB activation in HEK293T cells, IC50=20 μM | 17125270 |
| human 293 cells | - | Function assay | 24 h | Inhibition of TNFalpha induced NF-kappaB activation in human 293 cells after 24 hrs by luciferase reporter gene assay, IC50=16.1 μM | 18487053 |
| human A549 cells | - | Proliferation assay | 48 h | Antiproliferative activity against human A549 cells assessed as cell viability after 48 hrs by SRB assay, IC50=4.47e-05 | 21851083 |
| human Caco-2 cells | - | Cytotoxic assay | 3 days | Cytotoxicity against human Caco-2 cells after 3 days by [3H]thymidine incorporation assay | 20627379 |
| human CCD-18Co cells | 1 μM | Function assay | 18 h | Antiinflammatory activity in human CCD-18Co cells assessed inhibition of IL1beta-induced PGE2 production at 1 uM after 18 hrs | 20866032 |
| human COLO205 cells | - | Proliferation assay | - | Antiproliferative activity against human COLO205 cells by MTT assay , IC50=23.5 μM | 25455486 |
| human H460 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human H460 cells after 72 hrs, IC50=12.9 μM | 20409723 |
| human H460 cells | 20 μM | Function assay | 24 h | Cell cycle arrest in human H460 cells assessed as accumulation at G2/M phase at 20 uM after 24 hrs by propidium iodide staining-based flow cytometry | 20409723 |
| human HeLa cells | - | Proliferation assay | 48 h | Antiproliferative activity against human HeLa cells assessed as cell viability after 48 hrs by SRB assay, IC50=2.25e-05 μM | 21851083 |
| human HeLa cells | - | Function assay | 30 mins | Inhibition of Hedgehog signaling pathway in human HeLa cells assessed as inhibition of PMA induced luciferase treated 30 mins before induction | 23547843 |
| human HL60 cell line | - | Proliferation assay | - | Antiproliferative activity against human HL60 cell line, IC50=5 μM | 16686543 |
| human HT144 cells | - | Proliferation assay | 5 days | Antiproliferative activity against human HT144 cells after 5 days by acid phosphatase assay, IC50=40 μM | 19481462 |
| human HT-29 cells | - | Cytotoxic assay | 3 days | Cytotoxicity against human HT-29 cells after 3 days by [3H]thymidine incorporation assay, IC50=45.3 μM | 20627379 |
| human Huh7.5.1 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human Huh7.5.1 cells after 72 hrs, IC50=10.6 μM | 23673225 |
| human K562 cell line | - | Proliferation assay | - | Antiproliferative activity against human K562 cell line expressing Bcr-Abl, IC50=28 μM | 16686543 |
| human LS 174T cells | 10 μM | Cytotoxic assay | 4 days | Cytotoxicity against human LS 174T cells at 10 uM for 4 days by vi-cell cell viability analysis | 21291235 |
| human MCF7 cells | - | Proliferation assay | 48 h | Antiproliferative activity against human MCF7 cells assessed as cell viability after 48 hrs by SRB assay, IC50=7.91e-05 | 21851083 |
| human MCF7 cells | - | Function assay | 6 h | Inhibition of phorbol ester-induced ornithine decarboxylase in human MCF7 cells after 6 hrs, IC50=19 μM | 10075742 |
| human MDA468 cells | - | Proliferation assay | - | Antiproliferative activity against human MDA468 cells by MTT assay, IC50=45.2 μM | 25455486 |
| human MDA-MB-231 cells | - | Function assay | 6 days | Antitumor activity against human MDA-MB-231 cells after 6 days by SRB assay, IC50=20.5 μM | 20728369 |
| human MG63 cells | 0.1 to 10 uM | Function assay | 7 days | Increase in ALP activity in human MG63 cells at 0.1 to 10 uM after 7 days | 17513867 |
| human MGC803 cells | - | Proliferation assay | - | Antiproliferative activity against human MGC803 cells by MTT assay, IC50=42 μM | 25455486 |
| human SH-SY5Y cells | - | Function assay | - | Neuroprotective activity in human SH-SY5Y cells assessed as inhibition H2O2-induced reduction of cell viability at 10 uM after 24 hrs by phase-contrast micrograph analysis | 24269515 |
| human SK-MEL cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human SK-MEL cells assessed as growth inhibition measured after 48 hrs by XTT assay, IC50=47.3 μM | 23547843 |
| human SK-N-SH cells | - | Proliferation assay | 48 h | Antiproliferative activity against human SK-N-SH cells assessed as cell viability after 48 hrs by SRB assay, IC50=4.03e-05 | 21851083 |
| human SW480 cells | - | Proliferation assay | 48 h | Antiproliferative activity against human SW480 cells assessed as cell viability using propidium iodide staining after 48 hrs, IC50=20 μM | 20395019 |
| human SW480 cells | 10 μM | Cytotoxic assay | 2 days | Cytotoxicity against human SW480 cells at 10 uM for 2 days by vi-cell cell viability analysis | 21291235 |
| human T47D cells | 1 μM | Proliferation assay | - | Antiproliferative activity against human T47D cells at 1 uM by flow cytometry | 17513867 |
| human THP1 cells | 100 μM | Function assay | 1 h | Inhibition of FSL1-induced upregulation of dectin-1 mRNA expression in in human THP1 cells at 100 uM after 1 hr by RT-PCR | 17938187 |
| human U2OS cells | 100 μM | Function assay | - | Activation of SIRT1 in human U2OS cells assessed as decrease in p53 deacetylation level at 100 uM | 18046409 |
| human U937 cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human U937 cells after 48 hrs by WST1 test, IC50=17 μM | 16252924 |
| HUVEC | 100 μM | Function assay | 1 day | Induction of mitochondrial biogenesis in H2O2-induced HUVEC at 100 umol/L pretreated for 1 day followed by compound washout and H2O2-treatment measured after 10 days by MitoTracker Red staining-based assay | 23859249 |
| MCF7 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human estrogen receptor positive MCF7 cells after 72 hrs by MTT assay, IC50=28.07 μM | 23860590 |
| mouse 3T3L1 cells | - | Function assay | 7 days | Antiadipogenic activity against mouse 3T3L1 cells assessed as inhibition of differentiation after 7 days by oil-red O staining, IC50=31.4 μM | 19757853 |
| mouse C2C12 cells | 50 μM | Function assay | 16 h | Increase in mitochondrial function in mouse C2C12 cells assessed as enhancement of mitochondrial density at 50 uM after 16 hrs by green FM dye straining-based fluorescence assay | 19663498 |
| mouse Hepa1clc7 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against mouse Hepa1clc7 cells after 72 hrs, IC50=11 μM | 10075742 |
| mouse HT22 cells | - | Function assay | 24 h | Neuroprotective activity in mouse HT22 cells assessed as protection against glutamate-induced oxytosis after 24 hrs by MTT assay | 21129978 |
| mouse MC3T3-E1 cells | 1 μM | Apoptosis assay | 48 h | Inhibition of antiapoptotic activity in mouse MC3T3-E1 cells at 1 uM after 48 hrs in presence of BMP2-specific neutralizing antibody | 17513867 |
| mouse NIH/3T3 cells | - | Cytotoxic assay | 96 h | Cytotoxicity against mouse NIH/3T3 cells after 96 hrs by SRB assay, IC50=24.22 μM | 22749392 |
| RAW264.7 cells | - | Function assay | - | Inhibitory effect on PGE-2 production in LPS-stimulated RAW264.7 cells, IC50=4 μM | 15080988 |
| SKBR3 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human estrogen receptor negative SKBR3 cells after 72 hrs by MTT assay, IC50=41.42 μM | 23860590 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00920556 | Multiple Myeloma | Phase 2 | Terminated(Study terminated.24... more >> subjects enrolled;provided adequate data for decision making.) Collapse << | - | Denmark ... more >> GSK Investigational Site Vejle, Denmark, 7100 United Kingdom GSK Investigational Site Sutton, Surrey, United Kingdom, SM2 5NG GSK Investigational Site Birmingham, United Kingdom, B15 2TH GSK Investigational Site Leeds, United Kingdom, LS9 7TF GSK Investigational Site London, United Kingdom, EC1A 7BE Collapse << |
| NCT00654667 | Insulin Resistance | Phase 2 | Withdrawn(no participant enrol... more >>lment) Collapse << | - | United States, California ... more >> University of California San Francisco, California, United States, 94143-0608 Collapse << |
| NCT02256540 | Endothelial Dysfunction | Not Applicable | Active, not recruiting | January 2019 | United States, Colorado ... more >> University of Colorado Anschutz Medical Campus Aurora, Colorado, United States, 80045 Collapse << |
| NCT02944097 | Safety After Oral Intake ... more >> Pharmacokinetics After Oral Intake Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Collapse << |
| NCT01842399 | Vascular Resistance ... more >> Aging Hypertension Antioxidants Aerobic Capacity Collapse << | Phase 1 Phase 2 | Recruiting | September 1, 2019 | United States, Maryland ... more >> National Institute of Aging, Clinical Research Unit Recruiting Baltimore, Maryland, United States, 21224 Collapse << |
| NCT00721877 | Healthy, no Evidence of Diseas... more >>e Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Arizona Cancer Center - Tucson Tucson, Arizona, United States, 85724-5024 Collapse << |
| NCT00920803 | Neoplasms, Colorectal | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Leicester, Leicestershire, United Kingdom, LE1 5WW Collapse << |
| NCT03563365 | Acne Vulgaris | Phase 4 | Recruiting | June 4, 2019 | United States, Pennsylvania ... more >> Yardley Dermatology Associates, PC Recruiting Yardley, Pennsylvania, United States, 19067 Contact: Michelle Clinical Trial Site Manager 215-579-6155 ext 27 msibona@yardleyderm.com Principal Investigator: Richard G Fried, MD, PhD Sub-Investigator: Lauren Sternberg, MD Sub-Investigator: Judith Cenci, MD Sub-Investigator: Priya Dhanaraj, MD Collapse << |
| NCT01964846 | Effect of Resveratrol and Curc... more >>umin on Inflammation Collapse << | Not Applicable | Completed | - | Canada, Quebec ... more >> Institute on Nutrition and Functional Foods (INAF), Laval University Québec City, Quebec, Canada, G1V 0A6 Collapse << |
| NCT02523274 | Aging | Phase 2 | Active, not recruiting | July 2019 | United States, Alabama ... more >> UAB Center for Exercise Medicine Birmingham, Alabama, United States, 35294 Collapse << |
| NCT00743743 | Alzheimer Disease | Phase 3 | Withdrawn(PI has left institut... more >>ion) Collapse << | December 2010 | - |
| NCT01640197 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Brain performance and nutrition research centre, Northumbria university Newcastle upon Tyne, Tyne and Wear, United Kingdom, NE1 8ST Collapse << |
| NCT01881347 | Diabetes Mellitus | Not Applicable | Completed | - | United States, Massachusetts ... more >> Boston University Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT02130440 | Seasonal Allergic Rhinitis | Phase 3 | Completed | - | Italy ... more >> Second University of Naples Naples, Italy Collapse << |
| NCT01766180 | Memory Impairment | Not Applicable | Unknown | February 2016 | United States, Maryland ... more >> Memosyn Neurology Institute, 1205 York Road, Suite 11 Recruiting Lutherville, Maryland, United States, 21093 Contact: Majid Fotuhi, MD, PhD 443-275-1687 mfotuhi@jhu.edu Contact: Mehrnoosh Hadadi, MD, MPH (443) 275-1687 Mehr_Hadadi@yahoo.com Principal Investigator: Majid Fotuhi, MD, PhD Sub-Investigator: Mehrnoosh Hadadi, MD, MPH Collapse << |
| NCT01331382 | Healthy | Not Applicable | Completed | - | United Kingdom ... more >> Brain performance and nutrition research centre, Northumbria university Newcastle upon Tyne, Tyne and Wear, United Kingdom, NE1 8ST Collapse << |
| NCT03683108 | Upper Airways Disease | Phase 3 | Completed | - | Italy ... more >> Dept Of Obstetrics and Neonatology - Section of Neonatology University of Bari Policlinico Hospital Bari, Italy, 70124 Collapse << |
| NCT00433576 | Adenocarcinoma of the Colon ... more >> Adenocarcinoma of the Rectum Stage I Colon Cancer Stage I Rectal Cancer Stage II Colon Cancer Stage II Rectal Cancer Stage III Colon Cancer Stage III Rectal Cancer Collapse << | Phase 1 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01150955 | Metabolic Syndrome ... more >> Obesity Collapse << | Not Applicable | Completed | - | Denmark ... more >> Aarhus University Hospital, Department of Endocrinology, MEA Aarhus, Denmark, 8000 Collapse << |
| NCT01354977 | Type 2 Diabetes Mellitus ... more >> Insulin Resistance Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Albert Einstein College of Medicine Bronx, New York, United States, 10461 Collapse << |
| NCT01640197 | - | - | Completed | - | - |
| NCT01126229 | Memory | Phase 1 | Completed | - | United States, Florida ... more >> University of Florida, Aging and Geriatric Research Gainesville, Florida, United States, 32611 Collapse << |
| NCT01668836 | Vascular System Injuries ... more >> Lipid Metabolism Disorders Endothelial Disfunction Collapse << | Not Applicable | Completed | - | Brazil ... more >> INCOR - Heart Institute São Paulo, Brazil, 05403-900 Collapse << |
| NCT01668836 | - | - | Completed | - | - |
| NCT02766803 | PCOS, Insulin Resistance | Phase 4 | Unknown | June 2018 | Poland ... more >> Division of Infertility and Reproductive Endocrinology, Department of Gynecology and Obsterics Recruiting Poznan, Poland, 60-184 Contact: Beata Banaszewska, Md Ph D +48618419412 bbeata48@gmail.com Contact: Robert Z Spaczynski, Md PhD Prof +48618419412 rspaczynski@yahoo.com Principal Investigator: Leszek A Pawelczyk, MD PhD Prof Sub-Investigator: Antoni J Duleba, MD Prof Collapse << |
| NCT02123121 | Mitochondrial Function ... more >> Physical Function Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, Florida ... more >> UF Institute on Aging Clinical and Translational Research Building Gainesville, Florida, United States, 32611 Collapse << |
| NCT01370889 | Healthy, no Evidence of Diseas... more >>e Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Arizona Cancer Center - Tucson Tucson, Arizona, United States, 85724-5024 University of Arizona Health Sciences Center Tucson, Arizona, United States, 85724 Collapse << |
| NCT01593605 | Impaired Glucose Tolerance | Not Applicable | Completed | - | United States, Tennessee ... more >> Facility: Vanderbilt University Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02947932 | Pancreatitis, Acute ... more >> Gastrointestinal Disease Collapse << | Phase 4 | Unknown | September 2018 | - |
| NCT03349619 | Allergic Rhinitis | Phase 4 | Completed | - | Italy ... more >> Institute of Biomedicine and Molecular Immunology (IBIM) - National Research Council of Palermo Palermo, Italy, 90146 Collapse << |
| NCT02219906 | Metabolic Syndrome | Phase 3 | Enrolling by invitation | December 2018 | United States, Tennessee ... more >> Vanderbilt University Nashville, Tennessee, United States, 37232 United States, Texas Baylor University Houston, Texas, United States, 77004 Collapse << |
| NCT02062190 | Schizophrenia | Phase 2 | Completed | - | Brazil ... more >> HCPA Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT03509688 | Hepatitis | Not Applicable | Completed | - | - |
| NCT01446276 | Obesity Nonal... more >>coholic Fatty Liver Disease Collapse << | Not Applicable | Completed | - | Denmark ... more >> Department of Endocrinology and Internal Medicine Aarhus C, Denmark, 8000 Collapse << |
| NCT03090997 | Postmenopausal ... more >> Insulin Resistance Collapse << | Not Applicable | Recruiting | August 20, 2020 | Mexico ... more >> Instituto Nacional de Perinatología Isidro Espinosa de los Reyes Recruiting Mexico City, Mexico, 11000 Contact: Araceli Montoya-Estrada, MD +525555209900 ext 257 ara_mones@hotmail.com Contact: Enrique Reyes-Muñoz, MD, PhD. +525555209900 ext 307 dr.enriquereyes@gmail.com Collapse << |
| NCT03541993 | Hypoxia, Altitude ... more >> Brain Collapse << | Not Applicable | Completed | - | - |
| NCT03384329 | Depression | Phase 4 | Recruiting | February 2019 | Russian Federation ... more >> Lyubomir I. Aftanas Recruiting Novosibirsk, Russian Federation, 630117 Contact: Lyubomir I. Aftanas, MD 3359855 ext (007)383 liaftanas@gmail.com Collapse << |
| NCT01794351 | Cognitive Performance ... more >> Mood Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Brain performance and nutrition research centre, Northumbria university Newcastle upon Tyne, Tyne and Wear, United Kingdom, NE1 8ST Collapse << |
| NCT00256334 | Colon Cancer ... more >>Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> Chao Family Comprehensive Cancer Center Orange, California, United States, 92868 Collapse << |
| NCT03546075 | Cognitive Change | Not Applicable | Completed | - | - |
| NCT02216552 | NAFLD TYPE 2 ... more >>DIABETES METABOLIC SYNDROME Collapse << | Phase 2 Phase 3 | Completed | - | Canada, Manitoba ... more >> Children's Hospital Research Institute of Manitoba/University of Manitoba Winnipeg, Manitoba, Canada, R3E 3P4 Collapse << |
| NCT03100019 | Hypoxia, Brain | Not Applicable | Completed | - | - |
| NCT01615445 | Sedentary Lifestyle | Phase 2 | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital -General Campus Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT03062163 | Lipolysis | Not Applicable | Completed | - | Thailand ... more >> Faculty of Medicine, Chulalongkorn University Bangkok, Thailand, 10330 Collapse << |
| NCT03352895 | Chronic Kidney Disease | Not Applicable | Completed | - | Taiwan ... more >> Dalin Tzu Chi Hospital Chiayi City, Taiwan, 66247 Collapse << |
| NCT03546530 | Hepatitis B | Not Applicable | Completed | - | - |
| NCT01714102 | Obesity Insul... more >>in Resistance Metabolic Syndrome Collapse << | Not Applicable | Completed | - | United States, New York ... more >> The Rockefeller University New York, New York, United States, 10065 Collapse << |
| NCT02336633 | Huntington Disease | Not Applicable | Recruiting | January 2019 | France ... more >> Institut du Cerveau et de la Moelle, Hôpital de la Pitié Salpêtrière Recruiting Paris, France, 75013 Contact: Tiffany Monnier, M Scs Collapse << |
| NCT01324089 | Focus of the Study: Normal Vol... more >>unteers Collapse << | Not Applicable | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01504854 | - | - | Completed | - | - |
| NCT01244360 | Inflammation | Not Applicable | Unknown | June 2011 | United States, Pennsylvania ... more >> Marywood University Scranton, Pennsylvania, United States, 18509 Collapse << |
| NCT01321151 | Sports Concussion | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Collapse << |
| NCT01010009 | Cognitive and Cerebral Blood F... more >>low Effects of Resveratrol Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Northumbria University Newcastle upon Tyne, United Kingdom, NE1 8ST Collapse << |
| NCT01826279 | Obesity | Not Applicable | Unknown | August 2014 | Denmark ... more >> Department of Endocrinology and Internal Medicine Aarhus C, Denmark, 8000 Collapse << |
| NCT01010009 | - | - | Completed | - | - |
| NCT02137421 | Metabolic Syndrome ... more >> Coronary Artery Disease Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Islamic Republic of Iran Tehran, Iran, Islamic Republic of Collapse << |
| NCT01747252 | Healthy | Not Applicable | Completed | - | Israel ... more >> BioStudies Center Beer-Sheva, Israel, 84101 Collapse << |
| NCT02704494 | Diabetic Nephropathy | Early Phase 1 | Completed | - | Iran, Islamic Republic of ... more >> Shahid Motahhari Clinic, Shiraz University of Medical Sciences Shiraz, Fars, Iran, Islamic Republic of Collapse << |
| NCT01720459 | Polycystic Ovary Syndrome (PCO... more >>S) Collapse << | Not Applicable | Unknown | December 2014 | Poland ... more >> Poznan University of Medical Sciences Not yet recruiting Poznan, Poland, 60-535 Collapse << |
| NCT01158417 | Type 2 Diabetes ... more >> Obesity Insulin Resistance Collapse << | Phase 2 Phase 3 | Unknown | August 2014 | United States, New York ... more >> 115 Flint Road Buffalo, New York, United States, 14221 Collapse << |
| NCT01504854 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT02261844 | Liver Cancer | Phase 1 Phase 2 | Withdrawn(No funding) | - | United States, Kentucky ... more >> University of Louisville Louisville, Kentucky, United States, 40202 Collapse << |
| NCT02616822 | Endothelial Dysfunction | Phase 1 | Completed | - | - |
| NCT01412645 | Obesity Infla... more >>mmation Insulin Sensitivity Osteoporosis Collapse << | Not Applicable | Completed | - | Denmark ... more >> Clinical Institute, Aarhus University Aarhus, Denmark, 8000 Collapse << |
| NCT02030977 | Non Alcoholic Fatty Liver | Phase 2 Phase 3 | Completed | - | - |
| NCT02905799 | Knee Osteoarthritis | Phase 3 | Recruiting | August 2020 | France ... more >> Rehabilitation department , CHU Clermont-Ferrand Not yet recruiting Cebazat, France, 63118 Contact: Emmanuel Coudeyre, MD, PhD +33473750900 ecoudeyre@chu-clermontferrand.fr Rheumatology Department, Saint Antoine Hospital Not yet recruiting Paris, France, 75012 Contact: Francis Berenbaum, MD, PhD +33149282520 francis.berenbaum@aphp.fr Rehabilitation Department, Cochin Hospital Recruiting Paris, France, 75014 Contact: François Rannou, MD, PhD +33 1 58 41 25 35 francois.rannou@aphp.fr Collapse << |
| NCT01464801 | Fatty Liver | Not Applicable | Completed | - | Denmark ... more >> Aarhus University Hospital, Dept. of Hepatology and Gastroentology Aarhus, Denmark, 8000 Collapse << |
| NCT01038089 | Type 2 Diabetes Mellitus | Not Applicable | Completed | - | United States, Massachusetts ... more >> Boston Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT01564381 | Cardiovascular Disease | Phase 1 Phase 2 | Completed | - | United States, California ... more >> University of California, Davis - Ragle Human Nutrition Research Center Davis, California, United States, 95616 Collapse << |
| NCT01173640 | Healthy | Not Applicable | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01492114 | Chronic Subclinic Inflammation... more >> Redox Status Collapse << | Phase 3 | Completed | - | Italy ... more >> Simona Bo Turin, Italy, 10126 Collapse << |
| NCT01914081 | Dilated Cardiomyopathy | Phase 3 | Recruiting | November 2020 | Canada, Manitoba ... more >> St. Boniface General Hospital Recruiting Winnipeg, Manitoba, Canada, R2H 2A6 Contact: Wendy Janz, RN 204-237-2793 wjanz@hsc.mb.ca Sub-Investigator: Amrit Malik, MD Principal Investigator: Shelley Zieroth, MD Sub-Investigator: Thomas Netticadan, Ph. D. Collapse << |
| NCT01375959 | Impaired Glucose Tolerance | Not Applicable | Unknown | April 2014 | United States, New York ... more >> Albert Einstein College of Medicine of Yeshiva University Recruiting Bronx, New York, United States, 10461 Collapse << |
| NCT03728777 | Mitochondrial Myopathies ... more >> Fatty Acid Oxidation Defects Collapse << | Not Applicable | Recruiting | April 2021 | Denmark ... more >> Copenhagen Neuromuscular Center Recruiting Copenhagen, Denmark, 2100 Contact: Nicoline Løkken, MD Collapse << |
| NCT03597568 | Chronic Kidney Diseases ... more >> Endothelial Dysfunction Collapse << | Phase 1 Phase 2 | Not yet recruiting | August 1, 2020 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Not yet recruiting Iowa City, Iowa, United States, 52245 Contact: Katherine Geasland 319-353-6318 katherine-geasland@uiowa.edu Collapse << |
| NCT01997762 | Gestational Diabetes | Phase 4 | Unknown | December 2016 | Canada, Manitoba ... more >> Manitoba Institute of Child Health Recruiting Winnipeg, Manitoba, Canada, R3E 3P4 Principal Investigator: Shayne P Taback, MD Collapse << |
| NCT02114892 | - | - | Completed | - | - |
| NCT02415114 | CoQ10 Blood Levels ... more >> CoQ10 Blood Levels With Statin Use Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << |
| NCT01364961 | Dyslipidemia | Not Applicable | Completed | - | Netherlands ... more >> Maastricht University Medical Center Maastricht, Netherlands Collapse << |
| NCT02114892 | Metabolic Syndrome X | Phase 2 | Completed | - | Mexico ... more >> Intstituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 45037 Collapse << |
| NCT01638780 | Type 2 Diabetes | Not Applicable | Unknown | December 2014 | Netherlands ... more >> Maastricht University Medical Center Maastricht, Limburg, Netherlands, 6200MD Collapse << |
| NCT02246660 | - | - | Completed | - | - |
| NCT01339884 | Friedreich Ataxia | Phase 1 Phase 2 | Completed | - | Australia, Victoria ... more >> Monash Medical Centre, Southern Health Clayton, Melbourne, Victoria, Australia, 3168 Collapse << |
| NCT02625376 | AMD Age-relat... more >>ed Macular Degeneration Choroidal Neovascularisation Collapse << | Not Applicable | Recruiting | August 2019 | France ... more >> CHU de Poitiers - Ophtalmology Recruiting Poitiers, France, 86000 Contact: Nicolas leveziel, MD nicolas.leveziel@chu-poitiers.fr Collapse << |
| NCT02433925 | Chronic Renal Insufficiency | Phase 3 | Completed | - | - |
| NCT02246660 | Peripheral Arterial Disease | Not Applicable | Completed | - | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT02247596 | Obesity Diabe... more >>tes Collapse << | Phase 2 | Completed | - | Singapore ... more >> Khoo Teck Puat Hospital Singapore, Singapore, 768828 Collapse << |
| NCT02475564 | Endometriosis | Phase 4 | Completed | - | Brazil ... more >> HCPA Porto Alegre, RS, Brazil, 90035-903 Collapse << |
| NCT02549924 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(no financial suppor... more >>t) Collapse << | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT01476592 | Neuroendocrine Tumor | Not Applicable | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT02129595 | Pre-diabetes | Not Applicable | Completed | - | Netherlands ... more >> Maastricht University Medical Centre Maastricht, Limburg, Netherlands, 6200 MD Collapse << |
| NCT02244879 | Type 2 Diabetes Mellitus ... more >> Inflammation Insulin Resistance Other Disorders of Bone Density and Structure Collapse << | Phase 3 | Completed | - | Italy ... more >> University of Turin Turin, IT, Italy, 10126 Collapse << |
| NCT01782911 | Androgen Profile ... more >> Inflammatory Markers IVF Outcome Collapse << | Not Applicable | Suspended(Because the recruitm... more >>ent was difficult) Collapse << | - | Spain ... more >> Ivi Madrid Madrid, Spain, 28023 Collapse << |
| NCT01451918 | Dyslipidaemia ... more >> Insulin Resistance Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Toronto General Hospital Toronto, Ontario, Canada Collapse << |
| NCT02621554 | Healthy | Not Applicable | Completed | - | Germany ... more >> Department of Neurology, Max Planck Institute for Human Cognitive and Brain Sciences Leipzig, Germany Collapse << |
| NCT02321176 | Eye Disease | Not Applicable | Unknown | April 2015 | China, Hubei ... more >> Tongji Medical College of HUST Recruiting Wuhan, Hubei, China, 430030 Contact: ShuaiShuai Wang 15623422892 949990624@qq.com Collapse << |
| NCT03446625 | Infertility | Phase 3 | Recruiting | December 2018 | Spain ... more >> Ivi Madrid Recruiting Madrid, Spain, 28023 Contact: IVI MADRID 91 180 29 00 israel.ortega@ivi.es Principal Investigator: Israel Ortega, MD Collapse << |
| NCT03253913 | Lymphangioleiomyomatosis | Phase 2 | Recruiting | June 30, 2020 | United States, Ohio ... more >> University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45267 Contact: Tammy Roads, CCRP 513-558-2148 roadst@ucmail.uc.edu Principal Investigator: Nishant Gupta, MD Collapse << |
| NCT02475564 | - | - | Completed | - | - |
| NCT03448094 | Cognitive Function and Mood ... more >> Cerebral Blood Flow Systemic Inflammation Gut Microbiota Overweight and Obesity Collapse << | Not Applicable | Recruiting | October 2018 | United Kingdom ... more >> Northumbria University Recruiting Newcastle Upon Tyne, Tyne And Wear, United Kingdom, NE1 8ST Contact: Ellen Smith 01912437252 ellen.smith@northumbria.ac.uk Collapse << |
| NCT03665740 | Gulf War Illness | Phase 2 | Recruiting | August 31, 2022 | United States, Texas ... more >> VISN 17 Center of Excellence for Research on Returning War Veterans Recruiting Waco, Texas, United States, 76711 Contact: Tabitha Alverio, M.A. 254-297-3259 Tabitha.Alverio@va.gov Contact: Laura Constable, Ph.D. 254.297.3954 Laura.Constable@va.gov Principal Investigator: Dena Davidson, Ph.D. Sub-Investigator: Bryann DeBeer, Ph.D. Sub-Investigator: Geoffrey May, MD Sub-Investigator: Steven Nelson, Ph.D. Collapse << |
| NCT02245932 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Maastricht University Medical Center Maastricht, Limburg, Netherlands Collapse << |
| NCT03762096 | Diabetes Mellitus, Type 2 ... more >> Coronary Artery Disease Collapse << | Not Applicable | Recruiting | December 31, 2021 | United States, Maine ... more >> Maine Medical Center Recruiting Portland, Maine, United States, 04102 Collapse << |
| NCT03525379 | Congestive Heart Failure Chron... more >>ic Collapse << | Phase 2 | Recruiting | December 1, 2020 | Canada, Alberta ... more >> Alberta Cardiovascular and Stroke Research Centre Recruiting Edmonton, Alberta, Canada, T6G 2B7 Contact: Marleeni Irwin 7804922960 mirwin@ualberta.ca Principal Investigator: Justin Ezekowitz, MBBCh MSc Collapse << |
| NCT03743636 | Peripheral Artery Disease | Phase 3 | Recruiting | April 2022 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu Contact: Kathryn Domanchuk, BS 312-503-6438 k-domanchuk@northwestern.edu Principal Investigator: Mary McDermott, MD Collapse << |
| NCT01677611 | Type 2 Diabetes | Phase 1 | Completed | - | Singapore ... more >> Alexandra Health, Khoo Teck Puat Hospital Singapore, Singapore, 768828 Collapse << |
| NCT02565979 | Pre-diabetes | Not Applicable | Recruiting | December 2018 | Netherlands ... more >> Maastricht University and Medical Centre Recruiting Maastricht, Limburg, Netherlands, 6200 MD Contact: Marlies de Ligt, MSc +31 433 881587 marlies.deligt@maastrcihtuniversity.nl Contact: Patrick Schrauwen, Prof., Dr. +31 433 881502 p.schrauwen@maastrichtuniversity.nl Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
